
As filed with the Securities and Exchange Commission on August 14, 2015
Registration No. 333-
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM S-1
REGISTRATION STATEMENT
UNDER
THE SECURITIES ACT OF 1933
Penumbra, Inc.
(Exact Name of Registrant as Specified in Its Charter)
| Delaware | 3841 | 05-0605598 | ||
| (State or Other Jurisdiction of Incorporation or Organization) |
(Primary Standard Industrial Classification Code Number) |
(I.R.S. Employer Identification Number) |
One Penumbra Place
1351 Harbor Bay Parkway
Alameda, California 94502
(510) 748-3200
(Address, Including Zip Code, and Telephone Number, Including Area Code, of Registrant’s Principal Executive Offices)
Adam Elsesser
Chairman, Chief Executive Officer and President
Penumbra, Inc.
One Penumbra Place
1351 Harbor Bay Parkway
Alameda, California 94502
(510) 748-3200
(Name, Address, Including Zip Code, and Telephone Number, Including Area Code, of Agent For Service)
Copies to:
| Alan F. Denenberg Davis Polk & Wardwell LLP 1600 El Camino Real Menlo Park, California 94025 (650) 752-2000 |
Robert D. Evans Executive Vice President and General Counsel Penumbra, Inc. One Penumbra Place 1351 Harbor Bay Parkway Alameda, California 94502 (510) 748-3200 |
Rezwan D. Pavri Richard A. Kline Goodwin Procter LLP 135 Commonwealth Drive Menlo Park, California 94025 (650) 752-3100 |
Approximate date of commencement of proposed sale to the public: As soon as practicable after the effective date of this Registration Statement.
If any of the securities being registered on this form are to be offered on a delayed or continuous basis pursuant to Rule 415 under the Securities Act of 1933, check the following box. ¨
If this form is filed to register additional securities for an offering pursuant to Rule 462(b) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. ¨
If this form is a post-effective amendment filed pursuant to Rule 462(c) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. ¨
If this form is a post-effective amendment filed pursuant to Rule 462(d) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. ¨
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or a smaller reporting company. See the definitions of “large accelerated filer,” “accelerated filer” and “smaller reporting company” in Rule 12b-2 of the Exchange Act.
| Large accelerated filer | ¨ | Accelerated filer | ¨ | |||||
| Non-accelerated filer | x | (Do not check if a smaller reporting company) | Smaller reporting company | ¨ | ||||
CALCULATION OF REGISTRATION FEE
|
| ||||
| Title Of Each Class Of Securities To Be Registered |
Proposed Maximum Aggregate Offering Price(1)(2) |
Amount Of Registration Fee | ||
| Common Stock, par value $0.001 per share |
$115,000,000 | $13,363.00 | ||
|
| ||||
|
| ||||
| (1) | Includes additional shares that the underwriters have the right to purchase from us. |
| (2) | Estimated solely for the purpose of computing the amount of the registration fee pursuant to Rule 457(o) under the Securities Act of 1933. |
The Registrant hereby amends this registration statement on such date or dates as may be necessary to delay its effective date until the registrant shall file a further amendment which specifically states that this registration statement shall thereafter become effective in accordance with Section 8(a) of the Securities Act of 1933 or until the registration statement shall become effective on such date as the Commission, acting pursuant to said Section 8(a), may determine.
The information in this prospectus is not complete and may be changed. We may not sell these securities until the registration statement filed with the Securities and Exchange Commission is effective. This prospectus is not an offer to sell these securities and we are not soliciting offers to buy these securities in any state where the offer or sale is not permitted.
Subject to completion, dated , 2015
Preliminary Prospectus
Shares

Common Stock
$ per share
This is the initial public offering of common stock of Penumbra, Inc.
We are offering shares of our common stock. Prior to this offering, there has been no public market for our common stock. We anticipate that the initial public offering price will be between $ and $ per share.
We intend to apply to list our common stock on the New York Stock Exchange under the symbol “PEN.”
| Per Share | Total | |||||||
| Initial public offering price |
$ | $ | ||||||
| Underwriting discounts and commissions(1) |
$ | $ | ||||||
| Proceeds to Penumbra before expenses(1) |
$ | $ | ||||||
| (1) | See the section titled “Underwriting” for additional disclosure regarding underwriter compensation and offering expenses. |
We have granted the underwriters the right to purchase an additional shares of common stock from us.
We are an “emerging growth company” as defined under the federal securities laws, and as such, we have elected to comply with reduced reporting requirements for this prospectus and may elect to do so in future filings.
Investing in our common stock involves a high degree of risk. See the section titled “Risk Factors” beginning on page 10.
Neither the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities or determined if this prospectus is truthful or complete. Any representation to the contrary is a criminal offense.
The underwriters expect to deliver the shares of common stock to purchasers on or about , 2015.
| J.P. Morgan | BofA Merrill Lynch | |
| Wells Fargo Securities | Canaccord Genuity | |
, 2015
In this prospectus, “Penumbra,” “Penumbra, Inc.,” the “Company,” “we,” “us” and “our” refer to Penumbra, Inc. and its consolidated subsidiaries. We and the underwriters have not authorized anyone to provide any information or to make any representations other than those contained in this prospectus or in any free writing prospectuses we have prepared. We and the underwriters take no responsibility for, and can provide no assurance as to the reliability of, any other information that others may provide you. We are offering to sell, and seeking offers to buy, shares of common stock only in jurisdictions where offers and sales are permitted. The information contained in this prospectus is accurate only as of the date of this prospectus, regardless of the time of delivery of this prospectus or of any sale of the common stock.
This prospectus includes industry and market data that we obtained from industry publications, internal estimates and other third-party sources. These sources may include government and industry sources. Industry publications and surveys generally state that the information contained therein has been obtained from sources believed to be reliable. Although we believe the industry and market data to be reliable as of the date of this prospectus, this information could prove to be inaccurate. Industry and market data could be wrong because of the method by which sources obtained their data and because information cannot always be verified with complete certainty due to the limits on the availability and reliability of raw data, the voluntary nature of the data gathering process and other limitations and uncertainties. In addition, we do not know all of the assumptions regarding general economic conditions or growth that were used in preparing the forecasts from the sources relied upon or cited herein.
We also refer to certain studies in this prospectus. In certain cases, we may sponsor, fund and/or control the conduct of these studies, or may have other relationships with certain of the authors of such studies. Specifically, we sponsored, funded and controlled the THERAPY, Penumbra Pivotal and PRISM studies, and provided a modest grant, together with a number of other industry participants, relating to the MR CLEAN study, but did not control such study. We may also have or have had consulting relationships with or have provided grants to physicians who authored or co-authored some of such studies for matters unrelated to such studies, including the MR CLEAN, ESCAPE, SWIFT PRIME, REVASCAT, Kass-Hout T, et al., Humphries W, et al., ADAPT FAST, Mascitelli, Patel, et al. and Milburn, et al. studies cited in this prospectus.
i
Through and including , 2015 (the 25th day after the date of this prospectus), all dealers that buy, sell or trade our common stock, whether or not participating in this offering, may be required to deliver a prospectus. This is in addition to the dealers’ obligation to deliver a prospectus when acting as underwriters and with respect to their unsold allotments or subscriptions.
ii
This summary highlights information contained elsewhere in this prospectus. This summary may not contain all of the information that you should consider before deciding to invest in our common stock. You should read this entire prospectus carefully, including the sections titled “Risk Factors” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and the consolidated financial statements and the notes to those statements contained elsewhere in this prospectus.
Overview
Penumbra is a global interventional therapies company that designs, develops, manufactures and markets innovative medical devices. We have a broad portfolio of products that addresses challenging medical conditions and significant clinical needs across two major markets, neuro and peripheral vascular. The conditions that our products address include, among others, ischemic stroke and hemorrhagic stroke, which involve blockage or rupture of blood vessels in the brain, and various peripheral vascular conditions that can be treated through thrombectomy and embolization procedures, which involve the use of medical devices to remove or treat blockages or ruptures of blood vessels.
We are an established company focused on the neuro market, and we recently expanded our business to include the peripheral vascular market. We sell our products to hospitals, primarily through our salesforce, as well as through distributors in select international markets. We focus on developing, manufacturing and marketing products for use by specialist physicians, including interventional neuroradiologists, neurosurgeons, interventional neurologists, interventional radiologists and vascular surgeons. We design our products to provide these specialist physicians with a means to drive improved clinical outcomes through faster and safer procedures. Studies involving our Penumbra System ischemic stroke products and Penumbra Coil 400 neurovascular embolization products, as well as initial study results for our Indigo peripheral thrombectomy products, have shown that these products generate significant overall cost savings to the healthcare system relative to procedures using other products.
We attribute our success to our culture built on cooperation, our highly efficient product innovation process, our disciplined approach to product and commercial development, our deep understanding of our target end markets and our relationships with specialist physicians. We believe these factors have enabled us to rapidly innovate in a highly capital-efficient manner.
Since our founding in 2004, we have had a strong track record of organic product development and commercial expansion that has established the foundation of our global organization. Some of our key accomplishments include:
| • | launching our first product, for neurovascular access, in the United States in 2007; |
| • | establishing our direct neuro salesforce in the United States and Europe in 2008; |
| • | launching the first 510(k)-cleared, aspiration-based product for the treatment of ischemic stroke patients in 2008, and launching four subsequent generations of that product; |
| • | launching our first neurovascular coil for the treatment of brain aneurysms in 2011; |
| • | launching our first peripheral vascular product in 2013; and |
| • | establishing our direct peripheral vascular salesforce in the United States and Europe in 2014. |
As of June 30, 2015, we had approximately 1,000 employees worldwide. We sell our products to hospitals primarily through our direct sales organization in the United States, most of Europe, Canada and Australia, as well as through distributors in select international markets. For the year ended December 31, 2014, we generated revenue of $125.5 million, which represents a 41.3% increase over 2013, and $3.0 million in operating income as compared to an operating loss of $1.1 million in 2013. For the six months ended June 30, 2015, we generated revenue of $81.3 million, which represents a 41.0% increase over the six months ended June 30, 2014, and $0.2 million in operating income as compared to operating income of $2.4 million for the six months ended June 30, 2014.
1
Market Opportunity
We estimate that the market for our current neuro and peripheral vascular products in the United States and Europe combined was approximately $1.3 billion in 2014, which we estimate represents growth of approximately 3.2% per year from 2012. While reliable third-party data is not available for markets outside the United States and Europe, we believe that there is a substantial additional market for our neuro and peripheral vascular products in the rest of the world.
According to the American Heart Association (AHA), the worldwide incidence of all forms of stroke was 33 million in 2010. Furthermore, according to a paper published in the journal The Lancet, 202 million people globally were living with peripheral artery disease in 2010.
We believe the market for our products remains substantially under-penetrated today, and that this market will experience significant growth as we and our competitors:
| • | generate additional clinical evidence supporting endovascular treatment of vascular disease; |
| • | improve existing technologies to enable physicians to treat vascular disease faster and more safely than previously possible; |
| • | support and educate the growing number of specialist physicians who treat vascular disease in the use of endovascular treatment; |
| • | grow the number of hospitals where endovascular treatment of vascular disease is available; and |
| • | raise patient awareness of endovascular treatment of vascular disease. |
Industry Background
Vascular disease refers to any condition that affects the circulatory system and typically manifests as a blockage or rupture of an artery or a vein. It may occur in any part of the body, and is a condition that leads most often to blood vessel narrowing and obstruction, but can also lead to expansion of the blood vessel wall and blood vessel wall weakening and rupture. Vascular disease can cause a range of conditions, from pain to functional impairment, and it can require the amputation of a limb or result in death.
When the treatment for vascular disease is performed from within a vessel, it is referred to as an endovascular procedure. Endovascular device markets are conventionally classified according to the anatomic location of the disorder. We currently focus our efforts on the neuro and peripheral vascular markets.
| • | Neuro products. Our neuro products are used to treat patients with vascular disease and disorders in the brain, including patients with strokes caused by either vascular occlusion or rupture or weakening of the vessel walls. Our neuro products are generally catheter-based technologies that are administered by an interventional neuroradiologist, a neurosurgeon or an interventional neurologist. |
| • | Peripheral products. Our peripheral products are used to treat patients with vascular disease in all vasculature other than that which exists in the brain or the heart, including the upper and lower extremities, kidneys, neck and lungs. Our products that address peripheral vascular disorders are catheter-based technologies that are typically administered by an interventional radiologist or a vascular surgeon. |
Our Strengths
As we have grown as an organization, we have been able to scale our business from development stage in 2004 to a company with approximately 1,000 employees focused on multiple product categories in two target end markets. We believe the following strengths have enabled us to develop our broad and differentiated product portfolio and have been, and will continue to be, significant factors in our continued success and growth:
| • | our culture built on cooperation, which we have institutionalized through our unique organizational structure; |
2
| • | our highly efficient product innovation process; |
| • | our disciplined approach to clinical and commercial development; |
| • | our deep understanding of our target end markets; and |
| • | our relationships with specialist physicians. |
Our Products
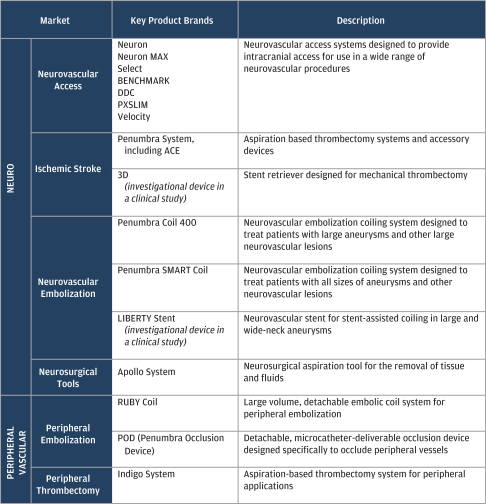
Since our founding in 2004, we have invested in expanding our product development and marketing capabilities. These investments have included engineering and materials science capabilities, pre-clinical and bench-testing infrastructure and in-house clinical and regulatory infrastructure. Our fully-integrated organization has enabled us to launch 14 product brands for access, thrombectomy and embolization since 2007 to service our two target end markets. Our current portfolio provides a range of products that are designed to enable specialist physicians to drive improved clinical outcomes in a cost effective manner.
The following table summarizes our product offerings in each of our target end markets:
3
Our Growth Strategies
We believe the following strategies will continue to play a critical role in our future growth:
| • | expanding the penetration of our products in our target end markets; |
| • | growing the acceptance of our innovative products as the standard of care in their targeted clinical applications; |
| • | continuing to leverage our development capabilities to drive efficient, rapid product development; and |
| • | scaling our organizational culture of cooperative product development and commercial execution. |
Risks Associated With Our Business
Our business is subject to numerous risks, as more fully described in the section titled “Risk Factors” immediately following this prospectus summary. These risks include, among others:
| • | we have a limited operating history and may not be able to sustain or grow our profitability or generate positive cash flows from operations; |
| • | our existing products may be rendered obsolete and we may be unable to effectively introduce and market new products or may fail to keep pace with advances in technology; |
| • | delays in product introductions could adversely affect our business, results of operations, financial condition or cash flows; |
| • | we face significant competition, and if we are unable to compete effectively, we may not be able to achieve or maintain significant market penetration or improve our results of operations; |
| • | our future growth depends, in part, on our ability to further penetrate our current customer base and increase the frequency of use of our products by our customers; |
| • | our future growth depends, in part, on significantly expanding our user base to include additional specialist physicians in both our existing and future target end markets; |
| • | the marketing and sales of our products require a significant amount of time and expense and we may not have the resources to successfully market and sell our products; |
| • | third-party reimbursement may not be available or adequate for the procedures in which our products are used; |
| • | we are subject to stringent domestic and foreign medical device regulation, which may impede the approval or clearance process for our products, hinder our development activities and manufacturing processes and, in some cases, result in the recall or seizure of previously approved or cleared products; |
| • | we rely on a variety of intellectual property rights, and if we are unable to maintain or protect our intellectual property, our business and results of operations will be harmed; and |
| • | we may become involved in lawsuits or other proceedings to protect or enforce our patents or other intellectual property rights or to defend against accusations of infringement, which could be expensive, time consuming and unsuccessful. |
Corporate Information
We were incorporated in 2004 as a Delaware corporation under the name Penumbra, Inc. Our principal executive offices are located at One Penumbra Place, 1351 Harbor Bay Parkway, Alameda, California 94502, and our telephone number is (510) 748-3200. Our website address is www.penumbrainc.com. The information on, or that can be accessed through, our website is not part of this prospectus. We have included our website address as an inactive textual reference only.
4
We use “Penumbra System®,” “ACE™,” “Penumbra Coil 400™,” “Penumbra SMART COIL™,” “LIBERTY™ Stent,” “Apollo™ System,” “Ruby® Coil,” “Indigo® System” and other marks as trademarks in the United States and other countries. This prospectus contains references to our trademarks and service marks and to those belonging to other entities. Solely for convenience, trademarks and trade names referred to in this prospectus, including logos, artwork and other visual displays, may appear without the ® or ™ symbols, but such references are not intended to indicate in any way that we will not assert, to the fullest extent under applicable law, our rights or the rights of the applicable licensor to these trademarks and trade names. We do not intend our use or display of other entities’ trade names, trademarks or service marks to imply a relationship with, or endorsement or sponsorship of us by, any other entity.
Implications of Being an Emerging Growth Company
We are an “emerging growth company,” as defined in the Jumpstart Our Business Startups Act of 2012. We will remain an emerging growth company until the earlier of (1) December 31, 2020 (the last day of the fiscal year following the fifth anniversary of our initial public offering), (2) the last day of the fiscal year in which we have total annual gross revenue of at least $1.0 billion, (3) the last day of the fiscal year in which we are deemed to be a large accelerated filer, which means the market value of our common stock that is held by non-affiliates is equal to or exceeds $700 million as of the prior June 30th, and (4) the date on which we have issued more than $1.0 billion in non-convertible debt during the prior three-year period. We refer to the Jumpstart Our Business Startups Act of 2012 herein as the “JOBS Act,” and any reference herein to “emerging growth company” has the meaning ascribed to it in the JOBS Act.
An emerging growth company may take advantage of reduced reporting requirements that are otherwise applicable to public companies. These provisions include, but are not limited to:
| • | being permitted to present only two years of audited financial statements and only two years of related “Management’s Discussion and Analysis of Financial Condition and Results of Operations” in this prospectus; |
| • | not being required to comply with the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act of 2002, as amended; |
| • | reduced disclosure obligations regarding executive compensation in our periodic reports, proxy statements and registration statements, including in this prospectus; and |
| • | exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and stockholder approval of any golden parachute payments not previously approved. |
We have elected to take advantage of certain of the reduced disclosure obligations in this prospectus and may elect to take advantage of other reduced reporting requirements in our future filings with the U.S. Securities and Exchange Commission (the SEC). As a result, the information that we provide to our stockholders may be different than you might receive from other public reporting companies in which you hold equity interests.
The JOBS Act also provides that an emerging growth company can take advantage of an extended transition period for complying with new or revised accounting standards. We have irrevocably elected not to avail ourselves of this exemption and, therefore, we will be subject to the same new or revised accounting standards as other public companies that are not emerging growth companies.
5
THE OFFERING
| Common stock offered by us |
shares |
| Common stock to be outstanding after this offering |
shares |
| Option to purchase additional shares of our common stock from us |
shares |
| Use of proceeds |
We estimate that the net proceeds to us from this offering will be approximately $ million, or approximately $ million if the underwriters exercise their option to purchase additional shares in full, assuming an initial public offering price of $ per share (which is the midpoint of the estimated initial public offering price range set forth on the cover page of this prospectus), after deducting underwriting discounts and commissions and estimated offering expenses payable by us. |
| The principal purposes of this offering are to increase our capitalization and financial flexibility, create a public market for our common stock and enable access to the public equity markets for us and our stockholders. We intend to use the net proceeds from this offering for product development, including research and development and clinical trials, expansion of our salesforce and working capital and general corporate purposes. From time to time, we may consider the acquisition of complementary technologies or businesses, though we have no agreements or understandings with respect to any such acquisitions at this time. Pending the use of the net proceeds from this offering, we intend to invest the net proceeds in investment grade, interest bearing securities. See the section titled “Use of Proceeds” for additional information. |
| Proposed NYSE stock symbol |
“PEN” |
The number of shares of common stock to be outstanding after this offering is based upon 26,038,637 shares outstanding (including preferred stock on an as-converted basis) as of June 30, 2015, and excludes:
| • | 2,460,574 shares of common stock issuable upon the exercise of options to purchase shares of our common stock outstanding as of June 30, 2015, at a weighted average exercise price of $5.27 per share; |
| • | 1,713,634 shares of common stock reserved for future grant or issuance under our 2014 Equity Incentive Plan as of June 30, 2015; |
| • | 867,000 shares of common stock issuable upon the exercise of options to purchase shares of our common stock at an exercise price of $22.04 per share and 5,000 shares of restricted stock, which were granted on August 12, 2015; |
| • | 3,000,000 shares of common stock initially reserved for future issuance under our Amended and Restated 2014 Equity Incentive Plan, which will become effective immediately prior to the completion of this offering, as well as any automatic increases in the number of shares of our common stock reserved for future issuance pursuant to this plan; and |
| • | 600,000 shares of common stock initially reserved for issuance under our 2015 Employee Stock Purchase Plan, or our ESPP, as well as any automatic increases in the number of shares of our common stock reserved for future issuance pursuant to this plan. |
6
Unless otherwise indicated, this prospectus reflects and assumes the following:
| • | outstanding shares include 24,818 shares of common stock issued upon early exercise of stock options and subject to repurchase; |
| • | outstanding shares include 755,771 shares of unvested restricted stock; |
| • | no exercise of options outstanding as of June 30, 2015, or subsequently issued; |
| • | the automatic conversion of all of our outstanding shares of preferred stock into an aggregate of 19,510,410 shares of common stock immediately prior to the completion of this offering, assuming a one-to-one conversion ratio of our outstanding shares of preferred stock into common stock; |
| • | no exercise by the underwriters of their option to purchase up to additional shares of our common stock from us; and |
| • | the filing and effectiveness of our amended and restated certificate of incorporation in Delaware and the adoption and effectiveness of our amended and restated bylaws immediately prior to the completion of this offering. |
7
SUMMARY CONSOLIDATED FINANCIAL DATA
The following tables summarize our consolidated financial data. We have derived the summary consolidated statement of operations data for the years ended December 31, 2013 and 2014, from our audited consolidated financial statements included elsewhere in this prospectus. We have derived the summary consolidated statement of operations data for the year ended December 31, 2012, from our audited consolidated financial statements not included in this prospectus. We have derived the summary consolidated statement of operations data for the six months ended June 30, 2014 and 2015, and our balance sheet data as of June 30, 2015, from our unaudited interim consolidated financial statements included elsewhere in this prospectus. The unaudited interim consolidated financial statements have been prepared on the same basis as the audited consolidated financial statements and reflect, in our opinion, all adjustments of a normal, recurring nature that are necessary for a fair statement of the unaudited interim consolidated financial statements. Our historical results are not necessarily indicative of the results that may be expected in the future and the results for the six months ended June 30, 2015 are not necessarily indicative of results to be expected for the full year or any other period. The following summary consolidated financial data should be read in conjunction with the section titled “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and our consolidated financial statements and related notes included elsewhere in this prospectus.
| Year Ended December 31, | Six Months Ended June 30, | |||||||||||||||||||
| 2012 | 2013 | 2014 | 2014 | 2015 | ||||||||||||||||
| (in thousands, except share and per share amounts) | ||||||||||||||||||||
| Consolidated Statement of Operations Data: |
||||||||||||||||||||
| Revenue |
$ | 73,141 | $ | 88,848 | $ | 125,510 | $ | 57,643 | $ | 81,263 | ||||||||||
| Cost of revenue |
24,178 | 30,972 | 42,668 | 19,489 | 27,160 | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Gross profit |
48,963 | 57,876 | 82,842 | 38,154 | 54,103 | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Operating expenses: |
||||||||||||||||||||
| Research and development |
12,548 | 14,084 | 15,575 | 7,538 | 7,983 | |||||||||||||||
| Selling, general and administrative |
32,987 | 44,918 | 64,258 | 28,240 | 45,943 | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Total operating expenses |
45,535 | 59,002 | 79,833 | 35,778 | 53,926 | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Income (loss) from operations |
3,428 | (1,126 | ) | 3,009 | 2,376 | 177 | ||||||||||||||
| Interest income (expense), net |
244 | 345 | 439 | 39 | 385 | |||||||||||||||
| Other income (expense), net |
220 | (474 | ) | (309 | ) | (92 | ) | (498 | ) | |||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Income (loss) before provision for (benefit from) income taxes |
3,892 | (1,255 | ) | 3,139 | 2,323 | 64 | ||||||||||||||
| Provision for (benefit from) income taxes |
1,934 | (5,354 | ) | 894 | 666 | 233 | ||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Net income (loss) |
$ | 1,958 | $ | 4,099 | $ | 2,245 | $ | 1,657 | $ | (169 | ) | |||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Net income (loss) attributable to common stockholders |
$ | 412 | $ | 887 | $ | (833 | ) | $ | 355 | $ | (34 | ) | ||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Net income (loss) per share attributable to common stockholders |
||||||||||||||||||||
| Basic |
$ | 0.10 | $ | 0.21 | $ | (0.18 | ) | $ | 0.08 | $ | (0.01 | ) | ||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Diluted |
$ | 0.07 | $ | 0.14 | $ | (0.18 | ) | $ | 0.05 | $ | (0.01 | ) | ||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Weighted average shares used to compute net income (loss) per share attributable to common stockholders |
||||||||||||||||||||
| Basic |
4,153,121 | 4,304,396 | 4,609,375 | 4,520,898 | 5,000,375 | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Diluted |
5,886,126 | 6,500,835 | 4,609,375 | 6,743,140 | 5,000,375 | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Pro forma net income (loss) per share — Basic |
$ | 0.10 | $ | (0.01 | ) | |||||||||||||||
|
|
|
|
|
|||||||||||||||||
| Pro forma net income (loss) per share — Diluted |
$ | 0.09 | $ | (0.01 | ) | |||||||||||||||
|
|
|
|
|
|||||||||||||||||
| Weighted average shares used to compute the pro forma net income (loss) per share |
||||||||||||||||||||
| —Basic (unaudited) |
22,680,810 | 24,510,785 | ||||||||||||||||||
|
|
|
|
|
|||||||||||||||||
| —Diluted (unaudited) |
25,037,541 | 24,510,785 | ||||||||||||||||||
|
|
|
|
|
|||||||||||||||||
8
| As of June 30, 2015 | ||||||||||||
| Actual | Pro Forma(1) | Pro Forma As Adjusted(2)(3) |
||||||||||
| (in thousands) | ||||||||||||
| Consolidated Balance Sheet Data: |
||||||||||||
| Cash and cash equivalents |
$ | 36,764 | $ | 36,764 | $ | |||||||
| Total assets |
$ | 129,070 | $ | 129,070 | $ | |||||||
| Long-term debt |
$ | — | $ | — | $ | |||||||
| Working capital |
$ | 91,298 | $ | 91,298 | $ | |||||||
| Preferred stock |
$ | 111,467 | $ | — | $ | |||||||
| Total stockholders’ equity (deficit) |
$ | (11,099 | ) | $ | 100,368 | $ | ||||||
| (1) | The pro forma column reflects (i) the automatic conversion of all outstanding shares of our preferred stock into an aggregate of 19,510,410 shares of our common stock, which conversion will occur immediately prior to the completion of this offering, as if such conversion had occurred on June 30, 2015, and (ii) the filing and effectiveness of our amended and restated certificate of incorporation. |
| (2) | The pro forma as adjusted column gives effect to (a) the pro forma adjustments set forth in (1) above and (b) the sale and issuance by us of shares of our common stock in this offering, assuming an initial public offering price of $ per share, which is the midpoint of the estimated initial public offering price range set forth on the cover page of this prospectus, after deducting underwriting discounts and commissions and estimated offering expenses payable by us. |
| (3) | Each $1.00 increase or decrease in the assumed initial public offering price of $ per share, which is the midpoint of the estimated initial public offering price range set forth on the cover page of this prospectus, would increase or decrease, as applicable, the amount of our cash and cash equivalents, total assets, working capital and stockholders’ equity by $ million, assuming that the number of shares offered by us, as set forth on the cover page of this prospectus, remains the same, after deducting underwriting discounts and commissions payable by us. An increase or decrease of 1.0 million shares in the number of shares offered by us would increase or decrease, as applicable, the amount of our cash and cash equivalents, total assets, working capital and stockholders’ equity by $ million, assuming an initial public offering price of $ per share, which is the midpoint of the estimated initial public offering price range set forth on the cover page of this prospectus, after deducting underwriting discounts and commissions payable by us. |
9
You should carefully consider the risks described below before making an investment decision. Additional risks not presently known to us or that we currently deem immaterial may also impair our business. Our business could be harmed by any of these risks. The trading price of our common stock could decline due to any of these risks, and you may lose all or part of your investment. In assessing these risks, you should also refer to the other information contained in this prospectus, including the section titled “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and our consolidated financial statements and related notes, before making a decision to invest in our common stock.
Business Risks
We have a limited operating history and may not be able to sustain or grow our profitability or generate positive cash flows from operations.
We were founded in 2004 and did not generate any revenue until 2007. Moreover, while we have successfully developed, obtained regulatory clearance or approval for, and introduced a number of products in the neuro market since 2007, we first introduced products in the peripheral vascular and neurosurgical markets in 2013 and 2014, respectively. Accordingly, we only have a limited operating history upon which investors can evaluate our business and prospects, and this limited operating history may not be indicative of our future results. Since 2009, we have been generally profitable on an annual basis; however, we incurred operating losses in 2013, and we are not currently cash-flow positive. We can give no assurance that we will be profitable or cash-flow positive in the future.
We expect that our general and administrative and sales and marketing expenses will increase to support our anticipated growth as well as the additional operational and reporting costs associated with being a public company. We have also expended significant amounts on research and development to develop and fund clinical testing of our products, and we expect to continue to do so. We also expend significant amounts on maintaining inventory levels of raw materials, components and finished products to meet anticipated customer demand. In addition, our coil products are sold on a consignment basis, which requires us to expend significant amounts on inventory that is placed at many customer locations. Our ability to sustain our growth and profitability and become cash-flow positive may be influenced by many factors, including:
| • | our ability to achieve and maintain market acceptance of our products; |
| • | unanticipated problems and additional costs relating to the development and testing of new products; |
| • | our ability to introduce, manufacture at scale and commercialize new products; |
| • | our ability to produce sufficient quantities of our products to meet demand and to smoothly transition to new products; |
| • | the impact of competition; |
| • | the timing and impact of market and regulatory developments; |
| • | our ability to expand into new markets; |
| • | pricing pressure from competitors; |
| • | the availability and adequacy of third-party reimbursement for procedures in which our products are used; and |
| • | our ability to obtain and maintain adequate intellectual property protection for our products and technologies. |
If we encounter difficulties with any of the foregoing or unexpected expenses, it could materially adversely affect our business, results of operations, financial condition or cash flows.
10
Our existing products may be rendered obsolete and we may be unable to effectively introduce and market new products or may fail to keep pace with advances in technology.
The medical device market is characterized by rapidly advancing technology. Our success depends, in part, on our ability to anticipate technological advancements and competitive innovations and introduce new products to adapt to these advancements and innovations. To compete in the marketplace, we have made, and we must continue to make, substantial investments in new product development, whether internally through research and development or externally through licensing or acquisitions. We can give no assurance that we will be successful in identifying, developing or acquiring, and marketing new products or enhancing our existing products. In addition, we can give no assurance that new products or alternative treatment techniques developed by competitors will not render our current or future products obsolete or inferior, technologically or economically.
The success of any new products that we develop or acquire depends on achieving and maintaining market acceptance. Market acceptance for our current and new products could be affected by a number of factors, including:
| • | our ability to market and distribute our products effectively; |
| • | the availability, perceived efficacy and pricing of alternative products from our competitors; |
| • | the development of new products or alternative treatments by others that render our products and technologies obsolete; |
| • | the price, quality, effectiveness and reliability of our products; |
| • | our customer service and reputation; |
| • | our ability to convince specialist physicians to use our products on their patients; and |
| • | the timing of market entry of new products or alternative treatments. |
Our competition may respond more quickly to new or emerging technologies or a changing clinical landscape, undertake more extensive marketing campaigns, have greater financial, marketing and other resources than us or be more successful in attracting potential customers and strategic partners. Given these factors, we cannot assure you that we will be able to continue or increase our level of success. Our failure to introduce new and innovative products in a timely manner, and our inability to maintain or grow the market acceptance of our existing products, could result in permanent write-downs or write-offs of our inventory and otherwise have a material and adverse effect on our business, results of operations, financial condition or cash flows.
Delays in product introductions could adversely affect our business, results of operations, financial condition or cash flows.
The medical device market is highly competitive and designs change often to adjust to shifting market preferences and other factors. Therefore, product life cycles are relatively short. As a result, any delays in our product launches may significantly impede our ability to enter or compete in a given market and may reduce the sales that we are able to generate from these products. We may experience delays in any phase of a product launch, including during research and development, clinical trials, regulatory review, manufacturing and marketing. Delays in product introductions could materially adversely affect our business, results of operations, financial condition or cash flows.
We face significant competition, and if we are unable to compete effectively, we may not be able to achieve or maintain significant market penetration or improve our results of operations.
The medical device industry is intensely competitive, subject to rapid change and significantly affected by new product introductions and other market activities of industry participants. We compete with a number of manufacturers and distributors of neuro and peripheral vascular devices. Our most notable competitors are Boston Scientific, Johnson & Johnson, Medtronic, Stryker and Terumo. All of these competitors are large, well-capitalized companies with longer operating histories and significantly greater resources than us. We also
11
compete with a number of smaller medical device companies that have a single product or a limited range of products. Our competitors may be able to spend more on product development, marketing, sales and other product initiatives, or be more focused in their spending and activities, than we can. Some of our competitors have:
| • | significantly greater name recognition; |
| • | broader or deeper relations with healthcare professionals, customers and third-party payors; |
| • | more established distribution networks; |
| • | additional lines of products and the ability to offer rebates or bundle products to offer greater discounts or other incentives to gain a competitive advantage; |
| • | greater experience in conducting research and development, manufacturing, clinical trials, marketing and obtaining regulatory clearance or approval for products; and |
| • | greater financial and human resources for product development, sales and marketing and patent litigation. |
We compete primarily on the basis that our products are able to treat patients with neurovascular and peripheral vascular diseases and disorders safely and effectively, with improved outcomes and procedural cost savings. Our continued success depends on our ability to:
| • | develop innovative, proprietary products that can cost-effectively address significant clinical needs; |
| • | continue to innovate and develop scientifically advanced technology; |
| • | obtain and maintain regulatory clearances or approvals; |
| • | demonstrate efficacy in Penumbra-sponsored and third-party clinical trials and studies; |
| • | apply technology across product lines and markets; |
| • | attract and retain skilled research and development and sales personnel; and |
| • | cost-effectively manufacture and successfully market and sell products. |
We cannot assure you that we will be able to compete effectively on the basis of these factors. Additionally, our competitors with greater financial resources could acquire or develop new technologies or products that effectively compete with our existing or future products. If we are unable to effectively compete, it would materially adversely affect our business, results of operations, financial condition and cash flows.
Our future growth depends, in part, on our ability to further penetrate our current customer base and increase the frequency of use of our products by our customers.
We will need to continue to make specialist physicians and other hospital staff aware of the benefits of our products to generate increased demand and frequency of use, and thus increase sales to our hospital customers. Although we are attempting to increase the number of patients treated with procedures that use our products through our established relationships and focused sales efforts, we cannot provide assurance that our efforts will increase the use of our products. If we are unable to increase the frequency of use of our products by specialist physicians, this could materially adversely affect our business, results of operations, financial condition or cash flows.
Our future growth depends, in part, on significantly expanding our user base to include additional specialist physicians in both our existing and future target end markets.
Currently, the primary users of our neuro products are neuro interventionalists who perform endovascular neuro interventions. We also began selling in the peripheral vascular market in 2013 with the introduction of our Ruby Coil and the neurosurgery market in 2014 with the introduction of our Apollo System, and we may enter new target end markets in the future. Our revenue growth will depend in part on our ability to convince
12
specialist physicians in our existing and future target end markets of our products’ efficacy, to educate them in the proper use of our products and to sell our products to their affiliated hospitals. Convincing specialist physicians to use new products and to dedicate the time and energy necessary for adequate education in the use of our products is challenging, especially in new markets where treatments using our products are not established. Expanding our customer base in existing or new target end markets may require, among other things, additional clinical evidence supporting patient benefits, training in a manner to which we are not accustomed, or other resources that we do not readily have available or are not cost effective for us to provide. If we are unable to convert specialist physicians in existing or new target end markets to the use of our products, our sales growth will be limited, which could materially adversely affect our business, results of operations, financial condition or cash flows.
The marketing and sales of our products require a significant amount of time and expense and we may not have the resources to successfully market and sell our products, which would adversely affect our business and results of operations.
The marketing and sales of our products requires us to invest in training and education and employ a salesforce that is large enough to interact with the specialist physicians who use our products. Entering new markets also requires a significant amount of time and expense in order to identify and establish relationships with key opinion leaders among the specialist physicians who may use our products in those markets. We may not have adequate resources to market and sell our products successfully against larger competitors. For example, when we began selling in the peripheral vascular market in 2013, we did not have a dedicated direct peripheral vascular sales team and our neuro sales team was required to dedicate a portion of its efforts to the sales of our peripheral vascular products. We subsequently expended significant sums to develop a direct salesforce focused on peripheral vascular product sales. If we do not have adequate resources to market and sell our products effectively, or cannot otherwise market and sell our products successfully, it could materially adversely affect our business, results of operations, financial condition or cash flows.
Third-party reimbursement may not be available or adequate for the procedures in which our products are used.
Our ability to commercialize new products successfully in both the United States and international markets depends in part on the availability of, and hospitals’ ability to obtain, adequate levels of third-party reimbursement for the procedures in which our products are used. In the United States, the cost of medical care is funded, in substantial part, by government insurance programs, such as Medicare and Medicaid, and private and corporate health insurance plans. Third-party payors may deny reimbursement if they determine that a device used in a procedure has not received appropriate FDA or other governmental regulatory clearances or approvals, is not used in accordance with cost-effective treatment methods as determined by the payor, or is experimental, unnecessary or inappropriate. Our ability to commercialize our products successfully will depend, in large part, on the extent to which adequate reimbursement levels for the cost of their use are obtained from government authorities, private health insurers and other organizations, such as health maintenance organizations. Further, healthcare in the United States and international markets is also being affected by economic pressure to contain reimbursement levels and costs. Changing reimbursement models could materially adversely affect our business, results of operations, financial condition or cash flows.
We have generated a significant portion of our revenue from products that are used in connection with the treatment of neurovascular diseases, and our revenue and business prospects would be adversely affected if our neuro product sales were to decline.
We have generated most of our revenue from our neurovascular products, including our Penumbra System, Penumbra Coil 400 and Neuron products. If any one or more of these products, or any successor products, were no longer available for sale in any key market because of regulatory, third-party reimbursement or intellectual property issues or any other reason, or if one of our competitors introduced one or more products that specialist physicians believe are superior to our products, our revenue from these products would
13
decline. A significant decline in our sales of neurovascular products could also negatively impact our financial condition and our ability to conduct product development activities, and therefore negatively impact our business prospects.
We must maintain and further develop relationships with specialist physicians. If specialist physicians do not recommend and endorse, or use, our products or if our relationships with specialist physicians deteriorate, our products may not be accepted or maintain acceptance in the marketplace, which would adversely affect our business and results of operations.
Our products are sold to hospitals for use by specialist physicians practicing at their facilities. In order for us to sell our products, specialist physicians must recommend and endorse them for the hospital to purchase them, and must use them in treating their patients to generate follow-on sales. We may not obtain the necessary recommendations or endorsements for new products from specialist physicians, nor may we be able to maintain the current or future level of acceptance and usage of our products. Acceptance of our products depends on educating the medical community as to the distinctive characteristics, perceived benefits, safety, clinical efficacy and cost-effectiveness of our products compared to products of our competitors or treatments that do not use our products, and on training specialist physicians in the proper application and use of our products. We invest in significant training and education of our sales representatives and specialist physicians to achieve market acceptance of our products, with no assurance of success. If we are not successful in obtaining and maintaining the recommendations or endorsements of specialist physicians for our products, if specialist physicians prefer our competitors’ products or other alternative treatments that do not use our products, or if our products otherwise do not gain or maintain market acceptance, our business could be adversely affected.
In addition, the research, development, marketing and sales of our products are dependent, in part, upon our working relationships with specialist physicians. We rely on them to provide us with knowledge and feedback regarding our products and the marketing of our products. If we are unable to develop or maintain strong relationships with specialist physicians and receive their advice and input, the development and marketing of our products could suffer, which could materially adversely affect our business, results of operations, financial condition or cash flows.
We may not be able to achieve or maintain satisfactory pricing and margins for our products.
Manufacturers of medical devices have a history of price competition, and we can give no assurance that we will be able to achieve satisfactory prices for our products or maintain prices at the levels we have historically achieved. If we are unable to achieve or maintain our prices, or if our costs increase and we are unable to offset such increase with an increase in our prices, our margins could erode and we may be unable to maintain profitable operations.
We cannot be certain that we will be able to manufacture our products in high volumes at commercially reasonable costs.
We currently maintain our manufacturing operations in buildings located at our campus in Alameda, California. We currently produce substantially all of our products at this facility, and we do not have redundant facilities. We may need to expend significant capital resources and increase the size of our manufacturing capabilities as we grow our business. We could, however, encounter problems related to:
| • | capacity constraints; |
| • | production yields; |
| • | quality control; |
| • | equipment availability; and |
| • | shortages of qualified personnel. |
14
Our continuous product innovation limits our ability to identify and implement manufacturing efficiencies. Failure to do so may reduce our ability to manufacture our products at commercially reasonable costs. If we are unable to manufacture our products in high volumes at commercially reasonable costs, it could materially affect our ability to adequately increase production of our products and fulfill customer orders on a timely basis, which could have a material adverse effect on our business, results of operations, financial condition or cash flows.
We are required to maintain high levels of inventory, which consume a significant amount of our working capital and could lead to permanent write-downs or write-offs of our inventory.
We maintain a significant inventory of raw materials, components and finished goods, which subjects us to a number of risks and challenges. Our hospital customers typically maintain only small quantities of our products at their facilities, so as products are used, they order replacements that typically require prompt delivery. As a result, we must maintain sufficient levels of finished goods to permit rapid shipment of products following receipt of a customer order. In turn, we must also maintain a sufficient supply of raw materials and components inventory to permit rapid manufacturing and re-stocking of finished goods. Furthermore, our coil inventory is supplied to hospital customers on a consignment basis, which means that it is classified as part of our inventory for financial reporting purposes but is maintained at the hospital location until it is used.
Maintaining a significant inventory of raw materials, components and finished goods consumes a significant amount of our working capital. This working capital could be used for other purposes, such as research and development or sales and marketing activities. As we grow our business, we may need substantial additional capital to fund higher levels of inventory, which may materially adversely affect our liquidity or result in dilution to our stockholders if we sell additional equity securities or leverage if we raise debt capital to finance our working capital requirements.
Maintaining a significant inventory of raw materials, components and finished goods also subjects us to the risk of inventory excess and obsolescence, which may lead to a permanent write-down or write-off of our inventory. While in inventory, our components and finished goods may become obsolete, and we may over-estimate the amount of inventory needed, which may lead to excessive inventory. In these circumstances, we would write-down or write-off our inventory, and we may be required to expend additional resources or be constrained in the amount of end product that we can produce. Furthermore, our products have a limited shelf life due to sterilization requirements, and part or all of a given product or component may expire, resulting in a decrease in value and potentially a permanent write-down of our inventory. For example, we recorded write downs of $0.9 million and $1.9 million for excess and obsolete inventory in 2013 and 2014, respectively. In the event that a substantial portion of our inventory becomes excess or obsolete, it could materially adversely affect our results of operations.
Defects or failures associated with our products could lead to recalls, safety alerts or litigation, as well as significant costs and negative publicity.
Manufacturing flaws, component failures, design defects, off-label uses or inadequate disclosure of product-related information could result in an unsafe condition or the injury or death of a patient. These problems could lead to a recall of, or issuance of a safety alert relating to, our products and result in significant costs, negative publicity and adverse competitive pressure. While we have had product recalls, they have all been voluntary, based on our own internal safety and quality monitoring and testing data, and none of our past product recalls has been material. The circumstances giving rise to recalls are, however, unpredictable, and any future recalls of existing or future products could materially adversely affect our business, results of operations, financial condition or cash flows.
The medical device industry has historically been subject to extensive litigation over product liability claims. There are high rates of mortality and other complications associated with some of the medical conditions suffered by the patients whom specialist physicians use our devices to treat, and we may be subject to product liability claims if our products cause, or merely appear to have caused, an injury or death. In addition, an injury or death that is caused by the activities of our suppliers, such as those that provide us with
15
components and raw materials, or by an aspect of a treatment used in combination with our products, such as a complementary drug or anesthesia, may be the basis for a claim against us by patients, hospitals, health-care providers or others purchasing or using our products, even if our products were not the actual cause of such injury or death. An adverse outcome involving one of our products could result in reduced market acceptance and demand for all of our products, and could harm our reputation and our ability to market our products in the future. In some circumstances, adverse events arising from or associated with the design, manufacture or marketing of our products could result in the suspension or delay of regulatory reviews of our premarket notifications or applications for marketing. Any of the foregoing problems could disrupt our business and have a material adverse effect on our business, results of operation, financial condition or cash flows.
Although we carry product liability insurance in the United States and in other countries in which we conduct business, including for clinical trials and product marketing, we can give no assurance that such coverage will be available or adequate to satisfy any claims. Product liability insurance is expensive, subject to significant deductibles and exclusions, and may not be available on acceptable terms, if at all. If we are unable to obtain or maintain insurance at an acceptable cost or on acceptable terms with adequate coverage or otherwise protect against potential product liability claims, we could be exposed to significant liabilities. A product liability claim, recall or other claim with respect to uninsured liabilities or for amounts in excess of insured liabilities could materially adversely affect our business, financial condition and results of operations. Defending a suit, regardless of its merit or eventual outcome, could be costly, could divert management’s attention from our business and might result in adverse publicity, which could result in reduced acceptance of our products in the market, product recalls or market withdrawals.
Our products are continually the subject of clinical trials conducted by us, our competitors, or other third parties, the results of which may be unfavorable, or perceived as unfavorable, and which could materially adversely affect our business, financial condition and results of operations.
As a part of the regulatory process of obtaining marketing clearance or approval for new products and new indications for existing products, as well as to provide specialist physicians with ongoing information regarding the efficacy of our products, we conduct and participate in numerous clinical trials with a variety of study designs, patient populations and trial endpoints. Our competitors and third parties also conduct clinical trials of our products without our participation. Unfavorable or inconsistent clinical data from existing or future clinical trials conducted by us, our competitors or third parties, or the market’s or regulators’ perception of clinical data, could materially adversely affect our business, results of operations, financial condition or cash flows.
Our future success depends in part upon establishing an interventional stroke care pathway in the United States that integrates the use of endovascular thrombectomy into the treatment of ischemic stroke.
The stroke care pathway in the United States generally begins with emergency responders who are responsible for transporting the patient to a hospital facility. With a small number of exceptions (such as for trauma), emergency responders in the United States generally operate under a protocol that transports patients to the nearest hospital, which decreases the likelihood that the patient will be transported to a stroke center that has a developed stroke team and an interventional approach to the treatment of stroke. Further, there is no agreed upon standard of care among physicians or hospitals regarding the treatment of ischemic stroke patients, and treatment protocols vary according to the particular hospital, often resulting in significant delays and gaps in patients being assessed for and receiving interventional treatment. The absence of a uniform protocol among hospitals and among physicians within the same hospital means that we have to educate each hospital and stroke center about protocols that integrate our products for the treatment of stroke.
We believe that the stroke care system in the United States has not been historically geared towards interventional treatment of stroke due to the absence of clinical evidence that interventional techniques were effective. Our and our competitors’ ability to alter the existing stroke care pathway may depend on whether we and our competitors are successful in using recent positive clinical studies to convince specialist physicians that intervention yields superior clinical results relative to cases where intervention is not used.
16
Establishing an interventional stroke pathway that integrates the use of interventional treatments, including our products, will depend upon many factors, including:
| • | continuing to educate hospitals and specialist physicians about the clinical evidence supporting intervention, as well as the use, benefits and cost-effectiveness of our products; |
| • | improving the speed with which patients are assessed for and receive interventional treatments; and |
| • | increasing the likelihood that patients are transported to a hospital or stroke center where interventional treatments are available. |
Even if these efforts are successful, it may be years before existing systems and care pathways are changed. These factors may make it difficult to grow our business.
Any data that is gathered in the course of clinical trials may be significantly more favorable than the typical results achieved by practicing specialist physicians, which could negatively impact rates of adoption of our products.
Even if the data collected from clinical trials indicates positive results, each specialist physician’s actual experience with our products will vary. Clinical trials often involve procedures performed by specialist physicians who are technically proficient and high volume users. Consequently, the results reported in clinical trials may be significantly more favorable than typical results of other users. If specialist physicians’ experiences indicate, or they otherwise believe, that our products are not as safe or effective as other treatment options with which they are more familiar, or clinical trial data indicates the same, adoption of our products may suffer, which could materially adversely affect our business, results of operations, financial condition or cash flows.
Negative publicity regarding our products or marketing tactics by competitors could reduce demand for our products, which would adversely affect sales and our financial performance.
We may experience, from time to time, negative exposure in clinical publications or in marketing campaigns of our competitors. Such publications or campaigns may present negative individual physician experience regarding the safety or effectiveness of our products or may suggest our competitors’ products are superior to ours, based on studies or clinical trials conducted or funded by competitors or that involved competitive products.
Our reputation and competitive position may also be harmed by other publicly available information suggesting that our products are not safe. For example, we file adverse event reports under Medical Device Reporting, or MDR, obligations with the FDA that are publicly available on the FDA’s website. We are required to file MDRs if our products may have caused or contributed to a serious injury or death or malfunctioned in a way that could likely cause or contribute to a serious injury or death if it were to recur. Any such MDR that reports a significant adverse event could result in negative publicity and could harm our reputation and future sales.
Our dependence on key suppliers puts us at risk of interruptions in the availability of our products, which could reduce our revenue and adversely affect our results of operations. In addition, increases in prices for raw materials and components used in our products could adversely affect our results of operations.
We require the timely delivery of sufficient amounts of components and materials to manufacture our products. For reasons of quality assurance, cost effectiveness or availability, we procure certain raw materials and components from a single or limited number of suppliers. We generally acquire such raw materials and components through purchase orders placed in the ordinary course of business, and as a result we may not have a significant inventory of these materials and components and generally do not have any guaranteed or contractual supply arrangements with many of these suppliers. Our reliance on these suppliers subjects us to risks that could harm our business, including, but not limited to, difficulty locating and qualifying alternative suppliers.
17
Our dependence on third-party suppliers involves several other risks, including limited control over pricing, availability, quality and delivery schedules. Suppliers of raw materials and components may decide, or be required, for reasons beyond our control, to cease supplying raw materials and components to us or to raise their prices. Shortages of raw materials, quality control problems, production capacity constraints or delays by our suppliers could negatively affect our ability to meet our production requirements and result in increased prices for affected materials or components. We may also face delays, yield issues and quality control problems if we are required to locate and secure new sources of supply. While we have not experienced any to date, any material shortage, constraint or delay may result in delays in shipments of our products, which could materially adversely affect our results of operations. Increases in prices for raw materials and components used in our products could also materially adversely affect our results of operations.
In addition, the FDA and regulators outside of the United States may require additional testing of any raw materials or components from new suppliers prior to our use of these materials or components. In the case of a device with clearance under section 510(k) of the U.S. Federal Food, Drug, and Cosmetic Act, referred to as a 510(k), we may be required to submit a new 510(k) if a change in a raw material or component supplier results in a change in a material or component supplied that is not within the 510(k) cleared device specifications. If we need to establish additional or replacement suppliers for some of these materials or components, our access to the materials or components might be delayed while we qualify such suppliers and obtain any necessary FDA approvals or clearances. Our suppliers may also be subject to regulatory inspection and scrutiny. Any adverse regulatory finding or action against those suppliers could impact their ability to supply us with raw materials and components for our products.
Our corporate culture has contributed to our success, and if we cannot maintain this culture as we grow, we could lose the innovative approach, creativity, and teamwork fostered by our culture, and our business may be harmed.
We believe that a critical contributor to our success has been our corporate culture, which we believe fosters innovation, teamwork, and a focus on execution, as well as facilitating critical knowledge transfer and knowledge sharing. As we grow, we may find it difficult to maintain these important aspects of our corporate culture, which could limit our ability to innovate and operate effectively. Any failure to preserve our culture could also negatively affect our ability to retain and recruit personnel or execute on our business strategy.
If our facilities were to become inoperable, we would be unable to continue to develop and manufacture our products until we were able to restore full research, manufacturing and administrative capabilities at our facilities or secure a new facility, and as a result, our business would be harmed.
We currently maintain our research and development, manufacturing and administrative operations in buildings located at our campus in Alameda, California, and we do not have redundant facilities. Alameda is situated on or near earthquake fault lines, and our facilities are built on filled land, which could be prone to liquefaction in a major earthquake. Should one or more of our buildings be significantly damaged or destroyed by natural or man-made disasters, such as earthquakes, fires or other events, it could take months to relocate or rebuild, during which time our employees may seek other positions, our research, development and manufacturing would cease or be delayed and our products may be unavailable. Moreover, because of the time required to approve and license a manufacturing facility under FDA and non-U.S. regulatory requirements, we may not be able to resume production on a timely basis even if we are able to replace production capacity in the event we lose manufacturing capacity. While we maintain property and business interruption insurance, such insurance has limits and would only cover the cost of rebuilding and relocating and lost profits, but not losses we may suffer due to our products being replaced by competitors’ products. The inability to perform our research, development and manufacturing activities, combined with our limited inventory of raw materials and components and manufactured products, may cause specialist physicians to discontinue using our products or harm our reputation, and we may be unable to reestablish relationships with those specialist physicians in the future. Consequently, a catastrophic event at our facility could materially adversely affect our business, results of operations, financial condition or cash flows.
18
To successfully market and sell our products internationally, we must address a number of unique challenges applicable to international markets.
For 2014 and the six months ended June 30, 2015, we derived 34% and 34%, respectively, of our revenue from international sales. International sales are subject to a number of risks and challenges, including:
| • | reliance on distributors; |
| • | varying coverage and reimbursement policies, processes and procedures; |
| • | difficulties in staffing and managing international operations from which sales are conducted; |
| • | difficulties in penetrating markets in which our competitors’ products or alternative procedures that do not use our products are more established; |
| • | reduced protection for intellectual property rights in some countries; |
| • | export licensing requirements or restrictions, trade regulations and foreign tax laws; |
| • | fluctuating foreign currency exchange rates; |
| • | foreign certification, regulatory requirements and legal requirements; |
| • | lengthy payment cycles and difficulty in collecting accounts receivable; |
| • | customs clearance and shipping delays; |
| • | pricing pressure in international markets; |
| • | political and economic instability; and |
| • | preference for locally produced products. |
If we are unable to successfully address these challenges, we may not be able to grow our international sales and our results of operations may suffer as a result.
Over the long term, we intend to grow our business internationally and to do so, we will need to either spend substantial sums to expand or develop direct sales capabilities in existing and new geographic areas or generate additional sales through existing distributors or attract additional distributors.
As a result of our international operations, we are required to comply with tax requirements in multiple jurisdictions, the scope and impact of which may be unclear. Moreover, tax authorities in jurisdictions in which we do business could disagree with tax positions that we take, including, for example, our inter-company pricing policies, or could assert that we owe more taxes than we currently pay due to the level and nature of our activities in such jurisdictions.
We rely on our distributors to market and sell our products in certain international markets.
We have established a direct sales capability in the United States, most of Europe, Canada and Australia, which we have complemented with distributors in Japan and certain other international markets. Sales to distributors represented 17.7% and 17.7% of our revenue in 2014 and the six months ended June 30, 2015, respectively. In addition, sales to our Japanese distributor, Medico’s Hirata Inc., represented approximately 11.7% of our revenue in 2014. Our success outside of the United States, most of Europe, Canada and Australia depends largely upon marketing arrangements with distributors, in particular their sales expertise and their relationships with specialist physicians and affiliated hospitals in their geographic areas. Distributors may terminate their relationship with us, sell competitive products or devote insufficient sales efforts or other resources to our products. We do not control our distributors, and they may not be successful in implementing our marketing plans. In addition, many of our distributors initially obtain and maintain foreign regulatory approval for the sale of our products in their respective countries, and their efforts in obtaining and maintaining regulatory approval may not be as robust as we desire or expect. Our failure to maintain our existing relationships with our distributors, or our failure to recruit and retain additional skilled distributors in
19
existing or new international markets, could have an adverse effect on our operations. If current or future distributors do not perform adequately, or if we lose a significant distributor, such as our Japanese distributor, we may not be able to maintain existing levels of international revenue or realize expected long term international revenue growth. We have also experienced turnover with some of our distributors in the past that has adversely affected sales in the countries in which those distributors operate. Similar occurrences could happen in the future.
Most of our customer relationships outside of the United States are with governmental entities, and we could be materially adversely affected by violations of the U.S. Foreign Corrupt Practices Act and similar anti-bribery laws in non-U.S. jurisdictions.
The FCPA and similar anti-bribery laws in non-U.S. jurisdictions generally prohibit companies and their intermediaries from making improper payments to non-U.S. officials for the purpose of obtaining or retaining business. Because of the predominance of government-sponsored healthcare systems around the world, most of our customer relationships outside of the United States are with governmental entities, and physicians practicing in those systems are considered “government officials;” our sale to these entities are therefore subject to such anti-bribery laws. Our policies mandate compliance with these anti-bribery laws. We operate in many parts of the world that have experienced governmental corruption, and in certain circumstances strict compliance with anti-bribery laws may be at variance with local customs and practices. Despite our training and compliance programs, our internal control policies and procedures may not always protect us from reckless or criminal acts committed by our employees, distributors or agents. Violations of the FCPA or other anti-bribery laws, or allegations of such violations, could disrupt our business and materially adversely affect our business, results of operations, financial condition or cash flows.
Foreign currency exchange rates may adversely affect our results.
We are exposed to the effects of changes in foreign currency exchange rates, and we have not historically hedged our foreign currency exposure. Approximately 34% and 34% of our revenue for 2014 and the six months ended June 30, 2015, respectively, were derived from sales in non-U.S. markets, and we expect sales from non-U.S. markets to continue to represent a significant portion of our revenue. For direct sales in our international markets, we are paid by our customers in their local currency, which is primarily euros. For sales to distributors in our international markets, we are paid in either U.S. dollars, euros or Japanese yen. Therefore, when the U.S. dollar strengthens relative to the euro, yen or other local currency, as it has in recent periods, our U.S. dollar reported revenue from non-U.S. dollar denominated sales will decrease, or we will need to increase our non-U.S. dollar denominated prices, which may not be commercially practical. Conversely, when the U.S. dollar weakens relative to the euro, yen or other local currency, our U.S. dollar reported expenses from non-U.S. dollar denominated operating costs will increase. Changes in the relative values of currencies occur regularly and, in some instances, could materially adversely affect our business, results of operations, financial condition or cash flows.
We have experienced rapid growth in recent periods, and if we fail to manage our growth effectively, our business and results of operations may suffer.
We have significantly expanded our overall business, research and development, customer base, product portfolio, employee headcount and operations in recent periods. We have also established new operations in other countries. We have increased our total number of full-time employees from 468 as of December 31, 2013, to approximately 1,000 as of June 30, 2015. Our expansion has placed, and our expected future growth will continue to place, a significant strain on our managerial, operational, product development, sales and marketing, administrative, financial and other resources. More systems, facilities, processes and management employees are needed to allow us to continue to grow successfully. We also plan to continue to increase our salesforce. Our experience has been that it takes at least six months, and often longer, before new sales personnel generate enough sales to cover their costs, resulting in increased costs without offsetting revenue during periods in which we are increasing the size of our salesforce. To meet anticipated demand for our products, we will also have to obtain additional space, buy additional equipment and hire additional research
20
and development and manufacturing employees, including quality control personnel and other personnel involved in the production process. If we are unable to manage our growth successfully, it could have a material and adverse effect on our business, results of operations, financial condition or cash flows.
We depend on key personnel to operate our business and develop our products, and if we are unable to retain, attract and integrate qualified personnel, our ability to develop and successfully grow our business could be harmed.
We believe that our future success is highly dependent on the contributions of our executive officers, particularly our chief executive officer, as well as our ability to attract and retain highly skilled and experienced sales and marketing, technical and other personnel in the United States and in international markets. Each of these persons’ efforts will be critical to us as we continue to develop our products and business. If we were to lose one or more of our key employees, including to competitors, we may experience difficulties in competing effectively, developing our products and implementing our business strategies.
Our research and development and sales and marketing programs depend on our ability to attract and retain highly skilled technicians, engineers and salespeople. We may not be able to attract or retain qualified employees in the future due to the intense competition for qualified personnel among life science businesses, particularly in the San Francisco Bay Area, where our corporate headquarters, research and development and manufacturing facilities are located. If we are not able to identify, recruit and retain highly qualified personnel, we may experience constraints that will adversely affect our ability to support our research, development, manufacturing and sales programs, and ultimately our ability to compete. If we are unable to identify, recruit and retain qualified salespeople, there could be a delay or decline in the adoption of our products. If key personnel were to leave Penumbra, either to join our competitors or otherwise, we may not be able to attract and retain equally qualified personnel to replace them.
We depend on information technology systems to operate our business and a cyber-attack or other breach of these systems could have a material adverse effect on our business.
We rely on information technology systems to process, transmit and store electronic information in our day-to-day operations. Our information technology systems are vulnerable to a cyber-attack, malicious intrusion, breakdown, destruction, loss of data privacy or other significant disruption. Any such successful attacks could result in the theft of intellectual property or other misappropriation of assets, or otherwise compromise our confidential or proprietary information and disrupt our operations. Cyber-attacks are becoming more sophisticated and frequent, and our systems could be the target of malware and other cyber-attacks. We have invested in our systems and the protection of our data to reduce the risk of an intrusion or interruption, and we monitor our systems on an ongoing basis for any current or potential threats. We can give no assurances that these measures and efforts will prevent interruptions or breakdowns. If we fail to maintain or protect our information technology systems and data integrity effectively or fail to anticipate, plan for or manage significant disruptions to these systems, our competitive position could be harmed, we could lose existing customers, have difficulty preventing, detecting, and controlling fraud, have disputes with customers, specialist physicians and other health care professionals, have regulatory sanctions or penalties imposed, incur expenses or lose revenues as a result of a data privacy breach or theft of intellectual property or suffer other adverse consequences, any of which could have a material adverse effect on our business, results of operations, financial condition or cash flows.
Cost-containment efforts of our customers, purchasing groups and governmental organizations could have a material adverse effect on our sales and profitability.
In an effort to reduce costs, many hospitals within the United States have become members of Group Purchasing Organizations (GPOs) and Integrated Delivery Networks (IDNs). GPOs and IDNs negotiate pricing arrangements with medical device companies and distributors and offer the negotiated prices to affiliated hospitals and other members. GPOs and IDNs typically award contracts on a category-by-category basis through a competitive bidding process. Bids are generally solicited from multiple providers with the intention of
21
driving down pricing or reducing the number of vendors. Due to the highly competitive nature of the GPO and IDN contracting processes, we may not be able to obtain or maintain contract positions with major GPOs and IDNs. Furthermore, the increasing leverage of organized buying groups may reduce market prices for our products, thereby reducing our profitability.
While having a contract with a GPO or IDN for a given product category can facilitate sales to members of that GPO or IDN, such contract positions can offer no assurance that any level of sales will be achieved, as sales are typically made pursuant to purchase orders. Even when a provider is the sole contracted supplier of a GPO or IDN for a certain product category, members of the GPO or IDN generally are free to purchase from other suppliers. Furthermore, GPO and IDN contracts typically are terminable without cause by the GPO or IDN upon 60 to 90 days’ notice. Accordingly, although we have multiple contracts with many major GPOs and IDNs, the members of such groups may choose to purchase from our competitors due to the price or quality offered by such competitors, which could result in a decline in our sales and profitability.
The successful use of our products depends, in part, on our ability to educate specialist physicians in the proper use of our products, which may be more complex than competitive products or alternative treatments that do not use our products. If we are unable to educate specialist physicians in the proper use of our products, we may experience a high risk of product liability.
The successful use of our products depends, in part, on our ability to educate specialist physicians in the proper use of our products, which may be more complex than competitive products or alternative treatments that do not use our products. We educate specialist physicians on the proper techniques in using our products to achieve the intended outcome. However, our products may be more complicated to operate than competitive products or alternative treatments that do not use our products. In the event that specialist physicians perceive that our products are complex relative to alternative products or established treatments that do not use our products, we may have difficulty gaining or increasing adoption of our products. Further, we may be unable to provide adequate education on the use of our products to specialist physicians, and some specialist physicians may not be willing to invest the time required to become properly educated on the use of our products. If we are unable to educate specialist physicians to properly use our products, this may lead to inadequate demand for our products and materially adversely affect our business, results of operations, financial condition or cash flows.
If we do not adequately educate specialist physicians on the use of our products, and our products are used incorrectly during procedures, we may also be subject to claims against us by such specialist physicians, their hospitals or their patients. Our business, including our reputation, may consequently be adversely affected by any litigation that may occur based on error in the use of our products, and such litigation could also materially adversely affect our results of operations, financial condition or cash flows.
Regulatory Risks
We are subject to stringent domestic and foreign medical device regulation, which may impede the approval or clearance process for our products, hinder our development activities and manufacturing processes and, in some cases, result in the recall or seizure of previously approved or cleared products.
Our products, development activities and manufacturing processes are subject to extensive and rigorous regulation by the U.S. Food and Drug Administration (FDA) and by comparable agencies in foreign countries and by other regulatory agencies and governing bodies. Manufacturers of medical devices such as us must comply with certain regulations that cover the composition, labeling, testing, clinical study, manufacturing, packaging and distribution of medical devices. In addition, medical devices must receive FDA clearance or approval before they can be commercially marketed in the United States. The FDA may require testing and surveillance programs to monitor the effects of cleared or approved products that have been commercialized and can prevent or limit further marketing of a product based on the results of these post-marketing programs. Furthermore, most major markets for medical devices outside the United States require clearance, approval or compliance with certain standards and requirements before a product can be commercially marketed. The process of obtaining marketing approval or clearance from the FDA and foreign regulatory agencies for new
22
products could take a significant period of time, require the expenditure of substantial resources, involve rigorous pre-clinical and clinical testing, require changes to our products and result in limitations on the indicated uses of our products. We cannot provide assurance that we will receive the required approval or clearance from the FDA and foreign regulatory agencies for future products on a timely basis. Results from pre-clinical studies and early clinical trials may not allow us to predict results in later-stage testing. We cannot be certain that our future clinical trials will demonstrate the safety and effectiveness of any of our future products or will result in clearance or approval to market any of these products. The failure to receive approval or clearance for significant new products on a timely basis could have a material adverse effect on our business, results of operation, financial condition or cash flows.
The FDA also conducts periodic inspections of our facilities to determine compliance with both the FDA’s Quality System Regulation (QSR) requirements and/or MDR regulations. Product approvals or clearances by the FDA can be withdrawn, and new product approvals or clearances by the FDA and foreign regulatory bodies can be delayed, due to failure to comply with regulatory requirements or the occurrence of unforeseen problems following initial approval or clearance of a product. The failure to comply with regulatory requirements or the discovery of previously unknown problems with a product or manufacturer could result in fines, delays or suspensions of regulatory approvals or clearances, seizures or recalls of products (with the attendant expenses and adverse competitive impact), the banning of a particular device, an order to replace or refund the cost of any device previously manufactured or distributed, operating restrictions and criminal prosecution, as well as decreased sales as a result of negative publicity and product liability claims, all of which could have a material adverse effect on our business, results of operation, financial condition or cash flows.
The implementation of healthcare reform in the United States could have a material adverse effect on our business.
In March 2010, the Patient Protection and Affordable Care Act, as amended by the Health Care and Education Reconciliation Act of 2010 (the Affordable Care Act) was enacted into law in the United States. The Affordable Care Act includes provisions that, among other things, reduce and/or limit Medicare reimbursement, require all individuals to have health insurance (with limited exceptions) and impose new and/or increased taxes. Specifically, the law imposes a 2.3% excise tax on the sale in the United States of certain medical devices by a manufacturer, producer or importer of such devices starting after December 31, 2012. The Affordable Care Act also reduces Medicare and Medicaid payments to hospitals and clinical laboratories, which could reduce medical procedure volumes and impact the demand for our products or the prices at which we sell them. While this legislation is intended to expand health insurance coverage to uninsured persons in the United States, the impact of any overall increase in access to healthcare on sales of our products remains uncertain. Various healthcare reform proposals have also emerged at the state level. The impact of the Affordable Care Act and these proposals could have a material adverse effect on our business, results of operation, financial condition or cash flows.
If we modify our FDA cleared products, we may need to seek and obtain new clearances, which, if not granted, would prevent us from selling our modified products or require us to redesign our products.
A component of our strategy is to continue to modify and upgrade our products that have been cleared by the FDA. The FDA requires device manufacturers to make a determination of whether or not a modification requires a clearance; however, the FDA can review a manufacturer’s decision not to submit for additional clearances. Any modifications to an FDA cleared device that would significantly affect its safety or effectiveness or that would constitute a major change in its intended use would require a new 510(k) clearance or possibly a premarket approval. We may not be able to obtain additional 510(k) clearances or premarket approvals for new products or for modifications to, or additional indications for, our existing products in a timely fashion, or at all. We also cannot provide any assurance that the FDA will agree with our decisions not to seek clearances for particular device modifications. Delays in obtaining future clearances would adversely affect our ability to introduce new or enhanced products in a timely manner, which in turn would harm our revenue and future profitability. We have made modifications to our products in the past and may make additional modifications in the future that we believe do not or will not require additional clearances. If the FDA disagrees, and requires new clearances or approvals for any modifications, and we fail to obtain such approvals or clearances or fail to secure approvals or clearances in a timely
23
manner, we may be required to recall and to stop the manufacturing and marketing of the modified device until we obtain FDA approval or clearance, and we may be subject to significant regulatory fines or penalties, all of which could harm our results of operations and require us to redesign our products.
We may not receive necessary foreign regulatory approvals or clearances or otherwise comply with foreign regulations.
In 2014 and for the six months ended June 30, 2015, sales outside the United States accounted for approximately 34% and 34%, respectively, of our total sales, and we expect this percentage to increase in future years. Foreign regulatory bodies have established varying regulations. Specifically, the European Union has promulgated rules that require that medical device products receive the right to affix the CE mark, an international symbol of adherence to quality assurance standards and compliance with applicable European medical device directives. Although we have received CE markings for all of the products we currently sell in the European Union, we can give no assurance that we will be able to obtain European Union approval for any of our future products. Our inability or failure, or the inability or failure of our international distributors, to comply with varying foreign regulations or the imposition of new regulations could restrict or, in certain countries, result in the prohibition of the sale of our products, and thereby adversely affect our business, financial condition and results of operations. In addition, our profitability from our international operations may be limited by risks and uncertainties related to economic conditions in these regions, foreign currency fluctuations, regulatory and reimbursement approvals, competitive offerings, infrastructure development, rights to intellectual property and our ability to implement our overall business strategy. Any significant changes in the competitive, political, legal, regulatory, reimbursement or economic environment where we conduct international operations may have a material adverse effect on our business, results of operation, financial condition or cash flows.
We may not be able to meet regulatory quality requirements applicable to our manufacturing process.
We are required to register with the FDA as a device manufacturer and as a result, we are subject to periodic inspection by the FDA for compliance with the FDA’s QSR requirements, which require manufacturers of medical devices to adhere to certain regulations, including testing, quality control and documentation procedures. In addition, the federal MDR regulations require us to provide information to the FDA whenever there is evidence that reasonably suggests that a device may have caused or contributed to a death or serious injury, or has malfunctioned, and if the malfunction were to recur, it would be likely to cause or contribute to a death or serious injury. Compliance with applicable regulatory requirements is subject to continual review and is rigorously monitored through periodic inspections by the FDA. In the European Community, we are required to maintain certain International Organization for Standardization (ISO) certifications in order to sell products and we undergo periodic inspections by notified bodies to obtain and maintain these certifications. Some foreign countries, most notably Japan and Brazil, have similar requirements or may require inspections of our manufacturing facilities before approving a product for sale in their country. Some of our suppliers are subject to the same or similar scrutiny. If we or our suppliers fail to adhere to QSR, ISO or similar requirements, this could delay production of our products and lead to fines, difficulties in obtaining regulatory clearances or approvals, recalls or other consequences, which could in turn have a material adverse effect on our business, results of operation, financial condition or cash flows.
We are subject to periodic inspections by the FDA and other regulatory bodies related to regulatory requirements that apply to medical devices designed and manufactured, and clinical trials sponsored, by us. We recently received notices of inspectional observations or deficiencies from the FDA, which require us to undertake corrective and preventive actions or other actions in order to address the FDA’s concerns, which could be expensive and time-consuming to complete and could impose additional burdens and expenses.
We are subject to periodic inspections by the FDA and other regulatory bodies. If we receive a notice of inspectional observations or deficiencies from the FDA following an inspection, we may be required to undertake corrective and preventive actions or other actions in order to address the FDA’s concerns, which could be expensive and time-consuming to complete and could impose additional burdens and expenses. Failure to adequately address the FDA’s concerns could expose us to enforcement and administrative actions.
24
For example, from June 24, 2015 to July 15, 2015 the FDA conducted an inspection of our records relating to certain investigational sites for two different clinical trials and from July 30, 2015 to August 4, 2015, the FDA conducted an inspection of our quality system. At the conclusion of the first inspection, a Form FDA 483 was issued with one observation. The 483 observation pertained to the failure to ensure proper monitoring at five of the investigational sites reviewed. Specifically, the observation noted that protocols relating to performing onsite monitoring visits at appropriate intervals and providing documentation to clearly address any repeated data problems and resolutions of noted deficiencies in written reports after each onsite visit were not properly followed. At the conclusion of the second inspection, a Form FDA 483 was also issued with one observation relating to our procedures for Corrective and Preventative Action (CAPA). Specifically, the observation noted our CAPA procedures do not require an effectiveness check in all cases.
We responded to the first 483 on August 4, 2015, and we have begun to take preventive actions to address the observation in that 483. We continue to review and enhance our investigational site monitoring to ensure compliance with regulatory requirements. We have opened a CAPA to address the observation in the second 483 and expect to respond to the second 483 later in August 2015. However, the FDA may conclude that we have not adequately responded to its observations, and could take action against us without further notice. Action by the FDA against us could result in monetary fines or require us to take further corrective actions, which could be expensive and time-consuming to complete and could impose additional burdens and expenses, and could even require us to discontinue our investigational studies.
We are subject to healthcare fraud and abuse regulations that could result in significant liability, require us to change our business practices and restrict our operations in the future.
We are subject to various federal, state and local laws targeting fraud and abuse in the healthcare industry, including anti-kickback and false claims laws. Violations of these laws are punishable by criminal or civil sanctions, including substantial fines, imprisonment and exclusion from participation in healthcare programs such as Medicare and Medicaid. These laws and regulations are wide ranging and subject to changing interpretation and application, which could restrict our sales or marketing practices. Furthermore, since many of our customers rely on reimbursement from Medicare, Medicaid and other governmental programs to cover a substantial portion of their expenditures, our exclusion from such programs as a result of a violation of these laws could have a material adverse effect on our business, results of operation, financial condition or cash flows.
The FDA and other regulatory agencies strictly regulate the promotional claims that may be made about medical devices. If we are found to have improperly promoted our products for off-label uses, we may become subject to significant fines and other liability.
The FDA and other regulatory agencies strictly regulate the promotional claims that may be made about medical devices. For example, devices cleared under section 510(k) cannot be marketed for any intended use that is outside of the FDA’s substantial equivalence determination for such devices. Physicians nevertheless may use our products on their patients in a manner that is inconsistent with the intended use cleared by the FDA. If we are found to have promoted such “off-label” uses, we may become subject to significant government fines and other related liability. The federal government has levied large civil and criminal fines against companies for alleged improper promotion and has enjoined several companies from engaging in off-label promotion. The FDA has also requested that companies enter into consent decrees or permanent injunctions under which specified promotional conduct is changed or curtailed.
Our operations are subject to environmental, health and safety laws and regulations, with which compliance may be costly.
Our business is subject to federal, state, and local laws and regulations relating to the protection of the environment, worker health and safety and the use, management, storage, and disposal of hazardous substances and wastes. Failure to comply with these laws and regulations may result in substantial fines, penalties or other sanctions. In addition, environmental laws and regulations could require us to pay for environmental remediation and response costs, or subject us to third party claims for personal injury, natural
25
resource or property damage, relating to environmental contamination. Liability may be imposed whether or not we knew of, or were responsible for, such environmental contamination. The cost of defending against environmental claims, of compliance with environmental, health and safety regulatory requirements or of remediating contamination could materially adversely affect our business, assets or results of operations and, consequently, amounts available for distribution to our stockholders.
Risks Related to Our Intellectual Property
We rely on a variety of intellectual property rights, and if we are unable to maintain or protect our intellectual property, our business and results of operations will be harmed.
Our commercial success will depend, in part, on our ability to obtain and maintain intellectual property protection for our products and related technologies both in the United States and elsewhere, successfully defend our intellectual property rights against third-party challenges and successfully enforce our intellectual property rights to prevent third-party infringement. While we rely primarily upon a combination of patents, trademarks and trade secret protection, as well as nondisclosure, confidentiality and other contractual agreements to protect the intellectual property related to our brands, products and other proprietary technologies, protection derived from patents is relatively limited.
The process of obtaining patent protection is expensive and time-consuming, and we may not be able to prosecute all necessary or desirable patent applications at a reasonable cost or in a timely manner. We may choose not to seek patent protection for certain innovations or products and may choose not to pursue patent protection in certain jurisdictions, and under the laws of certain jurisdictions, patents or other intellectual property rights may be unavailable or limited in scope and, in any event, any patent protection we obtain may be limited. As a result, some of our products are not, and in the future may not be, protected by patents. We generally apply for patents in those countries where we intend to make, have made, use or sell products and where we assess the risk of infringement to justify the cost of seeking patent protection. However, we do not seek protection in all countries where we sell products and we may not accurately predict all the countries where patent protection would ultimately be desirable. If we fail to timely file a patent application in any such country or major market, we may be precluded from doing so at a later date. Competitors may use our technologies in jurisdictions where we have not obtained patent protection to develop their own products and, further, may export otherwise infringing products to territories in which we have patent protection that may not be sufficient to terminate infringing activities.
Furthermore, we cannot guarantee that any patents will be issued from any pending or future owned or licensed patent applications, or if any current or future patents will provide us with any meaningful protection or competitive advantage. Even if issued, existing or future patents may be challenged, including with respect to ownership, narrowed, invalidated, held unenforceable or circumvented, any of which could limit our ability to prevent competitors and other third parties from developing and marketing similar products or limit the length of terms of patent protection we may have for our products and technologies. Other companies may also design around technologies we have patented, licensed or developed. In addition, the issuance of a patent does not give us the right to practice the patented invention. Third parties may have blocking patents that could prevent us from marketing our products or practicing our own patented technology.
The patent positions of medical device companies can be highly uncertain and involve complex legal, scientific and factual questions for which important legal principles remain unresolved. The standards that the United States Patent and Trademark Office (USPTO) and its foreign counterparts use to grant patents are not always applied predictably or uniformly. Changes in either the patent laws, implementing regulations or the interpretation of patent laws may diminish the value of our rights. The legal systems of certain countries do not protect intellectual property rights to the same extent as the laws of the United States, and many companies have encountered significant problems in protecting and defending such rights in foreign jurisdictions.
Because patent applications in the United States, Europe and many other jurisdictions are typically not published until 18 months after filing, or in some cases not at all, and because publications of discoveries in scientific literature lag behind actual discoveries, we cannot be certain that we were the first to make the
26
inventions claimed in our issued patents or pending patent applications, or that we were the first to file for protection of the inventions set forth in our patents or applications. We can give no assurance that all of the potentially relevant art relating to our patents and patent applications has been found; overlooked prior art could be used by a third party to challenge the validity, enforceability and scope of our patents or prevent a patent from issuing from a pending patent application. As a result, we may not be able to obtain or maintain protection for certain inventions. Therefore, the validity, enforceability and scope of our patents in the United States, Europe and in other countries cannot be predicted with certainty and, as a result, any patents that we own or license may not provide sufficient protection against our competitors.
Third parties may challenge any existing patent or future patent we own or license through adversarial proceedings in the issuing offices or in court proceedings, including as a response to any assertion of our patents against them. In any of these proceedings, a court or agency with jurisdiction may find our patents invalid and/or unenforceable, or even if valid and enforceable, insufficient to provide protection against competing products and services sufficient to achieve our business objectives. We may be subject to a third party pre-issuance submission of prior art to the USPTO, or reexamination by the USPTO if a third party asserts a substantial question of patentability against any claim of a U.S. patent we own or license. The adoption of the Leahy-Smith America Invents Act (Leahy-Smith Act) in September 2011 established additional opportunities for third parties to invalidate U.S. patent claims, including inter partes review and post-grant review proceedings. Outside of the United States, patents we own or license may become subject to patent opposition or similar proceedings, which may result in loss of scope of some claims or the entire patent. In addition, such proceedings are very complex and expensive, and may divert our management’s attention from our core business. If any of our patents are challenged, invalidated, circumvented by third parties or otherwise limited or expire prior to the commercialization of our product candidates, and if we do not own or have exclusive rights to other enforceable patents protecting our products or other technologies, competitors and other third parties could market products and use processes that are substantially similar to, or superior to, ours and our business would suffer.
The degree of future protection for our proprietary rights is uncertain because legal means afford only limited protection and may not adequately protect our rights or permit us to gain or keep a competitive advantage. For example:
| • | others may be able to develop products that are similar to, or better than, ours in a way that is not covered by the claims of our patents; |
| • | we might not have been the first to make the inventions covered by our patents or pending patent applications; |
| • | we might not have been the first to file patent applications for these inventions; |
| • | any patents that we obtain may not provide us with any competitive advantages or may ultimately be found invalid or unenforceable; or |
| • | we may not develop additional proprietary technologies that are patentable. |
We may file lawsuits or initiate other proceedings to protect or enforce our patents or other intellectual property rights, which could be expensive, time consuming and unsuccessful.
Competitors may infringe our issued patents or other intellectual property. To counter infringement or unauthorized use, we may be required to file infringement claims, which can be expensive and time-consuming. Any claims we assert against perceived infringers could provoke these parties to assert counterclaims against us alleging that we infringe their intellectual property. In addition, in a patent infringement proceeding, a court may decide that a patent of ours is invalid or unenforceable, in whole or in part, construe the patent’s claims narrowly or refuse to stop the other party from using the technology at issue on the grounds that our patents do not cover the technology in question. An adverse result in any litigation proceeding could put one or more of our patents at risk of being invalidated or interpreted narrowly, which could adversely affect our competitive business position, business prospects and financial condition.
27
Our commercial success depends significantly on our ability to operate without infringing upon the intellectual property rights of third parties.
The medical device industry is subject to rapid technological change and substantial litigation regarding patent and other intellectual property rights. Our competitors in both the United States and abroad, many of which have substantially greater resources and have made substantial investments in patent portfolios and competing technologies, may have applied for or obtained or may in the future apply for and obtain, patents that will prevent, limit or otherwise interfere with our ability to make, use and sell our products. Numerous third party patents exist in the fields relating to our products, and it is difficult for industry participants, including us, to identify all third-party patent rights relevant to our products and technologies. Moreover, because some patent applications are maintained as confidential for a certain period of time, we cannot be certain that third parties have not filed patent applications that cover our products and technologies.
Patents could be issued to third parties that we may ultimately be found to infringe. Third parties may have or obtain valid and enforceable patents or proprietary rights that could block us from developing product candidates using our technology. Our failure to obtain or maintain a license to any technology that we require may materially harm our business, financial condition and results of operations. Furthermore, we would be exposed to a threat of litigation.
From time to time, we may be party to, or threatened with, litigation or other proceedings with third parties, including non-practicing entities, who allege that our products, components of our products and/or proprietary technologies infringe, misappropriate or otherwise violate their intellectual property rights. The types of situations in which we may become a party to such litigation or proceedings include:
| • | we or our collaborators may initiate litigation or other proceedings against third parties seeking to invalidate the patents held by those third parties or to obtain a judgment that our products or processes do not infringe those third parties’ patents; |
| • | we or our collaborators may participate at substantial cost in International Trade Commission proceedings to abate importation of products that would compete unfairly with our products; |
| • | if our competitors file patent applications that claim technology also claimed by us or our licensors, we or our licensors may be required to participate in interference, derivation or opposition proceedings to determine the priority of invention, which could jeopardize our patent rights and potentially provide a third party with a dominant patent position; |
| • | if third parties initiate litigation claiming that our processes or products infringe their patent or other intellectual property rights, we and our collaborators will need to defend against such proceedings; |
| • | if third parties initiate litigation or other proceedings seeking to invalidate patents owned by or licensed to us or to obtain a declaratory judgment that their product or technology does not infringe our patents or patents licensed to us, we will need to defend against such proceedings; |
| • | we may be subject to ownership disputes relating to intellectual property, including disputes arising from conflicting obligations of consultants or others who are involved in developing our products; and |
| • | if a license to necessary technology is terminated, the licensor may initiate litigation claiming that our processes or products infringe or misappropriate their patent or other intellectual property rights and/or that we breached our obligations under the license agreement, and we and our collaborators would need to defend against such proceedings. |
These lawsuits and proceedings, regardless of merit, are time-consuming and expensive to initiate, maintain, defend or settle, and could divert the time and attention of managerial and technical personnel, which could materially adversely affect our business. Any such claim could also force use to do one or more of the following:
| • | incur substantial monetary liability for infringement or other violations of intellectual property rights, which we may have to pay if a court decides that the product or technology at issue infringes or violates the third party’s rights, and if the court finds that the infringement was willful, we could be ordered to pay treble damages and the third party’s attorneys’ fees; |
28
| • | pay substantial damages to our customers or end users to discontinue use or replace infringing technology with non-infringing technology; |
| • | stop manufacturing, selling, using, exporting or licensing the product or technology incorporating the allegedly infringing technology or stop incorporating the allegedly infringing technology into such product or technology; |
| • | obtain from the owner of the infringed intellectual property right a license, which may require us to pay substantial upfront fees or royalties to sell or use the relevant technology and which may not be available on commercially reasonable terms, or at all; |
| • | redesign our products and technology so they do not infringe or violate the third party’s intellectual property rights, which may not be possible or may require substantial monetary expenditures and time; |
| • | enter into cross-licenses with our competitors, which could weaken our overall intellectual property position; |
| • | lose the opportunity to license our technology to others or to collect royalty payments based upon successful protection and assertion of our intellectual property against others; |
| • | find alternative suppliers for non-infringing products and technologies, which could be costly and create significant delay; or |
| • | relinquish rights associated with one or more of our patent claims, if our claims are held invalid or otherwise unenforceable. |
Some of our competitors may be able to sustain the costs of complex intellectual property litigation more effectively than we can because they have substantially greater resources. In addition, intellectual property litigation, regardless of its outcome, may cause negative publicity, adversely impact prospective customers, cause product shipment delays, or prohibit us from manufacturing, marketing or otherwise commercializing our products and technology. Any uncertainties resulting from the initiation and continuation of any litigation could have a material adverse effect on our ability to raise additional funds or otherwise have a material adverse effect on our business, results of operation, financial condition or cash flows.
In addition, we may indemnify our customers and distributors against claims relating to the infringement of intellectual property rights of third parties related to our products. Third parties may assert infringement claims against our customers or distributors. These claims may require us to initiate or defend protracted and costly litigation on behalf of our customers or distributors, regardless of the merits of these claims. If any of these claims succeed, we may be forced to pay damages on behalf of our customers, suppliers or distributors, or may be required to obtain licenses for the products they use. If we cannot obtain all necessary licenses on commercially reasonable terms, our customers may be forced to stop using our products.
Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation, there is a risk that some of our confidential information could be compromised by disclosure during this type of litigation. There could also be public announcements of the results of hearings, motions or other interim proceedings or developments. If securities analysts or investors perceive these results to be negative, it could have a material adverse effect on the price of our common stock. The occurrence of any of these events may have a material adverse effect on our business, results of operation, financial condition or cash flows.
Changes in patent law could diminish the value of patents in general, thereby impairing our ability to protect our existing and future products.
Recent patent reform legislation could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents. On September 16, 2011, the Leahy-Smith Act was signed into law. The Leahy-Smith Act includes a number of significant changes to U.S. patent law. These include provisions that affect the way patent applications are prosecuted, redefine prior art, and may also affect patent litigation. The USPTO developed new regulations and procedures to govern
29
administration of the Leahy-Smith Act, including switching the United States patent system from a “first-to-invent” system to a “first-to-file” system. Under a “first-to-file” system, assuming the other requirements for patentability are met, the first inventor to file a patent application generally will be entitled to the patent on an invention regardless of whether another inventor had made the invention earlier. Many of the substantive changes to patent law associated with the Leahy-Smith Act, in particular, the first-to-file provisions, only became effective recently. Accordingly, it is not clear what, if any, impact the Leahy-Smith Act will have on the operation of our business. The Leahy-Smith Act and its implementation could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents, all of which could have a material adverse effect on our business, results of operation, financial condition or cash flows.
In addition, patent reform legislation may pass in the future that could lead to additional uncertainties and increased costs surrounding the prosecution, enforcement and defense of our patents and applications. Furthermore, the U.S. Supreme Court and the U.S. Court of Appeals for the Federal Circuit have made, and will likely continue to make, changes in how the patent laws of the United States are interpreted. Similarly, foreign courts have made, and will likely continue to make, changes in how the patent laws in their respective jurisdictions are interpreted. We cannot predict future changes in the interpretation of patent laws or changes to patent laws that might be enacted into law by United States and foreign legislative bodies. Those changes may materially affect our patents or patent applications and our ability to obtain additional patent protection in the future.
Obtaining and maintaining patent protection depends on compliance with various procedural, document submission, fee payment and other requirements imposed by governmental patent agencies, and our patent protection could be reduced or eliminated for non-compliance with these requirements.
The USPTO and various foreign patent offices require compliance with a number of procedural, documentary, fee payment and other similar provisions. In addition, periodic maintenance fees on our owned and in-licensed patents are due to be paid to governmental patent agencies over the lifetime of the patents. Future maintenance fees will also need to be paid on other patents that may be issued to us. We have systems in place to remind us to pay these fees, and we employ outside firms to remind us or our licensor to pay annuity fees due to patent agencies on our patents and pending patent applications. In certain cases, an inadvertent lapse can be cured by payment of a late fee or by other means in accordance with the applicable rules. However, there are situations in which noncompliance can result in abandonment or lapse of the patent or patent application, resulting in partial or complete loss of patent rights in the relevant jurisdiction. In such an event, our competitors might be able to enter the market and this circumstance would have a material adverse effect on our business, results of operation, financial condition or cash flows.
If our trademarks and trade names are not adequately protected, then we may not be able to build name recognition in our markets of interest and our business may be adversely affected.
We currently own eight trademarks, related to our company name, logo, products and technology, that are registered with the USPTO as well as six trademarks registered in Europe and Japan. Our registered or unregistered trademarks or trade names may be challenged, infringed, circumvented, declared generic or determined to be infringing on other marks or names. We may not be able to protect our rights in these trademarks and trade names, which we need in order to build name recognition with potential customers in our markets of interest. There is no guarantee we will be able to secure registration for any of our pending trademark applications with the USPTO or comparable foreign authorities. In addition, third parties have registered trademarks similar and identical to our trademarks, and may in the future file for registration of such trademarks. If they succeed in registering or developing common law rights in such trademarks, and if we are not successful in challenging such third-party rights, we may not be able to use these trademarks to market our products in those countries where such third parties have registered such trademarks or obtained such common law rights. In any case, if we are unable to establish name recognition based on our trademarks and trade names, then we may not be able to compete effectively and our business may be adversely affected.
30
In addition, we may be involved in litigation or other proceedings to protect our trademark rights associated with our company name or the names used with our products. For example, the United States application for registration of our Apollo trademark is currently subject to an opposition proceeding before the Trademark Trial and Appeal Board. An adverse decision in such proceeding could require us to establish an alternative name for our Apollo product line. Any objections we receive from the USPTO, foreign trademark authorities or third parties relating to our pending applications could require us to incur significant expense in defending the objections or establishing alternative names. Names used with our products may be claimed to infringe names held by others or to be ineligible for proprietary protection. If we have to change the name of our company or any product, we may experience a loss in goodwill associated with our brand name, customer confusion or a loss of sales.
If we are unable to protect the confidentiality of our trade secrets and other proprietary information, our business and competitive position may be harmed.
In addition to patent protection, we also rely on confidential proprietary information, including trade secrets and know-how, to develop and maintain our competitive position. We seek to protect our confidential proprietary information, in part, by entering into confidentiality agreements with our employees, consultants, collaborators and others upon the commencement of their relationships with us. These agreements require that all confidential information developed by the individual or made known to the individual by us during the course of the individual’s relationship with us be kept confidential. Our agreements with employees and our personnel policies also provide that any inventions conceived by the individual in the course of rendering services to us shall be our exclusive property. However, we may not obtain these agreements in all circumstances, and individuals with whom we have these agreements may not comply with their terms. Thus, despite such agreements, such inventions may become assigned to third parties. Monitoring unauthorized uses and disclosures of our intellectual property is difficult, and we do not know whether the steps we have taken to protect our intellectual property will be effective. In the event of unauthorized use or disclosure of our trade secrets or proprietary information, these agreements, even if obtained, may not provide meaningful protection, particularly for our trade secrets or other confidential information. To the extent that our employees, consultants or contractors use technology or know-how owned by third parties in their work for us, disputes may arise between us and those third parties as to the rights in related inventions. To the extent that an individual who is not obligated to assign rights in intellectual property to us is rightfully an inventor of intellectual property, we may need to obtain an assignment or a license to that intellectual property from that individual, or a third party or from that individual’s assignee. Such assignment or license may not be available on commercially reasonable terms or at all.
Adequate remedies may not exist in the event of unauthorized use or disclosure of our proprietary information. The disclosure of our trade secrets would impair our competitive position and may materially harm our business, financial condition and results of operations. Costly and time consuming litigation could be necessary to enforce and determine the scope of our proprietary rights, and failure to maintain trade secret protection could adversely affect our competitive business position. In addition, others may independently discover or develop our trade secrets and proprietary information, and the existence of our own trade secrets affords no protection against such independent discovery.
We may also employ individuals who were previously or concurrently employed at research institutions and/or other medical device companies, including our competitors or potential competitors. We may be subject to claims that these employees, or we, have inadvertently or otherwise used or disclosed trade secrets or other proprietary information of their former employers, or that patents and applications we have filed to protect inventions of these employees, even those related to one or more of our products, are rightfully owned by their former or concurrent employer. Litigation may be necessary to defend against these claims. Even if we are successful in defending against these claims, litigation could result in substantial costs and be a distraction to management.
31
Risks Related to Our Finances and Capital Requirements
It is difficult to forecast future performance, which may cause our financial results to fluctuate unpredictably.
A number of factors over which we have limited or no control may contribute to fluctuations in our financial results, such as:
| • | variations in revenue due to the unavailability of specialist physicians who use our products during certain times of the year, such as those periods when there are major conferences on conditions they treat or those periods when high volume users of our products take time off of work; |
| • | positive or negative media coverage of our products or the procedures or products of our competitors or our industry; |
| • | publication of clinical trial results or studies by us or our competitors; |
| • | changes in our sales process due to industry changes, such as changes in the stroke care pathway; |
| • | delays in receipt of anticipated purchase orders; |
| • | delays in customers receiving products; |
| • | performance of our independent distributors; |
| • | our ability to obtain further regulatory clearances or approvals; |
| • | the timing of product development and clinical trial activities; |
| • | delays in, or failure of, product and component deliveries by our suppliers; |
| • | changes in reimbursement policies or levels; |
| • | customer response to the introduction of new products or alternative treatments, and the degree to we which we are effective in transitioning customers to our products; and |
| • | fluctuations in foreign currency. |
In the event our actual revenue and results of operations do not meet our or others’ forecasts for a particular period, the market price of our common stock may decline substantially.
We may require additional financing in the future and may not be able to obtain such financing on favorable terms, if at all, which could force us to delay, reduce or eliminate our research and development activities or otherwise harm our business.
To date, we have financed our operations primarily through our operations, private placements of our equity securities and borrowings under a line of credit with a financial institution. We are unable to predict the extent of any future operating cash flows or whether we will be able to maintain or grow our profitability. If we require additional financing to continue or expand our operations, for research and development, for acquisitions or for other purposes, we may determine to engage in equity or debt financings or incur other indebtedness. We may not be able to timely secure additional debt or equity financing on favorable terms, or at all. If we raise additional funds through the issuance of equity or convertible debt or other equity-linked securities, our existing stockholders could suffer significant dilution. Any debt financing obtained by us in the future could involve restrictive covenants relating to our capital raising activities and other financial and operational matters, which may make it more difficult for us to obtain additional capital and to pursue business opportunities, including potential acquisitions. If needed funds are not available in adequate amounts or on acceptable terms from additional financing sources, our business will be materially adversely affected.
32
If we engage in any acquisitions, we will incur a variety of costs and may potentially face numerous risks that could adversely affect our business and operations.
If appropriate opportunities become available, we may seek to acquire additional businesses, assets, technologies or products to enhance our business. In connection with any acquisitions, we could issue additional equity securities or convertible debt or equity-linked securities, which would dilute our stockholders, cause us to incur substantial debt to fund the acquisitions, or assume significant liabilities.
Acquisitions involve numerous risks, including problems integrating the purchased operations, technologies or products, unanticipated costs and other liabilities, diversion of management’s attention from our core businesses, adverse effects on existing business relationships with current and/or prospective customers and/or suppliers, risks associated with entering markets in which we have no or limited prior experience and potential loss of key employees. Acquisitions may also require us to record goodwill and non-amortizable intangible assets that will be subject to impairment testing on a regular basis and potential periodic impairment charges, incur amortization expenses related to certain intangible assets, and incur write offs and restructuring and other related expenses, any of which could harm our results of operations and financial condition. If we fail in our integration efforts with respect to any of our acquisitions and are unable to efficiently operate as a combined organization, our business and financial condition may be adversely affected.
Risks Relating to Securities Markets and Investment in Our Common Stock
A viable trading market for our common stock may not develop or be sustained.
Prior to this offering, there has been no public market for shares of our common stock. Although we intend to list our common stock on the New York Stock Exchange (“NYSE”), an active trading market for our shares may never develop or be sustained following this offering. We and the underwriters will determine the initial public offering price of our common stock through negotiation. This price will not necessarily reflect the price at which investors in the market will be willing to buy and sell our shares following this offering. The initial public offering price may vary from the market price of our common stock after the offering. In addition, the trading volume of companies such as ours is often very low, and thus your ability to resell your shares may be severely constrained. As a result of these and other factors, you may be unable to resell your shares of our common stock at or above the initial public offering price.
The price of our common stock may be volatile, and you could lose all or part of your investment.
Stock markets have experienced extreme volatility that has often been unrelated to the operating performance of particular companies. These broad market fluctuations may adversely affect the trading price of our common stock. In addition, limited trading volume of our stock may contribute to its future volatility. Price declines in our common stock could result from general market and economic conditions, some of which are beyond our control, and a variety of other factors, including any of the risk factors described in this prospectus. These broad market and industry factors may harm the market price of our common stock, regardless of our operating performance, and could cause you to lose all or part of your investment in our common stock since you might be unable to sell your shares at or above the price you paid in this offering. Factors that could cause fluctuations in the market price of our common stock include the following:
| • | price and volume fluctuations in the overall stock market from time to time; |
| • | volatility in the market prices and trading volumes of medical device company stocks; |
| • | changes in operating performance and stock market valuations of other medical device companies generally, or those in our industry in particular; |
| • | sales of shares of our common stock by us or our stockholders; |
| • | failure of securities analysts to maintain coverage of us, changes in financial estimates by securities analysts who follow our company, or our failure to meet these estimates or the expectations of investors; |
33
| • | the financial projections we may provide to the public, any changes in those projections or our failure to meet those projections; |
| • | announcements by us or our competitors of new products or services; |
| • | the public’s reaction to our press releases, other public announcements and filings with the SEC; |
| • | rumors and market speculation involving us or other companies in our industry; |
| • | actual or anticipated changes in our results of operations or fluctuations in our results of operations; |
| • | actual or anticipated developments in our business, our competitors’ businesses or the competitive landscape generally; |
| • | litigation involving us, our industry or both, or investigations by regulators into our operations or those of our competitors; |
| • | developments or disputes concerning our intellectual property or other proprietary rights; |
| • | announced or completed acquisitions of businesses or technologies by us or our competitors; |
| • | new laws or regulations or new interpretations of existing laws or regulations applicable to our business; |
| • | changes in accounting standards, policies, guidelines, interpretations or principles; |
| • | any significant change in our management; and |
| • | general economic conditions and slow or negative growth of our markets. |
In addition, in the past, following periods of volatility in the overall market and the market price of a particular company’s securities, securities class action litigation has often been instituted against these companies. This litigation, if instituted against us, could result in substantial costs and a diversion of our management’s attention and resources.
Purchasers in this offering will experience immediate and substantial dilution in the book value of their investment.
The initial public offering price of our common stock is substantially higher than the net tangible book value per share of our outstanding common stock immediately prior to this offering. Therefore, if you purchase our common stock in this offering, you will incur an immediate dilution of $ in net tangible book value per share from the price you paid, based on an assumed initial public offering price of $ per share (which is the midpoint of the estimated initial public offering price range set forth on the cover page of this prospectus). In addition, purchasers who bought shares from us in this offering will have contributed % of the total consideration paid to us by our stockholders to purchase shares of our common stock, in exchange for acquiring approximately % of the outstanding shares of our capital stock as of June 30, 2015 after giving effect to this offering. The exercise of outstanding options will result in further dilution. For a further description of the dilution that you will experience immediately after this offering, see the section titled “Dilution.”
If our executive officers, directors and largest stockholders choose to act together, they may be able to significantly influence our management and operations, acting in their own best interests and not necessarily those of other stockholders.
As of June 30, 2015, our executive officers, directors and holders of 5% or more of our outstanding stock and their affiliates beneficially owned approximately 36.1% of our voting stock in the aggregate, and we expect that immediately following the completion of this offering the same group will continue to hold at least % of our outstanding voting stock, based on the number of shares outstanding as of June 30, 2015. As a result, these stockholders, acting together, would be able to significantly influence all matters requiring approval by our stockholders, including the election of directors and the approval of mergers or other business combination transactions. The interests of this group of stockholders may not always coincide with the interests of other
34
stockholders, and they may act in a manner that advances their best interests and not necessarily those of other stockholders. This concentration of ownership may have the effect of delaying, preventing or deterring a change in control of our company, could deprive our stockholders of an opportunity to receive a premium for their common stock as part of a sale of our company and might ultimately affect the market price of our common stock.
A significant portion of our outstanding shares of common stock is restricted from immediate resale but may be sold into the market in the near future. This could cause the market price of our common stock to drop significantly, even if our business is doing well.
Sales of a substantial number of shares of our common stock in the public market could occur at any time, subject to the 180-day lock-up periods under the lock-up agreements described in the section of this prospectus titled “Underwriting.” These sales, or the perception in the market that the holders of a large number of shares of common stock intend to sell shares, could reduce the market price of our common stock. As of June 30, 2015, our directors, executive officers and holders of 5% or more of our outstanding stock beneficially owned approximately 36.1% of our outstanding stock in the aggregate. If one or more of them were to sell a substantial portion of the shares they hold, it could cause our stock price to decline. Futhermore, the lock-up agreements mentioned above may be waived by the underwriters at any time which could lead to these shares being sold in the market prior to the expiration of this 180-day lock-up period.
In addition, as of June 30, 2015, there were 2,460,574 shares subject to outstanding options that will become eligible for sale in the public market upon exercise of such options to the extent permitted by any applicable vesting requirements, the lock-up agreements and Rules 144 and 701 under the Securities Act of 1933, as amended (Securities Act). Moreover, after this offering, holders of an aggregate of 21,617,845 shares of our common stock will have rights, subject to some conditions, to require us to file registration statements covering their shares or to include their shares in registration statements that we may file for ourselves or other stockholders.
We also intend to register all shares of common stock that we may issue under our Amended and Restated 2014 Equity Incentive Plan. Once we register these shares, they can be freely sold in the public market upon issuance and once vested and exercised, as applicable, subject to the 180-day lock-up periods under the lock-up agreements described in the section of this prospectus titled “Underwriting.”
Sales of our common stock as restrictions end or pursuant to registration rights may make it more difficult for us to sell equity securities in the future at a time and at a price that we deem appropriate. These sales also could cause the price of our common stock to fall and make it more difficult for you to sell shares of our common stock.
Our amended and restated certificate of incorporation, our amended and restated bylaws and Delaware law contain provisions that could discourage another company from acquiring us and may prevent attempts by our stockholders to replace or remove our current management.
Provisions of Delaware law (where we are incorporated), our amended and restated certificate of incorporation and amended and restated bylaws may discourage, delay or prevent a merger or acquisition that stockholders may consider favorable, including transactions in which you might otherwise receive a premium for your shares. In addition, these provisions may frustrate or prevent any attempts by our stockholders to replace or remove our current management by making it more difficult for stockholders to replace or remove our board of directors. These provisions include:
| • | authorizing the issuance of “blank check” preferred stock without any need for action by stockholders; |
| • | requiring supermajority stockholder voting to effect certain amendments to our amended and restated certificate of incorporation and amended and restated bylaws; |
| • | eliminating the ability of stockholders to call and bring business before special meetings of stockholders; |
| • | prohibiting stockholder action by written consent; |
35
| • | establishing advance notice requirements for nominations for election to the board of directors or for proposing matters that can be acted on by stockholders at stockholder meetings; |
| • | dividing our board of directors into three classes so that only one third of our directors will be up for election in any given year; and |
| • | providing that our directors may be removed by our stockholders only for cause. |
In addition, we are subject to Section 203 of the Delaware General Corporation Law, which may have an anti-takeover effect with respect to transactions not approved in advance by our board of directors, including discouraging takeover attempts that could have resulted in a premium over the market price for shares of our common stock.
These provisions apply even if a takeover offer may be considered beneficial by some stockholders and could delay or prevent an acquisition that our board of directors determines is not in our and our stockholders’ best interests and could also affect the price that some investors are willing to pay for our common stock. See the section titled “Description of Capital Stock.”
Our amended and restated certificate of incorporation will provide that the Court of Chancery of the State of Delaware will be the exclusive forum for substantially all disputes between us and our stockholders, which could limit our stockholders’ ability to obtain a favorable judicial forum for disputes with us or our directors, officers or employees.
Our amended and restated certificate of incorporation provides that the Court of Chancery of the State of Delaware is the exclusive forum for any derivative action or proceeding brought on our behalf; any action asserting a breach of fiduciary duty; any action asserting a claim against us arising pursuant to the Delaware General Corporation Law, our amended and restated certificate of incorporation or our amended and restated bylaws; or any action asserting a claim against us that is governed by the internal affairs doctrine. The choice of forum provision may limit a stockholder’s ability to bring a claim in a judicial forum that it finds favorable for disputes with us or our directors, officers or other employees, which may discourage such lawsuits against us and our directors, officers and other employees. If a court were to find the choice of forum provision contained in our amended and restated certificate of incorporation to be inapplicable or unenforceable in an action, we may incur additional costs associated with resolving such action in other jurisdictions, which could harm our business and financial condition.
As a public company, our expenses and administrative burden will increase.
As a public company, we will incur significant legal, accounting and other expenses that we did not incur as a private company. In addition, our administrative staff will be required to perform additional tasks. For example, in anticipation of becoming a public company, we will need to adopt additional internal controls and disclosure controls and procedures and bear all of the internal and external costs of preparing and distributing periodic public reports in compliance with our obligations under applicable securities laws.
In addition, laws, regulations and standards relating to corporate governance and public disclosure, including the Sarbanes-Oxley Act of 2002 and related regulations implemented by the SEC and the , have increased legal and financial compliance costs and made some regulatory and compliance activities more time consuming. We are currently evaluating and monitoring developments with respect to new and proposed rules and cannot predict or estimate the amount of additional costs we may incur or the timing of such costs. These laws, regulations and standards are subject to varying interpretations, in many cases due to their lack of specificity, and, as a result, their application in practice may evolve over time as new guidance is provided by regulatory and governing bodies. This could result in continuing uncertainty regarding compliance matters and higher costs necessitated by ongoing revisions to disclosure and governance practices. We intend to invest resources to comply with evolving laws, regulations and standards, and this investment may result in increased general and administrative expenses and a diversion of management’s time and attention from revenue-generating activities to compliance activities. We also expect that being a public company, and being subject to these new rules and regulations, will make it more expensive for us to obtain director and officer liability insurance, and we may be required to accept reduced coverage or incur substantially higher costs to obtain coverage. These factors could also make it more difficult for us to attract
36
and retain qualified members of our board of directors, particularly to serve on our audit committee and compensation committee, and attract and retain qualified executive officers.
The increased costs associated with operating as a public company may decrease our net income or increase any future net loss and may cause us to reduce costs in other areas of our business or increase the prices of our products to offset the effect of such increased costs. Additionally, if these requirements divert our management’s attention from other business concerns, they could have a material adverse effect on our business, results of operation, financial condition or cash flows.
We are an “emerging growth company,” and we cannot be certain if the reduced reporting requirements applicable to emerging growth companies will make our common stock less attractive to investors.
We are an “emerging growth company,” as defined in the JOBS Act. For as long as we continue to be an emerging growth company, we may take advantage of exemptions from various reporting requirements that are applicable to other public companies but not to “emerging growth companies,” including, but not limited to:
| • | being permitted to provide only two years of audited financial statements, in addition to any required unaudited interim financial statements, with correspondingly reduced “Management’s Discussion and Analysis of Financial Condition and Results of Operations” disclosure; |
| • | not being required to comply with the auditor attestation requirements in the assessment of our internal control over financial reporting under Section 404 of the Sarbanes-Oxley Act; |
| • | not being required to comply with any requirement that may be adopted by the Public Company Accounting Oversight Board regarding mandatory audit firm rotation or a supplement to the auditor’s report providing additional information about the audit and the financial statements; |
| • | reduced disclosure obligations regarding executive compensation in our periodic reports and proxy statements; and |
| • | exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and shareholder approval of any golden parachute payments not previously approved. |
We will remain an emerging growth company until the earlier of (1) the last day of the fiscal year (a) following the fifth anniversary of the completion of this offering, (b) in which we have total annual gross revenue of at least $1 billion, or (c) in which we are deemed to be a large accelerated filer, which means the market value of our common stock that is held by non-affiliates exceeds $700 million as of the prior June 30th, and (2) the date on which we have issued more than $1 billion in non-convertible debt during the prior three-year period. We cannot predict if investors will find our common stock less attractive if we choose to rely on these exemptions. If some investors find our common stock less attractive as a result of any choices to reduce future disclosure, there may be a less active trading market for our common stock and the price of our common stock may be more volatile.
Under the JOBS Act, emerging growth companies can also delay adopting new or revised accounting standards until such time as those standards apply to private companies. We have irrevocably elected not to avail ourselves of this exemption from new or revised accounting standards and, therefore, will be subject to the same new or revised accounting standards as other public companies that are not emerging growth companies.
If we fail to maintain an effective system of disclosure controls and internal control over financial reporting, our ability to produce timely and accurate financial statements or comply with applicable regulations could be impaired.
As a public company, we are subject to the reporting requirements of the Securities Exchange Act of 1934, as amended (the Exchange Act), the Sarbanes-Oxley Act, and the listing standards of the NYSE. We expect that the requirements of these rules and regulations will continue to increase our legal, accounting and financial compliance costs, make some activities more difficult, time consuming and costly, and place significant strain on our personnel, systems and resources.
37
The Sarbanes-Oxley Act requires, among other things, that we maintain effective disclosure controls and procedures and internal control over financial reporting. We are continuing to develop and refine our disclosure controls and other procedures that are designed to ensure that information required to be disclosed by us in the reports that we will file with the SEC is recorded, processed, summarized and reported within the time periods specified in SEC rules and forms, and that information required to be disclosed in reports under the Exchange Act is accumulated and communicated to our principal executive and financial officers. We are also continuing to improve our internal control over financial reporting. In order to maintain and improve the effectiveness of our disclosure controls and procedures and internal control over financial reporting, we have expended, and anticipate that we will continue to expend, significant resources, including accounting-related costs and significant management oversight.
Our current controls and any new controls that we develop may become inadequate because of changes in our business. Further, weaknesses in our disclosure controls or our internal control over financial reporting may be discovered in the future. Any failure to develop or maintain effective controls, or any difficulties encountered in their implementation or improvement, could harm our operating results or cause us to fail to meet our reporting obligations and may result in errors in our financial statements or a restatement of our financial statements for prior periods. Any failure to implement and maintain effective internal control over financial reporting also could adversely affect the results of management evaluations and independent registered public accounting firm audits of our internal control over financial reporting that we will eventually be required to include in our periodic reports that will be filed with the SEC. Ineffective disclosure controls and procedures and internal control over financial reporting could also cause investors to lose confidence in our reported financial and other information, which would likely have a negative effect on the trading price of our common stock. In addition, if we are unable to continue to meet these requirements, we may not be able to remain listed on the NYSE.
We are not currently required to comply with the SEC rules that implement Section 404 of the Sarbanes-Oxley Act, and are therefore not required to make a formal assessment of the effectiveness of our internal control over financial reporting for that purpose. As a public company, we will be required to provide an annual management report on the effectiveness of our internal control over financial reporting commencing with our second annual report on Form 10-K. Our independent registered public accounting firm is not required to audit the effectiveness of our internal control over financial reporting until after we are no longer an “emerging growth company,” as defined in the Jumpstart Our Business Startups Act of 2012, or the JOBS Act. At such time, our independent registered public accounting firm may issue a report that is adverse in the event it is not satisfied with the level at which our internal control over financial reporting is documented, designed or operating.
Any failure to maintain effective disclosure controls and internal control over financial reporting could have a material and adverse effect on our business and results of operations, and cause a decline in the price of our common stock.
We will have broad discretion in the use of the net proceeds from this offering and may not use them effectively.
We will have broad discretion in the use of the net proceeds from this offering and could spend the proceeds in ways that do not improve our results of operations or enhance the value of our common stock. Our failure to apply these funds effectively could have a material adverse effect on our business, delay the development and commercialization of our products and cause the price of our common stock to decline. Until the net proceeds are used, they may be placed in investments that do not produce significant income or that may lose value.
If securities or industry analysts do not publish or cease publishing research or publish inaccurate or unfavorable research about our business, our stock price and trading volume could decline.
The trading market for our common stock will depend in part on the research and reports that securities or industry analysts publish about us or our business. We may never obtain research coverage by securities and industry analysts. If no securities or industry analysts commence coverage of our company, the trading price for
38
our stock would be negatively impacted. In the event we obtain securities or industry analyst coverage, if one or more of the analysts who cover us downgrade our stock or publish inaccurate or unfavorable research about our business, our stock price would likely decline. If one or more of these analysts cease coverage of our company or fail to publish reports on us regularly, demand for our stock could decrease, which might cause our stock price and trading volume to decline.
We do not anticipate paying cash dividends, and accordingly, stockholders must rely on stock appreciation for any return on their investment.
We do not anticipate paying cash dividends in the future. As a result, only appreciation of the price of our common stock, which may never occur, would provide a return to stockholders. Investors seeking cash dividends should not invest in our common stock.
Impairment of our deferred tax assets could require a charge to earnings, which could result in a negative impact on our results of operations.
Primarily as a result of net operating losses, stock based compensation, various accruals and reserves, and tax credits, we maintain a deferred tax asset (an asset recognized to reflect an expected benefit to be realized in the future) that may be used to reduce the amount of tax that we would otherwise be required to pay in future periods. Deferred tax assets are reduced by a valuation allowance when it is more likely than not that the future realization of all or some of the deferred tax assets will not be achieved. Valuation allowances related to deferred tax assets can be affected by changes to tax laws, statutory tax rates, future taxable income levels and input from our tax advisors or regulatory authorities. At June 30, 2015, our net deferred tax asset was $6.8 million, after reduction of a valuation allowance of $2.9 million. If our management was to determine that we would not be able to realize all or a portion of our net deferred tax assets in the future, a valuation allowance and a related charge to earnings would be reflected in that period, which could have a material adverse impact on our financial condition and results of operations.
39
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS
We have made statements under the sections titled “Prospectus Summary,” “Risk Factors,” “Management’s Discussion and Analysis of Financial Condition and Results of Operations,” “Business” and in other sections of this prospectus that are forward-looking statements. In some cases, you can identify these statements by forward-looking words such as “may,” “might,” “will,” “should,” “expects,” “plans,” “anticipates,” “believes,” “estimates,” “predicts,” “potential” or “continue,” the negative of these terms and other comparable terminology. These forward-looking statements, which are subject to risks, uncertainties and assumptions about us, may include projections of our future financial performance, our anticipated growth strategies and anticipated trends in our business. Forward-looking statements contained in this prospectus include, but are not limited to statements about:
| • | our ability to design, develop, manufacture and market innovative products; |
| • | our expected future growth, including growth in international sales; |
| • | the size and growth potential of the markets for our products, and our ability to address those markets; |
| • | the market acceptance of our products; |
| • | the performance of our salesforce and distributors; |
| • | regulatory developments in the United States and other markets in which we sell our products; |
| • | our ability to obtain and maintain regulatory approval or clearance of our products on expected timelines; |
| • | our ability to scale our organizational culture; |
| • | competition; |
| • | our use of the proceeds from this offering; |
| • | our results of operations and capital requirements; and |
| • | our ability to obtain and maintain intellectual property protection for our products, as well as our ability to operate our business without infringing the intellectual property rights of others. |
These statements are only predictions based on our current expectations and projections about future events. There are important factors that could cause our actual results, level of activity, performance or achievements to differ materially from the results, level of activity, performance or achievements expressed or implied by the forward-looking statements, including those factors discussed in the section titled “Risk Factors.” You should specifically consider the numerous risks outlined in the section titled “Risk Factors.” Although we believe the expectations reflected in the forward-looking statements are reasonable, we cannot guarantee future results, level of activity, performance or achievements. We undertake no obligation to update any forward-looking statements made in this prospectus to reflect events or circumstances after the date of this prospectus or to reflect new information or the occurrence of unanticipated events, except as required by law.
40
We estimate that the net proceeds to us from this offering will be approximately $ million, or approximately $ million if the underwriters exercise their option to purchase additional shares from us in full, assuming an initial public offering price of $ per share, which is the midpoint of the estimated initial public offering price range set forth on the cover page of this prospectus, after deducting underwriting discounts and commissions and estimated offering expenses payable by us. Each $1.00 increase or decrease in the assumed initial public offering price of $ per share, which is the midpoint of the estimated initial public offering price range set forth on the cover page of this prospectus, would increase or decrease, as applicable, our net proceeds from this offering by $ million, assuming that the number of shares offered by us, as set forth on the cover page of this prospectus, remains the same, after deducting underwriting discounts and commissions payable by us. An increase or decrease of 1.0 million shares in the number of shares offered by us would increase or decrease, as applicable, our net proceeds from this offering by $ million, assuming an initial public offering price of $ per share, which is the midpoint of the estimated initial public offering price range set forth on the cover page of this prospectus, after deducting underwriting discounts and commissions payable by us.
The principal purposes of this offering are to increase our capitalization and financial flexibility, create a public market for our common stock and enable access to the public equity markets for us and our stockholders. We intend to use the net proceeds from this offering for product development, including research and development and clinical trials, expansion of our salesforce and for working capital and general corporate purposes. From time to time, we may consider the acquisition of complementary technologies or businesses, though we have no agreements or understandings with respect to any such acquisitions at this time. We currently have no specific plans for the use of the net proceeds that we receive from this offering. Accordingly, we will have broad discretion in using these proceeds. Pending the use of the net proceeds from this offering, we intend to invest the net proceeds in investment grade, interest bearing securities.
We have never declared or paid cash dividends on our capital stock. We currently intend to retain all available funds for use in the operation and expansion of our business, and do not anticipate paying any cash dividends in the foreseeable future. Any future determination related to dividend policy will be made at the discretion of our board of directors, subject to applicable laws, and will depend on a number of factors, including our financial condition, results of operations, capital requirements, contractual restrictions, general business conditions and other factors that our board of directors may deem relevant.
41
The following table sets forth our cash and cash equivalents and capitalization as of June 30, 2015:
| • | on an actual basis; |
| • | on a pro forma basis, giving effect to (i) the automatic conversion of all outstanding shares of our preferred stock into an aggregate of 19,510,410 shares of our common stock, which conversion will occur immediately prior to the completion of this offering, as if such conversion had occurred on June 30, 2015, and (ii) the filing and effectiveness of our amended and restated certificate of incorporation; and |
| • | on a pro forma as adjusted basis, giving effect to the pro forma adjustments set forth above, and the sale and issuance by us of shares of common stock in this offering, at an assumed initial public offering price of $ per share, which is the midpoint of the estimated initial public offering price range set forth on the cover page of this prospectus, and after deducting underwriting discounts and commissions and estimated offering expenses payable by us. |
This table should be read in conjunction with the section titled “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and the consolidated financial statements and notes thereto appearing elsewhere in this prospectus.
| June 30, 2015 | ||||||||||||
| Actual | Pro Forma | Pro Forma As Adjusted(1) |
||||||||||
| (in thousands, except share and per share data) |
||||||||||||
| Cash and cash equivalents and marketable investments |
$ | 36,764 | $ | 36,764 | $ | |||||||
|
|
|
|
|
|
|
|||||||
| Long-term debt |
$ | — | $ | — | $ | — | ||||||
|
|
|
|
|
|
|
|||||||
| Preferred stock, $0.001 par value per share, 25,000,000 shares authorized, 19,510,410 shares issued and outstanding, actual; no shares authorized, issued or outstanding, pro forma and pro forma as adjusted |
$ | 111,467 | $ | — | $ | — | ||||||
|
|
|
|
|
|
|
|||||||
| Stockholders’ equity (deficit): |
||||||||||||
| Preferred stock, $0.001 par value per share, no shares authorized, issued or outstanding, actual; 5,000,000 shares authorized, no shares issued or outstanding, pro forma and pro forma as adjusted |
— | — | — | |||||||||
| Common stock, $0.001 par value per share, 40,000,000 shares authorized, 5,747,638 shares issued and outstanding, actual; 300,000,000 authorized, 25,258,048 shares issued and outstanding, pro forma; 300,000,000 authorized, shares issued and outstanding, pro forma as adjusted |
6 | 25 | ||||||||||
| Additional paid-in capital |
10,169 | 121,617 | ||||||||||
| Notes receivable from stockholders |
(32 | ) | (32 | ) | ||||||||
| Accumulated other comprehensive loss |
(1,233 | ) | (1,233 | ) | ||||||||
| Accumulated deficit |
(20,009 | ) | (20,009 | ) | ||||||||
|
|
|
|
|
|
|
|||||||
| Total stockholders’ equity (deficit) |
$ | (11,099 | ) | $ | 100,368 | $ | ||||||
|
|
|
|
|
|
|
|||||||
| Total capitalization |
$ | 129,070 | $ | 100,368 | $ | |||||||
|
|
|
|
|
|
|
|||||||
| (1) | Each $1.00 increase or decrease in the assumed initial public offering price of $ per share, which is the midpoint of the estimated initial public offering price range set forth on the cover page of this prospectus, would increase or decrease, as applicable, the amount of our pro forma as adjusted cash and cash equivalents, additional paid-in capital, total stockholders’ equity and total capitalization by approximately $ million, assuming that the number of shares offered by us, as set forth on the cover page of this prospectus, remains the same, after deducting underwriting |
42
| discounts and commissions payable by us. An increase or decrease of 1.0 million shares in the number of shares offered by us would increase or decrease, as applicable, the amount of our pro forma as adjusted cash and cash equivalents, additional paid-in capital, total stockholders’ equity and total capitalization by approximately $ million, assuming an initial public offering price of $ per share, which is the midpoint of the estimated initial public offering price range set forth on the cover page of this prospectus, after deducting underwriting discounts and commissions payable by us. |
The actual column in the table above is based on 5,747,638 shares of our common stock issued and outstanding and excludes 24,818 shares of common stock issued upon early exercise of stock options and subject to repurchase and 755,771 shares of unvested restricted stock. See Notes 10 and 12 to our Consolidated Financial Statements. The pro forma and pro forma as adjusted columns in the table above are based on 25,258,048 shares of our common stock (including preferred stock on an as-converted basis) outstanding as of June 30, 2015, and exclude the following:
| • | 2,460,574 shares of common stock issuable upon the exercise of options to purchase shares of our common stock outstanding as of June 30, 2015, at a weighted average exercise price of $5.27 per share; |
| • | 1,713,634 shares of common stock reserved for future grant or issuance under our 2014 Equity Incentive Plan as of June 30, 2015; |
| • | 867, 000 shares of common stock issuable upon the exercise of options to purchase shares of our common stock at an exercise price of $22.04 per share and 5,000 shares of restricted stock, which were granted on August 12, 2015; |
| • | 24,818 shares of common stock issued upon early exercise of stock options and subject to repurchase; |
| • | 755,771 shares of unvested restricted stock; |
| • | 3,000,000 shares of common stock initially reserved for future issuance under our Amended and Restated 2014 Equity Incentive Plan, which will become effective immediately prior to the completion of this offering, as well as any automatic increases in the number of shares of our common stock reserved for future issuance pursuant to this plan; and |
| • | 600,000 shares of common stock initially reserved for issuance under our ESPP, as well as any automatic increases in the number of shares of our common stock reserved for future issuance pursuant to this plan. |
43
If you invest in our common stock in this offering, your ownership interest will be diluted to the extent of the difference between the initial public offering price per share of our common stock and the pro forma as adjusted net tangible book value per share of our common stock immediately after this offering.
Our pro forma net tangible book value as of , 2015 was $ or $ per share of common stock. Pro forma net tangible book value per share represents tangible assets, less liabilities, divided by the aggregate number of shares of common stock outstanding, after giving effect to the automatic conversion of all outstanding shares of our preferred stock into an aggregate of 19,510,410 shares of our common stock, which conversion will occur immediately prior to the completion of this offering. After giving effect to the sale by us of the shares of common stock in this offering, at an assumed initial public offering price of $ per share, which is the midpoint of the estimated initial public offering price range set forth on the cover page of this prospectus, and after deducting underwriting discounts and commissions and estimated offering expenses payable by us, our pro forma as adjusted net tangible book value as of , 2015, would be $ or $ per share. This represents an immediate increase in pro forma net tangible book value to existing stockholders of $ per share and an immediate dilution to new investors of $ per share. The following table illustrates this per share dilution:
| Assumed initial public offering price |
$ | |||||||
| Pro forma net tangible book value per share as of , 2015 |
$ | |||||||
| Increase in pro forma net tangible book value per share attributable to new investors |
||||||||
| Pro forma as adjusted net tangible book value per share after offering |
||||||||
|
|
|
|||||||
| Dilution per share to new investors |
$ | |||||||
|
|
|
Each $1.00 increase or decrease in the assumed initial public offering price of $ per share, which is the midpoint of the estimated initial public offering price range set forth on the cover page of this prospectus, would increase or decrease, as applicable, our pro forma as adjusted net tangible book value per share to new investors by $ , and would increase or decrease, as applicable, dilution per share to new investors in this offering by $ , assuming that the number of shares offered by us, as set forth on the cover page of this prospectus, remains the same and after deducting underwriting discounts and commissions and estimated offering expenses payable by us. Similarly, each increase or decrease of 1.0 million shares in the number of shares offered by us would increase or decrease, as applicable, our pro forma as adjusted net tangible book value by approximately $ per share and increase or decrease, as applicable, the dilution to new investors by $ per share, assuming the assumed initial public offering price remains the same, and after deducting underwriting discounts and commissions and estimated offering expenses payable by us.
If the underwriters’ option to purchase additional shares is exercised in full, the pro forma as adjusted net tangible book value per share of our common stock, as adjusted to give effect to this offering, would be $ per share, and the dilution in pro forma net tangible book value per share to new investors in this offering would be $ per share.
The following table sets forth, on a pro forma as adjusted basis, as of , 2015, the number of shares of common stock purchased from us, the total consideration paid, or to be paid, and the average price per share paid, or to be paid, by existing stockholders and by the new investors, at an assumed initial public offering price of $ per share, which is the midpoint of the estimated initial public offering range set forth on the cover page of this prospectus, before deducting underwriting discounts and commissions and estimated offering expenses payable by us:
| Shares Purchased | Total Consideration | Average Price Per Share |
||||||||||||||||
| Number |
Percent | Amount | Percent | |||||||||||||||
| Existing stockholders |
% | $ | % | $ | ||||||||||||||
| New investors |
$ | |||||||||||||||||
|
|
|
|
|
|
|
|
||||||||||||
| Total |
100.0 | % | $ | 100.0 | % | |||||||||||||
|
|
|
|
|
|
|
|
||||||||||||
44
Each $1.00 increase or decrease in the assumed initial public offering price of $ per share, which is the midpoint of the estimated initial public offering price range set forth on the cover page of this prospectus, would increase or decrease, as applicable, the total consideration paid by new investors and total consideration paid by all stockholders by approximately $ million, assuming that the number of shares offered by us, as set forth on the cover page of this prospectus, remains the same and after deducting underwriting discounts and commissions and estimated offering expenses payable by us.
The number of shares of common stock to be outstanding after this offering is based upon 26,038,637 shares outstanding (including preferred stock on an as-converted basis) as of June 30, 2015, and excludes:
| • | 2,460,574 shares of common stock issuable upon the exercise of options to purchase shares of our common stock outstanding as of June 30, 2015, at a weighted average exercise price of $5.27 per share; |
| • | 1,713,634 shares of common stock reserved for future grant or issuance under our 2014 Equity Incentive Plan as of June 30, 2015; |
| • | 867,000 shares of common stock issuable upon the exercise of options to purchase shares of our common stock at an exercise price of $22.04 per share and 5,000 shares of restricted stock, which were granted on August 12, 2015; |
| • | 3,000,000 shares of common stock initially reserved for future issuance under our Amended and Restated 2014 Equity Incentive Plan, which will become effective immediately prior to the completion of this offering, as well as any automatic increases in the number of shares of our common stock reserved for future issuance pursuant to this plan; and |
| • | 600,000 shares of common stock initially reserved for issuance under our ESPP, as well as any automatic increases in the number of shares of our common stock reserved for future issuance pursuant to this plan. |
To the extent options are exercised, there will be further dilution to new investors.
45
SELECTED CONSOLIDATED FINANCIAL DATA
The following selected consolidated financial data of Penumbra, Inc. should be read in conjunction with, and are qualified by reference to, the section titled “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and the consolidated financial statements and notes thereto included elsewhere in this prospectus. The consolidated statement of operations data for the years ended December 31, 2013 and 2014, and the consolidated balance sheet data as of December 31, 2013 and 2014, are derived from, and qualified by reference to, our audited consolidated financial statements included elsewhere in this prospectus and should be read in conjunction with those consolidated financial statements and notes thereto. The consolidated statement of operations data for the year ended December 31, 2012, are derived from our audited consolidated financial statements not included in this prospectus. The consolidated statement of operations data for the six month periods ended June 30, 2014 and 2015, and the consolidated balance sheet data as of June 30, 2015, are derived from our unaudited consolidated financial statements included elsewhere in this prospectus which, in our opinion, have been prepared on the same basis as the audited consolidated financial statements and reflect all adjustments, consisting only of normal recurring adjustments, necessary for a fair presentation of our results of operations and financial position. Our historical results are not necessarily indicative of the results that may be expected in the future and the results for the six months ended June 30, 2015 are not necessarily indicative of results to be expected for the full year or any other period.
| Year Ended December 31, | Six Months Ended June 30 | |||||||||||||||||||
| 2012 | 2013 | 2014 | 2014 | 2015 | ||||||||||||||||
| (in thousands, except share and per share amounts) | ||||||||||||||||||||
| Consolidated Statement of Operations Data: |
||||||||||||||||||||
| Revenue |
$ | 73,141 | $ | 88,848 | $ | 125,510 | $ | 57,643 | $ | 81,263 | ||||||||||
| Cost of revenue |
24,178 | 30,972 | 42,668 | 19,489 | 27,160 | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Gross profit |
48,963 | 57,876 | 82,842 | 38,154 | 54,103 | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Operating expenses: |
||||||||||||||||||||
| Research and development |
12,548 | 14,084 | 15,575 | 7,538 | 7,983 | |||||||||||||||
| Selling, general and administrative |
32,987 | 44,918 | 64,258 | 28,240 | 45,943 | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Total operating expenses |
45,535 | 59,002 | 79,833 | 35,778 | 53,926 | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Income (loss) from operations |
3,428 | (1,126 | ) | 3,009 | 2,376 | 177 | ||||||||||||||
| Interest income (expense), net |
244 | 345 | 439 | 39 | 385 | |||||||||||||||
| Other income (expense), net |
220 | (474 | ) | (309 | ) | (92 | ) | (498 | ) | |||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Income (loss) before provision for (benefit from) income taxes |
3,892 | (1,255 | ) | 3,139 | 2,323 | 64 | ||||||||||||||
| Provision for (benefit from) income taxes |
1,934 | (5,354 | ) | 894 | 666 | 233 | ||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Net income (loss) |
$ | 1,958 | $ | 4,099 | $ | 2,245 | $ | 1,657 | $ | (169 | ) | |||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Net income (loss) attributable to common stockholders |
$ | 412 | $ | 887 | $ | (833 | ) | $ | 355 | $ | (34 | ) | ||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Net income (loss) per share attributable to common stockholders |
||||||||||||||||||||
| Basic |
$ | 0.10 | $ | 0.21 | $ | (0.18 | ) | $ | 0.08 | $ | (0.01 | ) | ||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Diluted |
$ | 0.07 | $ | 0.14 | $ | (0.18 | ) | $ | 0.05 | $ | (0.01 | ) | ||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Weighted average shares used to compute net income (loss) per share attributable to common stockholders |
||||||||||||||||||||
| Basic |
4,153,121 | 4,304,396 | 4,609,375 | 4,520,898 | 5,000,375 | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Diluted |
5,886,126 | 6,500,835 | 4,609,375 | 6,743,140 | 5,000,375 | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Pro forma net income (loss) per share—Basic |
$ | 0.10 | $ | (0.01 | ) | |||||||||||||||
|
|
|
|
|
|||||||||||||||||
| Pro forma net income (loss) per share—Diluted |
$ | 0.09 | $ | (0.01 | ) | |||||||||||||||
|
|
|
|
|
|||||||||||||||||
| Weighted average shares used to compute the pro forma net income (loss) per share |
||||||||||||||||||||
| —Basic (unaudited) |
22,680,810 | 24,510,785 | ||||||||||||||||||
|
|
|
|
|
|||||||||||||||||
| —Diluted (unaudited) |
25,037,541 | 24,510,785 | ||||||||||||||||||
|
|
|
|
|
|||||||||||||||||
46
| As of December 31, | As of June 30, 2015 |
|||||||||||
| 2013 | 2014 | |||||||||||
| Consolidated Balance Sheet Data: |
||||||||||||
| Cash and cash equivalents |
$ | 4,131 | $ | 3,290 | $ | 36,764 | ||||||
| Marketable investments |
$ | 9,545 | $ | 48,253 | $ | — | ||||||
| Total assets |
$ | 71,147 | $ | 121,381 | $ | 129,070 | ||||||
| Long-term debt |
$ | — | $ | — | $ | — | ||||||
| Working capital |
$ | 46,401 | $ | 94,478 | $ | 91,298 | ||||||
| Preferred stock |
$ | 56,222 | $ | 111,467 | $ | 111,467 | ||||||
| Stockholders’ deficit |
$ | (8,062 | ) | $ | (12,370 | ) | $ | (11,099 | ) | |||
47
MANAGEMENT’S DISCUSSION AND ANALYSIS
OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
The following discussion and analysis contains forward-looking statements that involve risks and uncertainties. Our actual results may differ materially from those discussed in the forward-looking statements as a result of various factors, including those set forth in the sections titled “Risk Factors” and “Special Note Regarding Forward-Looking Statements.” The following discussion of our financial condition and results of operations should be read in conjunction with our consolidated financial statements included elsewhere in this prospectus, as well as the information presented under the section titled “Selected Consolidated Financial Data.”
Overview
Penumbra is a global interventional therapies company that designs, develops, manufactures and markets innovative medical devices. We have a broad portfolio of products that addresses challenging medical conditions and significant clinical needs across two major markets, neuro and peripheral vascular. The conditions that our products address include, among others, ischemic stroke, hemorrhagic stroke and various peripheral vascular conditions that can be treated through thrombectomy and embolization procedures.
We are an established company focused on the neuro market, and we recently expanded our business to include the peripheral vascular market. We focus on developing, manufacturing and marketing products for use by specialist physicians, including interventional neuroradiologists, neurosurgeons, interventional neurologists, interventional radiologists and vascular surgeons. We design our products to provide these specialist physicians with a means to drive improved clinical outcomes through faster and safer procedures. Studies involving our Penumbra System ischemic stroke products and Penumbra Coil 400 neurovascular embolization products, as well as initial study results for our Indigo peripheral thrombectomy products, have shown that these products generate significant overall cost savings to the healthcare system relative to procedures using other products.
Since our founding in 2004, we have invested heavily in our product development capabilities in our two key markets: neuro and peripheral vascular. We launched our first neurovascular product in 2007, our first peripheral vascular product in 2013 and our first neurosurgical product in 2014. To date, we have launched 14 product brands, and we expect to continue to develop and build our portfolio of products based on our thrombectomy, embolization and access technologies. Generally, when we introduce a next generation product or a new product designed to replace a current product, sales of the earlier generation product or the product replaced decline. Our research and development activities are centered around the development of new products and clinical activities designed to support our regulatory submissions and demonstrate the effectiveness of our products.
We manufacture substantially all of our products at our campus in Alameda, California and stock inventory of raw materials, components and finished goods at that location. We rely on a single or limited number of suppliers for certain raw materials and components, and we generally have no long-term supply arrangements with our suppliers, as we order on a purchase order basis. We ship all of our products from Alameda to our hospital customers and distributors worldwide pursuant to purchase orders. We typically recognize revenue when products are delivered to our hospital customers or distributors, other than our coils, which we ship to our hospital customers on a consignment basis, and for which we recognize revenue when the hospital customers utilize products in a procedure.
Hospitals purchase our products for use in procedures performed by their specialist physicians, generally seeking reimbursement from third party payors for procedures performed. We believe that the cost-effectiveness of our products is attractive to our hospital customers.
In 2014, 34% of our revenue was generated from customers located outside of the United States. Our sales outside of the United States are denominated principally in the euro and Japanese yen. As a result, we have foreign exchange exposure, but do not currently engage in hedging. In 2014, no single hospital and only one distributor accounted for more than 10% of our sales.
As of June 30, 2015, we had approximately 1,000 employees worldwide. We sell our products to hospitals primarily through our direct sales organization in the United States, most of Europe, Canada and Australia, as well as through distributors in select international markets. In 2014, we generated revenue of $125.5 million,
48
which represents a 41.3% increase over 2013, and $3.0 million in operating income as compared to an operating loss of $1.1 million in 2013. For the six months ended June 30, 2015, we generated revenue of $81.3 million, which represents a 41.0% increase over the six months ended June 30, 2014, and $0.2 million in operating income as compared to operating income of $2.4 million for the six months ended June 30, 2014.
Factors Affecting Our Performance
There are a number of factors that have impacted, and we believe will continue to impact, our results of operations and growth. These factors include:
| • | The rate at which we grow our salesforce and the speed at which newly hired salespeople become fully effective can impact our revenue growth or our costs incurred in anticipation of such growth. |
| • | Our industry is intensely competitive and, in particular, we compete with a number of large, well-capitalized companies. We must continue to successfully compete in light of our competitors’ existing and future products and their resources to successfully market to the specialist physicians who use our products. |
| • | We must continue to successfully introduce new products that gain acceptance with specialist physicians and successfully transition from existing products to new products, ensuring adequate supply while avoiding excess inventory of older products and resulting inventory write-downs or write-offs. In addition, as we introduce new products, we generally build our inventory of components and finished goods in advance of sales, which may cause quarterly fluctuations in our results of operations. |
| • | Publications of clinical results by us, our competitors and other third parties can have a significant influence on whether, and the degree to which, our products are used by specialist physicians and the procedures and treatments those physicians choose to administer for a given condition. |
| • | The specialist physicians who use our products may not perform procedures during certain times of the year, such as those periods when they are at major medical conferences or are away from their practices for other reasons, the timing of which occurs irregularly during the year and from year to year. |
In addition, we have experienced and expect to continue to experience meaningful variability in our quarterly revenue and gross profit as a result of a number of factors, including, but not limited to: the number of available selling days, which can be impacted by holidays; the mix of products sold; the geographic mix of where products are sold; the demand for our products and the products of our competitors; the timing of or failure to obtain regulatory approvals or clearances for products; increased competition; the timing of customer orders; inventory write-offs and write-downs; costs, benefits and timing of new product introductions; the availability and cost of components and raw materials; and fluctuations in foreign currency exchange rates. We experience quarters in which we have significant revenue growth sequentially followed by quarters of little or no revenue growth. Additionally, we experience quarters in which operating expenses, in particular research and development expenses, fluctuate depending on the stage and timing of product development.
Components of Results of Operations
Revenue. We sell our products directly to hospitals and through distributors for use in procedures performed by specialist physicians to treat patients in two key markets: neuro and peripheral vascular disease. We sell our products through purchase orders, and we do not have long term purchase commitments from our customers. We typically recognize revenue when products are delivered to our hospital customers or distributors. However, with respect to products that we consign to hospitals, which primarily consist of coils, we recognize revenue at the time hospitals utilize products in a procedure. Revenue also includes shipping and handling costs that we charge to customers.
Cost of Revenue. Cost of revenue consists primarily of the cost of raw materials and components, personnel costs, including stock-based compensation, inbound freight charges, receiving costs, inspection and testing costs, warehousing costs, royalty expense, shipping and handling costs and other labor and overhead costs incurred in the manufacturing of products. We manufacture substantially all of our products in our manufacturing facility at our campus in Alameda, California.
49
Operating Expenses
Research and Development. Research and development expenses include product development, clinical and regulatory expenses, materials, depreciation and other costs associated with the development of our products. Research and development expenses also include salaries, benefits and other related costs, including stock-based compensation, for personnel and consultants. We expense research and development costs as they are incurred.
We expect to incur additional research and development costs as we continue to innovate and develop new products and engage in ongoing clinical research. These costs will generally increase in absolute terms as we continue to expand our product pipeline and add personnel.
Sales, General and Administrative. Sales, general and administrative expenses primarily consist of salaries, benefits and other related costs, including stock-based compensation, for personnel and consultants engaged in sales, marketing, finance, legal, compliance, administrative, information technology, medical education and training and human resource activities. Our sales, general and administrative expenses also include commissions, generally based on a percentage of sales, to direct sales representatives and the medical device excise tax, which is approximately 2.3% of U.S. sales.
We expect our sales, general and administrative expenses to continue to increase in absolute terms as we expand our salesforce and operations. Additionally, we expect to incur increased expenses related to headcount, professional service fees, systems and other infrastructure related to operating as a public company.
Income Tax Expense. We are taxed at the rates applicable within each jurisdiction where we sell products. The composite income tax rate, tax provisions, deferred tax assets and deferred tax liabilities will vary according to the jurisdiction in which profits arise. Tax laws are complex and subject to different interpretations by management and the respective governmental taxing authorities, and require us to exercise judgment in determining our income tax provision, our deferred tax assets and liabilities and the potential valuation allowance recorded against our net deferred tax assets. Deferred tax assets and liabilities are determined using the enacted tax rates in effect for the years in which those tax assets are expected to be realized. A valuation allowance is established when it is more likely than not that the future realization of all or some of the deferred tax assets will not be achieved.
50
Results of Operations
The following table sets forth the components of our consolidated statements of operations in dollars and as a percentage of revenue for the periods presented:
| Year Ended December 31, | Six Months Ended June 30, | |||||||||||||||||||||||||||||||
| 2013 | 2014 | 2014 | 2015 | |||||||||||||||||||||||||||||
| (in thousands, except for percentages) | ||||||||||||||||||||||||||||||||
| Revenue |
$ | 88,848 | 100.0 | % | $ | 125,510 | 100.0 | % | $ | 57,643 | 100.0 | % | $ | 81,263 | 100.0 | % | ||||||||||||||||
| Cost of revenue |
30,972 | 34.9 | % | 42,668 | 34.0 | % | 19,489 | 33.8 | % | 27,160 | 33.4 | % | ||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
| Gross profit |
57,876 | 65.1 | % | 82,842 | 66.0 | % | 38,154 | 66.2 | % | 54,103 | 66.6 | % | ||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
| Operating expenses: |
||||||||||||||||||||||||||||||||
| Research and development |
14,084 | 15.9 | % | 15,575 | 12.4 | % | 7,538 | 13.1 | % | 7,983 | 9.8 | % | ||||||||||||||||||||
| Sales, general and administrative |
44,918 | 50.6 | % | 64,258 | 51.2 | % | 28,240 | 49.0 | % | 45,943 | 56.5 | % | ||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
| Total operating expenses |
59,002 | 66.4 | % | 79,833 | 63.6 | % | 35,778 | 62.1 | % | 53,926 | 66.4 | % | ||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
| Income (loss) from operations |
(1,126 | ) | (1.3 | )% | 3,009 | 2.4 | % | 2,376 | 4.1 | % | 177 | 0.2 | % | |||||||||||||||||||
| Interest income (expense), net |
345 | 0.4 | % | 439 | 0.3 | % | 39 | 0.1 | % | 385 | 0.5 | % | ||||||||||||||||||||
| Other income (expense), net |
(474 | ) | (0.5 | )% | (309 | ) | (0.2 | )% | (92 | ) | (0.2 | )% | (498 | ) | (0.6 | )% | ||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
| Income (loss) before provision for (benefit from) income taxes |
(1,255 | ) | (1.4 | )% | 3,139 | 2.5 | % | 2,323 | 4.0 | % | 64 | 0.1 | % | |||||||||||||||||||
| Provision for (benefit from) income taxes |
(5,354 | ) | (6.0 | )% | 894 | 0.7 | % | 666 | 1.2 | % | 233 | 0.3 | % | |||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
| Net income (loss) |
$ | 4,099 | 4.6 | % | $ | 2,245 | 1.8 | % | $ | 1,657 | 2.9 | % | $ | (169 | ) | (0.2 | )% | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
Six Months Ended June 30, 2015 Compared to Six Months Ended June 30, 2014
Revenue
| Six Months Ended June 30, | Change | |||||||||||||||
| 2014 | 2015 | $ | % | |||||||||||||
| (in thousands, except for percentages) | ||||||||||||||||
| Neuro |
$ | 50,068 | $ | 66,054 | $ | 15,986 | 31.9 | % | ||||||||
| Peripheral Vascular |
7,575 | 15,209 | 7,634 | 100.8 | % | |||||||||||
|
|
|
|
|
|
|
|||||||||||
| Total |
$ | 57,643 | $ | 81,263 | $ | 23,620 | 41.0 | % | ||||||||
|
|
|
|
|
|
|
|||||||||||
Revenue increased $23.6 million, or 41.0%, to $81.3 million in the six months ended June 30, 2015, from $57.6 million in the six months ended June 30, 2014. Our revenue growth was due to expansion of our salesforce headcount by 42.5%, further market penetration of our existing products and sales of new products. Increased sales of Penumbra System products accounted for more than half of the revenue increase in the six months ended June 30, 2015. Additionally, approximately 10.4% of the increase in revenue for the six months ended June 30, 2015 came from the sale of new products, including our Indigo peripheral thrombectomy product, our Apollo System and our Benchmark neuro access product.
Revenue from sales in the United States increased $17.0 million, or 46.0%, to $54.0 million in the six months ended June 30, 2015, from $37.0 million in the six months ended June 30, 2014. Revenue from sales in international markets increased $6.6 million, or 32.1%, to $27.3 million in the six months ended June 30, 2015, from $20.7 million in the six months ended June 30, 2014. Revenue from international sales represented 34% and 36% of our total revenue for the six months ended June 30, 2015 and 2014, respectively.
51
Revenue from our neuro products increased $16.0 million, or 31.9%, to $66.1 million in the six months ended June 30, 2015, from $50.1 million in the six months ended June 30, 2014. Our neuro product sales experienced strong momentum due to further market penetration and growth in the market following the presentation and publication of trial results in the fourth quarter of 2014, and the presentation and publication of additional trial results in the first quarter of 2015, each of which support endovascular treatment of stroke. We believe that these published trial results led to increases in the number of procedures performed by specialist physicians using our products. Increased sales of Penumbra System products accounted for most of the revenue increase in the six months ended June 30, 2015. Further, while our introduction of Benchmark in the fourth quarter 2014 was designed as a potential replacement for our Neuron access products, sales of our Neuron access products have increased slightly since Benchmark was introduced. The increase in revenue from our neuro products was partially offset by a decrease in sales of our neuro embolization products in the six months ended June 30, 2015. Prices for our neuro products remained substantially flat during the period.
Revenue from our peripheral vascular products increased $7.6 million, or 100.8%, to $15.2 million in the six months ended June 30, 2015, from $7.6 million in the six months ended June 30, 2014. Our peripheral embolization and peripheral thrombectomy products experienced strong volume growth in the period, primarily due to the focused efforts of our dedicated peripheral vascular salesforce, which was established in the second half of 2014, and further market penetration of our products. Prices for our peripheral vascular products remained substantially flat during the period.
Gross Profit and Gross Margin
| Six Months Ended June 30, | Change | |||||||||||||||
| 2014 | 2015 | $ | % | |||||||||||||
| (in thousands, except for percentages) | ||||||||||||||||
| Cost of revenue |
$ | 19,489 | $ | 27,160 | $ | 7,671 | 39.4 | % | ||||||||
|
|
|
|
|
|
|
|||||||||||
| Gross profit |
$ | 38,154 | $ | 54,103 | $ | 15,949 | 41.8 | % | ||||||||
|
|
|
|
|
|
|
|||||||||||
| Gross margin % |
66.2 | % | 66.6 | % | ||||||||||||
|
|
|
|
|
|||||||||||||
Gross profit increased $15.9 million, or 41.8%, to $54.1 million in the six months ended June 30, 2015, from $38.2 million in the six months ended June 30, 2014. The increase in gross profit was primarily due to an increase in revenue from sales of our neuro and peripheral vascular products.
Gross margin increased 0.4 percentage points to 66.6% in the six months ended June 30, 2015, from 66.2% in the six months ended June 30, 2014. The increase in gross margin was primarily due to geographic and product mix.
Research and Development (R&D)
| Six Months Ended June 30, | Change | |||||||||||||||
| 2014 | 2015 | $ | % | |||||||||||||
| (in thousands, except for percentages) | ||||||||||||||||
| R&D |
$ | 7,538 | $ | 7,983 | $ | 445 | 5.9 | % | ||||||||
|
|
|
|
|
|
|
|||||||||||
| R&D as a percentage of revenue |
13.1 | % | 9.8 | % | ||||||||||||
R&D expenses increased by $0.5 million, or 5.9%, to $8.0 million in the six months ended June 30, 2015, from $7.5 million in the six months ended June 30, 2014. The $0.5 million increase in R&D expenses was primarily due to a $0.9 million increase in compensation expense resulting from increased headcount to support continued investment in our products, a $0.2 million increase in travel related expenses and a $0.2 million increase in expenses related to demo products, partially offset by $0.8 million reduced R&D spend due to the stage and timing of development activities on our projects.
52
Sales, General and Administrative (SG&A)
| Six Months Ended June 30, | Change | |||||||||||||||
| 2014 | 2015 | $ | % | |||||||||||||
| (in thousands, except for percentages) | ||||||||||||||||
| SG&A |
$ | 28,240 | $ | 45,943 | $ | 17,703 | 62.7 | % | ||||||||
|
|
|
|
|
|
|
|||||||||||
| SG&A as a percentage of revenue |
49.0 | % | 56.5 | % | ||||||||||||
SG&A expenses increased by $17.7 million, or 62.7% to $45.9 million in the six months ended June 30, 2015, from $28.2 million in the six months ended June 30, 2014. Our sales and administrative headcount in the six months ended June 30, 2015 increased by 61.8%, which led to a $11.5 million increase in compensation expense. Additionally, SG&A expenses were impacted by a $2.5 million increase due to expanded marketing programs, a $1.7 million increase in legal, professional and consulting expenses and a $1.0 million increase in travel-related expenses of our salesforce to support our sales activities.
Provision for Income Taxes
| Six Months Ended June 30, | Change | |||||||||||||||
| 2014 | 2015 | $ | % | |||||||||||||
| (in thousands, except for percentages) | ||||||||||||||||
| Provision for income taxes |
666 | 233 | $ | (433 | ) | nm | ||||||||||
|
|
|
|
|
|
|
|||||||||||
| Effective tax rate |
28.7 | % | 364.1 | % | ||||||||||||
Our provision for income taxes decreased $0.4 million, to $0.2 million in the six months ended June 30, 2015, from $0.7 million in the six months ended June 30, 2014. Our effective tax rate increased to 364.1% for the six months ended June 30, 2015, compared to 28.7% for the six months ended June 30, 2014. The effective tax rate is based on a projection of our full year results. The higher effective tax rate for the six months ended June 30, 2015 was primarily due to the change in mix of jurisdictional profits.
Year Ended December 31, 2014 Compared to Year Ended December 31, 2013
Revenue
| Year Ended December 31, | Change | |||||||||||||||
| 2013 | 2014 | $ | % | |||||||||||||
| (in thousands, except for percentages) | ||||||||||||||||
| Neuro |
$ | 81,343 | $ | 106,242 | $ | 24,899 | 30.6 | % | ||||||||
| Peripheral Vascular |
7,505 | 19,268 | 11,763 | 156.7 | % | |||||||||||
|
|
|
|
|
|
|
|||||||||||
| Total |
$ | 88,848 | $ | 125,510 | $ | 36,662 | 41.3 | % | ||||||||
|
|
|
|
|
|
|
|||||||||||
Revenue increased $36.7 million, or 41.3%, to $125.5 million in 2014, from $88.8 million in 2013. Our revenue growth was primarily due to expansion of our salesforce headcount by 55.8%, further market penetration of our existing products and sales of new products. Increased sales of Penumbra System products accounted for approximately half of the revenue increase in 2014. With respect to the impact of the introduction of new products, 12.1% of the increase in 2014 revenues came from the sale of new products, including our Indigo peripheral thrombectomy product, our Apollo System and our Benchmark neuro access product.
Revenue from sales in the United States increased $24.7 million, or 42.3%, to $83.0 million in 2014, from $58.3 million in 2013. Revenue from sales in international markets increased $12.0 million, or 39.3%, to $42.5 million in 2014, from $30.5 million in 2013. Revenue from international sales represented 34% and 34% of our total revenue for 2014 and 2013, respectively.
Revenue from our neuro products increased $24.9 million, or 30.6%, to $106.2 million in 2014, from $81.3 million in 2013. Our neuro product sales experienced strong momentum due to further market penetration and growth in the market following presentation of trial results in October 2014, which support endovascular treatment of stroke. Additionally, our neuro product sales benefited from the launch of our ACE Reperfusion Catheter in July 2013. Prices for our neuro products remained substantially flat during the period.
53
Revenue from our peripheral vascular products increased $11.8 million, or 156.7%, to $19.3 million in 2014, from $7.5 million in 2013. Peripheral vascular product sales volume growth benefited from the focused efforts of our dedicated peripheral vascular salesforce, which was established in the second half of 2014, further market penetration and sales from our recently launched Indigo product. Prices for our peripheral vascular products remained substantially flat during the period.
Gross Profit and Gross Margin
| Year Ended December 31, | Change | |||||||||||||||
| 2013 | 2014 | $ | % | |||||||||||||
| (in thousands, except for percentages) | ||||||||||||||||
| Cost of revenue |
$ | 30,972 | $ | 42,668 | $ | 11,696 | 37.8 | % | ||||||||
| Gross profit |
$ | 57,876 | $ | 82,842 | $ | 24,966 | 43.1 | % | ||||||||
| Gross margin % |
65.1 | % | 66.0 | % | ||||||||||||
Gross profit increased $25.0 million, or 43.1%, to $82.8 million in 2014, from $57.9 million in 2013. The increase in gross profit was primarily driven by an increase in revenue from sales of our neuro and peripheral vascular products.
Gross margin increased 0.9 percentage points, to 66.0% in 2014, from 65.1% in 2013. The increase in gross margin is attributable to geographic and product mix and improved manufacturing efficiency.
Research and Development (R&D) Expenses
| Year Ended December 31, | Change | |||||||||||||||
| 2013 | 2014 | $ | % | |||||||||||||
| (in thousands, except for percentages) | ||||||||||||||||
| R&D |
$ | 14,084 | $ | 15,575 | $ | 1,491 | 10.6 | % | ||||||||
| R&D as a percentage of revenue |
15.9 | % | 12.4 | % | ||||||||||||
R&D expenses increased $1.5 million, or 10.6%, to $15.6 million in 2014, from $14.1 million in 2013. The $1.5 million increase was primarily due to a $1.2 million increase in compensation expense resulting from increased headcount to support continued research and development in our products and a $0.3 million increase in consulting costs.
Sales, General and Administrative (SG&A) Expenses
| Year Ended December 31, | Change | |||||||||||||||
| 2013 | 2014 | $ | % | |||||||||||||
| (in thousands, except for percentages) | ||||||||||||||||
| SG&A |
$ | 44,918 | $ | 64,258 | $ | 19,340 | 43.1 | % | ||||||||
| SG&A as a percentage of revenue |
50.6 | % | 51.2 | % | ||||||||||||
SG&A expenses increased by $19.3 million, or 43.1%, to $64.3 million in 2014, from $44.9 million in 2013. The $19.3 million increase was primarily due to a $10.9 million increase in compensation expense resulting from increased headcount, a $2.1 million increase in legal, professional and consulting fees, a $2.1 million increase in marketing expenses due to expanded marketing programs and a $1.5 million increase in travel-related expenses for our salesforce to support our sales activities.
Provision for Income Taxes
| Year Ended December 31, | Change | |||||||||||||||
| 2013 | 2014 | $ | % | |||||||||||||
| (in thousands, except for percentages) | ||||||||||||||||
| Provision for (benefit from) income taxes |
$ | (5,354 | ) | $ | 894 | $ | 6,248 | nm | ||||||||
| Effective tax rate |
nm | 28.5 | % | |||||||||||||
54
Our provision for income taxes was $0.9 million in 2014 compared to a tax benefit of $5.4 million in 2013. The higher tax provision in 2014 was due to the release of a valuation allowance of $5.0 million against net deferred tax assets in 2013 and the reduced availability of net operating loss carryforwards in 2014. Our effective tax rate was 28.5% in 2014. The effective tax rate in 2014 was lower than the U.S. federal statutory rate due to the impact of federal income tax credits.
Prior to 2013, we recorded a valuation allowance in the full amount of our net deferred tax assets, as we had assessed our cumulative loss position and determined that the future benefits were not more likely than not to be realized. As of December 31, 2013, we determined that it is more likely than not that a portion of the net deferred tax assets will be realized for federal and state income tax purposes in the U.S., except in California, and released $5.0 million of the valuation allowance. We continue to record a valuation allowance in the full amount of the net deferred tax assets attributable to California and certain foreign jurisdictions.
Liquidity and Capital Resources
Since our inception, we have financed our operations through private placements of preferred stock. As of June 30, 2015, we had $91.3 million in working capital, which included $36.8 million in cash and cash equivalents.
In addition to our existing cash and cash equivalents and marketable investment balances, our principal source of liquidity is our accounts receivable. We believe these sources will provide sufficient liquidity for us to meet our liquidity requirements for at least the next 12 months. Our principal liquidity requirements are to fund our operations, including our research and development, and capital expenditures. To facilitate our expansion, we may also lease or purchase additional facilities. We expect to continue to make investments as we launch new products, expand our manufacturing operations and further expand into international markets. We may, however, require or elect to secure additional financing as we continue to execute our business strategy. If we require additional funds, we may seek to raise capital through equity or debt financing, which may not be available on acceptable terms, could result in dilution to our stockholders and could require us to agree to covenants that limit our operating flexibility.
| December 31, | June 30, |
|||||||||||
| 2013 | 2014 | 2015 | ||||||||||
| (in thousands) | ||||||||||||
| Cash and cash equivalents |
$ | 4,131 | $ | 3,290 | $ | 36,764 | ||||||
| Marketable investments |
$ | 9,545 | $ | 48,253 | $ | — | ||||||
| Accounts receivable, net |
$ | 13,074 | $ | 18,912 | $ | 23,604 | ||||||
| Accounts payable |
$ | 1,312 | $ | 2,348 | $ | 3,721 | ||||||
| Accrued liabilities |
$ | 14,525 | $ | 18,475 | $ | 23,168 | ||||||
| Working capital(1) |
$ | 46,401 | $ | 94,478 | $ | 91,298 | ||||||
| (1) | Working capital consists of total current assets less total current liabilities. |
The following table sets forth, for the periods indicated, our beginning balance of cash and cash equivalents, net cash flows provided by (used in) operating, investing and financing activities and our ending balance of cash and cash equivalents:
| Year Ended December 31, | Six Months Ended June 30, | |||||||||||||||
| 2013 | 2014 | 2014 | 2015 | |||||||||||||
| (in thousands) | ||||||||||||||||
| Cash and cash equivalents at beginning of period |
$ | 7,435 | $ | 4,131 | $ | 4,131 | $ | 3,290 | ||||||||
| Net cash provided by (used in) operating activities |
$ | (3,396 | ) | $ | (6,389 | ) | $ | 1,992 | $ | (8,331 | ) | |||||
| Net cash provided by (used in) investing activities |
$ | (1,251 | ) | $ | (37,001 | ) | $ | (940 | ) | $ | 45,018 | |||||
| Net cash provided by (used in) financing activities |
$ | 2,178 | $ | 42,897 | $ | 50,495 | $ | (2,974 | ) | |||||||
| Cash and cash equivalents at end of period |
$ | 4,131 | $ | 3,290 | $ | 55,560 | $ | 36,764 | ||||||||
55
Net Cash Provided by (Used in) Operating Activities
Cash provided by (used in) operating activities consists primarily of net income adjusted for certain non-cash items (including depreciation and amortization, inventory write downs, stock-based compensation expense, provision for doubtful accounts, provision for sales returns, loss on minority investment, loss on disposal of property and equipment, provision for product warranty), and the effect of changes in working capital and other activities.
Net cash used in operating activities was $8.3 million during the six months ended June 30, 2015 and consisted of net loss of $0.2 million and net changes in operating assets and liabilities of $13.8 million, partially offset by non-cash items of $5.6 million. The change in operating assets and liabilities include the increase in inventories of $11.3 million to support our revenue growth, an increase in accounts receivable of $4.9 million, an increase in prepaid expenses and other current and non-current assets of $2.8 million, partially offset by an increase in accrued expenses and other non-current liabilities of $4.7 million and accounts payable of $0.5 million, as a result of the growth in our business activities.
Net cash provided by operating activities was $2.0 million during the six months ended June 30, 2014, and consisted of net income of $1.7 million and non-cash items of $1.7 million, partially offset by net changes in operating assets and liabilities of $1.3 million.
Net cash used by operating activities was $6.4 million in 2014 and consisted of net income of $2.2 million and non-cash items of $3.5 million, offset by net changes in operating assets and liabilities of $12.1 million. The change in operating assets and liabilities include an increase in inventories of $9.4 million to support our revenue growth and a corresponding increase in accounts receivable of $7.4 million and an increase in prepaid expenses and other current and non-current assets of $1.9 million partially offset by an increase in accrued expenses and other non current liabilities of $5.4 million and accounts payable of $1.3 million as a result of the growth in our business activities.
Net cash used by operating activities was $3.4 million in 2013 and consisted of net income of $4.1 million, and a change in non-cash items of $2.5 million, which includes a release of a valuation allowance of $5.0 million and a change in deferred tax benefits of $0.3 million, partially offset by net changes in operating assets and liabilities of $5.0 million. The significant items in the change in operating assets and liabilities include increases in inventories and accounts receivable of $3.8 million and $1.6 million, respectively, partially offset by increase in accrued expenses and other non-current liabilities of $1.7 million.
Net Cash Provided by (Used in) Investing Activities
Cash provided by (used in) investing activities relates primarily to divestures or purchases of marketable investments and capital expenditures.
Net cash provided by investing activities was $45.0 million during the six months ended June 30, 2015 and consisted of net proceeds from sales of marketable investments of $48.1 million partially offset by capital expenditures of $3.1 million.
Net cash used in investing activities was $0.9 million during the six months ended June 30, 2014 and consisted of capital expenditures of $0.8 million and net purchase of marketable investments of $0.1 million.
Net cash used in investing activities was $37.0 million in 2014 and consisted of net purchase of marketable investments of $33.1 million and capital expenditures of $3.9 million.
Net cash used in investing activities was $1.3 million during in 2013 and consisted of capital expenditures of $0.8 million and net purchase of marketable investments of $0.5 million.
Net Cash Provided by (Used in) Financing Activities
Cash from financing activities primarily relates to capital raising activities through equity or debt financing.
56
Financing activities in the six months ended June 30, 2015 used cash of $3.0 million and consisted of payment of employee taxes related to vested common and restricted stock of $2.5 million and payments of deferred issuance costs of $1.0 million, partially offset by proceeds from exercises of stock options of $0.5 million.
Financing activities in the six months ended June 30, 2014 provided $50.5 million and consisted of proceeds from the issuance of Series F Preferred Stock of $57.1 million, net of issuance costs and proceeds from exercises of stock options of $0.3 million. These proceeds were offset in part by the repayment of amounts outstanding under our credit facility of $6.0 million upon its termination in May 2014 and repurchases of common stock and stock options of $0.9 million.
Financing activities in 2014 provided $42.9 million and consisted of proceeds from issuance of Series F Preferred Stock of $57.2 million, net of issuance costs and proceeds from exercises of stock options of $1.0 million. These proceeds were partially offset by repurchases of preferred stock, common stock and stock options of $9.4 million and the repayment of amounts outstanding under our credit facility of $6.0 million upon its termination in May 2014.
Financing activities in 2013 provided $2.2 million and consisted of proceeds from our credit facility of $2.0 million and proceeds from exercises of stock options of $0.2 million.
Indebtedness
In May 2012, we entered into a $15.0 million revolving credit facility with Wells Fargo Bank, National Association. The credit facility was collateralized by our investment balances. The interest on the credit facility was based on the daily one-month London Inter-Bank Offered Rate (LIBOR), plus 1.75% and was payable monthly. The outstanding balance on the credit facility was due in full on June 1, 2015. The credit facility contained customary covenants for credit facilities of this type, including limitations on disposition of assets and changes in control. In May 2014, in conjunction with our Series F Preferred Stock financing, we paid the then outstanding balance on the credit facility and terminated the credit facility.
Contractual Obligations and Commitments
The following table summarizes our contractual obligations as of December 31, 2014:
| Payments Due by Period | ||||||||||||||||||||
| Total | Less Than One Year |
1-3 Years | 3-5 Years | More than Five Years |
||||||||||||||||
| (in thousands) | ||||||||||||||||||||
| Rent obligations(1) |
$ | 35,711 | $ | 2,010 | $ | 4,140 | $ | 4,405 | $ | 25,156 | ||||||||||
| Equipment lease obligations(2) |
333 | 138 | 192 | 3 | — | |||||||||||||||
| Purchase commitments(3) |
9,862 | 6,169 | 3,693 | — | — | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Total |
$ | 45,906 | $ | 8,317 | $ | 8,025 | $ | 4,408 | $ | 25,156 | ||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| (1) | We lease our corporate headquarters and a manufacturing facility at our campus in Alameda, California pursuant to lease agreements that expire in November 2029. Additionally, we lease offices in Germany, Australia and Brazil. In June 2015, a lease for additional space at our campus in Alameda, California commenced upon our landlord’s substantial completion of tenant improvements. This lease expires in November 2029 and, as of June 30, 2015, represented a contractual obligation of $13.9 million. |
| (2) | We lease equipment and automobiles under operating leases. These leases expire at various dates through 2018. |
| (3) | Purchase commitments consist of contracts with suppliers to purchase raw materials to be used to manufacture products. |
The amounts in the table above exclude $0.6 million of income tax liabilities included in current liabilities as we are unable to reasonably estimate the timing of settlement. See Note 15 to our Consolidated Financial Statements. In addition, the table above does not reflect royalty obligations under a license agreement as amounts due thereunder fluctuate depending on sales levels. See Note 7 to our Consolidated Financial Statements.
57
Off-Balance Sheet Arrangements
We do not have any off-balance sheet arrangements or any holdings in variable interest entities.
Quantitative and Qualitative Disclosure about Market Risk
We are exposed to various market risks, which may result in potential losses arising from adverse changes in market rates, such as interest rates and foreign exchange rates. We do not enter into derivatives or other financial instruments for trading or speculative purposes and do not believe we are exposed to material market risk with respect to our cash and cash equivalents and/or our marketable investments.
Interest Rate Risk. We had cash and cash equivalents of $3.3 million and $36.8 million as of December 31, 2014 and June 30, 2015, respectively, which consisted of cash and highly liquid money market funds.
Foreign Exchange Risk Management. We operate in countries other than the United States, and, therefore, we are exposed to foreign currency risks. We bill most sales outside of the United States in local currencies, primarily the euro and Japanese yen. We expect that the percentage of our sales denominated in foreign currencies will increase in the foreseeable future as we continue to expand into international markets. When sales or expenses are not denominated in U.S. dollars, a fluctuation in exchange rates could affect our net income. We do not believe an immediate 10% adverse change in foreign exchange rates would have a material effect on our results of operations. We do not currently hedge our exposure to foreign currency exchange rate fluctuations; however, we may choose to hedge our exposure in the future.
Related-Party Transactions
For a description of our related-party transactions, see the section titled “Certain Relationships and Related-Party Transactions.”
Critical Accounting Policies and Estimates
Our consolidated financial statements have been prepared in accordance with accounting principles generally accepted in the United States. The preparation of our consolidated financial statements requires management to make estimates, assumptions and judgments that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the consolidated financial statements and the reported amounts of revenues and expenses during the applicable periods. Management bases its estimates, assumptions and judgments on historical experience and on various other factors that it believes to be reasonable under the circumstances. Different assumptions and judgments would change the estimates used in the preparation of our consolidated financial statements, which, in turn, could materially change our results from those reported. Management evaluates its estimates, assumptions and judgments on an ongoing basis. Historically, our critical accounting estimates have not differed materially from actual results. However, if our assumptions change, we may need to revise our estimates, or take other corrective actions, either of which may also have a material adverse effect on our consolidated statements of operations, liquidity and financial condition.
We believe the following critical accounting policies involve significant areas where management applies judgments and estimates in the preparation of our consolidated financial statements.
Segments
We have determined our operating segment on the same basis that we use to evaluate our performance internally. We have one business activity: the design, development, manufacturing and marketing of innovative medical devices, and we operate as one operating segment. Our chief operating decision-maker, our Chief Executive Officer, reviews our operating results for the purpose of allocating resources and evaluating financial performance. We determine revenue by geographic area, based on the destination to which we ship our products.
58
Revenue Recognition
Revenue is comprised of product revenue net of returns, discounts and administration fees. We recognize revenue when persuasive evidence of an arrangement exists, delivery has occurred, the sales price is fixed or determinable and collectability is reasonably assured. Evidence of an arrangement consists of customer orders, and we typically consider delivery to have occurred once title and risk of loss has been transferred and the product has been delivered to our customers. We typically recognize revenue when products are delivered to our hospital customers or distributors. However, with respect to products that we consign to hospitals, which primarily consist of coils, we recognize revenue at the time hospitals utilize our products used in a procedure.
We defer revenue for amounts that we have already invoiced our customers for and are ultimately expected to be recognized as revenue, but for which not all revenue recognition criteria have been met.
Our terms and conditions permit product returns and exchanges, and we record returns reserves in the period when revenue is recognized. Estimates are based on actual historical returns over the prior three years and are recorded as reductions in revenue at the time of sale. Upon recognition, we reduce revenue and cost of revenue for the estimated return. Return rates can fluctuate over time, but are sufficiently predictable to allow us to estimate expected future product returns.
Accounts Receivable
Accounts receivable are stated at invoice value less estimated allowances for returns and doubtful accounts. We continually monitor customer payments and maintain a reserve for estimated losses resulting from our customers’ inability to make required payments. We consider factors such as historical experience, credit quality, age of the accounts receivable balances, geographic related risks and economic conditions that may affect a customer’s ability to pay. In cases where there are circumstances that may impair a specific customer’s ability to meet its financial obligations, a specific allowance is recorded against amounts due, and thereby reduces the net recognized receivable to the amount reasonably believed to be collectible.
Inventories
Inventories are stated at the lower of cost (determined under the first-in first-out method) or market. Write-downs are provided for finished goods expected to become nonsaleable and provisions are specifically made for excessive, slow-moving or obsolete items. Market value is determined as the lower of replacement cost or net realizable value. We regularly review inventory quantities in consideration of actual loss experiences, projected future demand, and remaining shelf life to record a provision for excess and obsolete inventory when appropriate.
The estimate of excess quantities is subjective and primarily dependent on our estimate of future demand for a particular product. If the estimate of future demand is inaccurate based on actual sales, we may increase the write down for excess inventory for that component and record a charge to inventory impairment in the accompanying consolidated statements of operations and comprehensive income. We periodically evaluate the carrying value of inventory on hand for potential excess amount over demand using the same lower of cost or market approach as that has been used to value the inventory. We also periodically evaluate inventory quantities in consideration of actual loss experience.
Cost of Revenue
Cost of revenue includes direct and indirect costs associated with the manufacture of our products. Direct costs include material and labor, while indirect costs include, but are not limited to, inbound freight charges, receiving costs, inspection and testing costs, warehousing costs, royalty expense and other labor and overhead costs incurred in the manufacturing of products. Cost of revenue also includes stock-based compensation, warranty replacement costs, cost of revenue related to product return reserves, and excess and obsolete inventory write-downs.
59
Research and Development (R&D) Expenses
R&D costs include personnel-related costs, including stock-based compensation, regulatory, supplies, services, depreciation, allocated facilities and information services, clinical trial and related clinical manufacturing expenses, fees paid to investigative sites and other indirect costs.
Our clinical trial accruals are based on estimates of patient enrollment and related costs at clinical investigator sites. We estimate preclinical and clinical trial expenses based on the services performed pursuant to contracts with research institutions and clinical research organizations that conduct and manage preclinical studies and clinical trials on our behalf. In accruing service fees, we estimate the time period over which services will be performed and the level of patient enrollment and activity expended in each period. If the actual timing of the performance of services or the level of effort varies from the estimate, we will adjust the accrual accordingly. Payments made to third parties under these arrangements in advance of the receipt of the related services are recorded as prepaid expenses until the services are rendered.
Stock-Based Compensation
Stock-based compensation costs related to stock options granted to employees are measured at the date of grant based on the estimated fair value of the award, net of estimated forfeitures. We estimate the grant date fair value, and the resulting stock-based compensation expense, using the Black-Scholes option-pricing model on a straight-line basis over the requisite service period of the award, which is generally the vesting term of four years.
The Black-Scholes option-pricing model requires the use of highly subjective assumptions which determine the fair value of stock-based awards. The assumptions used in our option-pricing model represent management’s best estimates. These estimates are complex, involve a number of variables, uncertainties and assumptions and the application of management’s judgment, so that they are inherently subjective. If factors change and different assumptions are used, our stock-based compensation expense could be materially different in the future. These assumptions are estimated as follows:
Fair Value of Common Stock. Because our stock is not publicly traded, we must estimate its fair value, as discussed in “Common Stock Valuations” below.
Risk-Free Interest Rate. We base the risk-free interest rate used in the Black-Scholes valuation model on the implied yield available on U.S. Treasury zero-coupon issues with an equivalent remaining term of the options for each option group.
Expected Term. The expected term represents the period that our stock-based awards are expected to be outstanding. Because of the limitations on the sale or transfer or our common stock as a privately held company, we do not believe our historical exercise pattern is indicative of the pattern we will experience as a publicly traded company. We have consequently used the Staff Accounting Bulletin, or SAB, 110, simplified method to calculate the expected term, which is the average of the contractual term and vesting period. We plan to continue to use the SAB 110 simplified method until we have sufficient trading history as a publicly traded company.
Volatility. We determine the price volatility factor based on the historical volatilities of our peer group as we do not have a trading history for our common stock. Industry peers consist of several public companies in the medical device technology industry with comparable characteristics including enterprise value, risk profiles and position within the industry. We intend to continue to consistently apply this process using the same or similar public companies until a sufficient amount of historical information regarding the volatility of our own common stock share price becomes available, or unless circumstances change such that the identified companies are no longer similar to us, in which case, more suitable companies whose share prices are publicly available would be utilized in the calculation.
Dividend Yield. The expected dividend assumption is based on our current expectations about our anticipated dividend policy. We currently do not expect to issue any dividends.
60
In addition to assumptions used in the Black-Scholes option-pricing model, we must also estimate a forfeiture rate to calculate the stock-based compensation for our awards. We will continue to use judgment in evaluating the assumptions related to our stock-based compensation on a prospective basis. As we continue to accumulate additional data, we may have refinements to our estimates, which could materially impact our future stock-based compensation expense.
We used the following assumptions in our Black-Scholes option pricing model to determine the fair value of employee stock options:
| Year Ended December 31, | Six Months Ended June 30, | |||||||
| 2013 | 2014 | 2014 | 2015 | |||||
| (unaudited) | ||||||||
| Expected term (in years) |
6.25 | 6.25 | 6.25 | 6.25 | ||||
| Expected volatility |
45% | 45% | 45% | 45% | ||||
| Risk-free interest rate |
0.63% - 0.90% | 1.76% - 2.02% | 1.76% - 2.02% | 1.56% - 1.78% | ||||
| Expected dividend rate |
0% | 0% | 0% | 0% | ||||
The following table sets forth the stock-based compensation expense included in the consolidated statements of operations (in thousands):
| Year Ended December 31, | Six Months Ended June 30, | |||||||||||||||
| 2013 | 2014 | 2014 | 2015 | |||||||||||||
| (unaudited) | ||||||||||||||||
| Cost of sales |
$ | 98 | $ | 267 | $ | 124 | $ | 130 | ||||||||
| Research and development |
84 | 96 | 47 | 182 | ||||||||||||
| Sales, general and administrative |
704 | 1,070 | 531 | 3,304 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| $ | 886 | $ | 1,433 | $ | 702 | $ | 3,616 | |||||||||
|
|
|
|
|
|
|
|
|
|||||||||
As of June 30, 2015, we had approximately $11.4 million of total unrecognized compensation expense, net of related forfeiture estimates, which we expect to recognize over a weighted average period of approximately 3.5 years.
The intrinsic value of all outstanding options as of was $ million based on the estimated fair value of our common stock of $ per share, which is the midpoint of the estimated initial public offering price range set forth on the cover page of this prospectus.
Common Stock Valuations
The fair value of the shares of common stock underlying our stock options has historically been determined by our board of directors. Because there has been no public market for our common stock and in the absence of recent arm’s-length cash sales transactions of our common stock with independent third parties, our board of directors has determined the fair value of our common stock by considering at the time of grant a number of objective and subjective factors. Our board of directors intends all options granted to be exercisable at a price per share not less than the per share fair value of our common stock underlying those options on the date of grant. The estimated fair value of our common stock was determined at each valuation date in accordance with the guidelines outlined in the American Institute of Certified Public Accountants Practice Aid, Valuation of Privately-Held-Company Equity Securities Issued as Compensation. Our board of directors, with the assistance of management, developed these valuations using significant judgment and taking into account numerous factors, including the following:
| • | independent third-party valuations; |
| • | progress of research and development activities; |
| • | our operating and financial performance, including our levels of available capital resources; |
| • | rights and preferences of our common stock compared to the rights and preferences of our other outstanding equity securities; |
61
| • | equity market conditions affecting comparable public companies, as reflected in comparable companies’ market multiples, IPO valuations and other metrics; |
| • | the achievement of enterprise milestones, including our progress in clinical trials; |
| • | the likelihood of achieving a liquidity event for the shares of common stock, such as an IPO given prevailing market and medical device sector conditions; |
| • | sales of our preferred stock in arms-length transactions; |
| • | the illiquidity of our securities by virtue of being a private company; |
| • | business risks; and |
| • | management and board experience. |
We considered the following approaches in the preparation of our valuations:
| • | Market Approach. The market approach values a business by reference to guideline companies, for which enterprise values are known. This approach has two principal methodologies. The guideline public company methodology derives valuation multiples from the operating data and share prices of similar publicly traded companies. The guideline acquisition methodology focuses on comparisons between the subject company and guideline acquired public or private companies. |
| • | Option-Pricing Method Backsolve, or OPM backsolve. The OPM backsolve method derives the implied equity value for a company from a recent transaction involving the company’s own securities issued on an arms-length basis. |
| • | Probability Weighted Expected Return Method. Using the probability weighted expected return, or PWERM, method, the value of a company’s common stock is estimated based upon the analysis of future values for the company assuming various possible future liquidity events like an initial public offering, sale or merger. Share value is based upon the probability-weighted present value of expected future net cash flows, considering each of the possible future events, as well as the rights and preferences of each share class. |
In addition, we also considered an enterprise value allocation method:
| • | Option-Pricing Method, or OPM. Under this method, each class of stock is modeled as a call option with a distinct claim on the enterprise value of the company. The option’s exercise prices would be based on a comparison with the enterprise value. The method assumes that a formula, such as the Black-Scholes model, would calculate the fair value when provided with certain values, including share price, expiration date, volatility and the risk free interest rate. |
The per share common stock value was estimated by allocating our enterprise value using the OPM method in October 1, 2013 , May 16, 2014, and September 30, 2014, which determined the common stock value to be $7.75, $9.06 and $10.92, respectively. The per share common stock value was estimated in December 31, 2014, March 31, 2015 and June 30, 2015, which utilized the PWERM method, which determined the common stock value to be $12.36, $14.46 and $20.51, respectively.
In determining the estimated fair value of our common stock, our board of directors also considered the fact that our stockholders could not freely trade our common stock in the public markets. Accordingly, we applied discounts to reflect the lack of marketability of our common stock based on the expected time to liquidity. The estimated fair value of our common stock at each grant date reflected a non-marketability discount partially based on the anticipated likelihood and timing of a future liquidity event.
The key subjective factors and assumptions used in our valuations primarily consisted of: (i) the selection of the appropriate market comparable transactions, (ii) the selection of the appropriate comparable publicly traded companies, (iii) the financial forecasts utilized to determine future cash balances and necessary capital requirements, (iv) the probability and timing of the various possible liquidity events, (v) the estimated weighted average cost of capital and (vi) the discount for lack of marketability of our common stock.
62
At each grant date the board of directors reviewed any recent events and their potential impact on the estimated fair value per share of the common stock. As is provided for in Internal Revenue Code Section 409A, we generally rely on our valuations for up to twelve months unless we have experienced a material event that would have affected the estimated fair value per common share.
Income Taxes
We account for income taxes using the asset and liability method, whereby deferred tax asset and liability account balances are determined based on differences between the financial reporting and tax bases of assets and liabilities, and are measured using the enacted tax rates and laws that will be in effect when the differences are expected to reverse. We provide a valuation allowance to reduce the net deferred tax assets to their estimated realizable value.
At December 31, 2014, we had approximately $17.8 million and $2.0 million of state and foreign net operating loss carryforwards, respectively, available to offset future taxable income. The state net operating loss carryforwards will begin to expire in 2017. At December 31, 2014, we had research credits available to offset federal and state tax liabilities in the amount of $0.7 million and $2.8 million, respectively. Federal credits will begin to expire in 2027 and California state tax credits have no expiration. As of December 31, 2014, we had fully used $22.6 million of federal net operating losses, which we had accumulated in our first six years of operations, from 2004 through 2009.
The calculation of our current provision for income taxes involves the use of estimates, assumptions and judgments while taking into account current tax laws, interpretation of current tax laws and possible outcomes of future tax audits. We have established reserves to address potential exposures related to tax positions that could be challenged by tax authorities. Although we believe our estimates, assumptions and judgments to be reasonable, any changes in tax law or its interpretation of tax laws and the resolutions of potential tax audits could significantly impact the amounts provided for income taxes in our consolidated financial statements.
At June 30, 2015, our net deferred tax asset was $6.8 million, after reduction of a valuation allowance of $2.9 million. The calculation of our deferred tax asset balance involves the use of estimates, assumptions and judgments while taking into account estimates of the amounts and type of future taxable income. Deferred tax assets are reduced by a valuation allowance when it is more likely than not that the future realization of all or some of the deferred tax assets will not be achieved. Valuation allowances related to deferred tax assets can be affected by changes to tax laws, statutory tax rates, future taxable income levels and input from our tax advisors or regulatory authorities. If our management was to determine that we would not be able to realize all or a portion of our net deferred tax assets in the future, a valuation allowance related charge to earnings would be reflected in that period, which could have a material adverse impact on our financial condition and results of operations.
We have adopted ASC 740-10 “Accounting for Uncertainty in Income Taxes” that prescribes a recognition threshold and measurement attribute for the financial statement recognition and measurement of uncertain tax positions taken or expected to be taken in our income tax return, and also provides guidance on derecognition, classification, interest and penalties, accounting in interim periods, disclosure and transition.
We include interest and penalties related to unrecognized tax benefits within income tax expense in the accompanying consolidated statements of operations. We have not incurred any interest or penalties related to unrecognized tax benefits in any of the periods presented.
Recently Issued Accounting Standards
In May 2014, the FASB issued ASU No. 2014-09, Revenue from Contracts with Customers, which outlines a comprehensive new revenue recognition model designed to depict the transfer of promised goods or services to customers in an amount that reflects the consideration to which the entity expects to be entitled in exchange for those goods or services. The accounting standard is effective for annual reporting periods (including interim reporting periods within those periods) beginning after December 15, 2016. Early adoption is not permitted. We are currently evaluating the impact of this accounting standard.
63
In July 2015, the FASB voted to defer the effective date by one year to December 15, 2017 for annual reporting periods beginning after that date. The FASB also voted to permit early adoption of the standard, but not before the original effective date of December 15, 2016. The final accounting standard formally amending the effective date is expected to be issued by the FASB by the end of the third quarter of 2015.
In November 2014, the FASB issued ASU No. 2014-17, Pushdown Accounting, which provides an acquired entity to elect the option to apply pushdown accounting in the reporting period in which the change-in-control event occurs and allows the acquired entity to determine whether to elect to apply pushdown accounting for each individual change-in-control event in which the acquirer obtains control of the acquired entity. This accounting standard is effective as of November 18, 2014 and we will apply the guidance to future change-in control events.
In July 2015, the FASB issued ASU No. 2015-11, Simplifying the Measurement of Inventory, which requires an entity to measure most inventory at the lower of cost and net realizable value, thereby simplifying the current guidance under which an entity must measure inventory at the lower of cost or market. The accounting standard is effective prospectively for annual periods beginning after December 15, 2016 and interim periods therein. Early adoption is permitted as of the beginning of an interim or annual reporting period. We are currently evaluating the impact of this accounting standard.
64
Overview
Penumbra is a global interventional therapies company that designs, develops, manufactures and markets innovative medical devices. We have a broad portfolio of products that addresses challenging medical conditions and significant clinical needs across two major markets, neuro and peripheral vascular. The conditions that our products address include, among others, ischemic stroke, hemorrhagic stroke and various peripheral vascular conditions that can be treated through thrombectomy and embolization procedures.
We are an established company focused on the neuro market, and we recently expanded our business to include the peripheral vascular market. We sell our products to hospitals, primarily through our salesforce, as well as through distributors in select international markets. We focus on developing, manufacturing and marketing products for use by specialist physicians, including interventional neuroradiologists, neurosurgeons, interventional neurologists, interventional radiologists and vascular surgeons. We design our products to provide these specialist physicians with a means to drive improved clinical outcomes through faster and safer procedures. Studies involving our Penumbra System ischemic stroke products and Penumbra Coil 400 neurovascular embolization products, as well as initial study results for our Indigo peripheral thrombectomy products, have shown that these products generate significant overall cost savings to the healthcare system relative to procedures using other products.
We attribute our success to our culture built on cooperation, our highly efficient product innovation process, our disciplined approach to product and commercial development, our deep understanding of our target end markets and our relationships with specialist physicians. We believe these factors have enabled us to rapidly innovate in a highly capital-efficient manner.
Since our founding in 2004, we have had a strong track record of organic product development and commercial expansion that has established the foundation of our global organization. Some of our key accomplishments include:
| • | launching our first product, for neurovascular access, in the United States in 2007; |
| • | establishing our direct neuro salesforce in the United States and Europe in 2008; |
| • | launching the first 510(k)-cleared, aspiration-based product for the treatment of ischemic stroke patients in 2008, and launching four subsequent generations of that product; |
| • | launching our first neurovascular coil for the treatment of brain aneurysms in 2011; |
| • | launching our first peripheral vascular product in 2013; and |
| • | establishing our direct peripheral vascular salesforce in the United States and Europe in 2014. |
As of June 30, 2015, we had approximately 1,000 employees worldwide. We sell our products to hospitals primarily through our direct sales organization in the United States, most of Europe, Canada and Australia, as well as through distributors in select international markets. For the year ended December 31, 2014, we generated revenue of $125.5 million, which represents a 41.3% increase over 2013, and $3.0 million in operating income as compared to an operating loss of $1.1 million in 2013. For the six months ended June 30, 2015, we generated revenue of $81.3 million, which represents a 41.0% increase over the six months ended June 30, 2014, and $0.2 million in operating income as compared to operating income of $2.4 million for the six months ended June 30, 2014.
Market Opportunity
We estimate that the market for our current neuro and peripheral vascular products in the United States and Europe combined was approximately $1.3 billion in 2014, which we estimate represents growth of approximately 3.2% per year from 2012. While reliable third-party data is not available for markets outside the United States and Europe, we believe that there is a substantial additional market for our neuro and peripheral vascular products in the rest of the world.
65
We believe the market for our products remains substantially underpenetrated today, and that this market will experience significant growth as we and our competitors:
| • | generate additional clinical evidence supporting endovascular treatment of vascular disease; |
| • | improve existing technologies to enable physicians to treat vascular disease faster and more safely than previously possible; |
| • | support and educate the growing number of specialist physicians who treat vascular disease in the use of endovascular treatment; |
| • | grow the number of hospitals where endovascular treatment of vascular disease is available; and |
| • | raise patient awareness of endovascular treatment of vascular disease. |
We estimate that the market for our neuro products in the United States and Europe combined was over $500 million in 2014. According to the American Heart Association (AHA), the worldwide incidence of all forms of stroke was 33 million in 2010. We believe the recent publication of several major clinical trials demonstrating the benefit of endovascular treatment of ischemic stroke patients over the standard medical therapy will significantly expand the neuro market and support increased future growth.
We estimate that the market for our peripheral vascular products in the United States and Europe combined was over $750 million in 2014. Furthermore, according to a paper published in the journal The Lancet, 202 million people globally were living with peripheral artery disease in 2010. We believe that expanded adoption of new technologies developed to treat peripheral vascular disease will drive continued growth in this market.
Industry Background
Vascular Disease
Vascular disease refers to any condition that affects the circulatory system and typically manifests as a blockage or rupture of an artery or a vein. It may occur in any part of the body and is a condition that leads most often to blood vessel narrowing and obstruction, but can also lead to expansion of the blood vessel wall and blood vessel wall weakening and rupture. Vascular disease can cause a range of conditions, from pain to functional impairment, and it can require the amputation of a limb or result in death.
When the treatment for vascular disease is performed from within a vessel, it is referred to as an endovascular procedure. Endovascular procedures are minimally invasive means of treating the two major vascular problems that can develop within blood vessels: an occlusion, where the vessel is blocked or narrowed, and an aneurysm, or weakening of the vessel wall that forms a balloon-like pouch that fills with blood.
Endovascular procedures are performed by utilizing an accessible artery to reach an occlusion or aneurysm. During most endovascular procedures, a catheter is placed into the femoral artery in the groin, and X-ray imaging or fluoroscopy is used to help the physician visually navigate the catheter to the area to be treated. Endovascular procedures are less invasive and require a smaller incision than conventional open surgery, and we believe they are safer procedures for patients, reduce hospital stays and recovery times, and are more cost effective to the healthcare system.
Endovascular device markets are conventionally classified according to the anatomic location of the disorder. We currently focus our efforts on the neuro and peripheral vascular markets.
Neurovascular Disease
Neurovascular diseases are vascular diseases and disorders in the brain. A devastating complication of neurovascular disease is stroke. A stroke is caused by either an occlusion or rupture of an artery or vein. According to the AHA, the worldwide incidence of all forms of stroke was 33 million in 2010, and stroke was the second leading cause of death globally. The AHA estimates that there are approximately 795,000 strokes
66
annually in the United States, making stroke the fourth leading cause of death, and the leading cause of long-term disability. The AHA estimates that stroke results in total direct and indirect costs of approximately $33 billion in the United States annually. There are two principal forms of stroke:
| • | ischemic strokes, which are caused by the blockage of an artery to the brain and affect approximately 690,000 patients annually in the United States; and |
| • | hemorrhagic strokes, which are caused by a sudden rupture of a brain artery that leads to bleeding into or around the brain and affect approximately 105,000 patients annually in the United States. |
Both forms of stroke may result in permanent brain damage or death. Patients who survive a stroke are often left with disabilities, including paralysis, coma, impaired cognition, decreased coordination, loss of visual acuity, loss of speech, loss of sensation or some combination of these conditions. The devastating effects of stroke are one of the greatest costs to the healthcare system.
Ischemic Stroke
Ischemic strokes can be caused by several kinds of disease and can be characterized as either thrombotic or embolic. A thrombotic stroke is the most common and occurs when arteries in the brain become blocked by the formation of a clot within the brain. An embolic stroke occurs when a clot or small piece of plaque formed in one of the arteries leading to the brain travels through the bloodstream where it lodges in narrower arteries in the brain and blocks blood flow.
Treatment for patients with a blocked blood vessel in the brain that has caused an ischemic stroke consists of two basic methodologies to restore blood flow to the brain: intravenous (IV) lysis of the clot with administration of tissue plasminogen activator (tPA), the standard medical therapy, and more recently, endovascular removal of the clot with catheter based devices, or thrombectomy.
tPA is a clot dissolving drug that is administered to patients presenting with ischemic stroke symptoms within 3 hours, in most patients, and up to 4.5 hours in certain eligible patients. However, a large percentage of stroke patients do not arrive at a hospital in time for tPA treatment, and a significant percentage of patients treated with tPA do not immediately experience improved blood flow, or perfusion, to the brain. Such delays in perfusion can lead to prolonging or exacerbating the symptoms of ischemic stroke.
Currently, thrombectomy products are 510(k) cleared by the FDA for use within eight hours of stroke onset to restore blood flow in patients experiencing an ischemic stroke. Thrombectomy is performed by advancing a catheter to the site of the blocked blood vessel in the brain and then aspirating the clot into an aspiration catheter at the site of the clot, deploying a stent-like device and retracting it through the cerebral vasculature into a large catheter in the neck, or a combination of the two approaches. Ischemic stroke patients with large vessel occlusions and those with sufficient salvageable tissue are candidates for endovascular thrombectomy. According to a paper published in the journal Stroke, only 0.6% of stroke patients in the United States in 2009 underwent such a procedure.
In the past year, several major clinical trials have produced clinical data demonstrating the benefit of endovascular treatment of stroke patients over standard medical therapy. The MR CLEAN study, published in The New England Journal of Medicine in January 2015, demonstrated the benefit of endovascular thrombectomy plus standard medical therapy over standard medical therapy alone. The MR CLEAN study demonstrated the benefit of endovascular treatment across the range of clinical outcomes at 90 days, judged by the modified Rankin Scale (mRS), which is a commonly used scale measuring the degree of disability or dependence in the daily activities of people who have suffered a stroke or other causes of neurological disability. In the MR CLEAN study, where patients received endovascular treatment for stroke, there was a significant increase in the number achieving functional independence at 90 days, with 32.6% of the endovascular treatment group achieving a 0 to 2 on the mRS, which indicates no symptoms or only slight disability, compared to 19.1% of the control group. The results of this study have now been independently confirmed by several additional clinical trials: ESCAPE (2015), SWIFT PRIME (2015), EXTEND IA (2015), REVASCAT (2015), THRACE (2015) and our own THERAPY (2015) study. These studies support endovascular stroke treatment (as compared to standard medical
67
therapy) across a range of different products and patient selection criteria. With the exception of our own THERAPY study, we do not have access to the raw data underlying the published results of these trials, and so we cannot state with certainty whether any of our products was used in any of the trials.
With the recent clinical data supporting the benefits of endovascular thrombectomy, the Society of NeuroInterventional Surgery (SNIS) has stated endovascular thrombectomy is a new standard of care for stroke patients who qualify for the procedure. SNIS published the Report of its Standard and Guidelines Committee in the Journal of NeuroInterventional Surgery in May 2015, including the following specific language:
“For patients with anterior circulation stroke and documented E[mergent] L[arge] V[essel] O[cclusion] affecting the ICA or M1 segment of the MCA and a corresponding clinical deficit, the addition of endovascular embolectomy results in superior clinical outcomes compared with best medical therapy alone. Embolectomy needs to be performed as rapidly as possible for the greatest clinical benefit, and is best when performed within 6 h[ours] from onset of symptoms (AHA Class I, level of evidence A.).”
The ICA or M1 Segment of the MCA refers to the specific anatomical location of the occlusion in the vasculature.
In addition, on June 29, 2015, the American Heart Association/American Stroke Association (AHA/ASA) issued its updated guidance on endovascular treatment of acute ischemic stroke—the 2015 AHA/ASA Focused Update of the 2013 Guidelines for the Early Management of Patients with Acute Ischemic Stroke Regarding Endovascular Treatment. It was published in Stroke, the medical journal of the AHA/ASA. The AHA/ASA guidelines focus on the use of stent retrievers as the primary method of endovascular treatment of acute ischemic stroke but also allow for the use of other devices. Recommendation 10 specifically states: “Use of stent retrievers is indicated in preference to the MERCI device. The use of mechanical thrombectomy devices other than stent retrievers may be reasonable in some circumstances.”
Given the recent clinical data supporting endovascular treatment of ischemic stroke patients and the substantial clinical need and economic burden of ischemic stroke, we believe that the market for endovascular treatment of ischemic stroke is in its early stages of development and will continue to expand significantly over time.
Hemorrhagic Stroke
Hemorrhagic strokes are caused by a sudden rupture of a brain artery leading to bleeding into or around the brain. A cerebral aneurysm is usually located along the major arteries deep within brain structures. A major cause of hemorrhagic stroke is the rupture of a cerebral aneurysm. Aneurysms can vary in shape and size, typically grow over time and, due to pressure placed on the wall of the aneurysm, are prone to rupture. Ruptured aneurysms can cause death as a result of massive intracranial bleeding and loss of perfusion to the brain in the area affected by the aneurysm rupture. If an aneurysm ruptures, it leaks blood into the space around the brain. This is called a subarachnoid hemorrhage. If the hemorrhage bleeds into the brain itself, causing damage to the brain directly, this results in a hemorrhagic stroke. According to the AHA, once an aneurysm bleeds, there is an approximately 30% to 40% likelihood of death and an approximately 20% to 35% likelihood of moderate to severe brain damage, even if the aneurysm is successfully treated. If the aneurysm is not treated quickly, additional bleeding may occur from the already ruptured aneurysm.
The optimal treatment for an aneurysm depends upon many factors, including whether the aneurysm has ruptured or not. If an aneurysm has not ruptured, the optimal treatment will depend upon its size, location and shape and the patient’s symptoms. Small, unruptured aneurysms that are not creating any symptoms may not need treatment unless they grow, trigger symptoms, or rupture. A ruptured aneurysm usually requires immediate treatment because the re-bleeding rate remains quite high. However, the treatment time and options for treatment depend upon the size, location and shape of the aneurysm, as well as the patient’s overall medical condition.
Depending on an individual’s risk factors, surgical clipping of an aneurysm had been traditionally recommended. In this open surgical procedure, patients are placed under general anesthesia, an opening is
68
made in the skull, the brain tissue is spread apart, and the aneurysm is surgically exposed. The neurosurgeon then places a clip around the aneurysm’s base. The clip seals off the aneurysm so that blood cannot enter. Possible complications from surgical clipping include infection at the incision site, rupturing of the aneurysm during surgery, damage to the artery and bleeding into the brain which could result in brain damage. As with other open surgical procedures, there are risks associated with the use of anesthesia during the procedure.
The primary endovascular procedure for treating both unruptured and ruptured aneurysms uses a repair technique called embolization. The objective of embolization is to induce a blood clot, or thrombus, in the diseased vasculature to limit blood flow through the diseased vascular anatomy, thereby reducing blood pressure and flow to a ruptured area or the likelihood of rupture in an unruptured area.
The endovascular embolization of cerebral aneurysms usually involves the deployment of small platinum coils into the aneurysm. During such an embolic coiling procedure, the physician accesses the femoral artery to allow the introduction of a catheter which is inserted and guided towards the brain. A microcatheter is then introduced through the larger catheter and used to deliver coils through the neck and into the sac of the aneurysm. They are individually placed and often detached by a small electric current or mechanical detachment. This process is generally repeated until the aneurysm is filled with many coils, generally in progressively smaller sizes. Coiling may be done under local or general anesthesia. Possible complications include rupture of the aneurysm during treatment and damage to the artery and bleeding into the brain that could result in brain damage or death, as well as risks from the anesthesia.
Following the release of results from the International Subarachnoid Aneurysm Trial (ISAT) which were published in the journal The Lancet in 2002 and did not involve our products, the treatment of aneurysms shifted from open surgical techniques, such as surgical clipping, to minimally invasive, endovascular techniques, such as embolic coiling. The ISAT was a clinical trial involving 2,143 patients in Europe, North America and Australia that compared aneurysm clipping with embolic coiling as a method of treating cerebral aneurysms. The trial concluded that endovascular intervention with detachable platinum coils resulted in a 23% relative and 7% absolute reduction in the risk of major brain injury or death at one-year follow up compared with surgical clipping of the aneurysm.
According to the American Association of Neurological Surgeons, up to 6% of the general population in the United States may be living with an unruptured aneurysm. In addition, an estimated 30,000 people per year in the United States suffer from a ruptured cerebral aneurysm. According to the AHA, annually, 0.5% to 3.0% of people with a brain aneurysm may suffer from bleeding. If a patient has one aneurysm, there is a 15% to 20% likelihood that the patient has at least one or more additional aneurysms.
Peripheral Vascular Disease
Peripheral vascular disease is defined as vascular disease occurring in vessels outside of the brain or heart. Peripheral vascular disease is characterized by the narrowing or occlusion of blood vessels and can cause pain or loss of function and lead to amputation and death. Mortality from peripheral vascular disease can occur as a result of stroke, kidney failure or diabetes-related vascular complications.
According to a paper published in the journal The Lancet, the worldwide prevalence of peripheral artery disease was 202 million in 2010. Within the United States, the AHA estimates that approximately 8 million adults have peripheral artery disease. Patients with peripheral artery disease have a nearly threefold increase in the risk of suffering a heart attack or stroke. Over half of the population with peripheral artery disease does not display classic symptoms of the disease, leading to substantial underdiagnosis. Massachusetts General Hospital estimates that only approximately 25% of patients with peripheral artery disease in the United States undergo treatment for the disease.
More particularly, in Deep Vein Thrombosis, Dr. Camerota estimated that approximately 1 million people in the United States are diagnosed with deep vein thrombosis (DVT) each year. Approximately 300,000 people die of a venous thromboembolism complication, which exceeds that of acute myocardial infarction or acute stroke. In addition, according to The New England Journal of Medicine, acute limb ischemia, a sudden decrease in limb perfusion that threatens the viability of the limb, is found in approximately 1.5 out of 10,000 people per year.
69
Peripheral thrombectomy
When blood is unable to flow smoothly through a vessel, it can begin to coagulate, turning from a free-flowing liquid to a semisolid gel, a process known as blood clotting. A blood clot, or thrombus, that forms within a blood vessel may continue to grow, blocking off the blood supply to certain parts of the body and causing damage to tissues and organs. In some patients, blood clots come from other sources and dislodge in relatively small vessels, thereby causing a blockage or embolization. If left untreated, a vascular blockage can lead to limb ischemia, a potentially lethal clinical event. Limb ischemia is a sudden decrease in blood supply to the limb that threatens the viability of the limb. The rapid onset of this condition reduces or prevents the delivery of nutrients to the metabolically active tissues of the limb, including skin, muscle, and nerves. Symptoms of limb ischemia include pain, pulselessness, pale color, perishing cold feeling and paraesthetic feeling, such as burning or tingling, and paralysis.
Thrombotic occlusion is the most common cause of limb ischemia. Immediate surgical or endovascular removal of the clot is recommended in order to restore blood flow to the limb and prevent amputation. In these situations, thrombectomy and thrombolysis are often performed as a less invasive alternative to bypass surgery, where clot removal is imperative for the patient’s survival.
Minimally invasive endovascular procedures for the treatment of blood clots in the peripheral vasculature consist primarily of thrombolytic drug infusion, syringe aspiration or mechanical removal. In thrombolytic drug infusion, a catheter with holes allowing for drug infusion is inserted into the blocked or narrowed part of the artery over a previously positioned guidewire that directs the catheter to the affected area. A thrombolytic drug, such as tPA, is then infused at a set infusion rate for a period of 18 to 48 hours, during which time, these patients are closely monitored in an Intensive Care Unit (ICU) to ensure there are no complications associated with infusion of the drug.
Clot removal using syringe aspiration is accomplished through the use of a catheter delivered to the clot, and then a syringe is hooked to the end of the catheter. This syringe creates an initial vacuum, and hence can remove very short, soft segments of the clot in small vessels. As clot segments get longer, and more fibrous in nature, these syringe aspiration techniques are no longer effective, and a more robust mode of endovascular treatment is required. Some new techniques are evolving, where there is now the ability to maintain a continuous vacuum using a mechanical pump, and the addition of another wire-based tool along with the catheter to help keep the catheter option for the duration of the procedure. This is proving to be able to handle longer segments of the clot, as well as a clot that has been in place for a longer period of time. Other techniques involve the combination of the tPA drug along with a mechanical component, with the risk of bleeding, albeit lower, as well as added procedural cost.
Percutaneous thrombectomy devices are increasingly used to remove clots from peripheral and coronary vessels because they offer a faster treatment option than the traditional standard of care, systemic thrombolysis. The advantages of thrombectomy over thrombolytic drug administration alone can include rapid reperfusion of ischemic limbs, reduced procedural times, potentially shorter hospital stays, and lower recurrence of thrombosis.
With proper endovascular or surgical care, acute limb ischemia is a highly treatable condition; however, delayed treatment beyond six to 12 hours can result in permanent disability, amputation and death. Despite currently available treatments, according to The New England Journal of Medicine, it is estimated that acute limb ischemia has an associated one-year mortality rate of between 15% and 20% and an in-hospital amputation rate of between 10% and 15%.
Peripheral embolization
Aneurysms are another potential result of peripheral vascular disease. Peripheral aneurysms vary in size, shape and location. Peripheral vascular aneurysms occur in the renal arteries, the lower extremities and the visceral arteries, which are located in the abdomen and feed several important organs, such as the liver and spleen. Ruptured aneurysms can result in death as a result of massive bleeding and loss of perfusion to the organ that the vessel feeds.
Internal hemorrhaging can be difficult to control, and interventional radiologists will often perform an embolization to occlude the bleeding vessel. Leading causes of internal hemorrhaging where embolization is used
70
include trauma (generally major organ or gastrointestinal), pre- and post-surgical organ removal, hemoptysis (respiratory tract bleeding resulting in the excessive coughing up of blood), epistaxis (excessive and uncontrollable nosebleed), pseudoaneurysm hemorrhage and pulmonary AVM (arterial venous malformation) hemorrhage.
A peripheral aneurysm requires surgical or endovascular repair because of the risk of rupture. If the aneurysm is small and there are no symptoms, the physician will monitor its size to determine when repair is needed. Endovascular repair makes use of a catheter to place embolization coils that occlude the aneurysm and prevent blood flow to the area, thereby preventing further growth of the aneurysm. Open surgical repair of a peripheral aneurysm may be recommended if the aneurysm anatomy does not allow for endovascular repair. In this procedure, the damaged vasculature is removed and replaced with a graft or surgically ligated.
Minimally invasive endovascular procedures for the treatment of peripheral aneurysms or blood vessel sacrifice involve the placement of a catheter at the targeted location. The interventional radiologist then delivers metallic occlusion devices such as embolization coils or plugs to the target location to obstruct the aneurysm or vessel. Coils can be either pushable or detachable. Pushable coils are pushed out of the catheter without exact control of placement and delivery. Detachable coils offer precise physician control and can be carefully positioned until the physician is satisfied with the coil placement at the desired location. At this point, the physician uses a trigger mechanism, which can be mechanical or electrolytic, to detach the coil and release it into the target vessel.
Peripheral vascular embolization coils (PV coils) are used in a variety of clinical applications. Detachable PV coils are becoming more popular among interventional radiologists due to their clinical benefits relative to pushable coils, their ease of use and their predictability. Clinical applications involving the use of coils include: active extravasations; selective embolization in patients with visceral aneurysms; exclusion of branches prior to chemoembolization and radioembolization; embolization in patients with gastrointestinal bleeding; embolization of branches prior to stent graft procedures; procedures after stent grafting in patients with persistent type II endoleaks and sac enlargement; treatment of patients with varicocele and pelvic congestion syndrome; high flow AVM; post trans intrahepatic shunt placement; balloon retrograde transvenous obliteration; and exclusion of hepatic branches prior to liver resection.
Our Strengths
We believe the following strengths have enabled us to develop our broad and differentiated product portfolio and will continue to be significant factors in our success and growth. We plan to continue investing in our organization to preserve these strengths as we grow our business.
| • | Our culture built on cooperation, which we have institutionalized through our unique organizational structure. From our inception, we structured Penumbra in a unique way, designed to optimize the level of cooperation and communication between functional areas of the organization. This structure is highlighted by the application of distinctive methodologies to almost every important aspect of management, including research and development (R&D) project selection, budgeting, hiring and compensation. We believe our success in maintaining this structure over the past ten years demonstrates that our corporate structure and approach to culture and cooperation should be scalable as we continue to grow. We also seek to ensure maximum control of our development, manufacturing and commercialization processes, and accordingly, we currently conduct substantially all of our own product development, manufacture substantially all of our own products and, where cost effective, sell our own products through our direct sales organization. |
| • | Our highly efficient product innovation process. Our differentiated products have been a significant factor in our commercial success. We believe our ability to rapidly develop innovative products is in large part attributable to the fully integrated product innovation process that we have implemented and the management philosophy behind that process. From project initiation, we staff projects with a development team that includes our R&D engineers, personnel with manufacturing and quality control competencies, as well as employees focused on clinical, regulatory and marketing. This enables us to have a broad perspective on the design, manufacturing and clinical needs for the product, and ensures that members of the team share in the ownership of any given project. In addition, we rotate our project team leaders from our various functional areas to ensure cooperation and communication. We empower each project team to foster and encourage long-term innovative success by discouraging the fear of failure. |
71
| • | Our disciplined approach to product and commercial development. One of the benefits of our organizational structure and product innovation process is our ability to approach our development and commercialization in a disciplined manner. In our product development, this discipline includes incorporating the feedback of our cross-functional teams to optimize the materials selection or physician ease-of-use objectives early in the process, which enables us to accelerate the development process. Of equal importance, this also enables us to eliminate a project more expeditiously should it not meet the requirements for success defined by one of our functional teams. In our commercial development, we believe that we have applied the same discipline to our selection of target end markets and our identification of new sales and marketing talent. |
| • | Our deep understanding of our target end markets. We have assembled a team with in-depth knowledge of both of the target end markets in which we currently compete. We entered the neuro market in 2007, and we have a highly experienced team with extensive expertise and long tenure in this market. We believe our experience and expertise in this market has enabled us to effectively address specific clinical challenges with innovative products and technologies. In our peripheral vascular business, we have built a team with vast experience in peripheral vascular disease. We have also sought expertise and gained insight from many of the physician thought leaders in the interventional and vascular surgery community. The combination of internal expertise and access to external expertise has enabled us to engage in focused product development. |
| • | Our relationships with specialist physicians. Many of the specialist physicians who use our products have been responsible for developing clinical treatments in the neuro and peripheral vascular markets. Given our deep understanding of the clinical challenges in these markets, we have been able to develop very strong relationships with many of the leading specialist physicians. These physicians range from those in the specialist residency programs in endovascular treatment of stroke to influential thought leaders who are developing some of the most innovative research and techniques in the field. |
Our Growth Strategies
We believe the following strategies will continue to play a critical role in our future growth:
| • | Expanding the penetration of our products in our target end markets. In order to grow our business, we plan to continue to establish and strengthen our presence in our target end markets of neuro and peripheral vascular disease. We sell our products through our direct sales organization in the United States, most of Europe, Canada and Australia and through distributors in select international markets. We plan to expand the reach and penetration of our products by growing our direct sales organization and establishing new relationships with specialist physicians who have not used our products. We also plan to establish deeper relationships with hospital customers for whom we believe the cost effectiveness of our products is very attractive relative to alternatives. |
| • | Growing the acceptance of our innovative products as the standard of care in their targeted clinical applications. We develop innovative products to improve the clinical outcomes in our targeted applications in a cost effective manner. Improving a clinical outcome can have many implications, ranging from improving post-procedure reperfusion in ischemic stroke or limb ischemia to generating rapid occlusion in hemorrhagic stroke patients and reducing procedure times. We have evaluated some of our products in clinical studies to obtain U.S. regulatory clearance, and many of our products have been studied post-marketing in either Penumbra or third-party sponsored clinical trials. We believe that our strategy of continuing to invest in clinical studies for our products will help us differentiate our products for specialist physicians, hospital customers and patients. |
| • | Continuing to leverage our product development capabilities to drive efficient, rapid product development. We believe our highly efficient product development process has been a significant factor in our success to date. We plan to continue investing in product development, applying our integrated development approach and core technology expertise, to drive efficient, rapid product development in our target end markets. We expect to maintain our disciplined approach to product development in markets where we believe we can improve clinical outcomes in a cost-effective manner. |
72
| • | Scaling our culture of cooperative product development and commercial execution. We believe that our organizational structure has been an important differentiating factor because it has enabled us to innovate and commercialize our products in a capital efficient manner. As we have grown as an organization, we have been able to scale our business from development stage in 2004 to a company with approximately 1,000 employees currently focused on multiple product categories in two target end markets. We believe our experience in maintaining our unique structure while scaling our business will help us considerably as we grow our business going forward. |
73
Our Products
Since our founding in 2004, we have invested in expanding our product development and marketing capabilities. These investments have included engineering and materials science capabilities, pre-clinical and bench-testing infrastructure and in-house clinical and regulatory infrastructure. Our fully-integrated organization has enabled us to launch 14 product brands for access, thrombectomy and embolization since 2007 to service our two target end markets. Our current portfolio provides a range of products that are designed to enable specialist physicians to drive improved clinical outcomes in a cost effective manner. Numerous independent clinical studies have supported the cost-effectiveness of our products for hospital customers through comparative cost data.
The following table summarizes our product offerings in each of our target end markets:
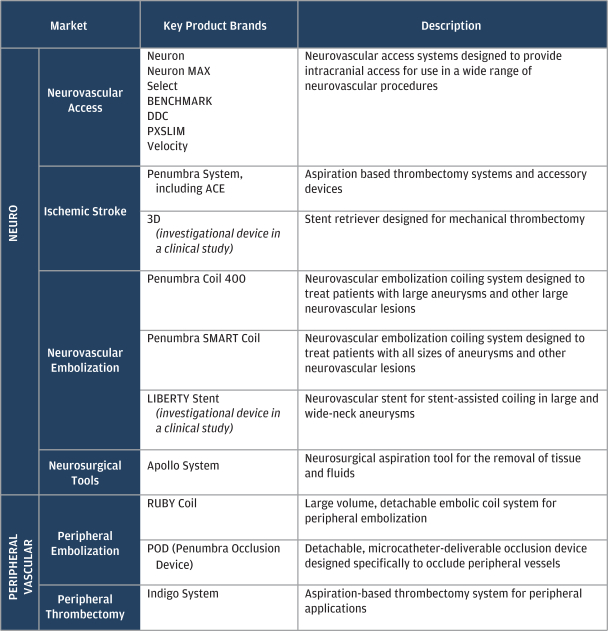
74
OUR NEURO PRODUCTS
Neurovascular Access
Accessing the brain through the tortuous neurovasculature has been a substantial challenge for physicians treating vascular disorders in the brain. Companies developing products for neurovascular applications have historically leveraged technologies developed for use in coronary or peripheral vascular interventions. This approach created challenges given the vastly different anatomy, structure and sizing of the neurovascular vessels.
Our portfolio of neurovascular access products includes our Neuron Access System catheters, BENCHMARK Intracranial Access System catheters and a variety of microcatheters.
Neuron Access System
We recognized the challenges posed by existing access technologies and focused our initial efforts on developing a guide catheter system designed specifically for neurointerventional procedures. Our Neuron delivery catheter is a variable stiffness guide catheter with increased support in the aortic arch, which enables trackability to access the intracranial vasculature. The design of Neuron enables physicians to position the catheter much higher in the anatomy than conventional guide catheters.
We believe the Neuron family of guide catheters and the Penumbra distal delivery catheters that we subsequently introduced have enabled many neuro-endovascular procedures that previously had not been possible in the tortuous anatomy of the neurovasculature. We have continued our development and currently offer a wide range of catheters that enable delivery of the different therapeutic catheters that are required for ischemic and hemorrhagic stroke interventions. Our Neuron products include the following:
| • | The Neuron Intracranial Access System is indicated for the introduction of interventional devices into the peripheral, coronary and neuro vasculature. The system is a two-catheter system comprised of the Neuron Delivery Catheter and the Select Catheter. |
| • | The Neuron Delivery Catheter is a variable stiffness, large lumen catheter that combines proximal arch support with a microcatheter-like distal segment that is designed to access the intracranial anatomy. The Neuron can be used individually with a 0.038 inch guidewire, or together with the Neuron Select Catheter, to access the desired location. |
| • | The Select Catheter is a single lumen, braid-reinforced, torquable catheter with a radiopaque distal end and a hub on the proximal end. The Select Catheter enables primary access with the Neuron Delivery Catheters, obviating the need for an extra guide catheter. |
| • | The Neuron MAX System is an additional configuration to the currently available Neuron Intracranial Access System. The Neuron MAX System is a long sheath catheter with a flexible distal tip for neurovascular use and provides a larger lumen to enable a wide range of device compatibility in neurovascular procedures. |
BENCHMARK Intracranial Access System
Advances in our catheter technology, driven largely by our advances in ischemic stroke therapy, have enabled us to further develop our intracranial access category of products. Our latest development in this category is the BENCHMARK catheter, which features additional improvements in ease-of-use, trackability, and aortic arch support that we believe will further enhance our position in the neurovascular access market.
The BENCHMARK catheter technology achieves these improvements by combining our advanced tracking technology with the original Neuron intracranial access concept. In addition to improved proximal support in the arch through multi-geometry metal reinforcement, the distal tip is softer and more trackable, while maintaining complete distal shaft radiopacity for improved visualization. The BENCHMARK also is available pre-packaged with a Select catheter to obviate the need for a neurovascular guide catheter exchange, which reduces the number of devices needed per procedure and shortens procedure times.
75
Ischemic Stroke
Penumbra System
We developed our aspiration-based Penumbra System family of products to enable specialist physicians to revascularize blood vessels that are blocked by clots in the intracranial vasculature. We launched our first Penumbra System product in 2008 in the United States. We believe ACE, launched in June 2013, and ACE 64, launched in May 2015, represent significant advancements over prior generations of the Penumbra System, enabling physicians to treat patients with ischemic stroke quickly, safely and cost effectively.
Overview of the Penumbra System
Our Penumbra System family of products is comprised of several principal components, which include:
| • | Penumbra Reperfusion Catheters are the cornerstone of the Penumbra System and are manufactured using a variety of proprietary processes and materials science innovations. We have launched five successive generations of Reperfusion Catheters since 2008. |
The latest generation of our Reperfusion Catheters, the ACE family of catheters, represents our most powerful and trackable Reperfusion Catheters launched to date. Its design enables specialist physicians to track these large bore aspiration catheters to the distal locations of occluded vessels. Once at the site of the occlusion, ACE provides significantly greater aspiration power than our prior Reperfusion Catheters, which we believe contributes to improved clinical outcomes and reduced procedure times.
ACE 64, our latest generation of ACE catheter, is designed to offer enhanced aspiration power relative to prior generations of the product, while maintaining trackability. ACE 64 launched in the United States in late May 2015.
| • | Penumbra Separators are a component of the earlier generations of the Penumbra System and enable a physician to remove an aspirated clot that has aggregated in the Reperfusion Catheter during the procedure. The Separators were an important component of our earlier Penumbra System due to the smaller diameter of our original Reperfusion Catheters, which resulted in frequent obstruction of the catheter. With the launch of our larger diameter ACE, Separators are less frequently used by physicians. |
| • | 3D is a stent retriever component of the Penumbra System that allows a physician to combine direct aspiration with stent retriever technology in a procedure commonly called “Solumbra.” 3D is currently being evaluated in a clinical study pursuant to an Investigational Device Exemption (IDE) to obtain 510(k) clearance. |
| • | Penumbra Aspiration Pumps are attached to our Reperfusion Catheters and provide the aspirating suction force. Our second generation MAX Aspiration Pump features increased aspiration capabilities and an improved, easier to use design. We have standardized the MAX Aspiration Pump to work with all generations of our Reperfusion Catheters. |
Evolution of Penumbra System’s Reperfusion Catheters
The Penumbra System Reperfusion Catheters are the foundation of the Penumbra System. The principal generations of our Reperfusion Catheters include the original Penumbra System, Penumbra System MAX, Penumbra System ACE and Penumbra System ACE 64. We have introduced five successive generations of these catheters since early 2008. Each subsequent generation of our Reperfusion Catheters has incorporated significant performance enhancements relative to prior generations with regard to trackability and aspiration power.
76
The Generations of the Penumbra System
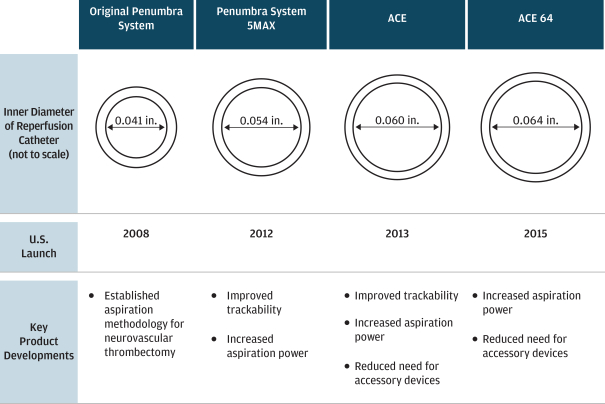
The Original Penumbra System
Our original Penumbra System was CE–marked in September 2006 and 510(k) cleared by the FDA in December 2007. The Penumbra System is intended for use in the revascularization of patients with ischemic stroke within eight hours of symptom onset.
Our original Penumbra System was evaluated in the Penumbra Pivotal study, a 125 patient clinical study to assess the safety and effectiveness of the Penumbra System in the revascularization of patients presenting with ischemic stroke. This study was sponsored by Penumbra to support and obtain regulatory clearance for the original Penumbra System. The Penumbra Pivotal study demonstrated an 81.6% success rate in achieving successful revascularization. The study was completed in 2007 and the results were published in the journal Stroke in 2009.
We had commissioned and subsequently evaluated the Penumbra System in our THERAPY study, a clinical study comparing the clinical outcomes in the medical management of stroke patients with IV recombinant tPA (rtPA) to stroke patients treated with a combination of IV rtPA and the Penumbra System. The THERAPY study was commenced in March 2012, and was designed to enroll up to 692 patients, but was stopped early in October 2014, because the positive results of the MR CLEAN study made it unethical to continue to treat the control group in the THERAPY study with medical management rather than with endovascular treatment. The MR CLEAN study demonstrated the superiority of endovascular treatment of stroke over medical management. As a result, the steering committee for THERAPY recommended stopping enrollment for the trial. The THERAPY study results, after the randomization of 108 patients, were presented in April 2015 at the European Stroke Organization Conference and the manuscript is being prepared for submission to a peer-reviewed journal. Despite the early termination of the study, the pre-specified per protocol analysis demonstrated a significant benefit of combined treatment with IV rtPA and the Penumbra System over IV rtPA alone.
77
Penumbra System MAX
The Penumbra System MAX applies our advanced tracking technology and improved aspiration power to the Penumbra System’s aspiration platform. Launched in 2011, the 3MAX and 4MAX Systems feature MAX Tracking Technology that allows access over a solo guidewire for an even faster, easier procedure than with our original Penumbra System. The proximal shaft of these specialized catheters incorporates tapering, larger diameters, enabling increased aspiration power. In August 2012, we launched the 5MAX, which added our MAX Tracking Technology to an even larger dimension Reperfusion Catheter.
ACE
Almost a decade of research and product development culminated in the introduction of our first ACE Reperfusion Catheter in July 2013. ACE features a unique tapered design, large lumen diameter and other developments that result in significantly greater aspiration power and improved trackability compared to our earlier original Penumbra System and Penumbra MAX products.
Given its improved aspiration power and larger lumen size, our ACE Reperfusion Catheter can enable the extraction of a fibrous thrombus without fragmentation and often in one solid piece. This leaves the longitudinal fibrin strands in the clot intact, allowing the thrombus to retain its integrity. We believe this is evidenced in our post-launch clinical experience, in which clinicians have seen high rates of TICI 3 revascularization, representing complete recanalization of the affected area, using our ACE catheters.
Our ACE 64 Reperfusion Catheter was launched in the United States in May 2015. It is built on our ACE platform and offers an increased lumen diameter, which leads to further increased aspiration power and which we believe will aid in the removal of clot from the neurovasculature.
ACE Reperfusion Catheter

78
Penumbra System Clinical Experience
There have been a number of clinical studies that have confirmed that mechanical thrombectomy results in generally better clinical outcomes than non-interventional treatments. Generations of our Penumbra System have been evaluated in several post-market clinical studies that demonstrate improved clinical outcomes and cost effectiveness. The following table summarizes certain key findings from certain clinical studies involving the Penumbra System and competitive technologies for treatment of ischemic stroke. While most of these studies were not designed to provide statistically significant comparisons of the various devices, these studies are highlighted as representative examples of the continuing evolution of mechanical thrombectomy in which clinical results are improving over time from original generation to subsequent generations across different products (including Penumbra products and competitor products). In these clinical studies, the Penumbra System demonstrated robust clinical performance.
| Product | MERCI(1) | Original Penumbra System |
Stent Retriever |
“Solumbra” | 5MAX | ACE | ||||||||||||||||||
| Author | Kass-Hout (JNIS 2014)(2) |
Humphries (JNIS 2014)(3) |
Turk (JNIS 2014)(4) |
|||||||||||||||||||||
| Number of patients |
81 | 91 | 115 | 105 | 44 | 44 | ||||||||||||||||||
| TICI 2b/3 (Successful revascularization (includes partial and complete revascularization)) |
70% | 78% | 86% | 88% | 96% | 98% | ||||||||||||||||||
| TICI 3 (Complete revascularization) |
14% | 9% | 37% | 44% | 41% | 61% | ||||||||||||||||||
| Puncture to revascularization (Time from groin puncture to successful revascularization) |
91 min | 75 min | 79 min | 57 min | 38 min | 36 min | ||||||||||||||||||
| sICH (Symptomatic intracranial hemorrhage) |
7% | 6% | 7% | 5% | 0% | 0% | ||||||||||||||||||
| mRS £ 2 at 90 days (Modified Rankin scale) |
25% | 41% | 36% | 44% | 34% | 50% | ||||||||||||||||||
| (1) | Merci is the marketing name for the “corkscrew” retriever device developed by Concentric Medical, Inc. |
| (2) | Kass-Hout T, Kass-Hout O, Sun C-H J, et al. (2014) Clinical, angiographic and radiographic outcome differences among mechanical thrombectomy devices: initial experience of a large-volume center. J NeuroIntervent Surg. Advance online publication doi:10.1136/neurintsurg-2013-011037. |
| (3) | Humphries W, Hoit D, Doss VT, et al. (2014) Distal aspiration with retrievable stent assisted thrombectomy for the treatment of ischemic stroke. J NeuroIntervent Surg. Advance online publication doi:10.1136/neurintsurg-2013-010986. |
| (4) | Turk AS, Frei D, Fiorella D, et al. (2014) ADAPT FAST study: a direct aspiration first pass technique for stroke thrombectomy. J NeuroIntervent Surg. Advance online publication doi:10.1136/neurintsurg-2014-011125. |
We believe the advancements offered by the Penumbra System, and in particular the ACE Reperfusion Catheter, have changed and will continue to change the way specialist physicians treat ischemic stroke. Some of the principal benefits include:
| • | Improved trackability: The ACE Reperfusion Catheter’s design features significant improvements in trackability, enabling greater ease-of-use in accessing the middle cerebral artery in a highly reproducible manner. |
| • | Reduced procedure times: We believe the improved trackability and the significantly greater aspiration power, which can be applied directly to the site of the clot, can enable clinicians using ACE to achieve revascularization faster than has been possible historically. Once our catheter has been navigated to the clot site, aspiration is typically rapid and the clot removed, often intact. The ADAPT FAST study demonstrated that ACE procedures averaged revascularization in 36 minutes from groin access. |
79
| • | Reduced need for general anesthesia: We believe ACE also may offer clinicians the ability to reduce the need for general anesthesia during the procedure. In some cases, ACE procedures can be performed without general anesthesia. It has been demonstrated that performing thrombectomy procedures without general anesthesia has been associated with better clinical outcomes for the patient. |
| • | Significant cost advantage: We designed ACE to offer significant economic advantages to our hospital customers relative to prior generations of the Penumbra System and our competitors’ offerings. These cost savings relative to competitive revascularization products are driven by lower total direct product costs as well as reduced procedure times. Based on current list prices, ACE procedures can be performed more cost effectively than procedures using stent retrievers based on device costs alone. |
3D
3D is a stent retriever that is designed to be used in conjunction with our Penumbra System. We believe that it will be useful in treating a small percentage of very difficult to treat ischemic clots that may not be removed with our catheters and aspiration pumps alone.
3D
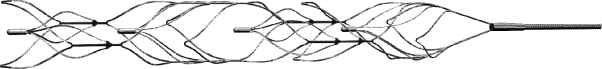
(side view of deployed 3D stent retriever)
We are currently evaluating 3D in a clinical trial pursuant to an Investigational Device Exemption (IDE) study to obtain 510(k) clearance. The 230 patient clinical study compares the safety and effectiveness of 3D as part of the Penumbra System with that of the standard Penumbra System alone. This study is actively enrolling patients. As of June 30, 2015, we have recruited 46 centers in the United States to participate in the trial and have enrolled 183 patients. We expect to complete enrollment of the study in 2016 and anticipate being in a position to file for 510(k) clearance by the end of 2016.
Neurovascular Embolization
Given the minimal product differentiation among the existing coils on the market, we concluded that to initially penetrate this market successfully we would have to develop a coil that was materially easier to deliver, and provided a procedural economic advantage. We also identified a segment of aneurysms that traditional neurovascular coils could not effectively treat on a cost effective basis. These included larger aneurysms and other larger, more complex lesions. We estimate that these aneurysms and lesions currently represent less than 10% of the addressable aneurysms.
The Penumbra Coil 400
We developed our Penumbra Coil 400 to offer an improved alternative for the treatment of larger aneurysms and other larger, more complex lesions. We implemented several proprietary design innovations to enable the coil to maintain shape while achieving biomechanically stable occlusion. Our coil system is composed of a platinum embolization coil complemented by a nitinol inner structure and stretch resistant nitinol wire. It is attached to a composite delivery pusher with a radiopaque positioning marker. The Penumbra Detachment Handle offers instant mechanical detachment of the coil and can be controlled by the physician in the sterile field.
80
We received 510(k) clearance for the Penumbra Coil 400 in 2011. The Penumbra Coil system is FDA cleared for endovascular embolization of intracranial aneurysms and other neurovascular abnormalities.
Penumbra Coil 400 Design Relative to Traditional Coils

Review of Penumbra Coil 400 Clinical Performance
Given the size and handling of the Penumbra Coil 400, it is able to achieve higher packing density with fewer coils compared to competitive coiling systems. These findings have been confirmed in numerous physician sponsored post-marketing studies. Collectively, the clinical studies have shown that use of the Penumbra Coil 400 resulted in:
| • | less retreatments or worsening occlusions; |
| • | larger aneurysm treatment capabilities; |
| • | higher packing density; and |
| • | fewer coils per aneurysm. |
Certain single center clinical studies evaluating the Penumbra Coil 400 have shown the clinical and economic benefit of using the large volume Penumbra Coil 400. Some of the principle conclusions include:
| • | In their 2014 study of 75 patients, Mascitelli, Patel, et al. reported their single-center experience with the Penumbra Coil 400 and compared it to their results with conventional coils. They concluded that the Penumbra Coil 400 is more efficient in the embolization of cerebral aneurysms, achieves greater packing density with fewer coils, and the related procedure takes less time to perform without compromising safety. |
| • | In their 2012 study of 58 patients, Milburn et al. reported their single-center experience with the Penumbra Coil 400 and compared it to their results with conventional coils. In consecutive cases, they reported a greater than 90% rate of stable occlusion on average follow-up time of 8.6 months. They evaluated and compared the number and total length of coils used per unit volume of aneurysm and experienced a 67% cost savings using the Penumbra Coil 400. |
Penumbra SMART Coil
Leveraging our initial experience treating larger aneurysms and more complex lesions with the Penumbra Coil 400, we turned our efforts to developing a standard sized coil to compete in the traditional, smaller neurovascular coil market. While the market has seen significant growth over the last 15 years, there has been
81
very little innovation in the last several years with regard to coil design, material science and performance. As a result, neurovascular coils built on the traditional, smaller-coil platform offer very little differentiation in terms of materials, ease-of-use and trackability.
In light of these dynamics, we focused our development efforts on a coil that would improve ease of delivery, or “feel” of the coil compared to the leading established coils. In order to accomplish this, our engineering team developed a highly sophisticated coil that dramatically changes its softness profile within the span of a single individual coil. This progressive softness feature enables physicians to pack the coil into a delicate lesion and mitigate catheter kick-back at the end of delivery, which can preclude the successful complete embolization of the lesion.
The Penumbra SMART Coil is designed to treat patients with a wide range of neurovascular lesions, including the small and medium sized aneurysms that comprise the majority of the neurovascular coiling market. Alternative products available to physicians in this market are offered in single levels of softness – standard, soft or extra soft.
The three principal levels of softness that competitors offer are derived from using smaller platinum filaments to increase the level of softness. However, this methodology does not allow for changes to the softness level within an individual coil. The design of the Penumbra SMART Coil allows the level of softness to be determined not only by the diameter of the platinum filament, but also by a structural component inside the coil itself. This development enables the Penumbra SMART Coil to become progressively softer within the span of an individual coil.
We anticipate launching our SMART Coil in the second half of 2015. We believe that it will provide us with another important opportunity to offer specialist physicians a broader suite of products to address their neurovascular coiling needs.
LIBERTY Stent
As the market for neurovascular coils grew over the past 15 years, the need arose to optimize coil performance using additional structural support devices. Neurovascular stents were initially developed as a means to complement these coil embolization procedures by enabling the treatment of wide-necked aneurysms that could not be treated with coils alone. These stents have historically had several shortcomings, which include lack of complete coverage of the neck of the aneurysm, challenging stent delivery through neurovascular tortuosity and limited vessel wall opposition.
To expand our presence in the market for the treatment of aneurysms, we began developing a next generation aneurysm stent that could overcome the limitations of existing stents. The objectives were to develop a stent that could:
| • | provide enhanced coverage of the neck of the aneurysm and increased flow diversion; |
| • | improve ease-of-use and stent delivery; and |
| • | provide improved opposition to the vessel wall through a self-expanding stent with excellent conformability. |
The LIBERTY stent is Penumbra’s product for stent-assisted coil embolization. The product incorporates a self-expanding nitinol stent technology developed from a laser-cut hypotube processed to create high coverage with many thin struts supported by a larger structure. This laser-cut nitinol design, complete with thin stent struts, address many of the limitations of other products in the market.
Unlike currently marketed flow-diverting stents, the LIBERTY stent allows the physician to penetrate the stent with a microcatheter and place coils for immediate protection from the possibility of aneurysm rupture, while establishing a scaffold for healing across the aneurysm neck.
82
LIBERTY Stent System
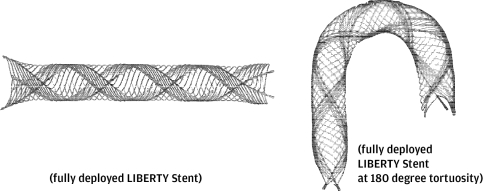
We are currently evaluating LIBERTY in a 120 patient international clinical study, the results of which, if positive, will be submitted to support our premarket approval application, or PMA. As of June 30, 2015, we had enrolled 113 patients in the study. We expect to complete enrollment of the study by the end of 2015 and anticipate being in a position to file for PMA approval by the FDA upon completion of the last patient’s 12-month follow-up.
Neurosurgical Tools
The Apollo System
We received 510(k) clearance from the FDA for our first neurosurgical product, the Apollo System, in 2014. The Apollo system leverages our expertise in thrombectomy and access to offer a minimally invasive approach to surgical removal of fluid and tissue from the ventricles in the brain.
The Apollo system is comprised of two primary components:
| • | the Apollo wand that is inserted into the brain through an endoscope, which, in turn, is inserted through a small burr hole into the skull; and |
| • | a reusable hardware device that delivers vacuum, irrigation and vibrational energy along the disposable wand to the site of the hemorrhage. |
83
| Apollo System | Apollo Wand | |
|
|
|
OUR PERIPHERAL VASCULAR PRODUCTS
After initially focusing our business on our neuro products, we identified the peripheral vascular market as an ideal opportunity to leverage our neuro experience and our core expertise in thrombectomy, embolization and access technologies to develop new products that could address significant clinical needs cost effectively.
The peripheral vasculature suffers from disorders that are very similar to those experienced in the neurovasculature that our products already successfully address. For example, weakening of the vascular walls can result in aneurysms, and blockages can form as the result of embolism or advanced atherosclerosis. Just as the disruption of blood flow to the brain has high mortality and morbidity, disruptions in the peripheral vasculature can also have serious adverse consequences.
The peripheral vasculature also presents unique challenges that do not apply to interventional efforts in the brain. Many peripheral arteries and veins are significantly larger than those found in the brain and therefore have higher blood flow rates. More importantly, they must be able to accommodate larger pressure gradients and sustain structural integrity despite substantial movement and flexing of the organs and musculature that surround them. Imaging can also be more challenging as physicians have to view their equipment through many more layers of organs and tissue than in the brain.
In 2012, we began investing further in research and development to evaluate and identify potential solutions to address significant clinical needs in the peripheral vasculature. Our products for the treatment of peripheral vascular disease focus on:
| • | peripheral vascular embolization; |
| • | vessel occlusion; and |
| • | peripheral vascular thrombectomy. |
Peripheral Embolization
Ruby Coil System
After completing research and development focusing on the specific requirements of the peripheral embolization market, we launched our Ruby Coil System for use in the peripheral vascular market in 2013. The
84
Ruby Coil System consists of detachable coils that are specifically designed for peripheral applications. The Ruby coils have a controlled mechanical detachment mechanism that permits the physician to deliver and reposition the coil until the final satisfactory position is reached before detachment. Compared to pushable coils, this minimizes costly complications like embolizing unintended vessels.
The Ruby Coil System is used in a variety of clinical applications, including:
| • | active extravasations, or the escape of blood into surrounding tissue; |
| • | selective embolization in patients with visceral aneurysms; |
| • | exclusion of branches prior to chemoembolization and radioembolization; |
| • | embolization in patients with gastrointestinal bleeding; |
| • | embolization of branches prior to stent graft procedures; |
| • | procedures after stent grafting in patients with persistent type II endoleaks and sac enlargement; |
| • | treatment of patients with varicocele and pelvic congestion syndrome; |
| • | high flow arterial venous malformations; |
| • | post trans intrahepatic shunt placement; |
| • | balloon retrograde transvenous obliteration; and |
| • | exclusion of hepatic branches prior to liver resection. |
We believe our Ruby Coil System offers specialist physicians a differentiated, cost-effective solution in the treatment of peripheral embolization patients.
POD (Penumbra Occlusion Device)
We developed POD, our peripheral vascular occlusion device, to address a specific need in the peripheral embolization market to rapidly and precisely occlude a target vessel. Current options for vessel occlusion in the periphery are limited, either requiring multiple devices or difficult to deliver vascular plugs. Microcatheter deliverable devices, such as coils, are not ideally suited for vessel embolization due to their tendency to migrate with antegrade flow and generally require the deployment of several devices to achieve occlusion. Vascular plug technology for larger peripheral vessels requires access with large diagnostic catheters or even larger bore sheaths. Additionally, these devices often require the placement of adjunctive devices, such as coils, to achieve complete occlusion. Our POD device utilizes technology that delivers both variable sizing and variable softness to provide a single device solution for rapid and precise embolization of the target vessel.
Unlike conventional vascular plugs, our POD technology enables the occlusive device to be delivered through a microcatheter. Additionally, a single POD can occlude a range of vessel diameters, reducing the need for sophisticated measurement prior to embolization.
Our POD technology leverages the key features of a dedicated vessel embolization device to improve ease-of-use. These include:
| • | microcatheter deliverability; |
| • | instant detachment; |
| • | immediate and precise anchoring; |
| • | a single device to treat a range of vessel diameters; and |
| • | dense occlusion in a short segment. |
The technology achieves this range of features through the design of a distal anchoring segment, thereby immediately anchoring the device in a range of vessel diameters. The proximal segment of the POD achieves
85
dense occlusion by packing a softer, smaller diameter segment tightly behind the anchored portion. Once POD is deployed, it can be detached instantly with the sterile detachment handle.
POD technology deployment

|

| |
| Step 1: Anchor | Step 2: Pack | |
Peripheral Thrombectomy
Indigo System
Our Indigo System, which we launched in 2014, was designed for continuous aspiration mechanical thrombectomy (CAT), leveraging the success of the Penumbra System in ischemic stroke. The Indigo System is designed to remove clots in the peripheral arteries, including in patients with limb ischemia.
Our Indigo System family of products and accessories is an easy to use thrombectomy system that is powerful, highly trackable, and suited to a wide range of clot morphology. The principal components include:
| • | Continuous Aspiration Mechanical Thrombectomy Catheters are the foundation of the system and are ideally suited to reach anatomy below the knee. Much like our MAX and ACE catheters, the CAT catheters are robust, durable, trackable and suited for the peripheral anatomy. The initial launch of the Indigo System included our CAT5 catheter and the device made for more distal access, CAT3, which is able to reach the distal peripheral vessels of the upper and lower extremities. On May 26, 2015, we received FDA clearance for two larger sizes of the Indigo System, as well as to market the Indigo System for use in the vessels of both the peripheral arterial and venous systems. |
| • | Indigo Separator enables the peripheral interventionalist to remove a difficult to aspirate clot from the CAT catheter. In the peripheral vessels, clots often form in long segments, and are more resistant to traditional aspiration techniques. The Indigo System with the Separator enables a wide range of clot morphology to be removed from the body. In clinical settings, we have demonstrated that the Indigo System with the Separator removes clots that were resistant to hours of revascularization attempts with other technologies and thrombolytic agents. |
| • | Penumbra Aspiration Pump is the power source that provides the aspirating suction force to remove waste, such as blood and clots. |
Our initial clinical experience with the Indigo System has demonstrated high rates of revascularization and reduced procedure times in a range of case applications including:
| • | revascularization of visceral vessels; |
| • | upper extremity revascularization in the brachial, radial, ulnar and digital arteries; |
| • | above and below the knee revascularization of arteries such as the superficial femoral, popliteal, peroneal, tibial and pedal arteries; and |
| • | rescue revascularization following failed revascularization or embolic showering during angioplasty/stenting, complete total occlusion (CTO) procedures, atherectomy, mechanical thrombectomy and thrombolytic infusion. |
86
Procedure times with current treatment options often range from several hours up to 24 to 72 hours when thrombolytic therapy is administered. Procedures using the Indigo Systems can generally achieve revascularization in times similar to the neuro experience with the Penumbra System in ischemic stroke.
We are working with leaders in the peripheral interventional field to collect clinical data in our PRISM study, which is a retrospective analysis of technical success using the Penumbra and Indigo Systems for mechanical thrombectomy in the periphery. Initial results of the PRISM study were presented at the 2015 Annual Scientific Meeting of the Society of Interventional Radiology (SIR). In these initial results based on 38 patients, 44.7% of patients were treated initially with Indigo and 44.7% of patients were treated following failed lytic therapy with the remaining patients treated following failed pharmaco-mechanical therapy. In the presented data, 97.3% of patients were successfully revascularized, and there were no device-related complications. The initial data illustrates the benefits of the Indigo System in achieving revascularization of these patients, many of whom had the Indigo System used in the first line therapy.
| CAT 5 & SEP 5, CAT 3 & SEP 3 | Indigo Pump | |
|
|
|
Research and Development
We direct our research efforts towards the development of clinical therapies that expand the therapeutic alternatives available to specialist physicians and improve upon our existing product offerings. Our research and development team has a track record of product innovation and significant product improvements. Since inception, we have introduced 14 products brands in either the United States, international markets, or both. Our research and development expenses totaled $15.6 million and $14.1 million for the years ended December 31, 2014 and 2013, respectively.
We believe our ability to rapidly develop innovative products is in large part attributable to the fully integrated product innovation process that we have implemented, and the management philosophy behind that process. In addition, we have recruited and retained engineers with both significant experience in the development of medical devices as well as engineers directly from undergraduate and graduate programs that have become immediately productive within our development process. We have a pipeline of products in various stages of development that are expected to provide additional commercial opportunities. All of our research and development efforts are based at our campus in Alameda, California.
Manufacturing
We currently maintain one manufacturing facility at our campus in Alameda, California, which, together with our research and development space, totals 180,000 square feet. The manufacturing facilities run two eight-hour shifts per weekday. In addition, we have capacity to increase production, and can expand adjacent to our current facilities. We currently produce substantially all of our products in house.
Our rigorous quality control management programs have earned us a number of quality-related manufacturing designations. Our manufacturing facilities are EN ISO 13485 compliant with ISO 13485-2003 certification achieved in 2005. In 2007, we achieved compliance with MDD standards, allowing our products to be
87
CE marked. We use annual internal audits, combined with external audits by regulatory agencies to help ensure strong quality control practices. An internal, on-going staff training and education program contributes to our quality assurance program; training is documented and considered part of the employee evaluation process.
Sales and Marketing
We have dedicated substantial resources to establish a direct sales capability in the United States, most of Europe, Canada and Australia, which we have complemented with distributors in Japan and certain other international markets. We have regulatory clearance to sell our neurovascular access, ischemic stroke, neurovascular embolization, peripheral embolization and peripheral thrombectomy products in two of our three major markets, the United States and Europe, except that neither DDC nor Apollo System has been cleared in Europe and 3D has been cleared in Europe but not the United States. In our third major market, Japan, we have regulatory clearance to sell our ischemic stroke, neurovascular embolization and peripheral embolization products. The only access product that has received regulatory clearance in Japan is PXSLIM. 3D, Penumbra SMART Coil, Ruby Coil, POD and Indigo System have not received regulatory clearance in Japan. Our Coil 400 products are also used for peripheral embolization in Japan, and have received regulatory clearance for that use in that market. Liberty Stent has not yet received regulatory clearance anywhere. We believe our global presence enables us to capitalize on the markets for neuro and peripheral vascular devices that exist outside of the United States.
We currently sell our products to hospitals in the United States through our dedicated salesforce in two target end markets, neuro and peripheral vascular. Our sales representatives and sales managers generally have substantial medical device experience and market our products directly to a variety of specialist physicians engaged in the treatment of neurovascular and peripheral vascular disorders, who are the end users of our products and significantly influence hospital buying decisions relating to medical devices. We are focused on developing strong relationships with specialist physicians and devote significant resources to training and educating physicians in the use and benefits of our products. The principal specialist physicians in our two target end markets include:
| • | Neuro: Interventional neuroradiologists, neurosurgeons and interventional neurologists. |
| • | Peripheral vascular: Interventional radiologists and vascular surgeons. |
In addition to our direct sales organizations, we work with distributors in certain geographies where we have determined that selling through distributors is likely to be more effective. The largest market where we sell our products through a distributor, Medico’s Hirata Inc., is Japan.
Our direct sales have been, and we anticipate will continue to represent, a majority of our revenues. In 2014, direct sales accounted for approximately 82.3% of our revenue, with the balance generated by independent distributors that sell our products outside of the United States.
Backlog
We typically accept and ship orders on the day purchase orders are received or the next business day. Furthermore, if requested, we generally permit customers to cancel or reschedule without penalty. As a result, we do not believe that our backlog at any particular time is material, nor is it a reliable indication of future revenue.
Reimbursement
In the United States, hospitals are the purchasers of our products. Hospitals in turn bill various third-party payors, such as Medicare, Medicaid and private health insurance plans, for the total healthcare services required to treat the patient. Government agencies, private insurers and other payors determine whether to provide coverage for a particular procedure and to reimburse hospitals for inpatient treatment at a fixed rate based on the diagnosis-related group (DRG) as determined by the U.S. Centers for Medicare and Medicaid Services (CMS). The fixed rate of reimbursement is based on the procedure performed, and is unrelated to the specific medical device used in that procedure. Medicare rates for the same or similar procedures vary due to geographic location, nature of facility in which the procedure is performed (i.e., teaching or community
88
hospital) and other factors. While private payors vary in their coverage and payment policies, most look to coverage and payment by Medicare as a benchmark by which to make their own decisions.
Some payors may deny reimbursement if they determine that the device used in a treatment was unnecessary, not cost-effective, or used for a non-approved indication. We cannot assure you that government or private third-party payors will cover and reimburse the procedures using our products in whole or in part in the future or that payment rates will be adequate.
Outside the United States, market acceptance of medical devices depends partly upon the availability of reimbursement within the prevailing healthcare payment system. Reimbursement levels vary significantly by country, and by region within some countries. Reimbursement is obtained from a variety of sources, including government-sponsored and private health insurance plans, and combinations of both. A small number of countries may require us to gather additional clinical data before recognizing coverage and reimbursement for our products. It is our intent to complete the requisite clinical studies and obtain coverage and reimbursement approval in countries where it makes economic sense to do so.
The increased emphasis on managed healthcare in the United States and on country and regional pricing and reimbursement controls in international markets will put additional pressure on product pricing, reimbursement and usage, which may adversely affect our product sales and results of operations. These pressures can arise from rules and practices of insurers and managed care organizations, judicial decisions and governmental laws and regulations related to Medicare, Medicaid and healthcare reform, medical device reimbursement policies and pricing in general. Our ability to achieve market acceptance or significant sales volume will depend in large part on the availability of coverage and the level of reimbursement for procedures performed using our products under healthcare payment systems in such markets.
All third-party reimbursement programs, whether government funded or insured commercially, whether in the United States or internationally, are developing increasingly sophisticated methods of controlling health care costs through prospective reimbursement and capitation programs, group purchasing, redesign of benefits, second opinions required prior to major surgery, careful review of bills, encouragement of healthier lifestyles and exploration of more cost-effective methods of delivering health care. These types of programs and legislative or regulatory changes to reimbursement policies could potentially limit the amount which healthcare providers may be willing to pay for medical devices.
Competition
The medical device industry is intensely competitive, subject to rapid change and significantly affected by new product introductions and other market activities of industry participants. We compete with a number of manufacturers and distributors of neurovascular and peripheral vascular medical devices. Our most notable competitors are Boston Scientific, Johnson & Johnson, Medtronic, Stryker and Terumo. All of these competitors are large, well-capitalized companies with significantly more market share and resources than we have. As a consequence, they are able to spend more on product development, marketing, sales and other product initiatives than we can. We also compete with a number of smaller medical device companies that have single products or a limited range of products. Some of our competitors have:
| • | significantly greater name recognition; |
| • | broader or deeper relations with healthcare professionals, customers and third-party payors; |
| • | more established distribution networks; |
| • | additional lines of products and the ability to offer rebates or bundle products to offer greater discounts or other incentives to gain a competitive advantage; |
| • | greater experience in conducting research and development, manufacturing, clinical trials, marketing and obtaining regulatory clearance or approval for products; and |
| • | greater financial and human resources for product development, sales and marketing and patent litigation. |
89
We compete primarily on the basis that our products are able to treat patients with neurovascular and peripheral vascular diseases and disorders safely and effectively, with improved outcomes and procedural cost savings. Our continued success depends on our ability to:
| • | develop innovative, proprietary products that can cost-effectively address significant clinical needs; |
| • | continue to innovate and develop scientifically advanced technology; |
| • | obtain and maintain regulatory clearances or approvals; |
| • | demonstrate efficacy in Penumbra-sponsored and third-party clinical trials and studies; |
| • | apply technology across product lines and markets; |
| • | attract and retain skilled research and development and sales personnel; and |
| • | cost-effectively manufacture and successfully market and sell products. |
Intellectual Property
Our success depends in part on our ability to protect our proprietary technology and intellectual property and operate without infringing the patents and other proprietary rights of third parties. We rely on a combination of patent, trademark, trade secret, copyright and other intellectual property rights and measures to protect our intellectual property rights that we consider important to our business. We also rely on know-how and continuing technological innovation to develop and maintain our competitive position. We do not have any material licenses to any technology or intellectual property rights.
As of June 30, 2015, we owned 18 issued patents globally, of which seven were U.S. patents. As of June 30, 2015, we owned 31 pending patent applications, of which 14 were patent applications pending in the United States. Subject to payment of required maintenance fees, annuities and other charges, eight of our issued patents are currently expected to expire between 2024 and 2025; five of these patents relate to components of the Penumbra System and the Indigo System and one of these patents relates to methods performed by the Apollo System. An additional four of our issued patents, which relate to components of devices that have not been commercialized, are expected to expire between 2026 and 2027. The remaining six of our issued patents, which relate to the components of the Penumbra Coil and Ruby Coil, are currently expected to expire after 2027. Our issued patents relate to the following main areas: mechanical thrombectomy, coil embolization, treatment of aneurysm and treatment of intracranial hemorrhage. Our pending patent applications relate primarily to the following five main areas: mechanical thrombectomy, coil embolization, coronary atherectomy, blood filtration and treatment of patients with intracranial hemorrhage. Some of our pending patent applications pertain to components and methods of use associated with currently commercialized products. Our pending patent applications may not result in issued patents and we can give no assurance that any patents that have issued or might issue in the future will protect our current or future products or provide us with any competitive advantage. See the section titled “Risk Factors—Risks Related to Our Intellectual Property” for additional information.
Additionally, we own or have rights to trademarks or trade names that are used in our business and in conjunction with the sale of our products, including eight U.S. trademark registrations and six foreign trademark registrations as of June 30, 2015. Included in the registered trademarks is a mark with our company name and logo.
We also seek to protect our proprietary rights through a variety of methods, including confidentiality agreements and proprietary information agreements with suppliers, employees, consultants and others who may have access to our proprietary information.
Government Regulation
United States
Our products are medical devices subject to extensive and ongoing regulation by the FDA under the Federal Food, Drug, and Cosmetic Act (FD&C Act), and its implementing regulations, as well as other federal and
90
state regulatory bodies in the United States and comparable authorities in other countries under other statutes and regulations. The laws and regulations govern, among other things, product design and development, pre- clinical and clinical testing, manufacturing, packaging, labeling, storage, recordkeeping and reporting, clearance or approval, marketing, distribution, promotion, import and export, and post-marketing surveillance. Failure to comply with applicable requirements may subject a device and/or its manufacturer to a variety of administrative sanctions, such as issuance of Warning letters, import detentions, civil monetary penalties, and/or judicial sanctions, such as product seizures, injunctions and criminal prosecution.
FDA’s Premarket Clearance and Approval Requirements
Each medical device we seek to commercially distribute in the United States will require either a prior 510(k) clearance, unless it is exempt, or a premarket approval from the FDA. Medical devices are classified into one of three classes—Class I, Class II or Class III—depending on the degree of risk associated with each medical device and the extent of control needed to provide reasonable assurance of safety and effectiveness. Class I devices are deemed to be low risk and are subject to the general controls of the FD&C Act, such as provisions that relate to adulteration; misbranding; registration and listing; notification, including repair, replacement, or refund; records and reports; and good manufacturing practices. Most Class I devices are classified as exempt from premarket notification under section 510(k) of the FD&C Act, and therefore may be commercially distributed without obtaining 510(k) clearance from the FDA., Class II devices are subject to both general controls and special controls to provide reasonable assurance of safety and effectiveness. Special controls include performance standards, postmarket surveillance, patient registries, and guidance documents. A manufacturer may be required to submit to the FDA a premarket notification requesting permission to commercially distribute some Class II devices. Devices deemed by the FDA to pose the greatest risk, such as life-sustaining, life-supporting or implantable devices, or devices deemed not substantially equivalent to a previously cleared 510(k) device, are placed in Class III. A Class III device cannot be marketed in the United States unless the FDA approves the device after submission of a premarket approval application, or PMA. However, there are some Class III devices for which FDA has not yet called for a PMA. For these devices, the manufacturer must submit a premarket notification and obtain 510(k) clearance in orders to commercially distribute these devices. The FDA can also impose sales, marketing or other restrictions on devices in order to assure that they are used in a safe and effective manner.
510(k) Clearance Pathway
When a 510(k) clearance is required, we must submit a premarket notification to the FDA demonstrating that our proposed device is substantially equivalent to a predicate device, which is a previously cleared and legally marketed 510(k) device or a device that was in commercial distribution before May 28, 1976. By regulation, a premarket notification must be submitted to the FDA at least 90 days before we intend to distribute a device. As a practical matter, clearance often takes significantly longer. To demonstrate substantial equivalence, the manufacturer must show that the proposed device has the same intended use as the predicate device, and it either has the same technological characteristics, or different technological characteristics and the information in the premarket notification demonstrates that the device is equally safe and effective and does not raise different questions of safety and effectiveness. The FDA may require further information, including clinical data, to make a determination regarding substantial equivalence. If the FDA determines that the device, or its intended use, is not substantially equivalent to a previously cleared device or use, the FDA will place the device into Class III.
There are three types of 510(k)s: traditional, special and abbreviated. Special 510(k)s are for devices that are modified and the modification needs a new 510(k) but does not affect the intended use or alter the fundamental scientific technology of the device. Abbreviated 510(k)s are for devices that conform to a recognized standard. The special and abbreviated 510(k)s are intended to streamline review, and the FDA intends to process special 510(k)s within 30 days of receipt.
Premarket Approval Pathway
A premarket approval application must be submitted to the FDA for Class III devices for which the FDA has required a PMA. The premarket approval application process is much more demanding than the 510(k)
91
premarket notification process. A premarket approval application must be supported by extensive data, including but not limited to technical, preclinical, clinical trials, manufacturing and labeling to demonstrate to the FDA’s satisfaction reasonable evidence of safety and effectiveness of the device.
After a premarket approval application is submitted, the FDA has 45 days to determine whether the application is sufficiently complete to permit a substantive review and thus whether the FDA will file the application for review. The FDA has 180 days to review a filed premarket approval application, although the review of an application generally occurs over a significantly longer period of time and can take up to several years. During this review period, the FDA may request additional information or clarification of the information already provided. Also, an advisory panel of experts from outside the FDA may be convened to review and evaluate the application and provide recommendations to the FDA as to the approvability of the device. Although the FDA is not bound by the advisory panel decision, the panel’s recommendations are important to the FDA’s overall decision making process. In addition, the FDA may conduct a preapproval inspection of the manufacturing facility to ensure compliance with the Quality System Regulation or QSR. The agency also may inspect one or more clinical sites to assure compliance with FDA’s regulations.
Upon completion of the PMA review, the FDA may: (i) approve the PMA which authorizes commercial marketing with specific prescribing information for one or more indications, which can be more limited than those originally sought; (ii) issue an approvable letter which indicates the FDA’s belief that the PMA is approvable and states what additional information the FDA requires, or the post-approval commitments that must be agreed to prior to approval; (iii) issue a not approvable letter which outlines steps required for approval, but which are typically more onerous than those in an approvable letter, and may require additional clinical trials that are often expensive and time consuming and can delay approval for months or even years; or (iv) deny the application. If the FDA issues an approvable or not approvable letter, the applicant has 180 days to respond, after which the FDA’s review clock is reset.
Clinical Trials
Clinical trials are almost always required to support premarket approval and are sometimes required for 510(k) clearance. In the United States, for significant risk devices, these trials require submission of an application for an Investigational Device Exemption, or IDE, to the FDA. The IDE application must be supported by appropriate data, such as animal and laboratory testing results, showing it is safe to test the device in humans and that the testing protocol is scientifically sound. The IDE must be approved in advance by the FDA for a specific number of patients at specified study sites. During the trial, the sponsor must comply with the FDA’s IDE requirements for investigator selection, trial monitoring, reporting, and recordkeeping. The investigators must obtain patient informed consent, rigorously follow the investigational plan and study protocol, control the disposition of investigational devices, and comply with all reporting and recordkeeping requirements. Clinical trials for significant risk devices may not begin until the IDE application is approved by the FDA and the appropriate institutional review boards, or IRBs, at the clinical trial sites. An IRB is an appropriately constituted group that has been formally designated to review and monitor medical research involving subjects and which has the authority to approve, require modifications in, or disapprove research to protect the rights, safety and welfare of human research subjects. A nonsignificant risk device does not require FDA approval of an IDE; however, the clinical trial must still be conducted in compliance with various requirements of FDA’s IDE regulations and be approved by an IRB at the clinical trials sites. We, the FDA or the IRB at each site at which a clinical trial is being performed may withdraw approval of a clinical trial at any time for various reasons, including a belief that the risks to study subjects outweigh the benefits or a failure to comply with FDA or IRB requirements. Even if a trial is completed, the results of clinical testing may not demonstrate the safety and effectiveness of the device, may be equivocal or may otherwise not be sufficient to obtain approval or clearance of the product.
Sponsors of clinical trials of devices are required to register with clinicaltrials.gov, a public database of clinical trial information. Information related to the device, patient population, phase of investigation, study sites and investigators, and other aspects of the clinical trial is made public as part of the registration.
92
Ongoing Regulation by the FDA
Even after a device receives clearance or approval and is placed on the market, numerous regulatory requirements apply. These include:
| • | establishment registration and device listing; |
| • | the QSR, which requires manufacturers, including third-party manufacturers, to follow stringent design, testing, control, documentation, and other quality assurance procedures during all aspects of the manufacturing process; |
| • | labeling regulations and the FDA prohibitions against the promotion of products for un-cleared, unapproved or “off-label” uses, and other requirements related to promotional activities; |
| • | medical device reporting regulations, which require that manufactures report to the FDA if their device may have caused or contributed to a death or serious injury or if their device malfunctioned and the device or a similar device marketed by the manufacturer would be likely to cause or contribute to a death or serious injury if the malfunction were to recur; |
| • | corrections and removal reporting regulations, which require that manufactures report to the FDA field corrections or removals if undertaken to reduce a risk to health posed by a device or to remedy a violation of the Federal Food, Drug, and Cosmetic Act that may present a risk to health; and |
| • | post market surveillance regulations, which apply to certain class II or III devices when necessary to protect the public health or to provide additional safety and effectiveness data for the device. |
After a device receives 510(k) clearance, any modification that could significantly affect its safety or effectiveness, or that would constitute a major change in its intended use, will require a new clearance or possibly a premarket approval. The FDA requires each manufacturer to make this determination initially, but the FDA can review any such decision and can disagree with a manufacturer’s determination. If the FDA disagrees with our determination not to seek a new 510(k) clearance, the FDA may retroactively require us to seek 510(k) clearance or possibly a premarket approval. The FDA could also require us to cease marketing and distribution and/or recall the modified device until 510(k) clearance or premarket approval is obtained. Also, in these circumstances, we may be subject to significant regulatory fines and penalties.
Some changes to an approved PMA device, including changes in indications, labeling, or manufacturing processes or facilities, require submission and FDA approval of a new PMA or PMA supplement, as appropriate, before the change can be implemented. Supplements to a PMA often require the submission of the same type of information required for an original PMA, except that the supplement is generally limited to that information needed to support the proposed change from the device covered by the original PMA. The FDA uses the same procedures and actions in reviewing PMA supplements as it does in reviewing original PMAs.
FDA regulations require us to register as a medical device manufacturer with the FDA. Additionally, the California Department of Health Services, or CDHS, requires us to register as a medical device manufacturer within the state. Because of this, the FDA and the CDHS inspect us on a routine basis for compliance with the QSR. These regulations require that we manufacture our products and maintain related documentation in a prescribed manner with respect to manufacturing, testing and control activities. We have undergone and expect to continue to undergo regular QSR inspections in connection with the manufacture of our products at our facilities. Further, the FDA requires us to comply with various FDA regulations regarding labeling. Failure by us or by our suppliers to comply with applicable regulatory requirements can result in enforcement action by the FDA or state authorities, which may include any of the following sanctions:
| • | warning or untitled letters, fines, injunctions, consent decrees and civil penalties; |
| • | customer notifications, voluntary or mandatory recall or seizure of our products; |
| • | operating restrictions, partial suspension or total shutdown of production; |
| • | delay in processing submissions or applications for new products or modifications to existing products; |
93
| • | withdrawing approvals that have already been granted; and |
| • | criminal prosecution. |
The Medical Device Reporting laws and regulations require us to provide information to the FDA when we receive or otherwise become aware of information that reasonably suggests our device may have caused or contributed to a death or serious injury as well as a device malfunction that likely would cause or contribute to death or serious injury if the malfunction were to recur. In addition, the FDA prohibits an approved device from being marketed for off-label use. The FDA and other agencies actively enforce the laws and regulations prohibiting the promotion of off-label uses, and a company that is found to have improperly promoted off-label uses may be subject to significant liability, including substantial monetary penalties and criminal prosecution.
Newly discovered or developed safety or effectiveness data may require changes to a product’s labeling, including the addition of new warnings and contraindications, and also may require the implementation of other risk management measures. Also, new government requirements, including those resulting from new legislation, may be established, or the FDA’s policies may change, which could delay or prevent regulatory clearance or approval of our products under development.
We are also subject to other federal, state and local laws, and regulations relating to safe working conditions, laboratory, and manufacturing practices.
Regulatory Inspections
We are subject to periodic inspections by the FDA and other regulatory bodies related to the regulatory requirements that apply to medical devices designed and manufactured, and clinical trials sponsored, by us. When the FDA conducts an inspection, the inspectors will identify any deficiencies they believe exist in the form of a notice of inspectional observations, or Form FDA 483. If we receive a notice of inspectional observations or deficiencies from the FDA following an inspection, we likely will be required to respond in writing, and may be required to undertake corrective and preventive actions or other actions in order to address the FDA’s concerns. Failure to address the FDA’s concerns may result in the issuance of a warning letter or other enforcement or administrative actions.
From June 24, 2015 to July 15, 2015 the FDA conducted an inspection of our records relating to certain investigational sites for two different clinical trials and from July 30, 2015 to August 4, 2015, the FDA conducted an inspection of our Quality System. At the conclusion of the first inspection, a Form FDA 483 was issued with one observation. The 483 observation pertained to the failure to ensure proper monitoring at five of the investigational sites reviewed. Specifically, the observation noted that protocols relating to performing onsite monitoring visits at appropriate intervals and providing documentation to clearly address any repeated data problems and resolutions of noted deficiencies in written reports after each onsite monitoring visit were not properly followed. At the conclusion of the second inspection, a Form FDA 483 was also issued with one observation relating to our procedures for Corrective and Preventative Action (CAPA). Specifically, the observation noted our CAPA procedures do not require an effectiveness check in all cases.
We responded to the first 483 on August 4, 2015, and we have begun to take preventive actions to address the observation in that 483. We continue to review and enhance our investigational site monitoring to ensure compliance with regulatory requirements. We have opened a CAPA to address the observation in the second 483 and expect to respond to the second 483 later in August 2015. However, the FDA may conclude in subsequent inspections that we have not adequately responded to its observations, and could take action against us without further notice. Action by the FDA against us could result in monetary fines or require us to take further corrective actions, which could be expensive and time-consuming to complete and could impose additional burdens and expenses, and could even require us to discontinue our investigational studies.
European Union
Our products are regulated in the European Union as medical devices per the European Union Directive (93/42/EEC), also known as the Medical Device Directive. An authorized third party, Notified Body, must
94
approve products for CE marking. The CE mark is contingent upon continued compliance to the applicable regulations and the quality system requirements of the ISO 13485 standard.
Other Regions
Most major markets have different levels of regulatory requirements for medical devices. Modifications to the cleared or approved products may require a new regulatory submission in all major markets. The regulatory requirements, and the review time, vary significantly from country to country. Products can also be marketed in other countries that have minimal requirements for medical devices.
Fraud and Abuse and Other Healthcare Regulation
Anti-Kickback Statute
We are subject to various federal and state healthcare laws, including, but not limited to, anti-kickback laws. In particular, the federal Anti-Kickback Statute prohibits persons or entities from knowingly and willfully soliciting, offering, receiving or paying any remuneration, directly or indirectly, overtly or covertly, in cash or in kind, in exchange for or to induce either the referral of an individual for the furnishing or arranging for a good or service, or for the purchasing, leasing, ordering, or arranging for or recommending any good, facility, service or item for which payment may be made in whole or in part under federal healthcare programs, such as the Medicare and Medicaid programs. The federal Anti-Kickback Statute is broad and prohibits many arrangements and practices that are lawful in businesses outside of the healthcare industry. The term “remuneration” expressly includes kickbacks, bribes, or rebates and also has been broadly interpreted to include anything of value, including, for example, gifts, discounts, the furnishing of supplies or equipment, credit arrangements, payments of cash, waivers of payments, ownership interests and providing anything at less than its fair market value.
There are a number of statutory exceptions and regulatory safe harbors protecting certain business arrangements from prosecution under the federal Anti-Kickback Statute. These statutory exceptions and safe harbors set forth provisions that, if all their applicable requirements are met, will assure healthcare providers and other parties that they may not be prosecuted under the federal Anti-Kickback Statute. The failure of a transaction or arrangement to fit precisely within one or more applicable statutory exceptions or safe harbors does not necessarily mean that it is illegal or that prosecution will be pursued. However, conduct and business arrangements that do not fully satisfy all requirements of an applicable safe harbor may result in increased scrutiny by government enforcement authorities and will be evaluated on a case-by-case basis based on a cumulative review of all of its facts and circumstances. Additionally, the intent standard under the federal Anti-Kickback Statute was amended under the Affordable Care Act, to a stricter standard such that a person or entity no longer needs to have actual knowledge of the statute or specific intent to violate it in order to have committed a violation. The Affordable Care Act provides that the government may assert that a claim including items or services resulting from a violation of the federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the federal civil False Claims Act which is discussed below. Penalties for violations of the anti-kickback statute include, but are not limited to, criminal, civil and/or administrative penalties, damages, fines, disgorgement, individual imprisonment, possible exclusion from Medicare, Medicaid and other federal healthcare programs, and the curtailment or restructuring of operations. Various states have adopted laws similar to the federal Anti-Kickback Statute, and some of these state laws may be broader in scope in that some of these state laws extend to all payors and may not contain safe harbors.
Federal Civil False Claims Act. The federal civil False Claims Act prohibits, among other things, persons or entities from knowingly presenting or causing to be presented a false or fraudulent claim to, or the knowing use of false statements to obtain payment from or approval by, the federal government. Suits filed under the federal civil False Claims Act, known as “qui tam” actions, can be brought by any individual on behalf of the government. These individuals, sometimes known as “relators” or, more commonly, as “whistleblowers,” may share in any amounts paid by the entity to the government in fines or settlement. The number of filings of qui tam actions has increased significantly in recent years, causing more healthcare companies to have to defend a
95
case brought under the federal civil False Claim Act. If an entity is determined to have violated the federal civil False Claims Act, it may be required to pay up to three times the actual damages sustained by the government, plus civil penalties for each separate false claim. Various states have adopted laws similar to the federal civil False Claims Act, and many of these state laws are broader in scope and apply to all payors, and therefore, are not limited to only those claims submitted to the federal government.
Federal Civil Monetary Penalties Statute. The federal Civil Monetary Penalties Statute, among other things, imposes fines against any person who is determined to have presented, or caused to be presented, claims to a federal healthcare program that the person knows, or should know, is for an item or service that was not provided as claimed or is false or fraudulent.
Sunshine Act. The Affordable Care Act also included a provision, commonly referred to as the Sunshine Act. This provision requires that any manufacturer of a covered device that provides payment or other transfer of value to a physician or teaching hospital, or to a third party at the request of a physician or teaching hospital, must submit to the Centers for Medicare and Medicaid Services information about the payment or other transfer of value annually, with the reported information to be made public on a searchable website.
Health Insurance Portability and Accountability Act of 1996. The federal Health Insurance Portability and Accountability Act (HIPAA) created several new federal crimes, including healthcare fraud and false statements relating to healthcare matters. The healthcare fraud statute prohibits knowingly and willfully executing a scheme to defraud any healthcare benefit program, including private third-party payors. The false statements statute prohibits knowingly and willfully falsifying, concealing or covering up a material fact or making any materially false, fictitious or fraudulent statement in connection with the delivery of or payment for healthcare benefits, items or services. In addition, HIPAA and its implementing regulations established uniform standards for certain covered entities, which are healthcare providers, health plans and healthcare clearinghouses, as well as their business associates, governing the conduct of specified electronic healthcare transactions and protecting the security and privacy of protected health information.
The American Recovery and Reinvestment Act of 2009, commonly referred to as the economic stimulus package, included an expansion of HIPAA’s privacy and security standards called the Health Information Technology for Economic and Clinical Health Act (HITECH). Among other things, HITECH created four new tiers of civil monetary penalties and gave state attorneys general new authority to file civil actions for damages or injunctions in federal courts to enforce the federal HIPAA laws and seek attorneys’ fees and costs associated with pursuing federal civil actions.
Employees
As of June 30, 2015, we had approximately 1,000 employees worldwide. None of our employees are represented by a collective bargaining agreement and we have never experienced a work stoppage. We believe our employee relations are good.
Facilities
We maintain a 180,000 square foot research and development and manufacturing facility in three buildings at our campus in Alameda, California. We leased the initial 75,000 square feet of the Alameda campus in 2008 and we added the additional space over several lease amendments and additional leases. The leases for all three buildings expire in 2029, subject to our option to renew any or all three leases for an additional ten years.
We also lease office space in Berlin, Germany; Sydney, Australia; and Sao Paulo, Brazil. The offices in Berlin and Sydney support our direct sales operations in Europe and Australia, respectively, and the office in Sao Paulo supports our Latin America marketing efforts through our distribution partners.
Legal Proceedings
We are not currently party to any material legal proceedings. We currently are and in the future may at times be involved in litigation and other legal claims in the ordinary course of business. When appropriate in our estimation, we may record reserves in our financial statements for pending litigation and other claims.
96
Executive Officers and Directors
The following table sets forth information regarding our executive officers and directors:
| Name |
Age | Position | ||||
| Adam Elsesser |
53 | Chairman, Chief Executive Officer and President | ||||
| Arani Bose, M.D. |
53 | Chief Innovator and Director | ||||
| Sri Kosaraju |
37 | Chief Financial Officer and Head of Strategy | ||||
| Daniel Davis |
37 | President, North America | ||||
| James Pray |
51 | President, International | ||||
| Lynn Rothman |
54 | Executive Vice President and Chief Business Officer | ||||
| Robert Evans |
68 | Executive Vice President, General Counsel and Secretary | ||||
| Don Kassing(1)(2)(3) |
73 | Director | ||||
| Walter Wang(1)(2)(3) |
47 | Director | ||||
| Harpreet Grewal(1)(2)(3) |
48 | Director | ||||
| (1) | Member of the audit committee. |
| (2) | Member of the compensation committee. |
| (3) | Member of the nominating and corporate governance committee. |
Executive Officers
Adam Elsesser co-founded Penumbra and has served as Chief Executive Officer and a member of our board of directors since our inception in 2004 and as President and Chairman of our board of directors since January 2015. Prior to Penumbra, Mr. Elsesser led SMART Therapeutics, Inc., a medical device company focused on devices for neuro-intervention, as its Chief Executive Officer from 2000 to 2002 and, after its acquisition by Boston Scientific Corporation, President of SMART Therapeutics within Boston Scientific Corporation from 2002 to 2005. Before his work in the medical device industry, Mr. Elsesser was a partner in the law firm of Shartsis Friese, LLP. Mr. Elsesser received a B.A. from Stanford University and a J.D. from Hastings College of the Law. Mr. Elsesser is qualified to serve on our board of directors based on his extensive knowledge of our company, the medical device industry and the competitive landscape, as well as his expertise in building and leading successful medical device companies and commercializing devices.
Arani Bose, M.D. co-founded Penumbra in 2004 and has served as a member of our board of directors since our inception. Dr. Bose was Chairman of our board of directors and Chief Medical Officer from 2005 until 2015 and currently serves as Chief Innovator. Prior to founding Penumbra, Dr. Bose was an Assistant Professor of Radiology and Neurology at New York University (NYU) School of Medicine from 1997 to 2004, where he also had a clinical practice. While at NYU, Dr. Bose co-founded SMART Therapeutics. Dr. Bose received a B.A. from Stanford University and a M.D. from the University of Colorado School of Medicine with residency and fellowships at Yale University School of Medicine and NYU Medical Center. Dr. Bose is qualified to serve on our board of directors based on his extensive knowledge of our company and the medical device industry, as well as his training and expertise in interventional radiology and neurology and his skills and experience in clinical research and device development and commercialization.
Sri Kosaraju joined Penumbra as Chief Financial Officer and Head of Strategy in 2015. Prior to joining Penumbra, Mr. Kosaraju worked in investment banking for J.P. Morgan Securities LLC (J.P. Morgan) from 1999 until 2015, where he held a variety of positions with successively greater responsibility, most recently Managing Director of Equity Capital Markets, Head of Healthcare Equity Capital Markets and co-Head of Technology, Media, Telecom Equity Capital Markets. Prior to entering J.P. Morgan’s equity capital markets group in 2006, Mr. Kosaraju served in various practice groups at J.P. Morgan, including Equity Derivatives from 2003 to 2006 and Technology, Media, Telecom Investment Banking Coverage from 1999 to 2003. Mr. Kosaraju received a B.S. from Massachusetts Institute of Technology in 1999.
97
Daniel Davis joined Penumbra in 2007. He has served as President, North America, since 2015. Mr. Davis previously served as Vice President of Strategy from 2013 to 2014 and as Director of Marketing from 2011 to 2012. Mr. Davis joined our sales team in 2007, serving as a Sales Trainer from 2008 to 2011. Before joining Penumbra, Mr. Davis held various sales and marketing positions at Boston Scientific Corporation. Mr. Davis received a B.A. from Duke University.
James Pray joined Penumbra in 2005. He has served as President, International, since 2015 and as President from 2005 to 2015. Mr. Pray joined Penumbra with over 15 years of experience in the medical device industry, much of that in the neurovascular arena. Prior to joining Penumbra, Mr. Pray worked for Boston Scientific Corporation, including as the Director of Marketing for the neurovascular division from 2000 to 2005. Mr. Pray also worked as a Research and Development Engineer and Engineering Manager for SCIMED Life Systems, a medical device company, from 1990 to 1996. Mr. Pray received a B.S. and M.S. from the University of Minnesota.
Lynn Rothman joined Penumbra in 2007. She has served as Executive Vice President and Chief Business Officer since January 2015; as Chief Financial Officer from March 2009 to January 2015; as Vice President, Administration from January 2009 to March 2009, and as Human Resources Manager from 2007 to January 2009. Ms. Rothman joined Penumbra with over 20 years of experience in finance and marketing of medical and emerging growth companies. Ms. Rothman served as Director of Corporate Marketing at Confer Software, Inc., a disease management company, from 1997 to 2000. Prior to that time, Ms. Rothman worked at Robertson Stephens & Company, a financial services firm, where she worked in both health care research and venture capital focused on potential medical device, service and software investment opportunities. She received an M.B.A. from The Wharton School, University of Pennsylvania and a B.A. from Stanford University.
Robert Evans joined Penumbra as Executive Vice President, General Counsel and Secretary in 2008. Prior to joining Penumbra, Mr. Evans was Executive Vice President, General Counsel and Secretary of Waste Connections, Inc., a publicly traded waste services company, from 2002 to 2008, and a partner in the law firm Shartsis Friese LLP from 1978 to 2002. Mr. Evans received a B.A. and J.D. from the University of California, Berkeley.
Nonemployee Directors
Don Kassing has served on our board of directors since 2008. Mr. Kassing is President Emeritus of San Jose State University. Mr. Kassing served as President of San Jose State University from 2004 to 2008, Interim President from 2010 to 2011, and Vice President, Administration and Finance and Chief Financial Officer from 1993 to 2004. Prior to his tenure at San Jose State University, Mr. Kassing spent 18 years in higher education and 11 years in private industry, including eight years in corporate finance and operations management at Brown Group, Inc., a retail real estate developer, and General Motors Corporation. Mr. Kassing received a B.A. and M.B.A. from Saint Louis University. Mr. Kassing is qualified to serve on our board of directors based on his extensive business and leadership experience, including valuable skills related to strategic planning, based on his long tenure leading a major educational institution, including overseeing the development and construction of two high-profile campus facilities and having primary responsibility for university business and financial affairs.
Walter Wang has served on our board of directors since January 2015. Mr. Wang has served as the Chief Executive Officer of Orlucent, Inc., an early stage diagnostics company, since 2012. From 1999 to the present, Mr. Wang has researched, advised and invested in medical device, financial and technology start-up companies through Wang Ventures, a family investment partnership and other related entities. In addition, Mr. Wang invests, develops and manages real estate through W5 Investments, a real estate company. Mr. Wang began his career in 1989 with U-Jin Enterprises, Inc., a food processing and import/export company. Mr. Wang received a B.S. from the University of Southern California. Mr. Wang is qualified to serve on our board of directors based on his extensive experience in analyzing, investing in and advising medical device and emerging growth companies and his strong financial background.
98
Harpreet Grewal has served on our board of directors since April 2015. Since 2010, Mr. Grewal has served as Executive Vice President and Chief Financial Officer of Constant Contact, Inc., a publicly-traded technology company primarily focused on marketing tools. From 2008 to 2009, Mr. Grewal worked as an independent consultant to small businesses and early-stage entrepreneurs. From 2006 through 2008, Mr. Grewal was Executive Vice President and Chief Financial Officer of VistaPrint, Ltd., a publicly-traded online printing and marketing services company. Prior to VistaPrint, Mr. Grewal was Senior Vice President and Chief Financial Officer of GoldenSource Corporation, a data management company, from 2002 to 2006, Chief Financial Officer of eGain Communications Corporation, a customer engagement services company, from 1999 to 2002, and held various financial and strategic planning positions with PepsiCo, Inc., a publicly-traded food and beverage company, from 1996 to 1999. Mr. Grewal received a B.A. from the University of California, Berkeley and a M.A. from Johns Hopkins University. Mr. Grewal is qualified to serve on our board of directors because of his extensive business and leadership experience, including financial expertise and strategic planning skills, based on his tenure as Chief Financial Officer at numerous companies, including several publicly-traded corporations.
Board Structure and Compensation of Directors
Upon completion of the offering, our board of directors will consist of five members. In accordance with our amended and restated certificate of incorporation and our amended and restated bylaws, immediately after the completion of this offering, our directors will be divided into three classes serving staggered three-year terms. At each annual meeting of stockholders, directors will be elected to succeed the class of directors whose terms have expired. Our current directors will be divided among the three classes as follows:
| • | the Class I directors will consist of Mr. Kassing, and his term will expire at the annual meeting of stockholders to be held in 2016; |
| • | the Class II directors will consist of Dr. Bose and Mr. Wang, and their terms will expire at the annual meeting of stockholders to be held in 2017; and |
| • | the Class III directors will consist of Messrs. Elsesser and Grewal, and their terms will expire at the annual meeting of stockholders to be held in 2018. |
This classification of our board of directors could have the effect of increasing the length of time necessary to change the composition of a majority of the board of directors. In general, at least two annual meetings of stockholders will be necessary for stockholders to effect a change in a majority of the members of the board of directors.
Director Independence
Our board of directors has undertaken a review of the independence of each director. Based on information provided by each director concerning his background, employment and affiliations, our board of directors has determined that each of Don Kassing, Walter Wang and Harpreet Grewal do not have a relationship that would interfere with the exercise of independent judgment in carrying out the responsibilities of a director and is independent under applicable NYSE rules. In making these determinations, our board of directors considered the current and prior relationships that each non-employee director has with our company and all other facts and circumstances our board of directors deemed relevant in determining their independence, including the beneficial ownership of our capital stock by each non-employee director, and the transactions involving them described in the section titled “Certain Relationships and Related Party Transactions.”
Director Compensation Policy
Directors who are also full-time officers or employees of our company will receive no additional compensation for serving as directors. All other directors will receive an annual retainer of $25,000. The chairman of the audit committee will receive an additional annual fee of $25,000. Each non-employee director also will receive an annual grant of 2,000 shares of restricted stock under our 2014 Equity Incentive Plan.
99
Board Committees
Audit Committee
The members of our audit committee are Don Kassing, Walter Wang and Harpreet Grewal. Harpreet Grewal is the chair of our audit committee. The composition of our audit committee meets the requirements for independence under the current NYSE listing standards and SEC rules and regulations. Each member of our audit committee is financially literate. In addition, our board of directors has determined that each of Mr. Grewal and Mr. Kassing is an “audit committee financial expert” as defined in Item 407(d)(5)(ii) of Regulation S-K promulgated under the Securities Act. This designation does not impose on either any duties, obligations or liabilities that are greater than are generally imposed on members of our audit committee and our board of directors. Our audit committee is directly responsible for, among other things:
| • | selecting a firm to serve as the independent registered public accounting firm to audit our financial statements; |
| • | ensuring the independence of the independent registered public accounting firm; |
| • | discussing the scope and results of the audit with the independent registered public accounting firm and reviewing, with management and that firm, our interim and year-end operating results; |
| • | establishing procedures for employees to anonymously submit concerns about questionable accounting or audit matters; |
| • | considering the adequacy of our internal controls and internal audit function; |
| • | reviewing material related party transactions or those that require disclosure; and |
| • | approving or, as permitted, pre-approving all audit and non-audit services to be performed by the independent registered public accounting firm. |
Our audit committee will operate under a written charter, to be effective prior to the completion of this offering, that satisfies the applicable rules of the SEC and the listing standards of the NYSE.
Compensation Committee
The members of our compensation committee are Don Kassing, Walter Wang and Harpreet Grewal. Walter Wang is the chair of our compensation committee. Each member of this committee is a non-employee director, as defined by Rule 16b-3 promulgated under the Securities Exchange Act of 1934, as amended (Exchange Act), and an outside director, as defined pursuant to Section 162(m) of the Code, and meets the requirements for independence under the current NYSE listing standards and SEC rules and regulations. Our compensation committee is responsible for, among other things:
| • | reviewing and approving, or recommending that our board of directors approve, the compensation of our executive officers; |
| • | reviewing and recommending to our board of directors the compensation of our directors; |
| • | administering our stock and equity incentive plans; |
| • | reviewing and approving, or making recommendations to our board of directors with respect to, incentive compensation and equity plans; and |
| • | reviewing our overall compensation philosophy. |
Our compensation committee will operate under a written charter, to be effective prior to the completion of this offering, that satisfies the applicable rules of the SEC and the listing standards of the NYSE.
Nominating and Governance Committee
The members of our nominating and governance committee are Don Kassing, Walter Wang and Harpreet Grewal. Don Kassing is the chair of our nominating and governance committee. Messrs. Kassing, Wang and
100
Grewal all meet the requirements for independence under the current NYSE listing standards. Our nominating and governance committee is responsible for, among other things:
| • | identifying and recommending candidates for membership on our board of directors; |
| • | reviewing and recommending our corporate governance guidelines and policies; |
| • | reviewing proposed waivers of the code of conduct for directors and executive officers; |
| • | overseeing the process of evaluating the performance of our board of directors; and |
| • | assisting our board of directors on corporate governance matters. |
Our nominating and governance committee will operate under a written charter, to be effective prior to the completion of this offering, that satisfies the applicable rules of the SEC and the listing standards of the NYSE.
Code of Ethics
In connection with this offering, our board of directors will adopt a code of ethics that applies to all of our employees, officers and directors, including our Chief Executive Officer, Chief Financial Officer and other executive and senior financial officers. Upon completion of this offering, the full text of our code of business conduct and ethics will be posted on the investor relations section of our website. We intend to disclose future amendments to our code of business conduct and ethics, or any waivers of such code, on our website or in public filings.
Limitation of Liability and Indemnification
Our amended and restated certificate of incorporation, which will be in effect immediately prior to the completion of this offering, contains provisions that limit the liability of our directors for monetary damages to the fullest extent permitted by Delaware law. Consequently, our directors will not be personally liable to us or our stockholders for monetary damages for any breach of fiduciary duties as directors, except liability for:
| • | any breach of the director’s duty of loyalty to us or our stockholders; |
| • | any act or omission not in good faith or that involves intentional misconduct or a knowing violation of law; |
| • | unlawful payments of dividends or unlawful stock repurchases or redemptions as provided in Section 174 of the Delaware General Corporation Law; or |
| • | any transaction from which the director derived an improper personal benefit. |
Our amended and restated certificate of incorporation to be in effect immediately prior to the completion of this offering will provide that we may indemnify our directors and officers, in each case to the fullest extent permitted by Delaware law. Our amended and restated bylaws to be in effect immediately prior to the completion of this offering will also provide that we are obligated to indemnify our directors and officers to the fullest extent permitted by Delaware law and advance expenses incurred by a director or officer in advance of the final disposition of any action or proceeding and permit us to secure insurance on behalf of any officer, director, employee or other agent for any liability arising out of his or her actions in that capacity regardless of whether we would otherwise be permitted to indemnify him or her under the provisions of Delaware law. We have entered into agreements to indemnify our directors, executive officers and other employees as determined by our board of directors. With specified exceptions, these agreements will provide for indemnification for related expenses including, among other things, attorneys’ fees, judgments, fines and settlement amounts incurred by any of these individuals in any action or proceeding. We believe these limitations of liability provisions and indemnification agreements are necessary to attract and retain qualified persons as directors and officers. We also maintain directors’ and officers’ liability insurance.
The limitation of liability and indemnification provisions in our amended and restated certificate of incorporation, amended and restated bylaws and indemnification agreements may discourage stockholders
101
from bringing a lawsuit against our directors and officers for breach of their fiduciary duty. Our amended and restated certificate of incorporation will provide that any such lawsuit must be brought in the Court of Chancery of the State of Delaware. The foregoing provisions may also reduce the likelihood of derivative litigation against our directors and officers, even though an action, if successful, might benefit us and other stockholders. Further, a stockholder’s investment may be adversely affected to the extent that we pay the costs of settlement and damage awards against directors and officers as required by these indemnification provisions. Insofar as indemnification for liabilities arising under the Securities Act may be permitted to our directors, officers and controlling persons pursuant to the foregoing provisions, or otherwise, we have been advised that, in the opinion of the SEC, such indemnification is against public policy as expressed in the Securities Act, and is, therefore, unenforceable. At present, there is no pending litigation or proceeding involving any of our directors, officers or employees for which indemnification is sought, and we are not aware of any threatened litigation that may result in claims for indemnification.
Compensation Committee Interlocks and Insider Participation
Our compensation committee has historically consisted solely of Adam Elsesser, our Chairman, Chief Executive Officer and President. As a result, Mr. Elsesser determined the compensation of our executive officers. Prior to the completion of this offering, we will appoint a compensation committee consisting solely of independent directors. None of our executive officers has served as a member of a compensation committee (or if no committee performs that function, the board of directors) of any other entity that has an executive officer serving as a member of our board of directors.
102
Summary Compensation Table
The following table sets forth information concerning the compensation paid to our principal executive officer, and our two most highly compensated executive officers other than the principal executive officer during our fiscal year ended December 31, 2014.
2014 SUMMARY COMPENSATION TABLE
| Name and Principal Position |
Year | Salary ($) | Stock Awards ($)(1) |
Option Awards ($)(1) |
All Other Compensation ($) |
Total ($) |
||||||||||||||||||
| Adam Elsesser, |
2014 | 511,250 | — | 1,162,500 | — | 1,673,750 | ||||||||||||||||||
| Chairman, Chief Executive Officer and President |
||||||||||||||||||||||||
| Daniel Davis, |
2014 | 300,000 | 387,500 | — | — | 687,500 | ||||||||||||||||||
| President, North America |
||||||||||||||||||||||||
| Lynn Rothman, |
2014 | 300,000 | 387,500 | — | — | 687,500 | ||||||||||||||||||
| Executive Vice President and Chief Business Officer |
||||||||||||||||||||||||
| (1) | The amounts reflect the aggregate grant date fair value for awards granted during 2014. The grant date fair value was computed in accordance with FASB ASC Topic 718, Compensation—Stock Compensation. Unlike the calculations contained in our financial statements, this calculation does not give effect to any estimate of forfeitures related to service-based vesting, but assumes that the executive will perform the requisite service for the award to vest in full. The assumptions we used in valuing options and stock awards are described in Note 12 to our Consolidated Financial Statements included in this prospectus. |
Executive Officer Arrangements
Our executive officers are employed on an at-will basis. Mr. Elsesser, Mr. Davis and Ms. Rothman have entered into at will employment agreements with us that set forth the general terms of their employment. We do not have agreements or policies that would require us to provide severance benefits or change-in-control benefits to our executive officers, other than the provisions under our equity plans described below.
Outstanding Equity Awards at Fiscal Year End
The following table sets forth information concerning unexercised options, stock that has not vested and equity incentive plan awards for the named executive officers named in the Summary Compensation Table as of the end of our fiscal year ended December 31, 2014.
103
OUTSTANDING EQUITY AWARDS AT 2014 FISCAL YEAR END
| Option Awards | Stock Awards | |||||||||||||||||||||||
| Name |
Numbers of Securities Underlying Unexercised Options (#) Exercisable(1) |
Numbers of Securities Underlying Unexercised Options (#) Unexercisable |
Option Exercise Price ($) |
Option Expiration Date |
Number of Shares or Units of Stock That Have Not Vested (#) |
Market Value of Shares or Units of Stock That Have Not Vested ($)(2) |
||||||||||||||||||
| Adam Elsesser |
300,000 | (3) | — | 0.57 | 04/01/2016 | — | — | |||||||||||||||||
| 200,000 | (3) | — | 1.26 | 05/01/2017 | — | — | ||||||||||||||||||
| 100,000 | (3) | — | 1.26 | 10/01/2017 | — | — | ||||||||||||||||||
| 5,000 | (4) | — | 7.75 | 03/21/2024 | — | — | ||||||||||||||||||
| 145,000 | (5) | — | 7.75 | 03/20/2024 | — | — | ||||||||||||||||||
| Daniel Davis |
— | — | — | — | 32 | (6) | 396 | |||||||||||||||||
| 65,000 | (7) | — | 3.98 | 09/30/2021 | — | — | ||||||||||||||||||
| 2,000 | (8) | — | 4.49 | 02/07/2022 | — | — | ||||||||||||||||||
| — | — | — | — | 50,000 | (9) | 618,000 | ||||||||||||||||||
| Lynn Rothman |
50,000 | (10) | — | 3.98 | 06/13/2021 | — | — | |||||||||||||||||
| — | — | — | — | 50,000 | (9) | 618,000 | ||||||||||||||||||
| (1) | Options may be exercised prior to vesting, subject to repurchase rights that expire over the vesting periods indicated in the footnotes below. Accordingly, all options outstanding as of December 31, 2014, were exercisable in full. |
| (2) | There was no public market for our common stock as of December 31, 2014. The fair value of our common stock as of December 31, 2014, was $12.36 per share. |
| (3) | The shares subject to this option were granted under the Penumbra 2005 Stock Plan and are fully vested as of December 31, 2014. Mr. Elsesser exercised options to purchase 300,000 shares of common stock on June 22, 2015. We withheld 52,337 of such shares for the payment of tax withholdings. Mr. Elsesser also tendered 11,825 of his existing shares of our common stock in payment of the exercise price. |
| (4) | Options were granted under the Penumbra 2005 Stock Plan and vest with respect to 1/4th of the shares subject to the option on March 21, 2015, and as to 1/48th of the total shares subject to the option on each month thereafter, subject to the executive continuing to provide services to us through each applicable vesting date. In the event of a Change in Control, as defined under the 2005 Stock Plan, the issued options will fully vest, subject to the executive continuing to provide services to us through the occurrence of the Change in Control. |
| (5) | Options were granted under the Penumbra 2011 Equity Incentive Plan and vest with respect to 1/4th of the shares subject to the option on March 21, 2015, and as to 1/48th of the total shares subject to the option on each month thereafter, subject to the executive continuing to provide services to us through each applicable vesting date. In the event of a Change in Control, as defined under the 2011 Equity Incentive Plan, the issued options will fully vest, subject to the executive continuing to provide services to us through the occurrence of the Change in Control. |
| (6) | The shares were issued upon the early exercise of options to purchase 1,500 shares of common stock. The original grant of options to purchase 1,500 shares of common stock was made under the Penumbra 2005 Stock Plan to vest as to 1/4th of the shares subject to the option on January 14, 2012, and as to 1/48th of the total shares subject to the option on each month thereafter, subject to the executive continuing to provide services to us through each applicable vesting date. Our repurchase rights with respect to issued restricted shares lapse over the vesting period. In the event of a Change in Control, as defined under the 2005 Stock Plan, the issued restricted shares will fully vest, subject to the executive continuing to provide services to us through the occurrence of the Change in Control. |
| (7) | Options were granted under the Penumbra 2005 Stock Plan and vest with respect to 1/4th of the shares subject to the option on September 30, 2012, and as to 1/48th of the total shares subject to the option on each month thereafter, subject to the executive continuing to provide services to us through each applicable vesting date. In the event of a Change in Control, as defined under the 2005 Stock Plan, the issued options will fully vest, subject to the executive continuing to provide services to us through the occurrence of the Change in Control. |
| (8) | Options were granted under the Penumbra 2005 Stock Plan and vest with respect to 1/4th of the shares subject to the option on January 1, 2013, and as to 1/4th of the total shares subject to the option on each January 2nd thereafter, |
104
| subject to the executive continuing to provide services to us through each applicable vesting date. In the event of a Change in Control, as defined under the 2005 Stock Plan, the issued options will fully vest, subject to the executive continuing to provide services to us through the occurrence of the Change in Control. |
| (9) | Mr. Davis and Ms. Rothman were each granted 50,000 restricted shares under the Penumbra 2011 Equity Incentive Plan on February 3, 2014. Our repurchase rights with respect to the issued restricted shares lapses as to 1/4th of the shares subject to the stock award on January 2, 2015, and as to 1/48th of the total shares subject to the award on each month thereafter, subject to the executive continuing to provide services to us through each applicable vesting date. In the event of a Change in Control, as defined under the 2011 Equity Incentive Plan, the issued restricted shares will fully vest, subject to the executive continuing to provide services to us through the occurrence of the Change in Control. |
| (10) | Options were granted under the Penumbra 2005 Stock Plan and vest with respect to 1/4th of the shares subject to the option on February 1, 2012, and as to 1/48th of the total shares subject to the option on each month thereafter, subject to the executive continuing to provide services to us through each applicable vesting date. In the event of a Change in Control, as defined under the 2005 Stock Plan, the issued options will fully vest, subject to the executive continuing to provide services to us through the occurrence of the Change in Control. |
Employee Benefit Plans
Our officers are entitled to participate in our equity incentive plans. All officers are eligible to participate in our 401(k) plan on the same terms as all other employees.
2014 Equity Incentive Plan
Our board of directors and our stockholders approved our 2014 Equity Incentive Plan (the 2014 plan) and it became effective in May, 2014. Our 2014 plan replaced our 2011 Equity Incentive Plan (the 2011 plan) and our 2005 Stock Plan (the 2005 plan). No further equity awards may be granted under our 2011 and 2005 plans. Our board of directors approved an amendment to the 2014 plan in August, 2015, which will become effective as of the business day immediately prior to the date of the effectiveness of the registration statement of which this prospectus is a part and will:
| • | increase the number of shares of our common stock initially reserved for issuance under the 2014 Plan to 3,000,000 shares; |
| • | include an “evergreen” provision which will automatically increase the shares of our common stock reserved for issuance under our 2014 Plan in an amount equal to the lesser of: (i) 2,500,000 shares of our common stock (after any adjustments for dividends or other distributions, stock splits, recapitalizations and certain other transactions in accordance with the plan); (ii) 5% of the number of outstanding shares of our common stock on the last day of the immediately preceding fiscal year; and (iii) an amount determined by the board of directors; |
| • | authorize the grant of performance awards which may be earned upon achievement or satisfaction of one or more performance conditions; and |
| • | authorize the grant of other awards that may be based on or related to shares of our common stock or other factors that may influence the value of our common stock. |
As of June 30, 2015, there were 1,713,634 shares remaining available for the grant of equity awards under our 2014 plan. As of June 30, 2015, we had granted options to purchase 536,650 shares of our common stock under our 2014 plan, of which 535,650 were outstanding and 1,000 options had been forfeited. We had also granted 662,361 shares of restricted stock under our 2014 plan, of which 503,646 shares were unvested and were subject to forfeiture.
Stock awards. The 2014 plan provides for the grant of incentive stock options (ISOs), nonstatutory stock options (NSOs), stock appreciation rights, restricted stock awards and restricted stock unit awards (collectively, “stock awards”). ISOs may be granted only to employees. All other awards may be granted to employees, directors and consultants.
Share reserve. The aggregate number of shares of our common stock initially reserved for issuance pursuant to stock awards under the 2014 plan was 2,700,000, plus (i) the shares reserved for issuance under
105
the 2005 plan and 2011 plan which were not issued or subject to awards granted under those plans and (ii) any shares subject to stock options or other stock awards granted under our 2011 and 2005 plans that expire or terminate for any reason, are forfeited or repurchased by us or are reacquired, withheld or not issued to satisfy a tax withholding obligation, up to a maximum of 3,007,276 shares added through clauses (i) or (ii). The maximum number of shares that may be issued upon the exercise of incentive stock options will equal this aggregate maximum number of shares plus other shares that become available upon lapsed awards or certain other conditions, to the extent allowed by section 422 of the Internal Revenue Code of 1986, as amended to the date hereof, and the regulations promulgated thereunder.
If a stock award granted under the 2014 plan is forfeited back to us because of the failure to meet a contingency or condition required to vest, such shares will become available for subsequent issuance under the 2014 plan. In addition, shares withheld to satisfy income or employment withholding taxes and shares used to pay the exercise price of a stock option will become available for the grant of new stock awards under the 2014 plan.
Administration. Our board of directors, or one or more duly authorized committees thereof, have the authority to administer the 2014 plan. Subject to the terms of the 2014 plan, our board of directors or the authorized committee, referred to herein as the plan administrator, determines recipients, dates of grant, the numbers and types of stock awards to be granted and the terms and conditions of the stock awards, including the period of their exercisability and vesting schedule applicable to a stock award. Subject to the limitations set forth below, the plan administrator will also determine the exercise price, strike price or purchase price of awards granted and the types of consideration to be paid for the award.
The plan administrator has the authority to modify outstanding awards under our 2014 plan. Subject to the terms of our 2014 plan, the plan administrator has the authority to extend the post-termination exercisability period of awards and to extend the maximum term of an option.
Stock Options. Incentive and nonstatutory stock options are granted pursuant to stock option agreements adopted by the plan administrator. The plan administrator determines the exercise price for a stock option, within the terms and conditions of the 2014 plan, provided that the exercise price of a stock option cannot be less than 100% of the fair market value of our common stock on the date of grant. In the case of an incentive stock option granted to an employee who owns stock representing more than 10% of the voting power of all classes of our stock or any parent or subsidiary of us, the exercise price will be no less 110% of the fair market value on the date of the grant. Options vest at the rate specified by the plan administrator. At the time an option is granted, the plan administrator will fix the period within which the option may be exercised and will determine any conditions that must be satisfied before the option may be exercised.
The plan administrator determines the term of stock options granted under the 2014 plan, up to a maximum of 10 years. In the case of an incentive stock option granted to an employee who owns stock representing more than 10% of the voting power of all classes of our stock or the stock of any of our parents or subsidiaries, the maximum term will be 5 years. Unless the terms of an option holder’s stock option agreement provide otherwise, if an option holder’s service relationship with us ceases for any reason other than disability or death, the option holder may generally exercise any vested options for a period of thirty days following the cessation of service. If an optionholder’s service relationship with us ceases due to disability or death, the optionholder or a beneficiary may generally exercise any vested options for a period of 6 months, or within such longer period of time as is specified in the award agreement. In no event may an option be exercised beyond the expiration of its term.
Acceptable consideration for the purchase of common stock issued upon the exercise of a stock option will be determined by the plan administrator and may include (1) cash, (2) check, (3) promissory note, (4) other shares, (5) a broker-assisted cashless exercise, (6) by net exercise, or (5) combination of the foregoing methods of payment.
Unless the plan administrator provides otherwise, options generally are not transferable except by will or by the laws of descent and distribution. An optionholder may designate a beneficiary, however, who may exercise the option following the optionholder’s death.
106
Stock Appreciation Rights. The 2014 plan permits the grant of stock appreciation rights. Stock appreciation rights give recipients the right to acquire a specified number of shares of stock at a predetermined price. The terms of the stock appreciation rights granted under the 2014 plan are determined by the plan administrator in the award agreement evidencing the award, including the number of shares, exercise price, expiration date and other terms.
Restricted Stock and Restricted Stock Units. The 2014 plan permits the grant of restricted stock and/or restricted stock units. Restricted stock awards are grants of shares of our common stock. Restricted stock units represent the right to receive shares of our common stock (or a cash amount equal to the value of our common stock) on future specified dates. The terms of the restricted stock and/or restricted stock units granted under the 2014 plan are determined by the plan administrator in the award agreement evidencing the award, including the number of shares, period of restriction or vesting schedule, and other terms.
Tax Limitations on Incentive Stock Options. The aggregate fair market value, determined at the time of grant, of our common stock with respect to ISOs that are exercisable for the first time by an optionholder during any calendar year under all of our stock plans may not exceed $100,000. Options or portions thereof that exceed such limit will generally be treated as NSOs. No ISO may be granted to any person who, at the time of the grant, owns or is deemed to own stock possessing more than 10% of our total combined voting power or that of any of our affiliates unless (1) the option exercise price is at least 110% of the fair market value of the stock subject to the option on the date of grant, and (2) the option is not exercisable after the expiration of five years from the date of grant.
Adjustments; Corporate Transactions. In the event of certain changes in our corporate structure, including any dividend or other distribution, recapitalization, stock split, reverse stock split, reorganization, merger, consolidation, split-up, spin-off, combination, repurchase, or exchange of shares of the company the administrator will make appropriate adjustments to outstanding awards to prevent diminution or enlargement of the benefits or potential benefits available under the plan.
Merger or Change of Control. In the event of certain corporate transactions specified in the plan, including a merger or change of control, as defined in the plan, each outstanding award will be treated as the administrator determines, without a participant’s consent, including that (i) awards will be assumed or substituted by the succeeding corporation; (ii) the awards will terminate; (iii) outstanding awards will vest and become exercisable, (iv) the awards will terminate in exchange for an amount of cash and/or property, if any, equal to the amount that would have been attained upon the exercise of the award or realization of the rights under the award as of the date of the transaction, (v) the replacement of any award with rights or property selected by the plan administrator or (vi) any combination of the above. In the event that the successor corporation does not assume or substitute the award, the participant will fully vest in and have the right to exercise all of his or her outstanding options and stock appreciation rights and all restrictions on restricted stock and restricted stock units will lapse and performance goals will be deemed achieved at 100% of the target levels. Our plan administrator is not obligated to treat all stock awards, even those that are of the same type, in the same manner. Under the 2014 plan, a change of control is generally (i) the acquisition by a person or entity of more than 50% of our combined voting power other than by private financing that is approved by the board, (ii) if we are public, the date on which a majority of the board has been replaced during any twelve month period by directors whose appointment or election is not endorsed by a majority of the members of the board prior to the date of appointment or election; or (iii) change in ownership of a substantial portion of our assets.
Amendment and Termination. The 2014 plan will terminate in 2024. However, our Board of Directors has the authority to amend, alter, or terminate our 2014 plan, provided that such action does not materially impair the existing rights of any participant without such participant’s written consent.
Employee Stock Purchase Plan
In August 2015, our board of directors adopted and we expect our stockholders to approve, our Employee Stock Purchase Plan, or the ESPP. The ESPP will become effective upon the effectiveness of the registration statement of which this prospectus forms a part. Unless otherwise determined by the Board, the ESPP will be administered by our compensation committee.
107
Share Reserve. The ESPP authorizes the initial issuance of up to a total of 600,000 shares of our common stock to participating employees. The ESPP provides that the number of shares reserved and available for purchase under the plan will automatically increase on the first day of each fiscal year beginning with the 2016 fiscal year and ending with the 2025 fiscal year, in an amount equal to the least of (i) 500,000 shares of our common stock, (ii) 1% of the outstanding shares of our common stock on the last day of immediately preceding fiscal year and (iii) such number of shares of our common stock determined by our board of directors. This number is subject to antidilution adjustment in the event of a stock split, stock dividend or other change in our capitalization.
Eligible Employees. All employees who have been employed by us or our designated subsidiaries are eligible to participate in the ESPP, provided that the plan administrator may determine from time to time in its discretion to not include in the ESPP or any particular offering period employees who work less than 20 hours per week or less than five months in any calendar year. Any employee who owns, or would own upon such purchase under the ESPP, 5% or more of the voting power or value of our stock is not eligible to purchase shares under our the ESPP.
Offering Periods. Unless otherwise determined by the administrator of the ESPP, each offering to our employees to purchase stock under the ESPP will begin on each May 20 and November 20 and will end on the following November 19 and May 19, respectively, each referred to as offering periods, provided that the first offering period will begin on the date that the registration statement of which this prospectus forms a part becomes effective and will end on May 19, 2016. The administrator may designate different offering periods prior to the beginning of an offering period in its discretion, but no offering shall exceed 27 months in duration.
Purchase Limits. Each employee who is a participant in the ESPP may purchase shares by authorizing payroll deductions at a minimum of 1% and up to 15% of his or her eligible compensation for each pay period. Unless the participating employee has previously withdrawn from the offering, his or her accumulated payroll deductions will be used to purchase our common stock on the last business day of the offering period at a price equal to 85% of the fair market value of the common stock on either the first or the last day of the offering period, whichever is lower, provided that no more than shares of our common stock or such other lesser maximum number established by the plan administrator may be purchased by any one employee during each offering period. Under applicable tax rules, an employee may purchase no more than $25,000 worth of common stock, valued at the start of the purchase period, under the ESPP in any calendar year.
The accumulated payroll deductions of any employee who is not a participant on the last day of an offering period will be refunded or retained for use in the next offering period. An employee’s rights under the ESPP terminate upon voluntary withdrawal from the plan or when the employee ceases employment for any reason. Eligible employees who have withdrawn from participation may reenroll effective the next offering period.
Corporate Reorganization. Prior to the effective time of any Corporate Reorganization, as defined in the ESPP, the ESPP will terminate and shares will be purchased prior to the effective time as determined by the plan administrator, unless the ESPP is continued by the company or assumed by the surviving corporation.
Non-Transferability. An employee may not transfer rights granted under the ESPP other than by beneficiary designation or the laws of descent and distribution.
Amendment and Termination. The ESPP may be terminated or amended by our board of directors at any time. Amendments that increase the number of shares of our common stock authorized under the ESPP and certain other amendments require the approval of our stockholders. The plan administrator may adopt and amend stock purchase subplans for employees of our non-U.S. subsidiaries.
2011 Equity Incentive Plan
Our board of directors approved our 2011 plan and it became effective in October 2011. As of June 30, 2015, we had granted options to purchase 145,000 shares of our common stock under our 2011 plan, all of which were outstanding. We had also granted 505,000 shares of restricted stock under our 2011 plan, of which 252,125 shares were unvested and were subject to forfeiture and 1,667 shares had been forfeited.
108
Stock awards. The 2011 plan provides for the grant of ISOs, NSOs, stock appreciation rights, restricted stock awards and restricted stock unit awards (collectively, stock awards). ISOs may be granted only to employees. All other awards may be granted to employees, directors and consultants.
Stock Options. No ISOs were issued under the 2011 plan. Nonstatutory stock options have been granted pursuant to stock option agreements adopted by the plan administrator. The terms of stock options granted under the 2011 plan are determined by the plan administrator in the award agreement evidencing the award, including the term of the option, exercise price of the option, vesting rate of the option, period within which the option may be exercised and any conditions that must be satisfied before the option may be exercised, provided that the exercise price of a stock option generally cannot be less than 100% of the fair market value of our common stock on the date of grant.
The plan administrator determines the term of stock options granted under the 2011 plan, up to a maximum of 10 years. Unless the terms of an option holder’s stock option agreement provide otherwise, if an option holder’s service relationship with us ceases for any reason other than termination for cause, disability or death, the option holder may generally exercise any vested options for a period of three months following the cessation of service. If an optionholder’s service relationship with us ceases due to termination for cause, the option terminates and ceases to be exercisable on termination of service. If an optionholder’s service relationship with us ceases due to disability or death, the optionholder or a beneficiary may generally exercise any vested options for a period of 12 months, or within such longer period of time as is specified in the award agreement. In no event may an option be exercised beyond the expiration of its term.
Unless the plan administrator provides otherwise, options generally are not transferable except by will or by the laws of descent and distribution. An optionholder may designate a beneficiary, however, who may exercise the option following the optionholder’s death.
Stock Appreciation Rights. The 2011 plan permits the grant of stock appreciation rights. The terms of the stock appreciation rights granted under the 2011 plan are determined by the plan administrator in the award agreement evidencing the award, including the number of shares, exercise price, expiration date and other terms.
Restricted Stock and Restricted Stock Units. The 2011 plan permits the grant of restricted stock and/or restricted stock units. The terms of the restricted stock and/or restricted stock units granted under the 2011 plan are determined by the plan administrator in the award agreement evidencing the award, including the number of shares, period of restriction or vesting schedule and other terms.
Adjustments; Corporate Transactions. In the event of certain changes in our corporate structure, including any dividend or other distribution, recapitalization, stock split, reverse stock split, reorganization, merger, consolidation, split-up, spin-off, combination, repurchase, or exchange of shares, the administrator will make appropriate adjustments to outstanding awards to prevent diminution or enlargement of the benefits or potential benefits available under the plan.
Merger or Change of Control. In the event of certain corporate transactions specified in the plan, including a merger or change of control, as defined in the plan, each outstanding award will be treated as the plan administrator determines, without a participant’s consent, including that (i) awards will be assumed or substituted by the succeeding corporation; (ii) the awards will terminate; (iii) outstanding awards will vest and become exercisable, (iv) the awards will terminate in exchange for an amount of cash and/or property, if any, equal to the amount that would have been attained upon the exercise of the award or realization of the rights under the award as of the date of the transaction, (v) the replacement of any award with rights or property selected by the plan administrator or (vi) any combination of the above. In the event that the successor corporation does not assume or substitute the award, the participant will fully vest in and have the right to exercise all of his or her outstanding options and stock appreciation rights and all restrictions on restricted stock and restricted stock units will lapse and performance goals will be deemed achieved at 100% of the target levels. Our plan administrator is not obligated to treat all stock awards, even those that are of the same type, in the same manner. Under the 2011 plan, a change of control is generally (i) the acquisition by a person or entity
109
of more than 50% of our combined voting power other than by private financing that is approved by the board, (ii) if we are public, the date on which a majority of the board has been replaced during any twelve month period by directors whose appointment or election is not endorsed by a majority of the members of the board prior to the date of appointment or election; or (iii) change in ownership of a substantial portion of our assets.
Amendment and Termination. The 2011 plan will terminate in October 2021. However, our board of directors has the authority to amend, alter, or terminate our 2011 plan, provided that such action does not materially impair the existing rights of any participant without such participant’s written consent. Termination of the 2011 plan will not affect the plan administrator’s ability to exercise its powers under the plan with respect to awards granted prior to termination of the plan.
Penumbra 2005 Stock Plan
Our board of directors and our stockholders approved our 2005 plan and it became effective in January 2005 and was subsequently amended and restated in 2006, 2007, 2008 and 2010. As of June 30, 2015, we had granted options to purchase 5,431,017 shares of our common stock under our 2005 plan, of which options to purchase 1,779,924 shares of our common stock were outstanding, and options to purchase 24,818 shares of our common stock had been early exercised and were unvested and subject to repurchase.
Stock awards. The 2005 plan provides for the grant of ISOs, NSOs, and stock purchase rights (collectively, stock awards). ISOs may be granted only to employees. All other awards may be granted to employees, directors and consultants.
Stock Options. Incentive and/or nonstatutory stock options have been granted pursuant to stock option agreements adopted by the plan administrator. The terms of stock options granted under the 2005 plan are determined by the board in the award agreement evidencing the award, including the term of the option, exercise price of the option, vesting schedule of the option, period within which the option may be exercised and any conditions that must be satisfied before the option may be exercised, provided that the exercise price of a stock option generally cannot be less than 100% of the fair market value of our common stock on the date of grant. In the case of an incentive stock option granted to an employee who owns stock representing more than 10% of the voting power of all classes of our stock or any parent or subsidiary of us, the exercise price will be no less 110% of the fair market value on the date of the grant.
The board determines the term of stock options granted under the 2005 plan, up to a maximum of 10 years. In the case of an incentive stock option granted to an employee who owns stock representing more than 10% of the voting power of all classes of our stock or the stock of any of our parents or subsidiaries, the maximum term will be 5 years. Unless the terms of an option holder’s stock option agreement provide otherwise, if an option holder’s service relationship with us ceases for any reason other than termination for cause, disability or death, the option holder may generally exercise any vested options for a period of three months following the cessation of service. If an optionholder’s service relationship with us ceases due to termination for cause, the option terminates and ceases to be exercisable on termination of service. If an optionholder’s service relationship with us ceases due to disability or death, the optionholder or a beneficiary may generally exercise any vested options for a period of 12 months, or within such longer period of time as is specified in the award agreement. In no event may an option be exercised beyond the expiration of its term.
Options generally are not transferable except by will or by the laws of descent and distribution.
Stock Purchase Rights. The 2005 plan permits the grant of stock purchase rights. The terms of the stock purchase rights granted under the 2005 plan are determined by the board in the award agreement evidencing the award, including the number of shares, purchase price, purchase period and other terms, provided that the purchase period is no longer than 30 days from the date of grant.
Adjustments; Corporate Transactions. In the event of certain changes in our corporate structure, including any dividend or other distribution, recapitalization, stock split, reverse stock split, reorganization, merger, consolidation, split-up, spin-off, combination, repurchase, or exchange of shares of the Company, the administrator will make appropriate adjustments to outstanding awards to prevent diminution or enlargement of the benefits or potential benefits available under the plan.
110
Merger or Change of Control. In the event of certain corporate transactions specified in the plan, including a merger or change of control, as defined in the plan, the surviving or purchasing corporation may, without the consent of the participants, either (i) assume our rights and obligations under the options or (ii) substitute for outstanding options substantially equivalent options of that company’s stock. Any unexercised options which are neither assumed by the surviving corporation nor substituted shall terminate as of the date of the change of control. In the event of a change of control, the board of directors may also cancel each or any option outstanding prior to the change of control in exchange for cash, equity or other property of the same value. In the event of a change in control, our board of directors may provide for the acceleration and vesting of any or all outstanding options and shares acquired upon the exercise of those options. Under the 2005 plan, a change of control is generally (i) the sale by our stockholders of more than 50% of the combined voting power of our outstanding shares, (ii) a merger or consolidation in which we are a party, (iii) the sale, exchange or transfer of substantially all of our assets, or (iv) our liquidation and dissolution, in each case where our stockholders after the transaction no longer beneficially own at least 50% of the combined voting power of our outstanding shares, or, in the case of an asset sale, beneficially own at least 50% of the combined voting power of the corporation to which our assets were transferred.
Amendment and Termination. The 2005 plan will continue in effect until the earlier of its termination by the board or all shares of stock available for issuance under the plan have been issued. All awards under the plan must be granted, if at all, within 10 years of its adoption. Termination or amendment of the 2005 plan will not affect any then outstanding award.
Pension Benefits
We do not maintain any defined benefit pension plans.
Nonqualified Deferred Compensation
We do not maintain any nonqualified deferred compensation plans.
111
2014 DIRECTOR COMPENSATION
None of our non-employee directors received any cash or equity compensation during fiscal 2014, or held any outstanding options or stock awards at the end of fiscal 2014. Mr. Elsesser, our Chairman, Chief Executive Officer and President, Dr. Bose, our Chief Innovator, and Mr. Krebs, our Treasurer and a director in 2014, did not receive additional compensation for their services as directors. For more information on Mr. Elsesser’s compensation as an officer, see the section titled “Executive Compensation.”
Benefit Plans for Directors
Our directors are entitled to participate in our equity incentive plans, described above.
112
CERTAIN RELATIONSHIPS AND RELATED PARTY TRANSACTIONS
We describe below transactions and series of similar transactions, during our last three fiscal years or currently proposed, to which we were a party or will be a party, in which:
| • | the amounts involved exceeds or will exceed $120,000; and |
| • | any of our directors, executive officers or beneficial holders of more than 5% of any class of our capital stock or any member of the immediate family of or entities affiliated with any of the forgoing persons had or will have a direct or indirect material interest. |
Other than as described below, there have not been, nor are there any currently proposed, transactions or series of similar transactions meeting this criteria to which we have been or will be a party other than compensation arrangements, which are described where required under the sections titled “Management—Board Structure and Compensation of Directors” and “Executive Compensation.”
Preferred Stock Financing and Tender Offer
On May 16, 2014, we sold a total of 4,545,455 shares of Series F preferred stock at a purchase price of $13.20 per share. Entities affiliated with Fidelity Investments, who currently beneficially own more than 5% of our outstanding capital stock, purchased 3,030,303 shares of Series F preferred stock in this offering. Prior to this investment, entities affiliated with Fidelity Investments were not affiliated with us.
In connection with our sale of Series F preferred stock in May 2014, we conducted a tender offer for up to 1,359,541 shares of our then-outstanding preferred stock at a price of $13.20 per share. Pursuant to the tender offer, a total of 584,052 shares of preferred stock were tendered by our stockholders. One of our executive officers, James Pray, and his immediate family or affiliated entities participated in the tender offer, tendering a total of 11,710 shares of preferred stock for aggregate consideration of $154,572.
Promissory Notes
In 2005, we made a loan to one of our executive officers, James Pray, in the form of a full-recourse secured promissory note in the amount of $85,000, to permit him to exercise stock options granted to him. This note was secured by a stock pledge agreement covering 500,000 shares of our common stock held by Mr. Pray. This note accrued interest at the rate of 2.92% per annum, with principal and interest due and payable on January 31, 2015, provided that upon the occurrence of a change of control of Penumbra, Mr. Pray would be required to pre pay principal in an amount equal to the product of the number of shares of our common stock that vested as a result of such change of control and $0.01. On April 14, 2015, our board of directors forgave this loan to Mr. Pray in its entirety, including accrued and unpaid interest thereon.
Investor Rights Agreement
We have entered into an agreement with our two founders, Adam Elsesser and Arani Bose, and the holders of our preferred stock including certain related persons, namely Daniel Davis, James Pray, Lynn Rothman, Robert Evans, Walter Wang, Harpreet Grewal and entities affiliated with FMR LLC, and certain members of the immediate family of the listed individuals, that provides for certain rights relating to the registration of their shares. See the section titled “Description of Capital Stock—Registration Rights” for additional information.
Director and Officer Indemnification
We have entered into an indemnification agreement with each of our directors and executive officers. These indemnification agreements and our amended and restated certificate of incorporation and amended and restated bylaws indemnify each of our directors and officers to the fullest extent permitted by the Delaware General Corporation Law. For information regarding these indemnification arrangements, please refer to the section titled “Management—Limitations on Liability and Indemnification of Directors and Officers.”
113
Policies and Procedures for Related Party Transactions
Our board of directors plans to adopt a written related party transaction policy setting forth the policies and procedures for the review and approval or ratification of related party transactions. This policy covers any transaction, arrangement or relationship, or any series of similar transactions, arrangements or relationships, in which we were or are to be a participant and a related party had or will have a direct or indirect material interest, as determined by the audit committee of our board of directors, including, without limitation, purchases of goods or services by or from the related party or entities in which the related party has a material interest, and indebtedness, guarantees of indebtedness or employment by us of a related party.
All related party transactions described in this section occurred prior to adoption of this policy and as such, these transactions were not subject to the approval and review procedures set forth in the policy. However, these transactions were reviewed and approved by our board of directors.
114
The following table sets forth information regarding beneficial ownership of our common stock as of June 30, 2015, by:
| • | each person whom we know to own beneficially more than 5% of our common stock; |
| • | each of our directors and named executive officers individually; and |
| • | all of our directors and executive officers as a group. |
In accordance with the rules of the SEC, beneficial ownership includes voting or investment power with respect to securities and includes the shares issuable pursuant to stock options that are exercisable within 60 days of June 30, 2015. Shares issuable pursuant to stock options are deemed outstanding for computing the percentage of the person holding such options but are not outstanding for computing the percentage of any other person. The number of shares of common stock outstanding after this offering includes shares of common stock being offered for sale by us in this offering. The percentage of beneficial ownership for the following table is based on 26,038,637 shares of common stock outstanding as of June 30, 2015, assuming the automatic conversion of all outstanding shares of our preferred stock as of June 30, 2015 into common stock, and shares of common stock outstanding after the completion of this offering assuming no exercise of the underwriters’ option to purchase additional shares.
Unless otherwise indicated, the address for each listed stockholder is: c/o Penumbra, Inc., One Penumbra Place, 1351 Harbor Bay Parkway, Alameda, California 94502. To our knowledge, except as indicated in the footnotes to this table and pursuant to applicable community property laws, the persons named in the table have sole voting and investment power with respect to all shares of common stock.
| Shares Beneficially Owned Before the Offering |
Shares Beneficially Owned After the Offering | |||||||||||
| Name and Address of Beneficial Owner |
Number | Percent | Number | Percent | ||||||||
| Greater than 5% Stockholders: |
||||||||||||
| Entities affiliated with FMR LLC(1) |
3,030,303 | 11.6 | % | |||||||||
| Directors and Named Executive Officers: |
||||||||||||
| Adam Elsesser(2) |
1,700,982 | 6.4 | % | |||||||||
| Daniel Davis(3) |
150,444 | * | ||||||||||
| Lynn Rothman(4) |
273,458 | 1.0 | % | |||||||||
| Arani Bose, M.D.(5) |
1,545,662 | 5.9 | % | |||||||||
| Don Kassing(6) |
16,000 | * | ||||||||||
| Walter Wang(7) |
1,480,931 | 5.7 | % | |||||||||
| Harpreet Grewal(8) |
14,139 | * | ||||||||||
| Directors and Officers as a Group |
6,671,256 | 24.8 | % | |||||||||
| * | Represents beneficial ownership of less than one percent (1%) of the outstanding common stock. |
| (1) | Consists of (i) 1,250,000 shares held by Fidelity Select Portfolios: Health Care Portfolio (“Fidelity Health”), (ii) 265,152 shares held by Fidelity Select Portfolios: Medical Equipment and Systems Portfolio (“Fidelity Medical Equipment”), (iii) 1,128,787 shares held by Fidelity Mt. Vernon Street Trust: Fidelity Growth Company Fund (“Fidelity Growth”), (iv) 257,576 shares held by Fidelity Mt. Vernon Street Trust: Fidelity Series Growth Company Fund (“Fidelity Series Growth”) and (v) 128,788 shares held by Fidelity Group Trust for Employee Benefit Plans: Fidelity Growth Company Commingled Pool (“Fidelity Group Trust”). Fidelity Health, Fidelity Medical Equipment, Fidelity Growth, Fidelity Series Growth and Fidelity Group Trust are managed by direct or indirect subsidiaries of FMR LLC. Edward C. Johnson 3d is a Director and the Chairman of FMR LLC and Abigail P. Johnson is a Director, the Vice Chairman and the President of FMR LLC. Members of the family of Edward C. Johnson 3d, including Abigail P. Johnson, are the predominant owners, directly or through trusts, of Series B voting common shares of FMR LLC, representing 49% of the voting power of FMR LLC. The Johnson family group and all other Series B shareholders have entered into a shareholders’ voting agreement under which all Series B voting common shares will be voted in accordance with the majority vote of Series B voting |
115
| common shares. Accordingly, through their ownership of voting common shares and the execution of the shareholders’ voting agreement, members of the Johnson family may be deemed, under the Investment Company Act of 1940, to form a controlling group with respect to FMR LLC. Neither FMR LLC nor Edward C. Johnson 3d nor Abigail P. Johnson has the sole power to vote or direct the voting of the shares owned directly by the various investment companies registered under the Investment Company Act (Fidelity Funds) advised by Fidelity Management & Research Company (FMR Co), a wholly owned subsidiary of FMR LLC, which power resides with the Fidelity Funds’ Boards of Trustees. Fidelity Management & Research Company carries out the voting of the shares under written guidelines established by the Fidelity Funds’ Boards of Trustees. The address for FMR LLC is 200 Seaport Blvd. V12G, Boston, Massachusetts 02210. |
| (2) | Consists of (i) 1,236,838 shares held by Mr. Elsesser, (ii) 14,144 shares held by the Siegel/Elsesser Revocable Trust and (iii) 450,000 shares issuable pursuant to options that are exercisable within 60 days of June 30, 2015 (of which 353,125 would be vested). |
| (3) | Consists of (i) 83,444 shares held by Mr. Davis, of which 37,500 shares are subject to our right of repurchase as of June 30, 2015, and (ii) 67,000 shares issuable pursuant to options that are exercisable within 60 days of June 30, 2015 (of which 64,083 would be vested). |
| (4) | Consists of (i) 200,000 shares held by Ms. Rothman, of which 37,500 shares are subject to our right of repurchase as of June 30, 2015, (ii) 23,458 shares held by Richard Koch & Lynn Rothman and (iii) 50,000 shares issuable pursuant to options that are exercisable within 60 days of June 30, 2015 (all of which would be vested). |
| (5) | Consists of (i) 731,518 shares held by Dr. Bose, (ii) 14,144 shares held by Arani & Shumita Bose, (iii) 250,000 shares held by the Arani Bose 2009 Family Trust, (iv) 250,000 shares held by the Shumita Bose 2009 Family Trust and (v) 300,000 shares issuable pursuant to options that are exercisable within 60 days of June 30, 2015 (all of which would be vested). |
| (6) | Consists of 16,000 shares held by The Kassing Family Trust. |
| (7) | Consists of (i) 517,071 shares held by Mr. Wang and (ii) 963,860 shares held by Wang Ventures LLC. Mr. Wang is the owner of a 25% interest in Wang Ventures LLC as a Member. Mr. Wang disclaims beneficial ownership of all shares held by Wang Ventures LLC. |
| (8) | Consists of 14,139 shares held by Mr. Grewal. |
| (9) | Consists of (i) 5,804,256 shares held by all current executive officers and directors as a group, of which 530,209 shares are subject to our right of repurchase as of June 30, 2015 and (ii) 867,000 shares issuable pursuant to options that are exercisable within 60 days of June 30, 2015 (of which 767,208 would be vested). |
116
The following descriptions are summaries of the material terms of our amended and restated certificate of incorporation and amended and restated bylaws that will be in effect upon the completion of this offering. Reference is made to the more detailed provisions of, and the descriptions are qualified in their entirety by reference to, these documents, copies of which will be filed with the SEC as exhibits to the registration statement of which this prospectus is a part. We refer in this section to our amended and restated certificate of incorporation as our certificate of incorporation, and we refer to our amended and restated bylaws as our bylaws.
General
Following this offering, our authorized capital stock will consist of 300,000,000 shares of common stock, par value $0.001 per share, and 5,000,000 shares of preferred stock, par value $0.001 per share.
Common Stock
Common stock outstanding. As of June 30, 2015, there were 26,038,637 shares of common stock outstanding (including preferred stock on an as-converted basis) which were held of record by 345 stockholders. There will be shares of common stock outstanding, assuming no exercise of the underwriters’ option to purchase additional shares and no exercise of outstanding options, after giving effect to the sale of the shares of common stock offered hereby. All outstanding shares of common stock are fully paid and non-assessable, and the shares of common stock to be issued upon completion of this offering will be fully paid and non-assessable.
Voting rights. The holders of common stock are entitled to one vote per share on all matters to be voted upon by the stockholders.
Dividend rights. Subject to preferences that may be applicable to any outstanding preferred stock, the holders of common stock are entitled to receive ratably such dividends, if any, as may be declared from time to time by the board of directors out of funds legally available therefor. See the section titled “Dividend Policy.”
Rights upon liquidation. In the event of liquidation, dissolution or winding-up of Penumbra, the holders of common stock are entitled to share ratably in all assets remaining after payment of liabilities, subject to prior distribution rights of preferred stock, if any, then outstanding.
Other rights. The holders of our common stock have no preemptive or conversion rights or other subscription rights. There are no redemption or sinking fund provisions applicable to the common stock.
Preferred Stock
Effective immediately prior to the completion of this offering, there will be no shares of preferred stock outstanding because all of our outstanding shares of preferred stock will have been automatically converted into an aggregate of 19,510,410 shares of common stock. Our board of directors has the authority to issue the preferred stock in one or more series and to fix the rights, preferences, privileges and restrictions thereof, including dividend rights, dividend rates, conversion rights, voting rights, terms of redemption, redemption prices, liquidation preferences and the number of shares constituting any series or the designation of such series, without further vote or action by the stockholders.
The issuance of preferred stock may have the effect of delaying, deferring or preventing a change in control of Penumbra without further action by the stockholders and may adversely affect the voting and other rights of the holders of common stock. At present, we have no plans to issue any of the preferred stock.
Registration Rights
After the closing of this offering, certain holders of our common stock will be entitled to rights with respect to the registration of their shares under the Securities Act. These registration rights are contained in our Fourth Amended and Restated Investors’ Rights Agreement, dated as of May 16, 2014, and are described in additional
117
detail below. With respect to any particular stockholder, when such stockholder is able to sell all of its shares pursuant to Rule 144 of the Securities Act without restriction (including any volume limitations), these registration rights will expire. We will pay the registration expenses (other than underwriting discounts and selling commissions) of the holders of the shares registered pursuant to the registrations describe below. In an underwritten offering, the managing underwriter, if any, has the right, subject to specified conditions, to limit the number of shares such holders may include. In connection with this offering, each stockholder that has registration rights agreed not to sell or otherwise dispose of any securities without the prior written consent of the underwriters for a period of 180 days after the date of this prospectus, subject to certain terms and conditions. For more information regarding such restrictions, see the section titled “Underwriting.”
Demand Registration Rights
Beginning 180 days following this offering through the date that is three years following this offering, the holders of approximately 19,641,495 shares of our common stock will be entitled to certain demand registration rights. The holders of at least 66 2/3% of these securities have the right to require us, on not more than two occasions, to file a registration statement under the Securities Act in order to register the resale of their shares of common stock. We may, in certain circumstances, defer such registrations and the underwriters have the right, subject to certain limitations, to limit the number of shares included in such registrations.
Piggyback Registration Rights
After the closing of this offering, if we propose to register the offer and sale of any of our securities under the Securities Act, in connection with the public offering of such securities the holders of approximately 21,617,845 shares of our common stock will be entitled to certain “piggyback” registration rights allowing the holders to include their shares in such registration, subject to certain marketing and other limitations. As a result, whenever we propose to file a registration statement under the Securities Act, other than with respect to a registration related to a company stock plan or on any form which does not include substantially the same information as would be required to be included in a registration statement covering the sale of the registrable securities or a registration that would not customarily provide for the sale of secondary equity shares for cash, the holders of these shares are entitled to notice of the registration and have the right, subject to limitations that the underwriters may impose on the number of shares included in the registration, to include their shares in the registration.
S-3 Registration Rights
After the closing of this offering, the holders of approximately 19,641,495 shares of our common stock may make a written request that we register the offer and sale of their shares on Form S-3 if we are eligible to file a registration statement on Form S-3 so long as the request covers at least that number of shares with an anticipated aggregate offering price of at least $1.0 million. These stockholders may make an unlimited number of requests for registration on Form S-3; however, we will not be required to effect a registration on Form S-3 if we have effected two such registrations in a given 12-month period. Additionally, if we determine that it would be seriously detrimental to us and our stockholders to effect such a registration, we have the right to defer such registration, not more than once in any 12-month period, for a period of up to 180 days.
Election and Removal of Directors
Immediately prior to the completion of this offering, our board of directors will consist of between five and nine directors. The exact number of directors will be fixed from time to time by resolution of the board. No director may be removed except for cause, and directors may be removed for cause by an affirmative vote of shares representing a majority of the shares then entitled to vote at an election of directors. Any vacancy occurring on the board of directors and any newly created directorship may be filled only by a majority of the remaining directors in office.
118
Staggered Board
Upon the closing of this offering, our board of directors will be will be divided into three classes serving staggered three-year terms. Class I, Class II and Class III directors will serve until our annual meetings of stockholders in 2016, 2017 and 2018, respectively. At each annual meeting of stockholders, directors will be elected to succeed the class of directors whose terms have expired. This classification of our board of directors could have the effect of increasing the length of time necessary to change the composition of a majority of the board of directors. In general, at least two annual meetings of stockholders will be necessary for stockholders to effect a change in a majority of the members of the board of directors.
Limits on Written Consents
Our certificate of incorporation and our bylaws will provide that holders of our common stock will not be able to act by written consent without a meeting.
Stockholder Meetings
Our certificate of incorporation and our bylaws will provide that special meetings of our stockholders may be called only by our board of directors acting pursuant to a resolution adopted by a majority of the board of directors.
Amendment of Certificate of Incorporation
The provisions of our certificate of incorporation described under the sections titled “Common Stock—Voting rights,” “Amendment of Bylaws,” “Election and Removal of Directors,” “Stockholder Meetings” and “Limits on Written Consents” may be amended only by the affirmative vote of holders of at least 662/3% of the voting power of our outstanding shares of voting stock, voting together as a single class. The affirmative vote of holders of at least a majority of the voting power of our outstanding shares of stock will generally be required to amend other provisions of our certificate of incorporation.
Amendment of Bylaws
Our bylaws may generally be altered, amended or repealed, and new bylaws may be adopted, with:
| • | the affirmative vote of a majority of directors present at any regular or special meeting of the board of directors called for that purpose, or without a meeting if all members of the board of directors consent to the taking of the action; or |
| • | the affirmative vote of holders of 66 2⁄3% of the voting power of our outstanding shares of voting stock, voting together as a single class. |
Other Limitations on Stockholder Actions
Our bylaws will also impose some procedural requirements on stockholders who wish to:
| • | make nominations in the election of directors; |
| • | propose any amendment to our bylaws; or |
| • | propose any other business to be brought before an annual or special meeting of stockholders. |
Under these procedural requirements, in order to bring a proposal before a meeting of stockholders, a stockholder must deliver timely notice of a proposal pertaining to a proper subject for presentation at the meeting to our corporate secretary along with the following:
| • | a brief description of any business to be brought before the meeting and the reasons for conducting such business at the meeting; |
| • | the text of any proposed amendments to our bylaws; |
119
| • | the stockholder’s name and address; |
| • | the number and type of shares beneficially owned by the stockholder and evidence of such ownership; |
| • | a description of any arrangement between the stockholder and other beneficial owners of our stock; |
| • | any hedging arrangements with respect to our securities entered into by the stockholder; |
| • | representations as to whether the stockholder intends to appear at the meeting, distribute a proxy statement or otherwise solicit proxies; and |
| • | any other information required to be disclosed under the Exchange Act or that we may reasonably require to determine that the proposed business is proper. |
In order to submit a nomination for our board of directors, a stockholder must also timely submit any information with respect to the nominee that we would be required to include in a proxy statement, as well as some other information. If a stockholder fails to follow the required procedures, the stockholder’s proposal or nominee will be ineligible and will not be voted on by our stockholders.
To be timely, a stockholder must generally deliver notice:
| • | in connection with an annual meeting of stockholders, not less than 120 nor more than 150 days prior to the date on which the annual meeting of stockholders was held in the immediately preceding year, but in the event that the date of the annual meeting is more than 30 days before or more than 70 days after the anniversary date of the preceding annual meeting of stockholders, a stockholder notice will be timely if received by us not later than the close of business on the later of (1) the 70th day prior to the annual meeting and (2) the 10th day following the day on which we first publicly announce the date of the annual meeting; or |
| • | in connection with the election of a director at a special meeting of stockholders, not earlier than 150 nor later than the later of (1) 120 days prior to the date of the special meeting and (2) the close of business on the 10th day following the day on which a notice of the date of the special meeting was mailed to the stockholders or the public disclosure of that date was made. |
Limitation of Liability of Directors and Officers
Our certificate of incorporation will provide that no director will be personally liable to us or our stockholders for monetary damages for breach of fiduciary duty as a director, except as required by applicable law, as in effect from time to time. Currently, Delaware law requires that liability be imposed for the following:
| • | any breach of the director’s duty of loyalty to our company or our stockholders; |
| • | any act or omission not in good faith or which involved intentional misconduct or a knowing violation of law; |
| • | unlawful payments of dividends or unlawful stock repurchases or redemptions as provided in Section 174 of the Delaware General Corporation Law; and |
| • | any transaction from which the director derived an improper personal benefit. |
As a result, neither we nor our stockholders have the right, through stockholders’ derivative suits on our behalf, to recover monetary damages against a director for breach of fiduciary duty as a director, including breaches resulting from grossly negligent behavior, except in the situations described above.
Our bylaws provide that, to the fullest extent permitted by law, we will indemnify any officer or director of our company against all damages, claims and liabilities arising out of the fact that the person is or was our director or officer, or served any other enterprise at our request as a director, officer, employee, agent or fiduciary. We will reimburse the expenses, including attorneys’ fees, incurred by a person indemnified by this provision when we receive an undertaking to repay such amounts if it is ultimately determined that the person is not entitled to be indemnified by us. Amending this provision will not reduce our indemnification obligations relating to actions taken before an amendment.
120
Forum Selection
The Court of Chancery of the State of Delaware will be the sole and exclusive forum for (i) any derivative action or proceeding brought on behalf of Penumbra, (ii) any action asserting a claim of breach of fiduciary duty owed by any director, officer or other employee of Penumbra to Penumbra or Penumbra’s stockholders, (iii) any action asserting a claim arising pursuant to any provision of the Delaware General Corporation Law, or (iv) any action asserting a claim governed by the internal affairs doctrine. Any person or entity purchasing or otherwise acquiring any interest in shares of capital stock of Penumbra shall be deemed to have notice of and consented to the foregoing forum selection provisions.
Delaware Business Combination Statute
We elect to be subject to Section 203 of the Delaware General Corporation Law, which regulates corporate acquisitions. Section 203 prevents an “interested stockholder,” which is defined generally as a person owning 15% or more of a corporation’s voting stock, or any affiliate or associate of that person, from engaging in a broad range of “business combinations” with the corporation for three years after becoming an interested stockholder unless:
| • | the board of directors of the corporation had previously approved either the business combination or the transaction that resulted in the stockholder’s becoming an interested stockholder; |
| • | upon completion of the transaction that resulted in the stockholder’s becoming an interested stockholder, that person owned at least 85% of the voting stock of the corporation outstanding at the time the transaction commenced, other than statutorily excluded shares; or |
| • | following the transaction in which that person became an interested stockholder, the business combination is approved by the board of directors of the corporation and holders of at least two-thirds of the outstanding voting stock not owned by the interested stockholder. |
Under Section 203, the restrictions described above also do not apply to specific business combinations proposed by an interested stockholder following the announcement or notification of designated extraordinary transactions involving the corporation and a person who had not been an interested stockholder during the previous three years or who became an interested stockholder with the approval of a majority of the corporation’s directors, if such extraordinary transaction is approved or not opposed by a majority of the directors who were directors prior to any person becoming an interested stockholder during the previous three years or were recommended for election or elected to succeed such directors by a majority of such directors.
Section 203 may make it more difficult for a person who would be an interested stockholder to effect various business combinations with a corporation for a three-year period. Section 203 also may have the effect of preventing changes in our management and could make it more difficult to accomplish transactions that our stockholders may otherwise deem to be in their best interests.
Anti-Takeover Effects of Some Provisions
Some provisions of our certificate of incorporation and bylaws could make an acquisition of control of us by means of a proxy contest or otherwise more difficult, including our classified board and ability to issue preferred stock. These provisions are designed to discourage coercive takeover practices and inadequate takeover bids. These provisions are also designed to encourage persons seeking to acquire control of us to first negotiate with our board of directors. We believe that the benefits of increased protection give us the potential ability to negotiate with the proponent of an unfriendly or unsolicited proposal to acquire or restructure us, and that the benefits of this increased protection outweigh the disadvantages of discouraging those proposals, because negotiation of those proposals could result in an improvement of their terms.
Listing
We expect to apply to list our common stock on the NYSE under the symbol “PEN.”
Transfer Agent and Registrar
The transfer agent and registrar for our common stock is American Stock Transfer & Trust Company, LLC.
121
MATERIAL U.S. FEDERAL INCOME AND ESTATE TAX CONSEQUENCES FOR
NON-U.S. HOLDERS OF COMMON STOCK
The following are the material U.S. federal income and estate tax consequences of the ownership and disposition of our common stock acquired in this offering by a “Non-U.S. Holder” that does not own, and has not owned, actually or constructively, more than 5% of our common stock. You are a Non-U.S. Holder if for U.S. federal income tax purposes you are a beneficial owner of our common stock that is:
| • | a nonresident alien individual; |
| • | a foreign corporation; or |
| • | a foreign estate or trust. |
You are not a Non-U.S. Holder if you are a nonresident alien individual present in the United States for 183 days or more in the taxable year of disposition, or if you are a former citizen or former resident of the United States for U.S. federal income tax purposes. If you are such a person, you should consult your tax adviser regarding the U.S. federal income tax consequences of the ownership and disposition of our common stock.
If you are a partnership for U.S. federal income tax purposes, the U.S. federal income tax treatment of a partner will generally depend on the status of the partner and your activities.
This discussion is based on the Internal Revenue Code of 1986, as amended to the date hereof (the Code), administrative pronouncements, judicial decisions and final, temporary and proposed Treasury regulations, changes to any of which subsequent to the date of this prospectus may affect the tax consequences described herein, possibly with retroactive effect. This discussion does not describe all of the tax consequences that may be relevant to you in light of your particular circumstances, including alternative minimum tax and Medicare contribution tax consequences, and does not address any aspect of state, local or non-U.S. taxation, or any taxes other than income and estate taxes. You should consult your tax adviser with regard to the application of the U.S. federal tax laws to your particular situation, as well as any tax consequences arising under the laws of any state, local or non-U.S. taxing jurisdiction.
Dividends
As discussed under “Dividend Policy” above, we do not currently expect to make distributions on our common stock. In the event that we do make distributions of cash or other property, those distributions will constitute dividends for U.S. federal income tax purposes to the extent paid from our current or accumulated earnings and profits, as determined under U.S. federal income tax principles. To the extent those distributions exceed our current and accumulated earnings and profits, they will constitute a return of capital, which will first reduce your basis in our common stock, but not below zero, and then will be treated as gain from the sale of our common stock, as described below under the section titled “—Gain on Disposition of Our Common Stock.”
Dividends paid to you generally will be subject to U.S. federal withholding tax at a 30% rate or a reduced rate specified by an applicable income tax treaty. In order to obtain a reduced rate of withholding, you will be required to provide a properly executed applicable Internal Revenue Service (IRS) Form W-8 certifying your entitlement to benefits under a treaty.
If dividends paid to you are effectively connected with your conduct of a trade or business in the United States (and, if required by an applicable income tax treaty, are attributable to a permanent establishment or fixed base maintained by you in the United States), you will generally be taxed on the dividends in the same manner as a U.S. person. In this case, you will be exempt from the withholding tax discussed in the preceding paragraph, although you will be required to provide a properly executed IRS Form W-8ECI in order to claim an exemption from withholding. You should consult your tax adviser with respect to other U.S. tax consequences of the ownership and disposition of our common stock, including the possible imposition of a branch profits tax at a rate of 30% (or a lower treaty rate) if you are a corporation.
122
Gain on Disposition of Our Common Stock
Subject to the discussions below under the sections titled “—Information Reporting and Backup Withholding” and “—FATCA,” you generally will not be subject to U.S. federal income or withholding tax on gain realized on a sale or other taxable disposition of our common stock unless:
| • | the gain is effectively connected with your conduct of a trade or business in the United States (and, if required by an applicable income tax treaty, is attributable to a permanent establishment or fixed base maintained by you in the United States), or |
| • | we are or have been a “United States real property holding corporation,” as defined in the Code, at any time within the five-year period preceding the disposition or your holding period, whichever period is shorter, and our common stock is not regularly traded on an established securities market during the calendar year in which the sale or disposition occurs. |
We believe that we are not, and do not anticipate becoming, a United States real property holding corporation.
If you recognize gain on a sale or other disposition of our common stock that is effectively connected with your conduct of a trade or business in the United States (and, if required by an applicable income tax treaty, is attributable to a permanent establishment or fixed base maintained by you in the United States), you will generally be taxed on such gain in the same manner as a U.S. person. You should consult your tax adviser with respect to other U.S. tax consequences of the ownership and disposition of our common stock, including the possible imposition of a branch profits tax at a rate of 30% (or a lower treaty rate) if you are a corporation.
Information Reporting and Backup Withholding
Information returns are required to be filed with the IRS in connection with payments of dividends on our common stock. Unless you comply with certification procedures to establish that you are not a U.S. person, information returns may also be filed with the IRS in connection with the payment of proceeds from a sale or other disposition of our common stock. You may be subject to backup withholding on payments on our common stock or on the proceeds from a sale or other disposition of our common stock unless you comply with certification procedures to establish that you are not a U.S. person or otherwise establish an exemption. Your provision of a properly executed applicable IRS Form W-8 (usually IRS Form W-8BEN or W-8BEN-E) certifying your non-U.S. status will permit you to avoid backup withholding. Amounts withheld under the backup withholding rules are not additional taxes and may be refunded or credited against your U.S. federal income tax liability, provided the required information is timely furnished to the IRS.
FATCA
Provisions of the Code commonly referred to as “FATCA” currently require withholding of 30% on payments of dividends on our common stock, and will require 30% withholding on payments of gross proceeds of dispositions of our common stock occurring after December 31, 2016, to “foreign financial institutions” (which is broadly defined for this purpose and in general includes investment vehicles) and certain other non-U.S. entities unless various U.S. information reporting and due diligence requirements (generally relating to ownership by U.S. persons of interests in or accounts with those entities) have been satisfied, or an exemption applies. An intergovernmental agreement between the United States and an applicable foreign country may modify these requirements. If FATCA withholding is imposed, a beneficial owner that is not a foreign financial institution generally may obtain a refund of any amounts withheld by filing a U.S. federal income tax return (which may entail significant administrative burden). You should consult your tax adviser regarding the effects of FATCA on your investment in our common stock.
Federal Estate Tax
Individual Non-U.S. Holders and entities the property of which is potentially includible in such an individual’s gross estate for U.S. federal estate tax purposes (for example, a trust funded by such an individual and with respect to which the individual has retained certain interests or powers), should note that, absent an applicable treaty exemption, our common stock will be treated as U.S. situs property subject to U.S. federal estate tax.
123
SHARES ELIGIBLE FOR FUTURE SALE
Prior to this offering, there has been no market for our common stock. Future sales of substantial amounts of our common stock in the public market could adversely affect market prices prevailing from time to time. Furthermore, because only a limited number of shares will be available for sale shortly after this offering due to existing contractual and legal restrictions on resale as described below, there may be sales of substantial amounts of our common stock in the public market after the restrictions lapse. This may adversely affect the prevailing market price and our ability to raise equity capital in the future.
Upon the completion of this offering, we will have shares of common stock outstanding assuming the exercise of the underwriters’ option to purchase additional shares, the automatic conversion of all outstanding shares of preferred stock and no exercise of any options outstanding as of June 30, 2015. Of these shares, the shares, or shares if the underwriters exercise their option to purchase additional shares in full, sold in this offering will be freely transferable without restriction or registration under the Securities Act, except for any shares purchased by one of our existing “affiliates,” as that term is defined in Rule 144 under the Securities Act. The remaining shares of common stock existing are “restricted shares” as defined in Rule 144. Restricted shares may be sold in the public market only if registered or if they qualify for an exemption from registration under Rules 144 or 701 of the Securities Act. As a result of the contractual 180-day lock-up period described below and the provisions of Rules 144 and 701, these shares will be available for sale in the public market as follows:
| Number of Shares |
Date | |
| On the date of this prospectus. | ||
| After 180 days from the date of this prospectus (subject, in some cases, to volume limitations). | ||
| At various times after 180 days from the date of this prospectus (subject, in some cases, to volume limitations). | ||
Rule 144
In general, a person who has beneficially owned restricted shares of our common stock for at least six months would be entitled to sell such securities, provided that (i) such person is not deemed to have been one of our affiliates at the time of, or at any time during the 90 days preceding, a sale and (ii) we are subject to the Exchange Act periodic reporting requirements for at least 90 days before the sale. Persons who have beneficially owned restricted shares of our common stock for at least six months but who are our affiliates at the time of, or any time during the 90 days preceding, a sale, would be subject to additional restrictions, by which such person would be entitled to sell within any three month period only a number of securities that does not exceed the greater of either of the following:
| • | 1% of the number of shares of our common stock then outstanding, which will equal approximately shares immediately after this offering, assuming no exercise of the underwriters’ option to purchase additional shares; or |
| • | the average weekly trading volume of our common stock on the during the four calendar weeks preceding the filing of a notice on Form 144 with respect to the sale; |
provided, in each case, that we are subject to the Exchange Act periodic reporting requirements for at least 90 days before the sale. Such sales both by affiliates and by non-affiliates must also comply with the manner of sale, current public information and notice provisions of Rule 144 to the extent applicable.
Rule 701
In general, under Rule 701, any of our employees, directors, officers, consultants or advisors who purchases shares from us in connection with a compensatory stock or option plan or other written agreement
124
before the effective date of this offering is entitled to resell such shares 90 days after the effective date of this offering in reliance on Rule 144, without having to comply with the holding period requirements or other restrictions contained in Rule 701.
The SEC has indicated that Rule 701 will apply to typical stock options granted by an issuer before it becomes subject to the reporting requirements of the Exchange Act, along with the shares acquired upon exercise of such options, including exercises after the date of this prospectus. Securities issued in reliance on Rule 701 are restricted securities and, subject to the contractual restrictions described above, beginning 90 days after the date of this prospectus, may be sold by persons other than “affiliates,” as defined in Rule 144, subject only to the manner of sale provisions of Rule 144 and by “affiliates” under Rule 144 without compliance with its one-year minimum holding period requirement.
Registration Rights
Upon the completion of this offering, the holders of 21,617,845 shares of common stock will be entitled to various rights with respect to the registration of these shares under the Securities Act. Registration of these shares under the Securities Act would result in these shares becoming freely tradable without restriction under the Securities Act immediately upon the effectiveness of the registration, except for shares purchased by affiliates.
Stock Options
As of June 30, 2015, options to purchase a total of 2,460,574 shares of common stock were outstanding. An additional 1,713,634 shares of common stock were available for future grants under our stock plans.
Upon the completion of this offering, we intend to file a registration statement under the Securities Act covering all shares of common stock subject to outstanding options or issuable pursuant to our 2014 Equity Incentive Plan. Subject to Rule 144 volume limitations applicable to affiliates, shares registered under any registration statements will be available for sale in the open market, beginning 90 days after the date of the prospectus, except to the extent that the shares are subject to vesting restrictions with us or the contractual restrictions described below.
Lock-up Agreements
We expect that all of our directors, executive officers and substantially all of the other holders of our equity securities will agree, subject to certain exceptions, not to offer, pledge, sell, contract to sell, sell any option or contract to purchase, purchase any option or contract to sell, grant any option, right or warrant to purchase, or otherwise transfer or dispose of, directly or indirectly, or enter into any swap or other arrangement that transfers to another, in whole or in part, any of the economic consequences of ownership of any shares of common stock or any securities convertible into or exercisable or exchangeable for shares of common stock for a period of 180 days after the date of this prospectus, without the prior written consent of the representatives of the underwriters. See the section titled “Underwriting.”
125
We are offering the shares of common stock described in this prospectus through a number of underwriters. J.P. Morgan Securities LLC and Merrill Lynch, Pierce, Fenner & Smith Incorporated are acting as joint book-running managers of the offering and as representatives of the underwriters. We will enter into an underwriting agreement with the underwriters. Subject to the terms and conditions of the underwriting agreement, we will agree to sell to the underwriters, and each underwriter will severally agree to purchase, at the public offering price less the underwriting discounts and commissions set forth on the cover page of this prospectus, the number of shares of common stock listed next to its name in the following table:
| Name |
Number of Shares | |
| J.P. Morgan Securities LLC |
||
| Merrill Lynch, Pierce, Fenner & Smith Incorporated |
||
| Wells Fargo Securities, LLC |
||
| Canaccord Genuity Inc. |
||
|
| ||
| Total |
||
|
|
The underwriters will be committed to purchase all the common shares offered by us if they purchase any shares. The underwriting agreement will also provide that if an underwriter defaults, the purchase commitments of non-defaulting underwriters may also be increased or the offering may be terminated.
The underwriters propose to offer the common shares directly to the public at the initial public offering price set forth on the cover page of this prospectus and to certain dealers at that price less a concession not in excess of $ per share. Any such dealers may resell shares to certain other brokers or dealers at a discount of up to $ per share from the initial public offering price. After the initial offering of the shares to the public, the offering price and other selling terms may be changed by the underwriters. Sales of shares made outside of the United States may be made by affiliates of the underwriters.
The underwriters will have an option to buy up to additional shares of common stock from us. The underwriters will have 30 days from the date of this prospectus to exercise this option to purchase additional shares. If any shares are purchased with this option to purchase additional shares, the underwriters will purchase shares in approximately the same proportion as shown in the table above. If any additional shares of common stock are purchased, the underwriters will offer the additional shares on the same terms as those on which the shares are being offered.
The underwriters do not expect to sell more than 5% of the common shares in the aggregate to accounts over which they exercise discretionary authority.
The underwriting fee is equal to the public offering price per share of common stock less the amount paid by the underwriters to us per share of common stock. The underwriting fee is $ per share. The following table shows the per share and total underwriting discounts and commissions to be paid to the underwriters assuming both no exercise and full exercise of the underwriters’ option to purchase additional shares.
| Without exercise of option to purchase additional shares |
With full exercise of option to purchase additional shares |
|||||||
| Per Share |
$ | $ | ||||||
| Total |
$ | $ | ||||||
Perella Weinberg Partners LP (Perella Weinberg), a Financial Industry Regulatory Association, Inc. (FINRA) member, is acting as our financial advisor in connection with the offering. We expect to pay Perella Weinberg, upon the successful completion of this offering, a fee of $1,250,000 for its services. The services provided to us by Perella Weinberg include, among other things, an independent financial valuation analysis; assisting in drafting our positioning and investment thesis; assisting us in our interactions with the underwriters; and
126
assisting us in crafting an appropriate aftermarket trading and investor relations strategy. Perella Weinberg will not sell or offer to sell any securities in this offering and will not identify, solicit or engage directly with potential investors in this offering. In addition, Perella Weinberg will not purchase any of the offered securities.
We estimate that the total expenses of this offering, including the fees of Perella Weinberg, registration, filing and listing fees, printing fees and legal and accounting expenses, but excluding the underwriting discounts and commissions, will be approximately $ . We have agreed to reimburse the underwriters for certain FINRA-related and other expenses incurred by them in connection with this offering in an amount up to $ . The underwriters have agreed to reimburse us for certain expenses we incur in connection with this offering up to $ .
A prospectus in electronic format may be made available on the web sites maintained by one or more underwriters, or selling group members, if any, participating in the offering. The underwriters may agree to allocate a number of shares to underwriters and selling group members for sale to their online brokerage account holders. Internet distributions will be allocated by the representatives to underwriters and selling group members that may make Internet distributions on the same basis as other allocations.
We will agree that, subject to limited exceptions, we will not (i) offer, pledge, sell, contract to sell, sell any option or contract to purchase, purchase any option or contract to sell, grant any option, right or warrant to purchase or otherwise dispose of, directly or indirectly, or file with the Securities and Exchange Commission a registration statement under the Securities Act relating to, any shares of our common stock or securities convertible into or exchangeable or exercisable for any shares of our common stock, or publicly disclose the intention to make any offer, sale, pledge, disposition or filing, or (ii) enter into any swap or other arrangement that transfers all or a portion of the economic consequences associated with the ownership of any shares of common stock or any such other securities (regardless of whether any of these transactions are to be settled by the delivery of shares of common stock or such other securities, in cash or otherwise), in each case without the prior written consent of J.P. Morgan Securities LLC and Merrill Lynch, Pierce, Fenner & Smith Incorporated for a period of 180 days after the date of this prospectus, other than the shares of our common stock to be sold hereunder and any shares of our common stock issued upon the exercise of options granted under our existing equity incentive plans.
Our directors and executive officers, and holders of substantially all of our outstanding shares have entered into or will enter into lock-up agreements with the underwriters prior to the commencement of this offering pursuant to which each of these persons or entities, with limited exceptions, for a period of 180 days after the date of this prospectus, may not, without the prior written consent of J.P. Morgan Securities LLC and Merrill Lynch, Pierce, Fenner & Smith Incorporated, (1) offer, pledge, sell, contract to sell, sell any option or contract to purchase, purchase any option or contract to sell, grant any option, right or warrant to purchase, or otherwise transfer or dispose of, directly or indirectly, any shares of our common stock or any securities convertible into or exercisable or exchangeable for our common stock (including, without limitation, common stock or such other securities which may be deemed to be beneficially owned by such directors, executive officers, or other stockholders in accordance with the rules and regulations of the SEC and securities which may be issued upon exercise of a stock option or warrant) or (2) enter into any swap or other agreement that transfers, in whole or in part, any of the economic consequences of ownership of the common stock or such other securities, whether any such transaction described in clause (1) or (2) above is to be settled by delivery of common stock or such other securities, in cash or otherwise, or (3) make any demand for or exercise any right with respect to the registration of any shares of our common stock or any security convertible into or exercisable or exchangeable for our common stock. The restrictions described above are subject to exceptions, including:
| • | open market transactions after the completion of this offering, provided that no filing under Section 16(a) of the Exchange Act, or other public announcement, shall be required or shall be made voluntarily in connection with any such transaction (other than a filing on a Form 5 made after the expiration of the restricted period); |
| • | transfers that occur by operation of law pursuant to a qualified domestic order or in connection with a divorce settlement, provided that any filing required to be made under Section 16(a) of the Exchange Act shall clearly indicate the purpose of the transfer in the footnotes thereto, no other public announcement shall be required or shall be made voluntarily in connection with such transfer and each transferee, |
127
| donee or distributee shall execute and deliver to the underwriters a lock-up agreement substantially in the form of the lock-up agreement entered into by the party subject to the lockup restrictions; |
| • | transfers to us pursuant to agreements under which we have the option to repurchase such shares or securities upon termination of service of the party subject to the lockup restrictions, provided that any public report or filing required to be made under Section 16(a) of the Exchange Act shall clearly indicate in the footnotes thereto the circumstances of such transfer and no other public announcement shall be required or shall be made voluntarily in connection with such transfer; |
| • | the exercise of stock options granted under a stock incentive plan or stock purchase plan described in this prospectus, provided that the shares received upon exercise shall continue to be subject to the restrictions on transfer set forth in the lock-up agreement and that any public report or filing required to be made under Section 16(a) of the Exchange Act shall clearly indicate in the footnotes thereto that the filing relates to the exercise of a stock option, that no shares were sold by the reporting person and that the shares received upon exercise of the stock option are subject to a lock-up agreement with the underwriters; |
| • | transfers to us upon the exercise of options to purchase our securities outstanding as of the date the party subject to the lockup restrictions entered into the lock-up agreement or pursuant to a stock incentive plan or stock purchase plan described in this prospectus, on a “cashless” or “net exercise” basis, provided that the shares received upon exercise shall continue to be subject to the restrictions on transfer set forth in the lock-up agreement and that any public report or filing required to be made under Section 16(a) of the Exchange Act shall clearly indicate in the footnotes thereto that the filing relates to the “cashless” or “net” exercise of a stock option, that no shares were sold by the reporting person and that the shares received upon exercise of the stock option are subject to a lock-up letter agreement with the underwriters; or |
| • | the establishment of a trading plan pursuant to Rule 10b5-1 under the Exchange Act for the transfer of shares of common stock, provided that (i) such plan does not provide for the transfer of common stock during the restricted period and (ii) no public announcement or filing shall be required or shall be made voluntarily in connection with the establishment of such plan during the restricted period. |
We will agree to indemnify the underwriters against certain liabilities, including liabilities under the Securities Act.
We will apply to have our common stock approved for listing on the NYSE under the symbol “PEN.”
In connection with this offering, the underwriters may engage in stabilizing transactions, which involves making bids for, purchasing and selling shares of common stock in the open market for the purpose of preventing or retarding a decline in the market price of the common stock while this offering is in progress. These stabilizing transactions may include making short sales of the common stock, which involves the sale by the underwriters of a greater number of shares of common stock than they are required to purchase in this offering, and purchasing shares of common stock on the open market to cover positions created by short sales. Short sales may be “covered” shorts, which are short positions in an amount not greater than the underwriters’ option to purchase additional shares referred to above, or may be “naked” shorts, which are short positions in excess of that amount. The underwriters may close out any covered short position either by exercising their option to purchase additional shares, in whole or in part, or by purchasing shares in the open market. In making this determination, the underwriters will consider, among other things, the price of shares available for purchase in the open market compared to the price at which the underwriters may purchase shares through the option to purchase additional shares. A naked short position is more likely to be created if the underwriters are concerned that there may be downward pressure on the price of the common stock in the open market that could adversely affect investors who purchase in this offering. To the extent that the underwriters create a naked short position, they will purchase shares in the open market to cover the position.
The underwriters have advised us that, pursuant to Regulation M of the Securities Act, they may also engage in other activities that stabilize, maintain or otherwise affect the price of the common stock, including the imposition of penalty bids. This means that if the representatives of the underwriters purchase common stock in the open market in stabilizing transactions or to cover short sales, the representatives can require the underwriters that sold those shares as part of this offering to repay the underwriting discount received by them.
128
These activities may have the effect of raising or maintaining the market price of the common stock or preventing or retarding a decline in the market price of the common stock, and, as a result, the price of the common stock may be higher than the price that otherwise might exist in the open market. If the underwriters commence these activities, they may discontinue them at any time. The underwriters may carry out these transactions on the or otherwise.
Prior to this offering, there has been no public market for our common stock. The initial public offering price will be determined by negotiations between us and the representatives of the underwriters. In determining the initial public offering price, we and the representatives of the underwriters expect to consider a number of factors including:
| • | the information set forth in this prospectus and otherwise available to the representatives; |
| • | our prospects and the history and prospects for the industry in which we compete; |
| • | an assessment of our management; |
| • | our prospects for future earnings; |
| • | the general condition of the securities markets at the time of this offering; |
| • | the recent market prices of, and demand for, publicly traded common stock of generally comparable companies; and |
| • | other factors deemed relevant by the underwriters and us. |
Neither we nor the underwriters can assure investors that an active trading market will develop for our common shares, or that the shares will trade in the public market at or above the initial public offering price.
An affiliate of Wells Fargo Securities, LLC, one of the underwriters for this offering, has provided us with commercial banking services in the past, for which it has received customary compensation. Certain of the underwriters or their affiliates may provide from time to time in the future certain commercial banking, financial advisory, investment banking and other services for us or our affiliates in the ordinary course of their business, for which they may receive customary fees and commissions. In addition, from time to time in the future, certain of the underwriters or their affiliates may effect transactions for their own account or the account of customers, and hold on behalf of themselves or their customers, long or short positions in our debt or equity securities or loans.
Selling Restrictions
General
Other than in the United States, no action has been taken by us or the underwriters that would permit a public offering of the securities offered by this prospectus in any jurisdiction where action for that purpose is required. The securities offered by this prospectus may not be offered or sold, directly or indirectly, nor may this prospectus or any other offering material or advertisements in connection with the offer and sale of any such securities be distributed or published in any jurisdiction, except under circumstances that will result in compliance with the applicable rules and regulations of that jurisdiction. Persons into whose possession this prospectus comes are advised to inform themselves about and to observe any restrictions relating to the offering and the distribution of this prospectus. This prospectus does not constitute an offer to sell or a solicitation of an offer to buy any securities offered by this prospectus in any jurisdiction in which such an offer or a solicitation is unlawful.
European Economic Area
In relation to each Member State of the European Economic Area (each, a Relevant Member State), no offer of shares may be made to the public in that Relevant Member State other than:
| A. | to any legal entity which is a qualified investor as defined in the Prospectus Directive; |
| B. | to fewer than 100 or, if the Relevant Member State has implemented the relevant provision of the 2010 PD Amending Directive, 150, natural or legal persons (other than qualified investors as defined |
129
| in the Prospectus Directive), as permitted under the Prospectus Directive, subject to obtaining the prior consent of the representatives; or |
| C. | in any other circumstances falling within Article 3(2) of the Prospectus Directive, |
provided that no such offer of shares shall require the Company or the representatives to publish a prospectus pursuant to Article 3 of the Prospectus Directive or supplement a prospectus pursuant to Article 16 of the Prospectus Directive.
Each person in a Relevant Member State who initially acquires any shares or to whom any offer is made will be deemed to have represented, acknowledged and agreed that it is a “qualified investor” within the meaning of the law in that Relevant Member State implementing Article 2(1)(e) of the Prospectus Directive. In the case of any shares being offered to a financial intermediary as that term is used in Article 3(2) of the Prospectus Directive, each such financial intermediary will be deemed to have represented, acknowledged and agreed that the shares acquired by it in the offer have not been acquired on a non-discretionary basis on behalf of, nor have they been acquired with a view to their offer or resale to, persons in circumstances which may give rise to an offer of any shares to the public other than their offer or resale in a Relevant Member State to qualified investors as so defined or in circumstances in which the prior consent of the representatives has been obtained to each such proposed offer or resale.
The Company, the representatives and their affiliates will rely upon the truth and accuracy of the foregoing representations, acknowledgements and agreements.
This prospectus has been prepared on the basis that any offer of shares in any Relevant Member State will be made pursuant to an exemption under the Prospectus Directive from the requirement to publish a prospectus for offers of shares. Accordingly any person making or intending to make an offer in that Relevant Member State of shares which are the subject of the offering contemplated in this prospectus may only do so in circumstances in which no obligation arises for the Company or any of the underwriters to publish a prospectus pursuant to Article 3 of the Prospectus Directive in relation to such offer. Neither the Company nor the underwriters have authorized, nor do they authorize, the making of any offer of shares in circumstances in which an obligation arises for the Company or the underwriters to publish a prospectus for such offer.
For the purpose of the above provisions, the expression “an offer to the public” in relation to any shares in any Relevant Member State means the communication in any form and by any means of sufficient information on the terms of the offer and the shares to be offered so as to enable an investor to decide to purchase or subscribe the shares, as the same may be varied in the Relevant Member State by any measure implementing the Prospectus Directive in the Relevant Member State and the expression “Prospectus Directive” means Directive 2003/71/EC (including the 2010 PD Amending Directive, to the extent implemented in the Relevant Member States) and includes any relevant implementing measure in the Relevant Member State and the expression “2010 PD Amending Directive” means Directive 2010/73/EU.
United Kingdom
In addition, in the United Kingdom, this document is being distributed only to, and is directed only at, and any offer subsequently made may only be directed at persons who are “qualified investors” (as defined in the Prospectus Directive) (i) who have professional experience in matters relating to investments falling within Article 19 (5) of the Financial Services and Markets Act 2000 (Financial Promotion) Order 2005, as amended (the “Order”) and/or (ii) who are high net worth companies (or persons to whom it may otherwise be lawfully communicated) falling within Article 49(2)(a) to (d) of the Order (all such persons together being referred to as “relevant persons”).
Any person in the United Kingdom that is not a relevant person should not act or rely on the information included in this document or use it as basis for taking any action. In the United Kingdom, any investment or investment activity that this document relates to may be made or taken exclusively by relevant persons. Any person in the United Kingdom that is not a relevant person should not act or rely on this document or any of its contents.
130
The validity of the issuance of the shares of common stock offered hereby will be passed upon for us by Davis Polk & Wardwell LLP. Goodwin Procter LLP is representing the underwriters.
The consolidated financial statements of Penumbra, Inc. and its subsidiaries as of December 31, 2013 and 2014, and for each of the two years ended December 31, 2014, included in this prospectus have been audited by Deloitte & Touche LLP, an independent registered public accounting firm, as stated in their report appearing herein. Such financial statements have been so included in reliance upon the report of such firm given upon their authority as experts in accounting and auditing.
131
WHERE YOU CAN FIND MORE INFORMATION
We have filed with the SEC a registration statement on Form S-1 under the Securities Act with respect to the common stock offered hereby. This prospectus does not contain all of the information set forth in the registration statement and the exhibits and schedules thereto. For further information with respect to the Company and its common stock, reference is made to the registration statement and the exhibits and any schedules filed therewith. Statements contained in this prospectus as to the contents of any contract or other document referred to are not necessarily complete and in each instance, if such contract or document is filed as an exhibit, reference is made to the copy of such contract or other document filed as an exhibit to the registration statement, each statement being qualified in all respects by such reference. A copy of the registration statement, including the exhibits and schedules thereto, may be read and copied at the SEC’s Public Reference Room at 100 F Street, N.E., Washington, D.C. 20549. Information on the operation of the Public Reference Room may be obtained by calling the SEC at 1-800-SEC-0330. In addition, the SEC maintains an Internet site at www.sec.gov, from which interested persons can electronically access the registration statement, including the exhibits and any schedules thereto.
As a result of the offering, we will be required to file periodic reports and other information with the SEC. We also maintain an Internet site at www.penumbrainc.com. Our website and the information contained therein or accessible therefrom shall not be deemed to be incorporated into this prospectus or the registration statement of which it forms a part.
132
INDEX TO CONSOLIDATED FINANCIAL STATEMENTS
Penumbra, Inc.
| Page | ||||
| F-2 | ||||
| F-3 | ||||
| Consolidated Statements of Operations and Comprehensive Income |
F-4 | |||
| Consolidated Statements of Preferred Stock and Stockholders’ Deficit |
F-5 | |||
| F-6 | ||||
| F-7 | ||||
F-1
Report of Independent Registered Public Accounting Firm
To the Board of Directors and Stockholders of
Penumbra, Inc.
Alameda, California
We have audited the accompanying consolidated balance sheets of Penumbra, Inc. and subsidiaries (the “Company”) as of December 31, 2013 and 2014, and the related consolidated statements of operations and comprehensive income, preferred stock and stockholders’ deficit, and cash flows for each of the two years in the period ended December 31, 2014. These consolidated financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the financial statements based on our audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. Our audits included consideration of internal control over financial reporting as a basis for designing audit procedures that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion. An audit also includes examining, on a test basis, evidence supporting the amounts and disclosures in the consolidated financial statements, assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement presentation. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, such consolidated financial statements present fairly, in all material respects, the consolidated financial position of the Company as of December 31, 2013 and 2014, and the consolidated results of its operations and its cash flows for each of the two years in the period ended December 31, 2014, in conformity with accounting principles generally accepted in the United States of America.
/s/ DELOITTE & TOUCHE LLP
San Francisco, California
June 9, 2015
F-2
CONSOLIDATED BALANCE SHEETS
(in thousands, except share and per share amounts)
| December 31, 2013 |
December 31, 2014 |
June 30, 2015 |
Pro Forma Stockholders’ Equity at June 30, 2015 (See Note 2) |
|||||||||||||
| (unaudited) | ||||||||||||||||
| Assets |
||||||||||||||||
| Current assets: |
||||||||||||||||
| Cash and cash equivalents |
$ | 4,131 | $ | 3,290 | $ | 36,764 | ||||||||||
| Marketable investments |
9,545 | 48,253 | — | |||||||||||||
| Accounts receivable, net of doubtful accounts of $471, $602 and $501 at December 31, 2013 and 2014 and June 30, 2015 (unaudited), respectively |
13,074 | 18,912 | 23,604 | |||||||||||||
| Inventories |
27,067 | 33,451 | 44,179 | |||||||||||||
| Deferred taxes |
5,134 | 6,280 | 5.720 | |||||||||||||
| Prepaid expenses and other current assets |
3,287 | 5,115 | 7,920 | |||||||||||||
|
|
|
|
|
|
|
|||||||||||
| Total current assets |
62,238 | 115,301 | 118,187 | |||||||||||||
| Property and equipment, net |
2,031 | 5,181 | 7,976 | |||||||||||||
| Restricted investments |
6,000 | — | — | |||||||||||||
| Deferred taxes |
424 | 571 | 1,039 | |||||||||||||
| Other non-current assets |
454 | 328 | 1,868 | |||||||||||||
|
|
|
|
|
|
|
|||||||||||
| Total assets |
$ | 71,147 | $ | 121,381 | $ | 129,070 | ||||||||||
|
|
|
|
|
|
|
|||||||||||
| Liabilities, Preferred Stock and Stockholders’ Deficit |
||||||||||||||||
| Current liabilities: |
||||||||||||||||
| Accounts payable |
$ | 1,312 | $ | 2,348 | $ | 3,721 | ||||||||||
| Accrued liabilities |
14,525 | 18,475 | 23,168 | |||||||||||||
|
|
|
|
|
|
|
|||||||||||
| Total current liabilities |
15,837 | 20,823 | 26,889 | |||||||||||||
| Credit facility |
6,000 | — | — | |||||||||||||
| Other non-current liabilities |
1,150 | 1,461 | 1,813 | |||||||||||||
|
|
|
|
|
|
|
|||||||||||
| Total liabilities |
22,987 | 22,284 | 28,702 | |||||||||||||
| Commitments and contingencies (Note 7) |
||||||||||||||||
| Preferred stock, $0.001 par value—25,000,000, 25,000,000 and 25,000,000 shares authorized at December 31, 2013 and 2014 and June 30, 2015 (unaudited) respectively; 15,594,618, 19,510,410 and 19,510,410 shares issued and outstanding at December 31, 2013 and 2014 and June 30, 2015 (unaudited), respectively; aggregate liquidation value $85,218, $149,361, and $153,738 at December 31, 2013 and 2014 and June 30, 2015 (unaudited), respectively; shares authorized, no shares issued or outstanding, pro forma (unaudited) |
56,222 | 111,467 | 111,467 | — | ||||||||||||
| Stockholders’ deficit: |
||||||||||||||||
| Common stock, $0.001 par value—40,000,000, 40,000,000 and 40,000,000 shares authorized at December 31, 2013, 2014 and June 30, 2015 (unaudited), respectively; 4,391,591, 4,736,689 and 5,747,638 shares issued and outstanding at December 31, 2013, 2014 and June 30, 2015 (unaudited), respectively; shares authorized, shares issued and outstanding pro forma (unaudited) |
4 | 5 | 6 | 25 | ||||||||||||
| Additional paid-in capital |
6,269 | 8,446 | 10,169 | 121,617 | ||||||||||||
| Notes receivable from stockholders |
(138 | ) | (117 | ) | (32 | ) | (32 | ) | ||||||||
| Accumulated other comprehensive income/(loss) |
796 | (864 | ) | (1,233 | ) | (1,233 | ) | |||||||||
| Accumulated deficit |
(14,993 | ) | (19,840 | ) | (20,009 | ) | (20,009 | ) | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total stockholders’ deficit |
(8,062 | ) | (12,370 | ) | (11,099 | ) | 100,368 | |||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total liabilities, preferred stock and stockholders’ deficit |
$ | 71,147 | $ | 121,381 | $ | 129,070 | ||||||||||
|
|
|
|
|
|
|
|||||||||||
The accompanying notes are an integral part of these financial statements.
F-3
CONSOLIDATED STATEMENTS OF OPERATIONS AND COMPREHENSIVE INCOME
(in thousands, except share and per share amounts)
| Year Ended December 31, | Six Months Ended June 30, | |||||||||||||||
| 2013 | 2014 | 2014 | 2015 | |||||||||||||
| (unaudited) | ||||||||||||||||
| Revenue |
$ | 88,848 | $ | 125,510 | $ | 57,643 | $ | 81,263 | ||||||||
| Cost of revenue |
30,972 | 42,668 | 19,489 | 27,160 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Gross profit |
57,876 | 82,842 | 38,154 | 54,103 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Operating expenses: |
||||||||||||||||
| Research and development |
14,084 | 15,575 | 7,538 | 7,983 | ||||||||||||
| Sales, general and administrative |
44,918 | 64,258 | 28,240 | 45,943 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total operating expenses |
59,002 | 79,833 | 35,778 | 53,926 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Income (loss) from operations |
(1,126 | ) | 3,009 | 2,376 | 177 | |||||||||||
| Interest income (expense), net |
345 | 439 | 39 | 385 | ||||||||||||
| Other income (expense), net |
(474 | ) | (309 | ) | (92 | ) | (498 | ) | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Income (loss) before provision for (benefit from) income taxes |
(1,255 | ) | 3,139 | 2,323 | 64 | |||||||||||
| Provision for (benefit from) income taxes |
(5,354 | ) | 894 | 666 | 233 | |||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Net income (loss) |
4,099 | 2,245 | 1,657 | (169 | ) | |||||||||||
| Foreign currency translation adjustments, net of tax |
(522 | ) | (1,423 | ) | (89 | ) | (589 | ) | ||||||||
| Unrealized gains (losses) on available-for-sale securities, net of tax |
(84 | ) | (237 | ) | 174 | 220 | ||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Comprehensive income (loss) |
$ | 3,493 | $ | 585 | $ | 1,742 | $ | (538 | ) | |||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Net income (loss) attributable to common stockholders (Note 16) |
$ | 887 | $ | (833 | ) | $ | 355 | $ | (34 | ) | ||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Net income (loss) per share attributable to common stockholders |
||||||||||||||||
| Basic |
$ | 0.21 | $ | (0.18 | ) | $ | 0.08 | $ | (0.01 | ) | ||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Diluted |
$ | 0.14 | $ | (0.18 | ) | $ | 0.05 | $ | (0.01 | ) | ||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Weighted average shares used to compute net income (loss) per share attributable to common stockholders |
||||||||||||||||
| Basic |
4,304,396 | 4,609,375 | 4,520,898 | 5,000,375 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Diluted |
6,500,835 | 4,609,375 | 6,743,140 | 5,000,375 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Pro forma net income (loss) per share (unaudited) (Note 16) |
||||||||||||||||
| Basic |
$ | 0.10 | $ | (0.01 | ) | |||||||||||
|
|
|
|
|
|||||||||||||
| Diluted |
$ | 0.09 | $ | (0.01 | ) | |||||||||||
|
|
|
|
|
|||||||||||||
| Weighted average shares used to compute pro forma net income (loss) per share (unaudited) (Note 16) |
||||||||||||||||
| Basic |
22,680,810 | 24,510,785 | ||||||||||||||
|
|
|
|
|
|||||||||||||
| Diluted |
25,037,541 | 24,510,785 | ||||||||||||||
|
|
|
|
|
|||||||||||||
The accompanying notes are an integral part of these financial statements.
F-4
CONSOLIDATED STATEMENTS OF PREFERRED STOCK AND STOCKHOLDERS’ DEFICIT
(in thousands, except share amounts)
| Additional Paid-in Capital |
Notes Receivable from Stockholders |
Accumulated Other Comprehensive Income/ (Loss) |
Accumulated Deficit |
Total Stockholders’ Deficit |
||||||||||||||||||||||||||||||||||
| Preferred Stock | Common Stock | |||||||||||||||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | |||||||||||||||||||||||||||||||||||
| Balance at December 31, 2012 |
15,594,618 | $ | 56,222 | 4,192,745 | $ | 4 | $ | 5,073 | $ | (138 | ) | $ | 1,402 | $ | (19,092 | ) | $ | (12,751 | ) | |||||||||||||||||||
| Issuance of common stock |
— | — | 198,843 | — | 310 | — | — | — | 310 | |||||||||||||||||||||||||||||
| Stock-based compensation |
— | — | — | — | 886 | — | — | — | 886 | |||||||||||||||||||||||||||||
| Foreign currency translation adjustment, net of tax of $213 |
— | — | — | — | — | — | (522 | ) | — | (522 | ) | |||||||||||||||||||||||||||
| Unrealized loss on investments, net of tax of $10 |
— | — | — | — | — | — | (84 | ) | — | (84 | ) | |||||||||||||||||||||||||||
| Net income |
— | — | — | — | — | — | — | 4,099 | 4,099 | |||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||
| Balance at December 31, 2013 |
15,594,618 | 56,222 | 4,391,588 | 4 | 6,269 | (138 | ) | 796 | (14,993 | ) | (8,062 | ) | ||||||||||||||||||||||||||
| Issuance of Series F preferred stock, net of issuance cost of $2,788 |
4,545,455 | 57,212 | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||
| Repurchase of preferred stock |
(629,663 | ) | (1,967 | ) | — | — | — | — | — | (6,344 | ) | (6,344 | ) | |||||||||||||||||||||||||
| Repurchase of common stock |
— | — | (115,612 | ) | (292 | ) | (748 | ) | (1,040 | ) | ||||||||||||||||||||||||||||
| Issuance of common stock |
— | — | 460,713 | 1 | 1,036 | — | — | — | 1,037 | |||||||||||||||||||||||||||||
| Stock-based compensation |
— | — | — | — | 1,433 | — | — | — | 1,433 | |||||||||||||||||||||||||||||
| Forgiven notes receivable from stockholders |
— | — | — | — | — | 21 | — | — | 21 | |||||||||||||||||||||||||||||
| Foreign currency translation adjustment, net of tax of $245 |
— | — | — | — | — | — | (1,423 | ) | — | (1,423 | ) | |||||||||||||||||||||||||||
| Unrealized loss on investments, net of tax of $168 |
— | — | — | — | — | — | (237 | ) | — | (237 | ) | |||||||||||||||||||||||||||
| Net income |
— | — | — | — | — | — | — | 2,245 | 2,245 | |||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||
| Balance at December 31, 2014 |
19,510,410 | 111,467 | 4,736,689 | 5 | 8,446 | (117 | ) | (864 | ) | (19,840 | ) | (12,370 | ) | |||||||||||||||||||||||||
| Issuance of common stock (unaudited) |
— | — | 1,034,599 | 1 | 974 | — | — | — | 975 | |||||||||||||||||||||||||||||
| Shares held for tax withholdings (unaudited) |
— | — | — | — | (2,525 | ) | — | — | — | (2,525 | ) | |||||||||||||||||||||||||||
| Repurchase of common stock (unaudited) |
— | — | (23,650 | ) | — | (342 | ) | — | — | — | (342 | ) | ||||||||||||||||||||||||||
| Stock-based compensation (unaudited) |
— | — | — | — | 3,616 | — | — | — | 3,616 | |||||||||||||||||||||||||||||
| Forgiven notes receivable from stockholders (unaudited) |
— | — | — | — | — | 85 | — | — | 85 | |||||||||||||||||||||||||||||
| Foreign currency translation adjustment, net of tax of $92 (unaudited) |
— | — | — | — | — | — | (589 | ) | — | (589 | ) | |||||||||||||||||||||||||||
| Unrealized gain on investments, net of tax of $ 159 (unaudited) |
— | — | — | — | — | — | 220 | — | 220 | |||||||||||||||||||||||||||||
| Net loss (unaudited) |
— | — | — | — | — | — | — | (169 | ) | (169 | ) | |||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||
| Balance at June 30, 2015 (unaudited) |
19,510,410 | $ | 111,467 | 5,747,638 | $ | 6 | $ | 10,169 | $ | (32 | ) | $ | (1,233 | ) | $ | (20,009 | ) | $ | (11,099 | ) | ||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||
The accompanying notes are an integral part of these financial statements.
F-5
CONSOLIDATED STATEMENTS OF CASH FLOWS
(in thousands)
| Year Ended December 31, |
Six Months Ended June 30, |
|||||||||||||||
| 2013 | 2014 | 2014 | 2015 | |||||||||||||
| (unaudited) | ||||||||||||||||
| CASH FLOWS FROM OPERATING ACTIVITIES: |
||||||||||||||||
| Net income (loss) |
$ | 4,099 | $ | 2,245 | $ | 1,657 | $ | (169 | ) | |||||||
| Adjustments to reconcile net income (loss) to net cash provided by (used in) operating activities: |
||||||||||||||||
| Depreciation and amortization |
677 | 751 | 328 | 745 | ||||||||||||
| Stock-based compensation |
886 | 1,433 | 702 | 3,616 | ||||||||||||
| Provision for doubtful accounts |
259 | 150 | 166 | (101 | ) | |||||||||||
| Inventory write downs |
892 | 1,852 | 481 | 304 | ||||||||||||
| Write off of note receivable |
— | 21 | — | 85 | ||||||||||||
| Provision for sales returns |
(212 | ) | (3 | ) | 79 | 223 | ||||||||||
| Loss on minority investment |
— | 150 | — | — | ||||||||||||
| Loss on disposal of property and equipment |
3 | 21 | 8 | 12 | ||||||||||||
| Realized loss on marketable investments |
160 | — | — | 541 | ||||||||||||
| Provision for product warranty |
— | (8 | ) | 17 | 176 | |||||||||||
| Release of valuation allowance |
(4,962 | ) | (321 | ) | — | — | ||||||||||
| Deferred taxes |
(335 | ) | (571 | ) | (107 | ) | — | |||||||||
| Issuance of common stock to third parties |
132 | — | — | — | ||||||||||||
| Changes in operating assets and liabilities: |
||||||||||||||||
| Accounts receivable |
(1,550 | ) | (7,426 | ) | (3,150 | ) | (4,940 | ) | ||||||||
| Inventories |
(3,779 | ) | (9,444 | ) | (2,864 | ) | (11,321 | ) | ||||||||
| Prepaid expenses and other current and non-current assets |
(961 | ) | (1,906 | ) | (235 | ) | (2,759 | ) | ||||||||
| Accounts payable |
(451 | ) | 1,299 | 1,237 | 548 | |||||||||||
| Accrued expenses and other non-current liabilities |
1,746 | 5,368 | 3,673 | 4,709 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Net cash provided by (used in) operating activities |
(3,396 | ) | (6,389 | ) | 1,992 | (8,331 | ) | |||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| CASH FLOWS FROM INVESTING ACTIVITIES: |
||||||||||||||||
| Purchase of marketable investments |
(6,858 | ) | (51,342 | ) | (4,428 | ) | (4,069 | ) | ||||||||
| Proceeds from sales of marketable investments |
6,405 | 18,229 | 4,306 | 52,160 | ||||||||||||
| Purchases of property and equipment |
(798 | ) | (3,888 | ) | (818 | ) | (3,073 | ) | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Net cash provided by (used in) investing activities |
(1,251 | ) | (37,001 | ) | (940 | ) | 45,018 | |||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| CASH FLOWS FROM FINANCING ACTIVITIES: |
||||||||||||||||
| Proceeds from issuance of preferred stock, net of issuance costs |
— | 57,212 | 57,112 | — | ||||||||||||
| Payments of deferred issuance costs |
— | — | — | (964 | ) | |||||||||||
| Proceeds from exercises of stock options |
178 | 1,036 | 264 | 515 | ||||||||||||
| Repurchase of preferred stock |
— | (8,311 | ) | — | — | |||||||||||
| Proceeds from credit facility |
2,000 | — | — | — | ||||||||||||
| Repayment of credit facility |
— | (6,000 | ) | (6,000 | ) | |||||||||||
| Repurchase of common stock and stock options |
— | (1,040 | ) | (881 | ) | — | ||||||||||
| Payment of employee taxes related to vested common and restricted stock |
— | — | — | (2,525 | ) | |||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Net cash provided by (used in) financing activities |
2,178 | 42,897 | 50,495 | (2,974 | ) | |||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Effect of foreign exchange rate changes on cash and cash equivalents |
(835 | ) | (348 | ) | (118 | ) | (239 | ) | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Net Increase (Decrease) In Cash And Cash Equivalents |
(3,304 | ) | (841 | ) | 51,429 | 33,474 | ||||||||||
| CASH AND CASH EQUIVALENTS—Beginning of period |
7,435 | 4,131 | 4,131 | 3,290 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| CASH AND CASH EQUIVALENTS—End of period |
$ | 4,131 | $ | 3,290 | $ | 55,560 | $ | 36,764 | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| SUPPLEMENTAL DISCLOSURES OF CASH FLOW INFORMATION: |
||||||||||||||||
| Cash paid for interest |
$ | 100 | $ | 160 | $ | 160 | $ | — | ||||||||
| Cash paid for income taxes |
$ | 37 | $ | 3,086 | $ | 107 | $ | 80 | ||||||||
| NONCASH INVESTING AND FINANCING ACTIVITIES: |
||||||||||||||||
| Purchase of property and equipment funded through accounts payable |
$ | 5 | $ | 44 | $ | — | $ | 483 | ||||||||
| Deferred issuance costs not yet paid |
$ | — | $ | — | $ | — | $ | 620 | ||||||||
The accompanying notes are an integral part of these financial statements.
F-6
Notes to Consolidated Financial Statements
1. Organization and Description of Business
Penumbra, Inc. (the “Company”) is a global interventional therapies company that designs, develops, manufactures and markets innovative medical devices. The Company has a broad portfolio of products that addresses challenging medical conditions and significant clinical needs across two major markets, neuro and peripheral vascular. The conditions that the Company’s products address include, among others, ischemic stroke, hemorrhagic stroke and various peripheral vascular conditions that can be treated through thrombectomy and embolization procedures.
2. Summary of Significant Accounting Policies
Basis of Presentation and Consolidation
The accompanying consolidated financial statements have been prepared in accordance with accounting principles generally accepted in the United States (U.S. GAAP). The consolidated financial statements include the accounts of the Company and its wholly-owned subsidiaries. All intercompany accounts and transactions have been eliminated in consolidation. Subsequent events were evaluated from the balance sheet dates of December 31, 2014 and June 30, 2015 (unaudited) through June 9, 2015 and August 14, 2015 (unaudited).
Use of Estimates
The preparation of financial statements in conformity with U.S. GAAP requires management to make estimates and assumptions that affect the reported amounts of assets, liabilities and equity accounts; disclosure of contingent assets and liabilities at the date of the financial statements; and the reported amounts of revenues and expenses during the reporting period. On an ongoing basis, the Company evaluates its estimates, including those related to provisions for doubtful accounts, sales return reserve, warranty reserves, valuation of inventories, useful lives of property and equipment, income taxes, the valuation of equity instruments and contingencies, among others. The Company bases its estimates on historical experience and on various other assumptions that are believed to be reasonable under the circumstances, the results of which form the basis for making judgments about the carrying values of assets and liabilities that are not readily apparent from other data. Actual results could differ from those estimates.
Unaudited Interim Financial Statements
The accompanying interim consolidated financial statements as of June 30, 2015 and for the six months ended June 30, 2014 and 2015 and the related interim information contained within the notes to the financial statements are unaudited. The unaudited interim consolidated financial statements have been prepared in accordance with U.S. GAAP for interim financial information and on the same basis as the audited financial statements. In the opinion of management, the accompanying unaudited interim consolidated financial statements contain all adjustments which are necessary to present fairly the Company’s financial position as of June 30, 2015, and the results of its operations and cash flows for the six months ended June 30, 2014 and 2015. Such adjustments are of a normal and recurring nature. The results for the six months ended June 30, 2015 are not necessarily indicative of the results to be expected for the year ending December 31, 2015 or for any future period.
Unaudited Pro Forma Stockholders’ Equity
The unaudited pro forma stockholders’ equity information in the accompanying consolidated balance sheet reflects that there are 25,258,048 shares of common stock outstanding and assumes the automatic
F-7
Penumbra, Inc.
Notes to Consolidated Financial Statements (Continued)
2. Summary of Significant Accounting Policies (Continued)
conversion of all outstanding shares of preferred stock into shares of common stock in connection with the Company’s proposed initial public offering (IPO). The unaudited pro forma stockholders’ equity does not assume any proceeds from the proposed IPO.
Segments
The Company determined its operating segment on the same basis that it uses to evaluate its performance internally. The Company has one business activity: the design, development, manufacturing and marketing of innovative medical devices, and operates as one operating segment. The Company’s chief operating decision-maker, its Chief Executive Officer, reviews its operating results for the purpose of allocating resources and evaluating financial performance. The Company determines revenue by geographic area, based on the destination to which it ships its products.
Foreign Currency Translation
The Company’s consolidated financial statements are prepared in United States Dollars (USD). Its foreign subsidiaries use their local currency as their functional currency and maintain their records in the local currency. Accordingly, the assets and liabilities of these subsidiaries are translated into USD using the current exchange rates in effect at the balance sheet date and equity accounts are translated into USD using historical rates. Revenues and expenses are translated using the average exchange rates in effect. The resulting foreign currency translation adjustments are recorded in other comprehensive income in the consolidated balance sheets. Transactions denominated in foreign currency are translated at exchange rates at the date of transaction with foreign currency gains (losses) recorded in other income (expense), net in the consolidated statements of operations and other comprehensive income. The Company recognized net foreign currency transaction gains (losses) of $(0.3) million, $(0.3) million, $(49,000) (unaudited) and $0.1 million (unaudited) during the year ended December 31, 2013 and 2014 and the six months ended June 30, 2014 (unaudited) and 2015 (unaudited), respectively.
As the Company’s international operations grow, its risks associated with fluctuation in currency rates will become greater, and the Company will continue to reassess its approach to managing this risk. In addition, currency fluctuations or a weakening USD can increase the costs of the Company’s international expansion. To date, the Company has not entered into any foreign currency hedging contracts, since exchange rate fluctuations have not had a material impact on its operating results and cash flows.
Concentration of Credit Risk
Financial instruments that potentially subject the Company to a concentration of credit risk consist of cash and cash equivalents, marketable investments and accounts receivable. The majority of the Company’s cash is held by one financial institution in the United States in excess of federally insured limits. The Company maintained investments in money market funds that were not federally insured during the year ended December 31, 2014 and held cash in foreign banks of approximately $0.6 million, $0.8 million and $3.0 million (unaudited) at December 31, 2013 and 2014 and June 30, 2015 (unaudited), respectively, that was not federally insured. The Company has not experienced any losses on its deposits of cash and cash equivalents.
All of the Company’s revenue has been derived from sales of its products in the United States and international markets. The Company uses both its own salesforce and independent distributors to sell its products. Concentrations of credit risk with respect to accounts receivable are limited due to the large number of entities comprising the Company’s customer base. The Company performs ongoing credit evaluations of its
F-8
Penumbra, Inc.
Notes to Consolidated Financial Statements (Continued)
2. Summary of Significant Accounting Policies (Continued)
customers, including its distributors, does not require collateral, and maintains allowances for potential credit losses on customer accounts when deemed necessary.
During the years ended December 31, 2013 and 2014 and the six months ended June 30, 2014 (unaudited) and 2015 (unaudited), one customer (a distributor) accounted for 14%, 12%, 13% and 11%, respectively, of the Company’s revenue. No customer accounted for greater than 10% of the Company’s accounts receivable balance as of December 31, 2013, 2014 or June 30, 2015 (unaudited).
Significant Risks and Uncertainties
The Company is subject to risks common to medical device companies including, but not limited to, new technological innovations, dependence on key personnel, protection of proprietary technology, compliance with government regulations, product liability, uncertainty of market acceptance of products and the potential need to obtain additional financing. The Company is dependent on third party suppliers, in some cases single-source suppliers.
There can be no assurance that the Company’s products will continue to be accepted in the marketplace, nor can there be any assurance that any future products can be developed or manufactured at an acceptable cost and with appropriate performance characteristics, or that such products will be successfully marketed, if at all.
The Company’s products require approval or clearance from the U.S. Food and Drug Administration prior to commencing commercial sales in the United States. There can be no assurance that the Company’s products will receive all of the required approvals or clearances. Approvals or clearances are also required in foreign jurisdictions in which the Company sells its products. If the Company is denied such approvals or clearances or such approvals or clearances are delayed, it may have a material adverse impact on the Company’s results of operations, financial position and liquidity.
Fair Value of Financial Instruments
Carrying amounts of certain of the Company’s financial instruments, including cash equivalents, accounts receivable, accounts payable and accrued liabilities, approximate fair value due to their relatively short maturities.
Cash, Cash Equivalents and Marketable Investments
The Company invests its cash primarily in money market funds and in highly liquid debt instruments of U.S. federal and municipal governments and their agencies and corporate debt securities. All highly liquid investments with stated maturities of three months or less from the date of purchase are classified as cash equivalents; all highly liquid investments with stated maturities of greater than three months are classified as marketable investments. The majority of the Company’s cash and investments are held in U.S. banks and its foreign subsidiaries maintain a limited amount of cash in their local banks to cover their short term operating expenses.
The Company determines the appropriate classification of its investments in marketable investments at the time of purchase and re-evaluates such designation at each balance sheet date. The Company’s marketable investments have been classified and accounted for as available-for-sale. Investments with remaining maturities of more than one year are viewed by the Company as available to support current operations and are classified as current assets under the caption marketable investments in the accompanying consolidated balance sheets. Investments in marketable investments are carried at fair value, with the unrealized gains and losses reported as a component of accumulated other comprehensive income. Any realized gains or losses on
F-9
Penumbra, Inc.
Notes to Consolidated Financial Statements (Continued)
2. Summary of Significant Accounting Policies (Continued)
the sale of marketable investments are determined on a specific identification method, and such gains and losses are reflected as a component of other income (expense), net.
Impairment of Marketable Investments
After determining the fair value of available-for-sales debt instruments, gains or losses on these securities are recorded to accumulated other comprehensive income until either the security is sold or the Company determines that the decline in value is other-than-temporary. The primary differentiating factors that the Company considers in classifying impairments as either temporary or other-than-temporary impairments is the intent and ability to retain the investment in the issuer for a period of time sufficient to allow for any anticipated recovery in market value, the length of the time and the extent to which the market value of the investment has been less than cost, the financial condition and near-term prospects of the issuer. There were no other-than-temporary impairments for the years ended December 31, 2013 and 2014 and for the six months ended June 30, 2014 (unaudited) and 2015 (unaudited). The Company did not have any marketable investments as of June 30, 2015 (unaudited).
Accounts Receivable
Accounts receivable are stated at invoice value less estimated allowances for doubtful accounts. The Company continually monitors customer payments and maintains a reserve for estimated losses resulting from its customers’ inability to make required payments. The Company considers factors such as historical experience, credit quality, age of the accounts receivable balances, geographic related risks and economic conditions that may affect a customer’s ability to pay. In cases where there are circumstances that may impair a specific customer’s ability to meet its financial obligations, a specific allowance is recorded against amounts due, and thereby reduces the net recognized receivable to the amount reasonably believed to be collectible.
Restricted Investments
Restricted investments of $6.0 million as of December 31, 2013, classified as available-for-sale investments in money market funds, are held with a major financial institution as security for the Company’s credit facility. Refer to Note 5. There are no restricted investments as of December 31, 2014 or June 30, 2015 (unaudited).
Inventories
Inventories are stated at the lower of cost (determined under the first-in first-out method) or market. Write downs are provided for raw materials, components or finished goods that are determined to be excessive or obsolete. Market value is determined as the lower of replacement cost or net realizable value. The Company regularly reviews inventory quantities in consideration of actual loss experience, projected future demand and remaining shelf life to record a provision for excess and obsolete inventory when appropriate.
The estimate of excess quantities is subjective and primarily dependent on the Company’s estimates of future demand for a particular product or components or raw materials used in the manufacturing of such product. If the estimate of future demand is inaccurate based on actual sales, the Company may increase the write down for excess inventory and record a charge to inventory impairment in the accompanying consolidated statements of operations and comprehensive income. The Company periodically evaluates the carrying value of inventory on hand for potential excess amounts over demand using the same lower of cost or market approach as that has been used to value the inventory. The Company also periodically evaluates inventory quantities in consideration of actual loss experience. As a result of these evaluations, the Company
F-10
Penumbra, Inc.
Notes to Consolidated Financial Statements (Continued)
2. Summary of Significant Accounting Policies (Continued)
recognized total write downs of $0.9 million and $1.9 million for the years ended December 31, 2013 and 2014, respectively, and $0.5 million and $0.3 million for the six months ended June 30, 2014 (unaudited) and 2015 (unaudited).
Property and Equipment, net
Property and equipment are stated at cost less accumulated depreciation. Depreciation is computed using the straight-line method over five years, which is the estimated useful lives of the assets. Leasehold improvements are amortized using the straight-line method over the shorter of the lease term or estimated useful life. Upon retirement or sale, the cost and the related accumulated depreciation are removed from the consolidated balance sheet and the resulting gain or loss is reflected in operations. Maintenance and repairs are charged to operations as incurred.
Impairment of Long-Lived Assets
The Company reviews long-lived assets for impairment whenever events or changes in circumstances indicate that the carrying amount of an asset may not be recoverable. When such an event occurs, management determines whether there has been impairment by comparing the anticipated undiscounted future net cash flows to the related asset group’s carrying value. If an asset is considered impaired, the asset is written down to fair value, which is determined based either on discounted cash flows or appraised value, depending on the nature of the asset. There was no impairment of long-lived assets during the year ended December 31, 2013 and 2014 and for the six months ended June 30, 2014 (unaudited) and 2015 (unaudited).
Preferred Stock
The Company has classified the preferred stock as temporary equity in the consolidated balance sheets due to the existence of certain change in control events that are not solely within the Company’s control, including liquidation, sale or transfer of the Company, that trigger the ability of the holders of the preferred stock to call for redemption of shares.
Revenue Recognition
Revenue is comprised of product revenue net of returns, discounts, administration fees and sales rebates. The Company recognizes revenue when persuasive evidence of an arrangement exists, delivery has occurred, the sales price is fixed or determinable and collectability is reasonably assured. Evidence of an arrangement consists of customer orders and the Company typically considers delivery to have occurred once title and risk of loss has been transferred and the product has been delivered to the customer. The Company typically recognizes revenue when products are delivered to hospital customers or distributors. However, with respect to products that the Company consigns to hospitals, which primarily consist of coils, the Company recognizes revenue at the time hospitals utilize products in a procedure.
Deferred revenue represents amounts that the Company has already invoiced its customers and are ultimately expected to be recognized as revenue, but for which not all revenue recognition criteria have been met. The Company had a deferred revenue balance of $1.1 million, $1.6 million and $0.8 million, as of December 31, 2013, 2014 and June 30, 2015 (unaudited), respectively.
The Company’s terms and conditions permit product returns and exchanges, and it records returns reserves in the period when revenue is recognized. Estimates are based on actual historical returns over the prior three years and are recorded as reductions in revenue at the time of sale. Upon recognition, the Company
F-11
Penumbra, Inc.
Notes to Consolidated Financial Statements (Continued)
2. Summary of Significant Accounting Policies (Continued)
reduces revenue and cost of revenue for the estimated return. Return rates can fluctuate over time, but are sufficiently predictable to allow the Company to estimate expected future product returns.
Cost of Revenue
Cost of revenue includes direct and indirect costs associated with the manufacture of the Company’s products. Direct costs include material and labor, while indirect costs include inbound freight charges, receiving costs, inspection and testing costs, warehousing costs, royalty expense and other labor and overhead costs incurred in the manufacturing of products. Cost of revenue also includes stock-based compensation, warranty replacement costs, cost of revenue related to product return reserves and excess and obsolete inventory write-downs.
Shipping Costs
Shipping and handling costs charged to customers are recorded as revenue. Shipping and handling costs are included in cost of revenue.
Research and Development (R&D) Expenses
R&D costs primarily consist of product development, clinical and regulatory expenses, materials, depreciation and other costs associated with the development of the Company’s products. R&D costs also include related personnel and consultants’ salaries, benefits and related costs, including stock-based compensation. The Company expenses R&D costs as they are incurred.
The Company’s clinical trial accruals are based on estimates of patient enrollment and related costs at clinical investigator sites. The Company estimates preclinical and clinical trial expenses based on the services performed pursuant to contracts with research institutions and clinical research organizations that conduct and manage preclinical studies and clinical trials on its behalf. In accruing service fees, the Company estimates the time period over which services will be performed and the level of patient enrollment and activity expended in each period. If the actual timing of the performance of services or the level of effort varies from the estimate, the Company will adjust the accrual accordingly. Payments made to third parties under these arrangements in advance of the receipt of the related services are recorded as prepaid expenses until the services are rendered.
Advertising Costs
Advertising costs are included in selling, general and administrative expenses and are expensed as incurred. Advertising costs consist primarily of trade show and booth costs, product demonstration, and marketing materials. Advertising costs for the years ended December 31, 2013 and 2014 and the six months ended June 30, 2014 (unaudited) and 2015 (unaudited) were $0.1 million, $0.3 million, $73,000, and $0.3 million, respectively.
Stock-Based Compensation
The Company accounts for stock-based compensation arrangements with employees in accordance with ASC 718, Compensation—Stock Compensation. ASC 718 requires the recognition of compensation expense, using a fair value-based method, for costs related to all share-based payments including stock options.
The Company’s determination of the fair value of stock options on the date of grant utilizes the Black-Scholes option-pricing model and is impacted by its common stock price as well as changes in assumptions
F-12
Penumbra, Inc.
Notes to Consolidated Financial Statements (Continued)
2. Summary of Significant Accounting Policies (Continued)
regarding a number of highly complex and subjective variables. These variables include, but are not limited to, expected term that options will remain outstanding, expected common stock price volatility over the term of the option awards, risk-free interest rates and expected dividends.
The fair value is recognized over the period during which an optionee is required to provide services in exchange for the option award, known as the requisite service period (usually the vesting period) on a straight-line basis. Stock-based compensation expense recognized at fair value includes the impact of estimated forfeitures. The Company estimates future forfeitures at the date of grant and revises the estimates, if necessary, in subsequent periods if actual forfeitures differ from those estimates. To the extent actual forfeiture results differ from the estimates, the difference is recorded as a cumulative adjustment in the period forfeiture estimates are revised. No compensation cost is recorded for options that do not vest.
Equity instruments issued to non-employees are recorded at their fair value on the measurement date and are subject to periodic adjustments as the underlying equity instruments vest. The fair value of options granted to consultants is expensed over the service period.
Estimating the fair value of equity-settled awards as of the grant date using valuation models, such as the Black-Scholes option pricing model, is affected by assumptions regarding a number of complex variables. Changes in the assumptions can materially affect the fair value and ultimately how much stock-based compensation expense is recognized. These inputs are subjective and generally require significant analysis and judgment to develop. For all stock options granted to date, the Company estimated the volatility data based on a study of publicly traded industry peer companies. For purposes of identifying these peer companies, the Company considered the industry, stage of development, size and financial leverage of potential comparable companies. The risk-free interest rate is based on the yield available on U.S. Treasury zero-coupon issues similar in duration to the expected term of the equity-settled award.
Income Taxes
The Company accounts for income taxes using the asset and liability method, whereby deferred tax asset and liability account balances are determined based on differences between the financial reporting and tax bases of assets and liabilities, and are measured using the enacted tax rates and laws that will be in effect when the differences are expected to reverse. The Company provides a valuation allowance to reduce the net deferred tax assets to their estimated realizable value.
The calculation of the Company’s current provision for income taxes involves the use of estimates, assumptions and judgments while taking into account current tax laws, interpretation of current tax laws and possible outcomes of future tax audits. The Company has established reserves to address potential exposures related to tax positions that could be challenged by tax authorities. Although the Company believes its estimates, assumptions and judgments to be reasonable, any changes in tax law or its interpretation of tax laws and the resolutions of potential tax audits could significantly impact the amounts provided for income taxes in the Company’s consolidated financial statements.
The calculation of the Company’s deferred tax asset balance involves the use of estimates, assumptions and judgments while taking into account estimates of the amounts and type of future taxable income. Actual future operating results and the underlying amount and type of income could differ materially from the Company’s estimates, assumptions and judgments thereby impacting the Company’s financial position and results of operations.
The Company follows ASC 740-10 “Accounting for Uncertainty in Income Taxes,” which prescribes a recognition threshold and measurement attribute for the financial statement recognition and measurement of
F-13
Penumbra, Inc.
Notes to Consolidated Financial Statements (Continued)
2. Summary of Significant Accounting Policies (Continued)
uncertain tax positions taken or expected to be taken in the Company’s income tax return, and also provides guidance on derecognition, classification, interest and penalties, accounting in interim periods, disclosure and transition.
The Company includes interest and penalties related to unrecognized tax benefits within income tax expense in the accompanying consolidated statements of operations. The Company has not incurred any interest or penalties related to unrecognized tax benefits in any of the periods presented.
Comprehensive Income
The Company is required to display comprehensive income and its components as part of the Company’s consolidated financial statements. Comprehensive income consists of net income, unrealized gains on available-for-sale investments and the effects of foreign currency translation.
Deferred Offering Costs
Deferred offering costs, which primarily consist of direct incremental legal and accounting fees relating to the proposed IPO, are capitalized. The deferred offering costs will be offset against IPO proceeds upon the consummation of the offering. In the event the offering is terminated, deferred offering costs will be expensed.
Net Income (Loss) Per Share and Unaudited Pro Forma Net Income (Loss) Per Share of Common Stock
The Company calculates its basic and diluted net income (loss) per share attributable to common stockholders in conformity with the two-class method required for companies with participating securities. Under the two-class method, the Company determines whether it has net income (loss) attributable to common stockholders, which includes the results of operations less current period preferred stock non-cumulative dividends. If it is determined that the Company does have net income (loss) attributable to common stockholders during a period, the related undistributed earnings are then allocated between common stock and the preferred stock based on the weighted average number of shares outstanding during the period to determine the numerator for the basic net income (loss) per share attributable to common stockholders. In computing diluted net income attributable to common stockholders, undistributed earnings are re-allocated to reflect the potential impact of dilutive securities to determine the numerator for the diluted net income per share attributable to common stockholders. The Company’s basic net income (loss) per share attributable to common stockholders is calculated by dividing the net income (loss) by the weighted average number of shares of common stock outstanding for the period. The diluted net income per share attributable to common stockholders is computed by giving effect to all potential dilutive common stock equivalents outstanding for the period. For purposes of this calculation, options to purchase common stock and common stock warrants are considered common stock equivalents.
The unaudited pro forma basic and diluted net income (loss) per share reflects the automatic conversion of all outstanding shares of preferred stock as if the conversion had occurred at the earlier of the beginning of the period or the date of issuance, if later.
The unaudited pro forma basic and diluted net income (loss) per share amounts do not give effect to the issuance of shares from the planned initial public offering nor do they give effect to potential dilutive securities where the impact would be anti-dilutive.
Recently Issued Accounting Standards
In May 2014, the FASB issued ASU No. 2014-09, Revenue from Contracts with Customers, which outlines a comprehensive new revenue recognition model designed to depict the transfer of promised goods or services to
F-14
Penumbra, Inc.
Notes to Consolidated Financial Statements (Continued)
2. Summary of Significant Accounting Policies (Continued)
customers in an amount that reflects the consideration to which the entity expects to be entitled in exchange for those goods or services. The accounting standard is effective for annual reporting periods (including interim reporting periods within those periods) beginning after December 15, 2016. Early adoption is not permitted. The Company is currently evaluating the impact of this accounting standard.
In July 2015, the FASB voted to defer the effective date by one year to December 15, 2017 for annual reporting periods beginning after that date. The FASB also voted to permit early adoption of the standard, but not before the original effective date of December 15, 2016. The final accounting standard formally amending the effective date is expected to be issued by the FASB by the end of the third quarter of 2015 (unaudited).
In November 2014, the FASB issued ASU No. 2014-17, Pushdown Accounting, which provides an acquired entity to elect the option to apply pushdown accounting in the reporting period in which the change-in-control event occurs and allows the acquired entity to determine whether to elect to apply pushdown accounting for each individual change-in-control event in which the acquirer obtains control of the acquired entity. This accounting standard is effective as of November 18, 2014 and the Company will apply the guidance to future change-in control events.
In July 2015, the FASB issued ASU No. 2015-11, Simplifying the Measurement of Inventory, which requires an entity to measure most inventory at the lower of cost and net realizable value, thereby simplifying the current guidance under which an entity must measure inventory at the lower of cost or market. The accounting standard is effective prospectively for annual periods beginning after December 15, 2016, and interim periods therein. Early adoption is permitted as of the beginning of an interim or annual reporting period. The Company is currently evaluating the impact of this accounting standard (unaudited).
3. Fair Value of Financial Instruments
Fair value is defined as the price that would be received to sell an asset or paid to transfer a liability (an exit price) in an orderly transaction between market participants at the reporting date. The accounting guidance establishes a three-tiered hierarchy, which prioritizes the inputs used in the valuation methodologies in measuring fair value:
Level 1—Quoted prices in active markets for identical assets or liabilities.
Level 2—Inputs other than Level 1 that are observable, either directly or indirectly, such as quoted prices in markets that are not active; or other inputs that are observable or can be corroborated by observable market data for substantially the full term of the assets or liabilities.
Level 3—Unobservable inputs that are supported by little or no market activity and that are significant to the fair value of the assets or liabilities.
The categorization of a financial instrument within the valuation hierarchy is based on the lowest level of input that is significant to the fair value measurement.
The Company classifies its cash equivalents and marketable investments within Level 1 and Level 2, as it uses quoted market prices or alternative pricing sources and models utilizing market observable inputs.
The Company determined the fair value of its Level 1 financial instruments, which are traded in active markets, using quoted market prices for identical instruments.
Marketable investments classified within Level 2 of the fair value hierarchy are valued based on other observable inputs, including broker or dealer quotations or alternative pricing sources. When quoted prices in
F-15
Penumbra, Inc.
Notes to Consolidated Financial Statements (Continued)
3. Fair Value of Financial Instruments (Continued)
active markets for identical assets or liabilities are not available, the Company relies on non-binding quotes from its investment managers, which are based on proprietary valuation models of independent pricing services. These models generally use inputs such as observable market data, quoted market prices for similar instruments, historical pricing trends of a security as relative to its peers and internal assumptions of the independent pricing services. To validate the fair value determination provided by its investment managers, the Company reviews the pricing movement in the context of overall market trends and trading information from its investment managers. In addition, the Company assesses the inputs and methods used in determining the fair value in order to determine the classification of securities in the fair value hierarchy.
The Company did not own any Level 3 financial assets or liabilities as of December 31, 2013 or 2014 or June 30, 2015 (unaudited).
During the years ended December 31, 2013 and 2014, and the six months ended June 30, 2014 (unaudited) and 2015 (unaudited), the Company did not record impairment charges related to its marketable investments, and the Company did not have any transfers between Level 1, Level 2 and Level 3 of the fair value hierarchy.
The following tables set forth the fair value of the Company’s financial assets and liabilities by level within the fair value hierarchy (in thousands):
| As of December 31, 2013 | ||||||||||||||||
| Level 1 | Level 2 | Level 3 | Fair Value | |||||||||||||
| Financial Assets |
||||||||||||||||
| Marketable investments: |
||||||||||||||||
| Mutual funds |
$ | 15,545 | $ | — | $ | — | $ | 15,545 | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total |
$ | 15,545 | $ | — | $ | — | $ | 15,545 | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
The Company’s marketable investments as of December 31, 2013 of $15.5 million included $6.0 million of restricted investments, which were held with a major financial institution as a security for the Company’s credit facility. Refer to Note 5.
| As of December 31, 2014 | ||||||||||||||||
| Level 1 | Level 2 | Level 3 | Fair Value | |||||||||||||
| Financial Assets |
||||||||||||||||
| Cash equivalents: |
||||||||||||||||
| Money market funds |
$ | 155 | $ | — | $ | — | $ | 155 | ||||||||
| Marketable investments: |
||||||||||||||||
| U.S. Agency securities |
— | 6,006 | — | 6,006 | ||||||||||||
| U.S. Treasury |
4,009 | — | — | 4,009 | ||||||||||||
| Corporate bonds |
— | 29,619 | — | 29,619 | ||||||||||||
| Mutual funds |
8,619 | — | — | 8,619 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total |
$ | 12,783 | $ | 35,625 | $ | — | $ | 48,408 | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
F-16
Penumbra, Inc.
Notes to Consolidated Financial Statements (Continued)
3. Fair Value of Financial Instruments (Continued)
| As of June 30, 2015 | ||||||||||||||||
| Level 1 | Level 2 | Level 3 | Fair Value | |||||||||||||
| (unaudited) | ||||||||||||||||
| Financial Assets |
||||||||||||||||
| Cash equivalents: |
||||||||||||||||
| Money market funds |
$ | 24,762 | $ | — | $ | — | $ | 24,762 | ||||||||
| Marketable investments: |
||||||||||||||||
| U.S. Agency securities |
— | — | — | — | ||||||||||||
| U.S. Treasury |
— | — | — | — | ||||||||||||
| Corporate bonds |
— | — | — | — | ||||||||||||
| Mutual funds |
— | — | — | — | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total |
$ | 24,762 | $ | — | $ | — | $ | 24,762 | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
During the years ended December 31, 2013 and 2014 and the six months ended June 30, 2014 (unaudited) and 2015 (unaudited), the Company did not have any transfers of financial assets measured at fair value on a recurring basis to or from Level 1, Level 2 or Level 3. The Company did not hold any Level 3 assets or liabilities as of December 31, 2013 and 2014 and June 30, 2015 (unaudited).
The carrying amounts of cash equivalents, accounts receivable, prepaid expenses and other current assets, accounts payable and accrued liabilities approximate fair value due to their relatively short maturities.
4. Balance Sheet Components
Accounts Receivable, Net
The Company’s allowance for doubtful accounts comprised of the following (in thousands):
| Balance At Beginning Of Year |
Charged To Costs And Expenses |
Deductions | Balance At End Of Year |
|||||||||||||
| Allowance for Doubtful Accounts |
||||||||||||||||
| For year ended: |
||||||||||||||||
| December 31, 2013 |
$ | 212 | $ | 259 | $ | — | $ | 471 | ||||||||
| December 31, 2014 |
471 | 150 | (19 | ) | 602 | |||||||||||
| June 30, 2015 (unaudited) |
602 | (101 | ) | — | 501 | |||||||||||
Prepaid Expenses and Other Current Assets
The Company’s prepaid expenses and other current assets comprised of the following (in thousands):
| December 31, | June 30, 2015 |
|||||||||||
| 2013 | 2014 | |||||||||||
| (unaudited) | ||||||||||||
| Prepaid expenses |
$ | 3,166 | $ | 3,130 | $ | 5,849 | ||||||
| Income tax receivable |
78 | 1,654 | 1,889 | |||||||||
| Other current assets |
43 | 331 | 182 | |||||||||
|
|
|
|
|
|
|
|||||||
| Prepaid expenses and other current assets |
$ | 3,287 | $ | 5,115 | $ | 7,920 | ||||||
|
|
|
|
|
|
|
|||||||
F-17
Penumbra, Inc.
Notes to Consolidated Financial Statements (Continued)
4. Balance Sheet Components (Continued)
Marketable Investments
The Company’s marketable investments as of December 31, 2013 were as follows (in thousands):
| Marketable Investments |
Cost | Gross Unrealized Gains |
Gross Unrealized Losses |
Fair Value | ||||||||||||
| Mutual funds |
$ | 15,519 | $ | 125 | $ | (99 | ) | $ | 15,545 | |||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total |
$ | 15,519 | $ | 125 | $ | (99 | ) | $ | 15,545 | |||||||
|
|
|
|
|
|
|
|
|
|||||||||
The Company’s marketable investments as of December 31, 2013 of $15.5 million included $6.0 million of restricted investments, which were held with a major financial institution as a security for the Company’s credit facility. Refer to Note 5.
The Company’s marketable investments as of December 31, 2014 were as follows (in thousands):
| Marketable Investments |
Cost | Gross Unrealized Gains |
Gross Unrealized Losses |
Fair Value | ||||||||||||
| U.S. Agency securities |
$ | 6,012 | $ | 3 | $ | (9 | ) | $ | 6,006 | |||||||
| U.S. Treasury |
4,011 | — | (2 | ) | 4,009 | |||||||||||
| Corporate bonds |
29,834 | 4 | (219 | ) | 29,619 | |||||||||||
| Mutual funds |
8,768 | — | (149 | ) | 8,619 | |||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total |
$ | 48,625 | $ | 7 | $ | (379 | ) | $ | 48,253 | |||||||
|
|
|
|
|
|
|
|
|
|||||||||
The Company did not have any marketable investments as of June 30, 2015 (unaudited).
For the years ended December 31, 2013 and 2014 and the six months ended June 30, 2014 (unaudited), gains or losses realized on the sale of investments were insignificant. During the six months ended June 30, 2015, the Company sold all of its marketable investments and recorded a realized loss of $0.5 million. As of December 31, 2014, there are no securities that have been in a loss position for more than twelve months.
The contractual maturities of the Company’s marketable investments as of December 31, 2014 were as follows (in thousands):
| December 31, 2014 |
||||
| Fair Value | ||||
| Due in one year |
$ | 16,442 | ||
| Due in one to five years |
31,811 | |||
|
|
|
|||
| Total |
$ | 48,253 | ||
|
|
|
|||
F-18
Penumbra, Inc.
Notes to Consolidated Financial Statements (Continued)
4. Balance Sheet Components (Continued)
Inventories
The components of inventories consisted of the following (in thousands):
| December 31, | June 30, 2015 |
|||||||||||
| 2013 | 2014 | |||||||||||
| (unaudited) | ||||||||||||
| Raw materials |
$ | 3,778 | $ | 5,105 | $ | 8,611 | ||||||
| Work in process |
614 | 543 | 1,094 | |||||||||
| Finished goods |
22,675 | 27,803 | 34,474 | |||||||||
|
|
|
|
|
|
|
|||||||
| Inventories |
$ | 27,067 | $ | 33,451 | $ | 44,179 | ||||||
|
|
|
|
|
|
|
|||||||
Property and Equipment, Net
Property and equipment, net comprised of the following (in thousands):
| December 31, | June 30 2015 |
|||||||||||
| 2013 | 2014 | |||||||||||
| (unaudited) | ||||||||||||
| Machinery and equipment |
$ | 3,466 | $ | 5,089 | $ | 7,272 | ||||||
| Furniture and fixtures |
418 | 519 | 1,845 | |||||||||
| Leasehold improvements |
387 | 379 | 967 | |||||||||
| Software |
482 | 599 | 769 | |||||||||
| Computers |
112 | 153 | 400 | |||||||||
| Construction in progress |
37 | 1,931 | 929 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total property and equipment |
4,902 | 8,670 | 12,182 | |||||||||
| Less: Accumulated depreciation and amortization |
(2,871 | ) | (3,489 | ) | (4,206 | ) | ||||||
|
|
|
|
|
|
|
|||||||
| Property and equipment, net |
$ | 2,031 | $ | 5,181 | $ | 7,976 | ||||||
|
|
|
|
|
|
|
|||||||
Depreciation and amortization expense for the years ended December 31, 2013 and 2014 and the six months ended June 30, 2014 (unaudited) and 2015 (unaudited) was $0.7 million, $0.8 million, $0.3 million and $0.8 million, respectively.
Accrued Liabilities
The following table shows the components of accrued liabilities (in thousands):
| December 31, | June 30, 2015 |
|||||||||||
| 2013 | 2014 | |||||||||||
| (unaudited) | ||||||||||||
| Payroll and employee-related expenses |
$ | 5,119 | $ | 8,221 | $ | 11,600 | ||||||
| Sales return reserve |
2,161 | 2,164 | 2,387 | |||||||||
| Preclinical and clinical trial cost |
2,267 | 2,319 | 1,661 | |||||||||
| Deferred revenue |
1,093 | 1,591 | 786 | |||||||||
| Product warranty |
323 | 314 | 490 | |||||||||
| Sales tax payable |
368 | 306 | 662 | |||||||||
| Income tax payable |
— | 332 | 672 | |||||||||
| Other accrued liabilities |
3,194 | 3,228 | 4,910 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total accrued liabilities |
$ | 14,525 | $ | 18,475 | $ | 23,168 | ||||||
|
|
|
|
|
|
|
|||||||
F-19
Penumbra, Inc.
Notes to Consolidated Financial Statements (Continued)
4. Balance Sheet Components (Continued)
The estimated product warranty accrual was as follows (in thousands):
| December 31, | June 30, 2015 |
|||||||||||
| 2013 | 2014 | |||||||||||
| (unaudited) | ||||||||||||
| Balance at the beginning of the period |
$ | 323 | $ | 323 | $ | 314 | ||||||
| Accruals of warranties issued |
100 | 149 | 351 | |||||||||
| Settlements of warranty claims |
(100 | ) | (158 | ) | (175 | ) | ||||||
|
|
|
|
|
|
|
|||||||
| Balance at the end of the period |
$ | 323 | $ | 314 | $ | 490 | ||||||
|
|
|
|
|
|
|
|||||||
5. Credit Facility
In May 2012, the Company entered into a revolving credit facility of $15.0 million with Wells Fargo Bank, National Association. The credit facility was collateralized by the Company’s investment balances. The interest on the credit facility was based on the daily one-month London Inter-Bank Offered Rate, plus 1.75% and was payable monthly. Any outstanding balance on the credit facility was due in full on June 1, 2015. The credit facility contained customary covenants for credit facilities of this type, including limitations on disposition of assets and changes in control. In May 2014, in conjunction with its Series F financing, the Company paid the then outstanding balance on the credit facility and terminated the credit facility. The Company was in compliance with the covenants as of December 31, 2013.
6. Related Party Transactions
Notes Receivable from Stockholders
In March 2005, options to purchase 875,000 shares of common stock, subject to repurchase by the Company, were exercised in exchange for full recourse promissory notes totaling $0.1 million. The notes bear interest at 2.92% per year, compounded annually with interest and principal due on the earlier to occur of January 31, 2015, or the date the employee ceases to be an employee, director or consultant of the Company.
In July 2011, options to purchase 5,000 shares of common stock were exercised in exchange for a full recourse promissory note totaling $10,250. The note is noninterest bearing and is due in full on December 31, 2016.
As of December 31, 2013 and 2014 and June 30, 2015 (unaudited), outstanding promissory notes were $0.1 million, $0.1 million and $31,500, respectively.
7. Commitments and Contingencies
Lease Commitments
The Company leases its offices and other equipment under non-cancelable operating leases that expire at various dates in 2029. Rent expense for the years ended December 31, 2013 and 2014 and the six months ended June 30, 2014 (unaudited) and 2015 (unaudited) was $1.7 million, $1.8 million, $0.9 million and $1.4 million, respectively.
F-20
Penumbra, Inc.
Notes to Consolidated Financial Statements (Continued)
7. Commitments and Contingencies (Continued)
Future minimum lease payments under the non-cancelable operating leases as of December 31, 2014
are as follows (in thousands):
| Year Ending December 31: |
Lease Payments | |||
| 2015 |
$ | 2,148 | ||
| 2016 |
2,158 | |||
| 2017 |
2,174 | |||
| 2018 |
2,166 | |||
| 2019 |
2,242 | |||
| Thereafter |
25,156 | |||
|
|
|
|||
| Total future minimum lease payments |
$ | 36,044 | ||
|
|
|
|||
Future minimum lease payments under the non-cancelable operating leases as of June 30, 2015 (unaudited) are as follows (in thousands):
| Quarter Ending June 30: |
Lease Payments | |||
| 2015 |
$ | 1,478 | ||
| 2016 |
3,026 | |||
| 2017 |
3,011 | |||
| 2018 |
3,010 | |||
| 2019 |
3,110 | |||
| Thereafter |
35,308 | |||
|
|
|
|||
| Total future minimum lease payments |
$ | 48,943 | ||
|
|
|
|||
Future minimum lease payments under the non-cancelable operating leases as of June 30, 2015 in the table above includes lease for additional space at the Company’s campus in Alameda, California. This lease commenced in June 2015 upon landlord’s substantial completion of tenant improvements and expires in November 2029.
Purchase Commitments
The Company had non-cancellable purchase obligations to suppliers for the year ended December 31, 2014 and the six months ended June 30, 2015 (unaudited) of $9.9 million and $6.3 million, respectively.
Royalty Obligations
In March 2005, the Company entered into a license agreement that requires the Company to make minimum royalty payments to the licensor, on a quarterly basis. As of December 31, 2014 and June 30, 2015, the license agreement requires minimum annual royalty payments of $0.1 million and $0.1 million in equal quarterly installments, respectively. On each January 1, the quarterly calendar year minimum royalty shall be adjusted to equal the prior year’s minimum royalty adjusted by a percentage equal to the percentage change in the “consumer price index for all urban consumers” for the prior calendar year as reported by the U.S. Department of Labor. Unless terminated earlier, the term of the license agreement shall continue until the expiration of the last to expire patent that covers that licensed product or for the period of 15 years following the first commercial sale of such licensed product, whichever is longer. The first commercial sale occurred in June 2007.
F-21
Penumbra, Inc.
Notes to Consolidated Financial Statements (Continued)
7. Commitments and Contingencies (Continued)
In April 2012, the Company entered into an agreement that requires the Company to pay a 5% royalty on sales of products covered under applicable patents, on a quarterly basis. The first commercial sale occurred in April 2014. Unless terminated earlier, the royalty term for each applicable product shall continue for fifteen years following the first commercial sale of such patented product, or when the applicable patent covering such product has expired, whichever is sooner.
In April 2015, the Company entered into a royalty agreement that requires the Company to pay a 2% royalty on sales of certain products covered by the agreement, on a quarterly basis. The Company anticipates that the first commercial sale of the covered Indigo products will occur in June 2015. Unless terminated earlier, the royalty term for each covered product shall continue for twenty years following the first commercial sale of the covered products.
Royalty expense included in cost of sales for the years ended December 31, 2013 and 2014 and the six months ended June 30, 2014 (unaudited) and 2015 (unaudited), was $0.7 million, $1.1 million, $0.5 million and $0.8 million, respectively.
Contingencies
From time to time, the Company may have certain contingent liabilities that arise in the ordinary course of business. The Company accrues a liability for such matters when it is probable that future expenditures will be made and such expenditures can be reasonably estimated. There have been no contingent liabilities requiring accrual or disclosure at December 31, 2013 and 2014 or June 30, 2015 (unaudited).
Indemnification
The Company enters into standard indemnification arrangements in the ordinary course of business. Pursuant to these arrangements, the Company indemnifies, holds harmless, and agrees to reimburse the indemnified parties for losses suffered or incurred by the indemnified party, in connection with any trade secret, copyright, patent or other intellectual property infringement claim by any third-party with respect to the Company’s technology. The term of these indemnification agreements is generally perpetual. The maximum potential amount of future payments the Company could be required to make under these agreements is not determinable because it involves claims that may be made against the Company in the future, but have not yet been made.
The Company has entered into indemnification agreements with its directors and officers that may require the Company to indemnify its directors and officers against liabilities that may arise by reason of their status or service as directors or officers, other than liabilities arising from willful misconduct of the individual.
The Company has not incurred costs to defend lawsuits or settle claims related to these indemnification agreements. No liability associated with such indemnifications has been recorded to date.
Litigation
From time to time, the Company is subject to claims and assessments in the ordinary course of business. The Company is not currently a party to any litigation matter that, individually or in the aggregate, is expected to have a material adverse effect on the Company’s business, financial condition, results of operations or cash flows.
F-22
Penumbra, Inc.
Notes to Consolidated Financial Statements (Continued)
8. Preferred Stock
The preferred stock at December 31, 2014 consisted of the following (in thousands, except shares):
| Series |
Shares Authorized |
Shares Issued and Outstanding |
Proceeds, Net of Issuance Costs |
Aggregate Liquidation Amount |
||||||||||||
| Series A Preferred Stock |
1,000,000 | 1,000,000 | $ | 299 | $ | 554 | ||||||||||
| Series B Preferred Stock |
4,287,486 | 4,005,338 | 6,536 | 11,725 | ||||||||||||
| Series C Preferred Stock |
4,388,715 | 4,168,218 | 13,266 | 22,238 | ||||||||||||
| Series D Preferred Stock |
3,944,733 | 3,881,459 | 19,647 | 30,976 | ||||||||||||
| Series E Preferred Stock |
1,973,684 | 1,909,940 | 14,507 | 21,609 | ||||||||||||
| Series F Preferred Stock |
5,303,031 | 4,545,455 | 57,212 | 62,259 | ||||||||||||
| Undesignated |
4,102,351 | — | — | — | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total preferred stock |
25,000,000 | 19,510,410 | $ | 111,467 | $ | 149,361 | ||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
The preferred stock at June 30, 2015 (unaudited) consisted of the following (in thousands, except shares):
| Series |
Shares Authorized |
Shares Issued and Outstanding |
Proceeds, Net of Issuance Costs |
Aggregate Liquidation Amount |
||||||||||||
| Series A Preferred Stock |
1,000,000 | 1,000,000 | $ | 299 | $ | 571 | ||||||||||
| Series B Preferred Stock |
4,287,486 | 4,005,338 | 6,536 | 12,072 | ||||||||||||
| Series C Preferred Stock |
4,388,715 | 4,168,218 | 13,266 | 22,893 | ||||||||||||
| Series D Preferred Stock |
3,944,733 | 3,881,459 | 19,647 | 31,886 | ||||||||||||
| Series E Preferred Stock |
1,973,684 | 1,909,940 | 14,507 | 22,246 | ||||||||||||
| Series F Preferred Stock |
5,303,031 | 4,545,455 | 57,212 | 64,070 | ||||||||||||
| Undesignated |
4,102,351 | — | — | — | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total preferred stock |
25,000,000 | 19,510,410 | $ | 111,467 | $ | 153,738 | ||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
The rights, preferences and privileges of the Series A Preferred Stock (Series A), Series B Preferred Stock (Series B), Series C Preferred Stock (Series C), Series D Preferred Stock (Series D), Series E Preferred Stock (Series E) and Series F Preferred Stock (Series F) are as follows:
Voting
The holders of each share of Series A, Series B, Series C, Series D, Series E and Series F preferred stock are entitled to the number of votes equal to the number of shares of common stock into which each share of preferred stock could be converted, and have voting rights and powers equal to the voting rights and powers of the holders of common stock.
Dividends
The holders of Series A, Series B, Series C, Series D, Series E and Series F preferred stock are entitled to receive dividends out of any assets legally available, prior, and in preference to, any declaration or payment of any dividend on the common stock of the Company, at the rate of 6% of the original purchase price (adjusted for any stock dividends, stock splits, or recapitalization) per share per annum. For any future dividends per annum, the preferred stock would participate with common stock on an as-converted basis. Such dividends are payable when, as, and if declared by the Board of Directors and are not cumulative. No dividends have been declared or paid since the issuance of preferred stock through December 31, 2014 or June 30, 2015 (unaudited).
F-23
Penumbra, Inc.
Notes to Consolidated Financial Statements (Continued)
8. Preferred Stock (Continued)
Conversion
Each share of Series A, Series B, Series C, Series D, Series E and Series F preferred stock, at the option of the holder, is convertible into the number of fully paid and nonassessable shares of common stock, which results from dividing the original purchase price of the Series A, Series B, Series C, Series D, Series E and Series F preferred stock by the conversion price applicable to such shares. The initial per-share conversion price of the Series A, Series B, Series C, Series D, Series E and Series F preferred stock is $0.30, $1.63, $3.19, $5.07, $7.60 and $13.20 per share, respectively.
Conversion is automatic at the then-effective conversion rate either (a) immediately upon the closing of a firm commitment underwritten public offering pursuant to an effective registration statement under the Securities Act of 1933, as amended, covering the offer and sale of common stock in which the aggregate proceeds raised, equal, or exceed $15,000,000 or (b) at the election of the holders of two-thirds of the outstanding Series A, Series B, Series C, Series D, Series E and Series F preferred stock voting together as a single class on an as-converted basis.
Liquidation
In the event of any liquidation, dissolution, or winding-up of the Company, either voluntary or involuntary, the holders of Series A, Series B, Series C, Series D, Series E and Series F preferred stock shall be entitled to receive, prior and in preference to any distribution of any of the assets of the Company to the holders of common stock by reason of their ownership, an amount per share equal to the original purchase price, plus 6% per annum compounded annually from original issuance date for each outstanding share of Series A, Series B, Series C, Series D, Series E and Series F preferred stock (as adjusted for any stock dividends, stock splits, or recapitalization), plus any declared, but unpaid, dividends on such share, respectively. As of December 31, 2014, the holders of Series A, Series B, Series C, Series D, Series E and Series F preferred stock are entitled to receive an amount per share equal to $0.55, $2.93, $5.34, $7.98, $11.31 and $13.70, respectively. As of June 30, 2015, the holders of Series A, Series B, Series C, Series D, Series E and Series F preferred stock are entitled to receive an amount per share equal to $0.57, $3.01, $5.49, $8.21, $11.65 and $14.10, respectively.
If upon the occurrence of such event, the assets and funds distributed among the holders of Series A, Series B, Series C, Series D, Series E and Series F preferred stock are insufficient to permit the payment to such holders of the full aforesaid preferential amounts, then, subject to the rights of series of preferred stock that may from time to time come into existence, the entire assets and funds of the Company legally available for distribution will be distributed ratably among the holders of Series A, Series B, Series C, Series D, Series E and Series F preferred stock, and holders of such series of preferred stock in proportion to the preferential amount each such holder is entitled to receive.
After payment has been made to the holders of Series A, Series B, Series C, Series D, Series E and Series F preferred stock, any remaining assets and funds are to be distributed among the holders of common stock ratably, based on the number of shares of common stock held by each stockholder.
Mergers
A merger, reorganization or sale of all or substantially all of the assets of the Company in which more than 50% of the voting power of the Company is disposed of shall be deemed to be a liquidation, dissolution or winding-up.
F-24
Penumbra, Inc.
Notes to Consolidated Financial Statements (Continued)
9. Common Stock
Each share of common stock is entitled to one vote. The holders of common stock are also entitled to receive dividends whenever funds are legally available and when declared by the board of directors, subject to the prior rights of holders of all classes of stock outstanding.
10. Common Stock Liability
As of December 31, 2013 and 2014 and June 30, 2015 (unaudited), the Company had outstanding options to purchase 3,092,176, 2,900,676 and 2,460,574 shares, respectively, of common stock. Proceeds from the exercise of unvested stock options are accounted for as a liability. The liability as of December 31, 2013 and 2014 and June 30, 2015 (unaudited), were $0.1 million, $0.2 million and $0.1 million, respectively.
The options are not considered exercised and the underlying shares are not considered issued until the Company’s right to repurchase lapses, when the employee vests in the award. Employees who have exercised shares prior to vesting are current employees of the Company. There were 16,305, 33,081 and 24,818 shares subject to repurchase as of December 31, 2013, 2014 and June 30, 2015 (unaudited), respectively.
11. Warrants
In connection with the sale of Series B preferred stock in 2004, the Company issued warrants to purchase 211,138 shares of common stock at a purchase price of $0.01 per share. The warrants were exercisable upon grant and had a term of 10 years from the date of grant, which expired on December 31, 2014. The value of the warrants was calculated using Black-Scholes option pricing model and was deemed to be immaterial. As of December 31, 2013, warrants to purchase 79,645 shares of common stock were outstanding. No warrants were outstanding as of December 31, 2014.
12. Stock Option Plans
2005 Stock Plan
The Company’s board of directors and stockholders approved the Company’s 2005 Stock Plan (the 2005 Plan) and it became effective in January 2005. The 2005 Plan was subsequently amended and restated in 2006, 2007, 2008 and 2010. As of December 31, 2014 and June 30, 2015 (unaudited), the Company had granted options to purchase 5,431,017 and 5,431,017 shares of common stock, respectively, under the 2005 Plan, of which options to purchase 2,707,176 and 1,779,924 shares of common stock were outstanding, and options to purchase 33,081 and 24,818 shares of common stock had been early exercised and were unvested and subject to repurchase, as of December 31, 2014 and June 30, 2015 (unaudited), respectively. Under the 2005 Plan, the board of directors could grant incentive stock options (ISO), nonqualified stock options (NSO), or stock awards to eligible persons, including employees, nonemployees, directors, consultants and other independent advisors who provide services to the Company. Stock purchase rights could also be granted under the Plan. The board of directors had the authority to determine to whom options would be granted, the number of options, the term and the exercise price. ISOs could only be granted to Company employees, which include officers and directors of the Company. NSOs and stock purchase rights could be granted to employees and consultants. For individuals holding more than 10% of the voting rights of all classes of stock, the exercise price for an ISO could not be less than 110% of fair market value. Options are exercisable immediately upon the optionee entering into a restricted stock purchase agreement with respect to any unvested options. Options generally vest annually at a rate of 1/4 after the first year and 1/48 per month thereafter. The term of the options is no longer
F-25
Penumbra, Inc.
Notes to Consolidated Financial Statements (Continued)
12. Stock Option Plans (Continued)
than five years for ISOs, for which the grantee owns greater than 10% of the voting power of all classes of stock and no longer than 10 years for all other options. Stock options granted under the 2005 Plan generally have a contractual life of ten years, and generally vest over a period of four years.
2011 Equity Incentive Plan
The Company’s board of directors approved the Company’s 2011 Equity Incentive Plan (the 2011 Plan) and it became effective in October 2011. As of December 31, 2014 and June 30, 2015 (unaudited), the Company had granted options to purchase 145,000 and 145,000 shares of common stock, respectively, under the 2011 Plan, of which options to purchase 145,000 and 145,000 shares of common stock were outstanding at December 31, 2014 and June 30, 2015 (unaudited), respectively. The Company had also granted 505,000 and 505,000 shares of restricted stock under the 2011 Plan, of which 367,126 and 252,125 shares were unvested and subject to forfeiture and 1,667 and 1,667 shares had been forfeited as of December 31, 2014 and June 30, 2015 (unaudited), respectively. Under the 2011 Plan, the board of directors could grant ISOs, NSOs, restricted stock, or restricted stock units (RSU) to eligible persons, including employees, directors and consultants who provide services to the Company. Stock Appreciation Rights (SAR) could also be granted under the 2011 Plan. The board of directors had the authority to determine to whom options would be granted, the number of options, the term and the exercise price. ISOs could only be granted to Company employees, which include officers and directors of the Company. NSOs, SARs, restricted stock and RSUs could be granted to employees and consultants. For individuals holding more than 10% of the voting rights of all classes of stock, the exercise price for an ISO could not be less than 110% of fair market value. Stock options granted under the 2011 Plan generally have a contractual life of ten years, and generally vest over a period of four years.
2014 Equity Incentive Plan
The Company’s board of directors and stockholders approved the Company’s 2014 Equity Incentive Plan (the 2014 Plan) and it became effective in May, 2014. The 2014 Plan replaced the 2011 Plan and the 2005 Plan. No further equity awards may be granted under the 2011 Plan or the 2005 Plan. As of December 31, 2014 and June 30, 2015 (unaudited), respectively there were 2,688,513 and 1,713,634 shares remaining available for the grant of equity awards under our 2014 Plan. As of December 31, 2014 and June 30, 2015 (unaudited), the Company had granted options to purchase 48,500 and 536,650 shares of common stock under the 2014 Plan, 48,500 and 535,650 of which were outstanding and 0 and 1,000 options had been forfeited as of December 31, 2014 and June 30, 2015 (unaudited), respectively. The Company had also granted 0 and 662,361 shares of restricted stock under the 2014 Plan, as of December 31, 2014 and June 30, 2015 (unaudited), respectively, of which 0 and 503,646 shares were unvested and subject to forfeiture as of such dates.
Early Exercises
Stock options granted under the 2005 Plan, 2011 Plan and 2014 Plan allow the board of directors to grant awards to provide employee option holders the right to elect to exercise unvested options in exchange for restricted common stock. Unvested shares, which amounted to 16,305, 33,081 and 24,818 as of December 31, 2013 and 2014 and June 30, 2015 (unaudited), respectively, were subject to a repurchase right held by the Company at the original issue price in the event the optionees’ employment was terminated either voluntarily or involuntarily. For exercises of employee options, this right lapses according to the vesting schedule designated on the associated option grant. The repurchase terms are considered to be a forfeiture provision. The shares purchased by the employees pursuant to the early exercise of stock options are not deemed to be issued or outstanding for accounting purposes until those shares vest, though they are legally issued and
F-26
Penumbra, Inc.
Notes to Consolidated Financial Statements (Continued)
12. Stock Option Plans (Continued)
outstanding. In addition, cash received from employees for exercise of unvested options is treated as a refundable deposit shown as a liability on the consolidated balance sheets. As of December 31, 2013 and 2014 and June 30, 2015 (unaudited), cash received related to unvested shares totaled $0.1 million, $0.2 million and $0.1 million, respectively. Amounts recorded are transferred into common stock and additional paid-in-capital as the shares vest.
Activity of stock options under 2005 Plan, 2011 Plan and 2014 Plan is set forth below:
| Shares Available for Grant |
Number of Shares |
Weighted- Average Exercise Price |
Weighted Average Remaining Contractual Life (in Years) |
Aggregate Intrinsic Value |
||||||||||||||||
| (in thousands) | ||||||||||||||||||||
| Balance, December 31, 2013 |
500,963 | 3,092,176 | $ | 2.23 | ||||||||||||||||
| Additional shares authorized |
2,700,000 | – | – | |||||||||||||||||
| Restricted stock granted |
(286,000 | ) | – | – | ||||||||||||||||
| Restricted stock cancelled |
1,667 | – | – | |||||||||||||||||
| Options granted |
(268,050 | ) | 268,050 | 7.99 | ||||||||||||||||
| Options exercised |
– | (421,159 | ) | 2.79 | ||||||||||||||||
| Options cancelled |
39,933 | (38,391 | ) | 3.89 | ||||||||||||||||
|
|
|
|
|
|
|
|||||||||||||||
| Balance, December 31, 2014 |
2,688,513 | 2,900,676 | 2.66 | |||||||||||||||||
| Restricted stock granted (unaudited) |
(662,361 | ) | – | – | ||||||||||||||||
| Restricted stock withheld for taxes (unaudited) |
65,638 | – | – | |||||||||||||||||
| Options granted (unaudited) |
(488,150 | ) | 488,150 | 12.57 | ||||||||||||||||
| Options exercised (unaudited) |
– | (927,252 | ) | 0.92 | ||||||||||||||||
| Options withheld for taxes (unaudited) |
108,994 | – | – | |||||||||||||||||
| Options cancelled (unaudited) |
1,000 | (1,000 | ) | 14.46 | ||||||||||||||||
|
|
|
|
|
|
|
|||||||||||||||
| Balance, June 30, 2015 (unaudited) |
1,713,634 | 2,460,574 | $ | 5.27 | ||||||||||||||||
|
|
|
|
|
|
|
|||||||||||||||
| Vested and expected to vest—December 31, 2014 |
2,890,852 | $ | 2.63 | 4.11 | $ | 28,126 | ||||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||||||
| Exercisable—December 31, 2014 |
2,852,176 | $ | 2.55 | 4.04 | $ | 27,987 | ||||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||||||
| Vested and expected to vest—June 30, 2015 (unaudited) |
2,408,967 | $ | 5.14 | 5.67 | $ | 37,027 | ||||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||||||
| Exercisable—June 30, 2015 (unaudited) |
1,939,479 | $ | 3.37 | 4.68 | $ | 33,237 | ||||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||||||
F-27
Penumbra, Inc.
Notes to Consolidated Financial Statements (Continued)
12. Stock Option Plans (Continued)
The following table summarizes information about stock options outstanding at December 31, 2014:
| Options Outstanding | ||||||||||||
| Exercise Price |
Stock Options Outstanding |
Weighted Average Remaining Contractual Life (Years) |
Options Exercisable |
|||||||||
| $0.17—$1.54 |
1,562,917 | 1.83 | 1,562,917 | |||||||||
| $2.05—$3.98 |
885,459 | 5.95 | 885,459 | |||||||||
| $4.49—$5.99 |
192,650 | 7.47 | 192,650 | |||||||||
| $7.75—$9.06 |
259,650 | 9.31 | 211,150 | |||||||||
| $12.36—$12.36 |
— | — | — | |||||||||
|
|
|
|
|
|||||||||
| $0.17—$12.36 |
2,900,676 | 4.13 | 2,852,176 | |||||||||
|
|
|
|
|
|||||||||
The following table summarizes information about stock options outstanding at June 30, 2015 (unaudited):
| Options Outstanding | ||||||||||||
| Exercise Price |
Stock Options Outstanding |
Weighted Average Remaining Contractual Life (in Years) |
Options Exercisable |
|||||||||
| $0.57—$1.54 |
731,050 | 1.97 | 731,050 | |||||||||
| $2.05—$3.98 |
834,459 | 5.56 | 834,459 | |||||||||
| $4.49—$5.99 |
159,740 | 7.06 | 159,740 | |||||||||
| $7.75—$9.06 |
248,175 | 8.82 | 212,842 | |||||||||
| $12.36—$14.46 |
487,150 | 9.74 | 1,388 | |||||||||
|
|
|
|
|
|||||||||
| 2,460,574 | 5.75 | 1,939,479 | ||||||||||
|
|
|
|
|
|||||||||
The Company uses the Black-Scholes option pricing model to determine the fair value of stock options. The valuation model for stock compensation expense requires the Company to make assumptions and judgments about the variables used in the calculation including the expected term (weighted average period of time that the options granted are expected to be outstanding); volatility of the Company’s common stock and an assumed-risk-free interest rate.
| Year Ended December 31, | Six Months Ended June 30, |
|||||||||||||||
| 2013 | 2014 | 2014 | 2015 | |||||||||||||
| (unaudited) | ||||||||||||||||
| Expected term (in years) |
6.25 | 6.25 | 6.25 | 6.25 | ||||||||||||
| Expected volatility |
45 | % | 45 | % | 45 | % | 45 | % | ||||||||
| Risk-free interest rate |
0.63%–0.90 | % | 1.76%–2.02 | % | 1.76%–2.02 | % | 1.56%–1.78 | % | ||||||||
| Expected dividend rate |
0 | % | 0 | % | 0 | % | 0 | % | ||||||||
Fair Value of Common Stock. The fair value of the shares of common stock underlying our stock options has historically been determined by the Company’s board of directors. Because there has been no public market for their common stock and in the absence of recent arm’s-length cash sales transactions of their common stock with independent third parties, the Company’s board of directors has determined the fair value of their common stock by considering at the time of grant a number of objective and subjective factors. The Company’s
F-28
Penumbra, Inc.
Notes to Consolidated Financial Statements (Continued)
12. Stock Option Plans (Continued)
board of directors intends all options granted to be exercisable at a price per share not less than the per share fair value of their common stock underlying those options on the date of grant. The estimated fair value of their common stock was determined at each valuation date in accordance with the guidelines outlined in the American Institute of Certified Public Accountants Practice Aid, Valuation of Privately-Held-Company Equity Securities Issued as Compensation. Their board of directors, with the assistance of management, developed these valuations using significant judgement and taking into account numerous factors, including the following:
| • | independent third party valuations; |
| • | progress of research and development activities; |
| • | their operating and financial performance, including levels of available capital resources; |
| • | rights and preferences of their common stock compared to the rights and preferences of their other outstanding equity securities; |
| • | equity market conditions affecting comparable public companies, as reflected in comparable companies; market multiples, IPO valuations and other metrics; |
| • | the achievement of enterprise milestones, including progress in clinical trials; |
| • | the likelihood of achieving a liquidity event for the shares of common stock, such as an IPO given prevailing market and medical device sector conditions; |
| • | sales of their preferred stock in arms-length transactions; |
| • | the illiquidity of their securities by virtue of being a private company; |
| • | business risks; and |
| • | management and board experience. |
The Company considered the following approaches in the preparation of their valuations:
| • | Market Approach. The market approach values a business by reference to guideline companies, for which enterprise values are known. This approach has two principal methodologies. The guideline public company methodology derives valuation multiples from the operating data and share prices of similar publicly traded companies. The guideline acquisition methodology focuses on comparisons between the subject company and guideline acquired public or private companies. |
| • | Option-Pricing Method Backsolve, or OPM backsolve. The OPM backsolve method derives the implied equity value for a company from a recent transaction involving the company’s own securities issued on an arm’s-length basis. |
| • | Probability Weighted Expected Return Method. Using the probability weighted expected return, or PWERM, method, the value of a company’s common stock is estimated based upon the analysis of future values for the company assuming various possible future liquidity events like an initial public offering, or IPO, sale or merger. Share value is based upon the probability-weighted present value of expected future net cash flows, considering each of the possible future events, as well as the rights and preferences of each share class. |
F-29
Penumbra, Inc.
Notes to Consolidated Financial Statements (Continued)
12. Stock Option Plans (Continued)
In addition, the Company also considered an enterprise value allocation method:
| • | Option-Pricing Method, or OPM. Under this method, each class of stock is modeled as a call option with a distinct claim on the enterprise value of the company. The option’s exercise prices would be based on a comparison with the enterprise value. The method assumes that a formula, such as the Black-Scholes model, would calculate the fair value when provided with certain values, including share price, expiration date, volatility and risk free interest rate. |
The per share common stock value was estimated by allocating the Company’s enterprise value using the OPM method in October 1, 2013, May 16, 2014, and September 30, 2014, which determined the common value to be $7.75, $9.06 and $10.92, respectively. The per share common stock value was estimated in December 31, 2014, March 31, 2015 and June 30, 2015, which utilized the PWERM method, which determined the common stock value to be $12.36, $14.46 and $20.51, respectively.
In determining the estimated fair value of their common stock, the Company’s board of directors also considered the fact that their stockholders could not freely trade their common stock in the public markets. Accordingly, the Company applied discounts to reflect the lack of marketability of their common stock based on the expected time to liquidity. The estimated fair value of their common stock at each grant date reflected a non-marketability discount partially based on the anticipated likelihood and timing of a future liquidity event.
The key subjective factors and assumptions used in their valuations primarily consisted of: (i) the selection of the appropriate market comparable transactions, (ii) the selection of the appropriate comparable publicly traded companies, (iii) the financial forecasts utilized to determine future cash balances and necessary capital requirements, (iv) the probability and timing of the various possible liquidity events, (v) the estimated weighted-average cost of capital and (vi) the discount for lack of marketability of our common stock.
At each grant date the board of directors reviewed any recent events and their potential impact on the estimated fair value per share of the common stock.
Weighted Average Expected Term. The Company derived the expected term using the “simplified” method (the expected term is determined as the average of the time-to-vesting and the contractual life of the options), as the Company had limited historical information to develop expectations about future exercise patterns and post vesting employment termination behavior.
Volatility. Since there has been no public market for the Company’s common stock and lack of company-specific historical volatility, it has determined the share price volatility for options granted based on an analysis of the volatility used by a peer group of publicly traded medical device companies. In evaluating similarity, the Company considers factors such as industry, stage of life cycle and size.
Risk-Free Interest Rate. The risk-free interest rate is based upon U.S. Treasury zero-coupon issues with remaining terms similar to the expected term of the options.
Dividend Yield. The Company has never paid any dividends and does not plan to pay dividends in the foreseeable future, and therefore, used an expected dividend yield of zero in the valuation model.
Forfeitures. The Company is required to estimate forfeitures at the time of grant, and revise those estimates in subsequent periods if actual forfeitures differ from those estimates. The Company uses historical
F-30
Penumbra, Inc.
Notes to Consolidated Financial Statements (Continued)
12. Stock Option Plans (Continued)
data to estimate pre-vesting option forfeitures and record stock based compensation expense only for those awards that are expected to vest. To the extent actual forfeitures differ from the estimates, the difference will be recorded as a cumulative adjustment in the period that the estimates are revised.
The total intrinsic value of stock options exercised during the year ended December 31, 2014 and the six months ended June 30, 2015 (unaudited) was $3.8 million and $17.7 million, respectively. The intrinsic value is calculated as the difference between the estimated fair value of the Company’s common stock at the exercise date and the exercise price of the stock option. As of December 31, 2014 and June 30, 2015 (unaudited), the Company’s common stock valuation was at $12.36 and $20.51 per share, respectively.
The weighted average grant date fair value of the employee stock options granted during the years ended December 31, 2013 and 2014 and the six months ended June 30, 2014 (unaudited) and 2015 (unaudited) was $2.63, $3.69, $3.58 and $5.72 per share, respectively.
The following table summarizes the activity of unvested restricted stock:
| Weighted Average | ||||||||
| Number | Grant Date | |||||||
| of Shares | Fair Value | |||||||
| Unvested at December 31, 2013 |
135,461 | $ | 5.45 | |||||
| Granted |
286,000 | 7.75 | ||||||
| Vested |
(52,668 | ) | 5.32 | |||||
| Cancelled/Forfeited |
(1,667 | ) | 4.49 | |||||
|
|
|
|||||||
| Unvested at December 31, 2014 |
367,126 | 7.26 | ||||||
| Granted (unaudited) |
662,361 | 14.30 | ||||||
| Vested (unaudited) |
(273,716 | ) | 11.28 | |||||
| Cancelled/Forfeited (unaudited) |
— | — | ||||||
|
|
|
|||||||
| Unvested and expected to vest at June 30, 2015 (unaudited) |
755,771 | $ | 11.98 | |||||
|
|
|
|||||||
As of December 31, 2014 and June 30, 2015 (unaudited), total unrecognized compensation cost was $2.9 million and $11.4 million related to unvested share-based compensation arrangements which is expected to be recognized over a weighted average period of 2.8 years and 3.5 years, respectively.
The following table sets forth the stock-based compensation expense included in the consolidated statements of operations (in thousands):
| Year Ended December 31, |
Six Months Ended June 30, |
|||||||||||||||
| 2013 | 2014 | 2014 | 2015 | |||||||||||||
| (unaudited) | ||||||||||||||||
| Cost of sales |
$ | 98 | $ | 267 | $ | 124 | $ | 130 | ||||||||
| Research and development |
84 | 96 | 47 | 182 | ||||||||||||
| Sales, general and administrative |
704 | 1,070 | 531 | 3,304 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| $ | 886 | $ | 1,433 | $ | 702 | $ | 3,616 | |||||||||
|
|
|
|
|
|
|
|
|
|||||||||
F-31
Penumbra, Inc.
Notes to Consolidated Financial Statements (Continued)
13. Common and Preferred Stock Repurchase
The Company’s board of directors approved the repurchase of 70,612 shares of common stock, 45,000 stock options and 45,611 of preferred stock from shareholders in May 2014 for $13.20 per share for a total purchase price of $2.0 million. For the repurchased shares of common stock and stock options, the Company charged the difference between the purchase and market prices of $0.5 million to expense. For the repurchased preferred shares, the excess between the purchase and the issuance price of $0.5 million was treated as a deemed dividend. In addition, the Company closed a tender offer in July 2014 to repurchase shares of preferred stock from existing shareholders at a purchase price of $13.20 per share. The Company repurchased 584,052 shares of preferred stock for a total purchase price of $7.7 million. The excess between the purchase and the issuance price of $5.8 million was treated as a deemed dividend. The repurchased shares of common and preferred stock were retired and remained as authorized but unissued.
14. Employee Benefit Plans
The Company offers a retirement savings plan under Section 401(k) of the Internal Revenue Code (the IRC) to its eligible U.S. employees whereby they may contribute up to the maximum amount permitted by IRC. The retirement savings plan provides for employer contributions to be made at the discretion of the Company’s board of directors. The Company has not provided any such matching contributions through June 30, 2015.
15. Income Taxes
The Company is incorporated in the United States and operates in various countries with differing tax laws and rates. A portion of the Company’s income or (loss) before taxes and the provision for income taxes are generated from international operations.
Income or (loss) before income taxes for the fiscal years ended December 31, 2013 and 2014 is summarized as follows:
| Year Ended |
||||||||
| 2013 | 2014 | |||||||
| United States |
$ | (1,142 | ) | $ | 2,230 | |||
| Foreign |
(113 | ) | 909 | |||||
|
|
|
|
|
|||||
| Total income (loss) before provision for (benefit from) income taxes |
$ | (1,255 | ) | $ | 3,139 | |||
|
|
|
|
|
|||||
Income tax provision in 2013 and 2014 is comprised of federal, state, and foreign taxes.
F-32
Penumbra, Inc.
Notes to Consolidated Financial Statements (Continued)
15. Income Taxes (Continued)
The components of the provision for (benefit from) income taxes are summarized as follows:
| Year Ended December 31, |
||||||||
| 2013 | 2014 | |||||||
| Current: |
||||||||
| Federal |
$ | (155 | ) | $ | 1,155 | |||
| State |
87 | 274 | ||||||
| Foreign |
— | 323 | ||||||
|
|
|
|
|
|||||
| Total current |
(68 | ) | 1,752 | |||||
|
|
|
|
|
|||||
| Deferred: |
||||||||
| Federal |
(5,087 | ) | (625 | ) | ||||
| State |
(199 | ) | (116 | ) | ||||
| Foreign |
— | (117 | ) | |||||
|
|
|
|
|
|||||
| Total deferred |
(5,286 | ) | (858 | ) | ||||
|
|
|
|
|
|||||
| Provision for (benefit from) income taxes |
$ | (5,354 | ) | $ | 894 | |||
|
|
|
|
|
|||||
The Company’s actual provision for tax differed from the amounts computed by applying the U.S. federal income tax rate of 34% to pretax income as a result of the following:
| Year Ended December 31, |
||||||||
| 2013 | 2014 | |||||||
| Income tax at federal statutory rate |
34.0 | % | 34.0 | % | ||||
| State income taxes, net of federal benefit |
(5.6 | ) | 3.2 | |||||
| Foreign taxes differential |
5.8 | 5.2 | ||||||
| APB 51 prepaid tax |
(2.7 | ) | (8.9 | ) | ||||
| Foreign exchange gains and losses |
11.7 | - | ||||||
| IRC 199 deduction |
- | (7.0 | ) | |||||
| Stock-based compensation |
(14.1 | ) | 6.0 | |||||
| Meals and entertainment |
(10.7 | ) | 5.8 | |||||
| Imputed interest |
(12.1 | ) | 6.6 | |||||
| Tax credits |
41.8 | (12.1 | ) | |||||
| Other |
0.3 | 2.8 | ||||||
| Change in valuation allowance |
378.2 | (7.1 | ) | |||||
|
|
|
|
|
|||||
| Effective tax rate |
426.6 | % | 28.5 | % | ||||
|
|
|
|
|
|||||
F-33
Penumbra, Inc.
Notes to Consolidated Financial Statements (Continued)
15. Income Taxes (Continued)
Deferred income tax assets and liabilities consist of the following:
| December 31, | ||||||||
| 2013 | 2014 | |||||||
| Deferred tax assets |
||||||||
| Net operating loss carryforwards |
$ | 3,067 | $ | 1,626 | ||||
| Tax credits |
1,890 | 1,779 | ||||||
| Accruals and reserves |
2,749 | 3,759 | ||||||
| Stock-based compensation |
192 | 416 | ||||||
| Translation adjustment |
271 | 674 | ||||||
| UNICAP adjustments |
1,729 | 2,227 | ||||||
|
|
|
|
|
|||||
| Gross deferred tax assets |
9,898 | 10,481 | ||||||
| Valuation allowance |
(3,860 | ) | (2,945 | ) | ||||
|
|
|
|
|
|||||
| Total deferred tax assets |
6,038 | 7,536 | ||||||
|
|
|
|
|
|||||
| Deferred tax liabilities |
||||||||
| Depreciation and amortization |
(480 | ) | (685 | ) | ||||
|
|
|
|
|
|||||
| Total deferred tax liabilities |
(480 | ) | (685 | ) | ||||
|
|
|
|
|
|||||
| Net deferred tax assets |
$ | 5,558 | $ | 6,851 | ||||
|
|
|
|
|
|||||
| Deferred income taxes, net - Balance Sheet Classification |
||||||||
| Current deferred income tax assets |
$ | 5,134 | $ | 6,280 | ||||
| Long-term deferred income tax assets |
424 | 571 | ||||||
|
|
|
|
|
|||||
| Net deferred tax assets |
5,558 | 6,851 | ||||||
|
|
|
|
|
|||||
The Company assesses the realizability of its net deferred tax assets by evaluating all available evidence, both positive and negative, including (1) cumulative results of operations in recent years, (2) sources of recent losses, (3) estimates of future taxable income and (4) the length of net operating loss carryforward periods. From inception through 2012, the Company had determined that it was more likely than not that all of the net deferred tax assets would not be realized. As a result, the Company had recorded a full valuation allowance against those net deferred tax assets to reduce them to their estimated net realizable value as of December 31, 2012. As of December 31, 2013, the Company determined that it is more likely than not that a portion of the net deferred tax assets will be realized for federal and U.S. states except California and released the valuation allowance of $5.0 million. The Company continued establishing a full valuation allowance against the net deferred tax assets of California, Germany, Australia and Canada. As of December 31, 2014, the Company determined that it is more likely than not that German net deferred tax assets will be realized and released the German valuation allowance of $0.3 million. As a result, the Company has continued establishing a full valuation allowance against the net deferred tax assets of California, Australia and Canada.
F-34
Penumbra, Inc.
Notes to Consolidated Financial Statements (Continued)
15. Income Taxes (Continued)
The valuation allowance against net deferred tax assets changed as follows:
| Balance at December 31, 2012 |
$ | 8,742 | ||||
| Release of valuation allowance |
(4,962 | ) | ||||
| Other reserves and deferrals |
80 | |||||
|
|
|
| ||||
| Balance at December 31, 2013 |
3,860 | |||||
| Release of valuation allowance |
(321 | ) | ||||
| Other reserves and deferrals |
(594 | ) | ||||
|
|
|
| ||||
| Balance at December 31, 2014 |
$ | 2,945 | ||||
|
|
|
|
At December 31, 2014, the Company had approximately $17.8 million and $2.0 million of state and foreign net operating loss carryforwards, respectively, available to offset future taxable income. The state net operating loss carryforwards will begin to expire in 2017. At December 31, 2014, the Company had research credits available to offset federal and state tax liabilities in the amount of $0.7 million and $2.8 million, respectively. Federal credits will begin to expire in 2027 and California state tax credits have no expiration. As of December 31, 2014, the Company had fully used $22.6 million of federal net operating losses, which the Company had accumulated in its first six years of operations, from 2004 through 2009.
Internal Revenue Code sections 382 and 383 limit the use of net operating losses and business credits if there is a change in ownership. In 2009, the Company determined there were changes in ownership in 2004 and 2008. As of December 31, 2014, the section 382 limitation has no impact on the Company’s deferred tax assets prior to the valuation allowance.
As of December 31, 2014, the Company has $1.7 million in unrecognized tax benefits, of which $0.7 million would affect the Company’s effective tax rate if recognized.
A reconciliation of the change in the gross unrecognized tax benefits from January 1, 2013 to December 31, 2014, is as follows:
| December 31, | ||||||||
| 2013 | 2014 | |||||||
| Beginning Balance |
$ | 838 | $ | 1,325 | ||||
| Gross increase for tax positions of current year |
324 | 401 | ||||||
| Gross increase for tax positions of prior years |
163 | — | ||||||
| Settlements |
— | — | ||||||
| Lapse of statute of limitations |
— | — | ||||||
|
|
|
|
|
|||||
| Ending Balance |
$ | 1,325 | $ | 1,726 | ||||
|
|
|
|
|
|||||
The Company recognizes interest and penalties related to uncertain tax positions in income tax expense. As of December 31, 2014, the Company had $69,000 accrued interest and penalties related to uncertain tax positions.
The Company files U.S., state and foreign income tax returns in jurisdictions with varying statutes of limitations. Due to net operating loss and credit carryovers, the tax years ending December 31, 2004 through December 31, 2014 remain subject to examination by federal and state tax authorities. In Australia and Canada, tax years ending December 31, 2008 through December 31, 2014 generally remain subject to examination by tax authorities. In Germany, tax years ending December 31, 2013 through December 31, 2014 remain subject to examination by tax authorities. As of December 31, 2014, the Company is under examination by the German tax authority for tax years ending December 31, 2010 through December 31, 2012.
F-35
Penumbra, Inc.
Notes to Consolidated Financial Statements (Continued)
15. Income Taxes (Continued)
The Company classifies its unrecognized tax benefits as non-current income taxes payable, as no amounts are expected to be payable within the next 12 months.
16. Net Income (Loss) per Share of Common Stock attributable to Common Stockholders and Unaudited Pro Forma Net Income (Loss) per Share of Common Stock
A reconciliation of the numerator and denominator used in the calculation of the basic and diluted net income (loss) per share attributable to common stockholders is as follows (in thousands except share and per share amounts):
| Year Ended December 31, | Six Months Ended June 30, | |||||||||||||||
| 2013 | 2014 | 2014 | 2015 | |||||||||||||
| (unaudited) | ||||||||||||||||
| Net income (loss) per share: |
||||||||||||||||
| Numerator |
||||||||||||||||
| Net income (loss) |
$ | 4,099 | $ | 2,245 | $ | 1,657 | $ | (169 | ) | |||||||
| Less: Deemed dividend paid to preferred stockholders upon repurchase |
— | (6,344 | ) | — | — | |||||||||||
| Less: Undistributed income (loss) attributable to preferred stockholders |
(3,212 | ) | — | (1,302 | ) | 135 | ||||||||||
| Add: Undistributed loss attributable to preferred stockholders |
— | 3,266 | — | — | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Net income (loss) attributable to common stockholders—basic and diluted |
$ | 887 | $ | (833 | ) | $ | 355 | $ | (34 | ) | ||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Denominator |
||||||||||||||||
| Weighted average shares used to compute net income (loss) attributable to common stockholders |
4,304,396 | 4,609,375 | 4,520,898 | 5,000,375 | ||||||||||||
| Potential dilutive options, as calculated using treasury stock method |
2,021,394 | — | 2,101,306 | — | ||||||||||||
| Potential dilutive common stock warrants, as calculated using treasury stock method |
86,985 | — | 77,339 | — | ||||||||||||
| Potential dilutive restricted stock, as calculated using treasury stock method |
88,060 | — | 43,597 | — | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Weighted average shares used to compute net income (loss) attributable to common stockholders —Diluted |
6,500,835 | 4,609,375 | 6,743,140 | 5,000,375 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Net income (loss) per share attributable to common stockholders |
$ | 0.21 | $ | (0.18 | ) | $ | 0.08 | $ | (0.01 | ) | ||||||
|
|
|
|
|
|
|
|
|
|||||||||
| —Diluted |
$ | 0.14 | $ | (0.18 | ) | $ | 0.05 | $ | (0.01 | ) | ||||||
|
|
|
|
|
|
|
|
|
|||||||||
F-36
Penumbra, Inc.
Notes to Consolidated Financial Statements (Continued)
16. Net Income (Loss) per Share of Common Stock attributable to Common Stockholders and Unaudited Pro Forma Net Income (Loss) per Share of Common Stock (Continued)
The following outstanding shares of potentially dilutive securities were excluded from the computation of diluted net income (loss) per share of common stock for the periods presented, because the effect of including them would have been anti-dilutive:
| Year Ended December 31, | Six Months Ended June 30, | |||||||||||||||
| 2013 | 2014 | 2014 | 2015 | |||||||||||||
| (unaudited) | ||||||||||||||||
| Options to purchase common stock |
63,750 | 2,933,757 | 217,650 | 2,485,392 | ||||||||||||
| Restricted stock |
— | 367,126 | 286,000 | 755,771 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total |
63,750 | 3,300,883 | 503,650 | 3,241,163 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
Unaudited pro forma basic and diluted income (loss) per share is computed as follows (in thousands, except share and per share data):
| Year Ended December 31, 2014 |
Pro Forma Six Months Ended June 30, 2015 |
|||||||
| (unaudited) | ||||||||
| Unaudited pro forma net income (loss) per share: |
||||||||
| Numerator |
||||||||
| Net income (loss) used in computing pro forma net income (loss) per share attributable to common stockholders |
$ | 2,245 | $ | (169 | ) | |||
|
|
|
|
|
|||||
| Denominator |
||||||||
| Weighted average shares used to compute net income (loss) attributable to common stockholders—basic |
4,609,375 | 5,000,375 | ||||||
| Pro forma adjustments to reflect weighted average effect of assumed automatic conversion of preferred stock |
18,071,435 | 19,510,410 | ||||||
|
|
|
|
|
|||||
| Denominator for pro forma basic net income (loss) per share of common stock |
22,680,810 | 24,510,785 | ||||||
| Potential dilutive options, as calculated using treasury stock method |
2,161,223 | — | ||||||
| Potential dilutive common stock warrants, as calculated using treasury stock method |
118,897 | — | ||||||
| Potential dilutive restricted stock, as calculated using treasury stock method |
76,611 | — | ||||||
|
|
|
|
|
|||||
| Denominator for pro forma diluted net income (loss) per share of common stock |
25,037,541 | 24,510,785 | ||||||
|
|
|
|
|
|||||
| Pro forma net income (loss) per share—Basic |
$ | 0.10 | $ | (0.01 | ) | |||
|
|
|
|
|
|||||
| Pro forma net income (loss) per share—Diluted |
$ | 0.09 | $ | (0.01 | ) | |||
|
|
|
|
|
|||||
F-37
Penumbra, Inc.
Notes to Consolidated Financial Statements (Continued)
17. Geographic Areas and Product Sales
The Company’s revenue by geographic area, based on the destination to which the Company ships its products, was as follows (in thousands):
| Year Ended December 31, | Six Months Ended June 30, | |||||||||||||||
| 2013 | 2014 | 2014 | 2015 | |||||||||||||
| (unaudited) | ||||||||||||||||
| United States |
$ | 58,305 | $ | 82,965 | $ | 36,976 | $ | 53,970 | ||||||||
| Japan |
12,668 | 14,699 | 7,329 | 8,610 | ||||||||||||
| Other International |
17,875 | 27,846 | 13,338 | 18,683 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total |
$ | 88,848 | $ | 125,510 | $ | 57,643 | $ | 81,263 | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
The following table sets forth revenue by product category (in thousands):
| Year Ended December 31, | Six Months Ended June 30, | |||||||||||||||
| 2013 | 2014 | 2014 | 2015 | |||||||||||||
| (unaudited) | ||||||||||||||||
| Neuro |
$ | 81,343 | $ | 106,242 | $ | 50,068 | $ | 66,054 | ||||||||
| Peripheral Vascular |
7,505 | 19,268 | 7,575 | 15,209 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total |
$ | 88,848 | $ | 125,510 | $ | 57,643 | $ | 81,263 | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
The Company does not have significant long-lived assets outside the U.S.
18. Subsequent Events
The Company has evaluated subsequent events that occurred after December 31, 2014 through June 9, 2015, the date that the audited annual consolidated financial statements were issued, and determined that no additional subsequent events had occurred that would require recognition in these consolidated financial statements and all material subsequent events that require disclosure have been disclosed. The Company has evaluated subsequent events and transactions for potential recognition or disclosure through August 14, 2015, the date the unaudited interim financial statements were available to be issued.
F-38
shares
Common Stock

Prospectus
| J.P. Morgan | BofA Merrill Lynch | |
| Wells Fargo Securities | Canaccord Genuity | |
, 2015
PART II
INFORMATION NOT REQUIRED IN PROSPECTUS
Item 13. Other Expenses of Issuance and Distribution
| Amount to Be Paid | ||||
| SEC registration fee |
$ | 13,363 | ||
| FINRA filing fee |
$ | 17,750 | ||
| NYSE listing fee |
* | |||
| Transfer agent’s fees |
* | |||
| Printing and engraving expenses |
* | |||
| Legal fees and expenses |
* | |||
| Accounting fees and expenses |
* | |||
| Blue Sky fees and expenses |
* | |||
| Miscellaneous |
* | |||
|
|
|
|||
| Total |
$ | * | ||
|
|
|
|||
| * | To be completed by amendment. |
Each of the amounts set forth above, other than the SEC registration fee and the FINRA filing fee, is an estimate.
Item 14. Indemnification of Directors and Officers
Section 145 of the Delaware General Corporation Law provides that a corporation may indemnify directors and officers as well as other employees and individuals against expenses (including attorneys’ fees), judgments, fines and amounts paid in settlement actually and reasonably incurred by such person in connection with any threatened, pending or completed actions, suits or proceedings in which such person is made a party by reason of such person being or having been a director, officer, employee or agent to the registrant. The Delaware General Corporation Law provides that Section 145 is not exclusive of other rights to which those seeking indemnification may be entitled under any bylaw, agreement, vote of stockholders or disinterested directors or otherwise. Article VIII of the registrant’s certificate of incorporation provides for indemnification by the registrant of its directors, officers and employees to the fullest extent permitted by the Delaware General Corporation Law. The registrant has entered into indemnification agreements with each of its current directors and executive officers to provide these directors and executive officers additional contractual assurances regarding the scope of the indemnification set forth in the registrant’s amended and restated certificate of incorporation and amended and restated bylaws and to provide additional procedural protections. There is no pending litigation or proceeding involving a director or executive officer of the registrant for which indemnification is sought.
Section 102(b)(7) of the Delaware General Corporation Law permits a corporation to provide in its certificate of incorporation that a director of the corporation shall not be personally liable to the corporation or its stockholders for monetary damages for breach of fiduciary duty as a director, except for liability (i) for any breach of the director’s duty of loyalty to the corporation or its stockholders, (ii) for acts or omissions not in good faith or which involve intentional misconduct or a knowing violation of law, (iii) for unlawful payments of dividends or unlawful stock repurchases, redemptions or other distributions, or (iv) for any transaction from which the director derived an improper personal benefit. The registrant’s certificate of incorporation provides for such limitation of liability.
The registrant maintains standard policies of insurance under which coverage is provided (a) to its directors and officers against loss rising from claims made by reason of breach of duty or other wrongful act, and (b) to the registrant with respect to payments which may be made by the registrant to such officers and directors pursuant to the above indemnification provision or otherwise as a matter of law.
The proposed form of underwriting agreement filed as Exhibit 1 to this registration statement provides for indemnification of directors and officers of the registrant by the underwriters against certain liabilities.
II-1
Item 15. Recent Sales of Unregistered Securities
Since January 1, 2012, the registrant has sold the following securities without registration under the Securities Act of 1933:
Preferred Stock Issuances
On May 16, 2014, we issued and sold an aggregate of 4,545,455 shares of our Series F preferred stock at a price of $13.20 per share to a total of 38 accredited investors for aggregate consideration of $60,000,006.
Option Grants and Common Stock and Restricted Stock Issuances
Since January 1, 2012, we granted to our directors, officers, employees and consultants options to purchase an aggregate of 1,862,300 shares of common stock under our equity compensation plans, at exercise prices ranging from $4.49 to $22.04 per share.
Since January 1, 2012, we issued and sold to our directors, officers, employees and consultants an aggregate of 1,444,496 shares of common stock upon the exercise of options under our equity compensation plans at exercise prices ranging from $0.17 to $7.75 per share, for an aggregate amount of approximately $2,275,176.
Since January 1, 2012, we granted to our officers and employees an aggregate of 1,164,861 shares of restricted stock under our equity compensation plans at a fair market value ranging from $4.49 to $22.04 per share, for an aggregate amount of approximately $12,804,050.
Since January 1, 2012, we issued to our consultants an aggregate of 55,000 shares of common stock at a fair market value ranging from $4.49 to $5.99 per share, for an aggregate amount of approximately $288,950.
Since January 1, 2012, we issued and sold an aggregate of 25,568 shares of common stock upon the exercise of warrants at an exercise price of $0.01 per share, for an aggregate amount of approximately $256.
On May 6, 2015, we issued 7,500 shares of restricted stock in connection with the acquisition of a privately held company at a fair market value of $14.46 per share, for an aggregate amount of approximately $108,450.
None of the foregoing transactions involved any underwriters, underwriting discounts or commissions, or any public offering. Unless otherwise stated, the sales of the above securities were deemed to be exempt from registration under the Securities Act in reliance upon Section 4(a)(2) of the Securities Act (or Regulation D or Regulation S promulgated thereunder) or Rule 701 promulgated under Section 3(b) of the Securities Act as transactions by an issuer not involving any public offering or pursuant to benefit plans and contracts relating to compensation as provided under Rule 701. The recipients of the securities in each of these transactions represented their intentions to acquire the securities for investment only and not with a view to or for sale in connection with any distribution thereof, and appropriate legends were placed upon the stock certificates issued in these transactions.
II-2
Item 16. Exhibits and Financial Statement Schedules
(a) The following exhibits are filed as part of this registration statement:
| Exhibit Number |
Description | |
| 1.1* | Form of Underwriting Agreement | |
| 3.1 | Amended and Restated Certificate of Incorporation of Penumbra, Inc., as currently in effect | |
| 3.2* | Form of Amended and Restated Certificate of Incorporation of Penumbra, Inc., to be in effect upon completion of this offering | |
| 3.3 | Amended and Restated Bylaws of Penumbra, Inc., as currently in effect | |
| 3.4* | Form of Amended and Restated Bylaws of Penumbra, Inc., to be in effect upon completion of this offering | |
| 4.1* | Form of Common Stock Certificate | |
| 4.2 | Fourth Amended and Restated Investors’ Rights Agreement, by and among Penumbra, Inc., Adam Elsesser and Arani Bose and the investors listed on Exhibit A thereto, dated May 16, 2014 | |
| 5.1* | Opinion of Davis Polk & Wardwell LLP | |
| 10.1 | Lease for facilities at 1351 Harbor Bay Parkway, Alameda, California, dated November 28, 2007 and amended on May 7, 2008 and June 23, 2011 | |
| 10.2 | Lease for facilities at 1411 Harbor Bay Parkway, Alameda, California, dated September 11, 2014 | |
| 10.3 | Lease for facilities at 1321 Harbor Bay Parkway, Alameda, California, dated September 11, 2014 | |
| 10.4# | Distribution Agreement between Penumbra, Inc. and Medico’s Hirata, dated August 2, 2009, as amended | |
| 10.5† | 2014 Equity Incentive Plan, and forms of Restricted Stock Agreement, Stock Option Agreement and Early Exercise Stock Option Agreement | |
| 10.6† | 2011 Equity Incentive Plan, and forms of Restricted Stock Agreement, Stock Grant Agreement, Stock Option Agreement and Early Exercise Stock Option Agreement | |
| 10.7† | 2005 Stock Plan, and forms of Notice of Grant and Early Exercise Stock Option Agreement | |
| 10.8† | Form of Indemnification Agreement by and between Penumbra, Inc. and each of its directors and executive officers, as currently in effect | |
| 10.9† | Form of Indemnification Agreement by and between Penumbra, Inc. and each of its directors and executive officers, to be in effect immediately prior to the consummation of the offering | |
| 10.10† | Offer Letter with Adam Elsesser | |
| 10.11† | Offer Letter with Arani Bose | |
| 10.12† | Offer Letter with Sri Kosaraju | |
| 10.13† | Offer Letter with Daniel Davis | |
| 10.14† | Offer Letter with James Pray | |
| 10.15† | Offer Letter with Lynn Rothman | |
| 10.16† | Offer Letter with Robert Evans | |
| 10.17† | Form of Employee Nondisclosure and Assignment Agreement | |
| 10.18† | 2015 Employee Stock Purchase Plan | |
| 10.19† | Amended and Restated 2014 Equity Incentive Plan, and forms of Restricted Stock Agreement, Stock Option Agreement and Early Exercise Stock Option Agreement | |
| 21.1 | Subsidiaries of the registrant | |
| 23.1 | Consent of Independent Registered Public Accounting Firm | |
| 23.2* | Consent of Davis Polk & Wardwell LLP (included in Exhibit 5.1) | |
| 24.1 | Power of Attorney (included on signature page) | |
| * | To be filed by amendment. |
| † | Indicates management contract or compensatory plan. |
| # | Portions of this exhibit (indicated by asterisks) have been omitted pursuant to a request for confidential treatment and this exhibit has been filed separately with the SEC. |
II-3
(b) Financial Statement Schedules.
All financial statement schedules are omitted because the information called for is not required or is shown either in the consolidated financial statements or in the notes thereto.
Item 17. Undertakings
The undersigned registrant hereby undertakes:
(a) The undersigned registrant hereby undertakes to provide to the underwriter at the closing specified in the underwriting agreement certificates in such denominations and registered in such names as required by the underwriter to permit prompt delivery to each purchaser.
(b) Insofar as indemnification for liabilities arising under the Securities Act of 1933 may be permitted to directors, officers and controlling persons of the registrant pursuant to the provisions referenced in Item 14 of this registration statement, or otherwise, the registrant has been advised that in the opinion of the Securities and Exchange Commission such indemnification is against public policy as expressed in the Act and is, therefore, unenforceable. In the event that a claim for indemnification against such liabilities (other than the payment by the registrant of expenses incurred or paid by a director, officer, or controlling person of the registrant in the successful defense of any action, suit or proceeding) is asserted by such director, officer or controlling person in connection with the securities being registered hereunder, the registrant will, unless in the opinion of its counsel the matter has been settled by controlling precedent, submit to a court of appropriate jurisdiction the question of whether such indemnification by it is against public policy as expressed in the Act and will be governed by the final adjudication of such issue.
(c) The undersigned registrant hereby undertakes that:
| (1) | For purposes of determining any liability under the Securities Act of 1933, the information omitted from the form of prospectus filed as part of this registration statement in reliance upon Rule 430A and contained in a form of prospectus filed by the registrant pursuant to Rule 424(b)(1) or (4) or 497(h) under the Securities Act shall be deemed to be part of this registration statement as of the time it was declared effective. |
| (2) | For the purpose of determining any liability under the Securities Act of 1933, each post-effective amendment that contains a form of prospectus shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof. |
II-4
SIGNATURES
Pursuant to the requirements of the Securities Act of 1933, the registrant has duly caused this registration statement to be signed on its behalf by the undersigned, thereunto duly authorized, in the City of Alameda, State of California, on the 14 day of August, 2015.
| PENUMBRA, INC. | ||||
| By: |
/s/ Adam Elsesser | |||
| Name: | Adam Elsesser | |||
| Title: | Chairman, Chief Executive Officer and President | |||
KNOW ALL MEN BY THESE PRESENTS, that each person whose signature appears below constitutes and appoints Adam Elsesser, Sri Kosaraju and Robert Evans, and each of them, his or her true and lawful attorneys-in-fact and agents, with full power of substitution and resubstitution, for him or her and in his or her name, place and stead, in any and all capacities, to sign any and all amendments (including post-effective amendments) to this registration statement and any and all additional registration statements pursuant to Rule 462(b) of the Securities Act of 1933, and to file the same, with all exhibits thereto, and all other documents in connection therewith, with the Securities and Exchange Commission, granting unto each said attorney-in-fact and agents full power and authority to do and perform each and every act in person, hereby ratifying and confirming all that said attorneys-in-fact and agents or either of them or their or his or her substitute or substitutes may lawfully do or cause to be done by virtue hereof.
Pursuant to the requirements of the Securities Act of 1933, this registration statement has been signed by the following persons in the capacities and on the dates indicated.
| Signature |
Title |
Date | ||
| /s/ Adam Elsesser Adam Elsesser |
Chairman, Chief Executive Officer and President (Principal Executive Officer) | August 14, 2015 | ||
| /s/ Sri Kosaraju Sri Kosaraju |
Chief Financial Officer and Head of Strategy (Principal Financial and Accounting Officer) | August 14, 2015 | ||
| /s/ Arani Bose Arani Bose, M.D. |
Chief Innovator and Director | August 14, 2015 | ||
| /s/ Don Kassing Don Kassing |
Director | August 14, 2015 | ||
| /s/ Walter Wang Walter Wang |
Director | August 14, 2015 | ||
| /s/ Harpreet Grewal Harpreet Grewal |
Director | August 14, 2015 | ||
II-5
EXHIBIT INDEX
| Exhibit Number |
Description | |
| 1.1* | Form of Underwriting Agreement | |
| 3.1 | Amended and Restated Certificate of Incorporation of Penumbra, Inc., as currently in effect | |
| 3.2* | Form of Amended and Restated Certificate of Incorporation of Penumbra, Inc., to be in effect upon completion of this offering | |
| 3.3 | Amended and Restated Bylaws of Penumbra, Inc., as currently in effect | |
| 3.4* | Form of Amended and Restated Bylaws of Penumbra, Inc., to be in effect upon completion of this offering | |
| 4.1* | Form of Common Stock Certificate | |
| 4.2 | Fourth Amended and Restated Investors’ Rights Agreement, by and among Penumbra, Inc., Adam Elsesser and Arani Bose and the investors listed on Exhibit A thereto, dated May 16, 2014 | |
| 5.1* | Opinion of Davis Polk & Wardwell LLP | |
| 10.1 | Lease for facilities at 1351 Harbor Bay Parkway, Alameda, California, dated November 28, 2007 and amended on May 7, 2008 and June 23, 2011 | |
| 10.2 | Lease for facilities at 1411 Harbor Bay Parkway, Alameda, California, dated September 11, 2014 | |
| 10.3 | Lease for facilities at 1321 Harbor Bay Parkway, Alameda, California, dated September 11, 2014 | |
| 10.4# | Distribution Agreement between Penumbra, Inc. and Medico’s Hirata, dated August 2, 2009, as amended | |
| 10.5† | 2014 Equity Incentive Plan, and forms of Restricted Stock Agreement, Stock Option Agreement and Early Exercise Stock Option Agreement | |
| 10.6† | 2011 Equity Incentive Plan, and forms of Restricted Stock Agreement, Stock Grant Agreement, Stock Option Agreement and Early Exercise Stock Option Agreement | |
| 10.7† | 2005 Stock Plan, and forms of Notice of Grant and Early Exercise Stock Option Agreement | |
| 10.8† | Form of Indemnification Agreement by and between Penumbra, Inc. and each of its directors and executive officers, as currently in effect | |
| 10.9† | Form of Indemnification Agreement by and between Penumbra, Inc. and each of its directors and executive officers, to be in effect immediately prior to the consummation of the offering | |
| 10.10† | Offer Letter with Adam Elsesser | |
| 10.11† | Offer Letter with Arani Bose | |
| 10.12† | Offer Letter with Sri Kosaraju | |
| 10.13† | Offer Letter with Daniel Davis | |
| 10.14† | Offer Letter with James Pray | |
| 10.15† | Offer Letter with Lynn Rothman | |
| 10.16† | Offer Letter with Robert Evans | |
| 10.17† | Form of Employee Nondisclosure and Assignment Agreement | |
| 10.18† | 2015 Employee Stock Purchase Plan | |
| 10.19† | Amended and Restated 2014 Equity Incentive Plan, and forms of Restricted Stock Agreement, Stock Option Agreement and Early Exercise Stock Option Agreement | |
| 21.1 | Subsidiaries of the registrant | |
| 23.1 | Consent of Independent Registered Public Accounting Firm | |
| 23.2* | Consent of Davis Polk & Wardwell LLP (included in Exhibit 5.1) | |
| 24.1 | Power of Attorney (included on signature page) | |
| * | To be filed by amendment. |
| † | Indicates management contract or compensatory plan. |
| # | Portions of this exhibit (indicated by asterisks) have been omitted pursuant to a request for confidential treatment and this exhibit has been filed separately with the SEC. |