

As filed with the U.S. Securities and Exchange Commission on December 23, 2024
Registration No. 333-
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM
REGISTRATION STATEMENT
UNDER
THE SECURITIES ACT OF 1933
(Exact name of Registrant as specified in its charter)
| 6770 | Not Applicable | |||
| (State
or other jurisdiction of incorporation or organization) |
(Primary
Standard Industrial Classification Code Number) |
(I.R.S.
Employer Identification No.) |
Wilayah
Persekutuan,
(Address, including zip code, and telephone number, including area code, of Registrant’s principal executive offices)
(Name, address, including zip code, and telephone number, including area code, of agent for service)
| Copies of communications to: | ||
Jenny Chen-Drake, Esq. The
Law Offices of Jenny Chen-Drake (310) 358-0880 |
Ying Li, Esq. Guillaume de Sampigny, Esq. Hunter Taubman Fischer & Li LLC 950 Third Avenue, 19th Floor New York, NY 10022 (212) 530-2206 | |
Approximate date of commencement of proposed sale to the public: As soon as practicable after the effective date of this registration statement and the satisfaction or waiver of all other conditions under the Merger Agreement described herein.
If the securities being registered on this Form are being offered in connection with the formation of a holding company and there is compliance with General Instruction G, check the following box: ☐
If this Form is filed to register additional securities for an offering pursuant to Rule 462(b) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering: ☐
If this Form is a post-effective amendment filed pursuant to Rule 462(d) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering: ☐
If applicable, place an X in the box to designate the appropriate rule provision relied upon in conducting this transaction: ☐
Exchange Act Rule 13e-4(i) (Cross-Border Issuer Tender Offer) ☐
Exchange Act Rule 14d-1(d) (Cross-Border Third-Party Tender Offer) ☐
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933.
Emerging
growth company
If
an emerging growth company that prepares its financial statements in accordance with U.S. GAAP, indicate by check mark if the registrant
has elected not to use the extended transition period for complying with any new or revised financial accounting standards† provided
pursuant to Section 7(a)(2)(B) of the Securities Act.
| ____________ |
| † | The term “new or revised financial accounting standard” refers to any update issued by the Financial Accounting Standards Board to its Accounting Standards Codification after April 5, 2012. |
The Registrant hereby amends this Registration Statement on such date or dates as may be necessary to delay its effective date until the Registrant shall file a further amendment which specifically states that this Registration Statement shall thereafter become effective in accordance with Section 8(a) of the Securities Act of 1933 or until the Registration Statement shall become effective on such date as the U.S. Securities and Exchange Commission, acting pursuant to said Section 8(a), may determine.
SCHEDULE A —TABLE OF ADDITIONAL REGISTRANTS
Name |
State
or Other Jurisdiction of Incorporation or Organization |
I.R.S.
Employer Identification Number |
Address and Telephone Number of Registrant’s Principal Executive Offices | |||
| Alps Life Sciences Inc | The Cayman Islands | Not Applicable | Unit E-18-01 & E-18-02, Level 18, Icon Tower (East) No. 1, Jalan 1/68F, Jalan Tun Razak 50400 Kuala Lumpur Wilayah Persekutuan, Malaysia +603-2163 1113 |
(1) The agent for service for the Co-Registrant is:
Cogency Global Inc.
122 East 42nd Street, 18th Floor
New York, NY 10168
(212) 947-7200
The information in this proxy statement/prospectus is not complete and may be changed. We may not sell these securities until the U.S. Securities and Exchange Commission declares our registration statement effective. This proxy statement/prospectus is not an offer to sell these securities and is not soliciting an offer to buy these securities in any jurisdiction or state where the offer or sale is not permitted.
PRELIMINARY PROXY STATEMENT/PROSPECTUS
SUBJECT TO COMPLETION DATED DECEMBER 23, 2024
 |
 |
PROXY STATEMENT FOR SPECIAL MEETING OF
GLOBALINK INVESTMENT INC.
AND PROSPECTUS FOR UP TO 218,964,511 ORDINARY SHARES (INCLUDING SHARES
UNDERLYING WARRANTS) AND 6,035,000 WARRANTS OF
ALPS GLOBAL HOLDING PUBCO
To the Stockholders of Globalink Investment Inc.:
You are cordially invited to attend the special meeting of the stockholders (the “Special Meeting”) of Globalink Investment Inc., (“Globalink”, “we”, “our”, or “us”), which will be held at , Eastern Time, on , 2025. We will be holding the Special Meeting in a virtual meeting format at . Stockholders will NOT be able to attend the Special Meeting in person. This proxy statement/prospectus includes instructions on how to access the Special Meeting and how to listen, vote, and submit questions from home or any remote location with Internet connectivity. You or your proxy holder will be able to attend and vote at the Special Meeting by visiting https://www.cstproxy.com/ and using a control number assigned by Continental Stock Transfer & Trust Company. To register and receive access to the Special Meeting, registered stockholders and beneficial stockholders (those holding shares through a stock brokerage account or by a bank or other holder of record) will need to follow the instructions applicable to them provided in this proxy statement/prospectus.
Globalink is a Delaware company incorporated as a blank check company for the purpose of effecting a merger, share exchange, asset acquisition, share purchase, reorganization or similar business combination with one or more businesses or entities. The business combination will be completed through a two-step process consisting of the Redomestication Merger (as defined below) and the Acquisition Merger (as defined below). The Redomestication Merger and the Acquisition Merger are collectively referred to herein as the “Business Combination.”
On January 30, 2024, Globalink entered into a merger agreement (as amended and restated on May 20, 2024 and as may be further amended, restated or supplemented from time to time, the “Merger Agreement”), by and among Globalink, GL Sponsor LLC, a Delaware limited liability company, in the capacity as the representative from and after the effective time of the Acquisition Merger (as defined below) (the “Effective Time”) in accordance with the terms and conditions of the Merger Agreement (the “Parent Representative” or the “Sponsor”), Alps Global Holding Pubco, a Cayman Islands exempted company (“PubCo”), Alps Biosciences Merger Sub, a Cayman Islands exempted company and wholly-owned subsidiary of PubCo (“Merger Sub”), Alps Life Sciences Inc, a Cayman Islands exempted company (“Alps Holdco”) and Dr. Tham Seng Kong, an individual, in the capacity as the representative from and after the Effective Time for the shareholders of Alps Holdco as of immediately prior to the Effective Time in accordance with the terms and conditions of the Merger Agreement (the “Seller Representative”).
Pursuant to the terms of the Merger Agreement, the Business Combination between Globalink and Alps Holdco will be effected in two steps: (i) subject to the approval and adoption of the Merger Agreement by the stockholders of Globalink, Globalink will be merged with and into PubCo, with PubCo remaining as the surviving publicly traded entity (the “Redomestication Merger”); and (ii) Merger Sub will merge with and into Alps Holdco, resulting in Alps Holdco remaining as the surviving entity and being a wholly-owned subsidiary of PubCo (the “Acquisition Merger”). PubCo after the Business Combination is referred to in this proxy statement/prospectus as the “Combined Company.”
As a result of and upon the Closing, pursuant to the terms of the Merger Agreement, all of the outstanding shares of Alps Holdco will be cancelled in exchange for the right to receive PubCo ordinary shares equal to the Conversion Ratio (as defined in the Merger Agreement). The aggregate consideration for the Business Combination is US$1.6 billion, payable at the Closing in the form of newly issued PubCo ordinary shares, at $10.00 per share, of US$0.0001 par value each (the “Merger Consideration Shares”). The Merger Consideration Shares will be allocated pro rata with each shareholder of Alps Holdco receiving a number of PubCo ordinary shares determined in accordance with the terms of the Merger Agreement. Moreover, Alps Holdco’s shareholders will be eligible to receive up to an aggregate of 48,000,000 additional ordinary shares (the “Earnout Shares”) of the Combined Company upon the completion of certain milestones relating to the consolidated revenue of the Combined Company for five fiscal years following the Closing in accordance with the terms set forth in the Merger Agreement.
Subsequent to the execution of the Merger Agreement and as a condition and an inducement to Globalink and Alps Holdco to consummate the Business Combination, PubCo, Globalink and Alps Holdco have entered into subscription agreements (collectively, the “Subscription Agreements”) with certain investors (the “PIPE Investors”) dated June 4, 2024, June 5, 2024 and August 27, 2024, for an aggregate subscription amount of US$40.2 million in PubCo ordinary shares in a private placement to be consummated substantially concurrently with the Closing (the “PIPE Investment”). The purpose of the PIPE Investment is to raise additional capital for the business operations of the Combined Company. The obligations to consummate the transactions contemplated by the Subscription Agreements are conditioned upon, among other things, customary closing conditions and the consummation of the transactions contemplated by the Merger Agreement. The PIPE Investors have not received any payments from the Sponsor in connection with the PIPE Investment.
At the Closing of the Business Combination, such number of ordinary shares the Combined Company representing five percent (5%) of the Merger Consideration Shares are to be issued and held in escrow to satisfy any indemnification obligations incurred under the Merger Agreement (the “Escrow Shares”).
At the Special Meeting, Globalink stockholders will also be asked to consider and vote upon the following proposals:
| ● | to approve the Redomestication Merger. This proposal is referred to as the “Redomestication Merger Proposal;” | |
| ● | to approve the Acquisition Merger, the Merger Agreement and such other transactions contemplated by the Merger Agreement. This proposal is referred to as the “Acquisition Merger Proposal;” |
| ○ | to approve separate proposals, for purposes of complying with the Nasdaq Listing Rule 5635 (the “Nasdaq Listing Rule”), to issue (i) up to 208,000,000 PubCo ordinary shares pursuant to the Merger Agreement (including the ordinary shares issuable pursuant to certain earnout provisions in the Merger Agreement), and (ii) up to 4,020,000 PubCo ordinary shares to the PIPE Investors. These proposals are referred to as the “Nasdaq Proposals;” and |
| ○ | to approve the adjournment of the Special Meeting by the chairman thereof to a later date, if necessary, under certain circumstances, including for the purpose of soliciting additional proxies in favor of the foregoing proposals, in the event that Globalink does not receive the requisite stockholder vote to approve the proposals. This proposal is referred to as the “Adjournment Proposal” and, together with the Redomestication Merger Proposal, the Acquisition Merger Proposal, and the Nasdaq Proposals, the “Proposals.” |
Globalink’s units, common stock, public warrants and public rights are traded on the OTC Pink under the symbols “GLLIU,” “GLLI,” “GLLIW,” and “GLLIR,” respectively. On , 2025, the record date for the Special Meeting, the closing sale prices of Globalink’s units, common stock, public warrants and public rights was $ , $ , $ and $ , respectively.
If the Globalink stockholders approve the Redomestication Merger Proposal and the Acquisition Merger Proposal, immediately prior to the consummation of the Business Combination, all outstanding units of Globalink will separate into their individual components of Globalink common stock, Globalink rights and Globalink warrants and will cease separate existence and trading.
Upon the consummation of the Business Combination the current equity holdings of the Globalink stockholders shall be exchanged as follows:
| (i) | Each share of Globalink common stock, par value $0.001 per share, issued and outstanding immediately prior to the effective time of the Redomestication Merger (other than any redeemed shares), will automatically be cancelled and cease to exist and for each share of Globalink common stock, PubCo shall issue to its holder (other than Globalink stockholders who exercise their redemption rights in connection with the Business Combination) one validly issued PubCo ordinary share, which shall be fully paid; | |
| (ii) | Each Globalink warrant to purchase one-half (1/2) of one share of Globalink common stock issued and outstanding immediately prior to effective time of the Redomestication Merger will convert into one warrant to purchase one-half (1/2) of one PubCo ordinary share (“PubCo warrant”) (or equivalent portion thereof). The PubCo warrants will have substantially the same terms and conditions as set forth in the Globalink warrants; and |
| (iii) | The holders of Globalink rights (convertible into one-tenth (1/10) of one share of Globalink common stock) issued and outstanding immediately prior to the effective time of the Redomestication Merger will obtain the right to receive one-tenth (1/10) of one PubCo ordinary share (“PubCo right”) in exchange for the cancellation of each Globalink right; provided, however, that no fractional shares will be issued and all fractional shares will be rounded to the nearest whole share. |
It is anticipated that upon completion of the Business Combination, assuming no redemptions, Globalink’s public stockholders would retain an ownership interest of approximately 0.2% in PubCo, Globalink public rights holders would retain an ownership interest of approximately 0.8% in PubCo for a total ownership interest held by Globalink’s public stockholders of 0.8%, the PIPE Investors will own approximately 2.4% of PubCo, assuming the PIPE Investors will hold 4,020,000 PubCo ordinary shares, the Parent Representative and officers and directors of Globalink will retain an ownership interest of approximately 2.4% of PubCo, Public Gold Marketing Sdn. Bhd., a Malaysian private limited company and an affiliate of the Sponsor (“PGM”), the holder of 570,000 private units, which include an aggregate of 627,000 shares of Globalink common stock, will retain an ownership interest of approximately 0.4% of PubCo, IBDC Asia Sdn. Bhd., an advisory firm to Alps, will own approximately 0.9% of PubCo, representing finder’s fees payable in PubCo ordinary shares with value equal to 1% of the aggregate consideration for the Business Combination of US$1.6 billion, and the Alps Holdco’ shareholders will own approximately 93.8% of PubCo. These percentages are calculated based on a number of assumptions (described in the accompanying proxy statement/prospectus) and are subject to adjustment in accordance with the terms of the Merger Agreement. If the actual facts are different from these assumptions (which they are likely to be), the percentage ownership retained by the respective parties will be different. See “Frequently Used Terms — Share Calculations and Ownership Percentages” and “Unaudited Pro Forma Condensed Combined Financial Information and Notes.”
PubCo intends to apply for the listing of its ordinary shares and warrants on the Nasdaq under the symbols “ALPS” and “ALPSW”, respectively, in connection with the closing of the Business Combination. We cannot assure you that the PubCo ordinary shares and PubCo warrants will be approved for listing on Nasdaq.
The approval of the Redomestication Merger Proposal, the Acquisition Merger Proposal, the Nasdaq Proposals, and the Adjournment Proposal each require the affirmative vote of the holders of a majority of the shares of Globalink common stock cast by the stockholders represented in person by virtual attendance or by proxy and entitled to vote thereon at the Special Meeting. If either of the Redomestication Merger Proposal or the Acquisition Merger Proposal is not approved, the Nasdaq Proposals will not be presented to the Globalink stockholders for a vote. The approval of the Redomestication Merger Proposal and the Acquisition Merger Proposal are preconditions to the consummation of the Business Combination.
Only holders of record of shares of Globalink common stock at the close of business on , 2025 (the “Record Date”), are entitled to notice of the Special Meeting and the right to vote and have their votes counted at the Special Meeting and any adjournments or postponements of the Special Meeting. Pursuant to the Globalink Charter, Globalink is providing its public stockholders with the opportunity to redeem, upon the closing of the Business Combination, shares of Globalink common stock then held by them for cash equal to their pro rata share of the aggregate amount on deposit (as of two business days prior to the closing of the Business Combination) in the trust account maintained by Continental Stock Transfer & Trust Company for Globalink’s public stockholders (the “Trust Account”) that holds the proceeds (including interest but less franchise and income taxes payable) of the initial public offering of Globalink consummated on December 9, 2021 (the “IPO”). For illustrative purposes, based on funds in the Trust Account of approximately $3.33 million as of December 6, 2024, the estimated per share redemption price would have been approximately $11.98.
Public stockholders may elect to redeem their shares regardless of whether they vote for, against or abstain on the Redomestication Merger Proposal or the Acquisition Merger Proposal. A public stockholder, together with any of his, her or its affiliates or any other person with whom it is acting in concert or as a “group” (as defined under Section 13 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), will be restricted from redeeming in the aggregate 20% or more of his, her or its shares of common stock or, if part of such a group, the group’s shares of common stock included in the units sold in the IPO of Globalink. Holders of outstanding units must separate the underlying securities prior to exercising redemption rights with respect to the public shares.
The Sponsor hold an aggregate of 2,835,000 shares of Globalink common stock as of the date of this proxy statement/prospectus. The Sponsor will receive a positive rate of return so long as the market price of the common stock is at least $0.01 per share, even if public stockholders experience a negative rate of return in the Combined Company. The Sponsor, its affiliates and promoters are not receiving compensation in connection with the Business Combination and the PIPE Investment. The Sponsor, its affiliates and promoters will not receive any securities of Globalink or PubCo in connection with the Business Combination and the PIPE Investment. However, the Sponsor is expected to own an equity interest of approximately 1.7% in the Combined Company after the consummation of the Business Combination. For an illustration of the dilutive effect the Business Combination, the PIPE Investment, and related transactions would have on the holders of public shares who hold the securities until the consummation of the Business Combination, see “Summary of the Proxy Statement/Prospectus — Ownership of the Combined Company After the Closing.” As of the date hereof, the deadline that Globalink has to consummate its initial business combination has been extended for a total of eighteen times, and a total of US$1,950,000 has been deposited into the Trust Account in connection with the previous extensions. In connection with funding the extensions, Globalink has issued an aggregate of US$3.85 million in promissory notes to PGM, which are payable upon the Closing. In the event of a liquidation, PGM has waived its rights to any liquidation distributions through the Insider Letter. PGM is an affiliate of the Sponsor as its 95% equity holder has a familial relationship with the control person of the Sponsor. As a result, the Sponsor has an additional interest in causing Globalink to consummate its initial business combination due to its familial relationship with PGM.
Upon the consummation of an initial business combination by Globalink, other than the PubCo ordinary shares that the Initial Stockholders are entitled to upon conversion of their shares of Globalink common stock at the completion of the Redomestication Merger, no additional compensation will be awarded to, earned by, or paid to the Sponsor, its affiliates or any promoters for all services rendered or to be rendered to Globalink and its affiliates. The Sponsor is expected to own an equity interest of approximately 1.7% in the Combined Company after the consummation of the Business Combination. See “The Special Meeting — Recommendation of the Board.”
To induce Chardan Capital Markets, LLC (“Chardan”), the representative of the underwriters for the initial public offering of Globalink (the “IPO”) to proceed with the IPO, and recognizing the economic benefit the IPO would confer upon the Sponsor, officers and directors of Globalink (the “Initial Stockholders”) and PGM, through a letter agreement dated December 6, 2021, by and among Globalink Investment Inc., the Sponsor, PGM and each of Globalink’s directors and officers (the “Insider Letter”), each of the Initial Stockholders and PGM have agreed to waive their redemption rights with respect to any shares of Globalink common stock they may hold in connection with the consummation of the Business Combination, and such shares will be excluded from the pro rata calculation used to determine the per-share redemption price. Currently, the Sponsor, the directors and officers of Globalink and PGM collectively own an aggregate of 92.6% of the issued and outstanding shares of Globalink common stock. The Sponsor and the directors and officers of Globalink have agreed to vote any shares of common stock owned by them in favor of the Redomestication Merger Proposal and the Acquisition Merger Proposal.
Globalink is providing the accompanying proxy statement/prospectus to its stockholders in connection with the solicitation of proxies to be voted at the Special Meeting and at any adjournments or postponements of the Special Meeting. Whether or not you plan to attend the Special Meeting, Globalink urges you to read the accompanying proxy statement/prospectus (and any documents incorporated into the accompanying proxy statement/prospectus by reference) carefully. Please pay particular attention to the section entitled “Risk Factors” beginning on page 52 for a discussion of information that should be considered in connection with an investment in the Combined Company’s securities.
After careful consideration, Globalink’s board of directors has unanimously approved the Merger Agreement and unanimously recommends that Globalink’s stockholders vote “FOR” approval of each of the Proposals. When you consider the recommendation by Globalink’s board of directors of these Proposals, you should keep in mind that the Sponsor and Globalink’s directors and officers have interests in the Business Combination that may conflict or differ from your interests as a stockholder of Globalink. See the section titled “The Acquisition Merger Proposal (Proposal 2) — Interests of Certain Persons in the Business Combination.” Additionally, directors and officers of Alps Holdco have interests that may conflict or differ from your interests as a stockholder of Globalink as certain directors and officers of Alps Holdco are expected to receive certain number of PubCo ordinary shares issued as part of the Merger Consideration Shares. The Globalink Board does not plan to obtain a final fairness opinion related to the Business Combination. The Board has unanimously determined that the Business Combination is in the best interests of, and advisable to, the Globalink stockholders and recommends that the Globalink stockholders adopt the Merger Agreement and approve the Business Combination.
As of the date of this proxy statement/prospectus, a total of 3,445,000 shares of common stock, or approximately 92.6% of the outstanding shares, were subject to the Insider Letter and the Globalink Support Agreements. The approval of the Redomestication Merger Proposal and the Acquisition Merger Proposal does not require the votes from at least a majority of unaffiliated security holders of Globalink. As such, as the vote to approve the Redomestication Merger Proposal and the Acquisition Merger Proposal is a majority of the then outstanding shares of common stock present and entitled to vote at the Special Meeting, assuming only the minimum number of shares of common stock to constitute a quorum is present, no public stockholder must vote in favor of the Redomestication Merger Proposal or the Acquisition Merger Proposal for them to be approved.
Your vote is very important. To ensure your representation at the Special Meeting, please complete and return the enclosed proxy card or submit your proxy by following the instructions contained in the accompanying proxy statement/prospectus and on your proxy card. Please submit your proxy promptly whether or not you expect to participate in the meeting. Submitting a proxy now will NOT prevent you from being able to vote online during the virtual Special Meeting. If you hold your shares in “street name,” you should instruct your broker, bank or other nominee how to vote in accordance with the voting instruction form you receive from your broker, bank or other nominee. If you return your proxy card signed and without indicating how you wish to vote, your proxy will be voted in favor of each of the Proposals presented at the Special Meeting and for the election of each of the directors proposed by Globalink for election. If you fail to return your proxy card or fail to submit your proxy by telephone or over the Internet, or fail to instruct your bank, broker or other nominee how to vote, and do not attend the Special Meeting in person online, the effect will be that your shares will not be counted for purposes of determining whether a quorum is present at the Special Meeting, and, if a quorum is present, will have no effect on the Proposals. If you are a stockholder of record and you attend the Special Meeting and wish to vote during the Special Meeting, you may withdraw your proxy and vote during the Special Meeting.
TO EXERCISE YOUR REDEMPTION RIGHTS, YOU MUST (1) IF YOU HOLD SHARES OF GLOBALINK COMMON STOCK THROUGH UNITS, SEPARATE YOUR UNITS INTO THE UNDERLYING SHARES OF GLOBALINK COMMON STOCK, WARRANTS AND RIGHTS PRIOR TO EXERCISING YOUR REDEMPTION RIGHTS WITH RESPECT TO THE PUBLIC SHARES, (2) SUBMIT A WRITTEN REQUEST, INCLUDING THE LEGAL NAME, PHONE NUMBER AND ADDRESS OF THE BENEFICIAL OWNER OF THE SHARES FOR WHICH REDEMPTION IS REQUESTED, TO THE TRANSFER AGENT AT LEAST TWO BUSINESS DAYS PRIOR TO THE DATE OF THE SPECIAL MEETING, THAT YOUR PUBLIC SHARES BE REDEEMED FOR CASH AND (3) DELIVER YOUR STOCK CERTIFICATES (IF ANY) AND OTHER REDEMPTION FORMS TO THE TRANSFER AGENT, PHYSICALLY OR ELECTRONICALLY USING THE DEPOSITORY TRUST COMPANY’S DWAC (DEPOSIT/WITHDRAWAL AT CUSTODIAN) SYSTEM, IN EACH CASE, IN ACCORDANCE WITH THE PROCEDURES AND DEADLINES DESCRIBED IN THE PROXY STATEMENT/PROSPECTUS. IF THE BUSINESS COMBINATION IS NOT CONSUMMATED, THEN THE PUBLIC SHARES WILL NOT BE REDEEMED FOR CASH. IF YOU HOLD THE SHARES IN STREET NAME, YOU WILL NEED TO INSTRUCT THE ACCOUNT EXECUTIVE AT YOUR BANK, BROKER OR OTHER NOMINEE TO WITHDRAW THE SHARES FROM YOUR ACCOUNT IN ORDER TO EXERCISE YOUR REDEMPTION RIGHTS. SEE “THE SPECIAL MEETING — REDEMPTION RIGHTS” IN THE PROXY STATEMENT /PROSPECTUS FOR MORE SPECIFIC INSTRUCTIONS.
PubCo will be a “foreign private issuer” as defined in the Securities Exchange Act of 1934, as amended (the “Exchange Act”) and will be exempt from certain rules under the Exchange Act that impose certain disclosure obligations and procedural requirements for proxy solicitations under Section 14 of the Exchange Act. In addition, PubCo’s officers, directors and principal shareholders will be exempt from the reporting and “short-swing” profit recovery provisions under Section 16 of the Exchange Act. Moreover, PubCo will not be required to file periodic reports and financial statements with the U.S. Securities and Exchange Commission as frequently or as promptly as U.S. companies whose securities are registered under the Exchange Act.
PubCo will be an “emerging growth company” as defined in the Jumpstart Our Business Startups Act of 2012 and is therefore eligible to take advantage of certain reduced reporting requirements otherwise applicable to other public companies.
Neither the U.S. Securities and Exchange Commission nor any state securities commission has approved or disapproved of the securities to be issued in the Business Combination or otherwise or passed upon the adequacy or accuracy of the accompanying proxy statement/prospectus. Any representation to the contrary is a criminal offense.
The accompanying proxy statement/prospectus is dated , 2025, and is first being mailed to stockholders of Globalink on or about , 2025.
On behalf of Globalink’s board of directors, I thank you for your support and we look forward to the successful consummation of the Business Combination.
| Sincerely, | |
Say Leong Lim | |
| Chief Executive Officer and Chairman of the Board of Directors | |
| Globalink Investment Inc. |
Globalink Investment Inc.
200 Continental Drive, Suite 401
Newark, Delaware
+6012 405 0015
NOTICE OF SPECIAL MEETING OF STOCKHOLDERS
TO BE HELD ON , 2025
To the Stockholders of Globalink Investment Inc.:
NOTICE IS HEREBY GIVEN that a special meeting of stockholders of Globalink Investment Inc., a Delaware corporation (“Globalink,” “we”, “our,” or “us”), will be held virtually on , 2025 at Eastern Time in a virtual meeting format at (the “Special Meeting”). The Globalink Board of Directors has determined to convene and conduct the Special Meeting in a virtual meeting format at . Stockholders will NOT be able to attend the Special Meeting in person. The accompanying proxy statement/prospectus includes instructions on how to access the virtual Special Meeting and how to listen and vote from home or any remote location with Internet connectivity. You or your proxy holder will be able to attend and vote at the Special Meeting by visiting https://www.cstproxy.com/ and using a control number assigned by Continental Stock Transfer & Trust Company. The Special Meeting will be held for the purpose of considering and voting on the proposals (the “Proposals”) described below and in the accompanying proxy statement/prospectus. To register and receive access to the virtual meeting, registered stockholders and beneficial stockholders of Globalink (those holding shares through a stock brokerage account or by a bank or other holder of record) will need to follow the instructions applicable to them provided in the accompanying joint proxy statement/consent solicitation statement/prospectus.
On January 30, 2024, Globalink entered into a merger agreement (as amended and restated on May 20, 2024 and as may be further amended, restated or supplemented from time to time, the “Merger Agreement”), by and among Globalink, GL Sponsor LLC, a Delaware limited liability company, in the capacity as the representative from and after the effective time of the Acquisition Merger (as defined below) (the “Effective Time”) in accordance with the terms and conditions of the Merger Agreement (the “Parent Representative” or the “Sponsor”), Alps Global Holding Pubco, a Cayman Islands exempted company (“PubCo”), Alps Biosciences Merger Sub, a Cayman Islands exempted company and wholly-owned subsidiary of PubCo (“Merger Sub”), Alps Life Sciences Inc, a Cayman Islands exempted company (“Alps Holdco”) and Dr. Tham Seng Kong, an individual, in the capacity as the representative from and after the Effective Time for the shareholders of Alps Holdco as of immediately prior to the Effective Time in accordance with the terms and conditions of the Merger Agreement (the “Seller Representative”).
Pursuant to the terms of the Merger Agreement, the Business Combination between Globalink and Alps Holdco will be effected in two steps: (i) subject to the approval and adoption of the Merger Agreement by the stockholders of Globalink, Globalink will be merged with and into PubCo, with PubCo remaining as the surviving publicly traded entity (the “Redomestication Merger”); and (ii) Merger Sub will merge with and into Alps Holdco, resulting in Alps Holdco remaining as the surviving entity and being a wholly-owned subsidiary of PubCo (the “Acquisition Merger”). PubCo after the Business Combination is referred to in this proxy statement/prospectus as the “Combined Company.”
As a result of and upon the Closing, pursuant to the terms of the Merger Agreement, all of the outstanding shares of Alps Holdco will be cancelled in exchange for the right to receive PubCo ordinary shares equal to the Conversion Ratio (as defined in the Merger Agreement). The aggregate consideration for the Business Combination is US$1.6 billion, payable at the Closing in the form of newly issued PubCo ordinary shares, at $10.00 per share (the “Merger Consideration Shares”). The Merger Consideration Shares will be allocated pro rata with each shareholder of Alps Holdco receiving a number of PubCo ordinary shares determined in accordance with the terms of the Merger Agreement. Moreover, Alps Holdco’s shareholders will be eligible to receive up to an aggregate of 48,000,000 additional ordinary shares (the “Earnout Shares”) of the Combined Company upon the completion of certain milestones relating to the consolidated revenue of the Combined Company for five fiscal years following the Closing in accordance with the terms set forth in the Merger Agreement.
Subsequent to the execution of the Merger Agreement and as a condition and an inducement to Globalink and Alps Holdco to consummate the Business Combination, PubCo, Globalink and Alps Holdco have entered into subscription agreements (collectively, the “Subscription Agreements”) with certain investors (the “PIPE Investors”) dated June 4, 2024, June 5, 2024 and August 27, 2024, for an aggregate subscription amount of US$40.2 million in ordinary shares of PubCo in a private placement to be consummated substantially concurrently with the Closing (the “PIPE Investment”). The purpose of the PIPE Investment is to raise additional capital for the business operations of the Combined Company. The obligations to consummate the transactions contemplated by the Subscription Agreements are conditioned upon, among other things, customary closing conditions and the consummation of the transactions contemplated by the Merger Agreement. The PIPE Investors have not received any payments from the Sponsor in connection with the PIPE Investment.
At the Closing of the Business Combination, such number of ordinary shares the Combined Company representing five percent (5%) of the Merger Consideration Shares are to be issued and held in escrow to satisfy any indemnification obligations incurred under the Merger Agreement (the “Escrow Shares”).
At the Special Meeting, Globalink’s stockholders will be asked to consider and vote upon the following Proposals):
| I. | The Redomestication Merger Proposal (Proposal 1) — To consider and vote upon a proposal to approve the merger of Globalink with and into PubCo, a Cayman Islands exempted company (“PubCo”), with PubCo remaining as the surviving publicly traded entity. This Proposal is referred to as the “Redomestication Merger Proposal.” | |
| II. | The Acquisition Merger Proposal (Proposal 2) — To consider and vote upon a proposal to approve and adopt the merger agreement dated January 30, 2024 (as amended and restated on May 20, 2024 and may be further amended, restated or supplemented from time to time, the “Merger Agreement”), by and among (i) Globalink, (ii) GL Sponsor LLC, a Delaware limited liability company, in the capacity as the representative from and after the effective time of the Business Combination (as defined below) (the “Effective Time”) in accordance with the terms and conditions of the Merger Agreement (the “Parent Representative” or the “Sponsor”), (iii) Alps Global Holding Pubco, a Cayman Islands exempted company (“PubCo”), (iv) Alps Biosciences Merger Sub, a Cayman Islands exempted company and wholly-owned subsidiary of PubCo (“Merger Sub”), (v) Alps Life Sciences Inc, a Cayman Islands company (“Alps Holdco”) and (vi) Dr. Tham Seng Kong, an individual, in the capacity as the representative from and after the Effective Time for the shareholders of Alps Holdco as of immediately prior to the Effective Time in accordance with the terms and conditions of the Merger Agreement (the “Seller Representative”) and (b) the transactions contemplated thereunder, including the merger of Merger Sub with and into Alps Holdco, with Alps Holdco continuing as the surviving company and as a wholly-owned subsidiary of PubCo, and the issuance of PubCo ordinary shares as merger consideration thereunder, as described in more detail in the accompanying proxy statement/prospectus (together with the Redomestication Merger and the other transactions contemplated by the Merger Agreement, the “Business Combination” and PubCo after the Business Combination, the “Combined Company”).
The Acquisition Merger Proposal is described in more detail in the accompanying proxy statement/prospectus under the heading “The Acquisition Merger Proposal (Proposal 2).” A copy of the Merger Agreement is attached to the accompanying proxy statement /prospectus as Annex A.
After the consummation of the Business Combination, |
| ● | Globalink shall merge with and into PubCo and Globalink, the Delaware corporation, shall cease to exist and PubCo shall be the surviving company and the name of the surviving company will be “ALPS Group;” | |
| ● | the authorized shares of the surviving company shall change from 500,000,000 shares of Globalink Common Stock and 5,000,000 shares of Globalink preferred stock to 495,000,000 PubCo ordinary shares of US$0.0001 par value each and 5,000,000 PubCo preferred shares of US$0.0001 par value each; | |
| ● | provisions specific to the Combined Company’s status as a special purpose acquisition company shall be eliminated making the Combined Company’s corporate existence of unlimited duration; and | |
| ● | certain other changes that our board of directors deems appropriate for a public operating company will be made. |
A summary of certain material differences between the amended and restated certificate of incorporation, as amended and currently in effect (the “Globalink Charter”) and the PubCo A&R Memorandum and Articles is presented separately and set forth in the section of this proxy statement/prospectus entitled “Comparison of Corporate Governance and Stockholders’ / Shareholders’ Rights.” A complete copy of the PubCo A&R Memorandum and Articles is attached to the proxy statement/prospectus as Annex B. You are encouraged to read them in their entirety.
| III. | The Nasdaq Proposals (Proposals 3) — To consider and approve upon separate proposals, for purposes of complying with the Nasdaq Listing Rule, to issue (i) up to 208,000,000 PubCo ordinary shares pursuant to the Merger Agreement and (ii) up to 4,020,000 PubCo ordinary shares to the PIPE Investors. The Nasdaq Proposals are described in more detail in the accompanying proxy statement/prospectus under the heading “The Nasdaq Proposals (Proposals 3).” | |
| IV. | The Adjournment Proposal (Proposal 4) — To consider and approve the adjournment of the Special Meeting to a later date, if necessary. We refer to this proposal as the “Adjournment Proposal.” The Adjournment Proposal is described in more detail in the accompanying proxy statement/prospectus under the heading “The Adjournment Proposal (Proposal 4).” |
Only holders of record of shares of Globalink common stock at the close of business on , 2025 (the “Record Date”), are entitled to notice of the Special Meeting and the right to vote and have their votes counted at the Special Meeting and any adjournments or postponements of the Special Meeting. Pursuant to the Globalink Charter, Globalink is providing its public stockholders with the opportunity to redeem, upon the closing of the Business Combination, shares of Globalink common stock then held by them for cash equal to their pro rata share of the aggregate amount on deposit (as of two business days prior to the closing of the Business Combination) in the Trust Account that holds the proceeds (including interest but less franchise and income taxes payable) of the IPO of Globalink. For illustrative purposes, based on funds in the Trust Account of approximately $3.33 million as of December 6, 2024, the estimated per share redemption price would have been approximately $11.98.
Public stockholders may elect to redeem their shares regardless of whether they vote for, against or abstain on the Redomestication Merger Proposal or the Acquisition Merger Proposal. A public stockholder, together with any of his, her or its affiliates or any other person with whom it is acting in concert or as a “group” (as defined under Section 13 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), will be restricted from redeeming in the aggregate 20% or more of his, her or its shares or, if part of such a group, the group’s shares of common stock included in the units sold in the IPO of Globalink. Holders of outstanding units must separate the underlying securities prior to exercising redemption rights with respect to the public shares. To induce Chardan, the representative of the underwriters for Globalink’s IPO to proceed with the IPO, and recognizing the economic benefit the IPO would confer upon the Initial Stockholders and PGM, through the Insider Letter, each of the Initial Stockholders and PGM have agreed to waive their redemption rights with respect to any shares of Globalink common stock they may hold in connection with the consummation of the Business Combination, and such shares will be excluded from the pro rata calculation used to determine the per-share redemption price. Currently, the Sponsor and the directors and officers of Globalink collectively own an aggregate of 92.6% of the issued and outstanding shares of Globalink common stock. The Sponsor, the directors and officers of Globalink, and PGM have agreed to vote any shares of common stock owned by them in favor of the Redomestication Merger Proposal, the Acquisition Merger Proposal, and the other Proposals.
Approval of the Redomestication Merger Proposal, the Acquisition Merger Proposal, the Nasdaq Proposals and the Adjournment Proposal will each require the affirmative vote of the holders of a majority of the issued and outstanding shares of common stock present in person by virtual attendance or represented by proxy and entitled to vote at the Special Meeting or any adjournment thereof. The Redomestication Merger Proposal and the Acquisition Merger Proposal are dependent upon each other. It is important for you to note that if either of the Redomestication Merger Proposal or the Acquisition Merger Proposal is not approved, Globalink will not consummate the Business Combination. If either the Redomestication Merger Proposal or the Acquisition Merger Proposal is not approved, the Redomestication Merger Proposal, and the Nasdaq Proposals will not be presented to the Globalink stockholders for a vote. It is important for you to note that, in the event that either of the Redomestication Merger Proposal or the Acquisition Merger Proposal is not approved, Globalink will not consummate the Business Combination.
Globalink currently has until January 9, 2025 to complete its initial business combination, or it will be required to dissolve and liquidate. If Globalink anticipates that it may not be able to consummate its initial business combination in time, Globalink may, by resolutions of its board of directors, if requested by the Sponsor, extend the period of time to consummate a business combination on a monthly basis to until June 9, 2025, subject to the Sponsor depositing additional funds into the Trust Account.
On December 10, 2024, Globalink received a delisting determination letter from the Nasdaq Stock Market (“Nasdaq”) as the 36-month anniversary from its IPO has passed on December 9, 2024, and Globalink are no longer in compliance with Nasdaq Listing Rule IM 5101-2(b). Globalink’s securities were suspended from trading and delisted from Nasdaq on December 17, 2024. As of the same date, Globalink’s securities started trading on OTC Pink. There could be substantial risks to Globalink and PubCo the as a result of Globalink’s non-compliance with this rule. See “Risk Factors — Risks Related to Globalink’s Business and the Business Combination — Our securities were suspended from trading and delisted from Nasdaq on December 17, 2024, following receipt of a delisting determination letter from Nasdaq on December 10, 2024. This could have significant material adverse consequences on us and our securities, including that it will negatively impact our ability to complete a business combination, will limit investors’ ability to make transactions in our securities and could subject us to additional trading restrictions.”
Investing in Globalink’s securities involves a high degree of risk. See “Risk Factors” beginning on page 52 for a discussion of information that should be considered in connection with an investment in Globalink’s securities.
YOUR VOTE IS VERY IMPORTANT. PLEASE VOTE YOUR SHARES PROMPTLY.
Whether or not you plan to participate in the virtual Special Meeting, please complete, date, sign and return the enclosed proxy card without delay, or submit your proxy through the internet or by telephone as promptly as possible in order to ensure your representation at the Special Meeting no later than the time appointed for the Special Meeting or adjourned meeting. Voting by proxy will not prevent you from voting your shares of common stock online if you subsequently choose to participate in the virtual Special Meeting. Please note, however, that if your shares are held of record by a broker, bank or other agent and you wish to vote at the Special Meeting, you must obtain a proxy issued in your name from that record. Only stockholders of record at the close of business on the record date may vote at the Special Meeting or any adjournment or postponement thereof. If you fail to return your proxy card or fail to instruct your bank, broker or other nominee how to vote, and do not participate in the virtual Special Meeting, your shares will not be counted for purposes of determining whether a quorum is present at, and the number of votes voted at, the Special Meeting.
You may revoke a proxy at any time before it is voted at the Special Meeting by executing and returning a proxy card dated later than the previous one, by participating in the virtual Special Meeting and casting your vote by hand or by ballot (as applicable) or by submitting a written revocation to Continental Stock Transfer & Trust Company, that is received by the proxy agent before we take the vote at the Special Meeting. If you hold your shares through a bank or brokerage firm, you should follow the instructions of your bank or brokerage firm regarding revocation of proxies.
Globalink’s board of directors has unanimously approved the Merger Agreement and unanimously recommends that Globalink’s stockholders vote “FOR” approval of each of the Proposals. When you consider the recommendation by Globalink’s board of directors of these Proposals, you should keep in mind that the Sponsor and Globalink’s directors and officers have interests in the Business Combination that may conflict or differ from your interests as a stockholder. See the section titled “The Acquisition Merger Proposal (Proposal 2) — Interests of Certain Persons in the Business Combination.”
On behalf of the Board of Directors of Globalink, I thank you for your support and we look forward to the successful consummation of the Business Combination.
| By order of the Board of Directors, | |
Say Leong Lim |
|
| Chief Executive Officer of Globalink Investment Inc. | |
| , 2025 |
IF YOU RETURN YOUR PROXY CARD WITHOUT AN INDICATION OF HOW YOU WISH TO VOTE, YOUR SHARES WILL BE VOTED IN FAVOR OF EACH OF THE PROPOSALS.
TO EXERCISE YOUR REDEMPTION RIGHTS, YOU MUST PRIOR TO P.M., EASTERN TIME, ON , 2025, (A) SUBMIT A WRITTEN REQUEST TO THE TRANSFER AGENT THAT GLOBALINK REDEEM YOUR PUBLIC SHARES FOR CASH AND (B) DELIVER YOUR PUBLIC SHARES TO THE TRANSFER AGENT, PHYSICALLY OR ELECTRONICALLY USING THE DEPOSITORY TRUST COMPANY’S DWAC (DEPOSIT WITHDRAWAL AT CUSTODIAN) SYSTEM, IN EACH CASE, IN ACCORDANCE WITH THE PROCEDURES DESCRIBED IN THE PROXY STATEMENT/PROSPECTUS. IF THE BUSINESS COMBINATION IS NOT CONSUMMATED, THEN THE PUBLIC SHARES WILL NOT BE REDEEMED FOR CASH. IF YOU HOLD THE SHARES IN STREET NAME, YOU WILL NEED TO INSTRUCT THE ACCOUNT EXECUTIVE AT YOUR BANK OR BROKER TO WITHDRAW THE SHARES FROM YOUR ACCOUNT IN ORDER TO EXERCISE YOUR REDEMPTION RIGHTS. SEE “THE SPECIAL MEETING — REDEMPTION RIGHTS” IN THE ACCOMPANYING PROXY STATEMENT/PROSPECTUS FOR MORE SPECIFIC INSTRUCTIONS.
TABLE OF CONTENTS
| i |
ABOUT THIS PROXY STATEMENT/PROSPECTUS
This document, which forms part of a registration statement on Form F-4 filed with the U.S. Securities and Exchange Commission (the “SEC”) by PubCo, constitutes a prospectus of PubCo under Section 5 of the Securities Act, with respect to the issuance of (i) the PubCo ordinary shares to Globalink stockholders, (ii) the PubCo warrants to holders of Globalink warrants in exchange for the Globalink warrants, (iii) the PubCo ordinary shares underlying the PubCo warrants, (iv) PubCo ordinary shares in exchange for Globalink rights, if the Business Combination is consummated, and (v) PubCo ordinary shares to existing shareholders of Alps Holdco. This document also constitutes a notice of meeting and a proxy statement under Section 14(a) of the Exchange Act, with respect to the Special Meeting at which Globalink stockholders will be asked to consider and vote upon the Proposals, including the Proposals approve the Redomestication Merger and the Acquisition Merger.
If you would like additional copies of this proxy statement/prospectus or if you have questions about the Redomestication Merger Proposal, the Acquisition Merger Proposal, or the Proposals to be presented at the Special Meeting, you should contact Globalink by telephone or in writing:
Globalink Investment Inc.
200 Continental Drive, Suite 401
Newark, Delaware 19713
+6012 405 0015
You may also obtain these documents by requesting them in writing or by telephone from Globalink’s proxy solicitor, Okapi Partners LLC, at 1212 Avenue of the Americas, 17th Floor, New York, New York 10036, or at (855) 305-0857, or banks and brokers can call collect at (212) 297-0720, or by emailing info@okapipartners.com.
If you are a stockholder of Globalink and would like to request documents, please do so by , 2025 to receive them before the Special Meeting. If you request any documents from Globalink, Globalink will mail them to you by first class mail, or another equally prompt means.
You should rely only on the information contained in this proxy statement/prospectus in deciding how to vote on the Redomestication Merger Proposal, the Acquisition Merger Proposal and other Proposals presented at the Special Meeting. Neither Globalink nor Alps Holdco has authorized anyone to give any information or to make any representations other than those contained in this proxy statement/prospectus. Do not rely upon any information or representations made outside of this proxy statement/prospectus. The information contained in this proxy statement/prospectus may change after the date of this proxy statement/prospectus. Do not assume after the date of this proxy statement/prospectus that the information contained in this proxy statement/prospectus is still correct.
Information contained in this proxy statement/prospectus regarding Globalink and its business, operations, management and other matters has been provided by Globalink, and information contained in this proxy statement/prospectus regarding Alps and its business, operations, management and other matters has been provided by Alps.
This proxy statement/prospectus does not constitute an offer to sell or a solicitation of an offer to buy any securities, or the solicitation of a proxy or consent, in any jurisdiction to or from any person to whom it is unlawful to make any such offer or solicitation in such jurisdiction.
MARKET AND INDUSTRY DATA
Certain information contained in this document relates to or is based on studies, publications, surveys and other data obtained from third-party sources and Globalink’s and Alps’ own internal estimates and research. While Globalink and Alps believe these third-party sources to be reliable as of the date of this proxy statement/prospectus, they have not independently verified the market and industry data contained in this proxy statement/prospectus or the underlying assumptions relied on therein. Finally, while Globalink and Alps believe their own internal research is reliable, such research has not been verified by any independent source. While Globalink and Alps are not aware of any misstatements regarding the industry data and internal research presented herein, these data and research are subject to assumptions and estimates and involve high degree of uncertainty and risk due to a variety of factors, including those discussed under the headings “Risk Factors,” “Cautionary Note Regarding Forward-Looking Statements” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations of Alps” in this proxy statement/prospectus.
| 1 |
TRADEMARKS AND SERVICE MARKS
This document contains references to trademarks, trade names and service marks belonging to other entities. Solely for convenience, trademarks, trade names and service marks referred to in this proxy statement/prospectus may appear without the ® or TM symbols, but such references are not intended to indicate, in any way, that the applicable licensor will not assert, to the fullest extent under applicable law, its rights to these trademarks and trade names. We do not intend our use or display of other companies’ trade names, trademarks or service marks to imply a relationship with, or endorsement or sponsorship of us by, any other companies.
FREQUENTLY USED TERMS
Unless otherwise stated or unless the context otherwise requires, the terms “we,” “us,” “our,” and the “Company” refer PubCo, and if the context requires, to the Combined Company following consummation of the Business Combination.
In this document:
| ● | “Alps” means Alps Global Holding Berhad, a Malaysian company. After the date of the Merger Agreement and prior to the Closing, Alps will become a subsidiary of Alps Holdco. When describing financial information, also includes Alps Holdco and Alps’ subsidiaries; | |
| ● | “Alps Holdco” means Alps Life Sciences Inc, a Cayman Islands exempted company; | |
| ● | “Alps Holdco Board” means the board of directors of Alps Holdco; | |
| ● | “Alps Holdco Ordinary Shares” means the ordinary shares of Alps Holdco of par value US$0.00001 each; | |
| ● | “Alps Holdco Group” means Alps Holdco, Alps and their subsidiaries; | |
| ● | “Alps Holdco Shareholders” means holders of Alps Holdco Ordinary Shares; | |
| ● | “Alps Holdco Support Agreements” means the agreements entered into after the execution of the Merger Agreement pursuant to which Alps Holdco, Globalink, and certain shareholders of Alps Holdco agreed to vote all of the Alps Holdco Ordinary Shares beneficially owned by them in favor of the Business Combination; | |
| ● | “APC” means Annual Practising Certificate. A certificate issued by the MMC for practitioner to legally practice medicine and has to be renewed every year. | |
| ● | “Authority” means any federal, state, local or foreign government or political subdivision thereof, or any agency or instrumentality of such government or political subdivision, or any self-regulated organization or other non-governmental regulatory authority or quasi-governmental authority exercising executive, legislative, judicial, regulatory or administrative functions (to the extent that the rules, regulations or orders of such organization or authority have the force of Law), or any arbitrator, court or tribunal of competent jurisdiction; | |
| ● | “Amended and Restated Registration Rights Agreement” means the Amended and Restated Registration Rights Agreement to be entered at the Closing by Globalink, certain stockholders of Globalink and certain shareholders of Alps Holdco; | |
| ● | “Board” or “Globalink Board” means the board of directors of Globalink; |
| 2 |
| ● | “Business Combination” means the Redomestication Merger and the Acquisition Merger as contemplated by the Merger Agreement; | |
| ● | “cGCP” means current good clinical practice, an international ethical and scientific quality standard for designing, conducting, recording, and reporting clinical trials that involve the participation of human subjects; | |
| ● | “cGMP” means current good manufacturing practice, a system for ensuring that products are consistently produced and controlled according to quality standards; | |
| ● | “Chardan” means Chardan Capital Markets, LLC, the representative of the underwriters for the IPO of Globalink; | |
| ● | “Closing” means the consummation of the Business Combination; | |
| ● | “Closing Date” means the date on which the Business Combination is consummated; | |
| ● | “Code” means the Internal Revenue Code of 1986, as amended; | |
| ● | “Combined Company” means the PubCo and its consolidated subsidiaries after the Business Combination; | |
| ● | “Combined Company Board” means the board of directors of the Combined Company; | |
| ● | “Companies Act” means the Companies Act (Revised) of the Cayman Islands; | |
| ● | “common stock” or “Globalink common stock” means the Globalink common stock; | |
| ● | “Continental” means Continental Stock Transfer & Trust Company, Globalink’s transfer agent, warrant agent, rights agent and trustee; | |
| ● | “COVID-19” means the worldwide novel coronavirus disease pandemic; | |
| ● | “CTIL” means the Clinical Trial Import License in Malaysia; | |
| ● | “CTX” means the Clinical Trial Exemption in Malaysia; | |
| ● | “Globalink Charter” means Globalink’s amended and restated certificate of incorporation, as amended, currently in effect; | |
| ● | “DGCL” means the General Corporation Law of the State of Delaware, as amended; | |
| ● | “DWAC” means The Depository Trust Company’s deposit withdrawal at custodian; | |
| ● | “Earnout Shares” means an aggregate of maximum 48,000,000 additional PubCo ordinary shares issuable to Alps Holdco Shareholders upon the completion of certain milestones set forth in the Merger Agreement; | |
| ● | “Effective Time” means the time at which the Business Combination becomes effective; | |
| ● | “Escrow Account” means the segregated escrow account in which the Escrow Property is maintained; | |
| ● | “Escrow Agent” means Continental or such other escrow agent mutually acceptable to Globalink, PubCo and Alps Holdco; | |
| ● | “Escrow Agreement” means an escrow agreement to be entered into by and among PubCo, Globalink, and the Escrow Agent, effective as of the Effective Time, in form and substance reasonably satisfactory to PubCo and Alps Holdco, pursuant to which PubCo shall issue to the Escrow Agent a number of PubCo ordinary shares (with each share valued at $10.00) equal to five percent (5%) of the Merger Consideration Shares; |
| 3 |
| ● | “Escrow Property” means Escrow Shares along with any other dividends, distributions or other income on the Escrow Shares; | |
| ● | “Escrow Shares” means a number of PubCo ordinary shares (with each share valued at $10.00) equal to five percent (5%) of the Merger Consideration Shares and any equity securities paid as dividends or distributions with respect to such shares or into which such shares are exchanged or converted; | |
| ● | “Exchange Act” means the Securities Exchange Act of 1934, as amended; | |
| ● | “FDA” means the U.S. Food and Drug Administration; | |
| ● | “Founder Shares” means the 2,875,000 outstanding shares of Globalink common stock held by the Sponsor and Globalink’s directors and officers; | |
| ● | “Fully Diluted Alps Holdco Shares” means all ordinary shares of Alps Holdco that are issued and outstanding immediately prior to the Effective Time; | |
| ● | “Globalink” means Globalink Investment Inc., a Delaware corporation or the reference to the current corporation as a special purpose acquisition corporation prior to the Business Combination; | |
| ● | “Globalink stockholders” means the holders of shares of Globalink common stock; | |
| ● | “Globalink Support Agreements” means the agreement entered into in connection with execution of the Merger Agreement pursuant to which Alps Holdco, Globalink, and certain stockholders of Globalink agreed to vote all of the shares of Globalink common stock beneficially owned by them in favor of the Business Combination; | |
| ● | “Initial Stockholders” means the Sponsor and Globalink’s directors and officers who are initial holders of the Founder Shares; | |
| ● | “Insider Letter” means the letter agreement dated December 6, 2021, by and among Globalink Investment Inc., the Sponsor, PGM and each of Globalink’s directors and officers; | |
| ● | “IPO” means the initial public offering of Globalink consummated on December 9, 2021; | |
| ● | “IRS” means the United States Internal Revenue Service; | |
| ● | “ISO” means the International Organization of Standardization; | |
| ● | “Law” means any domestic or foreign, federal, state, municipality or local law, statute, ordinance, code, rule, or regulation; | |
| ● | “LCP” means Letter of Credentialing and Privileging Medical Practice; | |
| ● | “Lock-Up Agreements” means the agreements to be entered into prior to the Closing, pursuant to which certain Alps Holdco Shareholders and certain Globalink stockholders agreed to certain restrictions on transfer of PubCo ordinary shares for a period of six months to fifteen months after the Closing Date. | |
| ● | “MAB” means Medicine Advertisements Board; | |
| ● | “Merger Agreement” means the Merger Agreement by and among Globalink, Alps Holdco, the Sponsor, PubCo, Merger Sub and Dr. Tham Seng Kong, dated January 30, 2024, as amended and restated on May 20, 2024 and may be further amended, restated or supplemented from time to time; |
| 4 |
| ● | “Merger Consideration Shares” means 160,000,000 PubCo ordinary shares, subject to the withholding of the Escrow Shares deposited in the Escrow Account, and after the Closing are subject to reduction for the indemnification obligations of the Indemnifying Parties set forth in the Merger Agreement; | |
| ● | “Merger Sub” means Alps Biosciences Merger Sub, a Cayman Islands exempted company and a wholly-owned subsidiary of PubCo; | |
| ● | “MMC” means Malaysian Medical Council; | |
| ● | “MOH” means Ministry of Health, Malaysia; | |
| ● | “MS” means Malaysian Standard; | |
| ● | “NPRA” means National Pharmaceutical Regulatory Agency, Malaysia; | |
| ● | “PGM” means Public Gold Marketing Sdn. Bhd., a Malaysian private limited company, an investor who purchased 570,000 private units in the private placement simultaneously with the consummation of the IPO. PGM is an affiliate of the Sponsor as its 95% equity holder has a familial relationship with the control person of the Sponsor; | |
| ● | “PIPE Investment” means the private placement contemplated by the subscription agreements dated June 4, 2024, June 5, 2024 and August 27, 2024, entered into by and between PubCo, Globalink, Alps Holdco, and certain investors for an aggregate of $40,200,000 for 4,020,000 PubCo ordinary shares, to be consummated simultaneously with the closing of the Business Combination; | |
| ● | “POC” means proof-of-concept; | |
| ● | “private rights” means the rights of Globalink included in the private units; | |
| ● | “private units” mean the 570,000 Globalink units issued to Public Gold Marketing Sdn. Bhd., a Malaysian private limited company, in a private placement; | |
| ● | “private warrants” means warrants of Globalink included in the private units, each entitling the holder thereof to purchase one-half (1/2) share of Globalink common stock at a purchase price of $11.50 per whole share; | |
| ● | “PubCo” means Alps Global Holding Pubco, a Cayman Islands exempted company; | |
| ● | “PubCo A&R Memorandum and Articles” means the Amended and Restated Memorandum and Articles of Association of PubCo to take effect at or immediately prior to the Effective Time, in the form included as Annex B to this proxy statement/prospectus, as further described in the “Comparison of Corporate Governance and Stockholders’/Shareholders’ Rights” section of this proxy statement/prospectus; | |
| ● | “PubCo Board” means the board of directors of PubCo; | |
| ● | “PubCo ordinary shares” means the ordinary shares of PubCo of par value US$0.0001 each; | |
| ● | “PubCo warrants” means warrants of PubCo each entitling the holder thereof to purchase one-half (1/2) of one PubCo ordinary share at a purchase price of $11.50 per whole share; | |
| ● | “public rights” means the rights which were sold as part of the Globalink units in the IPO; | |
| ● | “public shares” means shares of Globalink common stock underlying the Units sold in the IPO, including any overallotment securities acquired by Globalink’s underwriters; | |
| ● | “public stockholders” means the holders of Globalink public shares; | |
| ● | “public units” means the units of Globalink sold in the IPO, whether they were purchased in the IPO or thereafter in the open market, each consisting of one share of common stock, $0.001 par value, one right to receive one-tenth (1/10) of a share of common stock upon the consummation of an initial business combination and one redeemable warrant entitling the holder thereof to purchase one-half (1/2) of a share of common stock at a price of $11.50 per whole share; |
| 5 |
| ● | “public warrants” means warrants of Globalink included in the public units, each entitling the holder thereof to purchase one-half (1/2) share of Globalink common stock at a purchase price of $11.50 per whole share; | |
| ● | “R&D” means research and development; | |
| ● | “rights” or “Globalink rights” means the rights of Globalink included in the units, each entitling its holder to receive one-tenth (1/10) of a share of Globalink common stock upon the consummation of the Business Combination; | |
| ● | “RM” or “Ringgit Malaysia” means the lawful currency of Malaysia; | |
| ● | “SEC” means the U.S. Securities and Exchange Commission; | |
| ● | “Securities Act” means the Securities Act of 1933, as amended; | |
| ● | “Seller Representative” means Dr. Tham Seng Kong, an individual; | |
| ● | “Special Meeting” means the special meeting of the stockholders of Globalink, which will be held at , Eastern time, on , 2025; | |
| ● | “Sponsor” or “Parent Representative” means GL Sponsor LLC, a Delaware limited liability company; | |
| ● | “Subscription Agreements” means the subscription agreements with investors (the “PIPE Investors”) dated June 4, 2024, June 5, 2024 and August 27, 2024 in the PIPE Investment; | |
| ● | “Trust Account” means Globalink’s trust account maintained by Continental; | |
| ● | “units” or “Globalink units” means public units and private units, collectively; | |
| ● | “U.S. Dollars,” “US Dollars,” “$,” or “US$” means the legal currency of the United States; | |
| ● | “U.S. GAAP” or “GAAP” means accounting principles generally accepted in the United States; | |
| ● | “warrants” or “Globalink warrants” means private warrants and public warrants, collectively; and | |
| ● | “ZAR” means the legal currency of the Republic of South Africa. |
Share Calculations and Ownership Percentages
Unless otherwise specified (including in the sections entitled “Unaudited Pro Forma Condensed Combined Financial Information and Notes” and “Security Ownership of Certain Beneficial Owners and Management”), the share calculations and ownership percentages set forth in this proxy statement/prospectus with respect to shareholders of the Combined Company are for illustrative purposes only and assume the following (certain capitalized terms below are defined elsewhere in this proxy statement/prospectus):
| 1. | No public stockholders exercise their redemption rights in connection with the Closing of the Business Combination, and the balance of the Trust Account as of the Closing is the same as its balance on December 6, 2024 of approximately $3.33 million. Please see the section entitled “The Special Meeting — Redemption Rights.” | |
| 2. | There are no transfers by the Sponsor, or any of the officers and directors of Globalink of Globalink securities held by the Sponsor or any officer or director of Globalink on or prior to the Closing Date. | |
| 3. | The PIPE Investment is consummated in accordance with the terms of the Subscription Agreements for aggregated proceeds of $40,200,000, with PubCo issuing 4,020,000 ordinary shares to the PIPE Investors, in connection with which the PIPE Investors will own up to approximately 4,020,000 PubCo ordinary shares. Please see the section entitled “The Acquisition Merger Proposal (Proposal 2) — Related Agreements — Subscription Agreements; Lock-up Agreements.” | |
| 4. | There are no issuances of equity securities of Globalink prior to or in connection with the Closing. | |
| 5. | No Earnout Shares are issued and Alps Holdco Shareholders will receive 160,000,000 PubCo ordinary shares. | |
| 6. | That none of the Alps Holdco Shareholders exercises appraisal rights in connection with the Closing. | |
| 7. | That, the number of PubCo ordinary shares to be issued in connection with conversion of Globalink’s public and private rights is 1,207,000. Currently, PGM owns 570,000 private rights, or all of the outstanding private rights of Globalink. |
| 6 |
CAUTIONARY NOTE REGARDING FORWARD-LOOKING STATEMENTS
This proxy statement/prospectus contains forward-looking statements, including statements about the parties’ ability to close the Business Combination, the anticipated benefits of the Business Combination, and the financial condition, results of operations, earnings outlook and prospects of Globalink and/or Alps Holdco and may include statements for the period following the consummation of the Business Combination. Forward-looking statements appear in several places in this proxy statement/prospectus including, without limitation, in the sections titled “Management’s Discussion and Analysis of Financial Condition and Results of Operations of Alps” and “Information about Alps”. In addition, any statements that refer to projections, forecasts or other characterizations of future events or circumstances, including any underlying assumptions, are forward-looking statements. Forward-looking statements are typically identified by words such as “plan,” “believe,” “expect,” “anticipate,” “intend,” “outlook,” “estimate,” “forecast,” “project,” “continue,” “could,” “may,” “might,” “possible,” “potential,” “predict,” “should,” “would” and other similar words and expressions, but the absence of these words does not mean that a statement is not forward-looking.
The forward-looking statements are based on the current expectations of the management of Globalink and Alps Holdco as applicable and are inherently subject to uncertainties and changes in circumstances and their potential effects and speak only as of the date of such statement. There can be no assurance that future developments will be those that have been anticipated. These forward-looking statements involve several risks, uncertainties or other assumptions that may cause actual results or performance to be materially different from those expressed or implied by these forward-looking statements. These risks and uncertainties include, but are not limited to, those factors described in “Risk Factors,” those discussed and identified in public filings made with the SEC by Globalink and the following:
| ● | actual results may vary from expectations regarding Alps Holdco and Alps Holdco’s ability to meet expectations related to its business prospects; | |
| ● | the occurrence of any event, change or other circumstances that could give rise to the termination of the Merger Agreement; | |
| ● | the outcome of any legal proceedings that may be instituted against Globalink, Alps Holdco and others following announcement of the Merger Agreement and transactions contemplated therein; | |
| ● | the inability to complete the Business Combination due to the failure to obtain Globalink stockholders’ approval; | |
| ● | Alps Holdco’s failure to achieve or maintain profitability in the future; | |
| ● | Alps Holdco’s ability to execute its anticipated business plans and strategy; | |
| ● | Alps Holdco faces significant competition and expects to face increasing competition in many aspects of its business, which could cause its operating results to suffer; | |
| ● | the healthcare market is a new and rapidly changing industry and it is uncertain whether Alps Holdco’s business will be adversely affected by the changing industry and the development of technology and medical research; | |
| ● | the ability of the Combined Company following the Business Combination to receive listing approval from Nasdaq and maintain the listing of the Combined Company’s securities on Nasdaq; | |
| ● | the risk that the proposed Business Combination disrupts current plans and operations of Alps Holdco as a result of the announcement and consummation of the Business Combination; | |
| ● | the inability to recognize the anticipated benefits of the Business Combination; | |
| ● | the inability to consummate the PIPE Investment; |
| 7 |
| ● | the failure to satisfy the conditions to the consummation of the Business Combination, including the approval of the Merger Agreement by the stockholders of Globalink and the requirement, following any redemptions by Globalink’s public stockholders, Globalink have cash or cash equivalents equal to the aggregate subscriptions by investors in the PIPE Investment; | |
| ● | unexpected costs related to the proposed Business Combination; | |
| ● | the amount of any redemptions by existing Globalink stockholders being greater than expected; | |
| ● | the management and board composition of the Combined Company following the proposed Business Combination; | |
| ● | geopolitical risk and changes in applicable laws or regulations; | |
| ● | the possibility that Globalink and/or Alps Holdco may be adversely affected by other economic, business, and/or competitive factors; | |
| ● | litigation and regulatory enforcement risks, including the diversion of management time and attention and the additional costs and demands on the Combined Company’s resources; | |
| ● | the risks that the consummation of the Business Combination is substantially delayed or does not occur; | |
| ● | the impact from future regulatory, judicial, and legislative changes in Alps Holdco’s industry; | |
| ● | the uncertain effects of the COVID-19 pandemic; and | |
| ● | those factors set forth in documents of Globalink filed, or to be filed, with the SEC. |
All subsequent written and oral forward-looking statements concerning the Business Combination or other matters addressed in this proxy statement/prospectus and attributable to Globalink, Alps Holdco, or any person acting on their behalf are expressly qualified in their entirety by the cautionary statements contained or referred to in this proxy statement/prospectus. There may be additional risks that neither Globalink nor Alps Holdco presently know or that Globalink and Alps Holdco currently believe are immaterial that could also cause actual results to differ materially from those contained in the forward-looking statements. In addition, forward-looking statements reflect Globalink’s and Alps Holdco’s expectations, plans or forecasts of future events and views as of the date of this proxy statement/prospectus. Globalink and Alps Holdco’s anticipate that subsequent events and developments may cause Globalink’s and Alps Holdco’s assessments to change. However, while we may elect to update these forward-looking statements at some point in the future, Globalink and Alps Holdco specifically disclaim any obligation to do so. These forward-looking statements should not be relied upon as representing Globalink’s and Alps Holdco’s assessments as of any date subsequent to the date of this proxy statement/prospectus. Accordingly, undue reliance should not be placed upon the forward-looking statements.
If any of these risks materialize or any of Globalink’s or Alps Holdco’s assumptions prove incorrect, actual results could differ materially from the results implied by these forward-looking statements. Actual results, performance or achievements may, and are likely to, differ materially, and potentially adversely, from any projections and forward-looking statements and the assumptions on which those forward-looking statements were based. There can be no assurance that the data contained herein is reflective of future performance to any degree. You are cautioned not to place undue reliance on forward-looking statements as a predictor of future performance as projected financial information and other information are based on estimates and assumptions that are inherently subject to various significant risks, uncertainties and other factors, many of which are beyond Globalink’s and Alps Holdco’s control. Forward-looking statements are not guarantees of performance. All forward-looking statements attributable to Globalink or Alps Holdco or a person acting on their behalf are expressly qualified in their entirety by the foregoing cautionary statements.
| 8 |
QUESTIONS AND ANSWERS ABOUT THE SPECIAL MEETING AND THE PROPOSALS
The following are answers to some questions that you, as a stockholder of Globalink, may have regarding the Proposals being considered at the Special Meeting. The following questions and answers do not include all the information that is important to Globalink stockholders. We urge you to carefully read the remainder of this proxy statement/prospectus because the information in this section does not provide all the information that might be important to you with respect to the Business Combination, the Proposals and the other matters being considered at the Special Meeting. Additional important information is also contained in the annexes to and the documents incorporated by reference into this proxy statement/prospectus. See also the section of this proxy statement/prospectus titled “Where You Can Find More Information.”
| Q: | Why am I receiving this proxy statement/prospectus? |
| A | Globalink and Alps Holdco, among others, have agreed to the Business Combination under the terms of the Merger Agreement, which is attached to this proxy statement/prospectus as Annex A, and is incorporated into this proxy statement/prospectus by reference. The Globalink Board is soliciting your proxy to vote for the Business Combination, consisted of the Redomestication Merger and the Acquisition Merger, each of which are described in this proxy statement/prospectus, as well as other Proposals at the Special Meeting because you owned shares of common stock at the close of business on , 2025, the “Record Date” for the Special Meeting, and are therefore entitled to vote at the Special Meeting. This proxy statement/prospectus summarizes the information that you need to know in order to cast your vote. |
THE VOTE OF GLOBALINK STOCKHOLDERS IS IMPORTANT. GLOBALINK STOCKHOLDERS ARE URGED TO SUBMIT THEIR PROXIES AS SOON AS POSSIBLE AFTER CAREFULLY REVIEWING THIS PROXY STATEMENT/PROSPECTUS AND ITS ANNEXES AND CAREFULLY CONSIDERING EACH OF THE PROPOSALS BEING PRESENTED AT THE SPECIAL MEETING.
| Q: | What is being voted on at the Special Meeting? |
| A | During the Special Meeting, Globalink’s stockholders will be asked to consider and vote upon: |
| ● | The Redomestication Merger Proposal (Proposal 1) — to approve the Redomestication Merger. The Redomestication Merger Proposal is described in further detail in the accompanying proxy statement/prospectus under the heading “The Redomestication Merger Proposal (Proposal 1).” | |
| ● | The Acquisition Merger Proposal (Proposal 2) — To consider and vote upon a proposal to approve the Acquisition Merger, the Merger Agreement and such other transactions contemplated by the Merger Agreement. The Acquisition Merger Proposal is described in more detail in the accompanying proxy statement/prospectus under the heading “The Acquisition Merger Proposal (Proposal 2).” | |
| ● | The Nasdaq Proposals (Proposals 3) — To consider and vote upon separate proposals, for purposes of complying with The Nasdaq Listing Rule, to issue (i) up to 208,000,000 PubCo ordinary shares pursuant to the Merger Agreement (including the ordinary shares issuable pursuant to that certain earnout provisions in the Merger Agreement), and (ii) up to 4,020,000 PubCo ordinary shares to the PIPE Investors. | |
| ● | The Adjournment Proposal (Proposal 4) — To consider and vote upon a proposal to adjourn the Special Meeting to a later date or dates, if necessary or appropriate as determined by the Globalink Board. |
| 9 |
| Q: | What vote is required to approve the Proposals? |
| A: | The Redomestication Merger Proposal requires the affirmative vote of the majority of the issued and outstanding shares of common stock present by virtual attendance or represented by proxy and entitled to vote at the Special Meeting. As a result, a Globalink stockholder’s failure to vote by proxy or to vote in person by virtual attendance in respect of the Redomestication Merger Proposal at the Special Meeting, or to abstain, will not be counted for or against the number of shares of Globalink common stock required for the vote on this Proposal. |
The Acquisition Merger Proposal requires the affirmative vote of the majority of the issued and outstanding shares of common stock present by virtual attendance or represented by proxy and entitled to vote at the Special Meeting. As a result, a Globalink stockholder’s failure to vote by proxy or to vote in person by virtual attendance in respect of the Redomestication Merger Proposal at the Special Meeting, or to abstain, will not be counted for or against the number of shares of Globalink common stock required for the vote on this Proposal.
The Nasdaq Proposals require the affirmative vote of the majority of the issued and outstanding shares of common stock present by virtual attendance or represented by proxy and entitled to vote at the Special Meeting. As a result, a Globalink stockholder’s failure to vote by proxy or to vote in person by virtual attendance in respect of the Nasdaq Proposals at the Special Meeting, or to abstain, will not be counted for or against the number of shares of Globalink common stock required for the vote on these Proposals.
The Adjournment Proposal requires the affirmative vote of the majority of the issued and outstanding shares of common stock present in person by virtual attendance or represented by proxy and entitled to vote at the Special Meeting. As a result, a Globalink stockholder’s failure to vote by proxy or to vote in person by virtual attendance in respect of the Adjournment Proposal at the Special Meeting, or to abstain, will not be counted for or against the number of shares of Globalink common stock required for the vote on this Proposal.
| Q: | Are any of the proposals conditioned on one another? |
| A: | Yes, the Redomestication Merger Proposal and the Acquisition Merger Proposal are dependent upon each other. It is important for you to note that if either of the Redomestication Merger Proposal or the Acquisition Merger Proposal is not approved, Globalink will not consummate the Business Combination. If either of the Redomestication Merger Proposal or the Acquisition Merger Proposal is not approved, the Nasdaq Proposals will not be presented to the Globalink stockholders for a vote, although the Adjournment Proposal may be presented. Globalink currently has until January 9, 2025 to complete its initial business combination, or it will be required to dissolve and liquidate. If Globalink anticipates that it may not be able to consummate its initial business combination in time, Globalink may, by resolutions of its Board, if requested by the Sponsor, extend the period of time to consummate a business combination on a monthly basis to until June 9, 2025, subject to the Sponsor depositing additional funds into the Trust Account. |
| 10 |
The approval of the Redomestication Merger Proposal and the Acquisition Merger Proposal are preconditions to the consummation of the Business Combination. The Adjournment Proposal is not conditioned on the approval of any other proposal set forth in this proxy statement/prospectus.
It is important for you to note that if either the Redomestication Merger Proposal or the Acquisition Merger Proposal is not approved, Globalink will not consummate the Business Combination. Globalink currently has until January 9, 2025 to complete its initial business combination, or it will be required to dissolve and liquidate. If Globalink anticipates that it may not be able to consummate its initial business combination in time, Globalink may, by resolutions of its Board, if requested by the Sponsor, extend the period of time to consummate a business combination on a monthly basis to until June 9, 2025, subject to the Sponsor depositing additional funds into the Trust Account.
| Q: | What will happen in the Business Combination? |
| A: | The Business Combination will be effected with two steps: (i) the Redomestication Merger: Globalink will merge with and into PubCo, with PubCo surviving such merger as the surviving entity, and (ii) the Acquisition Merger: Merger Sub will merge with and into Alps Holdco, with Alps Holdco surviving such merger as the surviving entity. Upon consummation of the Business Combination, Alps Holdco will become a wholly owned subsidiary of PubCo. For details and more information please see the sections entitled “The Redomestication Merger Proposal (Proposal 1)” and “The Acquisition Merger Proposal (Proposal 2)”. In connection with the Business Combination, the cash held in the Trust Account after giving effect to any redemption of shares by Globalink’s public stockholders and the proceeds from the PIPE Investment will be used to pay certain fees and expenses in connection with the Business Combination, and for working capital and general corporate purposes of the Combined Company. |
| Q: | How will the Initial Stockholders and PGM vote? |
| A: | Pursuant to the Insider Letter, the Initial Stockholders and PGM agreed to vote their respective Founder Shares and any public shares of Globalink common stock in favor of the Redomestication Merger Proposal, the Acquisition Merger Proposal, and related proposals. In addition, in connection with the execution of the Merger Agreement, the Initial Stockholders and PGM entered into the Globalink Support Agreements with Alps Holdco pursuant to which they agreed to vote all shares of common stock beneficially owned by them in favor of the Business Combination and related transactions. As of the date of this proxy statement/prospectus, a total of 3,445,000 shares of common stock, or approximately 92.6% of the outstanding shares, were subject to the Insider Letter and the Globalink Support Agreements. As a result, no shares of common stock held by the public stockholders will need to be present in person by virtual attendance or by proxy to satisfy the quorum requirement for the Special Meeting. In addition, as the vote to approve the Redomestication Merger Proposal and the Acquisition Merger Proposal is a majority of the then outstanding shares of common stock present and entitled to vote at the Special Meeting, assuming only the minimum number of shares of common stock to constitute a quorum is present, no public stockholder must vote in favor of the Redomestication Merger Proposal or the Acquisition Merger Proposal for them to be approved. |
| Q: | How many votes do I and others have? |
| A: | You are entitled to one vote for each share of Globalink common stock that you held as of the Record Date. As of the close of business on the Record Date, there were 3,722,511 outstanding shares of Globalink common stock. |
| Q: | What is the consideration being paid to Alps Holdco’s shareholders? |
| A: | Subject to the terms of the Merger Agreement and customary adjustments set forth therein, the consideration to be delivered to Alps Holdco’s shareholders in connection with the Business Combination will consist of newly issued PubCo ordinary shares. |
| The number of newly-issued PubCo ordinary shares that each holder of Alps Holdco Ordinary Shares will receive for each Alps Holdco Ordinary Share as a result of the Business Combination (not including any Earnout Shares) will be equal to the (a) One Point Six Billion United States Dollars ($1,600,000,000), divided by (b) the number of Fully Diluted Alps Holdco Shares, then (c) divided by $10.00. At the Closing, the Alps Holdco Shareholders will receive the Merger Consideration Shares. |
| 11 |
PubCo will issue up to an additional 48,000,000 Earnout Shares to the Alps Holdco Shareholders if certain milestones are met as described below. Subject to the terms and conditions of the Merger Agreement, the Earnout Shares shall be earned and payable as follows:
(i) if, during the Combined Company’s first full fiscal year following the date of Closing, the Revenue exceeds $7,000,000, the Alps Holdco Shareholders shall be issued an additional 9,600,000 PubCo ordinary shares;
(ii) if during the Combined Company’s second full fiscal year following the date of Closing, the Revenue exceeds $14,000,000, the Alps Holdco Shareholders shall be issued an additional 9,600,000 PubCo ordinary shares;
(iii) if during the Combined Company’s third full fiscal year following the date of Closing, the Revenue exceeds $23,000,000, the Alps Holdco Shareholders shall be issued an additional 9,600,000 PubCo ordinary shares;
(iv) if during the Combined Company’s fourth full fiscal year following the date of Closing, the Revenue exceeds $34,000,000, the Alps Holdco Shareholders shall be issued an additional 9,600,000 PubCo ordinary shares; and
(v) if during the Combined Company’s fifth full fiscal year following the date of Closing, the Revenue exceeds $56,000,000, the Alps Holdco Shareholders shall be issued an additional 9,600,000 PubCo ordinary shares.
| Q: | What equity stake will current Globalink stockholders, Alps Holdco Shareholders and PIPE Investors hold in PubCo immediately after the Closing? |
| A: | Upon consummation of the Business Combination, based on the assumptions described under the heading “Frequently Used Terms — Share Calculations and Ownership Percentages”, (i) Globalink’s public stockholders are expected to own approximately 0.2% of the issued and outstanding PubCo ordinary shares under no redemption scenario, and 0.0% of the issued and outstanding PubCo ordinary shares under a maximum contractual redemption scenario, (ii) the Initial Stockholders are expected to own approximately 1.7% of the issued and outstanding PubCo ordinary shares under no redemption scenario and 1.7% of the issued and outstanding PubCo ordinary shares under a maximum contractual redemption scenario, (iii) Globalink public rights holders are expected to own approximately 0.6% of the PubCo ordinary shares under no redemption scenario and 0.7% of the issued and outstanding PubCo ordinary shares under a maximum contractual redemption scenario, (iv) PGM, an affiliate of the Sponsor, which holds private rights that entitle it to receive shares of Globalink common stock upon the consummation of the Business Combination, is expected to own approximately 0.4% of the issued and outstanding PubCo ordinary shares under no redemption scenario and 0.4% of the issued and outstanding PubCo ordinary shares under a maximum contractual redemption scenario, (v) the Alps Holdco Shareholders are expected to own approximately 93.8% of the issued and outstanding PubCo ordinary shares under no redemption scenario and 93.9% of the issued and outstanding PubCo ordinary shares under a maximum contractual redemption scenario, (vi) the PIPE investors are expected to own approximately 2.4% of the issued and outstanding PubCo ordinary shares under no redemption scenario and 2.4% of the issued and outstanding PubCo ordinary shares under a maximum contractual redemption scenario, assuming the PIPE Investors will hold 4,020,000 PubCo ordinary shares, and (vii) IBDC Asia Sdn. Bhd., an advisory firm to Alps, is expected to own approximately 0.9% of the issued and outstanding PubCo ordinary shares under no redemption scenario and 0.9% of the issued and outstanding PubCo ordinary shares under a maximum contractual redemption scenario, representing finder’s fees payable in PubCo ordinary shares with value equal to 1% of the aggregate consideration for the Business Combination of US$1.6 billion. |
| If any of Globalink’s public stockholders exercise redemption rights in connection with the Closing, the percentage of the outstanding PubCo ordinary shares held by Globalink’s public stockholders will decrease and the percentages of the outstanding PubCo ordinary shares held by the Sponsor, the officers and directors of Globalink and by the Alps Holdco Shareholders will increase, in each case, relative to the percentage held if none of the public shares are redeemed. |
| 12 |
| If any of Globalink’s public stockholders as of the Record Date redeem their public shares at Closing in accordance with the Globalink Charter but continue to hold public warrants after the Closing, the aggregate value of the public warrants that may be retained by them, based on the closing trading price per public warrant of $0.02 as of December 12, 2024, would be approximately $0.2 million, regardless of the number of shares redeemed by Globalink’s public stockholders. Upon the issuance of the PubCo ordinary shares in connection with the Business Combination, the percentage ownership of the Combined Company by Globalink’s public stockholders who do not redeem their public shares will be diluted. Globalink public stockholders that do not redeem their public shares in connection with the Business Combination will experience further dilution upon the exercise of public warrants that are retained after the Closing by redeeming Globalink public stockholders. The percentage of the total number of outstanding ordinary shares that will be owned by Globalink public stockholders as a group will vary based on the number of public shares for which the holders thereof elect to have redeemed in connection with the Business Combination. | |
| The following table illustrates varying ownership levels of PubCo immediately following the Business Combination, assuming that no Earnout Shares are issued. |
Assuming No Redemptions | Assuming 25% Redemptions | Assuming 50% Redemptions | Assuming 75% Redemptions | Assuming Maximum Contractual Redemptions (1) | ||||||||||||||||||||||||||||||||||||
Number of Shares | % | Number of Shares | % | Number of Shares | % | Number of Shares | % | Number of Shares | % | |||||||||||||||||||||||||||||||
| Alps Holdco Shareholders | 160,000,000 | 93.8 | % | 160,000,000 | 93.8 | % | 160,000,000 | 93.8 | % | 160,000,000 | 93.9 | % | 160,000,000 | 93.9 | % | |||||||||||||||||||||||||
| Globalink Public Shareholders | 277,511 | 0.2 | % | 208,133 | 0.1 | % | 138,755 | 0.1 | % | 69,377 | 0.0 | % | 0 | 0.0 | % | |||||||||||||||||||||||||
| Initial Stockholders | 2,875,000 | 1.7 | % | 2,875,000 | 1.7 | % | 2,875,000 | 1.7 | % | 2,875,000 | 1.7 | % | 2,875,000 | 1.7 | % | |||||||||||||||||||||||||
| Globalink Public Rights Holders | 1,150,000 | 0.6 | % | 1,150,000 | 0.7 | % | 1,150,000 | 0.7 | % | 1,150,000 | 0.7 | % | 1,150,000 | 0.7 | % | |||||||||||||||||||||||||
| PIPE Investors | 4,020,000 | 2.4 | % | 4,020,000 | 2.4 | % | 4,020,000 | 2.4 | % | 4,020,000 | 2.4 | % | 4,020,000 | 2.4 | % | |||||||||||||||||||||||||
| PGM | 627,000 | 0.4 | % | 627,000 | 0.4 | % | 627,000 | 0.4 | % | 627,000 | 0.4 | % | 627,000 | 0.4 | % | |||||||||||||||||||||||||
| IBDC Asia Sdn. Bhd. | 1,600,000 | 0.9 | % | 1,600,000 | 0.9 | % | 1,600,000 | 0.9 | % | 1,600,000 | 0.9 | % | 1,600,000 | 0.9 | % | |||||||||||||||||||||||||
| Total Shares | 170,549,511 | 100 | % | 170,480,133 | 100 | % | 170,410,755 | 100 | % | 170,341,377 | 100 | % | 170,272,000 | 100 | % | |||||||||||||||||||||||||
| (1) | Assumes 277,511 public shares of Globalink common stock are redeemed for the proceeds in the Trust Account and that no Earnout Shares are issued. The maximum contractual redemption scenario represents the maximum contractual redemptions that may occur, which would allow Globalink to have consolidated net tangible assets of at least $5,000,001 at the consummation of the Business Combination. The standalone requirement of 5,000,001 of Globalink net tangible assets is expected to be waived by the parties. |
The table below illustrates the potential dilutive impact, excluding the exercise of warrants, to the holders of securities across five different redemption scenarios based on the assumptions described above, except that these scenarios assume the Combined Company has achieved all revenue milestones and the maximum number of Earnout Shares (48,000,000) are issued according to the earnout arrangement:
Assuming No Redemptions | Assuming 25% Redemptions | Assuming 50% Redemptions | Assuming 75% Redemptions | Assuming Maximum Contractual Redemptions | ||||||||||||||||||||||||||||||||||||
Number of Shares | % | Number of Shares | % | Number of Shares | % | Number of Shares | % | Number of Shares | % | |||||||||||||||||||||||||||||||
| Alps Holdco Shareholders | 208,000,000 | 95.2 | % | 208,000,000 | 95.2 | % | 208,000,000 | 95.2 | % | 208,000,000 | 95.3 | % | 208,000,000 | 95.3 | % | |||||||||||||||||||||||||
| Globalink Public Shareholders | 277,511 | 0.1 | % | 208,133 | 0.1 | % | 138,755 | 0.1 | % | 69,377 | 0.0 | % | 0 | 0.0 | % | |||||||||||||||||||||||||
| Initial Stockholders | 2,875,000 | 1.3 | % | 2,875,000 | 1.3 | % | 2,875,000 | 1.3 | % | 2,875,000 | 1.3 | % | 2,875,000 | 1.3 | % | |||||||||||||||||||||||||
| Globalink Public Rights Holders | 1,150,000 | 0.5 | % | 1,150,000 | 0.5 | % | 1,150,000 | 0.5 | % | 1,150,000 | 0.5 | % | 1,150,000 | 0.5 | % | |||||||||||||||||||||||||
| PIPE Investors | 4,020,000 | 1.9 | % | 4,020,000 | 1.9 | % | 4,020,000 | 1.9 | % | 4,020,000 | 1.9 | % | 4,020,000 | 1.9 | % | |||||||||||||||||||||||||
| PGM | 627,000 | 0.3 | % | 627,000 | 0.3 | % | 627,000 | 0.3 | % | 627,000 | 0.3 | % | 627,000 | 0.3 | % | |||||||||||||||||||||||||
| IBDC Asia Sdn. Bhd. | 1,600,000 | 0.7 | % | 1,600,000 | 0.7 | % | 1,600,000 | 0.7 | % | 1,600,000 | 0.7 | % | 1,600,000 | 0.7 | % | |||||||||||||||||||||||||
| Total Shares | 218,549,511 | 100 | % | 218,480,133 | 100 | % | 218,410,755 | 100 | % | 218,341,377 | 100 | % | 218,272,000 | 100 | % | |||||||||||||||||||||||||
For additional information regarding assumptions incorporated into the information presented above, see the sections titled “Frequently Used Terms — Share Calculations and Ownership Percentages,” “Unaudited Pro Forma Condensed Combined Financial Information and Notes” and “The Acquisition Merger Proposal (Proposal 2) — The Merger Agreement — Merger Consideration.”
| Q: | Do any of Globalink’s Sponsor, directors or officers have interests that may conflict with my interests with respect to the Business Combination? |
| A: | In considering the recommendation of the Board to approve the Merger Agreement, Globalink stockholders should be aware that Globalink executive officers and directors may be deemed to have interests in the Business Combination that are different from, or in addition to, those of Globalink stockholders generally, including: |
| ● | The Initial Stockholders, including the officers and directors of Globalink, have waived their right to redeem any shares of Globalink common stock in connection with a stockholder vote to approve a proposed initial business combination or sell any shares of Globalink common stock to Globalink in a tender offer prior to a proposed initial business combination, or to receive distributions with respect to the Founder Shares upon the liquidation of the Trust Account if Globalink is unable to consummate a business combination. Globalink currently has until January 9, 2025 to complete its initial business combination, or it will be required to dissolve and liquidate. If Globalink anticipates that it may not be able to consummate its initial business combination in time, Globalink may, by resolutions of its Board, if requested by the Sponsor, extend the period of time to consummate a business combination on a monthly basis to until June 9, 2025, subject to the Sponsor depositing additional funds into the Trust Account. In such event, the 2,875,000 Founder Shares currently held by the Initial Stockholders, which were acquired prior to the IPO will be worthless because such holders have agreed to waive their rights to any liquidation distributions through the Insider Letter, in consideration for inducing Chardan, the representative of the underwriters for Globalink’s IPO to proceed with the IPO, and the economic benefit the IPO would confer upon the Initial Stockholders and PGM. The Founder Shares were purchased for an aggregate purchase price of $25,000, or less than $0.01 per share, and had a total market value of $34.36 million as of December 12, 2024. Accordingly, the Initial Stockholders will receive a positive rate of return so long as the market price of the common stock is at least $0.01 per share, even if public stockholders experience a negative rate of return in the Combined Company. |
| 13 |
| ● | The fact that the Initial Stockholders’ Founder Shares became subject to a lock-up whereby, subject to certain limited exceptions, the Initial Stockholders agree not to offer, sell, contract to sell, pledge or otherwise dispose of, directly or indirectly, any of the Lock-up Shares, enter into a transaction that would have the same effect, or enter into any swap, hedge or other arrangement that transfers, in whole or in part, any of the economic consequences of ownership of the Lock-Up Shares or otherwise, publicly disclose the intention to make any offer, sale, pledge or disposition, or to enter into any transaction, swap, hedge or other arrangement, or engage in any Short Sales (as defined in the Lock-up Agreement) with respect to the Lock-Up Shares until (i) (w) with respect to one hundred percent (100%) of the Lock-Up Shares, the date from the Closing until the last date that is six (6) months after the date of the Closing, (x) with respect to ninety percent (90%) of the Lock-Up Shares, the 1st day of the 7th month from the Closing until the date that is nine (9) months after the date of the Closing, (y) with respect to seventy percent (70%) of the Lock-Up Shares, the 1st day of the 10th month from the Closing until the date that is twelve (12) months after the date of the Closing, (z) with respect to forty percent (40%) of the Lock-Up Shares, the 1st day of the 13th month from the Closing until the date that is fifteen (15) months after the date of the Closing, or (ii) if sooner, the date after the Closing on which Globalink consummates a liquidation, merger, share exchange or other similar transaction with an unaffiliated third party that results in all of Globalink’s stockholders having the right to exchange their equity holdings in Globalink for cash, securities or other property. For the avoidance of doubt, no Lock-up Shares shall be subject to the terms in the Lock-up Agreement from and after the date that is fifteen (15) months after the date of the Closing; | |
| ● | The exercise of Globalink’s directors’ and officers’ discretion in agreeing to changes or waivers in the terms of the Business Combination may result in a conflict of interest when determining whether such changes or waivers are appropriate and in Globalink stockholders’ best interest. Globalink’s directors were aware of and considered these interests, among other matters, in evaluating the Business Combination, and in recommending to Globalink stockholders that they approve the Business Combination. Further, the interests of Globalink’s officers or directors may be different from or in addition to (and which may conflict with) your interests and may be incentivized to complete a less favorable business combination rather than liquidating Globalink. Public stockholders should take these interests into account in deciding whether to approve the Business Combination. | |
| ● | As of September 30, 2024, Globalink’s officers and directors have incurred an aggregate of $11,798 in out-of-pocket expenses. On each of September 5, 2023, September 29, 2023 and November 7, 2023, an affiliate of the Sponsor advanced $130,000 to the Company, for a total advance of $390,000, for Globalink’s extension payments. Unless Globalink consummates an initial business combination, Globalink’s officers, directors and the Sponsor (including its affiliates) will not receive reimbursement for any out-of-pocket expenses incurred by them to the extent that such expenses exceed the amount of available proceeds not deposited in the Trust Account. Upon the consummation of an initial business combination, Globalink’s offices, directors, and the Sponsor (including its affiliates) would be entitled to reimbursement for out-of-pocket expenses incurred by them from the Trust Account disbursements after redemption payouts and payments of deferred underwriting discounts. | |
| ● | The fact that, if the Trust Account is liquidated, including in the event Globalink is unable to complete an initial business combination within the required time period, the Sponsor has agreed to be liable to Globalink if and to the extent any claims by a vendor for services rendered or products sold to Globalink, or a prospective target business with which Globalink has discussed entering into a transaction agreement, reduce the amount of funds in the Trust Account. This liability will not apply with respect to any claims by a third party who executed a waiver of any right, title, interest or claim of any kind in or to any monies held in the Trust Account or to any claims under Globalink’s indemnity of the underwriters of the IPO against certain liabilities, including liabilities under the Securities Act. | |
| ● | As of the date hereof, the deadline that Globalink has to consummate its initial business combination has been extended for a total of eighteen times, and a total of US$1,950,000 has been deposited into the Trust Account in connection with the previous extensions. In connection with funding the extensions, Globalink has issued an aggregate of US$3.85 million in promissory notes to PGM, which are payable upon the Closing. In the event of a liquidation, PGM has waived its rights to any liquidation distributions through the Insider Letter. PGM is an affiliate of the Sponsor as its 95% equity holder has a familial relationship with the control person of the Sponsor. As a result, the Sponsor has an additional interest in causing Globalink to consummate its initial business combination due to its familial relationship with PGM. | |
| ● | After the Closing, the Sponsor will have the sole authority to handle various matters relating to the Escrow Account and indemnifications under the Merger Agreement. The Sponsor may have a conflict of interest when exercising such authority to handle matters relating to the Escrow Account and indemnifications after Closing, and they may not make decisions in the Globalink stockholders’ best interests. For details, see “Summary of the Proxy Statement/Prospectus—Parties to the Business Combination—Parent Representative.” |
| 14 |
These interests may have influenced Globalink’s directors in making their recommendation that you vote in favor of the approval of the Business Combination.
Upon the consummation of an initial business combination by Globalink, other than the PubCo ordinary shares that the Initial Stockholders are entitled to upon conversion of their shares of Globalink common stock at the completion of the Redomestication Merger, no additional compensation will be awarded to, earned by, or paid to the Sponsor, its affiliates or any promoters for all services rendered or to be rendered to Globalink and its affiliates. The Sponsor is expected to own an equity interest of approximately 1.7% in the Combined Company after the consummation of the Business Combination.
Prior to the consummation of an initial business combination by Globalink, the insider shares can only be transferred by the Initial Stockholders to (1) the Initial Stockholders or their respective affiliates or members, or to Globalink’s officers, directors, advisors and employees, (2) the Initial Stockholder’s affiliates or its members upon its liquidation, (3) to relatives and trusts for estate planning purposes, (4) by virtue of the laws of descent and distribution upon death of an Initial Stockholder, (5) pursuant to a qualified domestic relations order, or (6) by private sales made at prices no greater than the price at which the Insider Shares were originally purchased.
| Q: | Are there any arrangements to raise additional funds in connection with the Business Combination? |
| A: | Yes. Globalink, PubCo and Alps Holdco entered into Subscription Agreements, dated as of June 4, 2024, June 5, 2024, and August 27, 2024, respectively, with the PIPE Investors pursuant to which, among other things, PubCo agreed to issue and sell, in a private placement to close immediately prior to the Closing of the Business Combination, PubCo ordinary shares for a total of $40,200,000. To the extent not utilized to consummate the Business Combination, the proceeds from the Trust Account will be used for general corporate purposes, including, but not limited to, working capital for operations, capital expenditures and future acquisitions. PubCo agrees that it (or its successor) will file with the SEC a registration statement registering the resale of the shares purchased in the PIPE Investment and use its commercially reasonable efforts to have the registration statement declared effective as soon as practicable. Globalink’s Sponsor, directors, officers or their affiliates did not participate in the PIPE Investment. The PIPE Investors have not received any payments from the Sponsor in connection with the PIPE Investment. |
| Q: | What conditions must be satisfied to complete the Business Combination? |
| A: | In addition to the approval of the Redomestication Merger Proposal, the Acquisition Merger Proposal, and the Nasdaq Proposals, there are a number of closing conditions in the Merger Agreement, including the approval by Alps Holdco Shareholders of the Business Combination. For a summary of the conditions that must be satisfied or waived prior to the Closing of the Business Combination, see the section titled “The Acquisition Merger Proposal (Proposal 2) — The Merger Agreement” and “Summary of the Proxy Statement/Prospectus – The Proposals — The Acquisition Merger Proposal (Proposal 2) — Conditions to Closing.” |
| Q: | Did Globalink Board obtain a fairness opinion in determining whether to proceed with the Business Combination? |
| A: | The Globalink Board does not plan to obtain a final fairness opinion related to the Business Combination. The Globalink Board considered obtaining a fairness opinion as to the Merger Consideration paid to Alps Holdco Shareholders, and has reached out to Baker Tilly, a financial advisory, tax and assurance services firm, and Morison Advisory Sdn. Bhd., a financial advisory services firm based in Malaysia and a member firm of Morison Global, for a fairness opinion. Neither firm declined or were otherwise unable to or unwilling to provide a fairness opinion to the Globalink Board. On February 2, 2024, Globalink engaged Morison Advisory Sdn. Bhd. to provide a fairness opinion. The Globalink Board received a draft of fairness opinion from Morison Advisory Sdn. Bhd. in June 2024. For a description of the draft fairness opinion that the Globalink Board received, see “The Acquisition Merger Proposal (Proposal 2) — Fairness Opinion.” After a thorough review and internal discussions, the Globalink Board decided not to rely on the draft fairness opinion for determining the valuation of Alps Holdco, as the Globalink Board believed that the valuation methods adopted by Morison Advisory Sdn. Bhd., the income approach using Discounted Cash Flow (“DCF”) method, was reliant upon underlying key assumptions susceptible to material changes and uncertainties. The Globalink Board did not put much weight in considering the draft fairness opinion in determining the valuation of Alps Holdco. Instead, the Globalink Board primarily considered (i) the verbal commitment of Seller Representative and Alps Holdco during the negotiation stage of securing PIPE Investment of an aggregate amount of US$40.2 million or more, (ii) the financing completed by Alps in September 2023 of US$2.5 million, and (iii) the other factors disclosed under “Summary of the Proxy Statement/Prospectus—Globalink Board’s Reasons for the Approval of the Business Combination.” The Globalink Board intends to waive the fairness opinion as a closing condition of the Business Combination. |
| Q: | What happens if a substantial number of the public stockholders vote in favor of the Redomestication Merger Proposal and the Acquisition Merger Proposal, and exercise their redemption rights? |
| A: | Globalink stockholders who vote in favor of the Business Combination may nevertheless also exercise their redemption rights. Accordingly, the Business Combination may be consummated even though the funds available from the Trust Account and the number of public stockholders is reduced as a result of redemptions by public stockholders. Nonetheless, the consummation of the Business Combination is conditioned upon, among other things, the net tangible assets condition required in the Globalink Charter of having $5,000,001 immediately prior and upon consummation of the Business Combination. The immediately prior to consummation of the Business Combination condition is anticipated to be waived. In addition, with fewer public shares and public stockholders, the trading market for the PubCo ordinary shares may be less liquid than the market for Globalink common stock was prior to consummation of the Business Combination and PubCo may not be able to meet the listing standards of the Nasdaq. In addition, with less funds available from the Trust Account, the working capital infusion from the Trust Account into Alps Holdco’s business will be reduced. As a result, the proceeds will be greater in the event that no public stockholders exercise redemption rights with respect to their public shares for a pro rata portion of the Trust Account as opposed to the scenario in which Globalink’ public stockholders exercise the maximum allowed redemption rights. |
| 15 |
| The table below presents possible sources of dilution and the extent of such dilution that non-redeeming public stockholders could experience in connection with the closing of the Business Combination across a range of varying redemption scenarios. The maximum contractual redemption scenario represents the maximum contractual redemptions that may occur, which would allow Globalink to have consolidated net tangible assets of at least $5,000,001 at the consummation of the Business Combination. The standalone requirement of 5,000,001 of Globalink net tangible assets is expected to be waived by the parties. In an effort to illustrate the extent of such dilution, the table below assumes that no Earnout Shares are issued. |
Assuming No Redemptions | Assuming 25% Redemptions | Assuming 50% Redemptions | Assuming 75% Redemptions | Assuming Maximum Contractual Redemptions | ||||||||||||||||||||||||||||||||||||
Number of Shares | % | Number of Shares | % | Number of Shares | % | Number of Shares | % | Number of Shares | % | |||||||||||||||||||||||||||||||
| Alps Holdco Shareholders | 160,000,000 | 93.8 | % | 160,000,000 | 93.8 | % | 160,000,000 | 93.8 | % | 160,000,000 | 93.9 | % | 160,000,000 | 93.9 | % | |||||||||||||||||||||||||
| Globalink Public Shareholders | 277,511 | 0.2 | % | 208,133 | 0.1 | % | 138,755 | 0.1 | % | 69,377 | 0.0 | % | 0 | 0.0 | % | |||||||||||||||||||||||||
| Initial Stockholders | 2,875,000 | 1.7 | % | 2,875,000 | 1.7 | % | 2,875,000 | 1.7 | % | 2,875,000 | 1.7 | % | 2,875,000 | 1.7 | % | |||||||||||||||||||||||||
| Globalink Public Rights Holders | 1,150,000 | 0.6 | % | 1,150,000 | 0.7 | % | 1,150,000 | 0.7 | % | 1,150,000 | 0.7 | % | 1,150,000 | 0.7 | % | |||||||||||||||||||||||||
| PIPE Investors | 4,020,000 | 2.4 | % | 4,020,000 | 2.4 | % | 4,020,000 | 2.4 | % | 4,020,000 | 2.4 | % | 4,020,000 | 2.4 | % | |||||||||||||||||||||||||
| PGM | 627,000 | 0.4 | % | 627,000 | 0.4 | % | 627,000 | 0.4 | % | 627,000 | 0.4 | % | 627,000 | 0.4 | % | |||||||||||||||||||||||||
| IBDC Asia Sdn. Bhd. | 1,600,000 | 0.9 | % | 1,600,000 | 0.9 | % | 1,600,000 | 0.9 | % | 1,600,000 | 0.9 | % | 1,600,000 | 0.9 | % | |||||||||||||||||||||||||
| Total Shares | 170,549,511 | 100 | % | 170,480,133 | 100 | % | 170,410,755 | 100 | % | 170,341,377 | 100 | % | 170,272,000 | 100 | % | |||||||||||||||||||||||||
The table below illustrates the potential dilutive impact, excluding the exercise of warrants, to the holders of securities across five different redemption scenarios based on the assumptions described above, except that these scenarios assume the Combined Company has achieved all revenue milestones and the maximum number of Earnout Shares (48,000,000) are issued according to the earnout arrangement:
Assuming No Redemptions | Assuming 25% Redemptions | Assuming 50% Redemptions | Assuming 75% Redemptions | Assuming Maximum Contractual Redemptions | ||||||||||||||||||||||||||||||||||||
Number of Shares | % | Number of Shares | % | Number of Shares | % | Number of Shares | % | Number of Shares | % | |||||||||||||||||||||||||||||||
| Alps Holdco Shareholders | 208,000,000 | 95.2 | % | 208,000,000 | 95.2 | % | 208,000,000 | 95.2 | % | 208,000,000 | 95.3 | % | 208,000,000 | 95.3 | % | |||||||||||||||||||||||||
| Globalink Public Shareholders | 277,511 | 0.1 | % | 208,133 | 0.1 | % | 138,755 | 0.1 | % | 69,377 | 0.0 | % | 0 | 0.0 | % | |||||||||||||||||||||||||
| Initial Stockholders | 2,875,000 | 1.3 | % | 2,875,000 | 1.3 | % | 2,875,000 | 1.3 | % | 2,875,000 | 1.3 | % | 2,875,000 | 1.3 | % | |||||||||||||||||||||||||
| Globalink Public Rights Holders | 1,150,000 | 0.5 | % | 1,150,000 | 0.5 | % | 1,150,000 | 0.5 | % | 1,150,000 | 0.5 | % | 1,150,000 | 0.5 | % | |||||||||||||||||||||||||
| PIPE Investors | 4,020,000 | 1.9 | % | 4,020,000 | 1.9 | % | 4,020,000 | 1.9 | % | 4,020,000 | 1.9 | % | 4,020,000 | 1.9 | % | |||||||||||||||||||||||||
| PGM | 627,000 | 0.3 | % | 627,000 | 0.3 | % | 627,000 | 0.3 | % | 627,000 | 0.3 | % | 627,000 | 0.3 | % | |||||||||||||||||||||||||
| IBDC Asia Sdn. Bhd. | 1,600,000 | 0.7 | % | 1,600,000 | 0.7 | % | 1,600,000 | 0.7 | % | 1,600,000 | 0.7 | % | 1,600,000 | 0.7 | % | |||||||||||||||||||||||||
| Total Shares | 218,549,511 | 100 | % | 218,480,133 | 100 | % | 218,410,755 | 100 | % | 218,341,377 | 100 | % | 218,272,000 | 100 | % | |||||||||||||||||||||||||
| The deferred underwriting discounts in connection with the IPO will be paid to the underwriters only on completion of the Business Combination. The following table presents the deferred underwriting discounts as a percentage of the cash left in the Trust Account following redemptions across a range of varying redemption scenarios. The maximum contractual redemption scenario represents the maximum contractual redemptions that may occur, which would allow Globalink to have consolidated net tangible assets of at least $5,000,001 at the consummation of the Business Combination. The standalone requirement of 5,000,001 of Globalink net tangible assets is expected to be waived by the parties. For purposes of calculating the redemption scenarios, the trust value date as of December 6, 2024 is used. |
| Assuming No Redemptions | Assuming 25% Redemptions | Assuming 50% Redemptions | Assuming 75% Redemptions | Assuming Maximum Contractual Redemptions(1) | ||||||||||||||||
| Deferred underwriting discounts | $ | 4,025,000 | $ | 4,025,000 | $ | 4,025,000 | $ | 4,025,000 | $ | 4,025,000 | ||||||||||
| Deferred underwriting discounts as a percentage of cash left in the Trust Account following redemptions | 133 | % | 178 | % | 266 | % | 533 | % | 0 | % | ||||||||||
| (1) | The maximum contractual redemption scenario represents the maximum contractual redemptions that may occur but which would still allow Globalink to have consolidated net tangible assets of at least $5,000,001 at the consummation of the Business Combination. The standalone requirement of 5,000,001 of Globalink net tangible assets is expected to be waived by the parties. The maximum contractual redemption scenario assumes 277,511 public shares of Globalink common stock are redeemed for the proceeds in the Trust Account. |
| 16 |
| Q: | How do the public warrants differ from the private warrants and what are the related risks to any holders of public warrants following the Business Combination? |
| A: | The private warrants are identical to the public warrants in all material respects, except that the private warrants and the shares of common stock issuable upon the exercise of the private warrants will not be transferable, assignable or saleable until after the completion of a Business Combination, subject to certain limited exceptions. Additionally, the private warrants will be exercisable for cash or on a cashless basis, at the holder’s option, and be non-redeemable so long as they are held by the initial purchasers or their permitted transferees. If the private warrants are held by someone other than the initial purchasers or their permitted transferees, the private warrants will be redeemable by Globalink and exercisable by such holders on the same basis as the public warrants. |
Following the Business Combination, PubCo may redeem the public PubCo warrants, prior to their exercise at a time that is disadvantageous to the holder, thereby significantly impairing the value of such warrants. PubCo will have the ability to redeem outstanding public PubCo warrants at any time after they become exercisable and prior to their expiration, at a price of $0.01 per warrant, provided that the closing price of the PubCo ordinary shares equals or exceeds $16.50 per share (as adjusted for share splits, share dividends, reorganizations, recapitalizations and the like) for any 20 trading days within a 30 trading day period ending on the third trading day prior to the date on which a notice of redemption is sent to the warrant holders. PubCo will not redeem the warrants as described above unless a registration statement under the Securities Act covering the ordinary shares issuable upon exercise of such warrants is effective and a current prospectus relating to those ordinary shares is available throughout the 30-day redemption period, except if the warrants may be exercised on a cashless basis and such cashless exercise is exempt from registration under the Securities Act. If and when the public PubCo warrants become redeemable by PubCo, if PubCo has elected to require the exercise of public PubCo warrants on a cashless basis, PubCo will not redeem the warrants as described above if the issuance of ordinary shares upon exercise of public PubCo warrants is not exempt from registration or qualification under applicable state blue sky laws or PubCo is unable to effect such registration or qualification. Redemption of the outstanding public PubCo warrants could force you (i) to exercise your public PubCo warrants and pay the exercise price therefor at a time when it may be disadvantageous for you to do so, (ii) to sell your public PubCo warrants at the then-current market price when you might otherwise wish to hold your public PubCo warrants, or (iii) to accept the nominal redemption price which, at the time the outstanding public PubCo warrants are called for redemption, is likely to be substantially less than the market value of your public PubCo warrants.
In the event PubCo determines to redeem the public PubCo warrants, holders of public PubCo warrants would be notified of such redemption as described in Globalink’s warrant agreement. Specifically, in the event that PubCo elects to redeem all of the public warrants as described above, PubCo will fix a date for the redemption (the “Redemption Date”). Notice of redemption will be mailed by first class mail, postage prepaid, by PubCo not less than 30 days prior to the Redemption Date to the registered holders of the public PubCo warrants to be redeemed at their last addresses as they appear on the registration books. Any notice mailed in the manner provided in the warrant agreement will be conclusively presumed to have been duly given whether or not the registered holder received such notice. In addition, beneficial owners of the public PubCo warrants will be notified of such redemption via PubCo’s posting of the redemption notice to DTC.
The closing price for the Globalink’s common stock as of December 12, 2024 was $11.95 and has never exceeded the $16.50 threshold that would trigger the right to redeem the public PubCo warrants following the Closing.
| Q: | When and where is the Special Meeting? |
| A: | The Special Meeting will take place on , 2025, at a.m., Eastern Time. |
| Q: | Who may vote at the Special Meeting? |
| A: | Only holders of record of Globalink’s common stock as of the close of business on , 2025 may vote at the Special Meeting. As of , 2025, there were shares of Globalink common stock outstanding and entitled to vote. Please see “The Special Meeting — Record Date; Who is Entitled to Vote” for further information. |
| 17 |
| Q: | What is the quorum requirement for the Special Meeting? |
| A: | Stockholders representing a majority of the shares of common stock issued and outstanding as of the Record Date and entitled to vote at the Special Meeting must be present in person by virtual attendance or represented by proxy in order to hold the Special Meeting and conduct business. This is called a quorum. Shares of Globalink common stock will be counted for purposes of determining if there is a quorum if the stockholder (i) is present and entitled to vote at the meeting, or (ii) has properly submitted a proxy card or voting instructions through a broker, bank or custodian. In the absence of a quorum, stockholders representing a majority of the votes present in person or represented by proxy at such meeting may adjourn the meeting until a quorum is present. As of the Record Date, 1,861,256 shares of Globalink common stock would be required to achieve a quorum. |
| Q: | Am I required to vote against the Redomestication Merger Proposal or the Acquisition Merger Proposal in order to have my shares of Globalink common stock redeemed? |
| A: | No. You are not required to vote against the Redomestication Merger Proposal or the Acquisition Merger Proposal in order to have the right to demand that Globalink redeem your shares of Globalink common stock for cash equal to your pro rata share of the aggregate amount then on deposit in the Trust Account (before payment of deferred underwriting commissions and including interest earned on their pro rata portion of the Trust Account, net of taxes payable). These rights to demand redemption of public shares for cash are sometimes referred to herein as “redemption rights.” If the Business Combination is not completed, public stockholders electing to exercise their redemption rights will not be entitled to receive such payments and their shares of common stock will be returned to them. |
| Q: | How do I exercise my redemption rights? |
| A: | If you are a public stockholder and you seek to have your public shares redeemed, you must (i) demand, no later than p.m., Eastern Time on , 2025 (at least two business days before the Special Meeting), that Globalink redeem your shares into cash; and (ii) submit your request in writing to Continental, at the address listed at the end of this section and deliver your shares to Continental physically or electronically using The Depository Trust Company’s (“DTC”) Deposit/Withdrawal at Custodian (“DWAC”) System at least two business days before the Special Meeting. |
| Any corrected or changed written demand of redemption rights must be received by Continental two business days before the Special Meeting. No demand for redemption will be honored unless the holder’s shares have been delivered (either physically or electronically) at least two business days before the Special Meeting to Continental, Globalink’s transfer agent, at the following address: |
Continental Stock Transfer & Trust Company
One State Street Plaza, 30th Floor
New York, New York 10004
Attn: SPAC Redemption Team
E-mail: spacredemptions@continentalstock.com
| Please also affirmatively certify in your request to Continental for redemption if you “ARE” or “ARE NOT” acting in concert or as a “group” (as defined in Section 13d-3 of the Exchange Act) with any other stockholder with respect to shares of Globalink common stock. A holder of the public shares, together with any affiliate of his or any other person with whom he is acting in concert or as a “group” (as defined in Section 13d-3 of the Exchange Act) will be restricted from seeking redemption rights with respect to an aggregate of 20% or more of the public shares, which we refer to as the “20% threshold.” Accordingly, all public shares in excess of the 20% threshold beneficially owned by a public stockholder or group will not be redeemed for cash. |
| 18 |
| Public stockholders seeking to exercise their redemption rights and opting to deliver physical certificates should allot sufficient time to obtain physical certificates from the transfer agent and time to effect delivery. It is Globalink’s understanding that stockholders should generally allot at least two weeks to obtain physical certificates from the transfer agent. However, Globalink does not have any control over this process and it may take longer than two weeks. Stockholders who hold their shares in “street name” will have to coordinate with their bank, broker or other nominee to have the shares certificated or delivered electronically. | |
| Any demand for redemption, once made, may be withdrawn at any time until the deadline for exercising redemption requests and thereafter, with Globalink’s consent, until the consummation of the Business Combination, or such other date as determined by the Globalink Board. If you delivered your shares for redemption to Globalink’s transfer agent and decide within the required timeframe not to exercise your redemption rights, you may request that Globalink’s transfer agent return the shares (physically or electronically). You may make such request by contacting Globalink’s transfer agent at the phone number or address listed under the question “Who can help answer my questions?” below. |
| Q: | What are the U.S. federal income tax consequences of exercising my redemption rights? |
| A: | In the event that a U.S. Holder (as defined herein) elects to redeem its shares of common stock for cash, the treatment of the transaction for U.S. federal income tax purposes will depend on whether the redemption qualifies as a sale or exchange of common stock under Section 302 of the Code or is treated as a distribution under Section 301 of the Code. Whether the redemption qualifies as a sale or exchange or is treated as a distribution will depend on the facts and circumstances of each particular U.S. Holder at the time such holder exercises his, her, or its redemption rights. If the redemption qualifies as a sale or exchange of the shares of common stock, the U.S. Holder will be treated as recognizing capital gain or loss equal to the difference between the amount realized on the redemption and such U.S. Holder’s adjusted tax basis in the shares of common stock surrendered in such redemption transaction. Any such capital gain or loss generally will be long-term capital gain or loss if the U.S. Holder’s holding period for the shares of common stock redeemed exceeds one year. The deductibility of capital losses is subject to limitations. See “Material U.S. Federal Income Tax Consequences — Certain Material U.S. Federal Income Tax Consequences of Exercising Redemption Rights” for a more detailed discussion of the U.S. federal income tax consequences of a holder electing to redeem its shares of common stock for cash.
Subject to the PFIC rules, long-term capital gains recognized by non-corporate U.S. Holders will be eligible to be taxed at reduced rates. However, it is unclear whether the redemption rights with respect to the public shares may prevent a U.S. Holder from satisfying the applicable holding period requirements. Long-term capital gains recognized by non-corporate U.S. Holders will be eligible to be taxed at reduced rates. The deductibility of capital losses is subject to limitations. See “Material U.S. Federal Income Tax Consequences — Certain U.S. Federal Income Tax Consequences of Exercising Redemption Rights” and “Material U.S. Federal Income Tax Consequences — Passive Foreign Investment Company Status” for a more detailed discussion of the U.S. federal income tax consequences of a U.S. Holder electing to redeem its public shares for cash, including with respect to PubCo’s potential PFIC status and certain tax implications thereof.
Additionally, because the Redomestication Merger will occur prior to the redemption by U.S. Holders that exercise redemption rights with respect to Public Shares, U.S. Holders exercising such redemption rights will be subject to the potential tax consequences of section 367(a) of the Code and the PFIC rules. The tax consequences of the exercise of redemption rights, including pursuant to Section 367(a) of the Code and the PFIC rules, are discussed more fully below under “Material U.S. Federal Income Tax Consequences — Certain U.S. Federal Income Tax Consequences to U.S. Holders of Exercising Redemption Rights.” All holders of public shares considering exercising their redemption rights are urged to consult their tax advisor on the tax consequences to them of an exercise of redemption rights, including the applicability and effect of U.S. federal, state, local and non-U.S. income and other tax laws. |
| 19 |
| Q: | If I am a warrant or right holder, can I exercise redemption rights with respect to my warrants or rights? |
| A: | No. The holders of warrants and rights have no redemption rights with respect to warrants or rights. |
| Q: | If I am a public unit holder, can I exercise redemption rights with respect to my units? |
| A: | No. Holders of outstanding public units must separate the underlying public shares, public rights and public warrants prior to exercising redemption rights with respect to the public shares. |
If you hold units registered in your own name, you must deliver the certificate for such units to Continental, Globalink’s transfer agent, with written instructions to separate such units into public shares, public rights and public warrants. This must be completed far enough in advance to permit the mailing of the stock certificates for the public shares back to you so that you may then exercise your redemption rights upon the separation of the public shares from the units. See “How do I exercise my redemption rights?” above. The address of Continental is listed under the question “Who can help answer my questions?” below.
If a broker, dealer, commercial bank, trust company or other nominee holds your units, you must instruct such nominee to separate your units. Your nominee must send written instructions by facsimile to Continental, Globalink’s transfer agent. Such written instructions must include the number of units to be split and the nominee holding such units. Your nominee must also initiate electronically, using The DTC’s DWAC system, a withdrawal of the relevant units and a deposit of an equal number of public shares and public warrants. This must be completed far enough in advance to permit your nominee to exercise your redemption rights upon the separation of the public shares from the units. While this is typically done electronically the same business day, you should allow at least one full business day to accomplish the separation. If you fail to cause your public shares to be separated in a timely manner, you will likely not be able to exercise your redemption rights.
| Q: | What do I need to do now? |
| A: | We urge you to read carefully and consider the information contained in this proxy statement/prospectus, including the annexes, and consider how the Business Combination will affect you as a Globalink stockholder. You should vote as soon as possible in accordance with the instructions provided in this proxy statement/prospectus and on the enclosed proxy card or, if you hold your shares through a brokerage firm, bank or other nominee, on the voting instruction form provided by the broker, bank or nominee. |
| Q: | How can I vote? |
| A: | If you are a stockholder of record, you may vote online at the virtual Special Meeting or vote by proxy using the enclosed proxy card, the Internet or telephone. Whether or not you plan to participate in the Special Meeting, we urge you to vote by proxy to ensure your vote is counted. Even if you have already voted by proxy, you may still attend the virtual Special Meeting and vote online, if you choose. |
To vote online at the virtual Special Meeting, follow the instructions below under “How may I participate in the virtual Special Meeting?”
To vote using the proxy card, please complete, sign and date the proxy card and return it in the prepaid envelope. If you return your signed proxy card before the Special Meeting, we will vote your shares as you direct.
To vote via the telephone, you can vote by calling the telephone number on your proxy card. Please have your proxy card handy when you call. Easy-to-follow voice prompts will allow you to vote your shares and confirm that your instructions have been properly recorded.
To vote via the Internet, please go to https://www.cstproxy.com/ and follow the instructions. Please have your proxy card handy when you go to the website. As with telephone voting, you can confirm that your instructions have been properly recorded.
| 20 |
Telephone and Internet voting facilities for stockholders of record will be available 24 hours a day until 11:59 p.m. Eastern Time on , 2025. After that, telephone and Internet voting will be closed, and if you want to vote your shares, you will either need to ensure that your proxy card is received before the date of the Special Meeting or attend the virtual Special Meeting to vote your shares online.
If your shares are registered in the name of your broker, bank or other agent, you are the “beneficial owner” of those shares and those shares are considered as held in “street name.” If you are a beneficial owner of shares registered in the name of your broker, bank or other agent, you should have received a proxy card and voting instructions with these proxy materials from that organization rather than directly from Globalink. Simply complete and mail the proxy card to ensure that your vote is counted. You may be eligible to vote your shares electronically over the Internet or by telephone. A large number of banks and brokerage firms offer Internet and telephone voting. If your bank or brokerage firm does not offer Internet or telephone voting information, please complete and return your proxy card in the self-addressed, postage-paid envelope provided.
If you plan to vote at the virtual Special Meeting, you will need to contact Continental at the phone number or email below to receive a control number and you must obtain a legal proxy from your broker, bank or other nominee reflecting the number of shares of common stock you held as of the Record Date, your name and email address. You must contact Continental for specific instructions on how to receive the control number. Please allow up to 48 hours prior to the Special Meeting for processing your control number.
After obtaining a valid legal proxy from your broker, bank or other agent, to then register to attend the Special Meeting, you must submit proof of your legal proxy reflecting the number of your shares along with your name and email address to Continental. Requests for registration should be directed to 917-262-2373 or email proxy@continentalstock.com. Requests for registration must be received no later than p.m., Eastern Time, on , 2025.
You will receive a confirmation of your registration by email after Globalink receives your registration materials. We encourage you to access the Special Meeting prior to the start time leaving ample time for the check in.
| Q: | How may I participate in the virtual Special Meeting? |
| A. | If you are a stockholder of record as of the Record Date for the Special Meeting, you should receive a proxy card from Continental, containing instructions on how to attend the virtual Special Meeting including the URL address, along with your control number. You will need your control number for access. If you do not have your control number, contact Continental at 917-262-2373 or email proxy@continentalstock.com. |
| You can pre-register to attend the virtual Special Meeting starting on , 2025. Go to https://www.cstproxy.com/ , enter the control number found on your proxy card you previously received, as well as your name and email address. Once you pre-register you can vote or enter questions in the chat box. At the start of the Special Meeting you will need to re-log into https://www.cstproxy.com/ using your control number. | |
| If your shares are held in street name, and you would like to join and not vote, Continental will issue you a guest control number. Either way, you must contact Continental for specific instructions on how to receive the control number. Please allow up to hours prior to the Special Meeting for processing your control number. | |
| Q: | If my shares are held in “street name” by my bank, brokerage firm or nominee, will they automatically vote my shares for me? |
| A: | No. Your broker, bank or nominee cannot vote your shares of Globalink common stock with respect to non-discretionary matters unless you provide instructions on how to vote in accordance with the information and procedures provided to you by your broker, bank or nominee. If a Proposal is determined to be discretionary, your broker, bank or other holder of record is permitted to vote on the Proposal without receiving voting instructions from you. If a Proposal is determined to be non-discretionary, your broker, bank or other holder of record is not permitted to vote on the Proposal without receiving voting instructions from you. |
| 21 |
| Globalink believes that each of the Proposals presented at the Special Meeting except the Adjournment Proposal is a non-discretionary proposal and, therefore, your broker, bank or nominee cannot vote your shares without your instruction. Broker non-votes will not be considered present for the purposes of establishing a quorum and will have no effect on any of the Proposals. If you do not provide instructions with your proxy, your bank, broker or other nominee may submit a proxy card expressly indicating that it is NOT voting your shares of Globalink common stock; this indication that a bank, broker or nominee is not voting your shares of Globalink common stock is referred to as a “broker non-vote.” Your bank, broker or other nominee can vote your shares of Globalink common stock only if you provide instructions on how to vote. You should instruct your broker to vote your shares of Globalink common stock in accordance with directions you provide. However, the Adjournment Proposal is considered a routine proposal. Accordingly, your broker, bank or nominee may vote your shares with respect to such proposal without receiving voting instructions. | |
| Q: | What if I abstain from voting or fail to instruct my bank, brokerage firm or nominee? |
| A: | Globalink will count a properly executed proxy marked “ABSTAIN” with respect to a particular Proposal as present for the purposes of determining whether a quorum is present at the Special Meeting. For purposes of approval, an abstention will have no effect on the outcome of the Redomestication Merger Proposal, the Acquisition Merger Proposal, the Nasdaq Proposals and the Adjournment Proposal. |
| Q: | If I am not going to attend the Special Meeting, should I return my proxy card instead? |
| A. | Yes. Whether you plan to attend the Special Meeting virtually or not, please read the enclosed proxy statement/prospectus carefully, and vote your shares by completing, signing, dating and returning the enclosed proxy card in the postage-paid envelope provided. |
| Q: | How can I submit a proxy? |
| A. | You may submit a proxy by (a) visiting https://www.cstproxy.com/ and following the on screen instructions (have your proxy card available when you access the webpage), or (b) calling toll-free in the U.S. or from foreign countries from any touch-tone phone and follow the instructions (have your proxy card available when you call), or (c) submitting your proxy card by mail by using the previously provided self-addressed, stamped envelope. |
| Q: | Can I change my vote after I have mailed my proxy card? |
| A: | Yes. You may change your vote at any time before your proxy is voted at the Special Meeting. You may revoke your proxy by executing and returning a proxy card dated later than the previous one, or by attending the Special Meeting in person by virtual attendance and by casting your vote, voting again by telephone or Internet voting options described below, or by submitting a written revocation stating that you would like to revoke your proxy that Globalink’s proxy solicitor receives prior to the Special Meeting. If you hold your shares of Globalink common stock through a bank, brokerage firm or nominee, you should follow the instructions of your bank, brokerage firm or nominee regarding the revocation of proxies. If you are a record holder, you should send any notice of revocation or your completed new proxy card, as the case may be, to: |
| Continental Stock Transfer & Trust Company | |
| One State Street Plaza, 30th Floor | |
| New York, New York 10004 | |
| Tell: 917-262-2373 | |
| Email: proxy@continentalstock.com | |
| Unless revoked, a proxy will be voted at the virtual Special Meeting in accordance with the stockholder’s indicated instructions. In the absence of instructions, proxies will be voted FOR each of the Proposals. |
| 22 |
| Q: | What will happen if I return my proxy card without indicating how to vote? |
| A: | If you sign and return your proxy card without indicating how to vote on any particular Proposal, the shares of common stock represented by your proxy will be voted in favor of each Proposal. Proxy cards that are returned without a signature will not be counted as present at the Special Meeting and cannot be voted. |
| Q: | Should I send in my share certificates now to have my shares of common stock redeemed? |
| A: | Globalink stockholders who intend to have their shares redeemed should send their certificates to Continental at least two business days before the Special Meeting. Please see “The Special Meeting — Redemption Rights” for the procedures to be followed if you wish to redeem your shares for cash. |
| Q: | Who will solicit the proxies and pay the cost of soliciting proxies for the Special Meeting? |
| A: | Globalink will pay the cost of soliciting proxies for the Special Meeting. Globalink has engaged Okapi Partners LLC to assist in the solicitation of proxies for the Special Meeting. Globalink has agreed to pay Okapi Partners LLC a fee of $20,000, plus disbursements, and will reimburse for its reasonable out-of-pocket expenses and indemnify Okapi Partners LLC and its affiliates against certain claims, liabilities, losses, damages, and expenses. Globalink will also reimburse banks, brokers and other custodians, nominees and fiduciaries representing beneficial owners of common stock for their expenses in forwarding soliciting materials to beneficial owners of the common stock and in obtaining voting instructions from those owners. Globalink’s directors, officers and employees may also solicit proxies by telephone, by facsimile, by mail, on the Internet or in person. They will not be paid any additional amounts for soliciting proxies. |
| Q: | What happens if I sell my shares before the Special Meeting? |
| A: | The Record Date for the Special Meeting is earlier than the date of the Special Meeting, as well as the date that the Business Combination is expected to be consummated. If you transfer your shares of common stock after the Record Date, but before the Special Meeting, unless the transferee obtains from you a proxy to vote those shares, you would retain your right to vote at the Special Meeting, but will transfer ownership of the shares and will not hold an interest in Globalink after the Business Combination is consummated. |
| Q: | When is the Business Combination expected to occur? |
| A: | Assuming the requisite regulatory and stockholder approvals are received, Globalink expects that the Business Combination will occur as soon as possible following the Special Meeting. |
| Q: | Are Alps Holdco’s shareholders required to approve the Business Combination? |
| A: | Yes. The approval by Alps Holdco Shareholders of the Business Combination is a closing condition in the Merger Agreement. |
| Q: | Are there risks associated with the Business Combination that I should consider in deciding how to vote? |
| A: | Yes. There is a number of risks related to the Business Combination and other transactions contemplated by the Merger Agreement that are discussed in this proxy statement/prospectus. Please read with particular care the detailed description of the risks described in “Risk Factors” beginning on page 52 of this proxy statement/prospectus. |
| 23 |
| Q: | May I seek statutory appraisal rights or dissenter rights with respect to my shares of Globalink common stock? |
| A: | No. Appraisal rights are not available to holders of shares of Globalink common stock in connection with the proposed Business Combination. For additional information, see the section titled “The Special Meeting — Appraisal Rights and Dissenter’s Rights.” |
| Q: | What happens if the Business Combination is not consummated? |
| A: | If Globalink does not consummate the Business Combination by January 9, 2025, then pursuant to Article VI of the Globalink Charter, Globalink’s officers must take all actions necessary in accordance with the DGCL to dissolve and liquidate Globalink as soon as reasonably possible. If Globalink anticipates that it may not be able to consummate its initial business combination in time, Globalink may, by resolutions of its Board, if requested by the Sponsor, extend the period of time to consummate a business combination on a monthly basis to until June 9, 2025, subject to the Sponsor depositing additional funds into the Trust Account. Following dissolution, Globalink will no longer exist as a company. In any liquidation, the funds held in the Trust Account, plus any interest earned thereon (net of taxes payable), together with any remaining out-of-trust net assets, will be distributed pro-rata to holders of shares of common stock who acquired such shares in the IPO or in the aftermarket. The estimated consideration that each share of Globalink common stock would be paid at liquidation would be approximately $11.98 per share for stockholders based on amounts on deposit in the Trust Account as of December 6, 2024. The closing price of Globalink common stock on the Nasdaq as of December 12, 2024 was $11.95. The Initial Stockholders and PGM waived the right to any liquidation distribution with respect to any shares of common stock held by them at the time that the Founder Shares were purchased for no additional consideration. |
| In the event of liquidation, there will be no liquidating distributions with respect to Globalink’s outstanding rights and warrants. Accordingly, the rights and warrants will expire worthless. | |
| Q: | What happens to the funds deposited in the Trust Account following the Business Combination? |
| A: | Following the closing of the Business Combination, holders of public shares of Globalink common stock exercising redemption rights will receive their per share redemption price out of the funds in the Trust Account. The balance of the funds will be released to Alps Holdco and utilized to fund working capital needs of the Combined Company. As of December 6, 2024, there was approximately $3.33 million in the Trust Account. Globalink estimates that approximately $10.98 per outstanding share, net of interest available to pay taxes, will be paid to the public investors exercising their redemption rights. |
| Q: | Who will manage PubCo after the Business Combination? |
| A: | Upon the Closing of the Business Combination, all of the officers of Alps Holdco will become the initial officers of PubCo and shall hold office until their respective successors are duly elected or appointed and qualified, or until their earlier death, resignation or removal. Following the Business Combination, PubCo also expects to appoint a Chief Financial Officer. Alps is in the process of evaluating and seeking suitable candidates to serve as the Chief Financial Officer of the Combined Company. Alps Holdco will designate all members of the Combined Company’s Board of Directors, at least three (3) of whom shall qualify as independent directors under the Securities Act and the Nasdaq rules. For information on the anticipated management of the Combined Company, see the section titled “Directors and Executive Officers of the Combined Company” in this proxy statement/prospectus. |
| Q: | Is the consummation of the Business Combination subject to any conditions? |
| A: | Yes. The consummation of the Business Combination is subject to conditions, as more fully described in the section titled “The Acquisition Merger Proposal (Proposal 2) — Conditions to the Closing of the Business Combination” in this proxy statement/prospectus. |
| 24 |
| Q: | Who can help answer my questions? |
| A: | If you have questions about the Proposals or if you need additional copies of this proxy statement/prospectus or the enclosed proxy card you should contact: |
Say Leong Lim
Chief Executive Officer
200 Continental Drive, Suite 401
Newark, Delaware 19713
+6012 405 0015
You may also contact Globalink’s proxy solicitor at:
Okapi Partners LLC
1212 Avenue of the Americas, 17th Floor
New York, New York 10036
Individuals call toll-free (855) 305-0857
Banks and brokers call (212) 297-0720
Email: info@okapipartners.com
To obtain timely delivery, Globalink stockholders must request the materials no later than , 2025.
If you intend to seek redemption of your public shares, you will need to send a letter demanding redemption and deliver your shares (either physically or electronically) to Globalink’s transfer agent prior to the Special Meeting in accordance with the procedures detailed under the question “How do I exercise my redemption rights?” If you have questions regarding the certification of your position or delivery of your stock, please contact:
Continental Stock Transfer & Trust Company
One State Street Plaza, 30th Floor
New York, New York 10004
Attn: SPAC Redemption Team
E-mail: spacredemptions@continentalstock.com
You may also obtain additional information about Globalink from documents filed with the SEC by following the instructions in the section titled “Where You Can Find More Information.”
| 25 |
SUMMARY OF THE PROXY STATEMENT/PROSPECTUS
This summary highlights selected information from this proxy statement/prospectus but may not contain all of the information that may be important to you. Accordingly, we encourage you to carefully read this entire proxy statement/prospectus, including the Merger Agreement attached as Annex A. Please read these documents carefully as they are the legal documents that govern the Business Combination and your rights in the Business Combination. See also the section titled “Where You Can Find More Information” of this proxy statement/prospectus.
Unless otherwise indicated or the context otherwise requires, references in this summary to the “Combined Company” are to PubCo and its consolidated subsidiaries after giving effect to the Business Combination, including Alps Holdco. References to “Globalink” are to Globalink Investment Inc. and references to “Alps Holdco” are to Alps Life Sciences Inc.
Parties to the Business Combination
Globalink Investment Inc.
Globalink Investment Inc. was incorporated as a blank check company formed under the laws of the State of Delaware on March 24, 2021. Globalink Investment Inc. is a blank check company formed for the purpose of effecting a merger, share exchange, asset acquisition, share purchase, reorganization or similar business combination with one or more businesses.
The Globalink common stock, warrants, rights and units are currently quoted on the OTC Pink, under the symbols “GLLI,” “GLLIW,” “GLLIR,” and “GLLIU,” respectively. The Globalink common stock, warrants, rights and units commenced trading on the Nasdaq separately on or about December 22, 2021, and were delisted from Nasdaq on December 17, 2024.
If the Globalink stockholders approve the Redomestication Merger Proposal and the Acquisition Merger Proposal, immediately prior to the consummation of the Business Combination, all outstanding units of Globalink will separate into their individual components of Globalink common stock, Globalink rights and Globalink warrants and will cease separate existence and trading.
Upon the consummation of the Business Combination the current equity holdings of the Globalink stockholders shall be exchanged as follows:
| (i) | Each share of Globalink common stock, par value $0.001 per share, issued and outstanding immediately prior to the effective time of the Redomestication Merger (other than any redeemed shares), will automatically be cancelled and cease to exist and for each share of Globalink common stock, PubCo shall issue to its holder (other than Globalink stockholders who exercise their redemption rights in connection with the Business Combination) one validly issued PubCo ordinary share, which shall be fully paid; | |
| (ii) | Each Globalink warrant to purchase one-half (1/2) of one share of Globalink common stock issued and outstanding immediately prior to effective time of the Redomestication Merger will convert into one warrant to purchase one-half (1/2) of one PubCo ordinary share (“PubCo warrant”) (or equivalent portion thereof). The PubCo warrants will have substantially the same terms and conditions as set forth in the Globalink warrants; and | |
| (iii) | The holders of Globalink rights (convertible into one-tenth (1/10) of one share of Globalink common stock) issued and outstanding immediately prior to the effective time of the Redomestication Merger will obtain the right to receive one-tenth (1/10) of one PubCo ordinary share (“PubCo right”) in exchange for the cancellation of each Globalink right; provided, however, that no fractional shares will be issued and all fractional shares will be rounded to the nearest whole share. |
| 26 |
PubCo intends to apply for the listing of its ordinary shares and warrants on the Nasdaq under the symbols “ALPS” and “ALPSW”, respectively, in connection with the closing of the Business Combination. We cannot assure you that the PubCo ordinary shares and PubCo warrants will be approved for listing on Nasdaq.
Globalink’s principal executive offices are located at 200 Continental Drive, Suite 401, Newark, Delaware 19713 and its telephone number is +6012 405 0015.
Alps Holdco
Alps Life Sciences Inc is an exempted company incorporated in the Cayman Islands with limited liability on April 11, 2024 and Alps Global Holding Berhad (“Alps”) is a public limited company incorporated in Malaysia on April 14, 2017 and will be a subsidiary of Alps Holdco before Closing. Alps’ primary objective is to evolve into a fully integrated bench-to-bedside platform encompassing biotechnology research, medical services, and wellness solutions. Alps’ principal activities are research and development in various biotechnology-related products and services, with a focus on the use of precision and preventive medicine.
In pursuit of its vision, Alps has cultivated a diverse pipeline covering various biotechnology research areas. This includes initiatives focused on MyImmune (NK cells), CAR-T cells, COVID-19 mRNA vaccine, diabetes therapeutics, and advancements in stem cell technologies such as induced Pluripotent Stem Cells (iPSCs) and messenger ribonucleic acid (mRNA) platform technology. All of Alps’ current product candidates are currently in either the proof-of-concept or preclinical development stage.
As of the date of this proxy statement/prospectus, Alps does not have any product candidates that are approved for commercial sale and has not generated any revenue from the sale of product candidates. Currently, Alps’ revenue is primarily derived from aesthetic treatments, cosmetic procedures, general healthcare and wellness services.
MyGenome Sdn. Bhd., Alps’ subsidiary, operates a molecular laboratory and is accredited by the Malaysian Standard MS ISO 15189:2022, Medical laboratories – Requirements for quality and competence. In addition, Alps operates a cGMP cell and gene therapy research and cultivation laboratory under one of its wholly owned subsidiaries, Celestialab Sdn. Bhd., approved by the Ministry of Health in Malaysia. Alps’ multidisciplinary team oversees all critical cGMP activities, including manufacturing, quality control, quality assurance, and documentation.
Alps’ principal executive office is located at Unit E-18-01 & E-18-02 Level 18, ICON Tower (East), No.1, Jalan 1/68F, Jalan Tun Razak, 50400 Kuala Lumpur, Malaysia. Alps’ telephone number is +603 2163 1113. Investors should submit any inquiries to the address and telephone number of Alps’ principal executive office. Alps’ website address is https://alps-holdings.com/. Information contained on, or that can be accessed through, Alps’ website is not a part of, and shall not be incorporated by reference into, this proxy statement/prospectus.
For additional information about Alps, see the section entitled “Information about Alps”.
PubCo
PubCo is an exempted company incorporated in the Cayman Islands with limited liability on May 14, 2024 for the purpose of effecting the Business Combination and to serve as the publicly traded parent company of Alps following the Business Combination.
Merger Sub
Merger Sub is an exempted company incorporated in the Cayman Islands with limited liability on May 14, 2024, and is a wholly-owned subsidiary of PubCo incorporated for the purpose of effecting the Business Combination and to serve as the vehicle for, and be subsumed by, Alps pursuant to the Acquisition Merger.
| 27 |
Parent Representative
Globalink’s Sponsor, GL Sponsor LLC, is acting as the Parent Representative for the purpose of representing, from and after the Effective Time, the stockholders of Globalink (other than the Alps Holdco Shareholders as of immediately prior to the Effective Time and their successors and assigns). As the Parent Representative, the Sponsor negotiated the Merger Agreement and the ancillary agreements with Alps Holdco and the Seller Representative, for purposes of efficiency. To the extent that any disputes arise in connection with the Business Combination and/or Merger Agreement after the Closing, the Sponsor, as the Parent Representative, expects to act as the representative of the previous stockholders of Globalink in negotiating with PubCo and resolving such disputes. However, the Sponsor does not have any specific fiduciary or contractual duties to the previous stockholders of Globalink in its capacity as the Parent Representative and may have interests in addition to, or conflict with, those of the previous stockholders of Globalink. See “Summary of the Proxy Statement/Prospectus—Interests of Certain Persons in the Business Combination.”
Additionally, under the Merger Agreement, Globalink, on behalf of itself and its subsidiaries, successors and assigns, has appointed the Sponsor in the capacity as the Parent Representative, as each such person’s agent, attorney-in-fact and representative, with full power of substitution to act in the name, place and stead of such person, to act on behalf of such person from and after the Closing in connection with: (i) bringing, managing, controlling, defending and settling on behalf of Globalink any indemnification claims by any of them under the termination provision of the Merger Agreement; (ii) acting on behalf of such person under the Escrow Agreement; (iii) terminating, amending or waiving on behalf of such person any provision of the Merger Agreement or any ancillary agreement to which the Parent Representative is a party or otherwise has rights in such capacity (together with this Agreement, the “Parent Representative Documents”); (iv) signing on behalf of such person any releases or other documents with respect to any dispute or remedy arising under any Parent Representative Documents; (vi) employing and obtaining the advice of legal counsel, accountants and other professional advisors as the Parent Representative, in its reasonable discretion, deems necessary or advisable in the performance of its duties as the Parent Representative and to rely on their advice and counsel; (vi) incurring and paying reasonable out-of-pocket costs and expenses, including fees of brokers, attorneys and accountants incurred pursuant to the transactions contemplated hereby, and any other out-of-pocket fees and expenses allocable or in any way relating to such transaction or any indemnification claim; and (vii) otherwise enforcing the rights and obligations of any such Persons under any Parent Representative Documents, including giving and receiving all notices and communications hereunder or thereunder on behalf of such person; provided, that the parties acknowledge that the Parent Representative is specifically authorized and directed to act on behalf of, and for the benefit of, the holders of Globalink’s securities (other than the Alps Holdco Shareholders immediately prior to the Effective Time and their respective successors and assigns). All decisions and actions by the Parent Representative, including any agreement between the Parent Representative and Alps Holdco, Seller Representative, any Alps Holdco Shareholders or Indemnifying Party (as defined in the Merger Agreement) relating to the defense or settlement of any claims for which an Indemnifying Party may be required to indemnify Globalink pursuant to the indemnification provisions of the Merger Agreement, shall be binding upon Globalink and its subsidiaries, successors and assigns, and neither they nor any other party shall have the right to object, dissent, protest or otherwise contest the same.
Seller Representative
Dr. Tham Seng Kong is acting as the Seller Representative for the purpose of representing, from and after the Effective Time, the Alps Holdco Shareholders as of immediately prior to the Effective Time and their successors and assigns. As the Seller Representative, Dr. Tham Seng Kong, negotiated the Merger Agreement and the ancillary agreements with Globalink and the Sponsor. After the Business Combination, the Seller Representative shall be entitled to engage in the following on behalf of the Alps Holdco Shareholders as of immediately prior to the Effective Time and their successors and assigns:
| ● | making on behalf of such person and taking all actions on their behalf relating to the achievement of the requirements for the Earn-out Shares and any disputes with respect thereto; | |
| ● | managing, controlling, defending and settling on behalf of an Indemnifying Party any indemnification claims against any of them under the Merger Agreement, including controlling, defending, managing, settling and participating in any third party claim thereunder; | |
| ● | acting on behalf of such person under the Escrow Agreement; | |
| ● | terminating, amending or waiving on behalf of such person any provision of any Seller Representative Document (as defined in the Merger Agreement) (provided, that any such action, if material to the rights and obligations of the Alps Holdco Shareholders in the reasonable judgment of the Seller Representative, will be taken in the same manner with respect to all Alps Holdco Shareholders unless otherwise agreed by each Alps Holdco Shareholder who is subject to any disparate treatment of a potentially material and adverse nature); | |
| ● | signing on behalf of such person any releases or other documents with respect to any dispute or remedy arising under any Seller Representative Document; | |
| ● | employing and obtaining the advice of legal counsel, accountants and other professional advisors as the Seller Representative, in its reasonable discretion, deems necessary or advisable in the performance of its duties as the Seller Representative and to rely on their advice and counsel; | |
| ● | incurring and paying reasonable costs and expenses, including fees of brokers, attorneys and accountants incurred pursuant to the transactions contemplated hereby, and any other reasonable fees and expenses allocable or in any way relating to such transaction or any indemnification claim, whether incurred prior or subsequent to Closing; | |
| ● | receiving all or any portion of the consideration provided to the Alps Holdco Shareholders under the Merger Agreement and to distribute the same to the Alps Holdco Shareholders in accordance with their pro rata share; and |
otherwise enforcing the rights and obligations of any such persons under any Seller Representative Document, including giving and receiving all notices and communications hereunder or thereunder on behalf of such person.
The Merger Agreement and the Business Combination
On January 30, 2024, Globalink entered into the Merger Agreement (as amended and restated on May 20, 2024 and as may be further amended, restated or supplemented from time to time), with Alps Holdco, PubCo, Merger Sub, the Sponsor as the Parent Representative, and Dr. Tham Seng Kong, an individual, as the Seller Representative. Pursuant to the terms of the Merger Agreement, the Business Combination between Globalink and Alps Holdco will be effected in two steps: (i) subject to the approval and adoption of the Merger Agreement by the stockholders of Globalink, Globalink will be merged with and into PubCo, with PubCo remaining as the surviving publicly traded entity; and (ii) Merger Sub will merge with and into Alps Holdco, resulting in Alps Holdco remaining as the surviving entity and being a wholly-owned subsidiary of PubCo.
Subject to the terms and conditions set forth therein upon the Closing, the Alps Holdco Ordinary Share issued and outstanding immediately prior to the Effective Time will be converted into the right to receive PubCo ordinary shares equal to the Conversion Ratio (as defined in the Merger Agreement). The total consideration in the form of PubCo ordinary shares to be paid by Globalink to the Alps Holdco Shareholders at the Closing will be equal to US$1.6 billion, with an earnout provision permitting Alps Holdco Shareholders to receive up to 48 million additional shares of the Combined Company as and when the business meets certain incremental milestones for the consolidated revenue of the Combined Company for five fiscal years following the consummation of the Business Combination. The Merger Agreement is subject to certain customary closing conditions and contains customary representations, warranties, covenants and indemnity provisions. Capitalized terms used but not defined herein shall have the respective meanings set forth in the Merger Agreement. The respective boards of directors of Globalink and Alps Holdco have (i) approved and declared advisable the Merger Agreement and the transactions contemplated thereby and (ii) resolved to recommend approval of the Merger Agreement and related transactions by their respective stockholders.
| 28 |
Conditions to Closing
The consummation of the Business Combination is conditioned upon, among other things, (i) the absence of any applicable law or order restraining, prohibiting or imposing any condition on the consummation of the transactions contemplated by the Merger Agreement, (ii) the expiration or termination of the waiting period under the Hart-Scott-Rodino Antitrust Improvements Act of 1976, as amended (the “HSR Act”) (if applicable), (iii) receipt of any consent, approval or authorization required by any Authority (as such term is defined in the Merger Agreement), (iv) there being no action brought by any Authority to enjoin or otherwise restrict the consummation of the Closing, (v) Globalink having at least $5,000,001 of net tangible assets either immediately prior to (the immediately prior to the Business Combination condition is anticipated to be waived), and PubCo shall have net tangible assets of at least $5,000,001 upon consummation of the Business Combination, (vi) approval by the Alps Holdco Shareholders of the Business Combination and related transactions, (vii) approval by the stockholders of Globalink of the Business Combination and related transactions, (viii) the conditional approval for listing by Nasdaq of the PubCo ordinary shares and PubCo warrants and satisfaction of initial and continued listing requirements, (ix) the Form F-4 becoming effective in accordance with the provisions of the Securities Act, (x) solely with respect to Globalink, among other conditions, (A) Alps Holdco, PubCo and Merger Sub having duly performed or complied with all of their respective obligations under the Merger Agreement in all material respects, (B) the representations and warranties of Alps Holdco, PubCo and Merger Sub being true and correct in all respects unless failure would not have or reasonably be expected to have a Material Adverse Effect on Alps Holdco and its subsidiaries as a whole, (C) no event having occurred that has resulted in or would result in a Material Adverse Effect on Alps Holdco and its subsidiaries as a whole, (D) Globalink has obtained an opinion from an independent investment bank or other financial advisory firm mutually acceptable to Globalink and Alps Holdco as to the fairness from a financial point of view, as of the date of such opinion, of the Merger Consideration Shares to be paid to the Alps Holdco Shareholders pursuant to the Merger Agreement, and (E) the size and composition of the post-Closing PubCo Board being established as set forth in the Merger Agreement, and (xi) solely with respect to each of PubCo, Merger Sub, and Alps Holdco and its subsidiaries, among other conditions, (A) Globalink having duly performed or complied with all of its obligations under the Merger Agreement in all material respects, (B) the representations and warranties of Globalink being true and correct in all respects, (C) no event having occurred that has resulted in or would result in a Material Adverse Effect on Globalink which is continuing and uncured, and (D) Alps Holdco having received a copy of the Escrow Agreement duly executed by PubCo, Globalink and the Escrow Agent.
The Globalink Board intends to waive the fairness opinion as a closing condition of the Business Combination.
Termination
The Merger Agreement may be terminated at any time prior to the Effective Time as follows (i) by either Globalink or Alps Holdco if the transactions contemplated by the Merger Agreement are not consummated on or before the six (6)-month anniversary of May 20, 2024, the date of the Merger Agreement (the “Outside Closing Date”), provided that, if the SEC has not declared the Form F-4 effective on or prior to the five (5)-month anniversary of the Merger Agreement, then the Outside Closing Date will be extended by one (1) additional month, provided further that, the failure to consummate the transactions contemplated by the Merger Agreement by the Outside Closing Date is not due to a material breach by the party seeking to terminate the Agreement;
(ii) by either Globalink or Alps Holdco if any Authority has issued any final decree, order, judgment, award, injunction, rule or consent or enacted any law, having the effect of permanently enjoining or prohibiting the consummation of the Business Combination, provided that, the party seeking to terminate cannot have breached its obligations under the Merger Agreement and such breach was a substantial cause of, or substantially resulted in, such action by the Authority.
(iii) by mutual written consent of Globalink and Alps Holdco duly authorized by each of their respective board of directors;
(iv) by Globalink in the event that Alps Holdco does not deliver to Globalink the Final March 31, 2024 Financial Statements on or prior to May 31, 2024 or such further period as mutually agreed upon between Globalink and Alps Holdco in writing; and
(v) by either Globalink or Alps Holdco if the other party has breached any of its covenants or representations and warranties such that closing conditions would not be satisfied by the earlier of (A) the Closing Date and (B) 30 days following receipt by the breaching party of a written notice of the breach.
The Merger Agreement and other agreements described below have been included to provide investors with information regarding their respective terms. They are not intended to provide any other factual information about Globalink, Alps Holdco or the other parties thereto. In particular, the assertions embodied in the representations and warranties in the Merger Agreement were made as of a specified date, are modified or qualified by information in the disclosure schedules to the Merger Agreement, may be subject to a contractual standard of materiality different from what might be viewed as material to investors, or may have been used for the purpose of allocating risk between the parties. Accordingly, the representations and warranties in the Merger Agreement are not necessarily characterizations of the actual state of facts about Globalink, Alps Holdco or the other parties thereto at the time they were made or otherwise and should only be read in conjunction with the other information that Globalink makes publicly available in reports, statements and other documents filed with the SEC. Globalink or Alps Holdco investors and securityholders are not third-party beneficiaries under the Merger Agreement.
| 29 |
The Redomestication Merger
Immediately prior to the Acquisition Merger, Globalink will merge with and into PubCo, a Cayman Islands exempted company. The separate corporate existence of Globalink will cease and PubCo will continue as the surviving company in the Redomestication Merger. In connection with the Redomestication Merger, all outstanding Globalink units will separate into their individual components of shares of Globalink common stock, Globalink rights and Globalink warrants and will cease separate existence and trading. Upon the consummation of the Business Combination, the current equity holdings of the Globalink stockholders shall be exchanged as follows:
| (i) | Each share of Globalink common stock, par value $0.001 per share, issued and outstanding immediately prior to the effective time of the Redomestication Merger (other than any redeemed shares), will automatically be cancelled and cease to exist and for each share of Globalink common stock, PubCo shall issue to its holder (other than Globalink stockholders who exercise their redemption rights in connection with the Business Combination) one validly issued PubCo ordinary share, which shall be fully paid; | |
| (ii) | Each Globalink warrant to purchase one-half (1/2) of one share of Globalink common stock issued and outstanding immediately prior to effective time of the Redomestication Merger will convert into one PubCo warrant (or equivalent portion thereof). The PubCo warrants will have substantially the same terms and conditions as set forth in the Globalink warrants; and | |
| (iii) | The holders of Globalink rights (convertible into one-tenth (1/10) of one share of Globalink common stock) issued and outstanding immediately prior to the effective time of the Redomestication Merger will obtain the PubCo right in exchange for the cancellation of each Globalink right; provided, however, that no fractional shares will be issued and all fractional shares will be rounded to the nearest whole share. |
Globalink and Alps Holdco intends to enter into a supplemental warrant agreement with Continental (the “Supplemental Warrant Agreement”), pursuant to which, among other things, effective as of the Effective Time, Alps Holdco will assume the obligations of Globalink under that certain warrant agreement, dated December 6, 2021, by and between Globalink and Continental (the “Existing Warrant Agreement”). Pursuant to the Business Combination Agreement and the Supplemental Warrant Agreement, each issued and outstanding warrant of Globalink sold to the public and to PGM, an affiliate of the Sponsor, in a private placement in connection with Globalink’s initial public offering will be exchanged for a corresponding warrant exercisable for PubCo ordinary shares.
The Acquisition Merger
Immediately after and conditional upon the Redomestication Merger, Merger Sub, a Cayman Islands exempted company and wholly owned subsidiary of PubCo, will merge with and into Alps Holdco, with Alps Holdco being the surviving company in the merger, resulting in Alps Holdco being a wholly owned subsidiary of PubCo.
For more information about the Business Combination, please see the sections titled “The Redomestication Merger Proposal (Proposal 1)” and “The Acquisition Merger Proposal (Proposal 2).” A copy of the Merger Agreement is attached to this proxy statement/prospectus as Annex A.
Other Agreements Relating to the Business Combination
Globalink Support Agreements
In connection with the execution of the Merger Agreement, Globalink, Alps Holdco, and certain stockholders of Globalink have entered into a Globalink Support Agreement, pursuant to which the stockholders of Globalink that are parties to the Globalink Support Agreements have agreed to vote all shares of Globalink common stock beneficially owned by them in favor of the transactions contemplated by the Merger Agreement.
Alps Holdco Support Agreements
In connection with the execution of the Merger Agreement, Globalink, Alps Holdco and each of certain shareholders of Alps Holdco have entered into an Alps Holdco Support Agreement, pursuant to which the shareholders of Alps Holdco that are parties to the Alps Holdco Support Agreements have agreed to vote all ordinary shares of Alps Holdco beneficially owned by them in favor of the transactions contemplated by the Merger Agreement.
| 30 |
Lock-up Agreements
The Merger Agreement provides that, prior to the Closing, and effective as of the Closing, certain Alps Holdco Shareholders, the Parent Representative and certain other stockholders of Globalink will enter into the Lock-up Agreements, subject to certain customary exceptions, not to offer, sell, contract to sell, pledge or otherwise dispose of, directly or indirectly, any of the PubCo ordinary shares subject to lock-up, including the Merger Consideration Shares (the “Lock-up Shares”), enter into a transaction that would have the same effect, or enter into any swap, hedge or other arrangement that transfers, in whole or in part, any of the economic consequences of ownership of the Lock-Up Shares or otherwise, publicly disclose the intention to make any offer, sale, pledge or disposition, or to enter into any transaction, swap, hedge or other arrangement, or engage in any Short Sales (as defined in the Lock-up Agreement) with respect to the Lock-Up Shares until (i) (w) with respect to one hundred percent (100%) of the Lock-Up Shares, the date from the Closing until the last date that is six (6) months after the date of the Closing, (x) with respect to ninety percent (90%) of the Lock-Up Shares, the 1st day of the 7th month from the Closing until the date that is nine (9) months after the date of the Closing, (y) with respect to seventy percent (70%) of the Lock-Up Shares, the 1st day of the 10th month from the Closing until the date that is twelve (12) months after the date of the Closing, (z) with respect to forty percent (40%) of the Lock-Up Shares, the 1st day of the 13th month from the Closing until the date that is fifteen (15) months after the date of the Closing, or (ii) if sooner, the date after the Closing on which Globalink consummates a liquidation, merger, share exchange or other similar transaction with an unaffiliated third party that results in all of Globalink’s stockholders having the right to exchange their equity holdings in Globalink for cash, securities or other property. For the avoidance of doubt, no Lock-up Shares shall be subject to the terms in the Lock-up Agreement from and after the date that is fifteen (15) months after the date of the Closing.
Amended and Restated Registration Rights Agreement
At the Closing, Globalink will enter into the Amended and Restated Registration Rights Agreement with certain existing stockholders of Globalink with respect to the PubCo ordinary shares they own at the Closing, and with certain Alps Holdco Shareholders who will be holders of PubCo ordinary shares after the Closing. The Amended and Restated Registration Rights Agreement will require PubCo to, among other things, file a resale shelf registration statement on behalf of the stockholders no later than 60 days after the Closing. The Amended and Restated Registration Rights Agreement will also provide certain demand registration rights and piggyback registration rights to the stockholders, subject to underwriter cutbacks and issuer blackout periods. Globalink will agree to pay certain fees and expenses relating to registrations under the Amended and Restated Registration Rights Agreement.
Escrow Agreement
In connection with the Business Combination, PubCo, Globalink, and the Escrow Agent will enter into the Escrow Agreement, effective as of the Effective Time, in form and substance reasonably satisfactory to PubCo and Alps Holdco, pursuant to which PubCo shall issue to the Escrow Agent a number of PubCo ordinary shares (with each share valued at $10.00) equal to five percent (5%) of One Point Six Billion Dollars ($1,600,000,000), together with any equity securities paid as dividends or distributions with respect to the Escrow Shares and along with any other dividends, distributions or other income on the Escrow Shares in the Escrow Account.
The PIPE Investment
In connection with the execution of the Merger Agreement, PubCo, Globalink and Alps Holdco entered into the Subscription Agreements with certain accredited investors on June 4, 2024, June 5, 2024, and August 27, 2024, pursuant to which the investors have agreed to purchase, and PubCo has agreed to sell to the investors, an aggregate of $40,200,000 worth of PubCo ordinary shares, or 4,020,000 PubCo ordinary shares (the “PIPE Securities”). The purpose of the sale of the PIPE Securities is to raise additional capital for the business operations of the Combined Company. The obligations to consummate the transactions contemplated by the Subscription Agreements are conditioned upon, among other things, customary closing conditions and the consummation of the transactions contemplated by the Merger Agreement. The PIPE Investors have not received any payments from the Sponsor in connection with the PIPE Investment. For an illustration of the dilutive effect the Business Combination, the PIPE Investment, and related transactions would have on the holders of public shares who hold the securities until the consummation of the Business Combination, see “Summary of the Proxy Statement/Prospectus — Ownership of the Combined Company After the Closing.”
| 31 |
Structure of the Business Combination
The diagrams below depict simplified versions of the current organizational structures of Globalink and Alps, respectively.
Simplified Pre-Combination Structure
Globalink (Current Structure)
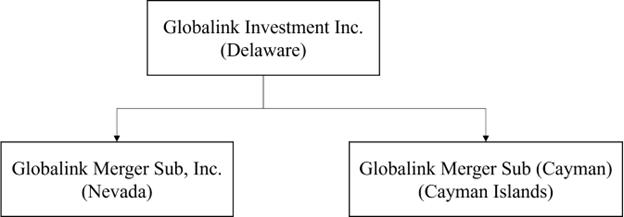
Alps (Current Structure)
Pre-Reorganization

Post-Reorganization

Simplified Post-Combination Structure
Immediately prior to the Acquisition Merger, Globalink will merge with and into PubCo, with PubCo to be the surviving company in the Redomestication Merger, and immediately after and conditional upon the Redomestication Merger, Merger Sub will merge with and into Alps Holdco, with Alps Holdco surviving the Acquisition Merger as a wholly-owned subsidiary of PubCo.
| 32 |

Executive Officers of the Combined Company
The executive officers of Alps Holdco immediately prior to the Closing will be the executive officers of the Combined Company upon the Closing. Following the Business Combination, the Combined Company also expects to appoint a Chief Financial Officer. Alps is in the process of evaluating and seeking suitable candidates to serve as the Chief Financial Officer of the Combined Company.
Board of Directors of the Combined Company
The Combined Company Board will consist of five directors, with Dr. Tham Seng Kong serving as the Chairman of the Combined Company Board.
Globalink Board’s Reasons for the Approval of the Business Combination
In reaching its decision with respect to the Business Combination, the Board considered views of Globalink’s management regarding the opportunity represented by the proposed transaction and evaluated the investor presentation provided by Alps. Globalink’s management discussed its diligence to the Board, which included:
| ● | review of Alps’ financial results; | |
| ● | review of Alps’ financial projections model; | |
| ● | review of Alps’ descriptions of its business operations, including its patents, premises and equipment; | |
| ● | review of financial data of public companies that are comparable to Alps; | |
| ● | review of Alps’ capitalization table; | |
| ● | review of Alps’ patents and pipelines, corporate records, material agreements, employment agreements, tax, litigation and other business-relevant materials; | |
| ● | discussions with Alps’ management; | |
| ● | third-party background checks on Alps’ management and directors, and each of the director nominees to the Combined Company Board nominated by Alps; | |
| ● | third-party verification of Alps’ inventory; and | |
| ● | discussions with specialists who are experienced professionals in the healthcare industry in Globalink’s directors’ and officers’ contacts. |
| 33 |
The Board supported the decision to enter into the Merger Agreement with its evaluation of the above due diligence and the following qualitative and quantitative evaluations regarding Alps:
Availability and terms of PIPE Investment
The Board also considered the availability and terms of the PIPE Investment prior to approving the Business Combination. Noting that PIPE Investment in the amount of approximately US$40.2 million from local investors from Malaysia would become available in connection with the Business Combination, the Board negotiated the terms of the PIPE Investment with Alps, and mutually agreed that the PIPE Investment would be used for PubCo’s working capital after the Business Combination. The Board determined that the terms of the PIPE Investment are fair and reasonable to Globalink. The determination was made unanimously in May 2024.
Prior to making such determination, the Globalink Board engaged in discussions and consultations with its management, financial and legal advisors of both Globalink and Alps Holdco, and reviewed the terms of the PIPE Investment. The Globalink Board considered matters such as (i) whether the terms of the PIPE subscription agreements are similar to the typical PIPE transactions entered into by other special purpose acquisition companies in connection with a De-SPAC transaction, and whether there are any usual terms not commonly observed, (ii) the potential investors’ profile, including each investor’s net worth and reputation, (iii) purpose of such investors’ the investment and utilization, including whether they intend to hold PubCo’s shares for long-term investment, and (iv) the amount of the financing and the timing such financing proceeds are expected to be available.
Industry trends
| ● | Advances in biotechnology: Rapid advancements in biotechnology, such as gene editing technologies like clustered regularly interspaced palindromic repeats (CRISPR-Cas9), have unlocked new possibilities in drug discovery, disease treatment, and personalized medicine. These biotechnological breakthroughs offer tremendous potential for innovation and growth within the industry. | |
| ● | Growing demand for personalized medicine: Personalized medicine, which tailors treatment plans based on an individual’s genetic makeup, is gaining popularity. This trend provides opportunities for biotech companies to develop targeted therapies, companion diagnostics, and genetic testing services to meet the demand for more precise and effective healthcare solutions. | |
| ● | Focus on rare diseases and orphan drugs: The biotech industry has shifted focus towards rare diseases, also known as orphan diseases, which affect a small percentage of the population. Governments and regulatory bodies have implemented incentives and streamlined approval processes to encourage the development of orphan drugs, making it an attractive area for biotech companies to invest in. | |
| ● | Increased investment in biotech startups: Venture capital firms, pharmaceutical companies, and government entities are increasingly investing in biotech startups. This influx of funding has created a supportive environment for innovative ideas and technologies to be developed, leading to a vibrant ecosystem of entrepreneurial ventures within the industry. | |
| ● | Convergence of biology and digital technology: The convergence of biology with digital technology, such as artificial intelligence (AI), big data analytics, and machine learning, is transforming the biotech and life sciences landscape. These digital technologies enable faster and more accurate drug discovery, clinical trials, and patient care, improving overall efficiency and outcomes. | |
| ● | Rising global healthcare expenditure: With the global population aging and the increasing prevalence of chronic diseases, healthcare expenditure is on the rise. Biotech and life sciences companies are well-positioned to benefit from this trend by developing innovative therapies and solutions that address the evolving healthcare needs of the population. |
These positive trends create an optimistic outlook for the biotech and life sciences industry, presenting numerous opportunities for growth, innovation, and impact.
For sources of the above statements and conclusions on the future trends in the biotechnology industry, please refer to the notes below.
Notes:
| 1. | Explore Emerging Biotech Trends in 2024 for Life Science Professionals. https://www.mrlcg.com/resources/blog/emerging-biotech-trends/ | |
| 2. | The Future of Biotech: Innovations and Trends in Science and Research. https://www.healthtechzone.com/topics/healthcare/articles/2024/06/21/459929-future-biotech-innovations-trends-science-research.htm. | |
| 3. | The Global Biotechnology Industry Outlook – 2024. https://www.marketsandmarkets.com/blog/HC/The-Global-Biotechnology-Industry-Outlook-2024 | |
| 4. | The Future of Biotech. https://www.dni.gov/index.php/gt2040-home/gt2040-deeper-looks/future-of-biotech | |
| 5. | Biotechnology Market Size, Share, and Trends 2024 to 2033. https://www.precedenceresearch.com/biotechnology-market | |
| 6. | Biotechnology Market Size & Trends. https://www.grandviewresearch.com/industry-analysis/biotechnology-market | |
| 7. | Biotechnology Industry Statistics: Market Data Report 2024. https://worldmetrics.org/biotechnology-industry-statistics/ |
Management team
The management team of Alps is composed of seasoned professionals with extensive experience and a track record of achievements in the field.
At the helm is Dr. Tham Seng Kong, a visionary and accomplished leader with a Bachelor of Medicine from Xiamen University, Doctor of Medicine (MD) Integrated Chinese & Western Clinical Medicine (Oncology) from Guangzhou University of Chinese Medicine (GUCM) and a Ph.D. in Clinical Discipline of Chinese and Western Medicine from GUCM. He also obtained his Masters of Business Administration from the Business Institute of Pennsylvania, United States. With his deep scientific expertise and keen business acumen, he has successfully spearheaded multiple R&D projects and breakthrough therapies to market. Driven by an unwavering passion for improving patient lives, Dr. Tham possesses the ability to inspire and motivate the entire team at Alps.
Supporting Dr. Tham is Lisa Teoh @ Teoh Lee Eng, a seasoned executive with more than 20 years of experience in finance and operations. As the chief operating officer of Alps, she brings a strong operational acumen to Alps. Ms. Teoh is a registered civil engineer with professional institutions in Malaysia and is an ASEAN Chartered Professional Engineer. She earned her Master’s Degree in Engineering Science (Civil) (M.Eng) in 1996 as well as a Master of Philosophy from the University of Oxford, United Kingdom in 1997. Ms. Teoh later obtained her Master of Business Entrepreneur Management in 2018 and Doctor of Business Administration in 2020 from the Institute of Entrepreneurship Malaysia.
Leading the research and development efforts is Professor Manickam Ravichandran, a renowned expert in molecular biology and vaccine discovery. He obtained his M.Sc.-Medical Microbiology from Christian Medical College, Vellore and was awarded his Ph.D. in Biotechnology from Anna University in Chennai, India. He has a track record of developing innovative therapeutic candidates and leading clinical trials. He was previously appointed as the former Vice-Chancellor of AIMST University, Malaysia. Professor Manickam Ravichandran’s strategic thinking and scientific expertise are instrumental in guiding Alps’ research and development pipeline.
Leading another pipeline in vaccine development is Alps’ Chief Vaccine Development Officer, Professor Chit Laa Poh. Professor Poh obtained her Ph.D. in Medical Microbiology and Bacteriology from Monash University (Australia) and completed postdoctoral training at Pasteur Institute, Cambridge University, and University College London. Professor Poh served as the Inaugural Dean of the Faculty of Science and Technology from 2012 to 2014 and currently holds the position of Distinguished Professor and Head of the Centre for Virus and Vaccine Research (CVVR) at Sunway University, Malaysia.
| 34 |
The commercialization strategy and marketing efforts are overseen by Ms. Chew Yoke Ling, a seasoned marketing professional with a deep understanding of the biotech market landscape. With her market research expertise and customer-centric approach, she ensures the effective positioning and successful commercial launch of Alps’ products.
Competition
Alps provides commercialized services and products in Malaysia. Alps has a group of skilled, multilingual employee able to contributes to the company’s success. Of all employees of Alps, 66% or more has a bachelor’s degree of above, and is comprised of individuals with diverse backgrounds and ethnicities, including Malay, Chinese, Indian and other ethnic backgrounds. This allows Alps to develop and communicate with customers from different backgrounds. Alps’ labor force also contributes to a low burn rate relative to many typical biotech companies. In the fiscal years ended March 31, 2023 and 2024, Alps incurred labor costs of US$1,136,059 and US$1,186,647, respectively, representing 31.6% and 49.37% of Alps’ total revenue in the respective period. Alps’ skilled, multilingual and cost-effective labor force gains the company a competitive advantage in its pipeline developments.
For the fiscal years ended March 31, 2024 and 2023, the burn rates of Alps approximately US$1,490,000 and US$224,000, respectively. Burn rates are largely driven by research and development expenses. For instance, Moderna spent US$4.85 billion on research and development in 2023, Biogen incurred US$2.46 billion in research and development expenses in 2023, and Novartis incurred US$11.37 billion in research and development expenses. Burn rates for biotechnology companies can vary widely from tens of millions to over $100 million per quarter, depending on their stage of development and level of commercial operations. For clarity, the companies referenced above are significantly larger than Alps, with significantly larger research and development plans and costs.
For sources of the above statements relating to Alps’ low burn rate relative to typical biotech companies, please refer to notes below:
Notes:
| 1. | The top pharmaceutical companies by R&D expenditure. https://www.pharmaceutical-technology.com/features/the-top-pharmaceutical-companies-by-rd-expenditure/ |
Significant growth potential
Southeast Asia is a dynamic region that offers numerous advantages for businesses seeking growth and expansion. With a rapidly growing economy, a large consumer base, and a strategic location, Southeast Asia has become an attractive destination for both established corporations and emerging startups.
The region has experienced robust economic growth in recent years, fueled by factors such as urbanization, rising middle-class population, and increased foreign investment. This growth has created a thriving market with a vast potential for businesses to tap into. Southeast Asia boasts a large and diverse consumer base. With a population of over 650 million people, the region offers businesses access to a vast market of potential customers. The middle class is expanding rapidly, leading to increased purchasing power and demand for products and services across various sectors. Additionally, with an increasingly aging population, Alps expects to observe increasing demand for its products and services.
Southeast Asia is also strategically located, serving as a gateway to the larger Asian market. Its proximity to China, India, and other emerging economies provides businesses with opportunities for regional integration and access to global supply chains.
Another advantage of Southeast Asia is its investment-friendly policies and business environment. Many countries in the region have implemented reforms to attract foreign investment, such as tax incentives, streamlined regulations, and improved infrastructure. This business-friendly ecosystem facilitates entry and expansion for companies seeking to establish a presence or invest in Southeast Asia. The region offers a large pool of young, educated, and motivated workers who are capable of driving innovation and productivity. With competitive labor costs compared to other regions, businesses can achieve greater cost efficiency and maintain competitiveness.
Additionally, Southeast Asia is a hub for innovation and entrepreneurship. The region has witnessed a burgeoning startup ecosystem, supported by incubators, accelerators, and venture capital investment. This vibrant startup culture fosters innovation, encourages collaboration, and provides opportunities for partnerships and synergies with local businesses.
For sources of the above statements and conclusions on the growth potential of Southeast Asia, please refer to the notes below.
Notes:
| 1. | The rise of the middle class and economic growth in ASEAN. https://documents.worldbank.org/en/publication/documents-reports/documentdetail/426681495130074444/the-rise-of-the-middle-class-and-economic-growth-in-asean | |
| 2. | A 3D view of Southeast Asia: demographics, digitisation and dynamism. https://www.business.hsbc.com/en-gb/insights/growing-my-business/a-3d-view-of-southeast-asia#:~:text=Southeast%20Asia%20is%20also%20home,5%25%20annually%20through%20to%202030.&text=This%20increase%20in%20purchasing%20power,foreign%20direct%20investment%20(FDI) | |
| 3. | Total population of the ASEAN countries from 2019 to 2029. https://www.statista.com/statistics/796222/total-population-of-the-asean-countries/ | |
| 4. | International Monetary Fund – Population. https://www.imf.org/external/datamapper/LP@WEO/VNM/IDN/PHL/MMR/MYS/KHM/LAO/THA/SGP/BRN/AUS | |
| 5. | Aging Southeast Asia grapples with weak social safety nets. https://asia.nikkei.com/Spotlight/Datawatch/Aging-Southeast-Asia-grapples-with-weak-social-safety-nets |
Fairness Opinion
The Globalink Board does not plan to obtain a final fairness opinion in connection with the Business Combination. The Globalink Board considered obtaining a fairness opinion as to the Merger Consideration paid to Alps Holdco Shareholders, and has reached out to Baker Tilly, a financial advisory, tax and assurance services firm, and Morison Advisory Sdn. Bhd., a financial advisory services firm based in Malaysia and a member firm of Morison Global, for a fairness opinion. Neither firm declined or were otherwise unable to unwilling to provide a fairness opinion to the Globalink Board. On February 2, 2024, Globalink engaged Morison Advisory Sdn. Bhd. to provide a fairness opinion. The Globalink Board received a draft of fairness opinion from Morison Advisory Sdn. Bhd. in June 2024. For a description of the draft fairness opinion that the Globalink Board received, see “The Acquisition Merger Proposal (Proposal 2) — Fairness Opinion.” After a thorough review and internal discussions, the Globalink Board decided not to rely on the fairness opinion for determining the valuation of Alps Holdco, as the Globalink Board believed that the valuation methods adopted by Morison Advisory Sdn. Bhd., the income approach using Discounted Cash Flow (“DCF”) method, was reliant upon underlying key assumptions susceptible to material changes and uncertainties. The Globalink Board did not put much weight in considering the draft fairness opinion in determining the valuation of Alps Holdco. Instead, the Globalink Board primarily considered (i) the verbal commitment of Seller Representative and Alps Holdco during the negotiation stage of securing PIPE Investment of an aggregate amount of US$40.2 million or more, (ii) the financing completed by Alps in September 2023, of US$2.5 million, and (iii) the other factors disclosed under “—Globalink Board’s Reasons for the Approval of the Business Combination.” The Globalink Board intends to waive the fairness opinion as a closing condition of the Business Combination.
Compensation of the Sponsor, its Affiliates and Promoters
The Sponsor holds an aggregate of 2,835,000 shares of Globalink common stock as of the date of this proxy statement/prospectus. The Sponsor will receive a positive rate of return so long as the market price of the common stock is at least $0.01 per share, even if public stockholders experience a negative rate of return in the Combined Company. The Sponsor, its affiliates and promoters are not receiving compensation in connection with the Business Combination and the PIPE Investment. The Sponsor, its affiliates and promoters will not receive any securities of Globalink or PubCo in connection with the Business Combination and the PIPE Investment. However, the Sponsor is expected to own an equity interest of approximately 1.7% in the Combined Company after the consummation of the Business Combination. For an illustration of the dilutive effect the Business Combination, the PIPE Investment, and related transactions would have on the holders of public shares who hold the securities until the consummation of the Business Combination, see “Summary of the Proxy Statement/Prospectus — Ownership of the Combined Company After the Closing.”
Recommendation of the Board
The Board has unanimously determined that the Business Combination is in the best interests of, and advisable to, the Globalink stockholders and recommends that the Globalink stockholders adopt the Merger Agreement and approve the Business Combination. The Board made its determination after consultation with its legal and financial advisors and consideration of a number of factors. The Globalink Board does not plan to obtain a final fairness opinion related to the Business Combination.
The Board unanimously recommends that you vote “FOR” the approval of the Redomestication Merger Proposal, “FOR” the approval of the Acquisition Merger Proposal, “FOR” the approval of the Nasdaq Proposals, and “FOR” the approval of the Adjournment Proposal.
The existence of financial and personal interests of Globalink’s directors and officers may result in a conflict of interest on the part of such director(s) and/or officer(s) between what he, she or they may believe is in the best interests of Globalink and its stockholders and what he, she or they may believe is best for himself, herself or themselves in determining to recommend that stockholders vote for the Proposals. See the section entitled “The Acquisition Merger Proposal (Proposal 2) — Interests of Certain Persons in the Business Combination” of this proxy statement/prospectus.
As of the date of this proxy statement/prospectus, a total of 3,445,000 shares of common stock, or approximately 92.6% of the outstanding shares, were subject to the Insider Letter and the Globalink Support Agreements. The approval of the Redomestication Merger Proposal and the Acquisition Merger Proposal does not require the votes from at least a majority of unaffiliated security holders of Globalink. As such, as the vote to approve the Redomestication Merger Proposal and the Acquisition Merger Proposal is a majority of the then outstanding shares of common stock present and entitled to vote at the Special Meeting, assuming only the minimum number of shares of common stock to constitute a quorum is present, no public stockholder must vote in favor of the Redomestication Merger Proposal or the Acquisition Merger Proposal for them to be approved.
| 35 |
Proposals to be Submitted at the Special Meeting
The Redomestication Merger Proposal
Globalink and Alps Holdco have agreed to the Business Combination under the terms of the Merger Agreement. Pursuant to the terms set forth in the Merger Agreement, subject to the satisfaction or waiver of the conditions to the Closing therein, Globalink will merge with and into PubCo, with PubCo surviving the Redomestication Merger.
If any proposal is not approved by the Globalink stockholders at the Special Meeting, the Board may submit the Adjournment Proposal for a vote.
For additional information, see “The Redomestication Merger Proposal (Proposal 1)” section of this proxy statement/prospectus.
The Acquisition Merger Proposal
Pursuant to the terms set forth in the Merger Agreement, subject to the satisfaction or waiver of the conditions to the Closing therein and conditioned upon the Redomestication Merger, Alps Holdco will merge with and into Merger Sub, with Alps Holdco surviving the Acquisition Merger.
After consideration of the factors identified and discussed in the section entitled “The Acquisition Merger Proposal (Proposal 2) — Interests of Certain Persons in the Business Combination,” the Board concluded that the Business Combination met all of the requirements disclosed in the prospectus for the Globalink IPO, including that the business of Alps Holdco had a fair market value of at least 80% of the balance of the funds in the Trust Account at the time of execution of the Merger Agreement. The Globalink Board does not plan to obtain a final fairness opinion in connection with the Business Combination.
For additional information, see “The Acquisition Merger Proposal (Proposal 2)” section of this proxy statement/prospectus.
The Nasdaq Proposals
Globalink stockholders are being asked to approve separate proposals, for purposes of complying with the Nasdaq Listing Rule, the issuance of (i) up to 208,000,000 PubCo ordinary shares pursuant to the Merger Agreement (including the ordinary shares issuable pursuant to that certain earnout provisions in the Merger Agreement), and (ii) up to 4,020,000 PubCo ordinary shares to the PIPE Investors. For more information, see the section entitled “The Nasdaq Proposals (Proposals 3)” of this proxy statement/prospectus.
| 36 |
The Adjournment Proposal
Globalink is proposing that its stockholders approve the adjournment of the Special Meeting to a later date or time, if necessary or appropriate.
For additional information, see “The Adjournment Proposal (Proposal 4)” section of this proxy statement/prospectus.
The Special Meeting
Date, Time and Place of the Special Meeting
The Special Meeting will be held virtually at , Eastern time, on , 2025 or at such other date and time to which such meeting may be adjourned or postponed, to consider and vote upon the Proposals.
Registering for the Special Meeting
As a registered Globalink stockholder, you received a proxy card from Continental. The form contains instructions on how to attend the virtual meeting including the URL address, along with your control number. You will need your control number for access. If you do not have your control number, contact Continental at the phone number or e-mail address below. Continental’s support contact information is as follows: 917-262-2373, or email proxy@continentalstock.com.
You can pre-register to attend the virtual meeting starting , 2025 at a.m. Eastern Time. Enter the URL address into your browser https://www.cstproxy.com/ , enter your control number, name and email address. At the start of the meeting you will need to re-log in using your control number and will also be prompted to enter your control number if you vote during the meeting.
A Globalink stockholder that holds such stockholder’s shares in “street name,” which means such stockholder’s shares are held of record by a broker, bank or other nominee, may need to contact Continental to receive a control number. If you plan to vote at the meeting, you will need to have a legal proxy from your bank or broker, or if you would like to join and not vote, Continental will issue you a guest control number with proof of ownership. Either way, you must contact Continental for specific instructions on how to receive the control number. They can be contacted at the number or email address above. Please allow up to 72 hours prior to the meeting for processing your control number.
If you do not have internet capabilities, you can listen only to the meeting by dialing +1 800-450-7155 within the U.S. and Canada (toll-free), or +1 857-999-9155 outside the U.S. and Canada (standard rates apply) when prompted enter the pin number . This is listen-only and you will not be able to vote or enter questions during the meeting.
Purpose of the Special Meeting
At the Special Meeting, Globalink is asking its stockholders to consider and vote upon:
| ● | The Redomestication Merger Proposal. | |
| ● | The Acquisition Merger Proposal. A copy of the Merger Agreement is attached to this proxy statement/prospectus as Annex A. | |
| ● | The Nasdaq Proposals. | |
| ● | The Adjournment Proposal, if presented at the Special Meeting. |
| 37 |
Voting Power and Record Date
You will be entitled to vote or direct votes to be cast at the Special Meeting if you owned shares of Globalink common stock at the close of business on , 2025, which is the Record Date. You are entitled to one vote for each share of Globalink common stock that you owned as of the close of business on the Record Date. If your shares are held in “street name” or are in a margin or similar account, you should contact your broker, bank or other nominee to ensure that votes related to the shares you beneficially own are properly counted. On the Record Date, there were shares of Globalink common stock outstanding, of which are public shares, are Founder Shares, and are shares included in the private units.
Quorum and Required Vote
A quorum of Globalink stockholders is necessary to hold a valid meeting. A quorum will be present at the Special Meeting if a majority of the issued and outstanding shares of Globalink common stock and entitled to vote at the Special Meeting is represented in person by virtual attendance or by proxy at the Special Meeting. Abstentions will count as present for the purposes of establishing a quorum. Broker non-votes will not be counted for purposes of establishing a quorum.
The approval of the Redomestication Merger Proposal, the Acquisition Merger Proposal, each of the Nasdaq Proposals, and the Adjournment Proposal each require the affirmative vote of the holders of a majority of the shares of Globalink common stock cast by the stockholders represented in person by virtual attendance or by proxy and entitled to vote thereon at the Special Meeting. A Globalink stockholder’s failure to vote by proxy or to vote in person online at the Special Meeting will not be counted towards the number of shares of Globalink common stock required to validly establish a quorum, and abstentions will count as present for the purposes of establishing a quorum. If a valid quorum is otherwise established, such failure to vote by proxy or in person or abstention will have no effect on the outcome of the vote on the Redomestication Merger Proposal, the Acquisition Merger Proposal, the Nasdaq Proposals and Adjournment Proposal
Each of the Nasdaq Proposals is conditioned on the approval of the Redomestication Merger Proposal and the Acquisition Merger Proposal. The Redomestication Merger Proposal and the Acquisition Merger Proposal are conditioned upon each other. Unless the each of the Redomestication Merger Proposal and the Acquisition Merger Proposal is approved, the Nasdaq Proposals will not be presented to the stockholders of Globalink at the Special Meeting. The Adjournment Proposal is not conditioned on any other proposal. It is important for you to note that in the event the Redomestication Merger Proposal and the Acquisition Merger Proposal do not receive the requisite vote for approval, Globalink will not consummate the Business Combination. If Globalink does not consummate the Business Combination and fails to complete an initial business combination by January 9, 2025, it will be required to dissolve and liquidate its Trust Account by returning the then-remaining funds in such account to its public stockholders. If Globalink anticipates that it may not be able to consummate its initial business combination in time, Globalink may, by resolutions of its Board, if requested by the Sponsor, extend the period of time to consummate a business combination on a monthly basis to until June 9, 2025, subject to the Sponsor depositing additional funds into the Trust Account.
On December 10, 2024, Globalink received a delisting determination letter from Nasdaq as the 36-month anniversary from its IPO has passed on December 9, 2024, and Globalink are no longer in compliance with Nasdaq Listing Rule IM 5101-2(b). Globalink’s securities were suspended from trading and delisted from Nasdaq on December 17, 2024. As of the same date, Globalink’s securities started trading on OTC Pink. There could be substantial risks to Globalink and PubCo the as a result of Globalink’s non-compliance with this rule. See “Risk Factors — Risks Related to Globalink’s Business and the Business Combination —Our securities were suspended from trading and delisted from Nasdaq on December 17, 2024, following receipt of a delisting determination letter from Nasdaq on December 10, 2024. This could have significant material adverse consequences on us and our securities, including that it will negatively impact our ability to complete a business combination, will limit investors’ ability to make transactions in our securities and could subject us to additional trading restrictions.”
| 38 |
In accordance with the Insider Letter entered into concurrently with the IPO, all shares of Globalink common stock owned by the Sponsor, Globalink’s directors and officers and PGM, an affiliate of the Sponsor, equal to 92.6% of the issued and outstanding shares of Globalink common stock, have agreed to vote in favor of each of the Proposals. Assuming all of the outstanding shares of Globalink common stock vote on each Proposal, no Proposal requires the affirmative vote of any additional shares of common stock in order to be approved.
For more information about these proposals, see the sections of this proxy statement/prospectus entitled “The Special Meeting — Quorum and Required Vote for Proposals.”
Proxy Solicitation
Proxies may be solicited by telephone, by facsimile, by mail, or on the Internet. Globalink has engaged Okapi Partners LLC to assist in the solicitation of proxies. If a stockholder grants a proxy, it may still vote its shares in person online if it revokes its proxy before the Special Meeting. A stockholder may also change its vote by submitting a later-dated proxy, as described in the section titled “The Special Meeting — Revoking Your Proxy.”
Implication of Being an Emerging Growth Company
As a company with less than US$1.235 billion in revenues for the last fiscal year, PubCo qualifies and has elected to be treated as an “emerging growth company” pursuant to the Jumpstart Our Business Startups Act of 2012, or the JOBS Act. As such, PubCo will be able to take advantage of specified reduced reporting and other requirements that are otherwise applicable generally to public companies. These provisions include exemption from the auditor attestation requirement under Section 404 of the Sarbanes-Oxley Act of 2002 in the assessment of its internal control over financial reporting. Under the JOBS Act, PubCo also does not need to comply with any new or revised financial accounting standards until the date that private companies are required to do so.
PubCo will remain an emerging growth company until the earliest of (1) the last day of its fiscal year during which it has total annual gross revenues of at least US$1.235 billion; (2) the last day of its fiscal year following the fifth anniversary of the closing of the Business Combination; (3) the date on which PubCo has, during the previous three-year period, issued more than US$1.0 billion in non-convertible debt; or (4) the date on which PubCo is deemed to be a “large accelerated filer” under the Securities Exchange Act of 1934, as amended, or the Exchange Act, which would occur if PubCo has been a public company for at least 12 months and the market value of its ordinary shares that are held by non-affiliates exceeds US$700 million as of the last business day of its most recently completed second fiscal quarter. Once PubCo ceases to be an emerging growth company, it will not be entitled to the exemptions provided in the JOBS Act discussed above.
We do not believe Pubco’s election as an emerging growth company would impact Pubco as an IFRS filer. Furthermore, even after Pubco no longer qualifies as an “emerging growth company,” as long as Pubco continues to qualify as a foreign private issuer under the Exchange Act, Pubco will be exempt from certain provisions of the Exchange Act that are applicable to U.S. domestic public companies.
Regulatory Approvals
Under the HSR Act, and the related rules and regulations issued by the Federal Trade Commission, which we refer to as the FTC, certain transactions, including the Business Combination, may not be consummated until notifications have been given and specified information and documentary material have been furnished to the FTC and the United States Department of Justice, which we refer to as the DOJ, and the applicable waiting periods have expired or been terminated. The completion of the Business Combination is conditioned upon the expiration or early termination of the HSR Act waiting period. The initial 30-day waiting period expired at 11:59 pm Eastern time on . See the section entitled “The Acquisition Merger Proposal (Proposal 2): — The Merger Agreement — Covenants of the Parties” for additional information.
Appraisal Rights
Globalink stockholders do not have appraisal rights in connection with the Business Combination or the other proposals under Delaware law.
Redemption Rights
Pursuant to Globalink Charter, holders of shares of Globalink common stock may elect to have their shares redeemed for cash at the applicable redemption price per share equal to the quotient obtained by dividing (i) the aggregate amount on deposit in the Trust Account as of two business days prior to the consummation of the Business Combination, including interest (net of taxes payable), by (ii) the total number of then-outstanding public shares. As of , 2025, this would have amounted to approximately $ per share.
| 39 |
You will be entitled to receive cash for any shares of Globalink common stock to be redeemed only if you:
| (i) | (a) hold shares of Globalink common stock, or (b) if you hold shares of Globalink common stock through units, you elect to separate your units into the underlying shares of Globalink common stock, rights and warrants prior to exercising your redemption rights with respect to the shares of Globalink common stock; and | |
| (ii) | prior to , Eastern Time, on , 2025, (a) submit a written request to Continental that Globalink redeem your shares of Globalink common stock for cash and (b) deliver your shares of Globalink common stock to Continental, physically or electronically through DTC. |
For illustrative purposes, based on funds in the Trust Account of approximately $3.33 million on December 6, 2024, the estimated per share redemption price would have been approximately $11. A public stockholder, together with any of such stockholder’s affiliates or any other person with whom it is acting in concert or as a “group” (as defined under Section 13 of Exchange Act) will be restricted from redeeming in the aggregate such stockholder’s shares or, if part of such a group, the group’s shares, with respect to 20% or more of the shares of Globalink common stock included in the units of Globalink sold in the IPO (including overallotment securities sold to Globalink’s underwriters after the IPO).
Holders of outstanding units must separate the underlying shares of Globalink common stock, the warrants and the rights prior to exercising redemption rights with respect to the shares of Globalink common stock. If units are registered in a holder’s own name, the holder must deliver the certificate for its units to the transfer agent with written instructions to separate the units into their individual component parts.
In order to exercise your redemption rights, you must:
| ● | prior to 5:00 p.m. Eastern time on , 2024 (two (2) business days before the Special Meeting), tender your shares physically or electronically using DTC’s DWAC system and submit a request in writing that Globalink redeems your public shares for cash to Continental Stock Transfer & Trust Company, Globalink’s transfer agent, at the following address: | |
| Continental Stock Transfer & Trust Company | ||
| One State Street Plaza, 30th Floor | ||
| New York, New York 10004 | ||
| Attn: SPAC Redemption Team | ||
| E-mail: spacredemptions@continentalstock.com | ||
| ● | In your request to Continental for redemption, you must also affirmatively certify if you “ARE” or “ARE NOT” acting in concert or as a “group” (as defined in Section 13d-3 of the Exchange Act) with any other stockholder with respect to shares of Globalink common stock; and | |
| ● | deliver your public shares either physically or electronically through DTC to Globalink’s transfer agent at least two (2) business days before the Special Meeting. Stockholders seeking to exercise their redemption rights and opting to deliver physical certificates should allot sufficient time to obtain physical certificates from the transfer agent and time to effect delivery. It is Globalink’s understanding that stockholders should generally allot at least two weeks to obtain physical certificates from the transfer agent. However, Globalink does not have any control over this process and it may take longer than two weeks. Stockholders who hold their shares in “street name” will have to coordinate with their bank, broker or other nominee to have the shares certificated or delivered electronically. If you do not submit a written request and deliver your public shares as described above, your shares will not be redeemed. |
| 40 |
Any demand for redemption, once made, may be withdrawn at any time until the deadline for exercising redemption requests (and submitting shares to the transfer agent) and thereafter, with Globalink’s consent, until the consummation of the Business Combinations as determined by the Board. If you delivered your shares for redemption to Globalink’s transfer agent and decide within the required timeframe not to exercise your redemption rights, you may request that Globalink’s transfer agent return the shares (physically or electronically). You may make such a request by contacting Globalink’s transfer agent at the phone number or address listed above.
Prior to exercising redemption rights, stockholders should verify the market price of Globalink common stock as they may receive higher proceeds from the sale of their shares of Globalink common stock in the public market than from exercising their redemption rights if the market price per share is higher than the redemption price. We cannot assure you that you will be able to sell your shares of Globalink common stock in the open market, even if the market price per share is higher than the redemption price stated above, as there may not be sufficient liquidity in Globalink common stock when you wish to sell your shares.
If you exercise your redemption rights, your shares of Globalink common stock will cease to be outstanding immediately prior to the Business Combination and will only represent the right to receive a pro rata share of the aggregate amount on deposit in the Trust Account, calculated as of two business days prior to the consummation of the Business Combination. You will no longer own those shares and will have no right to participate in, or have any interest in, the future growth of the Combined Company, if any. You will be entitled to receive cash for these shares only if you properly and timely demand redemption.
If the Business Combination is not consummated and Globalink otherwise does not consummate an initial business combination by January 9, 2025, Globalink will be required to dissolve and liquidate its Trust Account by returning the then-remaining funds in such account to the public stockholders and the warrants and rights will expire worthless. If Globalink anticipates that it may not be able to consummate its initial business combination in time, Globalink may, by resolutions of its Board, if requested by the Sponsor, extend the period of time to consummate a business combination on a monthly basis to until June 9, 2025, subject to the Sponsor depositing additional funds into the Trust Account.
On December 10, 2024, Globalink received a delisting determination letter from Nasdaq as the 36-month anniversary from its IPO has passed on December 9, 2024, and Globalink are no longer in compliance with Nasdaq Listing Rule IM 5101-2(b). Globalink’s securities were suspended from trading and delisted from Nasdaq on December 17, 2024. As of the same date, Globalink’s securities started trading on OTC Pink. There could be substantial risks to Globalink and PubCo the as a result of Globalink’s non-compliance with this rule. See “Risk Factors — Risks Related to Globalink’s Business and the Business Combination —Our securities were suspended from trading and delisted from Nasdaq on December 17, 2024, following receipt of a delisting determination letter from Nasdaq on December 10, 2024. This could have significant material adverse consequences on us and our securities, including that it will negatively impact our ability to complete a business combination, will limit investors’ ability to make transactions in our securities and could subject us to additional trading restrictions.”
For an illustration of the dilutive effect redemptions on non-redeeming Globalink public shareholders, see the section below titled “—Ownership of the Combined Company After the Closing.” Assuming the maximum number of public shares are redeemed, non-redeeming public shareholders of Globalink will own an aggregate equity interest representing 0.8% in the Combined Company.
Ownership of the Combined Company After the Closing
It is anticipated that, upon the Closing of the Business Combination, under a no redemption scenario, Globalink’s public stockholders and Globalink’s right holders (other than the PIPE Investors) will retain an ownership interest of approximately 0.8% in the Combined Company, the PIPE Investors will own approximately 2.4% of the Combined Company, assuming the PIPE Investors will hold 4,020,000 PubCo ordinary shares, the Sponsor and directors and officers of Globalink will retain an ownership interest of approximately 1.7% in the Combined Company, PGM, an affiliate of the Sponsor, will retain an ownership interest of approximately 0.4% in the Combined Company, IBDC Asia Sdn. Bhd., an advisory firm to Alps, will own approximately 0.9% of the outstanding PubCo ordinary shares, representing finder’s fees payable in PubCo ordinary shares with value equal to 1% of the aggregate consideration for the Business Combination of US$1.6 billion, and the Alps Holdco Shareholders will own approximately 93.8% of the outstanding PubCo ordinary shares (excluding the Earnout Shares). The ownership percentages with respect to the Combined Company does not take into account the redemption of any shares by Globalink’s public stockholders.
The following summarizes the pro forma ownership of shares of common stock as of December 6, 2024, including shares of common stock underlying the units following the Business Combination and the PIPE Investment under both the no redemption, 25% redemptions, 50% redemptions, 75% redemptions and maximum contractual redemption scenarios, assuming that no Earnout Shares are issued.
Assuming No Redemptions | Assuming 25% Redemptions | Assuming 50% Redemptions | Assuming 75% Redemptions | Assuming Maximum Contractual Redemptions | ||||||||||||||||||||||||||||||||||||
Number of Shares | % | Number of Shares | % | Number of Shares | % | Number of Shares | % | Number of Shares | % | |||||||||||||||||||||||||||||||
| Alps Holdco Shareholders | 160,000,000 | 93.8 | % | 160,000,000 | 93.8 | % | 160,000,000 | 93.8 | % | 160,000,000 | 93.9 | % | 160,000,000 | 93.9 | % | |||||||||||||||||||||||||
| Globalink Public Shareholders | 277,511 | 0.2 | % | 208,133 | 0.1 | % | 138,755 | 0.1 | % | 69,377 | 0.0 | % | 0 | 0.0 | % | |||||||||||||||||||||||||
| Initial Stockholders | 2,875,000 | 1.7 | % | 2,875,000 | 1.7 | % | 2,875,000 | 1.7 | % | 2,875,000 | 1.7 | % | 2,875,000 | 1.7 | % | |||||||||||||||||||||||||
| Globalink Public Rights Holders | 1,150,000 | 0.6 | % | 1,150,000 | 0.7 | % | 1,150,000 | 0.7 | % | 1,150,000 | 0.7 | % | 1,150,000 | 0.7 | % | |||||||||||||||||||||||||
| PIPE Investors | 4,020,000 | 2.4 | % | 4,020,000 | 2.4 | % | 4,020,000 | 2.4 | % | 4,020,000 | 2.4 | % | 4,020,000 | 2.4 | % | |||||||||||||||||||||||||
| PGM | 627,000 | 0.4 | % | 627,000 | 0.4 | % | 627,000 | 0.4 | % | 627,000 | 0.4 | % | 627,000 | 0.4 | % | |||||||||||||||||||||||||
| IBDC Asia Sdn. Bhd. | 1,600,000 | 0.9 | % | 1,600,000 | 0.9 | % | 1,600,000 | 0.9 | % | 1,600,000 | 0.9 | % | 1,600,000 | 0.9 | % | |||||||||||||||||||||||||
| Total Shares | 170,549,511 | 100 | % | 170,480,133 | 100 | % | 170,410,755 | 100 | % | 170,341,377 | 100 | % | 170,272,000 | 100 | % | |||||||||||||||||||||||||
The table below illustrates the potential dilutive impact, excluding the exercise of warrants, to the holders of securities across five different redemption scenarios based on the assumptions described above, except that these scenarios assume the Combined Company has achieved all revenue milestones and the maximum number of Earnout Shares (48,000,000) are issued according to the earnout arrangement:
Assuming No Redemptions | Assuming 25% Redemptions | Assuming 50% Redemptions | Assuming 75% Redemptions | Assuming Maximum Contractual Redemptions | ||||||||||||||||||||||||||||||||||||
Number of Shares | % | Number of Shares | % | Number of Shares | % | Number of Shares | % | Number of Shares | % | |||||||||||||||||||||||||||||||
| Alps Holdco Shareholders | 208,000,000 | 95.2 | % | 208,000,000 | 95.2 | % | 208,000,000 | 95.2 | % | 208,000,000 | 95.3 | % | 208,000,000 | 95.3 | % | |||||||||||||||||||||||||
| Globalink Public Shareholders | 277,511 | 0.1 | % | 208,133 | 0.1 | % | 138,755 | 0.1 | % | 69,377 | 0.0 | % | 0 | 0.0 | % | |||||||||||||||||||||||||
| Initial Stockholders | 2,875,000 | 1.3 | % | 2,875,000 | 1.3 | % | 2,875,000 | 1.3 | % | 2,875,000 | 1.3 | % | 2,875,000 | 1.3 | % | |||||||||||||||||||||||||
| Globalink Public Rights Holders | 1,150,000 | 0.5 | % | 1,150,000 | 0.5 | % | 1,150,000 | 0.5 | % | 1,150,000 | 0.5 | % | 1,150,000 | 0.5 | % | |||||||||||||||||||||||||
| PIPE Investors | 4,020,000 | 1.9 | % | 4,020,000 | 1.9 | % | 4,020,000 | 1.9 | % | 4,020,000 | 1.9 | % | 4,020,000 | 1.9 | % | |||||||||||||||||||||||||
| PGM | 627,000 | 0.3 | % | 627,000 | 0.3 | % | 627,000 | 0.3 | % | 627,000 | 0.3 | % | 627,000 | 0.3 | % | |||||||||||||||||||||||||
| IBDC Asia Sdn. Bhd. | 1,600,000 | 0.7 | % | 1,600,000 | 0.7 | % | 1,600,000 | 0.7 | % | 1,600,000 | 0.7 | % | 1,600,000 | 0.7 | % | |||||||||||||||||||||||||
| Total Shares | 218,549,511 | 100 | % | 218,480,133 | 100 | % | 218,410,755 | 100 | % | 218,341,377 | 100 | % | 218,272,000 | 100 | % | |||||||||||||||||||||||||
All of the relative percentages above are for illustrative purposes only and are based upon certain assumptions as described in the section entitled “Frequently Used Terms — Share Calculations and Ownership Percentages” and, with respect to the determination of the “maximum contractual redemptions,” the section entitled “Unaudited Pro Forma Condensed Combined Financial Statements.” Should one or more of the assumptions prove incorrect, actual ownership percentages may vary materially from those described in this proxy statement/prospectus as anticipated, believed, estimated, expected or intended. See “Unaudited Pro Forma Condensed Combined Financial Information.”
| 41 |
Interests of Certain Persons in the Business Combination
When you consider the recommendation of the Board in favor of adoption of the Redomestication Merger Proposal, the Acquisition Merger Proposal, and other Proposals, you should keep in mind that Globalink’s directors and officers have interests in the Business Combination that are different from, or in addition to, your interests as a stockholder, including:
| ● | The Initial Stockholders, including the officers and directors of Globalink, have waived their right to redeem any shares of Globalink common stock in connection with a stockholder vote to approve a proposed initial business combination or sell any shares of Globalink common stock to Globalink in a tender offer prior to a proposed initial business combination, or to receive distributions with respect to the Founder Shares upon the liquidation of the Trust Account if Globalink is unable to consummate a business combination. Globalink currently has until January 9, 2025 to complete its initial business combination, or it will be required to dissolve and liquidate. If Globalink anticipates that it may not be able to consummate its initial business combination in time, Globalink may, by resolutions of its Board, if requested by the Sponsor, extend the period of time to consummate a business combination on a monthly basis to until June 9, 2025, subject to the Sponsor depositing additional funds into the Trust Account. In the event of a liquidation, the 2,875,000 Founder Shares currently held by the Initial Stockholders, which were acquired prior to the IPO will be worthless because such holders have agreed to waive their rights to any liquidation distributions through the Insider Letter, in consideration for inducing Chardan, the representative of the underwriters for Globalink’s IPO to proceed with the IPO, and the economic benefit the IPO would confer upon the Initial Stockholders and PGM. The Founder Shares were purchased for an aggregate purchase price of $25,000, or less than $0.01 per share, and had a total market value of $34.36 million as of December 12, 2024. Accordingly, the Initial Stockholders will receive a positive rate of return so long as the market price of Globalink common stock is at least $0.01 per share, even if public stockholders experience a negative rate of return in the Combined Company. | |
| ● | The fact that the Initial Stockholders’ Founder Shares became subject to a lock-up whereby, subject to certain limited exceptions, the Initial Stockholders agree not to offer, sell, contract to sell, pledge or otherwise dispose of, directly or indirectly, any of the Lock-up Shares, enter into a transaction that would have the same effect, or enter into any swap, hedge or other arrangement that transfers, in whole or in part, any of the economic consequences of ownership of the Lock-Up Shares or otherwise, publicly disclose the intention to make any offer, sale, pledge or disposition, or to enter into any transaction, swap, hedge or other arrangement, or engage in any Short Sales (as defined in the Lock-up Agreement) with respect to the Lock-Up Shares until (i) (w) with respect to one hundred percent (100%) of the Lock-Up Shares, the date from the Closing until the last date that is six (6) months after the date of the Closing, (x) with respect to ninety percent (90%) of the Lock-Up Shares, the 1st day of the 7th month from the Closing until the date that is nine (9) months after the date of the Closing, (y) with respect to seventy percent (70%) of the Lock-Up Shares, the 1st day of the 10th month from the Closing until the date that is twelve (12) months after the date of the Closing, (z) with respect to forty percent (40%) of the Lock-Up Shares, the 1st day of the 13th month from the Closing until the date that is fifteen (15) months after the date of the Closing, or (ii) if sooner, the date after the Closing on which Globalink consummates a liquidation, merger, share exchange or other similar transaction with an unaffiliated third party that results in all of Globalink’s stockholders having the right to exchange their equity holdings in Globalink for cash, securities or other property. For the avoidance of doubt, no Lock-up Shares shall be subject to the terms in the Lock-up Agreement from and after the date that is fifteen (15) months after the date of the Closing; | |
| ● | The exercise of Globalink’s directors’ and officers’ discretion in agreeing to changes or waivers in the terms of the Business Combination may result in a conflict of interest when determining whether such changes or waivers are appropriate and in Globalink stockholders’ best interest. Globalink’s directors were aware of and considered these interests, among other matters, in evaluating the Business Combination, and in recommending to Globalink stockholders that they approve the Business Combination. Further, the interests of Globalink’s officers or directors may be different from or in addition to (and which may conflict with) your interests and may be incentivized to complete a less favorable business combination rather than liquidating Globalink. Public stockholders should take these interests into account in deciding whether to approve the Business Combination. | |
| ● | As of September 30, 2024, Globalink’s officers and directors have incurred an aggregate of $11,798 in out-of-pocket expenses. On each of September 5, 2023, September 29, 2023 and November 7, 2023, an affiliate of the Sponsor advanced $130,000 to the Company, for a total advance of $390,000, for Globalink’s extension payments. Unless Globalink consummates an initial business combination, Globalink’s officers, directors and the Sponsor (including its affiliates) will not receive reimbursement for any out-of-pocket expenses incurred by them to the extent that such expenses exceed the amount of available proceeds not deposited in the Trust Account. Upon the consummation of an initial business combination, Globalink’s offices, directors, and the Sponsor (including its affiliates) would be entitled to reimbursement for out-of-pocket expenses incurred by them from the Trust Account disbursements after redemption payouts and payments of deferred underwriting discounts. | |
| ● | The fact that, if the Trust Account is liquidated, including in the event Globalink is unable to complete an initial business combination within the required time period, the Sponsor has agreed to be liable to Globalink if and to the extent any claims by a vendor for services rendered or products sold to Globalink, or a prospective target business with which Globalink has discussed entering into a transaction agreement, reduce the amount of funds in the Trust Account. This liability will not apply with respect to any claims by a third party who executed a waiver of any right, title, interest or claim of any kind in or to any monies held in the Trust Account or to any claims under Globalink’s indemnity of the underwriters of the IPO against certain liabilities, including liabilities under the Securities Act. | |
| ● | As of the date hereof, the deadline that Globalink has to consummate its initial business combination has been extended for a total of eighteen times, and a total of US$1,950,000 has been deposited into the Trust Account in connection with the previous extensions. In connection with funding the extensions, Globalink has issued an aggregate of US$3.85 million in promissory notes to PGM, which are payable upon the Closing. In the event of a liquidation, PGM has waived its rights to any liquidation distributions through the Insider Letter. PGM is an affiliate of the Sponsor as its 95% equity holder has a familial relationship with the control person of the Sponsor. As a result, the Sponsor has an additional interest in causing Globalink to consummate its initial business combination due to its familial relationship with PGM. | |
| ● | After the Closing, the Sponsor will have the sole authority to handle various matters relating to the Escrow Account and indemnifications under the Merger Agreement. The Sponsor may have a conflict of interest when exercising such authority to handle matters relating to the Escrow Account and indemnifications after Closing, and they may not make decisions in the Globalink stockholders’ best interests. For details, see “Summary of the Proxy Statement/Prospectus—Parties to the Business Combination—Parent Representative.” |
| 42 |
These interests may have influenced Globalink’ directors in making their recommendation that you vote in favor of the approval of the Business Combination. See the section entitled “The Acquisition Merger Proposal (Proposal 2) — Interests of Certain Persons in the Business Combination” for additional information.
Prior to the consummation of an initial business combination by Globalink, the insider shares can only be transferred by the Initial Stockholders to (1) the Initial Stockholders or their respective affiliates or members, or to Globalink’s officers, directors, advisors and employees, (2) the Initial Stockholder’s affiliates or its members upon its liquidation, (3) to relatives and trusts for estate planning purposes, (4) by virtue of the laws of descent and distribution upon death of an Initial Stockholder, (5) pursuant to a qualified domestic relations order, or (6) by private sales made at prices no greater than the price at which the Insider Shares were originally purchased.
U.S. Federal Income Tax Consequences
The material U.S. federal income tax considerations that may be relevant to you in respect of the Business Combination are discussed in more detail in the section titled “Material U.S. Federal Income Tax Consequences” beginning on page 149, which contains a more detailed discussion of the U.S. federal income tax consequences of the Business Combination. You should also consult your tax advisor for a complete analysis of the effect of the Business Combination on your federal, state and local and/or foreign taxes.
Anticipated Accounting Treatment
The Business Combination will be accounted for as a “capital reorganization,” with no goodwill or other intangible assets recorded, in accordance with International Financial Reporting Standards (“IFRS”). A capital reorganization does not result in a new basis of accounting, and the financial statements of the combined entity represent the continuation of the financial statements of Alps Holdco in many respects. However, Globalink does not meet the definition of a “business” pursuant to IFRS 3 Business Combinations, and thus, for accounting purposes, the Business Combination will be accounted for as a capital reorganization.
This determination was primarily based on the assumption that Alps Holdco shareholders will hold the largest minority of the voting power of PubCo, Alps Holdco operations will substantially comprise the ongoing operations of PubCo, Alps Holdco designees are expected to comprise a majority of the governing body of PubCo, and Alps Holdco’s senior management will comprise the senior management of PubCo.
Under this method of accounting, Globalink will be treated as the “acquired” company for financial reporting purposes. For accounting purposes, Alps Holdco will be deemed to be the accounting acquirer in the transaction and, consequently, the transaction will be treated as a recapitalization of Alps Holdco (i.e., a capital transaction involving the issuance of shares by PubCo for the shares of Alps Holdco). Accordingly, the consolidated assets, liabilities and results of operations of Alps will become the historic financial statements of the Combined Company, and Globalink’s assets, liabilities and results of operations will be consolidated with Alps’ beginning on the acquisition date. Operations prior to the Business Combination will be presented as those of Alps Holdco in future reports. The net assets of Alps Holdco will be recognized at carrying value, with no goodwill or other intangible assets recorded.
The deemed costs of the shares issued by PubCo, which represents the fair value of the shares that Alps Holdco would have had to issue for the ratio of ownership interest in PubCo to be the same as if the Business Combination had taken the legal form of Alps Holdco acquiring shares of Globalink, in excess of the net assets of Globalink will be accounted for as stock-based compensation under IFRS 2 Share-Based Payment.
Recommendations of the Board and Reasons for the Business Combination
After careful consideration of the terms and conditions of the Merger Agreement, the Board has determined that Business Combination and the transactions contemplated thereby are fair to and in the best interests of Globalink and its stockholders. In reaching its decision with respect to the Business Combination, the Board reviewed various industry and financial data and the due diligence and evaluation materials provided by Alps. The Board recommends that Globalink stockholders vote:
| ● | FOR the Redomestication Merger Proposal; | |
| ● | FOR the Acquisition Merger Proposal; | |
| ● | FOR the Nasdaq Proposals; and | |
| ● | FOR the Adjournment Proposal. |
Summary of Risk Factors
In evaluating the Business Combination and the Proposals to be considered and voted on at the Special Meeting, you should carefully review and consider the risk factors set forth under the section titled “Risk Factors” beginning on page 52 of this proxy statement/prospectus. Some of these risks related to are summarized below. References in the summary below to “Alps” generally refer to Alps in the present tense or the Combined Company from and after the Business Combination.
The following summarizes certain principal factors that make an investment in the Combined Company speculative or risky, all of which are more fully described in the “Risk Factors” section below. This summary should be read in conjunction with the “Risk Factors” section and should not be relied upon as an exhaustive summary of the material risks related to Globalink’s, Alps’ and/or the Combined Company’s business.
| 43 |
Risks Related to Alps’ Business and Industry and Combined Company following the Business Combination
Alps’ business is subject to a number of risks of which you should be aware before making an investment decision. These risks are discussed more fully in the section of this proxy statement/prospectus titled “Risk Factors” immediately following this proxy statement/prospectus summary. These risks include, but are not limited to, the following:
| ● | Alps Life Sciences Inc is a recently formed holding company with no operating history. It expects to operate through Alps Global Holding Berhad which, in turn, is the holding company of several operating subsidiaries. Alps Global Holding Berhad has a limited operating history since its inception which may make it difficult to evaluate its current business and this makes predictions about its future success or viability subject to significant uncertainty. Alps Global Holding Berhad is currently not a subsidiary of Alps Life Sciences Inc but is in the process of becoming a subsidiary of Alps Life Sciences Inc. | |
| ● | Alps will need to obtain substantial additional funding to complete the development of its product candidates. | |
Should we fail to secure the necessary capital in adequate amounts or under acceptable terms, we might be compelled to reduce our workforce significantly, delay, downscale, or cease the development or commercialization of our product candidates, or explore less desirable collaborations for our product candidates. Such scenarios could severely impact our business prospects, financial health, operational results, and lead to a decline in PubCo’s share price after the Business Combination.
| ||
| ● | Alps has identified a material weakness in its internal control over financial reporting and may identify additional material weaknesses in the future that may cause it to fail to meet its reporting obligations or result in material misstatements in its financial statements. If Alps fails to remediate its material weakness, Alps may not be able to report its financial results accurately or to prevent fraud. | |
| ● | The selection and prioritization of our product candidates for development are subject to change based on various factors. Abandoning the development of a product candidate or altering Alps’ development priorities could potentially leave it without a viable replacement, thus impacting its business prospects. | |
| ● | Alps faces challenges in developing its manufacturing capabilities and may rely on third-party manufacturers, exposing it to various risks, exposing it to various risks including lack of accessibility, quality control, quantity limitations, manufacturing delays, contractual breaches or terminations. | |
| ● | Alps’ current product candidates are in the proof-of-concept and preclinical stage and have never been tested in humans. One or all of Alps’ current product candidates may fail in clinical development or suffer delays that materially and adversely affect their commercial viability. | |
| ● | Clinical trials are expensive, time-consuming and difficult to design and implement. There can be no assurance that Alps’ product candidates will be able to successfully complete clinical trials. The failure of Alps’ product candidates to complete clinical trials may have a material adverse effect on its business, financial condition, results of operations and prospects.
| |
| ● | If Alps encounter difficulties enrolling patients in clinical trials, Alps’ clinical development activities could be delayed or otherwise adversely affected. | |
| ● | Compassionate use of stem cells therapies and/or other medical treatments and procedures provided by Alps may expose Alps to medical malpractices claims, result in injuries that lead to costly liability suits, and/or subject it to penalties if Alps fails to comply with regulatory requirements or experience unanticipated problems with any products and/or product candidates.
| |
| ● | The commercial success of any of Alps’ current or future product candidates will depend upon the degree of market acceptance by physicians, patients, third-party payors, and others in the medical community. | |
| ● | If the use or misuse of Alps’ product candidates harm patients or is perceived to harm patients even when such harm is unrelated to Alps’ product candidates, Alps’ regulatory approvals could be revoked or could otherwise be negatively impacted, and Alps could be subject to costly and damaging product liability claims. | |
| ● | Any cell-based products that receive regulatory approval may be difficult and expensive to manufacture on a commercial scale. | |
| ● | The manufacturing operations of Alps’ potential product candidates including but not limited to cosmetics products infused with exosomes are themselves dependent upon third-party suppliers, making us vulnerable to supply shortages and price fluctuations, which could harm its business.
| |
| Alps relies on patents and intellectual property rights licensed from third parties to engage in research, development and commercialization activities. If Alps fails to maintain these licenses on terms favorable or otherwise acceptable to Alps, Alps’ ability to engage in research, development and commercialization activities may be materially impacted, resulting in a materially adverse effect on our business, financial condition and operational results. |
| 44 |
| ● | If Alps, its subsidiaries, or associate companies fail to meet their obligations under license agreements, they risk losing the rights to critical technologies upon which the business depends. | |
| ● | Alps relies on intellectual property that is jointly developed with third parties which exposes Alps to risks associated with the failure of joint owners to maintain, protect, and enforce such intellectual property and divergent interests arising during the course of joint ownership. | |
| ● | Upon the consummation of the Business Combination, PubCo expects to qualify as an “emerging growth company,” and may elect to comply with reduced public company reporting requirements applicable to emerging growth companies, which could make PubCo ordinary shares less attractive to investors. | |
| ● | Alps faces the risk that changes in the policies of the Malaysian government could have a significant impact upon the business it may be able to conduct in Malaysia and the profitability of such business. | |
| ● | The regulatory approval processes of the NPRA and comparable foreign authorities such as FDA are lengthy, time consuming and inherently unpredictable, and if Alps is ultimately unable to obtain regulatory approval for Alps’ product candidates, Alps’ business will be substantially harmed. | |
| ● | The FDA and other comparable foreign regulatory authorities may not accept data from trials or studies conducted in Malaysia or other locations outside of their jurisdiction.
| |
| ● | mRNA drug development has substantial clinical development and regulatory risks due to the novel and unprecedented nature of this new category of therapeutics.
| |
| ● | If Alps is unable to recruit and retain an adequate number of managers, doctors, nurses, consultants and other support staff in its treatment centres, our service quality and business strategy may suffer. | |
| ● | If Alps is unable to adapt to changing aesthetic medical trends and its customers’ changing needs, we will not be able to compete effectively, which may materially and adversely affect its business, financial condition and results of operations.
| |
| ● | Any failure in Alps’ efforts to train health practitioners could result in the misuse of its products, reduce the market acceptance of its products and have a material adverse effect on the business, financial condition, and results of operations. | |
| ● | In the future, if Alps’ customers cannot obtain third-party reimbursement for their use of Alps’ products, they could be less inclined to purchase Alps’ products. |
| 45 |
Risks Related to Globalink’s Business and the Business Combination
| ● | You must tender your shares of Globalink common stock in order to validly seek redemption at the Special Meeting. | |
| ● | If third parties bring claims against Globalink, the proceeds held in trust could be reduced and the per share liquidation price received by Globalink’s stockholders may be less. | |
| ● | Any distributions received by Globalink stockholders could be viewed as an unlawful payment if it was provided that immediately following the date on which the distribution was made, Globalink was unable to pay its debts as they fell due in the ordinary course of business and the value of its assets does not exceed its liabilities. | |
| ● | There is no assurance that Globalink’s diligence will reveal all material risks that may be present with regard to Alps. Subsequent to the completion of the Business Combination, the Combined Company may be required to take write-downs or write-offs, restructuring and impairment or other charges that could have a significant negative effect on its financial condition and its share price, which could cause you to lose some or all of your investment. |
Risks Related to the PubCo ordinary shares
| ● | The Combined Company’s share price may fluctuate significantly. | |
| ● | Your percentage ownership in the Combined Company will be diluted should the Earnout Shares and the PIPE Investment related shares be issued and may be diluted in the future due to the exercise of outstanding options, warrants and rights. | |
| ● | Future sales, or the perception of future sales, of the PubCo ordinary shares by the Combined Company or its existing stockholders in the public market could cause the market price for the PubCo ordinary shares to decline. |
Risks Related to Redemption
| ● | Globalink’s public stockholders who wish to redeem their public shares of Globalink common stock for a pro rata portion of the Trust Account must comply with specific requirements for redemption that may make it more difficult for them to exercise their redemption rights prior to the deadline. If stockholders fail to comply with the redemption requirements specified in this proxy statement/prospectus, they will not be entitled to redeem their public shares of Globalink common stock for a pro rata portion of the funds held in the Trust Account. | |
| ● | If a public stockholder of Globalink fails to receive notice of Globalink’s offer to redeem shares of Globalink common stock in connection with the Business Combination or fails to comply with the procedures for tendering its shares, such shares of Globalink common stock may not be redeemed. | |
| ● | If you or a “group” of stockholders of which you are a part are deemed to hold an aggregate of more than 20% of the public shares of Globalink common stock, you (or, if a member of such a group, all of the members of such group in the aggregate) will lose the ability to redeem all such shares in excess of 20% of the public shares of Globalink common stock. |
| 46 |
SELECTED HISTORICAL FINANCIAL DATA OF GLOBALINK
Globalink’s consolidated statements of operations data for the fiscal years ended December 31, 2023 and 2022 and consolidated balance sheet data as of December 31, 2023 and 2022 are derived from Globalink’s consolidated financial statements prepared under U.S. GAAP included elsewhere in this proxy statement/prospectus. Globalink’s consolidated statements of operations data for the fiscal quarters ended September 30, 2024 and 2023, March 31, 2024 and 2023 and consolidated balance sheet data as of September 30, 2024, March 31, 2024 are derived from Globalink’s unaudited consolidated financial statements included elsewhere in this proxy statement/prospectus.
The historical results of Globalink included below and elsewhere in this proxy statement/prospectus are not necessarily indicative of the future performance of Globalink. You should read the following selected financial data in conjunction with “Management’s Discussion and Analysis of Financial Condition and Results of Operations of Globalink” and the financial statements and the related notes appearing elsewhere in this proxy statement/prospectus.
| Three months period ended September 30, 2024 | Nine months period ended September 30, 2024 | Three months period ended September 30, 2023 | Nine months period ended September 30, 2023 | |||||||||||||
| Consolidated Statements of Operations Data | ||||||||||||||||
| Total operating expenses | $ | (337,590 | ) | $ | (1,404,334 | ) | $ | (309,829 | ) | $ | (849,621 | ) | ||||
| Total other income | $ | 311,930 | $ | 893,270 | $ | 591,464 | $ | 2,501,199 | ||||||||
| Net income (loss) | $ | (116,747 | ) | $ | (709,239 | ) | $ | 254,297 | $ | 1,224,868 | ||||||
| Three months period ended March 31, 2024 | Three months period ended March 31, 2023 | Year ended December 31, 2023 | Year ended December 31, 2022 | |||||||||||||
| Consolidated Statements of Operations Data | ||||||||||||||||
| Total operating expenses | $ | (597,133 | ) | $ | (321,537 | ) | $ | (1,175,824 | ) | $ | (1,259,743 | ) | ||||
| Total other income (expense) | $ | 295,117 | $ | 1,258,047 | $ | 3,025,653 | $ | 1,792,170 | ||||||||
| Net income (loss) | $ | (375,307 | ) | $ | 682,939 | $ | 1,320,324 | $ | 224,242 | |||||||
As of September 30, 2024 |
As of March 31, 2024 | As of December 31, 2023 | As of December 31, 2022 | |||||||||||||
| Consolidated Balance Sheets Data | ||||||||||||||||
| Total current assets | $ | 125,967 | $ | 142,158 | $ | 204,698 | $ | 289,208 | ||||||||
| Total assets | $ | 29,937,223 |
$ | 29,189,431 | $ | 28,872,916 | $ | 118,698,177 | ||||||||
| Total stockholders’ deficit | $ | (9,257,978 |
) | $ | (8,094,816 | ) | $ | (7,263,745 | ) | $ | (4,053,192 | ) | ||||
| Common stock subject to possible redemption | $ | 29,223,707 |
$ | 28,394,477 | $ | 27,938,713 | $ | 117,864,419 | ||||||||
| Total current liabilities | $ | 5,932,244 |
$ | 4,851,090 | $ | 4,171,067 | $ | 776,322 | ||||||||
| Total liabilities | $ | 9,971,494 |
$ | 8,889,770 | $ | 8,197,948 | $ | 4,886,950 | ||||||||
| 47 |
SELECTED HISTORICAL CONSOLIDATED FINANCIAL DATA OF ALPS
The following selected historical financial information for Alps set forth below should be read in conjunction with “Management’s Discussion and Analysis of Financial Condition and Results of Operations of Alps” and Alps’ historical financial statements and the related notes included elsewhere in this proxy statement/prospectus. Alps Global Holding Pubco is a newly incorporated exempted company in Cayman Islands on May 14, 2024 for the purpose of completing a business combinations with all of the shareholders of Alps Global Holding Berhad will exchange their equity ownership for identical interests in Alps Pubco. Alps Pubco has not commenced operations and has no assets or liabilities since its incorporation.
The selected historical financial information presented below for the year ended March 31, 2023 has been derived from Alps’ audited financial statements included elsewhere in this proxy statement/prospectus.
In the opinion of Alps management, such unaudited financial data reflect all adjustments, consisting only of normal and recurring adjustments necessary for a fair presentation of the results for those periods. The results of operations for the interim periods are not necessarily indicative of the results to be expected for the full year or any future period.
| For the year Ended March 31, 2024 | For the year Ended March 31, 2023 | |||||||
| $ | $ | |||||||
| Consolidated Statement of Profit or Loss and Other Comprehensive Income Data | ||||||||
| Revenue | 2,403,552 | 3,594,828 | ||||||
| Total operating expenses | (3,927,166 | ) | (3,378,589 | ) | ||||
| Total other income/(expenses) | (867,547 | ) | (946,101 | ) | ||||
| Net loss | (2,391,161 | ) | (729,862 | ) | ||||
| For the year Ended March 31, 2024 | For the year Ended March 31, 2023 | |||||||
| $ | $ | |||||||
| Consolidated Statement of Financial Position Data: | ||||||||
| Total current assets | 1,284,502 | 1,088,385 | ||||||
| Total assets | 6,700,933 | 4,802,896 | ||||||
| Total current liabilities | 5,055,579 | 2,982,923 | ||||||
| Total liabilities | 5,249,941 | 3,172,140 | ||||||
| Stockholders’ equity | 1,450,992 | 1,630,756 | ||||||
| 48 |
SUMMARY UNAUDITED PRO FORMA FINANCIAL STATEMENTS
SUMMARY UNAUDITED PRO FORMA CONDENSED
COMBINED FINANCIAL INFORMATION
The following summary unaudited pro forma condensed combined financial information (the “Summary Pro Forma Information”) gives effect to the transactions contemplated by the Business Combination and related transactions. The Business Combination will be accounted for as a reverse recapitalization in accordance with IFRS. Under this method of accounting, Globalink will be treated as the “acquired” company for financial reporting purposes. Accordingly, the Business Combination will be reflected as the equivalent of Alps Holdco issuing shares for the net assets of Globalink, followed by a recapitalization whereby no goodwill or other intangible assets are recorded. Operations prior to the Business Combination will be those of Alps. The summary unaudited pro forma condensed combined balance sheet data as of March 31, 2024 gives effect to the Business Combination and related transactions as if they had occurred on March 31, 2024. The summary unaudited pro forma condensed combined statements of operations data for the fiscal year ended March 31, 2024 give effect to the Business Combination and related transactions as if they had occurred on April 1, 2023.
The Summary Pro Forma Information has been derived from, and should be read in conjunction with, the more detailed unaudited pro forma condensed combined financial information included in the section titled “Unaudited Pro Forma Condensed Combined Financial Information” in this proxy statement/prospectus and the accompanying notes thereto. The unaudited pro forma condensed combined financial information is based upon, and should be read in conjunction with, the historical consolidated financial statements and related notes of Globalink and Alps for the applicable periods included in this proxy statement/prospectus. The Summary Pro Forma Information has been presented for informational purposes only and is not necessarily indicative of what Globalink’s financial position or results of operations actually would have been had the Business Combination and related transactions been completed as of the dates indicated. In addition, the Summary Pro Forma Information does not purport to project the future financial position or operating results of Globalink following the reverse recapitalization.
The Summary Pro Forma Information has been prepared using the assumptions below with respect to the potential redemption into cash of shares of Globalink common stock:
| 1. | Assuming No Redemptions: This scenario assumes that no public stockholders of Globalink exercise redemption rights with respect to their public shares for a pro rata share of the funds in Globalink’s Trust Account. | |
| 2. | Assuming Maximum Contractual Redemptions: This scenario assumes that 277,511 shares of Globalink common stock, the maximum contractual redemption of the outstanding Globalink common stock, are redeemed for their pro rata share of the cash in the Trust Account. This presentation assumes that Globalink stockholders exercise their redemption rights with respect to a maximum of 277,511 common stock for an aggregate payment of approximately $3.33 million upon consummation of the Business Combination at a redemption price of approximately $11.98 per share, determined based on funds in trust plus interest earned net of taxes payable. The maximum contractual redemption amount reflects the maximum number of Globalink public shares that can be redeemed without violating the requirement of Globalink Charter that Globalink will only redeem public shares if after such redemption the net tangible assets of the Corporation as a public company will be at least $5,000,001 upon consummation of the initial Business Combination. Scenario 2 includes all adjustments contained in Scenario 1 and presents additional adjustments to reflect the effect of the maximum contractual redemptions. |
Statement of Operations for the twelve months ended March 31, 2024
| PubCo (IFRS Historical) | Alps Holdco (IFRS Historical) | Globalink (US GAAP Historical) | Scenario 1: No Additional Redemption Scenario | Scenario 2: Maximum Contractual Redemption Scenario | ||||||||||||||||
| Revenue | $ | - | $ | 2,403,552 | $ | - | $ | 2,403,552 | $ | 2,403,552 | ||||||||||
| Cost of sales | $ | - | $ | (1,804,622 | ) | $ | - | $ | (1,804,622 | ) | $ | (1,804,622 | ) | |||||||
| Gross profit | $ | - | $ | 598,930 | $ | - | $ | 598,930 | $ | 598,930 | ||||||||||
| Operating profit | $ | - | $ | (2,359,152 | ) | $ | (1,451,420 | ) | $ | (86,547,577 | ) | $ | (86,840,113 | ) | ||||||
| Profit (loss) before income tax | $ | - | $ | (2,398,575 | ) | $ | 611,303 | $ | (86,856,274 | ) | $ | (87,148,810 | ) | |||||||
| Net loss | $ | - | $ | (2,391,161 | ) | $ | 262,078 | $ | (87,198,085 | ) | $ | (87,490,621 | ) | |||||||
| Net income (loss) attributed to shareholders | $ | - | $ | (2,304,026 | ) | $ | 262,078 | $ | (87,110,950 | ) | $ | (87,403,486 | ) | |||||||
| Balance sheet as of March 31, 2024 | ||||||||||||||||||||
| PubCo (IFRS Historical) | Alps Holdco (IFRS Historical) | Globalink (US GAAP Historical) | Scenario 1: No Additional Redemption Scenario | Scenario 2: Maximum Contractual Redemption Scenario | ||||||||||||||||
| Total current assets | $ | - | $ | 1,284,502 | $ | 142,158 | $ | 34,944,407 | $ | 31,618,676 | ||||||||||
| Total non-current assets | $ | - | $ | 5,416,431 | $ | 29,047,273 | $ | 5,416,431 | $ | 5,416,431 | ||||||||||
| Total assets | $ | - | $ | 6,700,933 | $ | 29,189,431 | $ | 40,360,838 | $ | 37,035,107 | ||||||||||
| Total equity (deficit) | $ | - | $ | 1,450,992 | $ | (8,094,816 | ) | $ | 14,910,104 | $ | 11,584,373 | |||||||||
| Common stock subject to possible redemption | $ | - | $ | - | $ | 28,394,477 | $ | - | $ | - | ||||||||||
| Total current liabilities | $ | - | $ | 5,055,579 | $ | 4,851,090 | $ | 24,966,692 | $ | 24,966,692 | ||||||||||
| Total liabilities | $ | - | $ | 5,249,941 | $ | 8,889,770 | $ | 25,450,734 | $ | 25,450,734 | ||||||||||
| 49 |
COMPARATIVE PER SHARE INFORMATION
The following table sets forth the per share data of each of Globalink and Alps on a stand-alone basis and the audited pro forma condensed combined per share data for years ended March 31, 2024 and after giving effect to the Business Combination assuming (i) no redemption of shares of Globalink common stock, and (ii) maximum contractual redemption of shares of Globalink common stock. The pro forma earnings information for the fiscal years ended March 31, 2024 were computed as if the Business Combination had been completed on April 1, 2023, and carried forward through the years.
The historical book value per share is computed by dividing total common stockholders’ equity by the number of shares of Globalink common stock outstanding at the end of the period. The pro forma combined book value per share of Globalink common stock is computed by dividing total pro forma common stockholders’ equity by the pro forma number of shares of Globalink common stock outstanding at the end of the period. The pro forma earnings per share of the Combined Company is computed by dividing the pro forma income available to the Combined Company’s shareholders by the pro forma weighted-average number of shares of Globalink common stock outstanding over the period.
You should read the information in the following table in conjunction with the selected historical financial information summary included elsewhere in this proxy statement/prospectus, and the historical financial statements of Globalink and Alps and related notes that are included elsewhere in this proxy statement/prospectus. The unaudited Globalink and Alps pro forma combined per share information is derived from, and should be read in conjunction with, the unaudited pro forma condensed combined financial statements and related notes included elsewhere in this proxy statement/prospectus.
The unaudited pro forma combined earnings per share information below does not purport to represent the earnings per share which would have occurred had the companies been combined during the periods presented, nor earnings per share for any future date or period. The unaudited pro forma combined book value per share information below does not purport to represent what the value of Globalink and Alps would have been had the companies been combined during the periods presented.
| PubCo (IFRS Historical) | ALPS Holdco (IFRS Historical) | Globalink (US GAAP Historical) | Scenario 1: No Additional Redemption Scenario | Scenario 2: Maximum Contractual Redemption Scenario | ||||||||||||||||
| March 31, 2024 | ||||||||||||||||||||
| Net Income (Loss) | $ | - | $ | (2,304,026 | ) | $ | 262,078 | $ | (87,110,950 | ) | $ | (87,403,486 | ) | |||||||
| Shareholders’ Equity (Deficit) | $ | - | $ | 1,567,183 | $ | (8,094,816 | ) | $ | 15,026,295 | $ | 11,700,564 | |||||||||
| Shareholders’ Equity per Share * | $ | 0.09 | $ | 0.07 | ||||||||||||||||
| Cash dividends | $ | - | $ | - | $ | - | $ | - | $ | - | ||||||||||
| Weighted average shares – Basic | 170,549,511 | 170,272,000 | ||||||||||||||||||
| Weighted average shares – Diluted | 170,549,511 | 170,272,000 | ||||||||||||||||||
| Net loss per share – Basic | $ | (0.51 | ) | $ | (0.51 | ) | ||||||||||||||
| Net loss per share – Diluted | $ | (0.51 | ) | $ | (0.51 | ) | ||||||||||||||
* Shareholders’ equity per share = shareholders’ equity/shares outstanding
| 50 |
TRADING MARKET AND DIVIDENDS
Globalink
Globalink’s units, common stock, warrants and rights are each quoted on the OTC Pink, under the symbols “GLLIU,” “GLLI,” “GLLIW,” and “GLLIR,” respectively. Each unit consists of one share of Globalink common stock, one redeemable warrant, each entitling its holder to purchase one-half (1/2) of one share of common stock at a price of $11.50 per whole share, and one right to receive one-tenth (1/10) of one share of Globalink common stock upon the consummation of the Business Combination. Globalink’s units commenced trading on Nasdaq on December 7, 2021. Globalink common stock, public rights and public warrants commenced trading on Nasdaq on December 21, 2021. Globalink’s units, common stock, public rights and public warrants were delisted from Nasdaq on December 17, 2024.
Holders
As of , 2025, the Record Date for the Special Meeting, there were holders of record of our units, holders of record of shares of common stock, holders of record for our rights and holders of record of Globalink warrants. The number of holders of record does not include a substantially greater number of “street name” holders or beneficial holders whose units, common stock, rights and warrants are held of record by banks, brokers and other financial institutions.
Dividend Policy
Globalink has not paid any cash dividends on its common stock to date and does not intend to pay cash dividends prior to the completion of a business combination. The payment of cash dividends in the future will be dependent upon Globalink’s revenues and earnings, if any, capital requirements and general financial condition subsequent to completion of a business combination. Further, if Globalink incurs any indebtedness, its ability to declare dividends may be limited by restrictive covenants Globalink may agree to in connection therewith. It is the present intention of the Board to retain all earnings, if any, for use in its business operations and, accordingly, the Board does not anticipate declaring any dividends in the foreseeable future.
The payment of any dividends subsequent to a business combination will be within the discretion of the Combined Company’s Board of Directors, subject to applicable laws and organizational documents. The Combined Company does not anticipate declaring or paying any dividends in the foreseeable future. It expects to retain its available funds and any future earnings for use in operations, pipeline development activities and expansion. Additionally, the Combined Company does not plan to use retained earnings for debt repayment.
Alps
There is no public market for Alps Holdco Ordinary Shares.
| 51 |
RISK FACTORS
You should carefully consider the following risk factors, as well as the other information set forth in this proxy statement/prospectus, before you decide whether to vote or direct your vote to be cast to approve the Business Combination.
The value of your investment following consummation of the Business Combination will be subject to significant risks affecting, among other things, the Combined Company’s business, financial condition or results of operations. If any of the events described below occur, the Combined Company’s post-Business Combination business and financial results could be adversely affected in material respects. This could result in a decline, which may be significant, in the trading price of the Combined Company’s securities and you therefore may lose all or part of your investment. The risk factors described below are not necessarily exhaustive and you are encouraged to perform your own investigation with respect to the businesses of Globalink and Alps.
Risks Related to Alps’ Business and Industry and the Combined Company
The following risk factors apply to the business and operations of Alps Life Sciences Inc and its operating subsidiaries, which are owned by Alps Global Holding Berhad, and will also apply to the business and operations of Alps Life Sciences Inc following the completion of the Business Combination. Unless the context otherwise requires, references to the “Company,” “Alps,” “we”, “us” and “our” in this subsection “— Risks Related to Alps’ Business and Industry and the Combined Company” generally refer to Alps Life Sciences Inc, Alps Global Holding Berhad and its subsidiaries as a whole. Alps Global Holding Berhad in the present tense and from and after the Business Combination. Where applicable, specific references to a company will refer to such company only.
Risks Related to Our Limited Operating History and Financial Condition
Alps Life Sciences Inc is a recently formed holding company with no operating history. It expects to operate through Alps Global Holding Berhad which, in turn, is a holding company of several operating subsidiaries. Alps Global Holding Berhad has a limited operating history since its inception which may make it difficult to evaluate its current business and this makes predictions about its future success or viability subject to significant uncertainty. Alps Global Holding Berhad is currently not a subsidiary of Alps Life Sciences Inc but is in the process of becoming a subsidiary of Alps Life Sciences Inc.
Alps Life Science Inc is a holding company formed for the purpose of the Business Combination. As such it has no operating history and its sole business will be the business of Alps Global Holding Berhad. As of the date of this registration statement, Alps Global Holding Berhad has not completed the process of becoming a subsidiary of Alps Life Science Inc. We expect this process to be completed in the near future. All risk factors relating to Alps’ operations relate to the business currently conducted by Alps Global Holding Berhad.
As a biotechnology entity with its pipeline of products all in the preclinical stage, combined with its limited operational history, determining the status and future trajectory of its business involves inherent uncertainties. Alps’ activities have been focused on organizational establishment, strategic planning, fundraising, intellectual property development, identifying new therapeutic targets, and advancing product candidates through various stages of research and development. Alps’ pipeline includes biologics and biopharmaceuticals, none of which have obtained regulatory approval.
To date, all of Alps’ product candidates in the pipeline are in the pre-clinical stage and have not proceeded to clinical trials.
As a result, Alps’ ability to successfully undertake and complete pivotal clinical trials, secure marketing approvals, scale up manufacturing, or engage in effective sales and marketing for product commercialization remains unproven. Despite Alps’ significant investments in research, development, and operations, forecasting our future with any degree of certainty is more challenging than it would be as compared to a company with a more extensive operational history or closer proximity to commercialization.
| 52 |
Moreover, Alps expects to face typical challenges and risks common to early-stage commercial ventures, including those related to organizational growth and the strategic prioritization of commercial, research, and business development initiatives. Alps is also likely to face unexpected expenses and obstacles that could adversely impact its business. Failure to effectively mitigate these risks could adversely impact our business.
Alps will need to obtain substantial additional funding to complete the development of its product candidates.
In addition to incurring substantial costs related to operating as a publicly listed company, Alps will require significant additional funds to further the development of its product candidates. However, Alps cannot assure that it will have adequate funds available in the future to further develop and bring to market its existing or potential future product candidates and technologies.
The development of biotechnology product candidates is inherently risky, capital-intensive and the unpredictable nature of candidate success. There is a considerable risk that our product candidates might not demonstrate the requisite efficacy or safety, achieve regulatory approval, or become commercially viable. Currently, University Science Malaysia (“USM”) has received external funding from the Ministry of Higher Education, Malaysia, and the Ministry of Science, Technology and Innovation, Malaysia, among others to develop a cholera vaccine. This cholera vaccine is being jointly developed by USM and our affiliate, Vax Biotech Sdn. Bhd., (“VaxBio”). As our product candidates progress through preclinical and clinical evaluations, we anticipate the need for significant additional funding to enhance our clinical, regulatory, quality, and manufacturing capabilities. Moreover, obtaining marketing approval for any of our candidates is likely to involve considerable expenses related to marketing, sales, manufacturing, and distribution.
With the successful completion of the Business Combination, any PIPE financing, combined with Alps’ existing resources, will be allocated to support the development of its product candidates, including MyImmune, MyCelest, CAR-T cells, iPSC, mRNA vaccine platform, and Celesome(+) as well as for other research and development activities, working capital, and general corporate purposes. While we believe our existing resources and proceeds from the PIPE financing will be sufficient for at least the next twelve (12) months, our assumptions may prove incorrect. We may need additional funding sooner than expected, necessitating public or private financing or strategic collaborations. However, obtaining additional financing may result in dilution to our stockholders or impose burdensome debt covenants and repayment obligations. Our future capital requirements and the timing and amount of our operating expenditures will depend largely on:
| ● | the timing and progress of preclinical and clinical development of our current and potential future product candidates; |
| ● | the number and scope of preclinical and clinical programs we decide to pursue; |
| ● | our ability to maintain our current licenses and collaborations, conduct our research and development programs and establish new strategic partnerships and collaborations; |
| ● | the progress of the development efforts of our existing strategic partners and third parties with whom we may in the future enter into collaboration and research and development agreements; |
| ● | the costs and timing of preparing, filing and prosecuting patent applications, maintaining and enforcing our intellectual property rights and defending any intellectual property-related claims, including any claims by third parties that we are infringing upon their intellectual property rights; |
| ● | our ability to establish and maintain strategic collaborations, licensing or other arrangements and the financial position, terms of any such agreements, including the timing and amount of any future milestone, royalty or other payments due under any such agreement; |
| ● | the cost and timing of regulatory approvals; and |
| ● | our efforts to enhance operational systems and to hire and retain personnel, including personnel to support development of our product candidates, the operation of our manufacturing facility, and to satisfy our obligations as a public company. |
| 53 |
The availability of additional funding on terms favorable to us is not guaranteed. Should we fail to secure the necessary capital in adequate amounts or under acceptable terms, we might be compelled to reduce our workforce significantly, delay, downscale, or cease the development or commercialization of our product candidates, or explore less desirable collaborations for our product candidates. Such scenarios could severely impact our business prospects, financial health, operational results, and lead to a decline in PubCo’s share price after the Business Combination.
Alps has identified a material weakness in its internal control over financial reporting and may identify additional material weaknesses in the future that may cause it to fail to meet its reporting obligations or result in material misstatements in its financial statements. If Alps fails to remediate its material weakness, Alps may not be able to report its financial results accurately or to prevent fraud.
Alps currently operates without a Chief Financial Officer (“CFO”) and is in the process of seeking suitable candidates to serve as a CFO after the completion of the Business Combination. The absence of a CFO may impede its ability to effectively manage and oversee its financial operations, including financial reporting, budgeting, and internal controls. The lack of a designated CFO may result in delays or errors in financial reporting, which could lead to non-compliance with regulatory requirements and investor uncertainty. Additionally, the absence of a CFO may hinder our ability to access capital markets, negotiate financing agreements, and maintain effective communication with investors and stakeholders. As a result, our financial management and strategic decision-making processes may be adversely affected, which could harm its business, financial condition, and results of operations.
Alps’ management bears the responsibility for establishing and maintaining internal control over financial reporting, disclosure controls, and compliance with applicable laws. Internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements in accordance with international financial reporting standards. A material weakness is defined as a deficiency, or a combination of deficiencies, in internal control over financial reporting, such that there is a reasonable possibility that a material misstatement of a company’s annual or interim financial statements will not be prevented or detected by the company’s internal controls on a timely basis.
Alps and its auditors identified a material weakness primarily related to (i) a lack of sufficient accounting and supervisory personnel who have the appropriate level of technical accounting experience and training, (ii) a lack of supervision over external consultants providing technical accounting services and (iii) a lack of consistent application of accounting processes and procedures by Alps’ accounting personnel. These deficiencies constitute a material weakness in Alps’ internal control over financial reporting in both design and operation. As a result of the material weakness, management failed to identify audit adjustments in various areas, including but not limited to revenue, capitalization of tangible and intangible assets, and share-based compensation. Alps has relied on the assistance of outside advisors with expertise in these matters to prepare its financial statements and comply with SEC reporting obligations, and Alps expects to continue to do so while it remediates this material weakness.
Alps is continuing to develop and implement a remediation plan to address the material weakness; however, Alps’ overall control environment still requires enhancement and may expose it to errors, losses or fraud. Alps’ remediation plan includes the hiring of additional suitably qualified staff. Additionally, Alps intends to document and implement consistent accounting policies and procedures and provide additional training to its accounting and finance staff. While Alps is working to remediate the material weakness as quickly and efficiently as possible, Alps cannot at this time provide an estimate of the costs it expects to incur or the expected timeline in connection with implementing its remediation plan. These remediation measures may be time-consuming and costly, and might place significant demands on its financial and operational resources. If Alps is unable to successfully remediate this material weakness or successfully supervise and rely on outside advisors with expertise in these matters to assist in the preparation of its financial statements, Alps’ financial statements could contain material misstatements that, when discovered in the future, could cause it to fail to meet its future reporting obligations.
| 54 |
Risks Related to Development of Our Product Candidates
The selection and prioritization of our product candidates for development are subject to change based on various factors. Abandoning a product candidate’s development or changing our development priorities could leave us without a viable replacement, impacting our business prospects.
Alps may determine to abandon the development of one or more of Alps’ product candidates, or we may change the prioritization of the development of certain product candidates, or Alps may select or acquire and prioritize the development of new product candidates. Alps’ choice and prioritization of product candidates for development will be influenced by a variety of factors, including but not limited to:
| ● | Alps may have limited capital to finance its development programs and projected costs may affect its ability to enter into licensing or collaborative arrangements with other biotechnology or biopharma companies or universities with their own laboratory facilities and research staffs to conduct research and development of one or more product candidates; |
| ● | competitors may develop alternatives that render Alps’ potential product candidates obsolete or less attractive; |
| ● | potential product candidates that Alps develop may nevertheless be covered by third parties’ patents or other exclusive rights; |
| ● | potential product candidates may not be effective in treating their targeted diseases; |
| ● | potential product candidates may, on further study, be shown to have harmful side effects, toxicities or other characteristics that indicate that they are unlikely to be products that will receive marketing approval and achieve market acceptance; |
| ● | Alps’ analysis of market demand and market prices for the products we plan to develop could lead us to conclude that market conditions are not favorable for receiving an adequate return on Alps’ investment in product development and commercialization; |
| ● | a potential product candidate may not be capable of being produced in commercial quantities at an acceptable cost, or at all; or |
| ● | the regulatory pathway for a potential product candidate is too complex and difficult to successfully navigate programs. |
Alps faces challenges in developing its manufacturing capabilities and currently relies may rely on third-party manufacturers, exposing it to various risks including lack of accessibility, quality control, quantity limitations, manufacturing delays, contractual breaches or terminations.
The manufacturing of biotechnology product candidates is complex, requiring significant expertise and capital, including the development of advanced manufacturing facilities, which we currently lack. Unless Alps we can finance and develop its own our manufacturing facilities, we will need to depend on third-party manufacturers, with no guarantee of favorable terms or availability. Whether Alps manufactures products or rely relies on third parties, it faces risks including manufacturing delays, inability to meet quality and quantity requirements, regulatory compliance challenges, and potential contract breaches or terminations. Regardless of whether Alps manufactures or relies on third parties to manufacture products for Alps, Alps we will face all risks related to the manufacture of therapeutic products for use in medicine including the following risks:
| ● | Alps or any third-party manufacturers might be unable to timely formulate and manufacture Alps’ products or produce the quantity and quality required to meet Alps’ clinical and commercial needs, if any; |
| ● | Alps or any third-party manufacturers may not be able to execute Alps’ manufacturing procedures appropriately; |
| 55 |
| ● | Any third-party manufacturers Alps engage may not perform as agreed or may not remain in the contract manufacturing business for the time required to supply Alps’ clinical trials or to successfully produce, store and distribute Alps’ products on a commercial scale; |
| ● | Alps or any third-party manufacturers will be subject to ongoing periodic unannounced inspection by the NPRA or other regulatory body, and corresponding state agencies to ensure strict compliance with cGMP and other government regulations and corresponding foreign standards. Alps will not have control over third-party manufacturers’ compliance with applicable regulations and standards; |
| ● | Alps may not own, or may have to share, the intellectual property rights to any improvements made by Alps’ third-party manufacturers in the manufacturing process for Alps’ product candidates; |
| ● | Third-party manufacturers could breach or terminate their agreements with Alps; and/or |
| ● | Alps or third-party manufacturers may experience manufacturing difficulties due to resource constraints or as a result of labor disputes or unstable political environments. |
In addition, Alps in the future may rely on third parties to perform release testing on Alps’ product candidates prior to delivery to patients. If these tests are not appropriately conducted and test data are not reliable, patients could be put at risk of serious harm which could result in product liability suits.
If Alps or any third-party manufacturers that Alps may engage were to encounter any of these difficulties, Alps’ ability to provide Alps’ product candidates to patients in clinical trials or to the medical marketplace would be jeopardized. Any delay or interruption in the supply of clinical trial supplies could delay the completion of clinical trials, increase the costs associated with maintaining clinical trial programs and, depending upon the period of delay, could require Alps to either commence new clinical trials at additional expense or terminate clinical trials completely.
The regulatory authorities also may, at any time following approval of a product for sale, audit Alps’ manufacturing facilities or those of Alps’ third-party contractors. If any such inspection or audit identifies a failure to comply with applicable regulations or if a violation of Alps’ product specifications or applicable regulations occurs independent of such an inspection or audit, Alps or the relevant regulatory authority may require remedial measures that may be costly and/or time-consuming for Alps or a third party to implement and that may include the temporary or permanent suspension of a clinical study or commercial sales or the temporary or permanent closure of a facility. Any such remedial measures imposed upon Alps or third parties with whom we contract could materially harm Alps’ business.
Additionally, if supply from one approved manufacturer is interrupted, there could be a significant disruption in commercial supply. The regulatory agencies may also require additional studies if a new manufacturer is relied upon for commercial production. Switching manufacturers may involve substantial costs and is likely to result in a delay in Alps’ desired clinical and commercial timelines.
Alps may not be able to manufacture product that meets release criteria due to sterility, identity or potency issues. Alps may not have access or be able to make the reagents necessary to manufacture the cells and Alps may not have access to adequate supply channels to transport and distribute the products. There are also risks that the cells may be destroyed by interruption in their cryopreservation by means of natural disasters such as earthquakes, power outages, or other unexpected events, or the cells may be determined to be unacceptable as a source of human cellular therapies for reasons Alps cannot envision. Alps cannot assure you that any stability or other issues relating to the manufacture of any of Alps’ product candidates or products will not occur in the future. If any of Alps’ master cell banks are lost or destroyed, including due to systems failure, Alps’ planned clinical trials would be severely delayed, and Alps would incur significant costs associated with obtaining new supply of cell banks. Accordingly, failures or difficulties faced at any level of Alps’ supply chain could adversely affect Alps’ business and delay or impede the development and commercialization of any of Alps’ product candidates or products and could have an adverse effect on Alps’ business, prospects, financial condition and results of operations.
| 56 |
There are a limited number of manufacturers that operate under cGMP regulations and that might be capable of manufacturing cell-based products for Alps. Accordingly, Alps’ cell-based products may have to compete with other cell-based products to access these manufacturing facilities. Future therapies that Alps may develop may compete with other product candidates and products for access to manufacturing facilities. Any performance failure on the part of Alps’ existing or future manufacturers could delay clinical development or marketing approval.
Each of these risks could delay Alps’ clinical trials, any regulatory approval of Alps’ product candidates, or the commercialization of Alps’ product candidates, and could result in higher costs or deprive Alps of potential product revenue.
Our current product candidates are in preclinical development and have never been tested in humans. One or all of our current product candidates may fail in clinical development or suffer delays that materially and adversely affect their commercial viability.
To date, none of our candidates has proceeded to clinical trials or been tested in humans. Our future profitability hinges on our ability to obtain regulatory approvals and successfully commercialize our product candidates, either independently or with partners.
Achieving commercial distribution for our product candidates requires extensive preclinical studies and clinical trials to prove their safety, purity, and efficacy in humans. Although we have observed positive outcomes observed in preclinical animal models for treatment of heart failure using iPSC-derived cardiomyocytes and the joint development of the oral Cholera vaccine VCUSM14P, these findings may not necessarily translate to similar success in future human clinical trials. There’s a risk that our product candidates may not exhibit the desired safety and efficacy in later stages of clinical development, even if they show promise in early trials. It is uncertain whether NPRA will authorize us to conduct clinical trials. Moreover, the completion and outcomes of our preclinical studies are unpredictable, and it is uncertain whether the NPRA or other regulatory bodies will approve our clinical programs, protocols, or support the further development of our preclinical programs for human testing.
Further, some or all of Alps’ product candidates under development may require the genetic modification of the pluripotent master cell banks. There is no certainty that a genetic modification will provide a long-term solution to transplant rejection, or that the modified cells will not cause unanticipated health risks to the patient that could delay or even halt the development of the products.
The preclinical development stage exposes us to inherent risks of failure associated with novel therapeutic approaches, targets, and action mechanisms. While we plan to initiate clinical trials for our leading candidates, there’s no assurance of progressing to clinical development for any candidate or of demonstrating clinical benefits in patient testing. Considering the costs, uncertainties, delays, and challenges typical for preclinical stage biotechnology company like us, our prospects should be evaluated accordingly. For example, the joint development of oral Cholera vaccine cannot be carried out in Malaysia, due to the relatively low incidence of cholera compared to endemic regions. The limited data available would hinder the accurate assessment of the cholera vaccine’s impact. Additionally, the low cholera burden in Malaysia makes it impractical and inefficient to conduct large-scale clinical trials and secure the necessary approvals.
We may not be able to access the financial resources to continue development of, or to enter into any collaborations for, any of our current or potential future product candidates. This may be exacerbated if we experience any issues that delay or prevent regulatory approval of, or our ability to commercialize, a product candidate, such as:
| ● | negative or inconclusive results from our preclinical studies or clinical trials or the clinical trials of others for product candidates similar to ours, leading to a decision or requirement to conduct additional preclinical studies or clinical trials or abandon any or all of our programs; |
| ● | adverse events experienced by participants in our clinical trials or by individuals using therapeutics similar to our product candidates; |
| ● | delays in submitting CTIL, CTX or comparable foreign applications, or delays or failures to obtain the necessary approvals from regulatory authorities to commence a clinical trial, or a suspension or termination of a clinical trial once commenced; |
| ● | conditions imposed by the NPRA or other regulatory authorities regarding the scope or design of our clinical trials; |
| 57 |
| ● | delays in enrolling research subjects in clinical trials; |
| ● | high drop-out rates of research subjects; |
| ● | inadequate supply or quality of product candidate components or materials or other supplies necessary for the conduct of our clinical trials; |
| ● | greater-than-anticipated clinical trial costs; |
| ● | poor potency or effectiveness of our product candidates during clinical trials; |
| ● | unfavorable NPRA or other regulatory authority inspection and review of a clinical trial or manufacturing site; |
| ● | delays as a result of the pandemic or other force majeure events or other associated events; |
| ● | failure of our third-party contractors or investigators to comply with regulatory requirements or otherwise meet their contractual obligations in a timely manner, or at all; or |
| ● | delays and changes in regulatory requirements, policies and guidelines. |
Clinical trials are expensive, time-consuming and difficult to design and implement. There can be no assurance that our product candidates will be able to successfully complete clinical trials. The failure of our product candidates to complete clinical trials may have a material adverse effect on our business, financial condition, results of operations and prospects.
Human clinical trials are expensive and difficult to design and implement, in part because they are subject to rigorous regulatory requirements. Because our current and potential future product candidates are based on new technologies and discovery approaches, we expect that they will require extensive research and development and have substantial manufacturing and processing costs. In addition, the NPRA or other regulatory authorities may require us to perform additional testing before commencing clinical trials and may be hesitant to allow us to enroll patients impacted with our targeted disease indications in our future clinical trials. If we are unable to enroll patients impacted by our targeted disease indications in our future clinical trials, we would be delayed in obtaining potential POC data in humans, which could extend our development timelines. In addition, costs to treat patients and to treat potential side effects that may result from our product candidates may be significant. Accordingly, our clinical trial costs are likely to be high and there is no assurance that our product candidates will successfully complete clinical trials. A failure to complete clinical trials could have a material adverse effect on our business, financial condition, results of operations and prospects.
If we encounter difficulties enrolling patients in clinical trials, our clinical development activities could be delayed or otherwise adversely affected.
The timely completion of clinical trials in accordance with their protocols depends, among other things, on our ability to enroll a sufficient number of patients who remain in the study until its conclusion. We may encounter delays in enrolling, or be unable to enroll, a sufficient number of patients to complete any of clinical trials of our product candidates, and even once enrolled we may be unable to retain a sufficient number of patients to complete the trials. The enrollment of patients depends on many factors, including:
| ● | the patient eligibility criteria defined in the protocol; | |
| ● | the size of the patient population required for analysis of the trial’s primary endpoints; | |
| ● | the proximity of patients to study sites; | |
| ● | the design of the trial; | |
| ● | our ability to recruit clinical trial investigators with the appropriate competencies and experience; |
| 58 |
| ● | clinicians’ and patients’ perceptions as to the potential advantages of the product candidate being studied in relation to other available therapies, including any new products that may be approved for the indications Alps is investigating; | |
| ● | our ability to obtain and maintain patient consents; and | |
| ● | the risk that patients enrolled in clinical trials will drop out of the trials before completion. |
In addition, clinical trials of our product candidates may compete with other clinical trials for product candidates of other companies that are in the same therapeutic areas as our product candidates, and this competition will reduce the number and types of patients available to us, because some patients who might have opted to enroll in trials of our product candidates may instead opt to enroll in a trial being conducted by one of our competitors. Since the number of qualified clinical investigators is limited, we expect that clinical trials or our product candidates may be conducted at the same clinical trial sites that some of our competitors use, which will reduce the number of patients who are available for our clinical trials in such clinical trial site.
Delays or failures in planned patient enrollment or retention may result in increased costs, program delays or both, which could have a harmful effect on our ability to develop our product candidates or could render further development impossible.
Compassionate use of stem cells therapies and/or other medical treatments and procedures provided by Alps may subject us to medical malpractices, result in injuries that lead to costly liability suits, and/or subject us to penalties if we fail to comply with regulatory requirements or experience unanticipated problems with any products and/or product candidates.
We have facilitated compassionate use of stem cells therapies in Malaysia, in accordance with the applicable laws and regulations established by the Malaysian regulatory authorities.
In Malaysia, the Control of Drugs and Cosmetics Regulations 1984, under the Sale of Drugs Act 1952, allows hospitals or physicians to apply for an exemption from license registration for importing or manufacturing products solely for treating patients with life-threatening conditions, as per Regulation 15(6). This regulatory pathway enables the compassionate use or named patient program, extending to patients already part of an approved clinical trial. The application criteria for such programs are stringent, necessitating the treatment to be for patients facing serious or immediate life-threatening conditions, with no alternative therapies available, among other considerations. While Alps strives to ensure its applications for compassionate use align with the requirements established by the Malaysian authorities, there remains a risk that regulatory perspectives may diverge from Alps’ interpretations. If Alps’ determination does not align with regulatory perspective and Alps becomes required to make compassionate use applications, Alps’ results of operations and project development timeline may be adversely affected.
The investigational nature of Alps’ products under compassionate use implies that their safety and efficacy profiles are not fully established, typically warranting their use within clinical trial settings. Post-trial access to investigational products is facilitated through the compassionate use program in Malaysia, which is strictly limited to participants of approved clinical trials involving CTIL and CTX, on a named patient basis. Our physicians are responsible for off-label compassionate use of our product candidates which requires comprehensive patient consent prior to treatment initiation.
Given these conditions, there is an inherent risk of future legal liabilities or damages stemming from the off-label compassionate use of our product candidates. Should adverse outcomes arise from such use, or if regulatory authorities interpret our compliance with compassionate use provisions differently, we may face lawsuits that could significantly impact our operations and financial standing. The legal and regulatory landscape for compassionate use is complex and subject to change, and our engagement in these programs, despite rigorous adherence to existing guidelines, may expose us to unforeseen liabilities. Our assumptions regarding the use of investigational products are based on the current interpretation of applicable regulations. However, if Alps fails to comply with the evolving legal and regulatory framework governing cell and gene therapy products, the Company may face regulatory scrutiny, investigations, fines, or other penalties, which could adversely affect our business, revenue projections, operations, and financial condition.
In addition, Alps’ provision of medical treatments and procedures entails inherent risks associated with medical malpractice, despite rigorous adherence to industry standards and best practices. There is a risk that patients may experience adverse outcomes, complications, or dissatisfaction with the provided medical care, leading to potential claims of medical malpractice. Such claims could arise from allegations of negligence, errors in diagnosis or treatment, failure to obtain informed consent, or breaches of duty of care. While Alps endeavors to maintain high standards of medical care and employs qualified healthcare professionals, there remains the possibility of human error, unforeseen complications, or adverse reactions to treatments. Moreover, factors beyond Alps’ control, such as patient-specific conditions, medical histories, or external environmental factors, may contribute to adverse outcomes or perceived medical malpractice.
| 59 |
Alps maintains medical malpractice insurance coverage to mitigate the financial risks associated with potential medical malpractice claims. However, there can be no guarantee that such insurance coverage will be adequate to fully indemnify Alps against all potential liabilities arising from medical malpractice claims. Such claim whether meritorious or not, could result in significant legal expenses, damage to reputation, and financial liabilities for Alps. Furthermore, adverse publicity stemming from medical malpractice claims may impact patient confidence, referral patterns, and overall business operations.
The commercial success of any of Alps’ current or future product candidates will depend upon the degree of market acceptance by physicians, patients, third-party payors, and others in the medical community.
Even with approvals from the NPRA and comparable foreign regulatory authorities such as the FDA, the commercial success of Alps’ products will depend in part on the health care providers, patients, insurers, and third-party payors accepting Alps’ product candidates as medically useful, cost-effective, and safe. Any product that Alps bring to the market may not gain market acceptance by physicians, patients, insurers, third-party payors and other health care providers. The clinical development, commercialization, and marketing of our product candidates are at an early-stage, substantially research-oriented, and financially speculative. It is important to recognize that developing and bringing cell therapies to market is a challenging endeavor, with only a few companies achieving success thus far. Cell therapies, in general, carry inherent risks, including potential side effects, immune system responses, limited therapeutic efficacy, and high costs, which may hinder their regulatory approval or commercial adoption. Alps’ success hinges on several factors, including the establishment of a robust global market for cell therapies and its ability to carve out a competitive position within this evolving landscape with its product candidates.
Even if Alps, a collaborator, or a licensee of Alps’ technology successfully develop and obtain regulatory approval for Alps’ product candidates, the market may not understand or accept them. Alps’ product candidates represent novel treatments and are expected to compete with a number of more conventional products and therapies manufactured and marketed by others, including major pharmaceutical and biotechnology companies. The degree of market acceptance of any of Alps’ products will depend on a number of factors, including without limitation:
| ● | the efficacy of the product as demonstrated in clinical studies and potential advantages over competing treatments; | |
| ● | the prevalence and severity of the disease and any side effects; | |
| ● | the clinical indications for which approval is granted, including any limitations or warnings contained in a product’s approved labeling; | |
| ● | the convenience and ease of administration; | |
| ● | the cost of treatment, particular if compared to existing treatments; | |
| ● | the willingness of the patients and physicians to accept and use these therapies and the perception of efficacy and safety of Alps’ approved products by such parties; | |
| ● | the marketing, sales and distribution support for the products; | |
| ● | the publicity and ethical, social and legal concerns regarding the use of embryonic stem cells for Alps’ products or competing products and treatments; | |
| ● | government regulations restricting or prohibiting Alps’ research or manufacturing processes for stem cells due to ethical, social and legal concerns regarding their use in medical research and treatment; and | |
| ● | the pricing and availability of third-party insurance coverage and reimbursement. |
Even if a product displays a favorable efficacy and safety profile upon approval, market acceptance of the product will initially remain uncertain. Efforts to educate the medical community and third-party payors on the benefits of the products may require significant investment and resources and may never be successful. If Alps’ products fail to achieve an adequate level of acceptance by physicians, patients, third-party payors, and other health care providers, Alps will not be able to generate sufficient revenue to become or remain profitable.
| 60 |
If the use or misuse of Alps’ product candidates harm patients or is perceived to harm patients even when such harm is unrelated to Alps’ product candidates, Alps’ regulatory approvals could be revoked or could otherwise be negatively impacted, and Alps could be subject to costly and damaging product liability claims.
The use or misuse of any product candidates in future clinical trials and the sale of any products for which Alps obtain marketing approval exposes Alps to the risk of product liability claims. Product liability claims might be brought against Alps by consumers, healthcare providers, pharmaceutical companies, or others selling or otherwise coming into contact with Alps’ products. There is a risk that Alps’ product candidates may induce adverse events. If Alps cannot successfully defend against product liability claims, Alps could incur substantial liability and costs. In addition, regardless of merit or eventual outcome, product liability claims may result in:
| ● | impairment of Alps’ business reputation; | |
| ● | initiation of investigations by regulators; | |
| ● | withdrawal of clinical trial participants; | |
| ● | costs due to related litigation; | |
| ● | distraction of management’s attention from Alps’ primary business; | |
| ● | substantial monetary awards to patients or other claimants; | |
| ● | the inability to commercialize Alps’ product candidates; | |
| ● | product recalls, withdrawals or labeling, and marketing or promotional restrictions; | |
| ● | loss of revenue; and | |
| ● | decreased demand for Alps’ product candidates, if approved for commercial sale. |
Alps may not be able to maintain insurance coverage at a reasonable cost or in sufficient amounts to protect Alps against losses due to liability. If and when Alps commence clinical trials or obtain regulatory and marketing approval for any product candidates, Alps intends to increase Alps’ insurance coverage to include clinical use or the sale of commercial products, as applicable; however, Alps may be unable to obtain product liability insurance on commercially reasonable terms or in adequate amounts. On occasion, large judgments have been awarded in lawsuits based on drugs or medical treatments that had unanticipated adverse effects. A successful product liability claim or series of claims brought against Alps could cause Alps’ stock price to decline and, if judgments exceed Alps’ insurance coverage or if the insurer refuses to cover and insure the claim, such factors could adversely affect Alps’ results of operations and business.
Alps’ business entails a significant risk of product liability, and its inability to obtain sufficient insurance coverage could have a material effect on its business, financial conditions, results of operations and prospects.
Alps does not currently have insurance coverage for all potential risks inherent in the business operations of its subsidiaries.
As Alps conducts preclinical studies and future clinical trials for its product candidates, it exposes itself to inherent risks associated with product development, testing, manufacturing, and marketing. Despite any future efforts to secure additional product liability insurance, there is no guarantee it will fully reimburse Alps for potential expenses or losses incurred from liability claims. If Alps obtains marketing approval for any of its product candidates, such claims could trigger regulatory investigations into the safety and effectiveness of the products, manufacturing processes, and facilities, or the marketing programs. This could potentially result in product recalls or more serious enforcement actions, limitations on approved indications, or even the suspension or withdrawal of approvals. A successful product liability claim or series of claims against Alps could cause a decline in its share price. If judgments exceed Alps’ insurance coverage, it could adversely affect its results of operations and business, including preventing or limiting the commercialization of any product candidates it develops.
Moreover, insurance coverage is becoming increasingly expensive, and in the future, Alps or any future collaborators may struggle to maintain adequate coverage at a reasonable cost or in sufficient amounts to protect against liability losses.
| 61 |
Any cell-based products that receive regulatory approval may be difficult and expensive to manufacture on a commercial scale.
Producing cell-based therapeutic products derived from pluripotent stem cells and mesenchymal cells for commercial use presents substantial challenges, as these cells have traditionally been generated only on a small scale. This scale is not sufficient for the mass commercialization required for widespread market penetration. Should Alps succeed in advancing cell-derived therapeutic products to the market, significant advancements in manufacturing facilities, processes, and technologies will be essential for commercial-level production. Given the complex nature of pluripotent stem cell and mesenchymal cell products, manufacturing them on a commercial scale is anticipated to be more costly compared to most existing drugs. This increased manufacturing cost necessitates setting higher prices for these products to cover costs and generate profit. However, if the pricing makes these products prohibitively expensive, healthcare institutions and professionals might hesitate to adopt them, potentially impacting the volume of sales needed for Alps to recover development costs and achieve profitability.
The manufacturing operations of some of our potential product candidates including but not limited to cosmetics products infused with exosomes are themselves dependent upon third-party suppliers, making us potentially vulnerable to supply shortages and price fluctuations, which could harm our business.
We are currently at the stage where we are able to derive exosomes from human Umbilical Cord Mesenchymal Stem Cells (hUMSCs) with our in-house facilities at MYCELEST. We are in the process of exploring the development of exosome infused products and have not developed or manufactured exosome infused products. At this time, we are not capable of manufacturing the finished cosmetics products such as facial masks and face serum infused with exosomes in-house. Accordingly, we expect to rely on third party manufacturers to produce any exosome infused products that we develop in the future. Such future reliance on third party manufacturers or suppliers could potentially expose us to risks that could harm our business, including:
| ● | interruption of supply resulting from modifications to or discontinuation of our products; | |
| ● | delays in product shipments resulting from uncorrected defects, reliability issues, or variation in a component; | |
| ● | a lack of long-term supply agreements; | |
| ● | inability to obtain adequate supply in a timely manner, or to obtain adequate supply on commercially reasonable terms; | |
| ● | difficulty and cost associated with locating and qualifying alternative suppliers in a timely manner; | |
| ● | production delays related to the evaluation and testing from alternative suppliers, and corresponding regulatory qualifications; and | |
| ● | damage to our brand reputation. |
Any interruption in the supply, or our inability to obtain substitute from alternate sources at acceptable prices in a timely manner, could harm our ability to perform procedures, fulfil customer orders and satisfy market demand until new sources of supply are identified and qualified. As of the date of this proxy statement/prospectus, we have not entered into any contracts with third parties for the manufacture of finished cosmetic products.
Alps’ inability to distinguish the product candidates from other similar treatment or solutions may restrict market acceptance of its products and market share.
While our line of product candidates represents a relatively new entry into the Southeast Asia market, it may not possess striking novelty against its predecessors. Alps’ future success will depend on our ability to increase demand for its products by demonstrating to a broad spectrum of healthcare providers the potential performance advantages and cost-effectiveness of our products, and our inability to do so could have a material adverse effect on the business, financial condition, and results of operations. As a result, we generally are required to invest a significant amount of time and resources to educate healthcare administrators and other purchasers about the benefits of a product in comparison to competing products and technologies before completing a sale, if any.
Several factors could hinder healthcare professionals’ adoption of our product candidates, including doubts about product efficacy, costs and reliability. Healthcare administrators considering our technology must allocate time to grasp the innovation, gauge physician receptiveness, evaluate the financial implications for their practice, and become adept at introducing and utilizing our products. Without a pressing competitive need, administrators may not prioritize learning about our products’ potential advantages. Acceptance and adoption by healthcare professionals may be contingent on additional clinical evidence supporting our products’ safety and effectiveness or endorsements from respected figures within the healthcare community. Moreover, economic pressures, such as downturns, shifts in healthcare reimbursement policies, or market-specific competitive challenges, might deter healthcare organizations from investing in significant capital equipment or new technologies.
| 62 |
Risks Related to Our Intellectual Property
Alps relies on patents and intellectual property rights licensed from third parties to engage in research, development and commercialization activities. If we fail to maintain these licenses on terms favorable or otherwise acceptable to Alps, Alps’ ability to engage in research, development and commercialization activities may be materially impacted, resulting in a materially adverse effect on our business, financial condition and operational results.
Alps, its affiliate and/or subsidiaries holds licenses granting them rights to third-party intellectual property that is necessary or useful to its business. In particular, Alps has obtained licenses from certain individuals for intellectual property concerning stem cell and/or exosome processing technologies, and VaxBio has granted a patent license covering protein mucosal delivery system method for use in developing cholera vaccine. Alps may enter into additional licenses to third-party intellectual property in the future.
Alps’ licenses to such intellectual property rights may not provide exclusive or unrestricted rights in all fields of use and in all territories in which Alps may wish to develop or commercialize its products in the future and may restrict its rights to offer certain products in certain markets, including through non-compete provisions, or impose other obligations on Alps in exchange for its rights to the licensed intellectual property. In addition, Alps may not have full control over the maintenance, protection, enforcement or use of the intellectual property rights in-licensed from licensors, and therefore Alps may be reliant on the licensors to conduct such activities.
These in-licensed patents are critical to our research, development, and commercialization activities. Our success will depend in part on the ability of our licensors to obtain, maintain, and enforce patent protection for the licensed intellectual property. While these arrangements enable access to essential technology and intellectual property, they also pose several risks that could adversely affect our business, financial condition, and operational results, as follows:
| ● | Our dependence on licensed intellectual property creates a risk of uncertainty regarding the continued availability of these rights. Licenses may be subject to termination under certain conditions, renegotiation, or may not be renewable on favorable terms, or at all. The loss of a critical license could result in the discontinuation of key research projects or the inability to continue manufacturing or selling certain products, which would negatively impact our business operations and financial performance. | |
| ● | Licensed intellectual property rights often come with financial obligations, including upfront payments, milestone payments, royalties on sales, and maintenance fees. These financial commitments can be substantial and may adversely affect our profitability. Additionally, disputes over royalty calculations or other financial terms can lead to litigation or arbitration, further imposing financial and operational burdens on our company. | |
| ● | Licenses typically contain restrictions on our ability to sublicense or otherwise transfer our rights to third parties. These restrictions can limit our flexibility in developing and commercializing our technology and products, potentially restricting our ability to enter into partnerships or collaborations that could be beneficial to our business. | |
| ● | We may also be unaware of existing patents that may be infringed upon by their respective patent owners. Our rights to use licensed intellectual property may be challenged by third parties, including challenges to the validity, enforceability, or scope of the underlying patents. Such challenges could jeopardize our ability to continue utilizing essential technologies or force us into costly litigation to defend our rights to use the licensed intellectual property. | |
| ● | Our licensors may not successfully prosecute the patent applications we have licensed. Our business may be adversely affected if licensors fail to adequately maintain or enforce the intellectual property rights we have licensed. Our ability to develop and commercialize our products depends on the licensors’ diligence in prosecuting patent applications, maintaining issued patents, and defending intellectual property against infringement or invalidity claims. Their failure to do so could materially and adversely affect our business and prospects. |
Alps relies on in-licensed patents registered in China for research and development activities even though Alps operates its business from Malaysia. As such, there is a risk that these in-licensed patents will not constitute adequate protection and affect our ability to successfully develop and commercialize our products.
Alps relies on in-licensed patents from China to support its research, development, and commercialization activities. However, these patents provide protection solely within China and may not offer adequate protection in other region(s) where Alps operates. Currently, Alps operates and conducts all of its research and development activities in Malaysia. It also works with strategic research partners that are based in Malaysia and also expects to develop products for commercialization in Malaysia and other territories outside of China. As these in-licensed patents do not extend protection beyond China, Alps faces the possibility that competitors could develop similar or derivative products, potentially leading to market saturation and increased competition, impacting its ability to commercialize its own products successfully. In addition, there will always be a risk that competitors or other third parties could challenge the validity or enforceability of any patent issued or licensed or sublicensed to Alps, Alps’ ability to defend its intellectual property rights could be compromised. This may lead to delays or difficulties in bringing products to market, or even the inability to commercialize products entirely, particularly in regions where patent protection is absent or weak. Such challenges could significantly impact Alps’ long-term growth and ability to compete effectively in global markets.
If Alps, its subsidiaries, or associate companies fail to meet their obligations under license agreements, they risk losing the rights to critical technologies upon which the business depends.
Certain product candidates, such as the cholera vaccine and iPSC, rely on technologies that Alps, its subsidiaries, or associate companies have obtained through licensing agreements with external parties, including USM and USCI University. For example, the licensing and commercialization agreement between VaxBio, Alps’ affiliate, and USM, dated September 12, 2023, imposes various obligations on VaxBio, including payment obligations and commitments to pursue the development and commercialization of products and technologies. Alps, its subsidiaries, and/or associate companies may enter into additional license agreements in the future. Alps’ existing license agreements impose, and it expects that future license agreements will impose various obligations including diligence, milestone payment, royalty, and other requirements. If Alps, its subsidiary, or associate company fails to comply with their obligations under these agreements, or they are subject to a bankruptcy, the licensor may have the right to terminate the license, in which event we would not be able to market products covered by the license.
If Alps, its subsidiary, or associate company fails to comply with its obligations under the agreement, or if Alps becomes subject to winding up or liquidation, the licensor may have the right to terminate the license. In such an event, Alps would be unable to market products covered by the license.
| 63 |
In some cases, patent prosecution of our licensed technology is controlled solely by the licensor. If our licensors fail to obtain and maintain patent or other protection for the proprietary intellectual property we license from them, we could lose our rights to the intellectual property and our competitors could market competing products using the intellectual property.
In addition, the agreements under which Alps, its affiliate and/or subsidiaries currently licenses or otherwise obtains rights to intellectual property from third parties are complex, and certain provisions in such agreements may be susceptible to multiple interpretations, which may lead to disputes between Alps and its licensor, including:
| ● | the scope of rights granted under the license agreement; | |
| ● | the extent to which Alps’ product infringe on intellectual property of the licensor that is not subject to the license agreement; | |
| ● | the right to sublicense patent and other rights under Alps’ collaborative development relationships; | |
| ● | Alps’ diligence and other obligations under the license agreement; and | |
| ● | the ownership of inventions and know-how resulting from the joint invention of intellectual property by Alps and its licensors and/or collaborators. |
The resolution of any contract disagreement that may arise could narrow what Alps believes to be the scope of its rights to the relevant intellectual property, or increase what Alps believes to be its financial or other obligations under the relevant agreement, either of which could have a material adverse effect on its business, financial condition, results of operations, and prospects. If Alps is required to engage in litigation to enforce or defend its rights under its license agreements, even if it is successful, such litigation could require significant financial resources, divert the attention of management and harm Alps’ business. Moreover, if disputes over intellectual property that Alps has licensed or otherwise obtained rights to prevent or impair Alps’ ability to maintain its current arrangements on commercially acceptable terms, or at all, Alps may be unable to successfully commercialize the affected product, which could have a material adverse effect on Alps’ business, financial condition, results of operations, and prospects.
Alps relies on intellectual property that is jointly developed with third parties which exposes Alps to risks associated with the failure of joint owners to maintain, protect, and enforce such intellectual property and divergent interests arising during the course of joint ownership.
Alps engages in collaborations and partnerships with other entities, including universities. Through these collaborations, we may jointly develop intellectual property that is crucial to our product development and commercialization efforts. An example of this is our collaboration with USM. While these partnerships can be instrumental in advancing our research and development capabilities, they also introduce significant risks associated with the joint ownership of intellectual property, including:
| ● | Joint ownership of intellectual property rights can lead to complexities in the management and decision-making processes regarding the exploitation, protection, and enforcement of these rights. Divergent interests among co-owners may result in delays or failure to effectively prosecute patent applications, maintain issued patents, or defend against challenges to our intellectual property rights. Alps Biotech Sdn. Bhd. jointly owns intellectual property with USM through a Research Collaboration Agreement dated 1 September 2020 (“Research Collaboration Agreement”). The Research Collaboration Agreement has expired, and Alps is in discussions to extend or modify this collaboration relationship with USM. Until such time as Research Collaboration Agreement has been extended and depending upon the terms of such extension, Alps may face difficulties relating to the use, protection, and commercialization of the intellectual property jointly owned with USM. | |
| ● | Agreements involving joint ownership often require detailed provisions on the allocation of revenues derived from the commercialization of jointly owned intellectual property. Disputes over revenue sharing or the allocation of expenses associated with the maintenance and protection of intellectual property may arise, potentially leading to litigation or arbitration that could be costly and time-consuming. | |
| ● | Joint ownership arrangements typically include provisions that restrict our ability to unilaterally license or transfer our interest in jointly owned intellectual property. Such restrictions could limit our flexibility in responding to market opportunities or challenges, potentially affecting our competitive position and financial prospects. | |
| ● | Our reliance on co-owners for the maintenance, protection, and enforcement of jointly owned intellectual property rights may expose us to risks associated with their solvency, willingness, and ability to fulfill these responsibilities. We are currently in discussions with USM to extend or otherwise modify the Research Collaboration Agreement. If we are not successful in extending our collaboration relationship with USM, Alps’ ability to license, sell, or otherwise exploit the intellectual property that is jointly owned with USM may be delayed or restricted and USM’s willingness to cooperate with us to maintain, protect and enforce such jointly owned intellectual property could be negative affected, which could in turn adversely impact our business operations and financial condition. |
There is no certainty that Alps’ future patent applications will result in the successful registration of patents.
Alps may file additional new patent applications in the future seeking patent protection for new technology or products that Alps develops itself or jointly with others. However, there is no assurance that any of Alps’ licensed patent applications, or any patent applications that Alps may file in the future in the Malaysia or abroad, will result in the successful registration of such patents.
Navigating the patent application and maintenance process demands considerable investment and time, characterized by:
| ● | The preparation and filing of patent applications, and the maintenance of patents that are issued, may require substantial time and money. | |
| ● | A patent interference proceeding may be instituted with the Intellectual Property Corporation of Malaysia (“MyIPO”) when more than one person files a patent application covering the same technology, or if someone wishes to challenge the validity of an issued patent. At the completion of the interference proceeding, MyIPO will determine which competing applicant is entitled to the patent, or whether an issued patent is valid. Patent interference proceedings are complex, highly contested legal proceedings, and MyIPO’s decision is subject to appeal. This means that if an interference proceeding arises with respect to any of Alps’ patent applications, Alps may experience significant expenses and delay in obtaining a patent, and if the outcome of the proceeding is unfavorable to Alps, the patent could be issued to a competitor rather than to Alps. | |
| ● | A derivation proceeding may be instituted by MyIPO or an inventor alleging that a patent or application was derived from the work of another inventor. | |
| ● | Oppositions to the issuance of patents may be filed under United States Patent and Trademark Office (USPTO), European patent law and the patent laws of certain other countries. As with MyIPO interference proceedings, these foreign proceedings can be very expensive to contest and can result in significant delays in obtaining a patent or can result in a denial of a patent application. |
These hurdles underscore the unpredictable and costly nature of securing patent protection, essential for safeguarding Alps’ proprietary technologies and innovations.
| 64 |
Alps’ patents may not adequately protect its technologies or products from competition.
| ● | Alps may struggle to secure additional patents beyond those already owned or have licensed or sublicensed, and any patents obtained might not offer comprehensive protection. | |
| ● | There will always be a risk that Alps’ competitors might be able to challenge the validity or enforceability of any patent issued or licensed or sublicensed to Alps. | |
| ● | In addition to interference proceedings, regulatory bodies like MyIPO can reexaminations of issued patents at the request of a third party. Alps’ patents may be subject to inter partes review (replacing the reexamination proceeding), a proceeding in which a third party can challenge the validity of one of Alps’ patents to have the patent invalidated. This means that patents owned or licensed by Alps may be subject to reexamination and may be lost if the outcome of the reexamination is unfavorable to Alps. | |
| ● | Pursuant to the Licensing and Commercialization Agreement dated September 12, 2023 between USM and VaxBio, an affiliate company of Alps which Alps holds a 30% shareholding, VaxBio has acquired an exclusive license to utilize the technical information for research and development, manufacturing, sales, or other dealings with the licensed products, subject to the terms and conditions specified in the agreement. However, there are potential regulatory challenges or compliance issues related to the licensing and commercialization agreement between USM and VaxBio. Any regulatory scrutiny, disputes, or breaches of terms and conditions outlined in the agreement could impact Alps’ ability to leverage the patented technology effectively and may result in financial or reputational consequences. Additionally, if USM were to develop competing products, it could lead to market saturation or increased competition for Alps, impacting its ability to commercialize its own products successfully. |
Alps may face challenges in enforcing its intellectual property rights throughout the world, which could adversely affect its business.
The cost of filing, prosecuting, and defending patents globally for Alps’ technology and product candidates would be excessively high. Patentability requirements vary across countries, especially in developing regions. Competitors may exploit our technology in areas where we lack patent coverage to develop rival products. Subsequently, they could potentially export these products to markets where we hold patents but have weaker enforcement mechanisms than Malaysia. This situation poses a risk of direct competition in regions where we cannot adequately protect our intellectual property rights.
Additionally, unexpected changes in foreign intellectual property laws may undermine our protection efforts. Legal systems in certain countries provide less robust intellectual property protection than Malaysia, presenting significant challenges in defending our rights there. Countries such as India and China, along with other developing nations, have legal frameworks less conducive to strong patent enforcement. This disparity in legal environments could hamper our ability to address infringement or prevent the unauthorized use of our intellectual assets.
Moreover, there are numerous countries possess compulsory licensing regulations, which may require patent holders to provide licenses to third parties, further complicating our control over our inventions outside Malaysia. In addition, certain countries impose restrictions on the enforceability of patents against government entities or contractors, potentially limiting available remedies for patent owners. In such jurisdictions, patent owners may have restricted remedies, potentially reducing the value of their patents significantly. If Alps or any of its licensors are compelled to grant licenses to third parties for patents relevant to its operations, its competitive stance in the respective jurisdiction could be compromised, adversely affecting our business prospects.
Proceedings to enforce our patent rights in foreign jurisdictions, whether or not successful, could result in substantial costs and divert our efforts and resources from other aspects of Alps’ business. Furthermore, while Alps intend to protect our intellectual property rights in major markets, we cannot ensure that Alps will be able to initiate or maintain similar efforts in all jurisdictions in which Alps may wish to market products or license Alps’ patented technologies. Accordingly, Alps’ efforts to protect Alps’ intellectual property rights in such countries may be inadequate.
| 65 |
Alps may be subject to patent infringement claims that could be costly to defend, which may limit Alps’ ability to use disputed technologies, and which could prevent Alps from pursuing research and development or commercialization of some of Alps’ technologies or products, require Alps to pay licensing fees to have freedom to operate and/or result in monetary damages or other liability for Alps.
Alps’ operational success is critically dependent on our ability to avoid infringing on the patents and proprietary rights of others. If the technology that Alps uses infringes a patent held by others, Alps could be sued for monetary damages by the patent holder or its licensee, or Alps could be prevented from continuing research, development, and commercialization of technologies and products that rely on that technology, unless Alps is able to obtain a license to use the patent. The cost and availability of a license to a patent cannot be predicted, and the likelihood of obtaining a license at an acceptable cost would be low if the patent holder or any of its licensees is using the patent to develop or market a technology or product with which Alps’ technologies or products would compete. If Alps could not obtain a necessary license, Alps would need to develop or obtain rights to alternative technologies, which could prove costly and could cause delays in developing Alps’ technologies or products, or Alps could be forced to discontinue the development or marketing of any technologies and products that were developed using the technology covered by the patent.
Risk Related to Government Regulation
Upon the consummation of the Business Combination, PubCo expects to qualify as an “emerging growth company,” and may elect to comply with reduced public company reporting requirements applicable to emerging growth companies, which could make PubCo ordinary shares less attractive to investors.
PubCo expects to qualify as an “emerging growth company,” as defined in the Securities Act of 1933, as amended, after the consummation of the Business Combination, and PubCo may take advantage of certain exemptions from various reporting requirements that are applicable to other public companies that are not “emerging growth companies”, including to provide less extensive narrative disclosure than required of other reporting companies, particularly in the description of executive compensation; and to provide audited financial statements for two fiscal years, in contrast to other reporting companies, which must provide audited financial statements for three fiscal years. PubCo cannot predict if investors will find the investment in PubCo riskier because PubCo may rely on these exemptions. If some investors find PubCo ordinary shares less attractive as a result, there may be a less active trading market for Alps Holdco ordinary shares and PubCo’s share price may be more volatile. PubCo may take advantage of these reporting exemptions until PubCo is no longer classified as an “emerging growth company”. PubCo will remain an “emerging growth company” unless one of the following occurs: (i) total annual gross revenues are $1.235 billion or more; (ii) Alps has issued more than $1 billion in non-convertible debt in the past three years; or (iii) becomes a “large accelerated filer,” as defined in Exchange Act of 1934 Rule 12b-2.
Alps’ operations are subject to various laws and regulations in Malaysia.
Alps’ business is based in Malaysia and is regulated by various laws and regulations in Malaysia such as regulations on business licenses, business centres, the quality and the licensing of medical facilities, equipment and services, practice of medical practitioners, manufacturing facilities, procurement and usage of drugs, intellectual property rights, employment, personal data and privacy. Accordingly, our business centres are subject to periodic licensing renewal requirements and inspections by various government agencies and departments at the federal, state and municipal level. In addition, any changes in laws and regulations could require us to obtain additional licenses, permits, approvals or certificates, impose additional conditions or requirements for the renewal of the licenses of the business centres, or result in the invalidation of our currently owned licenses.
Based on Alps’ experience, some of the laws and regulations of the place where Alps operates its business are subject to amendments, uncertainty in interpretation and administrative actions from time to time. Therefore, Alps faces a risk that, for the implementation of its business plans and the introduction of any new services or products, Alps may not be able to obtain all the necessary registrations, certificates and/or licenses. Any failure to comply with the above laws and regulations may give rise to fines, administrative penalties and/or prosecution against Alps, which may adversely affect its reputation, financial condition or results of operation.
| 66 |
Developments in the social, political, regulatory and economic environment in Malaysia may have a material adverse impact on us.
Our business, prospects, financial condition and results of operations may be adversely affected by social, political, regulatory and economic developments in Malaysia. Such political and economic uncertainties include, but are not limited to, the risks of war, terrorism, nationalism, nullification of contract, changes in interest rates, imposition of capital controls and methods of taxation.
Negative developments in Malaysia’s socio-political environment may adversely affect our business, financial condition, results of operations and prospects. Malaysia’s economy grew by 3% in the fourth quarter of 2023 (Q4 2023), supported by resilient domestic demand that rose by 5.2% in Q4 2023 (Q3 2023: 4.8%). Although the overall Malaysian economic environment (in which we predominantly operate) appears to be positive, there can be no assurance that this will continue to prevail in the future. Economic growth is determined by countless factors, and it is extremely difficult to predict with any level of absolute certainty.
Alps faces the risk that changes in the policies of the Malaysian government could have a significant impact upon the business we may be able to conduct in Malaysia and the profitability of such business.
The policies enacted by the Malaysian government have substantial effects on the country’s economic landscape. Our operations and financial performance are heavily influenced by the legal, regulatory, and policy environment in Malaysia, which governs various aspects of our business, including research and development activities, product approval, manufacturing, marketing, and sales. This regulatory framework is subject to change, particularly with shifts in political climate, economic priorities, or public health considerations. Any future alterations in Malaysian laws, regulations, or government policies, or their interpretation, could have adverse effects on our business, financial condition, and operational results. Such changes may result in increased compliance costs and necessitate modifications to our operations, products, or quality control and manufacturing processes. Adapting to these regulatory shifts may impose a financial burden that could impact our profitability.
There is also uncertainty regarding the continuity of current government policies, especially in the event of changes in leadership, social or political disruptions, or other circumstances affecting Malaysia’s political, economic, and social environment. We cannot guarantee that the government will maintain its current policies or that such policies will remain unchanged in the future.
Fluctuations in exchange rates could affect Alps’ business and the value of its securities.
We are exposed to fluctuations in the value of the RM. We currently rely on vendors and suppliers who operate in USD. Suppliers and vendors that operate in USD comprise approximately 2.48% and 2.14% of our expenses for financial year ended March 31, 2023 and March 2024, respectively. To the extent the USD increases in value relative to the RM, our margins may be affected. Foreign exchange rates may also impact trade between countries as fluctuations in currencies may impact the value of goods as between two trading countries. We do not take actions to hedge against foreign exchange and transaction risks and are therefore exposed to the swing in the value of the RM. Consequently, short-term or long-term exchange rate movements or controls may affect on our business, financial condition, results of operations and liquidity.
Alps is subject to laws and regulations governing corruption.
Alps is obligated to adhere to various international laws and regulations aimed at combating corruption, including the Malaysian Anti-Corruption Commission Act 2009, Anti-Money Laundering, Anti-Terrorism Financing and Proceeds of Unlawful Activities Act 2001.
Anti-bribery laws prohibit Alps, Alps’ employees, and some of Alps’ agents or representatives from offering or providing any personal benefit to covered government officials to influence their performance of their duties or induce them to serve interests other than the missions of the public organizations in which they serve. Certain commercial bribery rules also prohibit offering or providing any personal benefit to employees and representatives of commercial companies to influence their performance of their duties or induce them to serve interests other than their employers.
| 67 |
These anti-bribery regulations strictly prohibit Alps and its representatives from offering or delivering any undue advantage to government officials or commercial entity employees to improperly influence their official duties. This compliance is notably burdensome in regions where corruption is rampant and further complicated within the healthcare sector. Improper payments related to clinical trials, pharmaceutical procurement, or other activities have triggered significant enforcement actions and penalties under anti-bribery laws, especially in the United States and China.
Identifying and preventing violations of these laws is challenging. Despite preventative measures, it’s not always possible to curtail all illicit activities or manage the risks they pose. As a result, Alps could face government investigations, legal actions, or lawsuits due to non-compliance, leading to significant financial penalties and reputational harm.
In the medical field, corrupt practices such as kickbacks or bribes from pharmaceutical manufacturers or distributors to healthcare providers can lead to severe legal and financial repercussions. If Alps personnel or partners engage in illicit activities, Alps could face substantial fines across the jurisdictions it operates in, significantly impacting its financial health and operational outcomes. Recent trends show hospitals denying access to pharmaceutical sales representatives to avoid corruption perceptions, which could hinder Alps’ marketing efforts if this attitude becomes prevalent among potential clients.
As Alps contemplates international expansion, enhancing our compliance frameworks becomes imperative to mitigate the risks associated with the Foreign Corrupt Practices Act (FCPA) and similar anti-corruption statutes worldwide. Our comprehensive compliance programs must address a wide array of regulations, including meticulous record-keeping as a publicly traded entity, and ensure thorough training for all company personnel. The development and enforcement of these anti-corruption measures are costly and complex, particularly when dependent on third-party adherence. Violations of the FCPA and other anti-corruption laws can lead to severe penalties, including substantial fines, exclusion from government contracts, criminal charges against individuals, and potential suspension from U.S. securities exchanges. Even in the absence of penalties, the investigative and legal defense costs, along with the negative impact on our reputation, could significantly detract from our profitability and our ability to develop or market our product candidates. Furthermore, if our competitors are not subjected to the same stringent anti-corruption laws, they may adopt practices that afford them a competitive advantage in securing business deals with foreign healthcare institutions, positioning them unfavorably against us in the international market.
The regulatory approval processes of the NPRA and comparable foreign authorities such as FDA are lengthy, time consuming and inherently unpredictable, and if Alps is ultimately unable to obtain regulatory approval for Alps’ product candidates, Alps’ business will be substantially harmed.
The path to obtaining regulatory approvals from the NPRA and similar international bodies like the FDA is extensive, arduous, and fraught with uncertainty. This process, typically spanning several years post-initiation of clinical trials, hinges on multiple factors, including the regulatory authorities’ broad discretion. Moreover, policies, regulatory frameworks, and the specifics of clinical data required for approval are subject to change throughout a product candidate’s clinical journey and may differ across jurisdictions.
To date, Alps has not secured regulatory approval for any product candidate, and there is no assurance that any future product candidates Alps may develop will receive such approval. Both Alps and any potential collaborators are barred from marketing any of Alps’ product candidates in Malaysia without NPRA’s approval for biosimilar, gene, and cell therapy applications.
Securing commercialization approval in Malaysia or internationally necessitates demonstrating, through substantial evidence from controlled clinical trials, the safety and efficacy of a product candidate to the satisfaction of the NPRA and other equivalent foreign regulatory bodies such as FDA. Interpretations of nonclinical and clinical data can vary, and even promising data may not suffice for approval. Alps may face additional study requirements from the NPRA either before or after approval, potentially delaying the approval process by years or demanding resources beyond our current capacity.
| 68 |
Any therapeutic products that Alps and Alps’ subsidiaries may develop cannot be sold until the NPRA and corresponding foreign regulatory authorities approve the products for medical use. The need to obtain regulatory approval to market a new product means that:
| ● | Expensive and time-consuming clinical trials of new products will need to be conducted. The full cost of conducting and completing clinical trials necessary to obtain NPRA and equivalent foreign regulatory approval of a new product cannot be presently determined but could exceed Alps’ financial resources or could discourage any future licensees or collaborators from pursuing NPRA approval of Alps’ product candidates. | |
| ● | Clinical trials and the regulatory approval process for a pharmaceutical or cell-based product can take several years to complete. As a result, Alps will face expenses and delays inherent in seeking NPRA and foreign regulatory approval of new products, even if the results of clinical trials are favorable. | |
| ● | Data obtained from preclinical and clinical studies is susceptible to varying interpretations and regulatory changes that could delay, limit, or prevent regulatory agency approvals. | |
| ● | Because the therapeutic products Alps plan to develop with pluripotent stem cell technology involve the application of new technologies and approaches to medicine, NPRA or foreign regulatory agencies may subject those products to additional or more stringent review than drugs or biologicals derived from other technologies. | |
| ● | A product that is approved may be subject to restrictions on use. | |
| ● | The NPRA can recall or withdraw approval of a product, if it deems necessary. | |
| ● | Alps may face similar regulatory issues in foreign countries. |
Approval of Alps’ product candidates may be delayed or refused for many reasons, including the following:
| ● | The NPRA or comparable foreign regulatory authorities may disagree with the design or implementation of the applicable clinical trial. | |
| ● | A clinical trial might not demonstrate to the satisfaction of the NPRA or comparable foreign regulatory authorities that Alps’ product candidates are safe and effective for any of their proposed indications. | |
| ● | The results of clinical trials may not meet the level of statistical significance required by the NPRA or comparable foreign regulatory authorities for approval. | |
| ● | A clinical trial fail to demonstrate that Alps’ product candidates’ clinical and other benefits outweigh their safety risks. | |
| ● | The NPRA or comparable foreign regulatory authorities may disagree with Alps’ interpretation of data from preclinical programs or clinical trials. | |
| ● | The facilities of the any third-party manufacturers with which Alps may contract may not be adequate to support approval of Alps’ product candidates (for example, regulatory approval of cell and tissue-based products require high standards of quality control). | |
| ● | The approval policies or regulations of the NPRA or comparable foreign regulatory authorities may significantly change in a manner rendering Alps’ clinical data insufficient for approval. | |
| ● | Of the large number of potential products in development, only a small percentage successfully complete the NPRA or foreign regulatory approval processes and are commercialized. The lengthy approval process as well as the unpredictability of future clinical trial results may result in Alps’ failing to obtain regulatory approval to market Alps’ product candidates, which would significantly harm Alps’ business, results of operations and prospects. | |
| ● | Ethical, social and legal concerns about research regarding stem cells, could result in regulations restricting or prohibiting the processes Alps may use. Government agencies, parliamentary committees and foreign governments have expressed interest in further regulating biotechnology. More restrictive regulations or claims that Alps’ products are unsafe or pose a hazard could prevent Alps from commercializing any products. New government requirements may be established that could delay or prevent regulatory approval of Alps’ product candidates under development. It is impossible to predict whether legislative changes will be enacted, regulations, policies or guidance changed, or interpretations by agencies or courts changed, or what the impact of such changes, if any, may be. | |
| ● | Regulatory requirements governing gene and cell therapy products have changed frequently and may continue to change in the future. |
| 69 |
Additionally, ethical, social, and legal debates surrounding stem cell research could lead to restrictive regulations or outright bans on essential processes. Legislative changes, policy shifts, or new regulatory requirements could further complicate the approval landscape for gene and cell therapy products, emphasizing the volatile regulatory environment surrounding biotechnology.
The FDA and other comparable foreign regulatory authorities may not accept data from trials or studies conducted in Malaysia or other locations outside of their jurisdiction.
Alps intends to supply its product candidates to numerous clinics and medical centers outside of Malaysia. Such foreign clinics and medical centers may intend to use Alps’ product candidates, to conduct clinical studies for the potential treatment of a wide variety of indications, and Alps may choose to conduct international clinical trials or studies in the future. The primary purpose of these clinical studies is for the open-label treatment of the respective indication; accordingly, there is no randomized control group for patients treated in these foreign clinical studies.
The acceptance of study data by the NPRA, or other comparable foreign regulatory authority from clinical trials or studies approved by NPRA or jurisdictions outside of their respective jurisdictions may be subject to certain conditions. In cases where data from foreign clinical trials or studies are intended to serve as the basis for regulatory approval in the United States, the FDA will generally not approve the application on the basis of foreign data alone unless (1) the data are applicable to the United States population and United States medical practice; (2) the trials are performed by clinical investigators of recognized competence and pursuant to cGCP requirements; and (3) the FDA is able to validate the data through an on-site inspection or other appropriate means. The FDA may accept the use of some foreign data to support a marketing approval if the clinical trial meets certain requirements. Additionally, the FDA’s clinical trial requirements, including the adequacy of the subject population studied and statistical powering, must be met. Furthermore, such foreign trials or studies would be subject to the applicable local laws of the foreign jurisdictions where the trials or studies are conducted, including from our ongoing and planned pre-clinical studies. There can be no assurance that the FDA or any applicable foreign regulatory authority will accept data from trials or studies conducted outside of its respective jurisdiction. If the FDA, or any applicable foreign regulatory authority does not accept such data, it may result in the need for additional studies, which would be costly and time-consuming and delay aspects of our business plan, and which may result in our product candidates not receiving approval for commercialization in the applicable jurisdiction.
If we fail to comply with the extensive legal and regulatory requirements affecting the health care industry, Alps could face increased costs, penalties and a loss of business.
Alps’ activities, and the activities of any collaborators, distributors and other third-party providers that Alps may engage in the future, will be subject to extensive government regulation and oversight both in Malaysia and in foreign jurisdictions where Alps operates. The NPRA and comparable agencies in other jurisdictions will directly regulate many of Alps’ most critical business activities, including the conduct of preclinical and clinical studies, product manufacturing, advertising and promotion, product distribution, adverse event reporting, and product risk management. Alps’ interactions in Malaysia or abroad with physicians and other health care providers that prescribe or purchase Alps’ products will also be subject to government regulation designed to prevent fraud and abuse in the sale and use of the products and place greater restrictions on the marketing practices of health care companies. Biotechnology companies such as ours are facing heightened scrutiny of their relationships with health care providers from anti-corruption enforcement officials. In addition, biotechnology companies such as ours may be the target of lawsuits and investigations alleging violations of government regulation, including claims asserting submission of incorrect pricing information, impermissible off-label promotion of pharmaceutical products, payments intended to influence the referral of health care business, submission of false claims for government reimbursement, antitrust violations, and violations related to environmental matters. Risks relating to compliance with laws and regulations may be heightened if Alps operates in jurisdictions outside of Malaysia.
Regulations governing the health care industry are subject to change, with possibly retroactive effect, including:
| ● | new laws, regulations or judicial decisions, or new interpretations of existing laws, regulations or decisions, related to health care availability, pricing or marketing practices, compliance with wage and hour laws and other employment practices, method of delivery, payment for health care products and services, compliance with health information and data privacy and security laws and regulations, tracking and reporting payments and other transfers of value made to physicians and teaching hospitals, extensive anti-bribery and anti-corruption prohibitions, product serialization and labeling requirements and used product take-back requirements; |
| 70 |
| ● | changes in the NPRA and foreign regulatory approval processes that may delay or prevent the approval of new products and result in lost market opportunity; |
| ● | requirements that provide for increased transparency of clinical trial results and quality data, which could impact Alps’ ability to protect trade secrets and competitively-sensitive information contained in approval applications or could be misinterpreted leading to reputational damage, misperception, or legal action which could harm Alps’ business; and |
| ● | changes in NPRA and foreign regulations that may require additional safety monitoring, labeling changes, restrictions on product distribution or use, or other measures after the introduction of Alps’ products to market, which could increase Alps’ costs of doing business, adversely affect the future permitted uses of approved products, or otherwise adversely affect the market for Alps’ products. |
Violations of governmental regulation may be punishable by criminal and civil sanctions against Alps, including fines and civil monetary penalties and exclusion from participation in government programs, as well as sanctions against executives overseeing Alps’ business. In addition to penalties for violation of laws and regulations, Alps cannot ensure that Alps’ compliance controls, policies and procedures will in every instance protect Alps from acts committed by Alps’ employees, collaborators, partners or third-party providers that would violate the laws or regulations of the jurisdictions in which Alps operate. Whether or not Alps has complied with the law, an investigation into alleged unlawful conduct could increase Alps’ expenses, damage Alps’ reputation, divert management time and attention, and adversely affect Alps’ business.
mRNA drug development has substantial clinical development and regulatory risks due to the novel and unprecedented nature of this new category of therapeutics.
As a potential new category of therapeutics, to Alps’ knowledge, no mRNA therapies have been approved to date by the NPRA. Successful discovery and development of mRNA-based (and other) therapies by either Alps or its collaborators is highly uncertain and depends on numerous factors, many of which are beyond Alps’ or their control. Alps’ product candidates that appear promising in the early phases of development may fail to advance, experience delays in the clinic or clinical holds, or fail to reach the market for many reasons, including:
| ● | discovery efforts aimed at identifying potential immunotherapies may not be successful; | |
| ● | nonclinical or preclinical study results may show product candidates to be less effective than desired or have harmful or problematic side effects; | |
| ● | clinical trial results may show the product candidates to be less effective than expected, including a failure to meet one or more endpoints or have unacceptable side effects or toxicities; | |
| ● | manufacturing failures or insufficient supply of GMP materials for clinical trials, or higher than expected cost could delay or set back clinical trials, or make Alps’ product candidates commercially unattractive; | |
| ● | Alps’ improvements in the manufacturing processes may not be sufficient to satisfy the clinical or commercial demand of the product candidates or regulatory requirements for clinical trials; and | |
| ● | changes that Alps makes to optimize its manufacturing, testing or formulating of GMP materials could impact the safety, tolerability and efficacy of its product candidates. |
Risk Related to Our Healthcare, Wellness and Aesthetics Business
We are subject to customer complaints, claims and legal proceedings in the regular course of our operations from time to time, which could result in significant costs and materially and adversely affect our brand image, reputation and results of operations.
We rely heavily on our doctors and medical staff to make sound decisions regarding the treatment of our customers. However, we cannot guarantee that every employee at our treatment centers will consistently adhere to the appropriate professional standard of care. Any deviation from this standard by our medical staff, or any failure in managing our services and activities, may lead to unsatisfactory treatment outcomes, patient injuries, or in extreme cases, fatalities.
| 71 |
Given the subjective nature of the aesthetic medical industry, we are also vulnerable to various types of complaints related to our services. These may include dissatisfaction with customer service, disputes over charges, over-promising of treatment outcomes, dissatisfaction with post-treatment recovery periods, and general dissatisfaction with treatment results. Moreover, as the number of procedures we perform continues to increase with our growth, the absolute number of such complaints, allegations, and claims—regardless of merit—may also rise.
Failure to manage these complaints, allegations, and claims effectively could significantly damage our reputation, business, financial condition, and prospects. Even unfounded complaints or legal proceedings, if widely publicized, could tarnish our corporate image, divert management resources, and result in additional costs to address these matters. Furthermore, settlements or successful claims against us could entail substantial costs, damages, compensation, and reputational harm, ultimately impacting our business, financial performance, and operational outcomes.
If we are unable to recruit and retain an adequate number of managers, doctors, nurses, consultants and other support staff in our treatment centres, our service quality and business strategy may suffer.
Our performance hinges significantly on the expertise and dedication of highly skilled medical professionals. Securing and retaining top-tier talent across all areas of our treatment centres is crucial for our future success. However, the recruitment of qualified physicians in Malaysia is highly competitive due to their scarcity. Physicians typically consider several key factors when choosing medical institutions, including reputation, organizational culture, management efficiency, facility quality, patient volume, compensation, training opportunities, and location.
Competing with other aesthetic medical centers or clinics for qualified professionals may pose challenges for our company. If we fail to attract or retain seasoned and qualified physicians, it could adversely impact our business, financial stability, and operational outcomes.
Furthermore, recruiting and retaining qualified medical professionals have become increasingly costly in recent years, with no guarantee of success in the future. If we cannot attract a sufficient number of skilled professionals, our service quality and ability to execute our business strategy may suffer. Additionally, a shortage of qualified professionals may necessitate higher wage payments, leading to reduced profits and adversely affecting our operating results and financial performance.
Alps may fail to maintain the quality of the medical equipment, medical supplies, materials, skincare products, implants and consumables it uses. If these products do not meet the required standards, Alps could be exposed to liabilities and its business operations and reputation could suffer.
Despite implementing stringent measures for supplier selection, such as maintaining an updated list of qualified suppliers, Alps cannot guarantee the defect-free or substantial compliance of all medical equipment, supplies, materials, skincare products, implants, and consumables used in its operations. Defective or substandard products from Alps’ suppliers may expose Alps to liability claims, complaints, adverse publicity, penalties, license suspensions, or court-awarded compensations. In such cases, Alps may incur costs to replace products, impacting its profit margins and causing service delays for customers.
Alps’ suppliers are also governed by extensive laws and regulations, and any violations on their part may adversely affect its reputation or procurement processes. Applicable Malaysian laws mandate sourcing materials from licensed suppliers, and non-compliance could result in penalties or fines. Moreover, if Alps’ suppliers lose necessary qualifications without their knowledge, Alps may inadvertently breach regulatory requirements. This could lead to reputational damage, liabilities for defective goods, negative publicity, and adverse effects on its operational results.
| 72 |
Alps faces intense competition, and if it does not compete successfully against new or existing competitors, Alps may lose its market share and its profitability may be adversely affected.
Alps faces competition from private aesthetic hospitals, clinics, and public general hospitals’ aesthetic medical departments situated in the same geographical areas as our treatment centers. Additionally, the rapid expansion of the aesthetic medical industry in Malaysia may attract new domestic or international players, further intensifying competition. Some competitors, existing or potential, may possess competitive advantages such as greater financial resources or marketing capabilities, potentially replicating our business model. Alps’ competition for customers is primarily based on factors like location, pricing, service range, quality, and brand reputation. However, Alps cannot guarantee its ability to effectively compete against new or existing rivals. Failure to do so may impede its revenue and profitability growth and lead to a decline in market share.
If Alps is unable to adapt to changing aesthetic medical trends and its customers’ changing needs, we will not be able to compete effectively, which may materially and adversely affect its business, financial condition and results of operations.
In the aesthetic medical industry, it is essential to closely monitor market trends and customer needs, potentially leading to the introduction of new products, technologies, devices, solutions, service categories, and treatment procedures, as well as improvements to existing services. Alps participates in conferences organized by its suppliers in order to keep abreast of the latest aesthetic medical solutions, standards, and technologies. Investments in development and acquisitions may be necessary to stay abreast of new technologies, implement innovative solutions, or phase out outdated ones. Failure to identify, develop, and introduce new products, solutions, service categories, features, enhancements, and technologies in a timely and cost-effective manner could lead to a decline in demand for its services, hindering its ability to compete effectively and attract customers, thereby potentially impacting its business and financial performance negatively.
To successfully expand in markets outside of Malaysia, Alps must address many issues with which it has limited experience.
International expansion is subject to a number of risks, including:
| ● | difficulties in staffing and managing our international operations; | |
| ● | increased competition as a result of more procedures receiving regulatory approval or otherwise freedom to market in international markets; | |
| ● | reduced or varied protection for intellectual property rights in some countries; | |
| ● | foreign tax laws; | |
| ● | fluctuations in currency exchange rates; | |
| ● | foreign certification and regulatory clearance or approval requirements; | |
| ● | difficulties in developing effective marketing campaigns in unfamiliar foreign countries; | |
| ● | political, social, and economic instability abroad, terrorist attacks, and security concerns in general; | |
| ● | potentially adverse tax consequences, including the complexities of foreign value-added tax systems, tax inefficiencies related to our corporate structure, and restrictions on the repatriation of earnings; | |
| ● | the burdens of complying with a wide variety of foreign laws and different legal standards; and | |
| ● | increased financial accounting and reporting burdens and complexities. |
If one or more of these risks were realized, it could require Alps to dedicate significant financial and management resources and its revenue may decline.
Alps may determine to expand its business if it is able to raise sufficient capital to do so, and Alps may experience difficulties in managing this growth, which could disrupt Alps’ operations.
As of the date of this proxy statement/prospectus, Alps had 84 employees. Many of the biotechnology companies that Alps competes against for qualified personnel and consultants have greater financial and other resources, different risk profiles and a longer history in the industry than Alps do and are better positioned to attract and retain personnel and consultants. If Alps is unable to continue to attract and retain high-quality personnel and consultants, the rate and success at which Alps can discover and develop product candidates and operate Alps’ business will be limited.
| 73 |
Future growth would impose significant additional responsibilities on Alps’ management, including the need to identify, recruit, maintain, motivate and integrate additional employees, scientists, researches, doctors and consultants. Also, Alps’ management may need to divert a disproportionate amount of its attention away from Alps’ day-to-day activities and devote a substantial amount of time to managing these growth activities. Alps may face challenges in effectively managing the expansion of its operations, potentially leading to infrastructure weaknesses, operational errors, missed business opportunities, employee attrition, and decreased productivity among remaining staff. The growth of administrative resources could necessitate substantial capital investments and may divert financial resources from other projects, such as product candidate development. If Alps’ management fails to effectively handle its growth, expenses may exceed projections, hindering revenue generation and growth potential, and impeding the execution of its business strategy. Alps’ future financial performance and its ability to successfully commercialize product candidates and compete in the market will, in part, hinge on its capacity to manage future growth effectively.
Alps’ future success depends on its ability to retain key personnel and to attract, retain and motivate qualified personnel.
Alps’ industry has experienced a high rate of turnover of management personnel in recent years. Alps is highly dependent on our executive officers, senior management and key personnel. Despite having employment contracts with these executives, nothing stops them from ending their association with Alps at any time.
If Alps loses one or more of its executive officers, senior management or key employees, its ability to consummate the Business Combination and the transactions contemplated by the Business Combination could be adversely affected. Furthermore, replacing executive officers and key employees may be difficult, in particular during the pendency of the Business Combination, and may take an extended period of time because of the uncertainty about such persons roles following the Business Combination and the limited number of individuals in Alps’ industry with the breadth of skills and experience required to develop, gain regulatory approval of and commercialize product candidates successfully. Competition to hire from this limited pool is intense, and Alps may be unable to hire, train, retain or motivate these additional key personnel on acceptable terms given the competition among numerous pharmaceutical and biotechnology companies for similar personnel. Alps also will experience competition for the hiring of scientific and clinical personnel from universities and research institutions. In addition, Alps will rely on consultants and advisors, including scientific and clinical advisors, to assist us in formulating its research and development and commercialization strategy. Alps’ consultants and advisors may be engaged by entities other than Alps and may have commitments under consulting or advisory contracts with other entities that may limit their availability to Alps. If Alps is unable to continue to attract and retain high quality personnel, its ability to consummate the Business Combination and evaluating potential strategic alternatives with respect to its assets and development programs will be limited.
Alps may not be able to make acquisitions or investments, or successfully integrate them into Alps’ business.
In line with Alps’ research and expansion strategies, Alps actively engages in a variety of potential strategic transactions, such as investments, alliances, partnerships, joint ventures, and acquisitions. These endeavors target businesses, biological assets, research institutions, and other assets that are either complementary to its operations or are anticipated to foster business growth. Notably, Alps has sought and continue to explore strategic research collaborations with institutions across jurisdictions to advance its product candidates. For instance, Alps entered into a Memorandum of Agreement dated August 9, 2023 with UCSI Hospital Sdn. Bhd. and UCSI Education Sdn. Bhd., establishing a collaborative framework encompassing various initiatives, including the sourcing of cellular products, conducting genetic research, facilitating clinical trials, exploring immunotherapy trials, and the usage of laboratories.
These types of transactions involve numerous risks, including, among others:
| ● | intense competition for suitable targets and partners, which could increase prices and adversely affect Alps’ ability to consummate deals on favorable or acceptable terms; | |
| ● | complex technologies, terms and arrangements, which may be difficult to implement and manage; | |
| ● | failures or delays in closing transactions; |
| 74 |
| ● | difficulties integrating brand identity, technologies, operations, existing contracts, and personnel; | |
| ● | failure to realize the anticipated return on investment, benefits or synergies; | |
| ● | failure to identify the problems, liabilities, or other shortcomings or challenges of an acquired company, partner or technology, including but not limited to issues related to intellectual property, cybersecurity risks, regulatory compliance practices, litigation, security interests over assets, contractual issues, revenue recognition or other accounting practices, or employee or user issues; | |
| ● | regulatory changes that require adjustments to Alps’ business or shareholding or rights in relation to subsidiaries or joint ventures; and | |
| ● | adverse reactions to acquisitions by investors and other stakeholders. |
Navigating these risks is crucial for maintaining our strategic direction and safeguarding Alps’ intellectual property and business interests.
Alps’ business and operations could suffer in the event of system failures.
Despite the implementation of security measures, our internal computer systems and those of our contractors and consultants are vulnerable to damage from computer viruses, unauthorized access, natural disasters including earthquakes and tsunamis, terrorism, war, and telecommunication and electrical failures. A loss of or damage to our data, a disruption in access to our data, or inappropriate disclosure of confidential or proprietary information, could disrupt Alps’ operations, delay or otherwise adversely affect the development of Alps’ product candidates, significantly increase Alps’ costs, or result in delays in any future regulatory filings we may make.
In addition, our product candidates, which encompass mRNA profiling, Whole Genome Sequencing (WGS), as well as those manufactured using cells stored in a cryopreserved master cell bank, are crucial components of Alps’ operations. While Alps believes it has adequate backup measures in place to address potential data loss in the event of a catastrophic incident, there remains a risk that its third-party suppliers and manufacturers could experience loss or damage to multiple cell banks and other critical data, severely impacting our manufacturing capabilities and the data generated.
We cannot guarantee the absence of stability or other manufacturing issues with any of our product candidates or products in the future. Any delay or interruption in the supply of clinical trial materials could impede the completion of planned trials, escalate associated costs, and potentially necessitate the initiation of new trials at additional expense or even termination. Adverse developments affecting the clinical or commercial manufacturing of our product candidates or products may lead to shipment delays, inventory shortages, lot failures, product withdrawals, or recalls, disrupting the supply chain and the data generated. Consequently, challenges encountered at any level of our supply chain could negatively impact our business, delaying or obstructing the development and commercialization of our product candidates or products, thereby affecting our business, prospects, financial condition, and operational results.
Security breaches and other disruptions could compromise our information and expose us to liability, and could cause our business and reputation to suffer.
In the normal course of operations, Alps collects and stores sensitive data, which includes protected health information, personally identifiable information, financial details, intellectual property, and proprietary business information owned or controlled by the company, its customers, payers, and other involved parties. Alps manages its applications and data by utilizing a combination of on-site systems and cloud-based data centers.
The secure processing, maintenance, and transmission of this information are vital to our operations and business strategy. Despite the security measures in place, our information technology and infrastructure may be susceptible to cyberattacks or breaches caused by employee-related costs. Alps will also incur fees and costs related to integration and systems consolidation. The elimination of duplicative costs may not offset incremental transaction-related and other integration costs in the near-term errors, malfeasance, or other disruptions. Any such breach could compromise our networks, resulting in unauthorized access, public disclosure, loss, or theft of information. Such occurrences could lead to legal claims, liability under privacy protection laws, and disruptions to Alps’ operations, potentially tarnishing its reputation. Even without a direct interruption to its operations, fines, penalties, or financial liabilities resulting from a security breach could undermine confidence in our services, adversely impacting its business and competitive standing.
| 75 |
Any unauthorized access, loss, or dissemination of information could also trigger legal proceedings and liabilities under Malaysian laws and regulations governing personal data protection and cybersecurity, as well as those specifically addressing patient and medical data.
Unfavorable media coverage could harm Alps’ business, financial condition, results of operations and prospects.
Adverse publicity about aspects such as our business practices, customer support, product standards, privacy and security measures, compliance with regulations, financial or operational performance, accounting decisions, or leadership could damage Alps’ reputation. This negative attention could also negatively affect Alps’ credibility, business health, operational outcomes, and future prospects and the engagement and loyalty of Alps’ network of patients, scientific collaborators, and research partners relying on Alps’ assets. Moreover, adverse media attention could attract regulatory scrutiny, potentially leading to actions or the introduction of new laws or regulations affecting our operations. The risks associated with unfavorable publicity are amplified by the extensive use of social media and the rising occurrence of misleading or unfounded news, especially on social media and online platforms.
Any failure in Alps’ efforts to train health practitioners could result in the misuse of its products, reduce the market acceptance of its products and have a material adverse effect on the business, financial condition, and results of operations.
The proficiency of health practitioners in utilizing Alps’ products requires a significant learning curve. It is critical to the success of Alps’ sales efforts to adequately train a sufficient number of practitioners. Following completion of training, Alps relies on health practitioners and administrators to advocate the benefits of its products in the broader marketplace. Convincing practitioners to dedicate the time and energy necessary for adequate training and implementation is challenging, and Alps cannot provide assurance that it will be successful in these efforts. If practitioners are not properly trained, they may misuse or ineffectively use Alps’ products, leading to potential negative outcomes for patients, as well as negative publicity, regulatory actions, or lawsuits against Alps, all of which could adversely impact its reputation.
If future data proves to be inconsistent with Alps’ clinical results or if competitors’ products present more favorable results, Alps’ revenues could decline and Alps’ business, financial condition, and results of operations could be materially and adversely affected.
If future research contradicts Alps’ clinical outcomes or if competitors’ products demonstrate superior benefits, it could significantly impact Alps’ business, financial condition, and operational results. This may arise from new studies that diverge from our previous findings or from research favoring competitors’ products in terms of efficacy or reliability. Additionally, healthcare professionals may postpone purchasing Alps’ products until further long-term clinical evidence and endorsements by key professionals confirm the effectiveness of its products for clinical use. Such developments could lead to a decline in Alps’ revenues and have a material adverse effect on its overall performance.
In the future, if Alps’ customers cannot obtain third-party reimbursement for their use of Alps’ products, they could be less inclined to purchase Alps’ products.
Our products will generally purchase by medical professionals who have various billing practices and patient mixes. Such practices range from primarily private pay to those who rely heavily on third-party payers, such as private insurance or government programs. At present, our products are not generally claimable pursuant to any insurance policies. However, in the future, in the event our products and treatments are covered by insurance policies, we anticipate third-party payers to review and frequent challenge the prices charged for our medical products and/or services. Payers may deny coverage and reimbursement on various grounds, including if they determine that the procedure was not medically necessary. Accordingly, both coverage and reimbursement can be expected to vary significantly from payer to payer. For the portion of physicians who rely heavily on third-party reimbursement, the inability to obtain reimbursement for services using Alps’ products could deter them from purchasing or using Alps’ products. Alps cannot predict the effect that future health care reforms or changes in financing for health plans could have on Alps’ business. Any such changes could have an adverse effect on the ability of a physician or medical institution to generate a profit using Alps’ current or future products. In addition, such changes could act as disincentives for capital investments by medical professionals.
| 76 |
Risks Related to Globalink’s Business and the Business Combination
Unless the context otherwise requires, references to the “Company,” “we”, “us” and “our” in this subsection generally refer to Globalink Investment Inc.
Globalink will be forced to liquidate the Trust Account if it cannot consummate a business combination by January 9, 2025 (or up to June 9, 2025 upon extension). In the event of a liquidation, Globalink’s public stockholders will receive $11.98 per share of common stock and our warrants and rights will expire worthless.
Globalink currently has until January 9, 2025 to complete its initial business combination, or it will be required to dissolve and liquidate. If Globalink anticipates that it may not be able to consummate its initial business combination in time, Globalink may, by resolutions of its Board, if requested by the Sponsor, extend the period of time to consummate a business combination on a monthly basis to until June 9, 2025, subject to the Sponsor depositing additional funds into the Trust Account. In the event of a liquidation, the per-share liquidation distribution will be $11.98 based on the balance in the Trust Account as of December 6, 2024. Furthermore, our warrants and rights will expire worthless in the event Globalink fails to complete a business combination.
Our securities were suspended from trading and delisted from Nasdaq on December 17, 2024, following receipt of a delisting determination letter from Nasdaq on December 10, 2024. This could have significant material adverse consequences on us and our securities, including that it will negatively impact our ability to complete a Business Combination, will limit investors’ ability to make transactions in our securities and could subject us to additional trading restrictions.
We currently have up until June 9, 2025 (our 42-month anniversary) to complete an initial business combination. Nasdaq Listing Rule 5815, which was amended effective October 7, 2024, provides for the immediate suspension and desilting upon issuance of a listing determination letter for failure to meet the requirement in Nasdaq Listing Rule IM 5101-2(b), curtailing the ability of the Nasdaq hearings panel to give special purpose acquisition companies more time to complete an initial business combination beyond 36 months. Nasdaq Listing Rule IM 5101-2(b) requires a SPAC such as us to complete its initial business combination within 36 months of the effectiveness of its IPO registration statement, which, in our case, is December 9, 2024.
As such, following December 9, 2024 (our 36-month anniversary), we are no longer in compliance with Nasdaq listing rules. On December 10, 2024, we received a delisting determination letter from Nasdaq. As a result, our securities were immediately suspended from trading and delisted from Nasdaq on December 17, 2024, following our receipt of the delisting determination letter. Our securities are currently traded on OTC Pink.
In addition, in connection with any initial business combination, we would be required to demonstrate compliance with the applicable exchange’s initial listing requirements, which are more rigorous than the continued listing requirements, in order to continue to maintain the listing of our securities. We cannot assure you that we will be able to meet those initial listing requirements at that time, particularly if we are no longer listed on a stock exchange.
Following the suspension and delisting of our securities from Nasdaq, we and our securities are currently facing significant material adverse consequences, including:
| ● | being less attractive to potential business combination targets and therefore making it more difficult for us to complete an initial business combination; | |
| ● | a decreased ability to issue additional securities or obtain additional financing in the future; | |
| ● | a limited availability of market quotations for our securities, even if our securities were to be quoted on an over-the-counter market; | |
| ● | reduced liquidity and demand for our securities; | |
| ● | determination that our shares of common stock are a “penny stock” which will require brokers trading in our shares of common stock to adhere to more stringent rules and could result in a further reduced level of trading activity in the secondary trading market for our securities; | |
| ● | greater difficulty and cost at being able to satisfy any applicable stock exchange’s initial listing requirements for the post-business combination company; | |
| ● | our securities no longer qualifying as “covered securities” under the National Securities Markets Improvement Act of 1996 (“NSMIA”), meaning that sales of our securities would be subject to regulation in each state in which that sale occurs, including in connection with our initial business combination, which may negatively impact our ability to consummate our initial business combination or to otherwise issue additional securities or obtain additional financing in the future and could negatively impact the ability of our security holders to trade, and result in further reduced liquidity and demand for, our securities; and | |
| ● | a limited amount of news and analyst coverage. |
Additionally, under the Merger Agreement, one of the conditions to Closing is the listing by Nasdaq of the PubCo ordinary shares and PubCo warrants and satisfaction of initial and continued listing requirement. Following the desilting of our securities from the Nasdaq, PubCo may face increased difficulties and uncertainties in meeting the initial and continued listing requirement of Nasdaq, such as the requirements as to the market value of unrestricted publicly held shares and market value of listed securities, and therefore face increased uncertainties as to its ability to successfully consummate the Business Combination.
You must tender your shares of Globalink common stock in order to validly seek redemption at the Special Meeting.
In connection with tendering your shares for redemption, you must elect either to physically tender your share certificates to Globalink’s transfer agent by two (2) business days before the Special Meeting, or deliver your shares of Globalink common stock to the transfer agent electronically using The DTC’s DWAC System, which election would likely be determined based on the manner in which you hold your shares of Globalink common stock. Stockholders seeking to exercise their redemption rights and opting to deliver physical certificates should allot sufficient time to obtain physical certificates from the transfer agent and time to effect delivery. It is Globalink’s understanding that stockholders should generally allot at least two weeks to obtain physical certificates from the transfer agent. However, Globalink does not have any control over this process and it may take longer than two weeks. Stockholders who hold their shares in “street name” will have to coordinate with their bank, broker or other nominee to have the shares certificated or delivered electronically.
Any demand for redemption, once made, may be withdrawn at any time until the deadline for exercising redemption requests and thereafter, with Globalink’s consent, until the consummation of the Business Combination, or January 9, 2025 (or up to June 9, 2025 if Globalink further extends the timeline to complete its initial business combination) as determined by the Board. If you delivered your shares for redemption to Globalink’s transfer agent and decide within the required timeframe not to exercise your redemption rights, you may request that Globalink’s transfer agent return the shares (physically or electronically).
If third parties bring claims against Globalink, the proceeds held in trust could be reduced and the per-share liquidation price received by Globalink’s stockholders may be less.
Globalink’s placing of funds in trust may not protect those funds from third party claims against Globalink. Although Globalink has received from many of the vendors, service providers (other than its independent accountants) and prospective target businesses with which it does business executed agreements waiving any right, title, interest or claim of any kind in or to any monies held in the Trust Account for the benefit of Globalink’s public stockholders, they may still seek recourse against the Trust Account. Additionally, a court may not uphold the validity of such agreements. Accordingly, the proceeds held in trust could be subject to claims which could take priority over those of Globalink’s public stockholders. If Globalink liquidates the Trust Account before the completion of a business combination and distributes the proceeds held therein to its public stockholders, the Sponsor has contractually agreed that it will be liable to ensure that the proceeds in Globalink’s Trust Account are not reduced by the claims of target businesses or claims of vendors or other entities that are owed money by us for services rendered or contracted for or products sold to us, but only if such a vendor or prospective target business does not execute such a waiver. However, Globalink cannot assure you that they will be able to meet such obligation. Therefore, the per-share distribution from the Trust Account for our stockholders may be less than $11.98 calculated based on the balance of the Trust Account of $3.33 million as of December 6, 2024, due to such claims.
Additionally, if Globalink is forced to file a bankruptcy case or an involuntary bankruptcy case is filed against it which is not dismissed, the proceeds held in the Trust Account could be subject to applicable bankruptcy law, and may be included in Globalink’s bankruptcy estate and subject to the claims of third parties with priority over the claims of its stockholders. To the extent any bankruptcy claims deplete the Trust Account, Globalink may not be able to return the estimated redemption price of $11.98 to Globalink’s public stockholders.
| 77 |
We may be subject to U.S. foreign investment regulations which may impose conditions on or limit certain investors’ ability to purchase our securities or otherwise participate in the Business Combination, potentially making the securities less attractive to investors. Our future investments in U.S. companies may also be subject to U.S. foreign investment regulations.
We may not be able to complete an initial business combination with a U.S. target company since such initial business combination may be subject to U.S. foreign investment regulations and review by a U.S. government entity such as the Committee on Foreign Investment in the United States (“CFIUS”), or ultimately prohibited.
The Sponsor is a Delaware limited liability company, has equity holders that reside outside the United States. Globalink therefore may be considered a “foreign person” under the regulations administered by CFIUS and will continue to be considered as such in the future for so long as the Sponsor has the ability to exercise control over Globalink for purposes of CFIUS’s regulations. As such, an initial business combination with a U.S. business may be subject to CFIUS review, the scope of which was expanded by the Foreign Investment Risk Review Modernization Act of 2018 (“FIRRMA”), to include certain non-passive, non-controlling investments in sensitive U.S. businesses and certain acquisitions of real estate even with no underlying U.S. business. FIRRMA, and subsequent implementing regulations that are now in force, also subjects certain categories of investments to mandatory filings. If Globalink’s initial business combination with a U.S. business falls within CFIUS’s jurisdiction, Globalink may determine that it is required to make a mandatory filing or that it will submit a voluntary notice to CFIUS, or to proceed with the initial business combination without notifying CFIUS and risk CFIUS intervention, before or after closing the initial business combination. CFIUS may decide to block or delay Globalink’s initial business combination, impose conditions to mitigate national security concerns with respect to such initial business combination or order Globalink to divest all or a portion of a U.S. business of the combined company without first obtaining CFIUS clearance, which may limit the attractiveness of or prevent Globalink from pursuing certain initial business combination opportunities that it believes would otherwise be beneficial to Globalink and its stockholders.
Moreover, the process of government review, whether by the CFIUS or otherwise, could be lengthy and Globalink has limited time to complete its initial business combination. If Globalink cannot complete its initial business combination by January 9, 2025 (or up to June 9, 2025 if Globalink further extends the timeline to complete its initial business combination) because the review process drags on beyond such timeframe or because Globalink’s initial business combination is ultimately prohibited by CFIUS or another U.S. government entity, Globalink may be required to liquidate. If Globalink liquidates, based on the Trust Account balance as of December 6, 2024 Globalink’s public stockholders may only receive approximately $10.98 per share of common stock, net of taxes payable, and the warrants and rights will expire worthless. This will also cause you to lose the investment opportunity in a target company and the chance of realizing future gains on your investment through any price appreciation in the combined company.
The SEC issued final rules to regulate special purpose acquisition companies that may increase our costs and the time needed to complete our initial business combination.
On January 24, 2024, the SEC issued final rules and guidance relating to special purpose acquisition companies, like Globalink, regarding, among other things, disclosure in SEC filings in connection with initial business combination transactions; the financial statement requirements applicable to transactions involving shell companies; the use of projections in SEC filings in connection with proposed business combination transaction; and the potential liability of certain participants in proposed business combination transactions. The rules became effective as of July 1, 2024. This evolution may result in continuing uncertainty regarding compliance matters and additional costs necessitated by ongoing revisions to Globalink’s disclosure and governance practices. A failure to comply with applicable laws or regulations and any subsequent changes, as interpreted and applied, could have a material adverse effect on Globalink’s business, including its ability to complete the Business Combination, and results of operations.
| 78 |
If we are deemed to be an investment company for purposes of the Investment Company Act, we would be required to institute burdensome compliance requirements and our activities would be severely restricted. As a result, in such circumstances, unless we are able to modify our activities so that we would not be deemed an investment company, we would expect to abandon our efforts to complete an initial business combination and instead to liquidate the Company.
There is currently uncertainty concerning the applicability of the Investment Company Act to a SPAC. To mitigate the risk of being deemed to have been operating as an unregistered investment company (including under the subjective test of Section 3(a)(1)(A) of the Investment Company Act), on July 27, 2023, the Company instructed Continental, the trustee of the Trust Account, to liquidate the U.S. government securities or money market funds held in the Trust Account and thereafter to hold all funds in the Trust Account in cash (which may include demand deposit accounts) until the earlier of consummation of our Business Combination or liquidation. However, it is possible that a claim could be made that we were operating as an unregistered investment company prior to July 2023. This risk may be increased if we decide to hold the funds in the Trust Account in short-term U.S. government treasury obligations or in money market funds invested exclusively in such securities again, rather than continuing to hold the funds in the Trust Account in cash.
If we were deemed to be an investment company under the Investment Company Act, our activities would be severely restricted. In addition, we would be subject to burdensome compliance requirements. We do not believe that our principal activities will subject us to regulation as an investment company under the Investment Company Act. However, if we were deemed to be an investment company and subject to compliance with and regulation under the Investment Company Act, we would be subject to additional regulatory burdens and expenses for which we have not allotted funds. As a result, unless we can conduct our activities in such way that we would not be deemed an investment company, we may expect to abandon our efforts to complete the Business Combination and instead to liquidate. If we were required to liquidate, our stockholders would not be able to realize the benefits of owning shares of stock in the post-business combination company, including the potential appreciation in the value of our common stock and warrants following the Business Combination, and our warrants would expire worthless.
Any distributions received by Globalink’s stockholders could be viewed as an unlawful payment if it was proved that immediately following the date on which the distribution was made, Globalink was unable to pay its debts as they fell due in the ordinary course of business and the value of its assets does not exceed its liabilities.
Globalink currently has until January 9, 2025 to complete its initial business combination, or it will be required to dissolve and liquidate. If Globalink anticipates that it may not be able to consummate its initial business combination in time, Globalink may, by resolutions of its Board, if requested by the Sponsor, extend the period of time to consummate a business combination on a monthly basis to until June 9, 2025, subject to the Sponsor depositing additional funds into the Trust Account. If Globalink is unable to consummate a transaction within the required time period, upon notice from Globalink, the trustee of the Trust Account will distribute the amount in its Trust Account to its public stockholders. Concurrently, Globalink shall pay, or reserve for payment, from funds not held in trust, its liabilities and obligations, although Globalink cannot assure you that there will be sufficient funds for such purpose.
We expect that all costs and expenses associated with implementing our plan of dissolution, as well as payments to any creditors, will be funded from amounts remaining out of the approximately the proceeds of the promissory notes held outside of the Trust Account we issued to PGM as of the date of this proxy statement/prospectus, although we cannot assure you that there will be sufficient funds for such purpose. We will depend on sufficient interest being earned on the proceeds held in the Trust Account to pay any tax obligations we may owe or for working capital purposes.
However, we may not properly assess all claims that may be potentially brought against us. As such, our stockholders could potentially be liable for any claims to the extent of distributions received by them (but no more) and any liability of our stockholders may extend well beyond the third anniversary of the date of distribution. Accordingly, third parties may seek to recover from our stockholders amounts owed to them by us.
If, after we distribute the proceeds in the Trust Account to our public stockholders, we file a bankruptcy petition or an involuntary bankruptcy petition is filed against us that is not dismissed, any distributions received by stockholders could be viewed under applicable debtor/creditor and/or bankruptcy laws as either a “preferential transfer” or a “fraudulent conveyance.” As a result, a bankruptcy court could seek to recover all amounts received by our stockholders. In addition, our Board may be viewed as having breached its fiduciary duty to our creditors and/or having acted in bad faith, thereby exposing itself and us to claims of punitive damages, by paying public stockholders from the Trust Account prior to addressing the claims of creditors.
| 79 |
There is no assurance that Globalink’s diligence will reveal all material risks that may be present with regard to Alps. Subsequent to the completion of the Business Combination, the Combined Company may be required to take write-downs or write-offs, restructuring and impairment or other charges that could have a significant negative effect on its financial condition and its share price, which could cause you to lose some or all of your investment.
Even though Globalink conducted a due diligence investigation of Alps, it cannot be sure that this diligence uncovered all material issues that may be present inside Alps or its business, or that it would be possible to uncover all material issues through a customary amount of due diligence, or that factors outside of Alps and its business and outside of its control will not later arise. Alps is a privately held company that expects to operate its planned business and offer services that have not yet been fully developed or been commercialized and Globalink therefore has made its decision to pursue a business combination with Alps on the basis of limited information, which may result in a business combination that is not as profitable as expected, if at all. As a result of these factors, the Combined Company may be forced to later write-down or write-off assets, restructure operations, or incur impairment or other charges that could result in reporting losses. Even if Globalink’s due diligence successfully identified certain risks, unexpected risks may arise and previously known risks may materialize in a manner not consistent with our preliminary risk analysis. Accordingly, any stockholders of Globalink who choose to remain shareholders of the Combined Company following the Business Combination could suffer a reduction in the value of their shares. Such stockholders are unlikely to have a remedy for such reduction in value unless they are able to successfully claim that the reduction was due to the breach by Globalink’s officers or directors of a duty of care or other fiduciary duty owed by them to Globalink, or if they are able to successfully bring a private claim under securities laws that the proxy statement/prospectus relating to the Business Combination contained an actionable material misstatement or material omission.
Because the Combined Company will be a publicly traded company by virtue of mergers as opposed to an underwritten initial public offering, the process does not use the services of one or more underwriters, which could result in less diligence being conducted.
In an underwritten initial public offering, underwriters typically conduct due diligence on the company being taken public in order to establish a due diligence defense against liability claims under federal securities laws. Because Globalink is already a publicly traded company, an underwriter has not been engaged. While the Sponsor may have an inherent conflict of interest because its shares, rights and warrants will be worthless if a business combination is not completed, management and the board of directors of the acquirer, as well as private investors, undertake a certain level of due diligence. However, this due diligence is not necessarily the same level of due diligence undertaken by an underwriter in a traditional initial public offering. If such investigation had occurred, certain information in this proxy statement/prospectus may have been presented in a different manner or additional information may have been presented at the request of such underwriter.
Stockholder litigation and regulatory inquiries and investigations are expensive and could harm Globalink’s operating results and could divert management attention.
In the past, securities class action litigation and/or stockholder derivative litigation and inquiries or investigations by regulatory authorities have often followed certain significant business transactions, such as the sale of a company or announcement of any other strategic transaction, such as the Business Combination. Any stockholder litigation and/or regulatory investigations against Globalink, whether or not resolved in Globalink’s favor, could result in substantial costs and divert Globalink’s management’s attention from other business concerns, which could adversely affect Globalink’s business and cash resources and the ultimate value Globalink’s stockholders receive as a result of the Business Combination.
| 80 |
The Initial Stockholders, Globalink’s executive officers and directors who own shares of common stock will lose their entire investment in Globalink if the Business Combination or an alternative business combination is not completed, and because Globalink’s Initial Stockholders, executive officers and directors will not participate in liquidation distributions and will not be eligible to be reimbursed for their out-of-pocket expenses if the Business Combination is not completed, a conflict of interest may have arisen in determining whether Alps was appropriate for Globalink’s initial business combination.
As of the Record Date, the Initial Stockholders owned an aggregate of 2,875,000 shares of common stock. They have waived their right to redeem any shares of common stock in connection with a stockholder vote to approve a proposed initial business combination or sell any shares of common stock to Globalink in a tender offer in connection with a proposed initial business combination, or to receive distributions with respect to any shares of common stock upon the liquidation of the Trust Account if Globalink is unable to consummate a business combination. Based on a market price of $11.95 per unit on December 12, 2024, the value of the Founder Shares was $34.36 million. The Founder Shares acquired prior to the IPO will be worthless if Globalink does not consummate a business combination. The personal and financial interests of Globalink’s executive officers and directors may have influenced their motivation in identifying and selecting a target business combination, completing an initial business combination and influencing the operation of the business following the initial business combination. At the closing of Globalink’s initial business combination, its Initial Stockholders, executive officers and directors, or any of their respective affiliates, will be reimbursed for any out-of-pocket expenses incurred in connection with activities on Globalink’s behalf such as identifying potential target businesses and performing due diligence on suitable business combinations. In the event the Business Combination or an alternative business combination is completed, there is no cap or ceiling on the reimbursement of out-of-pocket expenses incurred in connection with activities on Globalink’s behalf. However, Globalink’s Initial Stockholders, executive officers and directors, or any of their respective affiliates will not be eligible for any such reimbursement if the Business Combination or an alternative business combination is not completed. Such financial interests of Globalink’s Initial Stockholders, executive officers and directors may have influenced their motivation in approving the Business Combination and may influence their motivation for completing the Business Combination. Consequently, our directors’ discretion in identifying and selecting Alps as a suitable target business may result in a conflict of interest when determining whether the terms, conditions and timing of the Business Combination are appropriate and in Globalink’s public stockholders’ best interest.
Globalink is requiring stockholders who wish to redeem their shares in connection with a proposed business combination to comply with specific requirements for redemption that may make it more difficult for them to exercise their redemption rights prior to the deadline for exercising their rights.
Globalink is requiring public stockholders who wish to redeem their shares of common stock to either tender their certificates to Continental or deliver their shares to Continental electronically using the Depository Trust Company’s, or DTC, DWAC System two (2) business days before the Special Meeting. In order to obtain a physical certificate, a stockholder’s broker and/or clearing broker, DTC and Continental will need to act to facilitate this request. It is Globalink’s understanding that stockholders should generally allot at least two weeks to obtain physical certificates from Continental. However, because Globalink does not have any control over this process or over the brokers or DTC, it may take significantly longer than two weeks to obtain a physical share certificate. While Globalink has been advised that it takes a short time to deliver shares through the DWAC System, Globalink cannot assure you of this fact. Accordingly, if it takes longer than Globalink anticipates for stockholders to deliver their shares, stockholders who wish to redeem may be unable to meet the deadline for exercising their redemption rights and thus may be unable to redeem their shares of common stock.
Globalink will require its public stockholders who wish to redeem their shares of common stock in connection with the Business Combination to comply with specific requirements for redemption described above, such redeeming stockholders may be unable to sell their securities when they wish to in the event that the Business Combination is not consummated.
If Globalink requires public stockholders who wish to redeem their shares of common stock in connection with the proposed Business Combination to comply with specific requirements for redemption as described above and the Business Combination is not consummated, Globalink will promptly return such certificates to its public stockholders. Accordingly, investors who attempted to redeem their shares of common stock in such a circumstance will be unable to sell their securities after the failed acquisition until Globalink has returned their securities to them. The market price for common stock may decline during this time and you may not be able to sell your securities when you wish to, even while other stockholders that did not seek redemption may be able to sell their securities.
| 81 |
If Globalink’s security holders exercise their registration rights with respect to their securities, it may have an adverse effect on the market price of the Combined Company’s securities.
Globalink’s Initial Stockholders are entitled to make a demand that it register the resale of their Founder Shares at any time commencing three months prior to the date on which their shares may be released from escrow. Additionally, the holder of the private units and any units the Sponsor, Initial Stockholders, officers, directors, or their affiliates may be issued in payment of working capital loans made to Globalink, are entitled to demand that Globalink register the resale of the private units and any other units Globalink issues to them (and the underlying securities) commencing at three months after Globalink consummates an initial business combination. If such persons exercise their registration rights with respect to all of their securities, then there will be at least an additional 2,875,000 PubCo ordinary shares and the securities underlying Globalink’s 570,000 units eligible for trading in the public market, including 570,000 ordinary shares and 57,000 ordinary shares upon conversion of private rights. The presence of these additional ordinary shares and securities underlying Globalink’s units trading in the public market may have an adverse effect on the market price of Combined Company’s securities.
If the Business Combination’s benefits do not meet the expectations of financial or industry analysts, the market price of Combined Company’s securities may decline.
The market price of Combined Company’s securities may decline as a result of the Business Combination if:
| ● | Combined Company does not achieve the perceived benefits of the acquisition as rapidly as, or to the extent anticipated by, financial or industry analysts; or | |
| ● | The effect of the Business Combination on the financial statements is not consistent with the expectations of financial or industry analysts. |
Accordingly, investors may experience a loss as a result of decreasing share prices of the Combined Company.
Globalink’s and Alps Holdco’s directors and officers may have certain conflicts in determining to recommend the acquisition of Alps, since certain of their interests, and certain interests of their affiliates and associates, are different from, or in addition to, your interests as a stockholder.
Globalink’s and Alps Holdco’s management and directors have interests in and arising from the Business Combination that are different from, or in addition to, your interests as a stockholder, including, among others, the continued service as an officer or director of the Combined Company, severance benefits, equity grants, continued indemnification and the potential ability to sell an increased number of PubCo ordinary shares, which could result in a real or perceived conflict of interest. These interests include the fact that certain of the shares of common stock are owned by Globalink’s management and directors, or their affiliates and associates, would become worthless if either the Redomestication Merger Proposal or the Acquisition Merger Proposal is not approved and Globalink otherwise fails to consummate a business combination prior to its liquidation. See “The Acquisition Merger Proposal (Proposal 2) — Interests of Certain Persons in the Business Combination” beginning on page 136 for additional information.
Globalink and Alps have incurred and expect to incur significant transaction costs in connection with the Business Combination. Whether or not the Business Combination is completed, the incurrence of these costs will reduce the amount of cash available to be used for other corporate purposes by Globalink.
Globalink and Alps have incurred and expect to incur significant costs associated with the Business Combination. Whether or not the Business Combination is completed, Globalink expects to incur approximately $1 million in expenses. These expenses will reduce the amount of cash available to be used for other corporate purposes by Globalink. If the Business Combination is not consummated, Globalink may not have sufficient funds to seek an alternative business combination and may be forced to voluntarily liquidate and subsequently dissolve.
The unaudited pro forma condensed combined financial information included in this proxy statement/prospectus may not be indicative of what the Combined Company’s actual financial position or results of operations would have been.
The unaudited pro forma condensed combined financial information in this proxy statement/prospectus is presented for illustrative purposes only and is not necessarily indicative of what the Combined Company’s actual financial position or results of operations would have been had the Business Combination been completed on the dates indicated. See the section titled “Unaudited Pro Forma Condensed Combined Financial Information and Notes” for more information. Differences between preliminary estimates in the pro forma financial information and the final acquisition accounting will occur and could have an adverse impact on the pro forma financial information and the Combined Company’s financial position and future results of operations.
| 82 |
In addition, the assumptions used in preparing the pro forma financial information may not prove to be accurate and other factors may affect the Combined Company’s financial condition or results of operations following the Closing. Any potential decline in the Combined Company’s financial condition or results of operations may cause significant variations in the stock price of the Combined Company.
In the event that a significant number of shares of common stock are redeemed, the PubCo ordinary shares may become less liquid following the Business Combination. The ability of Globalink stockholders to exercise redemption rights with respect to a large number of Public Shares or other factors may not allow Globalink to complete the Business Combination or optimize its capital structure.
If a larger number of shares are submitted for redemption than Globalink currently expects and such redemptions or other conditions such as the PIPE Investment are determined to be insufficient or result in a failure to satisfy the net tangible asset requirement set forth in Globalink’ Globalink Charter, Globalink may need to seek to restructure the transaction to reserve a greater portion of the cash in the Trust Account or arrange for additional third-party financing. Third-party financing may not be available to Globalink. Furthermore, raising additional third-party financing may involve dilutive equity issuances or the incurrence of indebtedness at higher than desirable levels.
If the Business Combination is unsuccessful, you would not receive your pro rata portion of the Trust Account until Globalink liquidates the Trust Account or consummates an alternative initial business combination or upon the occurrence of an extension or certain other corporation actions as set forth in the Globalink Charter. If you are in need of immediate liquidity, you could attempt to sell your stock in the open market; however, at such time Globalink’ stock may trade at a discount to the pro rata amount per share in the Trust Account or there may be limited market demand at such time. In either situation, you may suffer a material loss on your investment or lose the benefit of funds expected in connection with Globalink’ redemption until Globalink liquidates, consummates an alternative initial business combination, effectuates an extension or takes certain other actions set forth in the Globalink Charter or you are able to sell your share in the open market.
The Combined Company will be required to meet the initial listing requirements to be listed on the Nasdaq. However, the Combined Company may be unable to maintain the listing of its securities in the future.
If the Combined Company fails to meet the continued listing requirements and Nasdaq delists its securities, the Combined Company could face significant material adverse consequences, including:
| ● | a limited availability of market quotations for its securities; | |
| ● | a limited amount of news and analyst coverage for the Combined Company; and | |
| ● | a decreased ability to issue additional securities or obtain additional financing in the future. |
If the conditions to the Business Combination are not met, the Business Combination may not occur.
Even if the Business Combination is approved by the stockholders of Globalink and Alps Holdco Shareholders, specified conditions must be satisfied or waived to complete the Business Combination. These conditions are described in detail in the Merger Agreement. See “The Acquisition Merger Proposal (Proposal 2) — Conditions to the Closing of the Business Combination” below for a more complete summary. Globalink and Alps Holdco cannot assure you that all of the conditions will be satisfied. If the conditions are not satisfied or waived, the Business Combination may not occur, or may be delayed and such delay may cause Globalink and Alps Holdco to each lose some or all of the intended benefits of the Business Combination. If the Business Combination does not occur, Globalink may not be able to find another potential candidate for its initial business combination prior to Globalink’s deadline (currently January 9, 2025 and may be extended to June 9, 2025 on a monthly basis by resolutions of its Board, if requested by the Sponsor, and subject to the Sponsor depositing additional funds into the Trust Account), and Globalink will be required to liquidate.
| 83 |
Globalink may waive one or more of the conditions to the Business Combination without resoliciting stockholder approval for the Business Combination.
Globalink may agree to waive, in whole or in part, some of the conditions to its obligations to complete the Business Combination, to the extent permitted by applicable laws. The Board will evaluate the materiality of any waiver to determine whether an amendment of this proxy statement/prospectus and resolicitation of proxies is warranted. In some instances, if the Board determines that a waiver is not sufficiently material to warrant resolicitation of stockholders, Globalink has the discretion to complete the Business Combination without seeking further stockholder approval. For example, it is a condition to Globalink’s obligations to close the Business Combination that there be no applicable law and no injunction or other order restraining or imposing any condition on the consummation of the Business Combination. However, if the Board determines that any such order or injunction is not material to the business of Alps, then the Board may elect to waive that condition without stockholder approval and close the Business Combination.
Globalink’s stockholders will experience immediate and substantial dilution as a consequence of the issuance of PubCo ordinary shares as consideration in the Business Combination and the PIPE Investment. Having a minority share position may reduce the influence that Globalink’s current stockholders have on the management of the Combined Company.
It is anticipated that upon completion of the Business Combination, under a no redemption scenario, Globalink’s public stockholders and Globalink’s right holders (other than the PIPE Investors) will retain an ownership interest of approximately 0.8% in the Combined Company, the PIPE Investors will own approximately 2.4% of the Combined Company, assuming the PIPE Investors will hold 4,020,000 PubCo ordinary shares, the Sponsor, officers, directors and other holders of Founder Shares will retain an ownership interest of approximately 1.7% of the Combined Company, PGM, an affiliate of the Sponsor, will retain an ownership interest of approximately 0.4% in the Combined Company, IBDC Asia Sdn. Bhd., an advisory firm to Alps, will own approximately 0.9% of the Combined Company, representing finder’s fees payable in PubCo ordinary shares with value equal to 1% of the aggregate consideration for the Business Combination of US$1.6 billion, and the Alps Holdco Shareholders will own approximately 93.8% of the Combined Company.
At the Closing, all outstanding Alps Holdco ordinary shares will be exchanged for PubCo ordinary shares. As a result, it is anticipated that upon completion of the Business Combination, under a maximum contractual redemption scenario, Globalink’s public stockholders and Globalink’s right holders (other than the PIPE Investors) will retain voting power of approximately 0.7% in the Combined Company, the PIPE Investors will have voting power approximately 2.4% in the Combined Company, assuming the PIPE Investors will hold 4,020,000 PubCo ordinary shares, the Sponsor, officers, directors and other holders of Founder Shares will have voting power of approximately 1.7% in the Combined Company, PGM, an affiliate of the Sponsor, will retain an ownership interest of approximately 0.4% in the Combined Company, IBDC Asia Sdn. Bhd., an advisory firm to Alps, will own approximately 0.9% of the Combined Company, representing finder’s fees payable in PubCo ordinary shares with value equal to 1% of the aggregate consideration for the Business Combination of US$1.6 billion, and the Alps Holdco Shareholders will have voting power of approximately 93.9% of the Combined Company.
The ownership percentage with respect to the Combined Company does not take into account the redemption of any shares of common stock by the Globalink public stockholders. If the actual facts are different from these assumptions (which they are likely to be), the percentage ownership retained by the Globalink stockholders will be different. See “Unaudited Pro Forma Condensed Combined Financial Information and Notes.”
Our ability to consummate a business combination may be materially adversely affected by the COVID-19 pandemic.
The ongoing COVID-19 pandemic has resulted in a widespread health crisis that has adversely affected and could continue to adversely affect the economies and financial markets worldwide, and the business of Alps or any other target business with which we consummate a business combination has been and could continue to be materially and adversely affected. The extent to which COVID-19 may further impact our ability to consummate a business combination will depend on future developments, which are highly uncertain and cannot be predicted. If the disruptions posed by the COVID-19 pandemic or other matters of global concern continue for an extensive period of time, our ability to consummate a business combination, or the operations of a target business with which we ultimately consummate a business combination, may be materially adversely affected.
| 84 |
Cayman Islands law and the Combined Company’s PubCo A&R Memorandum and Articles will contain certain provisions, including anti-takeover provisions, which limit the ability of shareholders to take certain actions and could delay or discourage takeover attempts that shareholders may consider favorable.
The Proposed PubCo A&R Memorandum and Articles that will be in effect upon consummation of the Business Combination, and the Companies Act, contain provisions that could have the effect of rendering more difficult, delaying, or preventing an acquisition deemed undesirable by the Combined Company Board and therefore depress the trading price of the PubCo ordinary shares. These provisions could also make it difficult for shareholders to take certain actions, including electing directors who are not nominated by the current members of the Alps Holdco Board or taking other corporate actions, including effecting changes in the management of the Combined Company. Among other things, the Proposed PubCo A&R Memorandum and Articles include provisions regarding:
| ● | the ability of the Combined Company Board to issue preferred shares and to determine the price and other terms of those shares, including preferences and voting rights, without shareholder approval, which could be used to significantly dilute the ownership of a hostile acquirer; |
| ● | the limitation of the liability of, and the indemnification of, the Combined Company’s directors and officers; | |
| ● | controlling the procedures for the conduct and scheduling of board of directors and shareholder meetings; and | |
| ● | the requirement for the affirmative vote of holders of a majority of not less than two-thirds of the voting power of all of the then outstanding shares, voting together as a single class, to amend, alter, change or repeal any provision of the Proposed PubCo A&R Memorandum and Articles, which could preclude shareholders from bringing matters before annual or extraordinary general meeting of shareholders and delay changes in the Combined Company Board and also may inhibit the ability of an acquirer to effect such amendments to facilitate an unsolicited takeover attempt. |
These provisions, alone or together, could delay or prevent hostile takeovers and changes in control or changes in the Combined Company Board or management.
Any provision of the PubCo A&R Memorandum and Articles or Cayman Islands law that has the effect of delaying or preventing a change in control could limit the opportunity for stockholders to receive a premium for their shares of the Combined Company’s share capital and could also affect the price that some investors are willing to pay for the PubCo ordinary shares.
Risks Related to the PubCo ordinary shares
The Combined Company’s share price may fluctuate significantly.
The market price of the PubCo ordinary shares may fluctuate widely, depending on many factors, some of which may be beyond PubCo’s control, including:
| ● | actual or anticipated fluctuations in PubCo’s results of operations due to factors related to its business; | |
| ● | success or failure of its business strategies; | |
| ● | competition and industry capacity; |
| 85 |
| ● | changes in interest rates and other factors that affect earnings and cash flow; | |
| ● | its level of indebtedness, its ability to make payments on or service its indebtedness and its ability to obtain financing as needed; | |
| ● | its ability to retain and recruit qualified personnel; | |
| ● | its quarterly or annual earnings, or those of other companies in its industry; | |
| ● | announcements by PubCo or its competitors of significant acquisitions or dispositions; | |
| ● | changes in accounting standards, policies, guidance, interpretations or principles; | |
| ● | the failure of securities analysts to cover, or positively cover, its ordinary shares after the Business Combination; |
| ● | changes in earnings estimates by securities analysts or its ability to meet those estimates; | |
| ● | the operating and stock price performance of other comparable companies; | |
| ● | investor perception of the company and its industry; | |
| ● | overall market fluctuations unrelated to its operating performance; | |
| ● | results from any material litigation or government investigation; | |
| ● | changes in laws and regulations (including tax laws and regulations) affecting its business; | |
| ● | changes in capital gains taxes and taxes on dividends affecting shareholders; and | |
| ● | general economic conditions and other external factors. |
Low trading volume for the PubCo ordinary shares, which may occur if an active trading market does not develop, among other reasons, would amplify the effect of the above factors on share price volatility.
Should the market price of the Combined Company’s shares drop significantly, shareholders may institute securities class action lawsuits against the Combined Company. A lawsuit against the Combined Company could cause the Combined Company to incur substantial costs and could divert the time and attention of its management and other resources.
Your percentage ownership in the Combined Company may be diluted by the Earnout Shares, the PIPE Investment and other equity issuance in the future.
Shareholders’ percentage ownership in the Combined Company may be diluted in the future if the Earnout Shares are issued, or due to the exercise of outstanding options, warrants and rights, or as a result of equity issuances for acquisitions, capital market transactions or otherwise, including equity awards that the Combined Company will be granting to directors, officers and other employees.
Following the execution of the Merger Agreement, PubCo, Globalink and Alps Holdco entered into the Subscription Agreements with certain accredited investors on June 4, 2024, June 5, 2024 and August 27, 2024, pursuant to which the investors have agreed to purchase, and PubCo has agreed to sell to the PIPE Investors, an aggregate of $40,200,000 worth of PubCo ordinary shares, or 4,020,000 PubCo ordinary shares.
From time-to-time, the Combined Company may opportunistically evaluate and pursue acquisition opportunities, including acquisitions for which the consideration thereof may consist partially or entirely of newly-issued ordinary shares of Combined Company and, therefore, such transactions, if consummated, would dilute the voting power and/or reduce the value of the PubCo ordinary shares.
| 86 |
An active, liquid trading market for the PubCo ordinary shares may not develop, which may limit your ability to sell your shares.
An active trading market for the PubCo ordinary shares may never develop or be sustained following the consummation of the Business Combination. A public trading market having the desirable characteristics of depth, liquidity and orderliness depends upon the existence of willing buyers and sellers at any given time, such existence being dependent upon the individual decisions of buyers and sellers over which neither the Combined Company nor any market maker has control. The failure of an active and liquid trading market to develop and continue would likely have a material adverse effect on the value of the PubCo ordinary shares. An inactive market may also impair the Combined Company’s ability to raise capital to continue to fund operations by issuing shares and may impair the Combined Company’s ability to acquire other companies or technologies by using the Combined Company’s shares as consideration.
Because there are no current plans to pay cash dividends on the PubCo ordinary shares for the foreseeable future, you may not receive any return on investment unless you sell your PubCo ordinary shares at a price greater than what you paid for it.
The Combined Company intends to retain its future earnings, if any, for future operations, expansion and pipeline development activities, and there are no current plans to pay any cash dividends for the foreseeable future. Additionally, the Combined Company does not plan to use its retained earnings for debt repayment. As a result, you may not receive any return on an investment in the PubCo ordinary shares unless you sell your PubCo ordinary shares for a price greater than that which you paid for it.
The issuance of additional ordinary shares or convertible securities may dilute your ownership and could adversely affect the share price.
From time to time in the future, the Combined Company may issue additional ordinary shares or securities convertible into ordinary shares pursuant to a variety of transactions, including acquisitions. Additional ordinary shares may also be issued upon exercise of outstanding share options and warrants to purchase ordinary shares. The issuance by PubCo of additional ordinary shares or securities convertible into ordinary shares would dilute your ownership of the Combined Company and the sale of a significant amount of such shares in the public market could adversely affect prevailing market prices of PubCo ordinary shares. Subject to the satisfaction of vesting conditions and the expiration of lockup agreements, shares issuable upon exercise of options will be available for resale immediately in the public market without restriction.
Issuing additional shares of the Combined Company’s share capital, other equity securities, or securities convertible into equity may dilute the economic and voting rights of the Combined Company’s existing shareholders, reduce the market price of the PubCo ordinary shares, or both. Debt securities convertible into equity could be subject to adjustments in the conversion ratio pursuant to which certain events may increase the number of equity securities issuable upon conversion. Preferred shares, if issued, could have a preference with respect to liquidating distributions or a preference with respect to dividend payments that could limit the Combined Company’s ability to pay dividends to the holders of its ordinary shares. The Combined Company’s decision to issue securities in any future offering will depend on market conditions and other factors beyond its control, which may adversely affect the amount, timing, or nature of PubCo’s future offerings. As a result, holders of the PubCo ordinary shares bear the risk that the Combined Company’s future offerings may reduce the market price of the PubCo ordinary shares and dilute their percentage ownership.
| 87 |
Future sales, or the perception of future sales, of the PubCo ordinary shares by the Combined Company or its existing shareholders in the public market could cause the market price for the PubCo ordinary shares to decline.
The sale of substantial amounts of PubCo ordinary shares in the public market, or the perception that such sales could occur, could harm the prevailing market price of the PubCo ordinary shares. These sales, or the possibility that these sales may occur, also might make it more difficult for the Combined Company to sell equity securities in the future at a time and at a price that it deems appropriate.
In connection with the Business Combination, certain Alps Holdco Shareholders have agreed that, subject to certain exceptions, they will not, during the period beginning at the effective time of the Business Combination and the date that is six months after the date of the Business Combination (subject to early release if Alps Holdco consummates a liquidation, merger, share exchange or other similar transaction with an unaffiliated third party), directly or indirectly, offer, sell, contract to sell, pledge, grant any option to purchase, make any short sale, or otherwise dispose of any PubCo ordinary shares, or any options or warrants to purchase any PubCo ordinary shares, or any securities convertible into, exchangeable for, or that represent the right to receive PubCo ordinary shares, or any interest in any of the foregoing.
Upon the expiration or waiver of the lock-up described above, shares held by these shareholders will be eligible for resale, subject to, in the case of shareholders who are PubCo’s affiliates, volume, manner of sale, and other limitations under Rule 144 promulgated under the Securities Act.
In addition, certain shareholders of the Combined Company will have registration rights under a registration rights agreement to be entered into prior to the Closing pursuant to which the Combined Company is obligated to register such shareholders’ PubCo ordinary shares and other securities that such shareholders may acquire after the Closing. Upon the effectiveness of the applicable registration statement, these PubCo ordinary shares will be available for resale without restriction, subject to any lock-up agreement.
The market price of PubCo ordinary shares could drop significantly if the holders of the shares described above sell them or are perceived by the market as intending to sell them. The market price of the PubCo ordinary shares may also decline as a result of the Business Combination for a number of reasons including if:
● investors react negatively to the prospects of the Combined Company’s business and the prospects of the Business Combination;
● the effect of the Business Combination on the Combined Company’s business and prospects is not consistent with the expectations of financial or industry analysts; or
● the Combined Company does not achieve the perceived benefits of the Business Combination as rapidly or to the extent anticipated by financial or industry analysts.
These factors could also make it more difficult for the Combined Company to raise additional funds through future offerings of PubCo ordinary shares or other securities.
If securities or industry analysts publish inaccurate or unfavorable research or reports about the Combined Company’s business, its share price and trading volume could decline.
The trading market for the PubCo ordinary shares depends, in part, on the research and reports that third-party securities analysts publish about the Combined Company and the industries in which the Combined Company operates. The Combined Company may be unable or slow to attract research coverage and if one or more analysts cease coverage of the Combined Company, the price and trading volume of its securities would likely be negatively impacted. If any of the analysts that may cover the Combined Company change their recommendation regarding the PubCo ordinary shares adversely or provide more favorable relative recommendations about the Combined Company’s competitors, the price of its ordinary shares would likely decline. If any analyst that may cover the Combined Company ceases covering the Combined Company or fails to regularly publish reports on the Combined Company, the Combined Company could lose visibility in the financial markets, which could cause the price or trading volume of the PubCo ordinary shares to decline. Moreover, if one or more of the analysts who cover the Combined Company downgrades the PubCo ordinary shares, or if the Combined Company’s reporting results do not meet their expectations, the market price of the PubCo ordinary shares could decline.
| 88 |
The Combined Company may be subject to securities litigation, which is expensive and could divert management attention.
Following the Business Combination, the price of the PubCo ordinary shares may be volatile and, in the past, companies that have experienced volatility in the market price of their ordinary shares have been subject to securities litigation, including class action litigation. Litigation of this type could result in substantial costs and diversion of management’s attention and resources, which could have a material adverse effect on the Combined Company’s business, financial condition, and results of operations. Any adverse determination in litigation could also subject the Combined Company to significant liabilities.
As a foreign private issuer, PubCo is permitted to adopt certain home country practices in relation to corporate governance matters that differ significantly from the Nasdaq corporate governance listing standards. These practices may afford less protection to shareholders than they would enjoy if PubCo complies fully with corporate governance listing standards .
As a foreign private issuer, PubCo has the privilege of utilizing certain provisions within the Nasdaq listing rules that permit adherence to Cayman Islands law for specific governance matters. It is important to note that corporate governance practices in the Cayman Islands may differ markedly from those mandated by corporate governance listing standards. With the exception of general fiduciary duties and duties of care, Cayman Islands law lacks a prescribed corporate governance regime outlining specific standards.
Upon the Closing, PubCo intends to maintain adherence to Cayman Islands corporate governance practices, rather than conforming to the corporate governance requirements stipulated by the Nasdaq. Notably, there is no Cayman Islands legal requirement mandating that PubCo’s board of directors must comprise a majority of independent directors. Similarly, Cayman Islands law does not impose specific obligations regarding the establishment of a compensation committee, audit committee, or the nominating process.
By opting to follow its home country practices, PubCo’s shareholders may receive comparatively less protection than they would under corporate governance listing standards applicable to U.S. domestic issuers.
PubCo may lose its foreign private issuer status in the future, which could result in significant additional costs and expenses.
PubCo operates as a foreign private issuer, exempting it from full compliance with the periodic disclosure and current reporting obligations outlined in the Exchange Act applicable to U.S. domestic issuers. This designation is reassessed annually, typically on the final business day of PubCo’s most recently completed second fiscal quarter. PubCo would lose its foreign private issuer status if, for example, more than 50% of its ordinary shares are directly or indirectly held by residents of the U.S. and it fails to meet additional requirements necessary to maintain its foreign private issuer status.
Should this occur, PubCo would be compelled to adhere to SEC reporting standards applicable to U.S. domestic issuers, necessitating the submission of more comprehensive and detailed periodic reports and registration statements. Furthermore, PubCo would be subject to U.S. federal proxy requirements, and PubCo’s officers, directors, and principal shareholders would fall under the short-swing profit disclosure and recovery provisions outlined in Section 16 of the Exchange Act.
In addition, the loss of PubCo’s foreign private issuer status would result in the forfeiture of exemptions from specific corporate governance obligations outlined in the Nasdaq listing rules. Transitioning into a U.S. listed public company would entail substantial additional legal, accounting, and operational costs not incurred as a foreign private issuer. These include ongoing expenses related to compliance efforts and listing requirements on a U.S. securities exchange.
| 89 |
The consummation of the Business Combination is subject to various conditions, some beyond the involved parties’ control. If these conditions are not met, the Business Combination may not be consummated.
The Merger Agreement contains a number of conditions that must be fulfilled (or waived by the parties) to complete the Business Combination. These conditions include, among other customary conditions, (i) the absence of an enacted or promulgated law by any governmental entity of competent jurisdiction that remains in effect that precludes, restrains, enjoins or prohibits the consummation of the Business Combination, (ii) the absence of any temporary restraining order, preliminary or permanent injunction or any other order in effect precluding, restraining, enjoining, prohibiting, suspending or making illegal the consummation of the Business Combination, (iii) the SEC having declared effective a registration statement on Form F-4 of which this proxy statement/prospectus forms a part and the absence of a stop order suspending such effectiveness issued by the SEC and remaining in effect and the absence of any proceeding initiated for that purpose by the SEC and not subsequently withdrawn, (iv) the approval for listing of PubCo’s securities on Nasdaq, (v) subject to certain materiality exceptions, the accuracy of certain representations and warranties of each of the parties contained in the Merger Agreement and the compliance by each party with the covenants contained in the Merger Agreement, and (vi) the absence of a material adverse effect with respect to each of the parties thereto.
The required satisfaction (or waiver) of the foregoing conditions could delay the completion of the Business Combination for a significant period of time or prevent it from occurring. Further, there can be no assurance that the conditions to the closing of the Business Combination will be satisfied or waived or that the Business Combination will be completed.
The Combined Company may not fully realize the anticipated benefits of the Business Combination or realize such benefits within the timing anticipated.
The parties entered into the Merger Agreement because they believe that the Business Combination will be beneficial to each of Globalink and Alps Holdco and their respective stockholders/shareholders. The Combined Company may not be able to achieve the anticipated long-term strategic benefits of the Business Combination within the timing anticipated or at all. Any delays and challenges that may be encountered in completing the Business Combination or in the post-combination process of consolidation could have an adverse effect on the business and results of operations of the Combined Company, and the value of PubCo ordinary shares after the completion of the Business Combination may be materially and adversely affected.
You may face difficulties in protecting your interests, and your ability to protect your rights through U.S. courts may be limited, because PubCo is incorporated under the law of the Cayman Islands, and after the Business Combination, PubCo intends to conduct its operations and a majority of its directors and executive officers will reside outside of the United States.
PubCo is incorporated under the laws of the Cayman Islands. Upon the completion of the Business Combination, PubCo will conduct a majority of its operations and a majority of its directors and executive officers after the completion of the Business Combination will reside outside of the United States. PubCo’s corporate affairs are governed by PubCo A&R Memorandum and Articles of Association, the Companies Act and the common law of the Cayman Islands. The rights of shareholders to take action against the directors, actions by noncontrolling shareholders and the fiduciary responsibilities of PubCo’s directors to PubCo under Cayman Islands law are to a large extent governed by the common law of the Cayman Islands. The common law of the Cayman Islands is derived in part from comparatively limited judicial precedent in the Cayman Islands as well as from English common law, which has persuasive, but not binding, authority on a court in the Cayman Islands. The rights of PubCo’s shareholders and the fiduciary responsibilities of PubCo’s directors under Cayman Islands law are not as clearly established as they would be under statutes or judicial precedent in some jurisdictions in the United States. In particular, the Cayman Islands has a less developed body of securities laws than the United States and provides significantly less protection to investors. In addition, some U.S. states, such as Delaware, have more fully developed and judicially interpreted bodies of corporate law than the Cayman Islands.
| 90 |
We have been advised by our Cayman Islands legal counsel, that there is uncertainty as to whether the courts of the Cayman Islands would (i) to recognize or enforce against us, judgments of courts of the United States obtained against us or our directors or officers predicated upon the civil liability provisions of the securities laws of the United States or any state in the United States; and (ii) entertain original actions brought in the Cayman Islands, to impose liabilities against us or our directors or officers predicated upon the civil liability provisions of the securities laws of the United States or any state in the United States, so far as the liabilities imposed by those provisions are penal in nature. In those circumstances, although there is currently no statutory enforcement or treaty between the United States and the Cayman Islands providing for enforcement of judgments obtained in the United States, the courts of the Cayman Islands will in certain circumstances recognize and enforce a foreign money judgment of a foreign court of competent jurisdiction without retrial on the merits based on the principle that a judgment of a competent foreign court imposes upon the judgment debtor an obligation to pay the sum for which judgment has been given provided certain conditions are met. For a foreign judgment to be enforced in the Cayman Islands, such judgment must be final and conclusive, given by a court of competent jurisdiction (the courts of the Cayman Islands will apply the rules of Cayman Islands private international law to determine whether the foreign court is a court of competent jurisdiction), and must not be in respect of taxes or a fine or penalty, inconsistent with a Cayman Islands judgment in respect of the same matter, impeachable on the grounds of fraud or obtained in a manner, and or be of a kind the enforcement of which is, contrary to natural justice or the public policy of the Cayman Islands. Furthermore, it is uncertain that Cayman Islands courts would enforce: (1) judgments of U.S. courts obtained in actions against us or other persons that are predicated upon the civil liability provisions of the U.S. federal securities laws; or (2) original actions brought against us or other persons predicated upon the Securities Act.
Ogier (Cayman) LLP has informed us that there is uncertainty with regard to Cayman Islands law relating to whether a judgment obtained from the U.S. courts under civil liability provisions of the securities laws will be determined by the courts of the Cayman Islands as penal, punitive in nature. A Cayman Islands court may stay enforcement proceedings if concurrent proceedings are being brought elsewhere.
Shareholders of Cayman Islands exempted companies such as PubCo have limited rights under Cayman Islands laws to inspect corporate records and accounts or to obtain copies of lists of shareholders of these companies. PubCo’s directors have discretion under PubCo A&R Memorandum and Articles of Association to determine whether or not, and under what conditions, PubCo’s corporate records may be inspected by PubCo’s shareholders, but are not obliged to make them available to PubCo’s shareholders. This may make it more difficult for you to obtain the information needed to establish any facts necessary for a shareholder motion or to solicit proxies from other shareholders in connection with a proxy contest.
As a result of all of the above, public shareholders may have more difficulty in protecting their interests in the face of actions taken by management, members of the board of directors or controlling shareholders than they would as public shareholders of a U.S. company. Therefore, you may not be able to effectively enjoy the protection offered by the U.S. laws and regulations that intend to protect public investors.
Cayman Islands companies may not have standing to initiate a derivative action in a federal court of the United States. As a result, your ability to protect your interests if you are harmed in a manner that would otherwise enable you to sue in a United States federal court may be limited to direct shareholder lawsuits.
You may experience difficulties in effecting service of legal process, enforcing foreign judgments or bringing actions against PubCo or its management named in the proxy statement/prospectus based on foreign laws, and therefore you may not be afforded the same protection as provided to investors in U.S. domestic companies.
PubCo is an exempted company incorporated and registered under the laws of the Cayman Islands. Upon the completion of the Business Combination, PubCo will conduct its operations, through its subsidiary, Alps, which is located in Malaysia and a majority of its post-combination directors and executive officers, will also reside outside of the United States. As a result, it may be difficult or impossible for you to bring an action against PubCo or against these individuals in the United States in the event that you believe that your rights have been infringed under the U.S. federal securities laws or otherwise. It may also be difficult for you to enforce judgments obtained in U.S. courts based on the civil liability provisions of the U.S. federal securities laws against PubCo and its officers and directors. Even if you are successful in bringing an action of this kind, the laws of the Cayman Islands and of Malaysia may render you unable to enforce a judgment against PubCo, its assets, directors and officers or their assets. Therefore, you may not be able to enjoy the same protection provided by various U.S. authorities as it is provided to investors in U.S. domestic companies.
Management has been advised that the Cayman Islands and Malaysia do not have treaties with the United States for the reciprocal recognition and enforcement of court judgments. Additionally, it is uncertain whether the existing extradition treaties between the United States and Malaysia would allow for the effective enforcement of criminal penalties under U.S. federal securities laws.
Additionally, PubCo’s corporate affairs will be governed by the PubCo A&R Memorandum and Articles, the Companies Act, and the common law of the Cayman Islands. The rights of shareholders to take action against directors, the actions of minority shareholders, and the fiduciary duties of PubCo’s directors to PubCo are largely governed by Cayman Islands common law. The rights of PubCo’s shareholders and the fiduciary duties of PubCo’s directors under Cayman Islands law may not be as well-defined as under statutes or judicial precedent in certain U.S. jurisdictions. Specifically, the Cayman Islands has different securities laws than the United States. Some U.S. states, such as Delaware, have more extensively developed and judicially interpreted corporate laws than the Cayman Islands. Furthermore, Cayman Islands companies may not have the standing to initiate shareholder derivative actions in U.S. federal courts.
Shareholders of Cayman Islands exempted companies like PubCo have limited rights under Cayman Islands law to inspect corporate records. PubCo’s directors have discretion under the PubCo A&R Memorandum and Articles to decide whether and under what conditions PubCo’s corporate records may be inspected by its shareholders. PubCo is not required to make these records available to shareholders, except in limited circumstances as required by the Companies Act. This may make it more difficult for shareholders to obtain the information necessary to support a shareholder motion or to solicit proxies for a proxy contest. See “Description of PubCo’s Securities—Inspection of Books and Records.”
Certain corporate governance practices in the Cayman Islands, which is PubCo’s home country, differ significantly from requirements for companies incorporated in other jurisdictions such as the United States. If PubCo chooses to follow home country practices regarding corporate governance matters, its shareholders may be afforded less protection than they otherwise would under rules and regulations applicable to U.S. domestic issuers.
As a result of all the above, PubCo’s shareholders may have more difficulty in protecting their interests in the face of actions taken by management, members of the board of directors or controlling shareholders than they would as public shareholders of a company incorporated in the United States.
PubCo may be or become a passive foreign investment company (“PFIC”), which could result in adverse U.S. federal income tax consequences to U.S. Holders.
If PubCo or any of its subsidiaries is a PFIC for any taxable year, or portion thereof, that is included in the holding period of a U.S. Holder of PubCo ordinary shares or PubCo warrants, such U.S. Holder may be subject to certain adverse U.S. federal income tax consequences and may be subject to additional reporting requirements. There is no assurance that PubCo or its subsidiaries are not currently PFICs for U.S. federal income tax purposes for the taxable year of the Business Combination or for foreseeable future taxable years. Moreover, PubCo does not expect to provide a PFIC annual information statement for 2024 or going forward, which will preclude U.S. Holders from making or maintaining a “qualified electing fund” election under Section 1295 of the Code. Please see the section titled “Material U.S. Federal Income Tax Consequences — Passive Foreign Investment Company Status” for a more detailed discussion with respect to PubCo’s potential PFIC status and certain tax implications thereof. U.S. Holders are urged to consult their tax advisors regarding the possible application of the PFIC rules to holders of PubCo securities.
| 91 |
The IRS may not agree with the position that PubCo should be treated as a foreign corporation for U.S. federal income tax purposes following the Business Combination, which could have a material adverse effect on PubCo’s financial position and results from operations and on non-U.S. holders of PubCo securities.
Although PubCo will be incorporated under the laws of the Cayman Islands, the IRS may assert that PubCo should be treated as a U.S. corporation (and, therefore, a U.S. tax resident) for U.S. federal income tax purposes pursuant to section 7874 of the Code. For U.S. federal income tax purposes, a corporation is generally considered a tax resident in the jurisdiction of its organization or incorporation. Because PubCo will be incorporated under the laws of the Cayman Islands, PubCo would generally be classified as a foreign corporation (and, therefore, a non-U.S. tax resident) for U.S. federal income tax purposes. Section 7874 provides an exception pursuant to which a foreign incorporated entity may, in certain circumstances, be treated as a U.S. corporation for U.S. federal income tax purposes. These rules are complex and require analysis of all relevant facts and circumstances, and there is limited guidance and significant uncertainties as to their application. If it were determined that PubCo should be taxed as a U.S. corporation for U.S. federal income tax purposes under section 7874, PubCo would be subject to U.S. federal income tax on its taxable income like any other U.S. corporation and certain distributions made by PubCo to non-U.S. holders of PubCo securities would be subject to U.S. withholding tax at the rate of 30% or such lower rate as provided by an applicable treaty. Taxation as a U.S. corporation could also have a material adverse effect on PubCo’s financial position and results from operations.
As more fully described under “Material U.S. Federal Income Tax Consequences — Certain U.S. Federal Income Tax Consequences of the Business Combination to Globalink and PubCo — Tax Residence of PubCo for U.S. Federal Income Tax Purposes,” PubCo intends to take the position that section 7874 is currently expected to apply in a manner such that PubCo should not be treated as a U.S. corporation for U.S. federal income tax purposes, based on its position that after completion of the Business Combination, former Globalink stockholders will own, by reason of owning (or being treated as owning) shares of Globalink common stock, less than 80% of the voting power and value of the PubCo securities (the “Ownership Test”). However, PubCo’s position depends in part on the position that the Ownership Test is determined after the Business Combination rather than immediately after the Redomestication Merger for purposes of Section 7874 of the Code. Further, holders are cautioned that the application of section 7874 to PubCo is extremely complex and the applicable Treasury Regulations are subject to significant uncertainty and there is limited guidance regarding their application. Moreover, the application of section 7874 to the facts and circumstances of the proposed transaction are uncertain. In addition, there could be a future change in law under section 7874, the Treasury Regulations promulgated thereunder or otherwise that could have an effect on the application of section 7874 to PubCo. No IRS ruling has been requested or will be obtained regarding the U.S. federal income tax consequences of the Business Combination or any other matter described in this proxy statement/prospectus. The IRS may not agree with PubCo’s position that it should be treated as a non-U.S. corporation for U.S. federal income tax purposes, and there can be no assurance that, if challenged, such treatment will be sustained by a court.
Changes to, or changes in interpretations of, tax laws could have a material adverse effect on the PubCo’s business, financial condition and results of operations.
PubCo is subject to income taxes and non-income taxes in the United States and other countries in which it transacts or conducts business, and such laws and rates vary by jurisdiction. Tax laws and regulations, including at non-U.S. and U.S. federal and local jurisdictions, frequently change, especially in relation to the interpretation of existing tax laws for new and emerging industries, and PubCo cannot always reasonably predict the impact from, or the ultimate cost of compliance with, current or future tax laws.
| 92 |
Any changes in the taxation of PubCo’s business activities may increase its worldwide effective tax rate and harm its business, financial condition and results of operations. PubCo’s tax expense could also be impacted by the applicability of withholding taxes and the impact of changes in the evaluation of tax positions PubCo has taken in prior tax periods. The amount of taxes PubCo pays in these jurisdictions could increase substantially as a result of changes in the applicable tax principles, including increased tax rates, new tax laws or revised interpretations of existing tax laws and precedents, which could harm PubCo’s liquidity and results of operations.
All statements contained herein concerning U.S. federal income or other tax consequences are based on existing law and interpretations thereof. The tax regimes to which PubCo is subject or under which PubCo operates, including income and non-income taxes, are unsettled and may be subject to significant change. While some of these changes could be beneficial, others could negatively affect PubCo’s after-tax returns. Accordingly, no assurance can be given that the currently anticipated tax treatment will not be modified by legislative, judicial or administrative changes, possibly with retroactive effect. In addition, no assurance can be given that any tax authority or court will agree with any particular interpretation of the relevant laws.
PubCo may adopt share incentive plans in the future, which may adversely affect PubCo’s results of operations.
PubCo may adopt share-based incentive plan in the future, and grant share-based awards to its employees, directors and consultants to incentivize their performance and align their interests with that of PubCo. If PubCo adopts share-based incentive plan and grant share-based compensation in the future, it will be required to account such awards for share-based compensation expenses in accordance with the applicable accounting standards. The Financial Accounting Standards Board Accounting Standards Codification Topic 718, Compensation — Stock Compensation generally requires a company to recognize, as an expense, the fair value of share options and other equity incentives to employees based on the fair value of equity awards on the date of the grant, with the compensation expense recognized over the period in which the recipient is required to provide service in exchange for the equity award. If PubCo adopts any such share incentive plan and grant options or other equity incentives in the future, such grants could have dilutive impact on PubCo’s existing shareholders, and cause PubCo to incur significant compensation charges and its results of operations could be adversely affected.
Risks Related to Redemption
Globalink’s public stockholders who wish to redeem their public shares of Globalink common stock for a pro rata portion of the Trust Account must comply with specific requirements for redemption that may make it more difficult for them to exercise their redemption rights prior to the deadline. If stockholders fail to comply with the redemption requirements specified in this proxy statement/prospectus, they will not be entitled to redeem their public shares of Globalink common stock for a pro rata portion of the funds held in the Trust Account.
A public stockholder will be entitled to receive cash for any public shares to be redeemed only if such public stockholder: (1)(a) holds shares of Globalink common stock, or (b) if he, she or it holds shares of Globalink common stock through units, such public stockholder elects to separate its units into the underlying shares of Globalink common stock, rights and warrants prior to exercising his, her or its redemption rights with respect to the shares of Globalink common stock; (2) prior to , Eastern Time, on , 2025 (two business days before the scheduled date of the Special Meeting) submits a written request to Continental, Globalink’s transfer agent, that Globalink redeem all or a portion of his, her or its shares of Globalink common stock for cash; and (3) delivers his, her or its shares of Globalink common stock to Globalink’s transfer agent physically or electronically through DTC. In order to obtain a physical share certificate, a stockholder’s broker or clearing broker, DTC and Globalink’s transfer agent will need to act to facilitate this request. It is our understanding that stockholders should generally allot at least two weeks to obtain physical certificates from Globalink’s transfer agent. However, because we do not have any control over this process or over DTC, it may take significantly longer than two weeks to obtain a physical stock certificate. If it takes longer than anticipated to obtain a physical certificate, public stockholders who wish to redeem their shares of Globalink common stock may be unable to obtain physical certificates by the deadline for exercising their redemption rights and thus will be unable to redeem their shares of Globalink common stock.
| 93 |
If a public stockholder of Globalink fails to receive notice of Globalink’s offer to redeem shares of Globalink common stock in connection with the Business Combination or fails to comply with the procedures for tendering its shares, such shares of Globalink common stock may not be redeemed.
If, despite Globalink’s compliance with the proxy rules, a public stockholder fails to receive Globalink’s proxy materials, such public stockholder may not become aware of the opportunity to redeem his, her or its public shares of Globalink common stock. In addition, the proxy materials that Globalink is furnishing to holders of public shares in connection with the Business Combination describe the various procedures that must be complied with in order to validly redeem the public shares of Globalink common stock. In the event that a public stockholder fails to comply with these procedures, its public shares of Globalink common stock may not be redeemed. Please see the section entitled “The Special Meeting — Redemption Rights” for additional information on how to exercise your redemption rights.
If you or a “group” of stockholders of which you are a part are deemed to hold an aggregate of more than 20% of the public shares of Globalink common stock, you (or, if a member of such a group, all of the members of such group in the aggregate) will lose the ability to redeem all such shares in excess of 20% of the public shares of Globalink common stock.
A public stockholder of Globalink common stock, together with any of his, her or its affiliates or any other person with whom it is acting in concert or as a “group” (as defined under Section 13 of the Exchange Act), will be restricted from redeeming in the aggregate his, her or its shares or, if part of such a group, the group’s shares, in excess of 20% of the public shares of Globalink common stock. In order to determine whether a stockholder is acting in concert or as a group with another stockholder, we will require each public stockholder seeking to exercise redemption rights to certify to Globalink whether such stockholder is acting in concert or as a group with any other stockholder. Such certifications, together with other public information relating to stock ownership available to Globalink at that time, such as Section 13D, Section 13G and Section 16 filings under the Exchange Act, will be the sole basis on which Globalink makes the above-referenced determination. Your inability to redeem any such excess shares of Globalink common stock will reduce your influence over Globalink’s ability to consummate the Business Combination and you could suffer a material loss on your investment in Globalink if you sell such excess shares in open market transactions.
There is no guarantee that a stockholder’s decision whether to redeem its shares for a pro rata portion of the Trust Account will put the stockholder in a better future economic position.
We can give no assurance as to the price at which a shareholder may be able to sell his, her or its shares of the PubCo ordinary shares in the future following the Closing or any alternative business combination. Certain events following the consummation of any initial business combination, including the Business Combination, may cause an increase in the price of the PubCo ordinary shares, and may result in a lower value realized now than a shareholder of the Combined Company might realize in the future had the shareholder not redeemed its shares. Similarly, if a shareholder does not redeem its shares, the shareholder will bear the risk of ownership of the public ordinary shares after the consummation of any initial business combination, and there can be no assurance that a shareholder can sell its shares of the PubCo ordinary shares in the future for a greater amount than the redemption price set forth in this proxy statement/prospectus. A shareholder should consult the shareholder’s own tax and/or financial advisor for assistance on how this may affect his, her or its individual situation.
| 94 |
THE SPECIAL MEETING
General
We are furnishing this proxy statement/prospectus to the Globalink stockholders as part of the solicitation of proxies by the Board for use at the Special Meeting of Globalink stockholders to be held on , 2025 and at any adjournment or postponement thereof. This proxy statement/prospectus is first being furnished to Globalink’s stockholders on or about , 2025 in connection with the vote on the Proposals. This proxy statement/prospectus provides you with the information you need to know to be able to vote or instruct your vote to be cast at the Special Meeting.
Date, Time and Place
The Special Meeting will be held virtually at a.m., Eastern Time, on , 2025 and conducted exclusively via live webcast at , or such other date, time and place to which such meeting may be adjourned or postponed, for the purposes set forth in the accompanying notice. There will not be a physical location for the Special Meeting, and you will not be able to attend the meeting in person. We are pleased to utilize the virtual stockholder meeting technology to provide ready access and cost savings for Globalink and its stockholders. The virtual meeting format allows attendance from any location in the world. You will be able to attend, vote your shares, view the list of stockholders entitled to vote at the Special Meeting and submit questions during the Special Meeting via a live webcast available at .
Virtual Special Meeting Registration
To register for the virtual meeting, please follow these instructions as applicable to the nature of your ownership of shares of Globalink common stock.
If your shares are registered in your name with Continental and you wish to attend the online-only virtual meeting, go to , enter the control number you received on your proxy card and click on the “Click here” to preregister for the online meeting link at the top of the page. Just prior to the start of the meeting you will need to log back into the meeting site using your control number. Pre-registration is recommended but is not required in order to participate in the virtual Special Meeting.
Beneficial stockholders who wish to participate in the online-only virtual meeting must obtain a legal proxy by contacting their account representative at the bank, broker, or other nominee that holds their shares and email a copy (a legible photograph is sufficient) of their legal proxy to . Beneficial stockholders who email a valid legal proxy will be issued a meeting control number that will allow them to register to attend and participate in the online-only meeting. After contacting , a beneficial holder will receive an email prior to the meeting with a link and instructions for entering the virtual Special Meeting. Beneficial stockholders should contact at least two business days prior to the meeting date.
Accessing the Virtual Special Meeting Webcast
You will need your control number for access. If you do not have your control number, contact at the phone number or email address below. Beneficial investors who hold shares through a bank, broker or other intermediary, will need to contact them and obtain a legal proxy. Once you have your legal proxy, contact to have a control number generated. Continental contact information is as follows: or email .
Purpose of the Special Meeting
At the Special Meeting, Globalink is asking its stockholders to approve the following Proposals:
| ● | The Redomestication Merger Proposal (Proposal 1). |
| ● | The Acquisition Merger Proposal (Proposal 2). A copy of the Merger Agreement is attached to the proxy statement/prospectus as Annex A. |
| ● | The Nasdaq Proposals (Proposals 3). |
| ● | The Adjournment Proposal (Proposal 4). |
| 95 |
You will be entitled to vote or direct votes to be cast at the Special Meeting if you own shares of Globalink common stock at the close of business on , 2025, which is the Record Date. You are entitled to one vote for each share of Globalink common stock that you owned as of the close of business on the Record Date. If your shares are held in “street name” or are in a margin or similar account, you should contact your broker, bank or other nominee to ensure that votes related to the shares you beneficially own are properly counted. On the Record Date, there were 3,722,511 shares of Globalink common stock outstanding, of which 277,511 are public shares and 2,875,000 are Founder Shares held by the Initial Stockholders.
Vote of the Sponsor, Directors and Officers
In connection with the IPO, Globalink entered into agreements with each of its Initial Stockholders, private investor, directors and officers, pursuant to which each agreed to vote any shares of Globalink common stock owned by them in favor of the Redomestication Merger Proposal, the Acquisition Merger Proposal, and for all other Proposals presented at the Special Meeting. These agreements apply to Globalink’s initial stockholders as it relates to the Founder Shares and the requirement to vote such shares in favor of the Redomestication Merger Proposal, the Acquisition Merger Proposal and for all other Proposals presented to Globalink stockholders in this proxy statement/prospectus.
Globalink’s initial stockholders have waived any redemption rights, including with respect to shares of Globalink common stock issued or purchased in the IPO or in the aftermarket, in connection with Business Combination. The Founder Shares held by the Sponsor have no redemption rights upon Globalink’s liquidation and will be worthless if no business combination is effected by Globalink by January 9, 2025 (and may be extended to June 9, 2025 on a monthly basis by resolutions of the Globalink Board, if requested by the Sponsor, and subject to the Sponsor depositing additional funds into the Trust Account).
On December 10, 2024, Globalink received a delisting determination letter from Nasdaq as the 36-month anniversary from its IPO has passed on December 9, 2024, and Globalink are no longer in compliance with Nasdaq Listing Rule IM 5101-2(b). Globalink’s securities were suspended from trading and delisted from Nasdaq on December 17, 2024. As of the same date, Globalink’s securities started trading on OTC Pink. There could be substantial risks to Globalink and PubCo the as a result of Globalink’s non-compliance with this rule. See “Risk Factors — Risks Related to Globalink’s Business and the Business Combination —Our securities were suspended from trading and delisted from Nasdaq on December 17, 2024, following receipt of a delisting determination letter from Nasdaq on December 10, 2024. This could have significant material adverse consequences on us and our securities, including that it will negatively impact our ability to complete a business combination, will limit investors’ ability to make transactions in our securities and could subject us to additional trading restrictions.”
Quorum and Required Vote for Proposals
A quorum of Globalink stockholders is necessary to hold a valid meeting. A quorum will be present at the Special Meeting if a majority of the shares of Globalink common stock outstanding and entitled to vote at the Special Meeting is represented in person online or by proxy at the Special Meeting.
The approval of the remaining Proposals (including the Redomestication Merger Proposal, the Acquisition Merger Proposal, the Nasdaq Proposals, and the Adjournment Proposal) each require the affirmative vote of the holders of a majority of the shares of Globalink common stock cast by the stockholders represented in person online or by proxy and entitled to vote thereon at the Special Meeting.
If either the Redomestication Merger Proposal or the Acquisition Merger Proposal is not approved, the Nasdaq Proposals will not be presented to the Globalink stockholders for a vote. The Nasdaq Proposals are conditioned on the approval of the Redomestication Merger Proposal and the Acquisition Merger Proposal. The Redomestication Merger Proposal and the Acquisition Merger Proposal generally are preconditions to the consummation of the Business Combination. The Adjournment Proposal is not conditioned on the approval of any other Proposal set forth in this proxy statement/prospectus.
| 96 |
It is important for you to note that in the event either the Redomestication Merger Proposal or the Acquisition Merger Proposal does not receive the requisite vote for approval, Globalink will not consummate the Business Combination. If Globalink does not consummate the Business Combination and fails to complete an initial business combination by January 9, 2025 (and may be extended to June 9, 2025 on a monthly basis by resolutions of the Globalink Board, if requested by the Sponsor, and subject to the Sponsor depositing additional funds into the Trust Account), Globalink will be required to dissolve and liquidate its Trust Account by returning the then-remaining funds in such account to the public stockholders.
Abstentions and Broker Non-Votes
Abstentions will have no effect on the outcome of the vote on the Redomestication Merger Proposal, the Acquisition Merger Proposal, the Nasdaq Proposals and Adjournment Proposal.
Recommendation of the Board
The Board:
| ● | has determined that each of the Redomestication Merger Proposal, the Acquisition Merger Proposal, the Nasdaq Proposals, and the Adjournment Proposal, are fair to, and in the best interests of, Globalink and its stockholders; | |
| ● | has approved the Redomestication Merger Proposal, the Acquisition Merger Proposal, the Nasdaq Proposals, and the Adjournment Proposal; and | |
| ● | recommends that the Globalink stockholders vote “FOR” each of the Redomestication Merger Proposal, the Acquisition Merger Proposal, the Nasdaq Proposals, and the Adjournment Proposal. |
When you consider the recommendation of the Board in favor of approval of the Proposals, you should keep in mind that the Sponsor, members of the Board and Globalink’s officers have interests in the Business Combination that may be different from or in addition to (or which may conflict with) your interests as a stockholder. These interests include, among other things:
| ● | The Initial Stockholders, including the officers and directors of Globalink, have waived their right to redeem any shares of Globalink common stock in connection with a stockholder vote to approve a proposed initial business combination or sell any shares of Globalink common stock to Globalink in a tender offer prior to a proposed initial business combination, or to receive distributions with respect to the Founder Shares upon the liquidation of the Trust Account if Globalink is unable to consummate a business combination. If an initial business combination, such as the Business Combination, is not completed by January 9, 2025 (and may be extended to June 9, 2025 on a monthly basis by resolutions of the Globalink Board, if requested by the Sponsor, and subject to the Sponsor depositing additional funds into the Trust Account), Globalink will be required to dissolve and liquidate. In such event, the 2,875,000 Founder Shares currently held by the Initial Stockholders, which were acquired prior to the IPO will be worthless because such holders have agreed to waive their rights to any liquidation distributions through the Insider Letter, in consideration for inducing Chardan, the representative of the underwriters for Globalink’s IPO to proceed with the IPO, and the economic benefit the IPO would confer upon the Initial Stockholders and PGM. The Founder Shares were purchased for an aggregate purchase price of $25,000, or less than $0.01 per share, and had a total market value of $34.36 million as of December 12, 2024. Accordingly, the Initial Stockholders will receive a positive rate of return so long as the market price of the Globalink common stock is at least $0.01 per share, even if public stockholders experience a negative rate of return in the Combined Company. |
| 97 |
| ● | The fact that the Initial Stockholders’ Founder Shares became subject to a lock-up whereby, subject to certain limited exceptions, the Initial Stockholders agree not to offer, sell, contract to sell, pledge or otherwise dispose of, directly or indirectly, any of the Lock-up Shares, enter into a transaction that would have the same effect, or enter into any swap, hedge or other arrangement that transfers, in whole or in part, any of the economic consequences of ownership of the Lock-Up Shares or otherwise, publicly disclose the intention to make any offer, sale, pledge or disposition, or to enter into any transaction, swap, hedge or other arrangement, or engage in any Short Sales (as defined in the Lock-up Agreement) with respect to the Lock-Up Shares until (i) (w) with respect to one hundred percent (100%) of the Lock-Up Shares, the date from the Closing until the last date that is six (6) months after the date of the Closing, (x) with respect to ninety percent (90%) of the Lock-Up Shares, the 1st day of the 7th month from the Closing until the date that is nine (9) months after the date of the Closing, (y) with respect to seventy percent (70%) of the Lock-Up Shares, the 1st day of the 10th month from the Closing until the date that is twelve (12) months after the date of the Closing, (z) with respect to forty percent (40%) of the Lock-Up Shares, the 1st day of the 13th month from the Closing until the date that is fifteen (15) months after the date of the Closing, or (ii) if sooner, the date after the Closing on which Globalink consummates a liquidation, merger, share exchange or other similar transaction with an unaffiliated third party that results in all of Globalink’s stockholders having the right to exchange their equity holdings in Globalink for cash, securities or other property. For the avoidance of doubt, no Lock-up Shares shall be subject to the terms in the Lock-up Agreement from and after the date that is fifteen (15) months after the date of the Closing; | |
| ● | The exercise of Globalink’s directors’ and officers’ discretion in agreeing to changes or waivers in the terms of the Business Combination may result in a conflict of interest when determining whether such changes or waivers are appropriate and in Globalink stockholders’ best interest. Globalink’s directors were aware of and considered these interests, among other matters, in evaluating the Business Combination, and in recommending to Globalink stockholders that they approve the Business Combination. Further, the interests of Globalink’s officers or directors may be different from or in addition to (and which may conflict with) your interests and may be incentivized to complete a less favorable business combination rather than liquidating Globalink. Public stockholders should take these interests into account in deciding whether to approve the Business Combination. | |
| ● | As of September 30, 2024, Globalink’s officers and directors have incurred an aggregate of $11,798 in out-of-pocket expenses. On each of September 5, 2023, September 29, 2023 and November 7, 2023, an affiliate of the Sponsor advanced $130,000 to the Company, for a total advance of $390,000, for Globalink’s extension payments. Unless Globalink consummates an initial business combination, Globalink’s officers, directors and the Sponsor (including its affiliates) will not receive reimbursement for any out-of-pocket expenses incurred by them to the extent that such expenses exceed the amount of available proceeds not deposited in the Trust Account. Upon the consummation of an initial business combination, Globalink’s offices, directors, and the Sponsor (including its affiliates) would be entitled to reimbursement for out-of-pocket expenses incurred by them from the Trust Account disbursements after redemption payouts and payments of deferred underwriting discounts. | |
| ● | The fact that, if the Trust Account is liquidated, including in the event Globalink is unable to complete an initial business combination within the required time period, the Sponsor has agreed to be liable to Globalink if and to the extent any claims by a vendor for services rendered or products sold to Globalink, or a prospective target business with which Globalink has discussed entering into a transaction agreement, reduce the amount of funds in the Trust Account. This liability will not apply with respect to any claims by a third party who executed a waiver of any right, title, interest or claim of any kind in or to any monies held in the Trust Account or to any claims under Globalink’s indemnity of the underwriters of the IPO against certain liabilities, including liabilities under the Securities Act. | |
| ● | As of the date hereof, the deadline that Globalink has to consummate its initial business combination has been extended for a total of eighteen times, and a total of US$1,950,000 has been deposited into the Trust Account in connection with the previous extensions. In connection with funding the extensions, Globalink has issued an aggregate of US$3.85 million in promissory notes to PGM, which are payable upon the Closing. In the event of a liquidation, PGM has waived its rights to any liquidation distributions through the Insider Letter. PGM is an affiliate of the Sponsor as its 95% equity holder has a familial relationship with the control person of the Sponsor. As a result, the Sponsor has an additional interest in causing Globalink to consummate its initial business combination due to its familial relationship with PGM. | |
| ● | After the Closing, the Sponsor will have the sole authority to handle various matters relating to the Escrow Account and indemnifications under the Merger Agreement. The Sponsor may have a conflict of interest when exercising such authority to handle matters relating to the Escrow Account and indemnifications after Closing, and they may not make decisions in the Globalink stockholders’ best interests. For details, see “Summary of the Proxy Statement/Prospectus—Parties to the Business Combination—Parent Representative.” |
These interests may have influenced Globalink’ directors in making their recommendation that you vote in favor of the approval of the Business Combination.
Upon the consummation of an initial business combination by Globalink, other than the PubCo ordinary shares that the Initial Stockholders are entitled to upon conversion of their shares of Globalink common stock at the completion of the Redomestication Merger, no additional compensation will be awarded to, earned by, or paid to the Sponsor, its affiliates or any promoters for all services rendered or to be rendered to Globalink and its affiliates. The Sponsor is expected to own an equity interest of approximately 1.7% in the Combined Company after the consummation of the Business Combination.
Prior to the consummation of an initial business combination by Globalink, the insider shares can only be transferred by the Initial Stockholders to (1) the Initial Stockholders or their respective affiliates or members, or to Globalink’s officers, directors, advisors and employees, (2) the Initial Stockholder’s affiliates or its members upon its liquidation, (3) to relatives and trusts for estate planning purposes, (4) by virtue of the laws of descent and distribution upon death of an Initial Stockholder, (5) pursuant to a qualified domestic relations order, or (6) by private sales made at prices no greater than the price at which the Insider Shares were originally purchased.
Record Date; Who is Entitled to Vote
Globalink has fixed the close of business on , 2025, as the Record Date for determining those Globalink stockholders entitled to notice of and to vote at the Special Meeting. As of the close of business on , 2025, there were shares of Globalink common stock issued and outstanding and entitled to vote, of which are shares of Globalink common stock held by public stockholders, are shares held by the Initial Stockholders. Each holder of shares of Globalink common stock is entitled to one vote per share on each Proposal. If your shares are held in “street name,” you should contact your broker, bank or other nominee to ensure that shares held beneficially by you are voted in accordance with your instructions.
In connection with its IPO, Globalink entered into certain letter agreements pursuant to which the Initial Stockholders and PGM, an affiliate of the Sponsor, agreed to vote any shares of Globalink common stock owned by them in favor of Globalink’s initial business combination. The Initial Stockholders and PGM also entered into the Globalink Support Agreement, pursuant to which they agreed to, among other things, vote in favor of the Redomestication Merger Proposal, the Acquisition Merger Proposal, and the other Proposals. As of the date of this proxy statement/prospectus, the Initial Stockholders and PGM together hold approximately 92.6% of the outstanding shares of common stock.
| 98 |
Voting Your Shares
Each share of Globalink common stock that you own in your name entitles you to one vote on each Proposal for the Special Meeting. Your proxy card shows the number of shares of common stock that you own.
Each share of common stock that you own in your name entitles you to one vote. If you are a record owner of your shares, there are three ways to vote your shares of common stock at the Special Meeting:
| 1. | Vote by Internet. |
| ● | Before the meeting: Go online to www.cstproxyvote.com. Use the Internet to transmit your voting instructions and for electronic delivery information up until 11:59 p.m. Eastern Time the day before the meeting date. Have your proxy card in hand when you access the website and follow the instructions to obtain your records and to create an electronic voting instruction form. If you hold your shares in “street name,” which means your shares are held of record by a broker, bank or other nominee, you should follow the instructions provided by your broker, bank or nominee to ensure that votes related to the shares you beneficially own are properly counted. In this regard, you must provide the record holder of your shares with instructions on how to vote your shares. However, if your shares are held in the name of your broker, bank or other nominee, you must get a proxy from the broker, bank or other nominee. That is the only way we can be sure that the broker, bank or nominee has not already voted your shares of common stock. | |
| ● | During the meeting: Go online to https://www.cstproxy.com/ . You will be able to attend the Special Meeting online and vote your shares electronically until voting is closed. If you hold your shares in “street name,” which means your shares are held of record by a broker, bank or other nominee, you should follow the instructions provided by your broker, bank or nominee to ensure that votes related to the shares you beneficially own are properly counted. In this regard, you must provide the record holder of your shares with instructions on how to vote your shares. However, if your shares are held in the name of your broker, bank or other nominee, you must get a proxy from the broker, bank or other nominee. That is the only way we can be sure that the broker, bank or nominee has not already voted your shares of common stock. |
| 2. | Vote by mail. Mark, date, sign and mail promptly the enclosed proxy card (a postage-paid envelope is provided for mailing in the United States). By signing the proxy card and returning it in the enclosed prepaid and addressed envelope, you are authorizing the individuals named on the proxy card to vote your shares at the Special Meeting in the manner you indicate. You are encouraged to sign and return the proxy card even if you plan to attend the Special Meeting so that your shares will be voted if you are unable to attend the Special Meeting. If you receive more than one proxy card, it is an indication that your shares are held in multiple accounts. Please sign and return all proxy cards to ensure that all of your shares are voted. If you hold your shares in “street name” through a bank, broker or other nominee, you will need to follow the instructions provided to you by your bank, broker or other nominee to ensure that your shares are represented and voted at the Special Meeting. If you sign and return the proxy card but do not give instructions on how to vote your shares, your shares of common stock will be voted as recommended by the Board. The Board recommends voting “FOR” the Proposals. Votes submitted by mail must be received by 11:59 p.m., Eastern Time, , 2025. |
| 3. | Vote by telephone. You may vote by proxy by calling and following the instructions on the proxy card. If you hold your shares in “street name,” which means your shares are held of record by a broker, bank or other nominee, you should follow the instructions provided by your broker, bank or nominee to ensure that votes related to the shares you beneficially own are properly counted. In this regard, you must provide the record holder of your shares with instructions on how to vote your shares. However, if your shares are held in the name of your broker, bank or other nominee, you must get a proxy from the broker, bank or other nominee. That is the only way we can be sure that the broker, bank or nominee has not already voted your shares of common stock. |
| 99 |
Revoking Your Proxy
If you give a proxy, you may revoke it at any time before it is exercised by doing any one of the following:
| ● | submitting a valid, later-dated proxy card or voting instructions via the Internet or by telephone before 11:59 p.m., Eastern Time, on the calendar day immediately preceding the Special Meeting, or by mail that is received prior to the Special Meeting; or | |
| ● | sending a written revocation of a proxy to Globalink’s proxy agent at , that bears a date later than the date of the proxy you want to revoke and is received prior to the date of the Special Meeting; or Special Meeting you may attend the Special Meeting, revoke your proxy, and vote during the virtual Special Meeting, as indicated above. |
Who Can Answer Your Questions About Voting Your Shares
If you have any questions about how to vote or direct a vote in respect of your shares of Globalink common stock, you may call Okapi Partners LLC, Globalink’s proxy solicitor, with individual call toll-free at (855) 305-0857 and banks and brokers call at (212) 297-0720.
No Additional Matters May Be Presented at the Special Meeting
The Special Meeting has been called only to consider the approval of the Redomestication Merger Proposal, the Acquisition Merger Proposal, the Nasdaq Proposals, and the Adjournment Proposal. Under Globalink Charter, other than procedural matters incident to the conduct of the Special Meeting, no other matters may be considered at the Special Meeting if they are not included in the notice of the Special Meeting.
Redemption Rights
Pursuant to Globalink Charter, a holder of shares of Globalink common stock has the right to redeem such shares for cash equal to its pro rata share of the Trust Account (net of taxes payable) in connection with a business combination.
If you are a public stockholder and you seek to have your shares redeemed, you must submit your request in writing that Globalink redeems your shares of Globalink common stock for cash no later than p.m., Eastern Time on , 2025 (at least two business days before the Special Meeting). The request must be signed by the applicable stockholder in order to validly request redemption. A stockholder is not required to submit a proxy card or vote in order to validly exercise redemption rights. The request must identify the holder of shares of common stock to be redeemed and must be sent to Continental at the following address:
Continental Stock Transfer & Trust Company
1
State Street, 30th floor
New York, NY 10004
Attn: SPAC Redemption Team
E-mail: spacredemptions@continentalstock.com
You may tender the shares of Globalink common stock for which you are electing redemption by two (2) business days before the Special Meeting by either:
| ● | delivering certificates representing shares of Globalink common stock to Continental, or | |
| ● | delivering the shares of Globalink common stock electronically through the DWAC system. |
| 100 |
Globalink stockholders will be entitled to redeem their shares of Globalink common stock for a full pro rata share of the Trust Account (anticipated to be no less than approximately $11.98 per share based on the balance of the Trust Account as of December 6, 2024).
Any corrected or changed written demand of redemption rights must be received by Continental two (2) business days prior to the Special Meeting. No demand for redemption will be honored unless the holder’s shares have been delivered (either physically or electronically) to Continental at least two (2) business days prior to the vote at the Special Meeting.
Public stockholders may seek to have their shares redeemed regardless of whether they vote for or against the Business Combination and whether or not they are holders of shares of Globalink common stock as of the Record Date. Any public stockholder who holds shares of Globalink common stock on or before , 2025 (two (2) business days before the Special Meeting) will have the right to demand that his, her or its shares be redeemed for a pro rata share of the aggregate amount then on deposit in the Trust Account, less any taxes then due but not yet paid, at the consummation of the Business Combination.
In connection with tendering your shares for redemption, you must elect either to physically tender your share certificates to Globalink’s transfer agent or deliver your shares to the transfer agent electronically using The Depository Trust Company’s DWAC System, in each case, two (2) business days before the Special Meeting.
If you wish to tender through the DWAC system, please contact your broker and request delivery of your shares through the DWAC system. Delivering shares physically may take significantly longer. In order to obtain a physical certificate, a stockholder’s broker and/or clearing broker, DTC, and Continental will need to act together to facilitate this request. It is Globalink’s understanding that stockholders should generally allot at least two weeks to obtain physical certificates from Continental. Globalink does not have any control over this process or over the brokers or DTC, and it may take longer than two weeks to obtain a physical certificate. Stockholders who request physical certificates and wish to redeem may be unable to meet the deadline for tendering their shares before exercising their redemption rights and thus will be unable to redeem their shares.
In the event that a stockholder tenders its shares and decides prior to the consummation of the Business Combination that it does not want to redeem its shares, the stockholder may withdraw the tender. In the event that a stockholder tenders shares and the Business Combination is not completed, these shares will not be redeemed for cash and the physical certificates representing these shares will be returned to the stockholder promptly following the determination that the Business Combination will not be consummated. Globalink anticipates that a stockholder who tenders shares for redemption in connection with the vote to approve the Business Combination would receive payment of the redemption price for such shares soon after the completion of the Business Combination.
If properly demanded by Globalink’s public stockholders, Globalink will redeem each share of Globalink common stock into a pro rata portion of the funds available in the Trust Account, calculated as of two business days prior to the anticipated consummation of the Business Combination. As of December 6, 2024, this would amount to approximately $11.98 per share. If you exercise your redemption rights, you will be exchanging your shares of Globalink common stock for cash and will no longer own the shares.
Holders of outstanding units must separate the underlying shares of Globalink common stock, warrants and rights prior to exercising redemption rights with respect to the shares of Globalink common stock. If the units are registered in a holder’s own name, the holder must deliver the certificate for its units to Continental with written instructions to separate the units into their individual component parts. This must be completed far enough in advance to permit the mailing of the certificates back to the holder so that the holder may then exercise his, her or its redemption rights upon the separation of the shares of Globalink common stock from the units.
| 101 |
If a broker, dealer, commercial bank, trust company or other nominee holds the units for an individual or entity (such individual or entity, the “beneficial owner”), the beneficial owner must instruct such nominee to separate the beneficial owner’s units into their individual component parts. The beneficial owner’s nominee must send written instructions by facsimile to the transfer agent. Such written instructions must include the number of units to be separated and the nominee holding such units. The beneficial owner’s nominee must also initiate electronically, using DTC’s DWAC system, a withdrawal of the relevant units and a deposit of an equal number of shares of Globalink common stock, warrants and rights. This must be completed far enough in advance to permit the nominee to exercise the beneficial owner’s redemption rights upon the separation of the shares of Globalink common stock from the units. While this is typically done electronically the same business day, beneficial owners should allow at least one full business day to accomplish the separation. If beneficial owners fail to cause their shares of Globalink common stock to be separated in a timely manner, they will likely not be able to exercise their redemption rights.
Notwithstanding the foregoing, a holder of the shares of Globalink common stock, together with any affiliate of his or her or any other person with whom he or she is acting in concert or as a “group” (as defined in Section 13(d)-(3) of the Exchange Act) will be restricted from seeking redemption rights with respect to more than 20% of the issued and outstanding shares of Globalink common stock.
If too many public stockholders exercise their redemption rights, Globalink may not be able to meet certain closing conditions, and as a result, would not be able to proceed with the Business Combination.
Appraisal Rights and Dissenter’s Rights
Appraisal rights are not available to security holders of Globalink in connection with the proposed Business Combination.
Proxies and Proxy Solicitation Costs
Globalink is soliciting proxies on behalf of the Board. This solicitation is being made by mail but also may be made by telephone or in person. Globalink and its directors, officers and employees may also solicit proxies in person, by telephone or by other electronic means. Any solicitation made and information provided in such a solicitation will be consistent with the written proxy statement/prospectus and proxy card. Globalink will bear the cost of solicitation. Okapi Partners LLC, a proxy solicitation firm that Globalink has engaged to assist it in soliciting proxies, will be paid a fixed fee of approximately $20,000 and be reimbursed out-of-pocket expenses.
Globalink will ask banks, brokers and other institutions, nominees and fiduciaries to forward its proxy materials to their principals and to obtain their authority to execute proxies and voting instructions. Globalink will reimburse them for their reasonable expenses.
| 102 |
THE REDOMESTICATION MERGER PROPOSAL (PROPOSAL 1)
The discussion in this proxy statement/prospectus of the Business Combination and the principal terms of the Merger Agreement, is subject to, and is qualified in its entirety by reference to, the Merger Agreement. The full text of the Merger Agreement is attached hereto as Annex A, which is incorporated by reference herein.
Purpose of the Redomestication Merger Proposal
The purpose of the Redomestication Merger is to merge Globalink, a Delaware corporation, with an into PubCo, a Cayman Islands exempted company, with PubCo continuing as the surviving company in the merger. Following the Redomestication Merger, it is intended that PubCo will ultimately become the parent entity of Alps Holdco (subject to the completion of the Acquisition Merger) and will be a “foreign private issuer” as that term is defined under the Exchange Act. As a result of the Redomestication Merger, the Globalink stockholders will no longer be stockholders of Globalink and (other than the Globalink stockholders who exercise their redemption rights) will become shareholders of PubCo, a foreign private issuer. As a foreign private issuer, PubCo is exempt from compliance with certain of the rules under the Exchange Act.
As a foreign private issuer, PubCo will be exempt from the rules under the Exchange Act prescribing the furnishing and content of proxy statements, and its officers, directors and principal shareholders will be exempt from the reporting and short-swing profit recovery provisions contained in Section 16 of the Exchange Act. In addition, PubCo will not be required under the Exchange Act to file quarterly periodic reports and financial statements with the SEC and will not be required to disclose in its periodic reports all of the information that U.S. domestic issuers are required to disclose. PubCo will also be permitted to follow corporate governance practices in accordance with Cayman Islands law in lieu of most of the corporate governance rules set forth by Nasdaq. As a result, PubCo’s corporate governance practices differ in some respects from those required to be followed by U.S. companies listed on a national securities exchange.
Summary of the Redomestication Merger
The Merger Agreement was entered into by and among Globalink, PubCo, Alps Holdco, Merger Sub, the Seller Representative, and the Sponsor on January 30, 2024 and amended on May 20, 2024. Upon the approval of the Redomestication Merger by the Globalink stockholders, PubCo and Globalink will execute the Plan of Merger relating to the Redomestication Merger, which shall be filed with the Registrar of Companies in the Cayman Islands with certain other documents on or prior to the Closing Date. On the Closing Date and immediately prior to the Acquisition Merger, Globalink will merge with and into PubCo, a Cayman Islands exempted company. The separate corporate existence of Globalink will cease and PubCo will continue as the surviving company. In connection with the Redomestication Merger, all outstanding Globalink units will separate into their individual components of Globalink common stock, Globalink rights and Globalink warrants and will cease separate existence and trading. Upon the consummation of the Business Combination, the current equity holdings of the Globalink stockholders shall be exchanged as follows:
| (i) | Each share of Globalink common stock, par value $0.001 per share, issued and outstanding immediately prior to the effective time of the Redomestication Merger (other than any redeemed shares), will automatically be cancelled and cease to exist and for each share of Globalink common stock, PubCo shall issue to its holder (other than Globalink stockholders who exercise their redemption rights in connection with the Business Combination) one validly issued PubCo ordinary share, which shall be fully paid; |
| (ii) | Each Globalink warrant to purchase one-half (1/2) of one share of Globalink common stock issued and outstanding immediately prior to effective time of the Redomestication Merger will convert into one PubCo warrant (or equivalent portion thereof). The PubCo warrants will have substantially the same terms and conditions as set forth in the Globalink warrants; and |
| (iii) | The holders of Globalink rights (convertible into one-tenth (1/10) of one share of Globalink common stock) issued and outstanding immediately prior to the effective time of the Redomestication Merger will obtain the PubCo right in exchange for the cancellation of each Globalink right; provided, however, that no fractional shares will be issued and all fractional shares will be rounded to the nearest whole share. |
| 103 |
Differences between the PubCo A&R Memorandum and Articles and Globalink Charter
Following is a summary of the material differences between the PubCo A&R Memorandum and Articles to be in effect following the Business Combination and Globalink’s amended and restated certificate of incorporation, as amended:
| ● | The name of the new public entity will be “ALPS Group” as opposed to “Globalink Investment Inc.”; | |
| ● | The authorized share capital of PubCo will be US$50,000 divided into 500,000,000 shares comprising (i) 495,000,000 PubCo ordinary shares of US$0.0001 par value each, and 5,000,000 preferred shares of US$0.0001 par value each, as opposed to Globalink having 500,000,000 authorized shares of common stock and 5,000,000 authorized shares of preferred stock; | |
| ● | PubCo’s corporate existence is of unlimited duration as opposed to Globalink’s corporate existence terminating if a business combination is not consummated by Globalink within a specified period of time; and | |
| ● | PubCo’s constitutional documents do not include the various provisions applicable only to special purpose acquisition corporations that Globalink’s amended and restated certificate of incorporation, as amended, contains. |
Authorized but unissued shares may enable PubCo’s board of directors to render it more difficult or to discourage an attempt to obtain control of PubCo and thereby protect continuity of or entrench its management, which may adversely affect the market price of PubCo’s securities. For example, if, in the due exercise of its fiduciary obligations, for example, PubCo’s board of directors were to determine that a takeover proposal were not in the best interests of PubCo, such shares could be issued by the board of directors without shareholder approval in one or more private placements or other transactions that might prevent or render more difficult or make more costly the completion of any attempted takeover transaction by diluting voting or other rights of the proposed acquirer or insurgent stockholder group, by creating a substantial voting block in institutional or other hands that might support the position of the incumbent board of directors, by effecting an acquisition that might complicate or preclude the takeover, or otherwise. The authorization of additional shares will, however, enable PubCo to have the flexibility to authorize the issuance of shares in the future for financing its business, for acquiring other businesses, for forming strategic partnerships and alliances and for stock dividends and stock splits. PubCo currently has no such plans, proposals, or arrangements, written or otherwise, to issue any of the additional authorized shares for such purposes.
A copy of PubCo A&R Memorandum and Articles, as will be in effect upon consummation of the Business Combination, is attached to this proxy statement/prospectus as Annex B. See also the Section titled “Comparison of Corporate Governance and Stockholders’/Shareholder’s Rights” on page 141 of this proxy statement/prospectus.
Required Vote
Approval of the Redomestication Merger Proposal requires the affirmative vote of the holders of a majority of the shares of Globalink common stock as of the record date represented in person or by proxy at the Special Meeting and entitled to vote thereon. Adoption of the Redomestication Merger Proposal is conditioned upon the adoption of the Acquisition Merger Proposal. It is important for you to note that in the event that either of the Redomestication Merger Proposal or the Acquisition Merger Proposal is not approved, then Globalink will not consummate the Business Combination.
Recommendation of the Board
After careful consideration, the Globalink Board unanimously determined that the Redomestication Merger forming part of the Business Combination with Alps Holdco is in the best interests of Globalink and its stockholders. On the basis of the foregoing, the Globalink Board has approved and declared advisable the Business Combination with Alps Holdco and unanimously recommends that you vote or give instructions to vote “FOR” adoption of the Redomestication Merger Proposal.
As of the date of this proxy statement/prospectus, a total of 3,445,000 shares of common stock, or approximately 92.6% of the outstanding shares, were subject to the Insider Letter and the Globalink Support Agreements. The approval of the Redomestication Merger Proposal does not require the votes from at least a majority of unaffiliated security holders of Globalink. As such, as the vote to approve the Redomestication Merger Proposal is a majority of the then outstanding shares of common stock present and entitled to vote at the Special Meeting, assuming only the minimum number of shares of common stock to constitute a quorum is present, no public stockholder must vote in favor of the Redomestication Merger Proposal for it to be approved.
| 104 |
THE ACQUISITION MERGER PROPOSAL (PROPOSAL 2)
Globalink stockholders should read carefully this proxy statement/prospectus in its entirety, including the subsection below titled “The Merger Agreement,” for more detailed information concerning the Business Combination and the terms and conditions of the Merger Agreement. Globalink also urges its stockholders to read carefully the Merger Agreement in its entirety before voting on this Proposal. A copy of the Merger Agreement is attached as Annex A to this proxy statement/prospectus.
General Description of the Acquisition Merger
Immediately following and conditional upon the completion of the Redomestication Merger, Merger Sub, a Cayman Islands exempted company and wholly owned subsidiary of PubCo, will merge with and into Alps Holdco with Alps Holdco being the surviving company, resulting in Alps Holdco being a wholly owned subsidiary of PubCo.
In consideration of the Acquisition Merger, PubCo will issue 160,000,000 ordinary shares as the Merger Consideration Shares with a deemed price per share US$10.00 to the shareholders of Alps Holdco. At the closing of the Acquisition Merger, the issued and outstanding shares in Alps Holdco held by the former Alps Holdco Shareholders will be cancelled and ceased to exist and will be automatically converted into a right to receive the applicable number of the Merger Consideration Shares, with such number subject to adjustment based on the Conversion Ratio. At the closing of the Acquisition Merger, the one fully paid share in Merger Sub held by PubCo will become one fully paid share in the surviving company, so that Alps Holdco will become a wholly owned subsidiary of PubCo.
Upon the closing of the Business Combination, PubCo board of directors will consist of five directors, all of whom will be designated by Alps Holdco and a majority of whom will be “independent” under Nasdaq’s listing standards. See section titled “Directors and Executive Officers of the Combined Company” for additional information.
According to the PubCo A&R Memorandum and Articles, the authorized share capital of the post-Closing company is US$50,000 divided into 500,000,000 shares comprising (i) 495,000,000 PubCo ordinary shares of US$0.0001 par value each, and 5,000,000 preferred shares of US$0.0001 par value each. After the consummation of the Business Combination, PubCo will be a “foreign private issuer” under the U.S. securities laws and the rules of Nasdaq.
After the Business Combination, assuming there are no redemptions of outstanding shares of Globalink common stock, there is no exercise of the PubCo warrants, and the 4,020,000 PubCo ordinary shares underlying the PIPE Investment are issued to the PIPE Investors, (i) Globalink’s public stockholders and Globalink’s right holders are expected to own approximately 0.8% of the outstanding ordinary shares of Combined Company, (ii) the Sponsor, the Initial Stockholders and PGM are expected to own approximately 2.1% of the outstanding ordinary shares of Combined Company, (iii) the Alps Holdco Shareholders are expected to own approximately 93.8% of the outstanding ordinary shares of Combined Company, (iv) the PIPE Investors are expected to own approximately 2.4% of the outstanding ordinary shares of Combined Company, and (v) IBDC Asia Sdn. Bhd., an advisory firm to Alps, is expected to own approximately 0.9% of the outstanding ordinary shares of Combined Company, representing finder’s fees payable in PubCo ordinary shares with value equal to 1% of the aggregate consideration for the Business Combination of US$1.6 billion.
The Merger Agreement
This section describes the material provisions of the Merger Agreement but does not purport to describe all of the terms thereof. The following summary is qualified in its entirety by reference to the complete text of the Merger Agreement and the related agreements. A copy of the Merger Agreement is attached as Annex A hereto, which is incorporated herein by reference. Globalink stockholders and other interested parties are urged to read such agreement in its entirety because it is the primary legal document that governs the Business Combination. Unless otherwise defined herein, the capitalized terms used in this section “The Acquisition Merger Proposal (Proposal 2) — The Merger Agreement” are defined in the Merger Agreement.
The Merger Agreement contains representations, warranties and covenants that the respective parties made to each other as of the date of the Merger Agreement or other specific dates, including, in some cases, as of the Closing of the Business Combination. The assertions embodied in those representations, warranties and covenants were made for purposes of the contract among the respective parties and are subject to important qualifications and limitations agreed to by the parties in connection with negotiating the Merger Agreement. The representations, warranties and covenants in the Merger Agreement are also modified in important part by the disclosure schedules attached thereto which are not filed publicly and which are subject to a contractual standard of materiality different from that generally applicable to stockholders.
| 105 |
The Merger Agreement contains representations and warranties that Globalink, on the one hand, and PubCo, Alps Holdco and Merger Sub, on the other hand, have made to one another as of specific dates. The assertions embodied in the representations and warranties are qualified by information in confidential disclosure schedules exchanged by the parties. Some of these schedules contain information that modifies, qualifies and creates exceptions to the representations and warranties set forth in the Merger Agreement. You should not rely on the representations and warranties described below as current characterizations of factual information about Globalink or Alps Holdco, because they were made as of specific dates, may be intended merely as a risk allocation mechanism between Globalink and Merger Sub, and Alps Holdco and are modified by the disclosure schedules.
Merger Consideration
The aggregate consideration to be paid by Globalink to Alps Holdco Shareholders in the form of shares of PubCo ordinary shares at the Closing will be equal to US$1.6 billion, determined based on an enterprise value of Alps Holdco (the “Merger Consideration”), with an earnout provision permitting Alps Holdco Shareholders to receive up to 48 million additional shares as and when the business meets certain incremental milestones for the consolidated revenue of Globalink and Alps Holdco for five fiscal years following the consummation of the Business Combination.
In addition to the Merger Consideration set forth above payable at the Closing, the Alps Holdco Shareholders will also have a contingent right to receive up to an additional 48 million additional PubCo ordinary shares. Subject to the terms and when the business meets certain incremental milestones for the number of conditions of the Merger Agreement, the Earnout Shares shall be earned and payable as follows:
(i) if, during the Combined Company’s first full fiscal year following the date of Closing, the Revenue exceeds $7,000,000, the Alps Holdco Shareholders shall be issued an additional 9,600,000 PubCo ordinary shares;
(ii) if during the Combined Company’s second full fiscal year following the date of Closing, the Revenue exceeds $14,000,000, the Alps Holdco Shareholders shall be issued an additional 9,600,000 PubCo ordinary shares;
(iii) if during the Combined Company’s third full fiscal year following the date of Closing, the Revenue exceeds $23,000,000, the Alps Holdco Shareholders shall be issued an additional 9,600,000 PubCo ordinary shares;
(iv) if during the Combined Company’s fourth full fiscal year following the date of Closing, the Revenue exceeds $34,000,000, the Alps Holdco Shareholders shall be issued an additional 9,600,000 PubCo ordinary shares; and
(v) if during the Combined Company’s fifth full fiscal year following the date of Closing, the Revenue exceeds $56,000,000, the Alps Holdco Shareholders shall be issued an additional 9,600,000 PubCo ordinary shares.
Conversion of Alps Holdco Ordinary Shares
Each Alps Holdco Ordinary Share issued and outstanding immediately prior to the Effective Time (other than any such Alps Holdco Ordinary Shares held in treasury, which will be cancelled, and any dissenting shares) shall be converted into the right to receive a number of PubCo ordinary shares equal to $1.6 billion divided by the number of fully-diluted Alps Holdco Ordinary Shares divided by $10.00 (of which 5% of the Merger Consideration Shares are be issued and held in escrow to satisfy any indemnification obligations incurred under the Merger Agreement as the Escrow Shares).
| 106 |
Directors and Executive Officers of the Combined Company Following the Business Combination
Each current member of the PubCo Board will cease to be a director upon the consummation of the Business Combination. Effective as of the Closing, the board of directors of the Combined Company will initially consist of five members, all of whom will be designated by Alps Holdco.
Conditions to the Closing of the Business Combination
Mutual Conditions
The obligations of all of the parties to consummate the Closing are subject to the satisfaction or written waiver (where permissible) by Globalink and Alps Holdco of all the following conditions:
| (i) | the absence of any applicable law or order restraining, prohibiting or imposing any condition on the consummation of the transactions contemplated by the Merger Agreement; | |
| (ii) | the expiration or termination of the waiting period under the Hart-Scott-Rodino Antitrust Improvements Act of 1976, as amended (if applicable); | |
| (iii) | receipt of any consent, approval or authorization required by any Authority (as such term is defined in the Merger Agreement); | |
| (iv) | there being no action brought by any Authority to enjoin or otherwise restrict the consummation of the Closing; | |
| (v) | Globalink having at least $5,000,001 of net tangible assets immediately prior to, which is expected to be waived, and PubCo having at least $5,000,001 of net tangible assets upon consummation of the Business Combination; | |
| (vi) | approval by the Alps Holdco Shareholders of the Business Combination and related transactions; | |
| (vii) | approval by the stockholders of Globalink of the Business Combination and related transactions; | |
| (viii) | the conditional approval for listing by Nasdaq of the PubCo ordinary shares and PubCo warrants and satisfaction of initial and continued listing requirements; and | |
| (ix) | the Form F-4 becoming effective in accordance with the provisions of the Securities Act. |
Conditions to Obligations of Globalink
The obligation of Globalink to consummate the Closing is subject to the satisfaction, or the waiver in Globalink’s sole and absolute discretion, of all the following further conditions:
(a) Alps Holdco, PubCo and Merger Sub shall have duly performed or complied with, in all material respects, all their respective obligations and covenants hereunder required to be performed or complied with (without giving effect to any materiality or similar qualifiers contained therein) by Alps Holdco, PubCo and Merger Sub at or prior to the Closing Date.
(b) The representations and warranties of Alps Holdco, PubCo and Merger Sub contained in the Merger Agreement (disregarding all qualifications and exceptions contained therein relating to materiality or Material Adverse Effect, shall be true and correct in all respects at and as of the date of Agreement and as of the Closing Date as if made as of such date (except to the extent that any such representation and warranty is expressly made as of a specific date, in which case such representation and warranty shall be true and correct at and as of such specific date), except, in each case, for any failure of such representations and warranties (disregarding all qualifications and exceptions contained therein relating to materiality or Material Adverse Effect) to be so true and correct that would not in the aggregate have or reasonably be expected to have a Material Adverse Effect in respect of the Alps Holdco and its subsidiaries as a whole.
(c) Since the date of the Merger Agreement, there shall not have occurred any Material Adverse Effect in respect of Alps Holdco and its subsidiaries as a whole which is continuing and uncured.
(d) Globalink shall have received a certificate, dated as of the Closing Date, signed by the chief executive officer or other officer or director of Alps Holdco certifying the accuracy of the provisions of the foregoing paragraphs (a), (b) and (c).
| 107 |
(e) Globalink shall have received a certificate, dated as of the Closing Date, signed by the secretary or another officer or director of Alps Holdco attaching true, correct and complete copies of (i) Alps Holdco’s memorandum and articles of association as then in force, as filed with and stamped by the Registrar of Companies of the Cayman Islands; (ii) copies of resolutions duly passed by the board of directors of Alps Holdco authorizing the Merger Agreement, the additional agreements to which Alps Holdco is a party and the transactions contemplated thereby and written consent of Alps Holdco Shareholders; and (iii) a certificate of good standing of Alps Holdco, issued as of a recent date (not less than three (3) days before the Closing Date) by the Registrar of Companies of the Cayman Islands.
(f) Globalink shall have received a certificate, dated as of the Closing Date, signed by the secretary or another officer or a director of PubCo attaching true, correct and complete copies of (i) the memorandum and articles of association of PubCo as then in force, as filed with and stamped by the Cayman Registrar; (ii) copies of resolutions duly passed by the PubCo Board authorizing the Merger Agreement, the additional agreements to which PubCo is a party and the transactions contemplated thereby; (iii) the PubCo Shareholder Approval, in each case evidencing the approval of the shareholders of PubCo, and (iv) a certificate of good standing of PubCo, issued as of a recent date (not less than three (3) days before the Closing Date) by the Cayman Registrar.
(g) Globalink shall have received a certificate, dated as of the Closing Date, signed by the secretary or another officer or a director of Merger Sub attaching true, correct and complete copies of (i) the memorandum and articles of association of Merger Sub as then in force, as filed with and stamped by the Cayman Registrar; (ii) copies of resolutions duly passed by the board of directors of Merger Sub authorizing the Merger Agreement, the additional agreements to which Merger Sub is a party and the transactions contemplated thereby; (iii) the Merger Sub Shareholder Approval duly executed by all of the shareholders of Merger Sub, and (iv) a certificate of good standing of Merger Sub, issued as of a recent date (not less than three (3) days before the Closing Date) by the Cayman Registrar.
(h) Each of Alps Holdco and the Alps Holdco Shareholders, as applicable, shall have executed and delivered to Globalink a copy of each additional agreement to which Alps Holdco or such Alps Holdco Shareholder, as applicable, is a party and that was not otherwise executed and delivered prior to the Closing.
(i) The size and composition of the post-Closing Board of Directors of the Combined Company shall have been approved, with effect from the Effective Time, by resolutions of PubCo Board of Directors.
(j) Globalink shall have received a copy of the Escrow Agreement, duly executed by the Seller Representative and the Escrow Agent.
(k) Globalink shall have obtained an opinion from an independent investment bank or other financial advisory firm mutually acceptable to Globalink and Alps as to the fairness from a financial point of view, as of the date of such opinion, of the Merger Consideration Shares to be paid to Alps Holdco Shareholders pursuant to the Merger Agreement.
Conditions to Obligations of Alps Holdco, PubCo and Merger Sub
The obligations of Alps Holdco, PubCo and Merger Sub to consummate the Closing is subject to the satisfaction, or the waiver in Alps Holdco’s sole and absolute discretion, of all of the following further conditions:
(a) Globalink shall have duly performed or complied with, in all material respects, all of its obligations and covenants hereunder required to be performed or complied with (without giving effect to any materiality or similar qualifiers contained therein) by Globalink at or prior to the Closing Date.
(b) The representations and warranties of Globalink contained in the Merger Agreement (disregarding all qualifications contained therein relating to materiality or Material Adverse Effect) shall be true and correct as of the date of the Merger Agreement and as of the Closing Date in all material respects, as if made at and as of such date (except to the extent that any such representation and warranty is made as of an earlier date, in which case such representation and warranty shall be true and correct in all material respects at and as of such earlier date).
| 108 |
(c) Since the date of the Merger Agreement, there shall not have occurred any material adverse effect in respect of Globalink which is continuing and uncured.
(d) Alps Holdco shall have received a certificate signed by the Chief Executive Officer of Globalink certifying the accuracy of the provisions of the foregoing paragraphs (a), (b) and (c).
(e) Alps Holdco shall have received a certificate, dated as of the Closing Date, signed by the secretary or another officer or a director of PubCo attaching true, correct and complete copies of (i) the memorandum and articles of association of PubCo then in effect, as filed with and (if received prior to the date of such certificate) stamped by the Cayman Registrar; (ii) copies of resolutions duly adopted by the board of directors of Globalink authorizing the Merger Agreement, the additional agreements to which Globalink is a party and the transactions contemplated hereby and thereby and the Globalink Proposals; and (iii) a certificate of good standing of PubCo, certified as of a recent date by the Registrar of Companies in the Cayman Islands.
(g) Each of Globalink, Sponsor or other stockholder of Globalink, as applicable, shall have executed and delivered to Alps Holdco a copy of each additional agreement to which Globalink, Sponsor or such other stockholder of Globalink, as applicable, is a party.
(h) Alps Holdco shall have received a copy of the Escrow Agreement, duly executed by PubCo, Globalink and the Escrow Agent.
Representations and Warranties
The Merger Agreement contains customary representations and warranties of the parties thereto with respect to, among other things, (a) corporate existence and power, (b) authorization to enter into the Merger Agreement and related transactions, (c) governmental authorization, (d) non-contravention, (e) capital structure, (f) corporate records, (g) subsidiaries, (h) consents, (i) financial statements, (j) books and records, (k) internal accounting controls, (l) absence of changes, (m) property and title to assets, (n) litigation, (o) material contracts, (p) licenses and permits, (q) compliance with laws, (r) intellectual property, (s) accounts payable and affiliate loans, (t) employee and employment matters, (u) withholding, (v) employee benefits, (w) real property, (x) tax matters, (y) environmental laws, (z) finder’s fees, (aa) power of attorney, suretyships and bank accounts, (bb) directors and officers, (cc) anti-money laundering laws, (dd) insurance, (ee) related party transactions, (ff) information supplied, (gg) derogation, and (hh) disclosure letter.
The Merger Agreement contains a number of representations and warranties of the parties thereto, many of the representations and warranties are qualified by materiality or Material Adverse Effect. “Material Adverse Effect” as used in the Merger Agreement means with respect to any specified person or entity, any fact, event, occurrence, change or effect that has had or would reasonably be expected to have, individually or in the aggregate, a material adverse effect on the business, assets, liabilities, results of operations, or condition (financial or otherwise) of such person or entity and its subsidiaries, taken as a whole, or the ability of such person or entity or any of its subsidiaries on a timely basis to consummate the transactions contemplated by the Merger Agreement or the ancillary documents to which it is a party or bound or to perform its obligations thereunder, in each case subject to certain customary exceptions. Certain of the representations are subject to specified exceptions and qualifications contained in the Merger Agreement or in information provided pursuant to certain disclosure schedules to the Merger Agreement.
The representations and warranties made by Globalink, PubCo, Alps Holdco and Merger Sub are customary for transactions similar to the transactions contemplated by the Merger Agreement.
Certain Surviving Representations and Warranties
Certain representations or warranties made by Alps Holdco in the Merger Agreement shall survive until twelve (12) months after the Closing. Subject to the terms and conditions of the Merger Agreement, and from and after the date of Closing, the Alps Holdco Shareholders agree to indemnify and hold harmless Globalink against and in respect of any and all out-of-pocket loss incurred or sustained by Globalink as a result of or in connection with any breach or inaccuracy of the representations and warranties of Alps Holdco with respect to (a) Corporate Existence and Power, (b) Authorization, (c) Governmental Authorization, (d) Non-Contravention, (e) Capitalization, (f) Subsidiaries, (g) Consents, (h) Financial Statements, (i) Books and Records, (j) Absence of Certain Changes, (k) Properties; Title to the Company’s Assets, (l) Litigation, (m) Licenses and Permits, (n) Compliance with Laws, (o) Real Property, (p) Tax Matters, (q) Finders’ Fees, (r) Anti-Money Laundering Laws, and (s) Related Party Transactions. Except for fraud claims, Globalink shall not assert any claim for indemnification until the aggregate amount of all losses exceeds $1,000,000, in which event the Alps Holdco Shareholders shall be responsible for the aggregate amount of all losses from the first dollar, regardless of such threshold. Except for fraud claims, the maximum aggregate amount of indemnification payments shall not exceed the amount of the Escrow Property in the Escrow Account at such time.
| 109 |
Subject to the foregoing, the representations and warranties of the parties contained in the Merger Agreement terminate as of, and do not survive, the Closing, and there are no indemnification rights for another party’s breach. The covenants and agreements of the parties contained in the Merger Agreement do not survive the Closing, except those covenants and agreements to be performed after the Closing, which covenants and agreements will survive until fully performed.
Covenants; Conduct of Business Pending the Business Combination
Globalink and Alps Holdco have each agreed that, except as permitted by the Merger Agreement or the additional agreements or as required by law and the disclosure schedules, during the period commencing on the date of the Merger Agreement and continuing until the Closing Date or earlier termination, each party will (i) conduct its business only in the ordinary course (including the payment of accounts payable and the collection of accounts receivable), consistent with past practices, (ii) duly and timely file all material tax returns required to be filed (or obtain a permitted extension with respect thereto) with the applicable taxing Authorities and pay any and all taxes due and payable during such time period, (iii) duly observe and conform in with all applicable law, including the Exchange Act, and orders, in each case, in all material respects, and (iv) use commercially reasonable efforts to preserve intact its business relationships with employees, clients, suppliers, contract manufacturing organizations, contract research organizations and other third parties. Additionally, neither Alps Holdco nor Globalink shall, or permit its subsidiaries to:
(i) amend, modify or supplement its memorandum or articles of association, certificate of incorporation, or bylaws or other organizational or governing documents except as contemplated hereby, or engage in any reorganization, reclassification, liquidation, dissolution or similar transaction;
(ii) amend, waive any provision of, terminate prior to its scheduled expiration date, or otherwise compromise in any material way or relinquish any material right under, any material contract, agreement, lease, license or other right or asset of such party or its Subsidiaries;
(iii) other than in the ordinary course of business, modify, amend or enter into any contract, agreement, lease, license or commitment, including for capital expenditures, that extends for a term of one year or more and obligates the payment by it of more than $100,000 (individually or in the aggregate);
(iv) make any capital expenditures in excess of $100,000 (individually or in the aggregate);
(v) sell, lease, license or otherwise dispose of any of its material assets, except pursuant to existing contracts or commitments disclosed herein or in the ordinary course of business consistent with past practices;
(vi) solely in the case of Alps Holdco, sell, lease, license or otherwise dispose of any of any material Alps Holdco owned intellectual property (as defined in the Merger Agreement) outside of the ordinary course of business consistent with past practices;
(vii) solely in the case of Alps Holdco, permit any material registered owned intellectual property to go abandoned or expire for failure to make an annuity or maintenance fee payment, or file any necessary paper or action to maintain such rights;
| 110 |
(viii) (A) pay, declare or promise to pay any dividends or other distributions with respect to its capital stock or other equity securities; or (B) except as contemplated hereby or by any additional agreement, amend any material term, right or obligation with respect to any outstanding shares of its capital stock or other equity securities;
(ix) (A) make any loan, advance or capital contribution in excess of $100,000 to any person; (B) incur any indebtedness in excess of $100,000 including drawings under the lines of credit, if any, other than (1) loans evidenced by promissory notes made to Globalink as working capital advances as described in the Prospectus and (2) intercompany Indebtedness; or (C) repay or satisfy any Indebtedness in excess of $100,000, other than the repayment of Indebtedness in accordance with the terms thereof;
(x) suffer or incur any lien in excess of $100,000, except for Permitted Liens (as defined in the Merger Agreement), on its assets;
(xi) delay, accelerate or cancel, or waive any material right with respect to, any receivables or Indebtedness in excess of $100,000 owed to it, or write off or make reserves in excess of $250,000 against the same (other than, in the case of Alps Holdco and its subsidiaries, in the ordinary course of business consistent with past practices);
(xii) merge or consolidate or enter a similar transaction with, or acquire all or substantially all of the assets or business of, any other person; make any material investment in any person; or be acquired by any other person;
(xiii) terminate or allow to lapse any material insurance policy protecting any of its material assets, unless simultaneously with such termination or lapse, a replacement policy underwritten by an insurance company of nationally recognized standing having comparable deductions and providing coverage equal to or greater than the coverage under the terminated or lapsed policy for substantially similar premiums or less is in full force and effect;
(xiv) adopt any severance, retention or other employee plan in excess of $100,000 in the aggregate, or fail to continue to make timely contributions to each Plan (as defined in the Merger Agreement) in accordance with the terms thereof;
(xv) institute, settle or agree to settle any Action before any Authority, in each case in excess of $100,000 (exclusive of any amounts covered by insurance) or that imposes material injunctive or other material non-monetary relief on it;
(xvi) except as required by IFRS, make any material change in its accounting principles, methods or practices or write down in any material respect the value of its assets;
(xvii) change its principal place of business or jurisdiction of organization;
(xviii) issue, redeem or repurchase any shares, capital stock, membership interests or other securities, or issue any securities exchangeable for or convertible into any shares of its capital stock or other securities, other than with respect to Alps Holdco and its subsidiaries, the issuance of Alps Holdco Ordinary Shares upon the exercise or conversion of outstanding Alps Holdco convertible securities;
(xix) (A) make, change or revoke any material tax election; (B) change any annual tax accounting periods in any material respect; (C) settle or compromise any material claim, notice, audit report or assessment in respect of taxes of Alps Holdco and its subsidiaries; (D) enter into any tax allocation, tax sharing, tax indemnity or other closing agreement relating to any taxes of Alps Holdco and its subsidiaries; or (E) surrender or forfeit any right to claim a material tax refund;
(xx) enter into any material transaction with or distribute or advance any material assets or property to any of its affiliates (other than its Subsidiaries), other than the payment of salary and benefits in the ordinary course;
| 111 |
(xxi) other than as required by a Plan, (A) materially increase or change the compensation or benefits of any employee or service provider of Alps Holdco and its subsidiaries other than in the ordinary course of business, (B) accelerate the vesting or payment of any material compensation or benefits of any employee or service provider of Alps Holdco and its subsidiaries, (C) make any loan to any present or former employee or other individual service provider of Alps Holdco and its subsidiaries, other than advancement of expenses in the ordinary course of business consistent with past practices, or (D) enter into, amend or terminate any collective bargaining agreement or other material agreement with a labor union or labor organization;
(xxii) fail to duly observe and conform to any applicable laws and orders in any material respect; or
(xxiii) agree or commit to do any of the foregoing.
The Merger Agreement also contains additional covenants of the parties, including, among others, access to information, confidentiality, cooperation in the preparation of the Form F-4 and Proxy Statement (as defined in the Merger Agreement) required to be filed in connection with the Business Combination and to obtain all requisite approvals of each party’s respective stockholders.
Termination of the Merger Agreement
The Merger Agreement may be terminated at any time prior to the Effective Time as follows:
| (i) | by either Globalink or Alps Holdco if the transactions contemplated by the Merger Agreement are not consummated on or before the Outside Date, provided that, if the SEC has not declared the Form F-4 effective on or prior to the five month anniversary of the Merger Agreement, then the Outside Date will be extended by one additional month, provided further that, the failure to consummate the transactions contemplated by the Merger Agreement by the Outside Date is not due to a material breach by the party seeking to terminate the agreement; | |
| (ii) | by either Globalink or Alps Holdco if any Authority has issued any final decree, order, judgment, award, injunction, rule or consent or enacted any law, having the effect of permanently enjoining or prohibiting the consummation of the Business Combination, provided that, the party seeking to terminate cannot have breached its obligations under the Merger Agreement and such breach was a substantial cause of, or substantially resulted in, such action by the Authority; | |
| (iii) | by mutual written consent of Globalink and Alps Holdco duly authorized by each of their respective board of directors; | |
| (iv) | by Globalink in the event that Alps Holdco does not deliver to Globalink the Final March 31, 2024 Financial Statements on or prior to May 31, 2024 or such other date as mutually agreed upon by Globalink and Alps Holdco in writing; and | |
| (v) | by either Globalink or Alps Holdco if the other party has breached any of its covenants or representations and warranties as set forth in the Merger Agreement such that closing conditions would not be satisfied by the earlier of (A) the Closing Date and (B) 30 days following receipt by the breaching party of a written notice of the breach. |
The Merger Agreement and other agreements described below have been included to provide investors with information regarding their respective terms. They are not intended to provide any other factual information about Globalink, PubCo, Alps Holdco, Merger Sub or the other parties thereto. In particular, the assertions embodied in the representations and warranties in the Merger Agreement were made as of a specified date, are modified or qualified by information in in the disclosure schedules to the Merger Agreement, may be subject to a contractual standard of materiality different from what might be viewed as material to investors, or may have been used for the purpose of allocating risk between the parties. Accordingly, the representations and warranties in the Merger Agreement are not necessarily characterizations of the actual state of facts about Globalink, PubCo, Alps Holdco, Merger Sub or the other parties thereto at the time they were made or otherwise and should only be read in conjunction with the other information that Globalink makes publicly available in reports, statements and other documents filed with the SEC. Globalink or Alps Holdco investors and shareholders are not third-party beneficiaries under the Merger Agreement.
| 112 |
Certain Related Agreements
This section describes the material provisions of certain additional agreements entered into or to be entered into pursuant to the Merger Agreement (the “Related Agreements”) but does not purport to describe all of the terms thereof or include all of the additional agreements entered into or to be entered into pursuant to the Merger Agreement. The following summary is qualified in its entirety by reference to the complete text of each of the Related Agreements. Globalink stockholders and other interested parties are urged to read such Related Agreements in their entirety.
Globalink Support Agreements
Globalink, Alps Holdco, and certain stockholders of Globalink have entered into a Globalink Support Agreement, pursuant to which the stockholders of Globalink that are parties to the Globalink Support Agreements will agree to vote all shares of Globalink common stock beneficially owned by them in favor of the transactions contemplated by the Merger Agreement.
Alps Holdco Support Agreements
Globalink, Alps Holdco and certain shareholder of Alps Holdco have entered into an Alps Holdco Support Agreement, pursuant to which the shareholders of Alps Holdco that are parties to the Alps Holdco Support Agreements will agree to vote all Alps Holdco Ordinary Shares beneficially owned by them in favor of the transactions contemplated by the Merger Agreement.
Subscription Agreements
In connection with the execution of the Merger Agreement, PubCo, Globalink and Alps Holdco entered into the Subscription Agreements with certain accredited investors on June 4, 2024, June 5, 2024 and August 27, 2024, pursuant to which the investors have agreed to purchase, and PubCo has agreed to sell to the PIPE Investors, an aggregate of $40,200,000 worth of PubCo ordinary shares, or 4,020,000 PubCo ordinary shares. The purpose of the sale of the PIPE Securities is to raise additional capital for the business operations of the Combined Company. The obligations to consummate the transactions contemplated by the Subscription Agreements are conditioned upon, among other things, customary closing conditions and the consummation of the transactions contemplated by the Merger Agreement.
Lock-up Agreements
The Merger Agreement provides that, prior to the Closing, and effective as of the Closing, certain Alps Holdco Shareholders, the Parent Representative and certain other stockholders of Globalink will enter into the Lock-up Agreements, subject to certain customary exceptions, not to offer, sell, contract to sell, pledge or otherwise dispose of, directly or indirectly, any of the Lock-up Shares, enter into a transaction that would have the same effect, or enter into any swap, hedge or other arrangement that transfers, in whole or in part, any of the economic consequences of ownership of the Lock-Up Shares or otherwise, publicly disclose the intention to make any offer, sale, pledge or disposition, or to enter into any transaction, swap, hedge or other arrangement, or engage in any Short Sales (as defined in the Lock-up Agreement) with respect to the Lock-Up Shares until (i) (w) with respect to one hundred percent (100%) of the Lock-Up Shares, the date from the Closing until the last date that is six (6) months after the date of the Closing, (x) with respect to ninety percent (90%) of the Lock-Up Shares, the 1st day of the 7th month from the Closing until the date that is nine (9) months after the date of the Closing, (y) with respect to seventy percent (70%) of the Lock-Up Shares, the 1st day of the 10th month from the Closing until the date that is twelve (12) months after the date of the Closing, (z) with respect to forty percent (40%) of the Lock-Up Shares, the 1st day of the 13th month from the Closing until the date that is fifteen (15) months after the date of the Closing, or (ii) if sooner, the date after the Closing on which Globalink consummates a liquidation, merger, share exchange or other similar transaction with an unaffiliated third party that results in all of Globalink’s stockholders having the right to exchange their equity holdings in Globalink for cash, securities or other property. For the avoidance of doubt, no Lock-up Shares shall be subject to the terms in the Lock-up Agreement from and after the date that is fifteen (15) months after the date of the Closing.
| 113 |
Amended and Restated Registration Rights Agreement
At the Closing, PubCo will enter into the Amended and Restated Registration Rights Agreement with certain existing stockholders of Globalink with respect to the PubCo ordinary shares they own at the Closing, and with certain Alps Holdco Shareholders who will own PubCo ordinary shares after the Closing. The Amended and Restated Registration Rights Agreement will require Globalink to, among other things, file a resale shelf registration statement on behalf of the stockholders no later than 60 days after the Closing. The Amended and Restated Registration Rights Agreement will also provide certain demand registration rights and piggyback registration rights to the shareholders, subject to underwriter cutbacks and issuer blackout periods. PubCo will agree to pay certain fees and expenses relating to registrations under the Amended and Restated Registration Rights Agreement.
Escrow Agreement
The parties agreed that at or prior to the Closing, PubCo, Globalink and Continental Stock Transfer & Trust Company, as escrow agent (the “Escrow Agent”) will enter into the Escrow Agreement, effective as of the Effective Time, in form and substance reasonably satisfactory to PubCo and Alps Holdco, pursuant to which PubCo shall issue to the Escrow Agent a number of PubCo ordinary shares (with each share valued at $10.00) equal to five percent (5%) of One Point Six Billion Dollars ($1,600,000,000), together with any equity securities paid as dividends or distributions with respect to the Escrow Shares and along with any other dividends, distributions or other income on the Escrow Shares (collectively, “Escrow Property”). The Escrow Property will be held in a segregated escrow account for a period of six (6) months after the Closing Date (“Escrow Expiry Date”) and will serve as the sole and exclusive remedy and payment for indemnification obligations (other than fraud claims), if any, pursuant to the Merger Agreement. The Escrow Property will not be subject to indemnification claims to the extent made after the Escrow Expiry Date; provided, however, with respect to any indemnification claim made on or prior to the Escrow Expiry Date that remains unresolved at the time of the Escrow Expiry Date, all or a portion of the Escrow Property reasonably necessary to satisfy such unresolved claim will remain in the Escrow Account until such claim is resolved. After the Escrow Expiry Date, any remaining Escrow Property not required to satisfy pending or unresolved indemnification claims will be distributed pro rata to the Alps Holdco Shareholders. Once all pending claims are resolved, any remaining Escrow Property will also be distributed pro rata to the Alps Holdco Shareholders. The Alps Holdco Shareholders will remain liable for any indemnification claims made after the Escrow Expiry Date. Except for fraud claims, the maximum aggregate amount of indemnification payments shall not exceed the amount of the Escrow Property in the Escrow Account at such time. As such, assuming that all of the Escrow Property is distributed to the Alps Holdco Shareholders after the Escrow Expiry Date, other than fraud claims, the maximum amount of indemnification payments would be zero given there would no longer be any Escrow Property in the Escrow Account at such time. In such event, the Alps Holdco Shareholders would not have any indemnification liability in practice after the Escrow Expiry Date other than with respect to fraud claims.
Board of Directors and Management Following the Business Combination
The following persons are expected to be elected or appointed by the Board to serve as executive officers and directors following the Business Combination. For biographical information concerning the executive officers and directors following the Business Combination, see “Directors and Executive Officers of the Combined Company.”
Each director will hold office until his or her term expires at the next annual meeting of shareholders in the year following the year of such director’s election or until his or her death, resignation, removal or the earlier termination of his or her term of office.
The following table sets forth the name, age and position of each of the expected directors and executive officers of the Combined Company upon consummation of the Business Combination:
Combined Company
| Name | Age | Position | ||
| Dr. THAM Seng Kong | 55 | Director and Chief Executive Officer | ||
| CHEW Yoke Ling | 53 | Director | ||
| LOW Wei Sim | 29 | Chief Operating Officer | ||
| Professor Manickam RAVICHANDRAN | 57 | Chief Scientific Officer | ||
| Professor POH Chit Laa | 73 | Chief Vaccine Development Officer | ||
| Tan Sri Dato’ Seri Dr. Suleiman bin MOHAMED | 76 | Independent Director | ||
| CHUA Boon Ping | 40 | Independent Director | ||
Intan Ilyani binti Ghazali |
45 | Independent Director |
Alps Holdco
| Name | Age | Position | ||
| Dr. THAM Seng Kong | 55 | Director and Chief Executive Officer | ||
| CHEW Yoke Ling | 53 | Director | ||
| LOW Wei Sim | 29 | Chief Operating Officer | ||
| Professor Manickam RAVICHANDRAN | 57 | Chief Scientific Officer | ||
| Professor POH Chit Laa | 73 | Chief Vaccine Development Officer | ||
| Tan Sri Dato’ Seri Dr. Suleiman bin MOHAMED | 76 | Independent Director | ||
| CHUA Boon Ping | 40 | Independent Director | ||
| Intan Ilyani binti Ghazali | 45 | Independent Director |
Alps is in the process of evaluating and seeking suitable candidates to serve as the Chief Financial Officer of the Combined Company.
| 114 |
The Sponsor, its Affiliates, and Promoters
GL Sponsor LLC, Globalink’s sponsor, was formed in the State of Delaware as a limited liability company. The Sponsor has no business operations and only serves as a vehicle that holds equity interests in Globalink. In addition to participating in sponsoring Globalink, none of the Sponsor, its affiliate, which includes the Sponsor’s manager and controlling person, Ng Yan Xun, or any of the Sponsor’s promoters, namely Ng Yan Xun, Chan Yew Ping, and Lin Ding Jie, who participated in the founding of the Sponsor and hold a 55%, 22.5%, and 22.5% of the equity interest in the Sponsor, respectively, had any additional experience in organizing special purpose acquisition companies. To Globalink’s knowledge, each of the Sponsor, its affiliate, and its promoters are not involved in other special purpose acquisition companies.
As of the date of this proxy statement/prospectus, Ng Yan Xun is the controlling person of the Sponsor, holding 55% of the Sponsor’s equity interest. Each of Lin Ding Jie and Chan Yew Ping holds 22.5% of the equity interest in the Sponsor.
From the inception of Globalink to about October 2022, Lin Ding Jie, a promoter of the Sponsor, and Sally Lim, the former affiliate of the Sponsor prior to transferring her interests in the Sponsor to Ng Yan Xun in October 2022, participated in and facilitated with the discussions and negotiations between Globalink and the previous target company, Tomorrow Crypto Group Inc., a Nevada corporation (“Tomorrow Crypto”) regarding a proposed business combination transaction, by acting as representatives of Globalink. Globalink entered into a merger agreement with Tomorrow Crypto on August 3, 2022 and the merger agreement was terminated on March 8, 2023.
Other than the foregoing roles taken on by Lin Ding Jie and Sally Lim in Globalink’s negotiations with Tomorrow Crypto, none of the Sponsor, its affiliates, or promoters directly oversees or manages the business operations of Globalink. Since October 2022, Globalink’s management has played an active role in managing and overseeing Globalink’s activities, including but not limited to, engaging in preliminary discussions with potential target companies, including Alps Holdco, negotiating the terms of the Business Combination Agreement and ancillary agreements with Alps Holdco and the Seller Representative, and overseeing Globalink’s other activities including reporting obligations to the SEC. However, Globalink’s management reports updates on the proposed Business Combination and negotiation status to the Sponsor on a regular basis.
Shortly after the formation of Globalink, the Sponsor purchased the Founder Shares for an aggregate purchase price of $25,000, or less than $0.01 per share, and had a total market value of $34.36 million as of December 12, 2024. Subsequently, the Sponsor transferred 40,000 shares of Globalink common stock to Globalink’s directors and officers, and hold an aggregate of 2,835,000 shares of Globalink common stock as of the date of this proxy statement/prospectus. The Sponsor will receive a positive rate of return so long as the market price of the common stock is at least $0.01 per share, even if public stockholders experience a negative rate of return in the Combined Company.
Through the Insider Letter, each of the Initial Stockholders and PGM agreed to vote all of the shares of Globalink common stock owned by them in favor of an initial business combination of Globalink, including the Business Combination. Additionally, through a Globalink Support Agreement entered into among Globalink, Alps Holdco, and certain stockholders of Globalink, the stockholders of Globalink that are parties to the Globalink Support Agreements, including each of the Initial Stockholders and PGM, have agreed to vote all shares of Globalink common stock beneficially owned by them in favor of the transactions contemplated by the Merger Agreement.
Through the Insider Letter, the Sponsor has waived its right to redeem any shares of Globalink common stock in connection with a stockholder vote to approve a proposed initial business combination or sell any shares of Globalink common stock to Globalink in a tender offer prior to a proposed initial business combination, or to receive distributions with respect to the Founder Shares upon the liquidation of the Trust Account if Globalink is unable to consummate a business combination. No agreement, arrangement, or understanding, including any payments, has been made between the Sponsor and any unaffiliated security holder of Globalink regarding the redemption of outstanding shares of Globalink common stock.
The following table illustrates the material terms of any agreement, arrangement, or understanding regarding the restrictions on the ability of the Sponsor and its affiliates to sell shares of Globalink common stock and PubCo ordinary shares:
| Name of Agreement | Date of Agreement | Person(s) subject to the Agreement | Shares subject to the Agreement | Date of Expiry | Exceptions under the Agreement | Terms Resulting in An Earlier Expiration of the Agreement | ||||||
| IPO Escrow Agreement | December 6, 2021 | the Sponsor, Say Leong Lim, Kevin (Zeng Yenn) Chin, Hong Shien Beh, Hui Liang Wong, and Kian Huat Lai | the Founder Shares | Six months after the consummation of an initial business combination by Globalink | None. | 50% of the escrow shares are subject to early release on the date the closing price of the Combined Company Ordinary Shares exceeds $12.50 per share for any 20 trading days within any 30-trading day period commencing after Globalink’s initial business combination.
If Globalink is being liquidated at any time during the escrow period, the escrow agent shall promptly destroy certificates representing the escrow shares | ||||||
| Lock-up Agreement | To be entered into prior to the Closing | the Sponsor, Say Leong Lim, Kevin (Zeng Yenn) Chin, Hong Shien Beh, Hui Liang Wong, Kian Huat Lai, and certain Alps Holdco Shareholders | the Founder Shares and certain Merger Consideration Shares | (w) with respect to one hundred percent (100%) of the Lock-Up Shares, the date from the Closing until the last date that is six (6) months after the date of the Closing, (x) with respect to ninety percent (90%) of the Lock-Up Shares, the 1st day of the 7th month from the Closing until the date that is nine (9) months after the date of the Closing, (y) with respect to seventy percent (70%) of the Lock-Up Shares, the 1st day of the 10th month from the Closing until the date that is twelve (12) months after the date of the Closing, (z) with respect to forty percent (40%) of the Lock-Up Shares, the 1st day of the 13th month from the Closing until the date that is fifteen (15) months after the date of the Closing, or (ii) if sooner, the date after the Closing on which Globalink consummates a liquidation, merger, share exchange or other similar transaction with an unaffiliated third party that results in all of Globalink’s stockholders having the right to exchange their equity holdings in Globalink for cash, securities or other property. For the avoidance of doubt, no Lock-up Shares shall be subject to the terms in the Lock-up Agreement from and after the date that is fifteen (15) months after the date of the Closing | Holders may transfer to its direct or indirect affiliates, by bona fid gift to immediate family, by virtue of the laws of descent and distribution upon death, transfers to PubCo’s officers, directors or their affiliates, transfers as a dividend or distribution to holder’s limited partners, shareholders, members of, or owners of similar equity interests, pledges lock-up shares as security or collateral in connection with a borrowing or the incurrence of any indebtedness by the holder | The lock-up agreement will not take effect if the closing of an initial business combination does not take place. |
| 115 |
Interests of Globalink’s Initial Stockholders, Directors and Officers in the Business Combination
When you consider the recommendation of the Board in favor of approval of the Proposals, you should keep in mind that Globalink’s directors and officers have interests in the Business Combination that may be different from or in addition to (and which may conflict with) your interests as a stockholder and may be incentivized to complete a business combination on terms less favorable to stockholders rather than liquidating Globalink, including the aggregate amount at risk to the Globalink’s initial stockholders, directors and officers of $25,000, which is the amount paid for the Founder Shares and the private units. These interests include, among other things, the fact that:
| ● | The Initial Stockholders, including the officers and directors of Globalink, have waived their right to redeem any shares of Globalink common stock in connection with a stockholder vote to approve a proposed initial business combination or sell any shares of Globalink common stock to Globalink in a tender offer prior to a proposed initial business combination, or to receive distributions with respect to the Founder Shares upon the liquidation of the Trust Account if Globalink is unable to consummate a business combination. If an initial business combination, such as the Business Combination, is not completed by January 9, 2025 (and may be extended to June 9, 2025 on a monthly basis by resolutions of the Globalink Board, if requested by the Sponsor, and subject to the Sponsor depositing additional funds into the Trust Account), Globalink will be required to dissolve and liquidate. In such event, the 2,875,000 Founder Shares currently held by the Initial Stockholders, which were acquired prior to the IPO will be worthless because such holders have agreed to waive their rights to any liquidation distributions through the Insider Letter, in consideration for inducing Chardan, the representative of the underwriters for Globalink’s IPO to proceed with the IPO, and the economic benefit the IPO would confer upon the Initial Stockholders and PGM. The Founder Shares were purchased for an aggregate purchase price of $25,000, or less than $0.01 per share, and had a total market value of $34.36 million as of December 12, 2024. Accordingly, the Initial Stockholders will receive a positive rate of return so long as the market price of the common stock is at least $0.01 per share, even if public stockholders experience a negative rate of return in the Combined Company. | |
| ● | The fact that the Initial Stockholders’ Founder Shares became subject to a lock-up whereby, subject to certain limited exceptions, the Initial Stockholders agree not to offer, sell, contract to sell, pledge or otherwise dispose of, directly or indirectly, any of the Lock-up Shares, enter into a transaction that would have the same effect, or enter into any swap, hedge or other arrangement that transfers, in whole or in part, any of the economic consequences of ownership of the Lock-Up Shares or otherwise, publicly disclose the intention to make any offer, sale, pledge or disposition, or to enter into any transaction, swap, hedge or other arrangement, or engage in any Short Sales (as defined in the Lock-up Agreement) with respect to the Lock-Up Shares until (i) (w) with respect to one hundred percent (100%) of the Lock-Up Shares, the date from the Closing until the last date that is six (6) months after the date of the Closing, (x) with respect to ninety percent (90%) of the Lock-Up Shares, the 1st day of the 7th month from the Closing until the date that is nine (9) months after the date of the Closing, (y) with respect to seventy percent (70%) of the Lock-Up Shares, the 1st day of the 10th month from the Closing until the date that is twelve (12) months after the date of the Closing, (z) with respect to forty percent (40%) of the Lock-Up Shares, the 1st day of the 13th month from the Closing until the date that is fifteen (15) months after the date of the Closing, or (ii) if sooner, the date after the Closing on which Globalink consummates a liquidation, merger, share exchange or other similar transaction with an unaffiliated third party that results in all of Globalink’s stockholders having the right to exchange their equity holdings in Globalink for cash, securities or other property. For the avoidance of doubt, no Lock-up Shares shall be subject to the terms in the Lock-up Agreement from and after the date that is fifteen (15) months after the date of the Closing; |
| ● | The exercise of Globalink’s directors’ and officers’ discretion in agreeing to changes or waivers in the terms of the Business Combination may result in a conflict of interest when determining whether such changes or waivers are appropriate and in Globalink stockholders’ best interest. Globalink’s directors were aware of and considered these interests, among other matters, in evaluating the Business Combination, and in recommending to Globalink stockholders that they approve the Business Combination. Further, the interests of Globalink’s officers or directors may be different from or in addition to (and which may conflict with) your interests and may be incentivized to complete a less favorable business combination rather than liquidating Globalink. Public stockholders should take these interests into account in deciding whether to approve the Business Combination. | |
| ● | As of September 30, 2024, Globalink’s officers and directors have incurred an aggregate of $11,798 in out-of-pocket expenses. On each of September 5, 2023, September 29, 2023 and November 7, 2023, an affiliate of the Sponsor advanced $130,000 to the Company, for a total advance of $390,000, for Globalink’s extension payments. Unless Globalink consummates an initial business combination, Globalink’s officers, directors and the Sponsor (including its affiliates) will not receive reimbursement for any out-of-pocket expenses incurred by them to the extent that such expenses exceed the amount of available proceeds not deposited in the Trust Account. Upon the consummation of an initial business combination, Globalink’s offices, directors, and the Sponsor (including its affiliates) would be entitled to reimbursement for out-of-pocket expenses incurred by them from the Trust Account disbursements after redemption payouts and payments of deferred underwriting discounts. | |
| ● | The fact that, if the Trust Account is liquidated, including in the event Globalink is unable to complete an initial business combination within the required time period, the Sponsor has agreed to be liable to Globalink if and to the extent any claims by a vendor for services rendered or products sold to Globalink, or a prospective target business with which Globalink has discussed entering into a transaction agreement, reduce the amount of funds in the Trust Account. This liability will not apply with respect to any claims by a third party who executed a waiver of any right, title, interest or claim of any kind in or to any monies held in the Trust Account or to any claims under Globalink’s indemnity of the underwriters of the IPO against certain liabilities, including liabilities under the Securities Act. | |
| ● | As of the date hereof, the deadline that Globalink has to consummate its initial business combination has been extended for a total of eighteen times, and a total of US$1,950,000 has been deposited into the Trust Account in connection with the previous extensions. In connection with funding the extensions, Globalink has issued an aggregate of US$3.85 million in promissory notes to PGM, which are payable upon the Closing. In the event of a liquidation, PGM has waived its rights to any liquidation distributions through the Insider Letter. PGM is an affiliate of the Sponsor as its 95% equity holder has a familial relationship with the control person of the Sponsor. As a result, the Sponsor has an additional interest in causing Globalink to consummate its initial business combination due to its familial relationship with PGM. | |
| ● | After the Closing, the Sponsor will have the sole authority to handle various matters relating to the Escrow Account and indemnifications under the Merger Agreement. The Sponsor may have a conflict of interest when exercising such authority to handle matters relating to the Escrow Account and indemnifications after Closing, and they may not make decisions in the Globalink stockholders’ best interests. For details, see “Summary of the Proxy Statement/Prospectus—Parties to the Business Combination—Parent Representative.” |
These interests may have influenced Globalink’s directors in making their recommendation that you vote in favor of the approval of the Business Combination.
Upon the consummation of an initial business combination by Globalink, other than the PubCo ordinary shares that the Initial Stockholders are entitled to upon conversion of their shares of Globalink common stock at the completion of the Redomestication Merger, no additional compensation will be awarded to, earned by, or paid to the Sponsor, its affiliates or any promoters for all services rendered or to be rendered to Globalink and its affiliates. The Sponsor is expected to own an equity interest of approximately 1.7% in the Combined Company after the consummation of the Business Combination.
Prior to the consummation of an initial business combination by Globalink, the insider shares can only be transferred by the Initial Stockholders to (1) the Initial Stockholders or their respective affiliates or members, or to Globalink’s officers, directors, advisors and employees, (2) the Initial Stockholder’s affiliates or its members upon its liquidation, (3) to relatives and trusts for estate planning purposes, (4) by virtue of the laws of descent and distribution upon death of an Initial Stockholder, (5) pursuant to a qualified domestic relations order, or (6) by private sales made at prices no greater than the price at which the Insider Shares were originally purchased.
None of the Sponsor and Globalink’s directors and officers hold any interest in, or affiliation with, Alps Holdco. For a description of the fiduciary obligations held by Globalink’s officers and directors to other entities, see “Globalink’s Directors and Executive Officers — Conflict of Interest.”
| 116 |
Effects of the Business Combination
Effects on Globalink, the Sponsor, and their Respective Affiliates
Pursuant to the terms of the Merger Agreement, the Business Combination between Globalink and Alps Holdco will be effected in two steps: (i) subject to the approval and adoption of the Merger Agreement by the stockholders of Globalink, Globalink will be merged with and into PubCo, with PubCo remaining as the surviving publicly traded entity; and (ii) Merger Sub will merge with and into Alps Holdco, resulting in Alps Holdco remaining as the surviving entity and being a wholly-owned subsidiary of PubCo.
As part of the merger of Globalink and PubCo (a) Globalink, the Delaware corporation, shall cease to exist, (b) PubCo shall be the surviving company, (c) the name of the surviving company will be “ALPS Group”, (d) the authorized shares of the surviving company shall change from 500,000,000 shares of Globalink Common Stock and 5,000,000 shares of Globalink preferred stock to 495,000,000 PubCo ordinary shares of US$0.0001 par value each and 5,000,000 PubCo preferred shares of US$0.0001 par value each, (e) provisions specific to the Combined Company’s status as a special purpose acquisition company shall be eliminated making the Combined Company’s corporate existence of unlimited duration, (f) and certain other changes that PubCo Board deems appropriate for a public operating company will be made.
A summary of certain material differences between the amended and restated certificate of incorporation, as amended and currently in effect (the “Globalink Charter”) and the PubCo A&R Memorandum and Articles is presented separately and set forth in the section of this proxy statement/prospectus entitled “Comparison of Corporate Governance and Stockholders’ / Shareholders’ Rights.” A complete copy of the PubCo A&R Memorandum and Articles is attached to the proxy statement/prospectus as Annex B. You are encouraged to read them in their entirety.
The Initial Stockholders, including the Sponsor and the officers and directors of Globalink, have waived their right to redeem any shares of Globalink common stock in connection with a stockholder vote to approve a proposed initial business combination or sell any shares of Globalink common stock to Globalink in a tender offer prior to a proposed initial business combination, or to receive distributions with respect to the Founder Shares upon the liquidation of the Trust Account if Globalink is unable to consummate a business combination. If an initial business combination, such as the Business Combination, is not completed by January 9, 2025 (and may be extended to June 9, 2025 on a monthly basis by resolutions of the Globalink Board, if requested by the Sponsor, and subject to the Sponsor depositing additional funds into the Trust Account), Globalink will be required to dissolve and liquidate. In such event, the 2,875,000 Founder Shares currently held by the Initial Stockholders, which were acquired prior to the IPO will be worthless because such holders have agreed to waive their rights to any liquidation distributions through the Insider Letter, in consideration for inducing Chardan, the representative of the underwriters for Globalink’s IPO to proceed with the IPO, and the economic benefit the IPO would confer upon the Initial Stockholders and PGM. The Founder Shares were purchased for an aggregate purchase price of $25,000, or less than $0.01 per share, and had a total market value of $34.36 million as of December 12, 2024. Accordingly, the Initial Stockholders will receive a positive rate of return so long as the market price of Globalink common stock is at least $0.01 per share, even if public stockholders experience a negative rate of return in the Combined Company.
| 117 |
Effects on Holders of Globalink Public Shares
If Globalink is unable to complete a business combination by January 9, 2025 (and may be extended to June 9, 2025 on a monthly basis by resolutions of the Globalink Board, if requested by the Sponsor, and subject to the Sponsor depositing additional funds into the Trust Account), and is forced to liquidate, the per-share liquidation distribution will be $11.98 based on the balance in the Trust Account as of December 6, 2024. Furthermore, Globalink’s warrants and rights will expire worthless in the event Globalink fails to complete a business combination.
Upon the consummation of the Business Combination, the current equity holdings of the Globalink stockholders shall be exchanged as follows:
| (i) | Each share of Globalink common stock, par value $0.001 per share, issued and outstanding immediately prior to the effective time of the Redomestication Merger (other than any redeemed shares), will automatically be cancelled and cease to exist and for each share of Globalink common stock, PubCo shall issue to its holder (other than Globalink stockholders who exercise their redemption rights in connection with the Business Combination) one validly issued PubCo ordinary share, which shall be fully paid; | |
| (ii) | Each Globalink warrant to purchase one-half (1/2) of one share of Globalink common stock issued and outstanding immediately prior to effective time of the Redomestication Merger will convert into one PubCo warrant (or equivalent portion thereof). The PubCo warrants will have substantially the same terms and conditions as set forth in the Globalink warrants; and | |
| (iii) | The holders of Globalink rights (convertible into one-tenth (1/10) of one share of Globalink common stock) issued and outstanding immediately prior to the effective time of the Redomestication Merger will obtain the PubCo right in exchange for the cancellation of each Globalink right; provided, however, that no fractional shares will be issued and all fractional shares will be rounded to the nearest whole share. |
Upon the consummation of the Business Combination, non-redeeming public stockholders will experience substantial dilution to their interest in PubCo. The table below presents possible sources of dilution and the extent of such dilution that non-redeeming public stockholders could experience in connection with the closing of the Business Combination across a range of varying redemption scenarios. The maximum contractual redemption scenario represents the maximum contractual redemptions that may occur but which would still allow Globalink to have consolidated net tangible assets of at least $5,000,001 at the consummation of the Business Combination. The standalone requirement of 5,000,001 of Globalink net tangible assets is expected to be waived by the parties. In an effort to illustrate the extent of such dilution, the table below assumes that no Earnout Shares are issued.
Assuming No Redemptions | Assuming 25% Redemptions | Assuming 50% Redemptions | Assuming 75% Redemptions | Assuming Maximum Contractual Redemptions | ||||||||||||||||||||||||||||||||||||
Number of Shares | % | Number of Shares | % | Number of Shares | % | Number of Shares | % | Number of Shares | % | |||||||||||||||||||||||||||||||
| Alps Holdco Shareholders | 160,000,000 | 93.8 | % | 160,000,000 | 93.8 | % | 160,000,000 | 93.8 | % | 160,000,000 | 93.9 | % | 160,000,000 | 93.9 | % | |||||||||||||||||||||||||
| Globalink Public Shareholders | 277,511 | 0.2 | % | 208,133 | 0.1 | % | 138,755 | 0.1 | % | 69,377 | 0.0 | % | 0 | 0.0 | % | |||||||||||||||||||||||||
| Initial Stockholders | 2,875,000 | 1.7 | % | 2,875,000 | 1.7 | % | 2,875,000 | 1.7 | % | 2,875,000 | 1.7 | % | 2,875,000 | 1.7 | % | |||||||||||||||||||||||||
| Globalink Public Rights Holders | 1,150,000 | 0.6 | % | 1,150,000 | 0.7 | % | 1,150,000 | 0.7 | % | 1,150,000 | 0.7 | % | 1,150,000 | 0.7 | % | |||||||||||||||||||||||||
| PIPE Investors | 4,020,000 | 2.4 | % | 4,020,000 | 2.4 | % | 4,020,000 | 2.4 | % | 4,020,000 | 2.4 | % | 4,020,000 | 2.4 | % | |||||||||||||||||||||||||
| PGM | 627,000 | 0.4 | % | 627,000 | 0.4 | % | 627,000 | 0.4 | % | 627,000 | 0.4 | % | 627,000 | 0.4 | % | |||||||||||||||||||||||||
| IBDC Asia Sdn. Bhd. | 1,600,000 | 0.9 | % | 1,600,000 | 0.9 | % | 1,600,000 | 0.9 | % | 1,600,000 | 0.9 | % | 1,600,000 | 0.9 | % | |||||||||||||||||||||||||
| Total Shares | 170,549,511 | 100 | % | 170,480,133 | 100 | % | 170,410,755 | 100 | % | 170,341,377 | 100 | % | 170,272,000 | 100 | % | |||||||||||||||||||||||||
| 118 |
Ownership of the Combined Company after the Business Combination
Upon consummation of the Business Combination (assuming, among other things, that no additional public stockholders exercise redemption rights in connection with the Closing and the other assumptions described under the section with the heading “Frequently Used Terms — Share Calculations and Ownership Percentages”), (i) Globalink’s public stockholders and Globalink’s right holders are expected to own approximately 0.8% of the outstanding ordinary shares of Combined Company, (ii) the Sponsor, the Initial Stockholders and PGM, an affiliate of the Sponsor, are expected to own approximately 2.1% of the outstanding ordinary shares of Combined Company, (iii) the Alps Holdco Shareholders are expected to own approximately 93.8% of the outstanding ordinary shares of Combined Company, (iv) the PIPE Investors are expected to own approximately 2.4% of the outstanding ordinary shares of Combined Company, assuming the PIPE Investors will hold 4,020,000 PubCo ordinary shares, and (v) IBDC Asia Sdn. Bhd., an advisory firm to Alps, is expected to own approximately 0.9% of the outstanding ordinary shares of Combined Company, representing finder’s fees payable in PubCo ordinary shares with value equal to 1% of the aggregate consideration for the Business Combination of US$1.6 billion.
These percentages assume, among other assumptions, that at, or in connection with, the Closing, (i) the PIPE Investment is consummated in accordance with its terms for aggregate proceeds of $40,200,000, (ii) no additional public stockholders exercise their redemption rights in connection with the Business Combination, and (iii) 160,000,000 ordinary shares of Combined Company are issued to former Alps Holdco Shareholders in accordance with the Merger Agreement, with no Earnout Shares are issued. If actual facts are different from these assumptions, the percentage ownership retained by the Globalink stockholders and Alps Holdco Shareholders in the Combined Company, and associated voting power, will be different.
PubCo A&R Memorandum and Articles
Pursuant to the Merger Agreement, conditional upon approval of the Redomestication Merger Proposal, the Acquisition Merger Proposal, and upon the Closing, the PubCo will adopt the PubCo A&R Memorandum and Articles. We currently also expect that upon the Closing, PubCo’s memorandum and articles of association will be amended and restated promptly to:
| ● | reflect necessary changes and to be consistent with the PubCo A&R Memorandum and Articles; and | |
| ● | make certain other changes that our board of directors deems appropriate for a public operating company. |
Name and Headquarters of the Combined Company
The name of the Combined Company will be ALPS Group or such other name as determined by the Combined Company Board, and the headquarters of the Combined Company will be located at Kuala Lumpur, Malaysia.
Background of the Business Combination
Globalink is a blank check company incorporated in the State of Delaware for the purpose of effecting a merger, share exchange, asset acquisition, share purchase, recapitalization, reorganization or similar business combination with one or more target businesses. The proposed Business Combination was the result of an extensive search for potential transactions utilizing the network of Globalink’s management team, the Board and Globalink’s advisors.
Prior to the consummation of the IPO, neither Globalink, nor anyone on its behalf, engaged in any substantive discussions, directly or indirectly, with any business combination target regarding a business combination with Globalink.
After the completion of the IPO, Globalink assessed and analyzed multiple prospective business combination targets and opportunities based on strength of business model, management team and growth prospects, among other factors. Representatives of Globalink contacted, and were contacted by, a number of individuals and entities with respect to potential business combination opportunities. Globalink evaluated, conducted preliminary due diligence based on publicly available information and other market research and engaged in various levels of discussions with, a number of different potential business combination targets. In connection with such evaluation, preliminary due diligence and discussions, Globalink entered into discussions or negotiations with representatives of more than 30 potential business combination targets. After reviewing and considering these opportunities, Globalink management determined that Alps offered the most compelling business combination opportunity, and Globalink entered into a letter of intent with Alps on October 23, 2023.
Alongside the outreach and pre-letter of intent discussions with Alps, Globalink also met and conducted discussions with representatives of, and commenced preliminary due diligence on, several other potential target opportunities. In particular, Globalink entered into a merger agreement with Tomorrow Crypto on August 3, 2022 regarding the business combination between Globalink and Tomorrow Crypto. The merger agreement was subsequently terminated on March 8, 2023 by Globalink. The Globalink Board decided to terminate the proposed business combination with Tomorrow Crypto primarily due to concerns over low Bitcoin trading prices at that time and the tightening regulatory environment that cryptocurrency industry players face and the associated uncertainties and unpredictability of such companies’ business prospects.
| 119 |
Other major targets which Globalink met and conducted discussions with include: (i) an electric vehicle brand and manufacturing company based in Indonesia; (ii) an Australian copper mining company; (iii) a Japan headquartered blood diagnostic company that employs self-test kits and interactive I.T. technologies to provide consulting services; (iv) a new generation e-commerce business in Japan that empowers social media influencers to build independent businesses; and (v) a beauty and cosmetics company with original equipment manufacturer (“OEM”) and house brands targeting Japanese consumers. Globalink entered into non-binding and non-exclusive letters of intent with each of the five target companies referred to above, and Globalink ultimately decided to abandon each of such other potential target opportunities because of various reasons. Globalink terminated discussions with the Australia copper mining company in March 2023 because the potential target company could not reach an agreement with Globalink as to the distribution of money left in the Trust Account after redemption upon the consummation of an initial business combination. Globalink terminated discussions with the Indonesian electric vehicle brand in May 2023 because the Globalink Board was of the opinion that the electric vehicle market was highly competitive, and the success of an electric vehicle brand requires high investment and a long period of time to achieve profitability, and involves significant technological uncertainties, and the Globalink Board decided that the potential target was not an ideal target to conduct business combination with. The Globalink Board terminated negotiations with the Japanese blood diagnostic company and the Japanese e-commerce company in July 2023 and December 2023, respectively, because both potential targets did not have the capacity to secure financings for a substantial PIPE investment to be closed simultaneously with the closing of the Business Combination. The Globalink Board terminated negotiations with the Japanese beauty and cosmetics company in June 2023 because the potential target identified another special purpose acquisition company that better suited their needs.
On August 18, 2023, a business advisor, IBDC Asia Sdn. (IBDC”) arranged a meeting with Globalink’s Chairman of the Board of Directors and Chief Executive Officer, Mr. Say Leong Lim, regarding a potential De-SPAC transaction between Globalink and Alps. IBDC is a common business acquaintance of the Chief Executive Officers of both Globalink and Alps, and is not affiliated with either Globalink or Alps. IBDC briefed Mr. Lim on the profile and background of Alps and Alps’ plans to seek a public listing in the U.S. for its growth. Since the beginning of 2023, Alps had been preparing for a traditional initial public offering and listing on Nasdaq. No specific merger terms were discussed during this meeting, Mr. Say Leong Lim informed IBDC about Globalink’s objectives and timeline to consummate a business combination and expressed interest in exploring De-SPAC transaction and suggested to schedule a meeting between senior management teams of Alps and Globalink.
On September 19, 2023, Mr. Lim and representatives of Alps, including Dr. Tham Seng Kong, the founder and CEO of Alps, held a meeting to discuss a potential De-SPAC transaction between Globalink and Alps. In the meeting, Mr. Lim introduced Globalink and the concept of a De-SPAC transaction, indicating the criteria of Globalink’s target selections and expectations. In turn, Dr. Tham shared Alps’ strategic plans, including the reasons for considering a De-SPAC transaction as an option to go public and become listed on Nasdaq. Dr. Tham explained that the key objective of Alps is to have a publicly listed platform for growth, expansion, working capital and to increase investor confidence in their business. The listing will also enhance Alps’ international visibility, shareholder liquidity, and overall corporate governance. Both parties agreed that mutual interest are aligned, and Alps and Globalink would create a good partnership. Therefore, the Alps team expressed interest in exploring a De-SPAC transaction as an alternative to a traditional IPO for several strategic reasons, particularly considering the presence of PIPE investors.
The Alps management team conducted an evaluation of the proposed business combination and the advantages and disadvantages of a De-SPAC transaction. Alps management are of the view that a De-SPAC transaction offered more flexibility and favorable terms compared to a traditional IPO, particularly in terms of speed to market, valuation and investment conditions.
A De-SPAC transaction is typically faster than a traditional IPO. The timelier speed to market would allow Alps to accelerate its development plans, including advancing multiple product candidates and preparing for commercialization if any of these candidates are approved. For a De-SPAC, the transaction and valuation is agreed upon during negotiations with the SPAC. This provides greater pricing certainty for Alps.
Alps emphasized that listing on Bursa Exchange or the Singapore Exchange would confine Alps to a more regional presence, while Nasdaq provides a global platform. Alps chose Nasdaq to align with its ambition to become a global company and leverage the international visibility and opportunities that come with it.
Overall, Alps viewed the De-SPAC transaction as a strategically advantageous and efficient pathway to becoming a publicly traded company while securing the necessary capital to support its development objectives.
On September 25, 2023, Mr. Lim of Globalink, Mr. Kelvin Chin, the Chief Financial Officer of Globalink, visited Alps to view its office and facilities. Representatives of Alps, including Dr. Tham, had further questions about the differences between a traditional initial public offering and a De-SPAC transaction, particularly in regards to timing, dilution, raising additional capital, and due diligence. During the discussion, the representatives of Alps highlighted to the Globalink representatives the growth potential of the Southeast Asian market and Alps’ plan and strategies to grow its business and expand its operations post-Closing. The Alps representatives asked the Globalink representatives whether any mechanism could capture Alps’ growth potential which can be reflected in the terms of the Merger Agreement. Both parties explored the idea of using earn-out shares as a performance indicator and incentive for Alps in achieving revenue or share price milestones. Both parties also discussed how Globalink’s existing management team can support the De-SPAC process. Globalink indicated to Alps that an Insider Letter was already in place, and Globalink Support Agreement would be in place to demonstrate the Initial Stockholders’ commitment. In conclusion, Alps requested the execution of a non-disclosure agreement and a draft of letter of intent. During the same meeting, Globalink and Alps discussed the expected pre-money valuation, with Alps indicating an expected range of $1.5-1.7 billion.
On October 10, 2023, Globalink informed Alps that Chardan is the M&A and capital markets advisor to Globalink, and Hunter Taubman Fischer and Li LLC is the U.S. securities legal counsel of Globalink. Further discussions took place between Globalink and Alps regarding the proposed business combination and U.S. capital market outlook.
On October 23, 2023, Globalink and Alps entered into a confidentiality agreement and executed a non-binding letter of intent. Alps subsequently provided Globalink with access to an online data room and certain other information for purposes of GLLI conducting a business, legal and financial due diligence. This non-binding letter of intent included a $1.5 billion pre-money valuation, which was subject to due diligence reviews and board approvals. Representatives of Globalink performed a high-level due diligence on Alps and participated in phone calls and working sessions with representatives of Alps regarding diligence and other commercial and legal matters.
On November 8, 2023, representatives of Alps, including Dr. Tham, hosted meeting with representatives of Globalink at Alps’ office in Kuala Lumpur, including Mr. Lim and Mr. Kelvin Chin, discussing updates on Alps’ business, operations, the competitive environment, future growth opportunities, and Alps’ commitment to the environment, social and governance. Globalink was also introduced to Alps’ Malaysian legal counsel, Darryl, Edward & Co. It was noted in the meeting that the last private fund raise of Alps in September 2023 totaled US$2.5 million, at a valuation of US$1.0 billion. Dr. Tham informed the Globalink team that several potential investors had expressed verbal interest in investing in Alps at a valuation of US$1.6 billion. Alps committed to Globalink that the PIPE investment will be secured after a definitive business combination agreement is finalized. Consequently, both parties mutually agreed to increase the valuation of Alps from the initial valuation of US$1.5 billion in the letter of intent to US$1.6 billion in recognition of the PIPE commitments.
On November 13, 2023, Globalink and Alps had an online meeting to discuss the status of the due diligence and PCAOB audit as well as the timeline, investment highlights and mitigating factors of the potential De-SPAC transaction.
On November 16, 2023, the parties met and discussed cashflows, net assets, revenues, investor presentation, data room updates, potential earnouts and the composition of board, which consists of five seats, with one seat to be appointed by Globalink. During discussions, Globalink representatives acknowledged Alps’ growth potential and proposed including an earn-out provision in the Merger Agreement. Alps suggested the earn-out constitute 50% of the total merger consideration, based on a US$1.6 billion valuation, with milestones set over eight years following the Closing. Globalink countered with a proposal for the earn-out to represent 20% of the total merger consideration, with milestones over four years. Ultimately, both parties agreed on an earn-out of 30%, with milestones over five years, deeming this timeframe sufficient for Alps to implement its growth strategies post-Closing. Both parties agreed that revenue of the Combined Company would serve as the key performance indicator as it can represent the growth rate of a life science company reasonably accurately, and Globalink reviewed Alps’ historical revenues of the fiscal years ended March 31, 2022 and 2023. The Alps representatives proposed revenue milestones of US$5 million, US$10 million, US$16 million, US$23 million and US$31 million for the five years after the Closing. The Globalink representatives countered with higher targets of US$7 million, US$14 million, US$23 million, US$34 million and US$56 million, respectively. The Alps representatives accepted Globalink’s proposal, and both parties reached an agreement as to the size of the earn-out and the applicable milestones.
| 120 |
On November 23, 2023, Globalink and Alps discussed potential engagement of Investor Relations and Public Relations for the business combination.
On December 14, 2023, representatives of Globalink, including Mr. Say Leong Lim and Mr. Kelvin Chin, met with representatives of Alps, including Dr. Tham, to further discuss business, legal and financial due diligence matters. The Globalink team also went through the terms of the proposed non-binding letter of intent, including a revised valuation of $1.6 billion. Globalink team held an online meeting with Chardan providing an update on progress and status.
On January 5, 2024, Alps informed Globalink on the capital and targeted PIPE capital raise with A and B teams assigned to identify potential local and foreign investors. Team A was primarily focusing on investments from ultra-high net worth individuals, while Team B is focusing on potential investments from institutions and corporations. Team A, led by Alps, identified several potential PIPE investors who were not affiliated with Globalink, the Sponsor, Alps, or Alps’ affiliates. Globalink and Alps reached a preliminary agreement to the material terms and conditions governing the PIPE Investment, with the form of subscription agreement to be finalized by Globalink’s outside legal counsel, Hunter Taubman Fischer and Li LLC. A copy of the draft Business Combination Agreement and Alps’ Teaser Deck will be provided to the potential investors to evaluate the investment opportunity. Alps’ Teaser Deck includes certain information about Alps, including its corporate history, business model and operations, its growth strategies and visions, research and development team and projects, and introduction of its board of directors. Mr. Lim of Globalink met with Dr. Tham of Alps in person to have follow-up discussions on business, legal and financial due diligence matters.
On January 19, 2024, Hunter Taubman Fischer and Li LLC circulated a draft of the Merger Agreement to the team for review and finalization. The draft of the Merger Agreement included terms relating to the set-up of an escrow account, holding certain percentage of Merger Consideration Shares as the sole source of payment for the obligations of the Alps Holdco Shareholders for their indemnification obligations under the Merger Agreement, for the relevant parties’ consideration and discussions. Over the next few days, representatives of Globalink and its counsel and advisors liaised and discussed the draft Merger Agreement with Alps and its outside counsel and advisor, and held a number of phone calls and working sessions regarding diligence and other commercial and legal matters and agreed on various outstanding terms with respect to the De-SPAC transaction, including, among others, (i) closing conditions; (ii) transaction structure; (iii) valuation; (iv) financial reporting periods; (v) corporate governance matters; (vi) break-up fee arrangements; and (vii) representations, warranties, approvals and covenants to be provided by the parties under the Merger Agreement. Representatives of Globalink and Alps also considered the inclusion of an escrow account and determined it was reasonable to both parties. As of the date of this proxy statement/prospectus, no draft escrow agreement has been circulated and negotiated among Globalink, Alps and the escrow agent.
On January 23, 2024, the Globalink team, including its directors Beh Hong Shien, Lai Kian Huat, Agnes Wong, and management team, Say Leong Lim and Kelvin Chin, held an internal meeting to discuss and review the following items such as (i) the target’s business, growth potential of the industry, and the plan of expanding into Southeast Asia and Middle East markets; (ii) the target’s management team; (iii) due diligence results; (iv) the form, terms and conditions of the Merger Agreement, including all exhibits and schedules attached thereto and the transactions contemplated therein (including the Business Combination); (v) risks and mitigants; and (vi) the target’s valuation based on internal assessment using Alps’ forecasted discounted cashflow, price/revenue multiples, peer comparison, the special listing requirements of biotechnology companies under Regulation 18a of Hong Kong Stock Exchange, and the amount of PIPE Investment.
Regulation 18a of the Hong Kong Stock Exchange sets out additional listing conditions and disclosure requirements for biotech companies that seek to list publicly but are unable to satisfy the typical IPO profit, revenue/cashflow/market capital requirements. Under the regulation, a biotech company needs to meet certain key criteria, including the following:
| ● | developed at least one core product beyond the concept stage or if the applicant has not completed any clinical trials, the Hong Kong Stock Exchange will evaluate the reason why and whether any substantive research and development and other work equivalent to the completion of one clinical trial has been performed; | |
| ● | has been undertaking research and development activities since at least 12 months prior to proposed listing; | |
| ● | funds raised from listing is for research and development to bring core product(s) to commercialization; | |
| ● | the company has either registered patents, patent application(s) and/or intellectual property in relation to its core products or joint developments; and | |
| ● | have a sophisticated investor invested in the company at least six months before the proposed listing and will stay through the IPO, with a 1% or more equity interest if at a valuation of HKD8 billion or above. |
| 121 |
Globalink’s chief financial officer prepared the forecasted discounted cashflow projection, which was presented by Globalink’s chief executive officer and chief financial officer to the Globalink Board on January 23, 2024 in Kuala Lumpur, Malaysia. The forecasted discounted cashflow was prepared based on existing sources of revenue. The Globalink Board reviewed the presentation from Globalink’s chief executive officer and chief financial officer, followed by a Q&A and discussion session with the Globalink’s management. Alps’ forecasted discounted cashflow and price/revenue multiples tables are as follows:
A. Forecasted Discounted Cashflow (Rate of Discount – 14.00%)
| INCOME | EXPENSES | CASH OUTFLOW | NET CASH | DISCOUNTED CASH FLOW | ||||||||||||||||||||||||
| YEAR FOLLOWING CLOSING | CASH INFLOW | FIXED | VARIABLE | FIXED + VARIABLE | INFLOW LESS OUTFLOW | PRESENT VALUE | CUMULATIVE PRESENT VALUE | |||||||||||||||||||||
| 1 | USD | 13,629,636.00 | USD | 2,846,994.96 | USD | 7,087,410.72 | USD | 9,934,405.68 | USD | 3,695,230.32 | USD | 3,241,430.11 | USD | 3,241,430.11 | ||||||||||||||
| 2 | USD | 23,032,764.00 | USD | 2,846,994.96 | USD | 11,977,037.28 | USD | 14,824,032.24 | USD | 8,208,731.76 | USD | 6,316,352.54 | USD | 9,557,782.64 | ||||||||||||||
| 3 | USD | 39,246,092.00 | USD | 3,910,824.69 | USD | 21,192,889.68 | USD | 25,103,714.37 | USD | 14,142,377.63 | USD | 9,545,702.07 | USD | 19,103,484.72 | ||||||||||||||
| 4 | USD | 67,401,124.00 | USD | 3,910,824.69 | USD | 36,396,606.96 | USD | 40,307,431.65 | USD | 27,093,692.35 | USD | 16,041,640.88 | USD | 35,145,125.60 | ||||||||||||||
| 5 | USD | 116,613,743.00 | USD | 3,910,824.69 | USD | 62,971,421.22 | USD | 66,882,245.91 | USD | 49,731,497.09 | USD | 25,828,981.22 | USD | 60,974,106.82 | ||||||||||||||
The above table was prepared by Alps accounting and business development personnel for Globalink’s information and its review of Alps’ projected growth and performance. Alps used the estimated management accounts for the year ended March 31, 2024 as the base for the forecasts. Percentages of revenue and costs averages are from Alps’ historical financial statements for the fiscal years ended March 31, 2021, 2022 and 2023. Key assumptions of the projected financial results are as follows:
| i. | the forecasts are for the Malaysian market only (except for viii below which is targeted into the Indonesian market); | |
| ii. | discount rate of 14%; | |
| iii. | foreign exchange rate at USD1 to Malaysian Ringgit of 4.70; | |
| iv. | 83% increase p.a for sales of wellness products and services; | |
| v. | 66% increase p.a for sales of aesthetics and cosmetic products and services; | |
| vi. | 57% increase p.a for hair implants and transplant; | |
| vii. | 59% increase p.a for DNA and mRNA testing services; | |
| viii. | 50% increase p.a for blood diagnostic lab test services; | |
| ix. | where historical trend or data is unavailable, revenues are forecasted based on number of patients multiplied by the average costs of products/services; | |
| x. | variable costs are 52% of income for years 1 and 2 and 54% of income in Year 3, 4 and 5 following Closing respectively; | |
| xi. | fixed costs are USD1,889,548 with stepped increase of USD957,447 in Year 1 following Closing and USD1,063,830 in Year 3 following Closing; | |
| xii. | Alps has funds to execute its growth strategies; | |
| xiii. | As at March 31, 2024, Alps has discontinued Covid-19 screening services. Therefore, growth projections do not rely on these COVID-19 PCR testing services; | |
| xiv. | Alps’ NK Cell, Car-T and Mesenchymal Stem Cells treatments are able to treat certain targeted non communicable diseases such as pancreatic, prostate, lung, cervical, breast and colorectal cancers, non-Hodgkin’s lymphoma, kidney failure, lung disease, stroke and diabetes; | |
| xv. | For the NK Cell segment, projected revenue for Year 1 following Closing, is calculated based on an estimated total customer base of 22,348 patients, representing higher-income individuals with greater disposable income, with an assumed penetration rate of 1.6% and an anticipated price per treatment of USD11,500. Future revenue for this segment is projected to grow at an annual rate of 83.0% based on Alps’ estimate. As at March 31, 2024, Alps has not commenced operation for NK Cells segment; | |
| xvi. | For the Car-T segment, projected revenue is based on an estimated customer base of 1,940 patients, assuming a penetration rate of 1.6% and a projected price per treatment of USD46,250. Revenue for this segment is expected to grow at an annual rate of 83.0% in subsequent periods based on Alps’ estimate. As at March 31, 2024, Alps has not commenced operation for Car-T segment; | |
| xvii. | In arriving at the market that Alps can capture and service for NK Cell, Car-T and Mesenchymal Stem Cells treatments, the market size i.e potential number of patients in Malaysia affected with such identified conditions were identified. Alps has targeted the top 20 percentile patients by income profile, representing higher-income individuals with greater disposable income. Applying a conservative penetration rate of 8%, Alps projects a market capture rate of approximately 1.6%. This strategic focus aims to align Alps’ services with individuals more likely to afford advanced treatments; | |
| xviii. | Alps surveyed via calls and market surveys of other companies that provided similar products and services in order to gauge the ongoing market rates and the estimated costs of treatments; | |
| xix. | Alps expects its blood testing lab services to be a stable and growing revenue source due to the collaborations and partnerships that Alps intends to execute and further, the applicability of these services across various patient segments and its role in early diagnosis, regular monitoring, and post-treatment follow-ups. For this segment, projected revenue for Year 1 following Closing, is based on an anticipated volume of 182,500 tests, with an estimated price per test of USD24. Alps’ anticipates that revenue for this segment will grow at an annual rate of 50.0% thereafter. As at March 31, 2024, Alps has not commenced operation for blood testing services; | |
| xx. | Alps’ assumes that its NK Cell, Car-T and Mesenchymal Stem Cells treatments can be utilized in the Malaysian market under compassionate use, pending regulatory approval; and | |
| xxi. | the respective projected revenues from NK Cell, Car-T, Mesenchymal Stem Cells treatments and blood testing services have been determined based on Alps’ management assessment of the current market outlook and future strategic business plans. |
A higher discount rate will result in a lower present value of future cash flows, and vice versa. The 14% discount rate represents the opportunity costs of capital, reflecting the rate of return to compensate for the time value of money with the expected rate of inflation in Malaysia at 2.0% and the risks profile associated with Alps’ business.
The revenue growth rates took into consideration the trends and outlook of the markets that Alps operates in and Alps’ competitive position. Forecasted cost increases considered potential changes in material, labor and operating costs and also the impact of operational improvements or efficiency. The rates represent historical performances of Alps’ past growth trends and expectations provided by Alps’ management. In addition, the revenue growth is prudently in line with selected annual industry revenue averages of 117%.
The financial forecast reflects numerous estimates and assumptions with respect to the general business, economic, regulatory, market and financial conditions and other future events, as well as matters specific to Alps’ business, all of which are difficult to predict and many of which are beyond Alps’ and Globalink’s control.
There are factors that may impact the assumptions, including:
| 1. | Economic conditions: macroeconomic trends, such as inflation and interest rates, and industry-specific economic factors that can impact demand, pricing, and costs. | |
| 2. | Market conditions: competitive landscape, including the actions of competitors and their impact on market share, pricing, and profitability, and changes in consumer preferences and behavior. | |
| 3. | Company-specific factors: revenue drivers, such as sales volume, pricing, and customer base, and cost structure, including labor, materials, and overhead expenses; investments in new products, services, or technologies; and management strategies and decision-making. | |
| 4. | Financial factors: access to financing and tax implications, including changes in tax rates or regulations. | |
| 5. | Regulatory and legal environment: changes in industry regulations, government policies, or laws that can impact the business, and compliance requirements and associated costs. | |
| 6. | Risks and uncertainties: potential disruptions, such as supply chain issues, technological changes, or geopolitical events, and exposure to foreign exchange fluctuations. |
The Company has relied on the following Alps’ historical revenue to come into conclusion with the above projections: -
| USD unless otherwise specified | FY’2021 ($) | FY’2022 ($) | FY’2023 ($) | FY’2024 ($) | Year 1 following Closing ($) | |||||||||||||||
| Wellness and others | ||||||||||||||||||||
| ● MSC Cells | 173,434 | 396,128 | 790,692 | 959,706 | 1,756,262 | |||||||||||||||
| ● NK Cells (1) | 0 | 0 | 0 | 0 | 4,112,032 | |||||||||||||||
| ● CAR-T Cells (2) | 0 | 0 | 0 | 0 | 1,435,600 | |||||||||||||||
| Cosmetic surgery and others | 8,529 | 104,697 | 120,612 | 261,400 | 433,924 | |||||||||||||||
| Hair transplant | 22,159 | 332,743 | 493,454 | 817,398 | 1,283,315 | |||||||||||||||
| DNA testing and mRNA testing | 178,600 | 131,615 | 82,545 | 131,134 | 208,502 | |||||||||||||||
| Blood Diagnostics (3) | 0 | 0 | 0 | 0 | 4,440,000 | |||||||||||||||
| COVID-19 screening services (4) | 1,400,000 | 1,700,000 | 1,100,000 | 0 | 0 | |||||||||||||||
| Total | 13,629,635 | |||||||||||||||||||
| (1) | As at the date of March 31, 2024, Alps has not commenced operation for NK Cells treatment. The projected revenue for the Year 1 following Closing is based on a publicly available information and information from certain data sources. These sources provided the following number of patients: 1,0891, 2,1462, 5,1393, 1,7404, 8,4185 and 3,8166. The aggregated estimate indicates a patient’s number of approximately 22,348 and, with an assumed penetration rate of 1.6%, and an estimated price of $11,500 averaged from market enquiries for each treatment, totaling a total revenue of $4,112,000. | |
| (2) | As at the date of March 31, 2024, Alps has not commenced operation for Car-T Cells treatment. The projected revenue for Year 1 following Closing is based on an estimated patient’s number of approximately 1,9407, assuming a penetration rate of 1.6% and an estimated price of $46,250 for each treatment, totaling a total revenue of 1,436,000. The source of reference is made with reference to other 3rd party providers in similar scale and size with Alps. | |
| (3) | As at the date of March 31, 2024, Alps has not commenced operation for blood testing services. The projected revenue for Year 1 following Closing is based on an estimated patient’s number of 182,500 equivalent to 100% of Alps’ potential partner’s Indonesian market share, at an estimated price of $24 for each product and/or service, totaling a total revenue of $4,440,000. The source of reference is based on Alps’ potential partner’s market shares in the Indonesian market. Alps intends to collaborate with the potential partner to jointly operate the potential Indonesian partner’s existing laboratory business. | |
| (4) | As at the date of March 31, 2024, Alps has discontinued Covid-19 screening services. Therefore, Covid-19 screening services is not considered in Year 1 following Closing. |
For the computation of average historical growth rates above, Alps’ management has adopted the actual revenue figures in Ringgit Malaysia (RM), which is the functional currency. This reflects the actual historical transaction currencies and operating environment, where USD was previously used as the presentation currency.
| 122 |
In determining the future growth rates, Alps’ management has focused on certain products and services that have demonstrated a track record of substantial contributions. These products and services are anticipated to exhibit promising growth prospects in the future: -
Average Historical Growth Rate p.a. | FY’2021 | FY’2022 | FY’2023 | FY’2024 | ||||||||||||||||
| Wellness and others | 83.0 | % | N/A | 128.4 | % | 99.6 | % | 21.4 | % | |||||||||||
| Cosmetic surgery and others (1) | 66.0 | % | N/A | 1,127.5 | % | 15.2 | % | 116.7 | % | |||||||||||
| Hair transplant (2) | 57.0 | % | N/A | 1,401.6 | % | 48.3 | % | 65.6 | % | |||||||||||
| DNA testing and mRNA testing (3) | 59.0 | % | N/A | -26.3 | % | -37.3 | % | 59.0 | % | |||||||||||
| (1) | The growth rate for cosmetic surgery and others in FY’2022 is not considered in the computation of the average historical growth rate. | |
| (2) | The growth rate for hair transplant in FY’2022 is not considered in the computation of the average historical growth rate. | |
| (3) | The computation of the average historical growth rate for DNA testing and mRNA testing is based solely on the fiscal years 2023 and 2024. |
Alps’ management is of the opinion that CAR-T cells, NK cells and blood diagnostic services demonstrate a positive current market outlook and potential future business opportunities. The management’s assessment of these products and services is based on the following grounds: -
CAR-T Cell
| ● | CAR-T cell therapy have demonstrated its application in treating patients with select liquid tumors in the relapsed and refractory stage, and CAR-T are increasingly being investigated for solid tumors and other hematologic cancers. | |
| ● | The industry faces a viral-vector capacity constraint and a limited number of third-party suppliers. There is massive value for efficient gene-transfer tools that enable rapid modification of T-Cells with CARs (such as transposon, CRISPR, among other). | |
| ● | Allogenic CAR-T cell therapies can provide several significant advantages in manufacturing, including reduced costs of goods sold; a simplified supply chain and avoidance of issues with autologous CAR-T cells in harvesting, long vein-to-vein time and T-cell dysfunction. | |
| ● | There are growing efforts to explore combination therapies involving CAR-T with other treatments, such as checkpoint inhibitors, monoclonal antibodies, or targeted therapies. | |
| ● | Compared with inpatient reimbursement, treatment in outpatient facilities can increase number of patients who would benefit from CAR-T and improve economics for Alps. | |
| ● | Currently, CAR T therapies use centralized manufacturing sites that serve multiple clinical centers, presenting logistical challenges. Alps’ bench to bedside treatment of CAR-T cells, that is, manufacturing on-site at the facilities could massively increase capacity and reduce vein-to-vein time and costs. |
NK Cells
| ● | NK cell therapies are gaining attention as an effective form of immunotherapy, complementing T-cell therapies like CAR-T. | |
| ● | Innovations in genetic engineering are enhancing the efficacy and persistence of NK cells. Techniques such as CRISPR are being used to modify NK cells, allowing them to better target specific cancer antigens and evade tumor immune suppression. | |
| ● | A significant number of clinical trials are underway to evaluate the safety and efficacy of NK cell therapies across various types of cancers, including hematological malignancies and solid tumors. Positive early results are contributing to optimism about their future. | |
| ● | Development of scalable manufacturing processes for NK cells, such as large-scale expansion and cryopreservation techniques, is crucial for commercial viability and accessibility of these therapies. | |
| ● | The efficacy of next generation CAR-NK cells has been explored for several solid tumors, including ovarian and breast cancers, as well as glioblastoma. | |
| ● | NK cell therapies are anticipated to become an integral part of standard cancer treatment protocols, especially as combination therapies demonstrate synergies with existing modalities. |
Blood Diagnostics
| ● | Blood diagnostics shows potentials in adopting data-dependent products such as such as next-generation sequencing, companion diagnostics, and microfluidics. | |
| ● | A significant trend in the industry is the adoption of point-of-care testing, which facilitates immediate diagnostic results. | |
| ● | Diagnostic workflows are being enhanced through the application of AI and machine learning, which enable sophisticated data analysis, thereby improving Alps’ economics of dispensing blood testing services. | |
| ● | Blood diagnostics is able to identify genetic mutations or biomarkers that inform individualized treatment plans for cancer, cardiovascular diseases, and infectious diseases. | |
| ● | Blood diagnostics market is projected to experience substantial growth, driven by the increasing prevalence of chronic diseases, technological advancements, and the demands of an aging population. | |
| ● | Diagnostic labs are expanding their offerings by developing comprehensive test panels to simultaneously screen for multiple diseases. | |
| ● | The integration of telemedicine with blood diagnostics is gaining momentum, enabling remote monitoring and home-based testing options to meet evolving patient needs. | |
| ● | Growing awareness of health disparities is encouraging the development of equitable access to blood diagnostics, particularly in underserved communities, through innovative outreach and testing initiatives. |
Based on the above outlook, Alps’ management intends focus on its future strategic business plans that includes: -
| ● | improve operational agility and equipment utilization to manufacturing-site capacity for individual pipelines, introducing the ability to run multiple products in fewer lines, and thereby improving Alps’ readiness to respond quickly to the needs of a volatile market. | |
| ● | undertaking the right partnerships or collaboration decisions to expand capacity, which may encompass leveraging on shared technologies, asset deployment, or capital-expenditure efficiency. | |
| ● | improving manufacturing efficiency and supply chain to manage inventory, distribution logic, and the complexities of the cold chain network. | |
| ● | considering a presence and market awareness in emerging markets and the associated cost, regulatory and market-access implication with great care. | |
| ● | building or acquiring strong, competitive regional network with the right suppliers, manufacturing plants and distribution capabilities to balance cost, service and patient acceptance. | |
| ● | promote sustainable practices and reduce operating costs by methodically adopting lean practices and improving process technology. |
Alps’ revenue forecasts are grounded in the abovementioned current market outlook and future business plan. These revenue forecasts are underpinned by an analysis of its (1) future market size and growth potential, (2) patient demographics and potential penetration into emerging markets, (3) existing and potential partnerships, collaboration, and distribution networks, and (4) ongoing availability of financial resources.
The specific information estimates and assumption for this projection included in this proxy statement/prospectus were prepared at the request of and disclosed to Globalink as part of its overall evaluation of Alps. These projections were provided to the Globalink Board for consideration and evaluation purposes. Alps does not warrant the accuracy, reliability, appropriateness, or completeness of the projections, whether to Globalink or to any other party. Neither Alps’ management nor its representatives, advisors, or affiliates make any representations regarding the actual future performance of Alps compared to the projections provided. Additionally, Alps does not plan to reference these projections in its future periodic reports filed under the Exchange Act.
The financial projections are forward looking statements that are inherently subject significant uncertainties and contingencies, many of which are beyond Alps’ and Globalink’s control. The various risks and uncertainties include those set forth in “Risk Factors,” “Globalink’s Management’s Discussion and Analysis of Financial Condition and Results of Operation of Alps” and “Cautionary Note Regarding Forward-Looking Statements.” As a result, there can be no assurance that the projected results will be realized or that actual results will not be significantly higher or lower than projected. Since the financial projections cover multiple years, such information by its nature becomes less reliable with each successive year. Alps has affirmed to Globalink that its forecasts reflect the views of the management and board of directors of Alps about its future performance as of the date of this proxy statement/prospectus.
B. Peer Price/Revenue Multiples
| Company Code | Market Cap ($MM) | Revenue | Net Earnings | Market Cap / Revenue | ||||||||||
| ABCL (NSDQ) | 1.66B | 38,025,000.00 | -146,398,000.00 | 43.66 | ||||||||||
| ACAD (NSDQ) | 5.14B | 726,437,000.00 | -61,286,000.00 | 7.08 | ||||||||||
| AGIO (NSDQ) | 1.24B | 26,823,000.00 | -352,088,000.00 | 46.23 | ||||||||||
| ACCD (NSDQ) | 671.92M | 414,292,000.00 | -459,650,000.00 | 1.62 | ||||||||||
| ALNY (NSDQ) | 24.02B | 1,828,292,000.00 | -440,242,000.00 | 13.14 | ||||||||||
| FOLD (NSDQ) | 4.16B | 399,356,000.00 | -151,584,000.00 | 10.42 | ||||||||||
| BHVN (NYSE) | 3.43B | 0.00 | -408,168,000.00 | - | ||||||||||
| AMLX (NSDQ) | 993.86M | 380,786,000.00 | 49,271,000.00 | 2.61 | ||||||||||
| WOCK (NSE) | 732.96M | 322,817,118.00 | -68,070,452.00 | 2.27 | ||||||||||
| STAR.NS (NSE) | 715.92M | 449,141,630.00 | -24,675,235.00 | 1.59 | ||||||||||
| OBIO TECHNOLOGY (SSE) | 814.80 M | 28,882,794.00 | -18,042,608.00 | 28.21 | ||||||||||
| *the Highest & The Lowest was removed to get the average | ||||||||||||||
| Average | 2.09B | 259,713,212.44 | -179,557,340.00 | 15.79 | ||||||||||
| ALPS | 1.60 B | 3,594,828.00 | -729,862.00 | 445.08 | ||||||||||
| Conversion of currency as of December 2023 AVG | ||||||||||||||
| USD | HKD | CNY | INR | MYR | ||||||||||
| 1 | 0.13 | 0.14 | 0.012 | 0.21 | ||||||||||
| Legend | Market Cap | Date | Revenue/ Net Earnings 2023 | |||
| AbCellera Biologics Inc. (ABCL) | https://finance.yahoo.com/quote/ABCL/key-statistics/ | 12/31/2023 | https://www.marketscreener.com/quote/stock/ABCELLERA-BIOLOGICS-INC-116373768/finances-income-statement/ | |||
| ACADIA Pharmaceuticals Inc. (ACAD) | https://finance.yahoo.com/quote/ACAD/key-statistics/ | 12/31/2023 | https://www.marketscreener.com/quote/stock/ACADIA-PHARMACEUTICALS-IN-8222/finances-income-statement/ | |||
| Agios Pharmaceuticals, Inc. (AGIO) | https://finance.yahoo.com/quote/AGIO?.tsrc=fin-srch | 12/31/2023 | https://www.marketscreener.com/quote/stock/AGIOS-PHARMACEUTICALS-INC-13709228/finances-income-statement/ | |||
| Accolade, Inc. (ACCD) | https://finance.yahoo.com/quote/ACCD/key-statistics/ | 11/30/2023 | https://www.marketscreener.com/quote/stock/ACCOLADE-INC-109224285/finances-income-statement/ | |||
| Alnylam Pharmaceuticals, Inc. (ALNY) | https://finance.yahoo.com/quote/ALNY/key-statistics/ | 12/31/2023 | https://www.marketscreener.com/quote/stock/ALNYLAM--I-8322/finances-income-statement/ | |||
| Amicus Therapeutics, Inc. (FOLD) | https://finance.yahoo.com/quote/FOLD/key-statistics/ | 12/31/2023 | https://www.marketscreener.com/quote/stock/AMICUS-THERAPEUTICS-INC-40449562/finances-income-statement/ | |||
| Biohaven Ltd. (BHVN) | Biohaven Ltd. (BHVN) Valuation Measures & Financial Statistics | 12/31/2023 | https://www.marketscreener.com/quote/stock/BIOHAVEN-LTD-144171176/finances-income-statement/ | |||
| Amylyx Pharmaceuticals, Inc. (AMLX) | https://finance.yahoo.com/quote/AMLX/key-statistics/ | 12/31/2023 | https://www.marketscreener.com/quote/stock/AMYLYX-PHARMACEUTICALS-IN-131408793/finances-income-statement// | |||
| Wockhardt Limited (WOCKPHARMA.NS) | https://finance.yahoo.com/quote/WOCKPHARMA.NS/key-statistics/ | 12/31/2023 | https://www.marketscreener.com/quote/stock/WOCKHARDT-LIMITED-9059063/finances-income-statement/ | |||
| Strides Pharma Science Limited (STAR.NS) | https://finance.yahoo.com/quote/STAR.NS/financials | 12/31/2023 | https://www.marketscreener.com/quote/stock/STRIDES-PHARMA-SCIENCE-LI-9743222/finances-income-statement/ | |||
| Obio Technology (Shanghai) Corp., Ltd. (688238.SS) | https://finance.yahoo.com/quote/688238.SS/key-statistics/ | 12/31/2023 | https://www.marketscreener.com/quote/stock/OBIO-TECHNOLOGY-SHANGHAI--142488012/finances-income-statement/ |
1 Galen Centre for Health and Social Policy. (n.d.). Early detection: Key to outcome of pancreatic cancer treatment. Retrieved from https://ova.galencentre.org/early-detection-key-to-outcome-of-pancreatic-cancer-treatment/#:~:text=The
2 New Normal, Same Cancer. (n.d.). Early detection of prostate cancer. Retrieved from https://newnormalsamecancer.org/early-detection/prostate-cancer/
3 National Center for Biotechnology Information. (n.d.). Lung cancer insights. Retrieved from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC10938108/
4 The Star. (2024, January 14). Statistics on cervical cancer in Malaysia alarming. Retrieved from https://www.thestar.com.my/news/nation/2024/01/14/statistics-on-cervical-cancer-in-malaysia-alarming#:~:text=PETALING%20JAYA%3A%20Cervical%20cancer%20is,Global%20Cancer%20Observatory%20(GCO)
5 New Straits Times. (2023, October 10). Breast cancer rise among Asian women. Retrieved from https://www.nst.com.my/lifestyle/heal/2023/10/964762/health-breast-cancer-rise-among-asian-women
6 Academia.edu. (n.d.). Characterization of multiple omics signatures in relation to dietary patterns for in silico personalized colon cancer risk stratification study: Study protocol for a case-control study and the challenges faced during the COVID-19 pandemic. Retrieved from https://www.academia.edu/86340672/Characterization_of_Multiple_Omics_Signatures_in_Relation_to_Dietary_Pattern_for_in_Silico_Personalised_Colon_Cancer_Risk_Stratification_Study_Protocol_for_a_Case_control_Study_and_the_Challenges_Faced_During_the_COVID_19_Pandemic
7 Novotech CRO. (2021). Non-Hodgkin lymphoma landscape in Asia-Pacific. Retrieved from https://novotech-cro.com/sites/default/files/2021-02/NHL%20Landscape%20in%20Asia-Pacific_2021.pdf.
| 123 |
Despite Alps’ market cap/revenue ratio appears to be significantly higher than the peer companies listed above, the board of directors and the management of Alps recognize the fact that the Alps’ Market Cap/Revenue ratio is premised on the size of current business in Malaysia only and did not factor in any growth and expansion plans of the potential Southeast Asian markets that are approximately 20 times larger in terms of market size compared to that of Malaysia.
Alps selected the peer companies based on the following criteria:
| 1. | Industry Classification. Peer companies should operate within the same industry or sector. This helps ensure that the comparison is relevant, as companies in the same industry typically face similar market conditions, regulatory environments, and competitive dynamics. | |
| 2. | Geographic Focus. Peer companies should operate in the same geographic region and/or potential capital markets may be more relevant for comparison. | |
| 3. | Growth Stage. Peer companies should be at similar stages in their business life cycle (startup, growth, maturity, or decline) can lead to more accurate comparisons. | |
| 4. | Technology and Innovation. Where technology or pipelines plays a crucial role, peer companies should be ones that leverage similar technologies or have comparable levels of investment in research and development can yield more relevant comparisons. | |
| 5. | Customer Segmentation. If the peer companies serve similar customer bases or market segments, this can enhance the validity of the comparison. |
The board of directors and management of Alps used the above to serve as a guide and reference in order to have a better insight on the business and industry potentials as below.
| 1. | Valuation Benchmarking: The Market Cap/Revenue ratio can help assess whether a company is overvalued or undervalued relative to its peers. A significantly higher Market Cap/Revenue ratio may indicate that the market expects stronger growth or has greater confidence in that company’s future, while a lower ratio may suggest the opposite. | |
| 2. | Growth Potential Assessment: Companies with higher Market Cap/Revenue ratios may have stronger growth expectations from investors, indicating that they are anticipated to capture more market share or innovate effectively. This is particularly relevant in industries where future growth is pivotal, like technology and biotechnology. | |
| 3. | Comparative Analysis Across Different Firm Sizes: The Market Cap/Revenue ratio allows for comparisons between both large and small firms within the same industry, providing insights into how smaller or new companies are valued compared to established players, which can indicate market trends or investor preferences. | |
| 4. | Non-Profitability Indicator: In situations where traditional profitability metrics (like P/E ratios) may not apply—as seen in nascent or growth-oriented companies—the Market Cap/Revenue ratio becomes an alternative way to gauge market expectations for future earnings based on current revenues. | |
| 5. | Guiding Business Decisions: Understanding relative Market Cap/Revenue ratios can help in constructing a portfolio that balances risk and potential returns, serving either as a screening tool as part of growth strategies. |
While the Market Cap/Revenue ratio alone should not and does not dictate investment decisions, it offers valuable insights into company valuations, market perceptions, and industry dynamics. Employing it in conjunction with other financial metrics and qualitative assessments can enhance the overall analysis and aid in making more informed business decisions. The management and board of directors of Globalink relied moderately on it in considering the peer price/revenue multiples, noting the following potential limitations:
| 1. | Different Business Models: Companies in the same industry may have vastly different business models, leading to differences in revenue generation and expenditure. | |
| 2. | Growth Rates: Companies may be at different stages of growth. High-growth companies may have lower immediate profits but higher sales, while mature companies might generate less revenue growth. | |
| 3. | Capital Structure and Debt Levels: This means that two companies may have similar Market Cap/Revenue ratios while having very different risk profiles. | |
| 4. | Market Sentiment and Speculation: Market capitalization can be heavily influenced by market sentiment, speculation, and trends, which may not reflect the underlying financial health of the company. | |
| 5. | Timing Issues: Revenue can fluctuate due to seasonality or one-time events (e.g., a big contract or product launch), so momentary spikes or dips in revenue can distort the Market Cap/Revenue ratio at any given time. | |
| 6. | Different Accounting Policies: Companies may employ different accounting practices, which can influence reported revenues. | |
| 7. | Market Capitalization Volatility: Stock prices can be volatile, leading to significant fluctuations in market capitalization, while revenues tend to be more stable. This volatility can cause the Market Cap/Revenue ratio to vary widely over short periods. |
| 124 |
During the meeting, the Globalink Board also reviewed and compared certain information related to the following selected publicly traded biotechnology companies with operations similar to Alps for its analysis based on its experience and professional judgment (which companies are referred to as the “selected public companies”). Although none of the selected public companies is directly comparable to Alps, the companies listed below were chosen by Globalink because, among other reasons, they are publicly traded early-stage biotechnology companies with certain operational, business or financial characteristics that, for purposes of Globalink’s analysis, may be considered similar to those of Alps.
Although none of the selected public companies are directly comparable to Alps, they were selected as they share characteristics, such as:
| 1. | Industry Classification: Companies operating in the same industry or sector, which face similar market conditions, regulatory environments, and competitive dynamics. |
| 2. | Geographic Focus: Companies that operate in the same geographic region and/or similar potential capital markets. |
| 3. | Growth Stage: Companies at comparable stages in their business life cycle (e.g., startup, growth, maturity, or decline) were selected for more accurate comparisons. |
| 4. | Technology and Innovation: Companies leveraging similar technologies or pipelines or have comparable levels of investment in research and development to yield more relevant comparisons. |
| 5. | Customer Segmentation: Companies serve similar customer bases or market segments, this could enhance the validity of the comparison. |
Considering the relatively limited number of comparable business in the industry operating in the same geographical location and potential capital markets, Globalink selected as comparable companies some business with smaller market capitalization, such as WOCK, and STAR.NS which operate in India.
However, because none of the selected public companies is identical or directly comparable to Alps, Globalink believed that it was inappropriate to, and therefore did not, rely solely on the quantitative results of the selected public company analysis. Due to the early-stage status and the large number of product candidates in Alps’ pipelines as compared to the selected public companies, Globalink is of the view that the selected public company analysis is not a suitable approach to assess the valuation of Alps. As a result, Globalink did not conduct comprehensive valuation on Alps using the selected public companies analysis. The selected public company analysis was performed by Globalink only to review certain key data and inputs necessary to estimate Alps’ performance outlook relative to the industry. Accordingly, Globalink also made qualitative judgments, based on its experience and professional judgment, concerning differences between the operational, business and/or financial characteristics of Alps and the selected public companies that could affect their public trading values in order to provide a context in which to consider the results of the quantitative analysis.
Using publicly available information obtained from SEC filings and other publicly available data sources from January 5, 2024 to January 10, 2024, Globalink reviewed, for each selected public company, such selected public company’s market capitalization, which is referred to, with respect to the selected public companies, as “Market Value.” The rationale for including each of the selected public companies are listed in the following table:
| Company | Listing | Rationale | ||
| AbCellera Biologics Inc | U.S. | AbCellera operates a biotechnology platform similar to Alps, emphasizing early-stage biotech innovations.
| ||
| Acadia Pharmaceuticals Inc | U.S. | Acadia incurs ongoing losses, providing a relevant comparison to Alps’ financial position.
| ||
| Agios Pharmaceuticals Inc | U.S. | Agios is in a high-growth biotech company with early-stage revenue and significant losses, closely mirroring Alps’ financial structure and growth outlook.
| ||
| Accolade Inc | U.S. | Accolade is in a more mature revenue streams, giving Alps a more balanced outlook on revenue growth within biotech.
| ||
| Alnylam Pharmaceuticals Inc. | U.S. |
|
Alnylam is closely aligned with Alps’ focus on developing mRNA as its modality, demonstrating the market potential in the mRNA-based therapies.
| |
| Amicus Therapeutics Inc | U.S. | While Amicus primarily operates in developed markets, its biotechnology focus and reliance on significant R&D investment make it relevant for comparison with Alps.
| ||
| Biohaven Ltd | U.S. | Biohaven has yet to generate any revenue but its market cap is supported by future revenue potential rather than current earnings.
| ||
| Wockhardt Ltd | India | Wockhardt is based in India and serves multiple emerging markets. This geographic focus is similar to Alps’ operational environment in Malaysia and potential expansion within Southeast Asia
| ||
| Strides Pharma Science Ltd | India | Strides serves a diverse customer base, including hospitals, clinics, and direct consumers, which parallels Alps’ customer segmentation. Alps, with a focus on diagnostics and wellness, shares a customer-driven approach with Strides, catering to healthcare providers and end-users. | ||
| Obio Technology (Shanghai) Corp Ltd | Shanghai | Obio offer diagnostic products and services to Asian market, directly comparable to Alps’ offerings, especially in genetic testing and diagnostics. |
There was no publicly available transaction that was directly comparable in terms of the uniqueness of Alps’ business model, its user base and core assets as well as its stage of development; thus, Globalink is of the view that the selected public company analysis is not a suitable approach to assess the valuation of Alps. In applying the market approach, different value measures or market multiples of the comparable companies are calculated and analyzed to induce a series of multiples that are considered representative of the industry average. Three commonly used price multiples are (i) Price-to-earnings (“P/E”) ratio; (ii) Market Cap-to-Revenue ratio; and (iii) Price-to-book value (“P/B”) ratio. The pre-requisite for the P/E ratio is being profit-making. However, Alps’ business is in its investment period with high growth potential but it is not yet profitable. Likewise, as a biotech platform, Alps’ fair value does not correlate closely with its book value, making the P/B ratio less relevant. Consequently, the Market Cap/Revenue ratio was selected as the most suitable metric for this analysis. Globalink has considered the following factors to use and rely on Market Cap/Revenue ratio:
| 1. | Valuation Benchmarking: the Market Cap/Revenue ratio can help assess whether a company is overvalued or undervalued relative to its peers. A significantly higher Market Cap/Revenue ratio may indicate that the market expects stronger growth or has greater confidence in that company’s future, while a lower ratio may suggest the opposite. |
| 125 |
| 2. | Growth Potential Assessment: companies with higher Market Cap/Revenue ratios may have stronger growth expectations from investors, indicating that they are anticipated to capture more market share or innovate effectively. This is particularly relevant in industries where future growth is pivotal, like technology and biotechnology. |
| 3. | Comparative Analysis Across Different Firm Sizes: the Market Cap/Revenue ratio allows for comparisons between both large and small firms within the same industry, providing insights into how smaller or new companies are valued compared to established players, which can indicate market trends or investor preferences. |
| 4. | Non-Profitability Indicator: in situations where traditional profitability metrics (like P/E ratios) may not apply—as seen in nascent or growth-oriented companies—the Market Cap/Revenue ratio becomes an alternative way to gauge market expectations for future earnings based on current revenues. |
| 5. | Guiding Business Decisions: understanding relative Market Cap/Revenue ratios can help in constructing a portfolio that balances risk and potential returns, serving either as a screening tool as part of growth strategies. |
Globalink noted that the Market Cap/Revenue ratio alone should not and does not dictate investment decisions, it offers valuable insights into company valuations, market perceptions, and industry dynamics. Employing it in conjunction with other financial metrics and qualitative assessments can enhance the overall analysis and aid in making more informed business decisions. The Globalink Board relied moderately on it noting the following potential limitations:
| 1. | Different Business Models: companies in the same industry may have vastly different business models, leading to differences in revenue generation and expenditure. |
| 2. | Growth Rates: companies may be at different stages of growth. High-growth companies may have lower immediate profits but higher sales, while mature companies might generate less revenue growth. |
| 3. | Capital Structure and Debt Levels: this means that two companies may have similar Market Cap/Revenue ratios while having very different risk profiles. |
| 4. | Market Sentiment and Speculation: market capitalization can be heavily influenced by market sentiment, speculation, and trends, which may not reflect the underlying financial health of the company. |
| 5. | Timing Issues: revenue can fluctuate due to seasonality or one-time events (e.g., a big contract or product launch), so momentary spikes or dips in revenue can distort the Market Cap/Revenue ratio at any given time. |
| 6. | Different Accounting Policies: companies may employ different accounting practices, which can influence reported revenues. |
| 7. | Market Capitalization Volatility: stock prices can be volatile, leading to significant fluctuations in market capitalization, while revenues tend to be more stable. This volatility can cause the Market Cap/Revenue ratio to vary widely over short periods. |
Globalink analyzed the selected public companies based on their similarity to Alps, primarily in terms of certain quantitative and qualitative factors, including, but not limited to the total number of pipelines in development, the development stage of such pipelines, the estimated commercialization year and the estimated patient population size, based on consensus equity research analyst estimates, public filings and Globalink’s experience and professional judgment. Taking into account such quantitative and qualitative factors, Globalink observed the illustrative market value range of the selected public companies of $235 million to $5,034 million, the average market value of the selected companies of $1,723 million and the market value of Alps of $1,600 million.
The Globalink Board also noted that Alps and the Seller Representative verbally committed to securing PIPE Investment of an aggregate amount of US$40.2 million or more, which the Globalink Board considered to be the primary basis for the pre-money valuation of Alps of approximately $1.6 billion.
The Globalink’s board also considered various uncertainties, risks, and other potentially negative factors concerning the business combination, including but not limited to, the following:
| ● | Insufficient Funding Risks. The risk that Alps requires substantial additional funding to advance the development of its biotech product candidates, the costs associated with being a publicly listed company, combined with the capital-intensive nature of clinical development and expanding manufacturing capabilities, require ongoing investments. Failing to secure the PIPE Investment at Closing will hinder Alps’ long-term strategic plans and objectives. | |
| ● | Clinical-Stage Company Development Risks. The risk that Alps is a proof-of-concept and preclinical-stage company that has yet to commercialize its product candidates. Product development may be delayed due to difficulties in patient enrolment. Running multiple clinical trials is expensive and complex, especially for cell-based products. | |
| ● | Other Business and Regulatory Risks. The risk that the future financial performance of Alps may not meet the Globalink Board’s expectations due to factors both in and outside of Alps’ control, including change of policies by the Malaysian regulatory authorities regarding compassionate use of stem cell therapies. |
The Board was also informed that there were no discussions between Globalink’s management with Alps about continuing employment or involvement for any persons affiliated with Globalink before the Business Combination. There is no pre-existing relationship between Globalink, the Sponsor, and target and its affiliates, on one hand, and selected PIPE investors, on the other hand, and that there would not be a placement agent involved in the PIPE Investment. The above enabled the Board to make an informed business decision as recommended by Globalink’s management to support the business combination as it was concluded that the advantages of the proposed business outweigh potentially negative effects and it is in line with Globalink’s objective. The Board also agreed to engage an independent financial valuator to issue a fair value report.
| 126 |
On January 28, 2024, GLLI and the Alps team including Dr. Tham finalized and agreed on remaining terms and conditions of the Merger Agreement and ancillary agreements. Towards the end, additional deliberations were focused on key risks factors that could negatively impact Alps’ operations and performance, including Alps’ services and product quality, sales targets, pricing strategy, costs and expenses overruns, expected return of PIPE Investment, clinical trials results, marketing strategies, industry competition, and governmental regulations. During the meeting, both parties considered and discussed the composition of board of the Combined Company again. Alps proposed that Alps shall appoint all five directors of the Combined Company, and Globalink accepted the proposal.
On January 30, 2024, the parties entered into the Merger Agreement.
After entering into the Merger Agreement, the Alps team began reaching out to potential investors for PIPE Investment on a larger scale to discuss the investment opportunities, and presented potential investors with its Teaser Deck to facilitate discussions. For potential investors who indicated interest, the Alps team would schedule video conferences or face-to-face meetings with the potential investors and bring them over the wall for more in-depth discussions. Following video conferences or face-to-face meetings, the Alps team held pitching sessions and entered into non-disclosure agreement with potential investors before providing them with an investor deck. Those potential investors who remain interested asked questions to the Alps team and negotiated the terms of the PIPE Investment. During this process, the Alps team were in constant communications with a PIPE Investor (“Investor One”), who the Alps team initially approached in November 2023 and later entered into a Subscription Agreement.
In April 2024, the Globalink team, Alps team and their counsels agreed to amend the Merger Agreement, to change the merger structure of the Business Combination to include two mergers, namely the Redomestication Merger and the Acquisition Merger, for the purposes of setting up a foreign incorporated company as the PubCo, and allowing the Combined Company to qualify for the foreign private issuer status after the Closing.
On May 6, 2024, representative of Alps, Dr. Tham Seng Kong, met the other PIPE Investor (“Investor Two”) at the Royal Selangor Golf Club in Kuala Lumpur, Malaysia, who was introduced by a common friend, and the representative of Alps introduced Alps and its business to the PIPE Investors, and shared with the PIPE Investors copies of public news of Alps and Globalink and the teaser.
On May 17, 2024, the Alps team received the identification information from Investor One.
On May 20, 2024, the parties amended and restated the Merger Agreement.
On June 4, 2024, the Alps team had a phone call with Investor Two to finalize the subscription details and the investment amount. Subsequently, Investor Two sent over his personal profile to us and passport for identification purposes. On the same day, Alps Holdco, Globalink, PubCo and Investor Two entered into a Subscription Agreement.
On June 5, 2024, Alps Holdco, Globalink, PubCo and Investor One entered into a Subscription Agreement.
On July 17, 2024, a PIPE Investor (“Investor Three”) conducted a site visit at Alps, during which the Alps team presented and shared its Teaser Deck, the Business Combination, and shared recent public news about Alps to the Investor Three. The Investor Three has subsequently entered into a Subscription Agreement on August 27, 2024.
Finder’s Fee to IBDC Asia Sdn. Bhd.
IBDC Asia Sdn. Bhd. (“IBDC”) is a private company incorporated in Malaysia in 2005 and provides business advisory services and business searching and matching services, including identifying special purpose acquisition companies for target companies seeking local or overseas listings.
Mr. Ler Leong Keh, a director of IBDC Asia, and Mr. Benny Tan, an Associate Director of IBDC, are acquainted with both of Mr. Say Leong Lim, Globalink’s Chief Executive Officer, and Dr. Tham Seng Kong, the Seller Representative, through previous business projects and connections in Malaysia. IBDC approached Mr. Say Leong Lim on behalf of Alps for a proposed business combination in August 2023, before IBDC was officially engaged by Alps. On August 18, 2023, IBDC arranged a meeting with Globalink’s Chairman of the Board of Directors and Chief Executive Officer, Mr. Say Leong Lim, regarding a potential De-SPAC transaction between Globalink and Alps. See “—Background of the Business Combination.”
After receiving indication from Globalink that Globalink was willing to explore a business combination with Alps, Alps officially engaged IBDC as its financial advisor in October 2023. The market rate in Malaysia for similar advisory and business searching and matching services is approximately 3% of the deal size. However, due to the potentially high valuation of Alps, the success fees payable to IBDC was negotiated and agreed at 0.5%, plus another 0.5% if the business combination agreement includes an earnout provision to reward future performance by Alps.
In October 2023, Globalink and Alps entered into a non-binding and non-exclusive letter of intent and started engaging in more thorough and official discussions. In January 2024, Globalink and Alps entered into the original Business Combination Agreement, which provided for an earn-out provision. As such, in accordance with the terms of engagement between Alps and IBDC, IBDC would be entitled to a fee of 1% of the Merger Consideration at the consummation of the Business Combination.
Board’s Discussion of Valuation and Reasons for the Approval of the Business Combination
In reaching its decision with respect to the Business Combination, the Board considered views of Globalink’s management regarding the opportunity represented by the proposed transaction and evaluated the investor presentation provided by Alps. Globalink’s management discussed its diligence to the Board, which included:
| ● | review of Alps’ financial results; | |
| ● | review of Alps’ financial projections model; | |
| ● | review of Alps’ descriptions of its business operations; | |
| ● | review of financial data of public companies that are comparable to Alps; | |
| ● | review of Alps’ capitalization table; | |
| ● | review of Alps’ corporate records, material agreements, employee agreements, tax, litigations and other business-relevant materials; | |
| ● | discussions with Alps’ management; and | |
| ● | on-site inspection of Alps’ operational facilities. |
The Board reviewed and analyzed the financial statements of Alps for the last three fiscal years, and the financial projections prepared by Alps for the upcoming five years following Closing. The financial projections were prepared for the information and review by the Board. The Board reviewed the historic results and projected results, and the underlying assumptions of the projections, which were based on historical performance of Alps and the performance of peer or competitor companies. However, the Board believes that the financial projections may have contained estimates and assumptions that susceptible to changes and uncertainties, and did not put much weight in considering the financial projections of Alps in determining the valuation of Alps nor in the decision of recommending the Business Combination for stockholders’ approval.
The Board supported the decision to enter into the Merger Agreement based on the Board’s evaluation of the above other due diligence by Globalink’s management and the investor presentation, and on the following qualitative and quantitative evaluations regarding Alps:
Business model
The Board reviewed the business model of Alps and believes that Alps adopts a business model that generates revenue through multiple revenue streams with relatively low overhead costs. The Board placed a moderate significant weighting on this factor.
Product Pipeline
Alps has the right to use certain patents and has developed several product pipelines with commercial potential and opportunities for future success in regional and international markets. The Board placed a moderate significant weighting on this factor.
Industry trends
| ● | Advances in biotechnology: Rapid advancements in biotechnology, such as gene editing technologies like clustered regularly interspaced palindromic repeats (CRISPR-Cas9), have unlocked new possibilities in drug discovery, disease treatment, and personalized medicine. These biotechnological breakthroughs offer tremendous potential for innovation and growth within the industry. | |
| ● | Growing demand for personalized medicine: Personalized medicine, which tailors treatment plans based on an individual’s genetic makeup, is gaining popularity. This trend provides opportunities for biotech companies to develop targeted therapies, companion diagnostics, and genetic testing services to meet the demand for more precise and effective healthcare solutions. | |
| ● | Focus on rare diseases and orphan drugs: The biotech industry has shifted focus towards rare diseases, also known as orphan diseases, which affect a small percentage of the population. Governments and regulatory bodies have implemented incentives and streamlined approval processes to encourage the development of orphan drugs, making it an attractive area for biotech companies to invest in. | |
| ● | Increased investment in biotech startups: Venture capital firms, pharmaceutical companies, and government entities are increasingly investing in biotech startups. This influx of funding has created a supportive environment for innovative ideas and technologies to be developed, leading to a vibrant ecosystem of entrepreneurial ventures within the industry. |
| 127 |
| ● | Convergence of biology and digital technology: The convergence of biology with digital technology, such as artificial intelligence (AI), big data analytics, and machine learning, is transforming the biotech and life sciences landscape. These digital technologies enable faster and more accurate drug discovery, clinical trials, and patient care, improving overall efficiency and outcomes. | |
| ● | Rising global healthcare expenditure: With the global population aging and the increasing prevalence of chronic diseases, healthcare expenditure is on the rise. Biotech and life sciences companies are well-positioned to benefit from this trend by developing innovative therapies and solutions that address the evolving healthcare needs of the population. |
These positive trends create an optimistic outlook for the biotech and life sciences industry, presenting numerous opportunities for growth, innovation, and impact. The Board placed a high significance weighing on this factor.
For sources of the above statements and conclusions on the future trends in the biotechnology industry, please refer to the notes below.
Notes:
| 1. | Explore Emerging Biotech Trends in 2024 for Life Science Professionals. https://www.mrlcg.com/resources/blog/emerging-biotech-trends/ | |
| 2. | The Future of Biotech: Innovations and Trends in Science and Research. https://www.healthtechzone.com/topics/healthcare/articles/2024/06/21/459929-future-biotech-innovations-trends-science-research.htm. | |
| 3. | The Global Biotechnology Industry Outlook – 2024. https://www.marketsandmarkets.com/blog/HC/The-Global-Biotechnology-Industry-Outlook-2024 | |
| 4. | The Future of Biotech. https://www.dni.gov/index.php/gt2040-home/gt2040-deeper-looks/future-of-biotech | |
| 5. | Biotechnology Market Size, Share, and Trends 2024 to 2033. https://www.precedenceresearch.com/biotechnology-market | |
| 6. | Biotechnology Market Size & Trends. https://www.grandviewresearch.com/industry-analysis/biotechnology-market | |
| 7. | Biotechnology Industry Statistics: Market Data Report 2024. https://worldmetrics.org/biotechnology-industry-statistics/ |
Management team
The management team of Alps is composed of seasoned professionals with extensive experience and a track record of achievements in the field.
At the helm is Dr. Tham Seng Kong, a visionary and accomplished leader with a Bachelor of Medicine from Xiamen University, Doctor of Medicine (MD) Integrated Chinese & Western Clinical Medicine (Oncology) from Guangzhou University of Chinese Medicine (GUCM) and a Ph.D. in Clinical Discipline of Chinese and Western Medicine GUCM He also obtained his Masters of Business Administration from University of Pennsylvania, United States. With his deep scientific expertise and keen business acumen, he has successfully spearheaded multiple R&D projects and breakthrough therapies to market. Driven by an unwavering passion for improving patient lives, Dr. Tham possesses the ability to inspire and motivate the entire team at Alps.
Supporting Dr. Tham is Lisa Teoh @ Teoh Lee Eng, a seasoned executive with more than 20 years of experience in finance and operations. As the chief operating officer of Alps, she brings a strong operational acumen to Alps. Ms. Teoh is a registered civil engineer with professional institutions in Malaysia and is an ASEAN Chartered Professional Engineer. She earned her Master’s Degree in Engineering Science (Civil) (M.Eng) in 1996 as well as a Master of Philosophy from the University of Oxford, United Kingdom in 1997. Ms. Teoh later obtained her Master of Business Entrepreneur Management in 2018 and Doctor of Business Administration in 2020 from the Institute of Entrepreneurship Malaysia.
Leading the research and development efforts is Professor Manickam Ravichandran, a renowned expert in molecular biology and vaccine discovery. He obtained his M.Sc.-Medical Microbiology from Christian Medical College, Vellore and was awarded his Ph.D. in Biotechnology from Anna University in Chennai, India. He has a track record of developing innovative therapeutic candidates and leading clinical trials. He was previously appointed as the former Vice-Chancellor of AIMST University, Malaysia. Professor Manickam Ravichandran ‘s strategic thinking and scientific expertise are instrumental in guiding Alps’ research and development pipeline.
Leading another pipeline in vaccine development is Alps’ Chief Vaccine Development Officer, Professor Chit Laa Poh. Professor Poh obtained her Ph.D. in Medical Microbiology and Bacteriology from Monash University (Australia) and completed postdoctoral training at Pasteur Institute, Cambridge University, and University College London. Professor Poh served as the Inaugural Dean of the Faculty of Science and Technology from 2012 to 2014 and currently holds the position of Distinguished Professor and Head of the Centre for Virus and Vaccine Research (CVVR) at Sunway University, Malaysia.
The commercialization strategy and marketing efforts are overseen by Ms. Chew Yoke Ling, a seasoned marketing professional with a deep understanding of the biotech market landscape. With her market research expertise and customer-centric approach, she ensures the effective positioning and successful commercial launch of Alps’ products.
The Board placed a moderate significance weighing on this factor.
Reason for Alps to engage in the De-SPAC Transaction
Alps has expressed its interest in exploring a De-SPAC transaction as an alternative to a traditional IPO for several strategic reasons, particularly considering the presence of PIPE investors.
The management team of Alps has conducted an evaluation of the proposed business combination and the advantages and disadvantages of a De-SPAC transaction. They have determined that a De-SPAC provides greater flexibility and more favorable terms than a traditional IPO, particularly regarding speed to market, valuation, and investment conditions.
As compared to a traditional IPO, a De-SPAC transaction typically allows for a quicker transition to being a publicly traded entity. Alps believes that this accelerated timeline will enable Alps to fast-track its development plans, including advancing multiple product candidates and preparing for commercialization, contingent upon approval. Furthermore, in a De-SPAC, the transaction terms and valuation are negotiated directly with the SPAC, offering enhanced pricing certainty for Alps.
Alps emphasized that listing on Bursa Exchange or the Singapore Exchange would keep Alps to a more regional presence, while Nasdaq provides a global platform. Alps chose Nasdaq to align with its ambition to become a global company and leverage the international visibility and opportunities that come with it.
Overall, Alps considers the De-SPAC transaction to be a strategically advantageous and efficient method for going public, ensuring the necessary capital to support its development goals while achieving a faster market entry.
Competition
Alps provides commercialized services and products in Malaysia. Alps has a group of skilled, multilingual employee able to contribute to the company’s success. Out of the total employees of Alps, 66% or more has a bachelor’s degree or above and has an average working experience of 19 years each, and is comprised of individuals with diverse backgrounds and ethnicities, including Malay, Chinese, Indian and other ethnic backgrounds. This allows Alps to develop and communicate with customers from different backgrounds. Alps’ labor force also contributes to a low burn rate relative to many typical biotech companies. In addition, Alps operates in Malaysia, a market that provides a competitive advantage in terms of labor costs. For instance, the annual salary for a research scientist at Alps ranges from approximately USD 8,156 to USD 14,154, depending on experience and specialization. This is considerably lower than the salary range for similar positions in the United States, where salaries range from approximately USD 57,000 to USD 129,000 annually, depending on experience and specialization. Alps’ skilled, multilingual and cost-effective labor force gains the company a competitive advantage in its pipeline developments.
For the fiscal years ended March 31, 2024 and 2023, the burn rates of Alps were approximately US$1,490,000 and US$224,000, respectively. Burn rates are largely driven by research and development expenses. For instance, Moderna spent US$4.85 billion on research and development in 2023, Biogen incurred US$2.46 billion in research and development expenses in 2023, and Novartis incurred US$11.37 billion in research and development expenses. Burn rates for biotechnology companies can vary widely from tens of millions to over $100 million per quarter, depending on their stage of development and level of commercial operations. For clarity, the companies referenced above are significantly larger than Alps, with significantly larger research and development plans and costs. The Board placed a high significance weighing on this factor.
For sources of the above statements relating to Alps’ low burn rate relative to typical biotech companies, please refer to notes below:
Notes:
| 1. | The top pharmaceutical companies by R&D expenditure. https://www.pharmaceutical-technology.com/features/the-top-pharmaceutical-companies-by-rd-expenditure/ |
| 128 |
Significant growth potential
Southeast Asia is a dynamic region that offers numerous advantages for businesses seeking growth and expansion. With a rapidly growing economy, a large consumer base, and a strategic location, Southeast Asia has become an attractive destination for both established corporations and emerging startups.
The region has experienced robust economic growth in recent years, fueled by factors such as urbanization, rising middle-class population, and increased foreign investment. This growth has created a thriving market with vast potential for businesses to tap into. Southeast Asia boasts a large and diverse consumer base. With a population of over 650 million people, the region offers businesses access to a vast market of potential customers. The middle class is expanding rapidly, leading to increased purchasing power and demand for products and services across various sectors. Additionally, with an increasingly aging population, Alps expects to observe increasing demand for its products and services.
Southeast Asia is also strategically located, serving as a gateway to the larger Asian market. Its proximity to China, India, and other emerging economies provides businesses with opportunities for regional integration and access to global supply chains.
Another advantage of Southeast Asia is its investment-friendly policies and business environment. Many countries in the region have implemented reforms to attract foreign investment, such as tax incentives, streamlined regulations, and improved infrastructure. This business-friendly ecosystem facilitates entry and expansion for companies seeking to establish a presence or invest in Southeast Asia. The region offers a large pool of young, educated, and motivated workers who are capable of driving innovation and productivity. With competitive labor costs compared to other regions, businesses can achieve greater cost efficiency and maintain competitiveness.
Additionally, Southeast Asia is a hub for innovation and entrepreneurship. The region has witnessed a burgeoning startup ecosystem, supported by incubators, accelerators, and venture capital investment. This vibrant startup culture fosters innovation, encourages collaboration, and provides opportunities for partnerships and synergies with local businesses.
The Board placed a high significance weighing on this factor.
For sources of the above statements and conclusions on the growth potential of Southeast Asia, please refer to the notes below.
Notes:
| 1. | The rise of the middle class and economic growth in ASEAN. https://documents.worldbank.org/en/publication/documents-reports/documentdetail/426681495130074444/the-rise-of-the-middle-class-and-economic-growth-in-asean | |
| 2. | A 3D view of Southeast Asia: demographics, digitisation and dynamism. https://www.business.hsbc.com/en-gb/insights/growing-my-business/a-3d-view-of-southeast-asia#:~:text=Southeast%20Asia%20is%20also%20home,5%25%20annually%20through%20to%202030.&text=This%20increase%20in%20purchasing%20power,foreign%20direct%20investment%20(FDI) | |
| 3. | Total population of the ASEAN countries from 2019 to 2029. https://www.statista.com/statistics/796222/total-population-of-the-asean-countries/ | |
| 4. | International Monetary Fund – Population. https://www.imf.org/external/datamapper/LP@WEO/VNM/IDN/PHL/MMR/MYS/KHM/LAO/THA/SGP/BRN/AUS | |
| 5. | Aging Southeast Asia grapples with weak social safety nets. https://asia.nikkei.com/Spotlight/Datawatch/Aging-Southeast-Asia-grapples-with-weak-social-safety-nets |
Management team’s reasons for engaging in De-SPAC Transaction
The decision to engage in a De-SPAC transaction as an alternative to IPO was influenced by several strategic reasons, particularly the presence of PIPE investors. The Alps management team conducted an evaluation of the proposed business combination, determining that a De-SPAC transaction provides more flexibility and favorable terms compared to a traditional IPO in terms of valuation and investment conditions. A key advantage was the shorter timeline of a De-SPAC transaction. Alps believed that this expedited process could allow Alps to finalize a definitive business combination agreement more quickly than traditional IPO. Alps believes that the ability to secure predictable capital combined with the efficiency of the De-SPAC process, would enable Alps to accelerate its development plans, including advancing multiple product candidates and preparing for commercialization if any of these candidates are approved.
Availability and terms of PIPE Investment
The Board also considered the availability and terms of the PIPE Investment prior to approving the Business Combination. Noting that PIPE Investment in the amount of approximately US$40.2 million from local investors from Malaysia would become available in connection with the Business Combination, the Board negotiated the terms of the PIPE Investment with Alps, and mutually agreed that the PIPE Investment would be used for PubCo’s working capital after the Business Combination. The Board determined that the terms of the PIPE Investment are fair and reasonable to Globalink. The determination was made unanimously in May 2024.
Prior to making such determination, the Globalink Board engaged in discussions and consultations with its management, financial and legal advisors of both Globalink and Alps Holdco, and reviewed the terms of the PIPE Investment. The Globalink Board considered matters such as (i) whether the terms of the PIPE subscription agreements are similar to the typical PIPE transactions entered into by other special purpose acquisition companies in connection with a De-SPAC transaction, and whether there are any usual terms not commonly observed, (ii) the potential investors’ profile, including each investor’s net worth and reputation, (iii) purpose of such investors’ investment and utilization, including whether they intend to hold PubCo’s shares for long-term investment, and (iv) the amount of the financing and the timing such financing proceeds are expected to be available. The Board also considered the US$1.6 billion valuation for the PIPE Investment as part of its determination that the PIPE Investment are fair and reasonable to Globalink, and the Board was of the view that the US$1.6 billion valuation was fair and reasonable, because such valuation was also accepted by the PIPE Investors who are independent third parties to Globalink and Alps, and compared this valuation with the market value range of selected public biotech companies of US$235 million to $5,039 million, with an average market value of US$1.723 billion, which were discussed in greater detail under “—Background of the Business Combination.” Based on all the factors discussed, the Board determined that the terms of the PIPE Investment are fair and reasonable to Globalink.
The Board placed a high significance weighing on this factor.
Valuation and Globalink Board’s Decision to Recommend the Business Combination
The Globalink Board considered the valuation and Merger Consideration in favor of recommending the approval of the Redomestication Merger Proposal and the Acquisition Merger Proposal to its stockholders. In reaching the valuation of Alps Holdco, the Globalink Board considered (i) the verbal commitment of Seller Representative and Alps Holdco during the negotiation stage of securing PIPE Investment of an aggregate amount of US$40.2 million or more, (ii) the financing completed by Alps in September 2023, of US$2.5 million, and (iii) the other factors disclosed under “—Board’s Discussion of Valuation and Reasons for the Approval of the Business Combination.” The Globalink Board believed that the valuation of Alps Holdco and the Merger Consideration to be paid to Alps Holdco Shareholders are fair and reasonable to holders of Globalink common stock.
| 129 |
Fairness Opinion
The Globalink Board considered obtaining a final fairness opinion as to the Merger Consideration paid to Alps Holdco Shareholders, and has considered Baker Tilly, a financial advisory, tax and assurance services firm, and Morison Advisory Sdn. Bhd., a financial advisory services firm based in Malaysia and a member firm of Morison Global, for a fairness opinion. Neither firm declined or were otherwise unable or unwilling to provide a fairness opinion to the Globalink Board. On February 2, 2024, after considering the qualifications, past project experience, and indicative fees of both firms, Globalink engaged Morison Advisory Sdn. Bhd. to provide a fairness opinion. Morison Advisory Sdn. Bhd. is a recognized professional services firm in Malaysia and specializes in financial advisory services in Malaysia. During the past two years, there has been no relationship between Morison Advisory Sdn. Bhd., its affiliates, and/or unaffiliated representatives, on one hand, and Globalink, the Sponsor, and/or their respective affiliates, on the other hand, and no such material relationship is being contemplated.
The Globalink Board instructed Morison Advisory Sdn. Bhd. to prepare an opinion assessing the fairness of the Merger Consideration to be paid by Globalink to the Alps Holdco Shareholders in connection with the Business Combination, and instructed Morison Advisory Sdn. Bhd. to produce the opinion on an expedited basis. No other instructions were provided by Globalink or the Sponsor to Morison Advisory Sdn. Bhd. in connection with the draft fairness opinion, nor did Globalink or the Sponsor impose any limitation on the scope of the investigation. The Globalink Board received a draft of fairness opinion from Morison Advisory Sdn. Bhd. in June 2024. After a thorough review and internal discussions, the Globalink Board decided not to rely on the fairness opinion for determining the valuation of Alps Holdco, as the Globalink Board believed that the valuation methods adopted by Morison Advisory Sdn. Bhd., the income approach using Discounted Cash Flow (“DCF”) method, was reliant upon underlying key assumptions susceptible to material changes and uncertainties. The Globalink Board did not put much weight in considering the draft fairness opinion in determining the valuation of Alps Holdco. Instead, the Globalink Board primarily considered (i) the verbal commitment of Seller Representative and Alps Holdco during the negotiation stage of securing PIPE Investment of an aggregate amount of US$40.2 million or more, (ii) the financing completed by Alps in September 2023, of US$2.5 million, and (iii) the other factors disclosed under “—Board’s Discussion of Valuation and Reasons for the Approval of the Business Combination.” The Globalink Board intends to waive the fairness opinion as a closing condition of the Business Combination.
Even though the Globalink Board will not obtain a final version of the fairness opinion and intends to waive the fairness opinion as a closing condition of the Business Combination, as compensation for the services of Morison Advisory Sdn. Bhd. in connection with the rendering of its draft opinion to the Globalink Board, Globalink agreed to pay Morison Advisory Sdn. Bhd. an aggregate fee of US$35,000.
The Globalink Board determined the amount of consideration to be paid to Alps Holdco’s securityholders and the US$1.6 billion valuation of Alps Holdco, without first seeking the recommendation of Morison Advisory Sdn. Bhd.
Draft Opinion of Morison Advisory Sdn. Bhd. to the Globalink Board
Globalink has engaged Morison Advisory Sdn. Bhd. as an independent financial advisor to the Globalink Board, specifically to provide to the Globalink Board an opinion as to the fairness, from a financial point of view, to holders of shares of Globalink common stock of the Merger Consideration to be paid by Globalink in the Business Combination. In June 2024, Morison Advisory Sdn. Bhd. delivered a draft written opinion to the Globalink Board (solely in their capacity as members of the Globalink Board) that, as of the date of such opinion and subject to and based on the assumptions made, procedures followed, matters considered and limitations and qualifications on the review undertaken by Morison Advisory Sdn. Bhd., the Merger Consideration to be paid by Globalink in the Business Combination was fair, from a financial point of view, to holders of shares of Globalink common stock. The full text of Morison Advisory Sdn. Bhd.’s draft written opinion, which describes the assumptions made, procedures followed, matters considered and limitations and qualifications on the review undertaken by Morison Advisory Sdn. Bhd., is attached as Annex J to this proxy statement/prospectus and is incorporated into this proxy statement/prospectus by reference. For purposes of Morison Advisory Sdn. Bhd.’s analyses and opinion, Morison Advisory Sdn. Bhd. did not take into account the aggregate proceeds from the PIPE Investment and any issuance of Earnout Shares in connection with the Business Combination. The description of Morison Advisory Sdn. Bhd.’s draft opinion set forth herein is qualified in its entirety by reference to the full text of Morison Advisory Sdn. Bhd.’s draft opinion.
| 130 |
Morison Advisory Sdn. Bhd.’s draft opinion was furnished for the use and benefit of the Globalink Board (in its capacity as such) in connection with its evaluation of the Merger Consideration from a financial point of view and did not address any other terms, aspects or implications of the Business Combination or any related transactions, the underlying business decision of Globalink to effect or enter into the Business Combination or any related transactions, the relative merits of the Business Combination or any related transactions as compared to any alternative strategy or transaction or the effect of any other transaction. Morison Advisory Sdn. Bhd.’s draft opinion was not a recommendation as to how the Globalink Board, and is not a recommendation as to how any stockholder, should vote or act with respect to any matters relating to the Business Combination or any related transactions, including without limitation, whether stockholders of Globalink should redeem their shares in connection with the Business Combination, or whether to proceed with the Business Combination or any related transaction.
In connection with its draft opinion, Morison Advisory Sdn. Bhd. made such reviews, analyses and inquiries as it deemed necessary and appropriate under the circumstances. Morison Advisory Sdn. Bhd. also took into account its assessment of general economic, market and financial conditions, as well as its experience in securities and business valuation, in general, and with respect to similar transactions, in particular. Morison Advisory Sdn. Bhd.’s procedures, investigations and financial analysis with respect to the preparation of its draft opinion included, but were not limited to, the items summarized below:
| ● | a draft of registration statement on Form F-4 prepared by PubCo in connection with the Business Combination; | |
| ● | Alps Holdco’s presentation deck dated May 2024; | |
| ● | financial projections of Alps Holdco comprising projected forecast income statements for ten (10) financial years (the “Financial Forecast”); | |
| ● | contacts and agreements entered into by Alps Holdco and its subsidiaries; | |
| ● | representation and explanation by the management and directors of Alps Holdco (the “Target Management”); and | |
| ● | other publicly available information in connection with the fairness opinion. |
Financial Analysis
Set forth below under this heading “—Financial Analyses” is a summary of the material financial analyses performed by Morison Advisory Sdn. Bhd. in connection with the delivery of Morison Advisory Sdn. Bhd.’s draft opinion to the Globalink Board. The summary set forth below does not purport to be a complete description of the financial analyses performed by, and underlying the opinion of, Morison Advisory Sdn. Bhd. The financial analyses summarized below include information presented in tabular format. In order for Morison Advisory Sdn. Bhd.’s financial analyses to be fully understood, the tables must be read together with the text of each summary. The tables alone do not constitute a complete description of the financial analyses, nor does the order of the financial analyses described represent the relative importance or weight given to those financial analyses by Morison Advisory Sdn. Bhd. Considering the data below without considering the full narrative description of the financial analyses, including the methodologies and assumptions underlying the analyses, could create a misleading or incomplete view of Morison Advisory Sdn. Bhd.’s financial analyses. Future results may differ from those described and such differences may be material.
Discounted Cash Flow Analysis (Income Approach)
A discounted cash flow analysis is a valuation technique that provides an estimation of the value of a business based on the cash flows that such business is projected to generate. A discounted cash flow analysis begins with an estimation of the annual cash flows that the subject business is expected to generate over a discrete projection period. The estimated cash flows for each of the years in the discrete projection period are then discounted to their present value equivalents using a rate of return appropriate for the risk of achieving the projected cash flows. Morison Advisory Sdn. Bhd. believes that the discounted cash flow method is an appropriate valuation method as Alps Holdco was still early-stage as of March 31, 2024, or the Valuation Date, and historical earnings are loss-making with substantial uncertainties over Alps Holdco’s future cash flow. The discounted cash flow method provides a more comprehensive assessment of Alps Holdco’s value by taking into consideration its business model, growth prospects, and risk profile.
Morison Advisory Sdn. Bhd. performed discounted cash flow analyses for Alps Holdco based on Alps Holdco’s financial projections for the ten (10) years commencing upon consummation of the Business Combination.
| 131 |
Financial Forecast
| Year following Closing (USD) | Year 1 | Year 2 | Year 3 | Year 4 | Year 5 | Year 6 | Year 7 | Year 8 | Year 9 | Year 10 | ||||||||||||||||||||||||||||||
| Total Revenue | 13,629,636 | 23,032,764 | 39,246,092 | 67,401,124 | 116,613,743 | 203,145,196 | 356,108,468 | 627,790,302 | 1,112,354,442 | 1,979,781,088 | ||||||||||||||||||||||||||||||
| Growth (%) | 69.0 | % | 70.4 | % | 71.7 | % | 73.0 | % | 74.2 | % | 75.3 | % | 76.3 | % | 77.2 | % | 78.0 | % | ||||||||||||||||||||||
| Cost of Sales | 7,087,411 | 11,977,037 | 21,192,890 | 36,396,607 | 62,971,421 | 113,761,310 | 199,420,742 | 364,118,375 | 645,165,576 | 1,148,273,031 | ||||||||||||||||||||||||||||||
| Gross Profit | 6,542,225 | 11,055,727 | 18,053,202 | 31,004,517 | 53,642,322 | 89,383,887 | 156,687,726 | 263,671,927 | 467,188,866 | 831,508,057 | ||||||||||||||||||||||||||||||
| GP Margin | 48.0 | % | 48.0 | % | 46.0 | % | 46.0 | % | 46.0 | % | 44.0 | % | 44.0 | % | 42.0 | % | 42.0 | % | 42.0 | % | ||||||||||||||||||||
| Operating Expenses | 2,340,426 | 2,340,426 | 3,404,255 | 3,404,255 | 3,404,255 | 4,680,851 | 4,680,851 | 6,382,979 | 11,702,128 | 23,404,255 | ||||||||||||||||||||||||||||||
| EBITDA | 4,201,800 | 8,715,302 | 14,648,947 | 27,600,262 | 50,238,066 | 84,703,036 | 152,006,875 | 257,288,948 | 455,486,738 | 808,103,801 | ||||||||||||||||||||||||||||||
| EBITDA Margin | 30.8 | % | 37.8 | % | 37.3 | % | 40.9 | % | 43.1 | % | 41.7 | % | 42.7 | % | 41.0 | % | 40.9 | % | 40.8 | % | ||||||||||||||||||||
| Depreciation and Amortisation | - | - | - | - | - | - | - | - | - | - | ||||||||||||||||||||||||||||||
| EBIT | 4,201,800 | 8,715,302 | 14,648,947 | 27,600,262 | 50,238,066 | 84,703,036 | 152,006,875 | 257,288,948 | 455,486,738 | 808,103,801 | ||||||||||||||||||||||||||||||
| EBIT Margin | 30.8 | % | 37.8 | % | 37.3 | % | 40.9 | % | 43.1 | % | 41.7 | % | 42.7 | % | 41.0 | % | 40.9 | % | 40.8 | % | ||||||||||||||||||||
| Finance Costs | - | - | - | - | - | - | - | - | - | - | ||||||||||||||||||||||||||||||
| Net Operating Profit Before Tax | 4,201,800 | 8,715,302 | 14,648,947 | 27,600,262 | 50,238,066 | 84,703,036 | 152,006,875 | 257,288,948 | 455,486,738 | 808,103,801 | ||||||||||||||||||||||||||||||
| PBT Margin | 30.8 | % | 37.8 | % | 37.3 | % | 40.9 | % | 43.1 | % | 41.7 | % | 42.7 | % | 41.0 | % | 40.9 | % | 40.8 | % | ||||||||||||||||||||
| Income Tax | 1,008,432 | 2,091,672 | 3,515,747 | 6,624,063 | 12,057,136 | 20,328,729 | 36,481,650 | 61,749,348 | 109,316,817 | 193,944,912 | ||||||||||||||||||||||||||||||
| Net Operating Profit After Tax | 3,193,368 | 6,623,629 | 11,133,200 | 20,976,199 | 38,180,930 | 64,374,307 | 115,525,225 | 195,539,600 | 346,169,921 | 614,158,889 | ||||||||||||||||||||||||||||||
| PAT Margin | 23.4 | % | 28.8 | % | 28.4 | % | 31.1 | % | 32.7 | % | 31.7 | % | 32.4 | % | 31.1 | % | 31.1 | % | 31.0 | % | ||||||||||||||||||||
| Year following Closing (USD) | Year 1 | Year 2 | Year 3 | Year 4 | Year 5 | Year 6 | Year 7 | Year 8 | Year 9 | Year 10 | ||||||||||||||||||||||||||||||
| EBITDA | 4,201,800 | 8,715,302 | 14,648,947 | 27,600,262 | 50,238,066 | 84,703,036 | 152,006,875 | 257,288,948 | 455,486,738 | 808,103,801 | ||||||||||||||||||||||||||||||
| Adjustments: | ||||||||||||||||||||||||||||||||||||||||
| Less: Income Tax Expense | (1,008,432 | ) | (2,091,672 | ) | (3,515,747 | ) | (6,624,063 | ) | (12,057,136 | ) | (20,328,729 | ) | (36,481,650 | ) | (61,749,348 | ) | (109,316,817 | ) | (193,944,912 | ) | ||||||||||||||||||||
| Less: Incremental Net Working Capital | (1,127,256 | ) | (1,650,549 | ) | (2,138,293 | ) | (3,739,483 | ) | (6,536,459 | ) | (11,719,003 | ) | (20,576,031 | ) | (37,418,085 | ) | (65,565,228 | ) | (117,073,743 | ) | ||||||||||||||||||||
| Net Available Cash Flow | 2,066,111 | 4,973,080 | 8,994,906 | 17,236,716 | 31,644,472 | 52,655,304 | 94,949,194 | 158,121,515 | 280,604,693 | 497,085,147 | ||||||||||||||||||||||||||||||
| PV Factor @ 14.0% | 0.937 | 0.822 | 0.721 | 0.632 | 0.555 | 0.486 | 0.427 | 0.374 | 0.328 | 0.288 | ||||||||||||||||||||||||||||||
| PV Factor of Cash Flows | 1,935,091 | 4,085,716 | 6,482,381 | 10,896,513 | 17,547,935 | 25,613,294 | 40,514,432 | 59,184,037 | 92,130,556 | 143,164,288 | ||||||||||||||||||||||||||||||
| PV Factor @ 15.0% | 0.933 | 0.811 | 0.705 | 0.613 | 0.533 | 0.464 | 0.403 | 0.351 | 0.305 | 0.265 | ||||||||||||||||||||||||||||||
| PV Factor of Cash Flows | 1,926,659 | 4,032,540 | 6,342,377 | 10,568,469 | 16,871,650 | 24,412,036 | 38,278,536 | 55,431,568 | 85,538,826 | 131,765,375 | ||||||||||||||||||||||||||||||
The financial projections that Morison Advisory Sdn. Bhd.’s opinion were based on were prepared by Alps’ finance and business development team, analyzing Alps’ future growth potential and financial performance. The projections period covered the ten (10) years commencing upon consummation of the Business Combination.
The projections were primarily driven by historical track records achieved during the fiscal years ended March 31, 2021, 2022, 2023 and 2024 as well as future market outlooks based on current and future business plans. The financial projections are subject to material uncertainties and risks, as previous performances are not guarantees of future performance and are subject to inherent risks, uncertainties and other factors that could cause actual results to differ materially from the future results expressed or implied. These include but limited to factors such as regulatory approval processes in various jurisdictions, market acceptance, development and commercialization of technologies. Given the inherent risks associated with the bio-technology industry, actual outcomes may materially differ from the forecasts presented.
The following tables represents the revenue track records and annualized historical revenue growth rates achieved for the fiscal years ended March 31, 2021, 2022, 2023, and 2024. For the avoidance of doubt, the revenue figures set out below have been converted from RM to USD at the exchange rate of USD 1.00 to RM 4.70, being the applicable rate at the time of the projections preparation as determined by Alps’ management, as RM was the currency in which the transaction was denominated. Thus, the revenue figures below do not align to the reported audited revenue figures which are disclosed based on actual exchange rates effective as at respective reporting periods.
| USD unless otherwise specified | FY’2021 (Audited) | FY’2022 (Audited) | FY’2023 (Audited) | FY’2024 (Unaudited) | ||||||||||||
| Wellness and others | 173,434 | 396,128 | 790,692 | 959,706 | ||||||||||||
| Cosmetic surgery and others | 8,529 | 104,697 | 120,612 | 261,400 | ||||||||||||
| Hair transplant | 22,158 | 332,743 | 493,454 | 817,398 | ||||||||||||
| DNA testing and mRNA testing | 178,600 | 131,615 | 82,545 | 131,134 | ||||||||||||
| Aesthetic | 104,295 | 65,805 | 165,963 | 16,729 | ||||||||||||
| Therapeutic products | 306,822 | 221,638 | 139,502 | 165,323 | ||||||||||||
| Clinical trial | n.a. | 213,059 | 478,424 | 2,468 | ||||||||||||
| Covid-19 screening | 1,411,843 | 1,684,517 | 1,061,496 | 157,876 | ||||||||||||
| Others | 93,503 | 81,778 | 72,070 | 123,249 | ||||||||||||
| Total Revenue | 2,299,184 | 3,231,980 | 3,404,758 | 2,635,283 | ||||||||||||
In determining the future growth rates, Alps management have considered products and services with track record of significant contributions and anticipated to have strong growth prospects in the future.
Average Historical Growth Rate p.a. | FY’2021 | FY’2022 | FY’2023 | FY’2024 | ||||||||||||||||
| Wellness and others | 83.0 | % | n.a. | 128.4 | % | 99.6 | % | 21.4 | % | |||||||||||
| Cosmetic surgery and others | 66.0 | % | n.a. | 1,127.5 | % | 15.2 | % | 116.7 | % | |||||||||||
| Hair transplant | 57.0 | % | n.a. | 1,401.6 | % | 48.3 | % | 65.6 | % | |||||||||||
| DNA testing and mRNA testing | 59.0 | % | n.a. | -26.3 | % | -37.3 | % | 59.0 | % | |||||||||||
| 132 |
Based on discussions with Alps’ management, overall revenue projections of existing products and services are anchored in historical performance, utilizing the most recent data as of the fiscal year ended March 31, 2024, as the foundational baseline except for products and services not yet launched as of March 31, 2024. Revenue projections are forecasted to grow at a constant annual rate aligned with the average historical growth rate, as outlined in the table above.
In respect of revenue streams originating from the NK Cell, Car-T and blood testing segments where Alps has yet commence the operations as of March 31, 2024, the respective projected revenues have been determined based on Alps’ management assessment of the current market outlook and future strategic business plans.
For the NK Cell segment, projected revenue for the fiscal year ending March 31, 2025, is calculated based on an estimated total customer base of 22,348 patients, with an assumed penetration rate of 1.6% and an anticipated price per treatment of USD11,500. Future revenue for this segment is projected to grow at an annual rate of 83.0% based on management estimate. Similarly, for the Car-T segment, projected revenue is based on an estimated customer base of 1,940 patients, assuming a penetration rate of 1.6% and a projected price per treatment of USD46,250. Revenue for this segment is expected to grow at an annual rate of 83.0% in subsequent periods based on Alps’ management estimate.
For the blood testing segment, projected revenue for the fiscal year ending March 31, 2025, is based on an anticipated volume of 182,500 tests, with an estimated price per test of USD24. Alps’ management anticipates and estimates that revenue for this segment will grow at an annual rate of 50.0% thereafter.
The revenue breakdown is presented in the following table:
| USD unless otherwise specified | Annual Growth Rate p.a. | Year 1 | Year 2 | Year 3 | Year 4 | Year 5 | Year 6 | Year 7 | Year 8 | Year 9 | Year 10 | |||||||||||||||||||||||||||||||||
| Wellness and others | ||||||||||||||||||||||||||||||||||||||||||||
| Mesenchymal Stem Cells | 83.0 | % | 1,756,262 | 3,213,960 | 5,881,548 | 10,763,232 | 19,696,715 | 36,044,988 | 65,962,328 | 120,711,060 | 220,901,240 | 404,249,268 | ||||||||||||||||||||||||||||||||
| NK Cell | 83.0 | % | 4,112,032 | 7,525,019 | 13,770,784 | 25,200,535 | 46,116,978 | 84,394,070 | 154,441,148 | 282,627,300 | 517,207,959 | 946,490,565 | ||||||||||||||||||||||||||||||||
| Car-T | 83.0 | % | 1,435,600 | 2,627,148 | 4,807,681 | 8,798,056 | 16,100,443 | 29,463,811 | 53,918,773 | 98,671,355 | 180,568,580 | 330,440,501 | ||||||||||||||||||||||||||||||||
| Cosmetic surgery and others | 66.0 | % | 433,924 | 720,314 | 1,195,722 | 1,984,898 | 3,294,930 | 5,469,584 | 9,079,510 | 15,071,986 | 25,019,497 | 41,532,365 | ||||||||||||||||||||||||||||||||
| Hair transplant | 57.0 | % | 1,283,315 | 2,014,805 | 3,163,244 | 4,966,293 | 7,797,079 | 12,241,415 | 19,219,021 | 30,173,863 | 47,372,964 | 74,375,554 | ||||||||||||||||||||||||||||||||
| DNA testing and mRNA testing | 59.0 | % | 208,502 | 331,518 | 527,114 | 838,111 | 1,332,597 | 2,118,830 | 3,368,939 | 5,356,613 | 8,517,015 | 13,542,053 | ||||||||||||||||||||||||||||||||
| Blood testing | 50.0 | % | 4,400,000 | 6,600,000 | 9,900,000 | 14,850,000 | 22,275,000 | 33,412,500 | 50,118,750 | 75,178,125 | 112,767,188 | 169,150,781 | ||||||||||||||||||||||||||||||||
| 13,629,636 | 23,032,764 | 39,246,092 | 67,401,124 | 116,613,743 | 203,145,196 | 356,108,468 | 627,790,302 | 1,112,354,442 | 1,979,781,088 | |||||||||||||||||||||||||||||||||||
The revenue growth rates assumptions as per the table above was based on the historical track record of Alps during the fiscal years ended March 31, 2021, 2022, 2023 and 2024 as well as expectations provided by Alps’ management. Per our discussion with Alps’ management, the projected revenue growth is consistent with selected peer companies annualized revenue growth rate averages of approximately 117%.
Additionally, the revenue growth rates took into consideration the trends and outlook of the markets that Alps operates in and Alps’ competitive position. Alps relies on historical data and industry benchmarks to project revenue milestones. It is important to note that the revenue trajectory is based on key regulatory approval milestones being achieved, where these timelines are preliminary and may be subject to changes beyond the control of Alps. Various factors, including but not limited to regulatory review periods, patient recruitment, trial execution, and unforeseen circumstances during the development process, could potentially impact these timelines.
For avoidance of doubt, Alps’ management has explicitly excluded revenue contribution from products pipeline which are still in proof of concept and pre-clinical stages except for NK Cell, Mesenchymal Stem Cells and Car-T but only included those products and services with operational history.
In terms of the operating expenses, the Alps management has projected the variable costs as a percentage of revenue based on historical operating metrics and factoring future growth rates as the operations scale as follows:
| USD unless otherwise specified | Year 1 | Year 2 | Year 3 | Year 4 | Year 5 | Year 6 | Year 7 | Year 8 | Year 9 | Year 10 | ||||||||||||||||||||||||||||||
| Variable expenses | 7,087,411 | 11,977,037 | 21,192,890 | 36,396,607 | 62,971,421 | 113,761,310 | 199,420,742 | 364,118,375 | 645,165,576 | 1,148,273,031 | ||||||||||||||||||||||||||||||
| As a percentage of total revenue | 52.0 | % | 52.0 | % | 54.0 | % | 54.0 | % | 54.0 | % | 56.0 | % | 56.0 | % | 58.0 | % | 58.0 | % | 58.0 | % | ||||||||||||||||||||
| 133 |
With respect to the fixed expenses, the Alps management has projected the base fixed expense as of fiscal year 2025 with estimated step-up increase over the projection periods due to expected increase in administrative, staff costs, rental and other expenses as follows:
| USD unless otherwise specified | Year 1 | Year 2 | Year 3 | Year 4 | Year 5 | Year 6 | Year 7 | Year 8 | Year 9 | Year 10 | ||||||||||||||||||||||||||||||
| Fixed expenses | 2,340,426 | 2,340,426 | 3,404,255 | 3,404,255 | 3,404,255 | 4,680,851 | 4,680,851 | 6,382,979 | 11,702,128 | 23,404,255 | ||||||||||||||||||||||||||||||
Alps’ business is still in an early stage development biotechnology research, medical and wellness company with significant pipelines in pre-clinical testing phase, and if its business grows slower than Alp’s management expectations or current pipeline pre-clinical candidates fail to secure relevant regulatory approvals or experienced extended delays than beyond management expectation or fail to secure the necessary funding requirements in continuing the research and development, Alps’ business, financial condition, results of operations and prospects could be materially and adversely affected.
Morison Advisory Sdn. Bhd. strongly cautioned that these key assumptions are heavily dependent on a number of factors but not limited to:
| 1. | Economic conditions: broader macroeconomic factors, such as inflation, interest rate fluctuations, and general economic downturns, can significantly influence demand for biotechnology solutions, impact pricing strategies and operational costs. | |
| 2. | Market conditions: the competitive landscape within the emerging biotech sector is rapidly evolving, with actions by competitors affecting market share, pricing and profitability. Additionally, shifts in consumer preferences for healthcare and biotechnology solutions, alongside technological advancements, can affect the commercial viability of products. | |
| 3. | Company-specific factors: key drivers of revenue, such as patient volume, service pricing, and a diversified customer base, are crucial for financial stability. Cost structure, including labor, materials, and operational overhead, alongside significant investments in new biotechnology products, services, or platforms, can create financial strain. Early-stage clinical trials pose high uncertainty and setbacks could severely impact future revenue potential. | |
| 4. | Financial factors: access to financing remains a critical risk, especially for early-stage biotech companies that are heavily reliant on external funding needed for research and clinical trials. Any constraints in raising capital could delay or halt R&D activities and clinical trials. | |
| 5. | Regulatory and legal environment: the biotechnology sector is highly regulated and changes in industry regulations, government healthcare policies or intellectual property laws may introduce new compliance hurdles. Delays in regulatory approvals or unexpected shifts in legal requirements for clinical trials and certifications could impede product development milestone and increase development costs. | |
| 6. | Risks and uncertainties: disruptions such as supply chain issues or emerging technological innovations that may render current research development obsolete. Early clinical trials, face significant risks in R&D outcomes. Uncertain efficacy, unforeseen side effects or slow progress in technological advancements could delay the path to commercialisation or pathways to monetisation | |
| 7. | Intellectual Property Risks: protecting patents and proprietary assets are critical in the biotech industry as patent infringement could compromise the competitive edge, market entry or raise legal disputes. | |
| 8. | Talent Acquisition and Retention: ability to attract and retain skilled scientists, researchers, and management personnel is crucial as shortage of sufficient skilled talent or high employee turnover may jeopardise and adversely impact clinical trial progress. |
However, Morison Advisory Sdn. Bhd. understands that the management and board of directors of Alps have reviewed the forecasts and Alps has affirmed to Globalink that the forecasts represents the views of the management and board of directors of Alps about its future performance as of the date of this proxy statement/prospectus.
DCF Valuation
| USD mil unless otherwise specified | Low Case | High Case | ||
| Present Value of Discrete Cash Flow | 375.1 mil | 401.5 mil | ||
| Present Value of Terminal Value | 1,068.3 mil | 1,243.6 mil | ||
| Indicative Enterprise Value | 1,443.4 mil | 1,645.2 mil | ||
| Add: Surplus Cash and Bank Balances | 0.5 mil | 0.5 mil | ||
| Less: Amount due to Directors | 3.5 mil | 3.5 mil | ||
| Less: Loans and Borrowings | 0.5 mil | 0.5 mil | ||
| Indicative Equity Value | 1,439.8 mil | 1,641.5 mil |
Discounted Cash Flow Analysis
Morison Advisory Sdn. Bhd. determined the Enterprise Value (“EV”) by computing the net present value (“NPV”) of the free cash flow as of the Valuation Date. Based on the free cash flow, the indicative equity value of Alps Holdco as of March 31, 2024 ranges from USD1.4 billion to USD1.6 billion.
Globalink Board’s assessment of the pre-money valuation of Alps of approximately $1.6 billion based on the verbal commitment by Alps and the Seller Representative for PIPE Investment of an aggregate amount of US$40.2 million or more is within range of the computed indicative equity value of Alps Holdco as of March 31, 2024 ranges from USD1.4 billion to USD1.6 billion.
| 134 |
The key consideration in Morison Advisory Sdn. Bhd.’s valuation include:
| ● | nil terminal growth rate justified by the nature of the business as a loss-making biotech company. This conservative approach acknowledges the inherent risks and uncertainties associated with the biotech sector, particularly for companies that are currently incurring losses due to substantial investments in research and development; | |
| ● | Morison Advisory Sdn. Bhd. applied discount rates ranging from 14.0% to 15.0%, reflecting Alps Holdco’s current development phase and its associated risk profile. See below section for the computation of WACC; | |
| ● | although Target Management has not projected capital expenditures, Morison Advisory Sdn. Bhd. assumed that Target Management will replace and maintain its property, plant and equipment starting from the terminal year onwards; | |
| ● | the networking capital projections are based on the turnover days of trade receivables and trade payables; and | |
| ● | Morison Advisory Sdn. Bhd. considered and applied mid-point discounting as it believed that this was a better reflection of the cash flows received than end period discounting. |
The discount rate used in the DCF analysis is derived from the Alps Holdco’s WACC. The WACC reflects the cost of equity and debt financing, adjusted for Alps Holdco’s capital structure and risk profile.
WACC Computation (discount rates range between 14.0% and 15.0%)
| Key Parameters | Low Case | High Case | Source of Information and Comments | |||||||
| Malaysia Risk Free Rate | 4.1 | % | 4.1 | % | 20-Year MGS rate. Source: Bank Negara Malaysia (“BNM”) | |||||
| Equity Risk Premium | 6.4 | % | 6.4 | % | Source: Damodaran Jan 2024 | |||||
| Unlevered Beta | 0.44 | 0.44 | Unlevered Equity Beta = Levered Equity Beta / [1 + (1-Tax Rate) × Debt-to-Equity] | |||||||
| Relevered Equity Beta | 0.48 | 0.48 | Levered Equity Beta = Unlevered Equity Beta × [1 + (1-Tax Rate) × Debt-to-Equity] | |||||||
| Preliminary Cost of Equity | 7.1 | % | 7.1 | % | Calculated | |||||
| Size Premium | 5.0 | % | 5.0 | % | Estimated | |||||
| Company-Specific Risk | 3.0 | % | 4.0 | % | Estimated | |||||
| Cost of Equity Capital | 15.1 | % | 16.1 | % | Calculated | |||||
| Subject’s Estimated Pre-Tax Cost of Debt Capital | 6.5 | % | 7.9 | % | Based on A- to BBB rated corporate bonds in Malaysia. Source: BNM | |||||
| Tax Rate | 24.0 | % | 24.0 | % | Corporate tax rate of the subject company | |||||
| After-Tax Cost of Debt | 5.0 | % | 6.0 | % | Calculated | |||||
| Debt to Capital | 11.4 | % | 11.4 | % | Based on the median debt structure of the comparable companies | |||||
| Equity to Capital | 88.6 | % | 88.6 | % | Based on the median debt structure of the comparable companies | |||||
| WACC Computation | 14.0 | % | 15.0 | % | Calculated | |||||
Selected Public Comparable Companies
In validating the key assumptions and projections to the valuation, Morison Advisory Sdn. Bhd. reviewed publicly listed comparable companies globally that are principally involved in the provision of biotech research, medical and wellness services. These companies serve as benchmarks for assessing Alps Holdco’s growth prospects, profitability and risk factors. While the comparable companies provide valuable insights, they can differ from Alps Holdco in terms of business activities, scale of operations, geographical presence, historical track record, financial performance, capital structure, and other factors.
Morison Advisory Sdn. Bhd. reviewed certain financial information for 13 selected public companies (the “selected public companies”) with operations in the biotech industry. The selected public companies were as follow:
| ● | Stoke Therapeutic s, Inc. (Nasdaq: STOK) | ● | Accolade, Inc. (Nasdaq: ACCD) | |
| ● | Precigen, Inc. (Nasdaq: PGEN) | ● | Amicus Therapeutics, Inc. (Nasdaq: FOLD) | |
| ● | AbCellera Biologics Inc. (Nasdaq: ABCL) | ● | Wockhardt Limited (NSEI: WOCKPHARMA) | |
| ● | Hualan Biological Engineering Inc. (SZSE: 002007) | ● | Strides Pharma Science Limited (NSEI: STAR) | |
| ● | HLB bioStep Co.,Ltd. (KOSDAQ: A278650) | ● | SD Biosensor, Inc. (KOSE: A137310) | |
| ● | Acadia Pharmaceuticals Inc. (Nasdaq: ACAD) | ● | Obio Technology (Shanghai) Corp., Ltd. (SHSE: 688238) | |
| ● | Agios Pharmaceuticals, Inc. (Nasdaq: AGIO) |
Unaffiliated Representative
Globalink’s independent directors have not retained an unaffiliated representative to act solely on behalf of unaffiliated security holders of Globalink for purposes of negotiating the terms of the Business Combination and/or preparing a report concerning the approval of the Business Combination.
Satisfaction of 80% Test
It is a requirement under the Nasdaq Rules that the business or assets acquired in Globalink’s initial business combination have a fair market value equal to at least 80% of Globalink’s assets held in the Trust Account (excluding taxes payable on the income earned on the Trust Account) at the time of the execution of a definitive agreement for such initial business combination. As of January 30, 2024, the date of the execution of the Merger Agreement, the fair value of marketable securities held in the Trust Account was approximately $28.8 million and 80% thereof represents approximately $23.1 million. In reaching its conclusion that the Business Combination meets the 80% asset test, the Board reviewed the pre-money valuation of Alps of approximately $1.6 billion. In determining whether the pro forma total enterprise value described above represents the fair market value of Alps, the Board considered all of the factors described in this section and the section of this proxy statement/prospectus entitled “The Acquisition Merger Proposal (Proposal 2) — The Merger Agreement” and that the pre-money valuation of approximately $2.08 billion (including earnout, $1.6 billion excluding earnout) was determined as a result of arm’s length negotiations. As a result, the Board concluded that the fair market value of the equity acquired was significantly in excess of 80% of the assets held in the Trust Account (excluding taxes payable on the income earned on the Trust Account).
| 135 |
Interests of Certain Persons in the Business Combination
In considering the recommendation of the Board to approve the Merger Agreement, Globalink stockholders should be aware that certain Globalink executive officers and directors may be deemed to have interests in the Business Combination that are different from, or in addition to, those of Globalink stockholders generally, including:
| ● | The Initial Stockholders, including the officers and directors of Globalink, have waived their right to redeem any shares of Globalink common stock in connection with a stockholder vote to approve a proposed initial business combination or sell any shares of Globalink common stock to Globalink in a tender offer prior to a proposed initial business combination, or to receive distributions with respect to the Founder Shares upon the liquidation of the Trust Account if Globalink is unable to consummate a business combination. If an initial business combination, such as the Business Combination, is not completed by January 9, 2025 (and may be extended to June 9, 2025 on a monthly basis by resolutions of the Globalink Board, if requested by the Sponsor, and subject to the Sponsor depositing additional funds into the Trust Account), Globalink will be required to dissolve and liquidate. In such event, the 2,875,000 Founder Shares currently held by the Initial Stockholders, which were acquired prior to the IPO will be worthless because such holders have agreed to waive their rights to any liquidation distributions through the Insider Letter, in consideration for inducing Chardan, the representative of the underwriters for Globalink’s IPO to proceed with the IPO, and the economic benefit the IPO would confer upon the Initial Stockholders and PGM. The Founder Shares were purchased for an aggregate purchase price of $25,000, or less than $0.01 per share, and had a total market value of $34.36 million as of December 12, 2024. Accordingly, the Initial Stockholders will receive a positive rate of return so long as the market price of the common stock is at least $0.01 per share, even if public stockholders experience a negative rate of return in the Combined Company. | |
| ● | The fact that the Initial Stockholders’ Founder Shares became subject to a lock-up whereby, subject to certain limited exceptions, the Initial Stockholders agree not to offer, sell, contract to sell, pledge or otherwise dispose of, directly or indirectly, any of the Lock-up Shares, enter into a transaction that would have the same effect, or enter into any swap, hedge or other arrangement that transfers, in whole or in part, any of the economic consequences of ownership of the Lock-Up Shares or otherwise, publicly disclose the intention to make any offer, sale, pledge or disposition, or to enter into any transaction, swap, hedge or other arrangement, or engage in any Short Sales (as defined in the Lock-up Agreement) with respect to the Lock-Up Shares until (i) (w) with respect to one hundred percent (100%) of the Lock-Up Shares, the date from the Closing until the last date that is six (6) months after the date of the Closing, (x) with respect to ninety percent (90%) of the Lock-Up Shares, the 1st day of the 7th month from the Closing until the date that is nine (9) months after the date of the Closing, (y) with respect to seventy percent (70%) of the Lock-Up Shares, the 1st day of the 10th month from the Closing until the date that is twelve (12) months after the date of the Closing, (z) with respect to forty percent (40%) of the Lock-Up Shares, the 1st day of the 13th month from the Closing until the date that is fifteen (15) months after the date of the Closing, or (ii) if sooner, the date after the Closing on which Globalink consummates a liquidation, merger, share exchange or other similar transaction with an unaffiliated third party that results in all of Globalink’s stockholders having the right to exchange their equity holdings in Globalink for cash, securities or other property. For the avoidance of doubt, no Lock-up Shares shall be subject to the terms in the Lock-up Agreement from and after the date that is fifteen (15) months after the date of the Closing; |
| ● | The exercise of Globalink’s directors’ and officers’ discretion in agreeing to changes or waivers in the terms of the Business Combination may result in a conflict of interest when determining whether such changes or waivers are appropriate and in Globalink stockholders’ best interest. Globalink’s directors were aware of and considered these interests, among other matters, in evaluating the Business Combination, and in recommending to Globalink stockholders that they approve the Business Combination. Further, the interests of Globalink’s officers or directors may be different from or in addition to (and which may conflict with) your interests and may be incentivized to complete a less favorable business combination rather than liquidating Globalink. Public stockholders should take these interests into account in deciding whether to approve the Business Combination. |
| ● | As of September 30, 2024, Globalink’s officers and directors have incurred an aggregate of $11,798 in out-of-pocket expenses. On each of September 5, 2023, September 29, 2023 and November 7, 2023, an affiliate of the Sponsor advanced $130,000 to the Company, for a total advance of $390,000, for Globalink’s extension payments. Unless Globalink consummates an initial business combination, Globalink’s officers, directors and the Sponsor (including its affiliates) will not receive reimbursement for any out-of-pocket expenses incurred by them to the extent that such expenses exceed the amount of available proceeds not deposited in the Trust Account. Upon the consummation of an initial business combination, Globalink’s offices, directors, and the Sponsor (including its affiliates) would be entitled to reimbursement for out-of-pocket expenses incurred by them from the Trust Account disbursements after redemption payouts and payments of deferred underwriting discounts. | |
| ● | The fact that, if the Trust Account is liquidated, including in the event Globalink is unable to complete an initial business combination within the required time period, the Sponsor has agreed to be liable to Globalink if and to the extent any claims by a vendor for services rendered or products sold to Globalink, or a prospective target business with which Globalink has discussed entering into a transaction agreement, reduce the amount of funds in the Trust Account. This liability will not apply with respect to any claims by a third party who executed a waiver of any right, title, interest or claim of any kind in or to any monies held in the Trust Account or to any claims under Globalink’s indemnity of the underwriters of the IPO against certain liabilities, including liabilities under the Securities Act. | |
| ● | As of the date hereof, the deadline that Globalink has to consummate its initial business combination has been extended for a total of eighteen times, and a total of US$1,950,000 has been deposited into the Trust Account in connection with the previous extensions. In connection with funding the extensions, Globalink has issued an aggregate of US$3.85 million in promissory notes to PGM, which are payable upon the Closing. In the event of a liquidation, PGM has waived its rights to any liquidation distributions through the Insider Letter. PGM is an affiliate of the Sponsor as its 95% equity holder has a familial relationship with the control person of the Sponsor. As a result, the Sponsor has an additional interest in causing Globalink to consummate its initial business combination due to its familial relationship with PGM. | |
| ● | After the Closing, the Sponsor will have the sole authority to handle various matters relating to the Escrow Account and indemnifications under the Merger Agreement. The Sponsor may have a conflict of interest when exercising such authority to handle matters relating to the Escrow Account and indemnifications after Closing, and they may not make decisions in the Globalink stockholders’ best interests. For details, see “Summary of the Proxy Statement/Prospectus—Parties to the Business Combination—Parent Representative.” |
These interests may have influenced Globalink’ directors in making their recommendation that you vote in favor of the approval of the Business Combination.
Upon the consummation of an initial business combination by Globalink, other than the PubCo ordinary shares that the Initial Stockholders are entitled to upon conversion of their shares of Globalink common stock at the completion of the Redomestication Merger, no additional compensation will be awarded to, earned by, or paid to the Sponsor, its affiliates or any promoters for all services rendered or to be rendered to Globalink and its affiliates. The Sponsor is expected to own an equity interest of approximately 1.7% in the Combined Company after the consummation of the Business Combination.
Prior to the consummation of an initial business combination by Globalink, the insider shares can only be transferred by the Initial Stockholders to (1) the Initial Stockholders or their respective affiliates or members, or to Globalink’s officers, directors, advisors and employees, (2) the Initial Stockholder’s affiliates or its members upon its liquidation, (3) to relatives and trusts for estate planning purposes, (4) by virtue of the laws of descent and distribution upon death of an Initial Stockholder, (5) pursuant to a qualified domestic relations order, or (6) by private sales made at prices no greater than the price at which the Insider Shares were originally purchased.
| 136 |
Appraisal Rights
Appraisal rights are not available to security holders of Globalink in connection with the proposed Business Combination.
Total Ordinary Shares Outstanding upon Consummation of the Business Combination
It is anticipated that, upon the Closing of the Business Combination, Globalink’s public stockholders and Globalink’s right holders (other than the PIPE Investors) will retain an ownership interest of approximately 0.8% in the Combined Company, the PIPE Investors will own approximately 2.4% of the Combined Company, assuming that the PIPE Investors will hold 4,020,000 PubCo ordinary shares, the Sponsor and directors and officers of Globalink will retain an ownership interest of approximately 1.7% in the Combined Company, PGM, an affiliate of the Sponsor, will retain an ownership interest of approximately 0.4% in the Combined Company, IBDC Asia Sdn. Bhd., an advisory firm to Alps, will own approximately 0.9% in the Combined Company, representing finder’s fees payable in PubCo ordinary shares with value equal to 1% of the aggregate consideration for the Business Combination of US$1.6 billion, and the Alps Holdco Shareholders will own approximately 93.8% in the Combined Company. The ownership percentages with respect to the Combined Company does not take into account (i) the redemption level of any shares by the Globalink’s public stockholders other than the five scenarios described below or (ii) the Earnout Shares.
The following summarizes the pro forma ownership of ordinary shares as of December 6, 2024, including ordinary shares underlying units, of the Combined Company following the Business Combination and the PIPE Investment under the no redemption, 25% redemptions, 50% redemption, 75% redemption and maximum contractual redemption scenarios, assuming that no Earnout Shares are issued.
Assuming Redemptions | Assuming 25% Redemptions | Assuming 50% Redemptions | Assuming 75% Redemptions | Assuming Maximum Contractual Redemptions | ||||||||||||||||||||||||||||||||||||
Number of Shares | % | Number of Shares | % | Number of Shares | % | Number of Shares | % | Number of Shares | % | |||||||||||||||||||||||||||||||
| Alps Holdco Shareholders | 160,000,000 | 93.8 | % | 160,000,000 | 93.8 | % | 160,000,000 | 93.8 | % | 160,000,000 | 93.9 | % | 160,000,000 | 93.9 | % | |||||||||||||||||||||||||
| Globalink Public Shareholders | 277,511 | 0.2 | % | 208,133 | 0.1 | % | 138,755 | 0.1 | % | 69,377 | 0.0 | % | 0 | 0.0 | % | |||||||||||||||||||||||||
| Initial Stockholders | 2,875,000 | 1.7 | % | 2,875,000 | 1.7 | % | 2,875,000 | 1.7 | % | 2,875,000 | 1.7 | % | 2,875,000 | 1.7 | % | |||||||||||||||||||||||||
| Globalink Public Rights Holders | 1,150,000 | 0.6 | % | 1,150,000 | 0.7 | % | 1,150,000 | 0.7 | % | 1,150,000 | 0.7 | % | 1,150,000 | 0.7 | % | |||||||||||||||||||||||||
| PIPE Investors | 4,020,000 | 2.4 | % | 4,020,000 | 2.4 | % | 4,020,000 | 2.4 | % | 4,020,000 | 2.4 | % | 4,020,000 | 2.4 | % | |||||||||||||||||||||||||
| PGM | 627,000 | 0.4 | % | 627,000 | 0.4 | % | 627,000 | 0.4 | % | 627,000 | 0.4 | % | 627,000 | 0.4 | % | |||||||||||||||||||||||||
| IBDC Asia Sdn. Bhd. | 1,600,000 | 0.9 | % | 1,600,000 | 0.9 | % | 1,600,000 | 0.9 | % | 1,600,000 | 0.9 | % | 1,600,000 | 0.9 | % | |||||||||||||||||||||||||
| Total Shares | 170,549,511 | 100 | % | 170,480,133 | 100 | % | 170,410,755 | 100 | % | 170,341,377 | 100 | % | 170,272,000 | 100 | % | |||||||||||||||||||||||||
The table below illustrates the potential dilutive impact, excluding the exercise of warrants, to the holders of securities across five different redemption scenarios based on the assumptions described above, except that these scenarios assume the Combined Company has achieved all revenue milestones and the maximum number of Earnout Shares (48,000,000) are issued according to the earnout arrangement:
Assuming No Redemptions | Assuming 25% Redemptions | Assuming 50% Redemptions | Assuming 75% Redemptions | Assuming Maximum Contractual Redemptions | ||||||||||||||||||||||||||||||||||||
Number of Shares | % | Number of Shares | % | Number of Shares | % | Number of Shares | % | Number of Shares | % | |||||||||||||||||||||||||||||||
| Alps Holdco Shareholders | 208,000,000 | 95.2 | % | 208,000,000 | 95.2 | % | 208,000,000 | 95.2 | % | 208,000,000 | 95.3 | % | 208,000,000 | 95.3 | % | |||||||||||||||||||||||||
| Globalink Public Shareholders | 277,511 | 0.1 | % | 208,133 | 0.1 | % | 138,755 | 0.1 | % | 69,377 | 0.0 | % | 0 | 0.0 | % | |||||||||||||||||||||||||
| Initial Stockholders | 2,875,000 | 1.3 | % | 2,875,000 | 1.3 | % | 2,875,000 | 1.3 | % | 2,875,000 | 1.3 | % | 2,875,000 | 1.3 | % | |||||||||||||||||||||||||
| Globalink Public Rights Holders | 1,150,000 | 0.5 | % | 1,150,000 | 0.5 | % | 1,150,000 | 0.5 | % | 1,150,000 | 0.5 | % | 1,150,000 | 0.5 | % | |||||||||||||||||||||||||
| PIPE Investors | 4,020,000 | 1.9 | % | 4,020,000 | 1.9 | % | 4,020,000 | 1.9 | % | 4,020,000 | 1.9 | % | 4,020,000 | 1.9 | % | |||||||||||||||||||||||||
| PGM | 627,000 | 0.3 | % | 627,000 | 0.3 | % | 627,000 | 0.3 | % | 627,000 | 0.3 | % | 627,000 | 0.3 | % | |||||||||||||||||||||||||
| IBDC Asia Sdn. Bhd. | 1,600,000 | 0.7 | % | 1,600,000 | 0.7 | % | 1,600,000 | 0.7 | % | 1,600,000 | 0.7 | % | 1,600,000 | 0.7 | % | |||||||||||||||||||||||||
| Total Shares | 218,549,511 | 100 | % | 218,480,133 | 100 | % | 218,410,755 | 100 | % | 218,341,377 | 100 | % | 218,272,000 | 100 | % | |||||||||||||||||||||||||
All of the relative percentages above are for illustrative purposes only and are based upon certain assumptions as described in the section entitled “Frequently Used Terms — Share Calculations and Ownership Percentages” and, with respect to the determination of the “maximum contractual redemptions,” the section entitled “Unaudited Pro Forma Condensed Combined Financial Statements.” Should one or more of the assumptions prove incorrect, actual ownership percentages may vary materially from those described in this proxy statement/prospectus as anticipated, believed, estimated, expected or intended. See “Unaudited Pro Forma Condensed Combined Financial Information and Notes.”
Anticipated Accounting Treatment
The Business Combination will be accounted for as a “capital reorganization,” with no goodwill or other intangible assets recorded, in accordance with IFRS. A capital reorganization does not result in a new basis of accounting, and the financial statements of the combined entity represent the continuation of the financial statements of Alps Holdco in many respects. However, Globalink does not meet the definition of a “business” pursuant to IFRS 3 Business Combinations, and thus, for accounting purposes, the Business Combination will be accounted for as a capital reorganization.
| 137 |
This determination was primarily based on the assumption that Alps Holdco shareholders will hold the largest minority of the voting power of PubCo, Alps Holdco operations will substantially comprise the ongoing operations of PubCo, Alps Holdco designees are expected to comprise a majority of the governing body of PubCo, and Alps Holdco’s senior management will comprise the senior management of PubCo.
Under this method of accounting, Globalink will be treated as the “acquired” company for financial reporting purposes. For accounting purposes, Alps Holdco will be deemed to be the accounting acquirer in the transaction and, consequently, the transaction will be treated as a recapitalization of Alps Holdco (i.e., a capital transaction involving the issuance of shares by PubCo for the shares of Alps Holdco). Accordingly, the consolidated assets, liabilities and results of operations of Alps will become the historic financial statements of the Combined Company, and Globalink’s assets, liabilities and results of operations will be consolidated with Alps’ beginning on the acquisition date. Operations prior to the Business Combination will be presented as those of Alps Holdco in future reports. The net assets of Globalink will be recognized at carrying value, with no goodwill or other intangible assets recorded.
The deemed costs of the shares issued by PubCo, which represents the fair value of the shares that Alps Holdco would have had to issue for the ratio of ownership interest in PubCo to be the same as if the Business Combination had taken the legal form of Alps Holdco acquiring shares of Globalink, in excess of the net assets of Globalink will be accounted for as stock-based compensation under IFRS 2 Share-Based Payment.
Redemption Rights
Pursuant to Globalink Charter, a holder of shares of Globalink common stock has the right to redeem such shares for cash equal to its pro rata share of the Trust Account (net of taxes payable) in connection with a business combination.
If you are a public stockholder and you seek to have your shares redeemed, you must submit your request in writing that Globalink redeem your shares of Globalink common stock for cash no later than , Eastern Time on , 2025 (at least two business days before the Special Meeting). The request must be signed by the applicable stockholder in order to validly request redemption. A stockholder is not required to submit a proxy card or vote in order to validly exercise redemption rights. The request must identify the holder of shares of common stock to be redeemed and must be sent to Continental at the following address:
Continental
Stock Transfer & Trust Company
1 State Street, 30th floor
New York, NY 10004
Attn: SPAC Redemption Team
E-mail: spacredemptions@continentalstock.com
You may tender the shares of Globalink common stock for which you are electing redemption by two (2) business days before the Special Meeting by either:
| ● | delivering certificates representing shares of Globalink common stock to Continental, or | |
| ● | delivering the shares of Globalink common stock electronically through the DWAC system. |
Globalink stockholders will be entitled to redeem their shares of Globalink common stock for a full pro rata share of the Trust Account (anticipated to be no less than approximately $10.98 per share based on the balance in the Trust Account as of December 6, 2024) net of taxes payable.
Any corrected or changed written demand of redemption rights must be received by Continental two (2) business days prior to the Special Meeting. No demand for redemption will be honored unless the holder’s shares have been delivered (either physically or electronically) to Continental at least two (2) business days prior to the vote at the Special Meeting.
| 138 |
Public stockholders may seek to have their shares redeemed regardless of whether they vote for or against the Business Combination and whether or not they are holders of shares of Globalink common stock as of the Record Date. Any public stockholder who holds shares of Globalink common stock on or before , 2025 (two (2) business days before the Special Meeting) will have the right to demand that his, her or its shares be redeemed for a pro rata share of the aggregate amount then on deposit in the Trust Account, less any taxes then due but not yet paid, at the consummation of the Business Combination.
In connection with tendering your shares for redemption, you must elect either to physically tender your share certificates to Globalink’s transfer agent or deliver your shares to the transfer agent electronically using The Depository Trust Company’s DWAC System, in each case, two (2) business days before the Special Meeting.
If you wish to tender through the DWAC system, please contact your broker and request delivery of your shares through the DWAC system. Delivering shares physically may take significantly longer. In order to obtain a physical certificate, a stockholder’s broker and/or clearing broker, DTC, and Continental will need to act together to facilitate this request. It is Globalink’s understanding that stockholders should generally allot at least two weeks to obtain physical certificates from Continental. Globalink does not have any control over this process or over the brokers or DTC, and it may take longer than two weeks to obtain a physical certificate. Stockholders who request physical certificates and wish to redeem may be unable to meet the deadline for tendering their shares before exercising their redemption rights and thus will be unable to redeem their shares.
In the event that a stockholder tenders its shares and decides prior to the consummation of the Business Combination that it does not want to redeem its shares, the stockholder may withdraw the tender. In the event that a stockholder tenders shares and the Business Combination is not completed, these shares will not be redeemed for cash and the physical certificates representing these shares will be returned to the stockholder promptly following the determination that the Business Combination will not be consummated. Globalink anticipates that a stockholder who tenders shares for redemption in connection with the vote to approve the Business Combination would receive payment of the redemption price for such shares soon after the completion of the Business Combination.
If properly demanded by Globalink’s public stockholders, Globalink will redeem each share of Globalink common stock into a pro rata portion of the funds available in the Trust Account, calculated as of two business days prior to the anticipated consummation of the Business Combination. As of December 6, 2024, this would amount to approximately $11.98 per share. If you exercise your redemption rights, you will be exchanging your shares of Globalink common stock for cash and will no longer own the shares.
Holders of outstanding units must separate the underlying shares of Globalink common stock, warrants and rights prior to exercising redemption rights with respect to the shares of Globalink common stock. If the units are registered in a holder’s own name, the holder must deliver the certificate for its units to Continental with written instructions to separate the units into their individual component parts. This must be completed far enough in advance to permit the mailing of the certificates back to the holder so that the holder may then exercise his, her or its redemption rights upon the separation of the shares of Globalink common stock from the units.
If a broker, dealer, commercial bank, trust company or other nominee holds the units for an individual or entity (such individual or entity, the “beneficial owner”), the beneficial owner must instruct such nominee to separate the beneficial owner’s units into their individual component parts. The beneficial owner’s nominee must send written instructions by facsimile to the transfer agent. Such written instructions must include the number of units to be separated and the nominee holding such units. The beneficial owner’s nominee must also initiate electronically, using DTC’s DWAC system, a withdrawal of the relevant units and a deposit of an equal number of shares of Globalink common stock, warrants and rights. This must be completed far enough in advance to permit the nominee to exercise the beneficial owner’s redemption rights upon the separation of the shares of Globalink common stock from the units. While this is typically done electronically the same business day, beneficial owners should allow at least one full business day to accomplish the separation. If beneficial owners fail to cause their shares of Globalink common stock to be separated in a timely manner, they will likely not be able to exercise their redemption rights.
| 139 |
Notwithstanding the foregoing, a holder of the shares of Globalink common stock, together with any affiliate of his or her or any other person with whom he or she is acting in concert or as a “group” (as defined in Section 13(d)-(3) of the Exchange Act) will be restricted from seeking redemption rights with respect to more than 20% of the issued and outstanding shares of Globalink common stock.
If too many public stockholders exercise their redemption rights, we may not be able to meet certain closing conditions, and as a result, would not be able to proceed with the Business Combination.
Vote Required for Approval
Along with the approval of the Nasdaq Proposals, and the Adjournment Proposal, the approval of this Acquisition Merger Proposal is a condition to the consummation of the Business Combination. If this Acquisition Merger Proposal is not approved, the Business Combination will not take place. Approval of this Acquisition Merger Proposal is also a condition to the Redomestication Merger Proposal, the Nasdaq Proposals. If the Redomestication Merger Proposal is not approved, this Acquisition Merger Proposal will have no effect and the Business Combination will not occur.
This Acquisition Merger Proposal (and consequently, the Merger Agreement and the transactions contemplated thereby, including the Business Combination) will be approved and adopted only if holders of at least a majority of the shares of common stock present in person by virtual attendance or represented by proxy and entitled to vote at the Special Meeting vote “FOR” the Acquisition Merger Proposal.
Pursuant to the Insider Letter and the Globalink Support Agreement, the Initial Stockholders and PGM holding an aggregate of 3,445,000 shares (or approximately 92.6% of the outstanding shares) of common stock have agreed to vote their respective shares of common stock (including shares included in the private units) in favor of each of the Proposals. The approval of the Acquisition Merger Proposal does not require the votes from at least a majority of unaffiliated security holders of Globalink. As a result, no shares of common stock held by the public stockholders will need to be present in person by virtual attendance or by proxy to satisfy the quorum requirement for the Special Meeting. In addition, as the vote to approve the Acquisition Merger Proposal is a majority of the votes cast at a meeting at which a quorum is present, assuming only the minimum number of shares to constitute a quorum is present, no stockholder must vote in favor of the Acquisition Merger Proposal for it to be approved.
Board Recommendation
The Board has unanimously determined that the Business Combination is in the best interests of, and advisable to, the Globalink stockholders and unanimously recommends that the Globalink stockholders adopt the Merger Agreement and approve the Business Combination. The Board made its determination after consultation with its legal and financial advisors and consideration of a number of factors.
THE BOARD UNANIMOUSLY RECOMMENDS THAT GLOBALINK STOCKHOLDERS VOTE “FOR” THE BUSINESS COMBINATION.
| 140 |
COMPARISON OF CORPORATE GOVERNANCE AND STOCKHOLDERS’/SHAREHOLDERS’ RIGHTS
General
Globalink is incorporated under the laws of the State of Delaware and the rights of Globalink stockholders are governed by the laws of the State of Delaware, including the DGCL, Globalink Charter and Globalink’s current bylaws. As a result of the Business Combination, the rights of Combined Company shareholders are governed by the laws of the Cayman Islands, including the Companies Act, and the PubCo A&R Memorandum and Articles.
Set forth below is a summary comparison of material differences between the rights of Globalink stockholders under Globalink Charter and Globalink’s current bylaws, the rights of the shareholders of Alps Life Sciences Inc (“Target”) and the rights of Combined Company’s shareholders under the forms of the PubCo A&R Memorandum and Articles. The summary set forth below is not intended to be complete or to provide a comprehensive discussion of each of Globalink’s, Target’s or Combined Company’s governing documents. This summary is qualified in its entirety by reference to the full text of Globalink Charter and Globalink’s current bylaws, the Target’s memorandum and articles of association and the form of the PubCo A&R Memorandum and Articles, which is attached as Annex B to the registration statement of which this proxy statement/prospectus forms a part, as well as the relevant provisions of the Companies Act.
| Provision | Globalink | Target | The Combined Company | |||
| Title of Organizing Documents | Certificate of Incorporation and Amended and Restated Bylaws | Memorandum and Articles of Association
|
Amended and Restated Memorandum and Articles of Association
| |||
| Authorized Capital Stock | Under the Globalink Charter, the authorized capital stock of Globalink consists of 505,000,000 shares, consisting of 500,000,000 ordinary shares, par value $0.001 per share and 5,000,000 shares of preferred stock, par value $0.001 per share. | The authorized share capital of Target is US$5,000 divided into 500,000,000 ordinary shares of US$0.00001 par value each. | The authorized share capital of the Combined Company will be US$50,000 divided into 500,000,000 shares comprising (i) 495,000,000 ordinary shares of US$0.0001 par value each, and (ii) 5,000,000 preferred shares of US$0.0001 par value each. | |||
| Number of Directors | Under the Current Bylaws, the Board shall consist of one or more members. The number of directors shall be fixed by the Board and may thereafter be changed from time to time by resolution of the Board. | The board of directors of the Target shall be a minimum of one person and a maximum of ten persons. | The board of directors of the Combined Company shall consist of not less than one person. Unless otherwise fixed by ordinary resolution of the shareholders, the maximum number of directors shall be unlimited. | |||
| Classification and Election of the Board of Directors | Under the Globalink Charter, the Globalink board of directors is divided into three classes, Class I, Class II and Class III. The Current Bylaws provide that only one class of directors will be elected in each year and each class will serve a three-year term. Class II directors will initially serve until the second annual meeting of stockholders; Class III directors will initially serve until the third annual meeting of stockholders; and Class I directors will initially serve until the fourth annual meeting of stockholders. Directors elected to succeed those directors whose terms expire shall be elected for a term of office to expire at the third succeeding annual meeting of stockholders after their election in each case, or until such director’s earlier death, disqualification, resignation, removal or death. | There is only one class of directors.
A director may be appointed by ordinary resolution or by the directors. Any appointment may be to fill a vacancy or as an additional director.
A director’s office shall be terminated forthwith if:
|
The PubCo directors shall be divided into two classes: Class A and Class B. An executive director shall be a Class A director and an independent director shall be a Class B director. Any Director that is not an independent director but is non-executive shall also be a Class B director. Upon the adoption of the PubCo A&R Memorandum and Articles, the existing directors shall by resolution classify themselves as Class A or Class B as applicable.
Subject always to the applicable law and the rules of the applicable stock exchange, the Class A directors shall hold office until such director resigns, is removed from office, or otherwise vacates the office. There shall be no requirement for any Class A directors to retire or be re-elected at any annual general meeting of the company or upon any specified event. A Class A director elected to fill a vacancy resulting from the death, resignation or removal of a director shall serve for the remainder of the full term of the director whose death, resignation or removal shall have created such vacancy and until his successor shall have been elected and qualified.
An appointment of a Class B director may be on terms that the director shall automatically retire from office (unless he has sooner vacated office) at the next or a subsequent annual general meeting or upon any specified event or after any specified period in a written agreement between the Company and the Class B director, if any; but no such term shall be implied in the absence of express provision. Each Class B director whose term of office expires shall be eligible for re-election at a meeting of the shareholders or re-appointment by the board. |
| 141 |
| Amendment to Organizing Documents | Under the Globalink Charter, the Globalink Charter may be amended by Globalink in the manner prescribed by the DGCL. Any such amendment requires a majority vote of the Globalink stockholders, voting as a class. | Pursuant to the Companies Act, Target may at any time and from time to time by special resolution (as defined by the Companies Act) change or amend its memorandum and articles of association in whole or in part. | Pursuant to the Companies Act, PubCo may at any time and from time to time by special resolution (as defined by the Companies Act) change or amend the PubCo A&R Memorandum and Articles in whole or in part. | |||
| Special Meeting of Members | The Current Bylaws provide that Special Meetings of stockholders may be called only by a majority vote of the Board, by the Chief Executive Officer or by the Chairman of the Board. | The directors may call a general meeting at any time. If there are insufficient directors to constitute a quorum and the remaining directors are unable to agree on the appointment of additional directors, any one or more members who together hold at least 10% of the rights to vote at a general meeting may call a general meeting for the purpose of considering the business specified in the notice of meeting which shall include as an item of business the appointment of additional directors. | A majority of all the directors or all of the Class A directors may call a general meeting at any time. | |||
| Member Action by Written Consent | The Globalink Charter is silent on the ability for Globalink stockholders to take actions by written consent. Accordingly, Globalink stockholders may take action by written consent in lieu of a formal meeting as prescribed by the DGCL. | Members of the Target are entitled to take action by written consent in lieu of a formal meeting upon meeting certain notice and document delivery conditions. | Members of the Combined Company will be entitled to take action by written consent in lieu of a formal meeting upon meeting certain notice and document delivery conditions. | |||
| Limitation of Liability of Directors and Officers | The Globalink Charter provides that only directors of Globalink are exculpated from liability for breach of duty of care in certain actions, except for liability for breach of duty of loyalty, acts or omissions not in good faith, or that involve intentional misconduct or a knowing violation of law, unlawful payment of dividend or unlawful stock purchase or redemption, or any transaction in which the director derived an improper personal benefit. | The Cayman Islands law does not limit the extent to which a company’s memorandum and articles of association may provide for indemnification of officers and directors, except to the extent any such provision may be held by the Cayman Islands courts to be contrary to public policy, such as to provide indemnification against the indemnified person’s own fraud or dishonesty or against the consequences of committing a crime. | The Cayman Islands law does not limit the extent to which a company’s memorandum and articles of association may provide for indemnification of officers and directors, except to the extent any such provision may be held by the Cayman Islands courts to be contrary to public policy, such as to provide indemnification against the indemnified person’s own fraud or dishonesty or against the consequences of committing a crime. |
| 142 |
| Indemnification of Directors, Officers, Employees and Agents | The Globalink Charter provides that all persons whom it may indemnify, to the full extent permitted by Section 145 of the DGCL, shall be indemnified by Globalink in a civil, criminal, administrative or investigative action or suit | The Cayman Islands law does not limit the extent to which a company’s memorandum and articles of association may provide for indemnification of officers and directors, except to the extent any such provision may be held by the Cayman Islands courts to be contrary to public policy, such as to provide indemnification against the indemnified person’s own fraud or dishonesty or against the consequences of committing a crime.
The Target’s memorandum and articles of association provide that, to the extent permitted by law, the Target shall indemnify each existing or former secretary, director (including alternate director), and other officer of the Target (including an investment adviser or an administrator or liquidator) and their personal representatives against:
(a) all actions, proceedings, costs, charges, expenses, losses, damages or liabilities incurred or sustained by the existing or former secretary or officer in or about the conduct of the Target’s business or affairs or in the execution or discharge of the existing or former secretary’s or officer’s duties, powers, authorities or discretions; and
(b) without limitation to paragraph (a), all costs, expenses, losses or liabilities incurred by the existing or former secretary or officer in defending (whether successfully or otherwise) any civil, criminal, administrative or investigative proceedings (whether threatened, pending or completed) concerning the Target or its affairs in any court or tribunal, whether in the Cayman Islands or elsewhere.
No such existing or former secretary or officer, however, shall be indemnified in respect of any matter arising out of his own dishonesty.
To the extent permitted by law, the Target may make a payment, or agree to make a payment, whether by way of advance, loan or otherwise, for any legal costs incurred by an existing or former secretary or officer of the Target in respect of any matter identified in paragraph (a) or paragraph (b) of the preceding article on condition that the secretary or officer must repay the amount paid by the Target to the extent that it is ultimately found not liable to indemnify the secretary or that officer for those legal costs. |
The Cayman Islands law does not limit the extent to which a company’s memorandum and articles of association may provide for indemnification of officers and directors, except to the extent any such provision may be held by the Cayman Islands courts to be contrary to public policy, such as to provide indemnification against the indemnified person’s own fraud or dishonesty or against the consequences of committing a crime.
The proposed PubCo A&R Memorandum and Articles provide that, to the extent permitted by law, the PubCo shall indemnify each existing or former secretary, director (including alternate director) and any other officers (including an investment adviser or an administrator or liquidator) and their personal representatives against:
(a) all actions, proceedings, costs, charges, expenses, losses, damages or liabilities incurred or sustained by the existing or former director (including alternate director), secretary or officer in or about the conduct of the Pubco’s business or affairs or in the execution or discharge of the existing or former director (including alternate director), secretary’s or officer’s duties, powers, authorities or discretions; and
(b) without limitation to (a) above, all costs, expenses, losses or liabilities incurred by the existing or former director (including alternate director), secretary or officer in defending (whether successfully or otherwise) any civil, criminal, administrative or investigative proceedings (whether threatened, pending or completed) concerning the PubCo or its affairs in any court or tribunal, whether in the Cayman Islands or elsewhere.
No such existing or former director (including alternate director), secretary or officer, however, shall be indemnified in respect of any matter arising out of his own dishonesty, willful default, willful neglect or fraud.
To the extent permitted by law, the PubCo may make a payment, or agree to make a payment, whether by way of advance, loan or otherwise, for any legal costs incurred by an existing or former director (including alternate director), secretary or officer in respect of any matter identified in above on condition that the director (including alternate director), secretary or officer must repay the amount paid by us to the extent that it is ultimately found not liable to indemnify the director (including alternate director), the secretary or that officer for those legal costs. |
| 143 |
| Liquidation / Winding Up | Globalink Charter provides that if Globalink is unable to complete an initial business combination by January 9, 2025 9, 2024 (and may be extended to June 9, 2025 on a monthly basis by resolutions of the Globalink Board, if requested by the Sponsor, and subject to the Sponsor depositing additional funds into the Trust Account), Globalink will (i) cease all operations except for the purpose of winding up, (ii) as promptly as reasonably possible but not more than ten business days thereafter, redeem 100% of the outstanding public shares for a pro rata portion of the funds held in the Trust Account, which redemption will completely extinguish Globalink stockholders’ rights as shareholders (including the right to receive further liquidation distributions, if any), subject to applicable law, and (iii) as promptly as reasonably possible following such redemption, subject to the approval of Globalink’s remaining holders of common stock and the Board, dissolve and liquidate, subject (in the case of (ii) and (iii) above) to Globalink’s obligations under Delaware law to provide for claims of creditors and the requirements of other applicable law. | If the Target is wound up, the shareholders may, subject to the articles and any other sanction required by the Companies Act, pass a special resolution allowing the liquidator to do either or both of the following: (a) to divide in specie among the shareholders the whole or any part of the assets of the Target and, for that purpose, to value any assets and to determine how the division shall be carried out as between the shareholders or different classes of shareholders; (b) to vest the whole or any part of the assets in trustees for the benefit of shareholders and those liable to contribute to the winding up. The directors have the authority to present a petition for our winding up to the Grand Court of the Cayman Islands on the Target’s behalf without the sanction of a resolution passed at a general meeting. |
If the PubCo is wound up, the shareholders may, subject to the articles and any other sanction required by the Companies Act, pass a special resolution allowing the liquidator to do either or both of the following: (i) to divide in specie among the shareholders the whole or any part of the assets of the PubCo and, for that purpose, to value any assets and to determine how the division shall be carried out as between the shareholders or different classes of shareholders; and (ii) to vest the whole or any part of the assets in trustees for the benefit of shareholders and those liable to contribute to the winding up. The directors have the authority to present a petition for our winding up to the Grand Court of the Cayman Islands on the Pubco’s behalf without the sanction of a resolution passed at a general meeting | |||
| Supermajority Voting Provisions | The Current Bylaws provides that the Current Bylaws may be altered, amended, supplemented or repealed or new bylaws may be adopted (a) at any regular or Special Meeting of stockholders at which a quorum is present or represented, by the affirmative vote of the holders of 66-2/3% of the shares entitled to vote, provided notice of the proposed alteration, amendment or repeal be contained in the notice of such meeting, or (b) by a resolution adopted by a majority of the whole Board any regular or Special Meeting of the Board. The Globalink stockholders shall have authority to change or repeal any bylaws adopted by the directors, subject to compliance with the provisions of the Current Bylaws. | For the protection of shareholders, certain matters must be approved by special resolution of the shareholders as a matter of Cayman Islands law, including alteration of the memorandum or articles of association, appointment of inspectors to examine company affairs, reduction of share capital (subject, in relevant circumstances, to court approval), change of name, authorization of a plan of merger or transfer by way of continuation to another jurisdiction or consolidation or voluntary winding up of the company.
The Companies Act requires that a special resolution be passed by a majority of at least two-thirds or such higher percentage as set forth in the memorandum and articles of association, of shareholders being entitled to vote and do vote in person or by proxy at a general meeting, or by unanimous written consent of shareholders entitled to vote at a general meeting.
The Companies Act defines “special resolutions” only. A company’s memorandum and articles of association can therefore tailor the definition of “ordinary resolutions” as a whole, or with respect to specific provisions. |
For the protection of shareholders, certain matters must be approved by special resolution of the shareholders as a matter of Cayman Islands law, including alteration of the memorandum or articles of association, appointment of inspectors to examine company affairs, reduction of share capital (subject, in relevant circumstances, to court approval), change of name, authorization of a plan of merger or transfer by way of continuation to another jurisdiction or consolidation or voluntary winding up of the company.
The Companies Act requires that a special resolution be passed by a majority of at least two-thirds or such higher percentage as set forth in the memorandum and articles of association, of shareholders being entitled to vote and do vote in person or by proxy at a general meeting, or by unanimous written consent of shareholders entitled to vote at a general meeting.
The Companies Act defines “special resolutions” only. A company’s memorandum and articles of association can therefore tailor the definition of “ordinary resolutions” as a whole, or with respect to specific provisions.
|
| 144 |
| Duties of directors | Under Delaware law, the business and affairs of a corporation are managed by or under the direction of its board of directors. In exercising their powers, directors are charged with a fiduciary duty of care to protect the interests of the corporation and a fiduciary duty of loyalty to act in the best interests of its stockholders. The duty of care requires that directors act in an informed and deliberative manner and inform themselves, prior to making a business decision, of all material information reasonably available to them. The duty of care also requires that directors exercise care in overseeing and investigating the conduct of the corporation’s employees. The duty of loyalty may be summarized as the duty to act in good faith, not out of self-interest, and in a manner which the director reasonably believes to be in the best interests of the stockholders. | As a matter of Cayman Islands law, a director owes three types of duties to the company: (i) statutory duties, (ii) fiduciary duties, and (iii) common law duties. The Companies Act imposes a number of statutory duties on a director. A Cayman Islands director’s fiduciary duties are not codified, however the courts of the Cayman Islands have held that a director owes the following fiduciary duties (a) a duty to act in what the director bona fide considers to be in the best interests of the company, (b) a duty to exercise their powers for the purposes they were conferred, (c) a duty to avoid fettering his or her discretion in the future and (d) a duty to avoid conflicts of interest and of duty. The common law duties owed by a director are those to act with skill, care and diligence that may reasonably be expected of a person carrying out the same functions as are carried out by that director in relation to the company and, also, to act with the skill, care and diligence in keeping with a standard of care commensurate with any particular skill they have which enables them to meet a higher standard than a director without those skills. In fulfilling their duty of care to us, our directors must ensure compliance with the Target’s articles of association, as amended and restated from time to time. The Target has the right to seek damages where certain duties owed by any directors are breached. | As a matter of Cayman Islands law, a director owes three types of duties to the company: (i) statutory duties, (ii) fiduciary duties, and (iii) common law duties. The Companies Act imposes a number of statutory duties on a director. A Cayman Islands director’s fiduciary duties are not codified, however the courts of the Cayman Islands have held that a director owes the following fiduciary duties (a) a duty to act in what the director bona fide considers to be in the best interests of the company, (b) a duty to exercise their powers for the purposes they were conferred, (c) a duty to avoid fettering his or her discretion in the future and (d) a duty to avoid conflicts of interest and of duty. The common law duties owed by a director are those to act with skill, care and diligence that may reasonably be expected of a person carrying out the same functions as are carried out by that director in relation to the company and, also, to act with the skill, care and diligence in keeping with a standard of care commensurate with any particular skill they have which enables them to meet a higher standard than a director without those skills. In fulfilling their duty of care to us, our directors must ensure compliance with the PubCo’s amended articles of association, as amended and restated from time to time. The PubCo has the right to seek damages where certain duties owed by any directors are breached. | |||
| Interested Directors | Under Delaware law, a transaction in which a director who has an interest in such transaction would not be voidable if (i) the material facts as to such interested director’s relationship or interests are disclosed or are known to the board of directors and the board in good faith authorizes the transaction by the affirmative vote of a majority of the disinterested directors, even though the disinterested directors are less than a quorum, (ii) such material facts are disclosed or are known to the shareholders entitled to vote on such transaction and the transaction is specifically approved in good faith by vote of the shareholders, or (iii) the transaction is fair as to the corporation as of the time it is authorized, approved or ratified. Under Delaware law, a director could be held liable for any transaction in which such director derived an improper personal benefit. | Except
as expressly permitted in the Target’s articles of association, a director may not
have a direct or indirect interest or duty which conflicts or may possibly conflict with
the interests of the Target. If, notwithstanding the foregoing prohibition, a director discloses
to his fellow directors the nature and extent of any material interest or duty in accordance
with the notification provisions set forth in the articles of association, he may: (a) be
a party to, or otherwise interested in, any transaction or arrangement with the Target or
in which the Target is or may otherwise be interested; (b) be interested in another body
corporate promoted by the Target or in which the Target is otherwise interested. In particular,
the director may be a director, secretary or officer of, or employed by, or be a party to
any transaction or arrangement with, or otherwise interested in, that other body corporate.
A director may vote at a meeting of directors on any resolution concerning a matter in which that director has an interest or duty, whether directly or indirectly, so long as that director discloses any material interest pursuant to the Articles. The director shall be counted towards a quorum of those present at the meeting. If the director votes on the resolution, his vote shall be counted. |
Interested director transactions are governed by the terms of a company’s memorandum and articles of association.
Pursuant to PubCo A&R Memorandum and Articles, a director who is in any way, whether directly or indirectly, interested in a contract or transaction or proposed contract or transaction with the company shall declare the nature of his interest at a meeting of the directors. A general notice given to the directors by any director to the effect that he is a member of any specified company or firm and is to be regarded as interested in any contract or transaction which may thereafter be made with that company or firm shall be deemed a sufficient declaration of interest in regard to any contract so made or transaction so consummated. Subject to the rules of any applicable stock exchange and disqualification by the chairman of the relevant board meeting, a director may vote in respect of any contract or transaction or proposed contract or transaction notwithstanding that he may be interested therein and if he does so his vote shall be counted and he may be counted in the quorum at any meeting of the directors at which any such contract or transaction or proposed contract or transaction shall come before the meeting for consideration.
|
| 145 |
| Voting for Directors | Under Delaware law, unless otherwise specified in the certificate of incorporation or bylaws of the corporation, directors shall be elected by a plurality of the votes of the shares present in person or represented by proxy at the meeting and entitled to vote on the election of directors.
|
Pursuant to Target’s memorandum and articles of association, directors may be appointed by way of ordinary resolutions or by the directors.
|
Pursuant to PubCo A&R Memorandum and Articles, directors may be appointed by way of ordinary resolutions or by the directors. | |||
| Director Removal | Pursuant to the Globalink Charter and the Current Bylaws, directors may be removed with cause by the affirmative vote of holders of 66-2/3% of the voting power of all then outstanding shares entitled to vote generally in the election of directors, voting as a single class.
|
Directors may be removed by way of ordinary resolutions. | Directors may be removed by way of ordinary resolutions. | |||
| Cumulative Voting | No cumulative voting for the election of directors unless so provided in the certificate of incorporation. | There are no prohibitions in relation to cumulative voting under the Companies Act but the Target’s memorandum and articles of association do not provide for cumulative voting.
|
There are no prohibitions in relation to cumulative voting under the Companies Act but the PubCo A&R Memorandum and Articles do not provide for cumulative voting.
| |||
| Directors’ Powers Regarding Bylaws | The Globalink Charter grants the directors the power to adopt, amend or repeal bylaws without seeking a vote of the stockholders. | The memorandum and articles of association of Target may only be amended, in whole or in part, by a special resolution of the shareholders.
|
The memorandum and articles of association of PubCo may only be amended, in whole or in part, by a special resolution of the shareholders.
| |||
| Forum Selection | Subject to certain conditions, the Globalink Charter provides that the Delaware Court of Chancery shall serve as the sole and exclusive forum for derivative actions brought on behalf of Globalink, breach of fiduciary claims asserted against current or former officers, directors, employees or agents, claims arising out of the DGCL, Globalink Charter or Current Bylaws, or other actions against Globalink, or current and former directors, officers, employees governed by the internal affairs doctrine. | There is no forum selection provision. | There is no forum selection provision. |
| 146 |
THE NASDAQ PROPOSALS (PROPOSALS 3)
Overview
Assuming the Redomestication Merger Proposal and the Acquisition Merger Proposal are approved, Globalink stockholders are also being asked to approve separate proposals approving the issuance of:
| (A) | up to 208,000,000 PubCo ordinary shares pursuant to the Merger Agreement (including the ordinary shares issuable pursuant to that certain earnout provisions in the Merger Agreement), and | |
| (B) | up to 4,020,000 PubCo ordinary shares to the PIPE Investors. |
Why Globalink Needs Stockholder Approval
Globalink is seeking stockholder approval for the Nasdaq Proposals in order to comply with Nasdaq Listing Rule 5635.
Under Nasdaq Listing Rule 5635(a), stockholder approval is required prior to the issuance of securities in connection with the acquisition of the stock or assets of another company if, due to the present or potential issuance of shares of common stock, including shares issued pursuant to an earnout provision or similar type of provision, or securities convertible into or exercisable for common stock, other than a public offering for cash, (a) the common stock has or will have upon issuance voting power equal to or in excess of 20% of the voting power outstanding before the issuance of stock or securities convertible into or exercisable for common stock; or (b) the number of shares of common stock to be issued is or will be equal to or in excess of 20% of the number of shares of common stock outstanding before the issuance of the stock or securities.
Under Nasdaq Listing Rule 5635(b), stockholder approval is required when any issuance or potential issuance will result in a “change of control” of the issuer. Although Nasdaq has not adopted any rule on what constitutes a “change of control” for purposes of Rule 5635(b), Nasdaq has previously indicated that the acquisition of, or right to acquire, by a single investor or affiliated investor group, as little as 20% of the common stock (or securities convertible into or exercisable for common stock) or voting power of an issuer could constitute a change of control.
Currently, 3,722,511 shares of Globalink common stock are outstanding. Globalink anticipates that (i) the number of PubCo ordinary shares to be issued to Alps Holdco Shareholders pursuant to the Merger Agreement, and (ii) the number of ordinary shares of the Combined Company issuable to the PIPE Investors in the PIPE Investment will each be in excess of 20% of the number of shares of Globalink common stock outstanding before the Closing. We expect the issuances will result in a change in control of Globalink. Accordingly, Globalink is required to obtain stockholder approval of such issuances pursuant to Nasdaq Listing Rule 5635(a), (b) and (d).
Effect of the Proposals on Current Stockholders
The issuance of PubCo ordinary shares to Alps Holdco Shareholders pursuant to the Merger Agreement, and to the PIPE Investors pursuant to the PIPE Investment would result in further dilution to Globalink’s current stockholders in connection with the consummation of the Business Combination and would result in Globalink’s current stockholders holding a smaller percentage interest in the voting power, liquidation value and aggregate book value of the Combined Company. In addition, the resale of the PubCo ordinary shares to be issued to Alps Holdco Shareholders and the PIPE Investors could cause the market price of the PubCo ordinary shares to decline. However, the issuance of these shares is contemplated by the Merger Agreement and the Subscription Agreements. Accordingly, if the Nasdaq Proposals are not approved, Globalink cannot consummate the Business Combination.
Vote Required for Approval
The approval of each of the Nasdaq Proposals requires the affirmative vote of a majority of the votes cast by the Globalink stockholders present by virtual attendance or represented by proxy and entitled to vote thereon at the Special Meeting, assuming that a quorum is present. Abstentions and broker non-votes will have no effect on the approval of these Proposals.
The Nasdaq Proposals will not be submitted if either the Redomestication Merger Proposal or the Acquisition Merger Proposal is not approved. Approval of the Nasdaq Proposals may be necessary to enable Globalink to meet its obligations under the Merger Agreement.
Board Recommendation of the Board
THE BOARD UNANIMOUSLY RECOMMENDS THAT GLOBALINK STOCKHOLDERS VOTE “FOR” THE APPROVAL OF EACH OF THE NASDAQ PROPOSALS.
| 147 |
THE ADJOURNMENT PROPOSAL (PROPOSAL 4)
The Adjournment Proposal, if adopted, will approve the chairman’s adjournment of the Special Meeting to a later date to permit further solicitation of proxies. The Adjournment Proposal will only be presented to Globalink stockholders in the event, based on the tabulated votes, there are not sufficient votes received at the time of the Special Meeting to approve the other Proposals.
Consequences if the Adjournment Proposal is Not Approved
If the Adjournment Proposal is not approved by Globalink stockholders, the chairman will not adjourn the Special Meeting to a later date in the event, based on the tabulated votes, there are not sufficient votes received at the time of the Special Meeting to approve the Redomestication Merger Proposal, the Acquisition Merger Proposal, or the Nasdaq Proposals.
Required Vote
This Adjournment Proposal will be approved and adopted only if holders of at least a majority of the issued and outstanding shares of common stock present in person by virtual attendance or represented by proxy and entitled to vote at the Special Meeting vote “FOR” the Adjournment Proposal. The Adjournment Proposal is not conditioned on the approval of any other Proposal set forth in this proxy statement/prospectus.
Board Recommendation
THE BOARD UNANIMOUSLY RECOMMENDS A VOTE “FOR” THE ADJOURNMENT PROPOSAL.
| 148 |
MATERIAL U.S. FEDERAL INCOME TAX CONSEQUENCES
The following is a brief discussion of the material U.S. federal income tax consequences (i) of the exercise of redemption rights by U.S. Holders and Non-U.S. Holders (defined below) of Globalink common stock, (ii) of the Business Combination to U.S. Holders of Globalink common stock, (iii) Redomestication Merger, (iv) the Business Combination to Globalink and PubCo, (v) of ownership and disposition of PubCo securities, and (vi) of PubCo’s passive foreign investment company status. Subject to the limitations and qualifications set forth herein, the discussion of (a) the exercise of redemption rights by U.S. Holders and Non-U.S. Holders of Globalink common stock, (b) the tax consequences of the Business Combination to U.S. Holders of Globalink common stock, and (c) tax consequences of the Business Combination to Globalink is the opinion of Hunter Taubman Fischer & Li LLC.
This discussion is based on provisions of the Internal Revenue Code of 1986, as amended (the “Code”), the Treasury Regulations promulgated thereunder (whether final, temporary, or proposed), administrative rulings of the IRS, and judicial decisions, all as in effect on the date hereof, and all of which are subject to differing interpretations or change, possibly with retroactive effect. This discussion does not purport to be a complete analysis or listing of all potential U.S. federal income tax consequences that may apply to a holder as a result of the Business Combination. In addition, this discussion does not address all aspects of U.S. federal income taxation that may be relevant to particular holders nor does it take into account the individual facts and circumstances of any particular holder that may affect the U.S. federal income tax consequences to such holder, and accordingly, is not intended to be, and should not be construed as tax advice. This discussion does not address the U.S. federal 3.8% Medicare tax imposed on certain net investment income or any aspects of U.S. federal taxation other than those pertaining to the income tax, nor does it address any tax consequences arising under any U.S. state and local, or non-U.S. tax laws. Holders should consult their own tax advisors regarding such tax consequences in light of their particular circumstances.
No ruling has been requested or will be obtained from the IRS regarding the U.S. federal income tax consequences of the Business Combination or any other related matter; thus, there can be no assurance that the IRS will not challenge the U.S. federal income tax treatment described below or that, if challenged, such treatment will be sustained by a court.
U.S. Federal Income Tax Consequences to U.S. Holders of Alps Life Sciences Inc Excluded
Alps Holdco does not have any U.S. holders. Given that Alps Holdco is a non-U.S. entity with no U.S. holders, Alps Holdco does not foresee any material U.S. federal income tax consequences of the Business Combination (including the Redomestication Merger and the Acquisition Merger) to its pre-merger shareholders. Discussions on the “U.S. Federal Income Tax Consequences of the Business Combination to U.S. Holders of Alps Life Sciences Inc’s Shares” are excluded they are not relevant to Alps Holdco as shareholders of Alps Holdco are not subject to U.S. federal income taxes.
This summary is limited to considerations relevant to holders that hold common stock or PubCo ordinary shares as “capital assets” within the meaning of section 1221 of the Code (generally, property held for investment). This discussion does not address all aspects of U.S. federal income taxation that may be important to holders in light of their individual circumstances, including holders subject to special treatment under the U.S. tax laws, such as, for example:
| ● | banks or other financial institutions, underwriters, or insurance companies; | |
| ● | traders in securities who elect to apply a mark-to-market method of accounting; | |
| ● | real estate investment trusts and regulated investment companies; | |
| ● | tax-exempt organizations, qualified retirement plans, individual retirement accounts, or other tax-deferred accounts; | |
| ● | expatriates or former long-term residents of the United States; | |
| ● | subchapter S corporations, partnerships or other pass-through entities or investors in such entities; | |
| 149 |
| ● | dealers or traders in securities, commodities or currencies; | |
| ● | grantor trusts; | |
| ● | persons subject to the alternative minimum tax; | |
| ● | U.S. persons whose “functional currency” is not the U.S. dollar; | |
| ● | persons who received shares of Globalink common stock or PubCo ordinary shares through the issuance of restricted stock under an equity incentive plan or through a tax-qualified retirement plan or otherwise as compensation; | |
| ● | persons who own (directly or through attribution) 5% or more (by vote or value) of the outstanding shares of Globalink common stock or PubCo ordinary shares (excluding treasury shares); | |
| ● | holders holding Globalink common stock or PubCo ordinary shares as a position in a “straddle,” as part of a “synthetic security” or “hedge,” as part of a “conversion transaction,” or other integrated investment or risk reduction transaction; | |
| ● | controlled foreign corporations, passive foreign investment companies, or foreign corporations with respect to which there are one or more United States stockholders within the meaning of Treasury Regulation Section 1.367(b)-3(b)(1)(ii); or | |
| ● | the Sponsor or its affiliates. |
As used in this proxy statement/prospectus, the term “U.S. Holder” means a beneficial owner of Globalink common stock or PubCo ordinary shares that is, for U.S. federal income tax purposes:
| ● | an individual who is a citizen or resident of the United States; | |
| ● | a corporation (or other entity that is classified as a corporation for U.S. federal income tax purposes) that is created or organized in or under the laws of the United States or any State thereof or the District of Columbia; | |
| ● | an estate the income of which is subject to U.S. federal income tax regardless of its source; or | |
| ● | a trust (i) if a court within the United States is able to exercise primary supervision over the administration of the trust and one or more U.S. persons have the authority to control all substantial decisions of the trust, or (ii) that has a valid election in effect under applicable Treasury Regulations to be treated as a U.S. person for U.S. federal income tax purposes. |
A “Non-U.S. Holder” means a beneficial owner of Globalink common stock or PubCo ordinary shares that is neither a U.S. Holder nor a partnership (or an entity or arrangement treated as a partnership) for U.S. federal income tax purposes.
If a partnership, including for this purpose any entity or arrangement that is treated as a partnership for U.S. federal income tax purposes, holds Globalink common stock or PubCo ordinary shares, the U.S. federal income tax treatment of a partner in such partnership will generally depend on the status of the partner and the activities of the partnership. A holder that is a partnership and the partners in such partnership should consult their own tax advisors with regard to the U.S. federal income tax consequences of the Business Combination.
THIS SUMMARY DOES NOT PURPORT TO BE A COMPREHENSIVE ANALYSIS OR DESCRIPTION OF ALL POTENTIAL U.S. FEDERAL INCOME TAX CONSEQUENCES OF THE BUSINESS COMBINATION. IN ADDITION, THE U.S. FEDERAL INCOME TAX TREATMENT OF THE BENEFICIAL OWNERS OF SHARES OF GLOBALINK COMMON STOCK OR PUBCO ORDINARY SHARES MAY BE AFFECTED BY MATTERS NOT DISCUSSED HEREIN AND DEPENDS IN SOME INSTANCES ON DETERMINATIONS OF FACT AND INTERPRETATIONS OF COMPLEX PROVISIONS OF U.S. FEDERAL INCOME TAX LAW FOR WHICH NO CLEAR PRECEDENT OR AUTHORITY MAY BE AVAILABLE. HOLDERS OF PUBCO ORDINARY SHARES SHOULD CONSULT WITH THEIR TAX ADVISORS REGARDING THE PARTICULAR TAX CONSEQUENCES TO THEM OF THE BUSINESS COMBINATION, INCLUDING THE APPLICABILITY AND EFFECTS OF U.S. FEDERAL, STATE, LOCAL, AND OTHER TAX LAWS.
| 150 |
Certain Material U.S. Federal Income Tax Consequences of Exercising Redemption Rights
Subject to the limitations and qualifications set forth herein, the following discussion of certain material U.S. federal income tax consequences of exercising redemption rights is pursuant to the opinion of Hunter Taubman Fischer & Li LLC.
U.S. Federal Income Tax Consequences of Exercising Redemption Rights to U.S. Holders
In the event that a U.S. Holder elects to redeem its shares of Globalink common stock for cash, the treatment of the transaction for U.S. federal income tax purposes will depend on whether the redemption qualifies as a sale or exchange of the shares of Globalink common stock under Section 302 of the Code or is treated as a corporate distribution under Section 301 of the Code with respect to the U.S. Holder. If the redemption qualifies as a sale or exchange of the shares of Globalink common stock, the U.S. Holder will be treated as recognizing capital gain or loss equal to the difference between the amount realized on the redemption and such U.S. Holder’s adjusted tax basis in the shares of Globalink common stock surrendered in such redemption transaction. Any such capital gain or loss generally will be long-term capital gain or loss if the U.S. Holder’s holding period for the shares of Globalink common stock redeemed exceeds one year. It is unclear, however, whether the redemption rights with respect to the shares of Globalink common stock may suspend the running of the applicable holding period for this purpose. Long term capital gain realized by a non-corporate U.S. Holder is currently taxed at a reduced rate. The deductibility of capital losses is subject to limitations.
If the redemption does not qualify as a sale or exchange of Globalink common stock, the U.S. Holder will be treated as receiving a corporate distribution. Such distributions generally will constitute dividends for U.S. federal income tax purposes to the extent paid from Globalink’s current or accumulated earnings and profits, as determined under U.S. federal income tax principles. Distributions in excess of current and accumulated earnings and profits will constitute a return of capital that will be applied against and reduce (but not below zero) the U.S. Holder’s adjusted tax basis in the Globalink common stock. Any remaining excess will be treated as gain realized on the sale or other disposition of the common stock. Dividends paid to a U.S. Holder that is a taxable corporation generally will qualify for the dividends received deduction if the requisite holding period is satisfied. With certain exceptions (including, but not limited to, dividends treated as investment income for purposes of investment interest deduction limitations) and provided certain holding period requirements are met, dividends paid to a non-corporate U.S. Holder generally will constitute “qualified dividends” that will be subject to tax at the maximum tax rate accorded to long-term capital gains. However, it is unclear whether the redemption rights with respect to the shares of Globalink common stock may prevent a U.S. Holder from satisfying the applicable holding period requirements with respect to the dividends received deduction or the preferential tax rate on qualified dividend income, as the case may be.
Whether a redemption qualifies for sale or exchange treatment will depend largely on the total number of shares of Globalink common stock treated as held by the U.S. Holder (including any shares of Globalink common stock constructively owned by the U.S. Holder as a result of owning warrants) relative to all of the shares of Globalink common stock outstanding both before and after the redemption. The redemption of shares of Globalink common stock generally will be treated as a sale or exchange of the shares of Globalink common stock (rather than as a corporate distribution) if the redemption (i) is “substantially disproportionate” with respect to the U.S. Holder, (ii) results in a “complete termination” of the U.S. Holder’s interest in Globalink or (iii) is “not essentially equivalent to a dividend” with respect to the U.S. Holder. These tests are explained more fully below.
| 151 |
In determining whether any of the foregoing tests are satisfied, a U.S. Holder takes into account not only shares of Globalink common stock actually owned by the U.S. Holder, but also shares of Globalink common stock that are constructively owned by the U.S. Holder. A U.S. Holder may constructively own, in addition to shares of stock owned directly, shares of stock owned by certain related individuals and entities in which the U.S. Holder has an interest or that have an interest in such U.S. Holder, as well as any stock the U.S. Holder has a right to acquire by exercise of an option, which would generally include shares of Globalink common stock which could be acquired pursuant to the exercise of warrants. In order to meet the substantially disproportionate test, (i) the percentage of Globalink’s outstanding shares of voting stock actually and constructively owned by the U.S. Holder immediately following the redemption of Globalink common stock must be less than 80% of the percentage of its outstanding shares of voting stock actually and constructively owned by the U.S. Holder immediately before the redemption, (ii) the U.S. Holder’s percentage ownership (including constructive ownership) of Globalink’s outstanding shares of stock (both voting and nonvoting) immediately after the redemption must be less than 80% of such percentage ownership (including constructive ownership) immediately before the redemption; and (iii) the U.S. Holder must own (including constructive ownership), immediately after the redemption, less than 50% of the total combined voting power of all classes of Globalink’s shares of stock entitled to vote. There will be a complete termination of a U.S. Holder’s interest if either (i) all of the shares of the Globalink common stock actually and constructively owned by the U.S. Holder are redeemed or (ii) all of the shares of the Globalink common stock actually owned by the U.S. Holder are redeemed and the U.S. Holder is eligible to waive, and effectively waives in accordance with specific rules, the attribution of stock owned by certain family members and the U.S. Holder does not constructively own any other shares of Globalink common stock. The redemption of the shares of Globalink common stock will not be essentially equivalent to a dividend if a U.S. Holder’s redemption results in a “meaningful reduction” of the U.S. Holder’s proportionate interest in Globalink. Whether the redemption will result in a meaningful reduction in a U.S. Holder’s proportionate interest in Globalink will depend on the particular facts and circumstances. However, the IRS has indicated in a published ruling that even a small reduction in the proportionate interest of a small minority stockholder in a publicly held corporation who exercises no control over corporate affairs may constitute such a “meaningful reduction.” A U.S. Holder should consult with its own tax advisors as to the tax consequences of a redemption.
If none of the foregoing tests are satisfied, then the redemption will be treated as a corporate distribution. After the application of those rules regarding corporate distributions, any remaining tax basis of the U.S. Holder in the redeemed shares of Globalink common stock will be added to the U.S. Holder’s adjusted tax basis in its remaining shares of Globalink common stock, or, if it has none, to the U.S. Holder’s adjusted tax basis in its warrants or possibly in other shares of Globalink common stock constructively owned by it.
U.S. Federal Income Tax Consequences to Non-U.S. Holders
The characterization for U.S. federal income tax purposes of the redemption of a Non-U.S. Holder’s Globalink common stock as a sale or exchange under Section 302 of the Code or as a corporate distribution under Section 301 of the Code generally will correspond to the U.S. federal income tax characterization of such a redemption of a U.S. Holder’s shares of Globalink common stock, as described above, and the corresponding consequences will be as described below.
Redemption Treated as Sale or Exchange
Any gain realized by a Non-U.S. Holder on the redemption of shares of Globalink common stock that is treated as a sale or exchange under Section 302 of the Code generally will not be subject to U.S. federal income tax unless:
| ● | the gain is effectively connected with a trade or business of the Non-U.S. Holder in the United States (and, if required by an applicable income tax treaty, is attributable to a United States permanent establishment or fixed base of the Non-U.S. Holder); | |
| ● | the Non-U.S. Holder is an individual who is present in the United States for a period or periods aggregating 183 days or more in the taxable year of the disposition, and certain other conditions are met; or | |
| ● | Globalink is or has been a “United States real property holding corporation” for U.S. federal income tax purposes at any time during the shorter of the five-year period ending on the date of disposition or the Non-U.S. Holder’s holding period for such shares of Globalink common stock redeemed, and either (A) shares of Globalink common stock are not considered to be regularly traded on an established securities market or (B) such Non-U.S. Holder has owned or is deemed to have owned, at any time during the shorter of the five-year period preceding such disposition and such Non-U.S. Holder’s holding period more than 5% of the outstanding shares of Globalink common stock. There can be no assurance that shares of Globalink common stock will be treated as regularly traded on an established securities market for this purpose. |
| 152 |
A “Non-U.S. Holder” means a beneficial owner of Globalink common stock or PubCo ordinary shares that is neither a U.S. Holder nor a partnership (or an entity or arrangement treated as a partnership) for U.S. federal income tax purposes.
If a partnership, including for this purpose any entity or arrangement that is treated as a partnership for U.S. federal income tax purposes, holds Globalink common stock or PubCo ordinary shares, the U.S. federal income tax treatment of a partner in such partnership will generally depend on the status of the partner and the activities of the partnership. A holder that is a partnership and the partners in such partnership should consult their own tax advisors with regard to the U.S. federal income tax consequences of the Business Combination.
THIS SUMMARY DOES NOT PURPORT TO BE A COMPREHENSIVE ANALYSIS OR DESCRIPTION OF ALL POTENTIAL U.S. FEDERAL INCOME TAX CONSEQUENCES OF THE BUSINESS COMBINATION. IN ADDITION, THE U.S. FEDERAL INCOME TAX TREATMENT OF THE BENEFICIAL OWNERS OF SHARES OF GLOBALINK COMMON STOCK OR PUBCO ORDINARY SHARES MAY BE AFFECTED BY MATTERS NOT DISCUSSED HEREIN AND DEPENDS IN SOME INSTANCES ON DETERMINATIONS OF FACT AND INTERPRETATIONS OF COMPLEX PROVISIONS OF U.S. FEDERAL INCOME TAX LAW FOR WHICH NO CLEAR PRECEDENT OR AUTHORITY MAY BE AVAILABLE. HOLDERS OF PUBCO ORDINARY SHARES SHOULD CONSULT WITH THEIR TAX ADVISORS REGARDING THE PARTICULAR TAX CONSEQUENCES TO THEM OF THE BUSINESS COMBINATION, INCLUDING THE APPLICABILITY AND EFFECTS OF U.S. FEDERAL, STATE, LOCAL, AND OTHER TAX LAWS.
Certain Material U.S. Federal Income Tax Consequences of Exercising Redemption Rights
Subject to the limitations and qualifications set forth herein, the following discussion of certain material U.S. federal income tax consequences of exercising redemption rights is pursuant to the opinion of Hunter Taubman Fischer & Li LLC.
U.S. Federal Income Tax Consequences to U.S. Holders
In the event that a U.S. Holder elects to redeem its shares of Globalink common stock for cash, the treatment of the transaction for U.S. federal income tax purposes will depend on whether the redemption qualifies as a sale or exchange of the shares of Globalink common stock under Section 302 of the Code or is treated as a corporate distribution under Section 301 of the Code with respect to the U.S. Holder. If the redemption qualifies as a sale or exchange of the shares of Globalink common stock, the U.S. Holder will be treated as recognizing capital gain or loss equal to the difference between the amount realized on the redemption and such U.S. Holder’s adjusted tax basis in the shares of Globalink common stock surrendered in such redemption transaction. Any such capital gain or loss generally will be long-term capital gain or loss if the U.S. Holder’s holding period for the shares of Globalink common stock redeemed exceeds one year. It is unclear, however, whether the redemption rights with respect to the shares of Globalink common stock may suspend the running of the applicable holding period for this purpose. Long term capital gain realized by a non-corporate U.S. Holder is currently taxed at a reduced rate. The deductibility of capital losses is subject to limitations.
If the redemption does not qualify as a sale or exchange of Globalink common stock, the U.S. Holder will be treated as receiving a corporate distribution. Such distributions generally will constitute dividends for U.S. federal income tax purposes to the extent paid from Globalink’s current or accumulated earnings and profits, as determined under U.S. federal income tax principles. Distributions in excess of current and accumulated earnings and profits will constitute a return of capital that will be applied against and reduce (but not below zero) the U.S. Holder’s adjusted tax basis in the Globalink common stock. Any remaining excess will be treated as gain realized on the sale or other disposition of the common stock. Dividends paid to a U.S. Holder that is a taxable corporation generally will qualify for the dividends received deduction if the requisite holding period is satisfied. With certain exceptions (including, but not limited to, dividends treated as investment income for purposes of investment interest deduction limitations) and provided certain holding period requirements are met, dividends paid to a non-corporate U.S. Holder generally will constitute “qualified dividends” that will be subject to tax at the maximum tax rate accorded to long-term capital gains. However, it is unclear whether the redemption rights with respect to the shares of Globalink common stock may prevent a U.S. Holder from satisfying the applicable holding period requirements with respect to the dividends received deduction or the preferential tax rate on qualified dividend income, as the case may be.
| 153 |
Whether a redemption qualifies for sale or exchange treatment will depend largely on the total number of shares of Globalink common stock treated as held by the U.S. Holder (including any shares of Globalink common stock constructively owned by the U.S. Holder as a result of owning warrants) relative to all of the shares of Globalink common stock outstanding both before and after the redemption. The redemption of shares of Globalink common stock generally will be treated as a sale or exchange of the shares of Globalink common stock (rather than as a corporate distribution) if the redemption (i) is “substantially disproportionate” with respect to the U.S. Holder, (ii) results in a “complete termination” of the U.S. Holder’s interest in Globalink or (iii) is “not essentially equivalent to a dividend” with respect to the U.S. Holder. These tests are explained more fully below.
In determining whether any of the foregoing tests are satisfied, a U.S. Holder takes into account not only shares of Globalink common stock actually owned by the U.S. Holder, but also shares of Globalink common stock that are constructively owned by the U.S. Holder. A U.S. Holder may constructively own, in addition to shares of stock owned directly, shares of stock owned by certain related individuals and entities in which the U.S. Holder has an interest or that have an interest in such U.S. Holder, as well as any stock the U.S. Holder has a right to acquire by exercise of an option, which would generally include shares of Globalink common stock which could be acquired pursuant to the exercise of warrants. In order to meet the substantially disproportionate test, (i) the percentage of Globalink’s outstanding shares of voting stock actually and constructively owned by the U.S. Holder immediately following the redemption of Globalink common stock must be less than 80% of the percentage of its outstanding shares of voting stock actually and constructively owned by the U.S. Holder immediately before the redemption, (ii) the U.S. Holder’s percentage ownership (including constructive ownership) of Globalink’s outstanding shares of stock (both voting and nonvoting) immediately after the redemption must be less than 80% of such percentage ownership (including constructive ownership) immediately before the redemption; and (iii) the U.S. Holder must own (including constructive ownership), immediately after the redemption, less than 50% of the total combined voting power of all classes of Globalink’s shares of stock entitled to vote. There will be a complete termination of a U.S. Holder’s interest if either (i) all of the shares of the Globalink common stock actually and constructively owned by the U.S. Holder are redeemed or (ii) all of the shares of the Globalink common stock actually owned by the U.S. Holder are redeemed and the U.S. Holder is eligible to waive, and effectively waives in accordance with specific rules, the attribution of stock owned by certain family members and the U.S. Holder does not constructively own any other shares of Globalink common stock. The redemption of the shares of Globalink common stock will not be essentially equivalent to a dividend if a U.S. Holder’s redemption results in a “meaningful reduction” of the U.S. Holder’s proportionate interest in Globalink. Whether the redemption will result in a meaningful reduction in a U.S. Holder’s proportionate interest in Globalink will depend on the particular facts and circumstances. However, the IRS has indicated in a published ruling that even a small reduction in the proportionate interest of a small minority stockholder in a publicly held corporation who exercises no control over corporate affairs may constitute such a “meaningful reduction.” A U.S. Holder should consult with its own tax advisors as to the tax consequences of a redemption.
If none of the foregoing tests are satisfied, then the redemption will be treated as a corporate distribution. After the application of those rules regarding corporate distributions, any remaining tax basis of the U.S. Holder in the redeemed shares of Globalink common stock will be added to the U.S. Holder’s adjusted tax basis in its remaining shares of Globalink common stock, or, if it has none, to the U.S. Holder’s adjusted tax basis in its warrants or possibly in other shares of Globalink common stock constructively owned by it.
| 154 |
U.S. Federal Income Tax Consequences to Non-U.S. Holders
The characterization for U.S. federal income tax purposes of the redemption of a Non-U.S. Holder’s Globalink common stock as a sale or exchange under Section 302 of the Code or as a corporate distribution under Section 301 of the Code generally will correspond to the U.S. federal income tax characterization of such a redemption of a U.S. Holder’s shares of Globalink common stock, as described above, and the corresponding consequences will be as described below.
Redemption Treated as Sale or Exchange
Any gain realized by a Non-U.S. Holder on the redemption of shares of Globalink common stock that is treated as a sale or exchange under Section 302 of the Code generally will not be subject to U.S. federal income tax unless:
| ● | the gain is effectively connected with a trade or business of the Non-U.S. Holder in the United States (and, if required by an applicable income tax treaty, is attributable to a United States permanent establishment or fixed base of the Non-U.S. Holder); | |
| ● | the Non-U.S. Holder is an individual who is present in the United States for a period or periods aggregating 183 days or more in the taxable year of the disposition, and certain other conditions are met; or | |
| ● | Globalink is or has been a “United States real property holding corporation” for U.S. federal income tax purposes at any time during the shorter of the five-year period ending on the date of disposition or the Non-U.S. Holder’s holding period for such shares of Globalink common stock redeemed, and either (A) shares of Globalink common stock are not considered to be regularly traded on an established securities market or (B) such Non-U.S. Holder has owned or is deemed to have owned, at any time during the shorter of the five-year period preceding such disposition and such Non-U.S. Holder’s holding period more than 5% of the outstanding shares of Globalink common stock. There can be no assurance that shares of Globalink common stock will be treated as regularly traded on an established securities market for this purpose. |
A non-corporate Non-U.S. Holder described in the first bullet point immediately above will be subject to tax on the net gain derived from the sale under regular graduated U.S. federal income tax rates. An individual Non-U.S. Holder described in the second bullet point immediately above will be subject to a flat 30% tax on the gain derived from the sale, which may be offset by certain United States source capital losses, even though the individual is not considered a resident of the United States, provided that the individual has timely filed U.S. federal income tax returns with respect to such losses. If a Non-U.S. Holder that is a corporation falls under the first bullet point immediately above, it will be subject to tax on its net gain in the same manner as if it were a United States person as defined under the Code and, in addition, may be subject to the branch profits tax equal to 30% (or such lower rate as may be specified by an applicable income tax treaty) of its effectively connected earnings and profits, subject to adjustments.
If the last bullet point immediately above applies to a Non-U.S. Holder, gain recognized by such Non-U.S. Holder on the redemption of shares of Globalink common stock generally will be subject to tax at generally applicable U.S. federal income tax rates. In addition, Globalink may be required to withhold U.S. income tax at a rate of 15% of the amount realized upon such redemption. Globalink would generally be classified as a “U.S. real property holding corporation” if the fair market value of Globalink’s “United States real property interests” equals or exceeds 50% of the sum of the fair market value of Globalink’s worldwide real property interests and Globalink’s other assets used or held for use in a trade or business, as determined for U.S. federal income tax purposes. However, Globalink believes that it is not and has not been at any time since its formation a U.S. real property holding corporation and Globalink does not expect to be a U.S. real property holding corporation immediately after the Business Combination is completed.
Redemption Treated as Corporate Distribution
With respect to any redemption treated as a corporate distribution under Section 301 of the Code, provided such dividends are not effectively connected with the Non-U.S. Holder’s conduct of a trade or business within the United States, Globalink will be required to withhold U.S. tax from the gross amount of the dividend at a rate of 30%, unless such Non-U.S. Holder is eligible for a reduced rate of withholding tax under an applicable income tax treaty and provides proper certification of its eligibility for such reduced rate (usually on an IRS Form W-8BEN or W-8BEN-E). Any distribution not constituting a dividend will be treated first as reducing (but not below zero) the Non-U.S. Holder’s adjusted tax basis in its shares of the Globalink common stock and, to the extent such distribution exceeds the Non-U.S. Holder’s adjusted tax basis, as gain realized from the sale or other disposition of the common stock, which will be treated as described above.
| 155 |
This withholding tax does not apply to dividends paid to a Non-U.S. Holder who provides a Form W-8ECI, certifying that the dividends are effectively connected with the Non-U.S. Holder’s conduct of a trade or business within the United States. Instead, the effectively connected dividends will be subject to regular U.S. income tax as if the Non-U.S. Holder were a U.S. resident, subject to an applicable income tax treaty providing otherwise. A Non-U.S. corporation receiving effectively connected dividends may also be subject to an additional “branch profits tax” imposed at a rate of 30% (or a lower treaty rate).
U.S. Federal Income Tax Consequences of the Business Combination to U.S. Holders of Globalink’s Securities
The following discussion, “— U.S. Federal Income Tax Consequences of the Business Combination to U.S. Holders,” constitutes the opinion of Hunter Taubman Fischer & Li LLC, counsel to Globalink, as to the material U.S. federal income tax consequences of the Business Combination, including the Redomestication Merger and the Acquisition Merger, to U.S. Holders of Globalink’s securities, subject to the limitations, exceptions, beliefs, assumptions, and qualifications described in such opinion and otherwise herein.
If the Redomestication Merger Qualifies as a Reorganization
The Redomestication Merger should qualify as a “reorganization” within the meaning of Section 368 of the Code. However, the provisions of the Code that govern reorganizations are complex and are based on typical transaction structures effected under U.S. law. U.S. Holders should be aware that the completion of the Business Combination is not conditioned on the receipt of an opinion of counsel that the Redomestication Merger qualifies as a reorganization, and that none of Globalink, PubCo or Alps Holdco has requested nor intends to request a ruling from the IRS with respect to the U.S. federal income tax treatment of the Business Combination. There can be no assurance that the IRS will not take a contrary position to views expressed herein or that a court will not agree with a contrary position of the IRS.
Although U.S. persons generally do not recognize gain or loss on the receipt of stock pursuant to a reorganization under Section 368 of the Code, Section 367(a) of the Code and Treasury Regulations promulgated thereunder require, where applicable, U.S. persons to recognize gain (but not loss) with respect to certain cross-border reorganizations. However, Section 367(a) should not apply to the Redomestication Merger in a manner that causes gain recognition to the U.S. Holders, unless the exchange of Globalink securities for PubCo securities is considered to be an indirect stock transfer under the applicable Treasury Regulations. For this purpose, an indirect stock transfer may occur if PubCo transfers the assets it acquires from Globalink pursuant to the Redomestication Merger to certain subsidiary corporations in connection with the Business Combination. If the indirect stock transfer rules apply, then the requirements under Section 367(a) of the Code generally would require U.S. Holders to recognize gain, but not loss, in the Redomestication Merger. The rules under Section 367(a) of the Code and Section 368 of the Code are complex and there is limited guidance as to their application, particularly with regard to indirect stock transfers in cross-border reorganizations. Accordingly, no assurance can be given as to whether an indirect stock transfer will occur in connection with the Business Combination or that U.S. Holders will not recognize gain, if any, as a result of the exchange of Globalink securities for PubCo securities.
Because it is intended that the Redomestication Merger qualify as a reorganization under the provisions of Section 368 of the Code and provided that it is not treated as an indirect stock transfer, a U.S. Holder that exchanges its Globalink securities pursuant to the Business Combination should not recognize gain or loss on the exchange of Globalink securities for PubCo securities. The aggregate adjusted tax basis of a U.S. Holder in the PubCo ordinary shares received as a result of the Business Combination should equal the aggregate adjusted tax basis of the shares of Globalink common stock and Globalink rights surrendered in the exchange, and the aggregate adjusted tax basis in the PubCo warrants received as a result of such exchange should equal the aggregate adjusted tax basis of the Globalink warrants surrendered in the exchange. A U.S. Holder’s holding period for the PubCo securities received in the exchange should include the holding period for the Globalink securities surrendered in the exchange. If Section 367(a) of the Code applies to the Redomestication Merger, as described above, a U.S. Holder may be required to recognize gain (but not loss) as a result of the Redomestication Merger.
| 156 |
If the Redomestication Merger Does Not Qualify as a Reorganization
If the Redomestication Merger fails to qualify as a “reorganization” within the meaning of Section 368 of the Code, a U.S. Holder that exchanges its Globalink securities for the consideration under the Business Combination will recognize gain or loss equal to the difference between (i) the sum of the fair market value of the PubCo securities received and (ii) the U.S. Holder’s adjusted tax basis in the Globalink securities exchanged. A U.S. Holder’s aggregate tax basis in the PubCo securities received will be the fair market value of those securities on the date the U.S. Holder receives them. The U.S. Holder’s holding period for the PubCo securities received pursuant to the Business Combination will begin on the day after the date the U.S. Holder receives such PubCo securities.
Such gain or loss will be a capital gain or loss and will be a long-term capital gain or loss if the U.S. Holder’s holding period for the Globalink securities exceeds one year at the time of the Business Combination. Long-term capital gains of non-corporate U.S. Holders currently are subject to reduced rates of U.S. federal income taxation. The deductibility of capital losses is subject to limitations under the Code. Any such gain or loss recognized by a U.S. Holder will generally be treated as U.S. source gain or loss.
U.S. Holders should consult their own tax advisors as to the particular consequences to them of the exchange of Globalink securities for PubCo securities pursuant to the Business Combination, the qualification of the Redomestication Merger as a reorganization, and the potential application of Section 367(a) to the Redomestication Merger.
U.S. Federal Income Tax Consequences of the Acquisition Merger to U.S. Holders
Qualification of the Acquisition Merger as a Reorganization
The Acquisition Merger should qualify as a “reorganization” within the meaning of Section 368(a)(1)(A) of the Code. However, U.S. Holders should be aware that the completion of the Business Combination is not conditioned on the receipt of an opinion of counsel that the Acquisition Merger qualifies as a reorganization, and that none of Globalink, PubCo, or Alps Holdco has requested nor intends to request a ruling from the IRS with respect to the U.S. federal income tax treatment of the Acquisition Merger. There can be no assurance that the IRS will not take a contrary position to views expressed herein or that a court will not agree with a contrary position of the IRS.
Tax Treatment of U.S. Holders
As discussed above, because the Redomestication Merger should be treated separately as a “reorganization” under Section 368(a) of the Code, U.S. Holders should not be treated as exchanging stock or securities in the Acquisition Merger and therefore will not be subject to any separate U.S. federal income tax as a result of the Acquisition Merger. However, the rules under Section 368 of the Code are complex and there is limited guidance as to their application in the context of a transaction or series of transactions like those contemplated by the Business Combination. Accordingly, no assurance can be given as to whether an indirect stock transfer will occur in connection with the Business Combination or that U.S. Holders will recognize gain, if any, as a result of the merger of Merger Sub with and into Alps Holdco. U.S. Holders should consult their own tax advisors as to the particular consequences to them of the qualification of the Acquisition Merger as a reorganization.
Certain U.S. Federal Income Tax Consequences of the Business Combination to Globalink and PubCo
The following discussion is a brief summary of certain U.S. federal income tax consequences of the Business Combination to Globalink and PubCo.
Tax Residence of PubCo for U.S. Federal Income Tax Purposes
Under current U.S. federal income tax law, a corporation generally is considered a resident for U.S. federal income tax purposes in its place of organization or incorporation. Accordingly, under the generally applicable U.S. federal income tax rules, PubCo, which is a Cayman Islands-incorporated entity, would generally be classified as a non-U.S. corporation (and, therefore, not a U.S. tax resident). Section 7874 of the Code and the Treasury Regulations promulgated thereunder, however, contain specific rules (more fully discussed below) that may cause a non-U.S. corporation to be treated as a U.S. corporation for U.S. federal income tax purposes. If it were determined that PubCo should be taxed as a U.S. corporation for U.S. federal income tax purposes under Section 7874 of the Code, PubCo would be liable for U.S. federal income tax on its income like any other U.S. corporation, and certain distributions made by PubCo to non-U.S. holders of PubCo securities would be subject to U.S. withholding tax at the rate of 30% or such lower rate as provided by an applicable treaty. As a result, taxation as a U.S. corporation could have a material adverse effect on PubCo’s financial position and results from operations. The Section 7874 rules are complex and require analysis of all relevant facts and circumstances, and there is limited guidance and significant uncertainties as to their application.
Under Section 7874 of the Code, a corporation created or organized outside the United States (i.e., a non-U.S. corporation) will nevertheless be treated as a U.S. corporation for U.S. federal income tax purposes (and, therefore, be a U.S. tax resident subject to U.S. federal income tax on its worldwide income) if (1) the non-U.S. corporation directly or indirectly acquires substantially all of the assets held directly or indirectly by a U.S. corporation, (2) the non-U.S. corporation’s expanded affiliated group does not have substantial business activities in the non-U.S. corporation’s country of organization or incorporation relative to the expanded affiliated group’s worldwide activities (the “substantial business activities test”), and (3) the shareholders of the acquired U.S. corporation hold at least 80% (by either vote or value) of the stock of the non-U.S. acquiring corporation after the acquisition by reason of holding shares in the U.S. acquired corporation, as determined under complex share ownership rules described below, which are uncertain in their application in many circumstances and are intended to increase the percentage ownership for these purposes (the “Ownership Test”). For this purpose, “expanded affiliated group” generally means the foreign acquiring corporation and all subsidiary corporations in which such foreign corporation owns, directly or indirectly, more than 50% of the stock (by vote and value) after the foreign acquiring corporation’s acquisition of the assets of the U.S. corporation.
In the Business Combination, PubCo will directly acquire all of Globalink’s assets through Globalink merging with and into PubCo, with PubCo remaining as the surviving publicly traded entity. As a result, the determination of whether PubCo will be treated as a U.S. corporation for U.S. federal income tax purposes will depend on the application of the substantial business activities test and the Ownership Test. PubCo is not expected to satisfy the substantial business activities test based on its activities in the Cayman Islands after the completion of the Business Combination. Accordingly, PubCo must determine whether the Ownership Test has been met.
| 157 |
Based on the complex rules for determining share ownership under Section 7874 of the Code and Treasury Regulations promulgated thereunder and certain factual assumptions, PubCo expects to take the position that after completion of the Business Combination, former shareholders of Globalink will own, by reason of owning (or being treated as owning) stock of Globalink, less than 80% of the voting power and value of the PubCo securities. Therefore, PubCo is not expected to satisfy the Ownership Test, and PubCo is not expected to be treated as a U.S. corporation for U.S. federal income tax purposes under Section 7874 of the Code. However, PubCo’s position depends in part on the position that the Ownership Test is determined after the Business Combination rather than immediately after the Redomestication Merger for purposes of Section 7874 of the Code.
No IRS ruling has been requested or will be obtained regarding the U.S. federal income tax consequences of the Business Combination. If the IRS were to apply Section 7874 of the Code immediately after completion of the Redomestication Merger, but before the Business Combination, then Section 7874 of the Code is generally expected to treat PubCo as a U.S. corporation for U.S. federal income tax purposes.
The application of the Ownership Test is extremely complex, and the applicable Treasury Regulations relating to the Ownership Test are subject to significant uncertainty and there is limited guidance regarding their application. Moreover, the application of the Ownership Test to the facts and circumstances of the proposed transactions are uncertain. In addition, changes to the rules in Section 7874 of the Code or the Treasury Regulations promulgated thereunder, or other changes or interpretations in the law, could adversely affect PubCo’s status as a non-U.S. corporation for U.S. federal income tax purposes. Accordingly, PubCo’s expectation that Section 7874 of the Code will not apply to treat PubCo as a U.S. corporation for U.S. federal income tax purposes is subject to challenge, and there can be no assurance that the IRS will not take a contrary position to those described above or that a court will not agree with a contrary position of the IRS in the event of litigation.
Gain Recognized by Globalink as a Result of the Redomestication Merger
Although corporations generally do not recognize gain or loss on the transfer of assets pursuant to a reorganization under Section 368 of the Code, however, Section 367 of the Code and Treasury Regulations promulgated thereunder require, where applicable, U.S. corporations to recognize gain (but not loss) with respect to certain transfers to foreign corporations in certain cross-border reorganizations.
Even if the Redomestication Merger qualifies as a “reorganization” within the meaning of Section 368 of the Code, as a result of Section 367 of the Code, Globalink may recognize gain (but not loss) on the transfer of its assets to PubCo, to the extent that the fair market value of such assets exceeds Globalink’s adjusted basis in such assets.
U.S. Federal Income Tax Consequences of Ownership and Disposition of PubCo Securities
The following discussion is a summary of certain material U.S. federal income tax consequences of the ownership and disposition of PubCo securities to U.S. Holders who receive such PubCo securities pursuant to the Business Combination, assuming PubCo is not treated as a U.S. corporation for U.S. federal income tax purposes under Section 7874 of the Code.
Distribution on PubCo ordinary shares
Subject to the PFIC rules discussed below “— Passive Foreign Investment Company Status,” a U.S. Holder generally will be required to include in gross income any distribution of cash or property paid on PubCo ordinary shares that is treated as a dividend for U.S. federal income tax purposes. A distribution on such shares generally will be treated as a dividend for U.S. federal income tax purposes to the extent the distribution is paid out of PubCo’s current or accumulated earnings and profits (as determined under U.S. federal income tax principles). Such dividends paid by PubCo will be taxable to a corporate U.S. Holder at regular rates and will not be eligible for the dividends-received deduction generally allowed to domestic corporations in respect of dividends received from other domestic corporations.
| 158 |
Dividends received by non-corporate U.S. Holders from a “qualified foreign corporation” may be eligible for reduced rates of taxation, provided that certain holding period requirements and other conditions are satisfied. For these purposes, a non-U.S. corporation will be treated as a qualified foreign corporation with respect to dividends paid by that corporation on shares that are readily tradable on an established securities market in the United States. U.S. Treasury Department guidance indicates that shares listed on the Nasdaq (on which PubCo intends to apply to list the PubCo ordinary shares) will be considered readily tradable on an established securities market in the United States. Even if the PubCo ordinary shares are listed on Nasdaq, there can be no assurance that the PubCo ordinary shares will be considered readily tradable on an established securities market in future years. Non-corporate U.S. Holders that do not meet a minimum holding period requirement or that elect to treat the dividend income as “investment income” pursuant to Section 163(d)(4) of the Code (dealing with the deduction for investment interest expense) will not be eligible for the reduced rates of taxation regardless of PubCo’s status as a qualified foreign corporation. In addition, the rate reduction will not apply to dividends if the recipient of a dividend is obligated to make related payments with respect to positions in substantially similar or related property. This disallowance applies even if the minimum holding period has been met. Finally, PubCo will not constitute a qualified foreign corporation for purposes of these rules if it is a PFIC for the taxable year in which it pays a dividend or for the preceding taxable year. See the discussion below under “— Passive Foreign Investment Company Status.”
The amount of any dividend paid in foreign currency will be the U.S. dollar value of the foreign currency distributed by PubCo, calculated by reference to the spot exchange rate in effect on the date the dividend is includible in the U.S. Holder’s income, regardless of whether the payment is in fact converted into U.S. dollars on the date of receipt. Generally, a U.S. Holder should not recognize any foreign currency gain or loss if the foreign currency is converted into U.S. dollars on the date the payment is received. However, any gain or loss resulting from currency exchange fluctuations during the period from the date the U.S. Holder includes the dividend payment in income to the date such U.S. Holder actually converts the payment into U.S. dollars will be treated as ordinary income or loss.
To the extent that the amount of any distribution made by PubCo on the PubCo ordinary shares exceeds PubCo’s current and accumulated earnings and profits for a taxable year (as determined under U.S. federal income tax principles), the distribution will first be treated as a tax-free return of capital, causing a reduction in the adjusted basis of the U.S. Holder’s PubCo ordinary shares, and to the extent the amount of the distribution exceeds the U.S. Holder’s tax basis, the excess will be taxed as capital gain recognized on a sale or exchange as described below under “— Sale, Exchange, Redemption or Other Taxable Disposition of PubCo Securities.” However, PubCo may not calculate earnings and profits in accordance with U.S. federal income tax principles. In such event, a U.S. Holder should expect to generally treat distributions PubCo makes as dividends.
Sale, Exchange, Redemption or Other Taxable Disposition of PubCo Securities
Subject to the discussion below under “— Passive Foreign Investment Company Status,” a U.S. Holder will generally recognize gain or loss on any sale, exchange, or other taxable disposition of PubCo ordinary shares and PubCo warrants in an amount equal to the difference between the amount realized on the disposition and such U.S. Holder’s adjusted tax basis in such PubCo ordinary shares or PubCo warrants. Any gain or loss recognized by a U.S. Holder on a taxable disposition of PubCo ordinary shares or PubCo warrants will generally be capital gain or loss and will be long-term capital gain or loss if the holder’s holding period in the PubCo ordinary shares or PubCo warrants exceeds one year at the time of the disposition. Preferential tax rates may apply to long-term capital gains of non-corporate U.S. Holders. The deductibility of capital losses is subject to limitations. Any gain or loss recognized by a U.S. Holder on the sale or exchange of PubCo ordinary shares or PubCo warrants will generally be treated as U.S. source gain or loss.
Exercise or Lapse of a PubCo warrants
Except as discussed below with respect to the cashless exercise of a PubCo warrant, and subject to the PFIC rules discussed below, a U.S. Holder generally will not recognize gain or loss upon the acquisition of a PubCo ordinary share on the exercise of a PubCo warrant for cash. A U.S. Holder’s tax basis in a PubCo ordinary share received upon exercise of the PubCo warrant generally will be an amount equal to the sum of the U.S. Holder’s tax basis in the PubCo warrant exchanged therefor and the exercise price. The U.S. Holder’s holding period for a PubCo ordinary share received upon exercise of the PubCo warrant will begin on the date following the date of exercise (or possibly the date of exercise) of the PubCo warrants and will not include the period during which the U.S. Holder held the PubCo warrants. If a PubCo warrant is allowed to lapse unexercised, a U.S. Holder generally will recognize a capital loss equal to such holder’s tax basis in the PubCo warrant.
| 159 |
The tax consequences of a cashless exercise of warrants are not clear under current tax law. A cashless exercise may be tax-free, either because the exercise is not a realization event or because the exercise is treated as a recapitalization for U.S. federal income tax purposes. In either tax-free situation, a U.S. Holder’s basis in the PubCo ordinary shares received generally would equal the holder’s basis in the PubCo warrants exchanged therefor. If the cashless exercise were treated as not being a realization event, a U.S. Holder’s holding period in the PubCo ordinary shares would be treated as commencing on the date following the date of exercise (or possibly the date of exercise) of the PubCo warrant and will not include the period during which the U.S. Holder held the PubCo warrants. If the cashless exercise were treated as a recapitalization, the holding period of the PubCo ordinary share would include the holding period of the PubCo warrant.
It is also possible that a cashless exercise could be treated as a taxable exchange in which gain or loss would be recognized. In such event, a U.S. Holder could be deemed to have surrendered a number of PubCo warrants with a fair market value equal to the exercise price for the number of PubCo warrants deemed exercised. For this purpose, the number of warrants deemed exercised would be equal to the amount needed to receive on exercise the number of PubCo ordinary shares issued pursuant to the cashless exercise. In this situation, the U.S. Holder would recognize capital gain or loss in an amount equal to the difference between the fair market value of the PubCo warrants deemed surrendered to pay the exercise price and the U.S. Holder’s tax basis in the PubCo warrants deemed surrendered. Such gain or loss would be long-term or short-term depending on the U.S. Holder’s holding period in the PubCo warrants. In this case, a U.S. Holder’s tax basis in the PubCo ordinary shares received would equal the sum of the fair market value of the PubCo warrants deemed surrendered and the U.S. Holder’s tax basis in the PubCo warrants deemed exercised. A U.S. Holder’s holding period in the PubCo ordinary shares would be treated as commencing on the date following the date of exercise (or possibly the date of exercise) of the PubCo warrant.
Due to the absence of authority on the U.S. federal income tax treatment of a cashless exercise, there can be no assurance which, if any, of the alternative tax consequences and holding periods described above would be adopted by the IRS or a court of law. Accordingly, U.S. Holders should consult their tax advisors regarding the tax consequences of a cashless exercise.
Passive Foreign Investment Company (PFIC) Status
Certain adverse U.S. federal income tax consequences could apply to a U.S. Holder if PubCo, or any of its subsidiaries, is treated as a PFIC for any taxable year during which the U.S. Holder holds PubCo securities. A non-U.S. corporation will be classified as a PFIC for any taxable year (a) if at least 75% of its gross income in a taxable year, including its pro rata share of the gross income of any entity in which it is considered to own at least 25% of the interest by value, is passive income, or (b) if at least 50% of its assets in a taxable year of the foreign corporation, ordinarily determined based on fair market value and averaged quarterly over the year, including its pro rata share of the assets of any entity in which it is considered to own at least 25% of the interest by value, are held for the production of, or produce, passive income. Passive income generally includes dividends, interest, rents and royalties (other than rents or royalties derived from the active conduct of a trade or business) and gains from the disposition of passive assets.
Whether PubCo or any of its subsidiaries is treated as a PFIC for U.S. federal income tax purposes is a factual determination that must be made annually at the close of each taxable year and, thus, is subject to significant uncertainty. Among other factors, fluctuations in the market price of PubCo ordinary shares and how, and how quickly, PubCo uses liquid assets and cash obtained in the Business Combination may influence whether PubCo or any of its subsidiaries is treated as PFIC. Accordingly, we may not be able to determine whether PubCo or any of its subsidiaries will be treated as a PFIC for the taxable year of the Business Combination or for future taxable years, and there can be no assurance that PubCo or any of its subsidiaries will not be treated as a PFIC for any taxable year.
If PubCo were determined to be a PFIC for any taxable year (or portion thereof) that is included in the holding period of a U.S. Holder of PubCo securities and, in the case of PubCo ordinary Shares, the U.S. Holder did not make a valid “mark-to-market” election, such U.S. Holder generally will be subject to special rules with respect to: (i) any gain recognized by the U.S. Holder on the sale or other disposition of PubCo securities and (ii) any “excess distribution” made to the U.S. Holder (generally, any distributions to such U.S. Holder during a taxable year of the U.S. Holder that are greater than 125% of the average annual distributions received by such U.S. Holder in respect of the PubCo ordinary shares during the three preceding taxable years of such U.S. Holder or, if shorter, such U.S. Holder’s holding period for such ordinary shares).
| 160 |
Under these rules:
| ● | the U.S. Holder’s gain or excess distribution will be allocated ratably over the U.S. Holder’s holding period for PubCo securities; | |
| ● | the amount allocated to the U.S. Holder’s taxable year in which the U.S. holder recognized gain or received the excess distribution, or to the period in the U.S. Holder’s holding period before the first day of PubCo’s first taxable year in which PubCo is a PFIC, will be taxed as ordinary income; | |
| ● | the amount allocated to other taxable years (or portions thereof) of the U.S. Holder and included in its holding period will be taxed at the highest tax rate in effect for that year and applicable to the U.S. Holder; and | |
| ● | the interest charge generally applicable to underpayments of tax will be imposed in respect of the tax attributable to each such other taxable year of the U.S. Holder. |
Although a determination as to PubCo’s PFIC status will be made annually, an initial determination that PubCo is a PFIC will generally apply for subsequent years to a U.S. Holder who held PubCo securities while PubCo was a PFIC, whether or not PubCo meets the test for PFIC status in those subsequent years.
If a U.S. Holder, at the close of its taxable year, owns shares in a PFIC that are treated as marketable stock, the U.S. Holder may make a mark-to-market election with respect to such shares for such taxable year. If the U.S. Holder makes a valid mark-to-market election for the first taxable year of the U.S. Holder in which the U.S. Holder holds (or is deemed to hold) PubCo ordinary shares and for which PubCo is determined to be a PFIC, such holder generally will not be subject to the PFIC rules described above in respect to its PubCo ordinary shares as long as such shares continue to be treated as marketable stock. Instead, in general, the U.S. Holder will include as ordinary income each year that PubCo is treated as a PFIC the excess, if any, of the fair market value of its PubCo ordinary shares at the end of its taxable year over the adjusted basis in its PubCo ordinary shares. The U.S. Holder also will be allowed to take an ordinary loss in respect of the excess, if any, of the adjusted basis of its PubCo ordinary shares over the fair market value of its PubCo ordinary shares at the end of its taxable year (but only to the extent of the net amount of previously recognized income as a result of the mark-to-market election). The U.S. Holder’s adjusted tax basis in its PubCo ordinary shares will be adjusted to reflect any such income or loss amounts, and any further gain recognized on a sale or other taxable disposition of the PubCo ordinary shares in a taxable year in which PubCo is treated as a PFIC will be treated as ordinary income. Special tax rules may also apply if a U.S. Holder makes a mark-to-market election for a taxable year after the first taxable year in which the U.S. Holder holds (or is deemed to hold) its PubCo ordinary shares and for which PubCo is treated as a PFIC. Currently, a mark-to-market election may not be made with respect to PubCo warrants.
The mark-to-market election is available only for stock that is regularly traded on a national securities exchange that is registered with the U.S. Securities and Exchange Commission, including Nasdaq (on which PubCo intends to list the ordinary shares), or on a foreign exchange or market that the IRS determines has rules sufficient to ensure that the market price represents a legitimate and sound fair market value. Such stock generally will be “regularly traded” for any calendar year during which such stock is traded, other than in de minims quantities, on at least 15 days during each calendar quarter, but no assurances can be given in this regard with respect to the ordinary shares. U.S. Holders should consult their own tax advisors regarding the availability and tax consequences of a mark-to-market election in respect of PubCo ordinary shares under their particular circumstances.
If PubCo is a PFIC and, at any time, has a foreign subsidiary that is classified as a PFIC, U.S. Holders generally would be deemed to own a portion of the shares of such lower-tier PFIC, and generally could incur liability for the deferred tax and interest charge described above if PubCo were to receive a distribution from, or dispose of all or part of PubCo’s interest in, the lower-tier PFIC (even though such U.S. Holder would not receive the proceeds of those distributions or dispositions) or the U.S. Holders otherwise were deemed to have disposed of an interest in the lower-tier PFIC. A mark-to-market election generally would not be available with respect to such lower-tier PFIC. U.S. Holders are urged to consult their own tax advisors regarding the tax issues raised by lower-tier PFICs.
| 161 |
If U.S. Holders do not make a timely “mark-to-market” election (as described above), and if PubCo were a PFIC at any time during the period U.S. Holders hold PubCo ordinary shares, then such ordinary shares will continue to be treated as stock of a PFIC with respect to U.S. Holder even if PubCo cease to be a PFIC in a future year, unless U.S. Holders make a “purging election” for the year PubCo ceases to be a PFIC. A “purging election” creates a deemed sale of such ordinary shares at their fair market value on the last day of the last year in which PubCo is treated as a PFIC. The gain recognized by the purging election will be subject to the special tax and interest charge rules treating the gain as an excess distribution, as described above. As a result of the purging election, U.S. Holder will have a new basis (equal to the fair market value of the ordinary shares on the last day of the last year in which PubCo is treated as a PFIC) and holding period (which new holding period will begin the day after such last day) in U.S. Holder’s ordinary shares for tax purposes.
IRC Section 1014(a) provides for a step-up in basis to the fair market value for our ordinary shares when inherited from a decedent that was previously a holder of our ordinary shares. However, if PubCo is determined to be a PFIC and a decedent that was a U.S. Holder did not make either a timely qualified electing fund election for our first taxable year as a PFIC in which the U.S. Holder held (or was deemed to hold) our ordinary shares, or a mark-to-market election and ownership of those ordinary shares are inherited, a special provision in IRC Section 1291(e) provides that the new U.S. Holder’s basis should be reduced by an amount equal to the Section 1014 basis minus the decedent’s adjusted basis just before death. As such if PubCo is determined to be a PFIC at any time prior to a decedent’s passing, the PFIC rules will cause any new U.S. Holder that inherits PubCo’s ordinary shares from a U.S. Holder to not get a step-up in basis under Section 1014 and instead will receive a carryover basis in those ordinary shares.
A U.S. Holder that owns (or is deemed to own) shares in a PFIC during any taxable year of the U.S. Holder may have to file an IRS Form 8621 (whether or not a mark-to-market election is or has been made) with such U.S. Holder’s U.S. federal income tax return and provide any such other information as may be required by the U.S. Treasury Department. Failure to do so, if required, will extend the statute of limitations until such required information is furnished to the IRS.
The rules dealing with PFICs and mark-to-market elections are very complex and are affected by various factors in addition to those described above. Accordingly, U.S. Holders of PubCo securities should consult their own tax advisors concerning the application of the PFIC rules to PubCo securities under their particular circumstances.
Information Reporting and Backup Withholding
In general, information reporting requirements will apply to dividends (including constructive dividends) received by U.S. Holders of PubCo ordinary shares, and the proceeds received on the disposition of PubCo ordinary shares and PubCo warrants effected within the United States (and, in certain cases, outside the United States), in each case, other than U.S. Holders that are exempt recipients (such as corporations). Information reporting requirements will also apply to redemptions from U.S. Holders of shares of Globalink common stock. Backup withholding (currently at a rate of 24%) may apply to such amounts if the U.S. Holder fails to provide an accurate taxpayer identification number (generally on an IRS Form W-9 provided to the paying agent or the U.S. Holder’s broker) or is otherwise subject to backup withholding.
Certain U.S. Holders holding specified foreign financial assets with an aggregate value in excess of the applicable dollar threshold are required to report information to the IRS relating to PubCo securities, subject to certain exceptions (including an exception for PubCo securities held in accounts maintained by U.S. financial institutions), by attaching a complete IRS Form 8938, Statement of Specified Foreign Financial Assets, with their tax return, for each year in which they hold PubCo securities. In addition to these requirements, U.S. Holders may be required to annually file FinCEN Report 114 (Report of Foreign Bank and Financial Accounts) with the U.S. Department of Treasury. U.S. Holders who are required to report specified foreign financial assets on IRS Form 8938 and/or foreign bank and financial accounts on FinCEN Report 114 and fail to do so may be subject to substantial penalties. U.S. Holders should consult their own tax advisors regarding information reporting requirements relating to their ownership of PubCo securities.
Backup withholding is not an additional tax. Any amounts withheld under the backup withholding rules may be allowed as a refund or credit against a holder’s U.S. federal income tax liability, if any, by filing the appropriate claim for refund and timely providing the required information to the IRS.
| 162 |
DILUTION TO GLOBALINK’S STOCKHOLDERS
Globalink’s net tangible book value as of March 31, 2024 was US$(8,370,816), or US$2.25) per share, based on 3,722,511 shares of Globalink common stock outstanding as of that date. The net tangible book value as of March 31, 2024 based on condensed combined pro forma financial statements was US$13,592,178, or US$0.08 per share after giving effect to (i) the issuance of 160,000,000 PubCo ordinary shares at $10.00 per share, as Merger Consideration Shares, assuming no issuance of the Earnout Shares, (ii) the issuance of 4,020,000 PubCo ordinary shares at $10.00 per share, as shares issuable under the PIPE Investment, (iii) the issuance of 1,600,000 PubCo ordinary shares at $10.00 per share, IBDC Asia Sdn. Bhd., an advisory firm to Alps, and (iv) the automatic conversion of PubCo rights into PubCo ordinary shares upon the Closing, and assuming no PubCo warrants are exercised, Globalink’s as adjusted net tangible book value as of March 31, 2024.
The following table illustrates the changes in net tangible book value to existing shareholders and dilution to recipients of Merger Consideration Shares and PIPE Investors at varying redemption levels.
| Assuming No Redemption | Assuming 25% Redemption | Assuming 50% Redemption | Assuming 75% Redemption | Assuming Maximum Contractual Redemption | ||||||||||||||||
| Offering Price of the Securities in the Initial Registered offering price per share | $ | 10.00 | $ | 10.00 | $ | 10.00 | $ | 10.00 | $ | 10.00 | ||||||||||
| Pro forma net tangible book value, as adjusted (1) | $ | 13,592,178 | $ | 12,760,742 | $ | 11,929,307 | $ | 11,097,871 | $ | 10,266,447 | ||||||||||
| Total Shares (2) | 170,549,511 | 170,480,133 | 170,410,755 | 170,341,377 | 170,272,000 | |||||||||||||||
| Net tangible book value per share as of March 31, 2024 | $ | 0.08 | $ | 0.08 | $ | 0.07 | $ | 0.07 | $ | 0.06 | ||||||||||
| Dilution per share to recipients of Merger Consideration Shares and PIPE Investors | $ | (9.92 | ) | $ | (9.92 | ) | $ | (9.93 | ) | $ | (9.93 | ) | $ | (9.94 | ) | |||||
The following table illustrates the changes in net tangible book value to Globalink’s shareholders and increase in net tangible book value to Globalink’s shareholder as a result of net assets acquired of Alps Holdco with Merger Consideration Shares and PIPE Investors at varying redemption levels.
| Assuming No Redemption | Assuming 25% Redemption | Assuming 50% Redemption | Assuming 75% Redemption | Assuming Maximum Contractual Redemption | ||||||||||||||||
| Globalink’s net tangible book value as of March 31, 2024 | $ | (8,370,816 | ) | $ | (8,370,816 | ) | $ | (8,370,816 | ) | $ | (8,370,816 | ) | $ | (8,370,816 | ) | |||||
| Globalink’s shares outstanding | 3,722,511 | 3,722,511 | 3,722,511 | 3,722,511 | 3,722,511 | |||||||||||||||
| Globalink’s net tangible book value per share as of March 31, 2024 | $ | (2.25 | ) | $ | (2.25 | ) | $ | (2.25 | ) | $ | (2.25 | ) | $ | (2.25 | ) | |||||
| Increase in net tangible book value per share attributable to Globalink’s stockholders | $ | 2.33 | $ | 2.33 | $ | 2.32 | $ | 2.32 | $ | 2.31 | ||||||||||
The following table illustrates the as adjusted net tangible book value to Globalink’s shareholders and increase in net tangible book value to Globalink’s shareholder as a result of transaction cost incurred by Globalink, financing acquired to extend and deposit into trust, and funds released from the trust at de-SPAC, and reflecting the issuance of ordinary shares to the rights holders for rights issued at IPO.
| Assuming No Redemption | Assuming 25% Redemption | Assuming 50% Redemption | Assuming 75% Redemption | Assuming Maximum Contractual Redemption | ||||||||||||||||
| As adjusted net tangible book value per share after giving effect to the issuance of ordinary shares in connection with right issued at IPO to Globalink’s right holders | $ | 0.08 | $ | 0.08 | $ | 0.07 | $ | 0.07 | $ | 0.06 | ||||||||||
| Numerator adjustments | ||||||||||||||||||||
| Globalink’s net tangible book value | $ | (8,370,816 | ) | $ | (8,370,816 | ) | $ | (8,370,816 | ) | $ | (8,370,816 | ) | $ | (8,370,816 | ) | |||||
| Alp’s net tangible book value | 133,066 | 133,066 | 133,066 | 133,066 | 133,066 | |||||||||||||||
| PIPE proceeds | 40,200,000 | 40,200,000 | 40,200,000 | 40,200,000 | 40,200,000 | |||||||||||||||
| Transaction cost attributed to Globalink | (1,690,428 | ) | (1,690,428 | ) | (1,690,428 | ) | (1,690,428 | ) | (1,690,428 | ) | ||||||||||
| Transaction cost attributed to Alps | (813,540 | ) | (813,540 | ) | (813,540 | ) | (813,540 | ) | (813,540 | ) | ||||||||||
| Extension financing for deposits into trust | (540,000 | ) | (540,000 | ) | (540,000 | ) | (540,000 | ) | (540,000 | ) | ||||||||||
| Earnout liability | (18,522,340 | ) | (18,522,340 | ) | (18,522,340 | ) | (18,522,340 | ) | (18,522,340 | ) | ||||||||||
| Funds released from trust (3) | 2,770,102 | 2,494,295 | 1,662,861 | 831,424 | - | |||||||||||||||
| As adjusted net tangible book value | $ | 13,166,044 | $ | 12,890,237 | $ | 12,058,802 | $ | 11,227,366 | $ | 10,395,942 | ||||||||||
| Denominator adjustments | ||||||||||||||||||||
| Globalink’s shares outstanding | 3,722,511 | 3,653,133 | 3,583,755 | 3,514,377 | 3,445,000 | |||||||||||||||
| Globalink’s Public rights outstanding | 1,150,000 | 1,150,000 | 1,150,000 | 1,150,000 | 1,150,000 | |||||||||||||||
| PGM rights outstanding | 57,000 | 57,000 | 57,000 | 57,000 | 57,000 | |||||||||||||||
| Alps’ shareholders | 160,000,000 | 160,000,000 | 160,000,000 | 160,000,000 | 160,000,000 | |||||||||||||||
| IBDC Asia Sdn Bhd shareholders | 1,600,000 | 1,600,000 | 1,600,000 | 1,600,000 | 1,600,000 | |||||||||||||||
| PIPE investors | 4,020,000 | 4,020,000 | 4,020,000 | 4,020,000 | 4,020,000 | |||||||||||||||
| As adjusted Globalink’s shares outstanding | 170,549,511 | 170,480,133 | 170,410,755 | 170,341,377 | 170,272,000 | |||||||||||||||
| (1) | Net tangible book value is based on the pro forma net tangible asset after giving effect the adjustment disclosed in pro forma including the issuance of Merger consideration shares, PIPE investment and the settlement of transaction cost. The net tangible assets under the 25% redemption scenario was further adjusted for the redemptions of $831,436, under 50% redemption scenario was further adjusted for the redemptions of $1,662,871 and under 75% redemption scenario was further adjusted for the redemptions of $2,494,307. | |
| (2) | Excludes potentially dilutive outstanding securities consisting of 5,750,000 share count underlying the warrants held by Globlalink’s public shareholders, 285,000 share count underlying the warrants held by Private Placement Sponsor and 48,000,000 Earn-out shares. These sources of future dilution as the extent of their exercise is assumed not to be probable | |
| (3) | The funds released from trust was adjusted under the 25% redemption scenario for the redemptions of $831,436, under 50% redemption scenario for the redemptions of $1,662,871 and under 75% redemption scenario for the redemptions of $2,494,307. |
A US$1.00 increase or decrease in the price at which the shares are issued from the assumed issuance price of US$10.00 per share, would increase or decrease, respectively, our as adjusted net tangible book value per share by US$0.03 per share, and the dilution per share to Globalink’s stockholders in the Business Combination and recipients of Merger Consideration Shares and PIPE Investors by approximately US$0.03 per share, assuming no redemption. The as adjusted information provided above is illustrative only.
To the extent that additional shares are issued pursuant to the foregoing, Globalink’s stockholders will experience further dilution. In addition, Globalink may enter into other transactions. To the extent it issues such securities, investors and Globalink’s stockholders may experience further dilution.
Globalink issued shares in an initial registered offering at $10 per share. The issued and outstanding public shares of the Globalink as of the date of this proxy statement/prospectus are 277,511. In connection with the de-SPAC transaction, the Pubco will issue 160,000,000 shares to the Alps shareholders, 4,020,000 shares in the PIPE investment, and 1,600,000 to IBDC Asia Sdn Bhd, for a total of 165,897,511 outstanding shares. Redemption levels of zero, 25%, 50% 75% and Maximum Contractual Redemption have been disclosure in the table below as required by Item 1604(c).
For purposes of Item 1604(c)(1), Globalink would have 165,897,511 total shares after giving effect to the de-SPAC transaction and related financing under the zero-redemption scenario. Where redemptions are zero, the company valuation is based on Globalinks offering Price of the securities in the initial registered offering price per share of $10 is therefore calculated as: $10 (per share IPO price) times 165,897,511 shares, or $1,658,975,110. The following table illustrates the PubCo valuation at the offering Price of the securities in the initial registered offering price of $10 per share for each redemption scenario:
| Assuming No Redemption | Assuming 25% Redemption | Assuming 50% Redemption | Assuming 75% Redemption | Assuming Maximum Contractual Redemption | ||||||||||||||||
| Globalink shares Valuation based on offering price of the securities in the initial registered offering of $10.00 per share | $ | 2,775,110 | $ | 2,081,330 | $ | 1,387,550 | $ | 693,770 | $ | - | ||||||||||
| Globalink public shareholders Ordinary shares outstanding post de-SPAC | 277,511 | 208,133 | 138,755 | 69,377 | - | |||||||||||||||
| Alps shares Valuation based on offering price of the securities in the initial registered offering of $10.00 per share | $ | 1,600,000,000 | $ | 1,600,000,000 | $ | 1,600,000,000 | $ | 1,600,000,000 | $ | 1,600,000,000 | ||||||||||
| Alps shareholders Ordinary shares outstanding post de-SPAC | 160,000,000 | 160,000,000 | 160,000,000 | 160,000,000 | 160,000,000 | |||||||||||||||
| PIPE Investor Valuation based on offering price of the securities in the initial registered offering of $10.00 per share | $ | 40,200,000 | $ | 40,200,000 | $ | 40,200,000 | $ | 40,200,000 | $ | 40,200,000 | ||||||||||
| PIPE investor Ordinary shares outstanding post de-SPAC | 4,020,000 | 4,020,000 | 4,020,000 | 4,020,000 | 4,020,000 | |||||||||||||||
| IBDC Asia Sdn Bhd shares Valuation based on offering price of the securities in the initial registered offering of $10.00 per share | $ | 16,000,000 | $ | 16,000,000 | $ | 16,000,000 | $ | 16,000,000 | $ | 16,000,000 | ||||||||||
| IBDC Asia Sdn Bhd shareholders Ordinary shares outstanding post de-SPAC | 1,600,000 | 1,600,000 | 1,600,000 | 1,600,000 | 1,600,000 | |||||||||||||||
| Total PubCo Valuation based on offering price of the securities in the initial registered offering of $10.00 per share | $ | 1,658,975,110 | $ | 1,658,281,330 | $ | 1,657,587,550 | $ | 1,656,893,770 | $ | 1,656,200,000 | ||||||||||
| Total Pubco Ordinary shares outstanding post de-SPAC | 165,897,511 | 165,828,133 | 165,758,755 | 165,689,377 | 165,620,000 | |||||||||||||||
The required disclosure is not a guarantee that the trading price of the combined company will not be below the IPO offering price of Globalink, nor is the disclosure a guarantee the company valuation will attain one of the stated levels of valuation.
| 163 |
GLOBALINK’S BUSINESS
Unless the context otherwise requires, references to the “Company,” “we”, “us” and “our” in this section generally refer to Globalink Investment Inc.
Overview
Globalink was incorporated in Delaware on March 24, 2021 and was formed for the purpose of entering into a merger, share exchange, asset acquisition, share purchase, recapitalization, reorganization or other similar Business Combination with one or more businesses or entities. On December 9 2021, Globalink consummated its initial public offering (“IPO”) of 10,000,000 units. Globalink has until January 9, 2025 (and may be extended to June 9, 2025, on a monthly basis by resolutions of the Globalink Board, if requested by the Sponsor, and subject to the Sponsor depositing additional funds into the Trust Account) to consummate a Business Combination. If Globalink is unable to complete its initial business combination within this time period, it will (i) cease all operations except for the purpose of winding up, (ii) as promptly as reasonably possible but not more than ten business days thereafter, redeem 100% of the outstanding public shares for a pro rata portion of the funds held in the Trust Account, which redemption will completely extinguish public stockholders’ rights as stockholders (including the right to receive further liquidation distributions, if any), subject to applicable law and (iii) as promptly as reasonably possible following such redemption, subject to the approval of its remaining stockholders and its Board, dissolve and liquidate, subject (in the case of (ii) and (iii) above) to its obligations under Delaware law to provide for claims of creditors and the requirements of other applicable law.
If Globalink anticipates that it may not be able to consummate its initial business combination in time, Globalink may, by resolutions of its Board, if requested by its Sponsor, extend the period of time to consummate a business combination on a monthly basis to until June 9, 2025, subject to its Sponsor depositing additional funds into the Trust Account. For the time available for Globalink to consummate its initial business combination to be extended, its Sponsor or its affiliates or designees, upon five days advance notice prior to the applicable deadline, must deposit into the Trust Account $60,000 on or prior to the date of the applicable deadline, for each extension. In the event that Globalink receives notice from Globalink’s Sponsor five days prior to the applicable deadline of its wish for Globalink to effect an extension, Globalink intends to issue a press release announcing such intention at least three days prior to the applicable deadline. In addition, Globalink intends to issue a press release the day after the applicable deadline announcing whether or not the funds had been timely deposited. Globalink’s Sponsor and its affiliates or designees are not obligated to fund the Trust Account to extend the time for Globalink to complete the initial business combination.
On December 10, 2024, Globalink received a delisting determination letter from Nasdaq as the 36-month anniversary from its IPO has passed on December 9, 2024, and Globalink are no longer in compliance with Nasdaq Listing Rule IM 5101-2(b). Globalink’s securities were suspended from trading and delisted from Nasdaq on December 17, 2024. As of the same date, Globalink’s securities started trading on OTC Pink. There could be substantial risks to Globalink and PubCo the as a result of Globalink’s non-compliance with this rule. See “Risk Factors — Risks Related to Globalink’s Business and the Business Combination — Our securities were suspended from trading and delisted from Nasdaq on December 17, 2024, following receipt of a delisting determination letter from Nasdaq on December 10, 2024. This could have significant material adverse consequences on us and our securities, including that it will negatively impact our ability to complete a business combination, will limit investors’ ability to make transactions in our securities and could subject us to additional trading restrictions.”
Trust Account
Following the closing of the IPO (including over-allotment units), $116,725,000 ($10.15 per unit) from the net proceeds of the sale of the units in the IPO, over-allotment units, and the private units was placed in the Trust Account established for the benefit of the Company’s public stockholders at JPMorgan Chase Bank, N.A. maintained by Continental, acting as trustee and has been held in cash, until the earlier of: (i) the completion of an initial Business Combination and (ii) the distribution of the Trust Account. The money held in the Trust Account, if invested, may only be invested in United States “government securities” within the meaning of Section 2(a) (16) of the Investment Company Act having a maturity of 180 days or less or in money market funds meeting certain conditions under Rule 2a-7 promulgated under the Investment Company Act which invest only in direct U.S. government treasury obligations.
Through a total of five elections from March 2023 to December 2023, Globalink elected to extend the Termination Date to December 9, 2023 and deposited an aggregate of US$1.17 million into the Trust Account for its public stockholders. Globalink elected all of the five extensions permitted under the amended and restated certificate of incorporation of the Company, as first amended.
As of the date of this proxy statement/prospectus, the Company has extended the Termination Date one time under its amended and restated certificate of incorporation, as third amended (or eighteen times since the IPO), and has until January 9, 2025 to complete its initial business combination (or up to June 9, 2025 if Globalink further extends the timeline to complete its initial business combination), and has caused an aggregate of $1,950,000 to be deposited into the Trust Account in connection with these extensions.
On December 3, 2024, the Company’s stockholders approved an amendment to its certificate of incorporation, which allows the Company to continue to extend the Termination Date to up to June 9, 2025 through monthly extensions, if requested by the Board, and subject to the Sponsor depositing additional funds into the Trust Account.
On December 10, 2024, Globalink received a delisting determination letter from Nasdaq as the 36-month anniversary from its IPO has passed on December 9, 2024, and Globalink are no longer in compliance with Nasdaq Listing Rule IM 5101-2(b). Globalink’s securities were suspended from trading and delisted from Nasdaq on December 17, 2024. As of the same date, Globalink’s securities started trading on OTC Pink. There could be substantial risks to Globalink and PubCo the as a result of Globalink’s non-compliance with this rule. See “Risk Factors — Risks Related to Globalink’s Business and the Business Combination — Our securities were suspended from trading and delisted from Nasdaq on December 17, 2024, following receipt of a delisting determination letter from Nasdaq on December 10, 2024. This could have significant material adverse consequences on us and our securities, including that it will negatively impact our ability to complete a business combination, will limit investors’ ability to make transactions in our securities and could subject us to additional trading restrictions.”
| 164 |
Business Combination Activities
Agreement with Chardan
On February 4, 2022, Globalink entered into a letter agreement with Chardan pursuant to which Chardan agreed to act as Globalink’s M&A and capital markets advisor with respect to a business combination involving Globalink and a target company. Upon consummation of business combination, Chardan is entitled to M&A fee in amount of $1,000,000 and Financing fees of 6% of aggregate sales price of securities purchased in the Business Combination or related transactions by parties introduced by Chardan to Globalink. In addition, Globalink has agreed to reimburse $100,000 to Chardan for out-of-pocket expenses, subject to presentation of appropriate documentation evidencing such out-of-pocket expenses.
Subsequently, on February 2, 2024, Globalink entered into an amended agreement with Chardan. The purpose of the amended agreement was to update certain commercial arrangements relating to the engagement. The engagement was updated to include a right of first refusal (as described below) and increase the expense reimbursement from $100,000 to $250,000 to account for Chardan’s out-of-pocket expense including cost of retaining counsel.
In the February 2, 2024 amended agreement, Chardan shall have a right (but not an obligation) of first refusal to act (a) as sole/lead left/lead book-running manager (with at least 30% of the economics paid to all advisors and/or underwriters) for any and all future financings undertaken until the first (1st) anniversary of the date of the closing of the Business Combination and (b) as sole/lead left financial advisor with respect to any and all M&A transactions during such period.
| Agreement with Trunnion Bridge Sdn. Bhd. |
On June 1, 2022, Globalink entered into a letter agreement with Trunnion Bridge Sdn. Bhd. (“Trunnion”), pursuant to which Trunnion agreed to assist Globalink to identify potential target companies for a business combination and to provide general consultancy services to Globalink in connection with a business combination transaction until consummation of a business combination. As consideration for the services provided by Trunnion to Globalink under the letter agreement, Globalink agreed to issue 350,000 shares of Globalink common stock to Trunnion upon the closing a business combination. Trunnion is also entitled to reimbursement of out-of-pocket expenses.
As Alps Holdco was not introduced by Trunnion to Globalink, Globalink is not obligated under the letter agreement to issue 350,000 shares to Trunnion.
| Merger Agreement |
On January 30, 2024, Globalink entered into the Merger Agreement (as amended and restated on May 20, 2024) with PubCo, Merger Sub, Alps Holdco, the Parent Representative and Seller Representative. Pursuant to the terms of the Merger Agreement, the Business Combination between Globalink and Alps Holdco will be effected in two steps: (i) the Redomestication Merger, whereby, subject to the approval and adoption of the Merger Agreement by the stockholders of Globalink, Globalink will merge with and into PubCo, with PubCo remaining as the surviving publicly traded entity; and (ii) the Acquisition Merger, whereby Merger Sub will merge with and into Alps Holdco, resulting in Alps Holdco remaining as the surviving entity and being a wholly-owned subsidiary of PubCo. In the event that the Business Combination is not consummated by January 9, 2025 (and may be extended to June 9, 2025 on a monthly basis by resolutions of the Globalink Board, if requested by the Sponsor, and subject to the Sponsor depositing additional funds into the Trust Account), its corporate existence will cease and Globalink will distribute the proceeds held in the Trust Account to its public stockholders.
| 165 |
Redemption Rights
The public stockholders will be entitled to redeem their public shares for a pro rata portion of the amount then in the Trust Account (anticipated to be $11.98 per public share based on the balance of the Trust Account as of December 6, 2024). There will be no redemption rights with respect to Globalink’s warrants or rights.
Automatic Dissolution and Subsequent Liquidation of Trust Account if No Business Combination
If Globalink does not complete a business combination within January 9, 2025 (and may be extended to June 9, 2025 on a monthly basis by resolutions of the Globalink Board, if requested by the Sponsor, and subject to the Sponsor depositing additional funds into the Trust Account), Globalink will (i) cease all operations except for the purpose of winding up, (ii) as promptly as reasonably possible but not more than ten business days thereafter, redeem 100% of the outstanding public shares for a pro rata portion of the funds held in the Trust Account, which redemption will completely extinguish public stockholders’ rights as stockholders (including the right to receive further liquidation distributions, if any), subject to applicable law and (iii) as promptly as reasonably possible following such redemption, subject to the approval of Globalink’s remaining stockholders and Globalink Board, dissolve and liquidate, subject (in the case of (ii) and (iii) above) to Globalink’s obligations under Delaware law to provide for claims of creditors and the requirements of other applicable law. At such time, the rights will expire and holders of the rights will receive nothing upon a liquidation with respect to such rights, and the rights will be worthless.
Under the DGCL, stockholders may be held liable for claims by third parties against a corporation to the extent of distributions received by them in a dissolution. The pro rata portion of Globalink’s Trust Account distributed to Globalink’s public stockholders upon the redemption of 100% of Globalink’s outstanding public shares in the event Globalink does not complete Globalink’s initial business combination within the required time period may be considered a liquidation distribution under Delaware law. If the corporation complies with certain procedures set forth in Section 280 of the DGCL intended to ensure that it makes reasonable provision for all claims against it, including a 60-day notice period during which any third-party claims can be brought against the corporation, a 90-day period during which the corporation may reject any claims brought, and an additional 150-day waiting period before any redemptions are made to stockholders, any liability of stockholders with respect to a redemption is limited to the lesser of such stockholder’s pro rata share of the claim or the amount distributed to the stockholder, and any liability of the stockholder would be barred after the third anniversary of the dissolution.
Furthermore, if the pro rata portion of Globalink’s Trust Account distributed to Globalink’s public stockholders upon the redemption of 100% of Globalink’s public shares in the event Globalink does not complete Globalink’s initial business combination within the required time period is not considered a liquidation distribution under Delaware law and such redemption distribution is deemed to be unlawful, then pursuant to Section 174 of the DGCL, the statute of limitations for claims of creditors could then be six years after the unlawful redemption distribution, instead of three years, as in the case of a liquidation distribution. It is Globalink’s intention to redeem Globalink’s public shares as soon as reasonably possible following the 12th or 15th or 18th month from the closing of the IPO and, therefore, Globalink does not intend to comply with the above procedures. As such, Globalink’s stockholders could potentially be liable for any claims to the extent of distributions received by them (but no more) and any liability of Globalink’s stockholders may extend well beyond the third anniversary of such date.
Because Globalink will not be complying with Section 280 of the DGCL, Section 281(b) of the DGCL requires us to adopt a plan, based on facts known to us at such time that will provide for Globalink’s payment of all existing and pending claims or claims that may be potentially brought against us within the subsequent 10 years. However, because Globalink is a blank check company, rather than an operating company, and Globalink’s operations will be limited to seeking to complete an initial business combination, the only likely claims to arise would be from Globalink’s vendors (such as lawyers, investment bankers, etc.) or prospective target businesses.
| 166 |
Globalink will seek to have all third parties (including any vendors or other entities Globalink engages after the IPO) and any prospective target businesses enter into valid and enforceable agreements with us waiving any right, title, interest or claim of any kind they may have in or to any monies held in the Trust Account. The underwriters in the IPO have executed such a waiver agreement. As a result, the claims that could be made against us will be limited, thereby lessening the likelihood that any claim would result in any liability extending to the trust. Globalink therefore believes that any necessary provision for creditors will be reduced and should not have a significant impact on Globalink’s ability to distribute the funds in the Trust Account to Globalink’s public stockholders. Nevertheless, there is no guarantee that vendors, service providers and prospective target businesses will execute such agreements. In the event that a potential contracted party was to refuse to execute such a waiver, Globalink will execute an agreement with that entity only if Globalink’s management first determines that Globalink would be unable to obtain, on a reasonable basis, substantially similar services or opportunities from another entity willing to execute such a waiver. Examples of instances where Globalink may engage a third-party that refused to execute a waiver would be the engagement of a third-party consultant who cannot sign such an agreement due to regulatory restrictions, such as Globalink’s auditors who are unable to sign due to independence requirements, or whose particular expertise or skills are believed by management to be superior to those of other consultants that would agree to execute a waiver or a situation in which management does not believe it would be able to find a provider of required services willing to provide the waiver. There is also no guarantee that, even if they execute such agreements with us, they will not seek recourse against the Trust Account. Globalink’s Sponsor has agreed that it will be liable to us if and to the extent any claims by a vendor for services rendered or products sold to us, or a prospective target business with which Globalink has discussed entering into a transaction agreement, reduce the amount of funds in the Trust Account to the lesser of (i) $10.15 per public share and (ii) the actual amount per public share held in the Trust Account as of the date of the liquidation of the Trust Account if less than $10.15 per public share due to reductions in the value of the trust assets, in each case less taxes payable, provided that such liability will not apply to any claims by a third-party who executed a waiver of any and all rights to seek access to the Trust Account nor will it apply to any claims under Globalink’s indemnity of the underwriters of the IPO against certain liabilities, including liabilities under the Securities Act. In the event that an executed waiver is deemed to be unenforceable against a third-party, Globalink’s Sponsor will not be responsible to the extent of any liability for such third-party claims. However, Globalink’s Sponsor may not be able to satisfy its indemnification obligations, as Globalink has not required Globalink’s Sponsor to retain any assets to provide for its indemnification obligations, nor has Globalink taken any further steps to ensure that Globalink’s Sponsor will be able to satisfy any indemnification obligations that arise. Moreover, Globalink’s Sponsor will not be liable to Globalink’s public stockholders and instead will only have liability to us. None of Globalink’s officers or directors will indemnify us for claims by third parties, including, without limitation, claims by vendors and prospective target businesses. As a result, if Globalink liquidates, the per-share distribution from the Trust Account could be less than approximately $10.15 due to claims or potential claims of creditors. Globalink will distribute to all of Globalink’s public stockholders, in proportion to their respective equity interests, an aggregate sum equal to the amount then held in the Trust Account, inclusive of any interest not previously released to us, (subject to Globalink’s obligations under Delaware law to provide for claims of creditors as described below).
If Globalink is unable to consummate an initial business combination and are forced to redeem 100% of Globalink’s outstanding public shares for a portion of the funds held in the Trust Account, Globalink anticipates notifying the trustee of the Trust Account to begin liquidating such assets promptly after such date and anticipate it will take no more than 10 business days to effectuate the redemption of Globalink’s public shares. Globalink’s insiders have waived their rights to participate in any redemption with respect to their insider shares. Globalink will pay the costs of any subsequent liquidation from Globalink’s remaining assets outside of the Trust Account. If such funds are insufficient, Globalink’s insiders have agreed to pay the funds necessary to complete such liquidation (currently anticipated to be no more than approximately $15,000) and have agreed not to seek repayment of such expenses. Each holder of public shares will receive a full pro rata portion of the amount then in the Trust Account, plus any pro rata interest earned on the funds held in the Trust Account and not previously released to us or necessary to pay Globalink’s taxes. The proceeds deposited in the Trust Account could, however, become subject to claims of Globalink’s creditors that are in preference to the claims of public stockholders.
Globalink’s public stockholders shall be entitled to receive funds from the Trust Account only in the event of Globalink’s failure to complete Globalink’s initial business combination in the required time period or if the stockholders seek to have us convert their respective shares of common stock upon a business combination which is actually completed by us. In no other circumstances shall a stockholder have any right or interest of any kind to or in the Trust Account.
| 167 |
If Globalink is forced to file a bankruptcy case or an involuntary bankruptcy case is filed against us which is not dismissed, the proceeds held in the Trust Account could be subject to applicable bankruptcy law, and may be included in Globalink’s bankruptcy estate and subject to the claims of third parties with priority over the claims of Globalink’s stockholders.
If, after Globalink distributes the proceeds in the Trust Account to Globalink’s public stockholders, Globalink files a bankruptcy petition or an involuntary bankruptcy petition is filed against us that is not dismissed, any distributions received by stockholders could be viewed under applicable debtor/creditor and/or bankruptcy laws as either a “preferential transfer” or a “fraudulent conveyance.” As a result, a bankruptcy court could seek to recover all amounts received by Globalink’s stockholders. In addition, Globalink Board may be viewed as having breached its fiduciary duty to Globalink’s creditors and/or having acted in bad faith, thereby exposing itself and us to claims of punitive damages, by paying public stockholders from the Trust Account prior to addressing the claims of creditors. Claims may be brought against us for these reasons.
Facilities
Globalink maintains its principal executive offices at 200 Continental Drive, Suite 401, Newark, Delaware 19713. The rent for this space is $115 per month. Globalink considers Globalink’s current office space, combined with the other office space otherwise available to Globalink’s executive officers, adequate for Globalink’s current operations.
Employees
Globalink has two executive officers. These individuals are not obligated to devote any specific number of hours to its matters and intend to devote only as much time as they deem necessary to its affairs. The amount of time they will devote in any time period will vary based on whether a target business has been selected for the business combination and the stage of the business combination process the company is in. Accordingly, once a suitable target business to consummate its initial business combination with has been located, management will spend more time investigating such target business and negotiating and processing the business combination (and consequently spend more time on Globalink’s affairs) than had been spent prior to locating a suitable target business. Globalink presently expects its executive officers to devote an average of approximately 10 hours per week to Globalink’s business. Globalink does not intend to have any full-time employees prior to the consummation of Globalink’s initial business combination.
| 168 |
INFORMATION ABOUT ALPS
In this section, all references to “Alps”, the “Company”, “we,” “us” and “our” refer to Alps Global Holding Berhad, Alps Life Sciences Inc and its subsidiaries prior to consummation of the Business Combination, which will be the business of PubCo and its subsidiaries following the consummation of the Business Combination.
Glossary
For purposes of this section, unless otherwise stated or unless the context otherwise requires, the following terms shall have the following meanings:
| “CAGR” | means compound annual growth rate; |
| “CAR-T” | means chimeric antigen receptor T-cells; |
| “DNA” | means deoxyribonucleic acid; |
| “hiPSCs” | means human-induced pluripotent stem cells; |
| “KIRs” | means killer cell immunoglobulin-like receptors; |
| “iPSC” | means induced pluripotent stem cells; |
| “MHC” | means major histocompatibility complex; |
| “mRNA” | means messenger ribonucleic acid; |
| “NK” | means natural killer cells; |
| “WGS” | means whole genome sequencing; and |
| “GMP” | means good manufacturing practice. |
Mission
Our mission is to foster a fair healthcare ecosystem. Alps has taken steps to cultivate an ecosystem of collaboration among clinicians, stakeholders, and researchers who share our vision. Together, we are dedicated to expanding the range of diagnostic and treatment options available while simultaneously reducing costs, ensuring that cutting-edge medicine is more accessible.
Overview
Pursuant to the terms of the Merger Agreement, after the date of the Merger Agreement and prior to the consummation of the Business Combination, Alps Global Holding Berhad (“Alps”), will become a subsidiary of Alps Life Sciences Inc (“Alps Holdco”), a holding company incorporated on April 14, 2017 under the Malaysian Companies Act 2016 as a public limited company (“Proposed Reorganization”), which will be overseeing the operations in Malaysia. Our group will consist of Alps Life Sciences Inc, Alps Global Holding Berhad and our subsidiaries, namely, Alps Biotech Sdn. Bhd., Alps Wellness Centre Sdn. Bhd., Alpscap Berhad, Celebre Pro Medic Sdn. Bhd., Celestialab Sdn. Bhd., Mont Life (M) Sdn. Bhd., MyGenome Sdn. Bhd., TMC Global Holdings Sdn. Bhd., and Alps Insurance PCC Inc., and our affiliate, Vax Biotech Sdn. Bhd. (30% owned). We are focused on becoming a fully integrated platform for biotechnology research, medical and wellness company. Our principal activities are research and development in various biotechnology-related products and services, with a focus on the use of precision and preventive medicine, and that substantially all of our operations are conducted through Alps and our subsidiaries and affiliate.
Because all of our pipeline candidates are in the pre-clinical phase, we do not generate revenue from our research and development efforts. Rather, we provide aesthetic treatments, general healthcare and wellness services to generate revenue which is used to support and sustain our research and development activities. This revenue is crucial for covering ongoing operational expenses, funding clinical studies and developing innovative biotechnology solutions, including new product candidates and advancements in precision medicine. Alps believes that this approach ensures that Alps’ R&D activities are not only innovative but also aligned with practical market needs and trends.
| 169 |
Our corporate structure after the Proposed Reorganization as of the date of this proxy statement/prospectus is depicted below:

Figure 1. Organizational Structure of Alps.
Details of our subsidiaries are summarized below:
| Company Name | Principal Activities | Date of Incorporation | Principal place of business | |||
Alps Global Holding Berhad
|
Business of research and development (including stem cell) in all kinds of biotechnology related business, invest and manage business such as health and beauty centre, medical centre as well as acting as an investment holding company
|
April 14, 2017 | Malaysia | |||
Mont Life (M) Sdn. Bhd.
|
Skincare, aesthetic, health supplements, food and drinks supplements and household products
|
May 8, 2015 | Malaysia | |||
Celebre Pro Medic Sdn. Bhd.
|
Business of beauty, health consultant and therapist
|
October 31, 2011 | Malaysia | |||
TMC Global Holdings Sdn. Bhd.
|
Beauty and health care consultants | July 4, 2014 | Malaysia | |||
Alps Wellness Centre Sdn. Bhd.
|
Business of health screening, beauty aesthetics, and to act as wellness centre
|
August 18, 2017 | Malaysia | |||
Celestialab Sdn. Bhd. (“Celestialab”)
|
i) Acquire, hold investment and manage movable and immovable assets, landed properties. ii) Manufacture cellular products such as human stem cells and lymphocytes-products to develop, establish, maintain and aid in the development, establishment and maintenance of laboratories on medical science and biotechnology, research stations, containment facilities and programmes
|
November 27, 2017 | Malaysia |
| 170 |
MyGenome Sdn. Bhd. (“MyGenome”)
|
Manufacture of medicaments, medical laboratories, research and development on biotechnology
|
November 27, 2018 | Malaysia | |||
Alps Biotech Sdn. Bhd.(“Alps Biotech”)
|
Leasing of intellectual property and similar products, except copyrighted works, research and development on medical sciences, investment advisory services
|
May 9, 2019 | Malaysia | |||
Alpscap Berhad
|
Activities of holding companies, accounting, bookkeeping and auditing activities, tax consultancy
|
April 11, 2018 | Malaysia | |||
Alps Insurance PCC Inc.
|
Provision of unconventional healthcare financing solutions
|
December 24, 2020 | Malaysia |
Details of our affiliates are summarized below:
| Company Name | Principal Activities | Date of Incorporation | Principal place of business | |||
| Vax Biotech Sdn. Bhd. | Manufacture of medicaments | January 25, 2021 | Malaysia | |||
| Cilo Cybin Holdings Limited | Investment holding company | February 23, 2022 | South Africa |
Corporate History
We currently operate through Alps Global Holding Berhad and our subsidiaries, namely, Alps Biotech Sdn. Bhd., Alps Wellness Centre Sdn. Bhd., Alpscap Berhad, Celebre Pro Medic Sdn. Bhd., Celestialab Sdn. Bhd., Mont Life (M) Sdn. Bhd., MyGenome Sdn. Bhd., TMC Global Holdings Sdn. Bhd., and Alps Insurance PCC Inc., and our affiliates, Vax Biotech Sdn. Bhd. (30% owned) and Cilo Cybin Holdings Limited (40.5% owned).
Share Subscription Agreement with Cilo Cybin Holdings Limited
On September 15, 2023, Alps Global Holding Berhad entered into a share subscription agreement (as supplemented from time to time) (the “Cilo Cybin Subscription Agreement”) with Dr. Tham Seng Kong, Cilo Cybin Holdings Limited (the “Cilo Cybin”), and Gabriel Theron.
Under the terms of the Cilo Cybin Subscription Agreement, Alps Global Holding Berhad and Dr. Tham each subscribed for 28,762,252 of ordinary shares in Cilo Cybin (for an aggregate of 57,524,504 ordinary shares) at a price of ZAR 0.90 per share, upon the terms and conditions stated therein, constituting approximately 40.5% of the issued securities of Cilo Cybin. Cilo Cybin was listed on the JSE’s Alternative Exchange as a special purpose acquisition company on June 25, 2024, to pursue the acquisition of, and investments in, commercial enterprises operating in the biotechnology and pharmaceuticals industries. As a result, Dr. Tham and Alps Global Holding Berhad each hold a 40.5% stake in the Target Company. Cilo Cybin has identified potential opportunities for asset acquisitions both in South Africa and globally. Among these opportunities is Cilo Cybin Pharmaceutical Proprietary Limited (“Cilo Cybin Pharmaceutical”), which is considered a viable asset.
This investment aligns with Alps’ business model by providing an opportunity to collaborate and potentially integrate innovative biotechnology and pharmaceutical solutions that complement Alps’ existing focus on precision and preventive medicine. Cilo Cybin Pharmaceutical intends to develop its exclusive house brand featuring cannabis and psychedelic products. Alps believes that these products may potentially lead to future therapeutic development targeting conditions such as epilepsy and Parkinson’s disease. Alps also sees a potential for collaboration with Cilo Cybin Pharmaceutical in research and development endeavors to enable each company to leverage each other’s strengths to drive innovation and accelerate the development and commercialization of new therapeutic solutions.
Share Sale Agreement in relation to the acquisition of the shares in Alps Insurance PCC Inc.
On September 11, 2023, Alpscap Berhad entered into a share sale agreement (as supplemented from time to time) (the “SSA”) with Dr. Tham Seng Kong, Mohd Razef Bin Abdullah and Poon Kian Huat (the “Vendors”) to acquire 100% equity interest in Alps Insurance PCC Inc (“Sale Shares”). The acquisition of Sale Shares has been completed and successfully transferred from the Vendors to Alpscap Berhad.
Alps Insurance PCC Inc is a captive insurance company regulated by Labuan Financial Services Authority, specializing in providing both insurance and reinsurance services. Pursuant to the SSA, the Vendors will receive a consideration of RM686,275.00 (approximately US$ 145,397.25) upon the completion of the sale and purchase of the Sale Shares.
Share Split Involving the Subdivision of Every 1 Existing Ordinary Share in Alps Global Holding Berhad into Approximately 1.24 Ordinary Shares Each
On February 23, 2024, Alps Global Holding Berhad had, vide an Extraordinary General Meeting, resolved to approve the subdivision of 46,838,742 of the existing ordinary shares in Alps Global Holding Berhad, held by the shareholders of Alps Global Holding Berhad whose names appear in the Register of Member as at the close of business on February 23, 2024 into 58,041,998 ordinary shares in Alps Global Holding Berhad (“Subdivided Shares”).
The Subdivided Shares will, upon allotment and issuance, rank pari passu in all respects with each other, except that the Subdivided Shares will not be entitled to any dividends, rights, allotments and/or other forms of distribution that may be declared, made or paid for which the entitlement date precedes the date of allotment and issuance of the Subdivided Shares. The rationale for the share split is to provide a premium to existing shareholders through issuance of additional premium shares funded by the capital raised.
Business Milestones
Alps aspires to establish a fully integrated platform encompassing biotechnology research, medical, and wellness services specializing in predictive, precision and preventive health management. We intend to advance personalized medicine through ongoing innovation.
Alps aims to become a leading healthcare destination, offering its expertise at fair and accessible prices. Our scope of biotechnology platform extends to creating a comprehensive “bench to bedside” ecosystem. This includes both upstream activities, such as sourcing raw materials and production, and to downstream operations that cover distribution, marketing and fill-and-finish processes.
| 171 |
Situated in the heart of Kuala Lumpur’s central business district, the core establishments of Alps include Alps Medical Centre operated by our subsidiary, TMC Global Holdings Sdn. Bhd. while Alps Wellness Centre is operated by Alps Wellness Sdn. Bhd., offering a range of medical, aesthetic, and wellness services. Additionally, MyGenome operates a molecular laboratory and has been granted with a BioNexus status by the Malaysian government through the Malaysian Investment Development Authority (MIDA). The BioNexus status affords MyGenome with incentives and benefits, among others, an income tax exemption of 70% on statutory income for a period of ten (10) years. MyGenome’s molecular laboratory accredited by the Malaysian Standard MS ISO 15189:2022, Medical laboratories – Requirements for quality and competence, and specializes in genetics sequencing including WGS, leveraging expertise in DNA and mRNA analysis. Celestialab, our subsidiary, focuses on the cultivation, research and development of cellular products. Both MyGenome and Celestialab contribute to our precision and preventive ethos aimed at enhancing individual health and well-being.
Furthermore, we collaborate with universities for research and development to develop product pipelines that align with our company’s mission. Alps’ initiatives of creating a bench-to-bed platform with the competitive advantage and positioning set in place, allows the adoption of high technology pipelines at a low or conservative burn rate.
Our Research and Development
Leveraging on our network comprising professional providers, suppliers, and supporting services, Alps aims to provide its clientele with access to DNA and mRNA sequencing, cellular therapy, as well as wellness and anti-aging products and services.
Alps possesses in-house laboratories capable of performing a range of genomic and cellular research, including WGS, Whole Exome Sequencing (WES), targeted sequencing, mRNA tests. Alps also operates a cGMP accredited cell manufacturing laboratory. WGS, known as the most comprehensive clinical-grade DNA test available, provides invaluable insights into an individual’s inherited genetic predispositions. By unraveling the entire genetic makeup, this sequencing offers a wealth of information that can shape personalized medicine and enhance treatment efficacy.
In the realm of diagnostics and therapeutics, mRNA gene expression profiling is rapidly becoming a game-changer by acting as molecular biomarkers. These biomarkers are crucial for the early, accurate prediction and diagnosis of diseases, heralding a more proactive stance in healthcare management. mRNA tests provide a window into gene expression changes as they occur, facilitating prompt medical interventions to preempt disease progression.
Alps is committed to integrate WGS and mRNA expression profiling into routine diagnostic practices and to forge new paths in personalized medicine. Looking ahead, we aim to further enhance our service offerings by integrating the production and cultivation of autologous stem cells directly within our facilities. Additionally, we intend to establish an international research and development hub, collaborating with renowned scientists and institutions to advance cellular therapy as therapeutic entities.
| 172 |
Our Pipeline
We are advancing a broad portfolio of pre-clinical product candidates derived from the following drug classes focused on the treatment of cancer, infectious and non-communicable diseases:

Figure 2. Overview of product candidates’ pipeline.
* The timelines presented in Figure 1 for Phase 1, Phase 2, and Phase 3 clinical trials are derived from industry standards and typical study durations for comparable trials. These estimates are based on standard timelines but may vary due to factors such as patient recruitment rates, trial design complexities, and regulatory requirements. Alps relies on historical data and industry benchmarks to project these durations. It is important to note that these timelines are preliminary and may be subject to adjustments. Various factors, including regulatory review periods, patient recruitment challenges, trial execution, and unforeseen variables during the development process, could potentially influence these timelines.
** The bottom half of the pipeline table, specifically, the mRNA diagnostics for lung cancer, cervical cancer, breast cancer and colorectal cancer, as well as exosome applications for cosmetic and wound care, represent product candidates that are designed to be used as diagnostic tools and cosmetic treatments rather than therapeutic interventions. As such, they follow a different regulatory pathway. In Malaysia, regulatory approval for these types of products may not necessitate the same extensive pre-clinical or clinical testing required for new therapeutic drugs. Instead, these products must meet specific regulatory standards and provide sufficient statistical evidence of safety and efficacy through documentation and studies appropriate for regulatory approval.
Details of our pipeline development stages and next anticipated milestones are summarized below:
| No. | Pipeline | Current Phase | Next Anticipated Milestones | |||
| 1. | Natural Killer (“NK”) Cell Therapy–MyImmune | The POC for NK cell therapy comprises of discovery, optimization, and process validation phase. Celestialab has completed the optimization phase, which aimed at culturing NK cells to achieve high-yield and high-purity. Currently, Celestialab is preparing to commence the process validation phase, which will include the completion of consecutive process replication and media-fill tests. |
Upon completing the process validation phase of the POC, Celestialab will initiate pre-clinical trials. This phase will involve designing the trials, determining efficacy parameters, and conducting comprehensive studies on toxicology, dosage, and safety. | |||
| 2. | Chimeric Antigen Receptor (“CAR”)-T Cell | Celestialab has concluded the discovery phase of its POC, identifying (i) CD19, (ii) CD20, or (iii) dual targeting of CD19 and CD22, as key targets for B-cell acute lymphoblastic leukemia and B-cell lymphoma. Celestialab is now preparing to start the optimization phase, which will include creating plasmid DNA (“pDNA”) templates. |
Upon completing the optimization phase, Celestialab will initiate the validation phase for the pDNA templates targeting CD19 and/or CD20. | |||
| 3. | COVID-19 Vaccine (mRNA) | The POC for COVID-19 Vaccine (mRNA) consists of (i) plasmid DNA (“pDNA”) construct design, (ii) synthesis and verification, (iii) optimization, and (iv) validation and formulation phase. MyGenome is now conducting in vitro tests on the pDNA constructs, including pDNA transfection into mammalian cells for protein expression study, and lab-scale production and formulation. |
After completing in vitro validation and formulation stage, Alps will begin in vivo POC testing for the pDNA constructs. | |||
| 4. | Cholera Vaccine | VaxBio has completed the POC for its cholera vaccine and is moving to the preclinical stage. VaxBio currently conducting a preclinical acute toxicity study at Malaysian Institute of Pharmaceuticals and Nutraceuticals (“IPHARM”) to assess the vaccine’s safety.
|
After completing the preclinical acute toxicity study, VaxBio plans to submit a dossier to the NPRA to seek approval for starting clinical trials. | |||
| 5. | Diabetes – MYCELEST | Celestialab is completing the preclinical studies phase for MYCELEST and is currently preparing the preclinical data for submission of the dossier application to the NPRA. |
Once Celestialab obtains the necessary approvals from NPRA, Celestialab will initiate clinical trials on patients with diagnosed type 1 and type 2 diabetes mellitus. | |||
| 6. | Heart Failure – iPSC | This pipeline has implemented and proved the construct/design of the product candidate, which involved the differentiation of human induced pluripotent stem cells (iPSCs) into cardiomyocytes using cord blood, which was a step in validating the approach. Additionally, this pipeline completed the study to test the therapeutic potential of these iPSC-derived cardiomyocytes by applying them to treat cryoinjured rat hearts (manuscript under preparation)., The current focus involves generating human iPSC cells from adult peripheral blood and manufacturing clinical-grade iPSCs under GMP conditions. |
Upon completion of generating human iPSC cells, the next step will involve conducting pre-clinical trials. This phase includes preparing the trial design, and conducting studies to assess efficacy, toxicology, dosage, and safety. | |||
| 7. | mRNA (Diagnostic) | This pipeline is currently undergoing the POC stage, focusing on recruiting patients for breast, lung, cervical, and colorectal cancers and collecting data to develop diagnostic kits for these cancers. |
Upon completing the data gathering phase, MyGenome will commence the development of diagnostic prototypes for the identification of differentially expressed genes.
| |||
| 8. | CELESOME(+) | Alps has completed its POC phase, which involved evaluating and refining exosome isolation methods and characterization. Alps is now finalizing an in-house protocol for further exosome characterization. |
After characterizing the exosomes, Alps plans to conduct toxicity studies, including cytotoxicity tests and acute toxicity studies in animal models.
|
MyImmune - NK Cell Therapy
Development Status of NK Cells Therapy
Celestialab is currently conducting research and development on MyImmune with the aim of enhancing its effectiveness and safety parameters before progressing to pre-clinical testing. The POC for NK cell therapy comprises of discovery, optimization, and process validation phases. Celestialab has completed the optimization phase, which is aimed at culturing NK cells to achieve high-yield and high-purity. Currently, Celestialab is preparing to commence the process validation phase, which will include the completion of consecutive process replication and media-fill tests. Upon completing the process validation phase of the POC, Celestialab will initiate pre-clinical trials. This phase will involve designing the trials, determining efficacy parameters, and conducting comprehensive studies on toxicology, dosage, and safety. Upon successful completion of pre-clinical testing, MyImmune expects to advance to clinical trial phases. These trials will involve human subjects and will assess the therapy’s safety, dosage, and effectiveness in treating targeted conditions. Celestialab anticipates completing the pre-clinical trials by the end of the third quarter of 2025. Based on this timeline, we expect the transition to clinical trials to occur within one (1) year from the date of this proxy statement/prospectus.
Overview
Natural killer (NK) cells, a vital component of the lymphocyte group, play a pivotal role in the innate immune system. These cells are instrumental in inducing apoptosis in infected or abnormal cells without the need for prior sensitization to specific antigens. NK cells, integral to the human innate immune response, possess the unique ability to selectively identify and eliminate cancerous or diseased cells. This capability is distinguished from that of the adaptive immune response system’s cells, such as T cells, which necessitate prior sensitization or co-stimulation to activate. The autonomous activation of NK cells, without the requirement for prior exposure to their targets, earns them the designation of “natural” killers. This intrinsic property enables NK cells to rapidly initiate an immune response within the human body, contrasting with the adaptive response where T cells may require a week or more to proliferate to significant numbers capable of mounting an effective defense against new antigens.
The potential of NK cell therapy to treat disease and cancer
NK cell therapy holds significant promise in the treatment of various diseases due to the unique properties and functions of NK cells. These cells are a critical component of the innate immune system and play a vital role in recognizing and eliminating abnormal or infected cells in the body. The potential of NK cell therapy to treat disease lies in its ability to harness the natural cytotoxicity of NK cells to target and destroy cancer cells, viral infected cells, and other diseased cells while preserving normal tissue integrity. Ongoing research and clinical trials are expected to further elucidate the therapeutic potential of NK cell therapy and its application across a wide range of diseases.
One of the key advantages of NK cell therapy is its broad specificity, allowing NK cells to recognize a wide range of target cells without the need for prior sensitization or specific antigen recognition. This innate recognition enables NK cells to rapidly respond to threats and initiate immune responses against various diseased and abnormal cells.
One example of the potential of NK cell therapy is its application in cancer treatment. NK cells have shown efficacy in targeting a variety of cancers, including leukemia, lymphoma, and solid tumors. For instance, in acute myeloid leukemia (“AML”), NK cell therapy has demonstrated the ability to selectively kill leukemia cells while sparing healthy hematopoietic stem cells. Clinical trials investigating the use of NK cell therapy in hematological malignancies have shown promising results, with some patients achieving complete remission or prolonged disease stabilization. NK cell therapy offers a potential alternative or complementary approach to conventional cancer treatments, such as chemotherapy and radiation therapy, and may overcome limitations such as tumour heterogeneity and resistance to treatment.
Beyond cancer, NK cell therapy is being investigated for the treatment of viral infections and anti-aging. NK cells play a crucial role in controlling viral infections and clearing senescent cells, which contribute to aging and age-related diseases. Studies have shown that NK cells can target and eliminate senescent cells, reducing inflammation and promoting healthier cellular environments.1
1 Please refer to the study on the role of NK cells in anti-aging therapies available on Frontiers in Immunology, https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2024.1360687/full
| 173 |
Global Market Analysis of NK Cells Therapies
The global NK cells therapeutics market reached a value of US$2.1 billion in 2021 and is projected to expand to US$5.1 billion by 2027, with a CAGR of 15.94% during the period 2021-2027.2 The increasing adoption of NK cells for treating cancer, infections, and liver diseases is a primary driver of market growth.
NK cells play a crucial role in tumor immune-surveillance, leading to their widespread adoption. Additionally, growing awareness among the population about various immunotherapies and increased research and development for bi-specific antibodies that engage NK cells in eliminating tumor cells further propel market expansion.
In 2022, there were an estimated 20 million new cancer cases and 9.7 million deaths. This trend is expected to continue and increase, leading to maintain this trend due to the rising adoption of NK cell therapies for various cancers, including AML, Hodgkin’s lymphoma, hematologic malignancies, solid tumors, cutaneous T cell lymphoma, and head & neck cancer. However, the gastrointestinal disease segment is anticipated to witness rapid growth due to the increasing incidence of gastrointestinal tract-related diseases.
In hospitals, NK cell therapies are employed for cancer treatment through genetic engineering. With the capability to produce NK cells in an accredited environment, we believe that we are well-positioned to capitalize on this growing market opportunity.
Key Challenges to Existing Treatment
Diseases are characterized by complex biological interactions, which diminish the effectiveness of existing therapeutics that are restricted to single mechanisms of action and are incapable of adapting to the dynamic nature of disease progression. We have identified the following key challenges:
| ● | The immune systems of cancer patients are often weakened, impairing the body’s natural defenses and diminishing the effectiveness of treatments. For example, chemotherapy can suppress the immune response, rendering the patient more susceptible to opportunistic infections. | |
| ● | The current application of chemical therapies, like chemotherapy and radiotherapy agents, often results in significant harm to the patient’s body. These treatments lack specificity, affecting not only diseased or cancerous cells but also healthy cells throughout the body. As a result, patients frequently experience severe side effects including persistent body pain, hair loss, and weight loss. | |
| ● | The pharmacodynamics of each patient renders it impractical for one form of generalized drug or treatment to be effective in treating diseases. For example, adverse drug reactions occur when some patients develop allergic reactions to the same drug in which others have not showed any side effects (e.g. sulperazone that can induce skin and eye inflammations among some patients). |
Our Strategy and Solution - MyImmune
We aim to develop autologous NK cell therapy – MyImmune to overcome the limitations identified in previous approaches by concentrating on the optimization of parameters that we believe are critical for MyImmune to drive clinical and commercial success. These optimization parameters include, but are not limited to:
| ● | a high purity and viability rate of NK cells to maximize therapeutic efficacy; | |
| ● | ensuring the production process is scalable and cost-effective for widespread clinical use; and |
| ● | the ability to generate NK cells using cGMP processes at a commercial scale. |
Immunotherapy is regarded as the current, state-of-the-art weapon in combating cancers, both solid (e.g. breast cancer, colon cancer and ovarian cancer) and liquid tumors (e.g. leukemia). In this approach, autologous mode of treatment which uses/isolates the patients’ own immune cells, augmenting/expanding them in-vitro and re-infusing them back to the patients, represent the safest manner of immunotherapy. Although there exist many different human immune cells that are utilized for autologous immunotherapy, NK cells are widely sought-after as these immune cells are known to be capable of targeting diseased cells (i.e. cancer, viral-infected, unhealthy, damaged cells) without a directed immune sensitization or immunological priming, such as through the MHC-antigen presentation route. There happens to be several different routes of culturing and expanding NK cells in-vitro, such as using multiple combinations of cytokines, prior NK cell isolation through magnetic beads, antibodies which stimulate the cell expansions or using specific-growth medias which may exponentially increase end-user’s therapy cost. Hence, the need for a cost-effective, easy-to-handle and cGMP-compliant methodology that can yield higher folds of NK cell production and concurrently with high purity of NK cells per batch.
2 Business Wire. (July 4, 2022). Global Natural Killer Cells Therapeutics Market 2022 to 2027 - Industry Trends, Share, Size, Growth, Opportunity and Forecasts. Retrieved from Business Wire.
| 174 |
We are developing MyImmune for the potential treatment of oncological diseases based on activated NK cells. Within our MyImmune therapy protocol, blood samples will be obtained from the patient, from which NK cells will be isolated and purified. These NK cells will undergo a multiplication process through culturing and stimulation for a duration of three (3) weeks in the presence of cytokines. Subsequently, the expanded NK cells will be reinfused back into the patients.
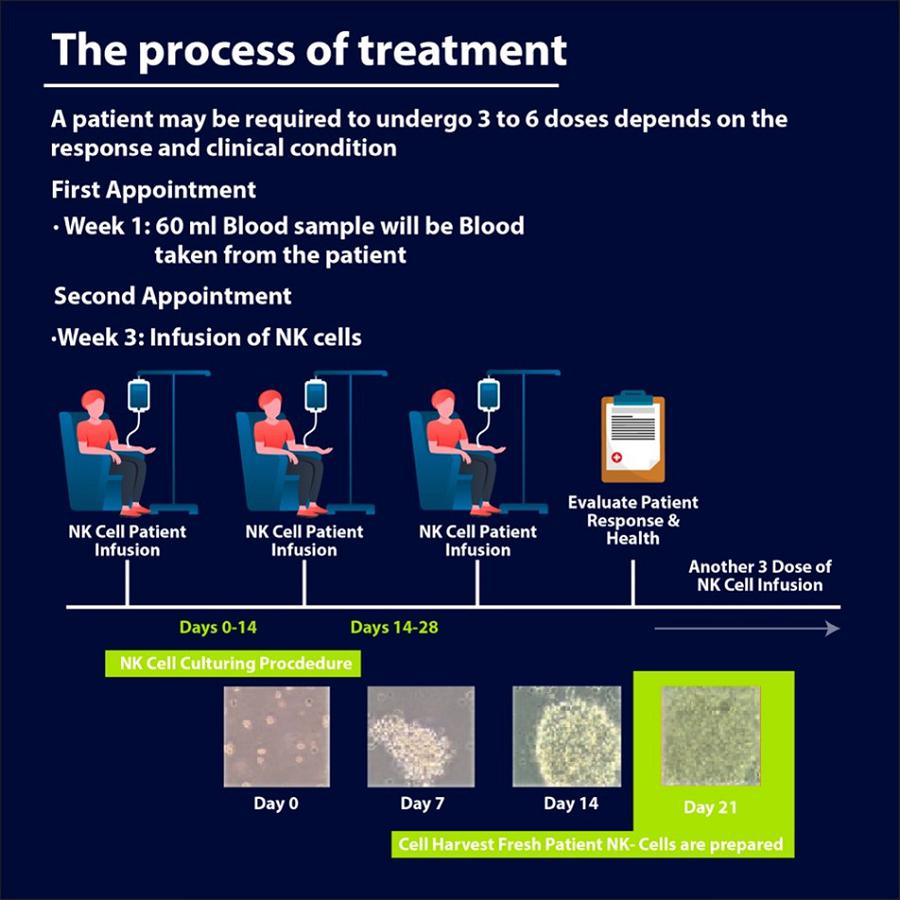
Figure 3. Overview of the autologous process of isolating, culturing, and treating patients with NK cells therapy.
Healthy cells present peptides derived from their internal proteins on their surface via MHC class I molecules. Upon encountering these molecules, NK cells utilize inhibitory receptors, including KIRs and CD94/NKG2A, to recognize and engage with them. This binding transmits inhibitory signals to the NK cell, thereby preventing it from destroying the healthy cell.
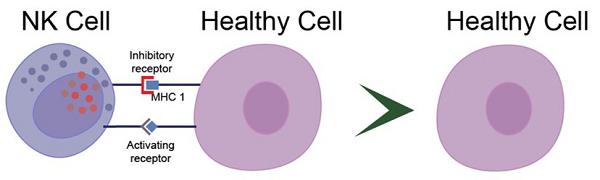
Figure 4. Mechanism of NK cells discrimination against healthy cells.
| 175 |
Cancer cells frequently exhibit a deficiency in MHC class I molecules on their surface. This reduced expression of MHC class I serves as a trigger for the activation of NK cells, which then proceed to target and eliminate the cancerous cells.
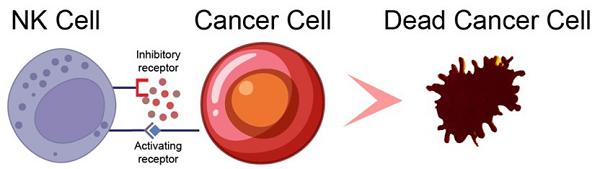
Figure 5. NK cell-induced apoptosis of a cancer cell.
The innovation presented herein describes a streamlined, cost-effective, and fully compliant methodology under cGMP for the production of NK cells with high purity and yield. This method produces cultured NK cells characterized by the expression of CD56+ and CD16+ markers and the absence of CD3, confirming the purity of the cell populations. The methodology employs interleukin-2 (IL-2) exclusively as the cytokine inducer and activator, yielding NK cells that maintain stable cytotoxicity against cancer cells and also a selective thermal induction step which induced the specific growth of NK cell populations, resulting in a NK cell batch with high purity. Our developed protocol eschews the need for prior selective isolation of NK cells from peripheral blood mononuclear cells (PBMCs) using antibodies, isolation kits, or magnetic bead-based purification methods. Furthermore, it avoids the use of antibody stimulants, antibody-coated flasks or plates, and additional cytokine combinations that could significantly increase the production costs of cGMP-grade cytokines. Instead, the protocol utilizes flask-based cultures, which eliminate the necessity for sophisticated bioreactors and antibiotics. Aseptic techniques are strictly followed, supported by sterility testing results that confirm the absence of contamination or toxins.
Proof-of-concept study of NK Cells Culture

Figure 6. Show an image of NK cells cluster formations cultured at magnification of 40X taken on the day of harvest, which conducted by Celestialab.
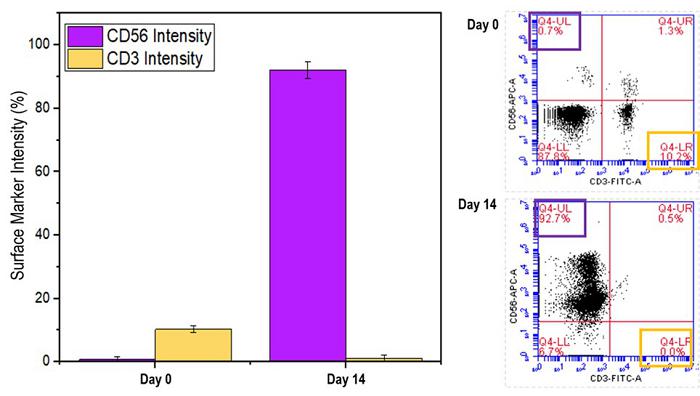
Figure 7. Shows the intensity of surface markers that indicate the presence of high-pure NK cells during product release.
| 176 |
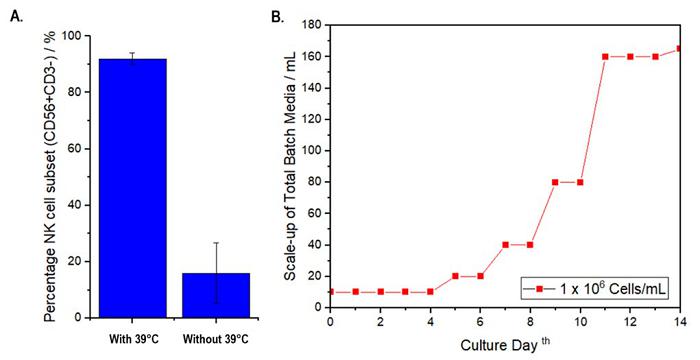
Figure 8 (A). Shows the percentage of NK cell subsets of CD56+CD3- with and without temperature-induction. Figure 8 (B). Depicts the yield of cultured cells through a period of 14 days.
The culture of NK cell at the end of batch production can be confirmed through both morphological and cell biology indications. From Figure 6, we can see that cellular clumping’s can be identified when viewed under the microscope. This translates to the presence of NK cells which have the natural ability to form clumping formations. From Figure 7, we have analyzed the surface maker CD56 which is used to identify the existence of NK cell populations while CD3 markers are used as negative indicators that corroborate to the existence of T-cells or non-relevant cells. The results indicate a high intensity of the CD56 marker which exceed more than 90% of the total cell population while the CD3 markers remains less than 1%, hence proving the purity of the NK cells cultured.
Furthermore, our optimizations shown in Figure 8 (A), reveal that our innovative approach of inducing a temperature of 39°C yields high-purity NK cells exceeding 90%, characterized by the phenotype CD56+CD3-. While without the temperature induction, the NK cell purity varies largely and falls below 50%. Figure 8 (B) also illustrated the media scale up of the culture immune cells or NK cells throughout a period of 14 days. The media is scaled up at an interval of 2 days by adjusting the cell density back to 1 million cells/ml facilitating media scale-up and high NK cell yield.
Competitive Analysis Industry Leaders
Incyte Corporation and Fate Therapeutics are notable industry leaders in pharmaceutical and cellular immunotherapy, respectively. While these companies focus on oncology and immune disorders, Alps aims to carve its niche in autologous NK cell therapy, offering a unique approach to disease treatment.
CAR-T Cell
Our subsidiary, Celestialab operates a manufacturing facility in accordance with cGMP standards and intends to equip such facility with the capability to manufacture CAR-T cells at a large scale. Our immediate focus is on advancing our CAR-T pipeline through clinical development stages. Celestialab has concluded the discovery phase of its POC, identifying CD19, CD20, and dual targeting of CD19 and CD22 as key targets for B-cell acute lymphoblastic leukemia and B-cell lymphoma. Celestialab is now preparing to start the optimization phase, which will include creating pDNA templates. Upon completing the optimization phase, Celestialab will initiate the validation phase for the pDNA templates targeting CD19 and CD20. Based on the foregoing developments, , we intend to conduct preclinical trials targeting CD19, CD20 and dual targeting of CD19 and CD22in chronic B cell leukemias.
Celestialab currently manufactures and produces umbilical cord-derived mesenchymal stem cells, NK cells, exosomes and induced pluripotent stem cells. Building on this experience, Celestialab also intends to develop and produce CAR-T cells using lentiviral vectors to introduce the genes encoding the CAR into the patient’s T cells, enabling them to recognize and target specific cancer cells. Although capable of producing CAR-T cells, Celestialab does not yet produce CAR-T cells at a large scale. Celestialab plans to commence full scale CAR-T cells production within 12 to 18 months following the Closing, eventually incorporating fully automated closed-system production technology. The planned timing to commence full-scale CAR-T cell production within 12 to 18 months following the Closing is underpinned by its existing infrastructure and expertise in manufacturing a variety of cell products. Celestialab’s experience in operating a CPC with cGMP facilities, which includes the necessary clean rooms, laboratory equipment, and controlled environment facilities, demonstrates its capability to adapt these resources for CAR-T cell production. Our strategy also involves developing CAR vectors specifically targeting CD and creating bispecific target CAR-T therapies, aiming to establish valuable in-house intellectual property. Additionally, we plan to design a viral-free delivery system for CAR-T cells using electroporation technology. This approach is intended to enhance the safety and efficacy of CAR-T cell therapies while potentially offering a unique solution in the field.
Our Strategy
Our objective is to commercialize CAR-T and related cell therapies for treating hematologic malignancies and solid tumors. Our strategy to achieve this goal is as follows: -
| ● | Advance our CAR-T pipeline into clinical development. Our immediate focus is on advancing our CAR-T pipeline through clinical development stages. Our CAR-T product candidate is currently undergoing POC validation and, subject to NPRA regulatory process for review of applications, we intend to conduct clinical trials targeting CD19 and CD20 within 12 months from the Closing, thereby progressing towards regulatory approval and eventual market entry. |
| ● | Expand our manufacturing capabilities: We currently have a manufacturing facility in Malaysia supplying clinical materials for our upcoming clinical trials. As we prepare for the potential commercialization of our products, we plan to further expand the commercial-scale manufacturing capacities by incorporating fully automated closed-system production technologies at our facility. This expansion is expected to enable rapid scale-up capabilities and ensure a steady supply of our products at a commercial scale, facilitating a smoother transition from clinical trials to market availability. |
Background of Cancer and CAR-T Cell Therapy
Cancer is characterized by the uncontrolled proliferation of abnormal cells. Cancer cells contain mutated proteins and may overexpress other proteins normally found in the body at low levels. The immune system typically recognizes abnormal protein expression and eliminates these cells in a highly efficient process known as immune surveillance. Cancer cells’ ability to evade immune surveillance is a key factor in their growth, spread, and persistence. There has been substantial scientific progress in countering these evasion mechanisms using immunotherapies, or therapies that activate the immune system. Immunotherapies are increasingly recognized as an important part of today’s frontier in the treatment of cancer.
The fundamental elements of the cellular aspect of the adaptive immune response revolve around T cells, named for their typical maturation site in the thymus. T cells play pivotal roles in detecting and eliminating infected or irregular cells, while also orchestrating the activation of other immune cells. These T cells can be categorized into two main subsets: CD4+ T cells and CD8+ T cells, distinguished by the presence of CD4 or CD8 glycoproteins on their cell surfaces. CD4+ T cells, also known as helper T cells, typically play a crucial role in orchestrating the immune response by amplifying the activation, proliferation, movement, and functional capabilities of various immune cell types. On the other hand, CD8+ T cells, referred to as cytotoxic T cells, possess the ability to directly target and eliminate cells identified as infected or irregular, with support from CD4+ T cells. Activation of both T cell types occurs when their T cell receptor interacts with and binds to a particular protein configuration displayed on the surface of another cell.
CAR-T cell therapy is a form of cancer immunotherapy, whereby a patient’s T cells are engineered to express a CAR that recognizes and binds to tumor cell surface antigens, resulting in their activation to target cancer cells for destruction. CAR-T signifies a type of cell therapy, or immunotherapy, harnessing genetically modified T cells sourced from the patient to combat cancer. CAR-T cell therapy has emerged as a revolutionary and potentially curative therapy for patients with certain hematologic cancers.
| 177 |
There are a number of FDA approved CAR-T cell therapies. This includes (A) Breyanzi (lisocabtagene maraleucel), which has been approved by the FDA for treatment of adult patients with r/r large B-cell lymphoma (LBCL) after two or more lines of systemic therapy; (B) Kymriah (tisagenlecleucel), which has been approved by the FDA for treatment of patients up to 25 years of age with r/r B-cell precursor acute lymphoblastic leukemia (“ALL”) that is refractory or in second or later relapse and adult patients with r/r LBCL after two or more lines of systemic therapy; (C) Tecartus (brexucabtagene autoleucel), which has been approved by the FDA for treatment of adult patients with r/r mantle cell lymphoma and adult patients with r/r ALL; and (D) Yescarta (axicabtagene ciloleucel), which has been approved by the FDA for treatment of adult patients with r/r LBCL after two or more lines of systemic therapy and adult patients with r/r follicular lymphoma after two or more lines of systemic therapy.
Market Analysis of CAR-T
The global CAR-T market is projected to witness a Compound Annual Growth Rate (CAGR) of 30.6%.3 Initially pioneered for the treatment of acute lymphoblastic leukemia (ALL) and B Cell Lymphoma, CAR-T therapy has strategically shifted its focus towards tackling challenging cancer indications, particularly solid tumors. The surge in cancer incidence rates, coupled with robust product pipelines, is anticipated to propel the global CAR-T cell therapy market forward.
Cancer stands as a pervasive cause of mortality worldwide. The majority of cancer cases stem from lifestyle-related factors such as alcohol and tobacco consumption, alongside dietary elements like nitrites and polyaromatic hydrocarbons. According to the World Health Organization (“WHO”), the global cancer burden is increasing, with an estimated 19.3 million new cancer cases and 10 million cancer deaths in 2023.4 The WHO article projects that by 2050, the global cancer burden will continue to increase significantly. The report estimates that new cancer cases could rise to 29.5 million annually, with 16.3 million cancer deaths. This projection underscores the urgent need for enhanced cancer control measures, including prevention, early detection, and access to treatment and palliative care, particularly in low- and middle-income countries. Key market players are heavily invested in research and development endeavors aimed at pioneering novel CAR T-cell therapy products tailored to combat various cancer types. This pursuit of innovation serves as a pivotal driver fueling the growth trajectory of the global CAR-T cell therapy market.
The landscape of CAR-T clinical research predominantly revolves around hematological cancers, accounting for 57% of ongoing investigations. Other targeted areas include the central nervous system (8%), gastrointestinal tract (6%), skin (5%), genitourinary system (4%), breast (4%), gynecologic system (4%), respiratory system (3%), sarcomas (2%), mesothelioma (2%), and miscellaneous cancers (5%).
Despite being in its infancy, CAR-T cell therapy continues to yield promising clinical outcomes. Notably, a systematic literature review by Shah et al. (2020), titled ‘Real-world evidence of tisagenlecleucel for pediatric acute lymphoblastic leukemia and non-Hodgkin lymphoma’, as published in Blood Advances, examined the effectiveness of tisagenlecleucel treatment. The review revealed that over 85% of children achieved complete remission immediately after treatment, and approximately 60% maintained a cancer-free status even 12 months post-treatment.
As research and development efforts intensify and clinical trials progress, the outlook for CAR-T therapy remains promising, offering renewed hope for patients grappling with various forms of cancer worldwide.
3 GlobeNewswire. (October 12, 2022). CAR-T cell Therapy Market Riding on the Wave of Growth and will Grow at a CAGR of 30.6% to 2031: TMR Study. Retrieved from https://www.globenewswire.com/en/news-release/2022/10/12/2533044/0/en/ CAR-T-cell-Therapy-Market-Riding-on-the-Wave-of-Growth-and-will-Grow-at-a-CAGR-of-30-6-to-2031-TMR-Study.html
4 World Health Organization. (February 1, 2024). Global cancer burden growing, amidst mounting need for services. Retrieved from https://www.who.int/news/item/01-02-2024-global-cancer-burden-growing--amidst-mounting-need-for-services
| 178 |
Key Challenges
The principal challenges stemming from conventional treatments are delineated as follows:
| ● | Relapse and Refractory Cases in Acute Lymphoblastic Leukemia and B Cell Lymphoma Patients. Addressing the recurrence and resistance observed in patients with acute lymphoblastic leukemia and B Cell Lymphoma poses a significant hurdle. Finding effective therapeutic approaches to combat relapse and refractory cases remains paramount in improving patient outcomes. | |
| ● | Limited Responsiveness to Standard Treatments in Acute Lymphoblastic Leukemia Patients. Many patients afflicted with acute lymphoblastic leukemia exhibit a lack of response to conventional or readily available treatments. Overcoming this challenge requires the development of innovative therapies capable of eliciting favorable responses in otherwise unresponsive patients. | |
| ● | Organ Shortage for Transplantation. The scarcity of organ donors presents a persistent obstacle in the realm of transplantation. The difficulty in sourcing organs for transplantation necessitates alternative strategies or technologies to alleviate the burden on patients awaiting organ transplants. |
Our Position in CAR-T Therapy
The biotechnology and pharmaceutical industries are increasingly engaged in T cell engineered therapies and sponsoring clinical trials. The production of CAR-T cells involves the utilization of lentiviruses in a process known as genetic engineering. Lentiviral vectors are commonly employed to introduce protein-encoding cDNAs. In the realm of gene therapy, lentiviral vectors have proven successful in modifying hematopoietic stem cells, leading to restored immune function or the correction of defects in hemoglobin, among other applications. The efficacy of CAR-T cells in treating B cell leukemias and lymphomas has been particularly noteworthy, with this approach heavily relying on lentivirus-mediated gene transfer.
The cell therapy industry in Malaysia is in its early stages, with only hospitals and biopharmaceutical companies currently involved in cellular immunotherapy for cancer treatment. We are inspired by the proven clinical efficacy of existing CAR-T cells in addressing hematological malignancies. In our pursuit, we are actively sourcing lentiviral vectors with potential partners and aiming to manufacture and produce CAR-T cells that are cost-effective and widely accessible. In line with this vision, we are strategically constructing our own pipeline to focus on revolutionizing medicine by re-engaging the body’s immune system to treat cancer through CAR-T therapy that will recognize and kill cancer cells.
We aim to develop a solution utilizing autologous CAR-T therapy to combat B cell leukemias. Our approach harnesses the power of existing CAR technology to stimulate a patient’s own T cells to target cancer cells, empowering them to target and destroy cancer cells with precision and efficacy. Autologous CAR-T therapy represents a personalized treatment strategy tailored to each patient’s unique biological makeup. By utilizing the patient’s own immune cells, we minimize the risk of rejection and maximize the potential for a targeted immune response against cancer.
| 179 |
As depicted below, our process initiates with the collection of the patient’s white blood cells, followed by the selection and activation of specific T cells. We will then genetically modify these cells to express CARs tailored to tumor-associated antigens. The gene sequences for the CAR construct are integrated into the T cell DNA via a lentiviral vector. These engineered CAR-T cells are subsequently expanded and reintroduced into the patient, where they target and destroy cancer cells while preserving healthy tissues.
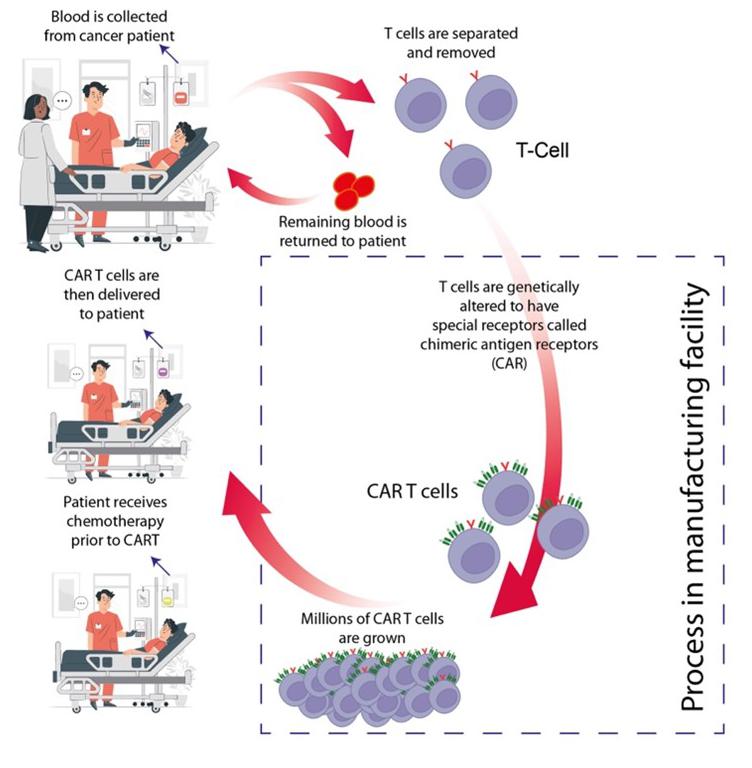
Figure 9. Diagrammatic representation of the CAR-T Cell Therapy Workflow.
| 180 |
Our solution not only leverages the proven effectiveness of CAR technology but also addresses key challenges associated with traditional cancer treatments, such as systemic toxicity and tumor heterogeneity. By precisely targeting cancer cells while minimizing damage to healthy tissues, autologous CAR-T therapy offers an avenue for improving patient outcomes and advancing the field of oncology.
To advance our product pipeline, we aim to develop three (3) CAR-T therapies targeting B-cell acute lymphoblastic leukemia (B-ALL) and B-cell lymphoma. Our CAR-T therapy clinical trial designs will target: (1) CD19, (2) CD20, and (3) dual targeting of CD19 and CD22, which Celestialab is in the POC stage. CAR-T therapy has demonstrated significant efficacy in targeting specific antigens on the surface of cancer cells, particularly in haematological malignancies5. CAR-T therapy targeting CD19 involves engineering T-cells to express receptors that specifically bind to the CD19 antigen, which is prevalent on the surface of B-cells. This approach has replicated in Malaysia6 and has shown high effectiveness in treating conditions such as ALL7 leading to substantial remission rates and prolonged responses in many patients.
Similarly, CAR-T therapy targeting CD20 operates on a comparable mechanism but focuses on the CD20 antigen. This therapy is beneficial for patients with CD19-B-cell malignancies or those who relapse after CD19 CAR-T therapy. Clinical trials conducted have shown positive results for CD20-targeted CAR-T therapy, providing alternative or complementary treatment option to CD19-targeted approaches.
To address CD19 antigen loss in haematological malignancies, we also aim to explore dual targeting of CD19 and CD22. Dual CAR-T therapies have shown promising efficacy in relapsed or refractory B-ALL, both alone and in combination with CD19 CAR-T cell therapies. Celestialab has initiated the POC stage for an autologous dual CAR T-cell targeting both CD19 and CD22 antigens, to overcome current mechanisms of resistance to CAR T-cell therapy in B-ALL, while providing a potential alternative to CD19 directed therapy.
Celestialab operates a Cell Processing Centre (CPC) with cGMP facility, and is our intention for this CPC to be well positioned to excel in the production of CAR-T cells.
Our Strength
| ● | cGMP-compliant Facility: Celestialab is the only facility within Alps that operates with cGMP certification. Celestialab adheres to strict cGMP guidelines to ensure the quality and safety. Equipped with industry-standard equipment and technology, Celestialab has the capability to produce CAR-T cell products efficiently and in compliance with regulatory requirements. |
| ● | Expert Scientific Support: Celestialab has assembled a team of experienced scientists and technicians that can provide expert scientific support throughout the manufacturing process. This pipeline is led by Dr Heilly Chong @ Azlinda Abd Halik, Ph.D. in Biochemistry/Biotechnology from University of Malaya, our Deputy Chief Scientific Officer. Dr Heilly has over 15 years of biotechnology research experience, including but not limited to, biologics (monoclonal antibody) drug development, antibody/protein engineering, assay/diagnostic development, molecular biology (high expression systems in mammalian cells), cell culture technology, cell characterization and banking, bioprocessing/biomanufacturing (upstream and downstream processes), and current good tissue practices manufacturing (CAR-T). |
| ● | Cost Effectiveness: Celestialab intends to position itself to offer CAR-T cells at a significantly lower cost in Malaysia compared to other countries. This cost advantage is a result of several factors, including efficient manufacturing processes, optimized supply chain management, and favorable regulatory environments. By providing affordable CAR-T cell production, Celestialab aims to improve accessibility and affordability for patients in Malaysia, ensuring that innovative therapies are accessible to those who need them most. |
COVID-19 Vaccine (mRNA)
Through our subsidiary, MyGenome, we are currently focused on developing three mRNA constructs for the COVID-19 vaccine. Accredited by the MS ISO 15189:2022, MyGenome operates as a molecular laboratory facility providing technical infrastructure for COVID-19 vaccine research. MyGenome has developed a total of eleven (11) mRNA constructs. Out of the eleven mRNA constructs, three (3) mRNA constructs have concluded the POC phase and the remaining eight (8) mRNA constructs are currently still undergoing the POC phase. The POC includes in vitro proof of concept and validation of plasmid DNA templates, followed by the optimization of the in vitro transcription process and mRNA formulation. This groundwork facilitates a transition to in vivo studies to confirm immunogenicity and efficacy. After completing in vitro validation and formulation stage, MyGenome will begin in vivo POC testing for pDNA constructs, which is projected to conclude by the end of 2025. Following this, the focus will shift to conducting a safety study under GLP guidelines, targeted to begin in early 2026. Once the safety study is completed, MyGenome will compile and submit a dossier to the NPRA seeking approval to initiate clinical trials. Based on the foregoing and subject to regulatory approval, MyGenome intends to carry out a series of clinical trials scheduled between 2026 and 2028, with target of eventual product registration by the end of 2029. MyGenome does not have the capacity to manufacture mRNA vaccines as of the date of this proxy statement/ prospectus.
Leveraging mRNA Technology for Global Health Security
MyGenome, in response to the need for rapid and extensive COVID-19 vaccination to mitigate the economic impact forecasted by the Economist Intelligence Unit8, is capitalizing on mRNA’s transformative potential in therapeutics to advance its vaccine development initiative. One of the proposed mRNA constructs, ME1.1, embodies a novel multi-epitope design, leveraging the versatility of mRNA technology to encode immunogenic viral peptides that has been previously studied by multiple research institutions including but not limited to Saudi Arabia and India in terms of their antigenicity, allergenicity and toxicity thus may provide protection against SARS-CoV-2 variants.
5 Ng BD, Rajagopalan A, Kousa AI, Fischman JS et al. IL-18-secreting multi-antigen targeting CAR T-cells eliminate antigen-low myeloma in an immunocompetent mouse model. Blood 2024 Apr 5. PMID: 38579288
6 Tan, S. M., Ong, T. C., Syed Abd Kadir, S. S., Jameela, S., Goh, A. S., Teo, C. S., Lim, S. M., Boo, Y. L., Wong, L. L. L., Wilfred, G., Chew, L. P., Yong, K. Y., Leong, T. S., Teh, A., Noor Hisham, A., & Lim, T. O. (2018). Real World Experience in Using CD19 CAR-T Therapy for Patients with Relapsed or Refractory B-cell Acute Lymphoblastic Leukemia (B-ALL) in Malaysia. International Academy for Clinical Hematology (IACH).
7 Dai Chihara, Laura Liao, Joseph Tkacz, Anjali Franco, Benjamin Lewing, Karl M. Kilgore, Loretta J. Nastoupil, Lei Chen; Real-world experience of CAR T-cell therapy in older patients with relapsed/refractory diffuse large B-cell lymphoma. Blood 2023; 142 (12): 1047–1055. doi: https://doi.org/10.1182/blood.2023020197.
8 Please refer to https://www.eiu.com/n/delayed-vaccination-timelines-will-cost-the-global-economy-us2-3trn/
| 181 |
Our scientific team is deploying a phased approach to vaccine development. We are now at the initial phase where it encompasses in vitro proof of concept and validation of plasmid DNA templates, followed by the optimization of the in vitro transcription process and mRNA formulation. This groundwork facilitates a seamless transition to in vivo studies to confirm immunogenicity and efficacy.
Over the past ten years, mRNA has emerged as a revolutionary medical modality with significant potential for addressing a broad spectrum of diseases that currently lack effective treatments. mRNA molecules are composed of a sequence of nucleotides, which serve as fundamental units that dictate the genetic instructions delivered to cells. These instructions direct the production of proteins that exert therapeutic actions within the body.
mRNA is generated with four main building blocks (Refer to Figure 10 below), but with unique Open Reading Frame (ORF) or Gene of Interest (GOI). This structure enables mRNA to encode a variety of different proteins. This structure enables the rapid development of mRNA-based therapeutics that are potentially applicable for treatment of many diseases, including cancer, infectious diseases and rare disease. Our mRNA pipeline plan intends to address these potential therapeutic areas.

Figure 10. Building blocks of a typical mRNA construct.
mRNA Drug Mechanisms: From Synthesis to Cellular Action
As a drug, manufactured mRNA provides instructions to a target cell to produce a desired therapeutic protein. The mRNA drug will temporarily change the status of the target cell where these instructions are translated into proteins. Based on the information encoded by the mRNA, the proteins will be either secreted or remain intracellular. The mRNA drug will eventually be degraded and eliminated from the body.
Our mRNA drugs are synthesized from a plasmid DNA template. With the exception of the 5’ cap, the template determines all structural elements of the mRNA. The mRNA molecule comprises (Refer to Figure 11):
| ● | an open reading frame, or ORF, which encodes for the protein of interest; | |
| ● | untranslated regions, or UTRs, which flank the ORF (5’ UTR and 3’-UTR); and | |
| ● | the cap and the poly(A) tail, which are the two terminal structures of the linear mRNA, and are responsible for increased stability and translational efficiency of mRNA. |
The mRNA drug needs to be appropriately formulated in order to protect it from breakdown by extracellular ribonucleases (RNases). The formulation is selected based on the intended application and route of delivery. After uptake into the target cell, the mRNA molecules are loaded into ribosomes, where translation into protein takes place. Subsequently, the mRNA is degraded by cellular mechanisms. In case of an immunotherapy application, the protein is degraded into immunogenic epitopes. These are loaded onto specialized molecules, namely MHC class I or MHC class II. These molecules present the epitopes to immune cells to provoke the desired immune response. In the case of other mRNA applications, the mRNA encodes proteins that are secreted from the cells, such as antibodies, and function extracellularly.
| 182 |
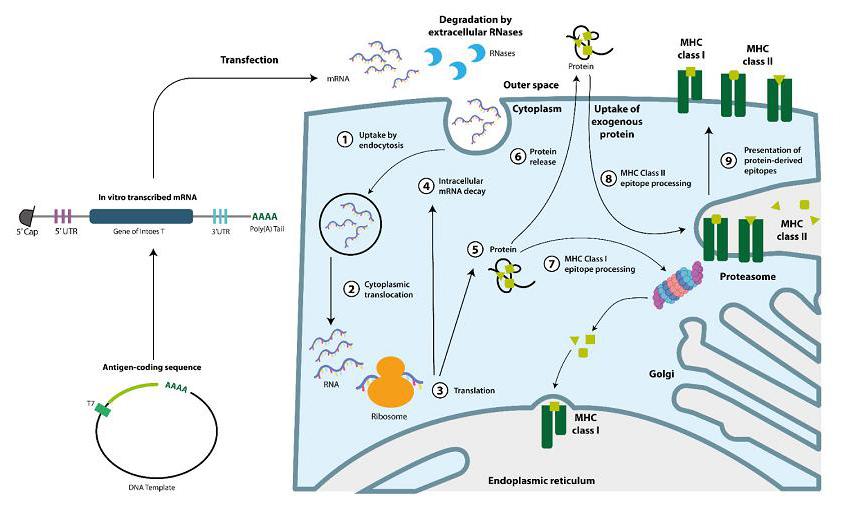
Figure 11. The MHC Class I Antigen Processing and Presentation Pathway
General principles of mRNA pharmacology involve several steps. First, mRNA is either delivered in a buffered solution as naked molecules or formulated as nano-particles to protect degradation by extracellular enzymes and is taken up by cells. Subsequently, mRNA is released from endosomes into the cytoplasm. The mRNA is then translated by the protein synthesis machinery of host cells. Translation termination with the translation by degradation of mRNA, and the translated protein product acts in the cell in which it has been generated. Alternatively, the protein product is secreted and may act via autocrine, paracrine or systemic, body-wide mechanisms. For vaccine activity, mRNA encoded antigens are degraded into shorter fragments and loaded onto MHC class I and class II molecules. Protein-derived epitopes can then be presented on the cell surface by both MHC class I and MHC class II molecules, enabling stimulation of CD8+ and CD4+ T cells.
The performance of mRNA is significantly influenced by its structural elements, including potential immunogenicity, translation efficacy, and molecule stability. MyGenome specializes in designing, synthesizing, and formulating therapeutic mRNA, leveraging its niche expertise. Our aim is to customize the composition of mRNA to meet the specific requirements of each application.
| 183 |
mRNA Backbone Concepts and Technologies
All mRNAs’ constructs contain basic structural elements, including the 5’ cap, the untranslated regions and the poly(A) tail, along with a coding sequence, all of which are encoded by the DNA template.
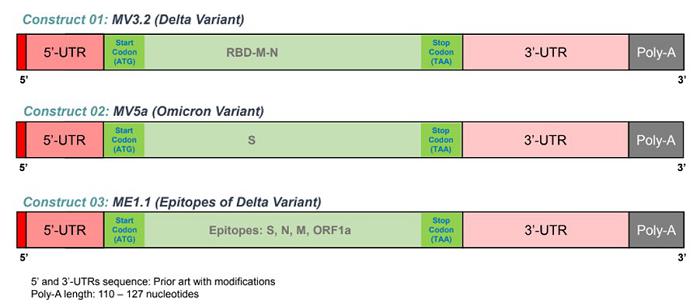
Figure 12. Schematic Representation of mRNA Vaccine Constructs for COVID-19 Variants
| ● | 5’ cap: For in vitro study, ARCA cap is added to the 5’ end of the mRNA during in vitro transcription (IVT). | |
| ● | Untranslated Regions (UTRs): The composition and structure of the 5’ and 3’ untranslated regions of the mRNA molecule are important determinants of the intracellular stability of mRNA. The 5’-and 3’-UTRs were based on generic sequences with slight modifications. | |
| ● | Poly(A) Tail: The length of the poly(A) tail is synthesized to range between 109 to 115 nucleotides, aiding in the stability and translation of the mRNA. |
Alps in Strategizing Multiepitope mRNA Vaccine Development for Enhanced Immunogenicity and Broad-Spectrum Efficacy
Our mRNA vaccine candidates are encapsulated in lipid nanoparticles for optimal delivery. Each mRNA sequence features an open reading frame that encodes antigenic proteins or peptides. These sequences are flanked by UTRs to enhance translational efficiency and capped to stabilize the molecule and modulate the immune response. The poly(A) tail is designed to an optimum length to provide stability.
The ME1.1 construct, containing epitopes from the SARS-CoV-2 spike (S), nucleocapsid (N), membrane (M) proteins, and ORF1a region, is designed to provide tailored immune defense against current and emergent viral strains. The vaccine aims to elicits a potent immune response, characterized by CD8+ and CD4+ T cell activation, ensuring comprehensive protection. The multiepitope selection were based on immunoinformatic analyses by researchers in the same fields.
The primary goal of this multiepitope mRNA vaccine strategy is to develop a highly effective and versatile vaccine that targets multiple epitopes from a pathogen. This approach triggers a robust immune response and offers broad protection against various strains and variants of the pathogen. This approach aims to enhance the efficacy, potency, and durability of the vaccine, ultimately leading to better protection against infectious diseases. Alps’ research team aims to enhance on an inclusive approach that encompasses the virus’s S (Spike), N (Nucleocapsid), M (Membrane), and ORF1a proteins (Refer to Figure 12). The potential final vaccine construct is likely to integrate LBL, CTL, and HTL epitopes linked by specific sequences (AAY, and GPGPG), encapsulating the strategic integration of S, N, M, and ORF1a proteins.
The end goal of multiepitope mRNA vaccine design is to create a highly effective and versatile vaccine that can provide broad protection against various strains and variants of the pathogen. This approach aims to enhance the efficacy, potency, and durability of the vaccine, ultimately leading to better protection against future SARS-CoV-2 virus strains.
| 184 |
MyGenome is developing a platform technology for mRNA vaccines, currently featuring 11 mRNA constructs aimed at combating COVID-19 vaccine The research indicates that three (3) mRNA constructs have completed the POC stage while the remaining eight (8) mRNA constructs are still in the POC stage. The R&D journey follows a structured development plan that includes the construction and verification of plasmid DNA templates, lab-scale optimization of the in vitro transcription process, both in vitro and in vivo POC studies, and subsequently nonclinical safety studies. The strategy focuses enhancing the immunogenicity, efficacy, and safety of the mRNA vaccines prior to moving into clinical trials and seeking regulatory approval.

Figure 13. Our mRNA Vaccine Development Road Map.
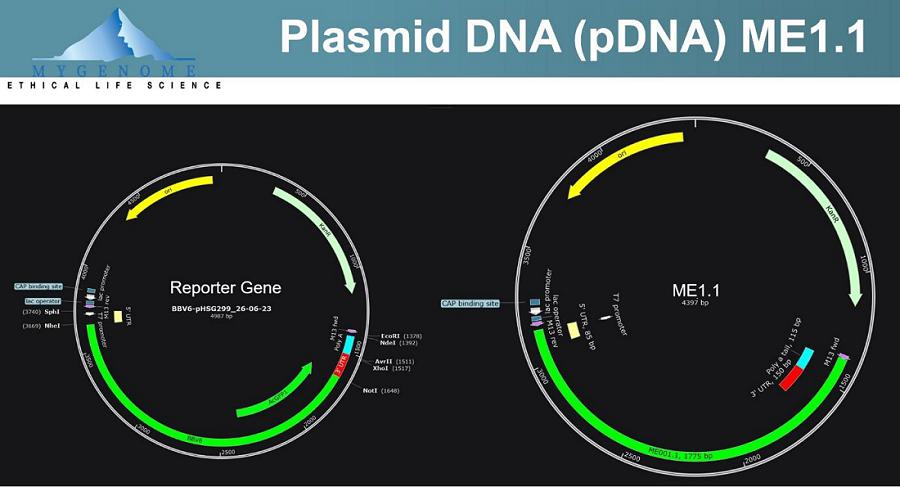
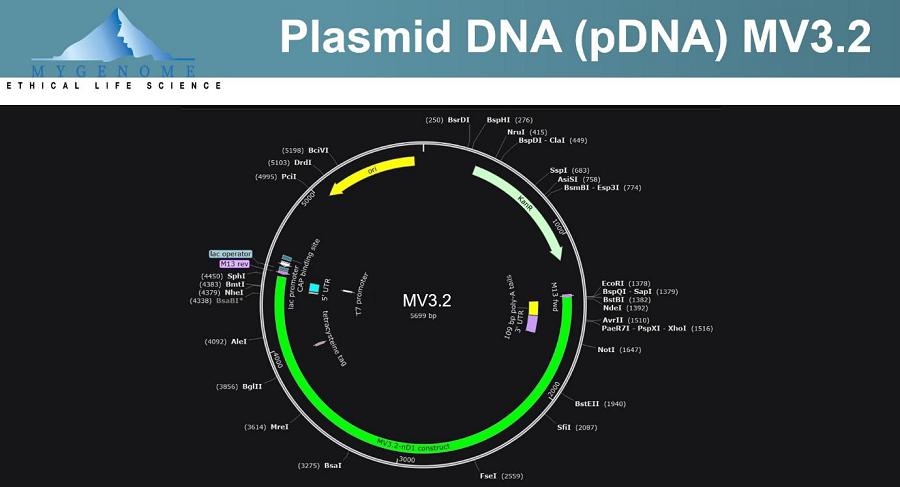

Figure 14. Images representing the plasmid DNA templates for the three (3) mRNA constructs. (Top to bottom: ME1.1; MV3.2 and MV5a)
| 185 |
Addressing
Technical Hurdles in the Advancement of COVID-19 mRNA Vaccine Development
In advancing the COVID-19 mRNA vaccine initiative, we encounter several technical challenges that are critical to the development process. A primary challenge is the meticulous verification of the constructs’ sequences following each laboratory procedure. The current infrastructure, lacking in specific advanced instruments, hampers the ability to conduct in-depth analyses internally. Consequently, this necessitates reliance on external third-party laboratories for comprehensive sequence analysis. We intend to establish partnerships with leading third-party laboratories, which will not only provide access to specialized equipment and expertise but also facilitate knowledge transfer, empowering the team with advanced analytical technique.
Moreover, the production of mRNA in milligram quantities, essential for conducting future in vivo studies, presents another significant obstacle. Given our existing laboratory limitations, including the absence of appropriate equipment, we are compelled to outsource the production of mRNA. This not only impacts our operational timelines but also underscores the necessity for strategic partnerships and investment in laboratory capabilities to support the large-scale production of mRNA. These steps are vital to overcoming the technical barriers currently faced and propelling our COVID-19 mRNA vaccine development towards the next phases of in vivo testing and clinical evaluation. Acknowledging the challenges in producing milligram quantities of mRNA with our current laboratory setup, we will identify and collaborate with CMO that specialize in mRNA production. This strategy ensures the availability of the required mRNA quantities for in vivo studies while maintaining the highest standards of quality and efficiency.
Strategic Alliance for National Vaccine Development and mRNA Therapeutics Expansion
As part of Malaysia’s National Vaccine Development Roadmap, we have submitted our application to the Malaysian Ministry of Science, Technology and Innovation to be a part of the vaccine development and self-sufficiency efforts within the country. Under this arrangement, we intend to conduct clinical trials, leveraging our subsidiary, MyGenome’s clinical development, regulatory, and commercial capabilities in the country. Upon regulatory approval, we will commercialize the vaccine in Malaysia, while also target to retain the full rights to develop and commercialize the vaccine in countries outside of Malaysia.
On March 19, 2024, Alps has entered into a memorandum of understanding (“MOU”) with JLand Group Sdn. Bhd. (“Jland”). Jland is a subsidiary of Johor Corporation as established under the Johor Corporation Enactment (No. 4, 1986) (as amended by Enactment No. 5, 1995), and control by the Johor State Government as a development agency and public enterprise with diverse operations in healthcare, plantations, property and food and beverages sectors across Malaysia, Singapore, Indonesia, Brunei, Cambodia and Bangladesh. This MOU outlines the parties non-binding joint commitment to develop and establish a biotechnology hub (“BioValley”). BioValley will serve a comprehensive center for biotech solutions, integrating biotechnology, pharmaceuticals, and medicine within a single facility. The aim is to create a global benchmark for biotech hubs in Malaysia. Alps aims to develop crucial infrastructure within BioValley, including a vaccine manufacturing plant, to support the advancement of our mRNA modality. Alps and Jland intend to assess the feasibility and perform due diligence on the envisioned establishment of the BioValley at Ibrahim Technopolis, Sedenak, Johor (“Collaboration Area”). Strategically located in southern Malaysia bordering Singapore, the Collaboration Area offers direct access to broader regional markets. Through the Biovalley, Alps intends to develop and implement several initiatives targets: -
| - | offering contract research organization services; | |
| - | providing clinical research associate services; | |
| - | establishing contract development and manufacturing organization services; | |
| - | hosting training programs for medical professionals and technical personnel; | |
| - | developing strategies to attract international medical and wellness clients; and | |
| - | implementing business incubation services for biotechnology startups and related enterprises. |
As part of Malaysia’s National Vaccine Development Roadmap, we are engaged in ongoing non-binding discussions with the Government of Malaysia to forge a strategic alliance, aimed at intensifying and developing COVID-19 vaccine development and self-sufficiency efforts within the country. Under this arrangement, we will conduct clinical trials, leveraging our subsidiary, MyGenome’s clinical development, regulatory, and commercial capabilities in the country. Upon regulatory approval, we will commercialize the vaccine in Malaysia, while also target to retain the full rights to develop and commercialize the vaccine in countries outside of Malaysia.
Leveraging from our experience in our subsidiary-owned biotechnology laboratory which has the capability to handle genetic sequencing/ screening and mRNA profiling/ sequencing, we also intend to leverage this to develop our mRNA platform for developing cancer vaccines by targeting tumor-specific antigens to stimulate an immune response. For gene therapy, the focus is on introducing mRNA sequences that encode therapeutic proteins to correct genetic disorders. In cell therapy, mRNA will be used to engineer cells ex vivo, enhancing their therapeutic potential when reintroduced into the patient. This strategy leverages the versatility of mRNA for precision medicine across multiple therapeutic areas.
We intend to validate our vaccine candidates through comprehensive in vitro and in vivo proof-of-concept studies, underpinned by a thorough nonclinical safety evaluation. This preparatory work is a precursor to our future plans to establish a cGMP-compliant manufacturing facility on a commercial scale to significantly elevate our production capabilities.
We intend to carry out a series of clinical trials scheduled between 2026 and 2028, with target of eventual product registration by the end of 2029. This will segue into an era of diligent post-market surveillance commencing in 2030. Through this timeline, we are dedicated not only to addressing current public health challenges but also to influencing the future direction of vaccine development and mRNA-based treatments.
Cholera Vaccine
Alps Global Holding Berhad has collaborated with researchers from USM to jointly develop an oral vaccine aimed at active immunization against cholera. This collaboration has led to the establishment of Vax Biotech Sdn. Bhd. (“VaxBio”), an affiliate company of Alps in which Alps holds a 30% shareholding.
In this collaboration, Alps Global Holding Berhad assumes the roles of securing funds, facilitating all future clinical phase studies, and undertaking the eventual commercialization of this vaccine. USM contributes the requisite talents, grants the right to use its intellectual property, and provide its facilities at IPHARM. Both parties are jointly involved in the research and development works for this cholera vaccine pipeline.
The cholera vaccine development journey started in the year 2000 at USM, under the leadership of Professor Manickam RAVICHANDRAN, our Chief Scientific Officer. The initial phase involved the isolation of the hemA gene from Vibrio cholerae (“V. Cholerae”), followed by development of a gene mutant vaccine candidate VCUSM2 against V. cholerae O139 in 2006. In 2010, VCUSM2 reactogenicity was reduced by replacing its two wild-type ctxA gene copies with mutated ctxA to produce strain VCUSM14. Introducing the hemA gene into VCUSM14 created VCUSM14P, a strain with the 5-aminolaevulinic acid (ALA) prototrophic trait and excellent colonisation and immunological properties (100% protection to wild-type challenged rabbits). The strain was further refined with completion of single- and repeated-dose toxicity evaluations in year 2019 in Sprague Dawley (SD) rats, followed by development of a novel cold-chain-free VCUSM14P formulation in year 2020. VCUSM14P is unique for its intact cholera toxin B, a known mucosal adjuvant.
As of the date of this proxy statement/prospectus, the development of the cholera vaccine has reached the conclusion of the proof-of-concept phase. VaxBio’s immediate focus is on the Good Laboratory Practice (“GLP”) animal toxicity study at IPHARM, which is a prerequisite for entering clinical development phases. This study is being conducted at IPHARM within USM’s facility, adhering to strict international regulatory standards. This transition aligns with international best practices and signifies a significant milestone in the vaccine’s development process. After completing the preclinical acute toxicity study, VaxBio plans to submit a dossier to the NPRA to seek approval for starting clinical trials.
| 186 |
Cholera Disease Profile and Vaccine Development Initiatives
Cholera, a global public health challenge, is an acute intestinal infection caused by the bacterium V. cholerae. It manifests predominantly as a diarrheal disease and affects an estimated 3 to 5 million individuals annually, resulting in over 100,000 fatalities worldwide. While a substantial proportion of infections may present as mild or asymptomatic, a notable minority, about 10%, progress to severe disease. This severe presentation is typified by copious watery diarrhea, emesis, and muscle cramps, precipitating rapid dehydration, shock, and, in the absence of medical intervention, potentially fatal outcomes within mere hours.
In response to this enduring health threat, the scientific community has commenced the development of a suite of vaccine formulations aimed at the prevention of cholera. These vaccines employ either inactivated or live attenuated strains of V. cholerae as their foundational mechanism of action. Specifically, these vaccine candidates, inclusive of both whole-killed and live attenuated variants, have demonstrated efficacy against the O139 serogroup of V. cholerae. They have been observed to induce robust antibody production and engender protective immune responses, highlighting their potential in the prophylactic landscape against cholera.
Despite these advances, the deployment of current cholera vaccines is not without its obstacles. Chief among these is the necessity for a reliable cold chain to preserve vaccine potency, an attribute that necessitates repeated dosing schedules. These logistical demands inherently elevate the costs associated with vaccination campaigns and may result in a shorter duration of immunity. The business is actively engaged in addressing these challenges, seeking to enhance vaccine accessibility and durability of immune protection, thereby reducing both the economic burden and health risk posed by cholera on a global scale.
This project not only aims to advance public health but also demonstrates its dedication to contributing scientifically sound and economically viable solutions to the global market.
Development of Oral Cholera Vaccine for Global Immunization Programs
This vaccine is particularly intended for adults and children aged two years and above who are traveling to, or residing in, areas where cholera is endemic or where outbreaks are active. The decision to administer the jointly developed cholera vaccine should align with official health recommendations, taking into consideration the varying epidemiological profiles and the inherent risks associated with different regions and travel circumstances.
Vaxbio expects to formulate a consumer friendly oral cholera vaccine that offers protection against cholera-induced diarrhea, as well as diarrhea caused by Enterotoxigenic Escherichia coli (ETEC), which is often a concern in the same regions affected by cholera. The cholera vaccine is intended to be conveniently administered by dissolving it in drinking water, facilitating ease of delivery and compliance.
VaxBio’s development objectives for the cholera vaccine were methodically outlined with a focus on several key characteristics:
| ● | Single Dose: The utilization of a live attenuated strain, negating the need for multiple doses. | |
| ● | Build-in Adjuvant: Ensuring robust colonization to closely replicate the natural infection pathway. | |
| ● | Cold Chain Free: Eliminating the requirement for cold-chain storage to alleviate logistical challenges in distribution. | |
| ● | Long Term Protection: Achieving high levels of protection, which is imperative for any vaccine. | |
| ● | Very Low Side Effects: Maintaining a low reactogenicity profile to ensure patient safety and comfort. |
| 187 |
VaxBio anticipates providing a vaccine that is not only effective and protective but also aligns with the practical demands of global immunization initiatives, particularly in regions with limited healthcare infrastructure.
Figure 15. VaxBio’s target to achieve all the key attributes of the innovative oral cholera vaccine.
Our Local Researchers and Collaborator Institutions
The development of this cholera vaccine has been supported by the Ministry of Science, Technology, and Innovation in Malaysia (MOSTI) and the Ministry of Higher Education through grants such as the Intensification of Research Priority Grant, Fundamental Research Grant Scheme and the Prototype Research Grant Scheme, provided to USM.
Strategic Intellectual Property Developments in Cholera Vaccine Research
There are two (2) key patents crucial for future research, development and commercialization of this cholera vaccine. The first patent, titled “Fusion Protein and Chimeric Protein as Mucosal Delivery System Method of Producing Same, and Biological Derivatives Thereof”, is owned by USM (“First Cholera Patent”). The First Cholera Patent focuses on a fusion protein and a chimeric protein used in mucosal delivery for the prohylaxis or treatment of a targeted disease, including V. cholerae. This patented formulation plays a pivotal role in cholera vaccine development by enabling mucosal delivery through fusion and chimeric proteins, which enhances targeted immunity at the primary infection site, improving the stability and potency of the vaccine. This approach improves the stability and potency of the vaccine, resulting in a more effective immune response against V. cholerae.
On September 12, 2023, VaxBio and USM has entered into a Licensing and Commercialization Agreement (“USM-VaxBio Agreement”), where USM granted a license to VaxBio to utilize the First Cholera Patent for purposes of research, development, manufacturing, selling and otherwise dealing with products made in accordance with the First Cholera Patent. The USM-VaxBio Agreement is effective from September 12, 2023 and it will remain in full force for a period of five (5) years from the effective date and may be renewed for another five (5) years.
The second patent, titled “Monovalent Vaccine Formulation and a Method for Preparation Thereof”, is owned by AIMST University (“Second Cholera Patent”). The Second Cholera Patent encompasses innovative approaches to vaccine development, specifically targeting the 0139 serogroup of V. cholerae. As at the date of this proxy statement/prospectus, VaxBio is pursuing a patent license from AIMST University for the Second Cholera Patent. VaxBio believes that the Second Cholera Patent will facilitate the formulation of a vaccine which has independence from cold-chain logistics, addressing a critical barrier in vaccine distribution but also ensuring the vaccine’s longevity, making it particularly suitable for deployment in regions where cholera poses a significant public health threat.
VaxBio plans to leverage this strategic collaboration and future licensing agreements to support broader cholera prevention efforts, particularly focusing on improving accessibility and effectiveness of cholera vaccines in regions most affected by the disease.
| 188 |
Navigating Challenges in Clinical Development for Cholera Vaccine
As VaxBio advances its cholera vaccine through the preclinical stages, it faces several challenges that necessitate strategic responses and collaboration. VaxBio’s current focus is on conducting rigorous pre-clinical assessments and preparing for the vaccine’s clinical trials, steps that are vital for ensuring its safety, efficacy, and compliance with international regulatory standards.
VaxBio’s immediate efforts are centered around the GLP animal toxicity study, a prerequisite for entering clinical development phases. This study is currently undertaken at IPHARM within USM’s facility, which adheres to stringent international regulatory guidelines. Simultaneously, the transition to GMP production represents a significant milestone, necessitating the establishment of facilities equipped for bacterial fermentation for human vaccines, in alignment with global best practices.
The path to clinical trials unveils several logistical and regulatory hurdles. Currently, the requisite GMP production and all three phases of clinical development is more feasible outside Malaysia, due to the lack of significant cholera cases in Malaysia and the absence of local facilities tailored for bacterial fermentation for human use. Additionally, the execution of Phase 1 clinical trials is impeded by readiness constraints, while the feasibility of conducting Phase 2 and 3 trials domestically is limited by the lower incidence of cholera in Malaysia as compared to other endemic regions. Given the collaborative nature of this cholera vaccine involving multiple parties in its development and research, the timing of subsequent clinical trials may not be solely within Alps’ control and is subject to the coordination efforts of USM and the approval process of governmental authorities.
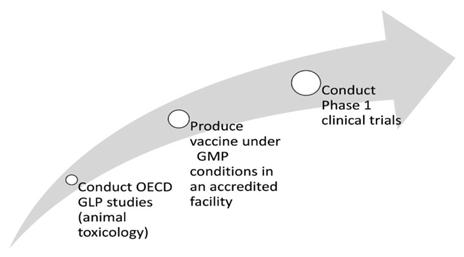
Figure 16. Overview of Vaccine Development Stages Prior to Clinical Efficacy Trials
VaxBio’s Strategic Market Entry
The current global health landscape presents an urgent demand for effective cholera vaccines amid escalating outbreaks and a pronounced vaccine shortage. With over 40,900 cases and 775 fatalities reported in the early months of 2024 alone, the need for a reliable vaccine supply is critical. This crisis is further exacerbated by the monopolization of vaccine production by a single manufacturer, highlighting the fragility of the existing supply chain.
Recognizing this dire need, VaxBio’s perceive a strategic market entry opportunity for our cholera vaccine. It intends to provide a cold-chain-free, single-dose solution not only positions it to address the immediate supply shortages but also to establish a sustainable presence in the global immunization landscape. By offering a cost-effective, easily distributable vaccine alternative, VaxBio aims to complement and reinforce the existing Water, Sanitation, and Hygiene (WASH) initiatives endorsed by the World Health Organization (WHO), thereby contributing to a holistic approach to cholera control and prevention.
As VaxBio progress towards clinical validation in accredited GLP facilities, its focus remains on ensuring the vaccine’s safety, efficacy, and regulatory compliance. VaxBio’s strategic direction is guided by a commitment to enhancing public health through innovative vaccine solutions, with the ultimate goal of mitigating the global burden of cholera and supporting the expansion of global immunization programs.
| 189 |
MYCELEST
Celestialab also engages in research and development for MYCELEST, a cell-based therapy specifically designed to target the underlying mechanisms of both type 1 and type 2 of diabetes mellitus. As the date of this proxy statement/prospectus, Celestialab is completing the pre-clinical studies phase, which includes initiating a randomized controlled trial to assess the safety, efficacy, and tolerability of intraperitoneal allogenic mesenchymal stem cell therapy in new-onset type 1 and type 2 diabetes mellitus. Celestialab and is currently preparing the preclinical data for submission of the dossier application to NPRA.
Once Celestialab obtains the necessary approvals from NPRA, Celestialab will initiate and conduct clinical trial on patients diagnosed with type 1 and type 2 diabetes for a future multicentre, randomized, double-blind, placebo-controlled study. Participants will further be stratified based on diabetes type and then randomized into six arms: T101 (standard treatment and low dose MyCelest), T102 (standard treatment and medium dose MyCelest), T201 (standard treatment and low dose MyCelest), T202 (standard treatment and medium dose MyCelest), PCB1 (Placebo/ Type 1), PCB2 (Placebo/Type 2).
Background of Diabetes
Diabetes mellitus (“DM”) is associated with enhanced reactive oxygen species (ROS) production. Type 1 diabetes (“T1DM”) is an autoimmune disease in which a person’s pancreas stops producing insulin, a hormone that enables glucose to enter cells and thereby allows people to get energy from food. It occurs when the body’s immune system attacks and destroys the insulin-producing cells in the islets of the pancreas, called beta cells. Type 2 diabetes mellitus (“T2DM”) is characterized by a combination of insulin resistance and pancreatic β-cell dysfunction due to metabolic exhaustion. Sustained hyperglycaemia may result in multi-system chronic complications, including micro- and macrovascular complications, which are associated with high morbidity and mortality. The prevalence of diabetes for all age-groups worldwide was estimated to be 4.4% in 2030, the total number of people with diabetes is projected to rise to 366 million in 2030.9
T1DM patients’ survival depends on daily doses of exogenous insulin (EI) to balance their blood glucose levels. Although this is the standard treatment, it does not provide an adequate physiological response and cannot prevent the progressive degeneration of islet β-cells; consequently, these patients might experience hypoglycemic episodes. Current therapies for T1DM have significant limitations. Insulin injections are the first-line treatment with different challenges:
| ● | long-term dependency, | |
| ● | difficulty with EI level management, and | |
| ● | potential insulin resistance with long-term usage. |
Pancreas transplantation as the second-line treatment has faced multiple obstacles such as donor shortages, the possibility of transplant rejection, usage of immunosuppressive drugs, and high risks with an organ transplantation surgery. Thus, improved diabetes treatment methods that can address these limitations will be applicable in diabetes research and future management.
T2DM is a chronic metabolic disorder characterized by elevated blood glucose levels resulting from insulin resistance and inadequate insulin production by the pancreas. This condition has become a significant global health concern, with its prevalence on the rise due to factors like sedentary lifestyles, unhealthy diets, obesity, aging populations, and genetic predisposition. T2DM poses substantial health risks, including cardiovascular disease, kidney failure, blindness, and neuropathy, making its effective management crucial.
9 Wild, S., Roglic, G., Green, A., Sicree, R., & King, H. (2004). Global prevalence of diabetes: estimates for the year 2000 and projections for 2030. Diabetes Care, 27(5), 1047-1053. https://doi.org/10.2337/diacare.27.5.1047
| 190 |
The historical approach to managing T2DM initially focused on dietary modifications and lifestyle changes. The introduction of insulin in the 1920s was a groundbreaking advancement, particularly for T1DM. However, its application in T2DM was limited due to a primary emphasis on symptom management rather than addressing the underlying insulin resistance. Over time, a range of oral antidiabetic medications was developed to target various aspects of glucose metabolism. These included biguanides like Metformin, which improve insulin sensitivity and reduce hepatic glucose production; sulfonylureas like Glipizide and Glyburide, which stimulate insulin secretion; and thiazolidinediones like Pioglitazone and Rosiglitazone, which enhance insulin sensitivity through PPAR activation. Other classes such as alpha-glucosidase inhibitors, DPP-4 inhibitors, and SGLT2 inhibitors also emerged, providing diverse options for glycemic control.
Despite the advancements in T2DM treatment, managing the disease remained challenging due to several reasons. First, the progressive nature of T2DM meant that over time, many patients required multiple medications or insulin therapy to maintain adequate glycemic control. Second, these older antidiabetic medications often came with significant side effects, including hypoglycemia, weight gain, gastrointestinal issues, and potential cardiovascular risks, limiting their long-term utility. Additionally, adherence to complex medication regimens, high treatment costs, and the lack of individualized treatment approaches further complicated T2DM management. Lastly, the predecessor treatments often fell short in addressing the underlying lifestyle factors contributing to T2DM, necessitating additional lifestyle modifications to achieve optimal outcomes. Despite these challenges, these early treatments laid the foundation for our understanding of T2DM and paved the way for the development of newer, more effective therapies.
Cell-based therapy offers promising approaches for both T1DM and T2DM by targeting the underlying mechanisms of the diseases. For T1DM, strategies like islet transplantation, cell regeneration, and immune system modulation using regulatory T cells aim to restore insulin production and prevent immune-mediated destruction of beta cells. In T2DM, the focus is on improving insulin sensitivity and secretion, as well as promoting beta-cell regeneration. Mesenchymal stem cells (“MSCs”) are being explored for their potential in enhancing insulin sensitivity and reducing inflammation in T2DM, while encapsulated islet cell transplantation and immune-modulating therapies are also under investigation. Despite the promising advancements, further research is needed to optimize these cell-based therapies, address challenges like immune rejection and long-term efficacy, and ensure their safe and effective integration into diabetes management strategies.
Mesenchymal Stem Cell in Reversing Insulin Resistance and Promotes Pancreatic β cell proliferation
To date, MSCs are one of the most studied cell population in clinical trials. The therapeutic capacity of MSCs is based on their ease of isolation, ability to differentiate into multiple cell types, low immunogenicity, abundant source, minimal ethical concerns, and, most notably, their release of biological factors that can alleviate impaired tissues. The findings of a preclinical study showed that MSCs could be effective as a distant immunomodulator in addition to their homing to injured sites.
MSCs have a mesodermal origin and are defined as multipotent cells that can adhere to plastic; self-renew; express specific surface antigen markers (CD73, CD90, CD105); lack hematopoietic antigen expressions (CD45, CD34, CD14 or CD11b, CD79α or CD19, HLA-DR (Human Leukocyte Antigen—DR isotype)); and can differentiate into osteoblasts, adipocytes, and chondroblasts. MSCs are derived from many sources such as the bone marrow, umbilical cord, umbilical cord blood, adipose tissue, and dental pulp.
Our Solution with Wharton’s jelly-derived mesenchymal stem cells
Wharton’s Jelly of the umbilical cord, previously deemed a medical waste of newborn deliveries, is becoming a preferred source of MSCs. Since the discovery of WJ-MSC by Friedenstein, repurposing the cord for tissue engineering and regenerative medicine has become a unique research interest. The Malaysia National Healthcare Establishments and Workforce Survey reported a total of 423,124 live births within Malaysia in 2023.10 This supersedes the concern for the scarcity of MSCs and long intervals between available tissue samples, which are a present challenge for bone marrow and adipose tissue.
10 Department of Statistics Malaysia, “The Population of Malaysia Dashboard,” updated October 17, 2023. Available at: DOSM Population Dashboard.
| 191 |
Over the past decade, various clinical trials have been conducted during the past decade to test the feasibility and efficacy of WJ-MSCs therapy for various disease conditions such as: neurological disorders, cancer, cardiac disease, liver disease, bone/cartilage disease, immunological diseases as well as DM and its complications. Many of these clinical trials have been completed and indeed demonstrated the safety and efficacy of WJ-MSCs. Public clinical trial databases currently show about 1458 investigating MSCs from various sources for a wide array of therapeutic application; 86 of which are diabetes mellitus related. Regarding the 86 studies currently registered on the public clinical trials database investigating MSCs (from various sources) for treating DM and/or its complications, only about 30 of these studies clearly state applying WJ-MSCs as an intervention.
Currently, we are preparing to commence our phase IIa/IIb randomized controlled trial to assess the safety, efficacy and tolerability of intraperitoneal allogenic mesenchymal stem cell therapy in new-onset T1DM and T2DM. This is a phase IIa/IIb randomized, double-blind, placebo-controlled study where we have identified at least 28 newly diagnosed adults with type 1 and type 2 diabetes mellitus to participate. Participants will be stratified based on T1DM or T2DM and then randomized to 3 arms – standard treatment + placebo, standard treatment + low dose human umbilical cord mesenchymal stem cells (hUCMSCs), standard treatment + moderate dose hUCMSCs.
Our team intends to apply WJ-MSCs transplantation in T2DM patients, and we target to demonstrate that treatment with WJ-MSCs can indeed improve metabolic control and β-cell function. Thus, it will reinforce that the mechanism of action may have involved improvements in systemic inflammation as well. Thus, WJ-MSCs proved to be safe and relatively effective in both T1DM and T2DM, as well as recently diagnosed or relatively long-standing diabetic patients.
When considering WJ-MSCs as a novel therapeutic intervention not only for DM, but also for other disease conditions, the majority of clinical trials reported the safety and absence of serious acute or chronic adverse effects. What makes MSCs, and especially WJ-MSCs an ideal candidate for regenerative medicine, with potential capabilities not only to regenerate damaged tissues but also to reduce rejection possibilities are their unique immunomodulatory properties. These properties provide some sort of relief regarding the concern of possible immuno-rejection.
cGMP Compliance
WJ-MSCs, like all other cell-based therapies are living products constantly interacting with their surrounding environment. This poses new challenges when looking from a biotechnology platform point of view. For example, they cannot be sterilized prior to use to provide strict protection from any contamination transmission to patients. In addition, in order to maintain a consistent product efficacy, with minimal batch-to-batch variation, this requires precise process control and avoidance of adverse changes in heterogeneous populations or the cell environments. Consistent cGMP-compliant manufacturing requires high reproducibility with a focus on safety, efficacy, and quality. Celestialab focuses on preventive medicine with a specialization in the production of cellular products adhering to cGMP standards. Alps is intending to apply for NPRA approval through Celestialab to produce stem cells and cell-based immunotherapy.
WJ-MSCs are considered an advanced therapy medicinal product, and should be produced in compliance with cGMP. We are looking at complying strictly with requirements include the following: (a) Tests for virology (HIV-1/2, HBV, HCV, HTLV-1/2, HPV, B-19, CMV, and EBV), syphilis, mycoplasma, and sterility being negative. (b) Phenotype: the percentages of CD73+, CD90+, and CD105+ cells ≥ 98% and the percentages of CD34−, CD45−, HLA-DR−, CD14− or CD11b−, CD79a−, or CD19− ≤2%. (c) Viability ≥ 80% after thawing. (d) The endotoxin content < 2 EU/mL. (e) No significant upregulation of telomerase reverse transcriptase (hTERT) gene and oncogenes during large-scale expansion. (f) No significant downregulation of tumor suppressor genes during large-scale expansion. (g) Confirmed potency.
Current Steps and Development
An estimated 130 million global annual births worldwide provide a unique opportunity as well as a readily available supply of life-saving stem cells, which could be recovered from WJ and UCB, then banked for future autologous or allogenic therapeutic application. Taking in consideration the ever-growing list of advantages and therapeutic effects of WJ-MSCs, not only for DM, but also for various disease conditions, banking of WJ-MSCs would indeed turn those UCs from just medical wastes into valuable priceless therapeutic tools.
| 192 |
Celestialab is completing the preclinical studies phase for MYCELEST and is currently preparing the preclinical data for submission of the dossier application to the NPRA. As such, once Celestialab obtains the necessary approvals from NPRA, we will progress the clinical phase, focusing on the safety, efficacy, tolerability, maximum tolerated dosage as well as pharmacology of UC-MSCs for diabetes mellitus. To facilitate commercialization in the market, maintaining cost-effectiveness is a crucial aspect for Celestialab. Furthermore, when considering scaling-up for the generation of efficient cellular therapies from WJ-MSCs, ensuring cGMP compliance while maintaining cost-effectiveness presents a significant challenge in the commercialization process.
iPSC (Heart Failure)
We have entered into two (2) research collaboration agreements with USM for the research and development of this iPSC product candidate. On December 16, 2019, Alps Biotech, one of our subsidiaries, entered into a research collaboration agreement with USM (as supplemented on March 15, 2023) to jointly undertake a research project titled “Establishing Heart Failure Model in Rats for Preclinical Assessment of Cardiovascular Regenerative Therapy” (“RCA for Preclinical Assessment of Cardiovascular Regenerative Therapy”). This project focuses on the development of an iPSC model for cardiac valve and cardiac muscle regeneration to address cardiovascular diseases. It is agreed that all rights, titles and interest of intellectual property created or developed as a result of this project shall be jointly owned by USM and Alps Biotech.
On February 10, 2020, Alps Global Holding Berhad entered into another research collaboration agreement with USM to jointly undertake a research titled “Cell Therapy for Heart Disease”. This project focuses on the development of pre-epicardial cells through the establishment of the USM-Alps Joint Laboratory for Cardiac Research. It is agreed that all rights, titles and interest of intellectual property created or developed as a result of this project shall be jointly owned by USM and Alps.
As of the date of this proxy statement/prospectus, both agreements have expired, and Alps is currently in negotiations with USM to consolidate all previous projects into a new research collaboration agreement for the next phase of research and development with respect to this iPSC pipeline.
The development effort of both Alps Biotech and USM (“IPSC Collaborators”) are currently involve generating human iPSC cells from adult peripheral blood and manufacturing clinical-grade iPSCs under GMP conditions. The IPSC Collaborators have established heart failure rat models by ligating the left anterior descending coronary artery and inducing cryoinjury. Furthermore, proof-of-concept studies using human iPSC-derived cardiomyocytes (“CMs”) to treat cryoinjured rat hearts have been successfully completed by Alps and USM. Early findings indicate that combining these CMs with hiPSC-differentiated pericytes (PECs) results in a superior regenerative effect, leading to enhanced vascularization and increased survival of CMs four weeks post-injection. These results represent an advancement as the research progresses towards assessing the long-term therapeutic impacts in larger animal models and verifying the safety of the product for human clinical trials. Upon completion of generating human iPSC cells, the next step will involve conducting pre-clinical trials. This phase includes preparing the trial design, and conducting studies to assess efficacy, toxicology, dosage, and safety.
Overview of Heart Disease
According to the WHO, non-communicable diseases (NCDs), including cardiovascular diseases (CVDs), are leading causes of mortality in the Western Pacific region, significantly impacting countries throughout Asia Pacific and Southeast Asia. Rapid urbanization, lifestyle changes, and aging populations contribute to the increasing burden of heart disease. Over 250 known genetically defined disorders primarily affect the heart, contributing to morbidity and mortality, yet few approved products target these genetic causes. Recent analyses have shown that after decades of declining mortality rates from heart failure, these rates are once again rising, underscoring the need for improved treatments.
| 193 |
The heart’s complexity, due to its biological structure and the tightly regulated coordination of its electrophysiological and biomechanical properties, complicates the treatment of heart disease. Heart disease manifests in various forms, affects individuals of many ages, and results from numerous factors. As illustrated in the Figure 17 below, heart disease can be categorized as either resulting directly from issues associated with the heart organ, such as heart failure, arrhythmia, and heart valve disease, or indirectly from vascular problems, such as coronary artery disease (CAD). The underlying causes can be genetic, due to aging, or environmental factors. The table below outlines four broad categories of heart disease:
| CATEGORIES | DESCRIPTION | |
Heart Failure |
Heart failure is a condition where the heart cannot pump enough blood and oxygen to meet the body’s needs. It can result from conditions that weaken the heart muscle, including heart attacks, uncontrolled high blood pressure, congenital heart defects, and genetic cardiomyopathies. | |
Arrhythmia |
Arrhythmia is a common heart condition, involves any deviation from the heart’s normal electrical impulses, affecting its pumping function. Arrhythmias can lead to a range of symptoms, sudden death, and stroke. | |
Heart Valve Disease |
Heart value disease occurs when one or more of the heart’s four valves malfunction, disrupting blood flow through the heart’s chambers. | |
Coronary Artery Disease (CAD) |
CAD is a prevalent form of heart disease, arises when plaque accumulates in the coronary arteries’ walls, restricting blood flow to the heart muscle and potentially leading to a heart attack. |
Figure 17. Heart disease can be categorized as either resulting directly from issues associated with the heart organ, such as heart failure, arrhythmia, and heart valve disease, or indirectly from vascular problems, such as coronary artery disease (CAD).
While there is a significant unmet need in the field of heart disease, challenges have historically hindered the development of novel therapies. The IPSC Collaborators aim to focus on heart failure, particularly myocardial infarction, as the most common cause of heart failure. If the dead, ischemic heart tissues could be regenerated with viable and functional cardiomyocytes, the disease could potentially be treated.
| 194 |
Advancements and Challenges in Cardiac Repair Post-Myocardial Infarction
Myocardial infarction (MI), a common cause of heart failure (HF), results from the partial or complete occlusion of the coronary artery, reducing oxygen and nutrient delivery to the myocardium. Approximately 25% of patients with myocardial infarction experience severe left ventricular dysfunction, risking progressive heart remodeling. The first non-invasive treatment option typically includes pharmacological approaches with drugs such as thrombolytic agents, β-blockers, and angiotensin-converting enzyme inhibitors. In more severe cases, such as ST-elevated myocardial infarction (STEMI), invasive procedures like balloon angioplasty and stent insertion may be recommended for myocardial reperfusion. Coronary artery bypass grafting, a highly invasive procedure, is advised for severe, irreversible coronary occlusion. These methods alleviate symptoms and enhance life quality, but none can remove fibrotic scars or replace lost myocardium with new cardiomyocytes. The presence of akinetic tissue limits cardiac performance, compelling the remaining myocytes to increase contractility to maintain adequate cardiac output. This trigger sudden changes in cardiac architecture, leading to cardiomyocyte hypertrophy, further myocyte loss, ventricular wall thinning, weakened contractility, and eventually, the cessation of cardiomyocyte function. To date, heart transplantation remains the only curative option. Despite successful heart transplants, the long waiting times, high patient-to-donor ratio, high incidence of post-procedural complications, and limited availability of transplantable hearts underscore the urgent need for alternative solutions.
Bone marrow-derived stem cells are the most common first-generation cell candidates used in clinical transplantation (Refer Figure 18 below). A weighted regression and meta-analysis of 49 trial reports using autologous bone marrow stem cells revealed discrepancies among these trials, with none showing benefits from bone marrow mononuclear cells (BMNCs). However, it is widely accepted that the therapeutic benefits of bone marrow-derived cells mainly result from a paracrine mechanism that activates endogenous healing. Reconstituting injured myocardium with cardiomyocytes may require second-generation cardiogenic cells, such as more defined, homogeneous cardiac-derived stem/progenitor cells or pluripotent stem cells, some of which have been used in clinical trials. Careful selection of cell candidates, delivery methods, cell engraftment enhancement strategies, thorough investigation of efficacy mechanisms, and clinically meaningful endpoints in experimental studies can advance cardiac cell therapy. To date, consensus is reached that pluripotent stem cells are by far the most promising cell source for deriving functional cardiomyocytes for testing and therapy.
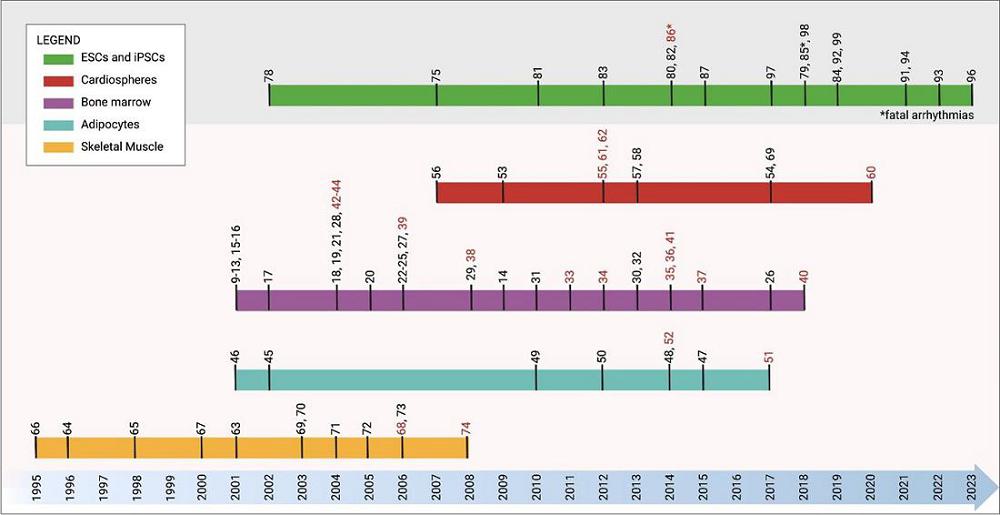
Figure 18. Timeline schematic portraying cell-based regenerative studies using both adult stem cells (skeletal muscle, adipocytes, bone marrow, and cardiosphere-derived cells) and ESCs/iPSCs.
[Pezhouman, Arash, et al. “Cardiac regeneration–Past advancements, current challenges, and future directions.” Journal of Molecular and Cellular Cardiology (2023)]
| 195 |
Potential Solution in Revolutionizing Cardiac Regeneration with Pluripotent Stem Cell Technologies
Stem cells offer hope for remuscularizing weakened, injured myocardium and reversing cardiac remodeling. Compelling evidence indicates that pluripotent stem cells (hPSCs), such as embryonic stem cells (ESCs) or human-induced pluripotent stem cells (hiPSCs), provide an indefinite source of CMs and are the only cells scalable to produce a clinically relevant number for myocardium remuscularization. Studies have demonstrated that intramyocardially injected hPSC-cardiomyocytes engraft, integrate synchronously with the host myocardium, regenerate remodeled, thin myocardium, and improve cardiac function.
While the clinical benefits of other adult stem cells, such as mesenchymal stromal cells or cardiac-derived progenitor cells, in cardiac regeneration are recognized, remuscularization in injured hearts using hPSC-cardiomyocytes has been more encouraging. Directed CMs differentiation of hiPSCs, employing systematic biochemical treatments, can generate spontaneously contracting cardiomyocytes with high efficiency in vitro. The application of hiPSC-derived CMs could have broad therapeutic applications in cardiac cellular therapy, myocardial tissue engineering, disease modeling, and drug screening for heart failure treatment. If hiPSC-derived epicardial cells can retain effects observed in the developing heart in vivo, they could theoretically enhance myocardial complexity and maturity in vitro.
Successful remuscularization requires the regeneration of dead myocardial tissue to restore muscle density and contractile strength. Various methods have been introduced to administer cells into the heart, aiming to maximize cell homing, retention, engraftment, survival, and function.
Generation of CM from autologous human induced pluripotent stem cells
Strategies to produce cardiomyocytes from hiPSCs have been well-established. However, the differentiation efficiency, purity, and homogeneity depend greatly on factors such as the choice of somatic cells used in reprogramming, the reprogramming method, the culture medium, colony selection, and expansion conditions. Here, we seek to develop a CM differentiation protocol that would consistently work for iPSCs reprogrammed from donor peripheral CD34 cells. This innovative approach aims to pinpoint specific iPSC colonies from reprogrammed populations, identify homogenous cardiac mesodermal cells through characterizing the metabolic attributes, surface markers and signaling, that could direct differentiate into homogenous, chamber specific CMs.
Allogenic human induced pluripotent stem cells remain the favorable cell source because they are cost effective, off-the-shelf cells with greater feasibility for clinical translation. However, patients receiving allogenic CMs are required to be on lifelong immunosuppression to ensure the transplanted CMs are not rejected by the host immunity. While manufacturing allogeneic cells for broad therapy is more cost-effective than producing personalized autologous cells, the long-term financial and health benefits for patients remain uncertain. Strategies to reduce the immunogenic reactivity of allogenic cells for transplantation, including the creation of hypoimmunogenic PSCs through gene editing, are still in the early phases of investigation, and the long-term consequences of gene-edited cell products have yet to be fully understood. Haplo-banking of iPSCs with selected haplotypes may be an alternative to serve a larger patient pool, if not all. However, this approach requires extensive donor cell typing and may pose challenges in nations with significant racial heterogeneity. Given these considerations, the refinement of existing methodologies to enable autologous cell therapy emerges as a compelling avenue. By refining and optimizing current processes, we aim to enhance the feasibility and effectiveness of personalized cell therapies, ultimately offering a more tailored and sustainable approach to regenerative medicine.
Potential Use of Non-Myocyte Cells to Enhance CM Functions: Key to Cardiac Regeneration
The IPSC Collaborators are building upon previous work in cardiac tissue formation to demonstrate a simple method for generating pre-epicardial cells (PECs) from hiPSCs (Refer Figure 19). PECs, a premature form of epicardial cells expressing typical epicardial genes WT1, TBX18, SEMA3D, and SCX, can develop into more mature epicardial cells (expressing additional markers UPK1B, ITGA4, ALDH1A2) after contact with CMs, recapitulating developmental roles in embryonic heart formations. Derived from the epicardium, a cell layer covering the heart’s surface, PECs significantly contribute to cardiac development and repair. Originating from the pro-epicardial organ, a transient organ from the lateral plate mesoderm (LPM) near the venous pole of the developing heart, these cells can transform into various cardiac cell types, including smooth muscle cells, fibroblasts, and endothelial cells, crucial for heart muscle regeneration, vascularization, and repair.
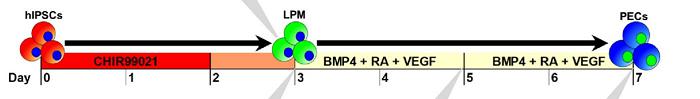
Figure 19. Stages of hiSPC Differentiation into Pre-Epicardial Cells (PECs) via Defined Growth Factor Modulation.
[Tan, J.J., Guyette, J.P., Miki, K. et al. Human iPS-derived pre-epicardial cells direct cardiomyocyte aggregation expansion and organization in vitro. Nat Commun 12, 4997 (2021).]
| 196 |
The current pipeline of the IPSC Collaborators focuses on developing a robust and efficient protocol to differentiate hiPSCs into highly pure and functional CMs and use PECs as the non-myocyte support cells to enhance the functionality and the outcome of CM therapy. This process involves precise manipulation of the cellular microenvironment, using specific growth factors and signaling molecules to mimic natural developmental cues directing PEC formation in the human heart.
Previous Success and Path to Clinical Application
The IPSC Collaborators have been working towards developing cardiomyocytes derived from human pluripotent stem cells that engraft, function, and persist in the human heart in vivo. The groundwork for potential future clinical development has been laid by key breakthroughs such as its preclinical studies have demonstrated the potential of hiPSC CMs to integrate into cardiac tissue, promoting repair and regeneration in damaged heart models. Combining non-myocyte support cells into CM therapy, the strategy is expected to not only contribute to reconstituting the infarct with functional cardiac muscle cells but also enhance the heart’s vascular network, improving blood supply and overall heart function.
Studies have shown that BMP4, RA, and Wnt signaling were key mechanisms driving PDGRFA+ and KDR+ LPM cells or cardiac progenitors, to an epicardial fate, some of which observed better efficiency with simultaneous activation of the signaling. The IPSC Collaborators have adapted the signaling model to develop an efficient BMP4/VEGF/RA based protocol, which generated >86% WT1+ pre-epicardial cells (PECs) in 7 days from monolayer hiPSC culture without the need of embryoid body generation, or fluorescence sorting. Taken together, this evidence suggests that hiPSC-derived PECs are functional and able to interact with CMs to enhance the function and structural organization in three-dimensional PEC/ CM microtissues. The IPSC Collaborators believe this work lends to continued efforts in directed morphogenesis, in which the strategic combination of early-stage cardiac cell types can enable the creation of more sophisticated and mature cardiac grafts.
Building upon these promising results, the IPSC Collaborators are advancing towards enabling the production of CMs and integrating non-myocyte support cells such as PECs into cellular therapy or the engineering of heart tissue for treating heart failure patients. The IPSC Collaborators’ objective is to develop a scalable and GMP-compliant production process that ensures the highest purity, potency, and safety of human induced pluripotent stem cell-derived cardiomyocytes (hiPSC-CMs), as well as the support cells, for therapeutic use.
Challenges and Innovations in hiPSC-PEC Therapy
Despite these achievements, hiPSC-derived CMs remain phenotypically immature, with underdeveloped organization and electromechanical function. Furthermore, bioengineered heart tissues using defined cardiac cells still lack the cellular and structural complexity of native myocardium, which could be greatly improved by implementing techniques that recapitulate and integrate important cues from heart development.
Developing hiPSC-PEC integrated CM tissue therapy involves overcoming several challenges, from ensuring the genetic stability and purity of the hiPSCs to achieving efficient differentiation into functional CM PECs. The Collaborators intend to pioneer innovative solutions to these challenges, including advanced genetic engineering techniques to enhance the safety profile of hiPSCs and novel bioreactor systems for large-scale PEC production.
| 197 |
Future Directions and Impact
Recent advancements in the worldwide scientific community’s iPSC technology have enabled the IPSC Collaborators to differentiate iPSCs into cardiac myocytes with high purity and consistency under GMP conditions—a significant step toward clinical application. Under the RCA for Preclinical Assessment of Cardiovascular Regenerative Therapy, the IPSC Collaborators have established heart failure rat models through ligating the left anterior descending coronary artery and cryoinjury. Additionally, the Collaborators have completed a preclinical trial using human iPSC-derived CMs to treat cryoinjured rat hearts. Early results demonstrate that the regenerative effect is superior when combining CMs with hiPSC-differentiated PECs, leading to enhanced regional vascularization and increased CM survival four weeks after injection (refer Figure 20 below). These findings represent a significant milestone, especially as the scientists extend the trial to examine the long-term therapeutic effects in large animals and ultimately prove the product’s safety in human trials. Nonetheless, the process of iPSC generation, the differentiation protocol, quality control, assessment of the cell product, and cell storage are marketable as services to customers and stakeholders (including research institutions, patients with special needs, mothers of newborn babies, etc.) during the development of the iPSC production pipeline on a large scale for human therapy.

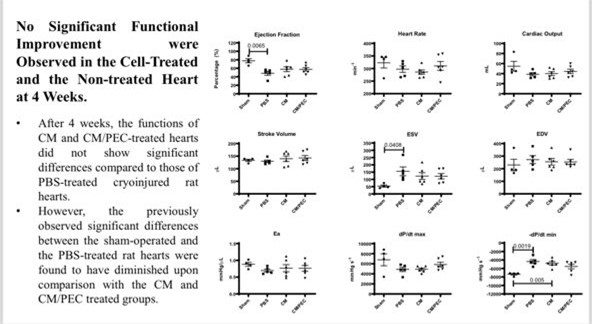
Figure 20. Regenerative Effect in Cell Treatment After 4 Weeks in Cryoinjured Rat Hearts.
Internationally, iPSC-based cardiac therapy research is evolving, with clinical trials in various phases. For instance, in Japan, pioneering efforts are underway to assess iPSC-derived cardiomyocytes in treating heart disease. The United States has seen substantial investment, as evidenced by a significant award granted for trials aimed at heart regeneration. Germany also contributes to this global effort, with advancements that may complement the wide-ranging international research landscape. Collectively, these efforts underline the potential of iPSCs to revolutionize cardiac therapy and highlight the substantial commercial and therapeutic promise of this technology.
The IPSC Collaborators’ objective is to establish the production of iPSCs derived from adult peripheral blood within a GMP-compliant framework, coupled with the strategic banking of these cells from donors with unique medical profiles. This endeavor will serve as the foundation for bespoke regenerative treatments. In the subsequent year, the IPSC Collaborators will focus on refining in-house protocols for the expansion and differentiation of iPSCs, with the aim of crafting cardiac cells of exceptional purity and reliability, again under cGMP guidelines. A patent application to safeguard our methods in iPSC production is slated for 2025. This lays the path for the commercial rollout of these groundbreaking cells over the next three to five years. The IPSC Collaborators’ commercialization efforts will include the provision of personalized cardiomyocyte production for therapy, development of robust heart cells for in vitro cardiotoxicity testing, and offering iPSC manufacturing services for individual or institutional needs, including cell storage and academic research. Additionally, the IPSC Collaborators aim to provide a comprehensive iPSC quality assessment service, meticulously evaluating iPSC karyotypes, genomic stability, and adaptations upon request. As the trial is collaborative effort between the IPSC Collaborators, the timing of subsequent clinical trial is not under our control, and is subject to change by USM.
| 198 |
mRNA (Diagnostic)
MyGenome is currently working on a comprehensive study and data collection on gene expression profiling, specifically targeting breast, lung, colorectal, and cervical cancers. The primary objective is to develop a diagnostic tool for these cancers, leveraging data from the nCounter PanCancer IO 360™ Panel. As of the date of this proxy statement/prospectus, Alps has not yet developed any diagnostic products or kits for commercial use. MyGenome is still in the process of collecting and analyzing data for the targeted breast, lung, colorectal and cervical cancers to support the development of the diagnostic tools.
One of our pipeline diagnostic candidates involves leveraging mRNA biomarkers for cancer diagnosis. Through our research and development efforts, this candidate focuses on analyzing specific mRNA molecules associated with cancerous cells to provide valuable insights into disease onset, progression, and treatment response. By examining the expression profiles of these mRNA biomarkers, we aim to develop highly sensitive and accurate diagnostic tools that can detect diseases at early stages with precision. Currently, we, in partnership with USM, are gathering specific cancer data for our mRNA diagnosis initiatives, with a targeted focus on breast, lung, colorectal, and cervical cancers. This pipeline is currently undergoing the POC stage, focusing on recruiting patients for breast, lung, cervical, and colorectal cancers and collecting data to develop diagnostic kits for these cancers. Upon completing the data gathering phase, MyGenome will commence the development of diagnostic prototypes for the identification of differentially expressed genes.
Overview of mRNA
Messenger RNA (mRNA) is a large family of RNA molecules that convey genetic information from DNA to specify the protein products of gene expression. mRNA gene expression profiling stands at the forefront of cutting-edge molecular biology, poised to revolutionize healthcare by offering advanced diagnostic and therapeutic solutions. mRNA biomarkers have the potential to shape the future of medical innovation and patient care. At its essence, mRNA serves as the conduit for translating genetic information from DNA into functional proteins, orchestrating fundamental cellular processes essential for health. Leveraging the power of molecular messengers, we are working to develop a cutting-edge platform that harnesses mRNA transcripts as dynamic biomarkers, providing unparalleled insights into disease pathology.
With one in four Malaysians affected by cancer, the urgency to advance diagnostic capabilities and therapeutic interventions are apparent.
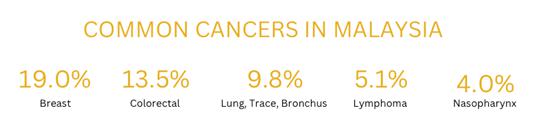
Figure 21. Prevalence of Common Cancers in Malaysia from 2012-2016 according to the National Cancer Registry (2019).
Breast cancer is the most common cancer in Malaysia. The common sites for invasive breast cancer to metastasize are the lung, liver, bone and brain while for cervical cancer, the frequent metastatic sites are the lungs, extra-pelvic nodes, liver and bones.
One of the standout advantages of mRNA biomarkers lies in their ability to deliver early and predictive insights, offering a proactive approach to healthcare management. mRNA testing serves several critical purposes in healthcare:
| ● | Early Disease Detection. mRNA testing enables the early detection of diseases, providing clinicians with valuable insights that can facilitate timely intervention and potentially prevent the onset or progression of illnesses. By detecting abnormalities in gene expression patterns, mRNA testing can identify subtle changes indicative of disease onset before symptoms manifest, allowing for proactive management and improved patient outcomes. This preemptive capability not only enhances treatment efficacy but also has the potential to mitigate disease burden and improve patient outcomes. |
| ● | Monitoring Disease Relapse. mRNA testing is beneficial in monitoring disease progression and detecting signs of relapse, particularly in conditions such as cancer. Changes in gene expression profiles can serve as early indicators of disease recurrence, prompting clinicians to adjust treatment strategies promptly and optimize patient care. Through regular mRNA testing, healthcare providers can closely monitor disease status and tailor therapeutic interventions to mitigate the risk of relapse. |
| ● | Personalized Treatment Approaches. mRNA testing facilitates personalized treatment approaches by analyzing dynamic gene expression patterns. Unlike static DNA tests, mRNA testing offers insights into real-time gene expression changes influenced by lifestyle and environmental factors. This dynamic nature allows for a more comprehensive understanding of disease progression and treatment response, enabling clinicians to tailor interventions to individual patient’s needs. |
| 199 |
There is a distinction between DNA and mRNA testing. While DNA tests offer insights into lifetime genetic risks, mRNA testing provides real-time assessments of present disease dynamics. This differentiation empowers healthcare providers with actionable information for personalized patient care, facilitating timely interventions and enhancing overall treatment efficacy.
| Aspect | DNA Testing | mRNA Testing | ||
| Genetic Material | DNA (Deoxyribonucleic Acid) | mRNA (Messenger Ribonucleic Acid) | ||
| Risk Assessment | Evaluates lifetime risks of cancer/disease | Evaluates current risk of cancer/disease | ||
| Genetic Stability | Genes do not change (static) | Gene expression can be influenced by lifestyle, diet, and other external factors | ||
| Test Consistency | Repeated tests will reveal the same results | Repeated tests will reveal current gene expression levels | ||
| Frequency of Testing | Typically performed once in a lifetime | Often performed annually to monitor improvements in gene expression levels |
Market Analysis
The global gene expression market, valued at USD 9.07 billion in 2020, is projected to exhibit a robust CAGR of 9.96% from 2021 to 2028.11 This growth trajectory is attributed to the emergence of technologically advanced solutions, offering promising avenues for market expansion.
The forecasted growth is underpinned by the development of sophisticated and specialized tests aimed at early disease detection and management, coupled with the rising demand for lab automation. Additionally, the increasing adoption of point-of-care diagnostic products has spurred a decentralization trend in the healthcare sector, empowering patients and healthcare facilities to avail medical services remotely, thus curbing costs. Other driving factors include the escalating prevalence of chronic diseases such as diabetes, heart failure, and colon cancer, alongside the surging demand for personalized medicine, expanding geriatric population, and growing patient awareness regarding disease diagnosis.
Our Solution
In the landscape of cancer diagnosis and monitoring, traditional approaches encounter significant challenges. These include limitations in clinical sampling for high-frequency detection and the constraints of early diagnosis and monitoring. Currently, reliance on tumor markers for screening and early cancer detection predominates in commercial labs’ practices. However, the inherent limitations of tumor markers underscore the urgent need for a more robust and reliable diagnostic paradigm.
Pursuant to the Research Collaboration Agreement, Alps Biotech has initiated a collaborative effort with USM to gather cancer data, specifically focusing on breast, lung, colorectal and cervical cancer. As of the date of this proxy statement/prospectus, the agreement has expired, and Alps is negotiation with USM to enter into a new research collaboration agreement for the next phase of research and development.
Our objective is to advance mRNA gene expression profiling, utilizing the latest advancements in molecular biology and bioinformatics to transform the standards of care, empower healthcare providers, and ultimately improve patient outcomes.
Our capabilities extend to investigating a multitude of gene expressions that intricately intertwine vital components within the complex dynamics of tumors, microenvironments, and immune responses in cancer. By employing the technology, it is possible to simultaneously identify over-expressed and under-expressed genes, offering a comprehensive view of gene expression dynamics. This unique capability enables for discerning genes undergoing significant changes during pivotal processes such as development, carcinogenesis, and disease progression.
11 MarketsandMarkets, “Gene Expression Analysis Market Size, Share, Trends and Revenue Forecast,” March 2024. Available at: Gene Expression Analysis Market Report.
| 200 |
Presently, the MyGenome biomarker laboratory uses the nCounter PanCancer IO 360™ Panel (the “Panel”) which is a unique 770 gene expression panel. According to the Panel’s manufacturer, the Panel integrates key elements essential for understanding the complex interactions between tumors, their surrounding environments, and the body’s immune response, thereby enabling a comprehensive analysis of disease biology and the exploration of immune evasion mechanisms. By leveraging the gene expression profiling capabilities of the Panel, MyGenome’s laboratory can predict the risk of incidence of the following 31 types of diseases:
| ● | Bladder cancer | |
| ● | Endometrial cancer | |
| ● | Liver cancer | |
| ● | Ovarian cancer | |
| ● | Brain Glioma | |
| ● | Esophagus cancer | |
| ● | Lung cancer | |
| ● | Pancreatic cancer | |
| ● | Breast cancer | |
| ● | Gastric cancer | |
| ● | Lymphoma | |
| ● | Prostate cancer | |
| ● | Cervical cancer | |
| ● | Head and neck cancer | |
| ● | Muscular sarcoma | |
| ● | Skin melanoma | |
| ● | Colorectal cancer | |
| ● | Kidney cancer | |
| ● | Nasopharyngeal cancer | |
| ● | Thyroid cancer | |
| ● | Alzheimer’s | |
| ● | Asthma | |
| ● | Atopic Eczema | |
| ● | Autism (Babies) | |
| ● | Cardiovascular disease | |
| ● | Diabetes | |
| ● | Hypertension | |
| ● | Obesity | |
| ● | Osteoarthritis | |
| ● | Parkinson’s | |
| ● | Bipolar disorder |
The Panel is not a standalone diagnostic tool and does not detect the presence of such 31 diseases. Instead, the data and information provided by the Panel, when integrated with additional bioinformatic data sequenced by MyGenome, will enhance its predictive ability. In addition to using the Panel to develop its mRNA diagnostic products, MyGenome also uses the Panel to provide genetic screening services in its wellness clinic.
MyGenome does not have any formal agreements, partnerships, or exclusive arrangements with the manufacturer of the Panel. Alps acquired the Panel as a standard purchaser and utilizes it within its facilities alongside other tools and methodologies to support its genetic screening services and mRNA diagnostic product development. The relationship with the Panel manufacturer is limited strictly to that of a buyer and manufacturer.
MyGenome’s mRNA diagnostic pipeline is currently dedicated to four (4) cancers – breast, lung, colorectal, and cervical cancers (the “Targeted Diseases”) – out of the 31 diseases mentioned above. To create its diagnostic products, in addition to relying on the gene expression capabilities of the Panel, MyGenome will need to accumulate data to enhance the precision of disease detection. As such, MyGenome selected the Targeted Diseases due to their high prevalence in Malaysia and clinical need for early detection. MyGenome’s current efforts are focused on gene expression profiling and data collection to advance the development of its proprietary diagnostic tool for the Targeted Diseases. While MyGenome is in the POC stage and actively gathering data, its studies and data indicate that each of the Targeted Disease exhibits distinct mRNA and genetic patterns, demonstrating correlations between the gene expression changes specifically associated with the Targeted Diseases. As MyGenome’s bioinformatics database expands, driven by the continuous accumulation of gene expression data, its ability to detect and characterize the molecular signatures of the Targeted Diseases will be further enhanced. This increase in bioinformatics data will provide deeper insights into the underlying gene expression patterns specific to each Targeted Disease, thereby improving the precision of MyGenome’s proprietary diagnostic tool for the Targeted Diseases.
Our mRNA Profiling Process
Our primary objective is the development of diagnostic tools for predicting the prognosis of the Targeted Diseases, thereby enabling early intervention for treatment. As at the date of this proxy statement/prospectus, MyGenome offers mRNA profiling services, which also serve as a channel for collecting bioinformatic data.
Our screening process begin with blood samples collected from the patient, either collected in-house or other healthcare providers.
| 201 |
Upon receipt of the blood sample, we initiate a meticulously structured workflow for mRNA testing, ensuring both accuracy and efficiency. The process unfolds as follows:
| 1. | Sample Logging and Identification: Each received blood sample is logged into our laboratory system, receiving a unique identifier to facilitate comprehensive tracking throughout the testing journey. |
| 2. | mRNA Extraction: Employing the QIAamp RNA Blood mini extraction kit and protocol (Qiagen), our scientists extract mRNA from the blood sample. This crucial step ensures the attainment of high-quality mRNA essential for subsequent analysis. |
| 3. | mRNA Quantification: The concentration and purity of the extracted mRNA are determined using the NanoDrop One (Thermo Scientific). This ensures that the mRNA sample meets stringent quality standards required for downstream analysis. |
| 4. | Hybridization and Detection: Following extraction, the mRNA undergoes hybridization with the probes in the nCounter PanCancer IO 360™ panel. Utilizing the cutting-edge NanoString nCounter sequencing platform, we accurately detect the expression levels of target genes. This analysis provides quantitative data on gene expression levels and the presence of disease biomarkers, furnishing crucial insights for precise diagnosis and treatment planning. |
| 5. | Analysis and Data Interpretation: The generated data then undergoes thorough interpretation utilizing the nSolver™ Analysis Software before uploading the results to Alps’ in-house mRNA system which generates the final report. |
| 6. | Reporting: A comprehensive report summarizing the mRNA testing results is generated by the Alps’ in-house mRNA system. This report encapsulates pertinent findings, insightful interpretations, and recommendations tailored to inform patient management. This report can be used by the patient and the patient’s physician to plan future tests and therapies. Figure 22 below details the diagnostics and recommendations the report provides based on the screening results. |
| 7. | Quality Control: Throughout the entire process, we adhere to stringent quality control measures to uphold the accuracy, reproducibility, and reliability of mRNA testing outcomes. This encompasses rigorous monitoring of instrument performance, adherence to validated protocols, and meticulous documentation of procedures. |



Figure 22. Sample pages from a report generated for the mRNA testing.
Through this systematic and meticulously executed process flow, we ensure the integrity and precision of mRNA testing results, thereby facilitating predictive disease detection and enabling personalized patient care.
| 202 |
Our Competitive Advantage
| ● | Established Wholly Owned Infrastructure: With a robust and fully owned infrastructure, Alps operates with autonomy and agility. Our dedicated team of scientists, renowned for their expertise in biomedicine and biotechnology, collaborates seamlessly within this infrastructure, enabling efficient execution of research and development initiatives. |
| ● | Facilitating Broader Access to Early-Stage Precision Medicine: We aim to broaden access to early-stage precision medicine, particularly in the realm of cancer treatments. Through efficient processes and resource optimization, we seek to offer cost-effective solutions that equip patients and healthcare providers with a range of diagnostic and therapeutic options. |
| ● | Maximizing Resources and Fostering Knowledge Sharing: At Alps, we recognize the importance of collaboration and knowledge exchange in driving innovation. Through strategic partnerships and alliances with external scientists and research centres, we maximize resources and leverage collective expertise to advance the frontier of WGS and mRNA gene expression profiling, ultimately enhancing patient care and outcomes globally. |
Development Status and Future Outlook
Currently, our mRNA profiling solely functions as a predictive test, with no ongoing development of new technologies or methodologies. Our primary focus is on accumulating sufficient data in respect of the Targeted Diseases, and we are actively engaged in the data collection process to ensure the accuracy of our tests and facilitate the development of its diagnostic products. MyGenome believes that with a sufficient volume of data, the diagnostic products will achieve higher accuracy in prognostic assessments of the Targeted Diseases. Our ultimate objective is to leverage these data to commercialize diagnostic kits for the Targeted Diseases, contingent upon obtaining necessary regulatory approvals.
CELESOME(+)
Exosome-infused cosmetic products hold potential for transforming skincare and beauty routines. By leveraging the potent properties of exosomes, we are committed to creating cosmetic formulations that enhance skin health, stimulate collagen production, alleviate inflammation, and enhance overall complexion. With Celestialab’s cGMP-accredited cell cultivation laboratory, Celestialab intends to develop, a robust and reproducible exosome manufacturing process. This process will be designed for large-scale operation, ensuring high productivity, purity, and quality, assuring compliance with regulatory standards, including good manufacturing practices.
Alps has completed its POC phase, which involved evaluating and refining exosome isolation methods and characterization. Alps is now finalizing an in-house protocol for further exosome characterization. After characterizing the exosomes, Alps plans to conduct comprehensive toxicity studies, including cytotoxicity tests and acute toxicity studies through animal models.
Exosomes in Cosmetic Industry
Exosomes, which are nano-sized vesicles ranging from 30 to 200 nanometers in diameter, are enriched with a diverse array of proteins, lipids, and genetic materials, including mRNA and microRNA (miRNA). These vesicles are produced by a wide range of cell types, notably stem cells, and are disseminated throughout the body via the bloodstream. The genesis of exosomes involves the inward budding of multivesicular bodies (MVBs), which contain intraluminal vesicles (ILVs); these are subsequently released into the extracellular space through the endosomal-lysosomal pathway. Playing a pivotal role in cellular communication, exosomes are instrumental in numerous biological functions, including the repair of tissue, modulation of the immune system, and facilitation of messaging between cells. Their anti-inflammatory and immunomodulatory properties are particularly valuable in procedures such as skin flap reconstructions.
The cosmetic industry is increasingly leveraging the potential of exosomes, incorporating them into various topical formulations such as creams, serums, and masks. These applications exploit the therapeutic and anti-aging properties of exosomes, delivering essential proteins, lipids, and other beneficial molecules that promote skin healing, hydration, and protection from environmental stressors. Notably, exosomes significantly contribute to the enhancement of collagen production, reduction of inflammation, and reinforcement of skin defense mechanisms. Additionally, the synergistic interaction of exosomes with other active ingredients, including hyaluronic acid, peptides, and antioxidants, potentiates their effectiveness.
| 203 |
Illustrative of their impact, adipose stem cell-derived conditioned media (ASC-CM) and bone marrow stem cell-derived (BMSC) exosomes have been shown to reduce levels of reactive oxygen species (ROS) and TNF-α while concurrently increasing TGF-β. This biochemical modulation results in elevated levels of MMP-1 and pro-collagen type I, leading to enhanced collagen synthesis, improved skin elasticity, and the visible reduction of wrinkles. Such outcomes establish a solid foundation for exosomes as an effective anti-aging treatment modality. By stimulating collagen production, exosomes aid in the diminishment of wrinkles and fine lines, support the repair of skin damage from sun exposure and acne scars, and contribute to overall skin plumpness and hydration. The inclusion of a spectrum of bioactive compounds within exosomes, such as cytokines, nucleic acids, and proteins, provides comprehensive protection against environmental stressors and aids in minimizing the appearance of dark spots and discoloration. Through these multifaceted therapeutic and aesthetic benefits, exosomes represent a ground-breaking and promising approach to enhancing skin tone, texture, and overall appearance in the field of regenerative medicine and cosmetic applications.
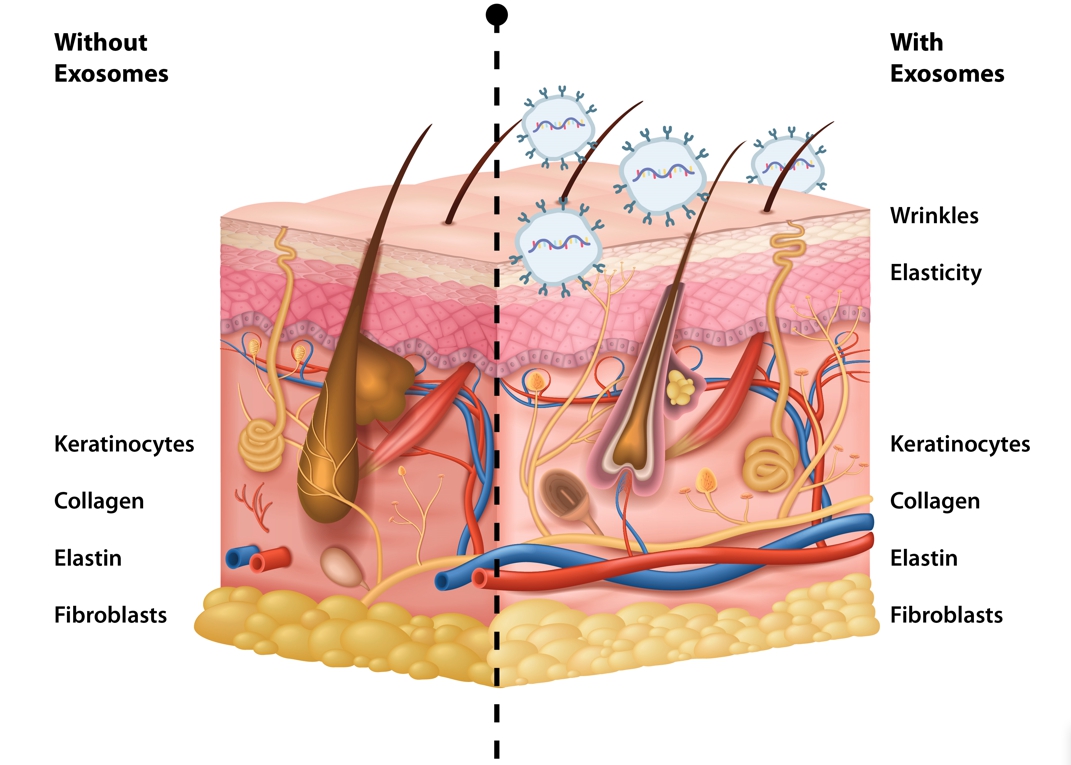
Figure 23. Role of exosomes for anti-aging treatment.
The application of adipose stem cell-derived conditioned media (ASC-CM) and bone marrow stem cell-derived exosomes (BMSC-exos) has demonstrated significant impacts on skin health, particularly in the context of anti-aging treatments. These interventions lead to a notable reduction in the production of reactive oxygen species (ROS), effectively minimizing oxidative stress within the skin. Concurrently, there’s a marked decrease in the expression levels of tumor necrosis factor-alpha (TNF-α), a cytokine known for its role in inflammation. In contrast, the expression of transforming growth factor-beta (TGF-β) is increased, which is crucial for its healing and regenerative properties.
The biochemical shifts induced by ASC-CM and BMSC-exos administration catalyze a series of downstream effects that are beneficial for skin rejuvenation. Specifically, there is an upregulation in the production of matrix metalloproteinase-1 (MMP-1) and pro-collagen type I. MMP-1 plays a pivotal role in the degradation of damaged collagen fibers, facilitating the turnover and renewal of the extracellular matrix. The increase in pro-collagen type I, a precursor of collagen, is particularly significant for the skin’s structural integrity and health.
These molecular and cellular responses cumulatively foster an environment conducive to enhanced collagen synthesis. As the foundational protein responsible for the skin’s strength and elasticity, collagen’s augmented production directly translates to improvements in skin elasticity and firmness. This not only aids in reducing the visibility of existing wrinkles but also prevents the formation of new ones, underscoring the effectiveness of ASC-CM and BMSC-exos as potent anti-aging therapies. By addressing the underlying causes of skin aging at a molecular level, these treatments offer a promising approach to achieving healthier, more youthful-looking skin, making them a cornerstone in the advancement of regenerative dermatology.
Evolving Demand in the Cosmetics Industry
The cosmetics industry is currently experiencing a significant surge in consumer demand, particularly in the realm of skincare products. This uptrend is attributed to a confluence of cultural and societal shifts, including a heightened emphasis on self-care, the pervasive influence of social media, and an increased awareness regarding the health implications of sun exposure. The philosophy underpinning the self-care movement posits skincare not just as a routine but as a form of personal stewardship for the body’s largest organ. Consequently, there’s a growing preference for products that are not only effective but also formulated with natural, organic, and ethically sourced ingredients.
| 204 |
Social media platforms have emerged as pivotal arenas for beauty discourse, with influencers and content creators sharing insights and recommendations that shape consumer preferences and behaviors. This digital proliferation of skincare knowledge has fostered a demand for products that cater to specific beauty ideals and skincare routines. Moreover, the recognition of sun damage’s long-term effects has catalyzed a demand for products offering robust protection against UV radiation, with sunscreens and SPF-infused moisturizers seeing heightened consumer interest.
Within this burgeoning market for skincare products, exosome-based therapies represent a novel frontier. Exosomes are being explored for their potential to rejuvenate facial skin and promote hair health. However, their integration into consumer products faces hurdles, primarily due to the absence of formal approval from regulatory bodies. Currently, the only stem cell treatments approved by FDA are products that treat certain cancers and disorders of the blood and immune system.
Our Ongoing Developments in Regenerative Dermatology and Cosmetology
Insights gathered from our own research endeavors, in conjunction with findings from parallel studies, underscore a probable correlative relationship between the specific composition of exosomes and their originating cells. This suggests that various cell types secrete uniquely characterized exosomes, each carrying distinct molecular payloads reflective of the parent cell’s function. Notably, human umbilical mesenchymal stem cells (hUMSCs) have been identified for their critical roles in modulating the immune response, facilitating cell growth, and managing cell turnover—key processes in the body’s healing, repair, and regeneration pathways. The remarkable capability of hUMSCs to produce functionally potent exosomes underpins our strategic decision to harness these cells for our advanced exosome-based therapies.
hUMSCs exhibit paracrine effects, releasing a variety of soluble factors that significantly enhance the body’s intrinsic repair and regeneration mechanisms upon topical application. This aspect of hUMSCs has paved the way for the development of cell therapies showing immense clinical promise, as evidenced by encouraging outcomes from pre-clinical and clinical evaluations. These studies have explored the efficacy and safety of hUMSC-based treatments across a spectrum of dermatological applications, including the management of skin burns, wounds, scars, wrinkles, and conditions such as psoriasis vulgaris and Romberg’s syndrome.
Our commitment to advancing regenerative dermatology and cosmetology solutions is supported by our in-house production facilities, which has the potential to source for high-quality hUMSCs exosomes. Our approach ensures a reliable and consistent supply of umbilical cords derived hUMSCs, ethically sourced in collaboration with our partner maternity clinics and based on informed consent from donors. The exosomes derived from these cells exhibit a concentration of 3.36e+10 +/- 1.96e+09 particles/ml, one of the highest concentration level of exosomes amongst the Malaysian players of similar scale and focus.
Potential products may be derived from this product candidate include facial masks, and treatments for both the face and scalp, incorporating our exosomes. By infusing such potential products with growth factor-rich exosomes from hUMSCs, we aim to redefine the standards of skincare and haircare.
The exosomes we utilize, approximately 62.1 nanometers in diameter, are enveloped in a lipid bilayer that not only safeguards their cargo but also enhances absorption by the skin, bolstering the skin’s moisture barrier. By delivering these growth factors topically, products can significantly rejuvenate the skin’s moisture barrier, promoting a healthier, more radiant complexion. Additionally, our ongoing research into the use of our exosomes in hair serums, face serums, ampoules, facial mesotherapy, and micro needling treatments is aimed at exploring their potential to visibly reduce hair shedding and improve scalp health.
We are also currently in the early stages of evaluating the adjunctive and topical use of our exosomes on through the use of pipeline hair serums, face serums, ampoules, facial mesotherapy, and facial micro needling. We hope our studies will show that if topically applied, our hUMSC’s exosomes will have a positive impact as it relates to noticeably reducing the appearance of hair shedding. We anticipate this effect will be primarily through hydrating and nourishing the scalp and topically applying nutrients to the scalp.
| 205 |
We operate a cGMP Stem Cell Research and Cultivation Laboratory, certified by the Ministry of Health in Malaysia. Our facility’s cleanrooms are classified as Grade B and C, as per the specified table of cleanroom classification:
| Cleanliness Level | Grade | Maximum concentration limits (particles/m3 of air) for particles equal to and larger than the considered sizes shown | ||||||
| At rest | In operation | |||||||
| ≥0.5 µm | ≥5.0 µm | ≥0.5 µm | ≥5.0 µm | |||||
| Extremely clean | A | 3,520 | 20 | 3520 | 20 | |||
| B | 3,520 | 29 | 352,000 | 2,900 | ||||
| Clean | C | 352,000 | 2,900 | 3,520,000 | 29,000 | |||
| D | 3,520,000 | 29,000 | Not Defined | Not Defined | ||||
Table 1: Cleanliness level classification and maximum particle concentration limits.
As part of our endeavour to introduce innovative exosome-infused cosmetic products into the Malaysian market, we are aligning with the regulatory framework established by the NPRA under the MOH. Unlike pharmaceuticals, clinical trials are not mandatory for cosmetic products. Our focus is on navigating the regulatory landscape efficiently, leveraging the unique positioning of cosmetic products within this framework.
Under the Malaysian regulatory framework, entities planning to introduce cosmetic products must follow a structured notification process. This entails formally declaring to the Director of Pharmaceutical Services (DPS) through the NPRA. The designated Cosmetic Notification Holder (CNH) must adhere to all specified requirements outlined in the relevant guidelines. Compliance encompasses various aspects, including product safety, quality, labeling, and advertising standards. Our potential product candidates, distinguished by their inclusion of in-house manufactured exosomes, benefit strategically from this regulatory pathway. As these products fall under the cosmetic category, they are exempt from the rigorous clinical trial requirements typically mandated for therapeutic products. This classification not only speeds up the time-to-market but also provides greater flexibility in product development and marketing strategies.
Microbiological Test
| TEST PARAMETER | UNIT | METHOD USED | RESULTS |
| Bacterial Endotoxin | EU/ml | In-house Method (ML-14) Based On USP<85> Using Limulus Amebocyte Lysate (LAL) kit by Cape Cod | ND(<0.25) |
Table 2: This table presents the results of a microbiological test for bacterial endotoxin levels in our exosomes sample. The sample tested indicating that the sample has very low to negligible levels of endotoxins.
Chemical/ Physical Test
| TEST PARAMETER | UNIT | METHOD USED | RESULTS |
| Mycoplasma | - | In-house Method, NA/PH/001, Using Real-time PCR | ABSENT |
Table 3: This table displays information about a microbiological test conducted to detect the presence of Mycoplasma in a sample. The result “ABSENT” indicates that the test did not detect any Mycoplasma DNA in the sample, suggesting that the sample is free from Mycoplasma contamination.
| 206 |
Microbiological Test
| MICROBIOLOGICAL TEST | RESULT | |||
| TEST PARAMETER | METHOD USED | Day 3 | Day 7 | Day 14 |
| Sterility Test | BP 2019, Vol. IV, Appendix XVIA | NO GROWTH | NO GROWTH | NO GROWTH |
Table 4: This table provides information about a sterility test performed on a sample, with results recorded over a 14-day period. The repeated confirmation of ‘NO GROWTH’ results across a two-week span is significant, as it not only supports the immediate sterility of the sample but also indicates stability in its sterility over time, which is essential for ensuring the safety of products with a longer shelf life.
Our Income Streams
Alps currently generates revenue through its subsidiaries, Alps Wellness Sdn. Bhd. and TMC Global Holdings Sdn. Bhd. These entities operate the Alps Wellness Centre and Alps Medical Centre, respectively. They offer a range of services, including surgical aesthetic treatments, non-surgical aesthetic treatments, and general healthcare and other wellness services. In the competitive biotechnology sector where high cash burn is typical for product and pipeline development, these revenue streams which allows Alps to sustain its operations and partially fund research and our product pipelines and reduce reliance on external funding. Additionally, Alps collaborates with third-party service providers to deliver certain services, enhancing its operational efficiency.
| (A) | Cosmetic and Aesthetic Services |
Our aesthetic services and cosmetic surgery services include:
Non-Surgical Aesthetic Services
Our non-surgical aesthetic services can be categorized into (i) skin treatments; (ii) facial sculpting; and (iii) body contouring. All treatments are performed by our qualified team of utilizing non-invasive or minimally-invasive procedures, and are personalized to each individual based on the analysis of their skin and personal objective.
| (i) | Skin Treatments | |
| Consistent facial and skin treatments are essential for maintaining optimal skin health. However, factors such as an unhealthy diet and the use of inappropriate off-the-shelf products can pose challenges in achieving and sustaining good skin health. Upon consultation, our doctors will create a customized treatment plan tailored to address each customer’s unique conditions and concerns. | ||
| To effectively address common skin issues such as acne, pigmentation, and scarring, we provide treatments that incorporate a variety of topical products and advanced medical equipment. For more complex procedures during skin treatments, we employ potent topical products like chemical solutions to induce skin peeling, along with specialized medical machinery and equipment. |
| 207 |
| Skin Treatments | Description | ||
| Chemical Peels | Chemical peels are effective in stimulating collagen and elastin production, thereby enhancing skin elasticity, reducing the appearance of fine lines, and promoting a more even skin tone by lightening dark spots and scars. These peels utilize an exfoliating acid solution that is both safe and gentle on the skin, making them a non-invasive option. This treatment is particularly beneficial for addressing deeper-set wrinkles and dark spots.
| ||
| Fractional CO2 Laser | The procedure involves directing short-pulsed energy onto the skin’s surface in a scanning pattern, removing damaged outer layers in thin increments. This process eliminates dead skin cells and activates the production of microthermal zones within the skin, stimulating the body’s natural healing response and promoting collagen production for skin remodeling. The goal of this treatment is to replace old and damaged skin cells with new, healthy ones.
| ||
| Radiofrequency (RF) | RF treatment is a non-invasive cosmetic procedure that uses radiofrequency energy to heat the skin’s deeper layers, promoting collagen production and tightening the skin. During the treatment, a device delivers controlled RF energy to the targeted area, generating heat that stimulates the production of new collagen fibers. This helps to improve skin elasticity, reduce the appearance of wrinkles and fine lines, and firm up sagging skin.
| ||
| Q-Switch Laser | This is a type of laser therapy that delivers short bursts of high-energy laser light to target specific pigments in the skin, such as melanin (responsible for pigmentation like freckles, age spots, and sun spots) or tattoo ink. In addition to targeting pigmentation, Q-switched lasers can also stimulate collagen production and improve overall skin texture, leading to a more youthful and radiant complexion. |
| (ii) | Facial Sculpting | |
| As we age, the natural contours of our face can diminish, leading to an older appearance or asymmetry. We provide non-invasive facial sculpting treatments aimed at restoring a youthful look and enhancing natural beauty. Through professional consultation and personalized treatment planning by our doctors, we offer treatments designed to rejuvenate and enhance natural beauty. Key treatments offered by Alps include: |
| ● | Dermal Fillers | |
| Hyaluronic Acid fillers are proven safe as dermal fillers and are effective to smoothen the appearance of deep grooves and facial wrinkles, enhancing facial features and restoring lost volume and facial structure. Dermal fillers give a natural result without affecting the facial expression. | ||
| ● | Botox | |
| Botox treatment is the single most popular aesthetic procedure worldwide and is incredibly safe. It helps to correct facial lines and dynamic wrinkles such as frown lines, forehead creases and crow’s feet. It also helps in treating hyperhidrosis. | ||
| ● | High Intensity Focused Ultrasound (HIFU) | |
| HIFU lifting is a non-invasive alternative to traditional facelift surgery to perform a skin tightening treatment. This procedure utilises ultrasound energy to target the layers of skin below the surface to encourage the production of collagen for firmer skin. |
| 208 |
| ● | Platelet Rich Plasma (PRP) | |
| PRP is effective for facial rejuvenation. It uses the patient’s own blood to obtain a high concentration of platelet and growth factors. It is injected into the whole face, and this ignites tissue regeneration, blood vessel formation and collagen production. Wrinkles, photodamaged skin, and skin discoloration can be treated by PRP injections. | ||
| ● | Non-Surgical Thread Lifts | |
| Thread lift is one of the most effective ways to lift the loose skin without surgery. This procedure is suitable for the eyebrows, areas around the eyes, the cheeks, the jowls and the neck. Thread lifts treatments result in an instant and natural looking improvement in the appearance of the sagging skin. |
| (iii) | Body Contouring |
| ● | Cryolipolysis | |
| Cryolipolysis treatment is a procedure commonly known as fat freezing, is a non-surgical fat reduction procedure that uses intense cold temperatures to freeze the layer of fat cells underneath the skin. The cold temperatures damage the fat cells which triggers an inflammatory response by the body which causes the body to dispel the fat cells. | ||
| ● | Fat Melting Injection | |
| Fat Melting Injection is a non-surgical cosmetic procedure aimed at reducing localized fat deposits in various areas of the body. It involves the injection of a solution directly into the targeted fatty tissue, which breaks down the fat cells over time. It offers a minimally invasive alternative to traditional liposuction, with minimal downtime. |
Surgical Aesthetic Services
We provide customary surgical aesthetic services, including double eye lid surgery, nasal augmentation, lip augmentation, breast augmentation, fat transfer, liposuction, lifts and tucks operation and thread lifts, among others. Surgical aesthetic treatments typically involve local or full anesthesia, and an extended recovery time. All of our surgical aesthetic treatments are performed in fully equipped operating rooms in our treatment center.
| Treatment | Description | |
| Double Eye Lids Surgery | Enhances the appearance of the eyes by creating or refining double eyelids for symmetry. | |
| Nose Surgery | Change the shape or appearance of the nose by adding fillers or cartilage. | |
| Lip Augmentation | Enhances the volume, shape, or definition of the lips using fillers or implants for a fuller appearance. | |
| Breast Augmentation | Procedures designed to enlarge/reduce or change the shape of the breasts. | |
| Fat Transfer | Transfers fat from one area of the body to another to add volume or improve contours, commonly used for the face, buttocks, or breasts. | |
| Liposuction | Procedure in which excess fatty tissue is removed from a specific part of the body through suction | |
| Lifts & Tucks Operation | Tightens and removes excess skin from various body areas to improve contours and firmness, commonly performed on the face, neck, arms, or abdomen. | |
| Thread lift | Uses dissolvable threads inserted under the skin to lift and tighten sagging facial tissues. |
Due to their complexity, we require all our surgical aesthetic procedures be conducted by highly experienced doctors. We implement a standardized process for treatments to uphold consistent quality and safety standards. Our surgeon, Dr. Benjamin GEORGE Jr., who is also the Medical Director for Alps Medical Centre, graduated from Royal College of Surgeons in Ireland with a Bachelor of Medicine and Surgery (M.B.B.Ch.) and obtained his Fellows of the Royal College of Surgeons FRCS (Ed) Surgery in the Royal College of Surgeons, Edinburgh. With over 30 years of experience in cosmetic, plastic, and reconstructive surgery, he holds an LCP certification and is an active member of several medical associations, including the Malaysian Association of Plastic and Cranio-Facial Surgeons, Academy of Medicine, Malaysia, and Malaysian Medical Association.
| 209 |
| (B) | Hair Transplant Solutions |
We provide hair transplant procedures, including techniques for natural looking hair, beard, scar revision on scalp, and eyebrow transplantation, as well as other non-surgical hair loss treatments. Factors such as stress, genetics, hormonal imbalances, and environmental pollutants contribute to hair loss. These modern influences, combined with lifestyle choices like poor diet and styling practices, play a significant role in causing hair loss among individuals.
At Alps, we specialize in the follicular unit extraction (FUE) method for hair restoration. This technique involves harvesting hair follicles from a donor area and transplanting them to areas experiencing hair loss. Our approach is tailored to each individual’s needs, ensuring personalized care and optimal results. Unlike the follicular unit transplantation (FUT) method, FUE leaves minimal scarring, making it a preferred option for many clients.
Our medical team consists of highly skilled specialists with extensive experience in hair transplantation. With a track record of thousands of successful procedures, we demonstrate our dedication to achieving excellence in every aspect of our practice. Each patient’s journey begins with a thorough private consultation, during which we conduct a detailed scalp analysis and develop a comprehensive plan tailored to promote hair regrowth.
Our facilities are equipped with technology to maintain the success rates for every graft implanted. Our methodology combines the latest medical advancements with a deep understanding of human biology to optimize the health and longevity of transplanted hair.
We distinguish ourselves from competitors through meticulous processes that include the use of optimal hair holding solutions, growth-promoting nutritional formulas, and precision temperature-controlled environments for graft preservation.
What makes Alps unique is our ability to conduct comprehensive genetic screenings to uncover underlying causes of hair loss related to inherited genes. This service allows us to offer personalized solutions and preventive strategies to slow down or reverse hair loss.
| (C) | General Medical and Wellness Services |
We offer a range of general health and wellness services, including medical consultation, health screenings, cellular therapy and research and COVID-19 testing. For the medical consultations, our doctors will conduct an assessment to provide a diagnosis, medical advice, and offer a relevant treatment plan for our customers. In addition, we offer a comprehensive range of health screenings – such as diabetic screening tests, liver function tests, mineral tests, general tumor markers, lipid profiles, and more, to identify and address potential health concerns.
Additionally, Alps Medical Center provides professional dietetic consultation and guidance. We offer personalized nutritional and dietary advice tailored to individual needs, along with lifestyle recommendations. Our services include specialized weight management programs and one-on-one nutrition coaching. To complement our consultations, we offer a range of supplements, designed to support various health needs and enhance overall well-being.
| (D) | Whole Genome Sequencing (WGS) and Pharmacogenomics |
Apart from the above, we currently offer comprehensive genomic services, specializing in WGS, and are strategically positioned to expand our offerings to include pharmacogenomics. Our services are aimed at personalized healthcare and precision medicine dedicated to empowering individuals and healthcare providers with actionable genetic insights. Leveraging our advance technology and expertise, we offer a wide range of genomic services tailored to meet the diverse needs of our clients.
| 210 |
Our pharmacogenomics analysis system is currently in the development stage, where we are currently in the process of refining the algorithm required for interpreting genetic data to ensure a comprehensive result generated. We anticipate launching our pharmacogenomics analysis service within a year from the date of this proxy statement/prospectus.
Background of Genetic Sequencing
In living organisms, genetic information is transmitted through deoxyribonucleic acid (DNA), which stores information in linear sequences of chemical bases (adenine (“A”), cytosine (“C”), guanine (“G”), and thymine (“T”)). Within cells, these sequences form pairs in a double helix structure by complementary base pairing. An organism’s entire DNA sequence, its genome, comprises approximately three billion base pairs in humans. The arrangement of these bases on the DNA molecule creates the genetic code, which contains instructions for protein synthesis and regulating biological functions. DNA replication, transcription, and translation are essential biological activities that depend on the structure and function of DNA.
Genetic sequencing involves determining the sequence of nucleotide bases (A, C, G, or T) in a sample, comprising three main phases: sample preparation, sequencing, and raw data analysis. Initially, during sample preparation, the target genome is fragmented into smaller pieces or amplified, depending on the available DNA amount. In the sequencing phase, individual bases within each fragment are identified, forming sequence reads, with the length of contiguous bases termed “read length”. Sequencing throughput is calculated as the product of the number of sequence reads and their average length. Subsequently, bioinformatics tools are used in the analysis phase to align overlapping reads, reconstructing the original genome sequence.
Exploring genomes aids in understanding biological inheritance, developmental processes, and normal and pathological cell and organism states. Genetic variability contributes to individual differences, including traits like eye color and blood type, and influences disease susceptibility, such as cancer, heart disease, or diabetes. Moreover, genetic variability affects individual responses to drug therapies.
Genetic sequencing, the foundation of WGS and Pharmacogenomics, provides insights into biological inheritance, disease susceptibility, and individual responses to drug therapies. As healthcare trends shift towards personalized medicine, integrating genomic data enables more precise predictive outcomes and treatment strategies. This transition towards precision-personalized medicine promises earlier disease detection, reduced healthcare costs, and optimized treatment efficacy, ultimately enhancing patient quality of life.
Current genomic testing offers insights into disease predisposition and medication response based on an individual’s unique genetic makeup. Researchers recognize the genomics potential, facilitating health assessment, early disease detection, treatment selection, monitoring treatment response, and identifying adverse effects.
Overview of Pharmacogenomics
Pharmacogenomics is an innovative approach that customizes treatment strategies to improve the effectiveness and safety of drug prescriptions. This field acknowledges the impact of genetic backgrounds on drug responses, highlighting the significance of personalized medicine. It offers the potential in optimizing drug therapy by tailoring treatments to each patient’s genetic profile based on the understanding that genetic differences among individuals influence how their body respond to medications of specific drug.
The primary goals and applications of pharmacogenomics are multifaceted. Firstly, it aims to optimize drug therapy by tailoring treatments based on a patient’s genotype, with the ultimate goal of achieving maximum efficacy while minimizing adverse effects. Additionally, pharmacogenomics contributes to the advancement of precision medicine, shifting away from the traditional “one-dose-fits-all” approach towards personalized medicine, where drugs are optimized for specific subsets of patients or even customized to an individual’s unique genetic profile. Furthermore, pharmacogenomics plays a crucial role in reducing trial and error in medication management by providing physicians with genetic insights to make informed decisions about drug dosages. This approach not only minimizes trial-and-error but also provides explanations for past treatment failures, paving the way for more effective and personalized healthcare interventions.
| 211 |
Market Industry
The global DNA sequencing market is projected to reach USD 23.56 billion by 2026, with a compound annual growth rate (CAGR) of 20.85% from 2021.12 The demand for services facilitating the sequencing of the entire human genome is increasing, driven by the need to convert vast amounts of raw genetic data into medically valuable insights. Automation in pre-sequencing protocols, post-sequencing protocols, and data analysis is expected to positively impact the whole genome sequencing (WGS) market. Additionally, the adoption of novel platforms for personalized medicine development at a genetic level is anticipated to drive demand for WGS in the coming years.
The demand for disease management, prevention, and treatment, along with advancements in bioinformatics, will further fuel the market, aligning with Alps’ core focus on utilizing in-house WGS capabilities. Additionally, the global rise in cancer prevalence and the availability of advanced healthcare facilities are expected to drive market growth. According to GLOBOCAN 2020, new cancer cases are projected to increase from 192,927,89 in 2020 to 288,879,40 by 2040, thereby stimulating cancer research and accelerating growth in the Next-Generation Sequencing (NGS) market.
The Problem
The challenges inherent in conventional treatments are multifaceted:
| ● | The pervasive use of conventional, uniform treatment modalities has led to widespread drug resistance, diminishing the efficacy of therapies provided to patients. This “one-size-fits-all” approach fails to account for the individual variability in drug response, leading to suboptimal outcomes. |
| ● | In oncology, the majority of diseases are associated with genetic mutations, some of which are hereditary. These life-threatening conditions are seldom detected during the initial stages when interventions could be more effective. Targeted treatment on specific genetic mutations potentially minimize treatment needed. |
| ● | Finding the right medication often involve a lengthy process of trial and error. Prolonged trial and error delay achieving the desired therapeutic outcomes, while patient’s condition may worsen and potentially leading to complications that could have been avoided with more immediate effective treatment. Furthermore, this can also put a strain on healthcare resources. |
Advancements in Genomic Sequencing
Personalized medicine represents a paradigm shift in healthcare, aiming to tailor medical treatments to the unique needs of individual patients. This specialized field holds promise for the future expansion of genomic sequencing technologies, offering unprecedented insights into disease susceptibility, treatment efficacy, and patient outcomes. Ongoing research in evolutionary biology is leveraging genomic sequencing to deepen our understanding of disease susceptibility and predict individual responses to medications. By deciphering the intricate genetic makeup of patients, genomic sequencing emerges as an invaluable tool in driving healthcare advancements and improving patient care.
Personalized targeted sequencing plays a crucial role in accelerating the identification of mutations and pinpointing cancer targets or pathways, facilitating the development of pharmacological treatments. The integration of NGS protocols into global-scale projects is leading to the generation of new foundational knowledge in oncology precision medicine.
12 Research and Markets. (November 16, 2022). Global DNA Sequencing Market to Grow by $23.56 Billion During 2022-2026 - ResearchAndMarkets.com. Business Wire. Retrieved from https://www.businesswire.com/news/home/20221116005663/en/ Global-DNA-Sequencing-Market-to-Grow-by-23.56-Billion-During-2022-2026---ResearchAndMarkets.com
13 Please refer to https://www.technavio.com/report/next-generation-sequencing-market-size-industry-analysis
| 212 |
Alps’ subsidiary, MyGenome, is leveraging on its current WGS analysis pipeline by integrating pharmacogenomics analysis into the existing framework. While our in-house WGS analysis has traditionally focused on analyzing patients’ traits and wellness, this integration represents a groundbreaking leap forward in genetic analysis capabilities. By incorporating insights into the characteristics of drug treatment for individual patients, we achieve a more comprehensive understanding of each individual’s genetic profile. This holistic approach not only enhances our ability to provide personalized healthcare but also enables us to offer tailored treatment strategies based on an individual’s unique genetic makeup. This integration promises to revolutionize genomic analysis, empowering both patients and healthcare providers with actionable insights for optimized treatment outcomes and patient safety.
Effectiveness of Pharmacogenomics
| 1. | Optimization of Therapeutics: Pharmacogenomics testing has demonstrated its potential in optimizing therapeutics. From genetic variations, pharmacogenomic analysis can enhance treatment efficacy and safety, ensuring that medications are tailored to individual genetic profiles for improved outcomes. |
| 2. | Reducing Adverse Drug Reactions (ADRs): Pharmacogenomics analysis enables the identification of risk of ADRs based on an individual’s genetic profiles. Predictive capability from pharmacogenomics analysis can significantly reduce the incidence of ADRs which may lead to hospitalization or death. |
| 3. | Personalized Medicine: Unlike traditional approaches that rely on population averages, pharmacogenomics testing predicts medication responses based on an individual’s genetic variations. This personalized approach ensures that treatments are tailored to each patient’s genetic makeup, optimizing effectiveness and minimizing adverse reactions. By offering insights into how genetic variations of an individual handle a specific drug differently, pharmacogenomics testing enables personalized medication selection and dosage, leading to improved treatment outcomes and patient safety. |
Our Methodology
WGS allows for genetic assessment, delivering comprehensive insights by mapping the entire DNA sequence. For individual’s intent on uncovering their complete genetic profile, WGS requires just a one-time analysis. This methodology is a foundational element in the evolution of personalized medicine, offering clarity on the functions and variations of genes. Our services efficiently furnish detailed genetic information that can be pivotal in transforming patient-specific treatment modalities, refining existing drugs and therapies, and even paving the way for curing diseases.
Leveraging public established genetic data, our WGS technology decodes every segment of the genome, including the often-overlooked intronic regions, as well as the coding exons. With the capacity to promptly generate data comprising billions of base pairs at a 30x read depth, we produce what can be considered clinical-grade data. We believe that the comprehensive scope and the non-discriminatory nature of our genomic analysis means that clinical-grade whole-genome sequencing (cWGS) is on the cusp of setting a new standard in medical practice, with the potential to be integrated as a routine element in clinical diagnostics.
| 213 |

Figure 24. Comparative Analysis of SNP Genotyping, Whole Exome Sequencing (WES), and WGS in Precision Medicine.
Moreover, we are utilizing a platform that merges raw genomic data with sophisticated bioinformatics software, known as novoClinic. This platform equips end users with the means to optimize health and wellness decisions by granting them access to an in-depth understanding of their genetic makeup. It provides actionable insights that are both relevant and tailored to individual health profiles, enabling users to make informed choices about their well-being.
Our process begins with the collection of a blood sample from the patient. Subsequently, the blood sample undergoes processing, during which DNA is extracted. This extracted DNA is then subjected to WGS to obtain the raw genetic data. Following this, a comprehensive genetic analysis is conducted on the obtained data, enabling the identification of genetic variations.
These genetic variations are then interpreted in the context of pharmacogenomics markers and drug-gene interactions. This interpretation process allows us to derive actionable insights that inform decision-making processes, including personalized medication selection, dosage adjustments, or tailored treatment plans for individual patients, optimizing therapeutic outcomes and enhancing patient safety and efficacy. A comprehensive report of the testing results will be generated. We utilize the findings from the pharmacogenetic test, in conjunction with other pertinent information about the patient’s condition, to tailor their treatment regimen or make adjustments as necessary. This may involve recommending the most suitable type of medication for their body and determining the optimal dosage to maximize therapeutic benefits. Additionally, the test results can help predict whether the client is likely to experience any adverse side effects from certain medications. Should the test results indicate that a particular medication is not well-suited, we will explore alternative treatment options that may better suit the genetic makeup and medical needs.
NovoClinic is the analysis software used for our whole genome sequencing data. NovoClinic is a platform for NGS targeted sequence analysis, focusing on personalized medicine. It is a patient-centric NGS targeted sequence analysis platform that provides integrated sample tracking for quality control and compliance. This software resolves the burden of data mining and interpretation, allowing researchers to focus on genetic changes and mutations. NovoClinic allows built-in customizable analysis of pipeline including data trimming, sequence alignment, variant calling and variant annotation from various established databases including GWAS, clinvar, dbSNP, COSMIC, etc., to generate both wellness and clinical reports.
| 214 |
Our Unique Capabilities
We believe our services impart the following unique capabilities, which serve as the core tenets for the development:
Built-in customizable analysis of pipeline: Having our built-in customizable analysis pipeline for whole genome sequencing empowers our researchers with:
| ● | Flexibility: customizable pipelines allow researchers to tailor the analysis workflow to their specific research questions, experimental design, and data characteristics; | |
| ● | Efficiency: customizable pipelines streamline the analysis process by automating repetitive tasks and integrating multiple analysis steps into a coherent workflow. This automation reduces the manual effort required for data processing, analysis, and result interpretation, allowing researchers to focus on higher-level tasks and insights, scalability, integration, community support, and adaptability, enabling comprehensive and customized analysis of genomic data for various research and clinical applications; and | |
| ● | Adaptability: our researchers are able to incorporate updates, improvements, and new features into the pipeline as advances in technology, algorithms, and genomic knowledge emerge, ensuring that the analysis workflow remains current and relevant. |
In-House Laboratory: Equipped with our own laboratory facilities, we have full control over the entire genetic testing process. This ensures the highest standards of quality control, confidentiality, and efficiency in delivering results to our clients. By managing the entire testing process in-house, we can expedite turnaround times and maintain the integrity of the genetic data, enhancing the overall reliability and accuracy of our services.
High accuracy: The DNBSEQ™ WGS sequencer, which is our current genetic sequencing tool is designed to provide high sensitivity and accuracy for each base detected during sequencing for reliable data generation. According to the specifications and performance benchmarks provided by MGI Tech Co., Ltd , the sequencer is anticipated to achieve an SNP and Indel calling rate of 99.9% and 99%, respectively, along with a decreased duplication rate of less than 2 percent, and an almost negligible Index mis-assignment rate.
Competition
The biotechnology sector, particularly the cell therapy segment, is characterized by rapid technological advancements, intense competition, and a strong focus on intellectual property. Despite our belief in the competitiveness of our approach, strategy, technology, knowledge, and experience, we face significant competition both in our current product development pipeline and in potential future product candidates. Competitors include major pharmaceutical and biotechnology companies, academic research institutions, governmental agencies, and public/private research institutions worldwide. Many of these competitors possess greater financial resources and expertise across various stages of product development, from research and development to regulatory approval and marketing. Mergers and acquisitions within the industry may further concentrate resources among fewer competitors, while smaller or early-stage companies could also pose significant competition, either independently or through partnerships with larger entities.
Competition extends beyond product development to areas such as recruitment and retention of scientific and management talent, establishing clinical trial sites, patient enrollment, and acquiring complementary technologies. Consequently, competitors may introduce products that are safer, more effective, have fewer side effects, are more convenient, or are less expensive than ours, potentially reducing or eliminating our commercial opportunity. Rapid regulatory approvals by competitors could also result in them establishing a dominant market position before we can enter. Key factors influencing the success of our programs include efficacy, safety, convenience, pricing, and reimbursement considerations.
We aim to research and develop cell-based therapies for a broader spectrum of diseases, including cancers, general wellness, and anti-aging, demonstrating a more expansive and inclusive approach to addressing health and wellness challenges.
| 215 |
Additionally, the aesthetic and beauty-related care industry in Malaysia is highly competitive. We face competition from various providers of cosmetic and aesthetic services, including aesthetic medical clinics and private hospitals. Furthermore, some of our competitors, such as private hospitals, have a longer operating history with stronger financial strength and more resources compared to us.
Competitive Strengths
We believe that we possess numerous competitive advantages. These strengths include:
| ● | Regional Efficacy and Efficiency: Alps is strategically positioned to deliver highly effective and efficient vaccines tailored for the Southeast Asian region. By understanding the unique epidemiological landscape and healthcare infrastructure of the region, Alps aim to develop vaccines that address specific regional needs, thereby enhancing their efficacy and impact. | |
| ● | Proprietary Platform Technology: Alps possesses its own development platform, enabling the rapid and streamlined production. This proprietary platform allows for faster turnaround times from product conceptualization to market launch, providing a competitive edge in responding to emerging infectious diseases and public health crises. | |
| ● | Strategic Location in Malaysia: Malaysia’s status as a member of the Pharmaceutical Inspection Cooperation Scheme (PICs) enhances Alps’ regulatory capabilities and facilitates smoother international collaborations. The country’s favorable regulatory environment and infrastructure support expedited approval processes, enabling Alps to bring product candidates to market more efficiently. | |
| ● | Cost Effectiveness: Alps prioritizes cost-effectiveness in product development and manufacturing processes across all pipelines and our services. Through efficient resource utilization, streamlined operations, and strategic partnerships, Alps aims to deliver high-quality products and services at competitive prices, making them accessible to a broader population. | |
| ● | Expertise and Talent: Alps boasts a multidisciplinary team comprising experts in various fields, including scientific research, clinical trials, manufacturing, business development, and leadership. This diverse talent pool brings together extensive experience and knowledge in biotechnology, platform research, drug discovery, and development, enabling Alps to innovate and excel in the various competitive markets. | |
| ● | Tax Incentives: Malaysia’s favourable tax policies and incentives further enhance Alps’ competitive advantage. Lower taxes and investment incentives encourage business growth and innovation, allowing Alps to allocate resources more efficiently towards research and development efforts. |
| 216 |
Medical Tourism in Malaysia
Malaysia has emerged as a premier destination for medical tourism, attributed to its provision of high-quality services, competitive medical rates, and access to advanced medical facilities. The government has implemented various initiatives to promote healthcare travel, aiming to bolster Malaysia’s reputation as a hub for top-notch healthcare by enhancing medical treatment quality, embracing medical digitization, and ensuring affordability.

Chart 1 and 2 illustrate a consistent upward trend in both Malaysia’s revenue and the number of tourists visiting for medical purposes from 2015 (859,000 travelers, USD 205 million) to 2019 (1.2 million tourists, USD 382 million) according to the statistics reported by the Malaysian Healthcare Travel Council.14
The majority of medical tourism in Malaysia is facilitated through private medical facilities, offering a wide range of services including dental care, cosmetic surgery, elective surgery, and reproductive therapy. Malaysia distinguishes itself by integrating both modern and traditional medical practices within its healthcare sector. The enactment of the Traditional and Complementary Medicine Act 2016 exemplifies Malaysia’s commitment to integrating traditional and complementary medicine into its healthcare system, ensuring the delivery of high-quality care through proper registration and education of practitioners. This has contributed to the growing popularity of traditional and complementary medicine among medical tourists seeking alternative yet safe treatments for various ailments.
In addition, the Malaysia Healthcare Travel Industry Blueprint 2021-2025 established by the Malaysia Healthcare Travel Council (MHTC) outlines a strategic roadmap for the development and enhancement of the healthcare travel sector in Malaysia over the next five years. This blueprint aims to leverage Malaysia’s strengths in healthcare to position the country as a leading destination for medical tourism globally. Its key strategies include improving the quality of medical care, digitizing the patient journey and continuing efforts to be a world-leading destination that provides affordable medical offerings to healthcare travelers.
Malaysia’s excellence in healthcare has garnered international recognition, with accolades such as being named the ‘Best Country in the World for Healthcare’ by US-based International Living magazine from 2015 to 2017 and in 2019. Additionally, the UK-based International Medical Travel Journal recognized Malaysia as the ‘Destination of the Year’ for healthcare travel from 2015 to 2017, coinciding with the milestone achievement of one million medical tourism arrivals in Malaysia in 2017.
14 Please see: https://www.mhtc.org.my/statistics/
| 217 |
Description of Properties
As of the date of this proxy statement/prospectus, we currently occupy the following properties:
| Company Name | Address | Purpose | Tenancy Period | Monthly Rental | ||||
| Alps Global Holding Berhad | Suites 02-07 & 02-08, 2nd Floor, Menara See Hoy Chan, No. 374, Jalan Tun Razak, 50400 Kuala Lumpur, Malaysia. | Holding and management company (approx. 3,606 sq. ft.) | 1 January 2022 to 31 December 2024 | RM 11,358.90 (approximately US$ 2,409.35) | ||||
| Celestialab Sdn. Bhd. | Suites G-05, Ground Floor Menara See Hoy Chan, No. 374, Jalan Tun Razak, 50400 Kuala Lumpur, Malaysia. | Cell manufacturing lab, R&D (approx. 5,301 sq. ft.) | 1 March 2022 to 28 February 2025 | RM 22,529.25 ((approximately US$ 4,778.71) | ||||
| MyGenome Sdn. Bhd. | Suites G-03 & G-04, Ground Floor, Menara See Hoy Chan, No. 374, Jalan Tun Razak, 50400, Kuala Lumpur, Malaysia. | Molecular lab, R&D (approx. 4,853 sq. ft.) | 1 March 2022 to 28 February 2025 | RM 20,625.25 (approximately US$ 4,368.37) | ||||
| Alpscap Berhad | E-18-01 & E-18-02, Level 18, The ICON, No 1, Jalan 1/68F, Jalan Tun Razak, 50400 Kuala Lumpur, Malaysia. | HQ office (approx. 14,180 sq. ft.) | 1 July 2023 to 30 June 2026 | RM 63,810.00 (approximately US$ 13,534.84) | ||||
| TMC Global Holding Sdn. Bhd. | Suite 02-06, 2nd Floor Menara See Hoy Chan , No. 374, Jalan Tun Razak, 50400 Kuala Lumpur, Malaysia. | Wellness center focusing on hair transplant, men’s health, joint & knee pain relief, aesthetics center (approx. 3,691 sq. ft.) | 1 December 2021 to 30 November 2024 | RM 11,626.65 (approximately US$ 2,464.19) | ||||
| TMC Global Holding Sdn Bhd | Suite 02-05, 2nd Floor Menara See Hoy Chan, No. 374, Jalan Tun Razak, 50400 Kuala Lumpur | Medical centre (approx. 2,168 sq. ft.) | 1 April 2024 to 31 March 2027 | RM11,444.90 (approximately US$ 2,424.77) | ||||
| TMC Global Holding Sdn. Bhd. | Suites 02-03 & 02-04, 2nd Floor Menara See Hoy Chan, No. 374, Jalan Tun Razak, 50400 Kuala Lumpur | Ambulatory care center for cellular therapy and plastic surgery (approx. 6,116 sq. ft.) | 1 December 2021 to 30 November 2024 | RM 11,626.65 (approximately US$ 2,464.19) |
[Exchange Rate: USD 1.00 = MYR 4.72]
| 218 |
cGMP Facility
Celestialab’s cGMP facility, located at Suites G-03 & G-04, Ground Floor, Menara See Hoy Chan, No. 374, Jalan Tun Razak, 50400 Kuala Lumpur, is equipped with the capacity to manufacture our cell therapy product candidates. Our cleanrooms for sterile manufacturing of cellular products adhere to the cGMP standards of the Pharmaceutical Inspection Co-operation Scheme, ISO cleanroom guidelines, and WHO guidelines on GMP, ensuring compliance and quality in our manufacturing processes.


Figure 25. Celestialab’s cGMP facility.
Intellectual Property
We strive to protect the proprietary technology that we believe is important to our business, including seeking and maintaining patents intended to cover our product candidates and technologies that are important to the development of our business, either directly or in collaboration with third parties. We also rely on trade secrets to protect aspects of our business that are not amenable to, or that we do not consider appropriate for, patent protection, as well as know-how, trademarks, continuing technological innovation and in-licensing opportunities to develop and maintain its proprietary position. We have not sought but may in the future seek appropriate patent protection for our product candidates, as well as other proprietary technologies and their uses by filing patent applications in Malaysia, the U.S. and other select countries.
Patent License
On March 31, 2021, Alps Global Holding Berhad entered into a lease agreement with Celestialab Sdn. Bhd. (“Lease Agreement”), pursuant to which Alps Global Holding Berhad granted Celestialab Sdn. Bhd. a right to use a patented process, specifically the dual channel – liposuction system for the collection and separation of adipose tissue-derived primary stem cells (the “Patent”), for a term of 10 years commencing on March 31, 2021 and expiring on March 31, 2031. Under this Lease Agreement, Celestialab Sdn. Bhd. is required to pay Alps Global Holding Berhad a monthly fee of RM12,500.00 (approximately US$2,612.88). Celestialab Sdn. Bhd. is a wholly-owned subsidiary of Alps Global Holding Berhad and utilizes the Patent in the course of its health and wellness business activities. The Lease Agreement was established and entered to ensure that this Celestialab’s usage of the patent is conducted on arm’s length, to comply with transfer pricing regulations in Malaysia.
On March 3, 2024, Alps entered into a patent license agreement with Dr. THAM Seng Kong, pursuant to which Alps received (i) an exclusive, royalty-free and non-transferable right and license ten (10) patents co-owned by Dr. THAM Seng Kong and the other individuals, as detailed in the table below, and (ii) the right to use, license, sublicense, manufacture, produce, develop, market and/or sell the licensed products and licensed processes. Unless otherwise agreed upon by the parties, the term of the patent license is for the life and duration of the licensed patents from the date of the patent license agreement, including any renewals subsequently granted for each licensed patent.
| Description of the Patent | Patent No./Application No. | Patentee | Expiration Date | Jurisdiction Covered | ||||
A kind of cell-penetrating peptide-acetyl group Argireline nano-emulsion and preparation method thereof |
CN108714111A | Yang Yong Peng Tham Seng Kong Ding KeXiang |
May 5, 2038 | China | ||||
| A hierarchical extraction element that is arranged in external cell culture liquid skin cell growth factor | CN214345637U | Yang Yong Peng Tham Seng Kong Ding KeXiang |
January 28, 2031 | China | ||||
| Instrument for rapidly extracting and separating stem cell exosomes | CN212476724U | Tham Seng Kong Yang Yong Peng Ding KeXiang |
June 1, 2030 | China | ||||
For the fat-derived primary stem cell collection piece-rate system of binary channels of human body liposuction |
CN207452106U | Tham Seng Kong Yang Yong Peng Ding KeXiang |
June 27, 2027 | China | ||||
Synchronous multi-person continuous automatic infusion system capable of keeping constant temperature for stem cell exosomes |
CN219963615U | Tham Seng Kong Ding KeXiang |
May 16, 2033 | China | ||||
Rapid preparation instrument for skin external stem cell membrane dressing loaded with cell growth support |
CN217014449U | Tham Seng Kong Ding KeXiang |
January 19, 2032 | China | ||||
| Device for simply and rapidly preparing nanoemulsion | CN219111470U | Tham Seng Kong Ding KeXiang Ding Yu |
December 28, 2032 | China | ||||
A continuous grading extraction system for natural bioactive peptide |
CN219804616U | Tham Seng Kong Ding KeXiang |
October 10, 2033 | China | ||||
Liquid gel spray preparation and treatment system for stem cell exosomes and secretion factors |
CN218687190U | Tham Seng Kong Ding KeXiang |
June 30, 2032 | China | ||||
Liquid stem cell temperature-sensitive gel spraying operation system with adjustable cell density |
CN217854156U | Tham Seng Kong Ding KeXiang |
April 28, 2032 | China |
On March 3, 2024, Alps entered into a patent license agreement with YANG YongPeng, pursuant to which Alps received (i) an exclusive, royalty-free and non-transferable right and license four (4) patents co-owned by YANG YongPeng and other individuals, as detailed in the table below, and (ii) the right to use, license, sublicense, manufacture, produce, develop, market and/or sell the licensed products and licensed processes. Unless otherwise agreed upon by the parties, the term of the patent license is for the life and duration of the licensed patents from the date of the patent license agreement, including any renewals subsequently granted for each licensed patent.
| Description of the Patent | Patent No./Application No. | Patentee | Expiration Date | Jurisdiction Covered | ||||
| A kind of cell-penetrating peptide-acetyl group Argireline nano-emulsion and preparation method thereof | CN108714111A | Yang Yong Peng Tham Seng Kong Ding KeXiang |
May 5, 2038 | China | ||||
| A hierarchical extraction element that is arranged in external cell culture liquid skin cell growth factor | CN214345637U | Yang Yong Peng Tham Seng Kong Ding KeXiang |
January 28, 2031 | China | ||||
| Instrument for rapidly extracting and separating stem cell exosomes | CN212476724U | Tham Seng Kong Yang Yong Peng Ding KeXiang |
June 1, 2030 | China | ||||
| For the fat-derived primary stem cell collection piece-rate system of binary channels of human body liposuction | CN207452106U | Tham Seng Kong Yang Yong Peng Ding KeXiang |
June 27, 2027 | China |
| 219 |
On March 3, 2024, Alps entered into a patent license agreement with DING KeXiang. In consideration for an aggregate payment of Five Million Chinese Yuan (CNY5,000,000.00) (approximately US$696,262) by Alps, DING KeXiang has granted Alps (i) an exclusive, royalty-free and non-transferable right and license fifteen (15) patents co-owned by DING KeXiang and other individuals, as detailed in the table below, and (ii) the right to use, license, sublicense, manufacture, produce, develop, market and/or sell the licensed products and licensed processes. Unless otherwise agreed upon by the parties, the term of the patent license is for the life and duration of the licensed patents from the date of the patent license agreement, including any renewals subsequently granted for each licensed patent.
| Description of the Patent | Patent No./Application No. | Patentee | Expiration Date | Jurisdiction Covered | ||||
A kind of cell-penetrating peptide-acetyl group Argireline nano-emulsion and preparation method thereof |
CN108714111A | Yang Yong Peng Tham Seng Kong Ding KeXiang |
May 5, 2038 | China | ||||
| A hierarchical extraction element that is arranged in external cell culture liquid skin cell growth factor | CN214345637U | Yang Yong Peng Tham Seng Kong Ding KeXiang |
January 28, 2031 | China | ||||
| Instrument for rapidly extracting and separating stem cell exosomes | CN212476724U | Tham Seng Kong Yang Yong Peng Ding KeXiang |
June 1, 2030 | China | ||||
For the fat-derived primary stem cell collection piece-rate system of binary channels of human body liposuction |
CN207452106U | Tham Seng Kong Yang Yong Peng Ding KeXiang |
June 27, 2027 | China | ||||
Synchronous multi-person continuous automatic infusion system capable of keeping constant temperature for stem cell exosomes |
CN219963615U | Tham Seng Kong Ding KeXiang |
May 16, 2033 | China | ||||
Rapid preparation instrument for skin external stem cell membrane dressing loaded with cell growth support |
CN217014449U | Tham Seng Kong Ding KeXiang |
January 19, 2032 | China | ||||
| Device for simply and rapidly preparing nanoemulsion | CN219111470U | Tham Seng Kong Ding KeXiang Ding Yu |
December 28, 2032 | China | ||||
A continuous grading extraction system for natural bioactive peptide |
CN219804616U | Tham Seng Kong Ding KeXiang |
October 10, 2033 | China | ||||
Liquid gel spray preparation and treatment system for stem cell exosomes and secretion factors |
CN218687190U | Tham Seng Kong Ding KeXiang |
June 30, 2032 | China | ||||
Liquid stem cell temperature-sensitive gel spraying operation system with adjustable cell density |
CN217854156U | Tham Seng Kong Ding KeXiang |
April 28, 2032 | China | ||||
Freezing-dried and non-freezing-dried hydroquinone mono-propionate beautifying effect nanometer emulsion and preparation method thereof |
CN100508952C | Ding KeXiang | October 24, 2027 | China | ||||
Rapid separation, purification and concentration system for preparing high-capacity stem cell exosomes |
CN215517397U | Ding KeXiang | June 25, 2031 | China | ||||
Skin externally-applied liquid state and solid state kreotoxin (liushengtai) composite nano emulsion and preparation thereof |
CN101214200B | Ding KeXiang | January 21, 2028 | China | ||||
Novel method for analyzing human thymidine kinase fluorescence immune based on magnetic nanometer particular |
CN101231288A | Ding KeXiang | January 31, 2028 | China | ||||
Breast cancer early warning chip for easily rapid measuring human I type thymidine kinase gene protein |
CN101275954A | Ding KeXiang | May 9, 2028 | China |
On March 3, 2024, Alps entered into a patent license agreement with DING Yu, pursuant to which Alps received (i) an exclusive, royalty-free and non-transferable right and license one (1) patent co-owned by DING Yu, Dr. THAM Seng Kong and DING Kexiang, as detailed in the table below, and (ii) the right to use, license, sublicense, manufacture, produce, develop, market and/or sell the licensed products and licensed processes. Unless otherwise agreed upon by the parties, the term of the patent license is for the life and duration of the licensed patent from the date of the patent license agreement, including any renewals subsequently granted for the licensed patent.
| Description of the Patent | Patent No./Application No. | Patentee | Expiration Date | Jurisdiction Covered | ||||
| Device for simply and rapidly preparing nanoemulsion | CN-219111470-U | Ding Yu; Tham Seng Kong; and Ding Kexiang |
December 28, 2032 | China |
Alps currently does not own any patents. However, pursuant to the patent license agreements dated March 3, 2024, as mentioned above, Alps has exclusively in-licensed fifteen (15) foreign patents in China. The actual protection afforded by a patent varies on a product-by-product basis and depends upon many factors, including the type of patent, the scope of its coverage, the availability of regulatory-related extensions, the availability of legal remedies in a particular country and the validity and enforceability of the patent. As of the date of this prospectus/proxy statement, Alps has identified five (5) of the fifteen (15) in-licensed patents as material and important to its business (“Material Patents”). These in-licensed patents cover stem cell and/or exosome processing technologies, which would be expected to expire between June 27, 2027 and January 19, 2032, without taking into account any possible patent term adjustment or extension and assuming payment of all appropriate maintenance, renewal, annuity or other governmental fees.
A tabular summary of the Material Patents is set forth below:
| Description of the Patent | Patent No./Application No. | Expiration Date | Jurisdiction Covered | |||
A rapid separation, purification and concentration system to prepare high-capacity stem cell exosomes |
CN215517397U | June 25, 2031 | China | |||
Rapid preparation instrument for skin external stem cell membrane dressing loaded with cell growth support |
CN217014449U | January 19, 2032 | China | |||
Instrument for rapidly extracting and separating stem cell exosomes |
CN212476724U | June 1, 2030 | China | |||
Liquid gel spray preparation and treatment system for stem cell exosomes and secretion factors |
CN218687190U | June 30, 2032 | China | |||
For the fat-derived primary stem cell collection piece-rate system of binary channels of human body liposuction |
CN207452106U | June 27, 2027 | China |
| 220 |
Trade Secrets
Various agreements among contracting member states and legislation in certain countries may provide some level of protection for trade secrets. Remedies available for breaches of such agreements vary, ranging from the destruction of products manufactured using illegally obtained proprietary information to punitive damages, injunctive relief, provisional relief to prevent infringement and preserve evidence, and even criminal sanctions. As our reliance on trade secrets to safeguard our proprietary information, know-how, and technologies is paramount, we are committed to continuously exploring and implementing proactive measures to protect them. This includes entering into confidentiality agreements with our employees and third parties and refraining from conducting business in environments where such protection is insufficient.
However, despite our efforts, breaches of these agreements may occur, and we may lack adequate remedies in such instances. Furthermore, there is a risk that our trade secrets could be acquired or independently discovered by competitors. Disputes may also arise regarding the rights to related or resulting know-how and inventions if our contractors, commercial partners, collaborators, employees, or consultants utilize intellectual property owned by others in their work for us. For more details, please refer to the section titled “Risk Factors—Risks Related to Alps’ Business and Industry and the Combined Company—Risks Related to Our Intellectual Property”.
We are also committed to maintaining the integrity and confidentiality of our data and trade secrets by ensuring continual physical security for our premises, enhancing the physical and electronic security of our information technology systems, and regularly backing up our information.
Trademarks
We have registered trademark protection for “Ethical Life Science” in Malaysia on March 11, 2021 and we have submitted applications to register the mark “MYGENOME” in Malaysia on March 17, 2021.
Domain Names
In addition, we also have registered domain names for websites that we use in our business, such as https://alps-holdings.com; https://alpsmedical.com; https://mygenome.asia; https://celestialab.com; https://celebre.com.my; https://alpscap.com.my; and https://alpsglobemedic.com.
Employees
As of March 31, 2023, Alps had 72 employees, and 88 employees as of March 31, 2024, all of whom are full-time, consisting of clinical, research, operations, regulatory, finance and business development personnel. There are no collective bargaining agreements that pertain to our employees, and we have never experienced a work stoppage. We contribute to social security insurance (SOCSO), the Employee Provident Fund (EPF). and Employment Insurance System (EIS). For the years ended March 31, 2023 and 2024, SOCSO contributions were $11,015.23 and $11,240.96, respectively, EPF contributions were $107,922.59 and $112,971.69, respectively, and EIS contributions were $1,227.16 and $1,551.09, respectively.
Insurance
Our employees presently are covered by SOCSO and EPF. We do not maintain business interruption insurance or key person insurance. Our insurance coverage is consistent with industry standards and is sufficient to cover our key assets, facilities, and liabilities. As part of our business, we also maintain burglary content risks, fire, malpractice liability, burglary money risk, public liability, special contingency, group health plan and group term life insurances.
Legal Proceedings
To the best of our knowledge, we are not subjected to nor engaged in any litigation, arbitration, or claim of material importance, and no litigation, arbitration, or claim of material importance is known to us to be pending or threatened by or against us that would have a material adverse effect on our Group’s results of operations or financial condition.
Regulatory Regime
Laws And Regulations Relating to Our Business and Operations
Local Government Act 1976 (“LGA 1976”)
According to the LGA 1976 and the by-laws of the relevant local authorities in Peninsular Malaysia, we are required to obtain and maintain business and advertisement licenses to carry out our operations and to display our signboard at our offices. A person to whom such license has been granted is generally required to exhibit the license at all times in some prominent place of the licensed premises and to produce the license if required to do so by any officer of the local authority authorized to demand the same. Any person who contravenes any of the provisions of the LGA 1976 and /or the by-laws of the relevant local authorities in Peninsular Malaysia shall be liable for an offence and shall on conviction, be liable to a fine not exceeding RM2,000 or to imprisonment for a term not exceeding one (1) year or to both.
Personal Data Protection Act 2010 (“PDPA 2010”)
The PDPA 2010 regulates the processing of personal data in commercial transactions and provides for connected and incidental matters. The PDPA 2010 applies to (a) any person who processes and (b) any person who has control over or authorizes the processing of any personal data in respect of commercial transactions (“Data User”).
Our business activities customarily involve the processing of personal data belonging to third parties. The processing of personal data by a Data User shall be in compliance with the personal data protection principles as stipulated in the PDPA 2010, namely (a) the General Principle; (b) the Notice and Choice Principle; (c) the Disclosure Principle; (d) the Security Principle; (e) the Retention Principle; (f) the Data Integrity Principle; and (g) the Access Principle (collectively, “the Personal Data Protection Principles”). A Data User who contravenes the Personal Data Protection Principles commits an offence and shall, on conviction, be liable to a fine not exceeding RM300,000.00 or to imprisonment for a term not exceeding two (2) years or to both.
| 221 |
Private Healthcare Facilities and Services Act 1998 (“PHFSA 1998”)
Private medical clinics are regulated by MOH in accordance with the PHFSA 1998 and its relevant regulations, including Private Healthcare Facilities and Services (Private Medical Clinics or Private Dental Clinics) Regulations 2006 (“Private Medical Clinic Regulation”). Under the PHFSA 1998, no one shall establish, maintain, operate or provide a private medical clinic or private dental clinic unless it is registered with the Director General of Health of Malaysia (“DG”). The PHFSA 1998 provides that a person who establishes, maintain, operate or provide any private medical clinic without a certificate of registration or approval from the DG shall commit an offence under the PHFSA 1998. The certificate of registration to operate private medical clinic will only be issued to a registered medical practitioner under the PHFSA 1998.
The holder of a certificate of registration or a person in charge of a private medical clinic shall ensure that all healthcare professional staff are registered under any law regulating their registration and in the absence of such law, hold such qualifications and possess such experience as are recognized by the DG and have such qualification, training, experience and skill to practice in his particular or other healthcare profession as are recognized by the DG.
Medical Act 1971 (“MA 1971”)
A medical practitioner is required to register with the MMC to practice medicine in Malaysia as per the Medical Act of 1971. According to this act, any fully registered individual who intends to practice as a medical practitioner after December 21st of any year must submit an application for a certificate to practice before December 1st of that same year. Upon application, the Registrar of Medical Practitioners will issue an Annual Practicing Certificate (APC) authorizing the applicant to practice as a medical practitioner for the following year.
Furthermore, the Medical Act of 1971 stipulates that only individuals whose names are entered into the register are allowed to practice as specialists in their respective specialties. Practicing medicine without registration with the MMC is considered a violation, and individuals who do so are subject to the disciplinary jurisdiction of the MMC.
Guidelines on Aesthetic Medical Practice for Registered Medical Practitioners (“Guidelines on Aesthetic Medical Practice”)
Guidelines on Aesthetic Medical Practice issued by the MOH provides that a medical practitioner who intends to practice aesthetic medical practice must be fully registered with the MMC and possess with a current and valid APC.
The Guidelines on Aesthetic Medical Practice also provides that all registered medical practitioners who intends to practice aesthetic medical practice to have a minimum of 2 years of clinical experience as a medical officer or equivalent after registration, and subsequently complete 2 years of relevant training in aesthetic medical practice under supervision/attachment and practicing as a general practitioner. Besides, the medical practitioner must be free from any disciplinary actions, exercise strict patient selection criteria, communicate to the potential patient the risks involved, the possible outcome, obtain valid consent for the aesthetic medical procedure planned, generally observe all aspects of the Code of Professional Conduct of the Malaysian Medical Council, place patient safety as the primary concern and should provide aesthetic medical services in a healthcare facility licensed or registered under the PHFSA 1998 and Private Medical Clinic Regulation.
All registered medical practitioners who qualify and wish to practice aesthetic medical practice are required to have an LCP for the aesthetic procedure(s) which he intends to perform and register under the National Registry of Registered Medical Practitioners Practicing Aesthetic Medical Practice, MOH. Pursuant to the Guidelines on Aesthetic Medical Practice, general practitioners who wish to embark on aesthetic medical practice as a new area of practice are required to undergo training.
| 222 |
Medicine (Advertisement and Sale) Act 1956 (“MA 1956”)
The MA 1956 and its relevant regulations and guidelines which include Medicine Advertisements Board Regulations 1976 that regulates the advertisement and dissemination of information in relation to healthcare matters to general public.
The information provided in the advertisements must be factually accurate and capable of being substantiated. It must not be exaggerated, false, misleading or deceptive. The authenticity and the accuracy of the information imparted in the advertisements should be verifiable by the Medicine Advertisements Board (“MAB”). The MA 1956 also provides that no person shall take part in the publication of any advertisement which refers to any skill or service relating to the treatment, prevention or diagnosis of any ailment, disease, injury, infirmity or condition affecting the human body and which is capable of inducing, or which contains an invitation, whether express or implied, to, any person to seek advice of the advertiser or any person referred to in the advertisement in connection with such skill or service provided that the requirements shall not apply to any advertisement published with the approval of MAB, by any professional body related to the medical profession or to any other allied profession which is established by or registered under any written law or with the approval of the MAB established by the MOH, by any private hospitals or by any private clinic, private radiological clinic, and private medical laboratory operated by a registered medical practitioner having a valid APC under the MA 1971.
Guidelines for Stem Cell Research and Therapy 2009 (“Guidelines for Stem Cell”)
The Guidelines for Stem Cell provide that all stem cell research and applications must be reviewed by the relevant institutional review board (IRB) or institutional ethics committee (IEC) for approval. Research on human adult stem cells, stem cells derived from fetal tissues from legally performed termination of pregnancies, non-human stem cells and human embryonic stem (HES) cell lines derived from surplus embryos are allowed. This soft law prohibits among others, the following matters:
| (i) | creation of human embryos by means such as assisted reproductive technology or somatic cell nuclear transfer for scientific research purposes; | |
| (ii) | research involving in vitro culture of any intact human embryo for longer than 14 days or until formation of the primitive streak begins; | |
| (iii) | research in which HES cells are introduced into non-human primate blastocysts or in which any embryonic stem cells are introduced into human blastocysts; | |
| (iv) | no animal into which HES cells have been introduced at any stage of development should be allowed to breed; and | |
| (v) | fusion of human stem cell or other cells of pluripotent nature with cells of non-human origin, longer than 14 days, or until the formation of the primitive streak begins, whichever occurs first. |
Sale of Drugs Act 1952 (“SDA 1952”)
In Malaysia, clinical trials are regulated by the NPRA, which is an agency under the MOH. The NPRA oversees the approval and conduct of clinical trials to ensure the safety, efficacy, and quality of investigational products, as well as the protection of participants’ rights and welfare. The IRB/IEC of the respective institutions also assist in the regulation of clinical trials in Malaysia, as their approval is mandatory before a trial can commence.
The primary legislation governing the regulation of clinical trials in Malaysia is the SDA 1952. Section 26 of the SDA 1952 empowers the MOH to impose regulations with respect to drugs, including the regulation of clinical trials as provided for under the Control of Drugs and Cosmetics Regulations 1984 (“Drugs and Cosmetics Regulations”).
Generally, clinical trials in Malaysia can be categorized in 4 phases:
| ● | For phase 1 trials, the aim is to determine the safety and tolerance of the investigational product. Examples of Phase 1 trials include first-in-human (FIH) studies, pharmacokinetics and pharmacodynamics studies, and drug-drug interaction studies. The number of subjects involved are usually small numbers of healthy volunteers or patients (10-100 subjects). | |
| ● | For phase 2 trials, the aim is to determine the efficacy and safety of the investigational product. Phase 2 trials explore the therapeutic dose range of the investigational product to treat a disease or condition and gathering more information on the investigational products’ safety in a larger group of patients (100-300 subjects). | |
| ● | For phase 3 trials, the aim is to determine the safety, efficacy or effectiveness of the investigational product. Phase 3 trials are confirmatory trials conducted to determine the therapeutic effect in patient populations for which the investigational product is intended. They also provide a definitive assessment of risk-benefit balance (to support drug registration or change in clinical practice). The number of subjects involved usually includes a large group of patients, ranging from several hundred to several thousand (300-3000 subjects). | |
| ● | For phase 4 trials, the aim is to conduct a post-marketing surveillance to monitor safety in real-world populations, including the long-term benefits and risks of the product. These trials take place after the product is approved and is on the market, and usually involve a large group of patients (1000 subjects). |
| 223 |
Drugs and Cosmetics Regulations 1984
Except as otherwise provided in the Drugs and Cosmetics Regulations, no person shall manufacture, sell, supply, import, possess or administer any medicinal product unless the product is registered or notified (for cosmetics) and the person holds a CTIL. Any person who wishes to manufacture any product solely for the purpose of producing samples for clinical trials, for registration or issuance of notification note under the Drugs and Cosmetics Regulations may on application be exempted from Regulation 7(1) (CTX). Only a person responsible for the conduct of the clinical trial at a trial site or an authorized person from a locally registered pharmaceutical company, sponsor or contract research organization with a permanent address in Malaysia can apply for a CTIL or CTX. The CTIL or CTX holder need not necessarily conduct the clinical trial him or herself. The NPRA shall not issue the approved CTIL or CTX application if the investigator fails to declare that he or she has attended any of the courses or training sessions certified by the National Committee for Clinical Research.
Guideline for Application of Clinical Trial Import License and Clinical Trial Exemption Eighth Edition
This guidance document provides a step by step procedure on submitting the documents for the CTIL/CTX application including a requirement to contact officers from the Investigational Product Evaluation and Safety Section (IPESS), Centre of Product and Cosmetic Evaluation (CPCE) to schedule for an appointment to submit the CTIL application in person. A flow chart of the CTIL/CTX application process and CTIL/CTX application process involving the First-in-Human Clinical Trial is included in the document for the applicants.
The Drug Control Authority (“DCA”) should be informed by the CTIL/CTX holder in case there are any changes in information or any information received by him/her that may cast doubt on the validity of the data which was submitted for the CTIL. This guidance document also entails information on how the applicants should proceed and engage with the DCA when the trial is completed and communicate with the authorities in the event of early termination of trials.
Guideline for Good Clinical Practice Forth Edition
The Malaysian Guideline for Good Clinical Practice mandates that all clinical trials in Malaysia should be conducted in accordance with the ethical principles of the Declaration of Helsinki. An IRB or IEC that reviews proposed clinical trials must consist of not fewer than five members with at least one member who specialises in a non-scientific field and one member who is independent of the trial site. Prior to commencing the trial, the investigator should have the IRB or IEC’s written approval or favourable opinion of the written informed consent form and any other written information to be provided to the subjects. A qualified physician (or dentist, when appropriate), who is an investigator or a sub-investigator for the trial, is responsible for all trial-related medical or dental decisions.
All serious adverse events (SAEs) detected or being notified should be reported immediately to the sponsor except for those SAEs that the protocol or other document identifies as not needing immediate reporting. If the trial is prematurely terminated or suspended, the investigator should promptly inform the subjects and provide them with the appropriate follow-up therapy and inform the regulatory authorities and the IRB or IEC. Upon completion of the trial, the investigator should inform the institution and where applicable provide the IRB or IEC with a summary of the trial’s outcome. If required by the applicable regulatory requirements, the sponsor should provide insurance or should indemnify the investigator against claims arising from the trial except for claims that arise from malpractice or negligence.
| 224 |
Laws and Regulations relating to Intellectual Property Rights
Intellectual property system in Malaysia is administered by the Intellectual Property Corporation of Malaysia (MyIPO), an agency under the Ministry of Domestic Trade and Consumer Affairs.
Trademarks Act 2019 (Act 815)
Trademark in Malaysia is governed by Trademarks Act 2019 (Act 815) (“TMA 2019”). Malaysia is also a member of various trademark-related treaties, including:
| (i) | Protocol relating to the Madrid Agreement concerning the International Registration of Marks since December 27, 2019; | |
| (ii) | Nice Agreement concerning the International Classification of Goods and Services since September 28, 2007; | |
| (iii) | Paris Convention for the Protection of Industrial Property since January 1, 1989; and | |
| (iv) | Agreement on Trade-related Aspects of Intellectual Property Rights (TRIPS) since January 1, 1995. |
The TMA 2019 provides that any person who claims to be the bona fide proprietor of a trademark may apply for the registration of the trademark if:
| (i) | the person is using or intends to use the trademark in the course of trade; or | |
| (ii) | the person has authorized or intends to authorize another person to use the trademark in the course of trade. |
The TMA 2019 has also expanded the types of trademarks recognized for registration to be more than just word, logo, numbers, name. signature, letter and to include shape of goods or their packaging, color, sound, scent, hologram, positioning marks and sequence of motion of any combination thereof; provided that they must be signs capable of being represented graphically.
In general, Malaysia provides for protection for both registered and unregistered trademarks. Unregistered trademarks are protected under common law rights, particularly in the tort of passing off. In fact, even during the examination of trademark, the Registrar shall refuse, under relative grounds of Section 24(4) of the TMA 2019, to register it if the mark’s use in Malaysia is prevented by virtue of any rule of law protecting an unregistered trademark or other sign used in the course of trade including the law of passing of.
The scope of trademark infringement and its exemptions has been substantially expanded by the TMA 2019. There could now be infringement even in the use of a similar mark on similar goods or services (as opposed to being identical). Secondary users who know or have reason to believe that such use is without authorization of the trademark proprietor may also become liable under TMA 2019.
The TMA 2019 and relevant regulation are applicable to our trademark, brand, word, logo, numbers, name. signature, letter and to include shape of goods or their packaging, color, sound, scent, hologram, positioning marks and/or sequence of motion of any combination.
Patents Act 1983 (“PA 1983”) and Patents Regulations 1986 (“PR 1986”)
The PA 1983 and PR 1986 are the laws and regulations which govern patent protection in Malaysia. PA 1983 would cover amongst others, the criteria for patentability, rights attached to patents, duration of patents and acts relating to infringement, whereas the PR 1986 predominantly encompass the procedures for the application of patents.
An invention is patentable if it is new, involves an inventive step, is industrially applicable and is not explicitly excluded by the PA 1983. An invention which may relate to a product or process means an idea of an inventor which permits in practice the solution to a specific problem in the field of technology. Registration of a patent grants the patent owner rights to exploit the patented invention, to assign or transmit the patent and to conclude license contacts.
A patent shall be deemed granted and shall take effect on the date the certificate of grant of patent is issued. The duration of a patent will generally be for a period of 20 years from the filing date of the application.
| 225 |
Copyright Act 1987 (“CA 1987”)
Copyright protection in Malaysia is governed by the CA 1987 which provides comprehensive protection for copyrightable works. The CA 1987 outlines the nature of works eligible for copyright (which includes computer software), the scope of protection, and the manner in which the protection is accorded. A unique feature of the CA 1987 is the inclusion of provisions for enforcing the Act, which include such powers to enter premises suspected of having infringing copies and to search and seize infringing copies and contrivances. Malaysia is a signatory of the Berne Convention. Foreign works of non-Berne member countries are also protected if they are made in Malaysia and are published in Malaysia within thirty days of their first publication in the country of origin.
Unlike trademarks, designs and patents (other intellectual property rights), there is no specific system of registration for copyright in Malaysia. Although copyright is a non-registrable right in Malaysia and enjoys automatic protection, ownership of copyright is difficult to establish. As such, proper documentation can be prepared to prove ownership. Copyright owners can claim ownership by way of a Statutory Declaration or by filing a Voluntary Notification at the MyIPO.
The definition of a literary work now includes table or compilations “whether or not expressed in words, figures or symbols and whether or not in a visible form”. The owner of copyright in a work including a derivative work, will have the exclusive right to control “the transmission of a work through wire or wireless means to the public, including the making available of a work to the public in such a way that members of the public may access the work from a place and at a time individually chosen by them”.
It is also an infringement of copyright to circumvent any effective technological measures aimed at restricting access to works, removal or alteration of any electronic rights management information without authority, or distribution, importation for distribution or communication to the public, without authority, works or copies of works in respect of which electronic rights management information has been removed or altered without authority.
The CA 1987 and the relevant regulations are benefited us which we are eligible to claim ownership by compiling proper documentation to prove ownership via a Statutory Declaration or by filing a Voluntary Notification at the MyIPO.
Laws and Regulations relating to Employment and Social Security
Employment Act 1955 (“EA 1955”)
The EA 1955 is the primary legislation on labor matters in Malaysia. The EA 1955 provides for minimum work requirements and benefits of employment, such as maximum working hours, overtime entitlement, leave entitlement, maternity protection and termination benefits.
Employees’ Provident Fund Act 1991 (“EPF 1991”)
The EPF 1991 imposes the statutory obligations on employers and employees to make contribution towards the Employees Provident Fund, which is essentially a fund established as a scheme of savings for employees’ retirement and the management of savings for the retirement purposes. Under the EPF 1991, any employer who fails to pay the necessary contributions shall be liable to imprisonment for a term not exceeding three years or to a fine not exceeding ten thousand ringgit or to both.
| 226 |
Employee’ Social Security Act 1969 (“ESSA 1969”)
The ESSA 1969 was implemented to provide protection for employees and their families against economic and social distress in situations where the employees sustain injury or death. The schemes of social security under the ESSA 1969 are administered by Social Security Organization (“SOCSO”) and are financed by compulsory contributions made by the employers and the employees. Under the ESSA 1969, any person who fails to make contribution shall be all be punishable with imprisonment for a term which may extend to two years, or with fine not exceeding ten thousand ringgit, or with both.
Employment Insurance System Act 2017 (“EIS 2017”)
EIS 2017 was introduced and enforced with the aim to provide protection and assist workers who have lost employment through two (2) main components namely, the Employment Insurance and Active Labor Market Policies. The Employment Insurance System (EIS) provides protection to workers who have lost their employment through income replacement, reskilling and upskilling training to enhance their employability as well as employment services so that they can secure other suitable jobs fast.
Our all employees which under definition of the EA 1955 are subject to the provision of EPF 1991, ESSA 1969, and EIS 2017.
Laws and Regulations relating to Consumer Protection
Consumer Protection Act 1999 (“CPA 1999”)
The principal law for consumer protection in Malaysia is the CPA 1999. The CPA 1999 establishes various consumer protection mechanisms in Malaysia, and bridge gaps that may occur in other major laws, which may be inadequate in protecting consumers. The government agency which is primarily responsible for policy-making and law enforcement on consumer protection in Malaysia and receiving consumer complaints is the Ministry of Domestic Trade and Consumer Affairs.
The CPA 1999 covers almost every aspects of consumer protection; ranging from misleading and deceptive conducts, false representation and unfair practices; safety of goods and services; unfair contract terms; guarantees in respect of the supply of goods and services; and product liability; to the establishment, structure and functions of the National Consumer Advisory Council; the Committee on Advertisement; the Tribunals for Consumer Claims; and other matters related to enforcement, offences, remedies, and compensation.
| 227 |
MANAGEMENT’S
DISCUSSION AND ANALYSIS OF
FINANCIAL CONDITION AND RESULTS OF OPERATIONS OF GLOBALINK
References in this section to “we,” “us,” or the “Globalink,” refer to Globalink Investment Inc., and when discussing consolidated financial information, also includes Globalink Investment Inc.’s wholly-owned subsidiary, Globalink Merger Sub, Inc., a Nevada corporation. References to “management,” or “management team” refer to Globalink’s officers and directors, and references to the “Sponsor” refer to GL Sponsor LLC. Certain information contained in the discussion and analysis set forth below includes forward-looking statements that involve risks and uncertainties.
Special Note Regarding Forward-Looking Statements
This Report includes “forward-looking statements” within the meaning of Section 27A of the Securities Act and Section 21E of the Exchange Act that are not historical facts and involve risks and uncertainties that could cause actual results to differ materially from those expected and projected. All statements, other than statements of historical fact included in this registration statement including, without limitation, statements in this “Management’s Discussion and Analysis of Financial Condition and Results of Operations” regarding the Company’s financial position, business strategy and the plans and objectives of management for future operations, are forward-looking statements. Words such as “expect,” “believe,” “anticipate,” “intend,” “estimate,” “seek” and variations and similar words and expressions are intended to identify such forward-looking statements. Such forward-looking statements relate to future events or future performance, but reflect management’s current beliefs, based on information currently available. A number of factors could cause actual events, performance or results to differ materially from the events, performance and results discussed in the forward-looking statements. The Company’s securities filings can be accessed on the EDGAR section of the SEC’s website at www.sec.gov. Except as expressly required by applicable securities law, the Company disclaims any intention or obligation to update or revise any forward-looking statements whether as a result of new information, future events or otherwise.
Overview
Globalink was formed on March 24, 2021 for the purpose of entering into a merger, share exchange, asset acquisition, stock purchase, recapitalization, reorganization or other similar business combination with one or more target businesses. Globalink’s efforts to identify a prospective target business will not be limited to any particular industry or geographic region, although we intend to focus our search on target businesses in in North America, Europe, South East Asia, and Asia (excluding mainland China and the Hong Kong and Macau special administrative regions), in the medical technology and green energy industry. Globalink shall not undertake its initial business combination with any entity with its principal business operations in China (including Hong Kong and Macau). We intend to complete the business acquisition in a combination of cash (whether cash from the trust account or cash from a debt or equity financing transaction that closes concurrently with the business combination) or Globalink’s equity securities.
The issuance of additional shares of common stock in connection with an initial business combination:
● may significantly dilute the equity interest of Globalink’s investors who would not have pre-emption rights in respect of any such issuance;
● may subordinate the rights of holders of shares of common stock if Globalink issue shares of preferred stock with rights senior to those afforded to our shares of common stock;
● could cause a change in control if a substantial number of shares of our common stock is issued, which may affect, among other things, Globalink’s ability to use its net operating loss carry forwards, if any, and could result in the resignation or removal of its present officers and directors;
● may have the effect of delaying or preventing a change of control of Globalink by diluting the stock ownership or voting rights of a person seeking to obtain control of Globalink; and
| 228 |
● may adversely affect prevailing market prices for Globalink’s common stock, rights and/or warrants. Similarly, if we issue debt securities or otherwise incur significant debt, it could result in:
● default and foreclosure on Globalink’s assets if operating revenues after an initial Business Combination are insufficient to repay its debt obligations;
● acceleration of Globalink’s obligations to repay the indebtedness even if it make all principal and interest payments when due if they breach certain covenants that require the maintenance of certain financial ratios or reserves without a waiver or renegotiation of that covenant;
● Globalink’s immediate payment of all principal and accrued interest, if any, if the debt security is payable on demand;
● Globalink’s inability to obtain necessary additional financing if the debt security contains covenants restricting our ability to obtain such financing while the debt security is outstanding;
● using a substantial portion of Globalink’s cash flow to pay principal and interest on its debt, which will reduce the funds available for dividends on our common stock if declared, its ability to pay expenses, make capital expenditures and acquisitions, and fund other general corporate purposes;
● limitations on Globalink’s flexibility in planning for and reacting to changes in its business and in the industry in which we operate;
● increased vulnerability to adverse changes in general economic, industry and competitive conditions and adverse changes in government regulation;
● limitations on Globalink’s ability to borrow additional amounts for expenses, capital expenditures, acquisitions, debt service requirements, and execution of its strategy; and
● other purposes and other disadvantages compared to Globalink’s competitors who have less debt.
Globalink expects to continue to incur significant costs in the pursuit of its acquisition plans. Globalink cannot assure you that its plans to complete a Business Combination will be successful.
Recent Developments
As of the date of this proxy statement/prospectus, Globalink has extended the date by which it must (i) consummate a merger, capital stock exchange, asset acquisition, stock purchase, reorganization or similar business combination involving Globalink and one or more businesses, (ii) cease its operations if it fails to complete such business combination, and (iii) redeem or repurchase 100% of the Globalink’s outstanding public shares of common stock included as part of the units sold in the Company’s IPO that closed on December 9, 2021 (the “Termination Date”) eighteen times since the IPO, and currently has until January 9, 2025 to complete its initial business combination. The Company may continue to extend the Termination Date under its amended and restated certificate of incorporation, as amended, to up to June 9, 2025 through monthly extensions by resolutions of its Board, if requested by the Sponsor, and subject to the Sponsor depositing additional funds into the Trust Account.
On January 29, 2024, Globalink submitted an application with Nasdaq for the transfer of its securities listed on the Nasdaq Global Market to Nasdaq Capital Market.
On March 6, 2024, Globalink received Nasdaq’s approval of its transfer application and on the same date, it was notified by Nasdaq that Globalink regained compliance with Nasdaq’s requirement of 300 public holders for continued listing on the Nasdaq Capital Market.
On December 10, 2024, Globalink received Nasdaq’s delisting determination letter from Nasdaq as the 36-month anniversary from its IPO has passed on December 9, 2024, and Globalink are no longer in compliance with Nasdaq Listing Rule IM 5101-2(b). Globalink’s securities were suspended from trading and delisted from Nasdaq on December 17, 2024.
On December 17, 2024, Globalink’s securities commenced trading on OTC Pink.
| 229 |
Results of Operations
As of September 30, 2024, Globalink had not commenced any operations. All activity through September 30, 2024 relates to Globalink’s formation and the initial public offering (the “IPO”) and search for a prospective initial business combination target. Globalink will not generate any operating revenues until after the completion of an initial business combination, at the earliest. Globalink generates non-operating income in the form of interest income from the proceeds derived from the IPO placed in the trust account established for the benefit of the Globalink’s public stockholders (the “Trust Account”).
For the three months ended September 30, 2024, we had a net loss of $116,747, all of which consisted of operating expenses incurred driven by general and administrative expenses of $287,590, franchise tax expense of $50,000, provision for income tax of $91,087, and $51,814 in interest expense, partially offset by change in fair value of warrants liabilities of $14,250 and interest income on cash held in the Trust Account of $349,494.
For the nine months ended September 30, 2024, we had a net loss of $709,239, all of which consisted of operating expenses incurred driven by general and administrative expenses of $1,310,672, franchise tax expense of $93,662, provision for income tax of $198,175, $128,811 in interest expense, penalties on franchise tax of $2,356 and change in fair value of warrants liabilities of $12,369, partially offset by interest income on cash held in the Trust Account of $1,036,806.
For the three months ended September 30, 2023, we had a net income of $254,297, all of which consisted of interest income on cash and investments held in the Trust Account of $621,793 and change in fair value of warrants liabilities of $1,824, partially offset by operating expenses incurred driven by general and administrative expenses of $259,829, provision for income tax of $27,338, accrual of Delaware franchise taxes of $50,000, $20,265 in interest expense and penalties on income tax of $11,888.
For the nine months ended September 30, 2023, we had a net income of $1,224,868, all of which consisted of interest income on cash and investments held in the Trust Account of $2,550,909, partially offset by operating expenses incurred driven by general and administrative expenses of $715,665, provision for income tax of $426,710, accrual of Delaware franchise taxes of $133,956, $33,376 in interest expense, penalties on income tax of $11,888 and change in fair value of warrants liabilities of $4,446.
For the three months ended March 31, 2024, Globalink had a net loss of $375,307, all of which consisted of operating expenses incurred driven by general and administrative expenses of $603,471, provision for income tax of $73,291, $33,395 in interest expense, penalties on franchise tax of $2,356 and change in fair value of warrants liabilities of $11,799, partially offset by reversal of Delaware franchise taxes of $6,338 as a result of a prior year true up and interest income on cash held in the Trust Account of $342,667.
For the three months ended March 31, 2023, Globalink had a net income of $682,939 all of which consisted of interest income on investments held in the Trust Account of $1,257,477 and change in fair value of warrants liabilities of $570, partially offset by operating expenses incurred driven by general and administrative expenses of $271,537, accrual of Delaware franchise taxes of $50,000, and provision for income tax of $253,571.
For the year ended December 31, 2023, Globalink had a net income of $1,320,324, all of which consisted of interest income on cash and investments held in the Globalink’s trust account of $3,090,407 and change in fair value of warrants liabilities of $4,389, partially offset by operating expenses incurred driven by general and administrative expenses of $991,868, provision for income tax of $529,505, accrual of Delaware franchise taxes of $183,956, $57,255 in interest expense, penalties on income tax of $11,888.
For the year ended December 31, 2022, Globalink had a net income of $224,242, all of which consisted of interest income on investments held in trust account of $1,683,870 and change in fair value of the warrant liabilities of $108,300, partially offset by operating expenses incurred driven by general and administrative expenses of $1,107,632, provision for income tax of $308,185 and accrual of Delaware franchise taxes of $152,111.
Liquidity, Capital Resources and Going Concern
The registration statement on Form S-1 for the Globalink’s IPO was declared effective on December 6, 2021. On December 9, 2021, Globalink consummated its IPO of 10,000,000 units. Each unit consists of one share of common stock, $0.001 par value, one right to receive one-tenth (1/10) of a share of common stock upon the consummation of an initial business combination and one redeemable warrant entitling the holder thereof to purchase one-half (1/2) of a share of common stock at a price of $11.50 per whole share. The units were sold at an offering price of $10.00 per unit, generating gross proceeds of $100,000,000. Simultaneously with the closing of the IPO, Globalink consummated the private placement of 517,500 private units at a price of $10.00 per unit, generating total proceeds of $5,175,000.
On December 9, 2021, the underwriters exercised the over-allotment option to purchase an additional 1,500,000 units in full (the “Over-Allotment Units”), and the closing of the Over-Allotment Units occurred on December 13, 2021. The total aggregate issuance by Globalink of 1,500,000 units at a price of $10.00 per unit resulted in total gross proceeds of $15,000,000. On December 13, 2021, simultaneously with the sale of the Over-Allotment Units, Globalink consummated the private sale of an additional 52,500 private units, generating gross proceeds of $525,000. Since the underwriter’s over-allotment was exercised in full, the Sponsor did not forfeit any insider shares.
Offering costs for the IPO and the exercise of the underwriters’ Over-allotment Option amounted to $6,887,896, consisting of $2,300,000 of underwriting fees, $4,025,000 of deferred underwriting fees payable (which are held in the Trust Account) and $562,896 of other costs. The $4,025,000 of deferred underwriting fee payable is contingent upon the consummation of an initial business combination by January 9, 2025 (and may be extended to June 9, 2025 on a monthly basis by resolutions of the Globalink Board, if requested by the Sponsor, and subject to the Sponsor depositing additional funds into the Trust Account), subject to the terms of the underwriting agreement.
| 230 |
Following the closing of the IPO (including the Over-Allotment Units), $116,725,000 ($10.15 per unit) from the net proceeds of the sale of the units in the IPO (including the Over-Allotment Units), and the private units was placed in the Trust Account and was initially invested in U.S. government securities, within the meaning set forth in Section 2(a)(16) of the Investment Company Act of 1940, as amended (the “Investment Company Act”), with a maturity of 180 days or less or in any open-ended investment company that holds itself out as a money market fund selected by the Company meeting the conditions of paragraphs (d)(2), (d)(3) and (d)(4) of Rule 2a-7 of the Investment Company Act.
To mitigate the risk of being deemed to have been operating as an unregistered investment company (including under the subjective test of Section 3(a)(1)(A) of the Investment Company Act), on July 27, 2023, Globalink instructed Continental Stock Transfer & Trust Company, as trustee of its Trust Account, to liquidate the U.S. government securities or money market funds held in the Trust Account and thereafter to hold all funds in the Trust Account in cash (which may include demand deposit accounts) until the earlier of consummation of the initial business combination or liquidation. Furthermore, such cash is held in bank accounts, which exceed federally insured limits as guaranteed by the Federal Deposit Insurance Corporation (the “FDIC”).
For the three months ended March 31, 2024, cash used in operating activities was $885,807. Net loss of $375,307 was affected by interest earned on cash held in the Trust Account of $342,667 and change in fair value of warrants liabilities of $11,799. Changes in operating assets and liabilities used $179,632 of cash for operating activities.
For the three months ended March 31, 2023, cash used in operating activities was $79,619. Net income of $682,939 was affected by interest earned on investments held in the Trust Account of $1,257,477 and change in fair value of warrants liabilities of $570. Changes in operating assets and liabilities used $495,489 of cash for operating activities.
For the nine months ended September 30, 2024, cash used in operating activities was $1,757,620. Net loss of $709,239 was affected by interest earned on cash held in the Trust Account of $1,036,806 and change in fair value of warrants liabilities of $12,369. Changes in operating assets and liabilities used $23,944 of cash for operating activities.
For the nine months ended September 30, 2023, cash used in operating activities was $1,174,478. Net income of $1,224,868 was affected by interest earned on cash and investments held in the Trust Account of $2,550,909 and change in fair value of warrants liabilities of $4,446. Changes in operating assets and liabilities reduced $147,117 of cash from operating activities.
For the year ended December 31, 2023, cash used in operating activities was $1,402,478. Net income of $1,320,324 was affected by interest earned on cash and investments held in the trust account of $3,090,407 and change in fair value of warrants liabilities of $4,389. Changes in operating assets and liabilities provided by $371,994 of cash from operating activities.
For the year ended December 31, 2022, cash used in operating activities was $730,469. Net income of $224,242 was impacted by interest earned on investments held in the trust account of $1,683,870, change in fair value of warrant liabilities of $108,300. Changes in operating assets and liabilities provided $837,459 of cash from operating activities.
Globalink had cash held in the Trust Account of $29,811,256, $29,047,273 and $28,668,218 as of September 30, 2024, March 31, 2024 and December 31, 2023, respectively. Interest income on the balance in the Trust Account of $1,036,806 for the nine months ended September 30, 2024 may be used by us to pay taxes. For the nine months ended September 30, 2024, $433,768 was withdrawn from the Trust Account to pay for taxes.
Globalink intend to use substantially all of the funds held in the Trust Account, including any amounts representing interest earned on the Trust Account (less income taxes payable), to complete a business combination. To the extent that our capital stock or debt is used, in whole or in part, as consideration to complete a business combination, the remaining proceeds held in the Trust Account will be used as working capital to finance the operations of the target business or businesses or make other acquisitions.
Globalink had $65,221, $6,878 and $79,073 of cash held outside of the Trust Account as of September 30, 2024, March 31, 2024 and December 31, 2023, respectively. Globalink intend to use the funds held outside the Trust Account primarily to identify and evaluate target businesses, perform business due diligence on prospective target businesses, travel to and from the offices, plants or similar locations of prospective target businesses or their representatives or owners, review corporate documents and material agreements of prospective target businesses, and structure, negotiate and complete initial business combination.
| 231 |
In order to finance transaction costs in connection with an initial business combination, the Sponsor or an affiliate of the Sponsor, or certain of Globalink’s officers and directors may, but are not obligated to, loan funds as may be required. If Globalink completes an initial business combination, Globalink will repay the working capital loans out of the proceeds of the Trust Account released to Globalink. Otherwise, the working capital loans would be repaid only out of funds held outside the Trust Account. In the event that an initial business combination does not close, Globalink may use a portion of proceeds held outside the Trust Account to repay the working capital loans, but no proceeds held in the Trust Account would be used to repay the working capital loans. Except for the foregoing, the terms of such working capital loans, if any, have not been determined and no written agreements exist with respect to such loans. The working capital loans would either be repaid upon consummation of an initial business combination, without interest, or, at the lender’s discretion, up to $1,500,000 of such working capital loans may be convertible into units of the post initial business combination entity at a price of $10.00 per unit. The units would be identical to the private units. As of September 30, 2024, there were no working capital loans outstanding.
On March 3, 2023, Globalink entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount of $390,000 for the purpose of extension fees payment. The promissory note bears an interest of 6% per annum and repayable upon consummation of an initial business combination. As of September 30, 2024, the full $390,000 had been borrowed and no amount was available under this note for borrowing.
On March 23, 2023, Globalink entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount of $250,000 for the purpose of extension fees payment. The promissory note bears an interest of 6% per annum and repayable upon consummation of an initial business combination. As of September 30, 2024, the full $250,000 had been borrowed and no amount was available under this note for borrowing.
On June 2, 2023, Globalink entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount of up to $700,000 for the purpose of extension fees payment. The promissory note bears an interest of 6% per annum and repayable upon consummation of an initial business combination. As of September 30, 2024, the full $700,000 had been borrowed and no amount was available under this note for borrowing.
On October 13, 2023, Globalink entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount of $250,000 for the purpose of working capital. The promissory note bears an interest of 6% per annum and repayable upon consummation of an initial business combination. As of September 30, 2024, the full $250,000 had been borrowed and no amount was available under this note for borrowing.
On December 8, 2023, Globalink entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount of $110,000 for the purpose of working capital. The promissory note bears an interest of 6% per annum and repayable upon consummation of an initial business combination. As of September 30, 2024, the full $110,000 had been borrowed and no amount was available under this note for borrowing.
On each of September 5, 2023, September 29, 2023 and November 7, 2023, an affiliate of the Sponsor advanced $130,000 to the Company, for a total advance of $390,000. As of September 30, 2024, $390,000 of advance is reflected in “Due to Affiliate” on the consolidated balance sheet.
On January 5, 2024, Globalink entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount of $250,000 for the purpose of working capital. The promissory note bears an interest of 6% per annum and repayable upon consummation of an initial business combination.
On January 25, 2024, Globalink entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount of $300,000 for the purpose of working capital. The promissory note bears an interest of 6% per annum and repayable upon consummation of an initial Business Combination. As of September 30, 2024, the full $300,000 had been borrowed and no amount was available under this note for borrowing.
On February 22, 2024, Globalink entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount of $300,000 for the purpose of working capital. The promissory note bears an interest of 6% per annum and repayable upon consummation of an initial Business Combination. As of September 30, 2024, the full $300,000 had been borrowed and no amount was available under this note for borrowing.
On April 4, 2024, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount of $300,000 for the purpose of working capital. The promissory note bears an interest of 6% per annum and repayable upon consummation of an initial Business Combination. As of September 30, 2024, the full $300,000 had been borrowed and no amount was available under this note for borrowing.
On June 5, 2024, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount of $400,000 for the purpose of working capital. The promissory note bears an interest of 6% per annum and repayable upon consummation of an initial Business Combination. As of September 30, 2024, the full $400,000 had been borrowed and no amount was available under this note for borrowing.
On August 14, 2024, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount of $300,000 for the purpose of working capital. The promissory note bears an interest of 6% per annum and repayable upon consummation of an initial Business Combination.
| 232 |
If the initial business combination is not consummated, Globalink will need to raise additional capital through loans or additional investments from its Sponsor, stockholders, officers, directors, or third parties. Globalink’s officers, directors and Sponsor may, but are not obligated to, loan funds, from time to time or at any time, in whatever amount they deem reasonable in their sole discretion, to meet the Globalink’s working capital needs. Accordingly, Globalink may not be able to obtain additional financing. If Globalink is unable to raise additional capital, it may be required to take additional measures to conserve liquidity, which could include, but not necessarily be limited to, curtailing operations, suspending the pursuit of a potential transaction, and reducing overhead expenses. Globalink cannot provide any assurance that new financing will be available to it on commercially acceptable terms, if at all.
In connection with Globalink’s assessment of going concern considerations in accordance with FASB ASU 2014-15, “Disclosures of Uncertainties about an Entity’s Ability to Continue as a Going Concern,” Globalink currently has until January 9, 2025 to consummate a business combination, or may face liquidation. If Globalink anticipates that it may not be able to consummate its initial business combination in time, Globalink may, by resolutions of its board of directors, if requested by the Sponsor, extend the period of time to consummate a business combination on a monthly basis to until June 9, 2025, subject to the Sponsor depositing additional funds into the Trust Account. It is uncertain that Globalink will be able to consummate a business combination by this time. If a business combination is not consummated by this date and an extension is not requested by the Sponsor, there will be a mandatory liquidation and subsequent dissolution of Globalink. Management has determined that the mandatory liquidation, should a business combination not occur, and an extension is not requested by the Sponsor, and potential subsequent dissolution as well as liquidity condition noted above raises substantial doubt about Globalink’s ability to continue as a going concern. No adjustments have been made to the carrying amounts of assets or liabilities should Globalink be required to liquidate after January 9, 2025 (and may be extended to June 9, 2025 on a monthly basis by resolutions of the Globalink Board, if requested by the Sponsor, and subject to the Sponsor depositing additional funds into the Trust Account). Globalink intends to complete a business combination before the mandatory liquidation date.
Off-Balance Sheet Arrangements
Globalink has no obligations, assets or liabilities, which would be considered off-balance sheet arrangements as of September 30, 2024, March 31, 2024 and December 31, 2023. Globalink does not participate in transactions that create relationships with entities or financial partnerships, often referred to as variable interest entities, which would have been established for the purpose of facilitating off-balance sheet arrangements. Globalink have not entered into any off-balance sheet financing arrangements, established any special purpose entities, guaranteed any debt or commitments of other entities, or purchased any non-financial assets.
Contractual obligations
Globalink does not have any long-term debt, capital lease obligations, operating lease obligations or long-term liabilities.
Registration Rights
The holders of the insider shares, the private units and any units that may be issued upon conversion of working capital loans or extension loans (and any securities underlying the private units or units issued upon conversion of the working capital loans or extension loans) will be entitled to registration rights pursuant to a registration rights agreement requiring us to register such securities for resale. The holders of these securities are entitled to make up to two demands, excluding short form demands, that we register such securities. In addition, the holders have certain “piggy-back” registration rights with respect to registration statements filed subsequent to our completion of our initial business combination and rights to require us to register for resale such securities pursuant to Rule 415 under the Securities Act.
Underwriting Agreement
The underwriters are entitled to a deferred underwriting discounts of $0.35 per unit, or $4,025,000 from the closing of the IPO and the Over-Allotment Units. The deferred discounts will become payable to the underwriters from the amounts held in the Trust Account solely if Globalink completes an initial business combination, subject to the terms of the underwriting agreement.
Right of First Refusal
Subject to certain conditions, Globalink granted Chardan for a period of 12 months after the date of the consummation of our initial business combination, a right of first refusal to act as sole/lead left/lead book-running manager, with at least 30% of the economics, for any and all future public and private equity and debt offerings, and as sole/lead left financial advisor with respect to any and all M&A Transactions. In accordance with FINRA Rule 5110(g)(6)(A), such right of first refusal shall not have a duration of more than three years from the effective date of this registration statement.
Promissory Notes
As of September 30, 2024, March 31, 2024 and December 31, 2023, the aggregate amounted owed in connection with the promissory notes was $3,736,066, $2,640,649 and $1,757,255, respectively, which includes interest accrued as reflected on the consolidated balance sheets.
| 233 |
Jumpstart Our Business Startups Act of 2012 (the “JOBS Act”)
On April 5, 2012, the JOBS Act was signed into law. The JOBS Act contains provisions that, among other things, relax certain reporting requirements for qualifying public companies. We will qualify as an “emerging growth company” and under the JOBS Act will be allowed to comply with new or revised accounting pronouncements based on the effective date for private (not publicly traded) companies. We are electing to delay the adoption of new or revised accounting standards, and as a result, we may not comply with new or revised accounting standards on the relevant dates on which adoption of such standards is required for non-emerging growth companies. As such, our consolidated financial statements may not be comparable to companies that comply with public company effective dates.
Additionally, Globalink is in the process of evaluating the benefits of relying on the other reduced reporting requirements provided by the JOBS Act. Subject to certain conditions set forth in the JOBS Act, if, as an “emerging growth company,” we choose to rely on such exemptions we may not be required to, among other things, (i) provide an auditor’s attestation report on our system of internal control over financial reporting pursuant to Section 404 of the Sarbanes-Oxley Act, (ii) provide all of the compensation disclosure that may be required of non-emerging growth public companies under the Dodd-Frank Wall Street Reform and Consumer Protection Act, (iii) comply with any requirement that may be adopted by the PCAOB regarding mandatory audit firm rotation or a supplement to the auditor’s report providing additional information about the audit and the consolidated financial statements (auditor discussion and analysis) and (iv) disclose certain executive compensation related items such as the correlation between executive compensation and performance and comparisons of executive compensation to median employee compensation. These exemptions will apply for a period of five years following the completion of our IPO or until we are no longer an “emerging growth company,” whichever is earlier.
Critical Estimates
Management’s discussion and analysis of Globalink’s results of operations and liquidity and capital resources are based on the financial information. The financial statements have been prepared in accordance with U.S. GAAP. Certain of our accounting policies require that management apply significant judgments in defining the appropriate assumptions integral to financial estimates. On an ongoing basis, management reviews the accounting policies, assumptions, estimates and judgments to ensure that our financial statements are presented fairly and in accordance with U.S. GAAP. Judgments are based on historical experience, terms of existing contracts, industry trends and information available from outside sources, as appropriate. Globalink have identified the following critical accounting policies:
Accounting for Warrants
Globalink accounts for warrants as either equity-classified or liability-classified instruments based on an assessment of the warrant’s specific terms and applicable authoritative guidance in FASB, ASC 480 and ASC 815. The assessment considers whether the warrants are freestanding financial instruments pursuant to ASC 480, meet the definition of a liability pursuant to ASC 480, and whether the warrants meet all of the requirements for equity classification under ASC 815, including whether the warrants are indexed to the Company’s own shares of common stock, among other conditions for equity classification. This assessment, which requires the use of professional judgment, is conducted at the time of warrant issuance and as of each subsequent quarterly period end date while the warrants are outstanding. Globalink accounts for the warrants issued in connection with our IPO in accordance with the guidance contained in ASC 815 under which the public warrants meet the criteria for equity treatment and the private warrants do not meet the criteria for equity treatment and must be recorded as liabilities. Accordingly, the private warrants are classified as liabilities at their fair value and adjust the private warrants to fair value at each reporting period. This liability is subject to re-measurement at each balance sheet date until exercised, and any change in fair value is recognized in our consolidated statements of operations. The fair value of the warrants was estimated using a binomial lattice model.
In determining the fair value of the Private Placement Warrants assumptions related to exercise price, market price of the public stock, expected life and risk-free interest rate are utilized. Globalink estimates the volatility of its common stock based on historical volatility that matches the expected remaining life of the warrants.
Recent Accounting Pronouncements
In December 2023, the FASB issued ASU 2023-09, Income Taxes (Topic 740): Improvements to Income Tax Disclosures (ASU 2023-09), which requires disclosure of incremental income tax information within the rate reconciliation and expanded disclosures of income taxes paid, among other disclosure requirements. ASU 2023-09 is effective for fiscal years beginning after December 15, 2024. Early adoption is permitted. The Company’s management does not believe the adoption of ASU 2023-09 will have a material impact on its consolidated financial statements and disclosures.
Management does not believe that any recently issued, but not yet effective, accounting standards, if currently adopted, would have a material effect on our unaudited condensed consolidated financial statements as of September 30, 2024.
| 234 |
MANAGEMENT’S
DISCUSSION AND ANALYSIS OF
FINANCIAL CONDITION AND RESULTS OF OPERATIONS OF ALPS
Unless the context otherwise requires, all references in this section to the “Company,” “Alps Group,” “we,” “us” or “our” refer to the business of Alps Life Sciences Inc, Alps Global Holding Berhad and its subsidiaries prior to the consummation of the Business Combination, which will be the business of Alps Global Holding Pubco and its subsidiaries following the consummation of the Business Combination. Alps Global Holding Pubco is a newly incorporated exempted company in Cayman Islands on May 14, 2024 for the purpose of completing a business combinations with all of the shareholders of Alps Global Holding Berhad will exchange their equity ownership for identical interests in Alps Pubco. Alps Pubco has not commenced operations and has no assets or liabilities since its incorporation.
You should read the following discussion and analysis of our financial condition and results of operations together with the “Selected Historical Consolidated Financial Data of Alps” and the historical audited consolidated financial statements as of and for the years ended March 31, 2023 and 2024 and the related notes included elsewhere in this proxy statement/prospectus. Some of the information contained in this discussion and analysis or set forth elsewhere in this proxy statement/prospectus, including information with respect to our plans and strategy for our business and related financing, includes forward-looking statements that involve risks and uncertainties. As a result of many factors, such as those set forth under the “Risk Factors” and “Cautionary Note Regarding Forward-Looking Statements” sections and elsewhere in this proxy statement/prospectus, our actual results may differ materially from those anticipated in these forward-looking statements.
Overview
Alps Life Sciences Inc (“Alps Holdco”) is an exempted company incorporated in the Cayman Islands with limited liability and Alps Global Holding Berhad (“Alps”) is a public limited company incorporated in Malaysia on April 14, 2017 and will be a subsidiary of Alps Holdco prior to Closing. Alps’ primary objective is to evolve into a fully integrated bench-to-bedside platform encompassing biotechnology research, medical services, and wellness solutions. Alps’ principal activities are research and development in various biotechnology-related products and services, with a focus on the use of precision and preventive medicine.
Alps intends to take a targeted approach to addressing challenges in the biotech and healthcare sector in ASEAN. Its efforts focus on the following areas:
| ● | Transitioning scientific discoveries into commercially viable products and treatments, thereby improving access to healthcare solutions. | |
| ● | Enhancing research capabilities by facilitating the development of infrastructure and establishing dependable supply chains to support innovation. | |
| ● | Implementing programs to recruit, train, and retain skilled professionals in specialized biotech disciplines. | |
| ● | Enabling collaboration among startups, corporations, and research institutions to build cohesive ecosystems that support growth. | |
| ● | Addressing funding gaps for early-stage research and development by working to reduce risk perceptions and facilitating investment opportunities. |
Financial Operations Overview
The following table summarizes our consolidated results of operations for the year ended March 31, 2023:
| For the Year Ended March 31, | ||||||||
| 2024 | 2023 | |||||||
| US$ | US$ | |||||||
| Revenue | 2,403,552 | 3,594,828 | ||||||
| Cost of sales | (1,804,622 | ) | (1,617,701 | ) | ||||
| Gross Profit | 598,930 | 1,977,127 | ||||||
| Other operating income | 49,977 | 4,594 | ||||||
| Distribution expenses | (160,855 | ) | (223,715 | ) | ||||
| Administrative expenses | (1,929,680 | ) | (1,344,398 | ) | ||||
| Other operating expenses | (917,524 | ) | (950,695 | ) | ||||
| Share results of associate | 3,421 | 1,635 | ||||||
| (2,355,731 | ) | (535,452 | ) | |||||
| Finance costs | (42,844 | ) | (46,608 | ) | ||||
| Loss before income tax | (2,398,575 | ) | (582,060 | ) | ||||
| Income tax expense | 7,414 | (147,802 | ) | |||||
| Loss for the financial year | (2,391,161 | ) | (729,862 | ) | ||||
| Other comprehensive loss, net of income tax | ||||||||
| Foreign currency translation, representing other comprehensive loss for the financial year | (121,289 | ) | (124,413 | ) | ||||
| Total comprehensive loss for the financial year | (2,512,459 | ) | (854,275 | ) | ||||
| Loss for the financial year attributable to: | ||||||||
| Owners of the Company | (2,304,026 | ) | (729,159 | ) | ||||
| Non-controlling interests | (87,135 | ) | (703 | ) | ||||
| (2,391,161 | ) | (729,862 | ) | |||||
| Total comprehensive loss for the financial year attributable to: | ||||||||
| Owners of the Company | (2,425,324 | ) | (853,572 | ) | ||||
| Non-controlling interests | (87,135 | ) | (703 | ) | ||||
| (2,512,459 | ) | (854,275 | ) | |||||
| 235 |
Revenue
During the years ended March 31, 2024 and 2023, Alps generated revenue from the provision of cellular therapy, medical testing, laboratory and aesthetics services as set forth below.
| For the Year Ended March 31, | ||||||||
| 2024 | 2023 | |||||||
| US$ | US$ | |||||||
| Cellular Therapy | 407,443 | 245,875 | ||||||
| Medical testing, laboratory and aesthetics beauty services | 1,755,910 | 3,009,485 | ||||||
| Consultation fee | 232,173 | 330,104 | ||||||
| Sale of medicine and healthcare product | 8,026 | 9,364 | ||||||
| 2,403,552 | 3,594,828 | |||||||
The decrease in revenue from $3,594,828 during the year ended March 31, 2023, to $2,403,552 during the year ended March 31, 2024, resulted primarily from the decrease in medical testing, laboratory and aesthetics beauty services as offset by an increase of $161,568 from the provision of cellular therapy services. The decrease in medical testing, laboratory and aesthetic beauty services was due primarily from a decrease in COVID-19 RT-PCR screening revenue of $1,105,415, from $1,120,219 in year ended March 31, 2023 to $14,804 in year ended March 31, 2024, and partially offset with the increase in hair implant service revenue of $316,880, from $521,000 in year ended March 31, 2023 to $837,880 in year ended March 31, 2024. In the near future, we expect to continue to derive the majority of our revenues from the provision of medical testing, laboratory and aesthetics beauty services as we continue to advance our product candidates through and beyond the pre-clinical phase.
Cost of sales
| For the Year Ended March 31, | ||||||||
| 2024 | 2023 | |||||||
| US$ | US$ | |||||||
| Cost of inventories | 833,774 | 919,726 | ||||||
| Direct labour costs | 432,315 | 357,226 | ||||||
| Profit sharing on hair implant services | 522,066 | 340,749 | ||||||
| Cellular therapy cost | 15,499 | - | ||||||
| Others | 968 | - | ||||||
| 1,804,622 | 1,617,701 | |||||||
Alps’ cost of sales includes cost of inventories for services performed and products sold, personnel-related expenses, profit sharing on hair implant services, cellular therapy cost and others as shown in the table above. The overall increase in cost of sales was mainly due to the increase in profit sharing on hair implant services which resulted from the increase in hair implant services revenue categorized under medical testing, laboratory and aesthetics beauty services, the increase in direct labor costs due to the increase in personnel to support the operational and business expansion of Alps as offset by the reduction in cost of inventories of $85,952.
Alps Wellness Centre Sdn. Bhd. (“Alps Wellness”) is a party to a Memorandum of Agreement, dated November 1, 2023 (the “MoA”), with Advanced Hair Transplant Specialist Sdn. Bhd. (“Advanced Hair”), pursuant to which Advanced Hair agreed to provide hair transplant, mesotherapy and hair filler services using the facilities and equipment of Alps Wellness. Pursuant to the MoA, Alps Wellness will provide the facilities and financial support for the hair transplant services, while Advanced Hair will be responsible for delivering hair transplant services and conducting marketing efforts. The MOA is for a term of two years, commencing on November 1, 2023, and ending on October 31 2025, with an option for mutual extension. In consideration of the collaboration, the profit-sharing arrangement stipulates that profits, after deducting the cost of goods sold for the hair transplant services, will be allocated as follows: 40% to Alps Wellness and 60% to Advanced Hair. Alps generates revenues through the provision of aesthetic services, including hair implants, to support its overall business and also to enable Alps to reinvest in its research and development initiatives.
| 236 |
Other Operating Income
| For the Year Ended March 31, | ||||||||
| 2024 | 2023 | |||||||
| US$ | US$ | |||||||
| Refund of research fees | 48,457 | - | ||||||
| Sundry income | 1,305 | 946 | ||||||
| Wages subsidy | - | 3,648 | ||||||
| Gain on disposal of investment | 215 | - | ||||||
| 49,977 | 4,594 | |||||||
Alps’ other operating income for the financial year ended March 31, 2024 mainly comprised of the refund of the research fees resulting from the restructuring of our collaboration arrangement with USM under the Research Collaboration Agreement dated September 1, 2020, which is currently expired. The one-time refund is related to our mRNA diagnostic research project, and is not expected to occur in the future.
Distribution expenses
| For the Year Ended March 31, | ||||||||
| 2024 | 2023 | |||||||
| US$ | US$ | |||||||
| Agent commission | 159,998 | 221,610 | ||||||
| Advertisement and promotion | 857 | 985 | ||||||
| Travelling expenses | - | 1,120 | ||||||
| 160,855 | 223,715 | |||||||
During the year ended March 31, 2024, we shifted our distribution agents from external sales agent to internal sales and marketing personnel. As a result, agent commissions for the year ended March 31, 2024, decreased $61,612 as compared to the same period ended March 31, 2023. In the near future, we expect to reduce our reliance on the external sales agents to internal sales agents.
Administrative expenses
| For the Year Ended March 31, | ||||||||
| 2024 | 2023 | |||||||
| US$ | US$ | |||||||
| Payroll costs | 962,102 | 772,431 | ||||||
| Research expenses* | 276,957 | 298,432 | ||||||
| Advisory Fee | 19,374 | 14,599 | ||||||
| Professional Fees - Audit Fee | 214,839 | 11,333 | ||||||
| Professional Fees - De-SPAC related | 102,090 | 869 | ||||||
| Professional Fees - Others | 28,725 | 20,415 | ||||||
| Professional Fees - Tax And Secretary Statutory Filling | 10,600 | 5,912 | ||||||
| Subscription & Membership Fees | 27,504 | 19,510 | ||||||
| Transportation Fee | 4,047 | 426 | ||||||
| Travelling Expenses - Lodging And Accommodation | 16,710 | 1,178 | ||||||
| Travelling Expenses - Air Fare | 19,332 | 24,051 | ||||||
| Telephone & Fax Charges | 12,894 | 11,152 | ||||||
| Water & Electricity | 58,299 | 130,361 | ||||||
| R&M - Laboratory Equipment | 66,898 | 33,729 | ||||||
| Miscellaneous | 109,309 | - | ||||||
| 1,929,680 | 1,344,398 | |||||||
*Alps has excluded salaries and payroll costs for employees engaged in research activities from the research expenses component and instead included them in the payroll costs component.
Administrative expenses primarily consist of expenses incurred on research expenses, salaries, welfare benefits, and other related costs for employees in finance, operations, and administrative roles. These expenses also include professional fees for legal, auditing, tax and consulting services.
Our research activities are central to our business and we anticipate that they will increase in the foreseeable future as we advance our pre-clinical development for product candidates and continue to make strides in our scientific research and product development.
The following table illustrates the total research expenses incurred by each pipeline that are captured in our profit or loss for the financial year ended March 31, 2023 and March 31, 2024.
| For the Year Ended March 31, | ||||||||
| 2024 | 2023 | |||||||
| US$ | US$ | |||||||
| NK Cell Therapy – MyImmune* | 22,195 | 8,095 | ||||||
| CAR-T Cell* | * | * | ||||||
| COVID-19 Vaccine (mRNA)* | 18,513 | 31,670 | ||||||
| Cholera Vaccine* | * | * | ||||||
| MYCELEST* | 221,291 | 245,528 | ||||||
| iPSC (Heart Failure)* | 218 | - | ||||||
| mRNA (Diagnostic)* | 11,845 | 9,562 | ||||||
| CELESOME(+)* | 2,895 | 3,577 | ||||||
| 276,957 | 298,432 | |||||||
*In these pipelines, certain expenses, particularly salaries and payroll costs, are not individually tracked. These expenses are shared across research and development of various pipelines, and Alps Group does not allocate them to any specific pipeline.
The increase in administrative expenses was primarily due to the increase in payroll costs as evidenced by the increase in personnel from 72 for the financial year ended March 31, 2023, to 83 for the year ended March 31, 2024, and the increase in de-SPAC related professional fees including audit fees, legal counsels and consultancy fees.
Other Operating Expenses
| For the Year Ended March 31, | ||||||||
| 2024 | 2023 | |||||||
| US$ | US$ | |||||||
| Amortisation of right-of-use assets | 403,019 | 326,992 | ||||||
| Depreciation of property, plant and equipment | 455,613 | 479,861 | ||||||
| Amortisation of intangible assets | 44,807 | 91,773 | ||||||
| Property, plant and equipment written off | 14,084 | 52,069 | ||||||
| 917,523 | 950,695 | |||||||
Other operating expenses of Alps for both financial years mainly comprised of amortization of right-of-use assets and intangible assets and depreciation charge of property, plant and equipment.
Finance Costs
| For the Year Ended March 31, | ||||||||
| 2024 | 2023 | |||||||
| US$ | US$ | |||||||
| Interest expense on lease liabilities | 42,250 | 45,497 | ||||||
| Hire purchase interests | 594 | 1,111 | ||||||
| 42,844 | 46,608 | |||||||
| 237 |
Income Tax Benefit (Expense)
During the year ended March 31, 2024, our income tax benefit was $7,414 as compared to income tax expense of $147,802 for the same period ended March 31, 2023 2023. The main factor contributing to the income tax expense for the year ended March 31, 2023 was derived from the under provision of income tax expense in TMC Global Holdings Sdn. Bhd. (“TMC”) in FY2022, by USD118,113. The under provision arises from the deferred income which should be taxed ahead in accordance to the Malaysian income tax laws.
In FY2024, income tax benefit resulted from the derecognition of deferred tax liabilities from TMC. TMC is in deferred tax assets position as of FY2024 resulting from the unabsorbed business losses during the financial year.
Liquidity and Capital Resources
Overview
Since our inception, we have incurred significant operating losses and expect to continue incurring substantial expenses and operating losses for the foreseeable future as we advance the preclinical and clinical development of our research programs and product candidates. Our research and development and general and administrative expenses are anticipated to increase due to the execution of clinical trials and preclinical studies, expansion of our intellectual property portfolio, and provision of general and administrative support for our operations. Consequently, we will require additional capital to fund our operations, which may be obtained through additional equity or debt financings, collaborations, licensing arrangements, or other sources. We expect that we will need approximately US$1,500,000 in the next 12 months to finance our working capital needs and an additional US$1,000,000 for the next 12 months following.
The table below is extracted from the unaudited pro forma condensed combined balance sheet, presenting the anticipated liquidity position of the Combined Company post-Business Combination, based on two potential redemption scenarios:
| Scenario 1: No Additional Redemption Scenario | Scenario 2: Maximum Contractual Redemption Scenario | |||||||
| US$ | US$ | |||||||
| Cash and cash equivalent | 60,534,501 | 46,835,214 | ||||||
| Total liabilities (excluding earnout liability) | (6,428,251 | ) | (6,428,251 | ) | ||||
| Anticipated Liquidity Position | 54,106,250 | 40,406,963 | ||||||
Cash Flows
The following table summarizes our cash flows for the year/period presented:
| For the Year Ended March 31, | ||||||||
| 2024 | 2023 | |||||||
| US$ | US$ | |||||||
| Net cash flows from (used in) | ||||||||
| Operating Activities | (2,859,698 | ) | (113,569 | ) | ||||
| Investing Activities | (221,158 | ) | (288,294 | ) | ||||
| Financing Activities | 3,300,614 | 212,921 | ||||||
| Exchange Rate Differences | (21,105 | ) | (24,161 | ) | ||||
| Net decrease in cash and cash equivalents | 198,653 | (213,103 | ) | |||||
Operating activities
The cash flows used in operating activities for the year ended March 31, 2024 of US$2,859,698 comprised loss before tax of US$2,407,317, positive non-cash adjustments of US$965,472, net negative change in assets and liabilities of US$1,301,162 and tax paid of US$116,691. Non-cash adjustments primarily included amortization of right-of-use assets, depreciation of property, plant and equipment, amortization of intangible assets and property, plant and equipment written off. The net negative change in assets and liabilities was primarily due to an increase in other receivables, deposits and prepayments.
The cash flows used in operating activities for the year ended March 31, 2023 of US$113,569 comprised loss before tax of US$582,060, positive non-cash adjustments of US$1,013,354, net negative change in assets and liabilities of US$394,596 and tax paid of US$150,267. Non-cash adjustments primarily included amortization of right-of-use assets, depreciation of property, plant and equipment, amortization of intangible assets and property, plant and equipment written off. The net negative change in assets and liabilities was primarily due to an increase in inventories and decrease in other payables and accruals.
| 238 |
Investing activities
The cash flows used in investing activities for the year ended March 31, 2024 of US$ 221,158 comprised of purchase of property, plant and equipment of US$221,317 and net advances to associate companies of US$159.
The cash flows used in investing activities for the year ended March 31, 2023 of US$228,294 comprised of purchase of property, plant and equipment of US$274,467, net advances to associate companies of US$13,153 and subscription of shares in an associate company of US$674.
Financing activities
The cash flows from financing activities for the year ended March 31, 2024 of US$3,300,614 comprised of proceeds from issuance of shares of $2,317,209, advances from directors of $1,451,612, repayment of hire purchase of US$10,544, repayment of lease liabilities of US$414,819 and interest paid of US$42,844.
The cash flows from financing activities for the year ended March 31, 2023 of US$212,921 comprised of repayment of hire purchase of US$22,677, repayment of lease liabilities of US$322,547, interest paid of US$46,608 and advances received from directors of US$604,753.
Off-Balance Sheet Arrangements
Alps does not have any off-balance sheet arrangements.
Recently Issued Accounting Pronouncements
For a discussion of new accounting pronouncements see “Notes to Consolidated Financial Statements” located elsewhere in this proxy statement/prospectus.
Quantitative and Qualitative Disclosures about Market Risks
Market risk is the risk that the fair value or future cash flows of a financial instrument will fluctuate due to changes in market prices. Market risk comprises of three types of risk: interest risk, foreign currency risk and other price risk. Alps’ exposure to these risks is not material.
Critical Accounting Policies
Alps prepares its consolidated financial statements in accordance with International Financial Reporting Standards (“IFRS”) issued by the International Accounting Standards Board. In doing so, it has to make estimates and assumptions that affect its reported amounts of assets, liabilities, revenues and expenses, as well as related disclosure of contingent assets and liabilities. To the extent that there are material differences between these estimates and actual results, Alps’ financial condition or operating results and margins would be affected. Alps bases its estimates on past experience and other assumptions that it believes are reasonable under the circumstances, and it evaluates these estimates on an ongoing basis. Alps refers to accounting estimates of this type as critical accounting policies and estimates, which is discussed further below.
Impairment of Financial Assets
Alps recognises a loss allowance for expected credit losses (“ECL”) on trade receivables, other receivables and deposits, amount owing due associates as well as cash and bank balances. The amount of ECL is updated at each reporting date to reflect changes in credit risk since initial recognition of the respective financial instrument.
Alps recognises lifetime ECL for trade receivables. Alps considers past loss experience, timing of billing and observable data such as current changes and future forecasts in economic conditions to estimate the amount of expected impairment loss. The methodology and assumptions including any forecasts of future economic conditions are reviewed regularly.
For all other financial instruments, Alps Group recognise lifetime ECL when there has been a significant increase in credit risk since initial recognition. However, if the credit risk on the financial instrument has not increased significantly since initial recognition, Alps measures the loss allowance for that financial instrument at an amount equal to 12-month ECL.
Lifetime ECL represents the expected credit losses that will result from all possible default events over the expected life of a financial instrument. In contrast, 12-month ECL represents the portion of lifetime ECL that is expected to result from default events on a financial instrument that are possible within 12 months after the reporting date.
Reversal of impairment loss to profit or loss, if any, is restricted to not exceeding what the amortised cost would have been had the impairment not been recognised previously.
| 239 |
Significant increase in credit risk
In assessing whether the credit risk on a financial asset has increased significantly since initial recognition, Alps compares the risk of a default occurring on the financial instrument at the reporting date with the risk of a default occurring on the financial instrument at the date of initial recognition. In making this assessment, Alps considers both quantitative and qualitative information that is reasonable and supportable, including historical experience and forward-looking information such as financial evaluation of the creditworthiness of the debtors, ageing of receivables, defaults and past due amounts, past experience with the debtors, current conditions and reasonable forecast of future economic conditions.
Alps presumes that the credit risk on a financial asset has increased significantly since initial recognition when contractual payments are more than 365 days past due, unless Alps has reasonable and supportable information that demonstrates otherwise.
Probability of default
Alps considers the information developed internally or obtained from external sources indicates that the debtor is unlikely to pay its creditors, including Alps, in full, as constituting an event of default for internal credit risk management purposes as historical experience indicates that financial assets are generally not recoverable.
Alps considers that default has occurred when a financial asset is more than 365 days past due unless Alps has reasonable and supportable information to demonstrate that a more lagging default criterion is more appropriate.
Write-off policy
Alps writes off a financial asset when there is information indicating that the debtor is in severe financial difficulty and there is no realistic prospect of recovery, with case-by-case assessment performed based on indicators such as insolvency or demise. Financial assets written off may still be subject to enforcement activities under Alps’ recovery procedures, taking into account legal advice where appropriate. Any recoveries made are recognised in profit or loss as bad debts recovered.
Measurement and recognition of expected credit losses
The measurement of expected credit losses is a function of the probability of default, loss given default (i.e. the magnitude of the loss if there is a default) and the exposure at default. The assessment of the probability of default and loss given default is based on historical data adjusted by forward-looking information as described above. As for the exposure at default, for financial assets, this is represented by the assets’ gross carrying amount at the reporting date.
For financial assets, the expected credit loss is estimated as the difference between all contractual cash flows that are due to Alps in accordance with the contract and all the cash flows that Alps expects to receive, discounted at the original effective interest rate.
If Alps has measured the loss allowance for a financial instrument at an amount equal to lifetime ECL in the previous reporting period, but determine at the current reporting date that the conditions for lifetime ECL are no longer met, Alps measures the loss allowance at an amount equal to 12-month ECL at the current reporting date, except for assets for which the simplified approach was used.
Alps recognizes an impairment gain or loss in profit or loss for all financial instruments with a corresponding adjustment to their carrying amount through a loss allowance account.
| 240 |
UNAUDITED PRO FORMA CONDENSED COMBINED FINANCIAL INFORMATION
On January 30, 2024, Globalink entered into the Merger Agreement with Alps Holdco, the Sponsor, and the Seller Representative. The Merger Agreement was amended and restated on May 20, 2024 and entered into by and among Globalink, PubCo, Alps Holdco, Merger Sub, the Sponsor, and the Seller Representative. Pursuant to the terms of the Merger Agreement, the Business Combination between Globalink and Alps Holdco will be effected in two steps: (i) the Redomestication Merger, whereby, subject to the approval and adoption of the Merger Agreement by the stockholders of Globalink, Globalink will merge with and into PubCo, with PubCo remaining as the surviving publicly traded entity; and (ii) the Acquisition Merger, whereby Merger Sub will merge with and into Alps Holdco, resulting in Alps Holdco remaining as the surviving entity and being a wholly-owned subsidiary of PubCo. Subject to the terms and conditions set forth therein upon the Closing, each Alps Holdco Ordinary Share issued and outstanding immediately prior to the Effective Time (other than treasury shares or dissenting shares) will be ultimately converted into the right to receive PubCo ordinary shares. The total consideration to be paid by Globalink to the Alps Holdco Shareholders in the form of PubCo ordinary shares at the Closing will be equal to $1.6 billion, with an earnout provision permitting the shareholders of Alps Holdco to receive up to 48 million additional shares of the Combined Company as and when the business meets certain incremental milestones relating to the consolidated revenue of the Combined Company for five fiscal years following the Closing.
The Merger Agreement is subject to certain customary closing conditions and contains customary representations, warranties, covenants and indemnity provisions. Capitalized terms used but not defined herein shall have the respective meanings set forth in the Merger Agreement. The respective boards of directors of Globalink and Alps Holdco have (i) approved and declared advisable the Merger Agreement, the Merger and the transactions contemplated by the Merger Agreement and (ii) resolved to recommend approval of the Merger Agreement and related transactions by their respective shareholders.
The unaudited pro forma condensed combined balance sheet as of March 31, 2024 combines the unaudited historical condensed consolidated balance sheet of Globalink as of March 31, 2024 with the audited historical consolidated balance sheet of Alps Holdco as of March 31, 2024, giving effect to the Business Combination, as if it had been consummated as of that date.
The unaudited pro forma condensed combined statement of operations for the fiscal year ended March 31, 2024 combines the unaudited historical condensed consolidated statement of operations of Globalink for the trailing twelve months ended March 31, 2024 (compiled with the audited consolidated statement of operations for the year ended December 31, 2023 less the unaudited condensed consolidated statement of operations for the three month period ended March 31, 2023 plus the unaudited condensed consolidated statement of operations for the three months ended March 31, 2024 of Globalink) with the audited historical consolidated statement of operations of Alps Holdco for the fiscal year ended March 31, 2024, giving effect to the Business Combination, as if it had been consummated as of April 1, 2023, the earliest period presented.
The historical financial information has been adjusted to give pro forma effect to events that relate to material financing transactions consummated after March 31, 2024 and pro forma adjustments that are directly attributable to the Business Combination. The adjustments presented on the unaudited pro forma condensed combined financial statements have been identified and presented to provide relevant information necessary for an accurate understanding of the combined company upon consummation of the Business Combination.
The historical financial consolidated statements of Alps Holdco have been prepared in accordance with IFRS as issued by the IASB. The historical consolidated financial statements of Globalink have been prepared in accordance with U.S. GAAP. The condensed combined pro forma financial information reflects IFRS and in USD, the basis of accounting used by the registrant, PubCo, and other than the reclassification and presentation of redeemable Globalink’s public shares as other liabilities and the reclassification and presentation of the public warrants as liabilities under IFRS, disclosed in the pro forma notes, no material accounting policy difference is identified in converting Globalink’s historical consolidated financial statements from U.S. GAAP to IFRS. The adjustments presented in the pro forma condensed combined financial information have been identified and presented to provide relevant information necessary for an accurate understanding of PubCo after giving effect to the Business Combination.
The unaudited pro forma condensed combined financial information is for illustrative purposes only. The financial results may have been different had the companies always been combined. You should not rely on the unaudited pro forma condensed combined financial information as being indicative of the historical results that would have been achieved had the companies always been combined or the future results that the combined company will experience. Globalink and Alps Holdco have not had any historical relationship prior to the Business Combination. Accordingly, no pro forma adjustments were required to eliminate activities between the companies.
Alps Holdco was created on April 11, 2024 as such the financial statement as of March 31, 2024 for Alps Holdco, reflected in these pro forma financial statement as of March 31, 2024, represent the financial statement of Alps Global Holding Berhad as if the common control transaction had occurred on April 1, 2023 the earlies period presented. The financial statements of Alps Holdco were deemed de-minimis since inception as such no incremental adjustment is required to reflect the common control transaction between Alps and Alps Holco. This information should be read together with the following:
| ● | the historical unaudited condensed consolidated financial statements of Globalink as of March 31, 2024 and for the three months ended March 31, 2024; | |
| ● | the historical audited financial statements of Alps Holdco as of March 31, 2024 and for the year ended March 31, 2024 |
| 241 |
| ● | the historical audited consolidated financial statements of Globalink as of December 31, 2023 and for the year ended December 31, 2023; | |
| ● | the historical unaudited condensed consolidated financial statements of Globalink as of March 31, 2023 and for the three months ended March 31, 2023; | |
| ● | the sections titled “Management’s Discussion and Analysis of Financial Condition and Results of Operations of Globalink,” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations of Alps Holdco” and other financial information included elsewhere in this proxy statement/prospectus; and | |
| ● | other information relating to Globalink and Alps Holdco included in this proxy statement/prospectus, including the Merger Agreement and the description of certain terms thereof set forth under the section entitled “The Business Combination.” |
Description of the Business Combination
As a result of and upon the Closing, pursuant to the terms of the Merger Agreement, all of the outstanding shares of Alps Holdco will be cancelled in exchange for the right to receive PubCo ordinary shares. The aggregate consideration for the Business Combination is $1.6 billion, payable at the Closing in the form of newly issued PubCo ordinary shares, par value $0.0001 per share. The Merger Consideration Shares will be allocated pro rata with each Alps Holdco Shareholder receiving a number of PubCo ordinary shares determined in accordance with the terms of the Merger Agreement. Moreover, Alps Holdco Shareholders will be eligible to receive up to an aggregate of 48,000,000 additional PubCo ordinary shares (the “Earnout Shares”) upon the completion of certain milestones relating to the consolidated revenue of the Combined Company for five fiscal years following the Closing.
Following the execution of the Merger Agreement, PubCo, Globalink and Alps Holdco entered into subscription agreements with certain investors for 4,020,000 PubCo ordinary shares for a total of $40,200,000 in a PIPE Investment to be consummated simultaneously with the Closing. Consummation of the PIPE Investment is conditioned on the concurrent Closing of the Business Combination and other customary closing conditions.
The Business Combination Agreement provides that the parties will use commercially reasonable efforts to obtain commitments for the PIPE Investment prior to Closing and the parties are currently pursuing subscriptions for the PIPE Investment. Parent shall have net tangible assets of at least $5,000,001 immediately prior to consummation of the merger, which is anticipated to be waived, and Pubco shall have net tangible assets of at least $5,000,001 upon consummation of the merger, at any level of redemptions.
At the Closing of the Business Combination, the former Alps Holdco Shareholders will receive an aggregate of 160,000,000 PubCo ordinary shares, among which 8,000,000 PubCo ordinary shares are to be issued and held in escrow to satisfy any indemnification obligations incurred under the Merger Agreement.
| 242 |
Anticipated Accounting Treatment
The Business Combination will be accounted for as capital reorganization with no goodwill or other intangible assets recorded, in accordance with IFRS. A capital reorganization does not result in a new basis of accounting, and the financial statements of the combined entity represent the continuation of the financial statements of Alps Holdco in many respects. However, Globalink does not meet the definition of a “business” pursuant to IFRS 3 Business Combinations, and thus, for accounting purposes, the Business Combination will be accounted for as a capital reorganization.
Under this method of accounting, Globalink will be treated as the “acquired” company for financial reporting purposes. For accounting purposes, Alps Holdco will be deemed to be the accounting acquirer in the transaction and, consequently, the transaction will be treated as a recapitalization of Alps Holdco (i.e., a capital transaction involving the issuance of shares by PubCo for the shares of Alps Holdco). Accordingly, the consolidated assets, liabilities and results of operations of Alps Holdco will become the historic financial statements of the Combined Company, and Globalink’s assets, liabilities and results of operations will be consolidated with Alps Holdco beginning on the acquisition date. Operations prior to the Business Combination will be presented as those of Alps Holdco in future reports. The net assets of Globalink will be recognized at carrying value, with no goodwill or other intangible assets recorded.
The deemed costs of the shares issued by PubCo, which represents the fair value of the shares that Alps Holdco would have had to issue for the ratio of ownership interest in PubCo to be the same as if the Business Combination had taken the legal form of Alps Holdco acquiring shares of Globalink, in excess of the net assets of Globalink will be accounted for as stock-based compensation under IFRS 2 Share-Based Payment.
Alps Holdco has been determined to be the accounting acquirer based on evaluation of the following facts and circumstances:
| ● | Alps Holdco Shareholders will have the largest voting interest in PubCo under both the no redemption and maximum contractual redemption scenarios; | |
| ● | The board of directors of the Combined Company will be designated solely by Alps Holdco, with at least three (3) directors qualifying as independent directors under the Securities Act and the Nasdaq rules); | |
| ● | Alps Holdco’s senior management will be the senior management of the Combined Company; | |
| ● | The business of PubCo will comprise the ongoing operations of Alps Holdco; and | |
| ● | Alps Holdco is the larger entity, in terms of substantive assets. |
Basis of Pro Forma Presentation
The unaudited pro forma condensed combined financial information has been prepared assuming two redemption scenarios as follows:
| ● | Assuming No Redemptions: This scenario assumes that no shares of Globalink common stock are redeemed; and | |
| ● | Assuming Maximum Contractual Redemptions: This scenario assumes that 277,511 shares of Globalink common stock, the maximum contractual redemption of the outstanding Globalink common stock, are redeemed for their pro rata share of the cash in the Trust Account. This presentation assumes that Globalink stockholders exercise their redemption rights with respect to a maximum of 277,511 common stock for an aggregate payment of approximately $3.33 million upon consummation of the Business Combination at a redemption price of approximately $11.98 per share, determined based on funds in trust plus interest earned net of taxes payable. The maximum contractual redemption amount reflects the maximum number of Globalink public shares that can be redeemed without violating the requirement of Globalink Charter that Globalink will only redeem public shares if after such redemption the net tangible assets of the Corporation as a public company will be at least $5,000,001 upon consummation of the initial Business Combination. Scenario 2 includes all adjustments contained in Scenario 1 and presents additional adjustments to reflect the effect of the maximum contractual redemptions. |
Included in the shares outstanding and weighted average shares outstanding as presented in the pro forma combined financial statements are an aggregate of 160,000,000 PubCo ordinary shares to be issued to shareholders of Alps Holdco and an aggregate of 4,020,000 PubCo ordinary shares to be issued to the PIPE Investors.
After the Business Combination, assuming no redemptions of Globalink shares for cash, Globalink’s current public stockholders and Globalink’s right holders will own approximately 0.8% of the outstanding PubCo ordinary shares, the Globalink founders will own approximately 1.7% of the outstanding PubCo ordinary shares, PGM, an affiliate of the Sponsor, will own approximately 0.4%, the PIPE Investors will own approximately 2.4%, assuming the PIPE Investors will hold 4,020,000 PubCo ordinary shares, IBDC Asia Sdn. Bhd., an advisory firm to Alps Holdco, will own approximately 0.9%, representing finder’s fees payable in PubCo ordinary shares with value equal to 1% of the aggregate consideration for the Business Combination of US$1.6 billion, and the former shareholders of Alps Holdco will own approximately 93.8% of the outstanding PubCo ordinary shares. Assuming maximum contractual redemption by holders of 277,511 Globalink shares, Globalink’s public stockholders and Globalink’s right holders will own approximately 0.7% of the outstanding PubCo ordinary shares, Globalink founders will own approximately 1.7% of the outstanding PubCo ordinary shares, PGM will own approximately 0.4%, the PIPE investors will own approximately 2.4%, assuming the PIPE Investors will hold 4,020,000 PubCo ordinary shares, IBDC Asia Sdn. Bhd. will own approximately 0.9%, and the former shareholders of Alps Holdco will own approximately 93.9% of the outstanding PubCo ordinary shares (in each case, not giving effect to any shares issuable upon the exercise or conversion of options or warrants).
The foregoing scenarios are for Illustrative purposes only as the actual number of redemptions by Globalink’s public stockholders is not able to be known prior to the Globalink stockholder vote with respect to the Business Combination.
The following presents the calculation of basic and diluted weighted average shares outstanding.
| Scenario
1 Combined (Assuming No Redemptions Into Cash) | Scenario
2 Combined (Assuming Maximum Contractual Redemptions Into Cash) | |||||||
| Weighted average shares calculation, basic and diluted | ||||||||
| Globalink public shares and rights | 1,427,511 | 1,150,000 | ||||||
| Globalink Founder and director shares | 2,875,000 | 2,875,000 | ||||||
| Private shares and rights owned by PGM | 627,000 | 627,000 | ||||||
| PIPE Investors | 4,020,000 | 4,020,000 | ||||||
| IBDC Asia Sdn. Bhd. | 1,600,000 | 1,600,000 | ||||||
| Alps Holdco Shareholders | 160,000,000 | 160,000,000 | ||||||
| Weighted average shares outstanding | 170,549,511 | 170,272,000 | ||||||
| Percent of shares owned by Alps Holdco Shareholders | 93.8 | % | 93.9 | % | ||||
| Percent of shares owned by IBDC Asia Sdn. Bhd. | 0.9 | % | 0.9 | % | ||||
| Percent of shares owned by PIPE Investors | 2.4 | % | 2.4 | % | ||||
| Percent of shares owned by Globalink public holders | 0.8 | % | 0.7 | % | ||||
| Percent of shares owned by Globalink Founders and directors | 1.7 | % | 1.7 | % | ||||
| Percent of shares owned by PGM | 0.4 | % | 0.4 | % | ||||
| 100.0 | % | 100.0 | % | |||||
| 243 |
UNAUDITED PRO FORMA CONDENSED COMBINED BALANCE SHEET
AS OF MARCH 31, 2024
| Scenario 1: No Additional Redemption Scenario | Scenario 2: Maximum Contractual Redemption Scenario | |||||||||||||||||||||||||||||||||||
| PubCo
(IFRS Historical) |
ALPS (IFRS |
Globalink (US GAAP Historical) |
IFRS
Conversion and Presentation Alignment (Note 4) |
Transaction Accounting Adjustments | Pro Forma Combined | Transaction Accounting Adjustments | Pro Forma Combined | |||||||||||||||||||||||||||||
| ASSETS | ||||||||||||||||||||||||||||||||||||
| Non-current assets | ||||||||||||||||||||||||||||||||||||
| Property, plant and equipment | $ | - | $ | 2,591,861 | $ | - | $ | - | $ | - | $ | 2,591,861 | $ | - | $ | 2,591,861 | ||||||||||||||||||||
| Right-of-use assets | - | 480,123 | - | - | - | 480,123 | - | 480,123 | ||||||||||||||||||||||||||||
| Intangible assets | - | 837,803 | - | - | - | 837,803 | - | 837,803 | ||||||||||||||||||||||||||||
| Investment in associates | - | 1,506,644 | - | - | - | 1,506,644 | - | 1,506,644 | ||||||||||||||||||||||||||||
| Cash held in Trust Account | - | - | 29,047,273 | - | 1,458,992 | I | - | - | - | |||||||||||||||||||||||||||
| (3,615,886 | ) | B | ||||||||||||||||||||||||||||||||||
| (26,890,379 | ) | P | ||||||||||||||||||||||||||||||||||
| Total non-current assets | - | 5,416,431 | 29,047,273 | - | (29,047,273 | ) | 5,416,431 | - | 5,416,431 | |||||||||||||||||||||||||||
| Current assets | ||||||||||||||||||||||||||||||||||||
| Cash and cash equivalents | - | 460,467 | 6,878 | - | 3,325,730 | B | 34,009,592 | (3,325,731 | ) | G | 30,683,861 | |||||||||||||||||||||||||
| (4,025,000 | ) | A | ||||||||||||||||||||||||||||||||||
| (2,170,834 | ) | C | ||||||||||||||||||||||||||||||||||
| (540,000 | ) | J | ||||||||||||||||||||||||||||||||||
| 40,200,000 | M | |||||||||||||||||||||||||||||||||||
| (3,247,649 | ) | K | ||||||||||||||||||||||||||||||||||
| Inventories | - | 364,137 | - | - | - | 364,137 | - | 364,137 | ||||||||||||||||||||||||||||
| Trade receivables | - | 74,016 | - | - | - | 74,016 | - | 74,016 | ||||||||||||||||||||||||||||
| Other receivables, deposits and prepayments | - | 234,873 | 135,280 | - | (24,500 | ) | C | 345,653 | - | 345,653 | ||||||||||||||||||||||||||
| Amount due from associates | - | 2,145 | - | - | - | 2,145 | - | 2,145 | ||||||||||||||||||||||||||||
| Tax recoverable | - | 148,864 | - | - | - | 148,864 | - | 148,864 | ||||||||||||||||||||||||||||
| Total current assets | - | 1,284,502 | 142,158 | - | 33,517,747 | 34,944,407 | (3,325,731 | ) | 31,618,676 | |||||||||||||||||||||||||||
| Total assets | $ | - | $ | 6,700,933 | $ | 29,189,431 | $ | - | $ | 4,470,474 | $ | 40,360,838 | $ | (3,325,731 | ) | $ | 37,035,107 | |||||||||||||||||||
| EQUITY | ||||||||||||||||||||||||||||||||||||
| ALPS Holdco issued capital | - | 10,464,560 | - | - | (10,464,560 | ) | F | - | - | - | ||||||||||||||||||||||||||
| PubCo ordinary shares | - | - | - | - | 16,000 | F | 17,055 | (28 | ) | G | 17,027 | |||||||||||||||||||||||||
| 28 | G | |||||||||||||||||||||||||||||||||||
| 344 | H | |||||||||||||||||||||||||||||||||||
| 402 | M | |||||||||||||||||||||||||||||||||||
| 121 | L | |||||||||||||||||||||||||||||||||||
| 160 | O | |||||||||||||||||||||||||||||||||||
| Globalink common stock | - | - | 3,445 | - | (3,445 | ) | H | - | - | - | ||||||||||||||||||||||||||
| Additional paid-in capital | - | - | - | 12,113,800 | (36,325 | ) | C | 104,953,194 | (3,325,703 | ) | G | 101,920,027 | ||||||||||||||||||||||||
| 10,448,560 | F | 292,536 | E | |||||||||||||||||||||||||||||||||
| (22,718,489 | ) | D | ||||||||||||||||||||||||||||||||||
| 2,770,074 | G | |||||||||||||||||||||||||||||||||||
| 61,710,697 | E | |||||||||||||||||||||||||||||||||||
| 3,101 | H | |||||||||||||||||||||||||||||||||||
| 40,199,598 | M | |||||||||||||||||||||||||||||||||||
| (121 | ) | L | ||||||||||||||||||||||||||||||||||
| 462,299 | O | |||||||||||||||||||||||||||||||||||
| Accumulated deficit | - | (8,651,666 | ) | (8,098,261 | ) | (12,389,800 | ) | (2,041,509 | ) | C | (89,698,243 | ) | (292,536 | ) | E | (89,990,779 | ) | |||||||||||||||||||
| 22,718,489 | D | |||||||||||||||||||||||||||||||||||
| (61,710,697 | ) | E | ||||||||||||||||||||||||||||||||||
| (540,000 | ) | I | ||||||||||||||||||||||||||||||||||
| (18,522,340 | ) | N | ||||||||||||||||||||||||||||||||||
| (462,459 | ) | O | ||||||||||||||||||||||||||||||||||
| Other comprehensive income (loss) | - | (245,711 | ) | - | - | (245,711 | ) | - | (245,711 | ) | ||||||||||||||||||||||||||
| Non-controlling interest | - | (116,191 | ) | - | - | - | (116,191 | ) | - | (116,191 | ) | |||||||||||||||||||||||||
| Total equity (deficit) | - | 1,450,992 | (8,094,816 | ) | (276,000 | ) | 21,829,928 | 14,910,104 | (3,325,731 | ) | 11,584,373 | |||||||||||||||||||||||||
| Common stock subject to possible redemption | - | - | 28,394,477 | (28,394,477 | ) | - | - | - | - | |||||||||||||||||||||||||||
| LIABILITIES | ||||||||||||||||||||||||||||||||||||
| Non-current liabilities | ||||||||||||||||||||||||||||||||||||
| Lease liabilities | - | 194,362 | - | - | - | 194,362 | - | 194,362 | ||||||||||||||||||||||||||||
| Warrant liability | - | - | 13,680 | 276,000 | - | 289,680 | - | 289,680 | ||||||||||||||||||||||||||||
| Deferred underwiring fee commission | - | - | 4,025,000 | - | (4,025,000 | ) | A | - | - | - | ||||||||||||||||||||||||||
| Common stock subject to possible redemption | - | - | - | 28,394,477 | (2,770,102 | ) | G | - | - | - | ||||||||||||||||||||||||||
| 1,266,004 | I | |||||||||||||||||||||||||||||||||||
| (26,890,379 | ) | P | ||||||||||||||||||||||||||||||||||
| - | ||||||||||||||||||||||||||||||||||||
| Total non-current liabilities | - | 194,362 | 4,038,680 | 28,670,477 | (32,419,477 | ) | 484,042 | - | 484,042 | |||||||||||||||||||||||||||
| Current liabilities | ||||||||||||||||||||||||||||||||||||
| Trade payables | $ | - | $ | 175,599 | $ | - | $ | - | $ | $ | 175,599 | $ | - | $ | 175,599 | |||||||||||||||||||||
| Other payables, accruals and deposits received | - | 956,063 | 15,431 | - | (117,500 | ) | C | 853,994 | - | 853,994 | ||||||||||||||||||||||||||
| Amount due to directors | - | 3,610,711 | - | - | - | 3,610,711 | - | 3,610,711 | ||||||||||||||||||||||||||||
| Hire purchase payables | - | 7,260 | - | - | - | 7,260 | - | 7,260 | ||||||||||||||||||||||||||||
| Lease liabilities | - | 305,946 | - | - | - | 305,946 | - | 305,946 | ||||||||||||||||||||||||||||
| Tax payables | - | - | 602,796 | - | (290,156 | ) | B | 505,628 | - | 505,628 | ||||||||||||||||||||||||||
| 192,988 | I | |||||||||||||||||||||||||||||||||||
| Franchise tax payable | - | - | 50,000 | - | - | 50,000 | - | 50,000 | ||||||||||||||||||||||||||||
| Excise tax liability | - | - | 935,214 | - | - | 935,214 | - | 935,214 | ||||||||||||||||||||||||||||
| Earnout liability | - | - | - | - | 18,522,340 | N | 18,522,340 | - | 18,522,340 | |||||||||||||||||||||||||||
| Promissory note - related party | - | - | 2,640,649 | (2,640,649 | ) | K | - | - | - | |||||||||||||||||||||||||||
| 540,000 | I | |||||||||||||||||||||||||||||||||||
| (540,000 | ) | J | ||||||||||||||||||||||||||||||||||
| Due to related party | - | - | 607,000 | - | (607,000 | ) | K | - | - | - | ||||||||||||||||||||||||||
| Total current liabilities | - | 5,055,579 | 4,851,090 | - | 15,060,023 | 24,966,692 | - | 24,966,692 | ||||||||||||||||||||||||||||
| Total liabilities | - | 5,249,941 | 8,889,770 | 28,670,477 | (17,359,454 | ) | 25,450,734 | - | 25,450,734 | |||||||||||||||||||||||||||
| Total liabilities, common stock subject to redemptions and equity | $ | - | $ | 6,700,933 | $ | 29,189,431 | $ | - | $ | 4,470,474 | $ | 40,360,838 | $ | (3,325,731 | ) | $ | 37,035,107 | |||||||||||||||||||
| 244 |
UNAUDITED PRO FORMA CONDENSED COMBINED STATEMENT OF OPERATIONS
FISCAL YEAR ENDED MARCH 31, 2024
| Scenario 1: No Redemption Scenario | Scenario 2: Maximum Contractual Redemption Scenario | |||||||||||||||||||||||||||||||||||
| PubCo
(IFRS Historical) |
ALPS (IFRS |
Globalink (US GAAP Historical) |
IFRS
Conversion and Presentation Alignment (Note 4) |
Transaction Accounting Adjustments | Pro Forma Combined | Transaction Accounting Adjustments | Pro Forma Combined | |||||||||||||||||||||||||||||
| Revenue | $ | - | $ | 2,403,552 | $ | - | $ | - | $ | - | $ | 2,403,552 | $ | - | $ | 2,403,552 | ||||||||||||||||||||
| Cost of sales | - | (1,804,622 | ) | - | - | - | (1,804,622 | ) | - | (1,804,622 | ) | |||||||||||||||||||||||||
| Gross profit | - | 598,930 | - | - | - | 598,930 | - | 598,930 | ||||||||||||||||||||||||||||
| Selling, general and administrative expenses | - | (1,929,680 | ) | (1,451,420 | ) | - | (61,710,697 | ) | BB | (86,118,105 | ) | (292,536 | ) | BB | (86,410,641 | ) | ||||||||||||||||||||
| (18,522,340 | ) | CC | ||||||||||||||||||||||||||||||||||
| (2,503,968 | ) | DD | ||||||||||||||||||||||||||||||||||
| Distribution expense | - | (160,855 | ) | - | - | - | (160,855 | ) | - | (160,855 | ) | |||||||||||||||||||||||||
| Other operating income | - | 49,977 | - | - | 49,977 | - | 49,977 | |||||||||||||||||||||||||||||
| Other operating expense | - | (917,524 | ) | - | - | - | (917,524 | ) | - | (917,524 | ) | |||||||||||||||||||||||||
| Operating profit | - | (2,359,152 | ) | (1,451,420 | ) | - | (82,737,005 | ) | (86,547,577 | ) | (292,536 | ) | (86,840,113 | ) | ||||||||||||||||||||||
| Penalties on taxes | - | - | (14,244 | ) | - | - | (14,244 | ) | - | (14,244 | ) | |||||||||||||||||||||||||
| Finance expenses | - | (42,844 | ) | (90,650 | ) | - | - | (133,494 | ) | - | (133,494 | ) | ||||||||||||||||||||||||
| Share result of associate | - | 3,421 | - | - | - | 3,421 | - | 3,421 | ||||||||||||||||||||||||||||
| Change in fair value of Common stock subject to possible redemption | - | - | - | (2,707,502 | ) | 2,707,502 | EE | - | - | - | ||||||||||||||||||||||||||
| Change in fair value of warrant liabilities | - | - | (7,980 | ) | (156,400 | ) | - | (164,380 | ) | - | (164,380 | ) | ||||||||||||||||||||||||
| Interest earned on investments held in Trust Account | - | - | 2,175,597 | - | (2,175,597 | ) | AA | - | - | - | ||||||||||||||||||||||||||
| Profit (loss) before income tax | - | (2,398,575 | ) | 611,303 | (2,863,902 | ) | (82,205,100 | ) | (86,856,274 | ) | (292,536 | ) | (87,148,810 | ) | ||||||||||||||||||||||
| Income tax expense | - | 7,414 | (349,225 | ) | - | - | (341,811 | ) | - | (341,811 | ) | |||||||||||||||||||||||||
| Net income (loss) | $ | - | $ | (2,391,161 | ) | $ | 262,078 | $ | (2,863,902 | ) | $ | (82,205,100 | ) | $ | (87,198,085 | ) | $ | (292,536 | ) | $ | (87,490,621 | ) | ||||||||||||||
| Non-controlling interest | - | (87,135 | ) | - | - | - | (87,135 | ) | - | (87,135 | ) | |||||||||||||||||||||||||
| Net income (loss) attributed to shareholders | $ | - | $ | (2,304,026 | ) | $ | 262,078 | $ | (2,863,902 | ) | $ | (82,205,100 | ) | $ | (87,110,950 | ) | $ | (292,536 | ) | $ | (87,403,486 | ) | ||||||||||||||
| Weighted average shares basic and diluted | $ | - | $ | (0.05) | ||||||||||||||||||||||||||||||||
| Basic and diluted net loss per share | $ | - | $ | (0.05) | ||||||||||||||||||||||||||||||||
| Basic and diluted net income per share, redeemable common stock | $ | 0.35 | ||||||||||||||||||||||||||||||||||
| Basic and diluted net loss per share, non-redeemable common stock | $ | (0.33 | ) | |||||||||||||||||||||||||||||||||
| Pro forma weighted average number of shares outstanding - basic and diluted | 170,549,511 | 170,272,000 | ||||||||||||||||||||||||||||||||||
| Pro forma loss per share - basic and diluted | $ | (0.51 | ) | $ | (0.51 | ) | ||||||||||||||||||||||||||||||
| 245 |
NOTES TO UNAUDITED PRO FORMA CONDENSED COMBINED FINANCIAL STATEMENTS
Note 1 — Description of the Business Combination
On January 30, 2024, Globalink entered into the Merger Agreement with Alps Holdco, the Sponsor, and the Seller Representative. The Merger Agreement was amended and restated on May 20, 2024 and entered into by and among Globalink, Alps Holdco, Merger Sub, the Sponsor, and the Seller Representative. Pursuant to the terms of the Merger Agreement, the Business Combination between Globalink and Alps Holdco will be effected in two steps: (i) the Redomestication Merger, whereby, subject to the approval and adoption of the Merger Agreement by the stockholders of Globalink, Globalink will merge with and into PubCo, with PubCo remaining as the surviving publicly traded entity; and (ii) the Acquisition Merger, whereby Merger Sub will merge with and into Alps Holco, resulting in Alps Holdco remaining as the surviving entity and being a wholly-owned subsidiary of PubCo. Subject to the terms and conditions set forth therein upon the Closing, each Alps Holdco Ordinary Share issued and outstanding immediately prior to the Effective Time (other than treasury shares or dissenting shares) will be ultimately converted into the right to receive PubCo ordinary shares. The total consideration to be paid by Globalink to the Alps Holdco Shareholders in the form of PubCo ordinary shares at the Closing will be equal to $1.6 billion, with an earnout provision permitting the shareholders of Alps Holdco to receive up to 48 million additional shares as and when the business meets certain incremental milestones relating to the consolidated revenue of the Combined Company for five fiscal years following the Closing.
The Merger Agreement is subject to certain customary closing conditions and contains customary representations, warranties, covenants and indemnity provisions. Capitalized terms used but not defined herein shall have the respective meanings set forth in the Merger Agreement. The respective boards of directors of Globalink and Alps Holdco have (i) approved and declared advisable the Merger Agreement, the Merger and the transactions contemplated by the Merger Agreement and (ii) resolved to recommend approval of the Merger Agreement and related transactions by their respective shareholders.
As a result of and upon the Closing, pursuant to the terms of the Merger Agreement, all of the outstanding shares of Alps Holdco will be cancelled in exchange for the right to receive PubCo ordinary shares. The aggregate consideration for the Business Combination is $1.6 billion, payable at the Closing in the form of newly issued PubCo ordinary shares, par value $0.0001 per share. The Merger Consideration Shares will be allocated pro rata with each Alps Holdco Shareholder receiving a number of PubCo ordinary shares determined in accordance with the terms of the Merger Agreement. Moreover, Alps Holdco Shareholders will be eligible to receive up to an aggregate of 48,000,000 additional PubCo ordinary shares (the “Earnout Shares”) upon the completion of certain milestones relating to the consolidated revenue of the Combined Company for five fiscal years following the Closing.
Following the execution of the Merger Agreement, PubCo, Globalink and Alps Holdco entered into the Subscription Agreements with certain investors for 4,020,000 PubCo ordinary shares for a total of $40,200,000 in a PIPE Investment to be consummated simultaneously with the Closing. Consummation of the PIPE Investment is conditioned on the concurrent Closing of the Business Combination and other customary closing conditions.
| 246 |
At the Closing of the Business Combination, the former Alps Holdco Shareholders will receive an aggregate of 160,000,000 PubCo ordinary shares, among which 8,000,000 PubCo ordinary shares are to be issued and held in escrow to satisfy any indemnification obligations incurred under the Merger Agreement.
Note 2 — Basis of Presentation
The adjustments presented on the pro forma combined financial statements have been identified and presented to provide an understanding of PubCo upon consummation of the Business Combination for illustrative purposes.
The following unaudited pro forma condensed combined financial information has been prepared in accordance with Article 11 of Regulation S-X as amended by the final rule, Release No. 33-10786 “Amendments to Financial Disclosures about Acquired and Disposed Businesses.” Release No. 33-10786 replaces the existing pro forma adjustment criteria with simplified requirements to depict the accounting for the transaction (“Transaction Accounting Adjustments”) and present the reasonably estimable synergies and other transaction effects that have occurred or are reasonably expected to occur (“Management’s Adjustments”). PubCo has elected not to present Management’s Adjustments and will only be presenting Transaction Accounting Adjustments in the following unaudited pro forma condensed combined financial information. The historical financial information has been adjusted to reflect the pro forma adjustments that are directly attributable to the Business Combination and the PIPE financing as described below.
The unaudited pro forma condensed combined financial information has been prepared assuming two redemption scenarios as follows:
| ● | Assuming No Redemptions: This scenario assumes that no shares of Globalink common stock are redeemed; and | |
| ● | Assuming Maximum Contractual Redemptions: This scenario assumes that 277,511 shares of Globalink common stock, the maximum contractual redemption of the outstanding Globalink common stock, are redeemed for their pro rata share of the cash in the Trust Account. This presentation assumes that Globalink stockholders exercise their redemption rights with respect to a maximum of 277,511 common stock for an aggregate payment of approximately $3.33 million upon consummation of the Business Combination at a redemption price of approximately $11.98 per share. The maximum contractual redemption amount reflects the maximum number of Globalink public shares that can be redeemed without violating the requirement of Globalink Charter that Globalink will only redeem public shares if after such redemption the net tangible assets of the Corporation as a public company will be at least $5,000,001 upon consummation of the initial Business Combination. Scenario 2 includes all adjustments contained in Scenario 1 and presents additional adjustments to reflect the effect of the maximum contractual redemptions. |
Included in the shares outstanding and weighted average shares outstanding as presented in the pro forma combined financial statements are an aggregate of 160,000,000 PubCo ordinary shares to be issued to shareholders of Alps Holdco, 1,600,000 PubCo ordinary shares to be issued to IBDC Asia Sdn. Bhd. as finder fees, and an aggregate of 4,020,000 PubCo ordinary shares to be issued to the PIPE Investors. Each scenario assumes that the parties secure $40.2 million of PIPE financing in order to ensure that there is sufficient funding to meet the $5 million Minimum Net Tangible Asset Condition. The standalone requirement of 5,000,001 of Globalink net tangible assets is expected to be waived by the parties. At this time, the parties have received a firm commitment of such funding.
The pro forma adjustments do not have an income tax effect as they are either (i) incurred by legal entities that are not subject to a corporate income tax, or (ii) permanently nondeductible or nontaxable based on the laws of the relevant jurisdiction.
| 247 |
Note 3 — Accounting for the Business Combination
The Business Combination will be accounted for as a capital reorganization with no goodwill or other intangible assets recorded, in accordance with IFRS. A capital reorganization does not result in a new basis of accounting, and the financial statements of the combined entity represent the continuation of the financial statements of Alps Holdco in many respects. However, Globalink does not meet the definition of a “business” pursuant to IFRS 3 Business Combinations, and thus, for accounting purposes, the Business Combination will be accounted for as a capital reorganization.
Under this method of accounting, Globalink will be treated as the “acquired” company for financial reporting purposes. For accounting purposes, Alps Holdco will be deemed to be the accounting acquirer in the transaction and, consequently, the transaction will be treated as a recapitalization of Alps Holdco (i.e., a capital transaction involving the issuance of shares by PubCo for the shares of Alps Holdco). Accordingly, the consolidated assets, liabilities and results of operations of Alps Holdco will become the historic financial statements of the Combined Company, and Globalink’s assets, liabilities and results of operations will be consolidated with Alps Holdco beginning on the acquisition date. Operations prior to the Business Combination will be presented as those of Alps Holdco in future reports. The net assets of Alps Holdco will be recognized at carrying value, with no goodwill or other intangible assets recorded.
The deemed costs of the shares issued by PubCo, which represents the fair value of the shares that Alps Holdco would have had to issue for the ratio of ownership interest in PubCo to be the same as if the Business Combination had taken the legal form of Alps Holdco acquiring shares of Globalink, in excess of the net assets of Globalink will be accounted for as stock-based compensation under IFRS 2 Share-Based Payment.
Alps Holdco has been determined to be the accounting acquirer based on evaluation of the following facts and circumstances:
| ● | Alps Holdco Shareholders will have the largest voting interest in PubCo under both the no redemption and maximum contractual redemption scenarios; | |
| ● | The board of directors of the Combined Company will be designated solely by Alps Holdco, with at least three (3) directors qualifying as independent directors under the Securities Act and the Nasdaq rules); | |
| ● | Alps Holdco’s senior management will be the senior management of the Combined Company; | |
| ● | The business of PubCo will comprise the ongoing operations of Alps Holdco; and | |
| ● | Alps Holdco is the larger entity, in terms of substantive assets. |
As further consideration in the transaction, Alps Holdco Shareholders will be eligible to receive up to an aggregate of 48,000,000 additional PubCo ordinary shares (the “Earnout Shares”) upon the completion of certain milestones relating to the consolidated revenue of the Combined Company for five fiscal years following the Closing. The Earnout shares are representative of a financial instrument that provides for the issuance of shares if certain revenue milestones are achieved as summarized within section 4.5 of the Merger Agreement, which is based on a contingent event that is outside of the control of the Company and the holder. The settlement of the Earnout provision could result in a variable number of shares being issued upon settlement based on the cumulative nature of the Earnout provision; therefore, the Company has concluded that the Earnout provision is required to be classified and recorded as a financial liability at inception and remeasured at fair value at each reporting period until settlement with the changes in fair value recorded within earnings.
Note 4 — U.S. GAAP to IFRS Conversion and Presentation Alignment
The historical financial information of Globalink has been adjusted to give effect to the differences between U.S. GAAP and IFRS as issued by the IASB for the purposes of the unaudited pro forma condensed combined financial information. One adjustment required to convert Globalink’s consolidated balance sheet from U.S. GAAP to IFRS for purposes of the unaudited pro forma condensed combined financial information was to reclassify Globalink’s Public Warrants and Common stock subject to redemption to non-current financial liabilities under IAS 32, as shareholders have the right to require Globalink to redeem Globalink Public Shares and Warrants and Globalink has an irrevocable obligation to deliver cash or another financial instrument for such redemption.
Further, as part of the preparation of the unaudited pro forma condensed combined financial information, certain reclassifications were made to align Globalink’s consolidated historical financial information in accordance with the presentation of Alps Holdco’s historical financial information, see below for effect of conversion on the financial statements.
| 248 |
Globalink’s Consolidated Balance Sheet as of March 31, 2024
Globalink’s consolidated financial statements have been prepared in accordance with U.S. GAAP and in USD currency and converted to IFRS as follows:
Globalink’s Consolidated Balance Sheet as of March 31, 2024
| US
GAAP March 31, 2024 |
IFRS Adjustments 2024 |
Ref |
IFRS March 31, 2024 |
|||||||||||||
| Assets | ||||||||||||||||
| Non -current assets: | ||||||||||||||||
| Cash held in Trust Account | $ | 29,047,273 | $ | - | $ | 29,047,273 | ||||||||||
| 29,047,273 | - | 29,047,273 | ||||||||||||||
| Current assets | ||||||||||||||||
| Cash | 6,878 | - | 6,878 | |||||||||||||
| Prepaid expense | 135,280 | - | 135,280 | |||||||||||||
| 142,158 | - | 142,158 | ||||||||||||||
| Total assets | $ | 29,189,431 | $ | - | $ | 29,189,431 | ||||||||||
| Liabilities and | ||||||||||||||||
| Stockholders’ Deficit: | ||||||||||||||||
| Common stock | $ | 3,445 | $ | - | $ | 3,445 | ||||||||||
| Additional paid-in-capital | - | 12,113,800 | b | 12,113,800 | ||||||||||||
| Accumulated deficit | (8,098,261 | ) | (12,389,800 | ) | b | (20,488,061 | ) | |||||||||
| Total stockholders’ deficit | (8,094,816 | ) | (276,000 | ) | (8,370,816 | ) | ||||||||||
| Liabilities | ||||||||||||||||
| Non-current liabilities | ||||||||||||||||
| Deferred underwriting commissions | 4,025,000 | - | 4,025,000 | |||||||||||||
| Warrant liabilities | 13,680 | 276,000 | b | 289,680 | ||||||||||||
| Common stock subject to possible redemption | - | 28,394,477 | a | 28,394,477 | ||||||||||||
| 4,038,680 | 28,670,477 | 32,709,157 | ||||||||||||||
| Common stock subject to possible redemption | 28,394,477 | (28,394,477 | ) | a | - | |||||||||||
| Current liabilities | ||||||||||||||||
| Accounts payable | 15,431 | - | 15,431 | |||||||||||||
| Franchise tax payable | 50,000 | - | 50,000 | |||||||||||||
| Income tax payable | 602,796 | - | 602,796 | |||||||||||||
| Promissory Note - Related Party | 2,640,649 | - | 2,640,649 | |||||||||||||
| Due to related party | 607,000 | - | 607,000 | |||||||||||||
| Excise tax liability | 935,214 | - | 935,214 | |||||||||||||
| 4,851,090 | - | 4,851,090 | ||||||||||||||
| Total liabilities | 8,889,770 | 28,670,477 | 37,560,247 | |||||||||||||
| Total Liabilities and Stockholders’ Deficit | $ | 29,189,431 | $ | - | $ | 29,189,431 | ||||||||||
(a) To reclassify and present redeemable common stock of Globalink as other liabilities under IFRS, as shareholders have the right to require Globalink to redeem the Globalink Public Shares and Globalink has an irrevocable obligation to deliver cash or another financial instrument for such redemption.
(b) To reclassify and present the Public Warrants of Globalink as other liabilities under IFRS, as warrant holders have the right to require Globalink to redeem the Globalink Public warrants and Globalink has an irrevocable obligation to deliver cash or another financial instrument for such redemption. Specifically, there are redemption provisions for the warrants whereby they may be redeemed on a cashless basis, in which case the holders will receive a variable number of common stock based on the then market value to result in settlement equivalent to $0.01 per warrant.
| 249 |
U.S. GAAP to IFRS Conversion of Globalink’s Consolidated Statement of Operations for the twelve months ended March 31, 2024
Globalink’s financial statements have been prepared in accordance with U.S. GAAP and in USD currency and is converted to IFRS as follows:
Globalink’s Consolidated Statement of Operations for the trailing twelve months ended March 31, 2024
| US
GAAP March 31, 2024 |
IFRS
Adjustments 2024 |
Ref |
IFRS
March 31, 2024 |
|||||||||||||
| General and administrative expenses | $ | (1,323,802 | ) | - | $ | (1,323,802 | ) | |||||||||
| Franchise tax expense | (127,618 | ) | - | (127,618 | ) | |||||||||||
| Total operating expenses | (1,451,420 | ) | - | (1,451,420 | ) | |||||||||||
| Operating loss | (1,451,420 | ) | - | (1,451,420 | ) | |||||||||||
| Non-operating income (expenses) | ||||||||||||||||
| Interest earned on cash and investments held in Trust Account | 2,175,597 | - | 2,175,597 | |||||||||||||
| Penalties on income tax | (14,244 | ) | - | (14,244 | ) | |||||||||||
| Interest expense | (90,650 | ) | - | (90,650 | ) | |||||||||||
| Change in fair value of warrants liabilities | (7,980 | ) | (156,400 | ) | a | (164,380 | ) | |||||||||
| Change in fair value of common stock | - | (2,707,502 | ) | a | (2,707,502 | ) | ||||||||||
| Total non-operating income (expenses) | 2,062,723 | (2,863,902 | ) | (801,179 | ) | |||||||||||
| Profit/(loss) before income tax | 611,303 | (2,863,902 | ) | (2,252,599 | ) | |||||||||||
| Income tax expenses | (349,225 | ) | - | (349,225 | ) | |||||||||||
| Net profit/(loss) | $ | 262,078 | $ | (2,863,902 | ) | $ | (2,601,824 | ) | ||||||||
(a) To recognize the changes in fair value of the warrant liability and common stock subject to redemption for the twelve months ended March 31, 2024, under IAS 32 changes in fair value through profit or loss.
| 250 |
Note 5 — Adjustments to Unaudited Pro Forma
Condensed Combined Balance Sheet as of March 31, 2024
The pro forma notes and adjustments, based on preliminary estimates that could change materially as additional information is obtained, are as follows:
| A. | Reflects the payment of deferred underwriting commissions upon the closing of the Business Combination. | |
| B. | Reflects the liquidation and reclassification of $3.33 million of funds held in the Trust Account to cash and bank balances that becomes available following the Business Combination and the payment of $0.29 million in income taxes paid subsequent to March 31, 2024. | |
| C. | Represents preliminary estimated transaction costs expected to be paid by Globalink and Alps Holdco of approximately $2.17 million for legal, accounting, underwriting, due diligence and printing fees incurred as part of the Business Combination. For the Globalink transaction costs of $1.69 million is reflected as an adjustment to accumulated deficit and $0.02 million included in prepaid expense are expensed at closing. For the Alps Holdco transaction costs of $0.50 million of which $0.04 million are allocated to new share issuance and recorded to additional paid-in capital and $0.35 million is related to listing cost and recorded to accumulated deficit and $0.12 million is a reduction of accrued transaction cost included in March 31, 2024 balance sheet. | |
| D. | Represents the elimination of Globalink’s historical accumulated deficit after recording the transaction costs to be incurred by Globalink as described in (C) above and the recording of the public warrants and Common stock subject to redemption as liabilities described in IFRS note 4 above. | |
| E. | In Scenario 1, represents the preliminary estimated expense recognized, in accordance with IFRS 2, for the excess of the deemed costs of the shares issued by PubCo and the fair value of Globalink’s identifiable net assets at the date of the Business Combination, resulting in a $61.71 million increase to accumulated deficit. In Scenario 2, represents the preliminary estimated expense recognized, in accordance with IFRS 2, for the excess of the deemed costs of the shares issued by PubCo and the carrying value of GLLI’s identifiable net assets at the date of the Business Combination, resulting in a $61.70 million increase to accumulated deficit. The fair value of shares issued was estimated based on the market price of Globalink common stock of $10.93 per share (as of March 28, 2024). The value is preliminary and will change based on fluctuations in the share price of the GLLI common stock through the closing date. In Scenario 1, one percent change in the market price per share would result in a change of $0.54 million in the estimated expense. In Scenario 2, one percent change in the market price per share would result in a change of $0.51 million in the estimated expense. |
| No Redemption Scenario | Maximum Contractual Redemption Scenario | |||||||||||||||
| Shares | Dollars | Shares | Dollars | |||||||||||||
| Globalink shareholders | ||||||||||||||||
| Public Shareholders | 1,427,511 | 1,150,000 | ||||||||||||||
| Sponsor and other shareholders | 3,502,000 | 3,502,000 | ||||||||||||||
| Fair value of shares to be issued to Globalink shareholders at $10.93 per share | $ | 53,879,555 | $ | 50,846,360 | ||||||||||||
| IFRS Net assets of Globalink as of March 31, 2024 | (8,370,816 | ) | (8,370,816 | ) | ||||||||||||
| Less: Globalink transaction costs, net | (1,690,428 | ) | (1,690,428 | ) | ||||||||||||
| Less: Extension financing | (540,000 | ) | (540,000 | ) | ||||||||||||
| Add: Release of redeemable Common Stock | 2,770,102 | 2,770,102 | ||||||||||||||
| Less: Effect of maximum contractual redemption of Globalink shares | - | (3,325,731 | ) | |||||||||||||
| Adjusted net assets of Globalink as of March 31, 2024 | (7,831,142 | ) | (11,156,873 | ) | ||||||||||||
| Difference - being IFRS 2 charge for listing services | $ | 61,710,697 | $ | 62,003,233 | ||||||||||||
| 251 |
| F. | Represents the exchange of outstanding shares into 160,000,000 PubCo ordinary shares at par value of $0.0001 per share upon the Business Combination. | |
| G. | In Scenario 1, reflects the no additional redemption scenario. In Scenario 2, reflects the maximum contractual redemption of Globalink’s shares for aggregate redemption payments of $3.33 million at a redemption price of approximately $11.98 per share. The Business Combination Agreement provides that consummating the Business Combination is conditioned on Globalink having a minimum of $5.0 million USD of net tangible assets after giving effect to share redemptions and payment of SPAC transaction expenses. Should the NTA condition not be met, Globalink would not be permitted to proceed with the Business Combination. The standalone requirement of 5,000,001 of Globalink net tangible assets is expected to be waived by the parties. | |
| H. | Reflects the conversion of Common Stock into PubCo ordinary shares on a one-for-one basis and reflect the issuance shares in exchange for the rights. | |
| I. | Reflects the additional borrowings subsequent to March 31, 2024 in a form of a Promissory Note in order to fund the extension payments into the Trust account and the interest earned in trust through August 31, 2024 the date of the latest extension deposit, net of tax effect. | |
| J. | Reflects the repayment of additional borrowings for the extension note to be paid at the closing of the business combination. | |
| K. | Reflects the repayment of the Sponsor loans of $2.64 million and Due to related party of $0.61 million. | |
| L. | Reflects the conversion of Globalink’s Public and Private Rights into 1,207,000 PubCo ordinary shares upon the Closing of the Business Combination, including 57,000 Private Rights of Globalink held by PGM | |
| M. | Reflects proceeds taking into account the subscription of $40.20 million in PIPE funding at Closing pursuant to the subscription agreements in connection with the PIPE Investment. | |
| N. | To record the earnout liability under IAS 32. The Earnout shares are representative of a financial instrument that provides for the issuance of shares if certain revenue milestones are achieved as summarized within section 4.5 of the Merger Agreement, which is based on a contingent event that is outside of the control of the Company and the holder. The settlement of the Earnout provision could result in a variable number of shares being issued upon settlement based on the cumulative nature of the Earnout provision; therefore, the Company has concluded that the Earnout provision is required to be classified and recorded as a financial liability at inception and remeasured at fair value at each reporting period until settlement with the changes in fair value recorded within earnings. |
The Earnout was valued with a Monte Carlo Model simulation utilizing the following assumptions: Valuation Date of March 31, 2024, Base Annual Revenue $2.87 million, Industry Revenue Growth 2.6%, Revenue Volatility 119%, GPC Volatility 133%, Risk-Free Rate 4.21%, Term in years 5 years.
The Earnout shares will be issued based on the following milestones:
| (a) | If during the Surviving Company’s first full fiscal year following the Closing Date, the Revenue exceeds $7,000,000, the Alps Holdco Shareholders shall be issued an additional 9,600,000 Reincorporation Merger Surviving Company Ordinary Shares. | |
| (b) | If during the Surviving Company’s second full fiscal year following the Closing Date, the Revenue exceeds $14,000,000, the Alps Holdco Shareholders shall be issued an additional 9,600,000 Reincorporation Merger Surviving Company Ordinary Shares. | |
| (c) | If during the Surviving Company’s third full fiscal year following the Closing Date, the Revenue exceeds $23,000,000, the Alps Holdco Shareholders shall be issued an additional 9,600,000 Reincorporation Merger Surviving Company Ordinary Shares. | |
| (d) | If during the Surviving Company’s fourth full fiscal year following the Closing Date, the Revenue exceeds $34,000,000, the Alps Holdco Shareholders shall be issued an additional 9,600,000 Reincorporation Merger Surviving Company Ordinary Shares. | |
| (e) | If during the Surviving Company’s fifth full fiscal year following the Closing Date, the Revenue exceeds $56,000,000, the Alps Holdco Shareholders shall be issued an additional 9,600,000 Reincorporation Merger Surviving Company Ordinary Shares. | |
| In the event the Surviving Company fails to achieve the Revenue target as set out above for any particular fiscal year (“Relevant Fiscal Year”), the actual Revenue achieved in the Relevant Fiscal Year and the Earn-Out Shares for the Relevant Fiscal year (“Relevant Earn-Out shares”) shall be brought forward to the subsequent fiscal year (“Subsequent Fiscal Year”) |
| O. | Reflects 1% finder fees to IBDC Asia Sdn. Bhd. for introducing Globalink to Alps Holdco for corporate exercise payable in shares and valued at $10 per share. The shares are issued as transaction cost was proportionally allocated against additional paid in capital and accumulated deficit based on the proportional issuance of New shares versus shares issued for listing exchange purposes. | |
P. |
Reflects the redemption in connection with the December 6, 2024 meeting for 2,285,056 shares tendered for redemption resulting redemption of $26,890,379 from the trust at $11.76 per redeemed share. |
Adjustments to Unaudited Pro Forma Condensed Combined Statement of Operations for the Year Ended March 31, 2024
The pro forma notes and adjustments, based on preliminary estimates that could change materially as additional information is obtained, are as follows:
(AA) Reflects the elimination of interest income generated from the cash and investments held in the Trust Account.
(BB) In Scenario 1, represents $61.71 million of expense recognized, in accordance with IFRS 2, for the difference between the deemed costs of the shares issued by PubCo and the carrying value of Globalink’s identifiable net assets, as described in (E) above. In Scenario 2, represents $62.00 million of expense recognized, in accordance with IFRS 2, for the difference between the deemed costs of the shares issued by PubCo and the carrying value of GLLI’s identifiable net assets, as described in (E) above. This cost is a nonrecurring item.
(CC) To record the initial fair value of the earnout liability as described in adjustment N above as part of the deemed cost in connection with the business combination. The fair value of the earnout liability for pro forma purposes was assumed to be the same for at April 1, 2023 and March 31, 2024, as such there was no change in the fair value of the earnout liability recognized. The actual results are subject to change.
(DD) To reflect the incremental transaction cost incurred of $2.50 million. This is a non-recurring item.
(EE) To reflect the reversal of the fair value change of financial liability in connection with Common Stock subject to redemption as the shares at closing are no longer redeemable and thus accounted for as equity instruments.
| 252 |
Note 6 — Net Earnings (Loss) per Share
Represents the earnings (loss) per share calculated using the historical weighted average shares outstanding, and the issuance of additional shares in connection with the Business Combination, assuming the shares were outstanding since April 1, 2023. As the Business Combination is being reflected as if it had occurred at the beginning of the period presented, the calculation of weighted average shares outstanding for basic and diluted earnings (loss) per share assumes that the shares issued in connection with the Business Combination have been outstanding for the entire period presented. If the number of Public Shares described under the Maximum Contractual Redemption Scenario are redeemed, this calculation is retroactively adjusted to eliminate such shares for the entire period.
The unaudited pro forma condensed combined financial information has been prepared assuming two alternative levels of redemption. The following table sets out the share ownership of PubCo following Closing on a pro forma basis under the No Redemption Scenario and the Maximum Contractual Redemption Scenario:
The following table sets out the share ownership of PubCo following Closing on a pro forma basis under the No Redemption Scenario and the Maximum Contractual Redemption Scenario:
| Net loss per share | No Redemption Scenario | Maximum Contractual Redemption Scenario | ||||||
| Net loss | $ | (87,110,950 | ) | $ | (87,403,486 | ) | ||
| Net loss per share – Basic | $ | (0.51 | ) | $ | (0.51 | ) | ||
| Net loss per share - Diluted (1) | $ | (0.51 | ) | $ | (0.51 | ) | ||
| No Redemption Scenario | Maximum Contractual Redemption Scenario | |||||||||||||||||||||||
| Percent | Percent | |||||||||||||||||||||||
| Pro forma Ownership | Number of Shares | Outstanding | Fully diluted | Number of Shares | Outstanding | Fully diluted | ||||||||||||||||||
| Globalink public shares and rights | 1,427,511 | 0.8 | % | 0.6 | % | 1,150,000 | 0.7 | % | 0.6 | % | ||||||||||||||
| Globalink Founder and director shares | 2,875,000 | 1.7 | % | 1.3 | % | 2,875,000 | 1.7 | % | 1.3 | % | ||||||||||||||
| shares and rights held by PGM | 627,000 | 0.4 | % | 0.3 | % | 627,000 | 0.4 | % | 0.3 | % | ||||||||||||||
| PIPE Investors | 4,020,000 | 2.4 | % | 1.8 | % | 4,020,000 | 2.4 | % | 1.8 | % | ||||||||||||||
| IBDC Asia Sdn. Bhd. | 1,600,000 | 0.9 | % | 0.7 | % | 1,600,000 | 0.9 | % | 0.7 | % | ||||||||||||||
| Alps Holdco Shareholders | 160,000,000 | 93.8 | % | 71.2 | % | 160,000,000 | 93.2 | % | 71.3 | % | ||||||||||||||
| Weighted average shares outstanding | 170,549,511 | 100 | % | 170,272,000 | 100 | % | ||||||||||||||||||
| Potential Sources of Dilution(1) | ||||||||||||||||||||||||
| Earnout shares | 48,000,000 | 21.4 | % | 48,000,000 | 21.4 | % | ||||||||||||||||||
| Public Warrants | 5,750,000 | 2.5 | % | 5,750,000 | 2.6 | % | ||||||||||||||||||
| Private Warrants | 285,000 | 0.1 | % | 285,000 | 0.1 | % | ||||||||||||||||||
| Fully diluted weighted average shares outstanding | 224,584,511 | 100 | % | 224,307,000 | 100 | % | ||||||||||||||||||
(1) The potentially dilutive outstanding securities were excluded from the computation of pro forma net loss per share, basic and diluted, because their effect would have been anti-dilutive.
| 253 |
GLOBALINK’S DIRECTORS AND EXECUTIVE OFFICERS
Unless the context otherwise requires, references to the “Company,” “we”, “us” and “our” in this section generally refer to Globalink Investment Inc.
Directors and Executive Officers
Globalink’s directors and executive officers are as follows as of the Record Date:
| Name | Age | Position | ||
| Say Leong Lim | 54 | Chairman of the Board of Directors and Chief Executive Officer | ||
| Kelvin (Zeng Yenn) Chin | 39 | Chief Financial Officer and Director | ||
| Hong Shien Beh | 37 | Independent Director | ||
| Kian Huat Lai | 57 | Independent Director | ||
| Hui Liang Wong | 41 | Independent Director |
Say Leong Lim has served as our Chairman of the Board and Chief Executive Officer since our inception. Mr. Lim has been involved in numerous corporate and operation transactions, amongst other IPOs, RTOs, M&A deals, restructuring and rightsizing, funding, training, management and operational controls in Malaysia, Singapore, Indonesia, Hong Kong, Mainland China and Australia over the last 30 years. Since April 2023, Mr. Lim has served as the chief executive officer of Xtend Digital Sdn. Bhd., a technology company based in Malaysia. Since June 2021, Mr. Lim has served as an independent director at LFE corporation Bhd., an engineering company in Malaysia. Since February 2019, Mr. Lim has served as the Independent Non-executive Director of Aurora Italia International Bhd., a public retail company in Malaysia. In May 2010, Mr. Lim co-founded Everise Concepts PLT and has since served as its advisor. Everise Concepts PLT is principally involved in the provision of corporate and business consultancy, real estate projects and the wholesale and distribution of fast-moving consumer goods via retail and online channels. From November 2020 to April 2022, Mr. Lim served as an independent director of Caely Holdings Bhd. Mr. Lim obtained his Chartered Management Accountant Degree in management accountancy from the Chartered Institute of Management Accountants (CIMA) United Kingdom in 1991 and was admitted as a Malaysian Institute of Accountants (MIA) in 1996. Mr. Lim obtained his Master of Business Administration from Heriot-Watt University in the United Kingdom in 1997.
Kelvin (Zeng Yenn) Chin serves as a member of our Board and our Chief Financial Officer since March 2023. Mr. Chin has over 15 years of audit experience, including auditing of issuers seeking initial public offerings. Since September 2021, Mr. Chin has served as the Financial Controller at PG Mall Sdn. Bhd., an e-commerce marketplace, responsible for overseeing financial related matters of the company. From September 2019 to August 2021, Mr. Chin served as the Disruptive Events Advisory Director at Deloitte Southeast Asia. He was an Operational Director with Herman Corporate Advisory Sdn. Bhd., a Malaysian local boutique advisory firm from December 2017 to August 2019. From May 2016 to December 2017, he was an Audit Senior Manager with Deloitte Southeast Asia after serving as an Assurance Manager in PricewaterhouseCoopers Malaysia from January 2012 to April 2016. Mr. Kelvin (Zeng Yenn) Chin received his bachelor’s degree in business from Victoria University in 2007. He obtained his Certified Public Accountant (“CPA”) status in 2012 from CPA Australia and subsequently admitted as a member of the Malaysian Institute of Accountants in 2013.
Hong Shien Beh serves as an independent member of our Board. Mr. Beh is a legal professional with vast experience in various area of dispute resolution such as defamation, contract, arbitration, construction, planning appeals, commercial and stockholders disputes, industrial accidents, employment, family law, inheritance and estate disputes. Mr. Beh has been a partner at Messrs. Y.C. Wong Advocates & Solicitors since May 2013. From August 2010 to April 2013, Mr. Beh served as an associate at Ismail, Khoo & Associates, a law firm. Mr. Beh received his Bachelor of Laws (LLB) degree from University of Northumbria, Newcastle United Kingdom in 2008.
Hui Liang Wong serves as an independent member of our Board. Ms. Wong has extensive experience in project management. Ms. Wong has served as an executive director at Seedset Sdn. Bhd., a consulting company in Malaysia since May 2018. Ms. Wong has also served as an executive director at Avoras Malaysia Sdn. Bhd., an IT service company since November 2020. Ms. Wong received her Bachelor of Information Technology (Management) degree from University of Malaya, Malaysia in 2006. Ms. Wong received her Foundation Certificate in IT Service Management in August 2007.
| 254 |
Kian Huat Lai serves as an independent member of our Board. Mr. Lai has been serving as an executive director at Ni Hsin Group Berhad (formerly known as Ni Hsin Resource Berhad), a public company in Malaysia since December 2020. From April 2018 to November 2020, Mr. Lai served as a non-independent and non-executive director at Classita Holdings Berhad (formerly known as Caely Holdings Berhad), a public company in Malaysia. From November 2017 to June 2018, Mr. Lai served as an independent and non-executive director at Ta Win Holding Berhad, a public company in Malaysia, and from February 2016 to July 2017, Mr. Lai served as an independent and non-executive director at Widad Group Berhad (formerly Ideal Jacobs (Malaysia) Corporation Berhad), a public company in Malaysia. Mr. Lai studied at Stamford College, Malaysia for General Certificate of education (GCE A Level) from 1985 to 1986. Mr. Lai received his degree in accountancy from the Association of International Accountants, United Kingdom in 1993.
Number, Terms of Office and Appointment of Officers and Directors
Our Board of Directors consists of five members, three of whom are deemed “independent” under SEC rules. Our Board of Directors has been divided into three classes with only one class of directors being elected in each year and each class serving a three-year term. The term of office of the second class of directors, consisting of Hui Liang Wong and Hong Shien Beh, will expire at the second annual meeting held in 2024. The term of office of the third class of directors, consisting of Say Leong Lim and Kelvin (Zeng Yenn) Chin, will expire at our third annual meeting of stockholders held in 2025. The term of office of the first class of directors, consisting of Kian Huat Lai, will expire at our fourth annual meeting of stockholders held in 2026. We may not hold an annual meeting of stockholders until after we consummate our initial business combination.
Our officers are appointed by the Board and serve at the discretion of the Board, rather than for specific terms of office. Our Board is authorized to appoint persons to the offices set forth in our bylaws as it deems appropriate. Our bylaws provide that our directors may consist of a chairman of the Board, and that our officer may consist of chief executive officer, president, chief financial officer, executive vice president(s), vice president(s), secretary, treasurer and such other officers as may be determined by the Board.
Director Independence
An “independent director” is defined generally as a person other than an officer or employee of the company or its subsidiaries or any other individual having a relationship which in the opinion of the company’s Board, would interfere with the director’s exercise of independent judgment in carrying out the responsibilities of a director. Our Board had determined that each of Hong Shien Beh, Hui Liang Wong, and Kian Huat Lai is an “independent director” as defined in applicable SEC rules. Our independent directors have regularly scheduled meetings at which only independent directors are present.
We will only enter into a business combination if it is approved by a majority of our independent directors. Additionally, we will only enter into transactions with our officers and directors and their respective affiliates that are on terms no less favorable to us than could be obtained from independent parties. Any related-party transactions must be approved by our audit committee and a majority of disinterested directors.
Audit Committee
We have established an audit committee of the Board consisting of Hong Shien Beh, Hui Liang Wong, and Kian Huat Lai, each of whom is an independent director. Mr. Kian Huat Lai serves as chairman of the audit committee.
| 255 |
The audit committee’s duties, which are specified in the charter of our audit committee, include, but are not limited to:
| ● | reviewing and discussing with management and the independent registered public accounting firm the annual audited financial statements, and recommending to the Board of Directors whether the audited financial statements should be included in our Form 10-K; | |
| ● | discussing with management and the independent registered public accounting firm significant financial reporting issues and judgments made in connection with the preparation of our financial statements; | |
| ● | discussing with management major risk assessment and risk management policies; | |
| ● | monitoring the independence of the independent registered public accounting firm; | |
| ● | verifying the rotation of the lead (or coordinating) audit partner having primary responsibility for the audit and the audit partner responsible for reviewing the audit as required by law; | |
| ● | reviewing and approving all related-party transactions; | |
| ● | inquiring and discussing with management our compliance with applicable laws and regulations; | |
| ● | pre-approving all audit services and permitted non-audit services to be performed by our independent registered public accounting firm, including the fees and terms of the services to be performed; | |
| ● | appointing or replacing the independent registered public accounting firm; | |
| ● | determining the compensation and oversight of the work of the independent registered public accounting firm (including resolution of disagreements between management and the independent registered public accounting firm regarding financial reporting) for the purpose of preparing or issuing an audit report or related work; | |
| ● | establishing procedures for the receipt, retention and treatment of complaints received by us regarding accounting, internal accounting controls or reports which raise material issues regarding our financial statements or accounting policies; and | |
| ● | approving reimbursement of expenses incurred by our management team in identifying potential target businesses. |
Financial Experts on Audit Committee
The audit committee will at all times be composed exclusively of “independent directors” who are “financially literate” as defined under the Nasdaq listing standards. The Nasdaq listing standards define “financially literate” as being able to read and understand fundamental financial statements, including a company’s balance sheet, income statement and cash flow statement.
In addition, the audit committee has, and will continue to have, at least one member who has past employment experience in finance or accounting, requisite professional certification in accounting, or other comparable experience or background that results in the individual’s financial sophistication. The Board has determined that Mr. Kian Huat Lai qualifies as an “audit committee financial expert,” as defined under rules and regulations of the SEC.
Compensation Committee
We have established a compensation committee of the Board consisting of Hong Shien Beh, Hui Liang Wong, and Kian Huat Lai, each of whom is an independent director. Ms. Hui Liang Wong serves as chairman of the compensation committee.
| 256 |
We adopted a compensation committee charter, which will detail the principal functions of the compensation committee, including:
| ● | reviewing and approving on an annual basis the corporate goals and objectives relevant to our Chief Executive Officer’s compensation, evaluating our Chief Executive Officer’s performance in light of such goals and objectives and determining and approving the remuneration (if any) of our Chief Executive Officer’s based on such evaluation; | |
| ● | reviewing and approving the compensation of all of our other executive officers and reviewing and making recommendations with respect to all non-executive officer compensation; | |
| ● | reviewing our executive compensation policies and plans; | |
| ● | implementing and administering our incentive compensation equity-based remuneration plans; | |
| ● | assisting management in complying with our proxy statement and annual report disclosure requirements; | |
| ● | approving all special perquisites, special cash payments and other special compensation and benefit arrangements for our executive officers and employees; | |
| ● | producing a report on executive compensation to be included in our annual proxy statement; and | |
| ● | reviewing, evaluating and recommending changes, if appropriate, to the remuneration for directors. |
The charter also provides that the compensation committee may, in its sole discretion, retain or obtain the advice of a compensation consultant, legal counsel or other adviser and will be directly responsible for the appointment, compensation and oversight of the work of any such adviser. However, before engaging or receiving advice from a compensation consultant, external legal counsel or any other adviser, the compensation committee will consider the independence of each such adviser, including the factors required by the SEC.
Director Nominations
We do not have a standing nominating committee, though we intend to form a corporate governance and nominating committee as and when required to do so by law. The Board believes that the independent directors can satisfactorily carry out the responsibility of properly selecting or approving director nominees without the formation of a standing nominating committee. Hong Shien Beh, Hui Liang Wong, and Kian Huat Lai will participate in the consideration and recommendation of director nominees. As there is no standing nominating committee, we do not have a nominating committee charter in place.
The Board will also consider director candidates recommended for nomination by our stockholders during such times as they are seeking proposed nominees to stand for election at the next annual meeting of stockholders (or, if applicable, a special meeting). Our stockholders that wish to nominate a director for election to the Board should follow the procedures set forth in our bylaws.
We have not formally established any specific, minimum qualifications that must be met or skills that are necessary for directors to possess. In general, in identifying and evaluating nominees for director, the Board considers educational background, diversity of professional experience, knowledge of our business, integrity, professional reputation, independence, wisdom, and the ability to represent the best interests of our stockholders.
Employment Agreements
Globalink has not entered into any employment agreements with its executive officers, and has not made any agreements to provide benefits upon termination of employment.
| 257 |
Executive Officers and Director Compensation
No executive officer has received any cash compensation for services rendered to us. From the date of our IPO to September 30, 2023, we paid to GL Sponsor LLC, a fee of $10,000 per month for providing us with office space and certain office and secretarial services. On September 30, 2023, Globalink terminated the administrative services agreement. As a result, Globalink is not required to pay the sponsor $10,000 monthly starting from September 30, 2023 However, pursuant to the terms of such agreement, we may delay payment of such monthly fee upon a determination by our audit committee that we lack sufficient funds held outside the trust to pay actual or anticipated expenses in connection with our initial business combination. Any such unpaid amount will accrue without interest and be due and payable no later than the date of the consummation of our initial business combination. Other than the $10,000 per month administrative fee, no compensation or fees of any kind, including finder’s fees, consulting fees and other similar fees, were paid and will be paid to our insiders or any of the members of our management team, for services rendered prior to or in connection with the consummation of our initial business combination (regardless of the type of transaction that it is). However, such individuals will receive reimbursement for any out-of-pocket expenses incurred by them in connection with activities on our behalf, such as identifying potential target businesses, performing business due diligence on suitable target businesses and business combinations as well as traveling to and from the offices, plants or similar locations of prospective target businesses to examine their operations. There is no limit on the amount of out-of-pocket expenses reimbursable by us; provided, however, that to the extent such expenses exceed the available proceeds not deposited in the Trust Account and the interest income earned on the amounts held in the Trust Account, such expenses would not be reimbursed by us unless we consummate an initial business combination.
After our initial business combination, members of our management team who remain with us may be paid consulting, management or other fees from the combined company with any and all amounts being fully disclosed to shareholders, to the extent then known, in the proxy solicitation materials furnished to our shareholders. It is unlikely the amount of such compensation will be known at the time of a shareholder meeting held to consider our initial business combination, as it will be up to the directors of the post-combination business to determine executive and director compensation. In this event, such compensation will be publicly disclosed at the time of its determination in a Current Report on Form 8-K, as required by the SEC.
There are no actual or potential material conflict of interest between Alps Holdco Group’s officers or directors and the unaffiliated security holders of Globalink.
| 258 |
Conflicts of Interest
Our directors and officers have following potential conflicts of interest:
| ● | None of our officers and directors is required to commit their full time to our affairs and, accordingly, they may have conflicts of interest in allocating their time among various business activities. |
| ● | In the course of their other business activities, our officers and directors may become aware of investment and business opportunities which may be appropriate for presentation to our company as well as the other entities with which they are affiliated. Thus, our officers and directors may have conflicts of interest in determining to which entity a particular business opportunity should be presented. |
| ● | Our officers and directors may in the future become affiliated with entities, including other blank check companies, engaged in business activities similar to those intended to be conducted by our company. |
| ● | Unless we consummate our initial business combination, our officers, directors and other insiders will not receive reimbursement for any out-of-pocket expenses incurred by them to the extent that such expenses exceed the amount of available proceeds not deposited in the trust account. |
| ● | The insider shares beneficially owned by our officers and directors will be released from escrow only if our initial business combination is successfully completed. Additionally, if we are unable to complete an initial business combination within the required time frame, our officers and directors will not be entitled to receive any amounts held in the trust account with respect to any of their insider shares or private units. Furthermore, GL Sponsor LLC has agreed that the private units will not be sold or transferred by it until after we have completed our initial business combination. For the foregoing reasons, our Board of Directors may have a conflict of interest in determining whether a particular target business is an appropriate business with which to effect our initial business combination. |
In general, officers and directors of a corporation incorporated under the laws of the State of Delaware are required to present business opportunities to a corporation if:
| ● | the corporation could financially undertake the opportunity; |
| ● | the opportunity is within the corporation’s line of business; and |
| ● | it would not be fair to the corporation and its stockholders for the opportunity not to be brought to the attention of the corporation. |
Accordingly, as a result of multiple business affiliations, our officers and directors may have similar legal obligations relating to presenting business opportunities meeting the above-listed criteria to multiple entities. Furthermore, our amended and restated certificate of incorporation provides that the doctrine of corporate opportunity will not apply with respect to any of our officers or directors in circumstances where the application of the doctrine would conflict with any fiduciary duties or contractual obligations they may have. In order to minimize potential conflicts of interest which may arise from multiple affiliations, our officers and directors (other than our independent directors) have agreed to present to us for our consideration, prior to presentation to any other person or entity, any suitable opportunity to acquire a target business, until the earlier of: (1) our consummation of an initial business combination and (2) January 9, 2025 (and may be extended to June 9, 2025 on a monthly basis by resolutions of the Globalink Board, if requested by the Sponsor, and subject to the Sponsor depositing additional funds into the Trust Account). This agreement is, however, subject to any pre-existing fiduciary and contractual obligations such officer or director may from time to time have to another entity. Accordingly, if any of them becomes aware of a business combination opportunity which is suitable for an entity to which he or she has pre-existing fiduciary or contractual obligations, he or she will honor his or her fiduciary or contractual obligations to present such business combination opportunity to such entity, and only present it to us if such entity rejects the opportunity. We do not believe, however, that the pre-existing fiduciary duties or contractual obligations of our officers and directors will materially undermine our ability to complete our business combination because in most cases the affiliated companies are closely held entities controlled by the officer or director or the nature of the affiliated company’s business is such that it is unlikely that a conflict will arise.
The following table summarizes the current material pre-existing fiduciary or contractual obligations of our officers and directors:
| Individual (1) (2) | Entity | Entity’s Business | Affiliation | |||
| Say Leong Lim | LFE Corporation Bhd. | Engineering | Independent Director | |||
| Aurora Italia International Bhd. | Retail | Independent Director | ||||
| Everise Concepts PLT | Consultancy | Advisor
| ||||
| Kelvin (Zeng Yenn) Chin | PG Mall Sdn Bhd. | e-Commerce marketplace | Financial Controller | |||
| Kian Huat Lai | Ni Hsin Group Berhad | Investment Holding | Executive Director | |||
| Hui Liang Wong | Seedset Sdn Bhd.
|
Consultancy
|
Executive Director | |||
| Avoras Malaysia Sdn Bhd. | IT Services | Executive Director | ||||
| Adansys Consulting Sdn. Bhd. | IT Consulting | Executive Director |
| (1) | Each of the entities listed in this table has priority and preference relative to our company with respect to the performance by each individual listed in this table of his obligations and the presentation by each such individual of business opportunities. |
| (2) | Each individual listed has a fiduciary duty with respect to each of the listed entities opposite from his or her name. |
Our insiders, including our officers and directors, have agreed to vote any shares of common stock held by them in favor of our initial business combination. In addition, to induce Chardan, the representative of the underwriters for Globalink’s IPO to proceed with the IPO, and recognizing the economic benefit the IPO would confer upon the Initial Stockholders and PGM, through the Insider Letter, they have agreed to waive their respective rights to receive any amounts held in the trust account with respect to their insider shares and private shares if we are unable to complete our initial business combination within the required time frame. The Initial Stockholders and PGM do not intend to acquire any shares of Globalink common stock in the open market.
All ongoing and future transactions between us and any of our officers and directors or their respective affiliates will be on terms believed by us to be no less favorable to us than are available from unaffiliated third parties. Such transactions will require prior approval by our audit committee and a majority of our uninterested “independent” directors, or the members of our Board of Directors who do not have an interest in the transaction, in either case who had access, at our expense, to our attorneys or independent legal counsel. We will not enter into any such transaction unless our audit committee and a majority of our disinterested “independent” directors determine that the terms of such transaction are no less favorable to us than those that would be available to us with respect to such a transaction from unaffiliated third parties.
To further minimize conflicts of interest, we have agreed not to consummate our initial business combination with an entity that is affiliated with any of our officers, directors or other insiders, unless we have obtained (i) an opinion from an independent investment banking firm that the business combination is fair to our unaffiliated stockholders from a financial point of view and (ii) the approval of a majority of our disinterested and independent directors (if we have any at that time). In no event will our insiders or any of the members of our management team be paid any finder’s fee, consulting fee or other similar compensation prior to, or for any services they render in order to effectuate, the consummation of our initial business combination (regardless of the type of transaction that it is).
| 259 |
DIRECTORS AND EXECUTIVE OFFICERS OF ALPS HOLDCO GROUP
Unless the context otherwise requires, references to “Alps”,” “we”, “us” and “our” in this section generally refer to the Alps Holdco Group.
Current Directors and Executive Officers
The following tables set forth the names, ages and positions of our directors and executive officers of Alps and Alps Holdco, respectively, as of the effectiveness of this proxy statement/prospectus:
Directors and Executive Officers of Alps Holdco
| Name | Age | Position | ||
| Dr. THAM Seng Kong | 55 | Managing Director; Group Chief Executive Officer; Group Chief Research Scientist | ||
| CHEW Yoke Ling | 53 | Director | ||
| Mohd Razef Bin ABDULLAH | 74 | Director | ||
| Lisa TEOH @ TEOH Lee Eng | 54 | Chief Operating Officer | ||
| LOW Wei Sim | 29 | Deputy Chief Operating Officer | ||
| Professor Manickam RAVICHANDRAN | 57 | Chief Scientific Officer | ||
| Professor POH Chit Laa | 73 | Chief Vaccine Development Officer |
Directors and Executive Officers of Alps Global Holding Berhad
| Name | Age | Position | ||
| Dr. THAM Seng Kong | 55 | Managing Director; Group Chief Executive Officer; Group Chief Research Scientist | ||
| CHEW Yoke Ling | 53 | Director | ||
| Mohd Razef Bin ABDULLAH | 74 | Director | ||
| Lisa TEOH @ TEOH Lee Eng | 54 | Chief Operating Officer | ||
| LOW Wei Sim | 29 | Deputy Chief Operating Officer | ||
| Professor Manickam RAVICHANDRAN | 57 | Chief Scientific Officer | ||
| Professor POH Chit Laa | 73 | Chief Vaccine Development Officer |
Dr. THAM Seng Kong has served as our Managing Director, Group Chief Executive Officer and Group Chief Research Scientist since our inception. Dr. Tham is a veteran in the field of life sciences, with a focus in the field of Cellular Therapy. He is an established member of the Malaysian Society for Stem Cell Research & Therapy (MSCRT) and the Malaysia Association for Cell Therapy (MACT). He has been involved in ongoing extensive research on anti-aging cellular therapy since 2006. Dr. Tham also actively participates in cancer research. Dr. Tham obtained his Bachelor of Medicine from Xiamen University in 1994, and Masters of Business Administration from the Business Institute of Pennsylvania in 2012. Dr. Tham obtained his Ph.D. in Integrated Chinese Medicine & Western Clinical Medicine (Oncology) from Guangzhou University of Chinese Medicine (GUCM) in 2017.
Lisa TEOH @ TEOH Lee Eng (Lisa) has served as our Chief Operating Officer since November 1, 2020. Prior to this role, Ms. Teoh served as the General Manager and was subsequently promoted to Project Director at Sunden Group of Companies from January 2013 to May 2020. The company specialized in delivering complex infrastructure and property projects across the country. Earlier in her career, Ms. Teoh served as Engineer at Putra Group of Companies from September 1997 to January 2013. She earned her Master’s Degree in Engineering Science (Civil) (M.Eng) in 1996 as well as a Master of Philosophy from the University of Oxford, United Kingdom in 1997. Ms. Teoh later obtained her Master of Business Entrepreneur Management in 2018 and Doctor of Business Administration in 2020 from the Institute of Entrepreneurship Malaysia.
Low Wei Sim (Amanda) has been appointed as our Deputy Chief Operating Officer from April 1, 2024. Prior to this role, she held a senior executive position within the same company. Since December 1, 2018, Ms. Amanda has served as a director at Aiiot Technologies Sdn. Bhd., which specializes in information system development and supporting services ancillary to the Internet of Things (IoT). Additionally, she also serves as the executive director in Alps Globemedic Sdn. Bhd., a company dedicated to healthcare business developments, since November 2018. Amanda obtained her Bachelor of Science (BSc) Hons. in Business Studies from Sunway University, Malaysia in 2018.
Professor Manickam RAVICHANDRAN has served as our Chief Scientific Officer since December 1, 2022. Professor Ravichandran serves as the Dean of the Faculty of Applied Sciences at AIMST University, and previously served as the Vice-Chancellor of AIMST University, Malaysia. Professor Ravichandran received his Bachelor of Science in Botany from Madurai Kamaraj University, India in 1988. He obtained his M.Sc. in Medical Microbiology from Christian Medical College, Vellore in 1991, and gained his Ph.D. in Biotechnology from Anna University, Chennai, India in 1977. Professor Ravichandran also contributes to the academic community as an Editorial Board Member of the Tropical Biomedicine Journal, a position he has held since July 7, 2020. Professor Ravichandran’s academic and research excellence has been recognized with 44 national and international awards, including honors from prestigious exhibitions such as IENA in Nuremberg, Germany, and the International Exhibition of Inventions in Geneva. He has also been awarded the Anugerah Inovasi Negara in Malaysia. As an associate member and committee member of ‘Top Research Scientists Malaysia’ at the Academy of Sciences Malaysia.
| 260 |
Professor POH Chit Laa has served as our Chief Vaccine Development Officer since April 1, 2024. Professor Poh has a prestigious career in higher education, focusing on medical microbiology, bacteriology, and biotechnology. She has served as distinguished professor and Head of the Centre for Virus and Vaccine Research at Sunway University from 2011 to 2024. Professor Poh held numerous academic positions, including Professor of Environmental Biotechnology at Swinburne University of Technology (Australia) from 2007 to 2009, and Professor of Biomedical Science at Curtin University of Technology (Australia) from 2005 to 2006. Earlier in her career, Professor Poh was an Associate Professor at the National University of Singapore from 1982 to 2007. Professor Poh obtained a Bachelor of Science (BSc) with First Class Honors in 1975, and a Ph.D. in Medical Microbiology and Bacteriology from Monash University, Australia in 1980. Additionally, Professor Poh contributes to the academic community as a member of the Editorial Committee of the Journal of Bioscience and Bioengineering, published by the Society for Biotechnology in Japan, a role she has held since 2023.
CHEW Yoke Ling has served on our board of directors since its inception. With a background in the beauty industry, Ms. Chew has leveraged her expertise to drive strategic initiatives within our organization. In her role as a marketing director, Ms. Chew oversees sales and marketing activities for Alps’ products and services. Her responsibilities extend to the planning and coordination of Alps medical tourism efforts, catering to both local and international clients. She obtained a Diploma in Marketing from Stamford Group of Colleges, in 1992, and a Diploma in Secretarial Studies from RIMA College, in 1990.
Mohd Razef Bin ABDULLAH has served on our board of directors since December 22, 2020. Datuk Razef has serves as chairman at Concord Link Realty Sdn. Bhd. since 2014, where he is responsible for securing investment for land acquisition and facilitating strategic partnerships. From January 1992 to June 2018, Datuk Razef was an investment director at Teambuild Land Pte Ltd, a property development arm of Teambuild Construction. Datuk Razef also contributed to the business development function on behalf of the Sabah State Government at Green Century Borneo (International) Sdn. Bhd. operating in the medical tourism sector, which involves providing healthcare services to international patients combined with rural-based tourism activities, from 1999 to 2008.
Number, Terms of Office and Appointment of Directors and Officers
Our board of directors consist of five (5) members. Our directors hold office until their successors have been elected and qualified or appointed, or the earlier of their death, resignation or removal. Our officers are elected by and serve at the discretion of the board of directors and the stockholders voting by ordinary resolution.
Scientific Advisory Board
We have established a Scientific Advisory Board (“SAB”) comprising leading experts who offer guidance on our scientific research, development, innovation and technological endeavors. Our SAB members comprised of the following individuals:
Dr. THAM Seng Kong, Bachelor of Medicine from Xiamen University, Doctor of Medicine (MD) Integrated Chinese Medicine & Western Clinical Medicine (Oncology) from Guangzhou University of Chinese Medicine (GUCM).
Professor NORAZMI Mohd Nor, Professor of Molecular Immunology and Dean of the School of Health Sciences, Universiti Sains Malaysia (USM), B.Sc (Hon) from Monash University in Australia, Ph.D. from Flinders University, Australia.
Professor DING KeXiang, professor and chief expert on research topics at the School of Public Health and Tropical Medicine of the Southern Medical University of China, Director of the American Institute of Biomedical Technology in Virginia, USA (ABTRI).
| 261 |
Professor Nik Soriani Yaacob, BSc. (Biochemistry & Pharmacology), Monash University, Melbourne, Australia; MSc. (Biochemical Pharmacology), South Australian Institute of Technology, Adelaide, Australia – now University of South Australia; Ph.D. (Molecular Toxicology), University of Surrey, UK.
Professor Manickam RAVICHANDRAN, Dean of Faculty of Applied Sciences, AIMST University, M.Sc.-Medical Microbiology from Christian Medical College, Vellore, Ph.D. in Biotechnology degree from Anna University, Chennai, INDIA.
Associate Professor Dr. TAN Jun Jie, BSc in Biomedical Science, Universiti Putra Malaysia; DPhil, University of Oxford; Visiting Research Fellow in King’s College London; postdoctoral research fellow at Massachusetts General Hospital, Harvard Medical School.
Professor POH Chit Laa, Ph.D. in Medical Microbiology and Bacteriology from Monash University, Australia in 1980.
Alps Research and Ethics Committee (AREC)
We have established Research and Ethics Committee consisting of the following members:
| No. | Designation | Name | Qualifications | Scientist/ Non-Scientist | Primary Specialty | |||||
| 1. | Chairperson | Dr. Basmullah Yusof | Medical
Degree (USM) MSc. Emergency Response & Planning (UPM) Cert. Diving –Medical Officer (MAFHS) MBA – Occupational Health Doctor (OHD) (CUMS) | Non-Scientist | Medical Health Doctor | |||||
| 2. | Deputy Chairperson | Dr. Benjamin George Jr. | B.BCh.BAO.
(Ireland), LRCP & SI (Ireland), FRCS (Edinburgh), AM (Malaysia), Dip. Psych. (Singapore), LCP. Plastic Surgery (Malaysia), National Specialist Register (Plastic Surgery) | Non-Scientist | Plastic,
Aesthetic, Cosmetic and Reconstructive Surgeon | |||||
| 3. | Legal Representative | Ong Soo Lee | LL.B
(Hons), University of Wolverhampton, UK Barrister at Law, (Lincoln’ Inn, UK) | Non-Scientist | Legal Practitioner | |||||
| 4. | Quality Assurance Personnel | Roshani Vijaya Kumar | BSc. Biomedical Sciences | Non-Scientist | Establish
QMS in compliance with international standard | |||||
| 5. | Secretary | Samantha Lo Yin Teng | BSc.
Biomedical Science, Newcastle MSc. Medical Science (Universiti Kebangsaan Malaysia) | Scientist | Cell culture |
| 262 |
| 6. | Deputy Secretary | Dr. Siti Muneerah Mohd Abd Rahman | Ph.D.
Biochemistry (UKM, Malaysia) MSc. Bioscience – Human Disease and Therapy (University of Leeds, UK) BSc. Biotechnology (IIUM, Malaysia) | Scientist | RNA
Profiling Good Clinical Practice (GCP) | |||||
| 7. | Deputy Secretary | Nur Farhana bt Mohamad Nizam | BSc.
Biomedical Science (Hons) (Universiti Kuala Lumpur) | Scientist | Molecular tests | |||||
| 8. | Subject Matter Expert | Dr. Heilly Chong @ Azlinda Abd. Halik | Ph.D.
Biochemistry/Biotechnology, BSc. Biochemistry, (University Malaya) | Scientist | Cell
Culture Immunology Vaccine development | |||||
| 9. | Subject Matter Expert | Professor Dr. Nik Soriani Yaacob | Sc
Biochemistry & Pharmacology (Monash University, Australia) MSc. Biochemical Pharmacology (South Australian Institute of Technology – now University of South Australia) Ph.D. Molecular Toxicity (University of Surrey, UK) | Scientist | Molecular Cell Biology | |||||
| 10. | Subject Matter Expert | Ong Hock Keat | BSc.
Applied Science (University Science Malaysia) Dip. in Rubber Processing (Rubber Research Institute of Malaysia) | Scientist | Quality
Control, Research & Development | |||||
| 11. | Subject Matter Expert | Zeinabsadat Masoudian | BSc.
General Biology (Azad University, North Tehran, Iran) MSc. Biotechnology (University Malaya) | Scientist | Cell Culture | |||||
| 12. | Subject Matter Expert | Associate Professor Siti Hawa Ali | BSoc.
Scn. Social Development and Administration, M.A (Social Work), Post grad. Certificate of Qualified Social Workers (CQSW, UK) | Non-Scientist | Human
Resource Ethics Committee |
| 263 |
| 13. | Subject Matter Expert | Dr. Amir Hamzah Abdul Latiff | MBBS,
MMed (Paeds), MRCP (Ire), DipRCPath (Immunology), AM, FACAAI, FAAAAI Fellowship in Clinical Immunology and Allergy (Leeds UK) | Non-Scientist | Immunology | |||||
| 14. | Subject Matter Expert | Associate Professor Dr. Najib Majdi Yaacob | Medical
Degree, MSc. Public Health Ph.D. Epidemiology (University Sains Malaysia) | Non-Scientist | Public
Health Medicine (Epidemiology) | |||||
| 15. | Paramedic | Wan Nor Hasmiza Binti Wan Mohd Roslan | Dip.
Nursing (Islamic College of Science and Technology) | Non-Scientist | Nursing | |||||
| 16. | Paramedic | Ain Safira Binti Zaidi | Dip.
Medical Assistant Pusrawi International College of Medical Sciences | Non-Scientist | Nursing | |||||
| 17. | Layperson | Yong Kok Thye | Dip.Computer
Programmer Informatic Computer Centre, Taiping, Perak | Non-Scientist | Software Programmer | |||||
| 18. | Layperson | Santhy
Devi Ayadurai | SPM level | Non-Scientist | Journalism Editor |
Our AREC’s duties, include, but are not limited to:
Ethical oversight for research and clinical trials. Upon the successful registration with the Drug Council Authority (DCA), AREC will provide diligent oversight to ensure that all research and clinical trials undertaken by Alps adhere strictly to established ethical standards. This includes thorough review and approval of research protocols to safeguard the rights and welfare of participants and trial subject, continuous monitoring of ongoing trials to uphold ethical compliance and rigorous evaluation of the risks and benefits associated with the research endeavors.
Enhancement of reputation and trust among stakeholders and clients. Through meticulous ethical oversight and unwavering commitment to integrity, the AREC will serves as a beacon of trust, enhancing Alps’ reputation among its stakeholders. By fostering a culture of ethical conduct, the committee instills confidence in investors, regulatory bodies, and the broader public, thereby bolstering the Alps’ standing in the industry.
Scientific and clinical quality assurance and compliance with regulations. AREC upholds the highest standards of scientific and clinical quality in all research and clinical trials conducted by Alps. This includes scrutinizing the credentials of researchers and clinicians involved in the trials, evaluating data collection and analysis methodologies, and ensuring strict adherence to relevant regulations and guidelines to maintain the integrity of the research.
Identification and mitigation of potential safety and legal risks. AREC proactively identifies potential safety and legal risks associated with research and clinical trials, developing comprehensive strategies to mitigate these risks effectively. This involves the implementation of robust safety monitoring mechanisms, provision of ongoing training on ethical practices, and establishment of policies and procedures to address legal and regulatory requirements, thereby safeguarding Alps’ interests and ensuring compliance with applicable laws.
Improvement of decision-making and transparency among members. AREC fosters a culture of ethical decision-making and transparency within our company, guiding members in navigating complex ethical issues with integrity and clarity. Through open and honest communication, the committee ensures that decisions are made in a fair and transparent manner, promoting a harmonious and ethical work environment.
| 264 |
Clinical Trial Application & Review Process. For each clinical trial conducted by Alps, AREC will conduct an application and review process during the 4 stages:
| Stage 1: Proposal for Clinical Trial |
A comprehensive and meticulously detailed plan for the clinical trial is proposed, including a definitive timeline, allocated responsibilities, and established procedures. This proposal is subject to thorough review and subsequent approval.
|
| Stage 2: Pre-Clinical Documents Submission, Review & Approval |
Prior to the initiation of the clinical phase, approximately 20 types of documents are required to be submitted for comprehensive review and approval, in accordance with Malaysian Good Clinical Practice (GCP) guidelines.
|
| Stage 3: During Clinical Conduct of Trial Document Submission, Review & Approval |
Throughout the clinical trial, continuous monitoring is conducted through the submission and review of approximately 25 types of documents, ensuring adherence to the Malaysian GCP standard.
|
| Stage 4: Post-Clinical Trial Documentation / Upon Trial Termination |
| Upon completion or termination of the trial, a set of approximately 8 types of documents must be compiled and submitted, adhering to the Malaysian GCP requirements. |
Ethics Committee Registered with Drug Control Authority (DCA)
AREC is currently in the process of submitting its application to become a registered ethics committee with the Drug Control Authority (DCA). Achieving registration under DCA will endorse AREC’s capacity to uphold the highest ethical standards in clinical research, aligning with national and international guidelines for the protection of human subjects. Registration will not only validate the committee’s commitment to ethical vigilance but will also facilitate the swift initiation of clinical trials, ensuring regulatory compliance and enhancing the credibility of the research findings. Moreover, it will position AREC as a trusted gatekeeper in the ethical review process of clinical trials conducted in Alps, streamlining Alps;’ trial approval process, potentially accelerating the development pipeline and facilitating access to broader research opportunities and partnerships. This validation by a national regulatory body can also serve as a benchmark of Alps’ commitment to ethical standards, reinforcing its stature in the biotechnology field both domestically and globally.
Family Relationships
There are no family relations among any of our executive officers or directors.
Involvement in Certain Legal Proceedings
No executive officer or director is a party in a legal proceeding adverse to us or any of our subsidiaries or has a material interest adverse to us or any of our subsidiaries.
No executive officer or director has been involved in the last ten years in any of the following:
| ● | Any bankruptcy petition filed by or against any business or property of such person, or of which such person was a general partner or executive officer either at the time of the bankruptcy or within two years prior to that time; | |
| ● | Any conviction in a criminal proceeding or being subject to a pending criminal proceeding (excluding traffic violations and other minor offenses); | |
| ● | Being subject to any order, judgment, or decree, not subsequently reversed, suspended or vacated, of any court of competent jurisdiction, permanently or temporarily enjoining, barring, suspending or otherwise limiting his involvement in any type of business, securities or banking activities; | |
| ● | Being found by a court of competent jurisdiction (in a civil action), the SEC or the Commodity Futures Trading Commission to have violated a federal or state securities or commodities law, and the judgment has not been reversed, suspended, or vacated; | |
| ● | Being the subject of or a party to any judicial or administrative order, judgment, decree or finding, not subsequently reversed, suspended or vacated relating to an alleged violation of any federal or state securities or commodities law or regulation, or any law or regulation respecting financial institutions or insurance companies, including but not limited to, a temporary or permanent injunction, order of disgorgement or restitution, civil money penalty or temporary or permanent cease-and-desist order, or removal or prohibition order, or any law or regulation prohibiting mail, fraud, wire fraud or fraud in connection with any business entity; or | |
| ● | Being the subject of or a party to any sanction or order, not subsequently reversed, suspended or vacated, of any self-regulatory organization (as defined in Section 3(a)(26) of the Exchange Act, any registered entity (as defined in Section 1(a)(29) of the Commodity Exchange Act), or any equivalent exchange, association, entity or organization that has disciplinary authority over its members or persons associated with a member. |
Duties of Directors
Under Malaysia Companies Act 2016, directors of Malaysian companies have both fiduciary duties and statutory duties to act in the company’s best interests. These duties encompass responsibilities such as exercising reasonable care, skill, and diligence in their roles, avoiding conflicts of interest, and acting in good faith for the benefit of the company.
| 265 |
EXECUTIVE COMPENSATION
Globalink
Executive Compensation
Globalink was incorporated on March 24, 2021 and none of Globalink’s executive officers, directors or industry advisors have received any cash compensation for services rendered to Globalink during the years ended December 31, 2022 and 2023, so there are no named executive officers for such years. Globalink also paid the Sponsor or its affiliate a total of $10,000 per month for office space, utilities, secretarial support and administrative services from the date of IPO to September 30, 2023. The Initial Stockholders, and Globalink’s officers, directors, industry advisor and any of their respective affiliates are also being reimbursed for any out-of-pocket expenses incurred in connection with activities on behalf of Globalink such as identifying potential target businesses and performing due diligence on suitable business combinations, including the Business Combination. Globalink’s independent directors review on a quarterly basis all payments that were made, directly or indirectly, to the Sponsor, officers, directors, industry advisors or Globalink’s or any of their affiliates.
Director Compensation
Globalink currently does not provide any compensation to its directors.
Alps Holdco Group
This section provides an overview of Alps Holdco Group’s executive compensation as it relates to the executive officers named below (the “named executive officers”), for the year ended March 31, 2023 and 2024, which consist of Alps Holdco Group’ principal executive officers:
| ● | Dr. THAM Seng Kong, M.D., Ph.D., Alps’ Group Chief Executive Officer and Group Chief Scientific Officer; | |
| ● | Lisa TEOH @ TEOH Lee Eng (Lisa), Alps’ Chief Operating Officer; | |
| ● | CHEW Yoke Ling, Alps’ Marketing Director; and | |
| ● | Professor Manickam RAVICHANDRAN, Alps’ Chief Scientific Officer. |
Summary Compensation Table
The following summary compensation table sets forth the aggregate compensation we paid or accrued during the fiscal years ended March 31, 2023 and 2024, to the named executive officers for services provided to Alps and its subsidiaries.
| Name and Position | Year Ended | Salary (RM) | Allowance (RM) | Exempted Allowance (RM) | Bonus (RM) | All Other Compensations(1) (RM) | Total (RM) | |||||||||||||||||||
| Dr. THAM Seng Kong | 2023 | 540,000.00 (approximately US$114,406.78) | - | - | - | - | 540,000.00 (approximately US$114,406.78) | |||||||||||||||||||
| 2024 | 540,000.00 (approximately US$114,406.78) | - | - | - | - | 540,000.00 (approximately US$114,406.78) | ||||||||||||||||||||
| Group Chief Executive Officer and Group Chief Scientific Officer | ||||||||||||||||||||||||||
| CHEW Yoke Ling | 2023 | 210,000.00
(approximately US$44,491.53) | 84,000.00 (approximately US$17,796.61) | - | - | - | 294,000.00 (approximately US$ 62,288.14) | |||||||||||||||||||
| 2024 | 210,000.00
(approximately US$44,491.53) | 84,000.00 (approximately US$17,796.61) | - | - | - | 294,000.00 (approximately US$62,288.14) | ||||||||||||||||||||
| Marketing Director | ||||||||||||||||||||||||||
| Lisa TEOH @ TEOH Lee Eng | 2023 | 113,000.00 (approximately US$23,940.68) | 22,000.00 (approximately US$4,661.02) | - | - | - | 135,000.00 (approximately US$28,601.69) | |||||||||||||||||||
| 2024 | 156,000.00 (approximately US$33,050.85) | 24,000.00 (approximately US$5,084.75) | 13,000.00 (approximately US$ 2,754.24) | - | - | 193,000.00 (approximately US$40,889.83) | ||||||||||||||||||||
| Chief Operation Officer | ||||||||||||||||||||||||||
| Professor Manickam RAVICHANDRAN | 2023 | 80,000.00 (approximately US$16,949.15) | 20,000.00 (approximately US$4,237.29) | - | - | - | 100,000.00 (approximately US$21,186.44) | |||||||||||||||||||
| 2024 | 240,000.00 (approximately US$50,847.46) | 60,000.00 (approximately US$12,711.86) | - | 5,000.00 (approximately US$ 1,059.32) | - | 305,000.00 (approximately US$64,618.64) | ||||||||||||||||||||
| Chief Scientific Officer | ||||||||||||||||||||||||||
[Exchange Rate: USD 1.00 = MYR 4.72]
(1) The named executive officers are entitled to certain group health and term life insurance, as well as medical reimbursement plans (collectively, “Benefits”), and contributions to the EPF, SOCSO, and EIS, which are not disclosed in the Summary Compensation Table above. The Benefits are generally available to all confirmed, full-time, and contract employees, while statutory contributions apply to all salaried employees.
| 266 |
Narrative Disclosure to Summary Compensation
Material Terms of Executive Officers’ Employment Agreements
Employment Arrangements with Dr. THAM Seng Kong
Dr. Tham entered into letters of appointment (“LOA”) (as amended and supplemented on April 25, 2024) with Alps Global Holding Berhad, TMC Global Holdings Sdn. Bhd. and Celebre Pro Medic Sdn. Bhd., commencing on July 1, 2017, July 4, 2014 and June 12, 2014, respectively. Pursuant to the respective LOA, Dr. Tham receives an aggregate sum of RM45,000.00 per month (approximately US$ 9,406.35) and is entitled to reimbursement for all reasonable and approved out-of-pocket expenses incurred in connection with his employment. The term of the employment continues until either party gives the other party at least two months’ written notice, or payment of salary-in-lieu, and Alps has the right to immediately terminate in the event of default, misconduct or any breach or non-observance by the employee. The LOA contain restrictive covenants governing confidentiality, a non-solicitation and non-competition restriction that applies to employees, client, customers and executives following termination, and confirmation that all intellectual properties developed during the employments belong exclusively to Alps.
Employment Arrangements with Lisa TEOH @ TEOH Lee Eng (Lisa)
Ms. Teoh entered into a LOA (as amended and supplemented on April 25, 2024) with Alps Global Holding Berhad commencing on November 1, 2020 for the appointment to serve as the Chief Operation Officer. Pursuant to the LOA, Ms. Teoh receives a sum of RM13,000 per month (approximately US$2,717.39) and is entitled to reimbursement for all reasonable and approved out-of-pocket expenses incurred in connection with her employment. The term of the employment continues until either party gives the other party at least two months’ written notice, or payment of salary-in-lieu, and Alps has the right to immediately terminate in the event of default, misconduct or any breach or non-observance by the employee. The LOA contain restrictive covenants governing confidentiality, a non-solicitation and non-competition restriction that applies to employees, client, customers and executives following termination, and confirmation that all intellectual properties developed during the employments belong exclusively to Alps.
Employment Arrangements with CHEW Yoke Ling
Ms. Chew entered into letters of appointment (“LOA”) with TMC Global Holdings Sdn. Bhd. and Celebre Pro Medic Sdn. Bhd., commencing on January 1, 2012 and March 1, 2016, respectively. Pursuant to the respective LOA, Ms. Chew receives an aggregate sum of RM17,500.00 per month (approximately US$ 3,715.43) and is entitled to reimbursement for all reasonable and approved out-of-pocket expenses incurred in connection with his employment. The term of the employment continues until either party gives the other party at least two months’ written notice, or payment of salary-in-lieu, and the Company has the right to immediately terminate in the event of default, misconduct or any breach or non-observance by the employee. The LOA contain restrictive covenants governing confidentiality, a non-solicitation and non-competition restriction that applies to employees, client, customers and executives following termination, and confirmation that all intellectual properties developed during the employments belong exclusively to the Company.
| 267 |
Employment Arrangements with Professor Manickam RAVICHANDRAN (Prof. Ravi)
Prof. Ravi entered into a LOA (as amended and supplemented on March 29, 2024) with Alps Global Holding Berhad for a fixed term of two (2) years, commencing on December 1, 2022, and expiring on November 30, 2024, for the appointment to serve as the Chief Scientific Officer. Pursuant to the LOA, Professor Ravi receives a sum of RM20,000.00 per month (approximately US$4,180.60) and is entitled to reimbursement for all reasonable and approved out-of-pocket expenses incurred in connection with his employment. The term of the employment continues until either party gives the other party at least two months’ written notice, or payment of salary-in-lieu, and Alps has the right to immediately terminate in the event of default, misconduct or any breach or non-observance by the employee. The LOA contain restrictive covenants governing confidentiality, a non-solicitation and non-competition restriction that applies to employees, client, customers and executives following termination, and confirmation that all intellectual properties developed during the employments belong exclusively to Alps.
Potential Payments Upon Termination or Change in Control
Dr. THAM Seng Kong
Dr. Tham’s LOA stipulates that if Alps terminates Dr. Tham’s employment at any time with two months’ prior written notice, or if he resigns with two months’ prior written notice, or if his employment terminates due to misconduct, death, or disability, he will receive his accrued salary up to his last day of employment but will not be entitled to any other form of compensation from Alps, including severance benefits. If Alps terminates Dr. Tham’s employment without two months’ prior written notice or if he resigns without two months’ prior written notice, then Alps or Dr. Tham (as the case may be) shall be liable to pay the salary in lieu of notice. Alps is entitled to immediately terminate the employee in the event of default, misconduct or any breach or non-observance of the terms of LOA by the employee. Severance and termination benefits are subject to Malaysian laws and regulations and the payout may be higher than aforementioned amounts.
Lisa TEOH @ TEOH Lee Eng
Ms. Teoh’s LOA stipulates that if Alps terminates Ms. Teoh’s employment at any time with two months’ prior written notice, or if she resigns with two months’ prior written notice, or if her employment terminates due to misconduct, death, or disability, she will receive her accrued salary up to her last day of employment but will not be entitled to any other form of compensation from Alps, including severance benefits. If Alps terminates Ms. Teoh’s employment without two months’ prior written notice or if she resigns without two months’ prior written notice, then Alps or Ms. Teoh (as the case may be) shall be liable to pay the salary in lieu of notice. Alps is entitled to immediately terminate the employee in the event of default, misconduct or any breach or non-observance of the terms of LOA by the employee. Severance and termination benefits are subject to Malaysian laws and regulations and the payout may be higher than aforementioned amounts.
Professor Manickam RAVICHANDRAN (Prof. Ravi)
Prof. Ravi’s LOA stipulates that if Alps terminates Prof. Ravi’s employment at any time with two months’ prior written notice, or if he resigns with two months’ prior written notice, or if his employment terminates due to misconduct, death, or disability, he will receive his accrued salary up to his last day of employment but will not be entitled to any other form of compensation from Alps, including severance benefits. If Alps terminates Professor Ravi’s employment without two months’ prior written notice or if he resigns without two months’ prior written notice, then Alps or Professor Ravi (as the case may be) shall be liable to pay the salary in lieu of notice. Alps is entitled to immediately terminate the employee in the event of default, misconduct or any breach or non-observance of the terms of LOA by the employee. Severance and termination benefits are subject to Malaysian laws and regulations and the payout may be higher than aforementioned amounts.
Other Benefits
Each of Alps’ named executive officers is eligible for reimbursement of travel and entertainment expenses in accordance with the terms and conditions outlined in the LOA.
Equity Awards
Except for securities of Pubco to be issued in connection with the Business Combination, there are no unvested options, warrants or convertible securities outstanding.
At no time during the last fiscal year with respect to any of any of our named executive officers was there:
| ● | any outstanding option or other equity-based award repriced or otherwise materially modified (such as by extension of exercise periods, the change of vesting or forfeiture conditions, the change or elimination of applicable performance criteria, or the change of the bases upon which returns are determined; | |
| ● | any waiver or modification of any specified performance target, goal or condition to payout with respect to any amount included in non-stock incentive plan compensation or payouts; | |
| ● | any option or equity grant; | |
| ● | any non-equity incentive plan award made to a named executive officer; | |
| ● | any nonqualified deferred compensation plans including nonqualified defined contribution plans; or | |
| ● | any payment for any item to be included under All Other Compensation in the Summary Compensation Table. |
| 268 |
Non-employee Director Compensation
The following table reflects the summary compensation for Alps’ non-employee director for fiscal year ended March 31, 2023 and 2024. Directors are entitled to reimbursement for reasonable travel and other out-of-pocket expenses incurred in connection with attendance at meetings of our board of directors.
| Name | Year Ended | Fee (RM) | Allowance (RM) | Exempted Allowance (RM) | Bonus (RM) | Total (RM) | ||||||||||||||||
| Mohd Razef Bin ABDULLAH | 2023 | 1,200.00 (approximately US$254.24) | - | - | - | 1,200.00 (approximately US$254.24) | ||||||||||||||||
| 2024 | 1,200.00 (approximately US$254.24) | - | - | - | 1,200.00 (approximately US$254.24) | |||||||||||||||||
| ONG Hock Keat | 2023 | 1,200.00 (approximately US$254.24) | - | - | - | 1,200.00 (approximately US$254.24) | ||||||||||||||||
| 2024 | 1,200.00 (approximately US$254.24) | - | - | - | 1,200.00 (approximately US$254.24) | |||||||||||||||||
| WONG Sui Ting | 2023 | 1,200.00 (approximately US$254.24) | - | - | - | 1,200.00 (approximately US$254.24) | ||||||||||||||||
| 2024 | 1,200.00 (approximately US$254.24) | - | - | - | 1,200.00 (approximately US$254.24) | |||||||||||||||||
[Exchange Rate: USD 1.00 = MYR 4.72]
Post-Merger Executive and Director Compensation
Following the completion of the Business Combination, the compensation arrangements for all directors and executive officers of the Combined Company shall initially remain unchanged. As its business develops and matures, the Combined Company intends to develop an executive compensation program that is designed to align compensation with business objectives and the creation of stockholder value, while enabling the Combined Company to attract, retain, incentivize and reward individuals who contribute to its long-term success. Decisions regarding the executive compensation program will be made by the compensation committee of the Combined Company’s board of directors.
Post-Closing Employment Agreements
Simultaneously with the Closing, the Combined Company intends to enter into an executive employment agreement, the form of which is attached hereto as Exhibit 10.34, with Dr. THAM Seng Kong as Chief Executive Officer, which shall supersede any and all prior employment contracts entered into between Alps Holdco Group and Dr. Tham. The executive employment agreement with Dr. Tham provides that Dr. Tham will hold the position of Chief Executive Officer of the Combined Company with a base annual salary of US$60,000.00. Under the agreement, Dr. Tham’s employment is at will and will continue until either Dr. Tham or the Combined Company notifies the other party at least sixty (60) days written notice of intent to terminate employment. If, Dr. Tham’s employment is terminated by the Combined Company without “cause” or by Dr. Tham for Just Cause, he will be entitled to receive (i) accrued but unpaid base salary through the termination date; (ii) reimbursement for any unreimbursed pre-approved reasonable business expenses incurred through the termination date; (iii) accrued but unused annual leave days; (iv) all other payments or benefits to which he shall be entitled as of the termination date under the terms of any applicable compensation arrangement or benefit, equity, or fringe benefit plan or program or grant; and (v) in lieu of benefits under any severance plan or policy of the Combined Company, any such amount as may be agreed between the Combined Company and Dr. Tham.
Simultaneously with the Closing, the Combined Company intends to enter into an executive employment agreement, the form of which is attached hereto as Exhibit 10.34, with Ms. CHEW Yoke Ling as Marketing Director, which shall supersede any and all prior employment contracts entered into between Alps Holdco Group and Ms. Chew. The executive employment agreement with Ms. Chew provides that Ms. Chew will hold the position of Marketing Director of the Combined Company with a base annual salary of US$60,000.00. Under the agreement, Ms. Chew’s employment is at will and will continue until either Ms. Chew or the Combined Company notifies the other party at least sixty (60) days written notice of intent to terminate employment. If, Ms. Chew’s employment is terminated by the Combined Company without “cause” or by Ms. Chew for Just Cause, she will be entitled to receive (i) accrued but unpaid base salary through the termination date; (ii) reimbursement for any unreimbursed pre-approved reasonable business expenses incurred through the termination date; (iii) accrued but unused annual leave days; (iv) all other payments or benefits to which she shall be entitled as of the termination date under the terms of any applicable compensation arrangement or benefit, equity, or fringe benefit plan or program or grant; and (v) in lieu of benefits under any severance plan or policy of the Combined Company, any such amount as may be agreed between the Combined Company and Ms. Chew.
| 269 |
Simultaneously with the Closing, the Combined Company intends to enter into an executive employment agreement, the form of which is attached hereto as Exhibit 10.34, with Ms. LOW Wei Sim (Amanda) as Chief Operating Officer, which shall supersede any and all prior employment contracts entered into between Alps Holdco Group and Ms. Amanda. The executive employment agreement with Ms. Amanda provides that Ms. Amanda will hold the position of Chief Operating Officer of the Combined Company with a base annual salary of US$60,000.00. Under the agreement, Ms. Amanda’s employment is at will and will continue until either Ms. Amanda or the Combined Company notifies the other party at least sixty (60) days written notice of intent to terminate employment. If, Ms. Amanda’s employment is terminated by the Combined Company without “cause” or by Ms. Amanda for Just Cause, she will be entitled to receive (i) accrued but unpaid base salary through the termination date; (ii) reimbursement for any unreimbursed pre-approved reasonable business expenses incurred through the termination date; (iii) accrued but unused annual leave days; (iv) all other payments or benefits to which she shall be entitled as of the termination date under the terms of any applicable compensation arrangement or benefit, equity, or fringe benefit plan or program or grant; and (v) in lieu of benefits under any severance plan or policy of the Combined Company, any such amount as may be agreed between the Combined Company and Ms. Amanda..
Simultaneously with the Closing, the Combined Company intends to enter into an executive employment agreement, the form of which is attached hereto as Exhibit 10.34, with Professor Manickam RAVICHANDRAN (Prof. Ravi) as Chief Scientific Officer, which shall supersede any and all prior employment contracts entered into between Alps Holdco Group and Prof. Ravi. The executive employment agreement with Prof. Ravi provides that Prof. Ravi will hold the position of Chief Scientific Officer of the Combined Company with a base annual salary of US$60,000.00. Under the agreement, Prof. Ravi’s employment is at will and will continue until either Prof. Ravi or the Combined Company notifies the other party at least sixty (60) days written notice of intent to terminate employment. If, Prof. Ravi’s employment is terminated by the Combined Company without “cause” or by Prof. Ravi for Just Cause, he will be entitled to receive (i) accrued but unpaid base salary through the termination date; (ii) reimbursement for any unreimbursed pre-approved reasonable business expenses incurred through the termination date; (iii) accrued but unused annual leave days; (iv) all other payments or benefits to which he shall be entitled as of the termination date under the terms of any applicable compensation arrangement or benefit, equity, or fringe benefit plan or program or grant; and (v) in lieu of benefits under any severance plan or policy of the Combined Company, any such amount as may be agreed between the Combined Company and Prof. Ravi.
Simultaneously with the Closing, the Combined Company intends to enter into an executive employment agreement, the form of which is attached hereto as Exhibit 10.34, with Professor POH Chit Laa (Prof. Poh) as Chief Vaccine Development Officer, which shall supersede any and all prior employment contracts entered into between Alps Holdco Group and Prof. Poh. The executive employment agreement with Prof. Ravi provides that Prof. Poh will hold the position of Chief Vaccine Development Officer of the Combined Company with a base annual salary of US$60,000.00. Under the agreement, Prof. Poh’s employment is at will and will continue until either Prof. Poh or the Combined Company notifies the other party at least sixty (60) days written notice of intent to terminate employment. If, Prof. Poh’s employment is terminated by the Combined Company without “cause” or by Prof. Poh for Just Cause, she will be entitled to receive (i) accrued but unpaid base salary through the termination date; (ii) reimbursement for any unreimbursed pre-approved reasonable business expenses incurred through the termination date; (iii) accrued but unused annual leave days; (iv) all other payments or benefits to which she shall be entitled as of the termination date under the terms of any applicable compensation arrangement or benefit, equity, or fringe benefit plan or program or grant; and (v) in lieu of benefits under any severance plan or policy of the Combined Company, any such amount as may be agreed between the Combined Company and Prof. Poh.
“Cause” is defined in the executive employment agreement as (i) a breach by the executive of his or her fiduciary duties to the Combined Company; (ii) the executive’s breach of the executive employment agreement, which, if curable, remains uncured or continues after ten days’ notice by the Combined Company thereof; (iii) the commission of (A) any crime constituting a felony in the jurisdiction in which committed, (B) any crime involving moral turpitude (whether or not a felony), or (C) any other criminal act involving embezzlement, misappropriation of money, fraud, theft, or bribery (whether or not a felony); (iv) illegal or controlled substance abuse or insobriety by the executive; (v) the executive’s material negligence or dereliction in the performance of, or failure to perform the executive’s duties of employment with the Combined Company, which remains uncured or continues after ten days’ notice by the Combined Company thereof; (vi) the executive’s refusal or failure to carry out a lawful directive of the Combined Company or any member of the board or any of their respective designees, which directive is consistent with the scope and nature of the executive’s responsibilities; or (vii) any conduct, action or behavior by the executive that is, or is reasonably expected to be, materially damaging to the Combined Company, whether to the business interests, finance or reputation. In addition, Further, the executive’s employment shall be deemed to have terminated for Cause if, on the date the executive’s employment terminates, facts and circumstances exist that would have justified a termination for Cause, even if such facts and circumstances are discovered after such termination.
“Just Cause” is defined in the executive employment agreement as a material breach by the Combined Company of its obligations under the executive employment agreement, upon which the executive notifies the board of directors in writing of such material breach within thirty (30) days of such occurrence and such material breach shall have not been cured within thirty (30) days after the Board’s receipt of written notice thereof from the executive.
| 270 |
DIRECTORS AND EXECUTIVE OFFICERS OF THE COMBINED COMPANY
The following table set forth the names, ages and positions of the directors and executive officers of the Combined Company upon Closing:
Combined Company
| Name | Age | Position | ||
| Dr. THAM Seng Kong | 55 | Managing Director; Chief Executive Officer | ||
| CHEW Yoke Ling | 53 | Director | ||
| LOW Wei Sim | 29 | Chief Operating Officer | ||
| Professor Manickam RAVICHANDRAN | 57 | Chief Scientific Officer | ||
| Professor POH Chit Laa | 73 | Chief Vaccine Development Officer | ||
| Tan Sri Dato’ Seri Dr. Suleiman bin Mohamed | 76 | Independent Director | ||
| CHUA Boon Ping | 40 | Independent Director | ||
| Intan Ilyani binti Ghazali | 45 | Independent Director |
Alps Holdco
| Name | Age | Position | ||
| Dr. THAM Seng Kong | 55 | Managing Director; Chief Executive Officer | ||
| CHEW Yoke Ling | 53 | Director | ||
| LOW Wei Sim | 29 | Chief Operating Officer | ||
| Professor Manickam RAVICHANDRAN | 57 | Chief Scientific Officer | ||
| Professor POH Chit Laa | 73 | Chief Vaccine Development Officer | ||
| Tan Sri Dato’ Seri Dr. Suleiman bin Mohamed | 76 | Independent Director | ||
| CHUA Boon Ping | 40 | Independent Director | ||
| Intan Ilyani binti Ghazali | 45 | Independent Director |
Dr. THAM Seng Kong will serve as Managing Director on the Combined Company’s board of director. Dr. Tham has served as Alps’ Managing Director, Group Chief Executive Officer and Group Chief Research Scientist since its inception. Dr. Tham is a veteran in the field of life sciences, with a focus in the field of Cellular Therapy. He is an established member of the Malaysian Society for Stem Cell Research & Therapy and the Malaysia Association for Cell Therapy. He has been involved in ongoing extensive research on anti-aging cellular therapy since 2006. Dr. Tham also actively participates in cancer research. Dr. Tham obtained his Bachelor of Medicine from Xiamen University in 1994, and Masters of Business Administration from the Business Institute of Pennsylvania in 2012. Dr. Tham obtained his Ph.D. in Integrated Chinese Medicine & Western Clinical Medicine (Oncology) from Guangzhou University of Chinese Medicine (GUCM) in 2017. Dr. Tham will bring to the board his deep expertise in the field of life sciences and cellular therapy.
CHEW Yoke Ling will serve as a Director on the Combined Company’s board of director. She has served as a Director of Alps since 2017. With a background in the beauty industry, Ms. Chew has leveraged her expertise to drive strategic initiatives within our organization. In her role as a marketing director, Ms. Chew oversees sales and marketing activities for Alps’ products and services. Her responsibilities extend to the planning and coordination of Alps medical tourism efforts, catering to both local and international clients. She obtained a Diploma in Marketing from Stamford Group of Colleges, in 1992, and a Diploma in Secretarial Studies from RIMA College, in 1990. Ms. Chew will bring to the board her expertise and knowledge in marketing and medical tourism
Low Wei Sim (Amanda) will serve as the Chief Operating Officer of the Combined Company. Ms. Amanda has been appointed as our Deputy Chief Operating Officer from April 1, 2024. Prior to this role, she held a senior executive position within the same company. Since December 1, 2018, Ms. Amanda has served as a director at Aiiot Technologies Sdn. Bhd., which specializes in information system development and supporting services ancillary to the Internet of Things (IoT). Additionally, she also serves as the executive director in Alps Globemedic Sdn. Bhd., a company dedicated to healthcare business developments, since November 2018. Amanda obtained her Bachelor of Science (BSc) Hons. in Business Studies from Sunway University, Malaysia in 2018. Ms. Low will bring to the board her expertise in healthcare and wellness operations.
Professor Manickam RAVICHANDRAN will serve as the Combined Company’s Chief Scientific Officer. Professor Ravichandran has been Alps’ Chief Scientific Officer since December 1, 2022. He is responsible for overseeing the scientific and research-related activities. He also serves as the Dean of the Faculty of Applied Sciences at AIMST University, and previously served as the Vice-Chancellor of AIMST University, Malaysia. Professor Ravichandran received his Bachelor of Science in Botany from Madurai Kamaraj University, India in 1988. He obtained his M.Sc. in Medical Microbiology from Christian Medical College, Vellore in 1991, and gained his Ph.D. in Biotechnology from Anna University, Chennai, India in 1977. Professor Ravichandran also contributes to the academic community as an Editorial Board Member of the Tropical Biomedicine Journal, a position he has held since July 7, 2020. Professor Ravichandran’s academic and research excellence has been recognized with 44 national and international awards, including honors from prestigious exhibitions such as IENA in Nuremberg, Germany, and the International Exhibition of Inventions in Geneva. He has also been awarded the Anugerah Inovasi Negara in Malaysia. As an associate member and committee member of ‘Top Research Scientists Malaysia’ at the Academy of Sciences Malaysia. Professor Ravichandran will bring to the board his deep knowledge and expertise in scientific and research related activities.
| 271 |
Professor POH Chit Laa will serve as the Combined Company’s Chief Vaccine Development Officer. Professor Poh is Alps’ Chief Vaccine Development Officer since 2024. Professor Poh has a prestigious career in higher education, focusing on medical microbiology, bacteriology, and biotechnology. She has served as distinguished professor and Head of the Centre for Virus and Vaccine Research at Sunway University from 2011 to 2024. Professor Poh held numerous academic positions, including Professor of Environmental Biotechnology at Swinburne University of Technology (Australia) from 2007 to 2009, and Professor of Biomedical Science at Curtin University of Technology (Australia) from 2005 to 2006. Earlier in her career, Professor Poh was an Associate Professor at the National University of Singapore from 1982 to 2007. Professor Poh obtained a Bachelor of Science (BSc) with First Class Honors in 1975, and a Ph.D. in Medical Microbiology and Bacteriology from Monash University, Australia in 1980. Additionally, Professor Poh contributes to the academic community as a member of the Editorial Committee of the Journal of Bioscience and Bioengineering, published by the Society for Biotechnology in Japan, a role she has held since 2023. Professor Poh will bring to the board her expertise and knowledge in medical microbiology, bacteriology and biotechnology.
Non-Executive and Independent Directors
Tan Sri Dato’ Seri Dr. Suleiman bin Mohamed has agreed to serve as an independent director after the Business Combination. Tan Sri Suleiman has had a distinguished career in journalism, government, and education. Since 2009, he has been the founder and Chairman of Koperasi Khazanah Suria (KL) Berhad, a company focused on business consulting and promoting financial literacy, education, and planning for its members. From 2012 to 2016, he served as the Chairman of UNITAR International University. Tan Sri Suleiman was elected as a Member of the Parliament of Malaysia for four consecutive terms from 1986 to 2004. During his tenure, he held several significant positions, Deputy Minister of Health from 1999 to 2004, Deputy Minister of Information from 1995 to 1999, and Deputy Minister of the Prime Minister’s Department from 1987 to 1995. Tan Sri Suleiman holds a Doctorate in Philosophy in Malay Literature Studies from Universiti Kebangsaan Malaysia. He obtained a Doctorate in Journalism and a Bachelor’s Degree in Communication Science from Universitas Negeri Padjadjaran, Bandung, Indonesia. Tan Sri Suleiman will bring to the board his deep experience and knowledge of Malaysia’s governmental policies and structure in the healthcare field.
Chua Boon Ping has agreed to serve as an independent director after the Business Combination. Mr. Chua is an experienced venture capitalist with a proven track record, currently spearheading Cento Ventures’ investments in early-stage startups in Southeast Asia. Since February 2023, he has been a Partner at Cento Ventures. Mr. Chua has served as a board member at Doxa Holdings International since November 2023. From May 2021 to August 2023, Mr. Chua has served as the chief executive officer of Straits Capital Investments Pte Ltd, the corporate ventures fund of Singapore Press Holdings. Mr. Chua obtained his Bachelor of Engineering (with Honours) and a Master in Business Administration (Banking & Finance) from Nanyang Technological University in 2002. Mr. Chua brings to the board his expertise in finance and investment in Southeast Asia.
Intan Ilyani binti Ghazali will serve as an independent director of the Combined Company after the Business Combination. Ms. Intan has extensive experience in strategic partnerships, branding, and stakeholder management. Since August 2024, she has served as Vice President of Retail & eCommerce at Shui Xing Group Ventures. From July 2021 to July 2024, Ms. Intan was the Head of Strategic Partnership & Branding at JLand Group, where she managed partnerships with companies including Petronas Gentari and Mitsui. Previously, she held roles at Maybank Group, including Special Officer to the Chairman from 2008 to 2014 and Executive, Corporate Services & Legal from 2006 to 2008. Ms. Intan also founded Face & Co, a skincare brand, and managed it from 2017 to 2020. Ms. Intan holds a Master of Business Administration from the University of Technology, Malaysia, awarded in 2023, and a Diploma in Executive Secretaryship from MARA University of Technology, Malaysia, awarded in 2000. She brings expertise in partnership development and branding to the board.
Family Relationships
There are no family relationships between any of the foregoing proposed directors or executive officers of the Combined Company.
Involvement in Certain Legal Proceedings
None of the foregoing proposed executive officer or director of the Combined Company is a party in a legal proceeding adverse to Alps or any of Alps’ subsidiaries or has a material interest adverse to Alps or any of Alps’ subsidiaries.
None of the foregoing proposed executive officer or director of the Combined Company has been involved in the last ten years in any of the following:
| ● | Any bankruptcy petition filed by or against any business or property of such person, or of which such person was a general partner or executive officer either at the time of the bankruptcy or within two years prior to that time; | |
| ● | Any conviction in a criminal proceeding or being subject to a pending criminal proceeding (excluding traffic violations and other minor offenses); | |
| ● | Being subject to any order, judgment, or decree, not subsequently reversed, suspended or vacated, of any court of competent jurisdiction, permanently or temporarily enjoining, barring, suspending or otherwise limiting his involvement in any type of business, securities or banking activities; | |
| ● | Being found by a court of competent jurisdiction (in a civil action), the SEC or the Commodity Futures Trading Commission to have violated a federal or state securities or commodities law, and the judgment has not been reversed, suspended, or vacated; | |
| ● | Being the subject of or a party to any judicial or administrative order, judgment, decree or finding, not subsequently reversed, suspended or vacated relating to an alleged violation of any federal or state securities or commodities law or regulation, or any law or regulation respecting financial institutions or insurance companies, including but not limited to, a temporary or permanent injunction, order of disgorgement or restitution, civil money penalty or temporary or permanent cease-and-desist order, or removal or prohibition order, or any law or regulation prohibiting mail, fraud, wire fraud or fraud in connection with any business entity; or | |
| ● | Being the subject of or a party to any sanction or order, not subsequently reversed, suspended or vacated, of any self-regulatory organization (as defined in Section 3(a)(26) of the Exchange Act, any registered entity (as defined in Section 1(a)(29) of the Commodity Exchange Act), or any equivalent exchange, association, entity or organization that has disciplinary authority over its members or persons associated with a member. |
Board of Directors
The board of directors of the Combined Company will initially consist of five directors immediately after the consummation of the Business Combination. Of these initial five directors, three will be independent. The PubCo A&R Memorandum and Articles provide that the maximum number of directors shall be unlimited unless otherwise fixed by ordinary resolution and the directors shall be divided into two classes: Class A and Class B. An executive director shall be a Class A director and an independent director shall be a Class B director. Upon the adoption of the PubCo A&R Memorandum and Articles, the existing directors shall by resolution classify themselves as Class A or Class B as applicable. Except as provided in the PubCo A&R Memorandum and Articles, a director may vote in respect of any contract or transaction in which he/she is interested provided that the nature of the interest of any director in any such contract or transaction is disclosed at or prior to its consideration and any vote thereon, and such director may be counted in the quorum at any meeting of directors at which any such contract or transaction is considered. A director who is interested in a contract or proposed contract with the Combined Company must declare the nature of his/her interest at a meeting of the directors. There is no non-employee director has a service contract with the Combined Company that provides for benefits upon termination of service.
| 272 |
Duties of Directors
Under Cayman Islands law, PubCo directors owe fiduciary duties to the company, including a duty of loyalty, a duty to act honestly, and a duty to act in good faith in what they consider to be in the company’s best interests. PubCo directors must also exercise their powers only for a proper purpose. PubCo directors also have a duty to exercise the skills they actually possess and such care and diligence that a reasonably prudent person would exercise in comparable circumstances.
In fulfilling the duty of care to the company, PubCo directors must ensure compliance with our memorandum and articles of association as may be amended from time to time. PubCo has a right to seek damages against any director who breaches a duty owed to us.
Committees of the Board of Directors
Audit Committee
After the consummation of the Business Combination, the Combined Company is required to maintain an audit committee consisting of at least three independent directors in accordance with the Nasdaq listing standards and applicable SEC rules. The Combined Company’s board of directors will subsequently identify which audit committee member meets the definition of an “audit committee financial expert” under Nasdaq listing standards and under Rule 10-A-3(b)(1) under the Exchange Act.
The audit committee’s role will be to compile the necessary audit committee report for the SEC, to be included in PubCo’s proxy statements, and to support the Combined Company’s board of directors in supervising (1) the integrity of the financial reports, (2) adherence to legal and statutory obligations, (3) the qualifications and autonomy of Alps’ independent accountants, (4) the internal audit function’s efficacy, and (5) the performance of the independent accounting firm engaged by the Combined Company.
Compensation Committee
Following the consummation of the Business Combination, the Combined Company is required to maintain a compensation committee of at least two independent directors, which will be subsequently identified by the Combined Company’s board of directors. The Combined Company’s board of directors has determined that each proposed member of PubCo’s remuneration committee is independent under the Nasdaq listing standards and applicable SEC rules, including the additional independence requirements applicable to the members of a remuneration committee.
The Combined Company’s board of directors will, prior to or concurrently with the listing of PubCo’s ordinary shares, adopt a term of reference setting forth the responsibilities of the committee, which are consistent with the Nasdaq listing standards and applicable SEC rules, and include among others:
| ● | recommending to the Combined Company’s board of directors for its approval a compensation policy in accordance with the requirements of Cayman Islands law as well as other compensation policies, incentive-based compensation plans and equity-based compensation plans, and overseeing the development and implementation of such policies and recommending to Combined Company’s board of directors any amendments or modifications the committee deems appropriate, including as required under Cayman Islands law;
| |
| ● | implementing and administering our incentive compensation equity-based remuneration plans;
| |
| ● | assisting management in complying with the proxy statement and annual report disclosure requirements;
| |
| ● | approving all special perquisites, special cash payments and other special compensation and benefit arrangements for the officers and employees;
| |
| ● | if required, producing a report on executive compensation to be included in the annual proxy statement; and
| |
| ● | reviewing, evaluating and recommending changes, if appropriate, to the remuneration for directors. |
| 273 |
Code of Business Conduct and Ethics
PubCo will, prior to or concurrently with the listing of PubCo’s Ordinary Shares on the Nasdaq, adopt a Code of Business Conduct and Ethics applicable to its directors, officers and employees in accordance with applicable securities laws. The code of ethics will codify the business and ethical principles that govern all aspects of our business.
Compensation of Directors and Executive Officers
The aggregate compensation paid to Alps’ executive officers as a group in cash compensation and benefits in kind was approximately RM1,337,400.00 (approximately US$ 283,847.07) for the year ended March 31, 2024. Alps’ executive officers do not receive pension, retirement or other similar benefits, and Alps and its subsidiaries have not set aside or accrued any amount to provide such benefits to its executive officers.
Alps’ subsidiaries in Malaysia are required by the applicable laws and regulations of Malaysia to make contributions, as employers, to the Employees Provident Fund (“EPF”) for their executive officers who are employed by Alps’ Malaysia subsidiaries as prescribed under the Employees Provident Fund Act 1991. The EPF contribution rates are 11% for employees and 12-13% for employers. The contribution rates vary, depending on the age of the executive officers, and whether such executive officer is a Malaysian citizen or permanent resident (contributions are not required or permitted in respect of a foreign employee or expatriates).
Employment Agreements and Indemnification Agreements
Alps, or certain of its subsidiaries, have entered into employment agreements with each of its executive officers, which set forth the terms and conditions of each executive’s employment. The employment of the executive officers under these employment agreements is for an indefinite period, but may be terminated by the employer for cause at any time with advance notice or for any other reason by giving prior written notice or by paying certain compensation in lieu of notice, and the executive officer may terminate his or her employment at any time by giving the employer prior written notice. The employment agreements with the executive officers also include confidentiality and non-competition and non-solicitation restrictions that apply during employment for certain periods following termination of employment.
PubCo will enter into indemnification agreements with each of its directors. Under these agreements, PubCo may agree to indemnify its director against certain liabilities and expenses incurred by such persons in connection with claims made by reason of their being a director of PubCo.
Directors’ Addresses
Each of the directors can be contacted at Unit E-18-01 & E-18-02, Level 18, Icon Tower (East), No. 1, Jalan 1/68F, Jalan Tun Razak, 50400 Kuala Lumpur, Wilayah Persekutuan, Malaysia.
| 274 |
SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT
The following table sets forth information regarding the beneficial ownership of shares of Globalink common stock as of September 26, 2024 pre-Business Combination and the beneficial ownership of PubCo ordinary shares immediately after the consummation of the Business Combination by:
● each person or “group” (as such term is used in Section 13(d)(3) of the Exchange Act) known by Globalink to be the beneficial owner of more than 5% of shares of Globalink common stock as of September 26, 2024 (pre-Business Combination) or of PubCo ordinary shares upon the Closing of the Business Combination;
● each of Globalink’s executive officers and directors;
● each person who will become an executive officer or director of the Combined Company upon the Closing of the Business Combination;
● all of Globalink’s current executive officers and directors as a group; and
● all executive officers and directors of the Combined Company as a group upon the Closing of the Business Combination.
As of the Record Date, Globalink had shares of common stock issued and outstanding.
Beneficial ownership is determined in accordance with SEC rules and includes voting or investment power with respect to securities. Except as indicated by the footnotes below, Globalink believes, based on the information furnished to it, that the persons and entities named in the table below have, or will have immediately after the consummation of the Business Combination, sole voting and investment power with respect to all PubCo ordinary shares that they beneficially own, subject to applicable community property laws. Any PubCo ordinary shares subject to rights or warrants exercisable within 60 days of the consummation of the Business Combination are deemed to be outstanding and beneficially owned by the persons holding those rights or warrants for the purpose of computing the number of shares beneficially owned and the percentage ownership of that person. They are not, however, deemed to be outstanding and beneficially owned for the purpose of computing the percentage ownership of any other person.
Subject to the paragraph above, percentage ownership of outstanding shares is based on PubCo ordinary shares to be outstanding upon consummation of the Business Combination, comprising 170,549,511 ordinary shares, and is inclusive of the PIPE Investment, but does not take into account any warrants, or other convertible securities of Alps issued and outstanding as of the date hereof. If the actual facts are different than these assumptions (which they are likely to be), the percentage ownership retained by Globalink’s existing stockholders in Globalink will be different.
The expected beneficial ownership of ordinary shares post-Business Combination under the header “Post-Business Combination — Assuming No Redemption” assumes none of the public shares of Globalink common stock having been redeemed. The expected beneficial ownership of common stock post-Business Combination under the header “Post-Business Combination — Assuming Maximum Contractual Redemption” assumes 277,511 public shares of Globalink having been redeemed.
| 275 |
| Pre-Business Combination | Post-Business Combination | |||||||||||||||||||||||
| Name and Address of | Number of | % of | Assuming No Redemptions Number of | % of | Assuming Maximum Contractual Redemptions Number of | % of | ||||||||||||||||||
| Beneficial Owner(1) | Shares | Shares | Shares | Shares | Shares | Shares | ||||||||||||||||||
| Directors and Executive Officers of Globalink | ||||||||||||||||||||||||
| Say Leong Lim (1) | 15,000 | * | 15,000 | * | 15,000 | * | ||||||||||||||||||
| Kelvin (Zeng Yenn) Chin (1) | 10,000 | * | 10,000 | * | 10,000 | * | ||||||||||||||||||
| Hong Shien Beh (1) | 5,000 | * | 5,000 | * | 5,000 | * | ||||||||||||||||||
| Kian Huat Lai (1) | 5,000 | * | 5,000 | * | 5,000 | * | ||||||||||||||||||
| Hui Liang Wong (1) | 5,000 | * | 5,000 | * | 5,000 | * | ||||||||||||||||||
| All officers and directors as a group (5 individuals) | 40,000 | * | 40,000 | * | 40,000 | * | ||||||||||||||||||
| 5% or Greater Beneficial Owners: | ||||||||||||||||||||||||
| GL Sponsor LLC(2) | 2,835,000 | 47.19 | % | 2,835,000 | 1.64 | % | 2,835,000 | 1.64 | % | |||||||||||||||
| Directors and Executive Officers of the Combined Company | ||||||||||||||||||||||||
| Dr. Tham Seng Kong (3) | Nil | Nil | 31,194,937 | 18.05 | % | 31,194,937 | 18.18 | % | ||||||||||||||||
| Chew Yoke Ling (3) | Nil | Nil | 2,491,789 | 1.44 | % | 2,491,789 | 1.45 | % | ||||||||||||||||
| Low Wei Sim (3) | Nil | Nil | 7,648 | * | 7,648 | * | ||||||||||||||||||
| Professor Manickam Ravichandran (3) | Nil | Nil | Nil | Nil | Nil | Nil | ||||||||||||||||||
| Professor Poh Chit Laa (3) | Nil | Nil | Nil | Nil | Nil | Nil | ||||||||||||||||||
| Tan Sri Dato’ Seri Dr. Suleiman bin Mohamed (3) | Nil | Nil | 2,000,000 | 1.16 | % | 2,000,000 | 1.17 | % | ||||||||||||||||
| Chua Boon Ping (3) | Nil | Nil | 34,178 | * | 34,178 | * | ||||||||||||||||||
| Intan Ilyani binti Ghazali (3) | Nil | Nil | Nil | Nil | Nil | Nil | ||||||||||||||||||
| All directors and executive officers of the Combined Company as a group | Nil | Nil | 37,728,552 | 21.83 | % | 37,728,552 | 21.99 | % | ||||||||||||||||
| Five Percent Holders of the Combined Company | ||||||||||||||||||||||||
| Dr. Tham Seng Kong (3) | Nil | Nil | 31,194,937 | 18.05 | % | 31,194,937 | 18.18 | % | ||||||||||||||||
| * | Less than one percent. |
| (1) | The business address of each of the individuals is 200 Continental Drive, Suite 401, Newark, Delaware 19713. |
| (2) | GL Sponsor LLC is the record holder of the shares reported herein. GL Sponsor LLC is managed by Ng Yan Xun. Ng Yan Xun may be deemed to beneficially own the shares held by the Sponsor by virtue of his control of the Sponsor. Ng Yan Xun disclaims beneficial ownership of the shares held by the Sponsor except to the extent of his pecuniary interest therein. The business address of the Sponsor is 1180 Avenue of the Americas, 8th Floor, New York, NY. |
| (3) | Unless otherwise noted, the address of each of the named directors, executive officers and holders is c/o Unit E-18-01 & E-18-02, Level 18, Icon Tower (East), No. 1, Jalan 1/68F, Jalan Tun Razak, 50400 Kuala Lumpur, Wilayah Persekutuan, Malaysia. |
| 276 |
DESCRIPTION OF GLOBALINK’S SECURITIES
Unless the context otherwise requires, references to the “Company,” “we”, “us” and “our” in this section generally refer to Globalink Investment Inc.
General
Globalink Charter authorizes the issuance of 500,000,000 shares of common stock, par value $0.001 per share and 5,000,000 shares of preferred stock, par value $0.001 per share. As of the date of this proxy statement/prospectus, there were 3,722,511 shares of common stock issued and outstanding. The following description summarizes all of the material terms of our securities. Because it is only a summary, it may not contain all the information that is important to you. For a complete description you should refer to Globalink Charter and bylaws, which are filed as exhibits to the registration statement of which this proxy statement/prospectus is a part.
Units
Each unit has an offering price of $10.00 and consists of one share of common stock, one right and one redeemable warrant. Each right entitles the holder thereof to receive one-tenth (1/10) of a share of common stock upon consummation of our initial business combination. We will not issue fractional shares in connection with an exchange of rights. As a result, you must hold rights in multiples of 10 in order to receive shares for all of your rights upon closing of a business combination. Each redeemable warrant entitles the registered holder to purchase one half (1/2) of one share of common stock at a price of $11.50 per full share, subject to adjustment as described in this proxy statement/prospectus, and shall expire five years after the completion of an initial business combination, or earlier upon redemption. Pursuant to the warrant agreement, a warrant holder may exercise its warrants only for a whole number of shares. This means that only warrants in multiples of two may be exercised at any given time by a warrant holder. For example, if a warrant holder holds one warrant to purchase one-half (1/2) of one share, such warrant shall not be exercisable. If a warrant holder holds two warrants, such warrants will be exercisable for one share. Fractional shares will either be rounded down to the nearest whole share or otherwise addressed in accordance with the applicable provisions of Delaware law.
Globalink’s common stock, rights and warrants comprising the units trade separately on OTC Pink. Unit holders have the option to continue to hold units or separate their units into the component pieces. Holders will need to have their brokers contact our transfer agent to separate the units into shares of common stock, rights and warrants.
Private Units
On December 9, 2021, the date we closed our IPO, we consummated the private placement of 517,500 private units at a price of $10.00 per unit, generating total proceeds of $5,175,000, with the private units sold to PGM, an affiliate of the Sponsor. Simultaneously with the full exercise of the over-allotment, Globalink consummated a private sale of an additional 52,500 private units to PGM at a price of $10.00 per unit, generating additional gross proceeds of $525,000. The private units are identical to the units sold in our IPO except that (a) the private units and their component securities will not be transferable, assignable or salable until 30 days after the consummation of our initial business combination except to permitted transferees, and (b) the private warrants, so long as they are held by our Sponsor or its permitted transferees, (i) will not be redeemable by us, (ii) may be exercised by the holders on a cashless basis, and (iii) will be entitled to registration rights.
Common Stock
Our holders of record of our common stock are entitled to one vote for each share held on all matters to be voted on by stockholders. In connection with any vote held to approve our initial business combination, the Initial Stockholders and PGM, an affiliate of the Sponsor, through the Insider Letter, have agreed to vote their respective shares of common stock owned by them and any shares subsequently acquired by them in favor of our initial business combination. The Initial Stockholders and PGM do not intend to acquire any shares of Globalink common stock in the open market.
We will consummate our initial business combination only if public stockholders do not exercise conversion rights in an amount that would cause our net tangible assets following the consummation of our initial business combination to be less than $5,000,001 the and, assuming a quorum is present at the meeting, the affirmative vote of a majority of the shares of common stock present in person by virtual attendance or represented by proxy and entitled to vote at the meeting are voted in favor of our initial business combination.
| 277 |
Our Board is divided into three classes, each of which will generally serve for a term of three years with only one class of directors being elected in each year. There is no cumulative voting with respect to the election of directors, with the result that the holders of more than 50% of the shares eligible to vote for the election of directors can elect all of the directors.
Pursuant to Globalink Charter, if we do not consummate our initial business combination by January 9, 2025 (and may be extended to June 9, 2025 on a monthly basis by resolutions of the Globalink Board, if requested by the Sponsor, and subject to the Sponsor depositing additional funds into the Trust Account), we will (i) cease all operations except for the purpose of winding up, (ii) as promptly as reasonably possible but not more than ten business days thereafter, redeem 100% of the outstanding public shares for a pro rata portion of the funds held in the Trust Account, which redemption will completely extinguish public stockholders’ rights as stockholders (including the right to receive further liquidation distributions, if any), subject to applicable law, and (iii) as promptly as reasonably possible following such redemption, subject to the approval of our remaining stockholders and our Board, dissolve and liquidate, subject (in the case of (ii) and (iii) above) to our obligations under Delaware law to provide for claims of creditors and the requirements of other applicable law. Through the Insider Letter, in consideration for inducing Chardan, the representative of the underwriters for Globalink’s IPO to proceed with the IPO, and the economic benefit the IPO would confer upon them, our insiders have agreed to waive their rights to share in any distribution with respect to their insider shares and private shares.
Our stockholders have no conversion, preemptive or other subscription rights and there are no sinking fund or redemption provisions applicable to the shares of common stock, except that public stockholders have the right to sell their shares to us in any tender offer or have their shares of common stock converted to cash equal to their pro rata share of the Trust Account if they vote on the initial business combination and the initial business combination is completed.
Preferred Stock
There are no shares of preferred stock outstanding. Globalink Charter provides that up to 5,000,000 shares of preferred stock may be issued from time to time in one or more series. Our Board will be empowered, without stockholder approval, to issue shares of preferred stock with dividend, liquidation, conversion, voting or other rights which could adversely affect the voting power or other rights of the holders of common stock. However, the underwriting agreement we entered into with Chardan in connection with the IPO prohibits us, prior to a business combination, from issuing shares of preferred stock which participates in any manner in the proceeds of the Trust Account, or which votes as a class with the common stock on our initial business combination. We may issue some or all of the shares of preferred stock to effect our initial business combination. In addition, the preferred stock could be utilized as a method of discouraging, delaying or preventing a change in control of us. Although we do not currently intend to issue any shares of preferred stock, we reserve the right to do so in the future.
Rights Included as Part of Units
Except in cases where we are not the surviving company in a business combination, each holder of a right will automatically receive one-tenth (1/10) of a share of common stock upon consummation of our initial business combination, even if the holder of a public right converted all shares of common stock held by him, her or it in connection with the initial business combination or an amendment to Globalink Charter with respect to our pre-business combination activities. In the event we will not be the surviving company upon completion of our initial business combination, each holder of a right will be required to affirmatively convert his, her or its rights in order to receive the one-tenth (1/10) of a share underlying each right upon consummation of the business combination. No additional consideration will be required to be paid by a holder of rights in order to receive his, her or its additional shares of common stock upon consummation of an initial business combination. The shares issuable upon exchange of the rights will be freely tradable (except to the extent held by affiliates of ours). If we enter into a definitive agreement for a business combination in which we will not be the surviving entity, the definitive agreement will provide for the holders of rights to receive the same per share consideration the holders of the shares of common stock will receive in the transaction on an as-converted into shares of common stock basis.
The rights will be issued in registered form under a rights agreement between Continental, as rights agent, and us. The rights agreement provides that the terms of the rights may be amended without the consent of any holder to cure any ambiguity or correct any defective provision, but requires the approval, by written consent or vote, of the holders of a majority of the then outstanding rights in order to make any change that adversely affects the interests of the registered holders.
| 278 |
We will not issue fractional shares in connection with an exchange of rights. Fractional shares will either be rounded down to the nearest whole share or otherwise addressed in accordance with the applicable provisions of Delaware law. As a result, you must hold rights in multiples of 10 in order to receive shares for all of your rights upon closing of a business combination. If we are unable to complete an initial business combination within the required time period and we liquidate the funds held in the Trust Account, holders of rights will not receive any of such funds with respect to their rights, nor will they receive any distribution from our assets held outside of the Trust Account with respect to such rights, and the rights will expire worthless. Further, there are no contractual penalties for failure to deliver securities to the holders of the rights upon consummation of an initial business combination. Additionally, in no event will we be required to net cash settle the rights. Accordingly, the rights may expire worthless.
We have agreed that, subject to applicable law, any action, proceeding or claim against us arising out of or relating in any way to the rights agreement, including under the Securities Act, will be brought and enforced in the courts of the State of New York or the United States District Court for the Southern District of New York, and we irrevocably submit to such jurisdiction, which jurisdiction will be the exclusive forum for any such action, proceeding or claim. This provision applies to claims under the Securities Act but does not apply to claims under the Exchange Act or any claim for which the federal district courts of the United States of America are the sole and exclusive forum. We note, however, that there is uncertainty as to whether a court would enforce these provisions and that investors cannot waive compliance with the federal securities laws and the rules and regulations thereunder. Section 22 of the Securities Act creates concurrent jurisdiction for state and federal courts over all suits brought to enforce any duty or liability created by the Securities Act or the rules and regulations thereunder.
Warrants
As of the date of this proxy statement/prospectus, the Company had 11,500,000 public warrants and 570,000 private warrants outstanding. Each whole warrant entitles the registered holder to purchase one half of a share of common stock at a price of $11.50 per full share, subject to adjustment as discussed below, at any time commencing on the later of the completion of an initial business combination and 12 months from the date of our IPO prospectus. However, except as set forth below, no warrants will be exercisable for cash unless we have an effective and current registration statement covering the shares of common stock issuable upon exercise of the warrants and a current prospectus relating to such shares. Notwithstanding the foregoing, if a registration statement covering the shares of common stock issuable upon exercise of the warrants is not effective within 90 days from the consummation of our initial business combination, warrant holders may, until such time as there is an effective registration statement and during any period when we shall have failed to maintain an effective registration statement, exercise warrants on a cashless basis pursuant to the exemption from registration provided by Section 3(a)(9) of the Securities Act provided that such exemption is available. If an exemption from registration is not available, holders will not be able to exercise their warrants on a cashless basis. The warrants will expire five years after the completion of an initial business combination at 5:00 p.m., Eastern Time, or earlier upon redemption or liquidation.
In addition, if (x) we issue additional shares of common stock or equity-linked securities for capital raising purposes in connection with the closing of our initial business combination at an issue price or effective issue price of less than $9.50 per share (with such issue price or effective issue price to be determined in good faith by our Board), (y) the aggregate gross proceeds from such issuances represent more than 60% of the total equity proceeds, and interest thereon, available for the funding of our initial business combination, and (z) the volume weighted average trading price of our shares of common stock during the 20 trading day period starting on the trading day prior to the day on which we consummate our initial business combination (such price, the “Market Price”) is below $9.50 per share, the exercise price of the warrants will be adjusted (to the nearest cent) to be equal to 115% of the Market Price, and the $16.50 per share redemption trigger price described above will be adjusted (to the nearest cent) to be equal to 165% of the Market Price.
The private warrants are identical to the public warrants underlying the units sold in our IPO except that such private warrants will be exercisable for cash (even if a registration statement covering the shares of common stock issuable upon exercise of such warrants is not effective) or on a cashless basis, at the holder’s option, and will not be redeemable by us, in each case so long as they are still held by our Sponsor or its permitted transferees.
| 279 |
We may call the warrants for redemption (excluding the private warrants), in whole and not in part, at a price of $0.01 per warrant:
| ● | at any time while the warrants are exercisable; | |
| ● | upon not less than 30 days’ prior written notice of redemption to each warrant holder; | |
| ● | if, and only if, the reported last sale price of the shares of common stock equals or exceeds $16.50 per share, for any 20 trading days within a 30 trading day period ending on the third business day prior to the notice of redemption to warrant holders (the “Force-Call Provision”); and | |
| ● | if, and only if, there is a current registration statement in effect with respect to the shares of common stock underlying such warrants at the time of redemption and for the entire 30-day trading period referred to above and continuing each day thereafter until the date of redemption. |
The right to exercise will be forfeited unless the warrants are exercised prior to the date specified in the notice of redemption. On and after the redemption date, a record holder of a warrant will have no further rights except to receive the redemption price for such holder’s warrant upon surrender of such warrant.
The redemption criteria for our warrants have been established at a price that is intended to provide warrant holders a reasonable premium to the initial exercise price and provide a sufficient differential between the then-prevailing share price and the warrant exercise price so that if the share price declines as a result of our redemption call, the redemption will not cause the share price to drop below the exercise price of the warrants.
If we call the warrants for redemption as described above, our management will have the option to require all holders that wish to exercise warrants to do so on a “cashless basis.” In such event, each holder would pay the exercise price by surrendering the whole warrants for that number of shares of common stock equal to the quotient obtained by dividing (x) the product of the number of shares of common stock underlying the warrants, multiplied by the difference between the exercise price of the warrants and the “fair market value” (defined below) by (y) the fair market value. The “fair market value” shall mean the volume weighted average trading price of our common stock for the 20 trading days ending on the third trading day prior to the date on which the notice of redemption is sent to the holders of warrants. Whether we will exercise our option to require all holders to exercise their warrants on a “cashless basis” will depend on a variety of factors, including the price of our shares of common stock at the time the warrants are called for redemption, our cash needs at such time and concerns regarding dilutive share issuances.
The warrants will be issued in registered form under a warrant agreement between Continental, as warrant agent, and us. The warrant agreement provides that the terms of the warrants may be amended without the consent of any holder to cure any ambiguity or correct any defective provision, but requires the approval, by written consent or vote, of the holders of a majority of the then outstanding warrants in order to make any change that adversely affects the interests of the registered holders.
The exercise price and number of shares of common stock issuable on exercise of the warrants may be adjusted in certain circumstances including in the event of a stock capitalizations, extraordinary dividend or our recapitalization, reorganization, merger or consolidation. However, the warrants will not be adjusted for issuances of shares of common stock at a price below their respective exercise prices.
The warrants may be exercised upon surrender of the warrant certificate on or prior to the expiration date at the offices of the warrant agent, with the exercise form on the reverse side of the warrant certificate completed and executed as indicated, accompanied by full payment of the exercise price, by certified or official bank check payable to us, for the number of warrants being exercised. The warrant holders do not have the rights or privileges of holders of shares of common stock and any voting rights until they exercise their warrants and receive shares of common stock. After the issuance of shares of common stock upon exercise of the warrants, each holder will be entitled to one vote for each share held of record on all matters to be voted on by stockholders.
| 280 |
Except as described above, no warrants will be exercisable and we will not be obligated to issue shares of common stock unless at the time a holder seeks to exercise such warrant, a prospectus relating to the shares of common stock issuable upon exercise of the warrants is current and the shares of common stock have been registered or qualified or deemed to be exempt under the securities laws of the state of residence of the holder of the warrants. Under the terms of the warrant agreement, we have agreed to use our best efforts to meet these conditions and to maintain a current prospectus relating to the shares of common stock issuable upon exercise of the warrants until the expiration of the warrants. However, we cannot assure you that we will be able to do so and, if we do not maintain a current prospectus relating to the shares of common stock issuable upon exercise of the warrants, holders will be unable to exercise their warrants and we will not be required to settle any such warrant exercise. If the prospectus relating to the shares of common stock issuable upon the exercise of the warrants is not current or if the shares of common stock is not qualified or exempt from qualification in the jurisdictions in which the holders of the warrants reside, we will not be required to net cash settle or cash settle the warrant exercise, the warrants may have no value, the market for the warrants may be limited and the warrants may expire worthless.
Warrant holders may elect to be subject to a restriction on the exercise of their warrants such that an electing warrant holder (and his, her or its affiliates) would not be able to exercise their warrants to the extent that, after giving effect to such exercise, such holder (and his, her or its affiliates) would beneficially own in excess of 9.99% of the shares of common stock issued and outstanding. Notwithstanding the foregoing, any person who acquires a warrant with the purpose or effect of changing or influencing the control of our company, or in connection with or as a participant in any transaction having such purpose or effect, immediately upon such acquisition will be deemed to be the beneficial owner of the underlying shares of common stock and not be able to take advantage of this provision.
No fractional shares will be issued upon exercise of the warrants. If, upon exercise of the warrants, a holder would be entitled to receive a fractional interest in a share (as a result of a subsequent share capitalizations payable in ordinary shares, or by a split up of the shares of common stock or other similar event), we will, upon exercise, round up or down to the nearest whole number the number of shares of common stock to be issued to the warrant holder.
We have agreed that, subject to applicable law, any action, proceeding or claim against us arising out of or relating in any way to the warrant agreement, including under the Securities Act, will be brought and enforced in the courts of the State of New York or the United States District Court for the Southern District of New York, and we irrevocably submit to such jurisdiction, which jurisdiction will be the exclusive forum for any such action, proceeding or claim. This provision applies to claims under the Securities Act but does not apply to claims under the Exchange Act or any claim for which the federal district courts of the United States of America are the sole and exclusive forum. We note, however, that there is uncertainty as to whether a court would enforce these provisions and that investors cannot waive compliance with the federal securities laws and the rules and regulations thereunder. Section 22 of the Securities Act creates concurrent jurisdiction for state and federal courts over all suits brought to enforce any duty or liability created by the Securities Act or the rules and regulations thereunder.
Contractual Arrangements with respect to the Certain Warrants
We have agreed that so long as the private warrants are still held by PGM or its permitted transferees, we will not redeem such warrants and we will allow the holders to exercise such warrants on a cashless basis (even if a registration statement covering the shares of common stock issuable upon exercise of such warrants is not effective). However, once any of the foregoing warrants are transferred from PGM or its affiliates, these arrangements will no longer apply. Furthermore, because the private warrants will be issued in a private transaction, the holders and their transferees will be allowed to exercise the private warrants for cash even if a registration statement covering the shares of common stock issuable upon exercise of such warrants is not effective and receive unregistered shares of common stock.
Dividends
We have not paid any cash dividends on our common stock to date and do not intend to pay cash dividends prior to the completion of a business combination. The payment of cash dividends in the future will be dependent upon our revenues and earnings, if any, capital requirements and general financial condition subsequent to completion of a business combination. The payment of any cash dividends subsequent to a business combination will be within the discretion of our Board at such time. In addition, our Board is not currently contemplating and does not anticipate declaring any stock dividends in the foreseeable future. Further, if we incur any indebtedness, our ability to declare dividends may be limited by restrictive covenants we may agree to in connection therewith.
| 281 |
Our Transfer Agent, Rights and Warrant Agent
The transfer agent for our common stock, rights agent for our rights and warrant agent for our warrants is Continental Stock Transfer & Trust Company, located at One State Street, 30th Floor, New York, New York 10004.
Certain Anti-Takeover Provisions of Delaware Law and Globalink Charter and By-Laws
We are subject to the provisions of Section 203 of the DGCL, regulating corporate takeovers upon completion of this offering. This statute prevents certain Delaware corporations, under certain circumstances, from engaging in a “business combination” with:
| ● | a stockholder who owns 10% or more of our outstanding voting stock (otherwise known as an “interested stockholder”); | |
| ● | an affiliate of an interested stockholder; or | |
| ● | an associate of an interested stockholder, for three years following the date that the stockholder became an interested stockholder. |
A “business combination” includes a merger or sale of more than 10% of our assets. However, the above provisions of Section 203 do not apply if:
| ● | our Board approves the transaction that made the stockholder an “interested stockholder,” prior to the date of the transaction; | |
| ● | after the completion of the transaction that resulted in the stockholder becoming an interested stockholder, that stockholder owned at least 85% of our voting stock outstanding at the time the transaction commenced, other than statutorily excluded shares of common stock; or | |
| ● | on or subsequent to the date of the transaction, the business combination is approved by our Board and authorized at a meeting of our stockholders, and not by written consent, by an affirmative vote of at least two-thirds of the outstanding voting stock not owned by the interested stockholder. |
Staggered Board of Directors
Globalink Charter provides that our Board is classified into three classes of directors. As a result, in most circumstances, a person can gain control of our Board only by successfully engaging in a proxy contest at two or more annual meetings.
Special Meeting of Stockholders
Our bylaws provide that Special Meetings of our stockholders may be called only by resolution of the Board, or by the Chairman or the Chief Executive Officer.
Advance Notice Requirements for Stockholder Proposals and Director Nominations
Our bylaws provide that stockholders seeking to bring business before our annual meeting of stockholders, or to nominate candidates for election as directors at our annual meeting of stockholders must provide timely notice of their intent in writing. To be timely, a stockholder’s notice will need to be delivered to our principal executive offices not later than the close of business on the 90th day nor earlier than the opening of business on the 120th day prior to the scheduled date of the annual meeting of stockholders. Our bylaws also specify certain requirements as to the form and content of a stockholders’ meeting. These provisions may preclude our stockholders from bringing matters before our annual meeting of stockholders or from making nominations for directors at our annual meeting of stockholders.
Exclusive Forum for Certain Lawsuits
Globalink Charter provides that, unless we consent in writing to the selection of an alternative forum, the Court of Chancery shall, to the fullest extent permitted by law, be the sole and exclusive forum for any (1) derivative action or proceeding brought on behalf of our company, (2) action asserting a claim of breach of a fiduciary duty owed by any director, officer, employee or agent of our company to our company or our stockholders, or any claim for aiding and abetting any such alleged breach, (3) action asserting a claim against our company or any director or officer of our company arising pursuant to any provision of the DGCL or Globalink Charter or Globalink’s bylaws, or (4) action asserting a claim against us or any director or officer of our company governed by the internal affairs doctrine except for, as to each of (1) through (4) above, any claim (A) as to which the Court of Chancery determines that there is an indispensable party not subject to the jurisdiction of the Court of Chancery (and the indispensable party does not consent to the personal jurisdiction of the Court of Chancery within ten days following such determination), (B) which is vested in the exclusive jurisdiction of a court or forum other than the Court of Chancery, or (C) arising under the federal securities laws, including the Securities Act as to which the Court of Chancery and the federal district court for the District of Delaware shall concurrently be the sole and exclusive forums. Notwithstanding the foregoing, the inclusion of such provision in Globalink Charter will not be deemed to be a waiver by our stockholders of our obligation to comply with federal securities laws, rules and regulations, and the provisions of this paragraph will not apply to suits brought to enforce any liability or duty created by the Exchange Act, or any other claim for which the federal district courts of the United States of America shall be the sole and exclusive forum. Although we believe this provision benefits us by providing increased consistency in the application of Delaware law in the types of lawsuits to which it applies, the provision may have the effect of discouraging lawsuits against our directors and officers. Furthermore, the enforceability of choice of forum provisions in other companies’ certificates of incorporation has been challenged in legal proceedings, and it is possible that a court could find these types of provisions to be inapplicable or unenforceable.
| 282 |
DESCRIPTION OF PUBCO’S SECURITIES
PubCo, or Alps Global Holding Pubco, is a Cayman Islands exempted company and its affairs are governed by the memorandum and articles of association, as amended and restated from time to time, and Companies Act, and the common law of the Cayman Islands.
PubCo currently has only one class of issued ordinary shares, which have identical rights in all respects and rank equally with one another. The current share capital of PubCo is US$50,000 divided into 500,000,000 ordinary shares of par value US$0.0001 each.
PubCo Ordinary Shares
The following includes a summary of the terms of PubCo ordinary shares, based on its memorandum and articles of association and Cayman Islands law. In connection with the Acquisition Merger, PubCo shall amend its memorandum and articles of association, which amendment is referred to herein as the “Memorandum and Articles of Association” and shall sub-divide its authorized share capital such that, the authorized share capital of the post-closing company will be US$50,000 divided into 500,000,000 shares comprising (i) 495,000,000 PubCo ordinary shares of US$0.0001 par value each, and (ii) 5,000,000 PubCo preferred shares of US$0.0001 par value each.
General. PubCo’s authorized share capital pre-closing and post is US$50,000 divided into 500,000,000 shares comprising (i) 495,000,000 PubCo ordinary shares of US$0.0001 par value each, and (ii) 5,000,000 PubCo preferred shares of US$0.0001 par value each. All of PubCo’s issued and outstanding ordinary shares are fully paid and non-assessable. Unless the board of directors determine otherwise, each holder of PubCo ordinary shares will not receive a certificate in respect of such ordinary shares. PubCo may not issue share or warrant to bearer. PubCo’s shareholders who are non-residents of the Cayman Islands may freely hold and transfer their ordinary shares.
Dividends. The holders of PubCo ordinary shares are entitled to such dividends as may be declared by its board of directors subject to its Memorandum and Articles of Association and the Companies Act. In addition, PubCo’s shareholders may by ordinary resolution declare a dividend, but no dividend may exceed the amount recommended by its directors. Dividends may also be declared and paid out of share premium account with the sanction of an ordinary resolution or any other lawfully available fund or account which can be authorized for this purpose in accordance with the Companies Act. No dividend may be declared and paid unless PubCo’s directors determine that, immediately following the date on which the dividend is proposed to be paid, PubCo will be able to pay its debts as they fall due in the ordinary course of business and PubCo has funds lawfully available for such purpose. Holders of PubCo ordinary shares will be entitled to the same amount of dividends, if declared.
Voting Rights. A resolution put to the vote of a meeting of shareholders shall be decided on a poll. Subject to any rights or restrictions as to voting attached to any shares, unless any share carries special voting rights, on a poll, every shareholder who is present in person and every person representing a shareholder by proxy shall have one vote for each share of which he or the person represented by proxy is the holder. In addition, all shareholders holding shares of a particular class are entitled to vote at a meeting of the holders of that class of shares. Votes may be given either personally or by proxy.
A quorum required for a meeting of shareholders consists of one or more shareholders holding not less than one-third of the votes attaching to the issued and outstanding shares entitled to vote at general meetings present in person or by proxy or, if a corporation or other non-natural person, by its duly authorized representative. As a Cayman Islands exempted company, PubCo is not obliged by the Companies Act to call shareholders’ annual general meetings. PubCo’s Memorandum and Articles of Association provide that PubCo may (but are not obliged to) in each year hold a general meeting as its annual general meeting in which case PubCo will specify the meeting as such in the notices calling it, and the annual general meeting will be held at such time and place as may be determined by its directors. We, however, will hold an annual shareholders’ meeting during each fiscal year, as required by the Nasdaq Listing Rules. Each general meeting, other than an annual general meeting, shall be an extraordinary general meeting. Shareholders’ annual general meetings and any other general meetings of PubCo’s shareholders may be called by the majority of all its directors or all of its Class A directors or, upon a requisition of one or more shareholders who together holding at least 10% of the rights to vote at such general meeting, in which case the directors are obliged to call such meeting and to put the resolutions so requisitioned to a vote at such meeting.
| 283 |
An ordinary resolution to be passed at a meeting by the shareholders requires the affirmative vote of a simple majority of the votes cast by those shareholders entitled to vote who are present in person or by proxy at a general meeting, while a special resolution also requires the affirmative vote of no less than two-thirds of the votes cast by those shareholders entitled to vote who are present in person or by proxy at a general meeting. A special resolution will be required for important matters such as a change of name or making changes to PubCo’s Memorandum and Articles of Association.
Transfer of Ordinary Shares. Subject to any applicable requirements set forth in the PubCo’s Memorandum and Articles of Association and provided that a transfer of ordinary shares complies with applicable rules of the Nasdaq, a shareholder may transfer ordinary shares to another person by completing an instrument of transfer in a common form or in a form prescribed by Nasdaq or in any other form approved by the directors, executed:
| ● | where the ordinary shares are fully paid, by or on behalf of that shareholder; and | |
| ● | where the ordinary shares are partly paid, by or on behalf of that shareholder and the transferee. |
The transferor shall be deemed to remain the holder of an ordinary share until the name of the transferee is entered into our register of members.
Where the ordinary shares in question are not listed on or subject to the rules of the Nasdaq, the board of directors may, in its absolute discretion, decline to register any transfer of any ordinary share that has not been fully paid up or is subject to a company lien. The board of directors may also decline to register any transfer of such Ordinary Share unless:
| ● | the instrument of transfer is lodged with the company, accompanied by the certificate for the ordinary shares to which it relates and such other evidence as the board of directors may reasonably require to show the right of the transferor to make the transfer; |
| ● | the instrument of transfer is in respect of only one class of ordinary shares; |
| ● | the instrument of transfer is properly stamped, if required; |
| ● | the ordinary share transferred is fully paid and free of any lien in favor of the company; |
| ● | any fee related to the transfer has been paid to us; and |
| ● | the transfer is not more than four joint holders. |
If the directors refuse to register a transfer, they are required, within one month after the date on which the instrument of transfer was lodged, to send to each of the transferor and the transferee notice of such refusal.
The registration of transfers may, on 14 days’ notice being given by advertisement in such one or more newspapers or by electronic means, be suspended and our register of members closed at such times and for such periods as the directors may, in their absolute discretion, from time to time determine. The registration of transfers, however, may not be suspended, and the register may not be closed, for more than 30 days in any year.
Liquidation. If the PubCo is wound up, the shareholders may, subject to the articles and any other sanction required by the Companies Act, pass a special resolution allowing the liquidator to do either or both of the following:
| ● | to divide in specie among the shareholders the whole or any part of assets of the company and, for that purpose, to value any assets and to determine how the division shall be carried out as between the shareholders or different classes of shareholders; and | |
| ● | to vest the whole or any part of the assets in trustees for the benefit of shareholders and those liable to contribute to the winding up. |
| 284 |
The directors have the authority to present a petition for our winding up to the Grand Court of the Cayman Islands on our behalf without the sanction of a resolution passed at a general meeting.
Redemption, Repurchase and Surrender of Ordinary Shares. PubCo may issue shares on terms that such PubCo ordinary shares are subject to redemption, at PubCo’s option or at the option of the holders thereof, on such terms and in such manner as may be determined, before the issue of such PubCo ordinary shares, by a board resolution of PubCo’s directors. PubCo may also repurchase any of its PubCo ordinary shares in such manner and on such other terms as agreed between the board of directors and the relevant shareholder. Under the Companies Act, the redemption or repurchase of any share may be paid out of PubCo’s profits, out of any combination of capital, profits and the proceeds of a fresh issue of shares if PubCo can, immediately following the date on which the payment is proposed to be paid, pay its debts as they fall due in the ordinary course of business.
In addition, under the Companies Act no such PubCo ordinary shares may be redeemed or repurchased (a) unless it is fully paid up, or (b) if such redemption or repurchase would result in there being no shares outstanding, other than shares held as treasury shares.
General Meetings of Shareholders. PubCo may, but shall not (unless required by the rules and regulations of the Nasdaq or any other national securities exchange on which the Shares are listed for trading) be obliged to, hold an annual general meeting in each year at such time and place as the board of directors of PubCo will determine. At least five (5) clear days’ notice shall be given for any general meeting. The majority of all the directors or all the Class A directors of PubCo may call general meetings, and they shall on a shareholders’ requisition forthwith proceed to convene an extraordinary general meeting. One or more shareholders who together hold not less than one-third of the issued and outstanding PubCo ordinary shares entitled to attend and vote at such meeting, being individuals present in person or by proxy shall be a quorum.
Variations of Rights of Shares. If at any time PubCo ordinary shares capital is divided into different classes of shares, the rights attached to any class of shares (unless otherwise provided by the terms of issue of the shares of that class) may be varied only with consent in writing of the holders of not less than two-thirds of the issued shares of that class, or with the approval of a special resolution passed by at a separate general meeting of the members holding the issued shares of such class. Unless the terms on which a class of shares was issued state otherwise, the rights conferred upon the holders of the shares of any class issued shall not be deemed to be varied by the creation or issue of further shares ranking pari passu therewith.
Inspection of Books and Records. The board of directors of PubCo or the shareholders by ordinary resolution will determine whether, to what extent, at what times and places and under what conditions or regulations the accounts and books of PubCo will be open to the inspection by PubCo shareholders, and no PubCo shareholder will otherwise have any right of inspecting any account or book or document of PubCo except as required by the Companies Act.
Changes in Capital
PubCo may from time to time by ordinary resolution:
| ● | increase its share capital by new shares of the amount fixed by that ordinary resolution; | |
| ● | consolidate and divide all or any of its share capital into shares of a larger amount than its existing shares; | |
| ● | convert all or any of its paid-up shares into stock and reconvert that stock into paid-up shares of any denomination; | |
| ● | sub-divide its shares or any of them into shares of a smaller amount; or | |
| ● | cancel any shares that at the date of the passing of the ordinary resolution have not been taken or agreed to be taken by any person and diminish the amount of its share capital by the amount of the shares so cancelled. |
Exempted Company
PubCo will be an exempted company with limited liability incorporated under the laws of Cayman Islands. The Companies Act distinguishes between ordinary resident companies and exempted companies. Any company that is registered in the Cayman Islands but conducts business mainly outside of the Cayman Islands may apply to be registered as an exempted company. The requirements for an exempted company are essentially the same as for an ordinary company except for the exemptions and privileges listed below:
| ● | an exempted company’s register of members is not open to inspection; | |
| ● | an exempted company does not have to hold an annual general meeting; | |
| ● | an exempted company may issue no par value shares; | |
| ● | an exempted company may obtain an undertaking against the imposition of any future taxation (such undertakings are usually given for 20 years in the first instance); | |
| ● | an exempted company may register by way of continuation in another jurisdiction and be deregistered in the Cayman Islands; | |
| ● | an exempted company may register as a limited duration company; and | |
| ● | an exempted company may register as a segregated portfolio company. |
| 285 |
SHARES ELIGIBLE FOR FUTURE SALE
Business Combination Shares
According to the Memorandum and Articles of Association of PubCo, the current authorized share capital of the PubCo is US$50,000 divided into 500,000,000 shares comprising (i) 495,000,000 PubCo ordinary shares of US$0.0001 par value each; and (ii) 5,000,000 preferred shares of US$0.0001 par value each. All of the PubCo ordinary shares issued in connection with the Redomestication Merger will be freely transferable by persons other than PubCo’s “affiliates” without restriction under the Securities Act, subject to the restrictions detailed below.
The PubCo ordinary shares issued in connection with the Acquisition Merger will also be registered at the Closing, but will be subject to the Lock-up Agreements described below. Sales of substantial amounts of PubCo ordinary shares in the public market could adversely affect prevailing market prices of the PubCo ordinary shares. Prior to the Business Combination, there has been no public market for PubCo ordinary shares. PubCo intends to apply for listing of the PubCo ordinary shares and PubCo warrants on Nasdaq, but it cannot be assured that a regular trading market will develop in the PubCo ordinary shares or PubCo warrants.
Transfer of PubCo Ordinary Shares
Subject to applicable securities laws in relevant jurisdictions and the PubCo Memorandum and Articles of Association, a shareholder may transfer ordinary shares to another person by completing an instrument of transfer in a common form or in a form prescribed by Nasdaq or in any other form approved by the directors, executed: (i) where the ordinary shares are fully paid, by or on behalf of that shareholder; and (ii) where the ordinary shares are partly paid, by or on behalf of that shareholder and the transferee. The transferor shall be deemed to remain the holder of an ordinary share until the name of the transferee is entered into our register of members.
Lock-up Agreements
At the Closing of the Business Combination, certain Alps Holdco Shareholders and certain Globalink stockholders shall execute Lock-up Agreements, pursuant to which such holders shall agree, subject to certain customary exceptions, not to (i) sell, offer to sell, contract or agree to sell, pledge or otherwise dispose of, directly or indirectly, any Lock-Up Shares held by them, (ii) enter into a transaction that would have the same effect, (iii) enter into any swap, hedge or other arrangement that transfers to another, in whole or in part, any of the economic consequences of ownership of the Lock-Up Shares or otherwise or engage in any short sales or other arrangement with respect to the Lock-Up Shares or (iv) publicly announce any intention to effect any transaction specified in clause (i) or (ii) for the duration of the six month Lock-Up Period.
Regulation S
Regulation S under the Securities Act provides an exemption from registration requirements in the United States for offers and sales of securities that occur outside the United States. Rule 903 of Regulation S provides the conditions to the exemption for a sale by an issuer, a distributor, their respective affiliates or anyone acting on their behalf, while Rule 904 of Regulation S provides the conditions to the exemption for a resale by persons other than those covered by Rule 903. In each case, any sale must be completed in an offshore transaction, as that term is defined in Regulation S, and no directed selling efforts, as that term is defined in Regulation S, may be made in the United States.
PubCo is a foreign issuer as defined in Regulation S. As a foreign issuer, securities that PubCo sells outside the United States pursuant to Regulation S are not considered to be restricted securities under the Securities Act, and, subject to the offering restrictions imposed by Rule 903, are freely tradable without registration or restrictions under the Securities Act, unless the securities are held by PubCo’s affiliates. Generally, subject to certain limitations, holders of PubCo’s restricted shares who are not affiliates of PubCo or who are affiliates of PubCo by virtue of their status as an officer or director of PubCo may, under Regulation S, resell their restricted shares in an “offshore transaction” if none of the shareholder, its affiliate nor any person acting on their behalf engages in directed selling efforts in the United States and, in the case of a sale of PubCo restricted shares by an officer or director who is an affiliate of PubCo solely by virtue of holding such position, no selling commission, fee or other remuneration is paid in connection with the offer or sale other than the usual and customary broker’s commission that would be received by a person executing such transaction as agent. Additional restrictions are applicable to a holder of PubCo restricted shares who will be an affiliate of PubCo other than by virtue of his or her status as an officer or director of PubCo.
| 286 |
PubCo is not claiming the potential exemption offered by Regulation S in connection with the offering of newly issued shares outside the United States and will register all of the newly issued shares under the Securities Act.
Rule 144
All of PubCo ordinary shares that will be outstanding upon the consummation of the Business Combination, other than those equity shares issued and registered in connection with the Business Combination, are “restricted securities” as that term is defined in Rule 144 under the Securities Act and may be sold publicly in the United States only if they are subject to an effective registration statement under the Securities Act or pursuant to an exemption from the registration requirement such as those provided by Rule 144 and Rule 701 promulgated under the Securities Act. In general, beginning 90 days after the date of this proxy statement/prospectus, a person (or persons whose shares are aggregated) who, at the time of a sale, is not, and has not been during the three months preceding the sale, an affiliate of PubCo and has beneficially owned PubCo’s restricted securities for at least six months will be entitled to sell the restricted securities without registration under the Securities Act, subject only to the availability of current public information about PubCo. Persons who are affiliates of PubCo and have beneficially owned PubCo’s restricted securities for at least six months may sell a number of restricted securities within any three-month period that does not exceed the greater of the following:
| ● | 1% of the then issued equity shares of the same class which, immediately after the Business Combination, will equal PubCo ordinary shares (not including the ordinary shares to be issued in the PIPE Investment and assuming no redemption scenario); or | |
| ● | the average weekly trading volume of PubCo ordinary shares of the same class during the four calendar weeks preceding the date on which notice of the sale is filed with the SEC. |
Sales by affiliates of PubCo under Rule 144 are also subject to certain requirements relating to manner of sale, notice and the availability of current public information about PubCo.
Rule 701
In general, under Rule 701 of the Securities Act, any of PubCo’s employees, directors, officers, consultants or advisors, other than PubCo’s affiliates, who purchased shares from PubCo in connection with a qualified compensatory stock plan or other written agreement in compliance with Rule 701 before the effective date of a registration statement under the Securities Act is entitled to rely on Rule 701 to resell such shares in reliance on Rule 144. An affiliate of PubCo can resell shares in reliance on Rule 144 without having to comply with the holding period requirements of Rule 144, and a non-affiliate of PubCo can resell shares in reliance on Rule 144 without having to comply with the holding period requirements of Rule 144 and without regard to the volume of such sales or the availability of public information about PubCo.
The SEC has indicated that Rule 701 will apply to typical stock options granted by an issuer before it becomes subject to the reporting requirements of the Exchange Act, along with the shares acquired upon exercise of such options, including exercises after an issuer becomes subject to the reporting requirements of the Exchange Act.
Registration Rights
PubCo has agreed to give holders of certain restricted securities registration rights to facilitate the resale of such restricted securities. Additional details of these rights can be found under the section titled “The Acquisition Merger Proposal (Proposal 2) — The Acquisition Merger Proposal — Certain Related Agreements — Amended and Restated Registration Rights Agreement.”
| 287 |
CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS
Globalink Related Person Transactions
On August 19, 2021, the Sponsor purchased an aggregate of 2,875,000 shares of Globalink common stock for an aggregate purchase price of $25,000, or approximately $0.009 per share. We refer to these shares throughout this proxy statement/prospectus as the “insider shares.” The number of insider shares issued was determined based on the expectation that such insider shares would represent 20% of the outstanding shares after the IPO (not including the shares underlying the private units). On October 14, 2021, the Sponsor transferred 15,000 insider shares to Globalink’s Chief Executive Officer, 10,000 insider shares to Mr. Cliff (Ming Hong) Chong, Globalink’s former Chief Financial Officer, and 5,000 insider shares to each of the independent directors at their original purchase price.
Globalink’s private investor and an affiliate of the Sponsor, PGM, purchased an aggregate of 570,000 private units at a price of $10.00 per unit ($5,700,000 in aggregate) in a private placement that closed simultaneously with the closing of the IPO. The private units are identical to the units sold in the IPO. Additionally, Globalink’s private investor has agreed not to transfer, assign or sell any of the private units or underlying securities (except to the same permitted transferees as the insider shares and provided the transferees agree to the same terms and restrictions as the permitted transferees of the insider shares must agree to, each as described above) until the completion of Globalink’s initial business combination.
In order to meet its working capital needs following the consummation of the IPO, Globalink’s insiders, officers and directors may, but are not obligated to, loan funds to Globalink, from time to time or at any time, in whatever amount they deem reasonable in their sole discretion. Each loan would be evidenced by a promissory note. The notes would either be paid upon consummation of Globalink’s initial business combination, without interest, or, at the lender’s discretion, up to $1,500,000 of the notes may be converted upon consummation of Globalink’s business combination into additional private units at a price of $10.00 per unit. Globalink stockholders have approved the issuance of the private units upon conversion of such notes, to the extent the holder wishes to so convert such notes at the time of the consummation of Globalink’s initial business combination. If Globalink does not complete a business combination, any outstanding loans from Globalink’s insiders or their affiliates, will be repaid only from amounts remaining outside the Trust Account, if any. As of December 31, 2022 and 2023, March 31, 2024 and September 30, 2024, no loans from Globalink’s insiders, officers and directors were outstanding to Globalink.
The holders of the insider shares, as well as the holders of the private units and any units Globalink’s insiders or their affiliates may be issued upon conversion of working capital loans or extension loans made to Globalink (and any securities underlying the private units or units issued upon conversion of the working capital loans or extension loans), will be entitled to registration rights pursuant to the terms of a registration rights agreement entered into with such holders. The holders of a majority of these securities are entitled to make up to two demands that Globalink registers such securities. The holders of the majority of the insider shares can elect to exercise these registration rights at any time commencing three months prior to the date on which these shares of common stock are to be released from escrow. The holders of a majority of the private units and any units issued upon conversion of working capital loans or extension loans made to Globalink can elect to exercise these registration rights at any time after Globalink consummates a business combination. In addition, the holders have certain “piggy-back” registration rights with respect to registration statements filed subsequent to the consummation of Globalink’s initial business combination. Globalink will bear the expenses incurred in connection with the filing of any such registration statements.
The Sponsor has agreed that, commencing on the date of the IPO prospectus through the earlier of the consummation of Globalink’s initial business combination or liquidation, it will make available to Globalink certain general and administrative services, including office space, utilities and administrative support, as Globalink may require from time to time. Globalink agreed to pay $10,000 per month for these services. However, pursuant to the terms of such agreement, Globalink may delay payment of such monthly fee upon a determination by its audit committee that Globalink lacks sufficient funds held outside the Trust Account to pay actual or anticipated expenses in connection with its initial business combination. Such unpaid amount accrues without interest and will be due and payable no later than the date of the consummation of Globalink’s initial business combination. On each of September 5, 2023, September 29, 2023 and November 7, 2023, an affiliate of the Company’s sponsor advanced $130,000 to the Company, for a total advance of $390,000. The $390,000 advance to fund trust extension deposits is reflected in “Due to related parties” on the consolidated balance sheets. As of December 31, 2023 and 2022 and March 31, 2024, the balance of unpaid amount due to the Sponsor was $607,000, $127,000, and $607,000, respectively. On September 30, 2023, Globalink terminated the administrative services agreement. As a result, Globalink is not required to pay the sponsor $10,000 monthly starting from September 30, 2023.
| 288 |
Simultaneously with the closing of the IPO, Globalink consummated the sale of 517,500 units, or private units, at a price of $10.00 per private unit in a private placement to PGM, generating gross proceeds of $5,175,000.
Simultaneously with the exercise of the over-allotment, Globalink consummated a private sale of an additional 52,500 private units to PGM at a price of $10.00 per unit, generating additional gross proceeds of $525,000. Since Chardan’s over-allotment was exercised in full, the Sponsor did not forfeit any Founder Shares.
On March 3, 2023, Globalink entered into a promissory note subscription term sheet with PGM for an amount of $390,000 for the purpose of extension fees payment. The promissory note bears an interest of 6% per annum and repayable upon consummation of an initial business combination. As of December 31, 2023, the full $390,000 had been borrowed and no amount was available under this note for borrowing.
On March 23, 2023, Globalink entered into a promissory note subscription term sheet with PGM for an amount of up to $250,000 for working capital needs. The promissory note bears an interest of 6% per annum and repayable upon consummation of an initial business combination. As of December 31, 2023, the full $250,000 had been borrowed and no amount was available under this note for borrowing.
On June 2, 2023, Globalink entered into a promissory note subscription term sheet with PGM for an amount of up to $700,000 for working capital needs. The promissory note bears an interest of 6% per annum and repayable upon consummation of an initial business combination. As of December 31, 2023, the full $700,000 had been borrowed and no amount was available under this note for borrowing.
On October 10, 2023, Globalink entered into a promissory note subscription term sheet with PGM for an amount of $250,000 for the purpose of working capital. The promissory note bears an interest of 6% per annum and repayable upon consummation of an initial business combination. As of December 31, 2023, the full $250,000 had been borrowed and no amount was available under this note for borrowing.
On December 8, 2023, Globalink entered into a promissory note subscription term sheet with PGM for an amount of $110,000 for the purpose of working capital. The promissory note bears an interest of 6% per annum and repayable upon consummation of an initial business combination. As of December 31, 2023, the full $110,000 had been borrowed and no amount was available under this note for borrowing.
On each of September 5, 2023, September 29, 2023 and November 11, 2023, an affiliate of the Sponsor, Ng Yan Xun, advanced $130,000 to Globalink, for a total advance of $390,000. As of December 31, 2023, $390,000 of advance is reflected in “Due to Affiliate” on Globalink’s consolidated balance sheet.
On January 5, 2024, Globalink entered into a promissory note subscription term sheet with PGM for an amount of $250,000 for the purpose of working capital.
On January 25, 2024, Globalink entered into a promissory note subscription term sheet with PGM for an amount of $300,000 for the purpose of working capital.
On February 22, 2024, Globalink entered into a promissory note subscription term sheet with PGM for an amount of $300,000 for the purpose of working capital.
On April 4, 2024, Globalink entered into a promissory note subscription term sheet with PGM for an amount of $300,000 for the purpose of working capital.
On June 5, 2024, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount of $400,000 for the purpose of working capital. The promissory note bears an interest of 6% per annum and repayable upon consummation of an initial Business Combination. As of September 30, 2024, the full $400,000 had been borrowed and no amount was available under this note for borrowing.
On August 14, 2024, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount of $300,000 for the purpose of working capital. The promissory note bears an interest of 6% per annum and repayable upon consummation of an initial Business Combination. As of September 30, 2024, the full $300,000 had been borrowed and no amount was available under this note for borrowing.
Globalink’s total promissory note liability as of December 31, 2023 and 2022, March 31, 2024 and September 30, 2024 is $1,757,255, zero, $2,640,649 and $3,736,066, respectively.
GL Sponsor LLC, the Sponsor, is the record holder of 2,835,000 shares of Globalink common stock. GL Sponsor LLC is managed by Ng Yan Xun. Ng Yan Xun may be deemed to beneficially own the shares held by the Sponsor by virtue of his control of the Sponsor. Ng Yan Xun disclaims beneficial ownership of the shares held by the Sponsor except to the extent of his pecuniary interest therein.
| 289 |
Other than the fees described above, no compensation or fees of any kind, including finder’s fees, consulting fees or other similar compensation, will be paid to Globalink’s insiders or any of the members of its management team, for services rendered to Globalink prior to, or in connection with the consummation of its initial business combination (regardless of the type of transaction that it is). However, such individuals will receive reimbursement for any out-of-pocket expenses incurred by them in connection with activities on behalf of Globalink, such as identifying potential target businesses, performing business due diligence on suitable target businesses and business combinations as well as traveling to and from the offices, plants or similar locations of prospective target businesses to examine their operations. There is no limit on the amount of out-of-pocket expenses reimbursable by Globalink; provided, however, that to the extent such expenses exceed the available proceeds not deposited in the Trust Account and the interest income earned on the amounts held in the Trust Account, such expenses would not be reimbursed by Globalink unless Globalink consummate an initial business combination.
After the initial business combination, members of Globalink’s management team who remain with Globalink, if any, may be paid consulting, Board, management or other fees from the Combined Company with any and all amounts being fully disclosed to shareholders, to the extent then known, in the proxy solicitation materials furnished to Globalink’s stockholders. It is unlikely the amount of such compensation will be known at the time of a stockholder meeting held to consider Globalink’s initial business combination, as it will be up to the directors of the post-combination business to determine executive and director compensation. In this event, such compensation will be publicly disclosed at the time of its determination in a current report as required by the SEC.
All ongoing and future transactions between Globalink and any of its officers and directors or their respective affiliates will be on terms believed by Globalink to be no less favorable to Globalink than are available from unaffiliated third parties. Such transactions will require prior approval by Globalink’s audit committee and a majority of its uninterested independent directors, in either case who had access, at its expense, to its attorneys or independent legal counsel. Globalink will not enter into any such transaction unless its audit committee and a majority of its disinterested independent directors determine that the terms of such transaction are no less favorable to Globalink than those that would be available to Globalink with respect to such a transaction from unaffiliated third parties.
Agreements Relating to the Business Combination
In connection with the execution of the Merger Agreement, Globalink, Alps Holdco, and each of certain stockholders of Globalink have entered into a Globalink Support Agreement, pursuant to which the stockholders of Globalink that are parties to the Globalink Support Agreements have agreed to vote all shares of Globalink common stock beneficially owned by them in favor of the transactions contemplated by the Merger Agreement.
In connection with the Business Combination, certain Globalink stockholders will enter into an Amended and Restated Registration Rights Agreement with PubCo at the Closing. See the subsection entitled “The Acquisition Merger Proposal (Proposal 2)—Certain Related Agreements—Amended and Restated Registration Rights Agreement” for more information relating to the Amended and Restated Registration Rights Agreement.
Globalink’s Related Party Policy
Globalink’s Code of Ethics requires Globalink to avoid, wherever possible, all related party transactions that could result in actual or potential conflicts of interests, except under guidelines approved by the Board (or the audit committee). Related party transactions are defined as transactions in which (1) the aggregate amount involved will or may be expected to exceed $120,000 in any calendar year, (2) Globalink or any of its subsidiaries is a participant, and (3) any (a) executive officer, director or nominee for election as a director, (b) greater than 5% beneficial owner of shares of Globalink common stock, or (c) immediate family member, of the persons referred to in clauses (a) and (b), has or will have a direct or indirect material interest (other than solely as a result of being a director or a less than 10% beneficial owner of another entity). A conflict-of-interest situation can arise when a person takes actions or has interests that may make it difficult to perform his or her work objectively and effectively. Conflicts of interest may also arise if a person, or a member of his or her family, receives improper personal benefits as a result of his or her position.
Globalink also requires each of its directors and executive officers to annually complete a directors’ and officers’ questionnaire that elicits information about related party transactions.
These procedures are intended to determine whether any such related party transaction impairs the independence of a director or presents a conflict of interest on the part of a director, employee or officer.
| 290 |
To further minimize conflicts of interest, Globalink has agreed not to consummate its initial business combination with an entity that is affiliated with any of its insiders, officers or directors unless Globalink has obtained an opinion from an independent investment banking firm and the approval of a majority of its disinterested and independent directors (if Globalink has any at that time) that the business combination is fair to Globalink’s unaffiliated stockholders from a financial point of view. In no event will Globalink’s insiders, or any of the members of its management team be paid any finder’s fee, consulting fee or other similar compensation prior to, or for any services they render in order to effectuate, the consummation of Globalink’s initial business combination (regardless of the type of transaction that it is).
Certain Relationships and Transactions of Alps
On March 31, 2021, Alps Global Holding Berhad entered into a Lease Agreement with Celestialab Sdn. Bhd. to lease a patent to Celestialab from Alps for a ten-year term with an annual payment of RM1,500,000.00.
On April 1, 2021, Alps Global Holding Berhad entered into a Management Service Agreement with Alpscap Berhad for the provision of management services for the establishment, development, operation, strategy formulation, financial, and human resources management for an initial term of 36 months, with a service fee of RM12,000.00 annually for the first year and RM24,000.00 annually for the second and third years.
On February 1, 2022, TMC Global Holdings Sdn. Bhd. entered into a Scientific Research Cooperation Agreement with Ding KeXiang, wherein the parties agreed to collaborate on a scientific research project focused on anti-aging between China and Malaysia for a term of three (3) years, from February 1, 2022 to January 31, 2025.
On November 1, 2022, Alps Global Holding Berhad entered into a Management Service Agreement with Celebre Pro Medic Sdn. Bhd. for the provision of management services for the establishment, development, operation, strategy formulation, financial, and human resources management for an initial term of 24 months, with a service fee of RM60,000.00 annually.
On November 1, 2022, Alps Global Holding Berhad entered into a Management Service Agreement with Alps Wellness Centre Sdn. Bhd. for the provision of management services for the establishment, development, operation, strategy formulation, financial, and human resources management for an initial term of 24 months, with a service fee of RM60,000.00 annually.
On November 1, 2022, Alps Global Holding Berhad entered into a Management Service Agreement with Alps Globemedic Sdn. Bhd. for the provision of management services for the establishment, development, operation, strategy formulation, financial, and human resources management for an initial term of 24 months, with a service fee of RM60,000.00 annually.
On April 1, 2021, Alps Global Holding Berhad entered into a Management Service Agreement with Mont Life (M) Sdn. Bhd. for the provision of management services for the establishment, development, operation, strategy formulation, financial, and human resources management for an initial term of 36 months, with a service fee of RM12,000.00 annually for the first year and RM24,000.00 annually for the second and third years.
On November 1, 2022, Alps Global Holding Berhad entered into a Management Service Agreement with Celetialab Sdn. Bhd., for the provision of management services for the establishment, development, operation, strategy formulation, financial, and human resources management for an initial term of 24 months, with a service fee of RM60,000.00 annually.
On November 1, 2022, Alps Global Holding Berhad entered into a Management Service Agreement with TMC Global Holdings Sdn. Bhd., for the provision of management services for the establishment, development, operation, strategy formulation, financial, and human resources management for an initial term of 24 months, with a service fee of RM96,000.00 annually.
On February 15, 2023, a Share Subscription Agreement was entered into between Alps Global Holding Berhad, Dr. Tham Seng Kong, Cilo Cybin Holdings Limited and Gabriel Theron for the subscription of Cilo Cybin Holdings Limited’s ordinary shares by Alps Global Holding Berhad and Dr. Tham Seng Kong for a total consideration of USD3,000,000.00.
| 291 |
On August 1, 2023, TMC Global Holdings Sdn. Bhd. entered into a Service Agreement with Alps Globemedic Sdn. Bhd. for the provision of medical aesthetics, wellness, cosmetic and plastic surgery, cellular therapy related products and services, as well as facility to use the premises for a term of two (2) years.
On September 11, 2023, Dr. Tham Seng Kong, Mohd. Razef Bin Abdullah, Poon Kian Huat (collectively, “Vendors”) and Alpscap Berhad entered into a Sale Shares (Share Swap) Agreement for sale of all shares held by the Vendors in Alps Insurance PCC Inc. to Alpscap Berhad.
On January 11, 2024, MyGenome Sdn. Bhd. entered into a Service Agreement with Celetialab Sdn. Bhd., wherein MyGenome Sdn. Bhd. provides Microbiology Examination services including Bacterial Endotoxin, Sterility Test, and Mycoplasma tests, while Celestialab Sdn. Bhd. is responsible for providing information, evaluating service provider competence, and ensuring proper sample packaging and labeling.
On March 3, 2024, Alps Global Holding Berhad entered into a Patent License Agreement with Ding KeXiang, wherein Ding KeXiang shall grant to Alps Global Holding Berhad the rights to exploit, use, license and sublicense all the patents owned by Ding KeXiang and develop, produce, manufacture, market, sell, import and distribute the products and practice the processes under such patents, at the consideration of CNY 5,000,000.00 (approximately US$ 690,684.05).
On March 3, 2024, Alps Global Holding Berhad entered into a Patent License Agreement with Dr. Tham Seng Kong, wherein Dr. Tham Seng Kong shall grant to Alps Global Holding Berhad an exclusive, royalty-free and non-transferable right and license all the patents stated therein.
During the year ended March 31, 2024, Dr. Tham Seng Kong, director and Chief Executive Officer of Alps, advanced to Alps, RM14,313,532.87 (approximately US$ 2,991,957.78) for working capital purposes. This advance is unsecured, interest free and repayable on demand.
During the year ended March 31, 2024, Chew Yoke Ling, director of Alps, advanced to Alps, RM611,305.58 (approximately US$ 127,781.21) for working capital purposes. This advance is unsecured, interest free and repayable on demand.
During the year ended March 31, 2024, Alps Global Holding Berhad advanced to ANH Healthcare Sdn. Bhd., RM15,815.14 (approximately US$ 3,305.84) for working capital purposes. This advance is unsecured, interest free and repayable on demand.
During the year ended March 31, 2024, Alps Global Holding Berhad advanced to Vax Biotech Sdn. Bhd., RM10,141.04 (approximately US$ 2,119.78) for working capital purposes. This advance is unsecured, interest free and repayable on demand.
During the year ended March 31, 2024, amounts due and owing to and/or from the related parties are as follows, for the working capital purposes:
| Amounts Due To | Amount in RM | Approximate Amount in USD | ||||||
| Amount Due To - TMC Global Holdings Sdn. Bhd. | 5,363,324.71 | 1,121,095.76 | ||||||
| Amount Due To - Celebre Pro Medic Sdn. Bhd. | 254,155.88 | 307,191.59 | ||||||
| Amount Due To Alps Global Holding Berhad | 135,678.20 | 28,360.81 | ||||||
The above advances are unsecured, interest free and repayable on demand.
| Amounts Due From | Amount in RM | Approximate Amount in USD | ||||||
| Amount Due From Mont Life (M) Sdn. Bhd. | 46,445.70 | 9,708.54 | ||||||
| Amount Due From Alps Wellness Centre Sdn. Bhd. | 514,202.89 | 107,483.83 | ||||||
| Amount Due From Celestialab Sdn. Bhd. | 6,903,475.99 | 1,443,033.59 | ||||||
| Amount Due From Mygenome Sdn. Bhd. | 5,122,662.31 | 1,070,790.10 | ||||||
| Amount Due From Alpscap Berhad | 1,302,914.45 | 289,070.61 | ||||||
| Amount Due From Alps Biotech Sdn. Bhd. | 109,105.96 | 22,806.42 | ||||||
| Amount Due From Alps Insurance Pcc Inc | 40,000.00 | 272,348.21 | ||||||
The above advances are unsecured, interest free and repayable on demand.
Related Person Transaction Policy
Upon the Closing of the Business Combination, PubCo intends to adopt a related party transaction policy requiring that all related party transactions required to be disclosed by a domestic public issuer pursuant to the Exchange Act be approved by the audit committee or another independent body of PubCo’s board of directors. The related party transaction policy expected to be adopted will provide for the consideration of the interests of its directors, executive officers and principal shareholders in its review and consideration of transactions and require each to obtain the approval of non-interested directors when it determines that such approval is appropriate under the circumstances of the particular transaction at hand.
| 292 |
LEGAL MATTERS
The validity of the PubCo ordinary shares to be issued pursuant to the Merger Agreement will be passed upon by Ogier, Cayman Islands legal counsel to PubCo. Certain U.S. federal income tax considerations of exercising redemption rights by Globalink stockholders as a result of the Business Combination will be passed upon by Hunter Taubman Fischer & Li LLC, U.S. securities counsel to Globalink.
EXPERTS
The consolidated financial statements of Globalink Investment Inc. for the years ended December 31, 2022 and 2023, appearing in this proxy statement/prospectus have been audited by Marcum LLP, independent registered public accounting firm, as set forth in their report thereon (which contains an explanatory paragraph relating to substantial doubt about the ability of Globalink Investment Inc. to continue as a going concern), appearing elsewhere in this proxy statement/prospectus, and are included in reliance upon such report given on the authority of such firm as experts in auditing and accounting.
The audited consolidated financial statements of Alps Global Holding Berhad for the year ended March 31, 2023 and balance sheet data as of March 31, 2023 have been included in this proxy statement/prospectus in reliance on the report of UHY LLP, an independent registered public accounting firm, appearing elsewhere in this Proxy statement/prospectus, and are included herein in reliance upon the authority of the said firm as experts in accounting and auditing.
APPRAISAL RIGHTS
Globalink stockholders do not have appraisal rights in connection with the Business Combination. However, Globalink stockholders are entitled to redeem their public shares. For details, see “The Special Meeting — Redemption Rights.”
DELIVERY OF DOCUMENTS TO GLOBALINK STOCKHOLDERS
Pursuant to the rules of the SEC, Globalink and servicers that Globalink employs to deliver communications to Globalink stockholders are permitted to deliver to two or more stockholders sharing the same address a single copy of the proxy statement. Upon written or oral request, Globalink will deliver a separate copy of the proxy statement/prospectus to any stockholder at a shared address to which a single copy of the proxy statement/prospectus was delivered and who wishes to receive separate copies in the future. Stockholders receiving multiple copies of the proxy statement/prospectus may likewise request that Globalink delivers single copies of the proxy statement/prospectus in the future. Stockholders may notify Globalink of their requests by calling or writing to Okapi Partners LLC, Globalink’s proxy solicitor at:
Okapi
Partners LLC
1212 Avenue of the Americas, 17th Floor
New York, New York 10036
Individuals call toll-free (855) 305-0857
Banks and brokers call (212) 297-0720
Email: info@okapipartners.com
TRANSFER AGENT AND REGISTRAR
The transfer agent for Globalink’s securities is Continental Stock Transfer & Trust Company.
SUBMISSION OF STOCKHOLDER PROPOSALS
Globalink Board is aware of no other matter that may be brought before the Special Meeting. Under Delaware law, only business that is specified in the notice of a Special Meeting to stockholders may be transacted at the Special Meeting.
| 293 |
FUTURE STOCKHOLDER PROPOSALS
Stockholder proposals, including director nominations, for the 2024 annual meeting must be delivered to, mailed and received, or (iii) electronically mailed and received at Globalink’s principal executive offices by not earlier than the opening of business on the 120th day before the 2024 annual meeting and not later than the later of (x) the close of business on the 90th day before the 2024 annual meeting or (y) the close of business on the 10th day following the first day on which Globalink publicly announces the date of the 2024 annual meeting, and must otherwise comply with applicable SEC rules and the advance notice provisions of Globalink’s bylaws, to be considered for inclusion in Globalink’s proxy materials relating to its 2024 annual meeting. You may contact Globalink’s secretary at its principal executive offices for a copy of the relevant bylaw provisions regarding the requirements for making stockholder proposals and nominating director candidates.
WHERE YOU CAN FIND MORE INFORMATION
Globalink must comply with the informational requirements of the Exchange Act and its rules and regulations, and in accordance with the Exchange Act, Globalink files annual, quarterly, and current reports, proxy statements, and other information with the SEC. You can read Globalink’s SEC filings, including this proxy statement/prospectus over the Internet at the SEC’s website at http://www.sec.gov. If you would like additional copies of this proxy statement/prospectus or if you have questions about the Business Combination or the Proposals to be presented at the Special Meeting, you should contact Globalink’s proxy solicitation agent at the following address and telephone number:
Okapi
Partners LLC
1212 Avenue of the Americas, 17th Floor
New York, New York 10036
Individuals call toll-free (855) 305-0857
Banks and brokers call (212) 297-0720
Email: info@okapipartners.com
If you are a shareholder of Globalink and would like to request documents, please do so by , 2025, in order to receive them before the Special Meeting. If you request any documents from Globalink, Globalink will mail them to you by first class mail, or another equally prompt means.
All information contained in this proxy statement/prospectus relating to Globalink has been supplied by Globalink, and all such information relating to Alps has been supplied by Alps. Information provided by either the Globalink or Alps does not constitute any representation, estimate or projection of any other party.
This document is a proxy statement of Globalink for the Special Meeting. Globalink has not authorized anyone to give any information or make any representation about the Business Combination, Globalink or Alps that is different from, or in addition to, that contained in this proxy statement/prospectus. Therefore, if anyone does give you information of this sort, you should not rely on it. The information contained in this proxy statement/prospectus speaks only as of the date of this proxy statement/prospectus unless the information specifically indicates that another date applies.
The information contained in this proxy statement/prospectus speaks only as of the date of this proxy statement/prospectus unless the information specifically indicates that another date applies.
| 294 |
INDEX TO FINANCIAL STATEMENTS
Globalink Investment Inc.
ALPS GLOBAL HOLDING BERHAD
CONSOLIDATED FINANCIAL STATEMENTS
FOR THE FINANCIAL YEAR ENDED MARCH 31, 2024
ALPS GLOBAL HOLDING BERHAD
TABLE OF CONTENTS
| Report of Independent Registered Public Accounting Firm | F-59 |
| Consolidated Financial Statements: | |
| Consolidated Statement of Financial Position | F-60 - F-61 |
| Consolidated Statement of Profit and Loss and Other Comprehensive | F-62 |
| Consolidated Statement of Changes in Equity | F-63 - F-64 |
| Consolidated Statement of Cash Flows | F-65 - F-66 |
| Notes to Consolidated Financial Statements | F-67 - F-100 |
ALPS GLOBAL HOLDING PUBCO
FINANCIAL STATEMENTS
FOR THE FINANCIAL PERIOD ENDED AUGUST 31, 2024
ALPS GLOBAL HOLDING BERHAD
TABLE OF CONTENTS
| Report of Independent Registered Public Accounting Firm | F-101 - F-102 |
| Financial Statements: | |
| Statement of Financial Position | F-103 |
| Statement of Profit and Loss and Other Comprehensive | F-104 |
| Statement of Changes in Equity | F-105 |
| Statement of Cash Flows | F-106 |
| Notes to Financial Statements | F-107 - F-113 |
ALPS LIFE SCIENCE INC
FINANCIAL STATEMENTS
FOR THE FINANCIAL PERIOD ENDED MAY 31, 2024
ALPS LIFE SCIENCE INC
TABLE OF CONTENTS
| Report of Independent Registered Public Accounting Firm | F-114 - F-115 |
| Financial Statements: | |
| Statement of Financial Position | F-116 |
| Statement of Profit and Loss and Other Comprehensive | F-117 |
| Statement of Changes in Equity | F-119 |
| Statement of Cash Flows | F-120 |
| Notes to Financial Statements | F-121 - F-126 |
| 295 |
GLOBALINK INVESTMENT INC.
CONDENSED CONSOLIDATED BALANCE SHEETS
| September 30, 2024 | December 31, 2023 | |||||||
| (Unaudited) | ||||||||
| ASSETS | ||||||||
| CURRENT ASSETS | ||||||||
| Cash | $ | $ | ||||||
| Prepaid expenses | ||||||||
| Total current assets | ||||||||
| Cash held in Trust Account | ||||||||
| TOTAL ASSETS | $ | $ | ||||||
| LIABILITIES, REDEEMABLE COMMON STOCK AND STOCKHOLDERS’ DEFICIT | ||||||||
| CURRENT LIABILITIES | ||||||||
| Accounts payable | $ | $ | ||||||
| Franchise tax payable | ||||||||
| Income tax payable | ||||||||
| Promissory note – related party | ||||||||
| Due to related parties | ||||||||
| Excise tax liability | ||||||||
| Total current liabilities | ||||||||
| Warrant liabilities | ||||||||
| Deferred underwriting fee payable | ||||||||
| Total Liabilities | ||||||||
| COMMITMENTS AND CONTINGENCIES | ||||||||
| REDEEMABLE COMMON STOCK | ||||||||
| Common stock subject to possible redemption, $ par value, shares at redemption value at September 30, 2024 and December 31, 2023 of $ and $ per share, respectively | ||||||||
| STOCKHOLDERS’ DEFICIT | ||||||||
| Common stock, $ par value; shares authorized; shares issued and outstanding at September 30, 2024 and December 31, 2023 (excluding shares subject to possible redemption) | ||||||||
| Accumulated deficit | ( | ) | ( | ) | ||||
| Total Stockholders’ Deficit | ( | ) | ( | ) | ||||
| LIABILITIES, REDEEMABLE COMMON STOCK AND STOCKHOLDERS’ DEFICIT | $ | $ | ||||||
The accompanying notes are an integral part of these unaudited condensed consolidated financial statements
| F-1 |
GLOBALINK INVESTMENT INC.
CONDENSED CONSOLIDATED STATEMENTS OF OPERATIONS (UNAUDITED)
| Three Months Ended September 30, | Nine Months Ended September 30, | |||||||||||||||
| 2024 | 2023 | 2024 | 2023 | |||||||||||||
| OPERATING EXPENSES | ||||||||||||||||
| General and administrative expenses | $ | $ | $ | $ | ||||||||||||
| Provision for franchise tax expense | ||||||||||||||||
| Total operating expenses | ||||||||||||||||
| OTHER INCOME (LOSS) | ||||||||||||||||
| Income on cash and investments held in Trust Account | ||||||||||||||||
| Penalties on franchise tax | ( | ) | ( | ) | ( | ) | ||||||||||
| Interest expense | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||
| Change in fair value of warrant liabilities | ( | ) | ( | ) | ||||||||||||
| Total other income, net | ||||||||||||||||
| (Loss) income before provision for income taxes | ( | ) | ( | ) | ||||||||||||
| Provision for income taxes | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||
| NET (LOSS) INCOME | $ | ( | ) | $ | $ | ( | ) | $ | ||||||||
| Weighted average shares outstanding Common stock – redeemable | ||||||||||||||||
| Basic and diluted net income per share, Common stock – redeemable | $ | $ | $ | $ | ||||||||||||
| Weighted average shares outstanding Common stock – non-redeemable | ||||||||||||||||
| Basic and diluted net loss per share, Common stock – non-redeemable | $ | ) | $ | ) | $ | ) | $ | ) | ||||||||
The accompanying notes are an integral part of these unaudited condensed consolidated financial statements
| F-2 |
GLOBALINK INVESTMENT INC.
CONDENSED CONSOLIDATED STATEMENTS OF CHANGES IN STOCKHOLDERS’ DEFICIT
(UNAUDITED)
FOR THE THREE AND NINE MONTHS ENDED SEPTEMBER 30, 2024
| Common Stock | Accumulated | Total Stockholders’ | ||||||||||||||
| Shares | Amount | Deficit | Deficit | |||||||||||||
| Balance - December 31, 2023 | $ | $ | ( | ) | $ | ( | ) | |||||||||
| Remeasurement of common stock subject to redemption | — | ( | ) | ( | ) | |||||||||||
| Net loss | — | ( | ) | ( | ) | |||||||||||
| Balance – March 31, 2024 | $ | $ | ( | ) | $ | ( | ) | |||||||||
| Remeasurement of common stock subject to redemption | — | ( | ) | ( | ) | |||||||||||
| Net loss | — | ( | ) | ( | ) | |||||||||||
| Balance - June 30, 2024 | $ | $ | ( | ) | $ | ( | ) | |||||||||
| Remeasurement of common stock subject to redemption | — | ( | ) | ( | ) | |||||||||||
| Net loss | — | ( | ) | ( | ) | |||||||||||
| Balance - September 30, 2024 | $ | $ | ( | ) | $ | ( | ) | |||||||||
FOR THE THREE AND NINE MONTHS ENDED SEPTEMBER 30, 2023
| Common Stock | Accumulated | Total Stockholders’ | ||||||||||||||
| Shares | Amount | Deficit | Deficit | |||||||||||||
| Balance, December 31, 2022 | $ | $ | ( | ) | $ | ( | ) | |||||||||
| Remeasurement of shares subject to possible redemption | — | ( | ) | ( | ) | |||||||||||
| Excise tax imposed on common stock redemptions | — | ( | ) | ( | ) | |||||||||||
| Net income | — | |||||||||||||||
| Balance, March 31, 2023 | ( | ) | ( | ) | ||||||||||||
| Remeasurement of shares subject to possible redemption | — | ( | ) | ( | ) | |||||||||||
| Net income | — | |||||||||||||||
| Balance, June 30, 2023 | ( | ) | ( | ) | ||||||||||||
| Remeasurement of shares subject to possible redemption | — | ( | ) | ( | ) | |||||||||||
| Net income | — | |||||||||||||||
| Balance, September 30, 2023 | $ | $ | ( | ) | $ | ( | ) | |||||||||
The accompanying notes are an integral part of these unaudited condensed consolidated financial statements
| F-3 |
GLOBALINK INVESTMENT INC.
CONDENSED CONSOLIDATED STATEMENTS OF CASH FLOWS
(UNAUDITED)
| For the Nine Months Ended September 30, | ||||||||
| 2024 | 2023 | |||||||
| CASH FLOWS FROM OPERATING ACTIVITIES: | ||||||||
| Net (loss) income | $ | ( | ) | $ | ||||
| Adjustments to reconcile net (loss) income to net cash used in operating activities: | ||||||||
| Interest income on cash and investments held in Trust Account | ( | ) | ( | ) | ||||
| Change in fair value of warrant liabilities | ||||||||
| Changes in operating assets and liabilities: | ||||||||
| Prepaid expenses | ||||||||
| Deferred tax liability | ( | ) | ||||||
| Due to related parties | ||||||||
| Income tax payable | ( | ) | ||||||
| Interest expense accrual | ||||||||
| Accounts payable | ( | ) | ( | ) | ||||
| Franchise tax payable | ( | ) | ( | ) | ||||
| Net cash used in operating activities | ( | ) | ( | ) | ||||
| Cash Flows from Investing Activities: | ||||||||
| Cash deposited to Trust Account | ( | ) | ||||||
| Cash withdrawn from Trust Account in connection with redemption of common stock | ||||||||
| Cash withdrawn from Trust Account to pay tax obligations | ( | ) | ||||||
| Net cash (used in) provided by investing activities | ( | ) | ||||||
| Cash Flows from Financing Activities: | ||||||||
| Proceeds from promissory note | ||||||||
| Due to affiliate - advance | ||||||||
| Common stock redemption | ( | ) | ||||||
| Net cash provided by (used in) financing activities | ( | ) | ||||||
| NET CHANGE IN CASH | ( | ) | ||||||
| CASH, BEGINNING OF PERIOD | ||||||||
| CASH, END OF PERIOD | $ | $ | ||||||
| Supplementary cash flow information: | ||||||||
Cash paid for interest expense | $ | $ | ||||||
| Cash paid for income taxes | $ | $ | ||||||
| Non-cash investing and financing activities: | ||||||||
| Excise tax accrued for common stock redemptions | $ | $ | ||||||
| Remeasurement of Common stock subject to redemption | $ | $ | ||||||
The accompanying notes are an integral part of these unaudited condensed consolidated financial statements
| F-4 |
GLOBALINK INVESTMENT INC
NOTES TO UNAUDITED CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
SEPTEMBER 30, 2024
Note 1 – Description of Organization and Business Operations and Liquidity
Globalink Investment Inc. (the “Company”) was incorporated in Delaware on March 24, 2021. The Company is a blank check company formed for the purpose of entering into a merger, share exchange, asset acquisition, stock purchase, recapitalization, reorganization or other similar business combination with one or more businesses or entities (the “Business Combination”). On July 27, 2022, Globalink Merger Sub, Inc., a Nevada corporation and a wholly-owned subsidiary of Globalink, was formed.
The Company is not limited to a particular industry or geographic region for purposes of consummating a Business Combination. The Company is an early stage and emerging growth company and, as such, the Company is subject to all of the risks associated with early stage and emerging growth companies.
As
of September 30, 2024, the Company had not commenced any operations. All activity through September 30, 2024 relates to the Company’s
formation and Initial Public Offering (“IPO”), which is described below and, since the offering, the search for a prospective
initial Business Combination. The Company will not generate any operating revenues until after the completion of its initial Business
Combination, at the earliest. The Company generates non-operating income in the form of interest income earned on investments from the
proceeds derived from the IPO. The registration statement for the Company’s IPO was declared effective on December 6, 2021. On
December 9, 2021, the Company consummated the IPO of units (“Units”) at $ per Unit generating gross proceeds
of $
Simultaneously
with the closing of the IPO, the Company consummated the sale of units (“Private Placement Units”) at a price of
$ per Private Placement Unit in a private placement to Public Gold Marketing Sdn. Bhd., a Malaysian private limited company, a related
party generating gross proceeds of $
Additionally
with the closing of the IPO, the Company granted the underwriters a 45-day option to purchase up to Units to cover over-allotment.
On December 13, 2021, the underwriters fully exercised the option and purchased additional Units (the “Over-allotment
Units”), generating additional gross proceeds of $
Simultaneously
with the exercise of the over-allotment, the Company consummated a private sale of an additional Private Placement Units to Public
Gold Marketing Sdn. Bhd. at a price of $ per Private Placement Unit, generating additional gross proceeds of $
Offering
costs for the IPO and the exercise of the underwriters’ over-allotment option amounted to $
Following
the closing of the IPO, $
To mitigate the risk of being deemed to have been operating as an unregistered investment company (including under the subjective test of Section 3(a)(1)(A) of the Investment Company Act), in July 2023, the Company instructed Continental Stock Transfer & Trust Company, the trustee of the Trust Account (the “Trustee” or “Continental”), to liquidate the U.S. government securities or money market funds held in the Trust Account and thereafter to hold all funds in the Trust Account in cash (which may include demand deposit accounts) until the earlier of consummation of our Business Combination or liquidation. Furthermore, such cash is held in bank accounts, which exceed federally insured limits as guaranteed by the Federal Deposit Insurance Corporation (the “FDIC”).
The
Company’s management has broad discretion with respect to the specific application of the net proceeds of the IPO and the sale
of the Private Placement Units, although substantially all of the net proceeds are intended to be applied generally toward consummating
a Business Combination. There is no assurance that the Company will be able to complete a Business Combination successfully.
The Company will provide the holders (the “Public Stockholders”) of the outstanding shares of common stock included in the Units, or the Public Shares with the opportunity to redeem all or a portion of their Public Shares upon the completion of a Business Combination either (i) in connection with a stockholder meeting called to approve the Business Combination or (ii) by means of a tender offer. The decision as to whether the Company will seek stockholder approval of a Business Combination or conduct a tender offer will be made by the Company. The Public Stockholders will be entitled to redeem their Public Shares for a pro rata portion of the amount then in the Trust Account (initially anticipated to be $ per Public Share, plus any pro rata interest then in the Trust Account, net of taxes payable). There will be no redemption rights with respect to the Company’s warrants.
| F-5 |
All
of the Public Shares contain a redemption feature, which allows for the redemption of such Public Shares in connection with the Company’s
liquidation, if there is a stockholder vote or tender offer in connection with the Company’s Business Combination and in connection
with certain amendments to the Company’s amended and restated certificate of incorporation (as amended and restated and may be
further amended and restated from time to time, the “Certificate of Incorporation”). In accordance with Financial Accounting
Standards Board (“FASB”) Accounting Standards Codification (“ASC”) 480-10-S99, redemption provisions not solely
within the control of a company require the Public Shares subject to redemption to be classified outside of permanent equity. Given that
the Public Shares will be issued with other freestanding instruments (i.e., public warrants and rights), the initial carrying value of
common stock classified as temporary equity will be the allocated proceeds determined in accordance with ASC 470-20. The Public Shares
are subject to ASC 480-10-S99. If it is probable that the equity instrument will become redeemable, the Company has the option to either
(i) accrete changes in the redemption value over the period from the date of issuance (or from the date that it becomes probable that
the instrument will become redeemable, if later) to the earliest redemption date of the instrument or (ii) recognize changes in the redemption
value immediately as they occur and adjust the carrying amount of the instrument to equal the redemption value at the end of each reporting
period. The Company has elected to recognize the changes immediately. While redemptions cannot cause the Company’s net tangible
assets to fall below $
Redemptions of the Company’s Public Shares may be subject to the satisfaction of conditions, including minimum cash conditions, pursuant to an agreement relating to the Company’s Business Combination. If the Company seeks stockholder approval of the Business Combination, the Company will proceed with a Business Combination if a majority of the shares voted are voted in favor of the Business Combination, or such other vote as required by law or stock exchange rule. If a stockholder vote is not required by applicable law or stock exchange listing requirements and the Company does not decide to hold a stockholder vote for business or other reasons, the Company will, pursuant to its Certificate of Incorporation, conduct the redemptions pursuant to the tender offer rules of the U.S. Securities and Exchange Commission (“SEC”) and file tender offer documents with the SEC prior to completing a Business Combination. If, however, stockholder approval of the transaction is required by applicable law or stock exchange listing requirements, or the Company decides to obtain stockholder approval for business or other reasons, the Company will offer to redeem shares in conjunction with a proxy solicitation pursuant to the proxy rules and not pursuant to the tender offer rules. If the Company seeks stockholder approval in connection with a Business Combination, the sponsor has agreed to vote its Founder Shares (as defined in Note 5) and any Public Shares purchased during or after the IPO in favor of approving a Business Combination. Additionally, each Public Stockholder may elect to redeem their Public Shares without voting, and if they do vote, irrespective of whether they vote for or against the proposed transaction.
Notwithstanding the foregoing, the Certificate of Incorporation provides that a Public Stockholder, together with any affiliate of such stockholder or any other person with whom such stockholder is acting in concert or as a “group” (as defined under Section 13 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”)), will be restricted from redeeming its shares with respect to more than an aggregate of % or more of the shares of common stock sold in the IPO, without the prior consent of the Company.
The
Company originally had until March 9, 2023, 15 months from the closing of the IPO to complete a Business Combination. On March 6, 2023,
the Company held a special meeting (the “March 2023 Special Meeting”), during which the stockholders of the Company approved
a proposal to amend the Company’s amended and restated certified articles of incorporation which included extending the time in
which the Company must complete a Business Combination (the “Extension Amendment Proposal”) and a proposal to amend the Company’s
investment management trust agreement, dated as of December 6, 2021 (the “Trust Agreement”), by and between the Company and
Continental (the “Trust Amendment Proposal”). The Company had the option of two (2) three-months extensions, followed by
three (3) one-month extensions, or until December 9, 2023, if all extensions are exercised. The Company exercised the option for two
three-month extensions and as a result the Company has deposited a total of $
On
March 6, 2023, in connection with the approval of the proposals presented at the March 2023 Special Meeting which extended the time in
which the Company must complete a Business Combination, holders of of the Company’s shares of common stock exercised
their right to redeem those shares for cash at an approximate price of $ per share, for an aggregate of approximately $
| F-6 |
On October 16, 2023, the Company received a written notice (the “Notice”) from the Nasdaq Listing Qualifications Department of The Nasdaq Stock Market (“Nasdaq”) indicating that the Company was not in compliance with Nasdaq Listing Rule 5450(a)(2), which requires the Company to maintain at least 400 total holders for continued listing on the Nasdaq Global Market (the “Minimum Total Holders Rule”). The Notice is only a notification of deficiency, not of imminent delisting, and has no current effect on the listing or trading of the Company’s securities on the Nasdaq Global Market. The Notice stated that the Company had 45 calendar days, or until November 30, 2023, to submit a plan to regain compliance with the Minimum Total Holders Rule. On January 29, 2024, the Company submitted an application to phase-down from The Nasdaq Global Market to The Nasdaq Capital Market. On March 6, 2024, the Company received a letter from the Nasdaq Listing Qualifications staff granting the Company’s request for transfer to The Nasdaq Capital Market. The Company’s securities were transferred to The Nasdaq Capital Market at the opening of business on March 12, 2024. In connection with the approval of the phase-down application, the staff indicated that the Company’s deficiency with the Minimum Total Holders Rule was cured and the matter was closed.
On
November 28, 2023,
On
November 28, 2023, the stockholders of the Company approved a proposal to amend the Company’s Certificate of Incorporation, allowing
the Company to the Extended Deadline from December 9, 2023 to up to December 9, 2024 through monthly Extensions. To obtain each Extension,
the Company, its sponsor or any of their affiliates or designees must deposit into the Company’s trust account with Continental
by the deadline applicable prior to such Extension, $
On April 3, 2024, Globalink Merger Sub (Cayman), was incorporated in the Cayman Islands as a wholly-owned subsidiary of Globalink.
As of the date of this report, the Company has extended the Termination Date twelve times under its Certificate of Incorporation, as amended (or seventeen times since the IPO), and has until December 9, 2024 to complete its initial business combination.
On
October 3, 2024, the Company deposited an aggregate of $
On
November 7, 2024, the Company deposited an aggregate of $
| F-7 |
The Initial Stockholders have agreed to waive their liquidation rights with respect to the Founder Shares if the Company fails to complete a Business Combination within the Combination Period. However, if the Initial Stockholders should acquire any Public Shares in or after the IPO, they will be entitled to liquidating distributions from the Trust Account with respect to such Public Shares if the Company fails to complete a Business Combination within the Combination Period. The underwriters have agreed to waive their rights to deferred underwriting discounts (see Note 6) held in the Trust Account in the event the Company does not complete a Business Combination within the Combination Period, and, in such event, such amounts will be included with the other funds held in the Trust Account that will be available to fund the redemption of the Public Shares. In the event of such distribution, it is possible that the per share value of the residual assets remaining available for distribution (including Trust Account assets) will be only $ per share held in the Trust Account. In order to protect the amounts held in the Trust Account, the sponsor has agreed to be liable to the Company if and to the extent any claims by a vendor for services rendered or products sold to the Company, or a prospective target business with which the Company has discussed entering into a transaction agreement, reduce the amount of funds in the Trust Account. This liability will not apply with respect to any claims by a third party who executed a waiver of any right, title, interest or claim of any kind in or to any monies held in the Trust Account or to any claims under the Company’s indemnity of the underwriters of the IPO against certain liabilities, including liabilities under the Securities Act of 1933, as amended (the “Securities Act”). Moreover, in the event that an executed waiver is deemed to be unenforceable against a third party, the sponsor will not be responsible to the extent of any liability for such third-party claims. The Company will seek to reduce the possibility that the sponsor will have to indemnify the Trust Account due to claims of creditors by endeavoring to have all vendors, service providers (except the Company’s independent registered public accounting firm), prospective target businesses or other entities with which the Company does business, execute agreements waiving any right, title, interest or claim of any kind in or to monies held in the Trust Account.
Business Combination
On August 3, 2022, the Company entered into an Agreement and Plan of Merger (the “Tomorrow Crypto Merger Agreement”) by and among Tomorrow Crypto Group Inc., a Nevada corporation (“Tomorrow Crypto”), Globalink Merger Sub, Inc., a Nevada corporation and a wholly-owned subsidiary of Globalink, GL Sponsor LLC, a Delaware limited liability company.
In accordance with the termination provisions under Section 10.1 of the Tomorrow Crypto Merger Agreement, the Tomorrow Crypto Merger Agreement was terminated on March 8, 2023 (the “Tomorrow Crypto Merger Agreement Termination Date”). In conjunction with the termination of the Tomorrow Crypto Merger Agreement, the Additional Agreements (as defined in the Tomorrow Crypto Merger Agreement) (including the Support Agreements) were also terminated in accordance with their respective terms as of March 8, 2023, the Tomorrow Crypto Merger Agreement Termination Date.
On January 30, 2024, the Company entered into a Merger Agreement (as amended and restated on May 20, 2024 and as may be further amended, restated or supplemented from time to time, the “Merger Agreement”), by and among GL Sponsor LLC, a Delaware limited liability company, in the capacity as the representative from and after the effective time of the Acquisition Merger (as defined below) (the “Effective Time”) in accordance with the terms and conditions of the Merger Agreement (the “Parent Representative” or the “Sponsor”), Alps Global Holding Pubco, a Cayman Islands exempted company (“PubCo”), Alps Biosciences Merger Sub, a Cayman Islands exempted company and wholly-owned subsidiary of PubCo (“Merger Sub”), Alps Life Sciences Inc, a Cayman Islands exempted company (“Alps Holdco”) and Dr. Tham Seng Kong, an individual, in the capacity as the representative from and after the Effective Time for the shareholders of Alps Holdco as of immediately prior to the Effective Time in accordance with the terms and conditions of the Merger Agreement (the “Seller Representative”). Pursuant to the terms of the Merger Agreement, the Business Combination between Globalink and Alps Holdco will be effected in two steps: (i) subject to the approval and adoption of the Merger Agreement by the stockholders of the Company, the Company will be merged with and into PubCo, with PubCo remaining as the surviving publicly traded entity and (ii) Merger Sub will merge with and into Alps Holdco, resulting in Alps Holdco remaining as the surviving entity and being a wholly-owned subsidiary of PubCo (the “Acquisition Merger”).
On
June 4, June 5 and August 27, 2024, Globalink, Alps Holdco and PubCo entered into subscription agreements (the “PIPE Subscription
Agreements”) with three investors on substantially same terms (the “PIPE Investors”), pursuant to which, among other
things, PubCo has agreed to issue and sell to the PIPE Investors, and the PIPE Investors have agreed to subscribe for and purchase certain
number of ordinary shares of PubCo (“PIPE Shares”) at a purchase price of $ per share for an aggregate purchase price
of $
The purpose of the PIPE Investment is to raise additional capital for use by PubCo following the closing of the Business Combination (the “Closing”). The PIPE Subscription Agreements contain customary representations and warranties of each of Globalink, Alps Holdco, PubCo and the PIPE Investors, and customary conditions to closing, including the consummation of the Business Combination. Under the terms of the PIPE Subscription Agreements, PubCo is obligated to file a registration statement to register for the resale of all the PIPE Shares within 60 days of the Closing (the “Filing Deadline”), and to use its commercially reasonable efforts to cause such registration statement to become effective as soon as practicable after the filing there of.
Risks and Uncertainties
The Company continues to evaluate the impact of increases in inflation and rising interest rates, financial market instability, including the recent bank failures, the potential government shutdown, the lingering effects of the COVID-19 pandemic and certain geopolitical events, including the wars in Ukraine and the surrounding region and between Israel and Hamas. The Company has concluded that while it is reasonably possible that the risks and uncertainties related to or resulting from these events could have a negative effect on its financial position, results of operations and/or ability to complete an initial Business Combination, the Company cannot at this time fully predict the likelihood of one or more of the above events, their duration or magnitude or the extent to which they may negatively impact the Company’s business and its ability to complete an initial Business Combination.
On
August 16, 2022, the Inflation Reduction Act of 2022 (the “IR Act”) was signed into federal law. The IR Act provides for,
among other things, a new U.S. federal
Any redemption or other repurchase that occurs after December 31, 2022, in connection with a Business Combination, extension vote or otherwise, may be subject to the excise tax. Whether and to what extent the Company would be subject to the excise tax in connection with a Business Combination, extension vote or otherwise would depend on a number of factors, including (i) the fair market value of the redemptions and repurchases in connection with the Business Combination, extension or otherwise, (ii) the structure of a Business Combination, (iii) the nature and amount of any “PIPE” or other equity issuances in connection with a Business Combination (or otherwise issued not in connection with a Business Combination but issued within the same taxable year of a Business Combination) and (iv) the content of regulations and other guidance from the Treasury. In addition, because the excise tax would be payable by the Company and not by the redeeming holder. The foregoing could cause a reduction in the cash available on hand to complete a Business Combination and in the Company’s ability to complete a Business Combination.
During the second quarter of 2024, the Internal Revenue Service issued final regulations with respect to the timing and payment of the Excise Tax. These regulations provided that the filing and payment deadline for any liability incurred during the period from January 1, 2023 to December 31, 2023 would be October 31, 2024.
The
Company is currently evaluating its options with respect to this obligation.
| F-8 |
Liquidity, Capital Resources and Going Concern
As
of September 30, 2024, the Company had $
Until the consummation of a Business Combination, the Company will be using the funds not held in the Trust Account for identifying and evaluating prospective acquisition candidates, performing due diligence on prospective target businesses, paying for travel expenditures, selecting the target business to acquire, and structuring, negotiating and consummating the Business Combination. The Company will need to raise additional capital through loans or additional investments from its sponsor, stockholders, officers, directors, or third parties. The Company’s officers, directors and the sponsor may, but are not obligated to, loan the Company funds, from time to time or at any time, in whatever amount they deem reasonable in their sole discretion, to meet the Company’s working capital needs. Accordingly, the Company may not be able to obtain additional financing.
If the Business Combination is not consummated, the Company will need to raise additional capital through loans or additional investments from its sponsor, stockholders, officers, directors, or third parties. The Company’s officers, directors and its sponsor may, but are not obligated to, loan the Company funds, from time to time or at any time, in whatever amount they deem reasonable in their sole discretion, to meet the Company’s working capital needs. Accordingly, the Company may not be able to obtain additional financing. If the Company is unable to raise additional capital, it may be required to take additional measures to conserve liquidity, which could include, but not necessarily be limited to, curtailing operations, suspending the pursuit of a potential transaction, and reducing overhead expenses. The Company cannot provide any assurance that new financing will be available to it on commercially acceptable terms, if at all.
In connection with the Company’s assessment of going concern considerations in accordance with FASB ASU 2014-15, “Disclosures of Uncertainties about an Entity’s Ability to Continue as a Going Concern,” the Company currently has until November 9, 2024 to consummate a business combination, or up to December 9, 2024 if the time to complete the initial business combination is extended as described herein. It is uncertain that the Company will be able to consummate a business combination by this time. If a business combination is not consummated by this date and an extension is not requested by the sponsor, there will be a mandatory liquidation and subsequent dissolution of the Company. Management has determined that the mandatory liquidation, should a business combination not occur, and an extension is not requested by the sponsor, and potential subsequent dissolution as well as liquidity condition noted above raises substantial doubt about the Company’s ability to continue as a going concern. No adjustments have been made to the carrying amounts of assets or liabilities should the Company be required to liquidate after November 9, 2024 (or up to December 9, 2024 if the time to complete the initial business combination is extended as described herein). The Company intends to complete a business combination before the mandatory liquidation date.
Note 2 — Summary of Significant Accounting Policies
Basis of Presentation
The accompanying unaudited condensed consolidated financial statements of the Company are presented in conformity with accounting principles generally accepted in the United States of America (“U.S. GAAP”) and pursuant to the rules and regulations of the SEC. Certain information or footnote disclosures normally included in unaudited condensed consolidated financial statements prepared in accordance with U.S. GAAP have been condensed or omitted, pursuant to the rules and regulations of the SEC for interim financial reporting. Accordingly, they do not include all the information and footnotes necessary for a complete presentation of financial position, results of operations, or cash flows. In the opinion of management, the accompanying unaudited condensed consolidated financial statements include all adjustments, consisting of a normal recurring nature, which are necessary for a fair presentation of the financial position, operating results and cash flows for the periods presented. The accompanying unaudited condensed consolidated financial statements should be read in conjunction with the Company’s Annual Report on Form 10-K for the fiscal year ended December 31, 2023, as filed with the SEC on April 2, 2024. The interim results for the three and nine months ended September 30, 2024 presented are not necessarily indicative of the results to be expected for the year ending December 31, 2024 or for any future interim periods.
Principles of Consolidation
The accompanying unaudited condensed consolidated financial statements include the accounts of the Company and its wholly owned subsidiary. All significant intercompany balances and transactions have been eliminated in consolidation.
| F-9 |
Emerging Growth Company
The Company is an emerging growth company as defined in Section 102(b)(1) of the Jumpstart Our Business Startups Act of 2012 (the “JOBS Act”), which exempts emerging growth companies from being required to comply with new or revised financial accounting standards until private companies (that is, those that have not had a Securities Act registration statement declared effective or do not have a class of securities registered under the Exchange Act) are required to comply with the new or revised financial accounting standards. The JOBS Act provides that an emerging growth company can elect to opt out of the extended transition period and comply with the requirements that apply to non-emerging growth companies but any such an election to opt out is irrevocable. The Company has elected not to opt out of such extended transition period, which means that when a standard is issued or revised, and it has different application dates for public or private companies, the Company, as an emerging growth company, can adopt the new or revised standard at the time private companies adopt the new or revised standard. This may make comparison of the Company’s consolidated financial statements with another public company that is neither an emerging growth company nor an emerging growth company that has opted out of using the extended transition period difficult or impossible because of the potential differences in accounting standards used.
Use of Estimates
The preparation of consolidated financial statements in conformity with U.S. GAAP requires the Company’s management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the financial statements. Making estimates requires management to exercise significant judgment. One of the more significant accounting estimates included in these consolidated financial statements is the determination of the fair value of the warrant liabilities. Such estimates may be subject to change as more current information becomes available and accordingly the actual results could differ significantly from those estimates. It is at least reasonably possible that the estimate of the effect of a condition, situation or set of circumstances that existed at the date of the financial statements, which management considered in formulating its estimate, could change in the near term due to one or more future confirming events.
Cash Held in Trust Account
As of September 30, 2024 and December 31, 2023, all of the assets held in the Trust Account were held in cash.
To mitigate the risk of being deemed to have been operating as an unregistered investment company (including under the subjective test of Section 3(a)(1)(A) of the Investment Company Act), on July 27, 2023, the Company instructed the Trustee of the Trust Account, to liquidate the U.S. government securities or money market funds held in the Trust Account and thereafter to hold all funds in the Trust Account in cash (which may include demand deposit accounts) until the earlier of consummation of our Business Combination or liquidation. Furthermore, such cash is held in bank accounts, which exceed federally insured limits as guaranteed by the FDIC.
Concentration of Credit Risk
Financial instruments that potentially subject the Company to concentrations of credit risk consist of cash accounts in a financial institution, which, at times, may exceed the Federal Depository Insurance Corporation coverage limit. As of September 30, 2024 and December 31, 2023, the Company had not experienced losses on these accounts.
Fair Value of Financial Instruments
The fair value of the Company’s assets and liabilities which qualify as financial instruments under the FASB ASC 820, “Fair Value Measurements and Disclosures,” approximate the carrying amounts represented in the accompanying consolidated balance sheets, primarily due to their short-term nature.
| F-10 |
Income Taxes
The Company complies with the accounting and reporting requirements of ASC 740, “Income Taxes,” which requires an asset and liability approach to financial accounting and reporting for income taxes. Deferred income tax assets and liabilities are computed for differences between the financial statement and tax bases of assets and liabilities that will result in future taxable or deductible amounts, based on enacted tax laws and rates applicable to the periods in which the differences are expected to affect taxable income. Valuation allowances are established, when necessary, to reduce deferred tax assets to the amount expected to be realized. As of September 30, 2024 and December 31, 2023, the Company’s deferred tax asset for start up organizational expenses had a full valuation allowance recorded against it.
While
ASC 740 identifies usage of an effective annual tax rate for purposes of an interim provision, it does allow for estimating individual
elements in the current period if they are significant, unusual or infrequent. Computing the effective tax rate for the Company is complicated
due to the potential impact of the timing of any Business Combination expenses and the actual interest income that will be recognized
during the year. The Company has taken a position as to the calculation of income tax expense in a current period based on ASC 740-270-25-3
which states, “If an entity is unable to estimate a part of its ordinary income (or loss) or the related tax (benefit) but is otherwise
able to make a reasonable estimate, the tax (or benefit) applicable to the item that cannot be estimated shall be reported in the interim
period in which the item is reported.” The Company believes its calculation to be a reliable estimate and allows it to properly
take into account the usual elements that can impact its annualized book income and its impact on the effective tax rate. As such, the
Company is computing its taxable income (loss) and associated income tax provision based on actual results through September 30, 2024.
The Company’s effective tax rate was (
FASB ASC 740 prescribes a recognition threshold and a measurement attribute for the financial statement recognition and measurement of tax positions taken or expected to be taken in a tax return. For those benefits to be recognized, a tax position must be more likely than not to be sustained upon examination by taxing authorities. There were no unrecognized tax benefits as of September 30, 2024 or December 31, 2023. No amounts were paid for interest and penalties for the three months ended September 30, 2024. The Company is currently not aware of any issues under review that could result in significant payments, accruals or material deviation from its position. The Company is subject to income tax examinations by major taxing authorities since inception.
Excise Tax
In
connection with the vote to approve the charter amendment proposal presented at the March 2023 Special Meeting, holders of
shares of common stock properly exercised their right to redeem their shares of common stock for an aggregate redemption amount of approximately
$
During the second quarter of 2024, the Internal Revenue Service issued final regulations with respect to the timing and payment of the Excise Tax. These regulations provided that the filing and payment deadline for any liability incurred during the period from January 1, 2023 to December 31, 2023 would be October 31, 2024.
The Company is currently
evaluating its options with respect to this obligation.
Franchise Tax
The
Company calculates Franchise Tax liability on a quarterly basis using the estimate calculator on the Delaware Franchise Tax website.
At December 31, 2023, the Company had over accrued the amount due by approximately $
The Company accounts for its common stock subject to possible redemption in accordance with the guidance in ASC Topic 480 “Distinguishing Liabilities from Equity.” Shares of common stock subject to mandatory redemption (if any) is classified as a liability instrument and is measured at fair value. Conditionally redeemable common stock (including common stock that features redemption rights that are either within the control of the holder or subject to redemption upon the occurrence of uncertain events not solely within the Company’s control) is classified as temporary equity. At all other times, common stock is classified as stockholders’ equity. The Company’s shares of common stock sold in the IPO and as a result of the exercise by the underwriters of their over-allotment option features certain redemption rights that are considered to be outside of the Company’s control and subject to occurrence of uncertain future events. Accordingly, on September 30, 2024 and December 31, 2023, shares of common stock subject to possible redemption were presented as temporary equity, outside of the stockholders’ deficit section of the Company’s consolidated balance sheets.
On
March 6, 2023, in connection with the approval of the extension amendment proposal and the trust amendment proposal presented at the
March 2023 Special Meeting, holders of of the Company’s shares of common stock exercised their right to redeem those
shares for cash at an approximate price of $ per share, for an aggregate of approximately $
On November 28, 2023, in connection with the approval of the Extension Amendment Proposal and the Trust Amendment Proposal at the November 2023 Special Meeting, holders of shares of the Company’s common stock exercised their right to redeem those shares for cash at an approximate price of $ per share, for an aggregate of approximately $ million.
| F-11 |
| Schedule of Common Stock Subject to Possible Redemption | ||||||||
| Shares | Amount | |||||||
| Common stock subject to possible redemption, December 31, 2022 | ||||||||
| Less: | ||||||||
| Redemptions (paid in April and November 2023) | ) | ) | ||||||
| Plus: | ||||||||
| Remeasurement of carrying value to redemption value | ||||||||
| Common stock subject to possible redemption, December 31, 2023 | ||||||||
| Plus: | ||||||||
| Remeasurement of carrying value to redemption value | ||||||||
| Common stock subject to possible redemption, March 31, 2024 | $ | |||||||
| Plus: | ||||||||
| Remeasurement of carrying value to redemption value | ||||||||
| Common stock subject to possible redemption, June 30, 2024 | $ | |||||||
| Plus: | ||||||||
| Remeasurement of carrying value to redemption value | ||||||||
| Common stock subject to possible redemption, September 30, 2024 | $ | |||||||
The Company complies with the accounting and disclosure requirements of FASB ASC Topic 260, “Earnings Per Share” and uses the two class method. Net (loss) income per share of common stock is computed by dividing net (loss) income by the weighted average number of shares of common stock outstanding for the period. Any remeasurement of the accretion to redemption value of the shares of common stock subject to possible redemption was considered to be dividends paid to the public stockholders.
The Company has one authorized class of common stock. Warrants included in the Units sold in the IPO (the “Public Warrants”) (see Note 3) and warrants included in the Private Placement Units (the “Private Placement Warrants,” together with the Public Warrants, the “warrants”) (see Note 4) to purchase shares of common stock of the Company at $ per share were issued on December 9, 2021. For the periods ended September 30, 2024 and 2023, no Public Warrants or Private Placement Warrants had been exercised. The potential shares of common stock underlying the outstanding Public Warrants and Private Placement Warrants to purchase the Company’s shares of common stock were excluded from diluted earnings per share for the periods ended September 30, 2024 and 2023 because they are contingently exercisable, and the contingencies have not yet been met. Additionally, the rights are able to be demanded on or any time after the Business Combination, and as the contingency has not been met, the rights are excluded from diluted earnings per share for the periods ended September 30, 2024 and 2023. As a result, diluted net income (loss) per share of common stock is the same as basic net income (loss) per share of common stock for the periods. The table below presents a reconciliation of the numerator and denominator used to compute basic and diluted net income per share for each class of stock.
| For the Three Months Ended September 30, | ||||||||
| 2024 | 2023 | |||||||
| Net (loss) income | $ | ( | ) | $ | ||||
| Remeasurement of common stock subject to redemption | ( | ) | ( | ) | ||||
| Net loss including remeasurement of common stock subject to redemption value | $ | ( | ) | $ | ( | ) | ||
| For the Three Months Ended September 30, | ||||||||||||||||
| 2024 | 2023 | |||||||||||||||
| Redeemable | Non-redeemable | Redeemable | Non-redeemable | |||||||||||||
| Basic and diluted net loss per share of common stock | ||||||||||||||||
| Numerator: | ||||||||||||||||
| Allocation of net loss including remeasurement of common stock subject to redemption value | $ | ( | ) | $ | ( | ) | $ | ( | ) | $ | ( | ) | ||||
| Remeasurement of common stock subject to redemption | ||||||||||||||||
| Allocation of net income (loss), as adjusted | $ | $ | ( | ) | $ | $ | ( | ) | ||||||||
| Denominator: | ||||||||||||||||
| Basic and diluted weighted average shares outstanding | ||||||||||||||||
| Basic and diluted net income (loss) per share of common stock | $ | $ | ) | $ | $ | ) | ||||||||||
| F-12 |
| For the Nine Months Ended September 30, | ||||||||
| 2024 | 2023 | |||||||
| Net (loss) income | $ | ( | ) | $ | ||||
| Remeasurement of common stock subject to redemption | ( | ) | ( | ) | ||||
| Net loss including remeasurement of common stock subject to redemption value | $ | ( | ) | $ | ( | ) | ||
| For the Nine Months Ended September 30, | ||||||||||||||||
| 2024 | 2023 | |||||||||||||||
| Redeemable | Non-redeemable | Redeemable | Non-redeemable | |||||||||||||
| Basic and diluted net loss per share of common stock | ||||||||||||||||
| Numerator: | ||||||||||||||||
| Allocation of net loss including remeasurement of common stock subject to redemption value | $ | ( | ) | $ | ( | ) | $ | ( | ) | $ | ( | ) | ||||
| Remeasurement of common stock subject to redemption | ||||||||||||||||
| Allocation of net income (loss), as adjusted | $ | $ | ( | ) | $ | $ | ( | ) | ||||||||
| Denominator: | ||||||||||||||||
| Basic and diluted weighted average shares outstanding | ||||||||||||||||
| Basic and diluted net income (loss) per share of common stock | $ | $ | ) | $ | $ | ) | ||||||||||
Accounting for Warrants
The Company accounts for warrants as either equity-classified or liability-classified instruments based on an assessment of the warrant’s specific terms and applicable authoritative guidance in FASB ASC 480, Distinguishing Liabilities from Equity (“ASC 480”) and ASC 815, Derivatives and Hedging (“ASC 815”). The assessment considers whether the warrants are freestanding financial instruments pursuant to ASC 480, meet the definition of a liability pursuant to ASC 480, and whether the warrants meet all of the requirements for equity classification under ASC 815, including whether the warrants are indexed to the Company’s own common stock, among other conditions for equity classification. This assessment, which requires the use of professional judgment, is conducted at the time of warrant issuance and as of each subsequent quarterly period end date while the warrants are outstanding. The Company accounts for the warrants issued in connection with the Company’s IPO in accordance with the guidance contained in ASC 815 under which the public warrants meet the criteria for equity treatment and the private warrants do not meet the criteria for equity treatment and must be recorded as liabilities. Accordingly, the Company classifies the private warrants as liabilities at their fair value and adjust the private warrants to fair value at each reporting period. This liability is subject to re-measurement at each balance sheet date until exercised, and any change in fair value is recognized in the Company’s consolidated statements of operations. The fair value of the warrants was estimated using a binomial lattice model.
Recent Accounting Pronouncements
In December 2023, the FASB issued ASU 2023-09, Income Taxes (Topic 740): Improvements to Income Tax Disclosures (ASU 2023-09), which requires disclosure of incremental income tax information within the rate reconciliation and expanded disclosures of income taxes paid, among other disclosure requirements. ASU 2023-09 is effective for fiscal years beginning after December 15, 2024. Early adoption is permitted. The Company’s management does not believe the adoption of ASU 2023-09 will have a material impact on its consolidated financial statements and disclosures.
Note 3 — Initial Public Offering and Over-allotment
Pursuant
to the IPO and the over-allotment in December 2021, the Company sold Units at a price of $ per Unit. Each Unit consists
of one share of common stock, one redeemable warrant (each, a “Public Warrant” and collectively, the “Public Warrants”)
and one right (each a “Public Right” and collectively, the “Public Rights”).
Note 4 — Private Placement
On
December 9, 2021 and December 13, 2021, simultaneously with the consummation of the IPO and the underwriters’ exercise of their
over-allotment option, the Company consummated the issuance and sale (“Private Placement”) of Private Placement Units
in a private placement transaction at a price of $ per Private Placement Unit, generating gross proceeds of $
| F-13 |
Note 5 — Related Party Transactions
Founder Shares
On
August 19, 2021, the Company’s sponsor purchased shares (the “Founder Shares”) of the Company’s common
stock, par value $, for an aggregate price of $
Private Placement
Simultaneously
with the closing of the IPO, the Company consummated the sale of units (“Private Placement Units”) at a price of
$ per Private Placement Unit in a private placement to Public Gold Marketing Sdn. Bhd., a Malaysian private limited company, a related
party generating gross proceeds of $
Related Party Loans
In
order to finance transaction costs in connection with a Business Combination, the Company’s sponsor or an affiliate of the sponsor,
or certain of the Company’s officers and directors may, but are not obligated to, loan the Company funds as may be required (“Working
Capital Loans”). If the Company completes a Business Combination, the Company will repay the Working Capital Loans out of the proceeds
of the Trust Account released to the Company. Otherwise, the Working Capital Loans would be repaid only out of funds held outside the
Trust Account. In the event that a Business Combination does not close, the Company may use a portion of proceeds held outside the Trust
Account to repay the Working Capital Loans, but no proceeds held in the Trust Account would be used to repay the Working Capital Loans.
Except for the foregoing, the terms of such Working Capital Loans, if any, have not been determined and no written agreements exist with
respect to such loans. The Working Capital Loans would either be repaid upon consummation of a Business Combination, without interest,
or, at the lender’s discretion, up to $
The
Company entered into promissory notes with Public Gold Marketing Sdn. Bhd., which is considered a related party due to a familial relationship
between the controlling member of the sponsor and a 95% shareholder of Public Gold Marketing Sdn. Bhd. The promissory notes bear an interest
of
Support Services
The
Company has entered into an administrative services agreement pursuant to which the Company will pay its sponsor a total of $
Advances
On
each of September 5, 2023, September 29, 2023 and November 7, 2023, an affiliate of the Company’s sponsor advanced $
Note 6 — Commitments and Contingencies
Registration Rights
The holders of Founder Shares, Private Placement Units and warrants that may be issued upon conversion of Working Capital Loans, if any, will be entitled to registration rights (in the case of the Founder Shares, only after conversion of such shares into shares of common stock) pursuant to a registration rights agreement signed on the date of the prospectus for the IPO. These holders are entitled to certain demand and “piggyback” registration rights. However, the registration rights agreement provides that the Company will not permit any registration statement filed under the Securities Act to become effective until the termination of the applicable lock-up period for the securities to be registered. The Company will bear the expenses incurred in connection with the filing of any such registration statements.
Underwriting Agreement
The
underwriters were paid a cash underwriting discount of $ per unit on the offering including the Units issued with the underwriter’s
exercise of their over-allotment option, or $
| F-14 |
Note 7 — Promissory Notes – Related Party
On
March 3, 2023, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount
of $
On
March 23, 2023, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount
of up to $
On
June 2, 2023, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount of
up to $
On
October 13, 2023, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount
of $
On
December 8, 2023, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount
of $
On
January 5, 2024, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount
of $
On
January 25, 2024, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount
of $
On
February 22, 2024, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount
of $
On
April 4, 2024, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount
of $
On
June 5, 2024, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount of
$
On
August 14, 2024, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount
of $
For
the three and nine months ended September 30, 2024, the above-mentioned notes have incurred $
Note 8 — Stockholders’ Deficit
Common stock
The Company is authorized to issue shares of common stock with a par value of $ per share. As of September 30, 2024 and December 31, 2023, there were (excluding shares of common stock subject to possible redemption) shares of common stock issued and outstanding.
Warrants:
As
of September 30, 2024 and December 31, 2023, the Company had
The Public Warrants are accounted for as equity instruments in the Company’s consolidated financial statements. Public Warrants may only be exercised for a whole number of shares. No fractional shares will be issued upon exercise of the Public Warrants. The Public Warrants will become exercisable on the later of the completion of an initial Business Combination and will expire five years after the completion of an initial Business Combination, or earlier upon redemption. No Public Warrants will be exercisable for cash unless the Company has an effective and current registration statement covering the shares of common stock issuable upon exercise of the Public Warrants and a current prospectus relating to such shares of common stock. Notwithstanding the foregoing, if a registration statement covering the shares of common stock issuable upon exercise of the Public Warrants is not effective within a specified period following the consummation of a Business Combination, warrant holders may, until such time as there is an effective registration statement and during any period when the Company shall have failed to maintain an effective registration statement, exercise warrants on a cashless basis pursuant to the exemption provided by Section 3(a)(9) of the Securities Act, provided that such exemption is available. If that exemption, or another exemption, is not available, holders will not be able to exercise their warrants on a cashless basis. The Public Warrants will expire five years after the completion of a Business Combination or earlier upon redemption or liquidation.
| F-15 |
Redemption of warrants when the price per common stock equals or exceeds $
Once the warrants become exercisable, the Company may redeem the outstanding warrants (except as described herein with respect to the private placement warrants):
| ● | in whole and not in part; | |
| ● | at
a price of $ | |
| ● | upon a minimum of 30 days’ prior written notice of redemption, which the Company refers to as the “30-day redemption period”; and | |
| ● | if, and only if, the last reported sale price (the “closing price”) of the Company’s common stock equals or exceeds $ per share (as adjusted for adjustments to the number of shares issuable upon exercise or the exercise price of a warrant as described under the heading “Description of Securities—Warrants”) for any 20 trading days within a 30-trading day period ending on the third trading day prior to the date on which the Company sends the notice of redemption to the warrant holders. |
The Company will not redeem the warrants as described above unless an effective registration statement under the Securities Act covering the shares of common stock issuable upon exercise of the warrants is effective and a current prospectus relating to those shares of common stock is available throughout the 30-day redemption period. If and when the warrants become redeemable by the Company, the Company may exercise its redemption right even if the Company is unable to register or qualify the underlying securities for sale under all applicable state securities laws.
If the Company calls the Public Warrants for redemption, management will have the option to require all holders that wish to exercise the Public Warrants to do so on a “cashless basis,” as described in the warrant agreement.
The Private Placement Warrants are substantially in the same form as the Public Warrants, except they (i) will be exercisable either for cash or on a cashless basis at the holder’s option pursuant and (ii) will not be redeemable by the Company, in either case as long as the Private Placement Warrants are held by the initial purchasers or any of their permitted transferees (as prescribed in the Subscription Agreement, dated December 6, 2021, by and between the Company and Public Gold Marketing Sdn. Bhd.). Once a Private Placement Warrant is transferred to a holder other than a permitted transferee, it shall be treated as a Public Warrant for all purposes. Due to these terms the Private Warrants are required to be liability classified.
The exercise price and number of shares of common stock issuable on exercise of the warrants may be adjusted in certain circumstances, including in the event of a share dividend, extraordinary dividend or recapitalization, reorganization, merger or consolidation. However, the warrants will not be adjusted for issuances of shares of common stock at a price below their respective exercise prices. Additionally, in no event will the Company be required to net cash settle the warrants. If the Company is unable to complete a Business Combination within the Combination Period and the Company liquidates the funds held in the Trust Account, holders of the warrants will not receive any of such funds with respect to their warrants, nor will they receive any distribution from the Company’s assets held outside of the Trust Account with the respect to such warrants. Accordingly, the warrants may expire worthless.
In
addition, if the Company issues additional shares of common stock or equity-linked securities for capital raising purposes in connection
with the closing of a Business Combination at an issue price or effective issue price of less than $
Rights
Except in cases where the Company is not the surviving company in a Business Combination, each holder of a Public Right will automatically receive one-tenth of one share of common stock upon consummation of a Business Combination, even if the holder of a Public Right converted all shares held by him, her or it in connection with a Business Combination or an amendment to the Company’s Certificate of Incorporation with respect to its pre-business combination activities. In the event that the Company will not be the surviving company upon completion of a Business Combination, each holder of a Public Right will be required to affirmatively convert his, her or its rights in order to receive the one-tenth of a share underlying each Public Right upon consummation of the Business Combination.
The Company will not issue fractional shares in connection with an exchange of Public Rights. Fractional shares will either be rounded down to the nearest whole share or otherwise addressed in accordance with the applicable provisions of the Delaware General Corporation Law. As a result, the holders of the Public Rights must hold rights in multiples of 10 in order to receive shares for all of the holders’ rights upon closing of a Business Combination.
| F-16 |
Note 9 — Fair Value Measurements
The fair value of the Company’s financial assets and liabilities reflects management’s estimate of amounts that the Company would have received in connection with the sale of the assets or paid in connection with the transfer of the liabilities in an orderly transaction between market participants at the measurement date. In connection with measuring the fair value of its assets and liabilities, the Company seeks to maximize the use of observable inputs (market data obtained from independent sources) and to minimize the use of unobservable inputs (internal assumptions about how market participants would price assets and liabilities). The following fair value hierarchy is used to classify assets and liabilities based on the observable inputs and unobservable inputs used in order to value the assets and liabilities:
Level 1: Quoted prices in active markets for identical assets or liabilities. An active market for an asset or liability is a market in which transactions for the asset or liability occur with sufficient frequency and volume to provide pricing information on an ongoing basis.
Level 2: Observable inputs other than Level 1 inputs. Examples of Level 2 inputs include quoted prices in active markets for similar assets or liabilities and quoted prices for identical assets or liabilities in markets that are not active.
Level 3: Unobservable inputs based on the Company’s assessment of the assumptions that market participants would use in pricing the asset or liability.
As of September 30, 2024 and December 31, 2023 the assets held in the Trust Account were held in cash.
The following table presents information about the Company’s liabilities that are measured at fair value on a recurring basis as of September 30, 2024 and December 31, 2023 and indicates the fair value hierarchy of the valuation inputs the Company utilized to determine such fair value.
| Quoted Prices in Active Markets | Significant Other Observable Inputs | Significant Other Unobservable Inputs | ||||||||||||||
| Level | (Level 1) | (Level 2) | (Level 3) | |||||||||||||
| September 30, 2024 | ||||||||||||||||
| Liabilities: | ||||||||||||||||
| Warrant Liabilities- Private Warrants | 3 | $ | ||||||||||||||
| December 31, 2023 | ||||||||||||||||
| Liabilities: | ||||||||||||||||
| Warrant Liabilities- Private Warrants | 3 | |||||||||||||||
| F-17 |
The Private Placement Warrants were valued using a binomial lattice model, which is considered to be a Level 3 fair value measurement. One of the more significant inputs is the implied volatility, which is based on the observed prices of the Company’s common stock and publicly-traded warrants. As of September 30, 2024 and December 31, 2023, the estimated fair value of Warrant Liabilities – Private Warrants were determined based on the following significant inputs and are expressed on the basis of each being exercisable for a one-half of one share of common stock:
As of September 30, 2024 | As of December 31, 2023 | |||||||
| Exercise price | $ | $ | ||||||
| Market price of public stock | $ | $ | ||||||
| Term (years) | ||||||||
| Volatility | ||||||||
| Risk-free rate | % | % | ||||||
| Dividend yield | % | % | ||||||
The following table presents the changes in the fair value of warrant liabilities for the three and nine months ended September 30, 2024 and 2023:
| Private Placement Warrants | ||||
| Fair value as of January 1, 2024 | $ | |||
| Change in valuation inputs or other assumptions | ||||
| Fair value as of March 31, 2024 | $ | |||
| Change in valuation inputs or other assumptions | ||||
| Fair value as of June 30, 2024 | $ | |||
| Change in valuation inputs or other assumptions | ( | ) | ||
| Fair value as of September 30, 2024 | $ | |||
| Private Placement Warrants | ||||
| Fair value as of January 1, 2023 | $ | |||
| Change in valuation inputs or other assumptions | ( | ) | ||
| Fair value as of March 31, 2023 | $ | |||
| Change in valuation inputs or other assumptions | ||||
| Fair value as of June 30, 2023 | $ | |||
| Change in valuation inputs or other assumptions | ( | ) | ||
| Fair value as of September 30, 2023 | $ | |||
Note 10 — Subsequent Events
The Company evaluated subsequent events and transactions that occurred after the balance sheet date up to the date that the unaudited condensed consolidated financial statements were issued. Based upon this review the Company did not identify any subsequent events, other than the below, that would have required adjustment or disclosure in the consolidated financial statements.
On
October 3, 2024, the Company deposited an aggregate of $
On
October 3, 2024, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn Bhd for an amount
of $
On
November 7, 2024, the Company deposited an aggregate of $
On November 8, 2024, the Company announced that a Special Meeting will be held on December 3, 2024 to vote on an extension to complete a business combination.
| F-18 |
GLOBALINK INVESTMENT INC.
CONDENSED CONSOLIDATED BALANCE SHEETS
| March 31, 2024 | December 31, 2023 | |||||||
| (Unaudited) | ||||||||
| ASSETS | ||||||||
| CURRENT ASSETS | ||||||||
| Cash | $ | $ | ||||||
| Prepaid expenses | ||||||||
| Total current assets | ||||||||
| Cash held in Trust Account | ||||||||
| TOTAL ASSETS | $ | $ | ||||||
| LIABILITIES, REDEEMABLE COMMON STOCK AND STOCKHOLDERS’ DEFICIT | ||||||||
| CURRENT LIABILITIES | ||||||||
| Accounts payable | $ | $ | ||||||
| Franchise tax payable | ||||||||
| Income tax payable | ||||||||
| Promissory note – related party | ||||||||
| Due to related parties | ||||||||
| Excise tax liability | ||||||||
| Total current liabilities | ||||||||
| Warrant liabilities | ||||||||
| Deferred underwriting fee payable | ||||||||
| Total Liabilities | ||||||||
| COMMITMENTS AND CONTINGENCIES | ||||||||
| REDEEMABLE COMMON STOCK | ||||||||
| Common stock subject to possible redemption, $par value, shares at redemption value at March 31, 2024 and December 31, 2023 of $and $ per share, respectively | ||||||||
| STOCKHOLDERS’ DEFICIT | ||||||||
| Common stock, $par value; shares authorized; shares issued and outstanding at March 31, 2024 and December 31, 2023 (excluding shares subject to possible redemption) | ||||||||
| Accumulated deficit | ( | ) | ( | ) | ||||
| Total Stockholders’ Deficit | ( | ) | ( | ) | ||||
| LIABILITIES, REDEEMABLE COMMON STOCK AND STOCKHOLDERS’ DEFICIT | $ | $ | ||||||
The accompanying notes are an integral part of these unaudited condensed consolidated financial statements
| F-19 |
GLOBALINK INVESTMENT INC.
CONDENSED CONSOLIDATED STATEMENTS OF OPERATIONS (UNAUDITED)
| For the Three Months Ended March 31, | ||||||||
| 2024 | 2023 | |||||||
| OPERATING EXPENSES | ||||||||
| General and administrative expenses | $ | $ | ||||||
| (Reversal) provision for franchise tax expense | ( | ) | ||||||
| Total operating expenses | ( | ) | ( | ) | ||||
| OTHER INCOME (EXPENSE) | ||||||||
| Income on cash and investments held in Trust Account | ||||||||
| Penalties on franchise tax | ( | ) | ||||||
| Interest expense | ( | ) | ||||||
| Change in fair value of warrant liabilities | ( | ) | ||||||
| Total other income, net | ||||||||
| (Loss) income before provision for income taxes | ( | ) | ||||||
| Provision for income taxes | ( | ) | ( | ) | ||||
| NET (LOSS) INCOME | $ | ( | ) | $ | ||||
| Weighted average shares outstanding Common stock – redeemable | ||||||||
| Basic and diluted net income per share, Common stock – redeemable | ||||||||
| Weighted average shares outstanding Common stock – non-redeemable | ||||||||
| Basic and diluted net loss per share, Common stock – non-redeemable | $ | ) | $ | ) | ||||
The accompanying notes are an integral part of these unaudited condensed consolidated financial statements
| F-20 |
GLOBALINK INVESTMENT INC.
CONDENSED CONSOLIDATED STATEMENTS OF CHANGES IN STOCKHOLDERS’ DEFICIT
(UNAUDITED)
FOR THE THREE MONTHS ENDED MARCH 31, 2024
| Common Stock | Accumulated | Total Stockholders’ | ||||||||||||||
| Shares | Amount | Deficit | Deficit | |||||||||||||
| Balance - December 31, 2023 | $ | $ | ( | ) | $ | ( | ) | |||||||||
| Remeasurement of common stock subject to redemption | — | ( | ) | ( | ) | |||||||||||
| Net loss | — | ( | ) | ( | ) | |||||||||||
| Balance - March 31, 2024 | $ | $ | ( | ) | $ | ( | ) | |||||||||
FOR THE THREE MONTHS ENDED MARCH 31, 2023
| Common Stock | Accumulated | Total Stockholders’ | ||||||||||||||
| Shares | Amount | Deficit | Deficit | |||||||||||||
| Balance - December 31, 2022 | $ | $ | ( | ) | $ | ( | ) | |||||||||
| Remeasurement of shares subject to possible redemption | — | ( | ) | ( | ) | |||||||||||
| Excise tax imposed on common stock redemptions | — | ( | ) | ( | ) | |||||||||||
| Net income | — | |||||||||||||||
| Balance, March 31, 2023 | $ | $ | ( | ) | $ | ( | ) | |||||||||
The accompanying notes are an integral part of these unaudited condensed consolidated financial statements
| F-21 |
GLOBALINK INVESTMENT INC.
CONDENSED CONSOLIDATED STATEMENTS OF CASH FLOWS
(UNAUDITED)
| For the Three Months Ended March 31, | ||||||||
| 2024 | 2023 | |||||||
| CASH FLOWS FROM OPERATING ACTIVITIES: | ||||||||
| Net (loss) income | $ | ( | ) | $ | ||||
| Adjustments to reconcile net (loss) income to net cash used in operating activities: | ||||||||
| Interest income on cash and investments held in Trust Account | ( | ) | ( | ) | ||||
| Change in fair value of warrant liabilities | ( | ) | ||||||
| Changes in operating assets and liabilities: | ||||||||
| Prepaid expenses | ( | ) | ||||||
| Deferred tax liability | ( | ) | ||||||
| Due to related parties | ||||||||
| Income tax payable | ||||||||
| Interest expense accrual | ||||||||
| Accounts payable | ( | ) | ||||||
| Franchise tax payable | ( | ) | ||||||
| Net cash used in operating activities | ( | ) | ( | ) | ||||
| Cash Flows from Investing Activities: | ||||||||
| Cash deposited to Trust Account | ( | ) | ( | ) | ||||
| Cash withdrawn from Trust Account to pay tax obligations | ||||||||
| Net cash used in investing activities | ( | ) | ( | ) | ||||
| Cash Flows from Financing Activities: | ||||||||
| Issuance of promissory notes – related party | ||||||||
| Net cash provided by financing activities | ||||||||
| NET CHANGE IN CASH | ( | ) | ( | ) | ||||
| CASH, BEGINNING OF PERIOD | ||||||||
| CASH, END OF PERIOD | $ | $ | ||||||
| Supplementary cash flow information: | ||||||||
| Cash paid for interest expense | $ | $ | ||||||
| Cash paid for income taxes | $ | $ | ||||||
| Non-cash investing and financing activities: | ||||||||
| Excise tax accrued for common stock redemptions | $ | $ | ||||||
| Remeasurement of Common stock subject to redemption | $ | $ | ||||||
| Reclassification of redeemable Common stock to redeemed stock payable | $ | $ | ||||||
The accompanying notes are an integral part of these unaudited condensed consolidated financial statements
| F-22 |
GLOBALINK INVESTMENT INC
NOTES TO UNAUDITED CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
MARCH 31, 2024
Note 1 – Description of Organization and Business Operations and Liquidity
Globalink Investment Inc. (the “Company”) was incorporated in Delaware on March 24, 2021. The Company is a blank check company formed for the purpose of entering into a merger, share exchange, asset acquisition, stock purchase, recapitalization, reorganization or other similar business combination with one or more businesses or entities (the “Business Combination”). On July 27, 2022, Globalink Merger Sub, Inc., a Nevada corporation and a wholly-owned subsidiary of Globalink, was formed.
The Company is not limited to a particular industry or geographic region for purposes of consummating a Business Combination. The Company is an early stage and emerging growth company and, as such, the Company is subject to all of the risks associated with early stage and emerging growth companies.
As
of March 31, 2024, the Company had not commenced any operations. All activity through March 31, 2024 relates to the Company’s formation
and Initial Public Offering (“IPO”), which is described below and, since the offering, the search for a prospective initial
Business Combination. The Company will not generate any operating revenues until after the completion of its initial Business Combination,
at the earliest. The Company generates non-operating income in the form of interest income earned on investments from the proceeds derived
from the IPO. The registration statement for the Company’s IPO was declared effective on December 6, 2021. On December 9, 2021,
the Company consummated the IPO of units (“Units”) at $ per Unit generating gross proceeds of $
Simultaneously
with the closing of the IPO, the Company consummated the sale of units (“Private Placement Units”) at a price of
$ per Private Placement Unit in a private placement to Public Gold Marketing Sdn. Bhd., a Malaysian private limited company, a related
party generating gross proceeds of $
Additionally
with the closing of the IPO, the Company granted the underwriters a 45-day option to purchase up to Units to cover over-allotment.
On December 13, 2021, the underwriters fully exercised the option and purchased additional Units (the “Over-allotment
Units”), generating additional gross proceeds of $
Simultaneously
with the exercise of the over-allotment, the Company consummated a private sale of an additional Private Placement Units to Public
Gold Marketing Sdn. Bhd. at a price of $ per Private Placement Unit, generating additional gross proceeds of $
Offering
costs for the IPO and the exercise of the underwriters’ over-allotment option amounted to $
Following
the closing of the IPO, $
To mitigate the risk of being deemed to have been operating as an unregistered investment company (including under the subjective test of Section 3(a)(1)(A) of the Investment Company Act), in July 2023, the Company instructed Continental Stock Transfer & Trust Company, the trustee of the Trust Account (the “Trustee” or “Continental”), to liquidate the U.S. government securities or money market funds held in the Trust Account and thereafter to hold all funds in the Trust Account in cash (which may include demand deposit accounts) until the earlier of consummation of our Business Combination or liquidation. Furthermore, such cash is held in bank accounts, which exceed federally insured limits as guaranteed by the Federal Deposit Insurance Corporation (the “FDIC”).
The
Company’s management has broad discretion with respect to the specific application of the net proceeds of the IPO and the sale
of the Private Placement Units, although substantially all of the net proceeds are intended to be applied generally toward consummating
a Business Combination. There is no assurance that the Company will be able to complete a Business Combination successfully.
The Company will provide the holders (the “Public Stockholders”) of the outstanding shares of common stock included in the Units, or the Public Shares with the opportunity to redeem all or a portion of their Public Shares upon the completion of a Business Combination either (i) in connection with a stockholder meeting called to approve the Business Combination or (ii) by means of a tender offer. The decision as to whether the Company will seek stockholder approval of a Business Combination or conduct a tender offer will be made by the Company. The Public Stockholders will be entitled to redeem their Public Shares for a pro rata portion of the amount then in the Trust Account (initially anticipated to be $ per Public Share, plus any pro rata interest then in the Trust Account, net of taxes payable). There will be no redemption rights with respect to the Company’s warrants.
| F-23 |
All
of the Public Shares contain a redemption feature, which allows for the redemption of such Public Shares in connection with the Company’s
liquidation, if there is a stockholder vote or tender offer in connection with the Company’s Business Combination and in connection
with certain amendments to the Company’s amended and restated certificate of incorporation (as amended and restated and may be
further amended and restated from time to time, the “Certificate of Incorporation”). In accordance with Financial Accounting
Standards Board (“FASB”) Accounting Standards Codification (“ASC”) 480-10-S99, redemption provisions not solely
within the control of a company require the Public Shares subject to redemption to be classified outside of permanent equity. Given that
the Public Shares will be issued with other freestanding instruments (i.e., public warrants and rights), the initial carrying value of
common stock classified as temporary equity will be the allocated proceeds determined in accordance with ASC 470-20. The Public Shares
are subject to ASC 480-10-S99. If it is probable that the equity instrument will become redeemable, the Company has the option to either
(i) accrete changes in the redemption value over the period from the date of issuance (or from the date that it becomes probable that
the instrument will become redeemable, if later) to the earliest redemption date of the instrument or (ii) recognize changes in the redemption
value immediately as they occur and adjust the carrying amount of the instrument to equal the redemption value at the end of each reporting
period. The Company has elected to recognize the changes immediately. While redemptions cannot cause the Company’s net tangible
assets to fall below $
Redemptions of the Company’s Public Shares may be subject to the satisfaction of conditions, including minimum cash conditions, pursuant to an agreement relating to the Company’s Business Combination. If the Company seeks stockholder approval of the Business Combination, the Company will proceed with a Business Combination if a majority of the shares voted are voted in favor of the Business Combination, or such other vote as required by law or stock exchange rule. If a stockholder vote is not required by applicable law or stock exchange listing requirements and the Company does not decide to hold a stockholder vote for business or other reasons, the Company will, pursuant to its Certificate of Incorporation, conduct the redemptions pursuant to the tender offer rules of the U.S. Securities and Exchange Commission (“SEC”) and file tender offer documents with the SEC prior to completing a Business Combination. If, however, stockholder approval of the transaction is required by applicable law or stock exchange listing requirements, or the Company decides to obtain stockholder approval for business or other reasons, the Company will offer to redeem shares in conjunction with a proxy solicitation pursuant to the proxy rules and not pursuant to the tender offer rules. If the Company seeks stockholder approval in connection with a Business Combination, the sponsor has agreed to vote its Founder Shares (as defined in Note 5) and any Public Shares purchased during or after the IPO in favor of approving a Business Combination. Additionally, each Public Stockholder may elect to redeem their Public Shares without voting, and if they do vote, irrespective of whether they vote for or against the proposed transaction.
Notwithstanding the foregoing, the Certificate of Incorporation provides that a Public Stockholder, together with any affiliate of such stockholder or any other person with whom such stockholder is acting in concert or as a “group” (as defined under Section 13 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”)), will be restricted from redeeming its shares with respect to more than an aggregate of % or more of the shares of common stock sold in the IPO, without the prior consent of the Company.
The
Company originally had until March 9, 2023, 15 months from the closing of the IPO to complete a Business Combination. On March 6, 2023,
the Company held a special meeting (the “March 2023 Special Meeting”), during which the stockholders of the Company approved
a proposal to amend the Company’s amended and restated certified articles of incorporation which included extending the time in
which the Company must complete a Business Combination (the “Extension Amendment Proposal”) and a proposal to amend the Company’s
investment management trust agreement, dated as of December 6, 2021 (the “Trust Agreement”), by and between the Company and
Continental (the “Trust Amendment Proposal”). The Company will have the option of two (2) three-months extensions, followed
by three (3) one-month extensions, or until December 9, 2023, if all extensions are exercised. The Company exercised the option for two
three-month extensions and as a result the Company has deposited a total of $
Through
a total of five elections from March 2023 to December 2023, Globalink elected to extend the Termination Date (as defined below) to December
9, 2023 and deposited an aggregate of US$
On
March 6, 2023, in connection with the approval of the proposals presented at the March 2023 Special Meeting which extended the time in
which the Company must complete a Business Combination, holders of of the Company’s shares of common stock exercised
their right to redeem those shares for cash at an approximate price of $ per share, for an aggregate of approximately $
| F-24 |
On October 16, 2023, the Company received a written notice (the “Notice”) from the Nasdaq Listing Qualifications Department of The Nasdaq Stock Market (“Nasdaq”) indicating that the Company was not in compliance with Nasdaq Listing Rule 5450(a)(2), which requires the Company to maintain at least 400 total holders for continued listing on the Nasdaq Global Market (the “Minimum Total Holders Rule”). The Notice is only a notification of deficiency, not of imminent delisting, and has no current effect on the listing or trading of the Company’s securities on the Nasdaq Global Market. The Notice stated that the Company had 45 calendar days, or until November 30, 2023, to submit a plan to regain compliance with the Minimum Total Holders Rule. On January 29, 2024, the Company submitted an application to phase-down from The Nasdaq Global Market to The Nasdaq Capital Market. On March 6, 2024, the Company received a letter from the Nasdaq Listing Qualifications staff granting the Company’s request for transfer to The Nasdaq Capital Market. The Company’s securities were transferred to The Nasdaq Capital Market at the opening of business on March 12, 2024. In connection with the approval of the phase-down application, the staff indicated that the Company’s deficiency with the Minimum Total Holders Rule was cured and the matter was closed.
On
November 28, 2023,
On
November 28, 2023, the stockholders of the Company approved a proposal to amend the Company’s Certificate of Incorporation, allowing
the Company to the Extended Deadline from December 9, 2023 to up to December 9, 2024 through monthly Extensions. To obtain each Extension,
the Company, its sponsor or any of their affiliates or designees must deposit into the Company’s trust account with Continental
by the deadline applicable prior to such Extension, $
As of the date of this report, the Company has extended the Termination Date six times under its Certificate of Incorporation, as amended (or eleven times since the IPO), and has until June 9, 2024 to complete its initial business combination. The Company may continue to extend the Termination Date to up to December 9, 2024 through monthly Extensions by resolutions of its Board, if requested by the Sponsor, and subject to the Sponsor depositing additional funds into the Trust Account.
Through the Insider Letter, in consideration for inducing Chardan, the representative of the underwriters for Globalink’s IPO to proceed with the IPO, and the economic benefit the IPO would confer upon the Initial Stockholders and PGM, each of the Initial Stockholders and PGM have agreed to waive their liquidation rights with respect to the Founder Shares if the Company fails to complete a Business Combination within the Combination Period. However, if the Initial Stockholders should acquire any Public Shares in or after the IPO, they will be entitled to liquidating distributions from the Trust Account with respect to such Public Shares if the Company fails to complete a Business Combination within the Combination Period. The underwriters have agreed to waive their rights to deferred underwriting discounts (see Note 6) held in the Trust Account in the event the Company does not complete a Business Combination within the Combination Period, and, in such event, such amounts will be included with the other funds held in the Trust Account that will be available to fund the redemption of the Public Shares. In the event of such distribution, it is possible that the per share value of the residual assets remaining available for distribution (including Trust Account assets) will be only $ per share held in the Trust Account. In order to protect the amounts held in the Trust Account, the sponsor has agreed to be liable to the Company if and to the extent any claims by a vendor for services rendered or products sold to the Company, or a prospective target business with which the Company has discussed entering into a transaction agreement, reduce the amount of funds in the Trust Account. This liability will not apply with respect to any claims by a third party who executed a waiver of any right, title, interest or claim of any kind in or to any monies held in the Trust Account or to any claims under the Company’s indemnity of the underwriters of the IPO against certain liabilities, including liabilities under the Securities Act of 1933, as amended (the “Securities Act”). Moreover, in the event that an executed waiver is deemed to be unenforceable against a third party, the sponsor will not be responsible to the extent of any liability for such third-party claims. The Company will seek to reduce the possibility that the sponsor will have to indemnify the Trust Account due to claims of creditors by endeavoring to have all vendors, service providers (except the Company’s independent registered public accounting firm), prospective target businesses or other entities with which the Company does business, execute agreements waiving any right, title, interest or claim of any kind in or to monies held in the Trust Account.
Business Combination
On August 3, 2022, the Company entered into an Agreement and Plan of Merger (the “Merger Agreement”) by and among Tomorrow Crypto Group Inc., a Nevada corporation (“Tomorrow Crypto”), Globalink Merger Sub, Inc., a Nevada corporation and a wholly-owned subsidiary of Globalink (“Merger Sub”), GL Sponsor LLC, a Delaware limited liability company.
In accordance with the termination provisions under Section 10.1 of the Merger Agreement, the Merger Agreement was terminated on March 8, 2023 (the “Merger Agreement Termination Date”). In conjunction with the termination of the Merger Agreement, the Additional Agreements (as defined in the Merger Agreement) (including the Support Agreements) were also terminated in accordance with their respective terms as of March 8, 2023, the Merger Agreement Termination Date.
On January 30, 2024, the Company entered into a Merger Agreement (the “Merger Agreement”) by and among Alps Global Holding Berhad, a Malaysian company (“Alps”), GL Sponsor LLC and Dr. Tham Seng Kong, an individual. Pursuant to the terms of the Merger Agreement, a business combination between Globalink and Alps through the merger of a to be incorporated subsidiary of Globalink in the Cayman Islands (the “Merger Sub”) with and into Alps, with Alps surviving the merger (the “Surviving Company”) as a wholly-owned subsidiary of Globalink (the “Merger”, and, together with the other transactions contemplated by the Merger Agreement, the “Transactions”). After the date of the Merger Agreement and prior to the consummation of the transactions contemplated by the Merger Agreement (the “Closing”), a company formed under the laws of the Cayman Islands will be incorporated (“Cayman Holdco”), whereupon it is envisaged that Alps will become a subsidiary of the Cayman Holdco.
| F-25 |
Risks and Uncertainties
The Company continues to evaluate the impact of increases in inflation and rising interest rates, financial market instability, including the recent bank failures, the potential government shutdown, the lingering effects of the COVID-19 pandemic and certain geopolitical events, including the wars in Ukraine and the surrounding region and between Israel and Hamas. The Company has concluded that while it is reasonably possible that the risks and uncertainties related to or resulting from these events could have a negative effect on its financial position, results of operations and/or ability to complete an initial Business Combination, the Company cannot at this time fully predict the likelihood of one or more of the above events, their duration or magnitude or the extent to which they may negatively impact the Company’s business and its ability to complete an initial Business Combination.
On
August 16, 2022, the Inflation Reduction Act of 2022 (the “IR Act”) was signed into federal law. The IR Act provides for,
among other things, a new U.S. federal
Any redemption or other repurchase that occurs after December 31, 2022, in connection with a Business Combination, extension vote or otherwise, may be subject to the excise tax. Whether and to what extent the Company would be subject to the excise tax in connection with a Business Combination, extension vote or otherwise would depend on a number of factors, including (i) the fair market value of the redemptions and repurchases in connection with the Business Combination, extension or otherwise, (ii) the structure of a Business Combination, (iii) the nature and amount of any “PIPE” or other equity issuances in connection with a Business Combination (or otherwise issued not in connection with a Business Combination but issued within the same taxable year of a Business Combination) and (iv) the content of regulations and other guidance from the Treasury. In addition, because the excise tax would be payable by the Company and not by the redeeming holder, the mechanics of any required payment of the excise tax have not been determined. The foregoing could cause a reduction in the cash available on hand to complete a Business Combination and in the Company’s ability to complete a Business Combination.
Liquidity, Capital Resources and Going Concern
As
of March 31, 2024, the Company had $
Until the consummation of a Business Combination, the Company will be using the funds not held in the Trust Account for identifying and evaluating prospective acquisition candidates, performing due diligence on prospective target businesses, paying for travel expenditures, selecting the target business to acquire, and structuring, negotiating and consummating the Business Combination. The Company will need to raise additional capital through loans or additional investments from its sponsor, stockholders, officers, directors, or third parties. The Company’s officers, directors and the sponsor may, but are not obligated to, loan the Company funds, from time to time or at any time, in whatever amount they deem reasonable in their sole discretion, to meet the Company’s working capital needs. Accordingly, the Company may not be able to obtain additional financing.
If the Business Combination is not consummated, the Company will need to raise additional capital through loans or additional investments from its sponsor, stockholders, officers, directors, or third parties. The Company’s officers, directors and its sponsor may, but are not obligated to, loan the Company funds, from time to time or at any time, in whatever amount they deem reasonable in their sole discretion, to meet the Company’s working capital needs. Accordingly, the Company may not be able to obtain additional financing. If the Company is unable to raise additional capital, it may be required to take additional measures to conserve liquidity, which could include, but not necessarily be limited to, curtailing operations, suspending the pursuit of a potential transaction, and reducing overhead expenses. The Company cannot provide any assurance that new financing will be available to it on commercially acceptable terms, if at all.
In connection with the Company’s assessment of going concern considerations in accordance with FASB ASU 2014-15, “Disclosures of Uncertainties about an Entity’s Ability to Continue as a Going Concern,” the Company currently has until June 9, 2024 to consummate a business combination, and may be extended to December 9, 2024 on a monthly basis by resolutions of the Globalink Board, if requested by the Sponsor, and subject to the Sponsor depositing additional funds into the Trust Account. It is uncertain that the Company will be able to consummate a business combination by this time. If a business combination is not consummated by this date and an extension is not requested by the sponsor, there will be a mandatory liquidation and subsequent dissolution of the Company. Management has determined that the mandatory liquidation, should a business combination not occur, and an extension is not requested by the sponsor, and potential subsequent dissolution as well as liquidity condition noted above raises substantial doubt about the Company’s ability to continue as a going concern. No adjustments have been made to the carrying amounts of assets or liabilities should the Company be required to liquidate after June 9, 2024 (and may be extended to December 9, 2024 on a monthly basis by resolutions of the Globalink Board, if requested by the Sponsor, and subject to the Sponsor depositing additional funds into the Trust Account). The Company intends to complete a business combination before the mandatory liquidation date.
Note 2 — Summary of Significant Accounting Policies
Basis of Presentation
The accompanying unaudited condensed consolidated financial statements of the Company are presented in conformity with accounting principles generally accepted in the United States of America (“U.S. GAAP”) and pursuant to the rules and regulations of the SEC. Certain information or footnote disclosures normally included in unaudited condensed consolidated financial statements prepared in accordance with U.S. GAAP have been condensed or omitted, pursuant to the rules and regulations of the SEC for interim financial reporting. Accordingly, they do not include all the information and footnotes necessary for a complete presentation of financial position, results of operations, or cash flows. In the opinion of management, the accompanying unaudited condensed consolidated financial statements include all adjustments, consisting of a normal recurring nature, which are necessary for a fair presentation of the financial position, operating results and cash flows for the periods presented. The accompanying unaudited condensed consolidated financial statements should be read in conjunction with the Company’s Annual Report on Form 10-K for the fiscal year ended December 31, 2023, as filed with the SEC on April 2, 2024. The interim results for the three months ended March 31, 2024 presented are not necessarily indicative of the results to be expected for the year ending December 31, 2024 or for any future interim periods.
| F-26 |
Principles of Consolidation
The accompanying unaudited condensed consolidated financial statements include the accounts of the Company and its wholly owned subsidiary. All significant intercompany balances and transactions have been eliminated in consolidation.
Emerging Growth Company
The Company is an emerging growth company as defined in Section 102(b)(1) of the Jumpstart Our Business Startups Act of 2012 (the “JOBS Act”), which exempts emerging growth companies from being required to comply with new or revised financial accounting standards until private companies (that is, those that have not had a Securities Act registration statement declared effective or do not have a class of securities registered under the Exchange Act) are required to comply with the new or revised financial accounting standards. The JOBS Act provides that an emerging growth company can elect to opt out of the extended transition period and comply with the requirements that apply to non-emerging growth companies but any such an election to opt out is irrevocable. The Company has elected not to opt out of such extended transition period, which means that when a standard is issued or revised, and it has different application dates for public or private companies, the Company, as an emerging growth company, can adopt the new or revised standard at the time private companies adopt the new or revised standard. This may make comparison of the Company’s consolidated financial statements with another public company that is neither an emerging growth company nor an emerging growth company that has opted out of using the extended transition period difficult or impossible because of the potential differences in accounting standards used.
Use of Estimates
The preparation of consolidated financial statements in conformity with U.S. GAAP requires the Company’s management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the financial statements. Making estimates requires management to exercise significant judgment. One of the more significant accounting estimates included in these consolidated financial statements is the determination of the fair value of the warrant liabilities. Such estimates may be subject to change as more current information becomes available and accordingly the actual results could differ significantly from those estimates. It is at least reasonably possible that the estimate of the effect of a condition, situation or set of circumstances that existed at the date of the financial statements, which management considered in formulating its estimate, could change in the near term due to one or more future confirming events.
Cash Held in Trust Account
As of March 31, 2024 and December 31, 2023, all of the assets held in the Trust Account were held in cash.
To mitigate the risk of being deemed to have been operating as an unregistered investment company (including under the subjective test of Section 3(a)(1)(A) of the Investment Company Act), on July 27, 2023, the Company instructed the Trustee of the Trust Account, to liquidate the U.S. government securities or money market funds held in the Trust Account and thereafter to hold all funds in the Trust Account in cash (which may include demand deposit accounts) until the earlier of consummation of our Business Combination or liquidation. Furthermore, such cash is held in bank accounts, which exceed federally insured limits as guaranteed by the FDIC.
Concentration of Credit Risk
Financial instruments that potentially subject the Company to concentrations of credit risk consist of cash accounts in a financial institution, which, at times, may exceed the Federal Depository Insurance Corporation coverage limit. As of March 31, 2024 and December 31, 2023, the Company had not experienced losses on these accounts.
Fair Value of Financial Instruments
The fair value of the Company’s assets and liabilities which qualify as financial instruments under the FASB ASC 820, “Fair Value Measurements and Disclosures,” approximate the carrying amounts represented in the accompanying consolidated balance sheets, primarily due to their short-term nature.
Income Taxes
The Company complies with the accounting and reporting requirements of ASC 740, “Income Taxes,” which requires an asset and liability approach to financial accounting and reporting for income taxes. Deferred income tax assets and liabilities are computed for differences between the financial statement and tax bases of assets and liabilities that will result in future taxable or deductible amounts, based on enacted tax laws and rates applicable to the periods in which the differences are expected to affect taxable income. Valuation allowances are established, when necessary, to reduce deferred tax assets to the amount expected to be realized. As of March 31, 2024 and December 31, 2023, the Company’s deferred tax asset for start up organizational expenses had a full valuation allowance recorded against it.
| F-27 |
While
ASC 740 identifies usage of an effective annual tax rate for purposes of an interim provision, it does allow for estimating individual
elements in the current period if they are significant, unusual or infrequent. Computing the effective tax rate for the Company is complicated
due to the potential impact of the timing of any Business Combination expenses and the actual interest income that will be recognized
during the year. The Company has taken a position as to the calculation of income tax expense in a current period based on ASC 740-270-25-3
which states, “If an entity is unable to estimate a part of its ordinary income (or loss) or the related tax (benefit) but is otherwise
able to make a reasonable estimate, the tax (or benefit) applicable to the item that cannot be estimated shall be reported in the interim
period in which the item is reported.” The Company believes its calculation to be a reliable estimate and allows it to properly
take into account the usual elements that can impact its annualized book income and its impact on the effective tax rate. As such, the
Company is computing its taxable income (loss) and associated income tax provision based on actual results through March 31, 2024. The
Company’s effective tax rate was (
FASB ASC 740 prescribes a recognition threshold and a measurement attribute for the financial statement recognition and measurement of tax positions taken or expected to be taken in a tax return. For those benefits to be recognized, a tax position must be more likely than not to be sustained upon examination by taxing authorities. There were no unrecognized tax benefits as of March 31, 2024 or December 31, 2023. No amounts were paid for interest and penalties for the three months ended March 31, 2024. The Company is currently not aware of any issues under review that could result in significant payments, accruals or material deviation from its position. The Company is subject to income tax examinations by major taxing authorities since inception.
Excise Tax
In
connection with the vote to approve the charter amendment proposal presented at the March 2023 Special Meeting, holders of
shares of common stock properly exercised their right to redeem their shares of common stock for an aggregate redemption amount of approximately
$
Franchise Tax
The
Company calculates Franchise Tax liability on a quarterly basis using the estimate calculator on the Delaware Franchise Tax website.
At December 31, 2023, the Company had over accrued the amount due by approximately $
The Company accounts for its common stock subject to possible redemption in accordance with the guidance in ASC Topic 480 “Distinguishing Liabilities from Equity.” Shares of common stock subject to mandatory redemption (if any) is classified as a liability instrument and is measured at fair value. Conditionally redeemable common stock (including common stock that features redemption rights that are either within the control of the holder or subject to redemption upon the occurrence of uncertain events not solely within the Company’s control) is classified as temporary equity. At all other times, common stock is classified as stockholders’ equity. The Company’s shares of common stock sold in the IPO and as a result of the exercise by the underwriters of their over-allotment option features certain redemption rights that are considered to be outside of the Company’s control and subject to occurrence of uncertain future events. Accordingly, on March 31, 2024 and December 31, 2023, shares of common stock subject to possible redemption were presented as temporary equity, outside of the stockholders’ deficit section of the Company’s consolidated balance sheets.
On
March 6, 2023, in connection with the approval of the extension amendment proposal and the trust amendment proposal presented at the
March 2023 Special Meeting, holders of of the Company’s shares of common stock exercised their right to redeem those
shares for cash at an approximate price of $ per share, for an aggregate of approximately $
On November 28, 2023, in connection with the approval of the Extension Amendment Proposal and the Trust Amendment Proposal at the November 2023 Special Meeting, holders of shares of the Company’s common stock exercised their right to redeem those shares for cash at an approximate price of $ per share, for an aggregate of approximately $ million.
Schedule of Common Stock Subject to Possible Redemption | ||||||||
| Shares | Amount | |||||||
| Common stock subject to possible redemption, December 31, 2022 | ||||||||
| Less: | ||||||||
| Redemptions (paid in April and November 2023) | ) | ) | ||||||
| Plus: | ||||||||
| Remeasurement of carrying value to redemption value | ||||||||
| Common stock subject to possible redemption, December 31, 2023 | ||||||||
| Plus: | ||||||||
| Remeasurement of carrying value to redemption value | ||||||||
| Common stock subject to possible redemption, March 31, 2024 | $ | |||||||
| F-28 |
The Company complies with the accounting and disclosure requirements of FASB ASC Topic 260, “Earnings Per Share” and uses the two class method. Net (loss) income per share of common stock is computed by dividing net (loss) income by the weighted average number of shares of common stock outstanding for the period. Any remeasurement of the accretion to redemption value of the shares of common stock subject to possible redemption was considered to be dividends paid to the public stockholders.
The Company has one authorized class of common stock. Warrants included in the Units sold in the IPO (the “Public Warrants”) (see Note 3) and warrants included in the Private Placement Units (the “Private Placement Warrants,” together with the Public Warrants, the “warrants”) (see Note 4) to purchase shares of common stock of the Company at $ per share were issued on December 9, 2021. For the periods ended March 31, 2024 and 2023, no Public Warrants or Private Placement Warrants had been exercised. The potential shares of common stock underlying the outstanding Public Warrants and Private Placement Warrants to purchase the Company’s shares of common stock were excluded from diluted earnings per share for the periods ended March 31, 2024 and 2023 because they are contingently exercisable, and the contingencies have not yet been met. Additionally, the rights are able to be demanded on or any time after the Business Combination, and as the contingency has not been met, the rights are excluded from diluted earnings per share for the periods ended March 31, 2024 and 2023. As a result, diluted net income (loss) per share of common stock is the same as basic net income (loss) per share of common stock for the periods. The table below presents a reconciliation of the numerator and denominator used to compute basic and diluted net income per share for each class of stock.
| For the Three Months Ended March 31, | ||||||||
| 2024 | 2023 | |||||||
| Net (loss) income | $ | ( | ) | $ | ||||
| Remeasurement of common stock subject to redemption | ( | ) | ( | ) | ||||
| Net loss including remeasurement of common stock subject to redemption value | $ | ( | ) | $ | ( | ) | ||
| For the Three Months Ended March 31, | ||||||||||||||||
| 2024 | 2023 | |||||||||||||||
| Redeemable | Non-redeemable | Redeemable | Non-redeemable | |||||||||||||
| Basic and diluted net income (loss) per share of common stock | ||||||||||||||||
| Numerator: | ||||||||||||||||
| Allocation of net loss including remeasurement of common stock subject to redemption value | $ | ( | ) | $ | ( | ) | $ | ( | ) | $ | ( | ) | ||||
| Remeasurement of common stock subject to redemption | ||||||||||||||||
| Allocation of net income (loss), as adjusted | $ | $ | ( | ) | $ | $ | ( | ) | ||||||||
| Denominator: | ||||||||||||||||
| Basic and diluted weighted average shares outstanding | ||||||||||||||||
| Basic and diluted net income (loss) per share of common stock | $ | $ | ) | $ | $ | ) | ||||||||||
| F-29 |
Accounting for Warrants
The Company accounts for warrants as either equity-classified or liability-classified instruments based on an assessment of the warrant’s specific terms and applicable authoritative guidance in FASB ASC 480, Distinguishing Liabilities from Equity (“ASC 480”) and ASC 815, Derivatives and Hedging (“ASC 815”). The assessment considers whether the warrants are freestanding financial instruments pursuant to ASC 480, meet the definition of a liability pursuant to ASC 480, and whether the warrants meet all of the requirements for equity classification under ASC 815, including whether the warrants are indexed to the Company’s own common stock, among other conditions for equity classification. This assessment, which requires the use of professional judgment, is conducted at the time of warrant issuance and as of each subsequent quarterly period end date while the warrants are outstanding. The Company accounts for the warrants issued in connection with the Company’s IPO in accordance with the guidance contained in ASC 815 under which the public warrants meet the criteria for equity treatment and the private warrants do not meet the criteria for equity treatment and must be recorded as liabilities. Accordingly, the Company classifies the private warrants as liabilities at their fair value and adjust the private warrants to fair value at each reporting period. This liability is subject to re-measurement at each balance sheet date until exercised, and any change in fair value is recognized in the Company’s consolidated statements of operations. The fair value of the warrants was estimated using a binomial lattice model.
Recent Accounting Pronouncements
In December 2023, the FASB issued ASU 2023-09, Income Taxes (Topic 740): Improvements to Income Tax Disclosures (ASU 2023-09), which requires disclosure of incremental income tax information within the rate reconciliation and expanded disclosures of income taxes paid, among other disclosure requirements. ASU 2023-09 is effective for fiscal years beginning after December 15, 2024. Early adoption is permitted. The Company’s management does not believe the adoption of ASU 2023-09 will have a material impact on its consolidated financial statements and disclosures.
Note 3 — Initial Public Offering and Over-allotment
Pursuant
to the IPO and the over-allotment in December 2021, the Company sold Units at a price of $ per Unit. Each Unit consists
of one share of common stock, one redeemable warrant (each, a “Public Warrant” and collectively, the “Public Warrants”)
and one right (each a “Public Right” and collectively, the “Public Rights”).
Note 4 — Private Placement
On
December 9, 2021 and December 13, 2021, simultaneously with the consummation of the IPO and the underwriters’ exercise of their
over-allotment option, the Company consummated the issuance and sale (“Private Placement”) of Private Placement Units
in a private placement transaction at a price of $ per Private Placement Unit, generating gross proceeds of $
| F-30 |
Note 5 — Related Party Transactions
Founder Shares
On
August 19, 2021, the Company’s sponsor purchased shares (the “Founder Shares”) of the Company’s common
stock, par value $, for an aggregate price of $
Related Party Loans
In
order to finance transaction costs in connection with a Business Combination, the Company’s sponsor or an affiliate of the sponsor,
or certain of the Company’s officers and directors may, but are not obligated to, loan the Company funds as may be required (“Working
Capital Loans”). If the Company completes a Business Combination, the Company will repay the Working Capital Loans out of the proceeds
of the Trust Account released to the Company. Otherwise, the Working Capital Loans would be repaid only out of funds held outside the
Trust Account. In the event that a Business Combination does not close, the Company may use a portion of proceeds held outside the Trust
Account to repay the Working Capital Loans, but no proceeds held in the Trust Account would be used to repay the Working Capital Loans.
Except for the foregoing, the terms of such Working Capital Loans, if any, have not been determined and no written agreements exist with
respect to such loans. The Working Capital Loans would either be repaid upon consummation of a Business Combination, without interest,
or, at the lender’s discretion, up to $
The
Company entered into promissory notes with Public Gold Marketing Sdn. Bhd., which is considered a related party due to a familial relationship
between the controlling member of the sponsor and a 95% shareholder of Public Gold Marketing Sdn. Bhd. The promissory notes bear an interest
of
Support Services
The
Company has entered into an administrative services agreement pursuant to which the Company will pay its sponsor a total of $
Advances
On
each of September 5, 2023, September 29, 2023 and November 7, 2023, an affiliate of the Company’s sponsor advanced $
| F-31 |
Note 6 — Commitments and Contingencies
Registration Rights
The holders of Founder Shares, Private Placement Units and warrants that may be issued upon conversion of Working Capital Loans, if any, will be entitled to registration rights (in the case of the Founder Shares, only after conversion of such shares into shares of common stock) pursuant to a registration rights agreement signed on the date of the prospectus for the IPO. These holders are entitled to certain demand and “piggyback” registration rights. However, the registration rights agreement provides that the Company will not permit any registration statement filed under the Securities Act to become effective until the termination of the applicable lock-up period for the securities to be registered. The Company will bear the expenses incurred in connection with the filing of any such registration statements.
Underwriting Agreement
The
underwriters were paid a cash underwriting discount of $ per unit on the offering including the Units issued with the underwriter’s
exercise of their over-allotment option, or $
Note 7 — Promissory Notes – Related Party
On
March 3, 2023, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount
of $
On
March 23, 2023, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount
of up to $
On
June 2, 2023, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount of
up to $
On
October 13, 2023, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount
of $
On
December 8, 2023, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount
of $
On
January 5, 2024, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount
of $
On
January 25, 2024, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount
of $
On
February 22, 2024, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount
of $
For
the three months ended March 31, 2024 and 2023, the notes have incurred $
Note 8 — Stockholders’ Deficit
Common stock
The Company is authorized to issue shares of common stock with a par value of $per share. As of March 31, 2024 and December 31, 2023, there were (excluding shares of common stock subject to possible redemption) shares of common stock issued and outstanding.
Warrants:
As
of March 31, 2024 and December 31, 2023, the Company had
The Public Warrants are accounted for as equity instruments in the Company’s consolidated financial statements. Public Warrants may only be exercised for a whole number of shares. No fractional shares will be issued upon exercise of the Public Warrants. The Public Warrants will become exercisable on the later of the completion of an initial Business Combination and will expire five years after the completion of an initial Business Combination, or earlier upon redemption. No Public Warrants will be exercisable for cash unless the Company has an effective and current registration statement covering the shares of common stock issuable upon exercise of the Public Warrants and a current prospectus relating to such shares of common stock. Notwithstanding the foregoing, if a registration statement covering the shares of common stock issuable upon exercise of the Public Warrants is not effective within a specified period following the consummation of a Business Combination, warrant holders may, until such time as there is an effective registration statement and during any period when the Company shall have failed to maintain an effective registration statement, exercise warrants on a cashless basis pursuant to the exemption provided by Section 3(a)(9) of the Securities Act, provided that such exemption is available. If that exemption, or another exemption, is not available, holders will not be able to exercise their warrants on a cashless basis. The Public Warrants will expire five years after the completion of a Business Combination or earlier upon redemption or liquidation.
| F-32 |
Redemption of warrants when the price per common stock equals or exceeds $
Once the warrants become exercisable, the Company may redeem the outstanding warrants (except as described herein with respect to the private placement warrants):
| ● | in whole and not in part; | |
| ● | at
a price of $ | |
| ● | upon a minimum of 30 days’ prior written notice of redemption, which the Company refers to as the “30-day redemption period”; and | |
| ● | if, and only if, the last reported sale price (the “closing price”) of the Company’s common stock equals or exceeds $ per share (as adjusted for adjustments to the number of shares issuable upon exercise or the exercise price of a warrant as described under the heading “Description of Securities—Warrants”) for any 20 trading days within a 30-trading day period ending on the third trading day prior to the date on which the Company sends the notice of redemption to the warrant holders. |
The Company will not redeem the warrants as described above unless an effective registration statement under the Securities Act covering the shares of common stock issuable upon exercise of the warrants is effective and a current prospectus relating to those shares of common stock is available throughout the 30-day redemption period. If and when the warrants become redeemable by the Company, the Company may exercise its redemption right even if the Company is unable to register or qualify the underlying securities for sale under all applicable state securities laws.
If the Company calls the Public Warrants for redemption, management will have the option to require all holders that wish to exercise the Public Warrants to do so on a “cashless basis,” as described in the warrant agreement.
The Private Placement Warrants are substantially in the same form as the Public Warrants, except they (i) will be exercisable either for cash or on a cashless basis at the holder’s option pursuant and (ii) will not be redeemable by the Company, in either case as long as the Private Placement Warrants are held by the initial purchasers or any of their permitted transferees (as prescribed in the subscription agreement, dated December 6, 2021, by and between the Company and Public Gold Marketing Sdn. Bhd.). Once a Private Placement Warrant is transferred to a holder other than a permitted transferee, it shall be treated as a Public Warrant for all purposes. Due to these terms the Private Warrants are required to be liability classified.
The exercise price and number of shares of common stock issuable on exercise of the warrants may be adjusted in certain circumstances, including in the event of a share dividend, extraordinary dividend or recapitalization, reorganization, merger or consolidation. However, the warrants will not be adjusted for issuances of shares of common stock at a price below their respective exercise prices. Additionally, in no event will the Company be required to net cash settle the warrants. If the Company is unable to complete a Business Combination within the Combination Period and the Company liquidates the funds held in the Trust Account, holders of the warrants will not receive any of such funds with respect to their warrants, nor will they receive any distribution from the Company’s assets held outside of the Trust Account with the respect to such warrants. Accordingly, the warrants may expire worthless.
In
addition, if the Company issues additional shares of common stock or equity-linked securities for capital raising purposes in connection
with the closing of a Business Combination at an issue price or effective issue price of less than $
Rights
Except in cases where the Company is not the surviving company in a Business Combination, each holder of a Public Right will automatically receive one-tenth of one share of common stock upon consummation of a Business Combination, even if the holder of a Public Right converted all shares held by him, her or it in connection with a Business Combination or an amendment to the Company’s Certificate of Incorporation with respect to its pre-business combination activities. In the event that the Company will not be the surviving company upon completion of a Business Combination, each holder of a Public Right will be required to affirmatively convert his, her or its rights in order to receive the one-tenth of a share underlying each Public Right upon consummation of the Business Combination.
The Company will not issue fractional shares in connection with an exchange of Public Rights. Fractional shares will either be rounded down to the nearest whole share or otherwise addressed in accordance with the applicable provisions of the Delaware General Corporation Law. As a result, the holders of the Public Rights must hold rights in multiples of 10 in order to receive shares for all of the holders’ rights upon closing of a Business Combination.
| F-33 |
Note 9 — Fair Value Measurements
The fair value of the Company’s financial assets and liabilities reflects management’s estimate of amounts that the Company would have received in connection with the sale of the assets or paid in connection with the transfer of the liabilities in an orderly transaction between market participants at the measurement date. In connection with measuring the fair value of its assets and liabilities, the Company seeks to maximize the use of observable inputs (market data obtained from independent sources) and to minimize the use of unobservable inputs (internal assumptions about how market participants would price assets and liabilities). The following fair value hierarchy is used to classify assets and liabilities based on the observable inputs and unobservable inputs used in order to value the assets and liabilities:
Level 1: Quoted prices in active markets for identical assets or liabilities. An active market for an asset or liability is a market in which transactions for the asset or liability occur with sufficient frequency and volume to provide pricing information on an ongoing basis.
Level 2: Observable inputs other than Level 1 inputs. Examples of Level 2 inputs include quoted prices in active markets for similar assets or liabilities and quoted prices for identical assets or liabilities in markets that are not active.
Level 3: Unobservable inputs based on the Company’s assessment of the assumptions that market participants would use in pricing the asset or liability.
As of March 31, 2024 and December 31, 2023 the assets held in the Trust Account were held in cash.
The following table presents information about the Company’s liabilities that are measured at fair value on a recurring basis as of March 31, 2024 and December 31, 2023 and indicates the fair value hierarchy of the valuation inputs the Company utilized to determine such fair value.
| Quoted Prices in Active Markets | Significant Other Observable Inputs | Significant Other Unobservable Inputs | ||||||||||||||
| Level | (Level 1) | (Level 2) | (Level 3) | |||||||||||||
| March 31, 2024 | ||||||||||||||||
| Liabilities: | ||||||||||||||||
| Warrant Liabilities- Private Warrants | 3 | $ | | |||||||||||||
| December 31, 2023 | ||||||||||||||||
| Liabilities: | ||||||||||||||||
| Warrant Liabilities- Private Warrants | 3 | |||||||||||||||
| F-34 |
The Private Placement Warrants were valued using a binomial lattice model, which is considered to be a Level 3 fair value measurement. One of the more significant inputs is the implied volatility, which is based on the observed prices of the Company’s common stock and publicly-traded warrants. As of March 31, 2024 and December 31, 2023, the estimated fair value of Warrant Liabilities – Private Warrants were determined based on the following significant inputs and are expressed on the basis of each being exercisable for a one-half of one share of common stock:
As of March 31, 2024 | As of December 31, 2023 | |||||||
| Exercise price | $ | $ | ||||||
| Market price of public stock | $ | $ | ||||||
| Term (years) | ||||||||
| Volatility | ||||||||
| Risk-free rate | % | % | ||||||
| Dividend yield | % | % | ||||||
The following table presents the changes in the fair value of warrant liabilities for the three months ended March 31, 2024 and 2023:
| Private Placement Warrants | ||||
| Fair value as of January 1, 2024 | $ | |||
| Change in valuation inputs or other assumptions | ||||
| Fair value as of March 31, 2024 | $ | |||
| Private Placement Warrants | ||||
| Fair value as of January 1, 2023 | $ | |||
| Change in valuation inputs or other assumptions | ( | ) | ||
| Fair value as of March 31, 2023 | $ | |||
Note 10 — Subsequent Events
The Company evaluated subsequent events and transactions that occurred after the balance sheet date up to the date that the unaudited condensed consolidated financial statements were issued. Based upon this review the Company did not identify any subsequent events, other than the below, that would have required adjustment or disclosure in the consolidated financial statements.
On April 3, 2024, Globalink Merger Sub (Cayman), was incorporated as a wholly-owned subsidiary of Globalink.
On
April 4, 2024, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount
of $
On
April 9, 2024, the Company deposited an aggregate of $
On
May 4, 2024, the Company deposited an aggregate of $
| F-35 |
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Shareholders and Board of Directors of
Globalink Investment Inc.
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheets of Globalink Investment Inc. (the “Company”) as of December 31, 2023 and 2022, the related consolidated statements of operations, changes in stockholders’ deficit and cash flows for each of the two years in the period ended December 31, 2023, and the related notes (collectively referred to as the “financial statements”). In our opinion, the financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 2023 and 2022, and the results of its operations and its cash flows for each of the two years in the period ended December 31, 2023, in conformity with accounting principles generally accepted in the United States of America.
Explanatory Paragraph – Going Concern
The accompanying financial statements have been prepared assuming that the Company will continue as a going concern. As described in Note 1 to the financial statements, the Company is a Special Purpose Acquisition Corporation that was formed for the purpose of entering into a merger, share exchange, asset acquisition, stock purchase, recapitalization, reorganization or other similar business combination with one or more businesses or entities on or before April 9, 2024 or December 9, 2024 by making an additional monthly contribution to the trust account subject to the approval of the board of directors. The Company entered into a definitive merger agreement with a business combination target on January 30, 2024; however, the completion of this transaction is subject to the approval of the Company’s stockholders among other conditions. There is no assurance that the Company will obtain the necessary approvals, satisfy the required closing conditions, raise the additional capital it needs to fund its operations, and complete the transaction prior to April 9, 2024, if at all. The Company also has no approved plan in place to extend the business combination deadline and fund operations for any period of time after April 9, 2024, in the event that it is unable to complete a business combination by that date. These matters raise substantial doubt about the Company’s ability to continue as a going concern. Management’s plans with regard to these matters are also described in Note 1. The financial statements do not include any adjustments that may be necessary should the Company be unable to continue as a going concern.
Basis for Opinion
These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (“PCAOB”) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audits to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
/s/ Marcum LLP
Marcum LLP
We have served as the Company’s auditor since 2021 (such date takes into account the acquisition of certain assets of Friedman LLP by Marcum LLP effective September 1, 2022)
Costa Mesa, CA
April 1, 2024
| F-36 |
GLOBALINK INVESTMENT INC.
CONSOLIDATED BALANCE SHEETS
| December 31, | ||||||||
| 2023 | 2022 | |||||||
| ASSETS | ||||||||
| CURRENT ASSETS | ||||||||
| Cash and cash in escrow account | $ | $ | ||||||
| Prepaid expenses – current | ||||||||
| Total current assets | ||||||||
| Cash and investments held in Trust Account | ||||||||
| TOTAL ASSETS | $ | $ | ||||||
LIABILITIES, REDEEMABLE COMMON STOCK AND STOCKHOLDERS’ DEFICIT | ||||||||
| CURRENT LIABILITIES | ||||||||
| Accounts payable | $ | $ | ||||||
| Franchise tax payable | ||||||||
| Income tax payable | ||||||||
| Promissory note – related party | ||||||||
| Due to related parties | ||||||||
| Excise tax liability | ||||||||
| Total current liabilities | ||||||||
| Deferred tax liability | ||||||||
| Warrant liabilities | ||||||||
| Deferred underwriting fee payable | ||||||||
| Total Liabilities | ||||||||
| COMMITMENTS AND CONTINGENCIES | ||||||||
| REDEEMABLE COMMON STOCK | ||||||||
Common stock subject to possible redemption, $ par value, and shares at redemption value at December 31, 2023 and 2022 of $ and $ per share, respectively | ||||||||
| STOCKHOLDERS’ DEFICIT | ||||||||
Common stock, $ par value; shares authorized; shares issued and outstanding at December 31, 2023 and 2022 (excluding and shares subject to possible redemption) | ||||||||
| Accumulated deficit | ( | ) | ( | ) | ||||
| Total Stockholders’ Deficit | ( | ) | ( | ) | ||||
| LIABILITIES, REDEEMABLE COMMON STOCK AND STOCKHOLDERS’ DEFICIT | $ | $ | ||||||
The accompanying notes are an integral part of the consolidated financial statements.
| F-37 |
GLOBALINK INVESTMENT INC.
CONSOLIDATED STATEMENTS OF OPERATIONS
| For the Year Ended December 31, | ||||||||
| 2023 | 2022 | |||||||
| OPERATING EXPENSES | ||||||||
| General and administrative expenses | $ | $ | ||||||
| Franchise tax expense | ||||||||
| Total operating expenses | ( | ) | ( | ) | ||||
| OTHER INCOME (EXPENSE) | ||||||||
| Income on cash and investments held in Trust Account | ||||||||
| Penalties on income tax | ( | ) | ||||||
| Interest expense | ( | ) | ||||||
| Change in fair value of warrant liabilities | ||||||||
| Total other income, net | ||||||||
| Income before provision for income taxes | ||||||||
| Provision for income taxes | ( | ) | ( | ) | ||||
| NET INCOME | $ | $ | ||||||
| Weighted average shares outstanding Common stock – redeemable | ||||||||
| Basic and diluted net income per share, Common stock – redeemable | ||||||||
| Weighted average shares outstanding Common stock – non-redeemable | ||||||||
| Basic and diluted net loss per share, Common stock – non-redeemable | $ | ) | $ | ) | ||||
The accompanying notes are an integral part of the consolidated financial statements.
| F-38 |
GLOBALINK INVESTMENT INC.
CONSOLIDATED STATEMENTS OF CHANGES IN STOCKHOLDERS’ DEFICIT
FOR THE YEARS ENDED DECEMBER 31, 2023 AND 2022
| Common Stock | Additional Paid-in | Accumulated | Total Stockholders’ | |||||||||||||||||
| Shares | Amount | Capital | Deficit | Deficit | ||||||||||||||||
| Balance - December 31, 2021 | $ | $ | $ | ( | $ | ( | ) | |||||||||||||
| Remeasurement of common stock subject to redemption | — | ( | ) | ( | ) | |||||||||||||||
| Net income | — | | ||||||||||||||||||
| Balance - December 31, 2022 | ( | ) | ( | ) | ||||||||||||||||
| Excise tax imposed on common stock redemptions | — | ( | ) | ( | ) | |||||||||||||||
| Remeasurement of common stock subject to redemption | — | ( | ) | ( | ) | |||||||||||||||
| Net income | — | |||||||||||||||||||
| Balance - December 31, 2023 | $ | $ | $ | ( | ) | $ | ( | ) | ||||||||||||
The accompanying notes are an integral part of the consolidated financial statements.
| F-39 |
GLOBALINK INVESTMENT INC.
CONSOLIDATED STATEMENTS OF CASH FLOWS
| For
the Year Ended December 31, | ||||||||
| 2023 | 2022 | |||||||
| CASH FLOWS FROM OPERATING ACTIVITIES: | ||||||||
| Net income | $ | $ | ||||||
| Adjustments to reconcile net income to net cash used in operating activities: | ||||||||
| Interest income on cash and investments held in Trust Account | ( | ) | ( | ) | ||||
| Change in fair value of warrant liabilities | ( | ) | ( | ) | ||||
| Changes in operating assets and liabilities: | ||||||||
| Prepaid expenses | ||||||||
| Deferred tax liability | ( | ) | ||||||
| Due to related parties | ||||||||
| Income tax payable | ||||||||
| Interest expense accrual | ||||||||
| Accounts payable | ( | ) | ||||||
| Franchise tax payable | ( | ) | ||||||
| Net cash used in operating activities | ( | ) | ( | ) | ||||
| Cash Flows from Investing Activities: | ||||||||
| Cash deposited to Trust Account | ( | ) | ||||||
| Cash withdrawn from Trust Account to pay tax obligations | ||||||||
| Cash withdrawn from trust in connection with redemption of common stock | ||||||||
| Net cash provided by investing activities | ||||||||
| Cash Flows from Financing Activities: | ||||||||
| Issuance of promissory notes – related party | ||||||||
| Due to affiliate - advance | ||||||||
| Redemption of common stock | ( | ) | ||||||
| Net cash used in financing activities | ( | ) | ||||||
| NET CHANGE IN CASH | ( | ) | ( | ) | ||||
| CASH, BEGINNING OF PERIOD | ||||||||
| CASH, END OF PERIOD | $ | $ | ||||||
| Supplementary cash flow information: | ||||||||
| Cash paid for interest expense | $ | $ | ||||||
| Cash paid for income taxes | $ | $ | ||||||
| Non-cash investing and financing activities: | ||||||||
| Excise tax accrued for common stock redemptions | $ | $ | ||||||
| Remeasurement of Common stock subject to redemption | $ | $ | ||||||
The accompanying notes are an integral part of the consolidated financial statements.
| F-40 |
Note 1 – Description of Organization and Business Operations and Liquidity
Globalink Investment Inc. (the “Company”) was incorporated in Delaware on March 24, 2021. The Company is a blank check company formed for the purpose of entering into a merger, share exchange, asset acquisition, stock purchase, recapitalization, reorganization or other similar business combination with one or more businesses or entities (the “Business Combination”). On July 27, 2022, Globalink Merger Sub, Inc., a Nevada corporation and a wholly-owned subsidiary of Globalink, was formed.
The Company is not limited to a particular industry or geographic region for purposes of consummating a Business Combination. The Company is an early stage and emerging growth company and, as such, the Company is subject to all of the risks associated with early stage and emerging growth companies.
As
of December 31, 2023, the Company had not commenced any operations. All activity through December 31, 2023 relates to the Company’s
formation and Initial Public Offering (“IPO”), which is described below and, since the offering, the search for a prospective
initial Business Combination. The Company will not generate any operating revenues until after the completion of its initial Business
Combination, at the earliest. The Company generates non-operating income in the form of interest income earned on investments from the
proceeds derived from the IPO. The registration statement for the Company’s IPO was declared effective on December 6, 2021. On
December 9, 2021, the Company consummated the IPO of units (“Units”) at $ per Unit generating gross proceeds
of $
Simultaneously
with the closing of the IPO, the Company consummated the sale of
units (“Private Placement Units”) at a price of $
per Private Placement Unit in a private placement to Public Gold Marketing Sdn. Bhd., a Malaysian private limited company, a related party, the sponsor or the underwriters, generating gross proceeds of $
Additionally
with the closing of the IPO, the Company granted the underwriters a 45-day option to purchase up to Units to cover over-allotment.
On December 13, 2021, the underwriters fully exercised the option and purchased additional Units (the “Over-allotment
Units”), generating additional gross proceeds of $
Simultaneously
with the exercise of the over-allotment, the Company consummated a private sale of an additional Private Placement Units to Public
Gold Marketing Sdn. Bhd at a price of $ per Private Placement Unit, generating additional gross proceeds of $
Offering
costs for the IPO and the exercise of the underwriters’ over-allotment option amounted to $
Following
the closing of the IPO, $
To mitigate the risk of being deemed to have been operating as an unregistered investment company (including under the subjective test of Section 3(a)(1)(A) of the Investment Company Act), on July 27, 2023, the Company instructed Continental Stock Transfer & Trust Company, the trustee of the Trust Account (the “Trustee” or “Continental”), to liquidate the U.S. government securities or money market funds held in the Trust Account and thereafter to hold all funds in the Trust Account in cash (which may include demand deposit accounts) until the earlier of consummation of our Business Combination or liquidation. Furthermore, such cash is held in bank accounts, which exceed federally insured limits as guaranteed by the Federal Deposit Insurance Corporation (the “FDIC”).
| F-41 |
The
Company’s management has broad discretion with respect to the specific application of the net proceeds of the IPO and the sale
of the Private Placement Units, although substantially all of the net proceeds are intended to be applied generally toward consummating
a Business Combination. There is no assurance that the Company will be able to complete a Business Combination successfully.
The Company will provide the holders (the “Public Stockholders”) of the outstanding shares of common stock included in the Units, or the Public Shares with the opportunity to redeem all or a portion of their Public Shares upon the completion of a Business Combination either (i) in connection with a stockholder meeting called to approve the Business Combination or (ii) by means of a tender offer. The decision as to whether the Company will seek stockholder approval of a Business Combination or conduct a tender offer will be made by the Company. The Public Stockholders will be entitled to redeem their Public Shares for a pro rata portion of the amount then in the Trust Account (initially anticipated to be $ per Public Share, plus any pro rata interest then in the Trust Account, net of taxes payable). There will be no redemption rights with respect to the Company’s warrants.
All
of the Public Shares contain a redemption feature, which allows for the redemption of such Public Shares in connection with the Company’s
liquidation, if there is a stockholder vote or tender offer in connection with the Company’s Business Combination and in connection
with certain amendments to the Company’s amended and restated certificate of incorporation. In accordance with Financial Accounting
Standards Board (“FASB”) Accounting Standards Codification (“ASC”) 480-10-S99, redemption provisions not solely
within the control of a company require the Public Shares subject to redemption to be classified outside of permanent equity. Given that
the Public Shares will be issued with other freestanding instruments (i.e., public warrants and rights), the initial carrying value of
common stock classified as temporary equity will be the allocated proceeds determined in accordance with ASC 470-20. The Public Shares
are subject to ASC 480-10-S99. If it is probable that the equity instrument will become redeemable, the Company has the option to either
(i) accrete changes in the redemption value over the period from the date of issuance (or from the date that it becomes probable that
the instrument will become redeemable, if later) to the earliest redemption date of the instrument or (ii) recognize changes in the redemption
value immediately as they occur and adjust the carrying amount of the instrument to equal the redemption value at the end of each reporting
period. The Company has elected to recognize the changes immediately. While redemptions cannot cause the Company’s net tangible
assets to fall below $
Redemptions of the Company’s Public Shares may be subject to the satisfaction of conditions, including minimum cash conditions, pursuant to an agreement relating to the Company’s Business Combination. If the Company seeks stockholder approval of the Business Combination, the Company will proceed with a Business Combination if a majority of the shares voted are voted in favor of the Business Combination, or such other vote as required by law or stock exchange rule. If a stockholder vote is not required by applicable law or stock exchange listing requirements and the Company does not decide to hold a stockholder vote for business or other reasons, the Company will, pursuant to its Certificate of Incorporation, conduct the redemptions pursuant to the tender offer rules of the U.S. Securities and Exchange Commission (“SEC”) and file tender offer documents with the SEC prior to completing a Business Combination. If, however, stockholder approval of the transaction is required by applicable law or stock exchange listing requirements, or the Company decides to obtain stockholder approval for business or other reasons, the Company will offer to redeem shares in conjunction with a proxy solicitation pursuant to the proxy rules and not pursuant to the tender offer rules. If the Company seeks stockholder approval in connection with a Business Combination, the sponsor has agreed to vote its Founder Shares (as defined in Note 5) and any Public Shares purchased during or after the IPO in favor of approving a Business Combination. Additionally, each Public Stockholder may elect to redeem their Public Shares without voting, and if they do vote, irrespective of whether they vote for or against the proposed transaction.
| F-42 |
Notwithstanding the foregoing, the amended and restated certificate of incorporation of the Company (the “Certificate of Incorporation”) provides that a Public Stockholder, together with any affiliate of such stockholder or any other person with whom such stockholder is acting in concert or as a “group” (as defined under Section 13 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”)), will be restricted from redeeming its shares with respect to more than an aggregate of % or more of the shares of common stock sold in the IPO, without the prior consent of the Company.
The
Company originally had until March 9, 2023, 15 months from the closing of the IPO to complete a Business Combination. On March 6, 2023,
the Company held a special meeting (the “March 2023 Special Meeting”), during which the stockholders of the Company approved
a proposal to amend the Company’s amended and restated certified articles of incorporation which included extending the time in
which the Company must complete a Business Combination (the “Extension Amendment Proposal”) and a proposal to amend the Company’s
investment management trust agreement, dated as of December 6, 2021 (the “Trust Agreement”), by and between the Company and
Continental (the “Trust Amendment Proposal”). The Company will have the option of two (2) three-months extensions, followed
by three (3) one-month extensions, or until December 9, 2023, if all extensions are exercised. The Company exercised the option for two
three-month extensions and as a result the Company has deposited a total of $
Through
a total of five elections from March 2023 to December 2023, Globalink elected to extend the Termination Date to December 9, 2023 and
deposited an aggregate of US $
On
March 6, 2023, in connection with the approval of the proposals presented at the March 2023 Special Meeting which extended the time in
which the Company must complete a Business Combination, holders of of the Company’s shares of common stock exercised
their right to redeem those shares for cash at an approximate price of $ per share, for an aggregate of approximately $
On October 16, 2023, the Company received a written notice (the “Notice”) from the Nasdaq Listing Qualifications Department of The Nasdaq Stock Market (“Nasdaq”) indicating that the Company was not in compliance with Nasdaq Listing Rule 5450(a)(2), which requires the Company to maintain at least 400 total holders for continued listing on the Nasdaq Global Market (the “Minimum Total Holders Rule”). The Notice is only a notification of deficiency, not of imminent delisting, and has no current effect on the listing or trading of the Company’s securities on the Nasdaq Global Market. The Notice states that the Company has 45 calendar days, or until November 30, 2023, to submit a plan to regain compliance with the Minimum Total Holders Rule. On January 29, 2024, the Company submitted an application to phase-down from The Nasdaq Global Market to The Nasdaq Capital Market. On March 6, 2024, the Company received a letter from the Nasdaq Listing Qualifications staff granting the Company’s request for transfer to The Nasdaq Capital Market. The Company’s securities will be transferred to The Nasdaq Capital Market at the opening of business on March 12, 2024. In connection with the approval of the phase-down application, the staff indicated that the Company’s deficiency with the Minimum Total Holders Rule was cured and the matter is closed.
| F-43 |
On
November 28, 2023,
On
November 28, 2023, the stockholders of the Company approved a proposal to amend the Company’s amended and restated certificate
of incorporation, allowing the Company to Extended Deadline from December 9, 2023 to up to December 9, 2024 through monthly extensions.
To obtain each extension, the Company, its sponsor or any of their affiliates or designees must deposit into the Company’s trust
account with Continental by the deadline applicable prior to the extension, $
As of the date of this report, the Company has extended the Termination Date seven times under its current amended and restated certificate of incorporation, as amended (or nine times since the IPO), and has until April 9, 2024 to complete its initial business combination. The Company may continue to extend the Termination Date to up to December 9, 2024 through monthly extensions.
| F-44 |
Through the Insider Letter, in consideration for inducing Chardan, the representative of the underwriters for Globalink’s IPO to proceed with the IPO, and the economic benefit the IPO would confer upon the Initial Stockholders and PGM, each of the Initial Stockholders and PGM have agreed to waive their liquidation rights with respect to the Founder Shares if the Company fails to complete a Business Combination within the Combination Period. However, if the Initial Stockholders should acquire any Public Shares in or after the IPO, they will be entitled to liquidating distributions from the Trust Account with respect to such Public Shares if the Company fails to complete a Business Combination within the Combination Period. The underwriters have agreed to waive their rights to deferred underwriting discounts (see Note 6) held in the Trust Account in the event the Company does not complete a Business Combination within the Combination Period, and, in such event, such amounts will be included with the other funds held in the Trust Account that will be available to fund the redemption of the Public Shares. In the event of such distribution, it is possible that the per share value of the residual assets remaining available for distribution (including Trust Account assets) will be only $ per share held in the Trust Account. In order to protect the amounts held in the Trust Account, the sponsor has agreed to be liable to the Company if and to the extent any claims by a vendor for services rendered or products sold to the Company, or a prospective target business with which the Company has discussed entering into a transaction agreement, reduce the amount of funds in the Trust Account. This liability will not apply with respect to any claims by a third party who executed a waiver of any right, title, interest or claim of any kind in or to any monies held in the Trust Account or to any claims under the Company’s indemnity of the underwriters of the IPO against certain liabilities, including liabilities under the Securities Act of 1933, as amended (the “Securities Act”). Moreover, in the event that an executed waiver is deemed to be unenforceable against a third party, the sponsor will not be responsible to the extent of any liability for such third-party claims. The Company will seek to reduce the possibility that the sponsor will have to indemnify the Trust Account due to claims of creditors by endeavoring to have all vendors, service providers (except the Company’s independent registered public accounting firm), prospective target businesses or other entities with which the Company does business, execute agreements waiving any right, title, interest or claim of any kind in or to monies held in the Trust Account.
Business Combination
On August 3, 2022, the Company entered into an Agreement and Plan of Merger (the “Merger Agreement”) by and among Tomorrow Crypto Group Inc., a Nevada corporation (“Tomorrow Crypto”), Globalink Merger Sub, Inc., a Nevada corporation and a wholly-owned subsidiary of Globalink (“Merger Sub”), GL Sponsor LLC, a Delaware limited liability company
In accordance with the termination provisions under Section 10.1 of the Merger Agreement, the Merger Agreement was terminated on March 8, 2023 (the “Merger Agreement Termination Date”). In conjunction with the termination of the Merger Agreement, the Additional Agreements (as defined in the Merger Agreement) (including the Support Agreements) were also terminated in accordance with their respective terms as of March 8, 2023, the Merger Agreement Termination Date.
On January 30, 2024, the Company entered into a Merger Agreement (the “Merger Agreement”) by and among Alps Global Holding Berhad, a Malaysian company (“Alps”), GL Sponsor LLC and Dr. Tham Seng Kong, an individual. Pursuant to the terms of the Merger Agreement, a business combination between Globalink and Alps through the merger of a to be incorporated subsidiary of Globalink in the Cayman Islands (the “Merger Sub”) with and into Alps, with Alps surviving the merger (the “Surviving Company”) as a wholly-owned subsidiary of Globalink (the “Merger”, and, together with the other transactions contemplated by the Merger Agreement, the “Transactions”). After the date of the Merger Agreement and prior to the consummation of the transactions contemplated by the Merger Agreement (the “Closing”), a company formed under the laws of the Cayman Islands will be incorporated (“Cayman Holdco”), whereupon it is envisaged that Alps will become a subsidiary of the Cayman Holdco.
| F-45 |
Risks and Uncertainties
The Company continues to evaluate the impact of increases in inflation and rising interest rates, financial market instability, including the recent bank failures, the potential government shutdown, the lingering effects of the COVID-19 pandemic and certain geopolitical events, including the wars in Ukraine and the surrounding region and between Israel and Hamas. The Company has concluded that while it is reasonably possible that the risks and uncertainties related to or resulting from these events could have a negative effect on its financial position, results of operations and/or ability to complete an initial Business Combination, the Company cannot at this time fully predict the likelihood of one or more of the above events, their duration or magnitude or the extent to which they may negatively impact the Company’s business and its ability to complete an initial Business Combination.
On
August 16, 2022, the Inflation Reduction Act of 2022 (the “IR Act”) was signed into federal law. The IR Act provides for,
among other things, a new U.S. federal
Any redemption or other repurchase that occurs after December 31, 2022, in connection with a Business Combination, extension vote or otherwise, may be subject to the excise tax. Whether and to what extent the Company would be subject to the excise tax in connection with a Business Combination, extension vote or otherwise would depend on a number of factors, including (i) the fair market value of the redemptions and repurchases in connection with the Business Combination, extension or otherwise, (ii) the structure of a Business Combination, (iii) the nature and amount of any “PIPE” or other equity issuances in connection with a Business Combination (or otherwise issued not in connection with a Business Combination but issued within the same taxable year of a Business Combination) and (iv) the content of regulations and other guidance from the Treasury. In addition, because the excise tax would be payable by the Company and not by the redeeming holder, the mechanics of any required payment of the excise tax have not been determined. The foregoing could cause a reduction in the cash available on hand to complete a Business Combination and in the Company’s ability to complete a Business Combination.
Liquidity, Capital Resources and Going Concern
As
of December 31, 2023, the Company had $
Until the consummation of a Business Combination, the Company will be using the funds not held in the Trust Account for identifying and evaluating prospective acquisition candidates, performing due diligence on prospective target businesses, paying for travel expenditures, selecting the target business to acquire, and structuring, negotiating and consummating the Business Combination. The Company will need to raise additional capital through loans or additional investments from its sponsor, stockholders, officers, directors, or third parties. The Company’s officers, directors and the sponsor may, but are not obligated to, loan the Company funds, from time to time or at any time, in whatever amount they deem reasonable in their sole discretion, to meet the Company’s working capital needs. Accordingly, the Company may not be able to obtain additional financing.
If the Business Combination is not consummated, the Company will need to raise additional capital through loans or additional investments from its sponsor, stockholders, officers, directors, or third parties. The Company’s officers, directors and its sponsor may, but are not obligated to, loan the Company funds, from time to time or at any time, in whatever amount they deem reasonable in their sole discretion, to meet the Company’s working capital needs. Accordingly, the Company may not be able to obtain additional financing. If the Company is unable to raise additional capital, it may be required to take additional measures to conserve liquidity, which could include, but not necessarily be limited to, curtailing operations, suspending the pursuit of a potential transaction, and reducing overhead expenses. The Company cannot provide any assurance that new financing will be available to it on commercially acceptable terms, if at all.
| F-46 |
In connection with the Company’s assessment of going concern considerations in accordance with FASB Accounting Standards Update (“ASU”) 2014-15, “Disclosures of Uncertainties about an Entity’s Ability to Continue as a Going Concern,” the Company has up until December 9, 2024 to consummate a Business Combination if it elects to extend the Termination Date in accordance with its Amended and Restated Certificate of Incorporation as currently in effect. It is uncertain that the Company will be able to consummate a Business Combination by this time. If a Business Combination is not consummated by this date and an extension is not requested by the Company’s sponsor, there will be a mandatory liquidation and subsequent dissolution of the Company. Management has determined that the mandatory liquidation, should a Business Combination not occur and an extension is not requested by the Company’s sponsor, and potential subsequent dissolution as well as liquidity condition noted above raise substantial doubt about the Company’s ability to continue as a going concern. No adjustments have been made to the carrying amounts of assets or liabilities should the Company be required to liquidate after December 9, 2024.
Note 2 — Summary of Significant Accounting Policies
Basis of Presentation
The accompanying consolidated financial statements are presented in U.S. dollars and have been prepared in accordance with accounting principles generally accepted in the United States of America (“U.S. GAAP”) and pursuant to the accounting and disclosure rules and regulations of the Securities and Exchange Commission (the “SEC”).
Principles of Consolidation
The accompanying consolidated financial statements include the accounts of the Company and its wholly owned subsidiary. All significant intercompany balances and transactions have been eliminated in consolidation.
Emerging Growth Company
The Company is an emerging growth company as defined in Section 102(b)(1) of the Jumpstart Our Business Startups Act of 2012 (the “JOBS Act”), which exempts emerging growth companies from being required to comply with new or revised financial accounting standards until private companies (that is, those that have not had a Securities Act registration statement declared effective or do not have a class of securities registered under the Exchange Act) are required to comply with the new or revised financial accounting standards. The JOBS Act provides that an emerging growth company can elect to opt out of the extended transition period and comply with the requirements that apply to non-emerging growth companies but any such an election to opt out is irrevocable. The Company has elected not to opt out of such extended transition period, which means that when a standard is issued or revised, and it has different application dates for public or private companies, the Company, as an emerging growth company, can adopt the new or revised standard at the time private companies adopt the new or revised standard. This may make comparison of the Company’s consolidated financial statements with another public company that is neither an emerging growth company nor an emerging growth company that has opted out of using the extended transition period difficult or impossible because of the potential differences in accounting standards used.
Use of Estimates
The preparation of consolidated financial statements in conformity with U.S. GAAP requires the Company’s management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the financial statements. Making estimates requires management to exercise significant judgment. One of the more significant accounting estimates included in these consolidated financial statements is the determination of the fair value of the warrant liabilities. Such estimates may be subject to change as more current information becomes available and accordingly the actual results could differ significantly from those estimates. It is at least reasonably possible that the estimate of the effect of a condition, situation or set of circumstances that existed at the date of the financial statements, which management considered in formulating its estimate, could change in the near term due to one or more future confirming events.
| F-47 |
Cash and cash held in escrow
The
Company had $
Cash and investments Held in Trust Account
As of December 31, 2022, substantially all of the assets held in the Trust Account were held in money market funds. Assets held in money market funds were invested primarily in U.S. Treasury securities. All of the Company’s investments held in the Trust Account were classified as trading securities. Trading securities are presented on the consolidated balance sheets at fair value at the end of each reporting period. Gains and losses resulting from the change in fair value of investments held in Trust Account are included in interest earned on investments held in Trust Account in the accompanying consolidated statements of operations.
To mitigate the risk of being deemed to have been operating as an unregistered investment company (including under the subjective test of Section 3(a)(1)(A) of the Investment Company Act), on July 27, 2023, the Company instructed the Trustee of the Trust Account, to liquidate the U.S. government securities or money market funds held in the Trust Account and thereafter to hold all funds in the Trust Account in cash (which may include demand deposit accounts) until the earlier of consummation of our Business Combination or liquidation. Furthermore, such cash is held in bank accounts, which exceed federally insured limits as guaranteed by the FDIC.
Concentration of Credit Risk
Financial instruments that potentially subject the Company to concentrations of credit risk consist of cash accounts in a financial institution, which, at times, may exceed the FDIC coverage limit. At December 31, 2023 and 2022, the Company has not experienced losses on these accounts.
Fair value of Financial Instruments
The fair value of the Company’s assets and liabilities which qualify as financial instruments under the FASB ASC 820, “Fair Value Measurements and Disclosures,” approximate the carrying amounts represented in the accompanying consolidated balance sheets, primarily due to their short-term nature.
Income Taxes
The Company complies with the accounting and reporting requirements of ASC 740, “Income Taxes,” which requires an asset and liability approach to financial accounting and reporting for income taxes. Deferred income tax assets and liabilities are computed for differences between the financial statement and tax bases of assets and liabilities that will result in future taxable or deductible amounts, based on enacted tax laws and rates applicable to the periods in which the differences are expected to affect taxable income. Valuation allowances are established, when necessary, to reduce deferred tax assets to the amount expected to be realized. As of December 31, 2023 and 2022, the Company’s deferred tax asset for start up organizational expenses had a full valuation allowance recorded against it.
| F-48 |
FASB ASC 740 prescribes a recognition threshold and a measurement attribute
for the financial statement recognition and measurement of tax positions taken or expected to be taken in a tax return. For those benefits
to be recognized, a tax position must be more likely than not to be sustained upon examination by taxing authorities. There were no unrecognized
tax benefits as of December 31, 2023 or December 31, 2022. No amounts were accrued for the payment of interest and penalties for the year
ended December 31, 2023. Interest and penalties related to the December 31, 2022 tax return of $
Excise Tax
In
connection with the vote to approve the charter amendment proposal presented at the March 2023 Special Meeting, holders of shares of common stock properly exercised their
right to redeem their shares of common stock for an aggregate redemption amount of $
The Company accounts for its common stock subject to possible redemption in accordance with the guidance in ASC Topic 480 “Distinguishing Liabilities from Equity.” Shares of common stock subject to mandatory redemption (if any) is classified as a liability instrument and is measured at fair value. Conditionally redeemable common stock (including common stock that features redemption rights that are either within the control of the holder or subject to redemption upon the occurrence of uncertain events not solely within the Company’s control) is classified as temporary equity. At all other times, common stock is classified as stockholders’ equity. The Company’s shares of common stock sold in the IPO and as a result of the exercise by the underwriters of their over-allotment option features certain redemption rights that are considered to be outside of the Company’s control and subject to occurrence of uncertain future events. Accordingly, on December 31, 2023 and 2022, and shares of common stock subject to possible redemption were presented as temporary equity, outside of the stockholders’ deficit section of the Company’s consolidated balance sheets.
On
March 6, 2023, in connection with the approval of the extension amendment proposal and the trust amendment proposal presented at the
March 2023 Special Meeting, holders of of the Company’s shares of common stock exercised their right to redeem those
shares for cash at an approximate price of $ per share, for an aggregate of approximately $
On November 28, 2023, in connection with the approval of the Extension Amendment Proposal and the Trust Amendment Proposal at the November 2023 Special Meeting, holders of shares of the Company’s common stock exercised their right to redeem those shares for cash at an approximate price of $ per share, for an aggregate of approximately $ million.
| F-49 |
Schedule of Subject to Possible Redemption | ||||||||
| Shares | Amount | |||||||
| Gross proceeds from the IPO | $ | |||||||
| Less: | ||||||||
| Proceeds allocated to Public Warrants | ) | |||||||
| Common stock issuance costs | ) | |||||||
| Plus: | ||||||||
| Remeasurement of carrying amount to redemption value | ||||||||
| Common stock subject to possible redemption, December 31, 2022 | ||||||||
| Less: | ||||||||
| Redemptions (paid in April and November 2023) | ) | ) | ||||||
| Plus: | ||||||||
| Remeasurement of carrying value to redemption value | ||||||||
| Common stock subject to possible redemption, December 31, 2023 | $ | |||||||
The Company complies with the accounting and disclosure requirements of FASB ASC Topic 260, “Earnings Per Share” and uses the two class method. Net income per share of common stock is computed by dividing net income by the weighted average number of shares of common stock outstanding for the period. Any remeasurement of the accretion to redemption value of the shares of common stock subject to possible redemption was considered to be dividends paid to the public stockholders.
The Company has one authorized class of common stock. Warrants included in the Units sold in the IPO (the “Public Warrants”) (see Note 3) and warrants included in the Private Placement Units (the “Private Placement Warrants,” together with the Public Warrants, the “warrants”) (see Note 4) to purchase shares of common stock of the Company at $ per share were issued on December 9, 2021. For the years ended December 31, 2023 and 2022, no Public Warrants or Private Placement Warrants had been exercised. The potential shares of common stock underlying the outstanding Public Warrants and Private Placement Warrants to purchase the Company’s shares of common stock were excluded from diluted earnings per share for the years ended December 31, 2023 and 2022 because they are contingently exercisable, and the contingencies have not yet been met. Additionally, the rights are able to be demanded on or any time after the Business Combination, and as the contingency has not been met, the rights are excluded from diluted earnings per share for the years ended December 31, 2023 and 2022. As a result, diluted net income per share of common stock is the same as basic net income per share of common stock for the periods. The table below presents a reconciliation of the numerator and denominator used to compute basic and diluted net income per share for each class of stock.
| For the Year Ended December 31, | ||||||||
| 2023 | 2022 | |||||||
| Net income | $ | $ | ||||||
| Remeasurement of common stock subject to redemption | ( | ) | ( | ) | ||||
| Net loss including remeasurement of common stock subject to redemption value | $ | ( | ) | $ | ( | ) | ||
| F-50 |
| For the Year Ended December 31, | ||||||||||||||||
| 2023 | 2022 | |||||||||||||||
| Redeemable | Non- redeemable | Redeemable | Non- redeemable | |||||||||||||
| Basic and diluted net income (loss) per share of common stock | ||||||||||||||||
| Numerator: | ||||||||||||||||
| Allocation of net loss including remeasurement of common stock subject to redemption value | $ | ( | ) | $ | ( | ) | $ | ( | ) | $ | ( | ) | ||||
| Remeasurement of common stock subject to redemption | ||||||||||||||||
| Allocation of net income (loss), as adjusted | $ | $ | ( | ) | $ | $ | ( | ) | ||||||||
| Denominator: | ||||||||||||||||
| Basic and diluted weighted average shares outstanding | ||||||||||||||||
| Basic and diluted net income (loss) per share of common stock | $ | $ | ) | $ | $ | ) | ||||||||||
Accounting for Warrants
The Company accounts for warrants as either equity-classified or liability-classified instruments based on an assessment of the warrant’s specific terms and applicable authoritative guidance in FASB ASC 480, Distinguishing Liabilities from Equity (“ASC 480”) and ASC 815, Derivatives and Hedging (“ASC 815”). The assessment considers whether the warrants are freestanding financial instruments pursuant to ASC 480, meet the definition of a liability pursuant to ASC 480, and whether the warrants meet all of the requirements for equity classification under ASC 815, including whether the warrants are indexed to the Company’s own common stock, among other conditions for equity classification. This assessment, which requires the use of professional judgment, is conducted at the time of warrant issuance and as of each subsequent quarterly period end date while the warrants are outstanding. The Company accounts for the warrants issued in connection with the Company’s IPO in accordance with the guidance contained in ASC 815 under which the public warrants meet the criteria for equity treatment and the private warrants do not meet the criteria for equity treatment and must be recorded as liabilities. Accordingly, the Company classifies the private warrants as liabilities at their fair value and adjust the private warrants to fair value at each reporting period. This liability is subject to re-measurement at each balance sheet date until exercised, and any change in fair value is recognized in the Company’s consolidated statements of operations. The fair value of the warrants was estimated using a binomial lattice model.
Recent Accounting Standards
In December 2023, the FASB issued ASU 2023-09, Income Taxes (Topic 740): Improvements to Income Tax Disclosures (ASU 2023-09), which requires disclosure of incremental income tax information within the rate reconciliation and expanded disclosures of income taxes paid, among other disclosure requirements. ASU 2023-09 is effective for fiscal years beginning after December 15, 2024. Early adoption is permitted. The Company’s management does not believe the adoption of ASU 2023-09 will have a material impact on its consolidated financial statements and disclosures.
Note 3 — Initial Public Offering and Over-allotment
Pursuant
to the IPO and the over-allotment in December 2021, the Company sold Units at a price of $ per Unit. Each Unit consists
of one share of common stock, one redeemable warrant (each, a “Public Warrant” and collectively, the “Public Warrants”)
and one right (each a “Public Right” and collectively, the “Public Rights”).
| F-51 |
Note 4 — Private Placement
On
December 9, 2021 and December 13, 2021, simultaneously with the consummation of the IPO and the underwriters’ exercise of their
over-allotment option, the Company consummated the issuance and sale (“Private Placement”) of Private Placement Units
in a private placement transaction at a price of $ per Private Placement Unit, generating gross proceeds of $
Note 5 — Related Party Transactions
Founder Shares
On
August 19, 2021, the Company’s sponsor purchased shares (the “Founder Shares”) of the Company’s common
stock, par value $, for an aggregate price of $
Related Party Loans
In
order to finance transaction costs in connection with a Business Combination, the Company’s sponsor or an affiliate of the
sponsor, or certain of the Company’s officers and directors may, but are not obligated to, loan the Company funds as may be
required (“Working Capital Loans”). If the Company completes a Business Combination, the Company will repay the Working
Capital Loans out of the proceeds of the Trust Account released to the Company. Otherwise, the Working Capital Loans would be repaid
only out of funds held outside the Trust Account. In the event that a Business Combination does not close, the Company may use a
portion of proceeds held outside the Trust Account to repay the Working Capital Loans, but no proceeds held in the Trust Account
would be used to repay the Working Capital Loans. Except for the foregoing, the terms of such Working Capital Loans, if any, have
not been determined and no written agreements exist with respect to such loans. The Working Capital Loans would either be repaid
upon consummation of a Business Combination, without interest, or, at the lender’s discretion, up to $
The
Company entered into promissory notes with Public Gold Marketing Sdn. Bhd., which is considered a related party due to a familial relationship between the Sponsor and a 95% shareholder of Public
Gold Marketing Sdn. Bhd. The promissory notes bear an interest of
Support Services
The
Company has entered into an administrative services agreement pursuant to which the Company will pay its sponsor a total of $
On
September 30, 2023, the Company terminated the administrative services agreement. As a result, the Company will no longer be required
to pay the sponsor $
| F-52 |
Advances
On
each of September 5, 2023, September 29, 2023 and November 7, 2023, an affiliate of the Company’s sponsor advanced $
Note 6 — Commitments and Contingencies
Registration Rights
The holders of Founder Shares, Private Placement Units and warrants that may be issued upon conversion of Working Capital Loans, if any, will be entitled to registration rights (in the case of the Founder Shares, only after conversion of such shares into shares of common stock) pursuant to a registration rights agreement signed on the date of the prospectus for the IPO. These holders are entitled to certain demand and “piggyback” registration rights. However, the registration rights agreement provides that the Company will not permit any registration statement filed under the Securities Act to become effective until the termination of the applicable lock-up period for the securities to be registered. The Company will bear the expenses incurred in connection with the filing of any such registration statements.
Underwriting Agreement
The
underwriters were paid a cash underwriting discount of $ per unit on the offering including the Units issued with the underwriter’s
exercise of their over-allotment option, or $
Note 7 — Promissory Notes – Related Party
On
March 3, 2023, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount
of $
On
March 23, 2023, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount
of up to $
On
June 2, 2023, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount
of up to $
On
October 13, 2023, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount
of $
On
December 8, 2023, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd. for an amount
of $
For
the year ended December 31, 2023, the notes have incurred $
| F-53 |
Note 8 — Stockholders’ Deficit
Common stock
The Company is authorized to issue shares of common stock with a par value of $ per share. As of December 31, 2023 and 2022, there were (excluding and shares of common stock subject to possible redemption, respectively) shares of common stock issued and outstanding.
Warrants:
As
of December 31, 2023 and 2022, the Company had
The Public Warrants are accounted for as equity instruments in the Company’s consolidated financial statements. Public Warrants may only be exercised for a whole number of shares. No fractional shares will be issued upon exercise of the Public Warrants. The Public Warrants will become exercisable on the later of the completion of an initial Business Combination and will expire five years after the completion of an initial Business Combination, or earlier upon redemption. No Public Warrants will be exercisable for cash unless the Company has an effective and current registration statement covering the shares of common stock issuable upon exercise of the Public Warrants and a current prospectus relating to such shares of common stock. Notwithstanding the foregoing, if a registration statement covering the shares of common stock issuable upon exercise of the Public Warrants is not effective within a specified period following the consummation of a Business Combination, warrant holders may, until such time as there is an effective registration statement and during any period when the Company shall have failed to maintain an effective registration statement, exercise warrants on a cashless basis pursuant to the exemption provided by Section 3(a)(9) of the Securities Act, provided that such exemption is available. If that exemption, or another exemption, is not available, holders will not be able to exercise their warrants on a cashless basis. The Public Warrants will expire five years after the completion of a Business Combination or earlier upon redemption or liquidation.
Redemption of warrants when the price per common stock equals or exceeds $
Once the warrants become exercisable, the Company may redeem the outstanding warrants (except as described herein with respect to the private placement warrants):
| ● | in whole and not in part; | |
| ● | at
a price of $ | |
| ● | upon a minimum of 30 days’ prior written notice of redemption, which the Company refers to as the “30-day redemption period”; and | |
| ● | if, and only if, the last reported sale price (the “closing price”) of our common stock equals or exceeds $ per share (as adjusted for adjustments to the number of shares issuable upon exercise or the exercise price of a warrant as described under the heading “Description of Securities—Warrants”) for any 20 trading days within a 30-trading day period ending on the third trading day prior to the date on which the Company sends the notice of redemption to the warrant holders. |
The Company will not redeem the warrants as described above unless an effective registration statement under the Securities Act covering the shares of common stock issuable upon exercise of the warrants is effective and a current prospectus relating to those shares of common stock is available throughout the 30-day redemption period. If and when the warrants become redeemable by the Company, the Company may exercise its redemption right even if the Company is unable to register or qualify the underlying securities for sale under all applicable state securities laws.
If the Company calls the Public Warrants for redemption, management will have the option to require all holders that wish to exercise the Public Warrants to do so on a “cashless basis,” as described in the warrant agreement.
| F-54 |
The Private Placement Warrants are substantially in the same form as the Public Warrants, except they (i) will be exercisable either for cash or on a cashless basis at the holder’s option pursuant and (ii) will not be redeemable by the Company, in either case as long as the Private Placement Warrants are held by the initial purchasers or any of their permitted transferees (as prescribed in the subscription agreement, dated December 6, 2021, by and between the Company and Public Gold Marketing Sdn. Bhd.). Once a Private Placement Warrant is transferred to a holder other than a permitted transferee, it shall be treated as a Public Warrant for all purposes. Due to these terms the Private Warrants are required to be liability classified.
The exercise price and number of shares of common stock issuable on exercise of the warrants may be adjusted in certain circumstances, including in the event of a share dividend, extraordinary dividend or recapitalization, reorganization, merger or consolidation. However, the warrants will not be adjusted for issuances of shares of common stock at a price below their respective exercise prices. Additionally, in no event will the Company be required to net cash settle the warrants. If the Company is unable to complete a Business Combination within the Combination Period and the Company liquidates the funds held in the Trust Account, holders of the warrants will not receive any of such funds with respect to their warrants, nor will they receive any distribution from the Company’s assets held outside of the Trust Account with the respect to such warrants. Accordingly, the warrants may expire worthless.
In
addition, if the Company issues additional shares of common stock or equity-linked securities for capital raising purposes in connection
with the closing of a Business Combination at an issue price or effective issue price of less than $
Rights
Except in cases where the Company is not the surviving company in a Business Combination, each holder of a Public Right will automatically receive one-tenth of one share of common stock upon consummation of a Business Combination, even if the holder of a Public Right converted all shares held by him, her or it in connection with a Business Combination or an amendment to the Company’s Amended and Restated Certificate of Incorporation with respect to its pre-business combination activities. In the event that the Company will not be the surviving company upon completion of a Business Combination, each holder of a Public Right will be required to affirmatively convert his, her or its rights in order to receive the one-tenth of a share underlying each Public Right upon consummation of the Business Combination.
The Company will not issue fractional shares in connection with an exchange of Public Rights. Fractional shares will either be rounded down to the nearest whole share or otherwise addressed in accordance with the applicable provisions of the Delaware General Corporation Law. As a result, the holders of the Public Rights must hold rights in multiples of 10 in order to receive shares for all of the holders’ rights upon closing of a Business Combination.
| F-55 |
Note 9. Income Tax
The Company’s net deferred tax liabilities are as follows:
| December 31, | December 31, | |||||||
| 2023 | 2022 | |||||||
| Deferred tax assets | ||||||||
| Startup Costs | $ | $ | ||||||
| Accrued interest on investments held in Trust Account | ( | ) | ||||||
| Total deferred tax assets | ||||||||
| Valuation allowance | ( | ) | ( | ) | ||||
| Deferred tax asset (liabilities), net of allowance | $ | $ | ( | ) | ||||
The income tax provision for the years ended December 31, 2023 and 2022 consists of the following:
| December 31, | December 31, | |||||||
| 2023 | 2022 | |||||||
| Federal | ||||||||
| Current | $ | $ | ||||||
| Deferred | ( | ) | ( | ) | ||||
| State | ||||||||
| Current | $ | $ | ||||||
| Deferred | ||||||||
| Change in valuation allowance | ||||||||
| Income tax provision | $ | $ | ||||||
In
assessing the realization of deferred tax assets, management considers whether it is more likely than not that some portion or all of
the deferred tax assets will not be realized. The ultimate realization of deferred tax assets is dependent upon the generation of future
taxable income during the periods in which temporary differences representing net future deductible amounts become deductible. Management
considers the scheduled reversal of deferred tax assets, projected future taxable income and tax planning strategies in making this assessment.
For the years ended December 31, 2023 and 2022, the valuation allowance was $
A reconciliation of the statutory federal income tax rate to the Company’s effective tax rate is as follows:
| December 31, | December 31, | |||||||
| 2023 | 2022 | |||||||
| Statutory federal income tax rate | % | % | ||||||
| Delaware franchise tax penalties | % | % | ||||||
| Change in fair value of warrants | ( | )% | ( | )% | ||||
| Business combination expenses | % | % | ||||||
| Broken Deal | ( | )% | % | |||||
| Change in valuation allowance | % | % | ||||||
| Income tax provision expense | % | % | ||||||
The
Company’s effective tax rate was
The Company files income tax returns in the U.S. federal jurisdiction in various state and local jurisdictions and is subject to examination by the various taxing authorities.
| F-56 |
Note 10 — Fair Value Measurements
The fair value of the Company’s financial assets and liabilities reflects management’s estimate of amounts that the Company would have received in connection with the sale of the assets or paid in connection with the transfer of the liabilities in an orderly transaction between market participants at the measurement date. In connection with measuring the fair value of its assets and liabilities, the Company seeks to maximize the use of observable inputs (market data obtained from independent sources) and to minimize the use of unobservable inputs (internal assumptions about how market participants would price assets and liabilities). The following fair value hierarchy is used to classify assets and liabilities based on the observable inputs and unobservable inputs used in order to value the assets and liabilities:
Level 1: Quoted prices in active markets for identical assets or liabilities. An active market for an asset or liability is a market in which transactions for the asset or liability occur with sufficient frequency and volume to provide pricing information on an ongoing basis.
Level 2: Observable inputs other than Level 1 inputs. Examples of Level 2 inputs include quoted prices in active markets for similar assets or liabilities and quoted prices for identical assets or liabilities in markets that are not active.
Level 3: Unobservable inputs based on the Company’s assessment of the assumptions that market participants would use in pricing the asset or liability.
As of December 31, 2022, the assets held in the Trust Account were held in money market funds invested in U.S. Treasury Securities. All of the Company’s investments held in the Trust Account were classified as trading securities.
The following table presents information about the Company’s liabilities that are measured at fair value on a recurring basis as of December 31, 2023 and December 31, 2022 and indicates the fair value hierarchy of the valuation inputs the Company utilized to determine such fair value.
| Quoted Prices in Active Markets | Significant Other Observable Inputs | Significant Other Unobservable Inputs | ||||||||||||||
| Level | (Level 1) | (Level 2) | (Level 3) | |||||||||||||
| December 31, 2023 | ||||||||||||||||
| Liabilities: | ||||||||||||||||
| Warrant Liabilities- Private Warrants | 3 | $ | ||||||||||||||
| December 31, 2022 | ||||||||||||||||
| Assets: | ||||||||||||||||
| Money market funds invested in U.S. Treasury Securities | 1 | $ | ||||||||||||||
| Liabilities: | ||||||||||||||||
| Warrant Liabilities- Private Warrants | 3 | |||||||||||||||
The Private Placement Warrants were valued using a binomial lattice model, which is considered to be a Level 3 fair value measurement. One of the more significant inputs is the implied volatility, which is based on the observed prices of the Company’s common stock and publicly-traded warrants. As of December 31, 2023 and 2022, the estimated fair value of Warrant Liabilities – Private Warrants were determined based on the following significant inputs and are expressed on the basis of each being exercisable for a one-half of one share of common stock:
As of December 31, 2023 | As of December 31, 2022 | |||||||
| Exercise price | $ | $ | ||||||
| Market price of public stock | $ | $ | ||||||
| Term (years) | ||||||||
| Volatility | immaterial | % | % | |||||
| Risk-free rate | % | % | ||||||
| Dividend yield | % | % | ||||||
| F-57 |
The following table presents the changes in the fair value of warrant liabilities for the three and nine months ended December 31, 2023 and 2022:
| Private Placement Warrants | ||||
| Fair value as of January 1, 2023 | $ | |||
| Change in valuation inputs or other assumptions | ( | ) | ||
| Fair value as of December 31, 2023 | $ | |||
| Private Placement Warrants | ||||
| Fair value as of January 1, 2022 | $ | |||
| Change in valuation inputs or other assumptions | ( | ) | ||
| Fair value as of December 31, 2022 | $ | |||
NOTE 11. SUBSEQUENT EVENTS
The Company evaluated subsequent events and transactions that occurred after the balance sheet date up to the date that the consolidated financial statements were issued. Based upon this review, the Company did not identify any subsequent events, other than below, that would have required adjustment or disclosure in the consolidated financial statements.
On
January 5, 2024, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd for an
amount of $
On
January 5, 2024, Globalink elected to extend the Termination
Date by another month until February 9, 2024, and deposited an aggregate $
On
January 25, 2024, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd for an amount
of $
On January 30, 2024, the Company entered into a Merger Agreement by and among Alps, GL Sponsor LLC and Dr. Tham Seng Kong, an individual. Pursuant to the terms of the Merger Agreement, a business combination between Globalink and Alps through the merger of a Merger Sub with and into Alps, with the Surviving Company as a wholly-owned subsidiary of Globalink (the “Merger”, and, together with the other transactions contemplated by the Merger Agreement, the “Transactions”). After the date of the Merger Agreement and prior to the consummation of the transactions contemplated by the Merger Agreement (the “Closing”), a company formed under the laws of the Cayman Islands will be incorporated (“Cayman Holdco”), whereupon it is envisaged that Alps will become a subsidiary of the Cayman Holdco.
On
February 6, 2024, Globalink elected to extend the Termination
Date by another month until March 9, 2024, and deposited an aggregate $
On
February 22, 2024, the Company entered into a promissory note subscription term sheet with Public Gold Marketing Sdn. Bhd for an
amount of $
On
March 6, 2024, Globalink elected to extend the Termination
Date by another month until April 9, 2024, and deposited an aggregate $
| F-58 |
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING
FIRM TO THE SHAREHOLDERS AND THE BOARD OF DIRECTORS
OF ALPS GLOBAL HOLDING BERHAD
[Registration No.: 201701012867 (1227032 - V)]
(Incorporated in Malaysia)
Opinion on the Consolidated Financial Statements
We have audited the accompanying consolidated statement of financial position of ALPS Global Holding Berhad and its subsidiaries (the “Group”) as of March 31, 2024 and 2023, and the consolidated statement of profit or loss and other comprehensive income, statement of changes in equity and statement of cash flows for each of the years in the two-year period ended March 31, 2024 and 2023, including the related notes (collectively referred to as the “consolidated financial statements”). In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position of the Group as of March 31, 2024 and 2023, and the results of its operations and its cash flows for each of the years in the two-year period ended March 31, 2024 and 2023, in conformity with International Accounting Standards and International Financial Reporting Standards as issued by the International Accounting Standards Board.
Restatement of 2024 Financial Statements
As discussed in Notes 1 and 32 to the consolidated financial statements, the accompanying 2024 financial statements have been restated to correct misstatements.
Basis for Opinion
These consolidated financial statements are the responsibility of the Group’s management. Our responsibility is to express an opinion on the Group’s consolidated financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Group in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits of these consolidated financial statements in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the consolidated financial statements are free of material misstatement, whether due to error or fraud.
The Group is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits, we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Group’s internal control over financial reporting. Accordingly, we express no such opinion.
Our audit included performing procedures to assess the risks of material misstatement of the consolidated financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the consolidated financial statements. Our audit also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements. We believe that our audit provides a reasonable basis for our opinion.
| UHY Malaysia | |
| Firm Number: AF 1411 | |
| Chartered Accountants | |
| KUALA LUMPUR, MALAYSIA | |
| June 18, 2024, except for the effects of the restatement discussed in Note 1 and Note 32 to the consolidated financial statements, as to which the date is August 12, 2024 and December 4, 2024 |
| F-59 |
ALPS GLOBAL HOLDING BERHAD
CONSOLIDATED STATEMENT OF FINANCIAL POSITION AS AT 31 MARCH 2024
| Note | 2024 | 2023 | ||||||||
| USD | USD | |||||||||
| ASSETS | ||||||||||
| Non-Current Assets | ||||||||||
| Property, plant and equipment | 5 | |||||||||
| Right-of-use assets | 6 | |||||||||
| Intangible assets | 7 | |||||||||
| Other investments | 8 | |||||||||
| Investment in associates | 9 | |||||||||
| Total Non-Current Assets | ||||||||||
| Current Assets | ||||||||||
| Inventories | 10 | |||||||||
| Trade receivables | 11 | |||||||||
| Other receivables, deposits and prepayments | 12 | |||||||||
| Amount due from associates | 13 | |||||||||
| Tax recoverable | ||||||||||
| Cash and bank balances | ||||||||||
| Total Current Assets | ||||||||||
| Total Assets | ||||||||||
| EQUITY AND LIABILITIES | ||||||||||
| Capital and Reserves | ||||||||||
| Share capital | 14 | |||||||||
| Share application money | 15 | |||||||||
| Merger reserves | 16 | ( | ) | ( | ) | |||||
| Accumulated losses | ( | ) | ( | ) | ||||||
| Foreign translation reserve | 17 | ( | ) | ( | ) | |||||
| Equity Attributable to the Owners of the Company | ||||||||||
| Non-controlling interests | ( | ) | ( | ) | ||||||
| Total Equity | ||||||||||
| Non-Current Liabilities | ||||||||||
| Hire purchase liabilities | 18 | |||||||||
| Lease liabilities | 19 | |||||||||
| Deferred tax liabilities | 20 | |||||||||
| Total Non-Current Liabilities | ||||||||||
| F-60 |
ALPS GLOBAL HOLDING BERHAD
CONSOLIDATED STATEMENT OF FINANCIAL POSITION AS AT 31 MARCH 2024 (CONT’ D)
| Note | 2024 | 2023 | ||||||||
| USD | USD | |||||||||
| Current Liabilities | ||||||||||
| Trade payables | 21 | |||||||||
| Other payables and accruals | 22 | |||||||||
| Amount due to directors | 13 | |||||||||
| Hire purchase liabilities | 18 | |||||||||
| Lease liabilities | 19 | |||||||||
| Tax liabilities | ||||||||||
| Total Current Liabilities | ||||||||||
| Total Liabilities | ||||||||||
| Total Equity and Liabilities | ||||||||||
The accompanying notes are an integral part of the consolidated financial statements
| F-61 |
ALPS GLOBAL HOLDING BERHAD
CONSOLIDATED STATEMENT OF PROFIT OR LOSS AND OTHER COMPREHENSIVE INCOME
FOR THE FINANCIAL YEAR ENDED 31 MARCH 2024
| Note | 2024 | 2023 | ||||||||
| USD | USD | |||||||||
| Revenue | 23 | |||||||||
| Cost of sales | ( | ) | ( | ) | ||||||
| Gross profit | ||||||||||
| Other operating income | ||||||||||
| Distribution expenses | ( | ) | ( | ) | ||||||
| Administrative expenses | ( | ) | ( | ) | ||||||
| Other operating expenses | ( | ) | ( | ) | ||||||
| Share result of associates | ||||||||||
| Loss from operations | ( | ) | ( | ) | ||||||
| Finance costs | 24 | ( | ) | ( | ) | |||||
| Loss before tax | 25 | ( | ) | ( | ) | |||||
| Income tax credit/(expense) | 26 | ( | ) | |||||||
| Loss for the financial year | ( | ) | ( | ) | ||||||
| Other comprehensive loss, net of income tax | ||||||||||
| Foreign currency translation, representing other comprehensive loss for the financial year | ( | ) | ( | ) | ||||||
| Total comprehensive loss for the financial year | ( | ) | ( | ) | ||||||
| Loss for the financial year attributable to: | ||||||||||
| Owners of the Company | ( | ) | ( | ) | ||||||
| Non-controlling interests | ( | ) | ( | ) | ||||||
| ( | ) | ( | ) | |||||||
| Total comprehensive loss for the financial year attributable to: | ||||||||||
| Owners of the Company | ( | ) | ( | ) | ||||||
| Non-controlling interests | ( | ) | ( | ) | ||||||
| ( | ) | ( | ) | |||||||
| Loss per ordinary share attributable to owners of the Company: | ||||||||||
| Basic/Diluted | 27 | ) | ) | |||||||
The accompanying notes are an integral part of the consolidated financial statements
| F-62 |
ALPS GLOBAL HOLDING BERHAD
CONSOLIDATED STATEMENTS OF CHANGES IN EQUITY
FOR THE FINANCIAL YEAR ENDED 31 MARCH 2024
| Note | Share capital |
Share application money |
Non-distributable reserve - Merger reserves | Non-distributable reserve - Foreign translation reserve | Distributable reserve - Accumulated losses | Equity attributable to owners of the Company | Non-controlling interests | Total equity | |||||||||||||||||||||||||||
| USD | USD | USD | USD | USD | USD | USD | USD | ||||||||||||||||||||||||||||
| As at 1 April 2022 | ( | ) | ( | ) | ( | ) | |||||||||||||||||||||||||||||
| Contributions by and distribution to owners of the Company | |||||||||||||||||||||||||||||||||||
| Issuance of shares | 14 | ( | ) | ||||||||||||||||||||||||||||||||
| Loss for the financial year | ( | ) | ( | ) | ( | ) | ( | ) | |||||||||||||||||||||||||||
| Other comprehensive loss | ( | ) | ( | ) | ( | ) | |||||||||||||||||||||||||||||
| Total comprehensive loss for the financial year | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | |||||||||||||||||||||||||
| As at 31 March 2023 | ( | ) | ( | ) | ( | ) | ( | ) | |||||||||||||||||||||||||||
| F-63 |
ALPS GLOBAL HOLDING BERHAD
CONSOLIDATED STATEMENTS OF CHANGES IN EQUITY
FOR THE FINANCIAL YEAR ENDED 31 MARCH 2024 (CONT’ D)
| Note | Share capital | Share application money | Non-distributable reserve - Merger reserves | Non-distributable reserve - Foreign translation reserve | Distributable reserve - Accumulated losses | Equity attributable to owners of the Company | Non-controlling interests | Total equity | |||||||||||||||||||||||||||
| USD | USD | USD | USD | USD | USD | USD | USD | ||||||||||||||||||||||||||||
| As at 1 April 2023 | ( | ) | ( | ) | ( | ) | ( | ) | |||||||||||||||||||||||||||
| Contributions by and distribution to owners of the Company | |||||||||||||||||||||||||||||||||||
| Issuance of shares | 14 | ( | ) | ||||||||||||||||||||||||||||||||
| Arising from part dilution of equity interest in a subsidiary | ( | ) | |||||||||||||||||||||||||||||||||
| Loss for the financial year | ( | ) | ( | ) | ( | ) | ( | ) | |||||||||||||||||||||||||||
| Other comprehensive loss | ( | ) | ( | ) | ( | ) | |||||||||||||||||||||||||||||
| Total comprehensive loss for the financial year | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | |||||||||||||||||||||||||
| As at 31 March 2024 | ( | ) | ( | ) | ( | ) | ( | ) | |||||||||||||||||||||||||||
The accompanying notes are an integral part of the consolidated financial statements
| F-64 |
ALPS GLOBAL HOLDING BERHAD
CONSOLIDATED STATEMENT OF CASH FLOWS
FOR THE FINANCIAL YEAR ENDED 31 MARCH 2024
| 2024 | 2023 | |||||||
| USD | USD | |||||||
| CASH FLOWS USED IN OPERATING ACTIVITIES | ||||||||
| Loss before tax | ( | ) | ( | ) | ||||
| Adjustments for: | ||||||||
| Amortisation of intangible assets | ||||||||
| Amortisation of right-of-use assets | ||||||||
| Depreciation of property, plant and equipment | ||||||||
| Gain on disposal of other investment | ( | ) | ||||||
| Interest expenses | ||||||||
| Impairment losses on investment in associates | ||||||||
| Impairment losses on investment in unquoted shares | ||||||||
| Property, plant and equipment written off | ||||||||
| Share of results of associates | ( | ) | ( | ) | ||||
| Operating (loss)/profit before changes in working capital | ( | ) | ||||||
| Changes in working capital: | ||||||||
| Decrease/(Increase) in: | ||||||||
| Inventories | ||||||||
| Trade receivables | ( | ) | ||||||
| Other receivables, deposits and prepayments | ||||||||
| Increase/(Decrease) in: | ||||||||
| Trade payables | ( | ) | ||||||
| Other payables and accruals | ( | ) | ||||||
| Cash (Used In)/From Operations | ( | ) | ||||||
| Tax paid | ( | ) | ( | ) | ||||
| Net Cash Used In Operating Activities | ( | ) | ( | ) | ||||
| CASH FLOWS USED IN INVESTING ACTIVITIES | ||||||||
| Acquisition of associates | ( | ) | ( | ) | ||||
| Repayment from/(Advance to) associates | ( | ) | ||||||
| Purchase of property, plant and equipment | ( | ) | ( | ) | ||||
| Net Cash Used In Investing Activities | ( | ) | ( | ) | ||||
| CASH FLOWS FROM FINANCING ACTIVITIES | ||||||||
| Proceed from issuance of shares | ||||||||
| Advances from directors | ||||||||
| Interest paid | ( | ) | ( | ) | ||||
| Repayment of hire purchase liabilities | ( | ) | ( | ) | ||||
| Repayment of lease liabilities | ( | ) | ( | ) | ||||
| Net Cash From Financing Activities | ||||||||
| F-65 |
ALPS GLOBAL HOLDING BERHAD
CONSOLIDATED STATEMENT OF CASH FLOWS
FOR THE FINANCIAL YEAR ENDED 31 MARCH 2024 (CONT’D)
| 2024 | 2023 | |||||||
| USD | USD | |||||||
| NET DECREASE IN CASH AND CASH EQUIVALENTS | ( | ) | ||||||
| Effect of exchange differences | ( | ) | ( | ) | ||||
| CASH AND CASH EQUIVALENTS AT BEGINNING OF THE FINANCIAL YEAR | ||||||||
| CASH AND CASH EQUIVALENTS AT END OF THE FINANCIAL YEAR | ||||||||
The accompanying notes are an integral part of the consolidated financial statements
| F-66 |
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
FOR THE FINANCIAL YEAR ENDED 31 MARCH 2024
| 1. | Corporate Information |
Business Description
ALPS Global Holding Berhad (the “Company”) is a private limited liability company incorporated and domiciled in Malaysia. The registered office of the Company is located at Unit 20-5, Level 20, Q Sentral, 2A Jalan Stesen Sentral 2, Kuala Lumpur Sentral, 50470 Kuala Lumpur, Wilayah Persekutuan Kuala Lumpur, Malaysia. The principal place of business is at Unit E-18-01 & E-18-02, Level 18, Icon Tower (East), No 1 Jalan 1/68F, Jalan Tun Razak, 50400 Kuala Lumpur, Wilayah Persekutuan Kuala Lumpur, Malaysia.
The principal activities of the Company and its subsidiaries (together referred to as the Group) are involving in the business of research and development (including stem cell) in all kinds of biotechnology related business, invest and manage business such as health and beauty centre, medical centre as well as acting as an investment holding company.
These consolidated financial statements comprise the results of the Group.
Restatement of Current Year Consolidated Financial Statements
The Group identified certain misstatements in the consolidated statement of financial position and the consolidated statement of cash flows. These adjustments were made to reflect the classification of the investment in unquoted shares and the recognition of the patent license agreement in intangible assets. The effects of these restatements on the financial statements are disclosed in Note 32.
| 2. | Basis of Presentation |
Statement of Compliance
The financial statements of the Group have been prepared in accordance with in accordance with International Accounting Standards (“IAS”) and International Financial Reporting Standard (“IFRS”) as issued by the International Accounting Standards Board (“IASB”).
Adoption of amendments to IAS and IFRS
During the financial year, the Group adopted the following applicable amendments to IFRS issued by IASB:
| IFRS 17 | Insurance Contracts | |
| Amendments to IFRS 17 | Insurance Contracts | |
| Amendments to IFRS 17 | Initial Application of MFRS 17 and MFRS 9 - Comparative information | |
| Amendments to IAS 1 and IFRS Practice Statement 2 | Disclosure of Accounting Policies | |
| Amendments to IAS 8 | Definition of Accounting Estimates | |
| Amendments to IFRS 1 and IAS 12 | Deferred Tax related to Assets and Liabilities arising from a Single Transaction |
| F-67 |
| 2. | Basis of Presentation (Cont’d) |
Adoption of amendments to IAS and IFRS (Cont’d)
The adoption of these amendments to standards did not have any significant impact on the financial statements of the Group except for:
Amendments to IAS 1 and IFRS Practice Statement 2 Disclosure of Accounting Policies
The Group has adopted the amendments to IAS 1 Presentation of Financial Statements and IFRS Practice Statement 2 Materiality Practice Statement for the first time in the current financial year. The amendments change the requirements in IAS 1 Presentation of Financial Statements with regard to disclosure of accounting policies. The amendments replace all instances of the term “significant accounting policies” with “material accounting policy information”. Accounting policy information is material if, when considered together with other information included in an entity’s financial statements, it can reasonably be expected to influence decisions that the primary users of general purpose financial statements make on the basis of those financial statements.
Amendments to IAS 1 Presentation of Financial Statements are also amended to clarify that accounting policy information that relates to immaterial transactions, other events or conditions is immaterial and need not be disclosed. Accounting policy information may be material because of the nature of the related transactions, other events or conditions, even if the amounts are immaterial. However, not all accounting policy information relating to material transactions, other events or conditions is itself material.
The amendments have no effect on the measurement, recognition or presentation of any items in the Group’s financial statements but affect the disclosure of accounting policies.
New IFRS and amendments to IAS and IFRSs in issue but not yet effective
The Group has not applied the following new IFRS and amendments to IAS and IFRS that have been issued by the IASB but are not yet effective for the Group.
| Effective
dates for financial periods beginning on or after | ||||
| Amendments to IFRS 16 | Lease Liability in a Sale and Leaseback | 1 January 2024 | ||
| Amendments to IAS 1 | Classification of Liabilities as Current or Non-Current | 1 January 2024 | ||
| Amendments to IAS 1 and Practice Statement 2 | Non-current Liabilities with Covenants | 1 January 2024 | ||
| Amendments to IFRS 107 and IFRS 7 | Supplier Finance Agreements | 1 January 2024 | ||
| Amendments to IFRS 121 | Lack of Exchangeability | 1 January 2025 | ||
| Amendments to IFRS 9 and IFRS 7 | Amendments to the Classification and Measurement of Financial Instruments | 1 January 2026 | ||
| IFRS 18 | Presentation and Disclosure in Financial Statements | 1 January 2027 | ||
| IFRS 19 | Subsidiaries without Public Accountability: Disclosures | 1 January 2027 | ||
| Amendments to IFRS 10 and IAS 28 | Sales or Contribution of between an Investor and its Associate or Joint Venture | Deferred until further notice |
| F-68 |
| 2. | Basis of Presentation (Cont’d) |
New IFRS and amendments to IAS and IFRSs in issue but not yet effective (Cont’d)
The directors anticipate that the abovementioned new IFRS and amendments to IFRSs will be adopted in the annual financial statements of the Group when they become effective, if applicable, and that the adoption of the new IFRS and amendments to IFRS will have no material impact on the financial statements of the Group in the period of initial application.
| 3. | Material Accounting Policy Information |
Basis of accounting
The financial statements of the Group have been prepared under the historical cost unless otherwise indicated in the accounting policy information below.
Functional and presentation currency
The functional currency of the Group is Ringgit Malaysia (“RM”). The consolidated financial statements are presented in United States Dollar (“USD”), which is the presentation currency of the Group. All financial information is presented in USD.
Basis of consolidation
The Group adopts the merger method of accounting for all its subsidiaries.
A business combination involving entities under common control is a business combination in which all the combining subsidiaries are ultimately controlled by the same party and parties both before and after the business combination, and that control is not transitory. Under the merger method of accounting, the results of subsidiaries are presented as if the business combination had been affected throughout the current and previous financial years. The assets and liabilities combine are accounted for based on the carrying amounts from the perspective of the common control shareholder at the date of transfer. On consolidation, the difference between the carrying value of the investment in subsidiaries and the share capital of the Group’s subsidiaries is taken to merger reserve.
Non-controlling interests
At the acquisition date, components of non-controlling interests of the Group are measured at the non-controlling interest’s proportionate share of the acquiree’s identifiable assets.
Investment in subsidiaries
In the Group’s separate financial statements, investment in subsidiaries is stated at cost less accumulated impairment losses.
Investment in associates
Investment in associates is accounted for in the consolidated financial statements of the Group using the equity method.
Revenue
Revenue is recognised at a point in time when they transfer control over a product or service to a customer and satisfy their performance obligation to a customer. Where applicable, rebates and discounts to customers are accounted as net of revenue according to contract. The revenue is recognised to the extent that it is highly probable that a significant reversal in the amount of cumulative revenue recognised will not occur when the uncertainty associated with the variable consideration is subsequently resolved.
| F-69 |
| 3. | Material Accounting Policy Information (Cont’d) |
Revenue (Cont’d)
The Group recognises revenue from the following major sources:
| (a) | Cellular therapy |
Cellular therapy uses living cells to treat diseases by repairing damaged tissues, replacing lost cells, or boosting the body’s ability to fight illness.
Revenue is recognised at a point in time when the services have been rendered to the customers and coincide with the delivery of services and acceptance by customers.
| (b) | Medical testing, laboratory and aesthetics beauty services |
Medical testing, laboratory, and aesthetics beauty services cover a variety of procedures for health assessment and cosmetic enhancement.
Included in aesthetics beauty services are hair implant services, whereby, the revenue recognised are based on profit sharing basis as agreed with another counterparty.
Principal versus agent
When another party is involved in providing services to a customer, the Group determines whether the nature of its promise is a performance obligation to provide the specified services itself (i.e. the Group is a principal) or to arrange for those services to be provided by the other party (i.e. the Group is an agent).
The Group is classified as a principal when it controls the specified service before it is transferred to the customer. This control implies that the Group is responsible for fulfilling the service and bears the associated risks and rewards. Conversely, the Group is an agent if its obligation is to arrange for the service to be provided by another party without controlling it before transfer. In this case, the Group recognises revenue as a fee or commission for facilitating the service.
For hair implant services, the Group exercises control over the delivery of the service before it reaches the customer. Consequently, the Group is deemed a principal for these services and recognises revenue based on the total amount charged to the customer.
Revenue is recognised at a point in time when the services have been rendered to the customers and coincide with the delivery of services and acceptance by customers.
| (c) | Consultation fee |
Consultation fee is the charge imposed by a doctor for providing medical advice, diagnosis, and treatment recommendations during a visit. This fee covers the time and expertise of the healthcare professional and may vary based on the complexity of the case and the doctor’s experience.
Revenue is recognised at a point in time when the services have been rendered to the customers and coincide with the delivery of services and acceptance by customers.
| F-70 |
| 3. | Material Accounting Policy Information (Cont’d) |
Revenue (Cont’d)
| (d) | Sale of medicine and healthcare product |
The sale of medicine and healthcare products involves distributing pharmaceuticals and wellness items to improve health, with strict regulations to ensure safety and effectiveness.
Revenue is recognised when the transfer of significant risk and rewards of ownership of the goods to the customer, recovery of the consideration is probable, the associated costs and possible return of goods can be estimated reliably.
No element of financing is deemed present as the sales are made with credit term ranges from 30 days to 60 days, which is consistent with the market practice. Furthermore, the Group does not have any obligations related to returns, refunds or warranties.
Current and deferred income tax
Tax expenses for the year comprises current and deferred tax. Tax is recognised in profit or loss, except to the extent that it relates to items recognised in other comprehensive income or directly in equity. In this case, the tax is also recognised in other comprehensive income or directly in equity, respectively.
Current and deferred tax is measured using the tax rates that have been enacted or substantially enacted at the statement of financial position date in the countries where the Group’s subsidiaries and associates operate generate taxable income.
Deferred tax is provided for on temporary differences arising between the tax bases of assets and liabilities and their carrying amounts in the financial statements. Deferred tax assets are recognised to the extent that it is probable that future taxable profit will be available against which the deductible temporary differences and unused tax losses can be utilised.
Deferred tax is not accounted for if it arises from initial recognition of an asset or liability in a transaction other than a business combination that at the time of the transaction affects neither accounting nor taxable profit or loss.
Deferred income tax is provided on temporary differences arising on investments in subsidiaries and associates, except for deferred income tax liability where the timing of the reversal of the temporary difference is controlled by the Group and it is probable that the temporary difference will not reverse in the foreseeable future.
Deferred and income tax assets and liabilities are offset when there is a legally enforceable right to offset current tax assets against current tax liabilities and when the deferred income tax assets and liabilities relate to taxes levied by the same taxation authority on either the taxable entity or different taxable entities where there is an intention to settle the balances on a net basis.
Leases
Lessee accounting
The Group recognises a right-of-use asset and a lease liability at the lease commencement date. Right-of-use assets are initially measured at cost less any accumulated amortisation and accumulated impairment losses, and adjusted for any remeasurement of the lease liabilities. Amortisation is computed on the straight-line basis over the lease period.
| F-71 |
| 3. | Material Accounting Policy Information (Cont’d) |
Leases (Cont’d)
Lessee accounting (Cont’d)
The lease liabilities are initially measured at the present value of the lease payments that are paid at commencement date, discounted using the interest rate implicit in the lease contract. If that rate cannot be readily determined, the Group uses its incremental borrowing rate.
The Group has elected not to recognise right-of-use assets and lease liabilities for short-term leases that have a lease term of 12 months or less and leases of low value assets. The Group recognises the lease payments associated with these leases as an operating expense on a straight-line basis over the lease term.
Property, plant and equipment
Property, plant and equipment are measured at cost less any accumulated depreciation and accumulated impairment losses.
Property, plant and equipment are depreciated on straight-line basis over the estimated useful lives of the assets, at the following annual rates:
| Furniture & fittings | ||||
| Office equipment | ||||
| Medical equipment | ||||
| Computer equipment | ||||
| Renovation | ||||
| Laboratory | ||||
| Air conditioner | ||||
| Mobile device | ||||
| Laboratory equipment | ||||
| Plant and machinery | ||||
| Computer and ICT | ||||
| Signboard | ||||
| Motor vehicles |
Intangible assets
Intangible assets are measured at cost less any accumulated amortisation and accumulated impairment losses.
The amortisation of the intangible assets of the Group included in “other operating expenses” in the consolidated statements of profit or loss and other comprehensive income of the Group and the amortisation rate used was based on the estimated useful lives of the intangible assets, at the following rates:
| Trademark | ||
| Patent | ||
| Technical know-how of stem cells cultivation |
Inventories
Inventories are stated at lower of cost or net realisable value. Cost of inventories comprises the original cost of purchase plus incidental costs incurred in bringing the inventories to their present location, and accounted for on a first-in, first-out basis.
| F-72 |
| 3. | Material Accounting Policy Information (Cont’d) |
Financial instruments
Financial assets and financial liabilities are recognised in the Group’s statements of financial position when the Group becomes a party to the contractual provisions of the instrument.
Financial assets and financial liabilities are initially measured at fair value. Transaction costs that are directly attributable to the acquisition or issue of financial assets and financial liabilities (other than financial assets and financial liabilities at fair value through profit or loss) are added to or deducted from the fair value of the financial assets or financial liabilities, as appropriate, on initial recognition. Transaction costs directly attributable to the acquisition of financial assets or financial liabilities at fair value through profit or loss are recognised immediately in profit or loss.
Financial assets
All regular way purchases or sales of financial assets are recognised and derecognised on a trade date basis. Regular way purchases or sales are purchases or sales of financial assets that require delivery of assets within the time frame established by regulation or convention in the marketplace.
All recognised financial assets are measured subsequently in their entirety at either amortised cost or fair value, depending on the classification of the financial assets.
Classification of financial assets
Debt instruments that meet the following conditions are measured subsequently at amortised cost:
| ● | the financial asset is held within a business model whose objective is to hold financial assets in order to collect contractual cash flows; and | |
| ● | the contractual terms of the financial asset give rise on specified dates to cash flows that are solely payments of principal and interest on the principal amount outstanding. |
Debt instruments that meet the following conditions are measured subsequently at fair value through other comprehensive income (“FVTOCI”):
| ● | the financial asset is held within a business model whose objective is achieved by both collecting contractual cash flows and selling the financial assets; and | |
| ● | the contractual terms of the financial asset give rise on specified dates to cash flows that are solely payments of principal and interest on the principal amount outstanding. |
By default, all other financial assets are measured subsequently at fair value through profit or loss (“FVTPL”).
Despite the foregoing, the Group may make the following irrevocable election/designation at initial recognition of a financial asset:
| ● | the Group may irrevocably elect to present subsequent changes in fair value of an equity investment in other comprehensive income if certain criteria are met; and | |
| ● | the Group may irrevocably designate a debt instrument that meets the amortised cost or FVTOCI criteria as measured at FVTPL if doing so eliminates or significantly reduces an accounting mismatch (see (ii) below). |
| F-73 |
| 3. | Material Accounting Policy Information (Cont’d) |
Financial assets (Cont’d)
| (i) | Amortised cost and effective interest method |
The effective interest method is a method of calculating the amortised cost of a debt instrument and of allocating interest income over the relevant period.
For financial assets other than purchased or originated credit-impaired financial assets (i.e. assets that are credit-impaired on initial recognition), the effective interest rate is the rate that exactly discounts estimated future cash receipts (including all fees and points paid or received that form an integral part of the effective interest rate, transaction costs and other premiums or discounts) excluding expected credit losses, through the expected life of the debt instrument, or, where appropriate, a shorter period, to the gross carrying amount of the debt instrument on initial recognition. For purchased or originated credit-impaired financial assets, a credit-adjusted effective interest rate is calculated by discounting the estimated future cash flows, including expected credit losses, to the amortised cost of the debt instrument on initial recognition.
The amortised cost of a financial asset is the amount at which the financial asset is measured at initial recognition minus the principal repayments, plus the cumulative amortisation using the effective interest method of any difference between that initial amount and the maturity amount, adjusted for any loss allowance. The gross carrying amount of a financial asset is the amortised cost of a financial asset before adjusting for any loss allowance.
Interest income is recognised using the effective interest method for debt instruments measured subsequently at amortised cost and at FVTOCI. For financial assets other than purchased or originated credit-impaired financial assets, interest income is calculated by applying the effective interest rate to the gross carrying amount of a financial asset, except for financial assets that have subsequently become credit-impaired (see below). For financial assets that have subsequently become credit-impaired, interest income is recognised by applying the effective interest rate to the amortised cost of the financial asset. If, in subsequent reporting periods, the credit risk on the credit-impaired financial instrument improves so that the financial asset is no longer credit-impaired, interest income is recognised by applying the effective interest rate to the gross carrying amount of the financial asset.
For purchased or originated credit-impaired financial assets, the Group recognises interest income by applying the credit-adjusted effective interest rate to the amortised cost of the financial asset from initial recognition. The calculation does not revert to the gross basis even if the credit risk of the financial asset subsequently improves so that the financial asset is no longer credit-impaired.
| (ii) | Financial assets at FVTPL |
Financial assets that do not meet the criteria for being measured at amortised cost or FVTOCI are measured at FVTPL. Specifically:
| ● | Investments in equity instruments are classified as at FVTPL, unless the Group designates an equity investment that is neither held for trading nor a contingent consideration arising from a business combination as at FVTOCI on initial recognition. | |
| ● | Financial assets that do not meet the amortised cost criteria or the FVTOCI criteria are classified as at FVTPL. In addition, debt instruments that meet either the amortised cost criteria or the FVTOCI criteria may be designated as at FVTPL upon initial recognition if such designation eliminates or significantly reduces a measurement or recognition inconsistency (so called “accounting mismatch”) that would arise from measuring assets or liabilities or recognising the gains and losses on them on different bases. The Group has designated investment in unquoted shares as at FVTPL. |
| F-74 |
| 3. | Material Accounting Policy Information (Cont’d) |
Financial assets (Cont’d)
| (ii) | Financial assets at FVTPL (Cont’d) |
Financial assets at FVTPL are measured at fair value at the end of each reporting period, with any fair value gains or losses recognised in profit or loss to the extent they are not part of a designated hedging relationship. The net gain or loss from other financial assets are recognised in profit or loss includes any dividend or interest earned on the financial asset and is included in the “other operating income” or “other operating expenses” line item.
Financial liabilities
All financial liabilities are measured subsequently at amortised cost using the effective interest method.
Financial liabilities measured subsequently at amortised cost
Financial liabilities that are not (i) contingent consideration of an acquirer in a business combination, (ii) held-for-trading, or (iii) designated as at FVTPL, are measured subsequently at amortised cost using the effective interest method.
The effective interest method is a method of calculating the amortised cost of a financial liability and of allocating interest expense over the relevant period. The effective interest rate is the rate that exactly discounts estimated future cash payments (including all fees and points paid or received that form an integral part of the effective interest rate, transaction costs and other premiums or discounts) through the expected life of the financial liability, or (where appropriate) a shorter period, to the amortised cost of a financial liability.
Impairment of non-financial assets
The carrying amounts of non-financial assets are reviewed at the end of each reporting period to determine whether there is any indication of impairment. If any such indication exists, the Group make an estimate of the asset’s recoverable amount. For assets that has indefinite useful life, the recoverable amount is estimated at each reporting date.
For the purpose of impairment testing, assets are grouped together into the smallest group of assets that generates cash inflows from continuing use that are largely independent of the cash inflows of non-financial assets or cash-generating units (“CGUs”).
The recoverable amount of an asset or a CGU is the higher of its fair value less costs of disposal and its value-in-use. In assessing value-in-use, the estimated future cash flows are discounted to their present value using a pre-tax discount rate that reflects current market assessments of the time value of money and the risks specific to the asset or CGU. In determining the fair value less costs of disposal, recent market transactions are taken into account. If no such transactions can be identified, an appropriate valuation model is used.
Where the carrying amount of an asset exceed its recoverable amount, the carrying amount of asset is reduced to its recoverable amount. Impairment losses recognised in respect of a CGU or groups of CGUs are allocated first to reduce the carrying amount of any goodwill allocated to those units or groups of units and then, to reduce the carrying amount of the other assets in the unit or groups of units on a pro-rata basis.
Impairment losses are recognised in profit or loss, except for assets that were previously revalued with the revaluation surplus recognised in other comprehensive income. In the latter case, the impairment is recognised in other comprehensive income up to the amount of any previous revaluation.
| F-75 |
| 3. | Material Accounting Policy Information (Cont’d) |
Impairment of non-financial assets (Cont’d)
An assessment is made at each reporting date as to whether there is any indication that previously recognised impairment losses may no longer exist or may have decreased. An impairment loss is reversed only if there has been a change in the estimates used to determine the assets recoverable amount since the last impairment loss was recognised. An impairment loss is reversed only to the extent that the asset’s carrying amount does not exceed the carrying amount that would have been determined, net of depreciation or amortisation, if no impairment loss had been recognised previously. Such reversal is recognised in profit or loss unless the asset is measured at revalued amount, in which case the reversal is treated as a revaluation increase.
Impairment of financial assets
The Group recognise a loss allowance for expected credit losses (“ECL”) on trade receivables, other receivables and deposits, amount owing due associates as well as cash and bank balances. The amount of ECL is updated at each reporting date to reflect changes in credit risk since initial recognition of the respective financial instrument.
The Group recognises lifetime ECL for trade receivables. The Group considers past loss experience, timing of billing and observable data such as current changes and future forecasts in economic conditions to estimate the amount of expected impairment loss. The methodology and assumptions including any forecasts of future economic conditions are reviewed regularly.
For all other financial instruments, the Group recognise lifetime ECL when there has been a significant increase in credit risk since initial recognition. However, if the credit risk on the financial instrument has not increased significantly since initial recognition, the Group measure the loss allowance for that financial instrument at an amount equal to 12-month ECL.
Lifetime ECL represents the expected credit losses that will result from all possible default events over the expected life of a financial instrument. In contrast, 12-month ECL represents the portion of lifetime ECL that is expected to result from default events on a financial instrument that are possible within 12 months after the reporting date.
Reversal of impairment loss to profit or loss, if any, is restricted to not exceeding what the amortised cost would have been had the impairment not been recognised previously.
Significant increase in credit risk
In assessing whether the credit risk on a financial asset has increased significantly since initial recognition, the Group compare the risk of a default occurring on the financial instrument at the reporting date with the risk of a default occurring on the financial instrument at the date of initial recognition. In making this assessment, the Group consider both quantitative and qualitative information that is reasonable and supportable, including historical experience and forward-looking information such as financial evaluation of the creditworthiness of the debtors, ageing of receivables, defaults and past due amounts, past experience with the debtors, current conditions and reasonable forecast of future economic conditions.
The Group presume that the credit risk on a financial asset has increased significantly since initial recognition when contractual payments are more than 365 days past due, unless the Group have reasonable and supportable information that demonstrates otherwise.
| F-76 |
| 3. | Material Accounting Policy Information (Cont’d) |
Impairment of financial assets (Cont’d)
Probability of default
The Group consider the information developed internally or obtained from external sources indicates that the debtor is unlikely to pay its creditors, including the Group, in full, as constituting an event of default for internal credit risk management purposes as historical experience indicates that financial assets are generally not recoverable.
The Group consider that default has occurred when a financial asset is more than 365 days past due unless the Group have reasonable and supportable information to demonstrate that a more lagging default criterion is more appropriate.
Write-off policy
The Group write off a financial asset when there is information indicating that the debtor is in severe financial difficulty and there is no realistic prospect of recovery, with case-by-case assessment performed based on indicators such as insolvency or demise. Financial assets written off may still be subject to enforcement activities under the Group’s recovery procedures, taking into account legal advice where appropriate. Any recoveries made are recognised in profit or loss as bad debts recovered.
Measurement and recognition of expected credit losses
The measurement of expected credit losses is a function of the probability of default, loss given default (i.e. the magnitude of the loss if there is a default) and the exposure at default. The assessment of the probability of default and loss given default is based on historical data adjusted by forward-looking information as described above. As for the exposure at default, for financial assets, this is represented by the assets’ gross carrying amount at the reporting date.
For financial assets, the expected credit loss is estimated as the difference between all contractual cash flows that are due to the Group in accordance with the contract and all the cash flows that the Group expect to receive, discounted at the original effective interest rate.
If the Group have measured the loss allowance for a financial instrument at an amount equal to lifetime ECL in the previous reporting period, but determine at the current reporting date that the conditions for lifetime ECL are no longer met, the Group measure the loss allowance at an amount equal to 12-month ECL at the current reporting date, except for assets for which the simplified approach was used.
The Group recognise an impairment gain or loss in profit or loss for all financial instruments with a corresponding adjustment to their carrying amount through a loss allowance account.
Segments reporting
Operating segments are reported in a manner consistent with the internal reporting provided to the chief operating decision-maker. The chief operating decision-maker is responsible for allocating resources and assessing performance of the operating segments and make overall strategic decisions. The Group’s operating segments are organised and managed separately according to the nature of the products and services provided, with each segment representing a strategic business unit that offers different products and serves different markets.
| F-77 |
| 3. | Material Accounting Policy Information (Cont’d) |
Statement of cash flows
The Group adopt the indirect method in the preparation of the statements of cash flows.
For the purpose of the statements of cash flows, cash and cash equivalents are cash and bank balances.
| 4. | Critical accounting judgements and key sources of estimation uncertainty |
Directors have used estimates and assumptions in measuring the reported amounts of assets and liabilities at the end of the reporting period and the reported amounts of expenses during the reporting period. Judgements and assumptions are applied in the measurement, and hence, the actual results may not coincide with the report amounts.
| (a) | Critical judgements in applying the Group’s accounting policies |
In the process of applying the Group’s accounting policies, the directors are of the opinion that there are no instances of application of judgements which are expected to have a significant effect on the amounts recognised in the financial statements.
| (b) | Key sources of estimation uncertainty |
Directors believe that there are no key assumptions made concerning the future, and other key sources of estimation uncertainty at the end of the reporting period, that have a significant risk of causing a material adjustment to the carrying amounts of assets and liabilities within the next reporting period, except for:
Impairment of financial assets
The impairment provisions for trade receivables are based on assumptions about risk of default and expected loss rate. The Group uses judgement in making these assumptions and selecting inputs to the impairment calculation, based on the Group’s past history, existing market conditions as well as forward looking estimates at the end of each reporting period.
The Group uses a provision matrix to calculate expected credit losses for trade receivables. The provision rates are depending on the number of days that a trade receivable is past due. The Group uses the grouping according to the customer segments that have similar loss patterns.
The provision matrix is initially based on the Group’s historical observed default rates. The Group will calibrate the matrix to adjust the historical credit loss experience with forward-looking information. At the end of each reporting period, the historical observed default rates are updated and changes in the forward-looking estimates are analysed.
The assessment of the correlation between historical observed default rates, forward-looking estimates and expected credit losses is a significant estimate. The amount of expected credit losses is sensitive to changes in circumstances and of forecast economic conditions over the expected lives of the financial assets. The Group’s historical credit loss experience and forecast of economic conditions may also not be representative of customer’s actual default in the future.
| F-78 |
| 5. | Property, plant and equipment |
| Furniture and fittings | Office equipment | Medical equipment | Computer equipment | Renovation | Laboratory | Air conditioner | Mobile device | Laboratory equipment | Plant and machinery | Computer and ICT |
Signboard | Motor vehicles |
Total | |||||||||||||||||||||||||||||||||||||||||||
| USD | USD | USD | USD | USD | USD | USD | USD | USD | USD | USD | USD | USD | USD | |||||||||||||||||||||||||||||||||||||||||||
| Cost | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| As at 1 April 2022 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Additions | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Written off | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||||||||||||||||||||||||||||||||||||||||
| Exchange difference | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||||||||||||||||||||||||||
As at 31 March 2023/ 1 April 2023 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Additions | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Written off | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||||||||||||||||||||||||||||||||||||||||||
| Exchange difference | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||||||||||||||||||||||
| As at 31 March 2024 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Accumulated depreciation | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| As at 1 April 2022 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Charge for the financial year | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Written off | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||||||||||||||||||||||||||||||||||||||||
| Exchange difference | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||||||||||||||||||||||||
As at 31 March 2023/ 1 April 2023 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Charge for the financial year | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Written off | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||||||||||||||||||||||||||||||||||||||||||
| Exchange difference | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||||||||||||||||||||||
| As at 31 March 2024 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Carrying amount | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| As at 31 March 2023 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| As at 31 March 2024 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Included
in property, plant and equipment of the Group is motor vehicles under hire-purchase with net book value of approximately USD (2023:
USD
| F-79 |
| 6. | Right-of-use assets |
Information about leases for which the Group is lessees are presented below:
| Office premises | ||||
| USD | ||||
| Cost | ||||
| As at 1 April 2022 | ||||
| Exchange difference | ( | ) | ||
| As at 31 March 2023/1 April 2023 | ||||
| Additions | ||||
| Derecognition arising from lease termination | ( | ) | ||
| Exchange difference | ( | ) | ||
| As at 31 March 2024 | ||||
| Accumulated amortisation | ||||
| As at 1 April 2022 | ||||
| Charge for the financial year | ||||
| Exchange difference | ( | ) | ||
| As at 31 March 2023/1 April 2023 | ||||
| Charge for the financial year | ||||
| Derecognition arising from lease termination | ( | ) | ||
| Exchange difference | ( | ) | ||
| As at 31 March 2024 | ||||
| Carrying amount | ||||
| As at 31 March 2023 | ||||
| As at 31 March 2024 | ||||
| F-80 |
| 7. | Intangible assets |
| Trademark | Patent | Technical know-how of stem cells cultivation | Total | |||||||||||||
| USD | USD | USD | USD | |||||||||||||
| Cost | ||||||||||||||||
| As at 1 April 2022 | ||||||||||||||||
| Exchange difference | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||
| As at 31 March 2023/1 April 2023 | ||||||||||||||||
| Additions | ||||||||||||||||
| Exchange difference | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||
| As at 31 March 2024 | ||||||||||||||||
| Accumulated amortisation | ||||||||||||||||
| As at 1 April 2022 | ||||||||||||||||
| Charge of the financial year | ||||||||||||||||
| Exchange difference | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||
| As at 31 March 2023/1 April 2023 | ||||||||||||||||
| Charge of the financial year | ||||||||||||||||
| Exchange difference | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||
| As at 31 March 2024 | ||||||||||||||||
| Carrying amount | ||||||||||||||||
| As at 31 March 2023 | ||||||||||||||||
| As at 31 March 2024 | ||||||||||||||||
During
the financial year ended 31 March 2024, the Group has entered into a patent license agreement with Ding KeXiang, wherein Ding KeXiang
shall grant to the Group the rights to exploit, use, license and sublicense all the patents owned by Ding KeXiang and develop, produce,
manufacture, market, sell, import and distribute the products and practice the processes under such patents, at the consideration of
CNY
| F-81 |
| 8. | Other investments |
| 2024 | 2023 | |||||||
| USD | USD | |||||||
| Unquoted shares - at cost | ||||||||
| As at 1 April/31 March | ||||||||
| Disposal during year | ( | ) | ||||||
| As at 31 March | ||||||||
| Less: Accumulated impairment loss | ||||||||
| As at 1 April | ( | ) | ( | ) | ||||
| Addition during the financial year | ( | ) | ||||||
| Reversal of accumulated impairment loss | ||||||||
| As at 31 March | ( | ) | ( | ) | ||||
| Exchange differences | ( | ) | ( | ) | ||||
As
of 31 March 2023, the Group had carried out an assessment on fair value of these balances and the corresponding fair value loss of USD
| F-82 |
| 9. | Investment in associates |
| 2024 | 2023 | |||||||
| USD | USD | |||||||
| Unquoted shares - at cost | ||||||||
| As at 1 April | ||||||||
| Addition during the financial year | ||||||||
| As at 31 March | ||||||||
| Less: Accumulated impairment loss | ||||||||
| As at 1 April | ( | ) | ( | ) | ||||
| Addition during the financial year | ( | ) | ||||||
| As at 31 March | ( | ) | ( | ) | ||||
| Share of post-acquisition reserve | ||||||||
| As at 1 April | ||||||||
| Share of profit during the financial year | ||||||||
| As at 31 March | ||||||||
| Exchange differences | ( | ) | ( | ) | ||||
The details of associates are as follows:
| Effective equity | ||||||||
| Place of | interest | |||||||
| Name of Company | incorporation | 2024 | 2023 | Principal activities | ||||
| % | % | |||||||
| Vax Biotech Sdn. Bhd. (“Vax Biotech”) | ||||||||
| Alps Globemedics Sdn. Bhd. (“Alps Globemedics”) | ||||||||
| Cilo Cybin Holdings Limited (“Cilo Cybin”) | ||||||||
Investment in Vax Biotech
As at 31 March 2023, the Group purchased ordinary shares of USD each in Vax Biotech Sdn. Bhd. Pursuant to share allotment exercise for a total consideration of USD in cash.
| F-83 |
| 9. | Investment in associates (Cont’d) |
Summarised financial information of Vax Biotech is set out below. The summarised financial information represents the amount in the financial statements of the associate and not the Group’s share of those amounts.
| 2024 | 2023 | |||||||
| USD | USD | |||||||
| Statement of financial position | ||||||||
| Current assets, representing total assets | ||||||||
| Current liabilities, representing total liabilities | ( | ) | ( | ) | ||||
| Net assets of the associate | ( | ) | ( | ) | ||||
| Statement of comprehensive income | ||||||||
| Expenses for the financial year | ( | ) | ( | ) | ||||
| Loss for the financial year | ( | ) | ( | ) | ||||
| Share of loss of the associate | ( | ) | ( | ) | ||||
Reconciliation of the above summarised financial information to the carrying amount of the interest in associate recognised in the consolidated financial statements:
| 2024 | 2023 | |||||||
| USD | USD | |||||||
| Proportion of net assets of the associate | ||||||||
| Exchange differences | ( | ) | ||||||
| Carrying amount of the Group’s interest in associate | ||||||||
Investment in Alps Globemedics
Summarised financial information of Alps Globemedics is set out below. The summarised financial information represents the amount in the financial statements of the associate and not the Group’s share of those amounts.
| 2024 | 2023 | |||||||
| USD | USD | |||||||
| Statement of financial position | ||||||||
| Non-current assets | ||||||||
| Current assets | ||||||||
| Total assets | ||||||||
| Non-current liabilities | ( | ) | ( | ) | ||||
| Current liabilities | ( | ) | ( | ) | ||||
| Total liabilities | ( | ) | ( | ) | ||||
| Net assets of the associate | ||||||||
| Statement of comprehensive income | ||||||||
| Revenue for the financial year | ||||||||
| (Loss)/Profit for the financial year | ( | ) | ||||||
| Share of (loss)/profit of the associate | ( | ) | ||||||
| F-84 |
| 9. | Investment in associates (Cont’d) |
Reconciliation of the above summarised financial information to the carrying amount of the interest in associate recognised in the consolidated financial statements:
| 2024 | 2023 | |||||||
| USD | USD | |||||||
| Proportion of net assets of the associate | ||||||||
| Exchange differences | ( | ) | ( | ) | ||||
| Carrying amount of the Group’s interest in associate | ||||||||
Investment in Cilo Cybin
On
6 February 2024, the Group had subscribed for of ordinary shares in Cilo Cybin Holdings Limited (the “Cilo Cybin”)
(for an aggregate of ordinary shares) at a price of ZAR per share. As a result, the Group hold a
Summarised financial information of Cilo Cybin is set out below. The summarised financial information represents the amount in the financial statements of the associate and not the Group’s share of those amounts.
| 2024 | 2023 | |||||||
| USD | USD | |||||||
| Statement of financial position | ||||||||
| Current assets, representing total assets | ||||||||
| Current liabilities, representing total liabilities | ( | ) | ||||||
| Net assets of the associate | ||||||||
| Statement of comprehensive income | ||||||||
| Profit for the financial period | ||||||||
| Share of profit of the associate | ||||||||
Reconciliation of the above summarised financial information to the carrying amount of the interest in associate recognised in the consolidated financial statements:
| 2024 | 2023 | |||||||
| USD | USD | |||||||
| Proportion of net assets of the associate | ||||||||
| Exchange differences | ( | ) | ||||||
| Carrying amount of the Group’s interest in associate | ||||||||
| 10. | Inventories |
| 2024 | 2023 | |||||||
| USD | USD | |||||||
| At cost: | ||||||||
| Trading | ||||||||
| Chemical reagents and products | ||||||||
| Medical substances and chemical stocks | ||||||||
| Consumables | ||||||||
| Beauty and healthcare product | ||||||||
| Work-in-progress | ||||||||
| Stem cells | ||||||||
The
cost of inventories recognised by the Group as an expense during the financial year is USD
| F-85 |
| 11. | Trade receivables |
The credit period granted to customers ranges from 30 to 60 days (2023: 30 to 60 days).
The Group applies a simplified approach in calculating loss allowances for trade receivable at an amount equal to lifetime ECL.
The Group does not hold any collateral or other credit enhancements over trade receivable balances.
The aged analysis of trade receivables at the end of the reporting period:
| 2024 | 2023 | |||||||
| USD | USD | |||||||
| Neither past due nor impair | ||||||||
| Past due but not impair: | ||||||||
| 1 to 30 days | ||||||||
| 30 to 60 days | ||||||||
| More than 60 days | ||||||||
Receivables that are neither past due nor impaired
Trade receivables that are neither past due nor impaired are creditworthy receivables with good payment records with the Group.
Receivables that are past due but not impaired
Trade receivables that were past due but not impaired relate to customers that have a good track record with the Group. Based on past experience and no adverse information to date, the directors of the Group are of the opinion that no provision for impairment is necessary in respect of these balances as there has not been a significant change in the credit quality and the balances are still considered recoverable.
| 12. | Other receivables, deposits and prepayments |
| 2024 | 2023 | |||||||
| USD | USD | |||||||
| Other receivables | ||||||||
| Deposits | ||||||||
| Prepayments | ||||||||
| F-86 |
| 13. | Related party transactions |
| (a) | Identify related parties |
For the purposes of the consolidated financial statements, parties are considered to be related to the Group if the Group has the ability, directly or indirectly, to control the party or exercise significant influence over the party in making financial and operating decisions, or vice versa, or where the Group and the party are subject to common control. Related parties may be individuals or other entities.
Related parties also include key management personnel. Key management personnel are defined as those persons having authority and responsibility for planning, directing and controlling the activities of the Group either directly or indirectly. The key management personnel of the Group include directors of the Group.
The related parties of the Group and of the Company are disclosed in the Note 9 and 31.
| (b) | Significant related party transactions and balances |
Amount due from associates represent non-trade balances which are unsecured, interest free and receivable on demand.
Amount due to directors represent non-trade advances for working capital purposes in meeting the operational needs of the Group, which are unsecured, interest free and repayable on demand. The amount due to directors are expected to be settled progressively in cash within the next two (2) years from the effectiveness of this proxy statement/ prospectus.
Related party transactions have been entered in the normal course of business under negotiated terms. In addition to the related party balances disclosed elsewhere in the consolidated financial statements, the related party transactions of the Group are as follows:
| 2024 | 2023 | |||||||
| USD | USD | |||||||
| Associates: | ||||||||
| Alps Globemedics | ||||||||
| Agent commission | ||||||||
| Management fee income | ||||||||
| Purchases | ||||||||
| Related party: | ||||||||
| Director | ||||||||
| Agent commission | ||||||||
| Consultancy fee | ||||||||
| Acquisition of patent | ||||||||
| (c) | Compensation of key management personnel |
The remuneration of key management personnel during the financial year are as follows:
| 2024 | 2023 | |||||||
| USD | USD | |||||||
| Fee | ||||||||
| Salaries and other emoluments | ||||||||
| Defined contribution plans | ||||||||
| F-87 |
| 14. | Share capital |
| Number of ordinary shares | Amount | |||||||||||||||
| 2024 | 2023 | 2024 | 2023 | |||||||||||||
| USD | USD | |||||||||||||||
| Issued and fully paid: | ||||||||||||||||
| Ordinary shares | ||||||||||||||||
| As at 1 April | ||||||||||||||||
| Issuance of shares during the financial year | ||||||||||||||||
| Subdivision of shares during the financial year | - | - | ||||||||||||||
| As at 31 March | ||||||||||||||||
On 20 April 2022, the issued and paid-up capital of the Company was increased from ordinary shares to ordinary shares by way of issuance and allotment of new ordinary shares at an issue price of approximately USD per ordinary share.
On 23 September 2022, the Company subdivided existing ordinary shares in the Company, held by the shareholders of the Company as at the close business on 23 September 2022 into ordinary shares in the Company.
On 29 January 2024, the issued and paid-up capital of the Company was increased from ordinary shares to ordinary shares by way of issuance and allotment of new ordinary shares at an issue price of approximately USD per ordinary share.
On 23 February 2024, the Company subdivided existing ordinary shares in the Company, held by the shareholders of the Company as at the close business on 23 February 2024 into ordinary shares in the Company.
The new ordinary shares issued rank pari passu with the then existing ordinary shares of the Company.
The holders of ordinary shares are entitled to receive dividends as declared from time to time and are entitle to one vote per ordinary share at meeting of the Company. All ordinary shares rank equally with regard to the Company’s residual assets.
| 15. | Share application monies |
As at 31 March 2023, the Company received USD towards allotment of equity shares which have been allocated to the applicants and subject to the Company’s shareholder’s agreements and term sheet of the Company, where applicable. The directors had completed the share allotment exercise as at 31 March 2024.
| 16. | Merger reserves |
The merger reserves represent the difference between the carrying value of the investment in subsidiaries and the share capital of the Company’s subsidiaries upon consolidation under the merger accounting principle.
| F-88 |
| 17. | Foreign translation reserve |
The foreign currency translation reserve is used to record exchange differences arising from the translation of the financial statements from functional currency to presentation currency.
| 18. | Hire purchase liabilities |
| 2024 | 2023 | |||||||
| USD | USD | |||||||
| Total outstanding | ||||||||
| Less: Interest-in-suspense | ( | ) | ( | ) | ||||
| Principal outstanding | ||||||||
| Less: Amount due within 12 months (shown under current liabilities) | ( | ) | ( | ) | ||||
The
interest rates implicit in these hire-purchase obligations at
| 19. | Lease liabilities |
The maturity analysis of the lease liabilities are as follows:
| 2024 | 2023 | |||||||
| USD | USD | |||||||
| Minimum lease payments: | ||||||||
| Less than one year | ||||||||
| Later than one year but not later than five years | ||||||||
| Total minimum lease payments | ||||||||
| Less: Unearned interest | ( | ) | ( | ) | ||||
| Present value of lease liabilities | ||||||||
The lease liabilities component is analysed as follows:
| 2024 | 2023 | |||||||
| USD | USD | |||||||
| Current | ||||||||
| Non-current | ||||||||
The Group does not face a significant liquidity risk with regard to its lease liabilities.
The
lease liabilities comprise of office premises. The Group applied the incremental borrowing rate to the lease liabilities of
| F-89 |
| 20. | Deferred tax liabilities |
Deferred tax assets and liabilities are offset when there is a legally enforceable right to set off current tax assets against current tax liabilities and when the deferred taxes relate to the same tax authority. The movements in deferred tax, determined after appropriate offsetting, during the financial year are as follows:
| 2024 | 2023 | |||||||
| USD | USD | |||||||
| As at 1 April | ||||||||
| Credited to profit or loss (Note 26): | ||||||||
| Property, plant and equipment | ( | ) | ||||||
| Exchange differences | ( | ) | ( | ) | ||||
| As at 31 March | ||||||||
As at 31 March 2023, the deferred tax in the financial statements is in respect of tax effects arising from deductible temporary differences on property, plant, and equipment.
| 21. | Trade payables |
The normal credit terms granted to the Group ranges from 30 to 120 days (2023: 30 to 120 days).
| 22. | Other payables and accruals |
| 2024 | 2023 | |||||||
| USD | USD | |||||||
| Other payables | ||||||||
| Accruals | ||||||||
| Deposit received | ||||||||
As
of financial year ended 31 March 2024, included in other payables are amount due to a shareholder amounting to USD
| 23. | Revenue |
| 2024 | 2023 | |||||||
| USD | USD | |||||||
| Cellular therapy | ||||||||
| Medical testing, laboratory and aesthetics beauty services | ||||||||
| Consultation fee | ||||||||
| Sale of medicine and healthcare product | ||||||||
| Timing of revenue recognition: | ||||||||
| At a point in time | ||||||||
| F-90 |
| 24. | Finance costs |
| 2024 | 2023 | |||||||
| USD | USD | |||||||
| Interest expenses on: | ||||||||
| Hire purchase liabilities | ||||||||
| Lease liabilities | ||||||||
| 25. | Loss before tax |
Loss before tax for the financial year is arrived at after charging/(crediting):
| 2024 | 2023 | |||||||
| USD | USD | |||||||
| Auditors’ remuneration: | ||||||||
| Current financial year | ||||||||
| Underprovision in previous financial year | ||||||||
| Amortisation of intangible assets | ||||||||
| Amortisation of right of use assets | ||||||||
| Depreciation of property, plant and equipment | ||||||||
| Expenses relating to short-term leases | ||||||||
| Gain on disposal of other investment | ( | ) | ||||||
| Impairment losses on investment in unquoted shares | ||||||||
| Impairment losses on investment in associates | ||||||||
| Property, plant and equipment written off | ||||||||
| Staff costs: | ||||||||
| Salaries, wages and allowances | ||||||||
| Employees Provident Fund | ||||||||
| Other employees’ benefit | ||||||||
Included
in staff costs of the Group is directors’ remuneration of USD
| F-91 |
| 26. | Income tax (credit)/expense |
| 2024 | 2023 | |||||||
| USD | USD | |||||||
| Estimated tax payable: | ||||||||
| Current financial year | ||||||||
| Under provision in prior financial years | ||||||||
| Deferred tax (Note 20): | ||||||||
| Current financial year | ( | ) | ||||||
| Over provision in prior financial years | ( | ) | ||||||
| ( | ) | ( | ) | |||||
| ( | ) | |||||||
A reconciliation of income tax credit applicable to loss before tax at the applicable statutory income tax rate to income tax expense at the effective income tax rate of the Group is as follows:
| 2024 | 2023 | |||||||
| USD | USD | |||||||
| Loss before tax | ( | ) | ( | ) | ||||
| Less: Share result of associates | ( | ) | ( | ) | ||||
| ( | ) | ( | ) | |||||
| Taxation at statutory rate of | ( | ) | ( | ) | ||||
| Tax effects of: | ||||||||
| Non-taxable income | ( | ) | ||||||
| Expenses not deductible | ||||||||
| Utilisation of deferred tax assets previously not recognised | ( | ) | ( | ) | ||||
| Deferred tax assets not recognised | ||||||||
| Under/(Over) provision in prior financial years: | ||||||||
| Estimated tax payable | ||||||||
| Deferred tax | ( | ) | ||||||
| ( | ) | |||||||
At the end of the reporting period, the estimated amount of deductible temporary differences, unabsorbed capital allowances and unutilised tax losses of the Group, for which the deferred tax assets have not been recognised in the financial statements due to uncertainty of their realisation, is as follows:
| 2024 | 2023 | |||||||
| USD | USD | |||||||
| Temporary differences arising from: | ||||||||
| Property, plant and equipment | ( | ) | ( | ) | ||||
| Right-of-use assets and lease liabilities | ||||||||
| Unabsorbed capital allowances | ||||||||
| Unutilised tax losses | ||||||||
| F-92 |
| 26. | Income tax (credit)/expense (Cont’d) |
At the end of the reporting period, the Group has deductible temporary differences, unabsorbed capital allowances and unutilised tax losses that are available for offsetting against future taxable profits, subject to the agreement with the tax authorities.
Expiry date of the unutilised tax losses is summarised below:
| 2024 | 2023 | |||||||
| USD | USD | |||||||
| Year of assessment: | ||||||||
| 2028 | ||||||||
| 2029 | ||||||||
| 2030 | ||||||||
| 2031 | ||||||||
| 2032 | ||||||||
| 2033 | ||||||||
| 2034 | ||||||||
| 27. | Loss per ordinary share |
Basic and diluted loss per ordinary share attributable to owner of the Company are computed by dividing the loss for the financial year attributable to owners of the Company by the weighted average number of ordinary shares in issue during the financial year.
Basis loss per ordinary share
| 2024 | 2023 | |||||||
| USD | USD | |||||||
| Loss attributable to owners of the Company (USD) | ( | ) | ( | ) | ||||
| Weighted average number of ordinary shares (units) |
| Basic and diluted loss per ordinary share (USD) | ) | ) |
Diluted loss per ordinary share
There were no dilutive potential equity instruments in issue as at financial year ended that have dilutive effect to the loss per ordinary share.
| F-93 |
| 28. | Operating segments |
The Group is organised into business units based on its products and services, and has three reportable operating segments as follow:
| Medical and Wellness | Non-surgical aesthetic services, surgical aesthetic services, medical consultation, health screenings, cellular therapy and research and COVID-19 testing
| |
| Life Science | Whole genome sequencing and pharmacogenomics
| |
| Others | Investment holding and provision for management services |
Management monitors the operating results of its business units separately for the purpose of making decisions about resource allocation and performance assessment. Segment performance is evaluated based on operating profit or loss which, in certain respects as explained in the table below, is measured differently from operating profit or loss in the consolidated statement of profit or loss and other comprehensive income. Group financing (including finance costs) and income taxes are managed on a group basis and are not allocated to operating segments
The following is an analysis of the Group’s revenue and results by the operating segments.
| Medical and Wellness | Life Science | Others |
Eliminations | Consolidated | ||||||||||||||||
| USD | USD | USD | USD | USD | ||||||||||||||||
| The Group | ||||||||||||||||||||
| 2024 | ||||||||||||||||||||
| Revenue | ||||||||||||||||||||
| External sales | ||||||||||||||||||||
| Inter-segment sales | ( | ) | ||||||||||||||||||
| Total revenue | ( | ) | ||||||||||||||||||
| Result | ||||||||||||||||||||
| Loss from operations | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||||||
| Finance costs | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||||||
| Loss before tax | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||||||
| Income tax expense | ||||||||||||||||||||
| Loss for the financial year | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||||||
| Segment assets | ( | ) | ||||||||||||||||||
| Segment liabilities | ( | ) | ||||||||||||||||||
(Forward)
| F-94 |
| 28. | Operating segments (Cont’d) |
| Medical and Wellness | Life Science | Others | Eliminations | Consolidated | ||||||||||||||||
| USD | USD | USD | USD | USD | ||||||||||||||||
| The Group | ||||||||||||||||||||
| 2024 | ||||||||||||||||||||
| Other information | ||||||||||||||||||||
| Amortisation of intangible assets | ||||||||||||||||||||
| Amortisation of right-of-use assets | ||||||||||||||||||||
| Additional of property, plant and equipment | ||||||||||||||||||||
| Depreciation of property, plant and equipment | ||||||||||||||||||||
| Property, plant and equipment written off | ||||||||||||||||||||
| 2023 | ||||||||||||||||||||
| Revenue | ||||||||||||||||||||
| External sales | ||||||||||||||||||||
| Inter-segment sales | ( | ) | ||||||||||||||||||
| Total revenue | ( | ) | ||||||||||||||||||
| Result | ||||||||||||||||||||
| Profit/(Loss) from operations | ( | ) | ( | ) | ( | ) | ||||||||||||||
| Finance costs | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||||||
| Profit/(Loss) before tax | ( | ) | ( | ) | ( | ) | ||||||||||||||
| Income tax expense | ( | ) | ( | ) | ||||||||||||||||
| Profit/(Loss) for the financial year | ( | ) | ( | ) | ( | ) | ||||||||||||||
| Segment assets | ( | ) | ||||||||||||||||||
| Segment liabilities | ( | ) |
(Forward)
| F-95 |
| 28. | Operating segments (Cont’d) |
| Medical and Wellness | Life Science | Others | Eliminations | Consolidated | ||||||||||||||||
| USD | USD | USD | USD | USD | ||||||||||||||||
| The Group | ||||||||||||||||||||
| 2023 | ||||||||||||||||||||
| Other information | ||||||||||||||||||||
| Amortisation of intangible assets | ||||||||||||||||||||
| Amortisation of right-of-use assets | ||||||||||||||||||||
| Depreciation of property, plant and equipment | ||||||||||||||||||||
| Additional of property, plant and equipment | ||||||||||||||||||||
| Property, plant and equipment written off | ||||||||||||||||||||
| Impairment losses on investment in associates | ||||||||||||||||||||
| Impairment losses on investment in unquoted shares | ||||||||||||||||||||
Geographical segments
The Group’s revenue from contracts with customers were generated within Malaysia,
| 29. | Financial instrument |
The following table analyses the financial assets and liabilities in the statement of financial position by the class of financial instruments to which they are assigned, and therefore by the measurement basis:
| 2024 | 2023 | |||||||
| USD | USD | |||||||
| Financial assets | ||||||||
| Fair value through profit or loss | ||||||||
| Other investments | ||||||||
| At amortised cost | ||||||||
| Trade receivables | ||||||||
| Other receivables and deposits | ||||||||
| Amount due from associates | ||||||||
| Cash and bank balances | ||||||||
(Forward)
| F-96 |
| 29. | Financial instrument (Cont’d) |
| 2024 | 2023 | |||||||
| USD | USD | |||||||
| Financial liabilities | ||||||||
| At amortised cost | ||||||||
| Trade payables | ||||||||
| Other payables and accruals | ||||||||
| Amount due to directors | ||||||||
| Hire purchase liabilities | ||||||||
| Lease liabilities | ||||||||
Financial risk management objectives and policies
The Group is exposed to financial risk arising from its operations and the use of financial instruments. The key financial risks include credit risk and liquidity risk.
The following sections provide details regarding the Group’s exposure to the above-mentioned financial risks and the objectives, policies and processes for the management of those risks.
| (i) | Credit risk |
Credit risk is the risk of a financial loss to the Group that may arise if a customer or counterparty to a financial instrument fails to meet its contractual obligations. The Group’s exposure to credit risk mainly from trade receivables, other receivables and refundable deposits and amount due from associates.
The management has in place a credit procedure to monitor and minimises the exposure of default. Receivables are monitored on a regular and an ongoing basis. Credit evaluations are performed on all customers requiring credit over certain amount.
For cash and cash equivalents, the Group minimises credit risk by dealing exclusively with high credit rating counterparties.
The Group provides advances to associates. The Group monitors the results of the associates regularly.
Exposure to credit risk
The carrying amount of the financial assets recorded on the consolidated statement of financial position at the end of the reporting period represents the Group’s maximum exposure to credit risk in relation to financial assets. No financial assets carry a significant exposure to credit risk.
| F-97 |
| 29. | Financial instrument (Cont’d) |
| (ii) | Liquidity risk |
Liquidity risk is the risk that the Group encounters difficulty in meeting its obligations due to shortage of funds. The Group’s exposure to liquidity risk arises primarily from trade payables, other payables and accruals, amounts due to directors, hire purchase liabilities and lease liabilities.
The Group’s funding requirements and liquidity risk are managed with the objective of meeting business obligations on a timely basis. The Group manages liquidity risk by maintaining adequate reserves and banking facilities by continuously monitoring forecast and actual cash flows, and by matching the maturity profiles of financial assets and liabilities.
Analysis of financial instruments by remaining contractual maturities
The table below summarises the maturity profile of the Group’s financial liabilities at the end of the reporting period based on undiscounted contractual payments:
| Weighted average effective interest rate | Total carrying amount | On demand or within 1 year | Within
2 to | Total undiscounted cash flow | ||||||||||||||||
| % | USD | USD | USD | USD | ||||||||||||||||
| 2024 | ||||||||||||||||||||
| Non-interest bearing: | ||||||||||||||||||||
| Trade payables | ||||||||||||||||||||
| Other payables and accruals | ||||||||||||||||||||
| Amount due to directors | ||||||||||||||||||||
| Interest bearing: | ||||||||||||||||||||
| Hire purchase liabilities | ||||||||||||||||||||
| Lease liabilities | ||||||||||||||||||||
| 2023 | ||||||||||||||||||||
| Non-interest bearing: | ||||||||||||||||||||
| Trade payables | ||||||||||||||||||||
| Other payables and accruals | ||||||||||||||||||||
| Amount due to directors | ||||||||||||||||||||
| Interest bearing: | ||||||||||||||||||||
| Hire purchase liabilities | ||||||||||||||||||||
| Lease liabilities | ||||||||||||||||||||
Fair values of financial instruments
The fair values of other investment are measured at fair value in the statements of financial position as at the end of the reporting period using Level 3 inputs for the purpose of fair value hierarchy in accordance with the generally accepted pricing model based on the net asset value of the investments.
The carrying amounts of short-term receivables and payables and cash and cash equivalents approximate their fair value due to the relatively short-term nature of these financial instruments and insignificant impact of discounting.
The fair value of hire purchase liabilities and lease liabilities are determined by discounting the relevant cash flows using current interest rates for similar instruments as at the end of the financial reporting period.
| 30. | Capital management |
The Group’s objectives when managing capital are to safeguard the Group’s ability to continue as going concern in order to provide returns for shareholders and benefits for other stakeholders and to maintain an optimal capital structure to reduce the cost of capital.
In order to maintain or adjust the capital structure, the Group may adjusts the amount of dividends paid to shareholders, return capital to shareholders, issue new shares or sell assets to reduce debt.
The Group monitors capital on the basis of its business and operating requirements.
| F-98 |
| 31. | List of subsidiaries are as follows: |
| Name of | Date of | Place of | Effective interest | |||||||
| Company | acquisition | incorporation | 2024 | 2023 | Principal activities | |||||
| % | % | |||||||||
| ||||||||||
|
| |||||||||
|
| |||||||||
| ||||||||||
| ||||||||||
| ||||||||||
| ||||||||||
|
||||||||||
On 10 April 2023, a subsidiary of the Company, Alpscap Berhad entered into a Share Sale Agreement with the directors of the Group to acquire ordinary shares, representing % of the equity interest, in Alps Insurance PCC INC, for a total cash consideration of RM. Consequently, The Company’s effective equity interest in Alps Insurance PCC INC is %.
On
2 June 2023, the Company had transferred ordinary shares of Alpscap Berhad to Dr. Tham Seng Kong. Consequently, The Company’s
effective equity interest in Alpscap Berhad and Alps Insurance PCC INC are
| F-99 |
| 32. | Restatement of current year financial statements |
As described in Note 1 and detailed below, the Group identified certain misstatements in the historical presentation of the consolidated statement of financial position and the consolidated statement of cash flows. These adjustments were made to ensure:
| (a) | the classification of the investment in unquoted shares; | |
| (b) | the recognition of the patent license agreement in intangible assets; and | |
| (c) | the classification of the investment in an associate |
The effects of the restatements to the amounts presented in the consolidated statement of financial position and the consolidated statement of cash flow are summarised as below.
As previously | Adjustments | As | ||||||||||||||||||
| stated | (a) | (b) | (c) | restated | ||||||||||||||||
| Consolidated statement of financial position as at 30 March 2024 | ||||||||||||||||||||
| Non-Current Assets | ||||||||||||||||||||
| Intangible assets | ||||||||||||||||||||
| Other investments | ( | ) | ||||||||||||||||||
| Investment in associates | ||||||||||||||||||||
| Current Assets | ||||||||||||||||||||
| Other receivables, deposits and prepayments | ( | ) | ||||||||||||||||||
| Current Liabilities | ||||||||||||||||||||
| Other payables and accruals | ||||||||||||||||||||
| Amount due to directors | ||||||||||||||||||||
| Capital and Reserves | ||||||||||||||||||||
| Accumulated losses | ( | ) | ( | ) | ||||||||||||||||
| Foreign translation reserve | ( | ) | ( | ) | ( | ) | ||||||||||||||
| Consolidated statement of profit or loss and other comprehensive income for the financial year ended 30 March 2024 | ||||||||||||||||||||
| Share result of associate | ( | ) | ||||||||||||||||||
| Other comprehensive loss, net of income tax | ||||||||||||||||||||
| Foreign currency translation, representing other comprehensive loss for the financial year | ( | ) | ( | ) | ( | ) | ||||||||||||||
As previously | Adjustments | As | ||||||||||||||||||
| stated | (a) | (b) | (c) | restated | ||||||||||||||||
| Consolidated statement of cash flows for the financial year ended 31 March 2024 | ||||||||||||||||||||
| Cash flows used in operating activities | ||||||||||||||||||||
| Loss before tax | ( | ) | ( | ) | ||||||||||||||||
| Adjustments for: | ||||||||||||||||||||
| Share of results of associates | ( | ) | ( | ) | ||||||||||||||||
| Change in working capital | ||||||||||||||||||||
| Decrease in other receivables, deposits and prepayments | ( | ) | ||||||||||||||||||
| Cash flows used in investing activities | ||||||||||||||||||||
| Investment in unquoted shares | ( | ) | ||||||||||||||||||
| Investment in an associate | ( | ) | ( | ) | ||||||||||||||||
| 33. | Date of authorisation for issue |
The consolidated financial statements of the ALPS Global Holding Berhad for the financial year ended March 31, 2024 were authorised for issue in accordance with a resolution of the Board of Directors on December 4, 2024.
| F-100 |
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING
FIRM TO THE SHAREHOLDERS AND THE BOARD OF DIRECTORS
OF ALPS GLOBAL HOLDING PUBCO
[Company No.: OC-4099]
(Incorporated in the Cayman Islands)
Opinion on the Financial Statements
We have audited the accompanying statement of financial position of ALPS Global Holding Pubco as of August 31, 2024, and the statement of profit or loss and other comprehensive income, statement of changes in equity and statement of cash flows for the financial period May, 14 2024 (date of incorporation) to August 31, 2024, including the related notes (collectively referred to as the “financial statements”). In our opinion, the financial statements present fairly, in all material respects, the financial position of the Company as of August 31, 2024, and the results of its operations and its cash flows for the financial period May, 14 2024 (date of incorporation) to August 31, 2024, in conformity with International Accounting Standards and International Financial Reporting Standards as issued by the International Accounting Standards Board.
Basis for Opinion
These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits of these financial statements in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud.
The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits, we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
| F-101 |
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING
FIRM TO THE SHAREHOLDERS AND THE BOARD OF DIRECTORS
OF ALPS GLOBAL HOLDING PUBCO
[Company No.: OC-4099]
(Incorporated in the Cayman Islands)
Basis for Opinion (Cont’d)
Our audit included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audit also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audit provides a reasonable basis for our opinion.
UHY
Firm Number: AF 1411
Chartered Accountants
KUALA LUMPUR, MALAYSIA
September 27, 2024
| F-102 |
ALPS GLOBAL HOLDING PUBCO
STATEMENT OF FINANCIAL POSITION AS AT 31 AUGUST 2024
| 2024 | |||||
| Note | USD | ||||
| ASSET | |||||
| Current Asset | |||||
| Cash on hand, representing total asset | |||||
| EQUITY AND LIABILITIES | |||||
| Capital and Reserves | |||||
| Share capital | 5 | ||||
| Accumulated losses | ( | ) | |||
| Capital Deficiency | ( | ) | |||
| Current Liabilities | |||||
| Other payable and accrued expense, representing total liabilities | 6 | ||||
| Total Equity and Liabilities | |||||
The accompanying notes are an integral part of the financial statements
| F-103 |
ALPS GLOBAL HOLDING PUBCO
STATEMENT OF PROFIT OR LOSS AND OTHER COMPREHENSIVE INCOME
FOR THE FINANCIAL PERIOD 14 MAY 2024 (DATE OF INCORPORATION) TO 31 AUGUST 2024
14.05.2024 to 31.08.2024 | |||||
| (4 months) | |||||
| Note | USD | ||||
| Revenue | |||||
| Other operating income | |||||
| Other operating expenses | ( | ) | |||
| Loss before tax | 7 | ( | ) | ||
| Income tax expense | |||||
| Loss for the financial period and total comprehensive loss for the financial period | ( | ) | |||
The accompanying notes are an integral part of the financial statements
| F-104 |
ALPS GLOBAL HOLDING PUBCO
STATEMENTS OF CHANGES IN EQUITY
FOR THE FINANCIAL PERIOD ENDED 31 AUGUST 2024
Share capital | Accumulated losses |
Total | ||||||||||
| USD | USD | USD | ||||||||||
| As at 14 May 2024 (date of incorporation) | | |||||||||||
| Loss for the financial period, representing total comprehensive loss for the financial period | ( | ) | ( | ) | ||||||||
| As at 31 August 2024 | ( | ) | ( | ) | ||||||||
The accompanying notes are an integral part of the financial statements
| F-105 |
ALPS GLOBAL HOLDING PUBCO
STATEMENT OF CASH FLOWS
FOR THE FINANCIAL PERIOD 14 MAY 2024 (DATE OF INCORPORATION) TO 31 AUGUST 2024
14.05.2024 to 31.08.2024 | ||||
| (4 months) | ||||
| USD | ||||
| CASH FLOWS USED IN OPERATING ACTIVITIES | ||||
| Loss before tax | ( | ) | ||
| Changes in working capital: | ||||
| Increase in: | ||||
| Other payable and accrued expense | ||||
| Net Cash Used In Operating Activities | ||||
| NET MOVEMENT OF CASH AND CASH EQUIVALENTS | ||||
| CASH AND CASH EQUIVALENTS AT DATE OF INCORPORATION | ||||
| CASH AND CASH EQUIVALENTS AT END OF THE FINANCIAL PERIOD | ||||
The accompanying notes are an integral part of the financial statements
| F-106 |
NOTES TO THE FINANCIAL STATEMENTS
FOR THE FINANCIAL PERIOD ENDED 31 AUGUST 2024
| 1. | Corporate Information |
Business Description
ALPS Global Holding Pubco (the “Company”) is a private limited liability company incorporated and domiciled in Cayman Islands. The registered office of the Company is located at 89 Nexus Way, Camana Bay, Grand Cayman, KY1-9009, Cayman Islands.
The Company was incorporated on 14 May 2024 and is principally engaged in the business of investment holding.
| 2. | Basis of Presentation |
Statement of Compliance
The financial statements of the Company have been prepared in accordance with in accordance with International Accounting Standards (“IAS”) and International Financial Reporting Standard (“IFRS”) as issued by the International Accounting Standards Board (“IASB”).
Adoption of amendments to IAS and IFRS
During the financial period, the Company adopted the following applicable amendments to IFRS issued by IASB:
| IFRS 17 | Insurance Contracts |
| Amendments to IFRS 17 | Insurance Contracts |
| Amendments to IFRS 17 | Initial Application of IFRS 17 and IFRS 9 - Comparative information |
| Amendments to IAS 1 and IFRS Practice Statement 2 | Disclosure of Accounting Policies |
| Amendments to IAS 8 | Definition of Accounting Estimates |
| Amendments to IFRS 1 and IAS 12 | Deferred Tax related to Assets and Liabilities arising from a Single Transaction |
The adoption of these amendments to standards did not have any significant impact on the financial statements of the Company except for:
Amendments to IAS 1 and IFRS Practice Statement 2 Disclosure of Accounting Policies
The Company has adopted the amendments to IAS 1 Presentation of Financial Statements and IFRS Practice Statement 2 Materiality Practice Statement for the first time in the current financial period. The amendments change the requirements in IAS 1 Presentation of Financial Statements with regard to disclosure of accounting policies. The amendments replace all instances of the term “significant accounting policies” with “material accounting policy information”. Accounting policy information is material if, when considered together with other information included in an entity’s financial statements, it can reasonably be expected to influence decisions that the primary users of general purpose financial statements make on the basis of those financial statements.
| F-107 |
| 2. | Basis of Presentation (Cont’d) |
Adoption of amendments to IAS and IFRS (Cont’d)
Amendments to IAS 1 Presentation of Financial Statements are also amended to clarify that accounting policy information that relates to immaterial transactions, other events or conditions is immaterial and need not be disclosed. Accounting policy information may be material because of the nature of the related transactions, other events or conditions, even if the amounts are immaterial. However, not all accounting policy information relating to material transactions, other events or conditions is itself material.
The amendments have no effect on the measurement, recognition or presentation of any items in the Company’s financial statements but affect the disclosure of accounting policies.
New IFRS and amendments to IAS and IFRSs in issue but not yet effective
The Company has not applied the following new IFRS and amendments to IAS and IFRS that have been issued by the IASB but are not yet effective for the Company.
| Effective dates for financial periods beginning on or after | ||||
| Amendments to IFRS 16 | Lease Liability in a Sale and Leaseback | 1 January 2024 | ||
| Amendments to IAS 1 | Classification of Liabilities as Current or Non-Current | 1 January 2024 | ||
| Amendments to IAS 1 and Practice Statement 2 | Non-current Liabilities with Covenants | 1 January 2024 | ||
| Amendments to IFRS 107 and IFRS 7 | Supplier Finance Agreements | 1 January 2024 | ||
| Amendments to IFRS 121 | Lack of Exchangeability | 1 January 2025 | ||
| Amendments to IFRS 9 and IFRS 7 | Amendments to the Classification and Measurement of Financial Instruments | 1 January 2026 | ||
| IFRS 18 | Presentation and Disclosure in Financial Statements | 1 January 2027 | ||
| IFRS 19 | Subsidiaries without Public Accountability: Disclosures | 1 January 2027 | ||
| Amendments to IFRS 10 and IAS 28 | Sales or Contribution of between an Investor and its Associate or Joint Venture | Deferred until further notice |
The directors anticipate that the abovementioned new IFRS and amendments to IFRSs will be adopted in the annual financial statements of the Company when they become effective, if applicable, and that the adoption of the new IFRS and amendments to IFRS will have no material impact on the financial statements of the Company in the period of initial application.
| F-108 |
| 3. | Material Accounting Policy Information |
Basis of accounting
The financial statements of the Company have been prepared under the historical cost unless otherwise indicated in the accounting policy information below.
Functional and presentation currency
The financial statements of the Company are presented in United States Dollar (“USD”), which is the Company’s functional and presentation currency. All financial information is presented in USD.
Financial instruments
Financial assets and financial liabilities are recognised in the Company’s statements of financial position when the Company becomes a party to the contractual provisions of the instrument.
Financial assets and financial liabilities are initially measured at fair value. Transaction costs that are directly attributable to the acquisition or issue of financial assets and financial liabilities (other than financial assets and financial liabilities at fair value through profit or loss) are added to or deducted from the fair value of the financial assets or financial liabilities, as appropriate, on initial recognition. Transaction costs directly attributable to the acquisition of financial assets or financial liabilities at fair value through profit or loss are recognised immediately in profit or loss.
Financial assets
All regular way purchases or sales of financial assets are recognised and derecognised on a trade date basis. Regular way purchases or sales are purchases or sales of financial assets that require delivery of assets within the time frame established by regulation or convention in the marketplace.
All recognised financial assets are measured subsequently in their entirety at either amortised cost or fair value, depending on the classification of the financial assets.
Classification of financial assets
Debt instruments that meet the following conditions are measured subsequently at amortised cost:
| ● | the financial asset is held within a business model whose objective is to hold financial assets in order to collect contractual cash flows; and | |
| ● | the contractual terms of the financial asset give rise on specified dates to cash flows that are solely payments of principal and interest on the principal amount outstanding. |
Debt instruments that meet the following conditions are measured subsequently at fair value through other comprehensive income (“FVTOCI”):
| ● | the financial asset is held within a business model whose objective is achieved by both collecting contractual cash flows and selling the financial assets; and | |
| ● | the contractual terms of the financial asset give rise on specified dates to cash flows that are solely payments of principal and interest on the principal amount outstanding. |
By default, all other financial assets are measured subsequently at fair value through profit or loss (“FVTPL”).
| F-109 |
| 3. | Material Accounting Policy Information (Cont’d) |
Financial assets (Cont’d)
Despite the foregoing, the Company may make the following irrevocable election/designation at initial recognition of a financial asset:
| ● | the Company may irrevocably elect to present subsequent changes in fair value of an equity investment in other comprehensive income if certain criteria are met; and | |
| ● | the Company may irrevocably designate a debt instrument that meets the amortised cost or FVTOCI criteria as measured at FVTPL if doing so eliminates or significantly reduces an accounting mismatch (see (ii) below). |
| (i) | Amortised cost and effective interest method |
The effective interest method is a method of calculating the amortised cost of a debt instrument and of allocating interest income over the relevant period.
For financial assets other than purchased or originated credit-impaired financial assets (i.e. assets that are credit-impaired on initial recognition), the effective interest rate is the rate that exactly discounts estimated future cash receipts (including all fees and points paid or received that form an integral part of the effective interest rate, transaction costs and other premiums or discounts) excluding expected credit losses, through the expected life of the debt instrument, or, where appropriate, a shorter period, to the gross carrying amount of the debt instrument on initial recognition. For purchased or originated credit-impaired financial assets, a credit-adjusted effective interest rate is calculated by discounting the estimated future cash flows, including expected credit losses, to the amortised cost of the debt instrument on initial recognition.
The amortised cost of a financial asset is the amount at which the financial asset is measured at initial recognition minus the principal repayments, plus the cumulative amortisation using the effective interest method of any difference between that initial amount and the maturity amount, adjusted for any loss allowance. The gross carrying amount of a financial asset is the amortised cost of a financial asset before adjusting for any loss allowance.
Interest income is recognised using the effective interest method for debt instruments measured subsequently at amortised cost and at FVTOCI. For financial assets other than purchased or originated credit-impaired financial assets, interest income is calculated by applying the effective interest rate to the gross carrying amount of a financial asset, except for financial assets that have subsequently become credit-impaired (see below). For financial assets that have subsequently become credit-impaired, interest income is recognised by applying the effective interest rate to the amortised cost of the financial asset. If, in subsequent reporting periods, the credit risk on the credit-impaired financial instrument improves so that the financial asset is no longer credit-impaired, interest income is recognised by applying the effective interest rate to the gross carrying amount of the financial asset.
For purchased or originated credit-impaired financial assets, the Company recognises interest income by applying the credit-adjusted effective interest rate to the amortised cost of the financial asset from initial recognition. The calculation does not revert to the gross basis even if the credit risk of the financial asset subsequently improves so that the financial asset is no longer credit-impaired.
| F-110 |
| 3. | Material Accounting Policy Information (Cont’d) |
Financial assets (Cont’d)
| (ii) | Financial assets at FVTPL |
Financial assets that do not meet the criteria for being measured at amortised cost or FVTOCI are measured at FVTPL. Specifically:
| ● | Investments in equity instruments are classified as at FVTPL, unless the Company designates an equity investment that is neither held for trading nor a contingent consideration arising from a business combination as at FVTOCI on initial recognition. | |
| ● | Financial assets that do not meet the amortised cost criteria or the FVTOCI criteria are classified as at FVTPL. In addition, debt instruments that meet either the amortised cost criteria or the FVTOCI criteria may be designated as at FVTPL upon initial recognition if such designation eliminates or significantly reduces a measurement or recognition inconsistency (so called “accounting mismatch”) that would arise from measuring assets or liabilities or recognising the gains and losses on them on different bases. The Company has designated investment in quoted unit trust and other investment as at FVTPL. |
Financial assets at FVTPL are measured at fair value at the end of each reporting period, with any fair value gains or losses recognised in profit or loss to the extent they are not part of a designated hedging relationship. The net gain or loss from other financial assets are recognised in profit or loss includes any dividend or interest earned on the financial asset and is included in the “other operating income” or “other operating expenses” line item.
Financial liabilities
All financial liabilities are measured subsequently at amortised cost using the effective interest method.
Financial liabilities measured subsequently at amortised cost
Financial liabilities that are not (i) contingent consideration of an acquirer in a business combination, (ii) held-for-trading, or (iii) designated as at FVTPL, are measured subsequently at amortised cost using the effective interest method.
The effective interest method is a method of calculating the amortised cost of a financial liability and of allocating interest expense over the relevant period. The effective interest rate is the rate that exactly discounts estimated future cash payments (including all fees and points paid or received that form an integral part of the effective interest rate, transaction costs and other premiums or discounts) through the expected life of the financial liability, or (where appropriate) a shorter period, to the amortised cost of a financial liability.
Statement of cash flow
The Company adopt the indirect method in the preparation of the statements of cash flow.
For the purpose of the statements of cash flows, cash and cash equivalents are cash on hand.
| F-111 |
| 4. | Critical accounting judgements and key sources of estimation uncertainty |
Directors have used estimates and assumptions in measuring the reported amounts of assets and liabilities at the end of the reporting period and the reported amounts of expenses during the reporting period. Judgements and assumptions are applied in the measurement, and hence, the actual results may not coincide with the report amounts.
| (a) | Critical judgements in applying the Company’s accounting policies |
In the process of applying the Company’s accounting policies, the directors are of the opinion that there are no instances of application of judgements which are expected to have a significant effect on the amounts recognised in the financial statements.
| (b) | Key sources of estimation uncertainty |
Directors believe that there are no key assumptions made concerning the future, and other key sources of estimation uncertainty at the end of the reporting period, that have a significant risk of causing a material adjustment to the carrying amounts of assets and liabilities within the next reporting period.
| 5. | Share capital |
Number of ordinary shares | Amount | |||||||
| 2024 | 2024 | |||||||
| Units | USD | |||||||
| Issued and fully paid: | ||||||||
| Ordinary shares | ||||||||
| As at 14 May 2024 (date of incorporation)/31 August | ||||||||
The Company was incorporated on 14 May 2024 with an issued and paid up share capital of USD consisting of ordinary shares.
| 6. | Other payables and accrued expense |
| 2024 | ||||
| USD | ||||
| Other payable | ||||
| Accrued expense | ||||
| F-112 |
| 7. | Loss before tax |
Loss before tax for the financial period is arrived at after charging:
14.05.2024 to 31.08.2024 | ||||
| (4 months) | ||||
| USD | ||||
| Auditors’ remuneration | ||||
| Incorporation expenses | ||||
| 8. | Financial instrument |
The following table analyses the financial assets and liabilities in the statement of financial position by the class of financial instruments to which they are assigned, and therefore by the measurement basis:
| 2024 | ||||
| USD | ||||
| Financial assets | ||||
| At amortised cost | ||||
| Cash on hand | ||||
| Financial liabilities | ||||
| At amortised cost | ||||
| Other payable and accrued expense | ||||
Financial risk management objectives and policies
The Company is exposed to financial risk arising from its operations and the use of financial instruments. The key financial risk is liquidity risk.
The following sections provide details regarding the Company’s exposure to the above-mentioned financial risks and the objectives, policies and processes for the management of the risk.
Liquidity risk
Liquidity risk is the risk that the Company encounters difficulty in meeting its obligations due to shortage of funds. The Company’s exposure to liquidity risk arises primarily from other payables.
The Company’s funding requirements and liquidity risk are managed with the objective of meeting business obligations on a timely basis. The Company manages liquidity risk by maintaining adequate reserves and banking facilities by continuously monitoring forecast and actual cash flows, and by matching the maturity profiles of financial assets and liabilities.
All financial liabilities of the Company are assessed as current and correspondingly, no detailed maturity analysis is deemed necessary.
Fair values of financial instruments
The carrying amounts of short-term payable and cash and cash equivalents approximate their fair value due to the relatively short-term nature of these financial instruments and insignificant impact of discounting.
| 9. | Capital management |
The Company’s objectives when managing capital are to safeguard the Company’s ability to continue as going concern in order to provide returns for shareholders and benefits for other stakeholders and to maintain an optimal capital structure to reduce the cost of capital.
In order to maintain or adjust the capital structure, the Company may adjusts the amount of dividends paid to shareholders, return capital to shareholders, issue new shares or sell assets to reduce debt.
The Company monitors capital on the basis of its business and operating requirements.
| 10. | Date of authorisation for issue |
The financial statements of the ALPS Global Holding Pubco for the financial period ended August 31, 2024 were authorised for issue in accordance with a resolution of the Board of Directors on September 27, 2024.
| F-113 |
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING
FIRM TO THE SHAREHOLDERS AND THE BOARD OF DIRECTORS
OF ALPS LIFE SCIENCE INC
[Company No.: OC-408826]
(Incorporated in the Cayman Islands)
Opinion on the Financial Statements
We have audited the accompanying statement of financial position of Alps Life Sciences Inc as of May 31, 2024, and the statement of profit or loss and other comprehensive income, statement of changes in equity and statement of cash flows for the financial period April, 11 2024 (date of incorporation) to May 31, 2024, including the related notes (collectively referred to as the “financial statements”). In our opinion, the financial statements present fairly, in all material respects, the financial position of the Company as of May 31, 2024, and the results of its operations and its cash flows for the financial period April, 11 2024 (date of incorporation) to May 31, 2024, in conformity with International Accounting Standards and International Financial Reporting Standards as issued by the International Accounting Standards Board.
Basis for Opinion
These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits of these financial statements in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud.
The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits, we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
| F-114 |
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING
FIRM TO THE SHAREHOLDERS AND THE BOARD OF DIRECTORS
OF ALPS LIFE SCIENCE INC
[Company No.: OC-408826]
(Incorporated in the Cayman Islands)
Basis for Opinion (Cont’d)
Our audit included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audit also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audit provides a reasonable basis for our opinion.
UHY
Firm Number: AF 1411
Chartered Accountants
KUALA LUMPUR, MALAYSIA
November 4, 2024
| F-115 |
ALPS LIFE SCIENCE INC
STATEMENT OF FINANCIAL POSITION AS AT 31 MAY 2024
| 2024 | |||||||
| Note | USD | ||||||
| EQUITY AND LIABILITIES | |||||||
| Capital and Reserves | |||||||
| Share capital | 5 | * | |||||
| Accumulated losses | ( | ) | |||||
| Capital Deficiency | ( | ) | |||||
| Current Liabilities | |||||||
| Other payable and accrued expense, representing total liabilities | 6 | ||||||
| Total Equity and Liabilities | |||||||
| * |
The accompanying notes are an integral part of the financial statements
| F-116 |
ALPS LIFE SCIENCE INC
STATEMENT OF PROFIT OR LOSS AND OTHER COMPREHENSIVE INCOME
FOR THE FINANCIAL PERIOD 11 APRIL 2024 (DATE OF INCORPORATION) TO 31 MAY 2024
11.04.2024 to 31.5.2024 | |||||||
| (2 months) | |||||||
| Note | USD | ||||||
| Revenue | |||||||
| Other operating income | |||||||
| Other operating expenses | ( | ) | |||||
| Loss before tax | 7 | ( | ) | ||||
| Income tax expense | |||||||
| Loss for the financial period and total comprehensive loss for the financial period | ( | ) | |||||
The accompanying notes are an integral part of the financial statements
| F-117 |
ALPS LIFE SCIENCE INC
STATEMENTS OF CHANGES IN EQUITY
FOR THE FINANCIAL PERIOD ENDED 31 MAY 2024
Share capital | Accumulated losses |
Total | ||||||||||
| USD | USD | USD | ||||||||||
| As at 11 April 2024 (date of incorporation) | * | |||||||||||
| Loss for the financial period, representing total comprehensive loss for the financial period | ( | ) | ( | ) | ||||||||
| As at 31 May 2024 | * | ( | ) | ( | ) | |||||||
| * |
The accompanying notes are an integral part of the financial statements
| F-118 |
ALPS LIFE SCIENCE INC
STATEMENT OF CASH FLOWS
FOR THE FINANCIAL PERIOD 11 APRIL 2024 (DATE OF INCORPORATION) TO 31 MAY 2024
11.04.2024 to 31.5.2024 | ||||
| (2 months) | ||||
| USD | ||||
| CASH FLOWS USED IN OPERATING ACTIVITIES | ||||
| Loss before tax | ( | ) | ||
| Changes in working capital: | ||||
| Increase in: | ||||
| Other payable and accrued expense | ||||
| Net Cash Used In Operating Activities | ||||
| NET MOVEMENT OF CASH AND CASH EQUIVALENTS | ||||
| CASH AND CASH EQUIVALENTS AT DATE OF INCORPORATION | ||||
| CASH AND CASH EQUIVALENTS AT END OF THE FINANCIAL PERIOD | ||||
The accompanying notes are an integral part of the financial statements
| F-119 |
NOTES TO THE FINANCIAL STATEMENTS
FOR THE FINANCIAL PERIOD ENDED 31 MAY 2024
| 1. | Corporate Information |
Business Description
Alps Life Sciences Inc (the “Company”) is a private limited liability company incorporated and domiciled in Cayman Islands. The registered office of the Company is located at 89 Nexus Way, Camana Bay, Grand Cayman, KY1-9009, Cayman Islands.
The Company was incorporated on 11 April 2024 and is principally engaged in the business of investment holding.
| 2. | Basis of Presentation |
Statement of Compliance
The financial statements of the Company have been prepared in accordance with in accordance with International Accounting Standards (“IAS”) and International Financial Reporting Standard (“IFRS”) as issued by the International Accounting Standards Board (“IASB”).
Adoption of amendments to IAS and IFRS
During the financial period, the Company adopted the following applicable amendments to IFRS issued by IASB:
| IFRS 17 | Insurance Contracts |
| Amendments to IFRS 17 | Insurance Contracts |
| Amendments to IFRS 17 | Initial Application of IFRS 17 and IFRS 9 - Comparative information |
| Amendments to IAS 1 and IFRS Practice Statement 2 | Disclosure of Accounting Policies |
| Amendments to IAS 8 | Definition of Accounting Estimates |
| Amendments to IFRS 1 and IAS 12 | Deferred Tax related to Assets and Liabilities arising from a Single Transaction |
The adoption of these amendments to standards did not have any significant impact on the financial statements of the Company except for:
Amendments to IAS 1 and IFRS Practice Statement 2 Disclosure of Accounting Policies
The Company has adopted the amendments to IAS 1 Presentation of Financial Statements and IFRS Practice Statement 2 Materiality Practice Statement for the first time in the current financial period. The amendments change the requirements in IAS 1 Presentation of Financial Statements with regard to disclosure of accounting policies. The amendments replace all instances of the term “significant accounting policies” with “material accounting policy information”. Accounting policy information is material if, when considered together with other information included in an entity’s financial statements, it can reasonably be expected to influence decisions that the primary users of general purpose financial statements make on the basis of those financial statements.
| F-120 |
| 2. | Basis of Presentation (Cont’d) |
Adoption of amendments to IAS and IFRS (Cont’d)
Amendments to IAS 1 Presentation of Financial Statements are also amended to clarify that accounting policy information that relates to immaterial transactions, other events or conditions is immaterial and need not be disclosed. Accounting policy information may be material because of the nature of the related transactions, other events or conditions, even if the amounts are immaterial. However, not all accounting policy information relating to material transactions, other events or conditions is itself material.
The amendments have no effect on the measurement, recognition or presentation of any items in the Company’s financial statements but affect the disclosure of accounting policies.
New IFRS and amendments to IAS and IFRSs in issue but not yet effective
The Company has not applied the following new IFRS and amendments to IAS and IFRS that have been issued by the IASB but are not yet effective for the Company.
| Effective dates for financial periods beginning on or after | ||||
| Amendments to IFRS 16 | Lease Liability in a Sale and Leaseback | 1 January 2024 | ||
| Amendments to IAS 1 | Classification of Liabilities as Current or Non-Current | 1 January 2024 | ||
| Amendments to IAS 1 and Practice Statement 2 | Non-current Liabilities with Covenants | 1 January 2024 | ||
| Amendments to IFRS 107 and IFRS 7 | Supplier Finance Agreements | 1 January 2024 | ||
| Amendments to IFRS 121 | Lack of Exchangeability | 1 January 2025 | ||
| Amendments to IFRS 9 and IFRS 7 | Amendments to the Classification and Measurement of Financial Instruments | 1 January 2026 | ||
| IFRS 18 | Presentation and Disclosure in Financial Statements | 1 January 2027 | ||
| IFRS 19 | Subsidiaries without Public Accountability: Disclosures | 1 January 2027 | ||
| Amendments to IFRS 10 and IAS 28 | Sales or Contribution of between an Investor and its Associate or Joint Venture | Deferred until further notice |
The directors anticipate that the abovementioned new IFRS and amendments to IFRSs will be adopted in the annual financial statements of the Company when they become effective, if applicable, and that the adoption of the new IFRS and amendments to IFRS will have no material impact on the financial statements of the Company in the period of initial application.
| 3. | Material Accounting Policy Information |
Basis of accounting
The financial statements of the Company have been prepared under the historical cost unless otherwise indicated in the accounting policy information below.
| F-121 |
| 3. | Material Accounting Policy Information (Cont’d) |
Functional and presentation currency
The financial statements of the Company are presented in United States Dollar (“USD”), which is the Company’s functional and presentation currency. All financial information is presented in USD.
Financial instruments
Financial assets and financial liabilities are recognised in the Company’s statements of financial position when the Company becomes a party to the contractual provisions of the instrument.
Financial assets and financial liabilities are initially measured at fair value. Transaction costs that are directly attributable to the acquisition or issue of financial assets and financial liabilities (other than financial assets and financial liabilities at fair value through profit or loss) are added to or deducted from the fair value of the financial assets or financial liabilities, as appropriate, on initial recognition. Transaction costs directly attributable to the acquisition of financial assets or financial liabilities at fair value through profit or loss are recognised immediately in profit or loss.
Financial assets
All regular way purchases or sales of financial assets are recognised and derecognised on a trade date basis. Regular way purchases or sales are purchases or sales of financial assets that require delivery of assets within the time frame established by regulation or convention in the marketplace.
All recognised financial assets are measured subsequently in their entirety at either amortised cost or fair value, depending on the classification of the financial assets.
Classification of financial assets
Debt instruments that meet the following conditions are measured subsequently at amortised cost:
| ● | the financial asset is held within a business model whose objective is to hold financial assets in order to collect contractual cash flows; and | |
| ● | the contractual terms of the financial asset give rise on specified dates to cash flows that are solely payments of principal and interest on the principal amount outstanding. |
Debt instruments that meet the following conditions are measured subsequently at fair value through other comprehensive income (“FVTOCI”):
| ● | the financial asset is held within a business model whose objective is achieved by both collecting contractual cash flows and selling the financial assets; and | |
| ● | the contractual terms of the financial asset give rise on specified dates to cash flows that are solely payments of principal and interest on the principal amount outstanding. |
By default, all other financial assets are measured subsequently at fair value through profit or loss (“FVTPL”).
| F-122 |
| 3. | Material Accounting Policy Information (Cont’d) |
Financial assets (Cont’d)
Despite the foregoing, the Company may make the following irrevocable election/designation at initial recognition of a financial asset:
| ● | the Company may irrevocably elect to present subsequent changes in fair value of an equity investment in other comprehensive income if certain criteria are met; and | |
| ● | the Company may irrevocably designate a debt instrument that meets the amortised cost or FVTOCI criteria as measured at FVTPL if doing so eliminates or significantly reduces an accounting mismatch (see (ii) below). |
| (i) | Amortised cost and effective interest method |
The effective interest method is a method of calculating the amortised cost of a debt instrument and of allocating interest income over the relevant period.
For financial assets other than purchased or originated credit-impaired financial assets (i.e. assets that are credit-impaired on initial recognition), the effective interest rate is the rate that exactly discounts estimated future cash receipts (including all fees and points paid or received that form an integral part of the effective interest rate, transaction costs and other premiums or discounts) excluding expected credit losses, through the expected life of the debt instrument, or, where appropriate, a shorter period, to the gross carrying amount of the debt instrument on initial recognition. For purchased or originated credit-impaired financial assets, a credit-adjusted effective interest rate is calculated by discounting the estimated future cash flows, including expected credit losses, to the amortised cost of the debt instrument on initial recognition.
The amortised cost of a financial asset is the amount at which the financial asset is measured at initial recognition minus the principal repayments, plus the cumulative amortisation using the effective interest method of any difference between that initial amount and the maturity amount, adjusted for any loss allowance. The gross carrying amount of a financial asset is the amortised cost of a financial asset before adjusting for any loss allowance.
Interest income is recognised using the effective interest method for debt instruments measured subsequently at amortised cost and at FVTOCI. For financial assets other than purchased or originated credit-impaired financial assets, interest income is calculated by applying the effective interest rate to the gross carrying amount of a financial asset, except for financial assets that have subsequently become credit-impaired (see below). For financial assets that have subsequently become credit-impaired, interest income is recognised by applying the effective interest rate to the amortised cost of the financial asset. If, in subsequent reporting periods, the credit risk on the credit-impaired financial instrument improves so that the financial asset is no longer credit-impaired, interest income is recognised by applying the effective interest rate to the gross carrying amount of the financial asset.
For purchased or originated credit-impaired financial assets, the Company recognises interest income by applying the credit-adjusted effective interest rate to the amortised cost of the financial asset from initial recognition. The calculation does not revert to the gross basis even if the credit risk of the financial asset subsequently improves so that the financial asset is no longer credit-impaired.
| F-123 |
| 3. | Material Accounting Policy Information (Cont’d) |
Financial assets (Cont’d)
| (ii) | Financial assets at FVTPL |
Financial assets that do not meet the criteria for being measured at amortised cost or FVTOCI are measured at FVTPL. Specifically:
| ● | Investments in equity instruments are classified as at FVTPL, unless the Company designates an equity investment that is neither held for trading nor a contingent consideration arising from a business combination as at FVTOCI on initial recognition. | |
| ● | Financial assets that do not meet the amortised cost criteria or the FVTOCI criteria are classified as at FVTPL. In addition, debt instruments that meet either the amortised cost criteria or the FVTOCI criteria may be designated as at FVTPL upon initial recognition if such designation eliminates or significantly reduces a measurement or recognition inconsistency (so called “accounting mismatch”) that would arise from measuring assets or liabilities or recognising the gains and losses on them on different bases. The Company has designated investment in quoted unit trust and other investment as at FVTPL. |
Financial assets at FVTPL are measured at fair value at the end of each reporting period, with any fair value gains or losses recognised in profit or loss to the extent they are not part of a designated hedging relationship. The net gain or loss from other financial assets are recognised in profit or loss includes any dividend or interest earned on the financial asset and is included in the “other operating income” or “other operating expenses” line item.
Financial liabilities
All financial liabilities are measured subsequently at amortised cost using the effective interest method.
Financial liabilities measured subsequently at amortised cost
Financial liabilities that are not (i) contingent consideration of an acquirer in a business combination, (ii) held-for-trading, or (iii) designated as at FVTPL, are measured subsequently at amortised cost using the effective interest method.
The effective interest method is a method of calculating the amortised cost of a financial liability and of allocating interest expense over the relevant period. The effective interest rate is the rate that exactly discounts estimated future cash payments (including all fees and points paid or received that form an integral part of the effective interest rate, transaction costs and other premiums or discounts) through the expected life of the financial liability, or (where appropriate) a shorter period, to the amortised cost of a financial liability.
Statement of cash flow
The Company adopt the indirect method in the preparation of the statements of cash flow.
For the purpose of the statements of cash flows, cash and cash equivalents are cash on hand.
| F-124 |
| 4. | Critical accounting judgements and key sources of estimation uncertainty |
Directors have used estimates and assumptions in measuring the reported amounts of assets and liabilities at the end of the reporting period and the reported amounts of expenses during the reporting period. Judgements and assumptions are applied in the measurement, and hence, the actual results may not coincide with the report amounts.
| (a) | Critical judgements in applying the Company’s accounting policies |
In the process of applying the Company’s accounting policies, the directors are of the opinion that there are no instances of application of judgements which are expected to have a significant effect on the amounts recognised in the financial statements.
| (b) | Key sources of estimation uncertainty |
Directors believe that there are no key assumptions made concerning the future, and other key sources of estimation uncertainty at the end of the reporting period, that have a significant risk of causing a material adjustment to the carrying amounts of assets and liabilities within the next reporting period.
| 5. | Share capital |
| Number of ordinary shares | Amount | |||||||
| 2024 | 2024 | |||||||
| Units | USD | |||||||
| Issued and fully paid: | ||||||||
| Ordinary shares | ||||||||
| As at 11 April 2024 (date of incorporation)/31 May | * | |||||||
The Company was incorporated on 11 April 2024 with an issued and paid up share capital of USD consisting of ordinary shares.
| * |
| 6. | Other payable |
| 2024 | ||||
| USD | ||||
| Other payable | ||||
| Accruals | ||||
| F-125 |
| 7. | Loss before tax |
Loss before tax for the financial period is arrived at after charging:
11.04.2024 to 31.5.2024 | ||||
| (2 months) | ||||
| USD | ||||
| Auditors’ remuneration | ||||
| Incorporation expenses | ||||
| 8. | Financial instrument |
The following table analyses the financial assets and liabilities in the statement of financial position by the class of financial instruments to which they are assigned, and therefore by the measurement basis:
| 2024 | ||||
| USD | ||||
| Financial liabilities | ||||
| At amortised cost | ||||
| Other payable and accrued expense | ||||
Financial risk management objectives and policies
The Company is exposed to financial risk arising from its operations and the use of financial instruments. The key financial risk is liquidity risk.
The following sections provide details regarding the Company’s exposure to the above-mentioned financial risks and the objectives, policies and processes for the management of the risk.
Liquidity risk
Liquidity risk is the risk that the Company encounters difficulty in meeting its obligations due to shortage of funds. The Company’s exposure to liquidity risk arises primarily from other payables.
The Company’s funding requirements and liquidity risk are managed with the objective of meeting business obligations on a timely basis. The Company manages liquidity risk by maintaining adequate reserves and banking facilities by continuously monitoring forecast and actual cash flows, and by matching the maturity profiles of financial assets and liabilities.
All financial liabilities of the Company are assessed as current and correspondingly, no detailed maturity analysis is deemed necessary.
Fair values of financial instruments
The carrying amounts of short-term payable and cash and cash equivalents approximate their fair value due to the relatively short-term nature of these financial instruments and insignificant impact of discounting.
| 9. | Capital management |
The Company’s objectives when managing capital are to safeguard the Company’s ability to continue as going concern in order to provide returns for shareholders and benefits for other stakeholders and to maintain an optimal capital structure to reduce the cost of capital.
In order to maintain or adjust the capital structure, the Company may adjusts the amount of dividends paid to shareholders, return capital to shareholders, issue new shares or sell assets to reduce debt.
The Company monitors capital on the basis of its business and operating requirements.
| 10. | Date of authorisation for issue |
The financial statements of the Alps Life Sciences Inc for the financial period ended May 31, 2024 were authorised for issue in accordance with a resolution of the Board of Directors on November 4, 2024.
| F-126 |
ANNEX A
AMENDED AND RESTATED MERGER AGREEMENT
dated as of
May 20, 2024
by and among
Globalink Investment Inc.,
as Parent,
GL Sponsor LLC,
in the capacity as the Parent Representative,
Alps Global Holding Pubco,
as Pubco,
Alps Biosciences Merger Sub,
as Merger Sub
Alps Life Sciences Inc,
as Alps Holdco
and
Dr. Tham Seng Kong,
in the capacity as the Seller Representative
TABLE OF CONTENTS
| Page | |||
| ARTICLE I DEFINITIONS | 2 | ||
| 1.1 | Definitions | 2 | |
| 1.2 | Construction | 17 | |
| ARTICLE II REINCORPORATION MERGER | 18 | ||
| 2.1 | Reincorporation Merger | 18 | |
| 2.2 | Reincorporation Merger Effective Time | 19 | |
| 2.3 | Effect of Reincorporation Merger | 19 | |
| 2.4 | Charter Documents | 19 | |
| 2.5 | Directors and Officers of the Reincorporation Merger Surviving Company | 19 | |
| 2.6 | Effect on Issued Securities of Parent | 19 | |
| 2.7 | Surrender of Parent Common Stock | 21 | |
| 2.8 | Lost, Stolen or Destroyed Certificates | 21 | |
| 2.9 | Reincorporation Intended Tax Treatment | 21 | |
| 2.10 | Taking of Necessary Action; Further Action | 21 | |
| ARTICLE III MERGER | 21 | ||
| 3.1 | Merger | 21 | |
| 3.2 | Merger Effective Time | 22 | |
| 3.3 | Effect of the Merger | 22 | |
| 3.4 | Tax Opinion | 22 | |
| 3.5 | Memorandum and Articles of Association | 22 | |
| 3.6 | Closing | 22 | |
| 3.7 | Directors and Officers of Surviving Company | 22 | |
| 3.8 | Directors of Pubco | 23 | |
| 3.9 | Taking of Necessary Action; Further Action | 23 | |
| 3.10 | No Further Ownership Rights in Alps Holdco Ordinary Shares | 23 | |
| ARTICLE IV EFFECT OF THE MERGER | 23 | ||
| 4.1 | Effect of the Merger on Alps Holdco Ordinary Shares and Merger Sub Ordinary Shares | 23 | |
| 4.2 | Dissenting Shares | 24 | |
| 4.3 | Surrender and Payment | 25 | |
| 4.4 | Consideration Spreadsheet | 25 | |
| 4.5 | Earn-out Shares | 26 | |
| 4.6 | Adjustment | 27 | |
| 4.7 | No Fractional Shares | 27 | |
| 4.8 | Withholding | 27 | |
| 4.9 | Lost, Stolen or Destroyed Certificates | 27 | |
| 4.10 | Escrow | 28 | |
| ARTICLE V REPRESENTATIONS AND WARRANTIES OF ALPS HOLDCO, PUBCO AND MERGER SUB | 28 | ||
| 5.1 | Corporate Existence and Power | 29 | |
| 5.2 | Authorization | 29 | |
| i |
TABLE OF CONTENTS CONTINUED
| Page | |||
| 5.3 | Governmental Authorization | 30 | |
| 5.4 | Non-Contravention | 31 | |
| 5.5 | Capitalization | 31 | |
| 5.6 | Corporate Records | 32 | |
| 5.7 | Subsidiaries | 32 | |
| 5.8 | Consents | 33 | |
| 5.9 | Financial Statements | 33 | |
| 5.10 | Books and Records | 33 | |
| 5.11 | Internal Accounting Controls | 33 | |
| 5.12 | Absence of Certain Changes | 34 | |
| 5.13 | Properties; Title to Alps Holdco’s Assets | 34 | |
| 5.14 | Litigation | 34 | |
| 5.15 | Contracts | 34 | |
| 5.16 | Licenses and Permits | 36 | |
| 5.17 | Compliance with Laws | 37 | |
| 5.18 | Intellectual Property | 37 | |
| 5.19 | Accounts Payable; Affiliate Loans | 40 | |
| 5.20 | Employees; Employment Matters | 40 | |
| 5.21 | Withholding | 42 | |
| 5.22 | Employee Benefits | 43 | |
| 5.23 | Real Property | 43 | |
| 5.24 | Tax Matters | 44 | |
| 5.25 | Environmental Laws | 45 | |
| 5.26 | Finders’ Fees | 45 | |
| 5.27 | Powers of Attorney, Suretyships and Bank Accounts | 45 | |
| 5.28 | Directors and Officers | 45 | |
| 5.29 | Anti-Money Laundering Laws | 46 | |
| 5.30 | Insurance | 47 | |
| 5.31 | Related Party Transactions | 47 | |
| 5.32 | Information Supplied | 47 | |
| 5.33 | Derogation | 47 | |
| 5.34 | Disclosure Letter | 47 | |
| ARTICLE VI REPRESENTATIONS AND WARRANTIES OF PARENT | 48 | ||
| 6.1 | Corporate Existence and Power | 48 | |
| 6.2 | Corporate Authorization | 48 | |
| 6.3 | Governmental Authorization | 48 | |
| 6.4 | Non-Contravention | 49 | |
| 6.5 | Finders’ Fees | 49 | |
| 6.6 | Issuance of Shares | 49 | |
| 6.7 | Capitalization | 49 | |
| 6.8 | Information Supplied | 49 | |
| 6.9 | Trust Fund | 50 | |
| 6.10 | Listing | 50 | |
| 6.11 | Board Approval | 50 | |
| 6.12 | Parent SEC Documents and Financial Statements | 50 | |
| 6.13 | Certain Business Practices | 51 | |
| 6.14 | Anti-Money Laundering Laws | 51 | |
| 6.15 | Compliance with Laws | 52 | |
| 6.16 | Affiliate Transactions | 52 | |
| ii |
TABLE OF CONTENTS CONTINUED
| Page | |||
| 6.17 | Litigation; Permits | 52 | |
| 6.18 | Indebtedness | 52 | |
| 6.19 | Absence of Certain Changes | 52 | |
| 6.20 | Employees and Employee Benefit Plans | 52 | |
| 6.21 | Properties | 52 | |
| 6.22 | Parent Material Contracts | 52 | |
| 6.23 | Insurance | 53 | |
| 6.24 | Investment Company Act | 53 | |
| 6.25 | Tax Matters | 53 | |
| 6.26 | PIPE Financing | 54 | |
| 6.27 | CFIUS Foreign Person Status | 55 | |
| ARTICLE VII COVENANTS OF THE PARTIES PENDING CLOSING | 55 | ||
| 7.1 | Conduct of the Business | 55 | |
| 7.2 | Exclusivity | 58 | |
| 7.3 | Access to Information | 58 | |
| 7.4 | Notices of Certain Events | 59 | |
| 7.5 | Cooperation with Form F-4/Proxy Statement; Other Filings | 59 | |
| 7.6 | Trust Account | 62 | |
| 7.7 | Obligations of Merger Sub and Pubco | 62 | |
| 7.8 | Cooperation with Antitrust Law Approvals | 62 | |
| 7.9 | Subscription Agreements | 63 | |
| ARTICLE VIII COVENANTS OF ALPS HOLDCO | 63 | ||
| 8.1 | No Insider Trading | 63 | |
| 8.2 | Alps Holdco’s Shareholders Approval | 63 | |
| 8.3 | Additional Financial Information | 64 | |
| 8.4 | Lock-Up Agreements | 64 | |
| ARTICLE IX COVENANTS OF ALL PARTIES HERETO | 64 | ||
| 9.1 | Commercially Reasonable Efforts; Further Assurances; Governmental Consents | 64 | |
| 9.2 | Compliance with SPAC Agreements | 66 | |
| 9.3 | Confidentiality | 66 | |
| 9.4 | Directors’ and Officers’ Indemnification and Liability Insurance | 66 | |
| 9.5 | Parent Public Filings; NASDAQ | 67 | |
| 9.6 | Certain Tax Matters | 67 | |
| 9.7 | [Intentionally Omitted.] | 67 | |
| 9.8 | Parent Expenses | 67 | |
| ARTICLE X CONDITIONS TO CLOSING | 67 | ||
| 10.1 | Condition to the Obligations of the Parties | 67 | |
| 10.2 | Conditions to Obligations of Parent | 68 | |
| 10.3 | Conditions to Obligations of Alps Holdco | 70 | |
| iii |
TABLE OF CONTENTS CONTINUED
| Page | |||
| ARTICLE XI TERMINATION | 70 | ||
| 11.1 | Termination Without Default | 70 | |
| 11.2 | Termination Upon Default | 71 | |
| 11.3 | Effect of Termination | 72 | |
| ARTICLE XII INDEMNIFICATION | 72 | ||
| 12.1 | Indemnification of Parent | 72 | |
| 12.2 | Procedure | 72 | |
| 12.3 | Insurance | 74 | |
| 12.4 | Survival of Indemnification Rights | 74 | |
| 12.5 | Sole and Exclusive Remedy | 74 | |
| ARTICLE XIII MISCELLANEOUS | 75 | ||
| 13.1 | Notices | 75 | |
| 13.2 | Amendments; No Waivers; Remedies | 76 | |
| 13.3 | Arm’s Length Bargaining; No Presumption Against Drafter | 77 | |
| 13.4 | Publicity | 77 | |
| 13.5 | Expenses | 77 | |
| 13.6 | No Assignment or Delegation | 77 | |
| 13.7 | Governing Law | 77 | |
| 13.8 | Counterparts; Facsimile Signatures | 77 | |
| 13.9 | Entire Agreement | 77 | |
| 13.10 | Severability | 78 | |
| 13.11 | Further Assurances | 78 | |
| 13.12 | Third Party Beneficiaries | 78 | |
| 13.13 | Waiver | 78 | |
| 13.14 | No Other Representations; No Reliance | 79 | |
| 13.15 | Waiver of Jury Trial | 80 | |
| 13.16 | Submission to Jurisdiction | 81 | |
| 13.17 | Arbitration | 81 | |
| 13.18 | Remedies | 82 | |
| 13.19 | Non-Recourse | 82 | |
| 13.20 | Parent Representative | 82 | |
| 13.21 | Seller Representative | 84 | |
| Exhibit A – | Form of Alps Holdco Support Agreement |
| Exhibit B – | Form of Parent Support Agreement |
| Exhibit C – | Form of Subscription Agreement |
| Exhibit D – | Form of Lock-Up Agreement |
| Exhibit E – | Form of Amended and Restated Registration Rights Agreement |
| Exhibit F – | Form of New Pubco M&A |
| iv |
AMENDED AND RESTATED MERGER AGREEMENT
This AMENDED AND RESTATED MERGER AGREEMENT dated as of May 20, 2024 (this “Agreement”), by and among (i) Alps Global Holding Pubco, a Cayman Islands exempted company (“Pubco”); (ii) Alps Biosciences Merger Sub, a Cayman Islands exempted company and a wholly owned subsidiary of Pubco (“Merger Sub”); (iii) Alps Life Sciences Inc, a Cayman Islands exempted company (“Alps Holdco”); (iv) Globalink Investment Inc., a Delaware corporation (“Parent”), (v) GL Sponsor LLC, a Delaware limited liability company, in the capacity as the representative from and after the Effective Time (as defined below) other than Alps Holdco Shareholders (as defined below) as of immediately prior to the Effective Time and their successors and assignees) in accordance with the terms and conditions of this Agreement (the “Parent Representative”), and (vi) Dr. Tham Seng Kong, an individual, in the capacity as the representative from and after the Effective time for Alps Holdco Shareholders (as defined below) as of immediately prior to the Effective Time in accordance with the terms and conditions of this Agreement (the “Seller Representative”).
W I T N E S S E T H:
A. Parent, Alps Global Holding Berhad, a company formed under the laws of Malaysia (“Alps Malaysia”), the Parent Representative and the Seller Representative previously entered into a Merger Agreement, dated January 30, 2024 (“Principal Agreement”), which they desire to amend and restate in accordance with the terms of this Agreement. With effect from the date of this Agreement, the Principal Agreement shall be amended and superseded by this Agreement;
B. Alps Holdco and its Subsidiaries (collectively, the “Alps Holdco Group”) are a fully-integrated biotechnology research, medical and wellness services group of companies (such business as conducted or currently proposed to be conducted by the Alps Holdco Group, the “Business”);
C. Parent is a blank check company formed for the sole purpose of entering into a share exchange, asset acquisition, share purchase, recapitalization, reorganization or other similar business combination with one or more businesses or entities;
D. PubCo is an entity newly formed for the purposes of the merger of Parent with and into Pubco (the “Reincorporation Merger”), in which Pubco will be the surviving company, in accordance with this Agreement, the Reincorporation Plan of Merger, the Companies Act, and a certificate of merger to be filed with the Secretary of State of the State of Delaware, in accordance with the relevant provisions of the Delaware General Corporation Law (the “DGCL”);
E. Concurrently therewith or immediately after the Reincorporation Merger, Merger Sub will merge with and into Alps Holdco (the “Merger”, the Reincorporation Merger and the Merger are collectively referred to as the “Mergers”), with Alps Holdco being the surviving company (the “Surviving Company”) and a wholly-owned subsidiary of Pubco, in accordance with this Agreement, the Plan of Merger and the Companies Act;
F. As promptly as practicable following the execution of, and as a condition and an inducement to Parent and Alps Holdco entering into this Agreement, certain Alps Holdco Shareholders have entered into and delivered Support Agreements, substantially in the form attached hereto as Exhibit A (each, a “Alps Holdco Support Agreement”), pursuant to which such Alps Holdco Shareholders have agreed to vote in favor of this Agreement and the Mergers and the other transactions contemplated hereby;
G. As promptly as practicable following the execution of, and as a condition and an inducement to Parent and Alps Holdco entering into this Agreement, Sponsor and certain other stockholders of Parent that are officers or directors of Parent have entered into and delivered Support Agreements (collectively, the “Identified Parent Stockholders”), substantially in the form attached hereto as Exhibit B (each, a “Parent Support Agreement”), pursuant to which each such Identified Parent Stockholder has agreed (i) not to transfer or redeem any shares of Parent Common Stock held by such Identified Parent Stockholder, and (ii) to vote in favor of this Agreement and the Mergers at the Parent Stockholder Meeting;
H. Subsequent to the execution of, and as a condition and an inducement to Parent and Alps Holdco to consummate the Mergers, Parent will enter into subscription agreements in the form attached as Exhibit C hereto (each, a “Subscription Agreement”) with certain investors (the “PIPE Investors”) for an aggregate investment to be determined (the “PIPE Financing Amount”) in shares of Parent Common Stock (the “PIPE Shares”) in a private placement to be consummated substantially concurrently with the Closing (the “PIPE Financing”);
I. For U.S. federal income tax purposes, the parties hereto intend that the Reincorporation Merger will qualify as a reorganization within the meaning of Section 368(a) of the Code and Alps Holdco’s Board of Directors and the Boards of Directors of Parent and Pubco have approved this Agreement and intend that it constitute a plan of reorganization within the meaning of Treasury Regulation Section 1.368-2(g); and
J. The boards of directors of each of Alps Holdco, Parent, Pubco and Merger Sub have (i) approved and declared advisable this Agreement and the transactions contemplated by this Agreement and the Additional Agreements to which they are or will be party, including the Mergers, and the performance of their respective obligations hereunder or thereunder, on the terms and subject to the conditions set forth herein or therein, (ii) determined that this Agreement and such transactions are fair to, and in the best interests of, them and their respective stockholders or shareholders, as applicable, and (iii) resolved to recommend that their stockholders or shareholders, as applicable, approve the Mergers and such other transactions and adopt this Agreement and the Additional Agreements to which they are or will be a party and the performance of such party of its obligations hereunder and thereunder.
K. As at the date of this Agreement, the shares of Alps Malaysia have not been transferred to Alps Holdco. After the date of this Agreement and prior to the Closing, Alps Malaysia shall become a wholly-owned Subsidiary of Alps Holdco (the “Alps Restructuring”).
In consideration of the foregoing and the mutual covenants and promises set forth in this Agreement, and other good and valuable consideration, the parties hereto hereby amend and restate the Principal Agreement in its entirety and agree as follows:
ARTICLE I
DEFINITIONS
1.1 Definitions.
“AAA” has the meaning set forth in Section 13.17.
“AAA Procedures” has the meaning set forth in Section 13.17.
“Action” means any legal action, litigation, suit, claim, hearing, proceeding or investigation, including any audit, claim or assessment for Taxes or otherwise, by or before any Authority.
| 2 |
“Additional Agreements” means the Amended and Restated Registration Rights Agreement, Alps Holdco Support Agreements, the Subscription Agreements, the Escrow Agreement, the Parent Support Agreements and the Lock-Up Agreements.
“Additional Parent SEC Documents” has the meaning set forth in Section 6.12(a).
“Affiliate” means, with respect to any Person, any other Person directly or indirectly Controlling, Controlled by or under common Control with such Person.
“Agreement” has the meaning set forth in the Preamble.
“Alps Holdco” has the meaning set forth in the Preamble.
“Alps Holdco Ordinary Shares” means the ordinary shares of par value US$0.00001 per share in the capital of Alps Holdco.
“Alps Holdco Consent” has the meaning set forth in Section 5.8.
“Alps Holdco Exclusively Licensed IP” means all Alps Holdco Licensed IP that is exclusively licensed to or purported to be exclusively licensed to any member of the Alps Holdco Group.
“Alps Holdco Financial Statements” has the meaning set forth in Section 5.9(a).
“Alps Holdco Group” has the meaning set forth in the recitals to this Agreement.
“Alps Holdco Information Systems” has the meaning set forth in Section 5.18(k).
“Alps Holdco IP” means, collectively, all Alps Holdco Owned IP and Alps Holdco Licensed IP.
“Alps Holdco Licensed IP” means all Intellectual Property owned by a third Person and licensed to or purported to be licensed to any member of the Alps Holdco Group or that any member of the Alps Holdco Group otherwise has a right to use or purports to have a right to use.
“Alps Holdco Owned IP” means all Intellectual Property owned or purported to be owned by any member of the Alps Holdco Group, in each case, whether exclusively, jointly with another Person or otherwise.
“Alps Holdco Share Certificate” has the meaning set forth in Section 3.10.
“Alps Holdco Shareholders” means, at any given time, the holders of Alps Holdco Ordinary Shares, provided that following Closing it shall mean those persons who were holders of Alps Holdco Ordinary Shares (other than Alps Holdco or any Subsidiary of Alps Holdco) immediately prior to the Effective Time and their respective successors and assigns.
“Alps Holdco Shareholder Approval” has the meaning set forth in Section 5.2(b).
“Alps Holdco Shareholder Written Consent” has the meaning set forth in Section 8.2(a).
“Alps Holdco Shareholder Written Consent Deadline” has the meaning set forth in Section 8.2(a).
| 3 |
“Alps Holdco Support Agreement” has the meaning set forth in the recitals to this Agreement.
“Alps Holdco Surviving Representations” has the meaning set forth in Section 12.1.
“Alps Holdco Transaction Expenses” means, without duplication, the fees, costs, expenses, brokerage fees, commissions, finders’ fees and disbursements of financial advisors, investment banks, data room administrators, attorneys, accountants, consultants and other advisors and service providers in connection with the negotiation, documentation and consummation of this Agreement and the transactions contemplated hereby by the Alps Holdco Group, provided, however, that “Alps Holdco Transaction Expenses” shall not include (i) any premiums payable by Alps Holdco in connection with the “tail” policy pursuant to Section 8.4(d), (ii) the filing fee of the HSR Act filing, and (iii) any litigation expenses arising from the transactions contemplated hereby including any litigation related fees and expenses of attorneys, accountants and other.
“Alps Malaysia” has the meaning set forth in the recitals to this Agreement.
“Alps Restructuring” has the meaning set forth in the Preamble.
“Alternative Proposal” has the meaning set forth in Section 7.2(b).
“Alternative Transaction” has the meaning set forth in Section 7.2(a).
“Amended and Restated Registration Rights Agreement” means the amended and restated registration rights agreement, in substantially the form attached hereto as Exhibit E.
“Annual Financial Statements” has the meaning set forth in Section 5.9(a).
“Anti-Corruption Laws” has the meaning set forth in Section 5.29(a).
“Antitrust Laws” means any applicable Laws that are designed to prohibit, restrict or regulate actions having the purpose or effect of monopolization or restraint of trade, including the HSR Act.
“Applicable Taxes” mean such Taxes as defined in IRS Notice 2020-65 (and any corresponding Taxes under state or local tax applicable Laws).
“Applicable Wages” mean such wages as defined in IRS Notice 2020-65 (and any corresponding wages under state or local tax applicable Laws).
“Authority” means any federal, state, local or foreign government or political subdivision thereof, or any agency or instrumentality of such government or political subdivision, or any self-regulated organization or other non-governmental regulatory authority or quasi-governmental authority exercising executive, legislative, judicial, regulatory or administrative functions (to the extent that the rules, regulations or orders of such organization or authority have the force of Law), or any arbitrator, court or tribunal of competent jurisdiction.
“Balance Sheets” means the audited consolidated annual balance sheets of Alps Holdco Group as of March 31, 2023 and 2022.
“Balance Sheet Date” has the meaning set forth in Section 5.9(a).
| 4 |
“Books and Records” means all books and records, ledgers, employee records, customer lists, files, correspondence, and other records of every kind (whether written, electronic, or otherwise embodied) owned or controlled by a Person in which a Person’s assets, the business or its transactions are otherwise reflected, other than stock books and minute books.
“Business” has the meaning set forth in the recitals to this Agreement.
“Business Combination” has the meaning set forth in Section 6.11(a).
“Business Day” means any day other than a Saturday, Sunday or a legal holiday on which commercial banking institutions in New York, New York are authorized to close for business, excluding as a result of “stay at home”, “shelter-in-place”, “non-essential employee” or any other similar orders or restrictions or the closure of any physical branch locations at the direction of any governmental authority so long as the electronic funds transfer systems, including for wire transfers, of commercially banking institutions in New York, New York are generally open for use by customers on such day.
“Closing” has the meaning set forth in Section 3.6.
“Closing Date” has the meaning set forth in Section 3.6.
“COBRA” means collectively, the requirements of Sections 601 through 606 of ERISA and Section 4980B of the Code.
“Code” means the Internal Revenue Code of 1986.
“Companies Act” means the Companies Act (Revised) of the Cayman Islands.
“Confidential Information” means any information, knowledge or data concerning the businesses and affairs of the Alps Holdco Group, or any suppliers, customers or agents of the Alps Holdco Group that is not already generally available to the public, including any Intellectual Property.
“Confidentiality Agreement” means the Confidentiality Agreement dated as of October 2, 2023 by and between Alps Malaysia and Parent.
“Consideration Spreadsheet” has the meaning set forth in Section 4.4(a).
“Contracts” means all contracts, agreements, leases (including equipment leases, car leases and capital leases), licenses, Permits, commitments, client contracts, statements of work (SOWs), sales and purchase orders and similar instruments, oral or written, to which any member of the Alps Holdco Group is a party or by which any of its respective properties or assets is bound.
“Control” of a Person means the possession, directly or indirectly, of the power to direct or cause the direction of the management and policies of such Person, whether through the ownership of voting securities, by contract, or otherwise. “Controlled”, “Controlling” and “under common Control with” have correlative meanings.
“Conversion Ratio” means an amount equal to (a) the Per Share Merger Consideration Amount, divided by (b) $10.00.
| 5 |
“Copyleft Licenses” means all licenses or other Contracts to Software that requires as a condition of use, modification, or distribution of such Software that other Software or technology incorporated into, derived from, or distributed with such Software (i) be disclosed or distributed in source code form, (ii) be licensed for the purpose of making derivative works or (iii) be redistributable at no or minimal charge.
“Copyrights” has the meaning set forth in the definition of “Intellectual Property.”
“Data Protection Laws” means all applicable Laws in any applicable jurisdiction relating to the Processing, privacy, security, or protection of Personal Information, and all regulations or guidance issued thereunder.
“DGCL” has the meaning set forth in the Preamble.
“Disclosure Schedules” means the schedules, delivered by Alps Holdco to the Parent, disclosing information constituting exceptions to the representations and warranties by Alps Holdco as set forth under this Agreement, accurate as of January 30, 2024.
“Dispute” has the meaning set forth in Section 13.17.
“Dissenting Shares” has the meaning set forth in Section 4.2.
“Domain Names” has the meaning set forth in the definition of “Intellectual Property.”
“Earn-out Shares” has the meaning set forth in Section 4.5.
“Effective Time” has the meaning set forth in Section 3.2.
“Enforceability Exceptions” has the meaning set forth in Section 5.2(a).
“Environmental Laws” shall mean all applicable Laws that prohibit, regulate or control any Hazardous Material or any Hazardous Material Activity, including Environmental Quality Act 1974 and its relevant regulation, Environmental Quality (Scheduled Wastes) Regulations 2005.
“ERISA” means the Employee Retirement Income Security Act of 1974.
“ERISA Affiliate” means each entity, trade or business that is, or was at the relevant time, a member of a group described in Section 414(b), (c), (m) or (o) of the Code or Section 4001(b)(1) of ERISA that includes or included the Alps Holdco Group, or that is, or was at the relevant time, a member of the same “controlled group” as the Alps Holdco Group pursuant to Section 4001(a)(14) of ERISA.
“Escrow Account” has the meaning set forth in Section 4.10(a).
“Escrow Agent” has the meaning set forth in Section 4.10(a).
“Escrow Agreement” has the meaning set forth in Section 4.10(a).
“Escrow Amount” has the meaning set forth in Section 4.10(a).
“Escrow Property” has the meaning set forth in Section 4.10(a).
“Escrow Shares” has the meaning set forth in Section 4.10(a).
“Exchange Act” means the Securities Exchange Act of 1934, as amended.
| 6 |
“Exchange Agent” has the meaning set forth in Section 4.3(a).
“Exchange Fund” has the meaning set forth in Section 4.3(a).
“Excluded Matter” means any one or more of the following: (a) general economic or political conditions; (b) conditions generally affecting the industries in which such Person or its Subsidiaries operates; (c) any changes in financial, banking or securities markets in general, including any disruption thereof and any decline in the price of any security or any market index or any change in prevailing interest rates; (d) acts of war (whether or not declared), armed hostilities or terrorism, or the escalation or worsening thereof; (e) any action required or permitted by this Agreement or any action or omission taken by Alps Holdco or its Subsidiaries with the written consent or at the request of Parent or any action or omission taken by Parent or Merger Sub with the written consent or at the request of Alps Holdco; (f) (i) any changes in applicable Laws or accounting rules (including IFRS) or the enforcement, implementation or interpretation thereof, or (ii) in the case of Parent, new pronouncements by the SEC or other U.S. federal regulators with respect to prior accounting rules, including changes to, and the restatement of Parent’s audited financial statements as of and for the fiscal year ended December 31, 2020 or for future periods, as a result of the SEC pronouncement on April 12, 2021 relating to the accounting of warrants (the “SEC Warrant Pronouncement”); (g) the announcement, pendency or completion of the transactions contemplated by this Agreement; (h) any natural or man-made disaster, acts of God or pandemics; or (i) any failure by a party to meet any internal or published projections, forecasts or revenue or earnings predictions (it being understood that the facts or occurrences giving rise or contributing to such failure that are not otherwise excluded from the definition of a “Material Adverse Effect” may be taken into account in determining whether there has been a Material Adverse Effect); provided, however, that the exclusions provided in the foregoing clauses (a) through (d), clause (f) and clause (h) shall not apply to the extent that Parent and Merger Sub, taken as a whole, on the one hand, or the Alps Holdco Group, taken as a whole, on the other hand, is disproportionately affected by any such exclusions or any change, event or development to the extent resulting from any such exclusions relative to all other similarly situated companies that participate in the industry in which they operate.
“Expiration Date” has the meaning set forth in Section 4.10(b).
“Export Control Laws” has the meaning set forth in Section 5.29(a).
“F-4 Effective Date” has the meaning set forth in Section 7.5(c).
“Final March 31, 2024 Financial Statements” has the meaning set forth in Section 8.3.
“Foreign Corrupt Practices Act” has the meaning set forth in Section 5.17(a).
“Form F-4” has the meaning set forth in Section 7.5(a).
“Fraud Claim” means any claim based in whole or in part upon fraud, willful misconduct or intentional misrepresentation.
“Fully Diluted Alps Holdco Shares” means all Alps Holdco Ordinary Shares that are issued and outstanding immediately prior to the Effective Time.
“Hazardous Material” shall mean any material, emission, chemical, substance or waste that has been designated by any Authority to be radioactive, toxic, hazardous, a pollutant or a contaminant.
| 7 |
“Hazardous Material Activity” shall mean the transportation, transfer, recycling, storage, use, treatment, manufacture, removal, remediation, release, exposure of others to, sale, labeling, or distribution of any Hazardous Material or any product or waste containing a Hazardous Material, or product manufactured with ozone depleting substances, including any required labeling, payment of waste fees or charges (including so-called e-waste fees) and compliance with any recycling, product take-back or product content requirements.
“HSR Act” shall mean the Hart-Scott-Rodino Antitrust Improvements Act of 1976, as amended, and any rules or regulations promulgated thereunder.
“IFRS” means International Financial Reporting Standards as issued by the International Accounting Standards Board in effect from time to time applied consistently throughout the periods involved.
“Indebtedness” means with respect to any Person, (a) all obligations of such Person for borrowed money, including with respect thereto, all interests, fees and costs, (b) all obligations of such Person evidenced by bonds, debentures, notes or similar instruments, (c) all obligations of such Person under conditional sale or other title retention agreements relating to property purchased by such Person, (d) all obligations of such Person issued or assumed as the deferred purchase price of property or services (other than accounts payable to creditors for goods and services incurred in the ordinary course of business consistent with past practices), (e) all Indebtedness of others secured by (or for which the holder of such Indebtedness has an existing right, contingent or otherwise, to be secured by) any lien or security interest on property owned or acquired by such Person, whether or not the obligations secured thereby have been assumed, (f) all obligations of such Person under leases required to be accounted for as capital leases under U.S. IFRS, (g) all guarantees by such Person of the Indebtedness of another Person, (h) all liability of such Person with respect to any hedging obligations, including interest rate or currency exchange swaps, collars, caps or similar hedging obligations, (i) any obligations that Alps Holdco has elected to defer pursuant to the CARES Act or as a result of COVID-19, including any deferred rent or deferred Taxes, and any liabilities associated with any loans or other stimulus packages received by Alps Holdco under the CARES Act and applicable rules and regulations thereunder, and (j) any agreement to incur any of the same.
“Indemnification Notice” has the meaning set forth in Section 12.1.
“Indemnified Party” has the meaning set forth in Section 12.1.
“Indemnifying Parties” has the meaning set forth in Section 12.1.
“Intellectual Property” means all of the worldwide intellectual property rights and proprietary rights associated with any of the following, whether registered, unregistered or registrable, to the extent recognized in a particular jurisdiction: discoveries, inventions, ideas, technology, know-how, trade secrets, and Software, in each case whether or not patentable or copyrightable (including proprietary or confidential information, systems, methods, processes, procedures, practices, algorithms, formulae, techniques, knowledge, results, protocols, models, designs, drawings, specifications, materials, technical data or information, and other information related to the development, marketing, pricing, distribution, cost, sales and manufacturing) (collectively, “Trade Secrets”); trade names, trademarks, service marks, trade dress, product configurations, other indications of origin, registrations thereof or applications for registration therefor, together with the goodwill associated with the foregoing (collectively, “Trademarks”); patents, patent applications, utility models, industrial designs, supplementary protection certificates, and certificates of inventions, including all re-issues, continuations, divisionals, continuations-in-part, re-examinations, renewals, counterparts, extensions, and validations thereof (“collectively, “Patents”); works of authorship, copyrights, copyrightable materials, copyright registrations and applications for copyright registration (collectively, “Copyrights”); domain names and URLs (collectively, “Domain Names”), social media accounts, and other intellectual property, and all embodiments and fixations thereof and related documentation and registrations and all additions, improvements and accessions thereto.
| 8 |
“Interim Period” has the meaning set forth in Section 7.1(a).
“International Trade Control Laws” has the meaning set forth in Section 5.29(a).
“IP Contracts” means, collectively, any and all Contracts to which any member of the Alps Holdco Group is a party or by which any of its respective properties or assets is bound, in any case under which the Alps Holdco Group (i) is granted a right (including option rights, rights of first offer, first refusal, first negotiation, etc.) in or to any Intellectual Property of a third Person, (ii) grants a right (including option rights, rights of first offer, first refusal, first negotiation, etc.) to a third Person in or to any Intellectual Property owned or purported to be owned by the Alps Holdco Group or (iii) has entered into an agreement not to assert or sue with respect to any Intellectual Property (including settlement agreements and co-existence arrangements), in each case other than (A) “shrink wrap” or other licenses for generally commercially available software (including Publicly Available Software) or hosted services, (B) customer, distributor or channel partner Contracts on Alps Holdco’s standard forms, (C) Contracts with the Alps Holdco Group’s employees or contractors on Alps Holdco’s standard forms, and (D) customary non-disclosure agreements entered into in the ordinary course of business consistent with past practices (subparts (A)-(D) collectively, the “Standard Contracts”).
“IPO” means the initial public offering of Parent pursuant to the Prospectus.
“Knowledge” (i) with respect to Alps Holdco means the actual knowledge after reasonable inquiry of the Key Management of Alps Holdco; and (ii) with respect to Parent means the actual knowledge after reasonable inquiry of Say Leong Lim and Kelvin (Zeng Yenn) Chin.
“Key Management of Alps Holdco” means Dr. Tham Seng Kong and Chew Yoke Ling.
“Law” means any domestic or foreign, federal, state, municipality or local law, statute, ordinance, code, rule, or regulation.
“Letter of Transmittal” has the meaning set forth in Section 4.3(b).
“Lien” means, with respect to any property or asset, any mortgage, lien, pledge, charge, claim, security interest or encumbrance of any kind in respect of such property or asset, and any conditional sale or voting agreement or proxy, including any agreement to give any of the foregoing.
“Lock-Up Agreement” means the agreement, in substantially the form attached hereto as Exhibit D, restricting the sale, transfer or other disposition of Reincorporation Merger Surviving Company Ordinary Shares to be received by certain Alps Holdco Shareholders and the Identified Parent Stockholders at the Closing in connection with the Mergers.
“Losses” has the meaning set forth in Section 12.1.
“Material Adverse Effect” means any fact, effect, event, development, change, state of facts, condition, circumstance, violation or occurrence (an “Effect”) that, individually or together with one or more other contemporaneous Effect, (i) has or would reasonably be expected to have a materially adverse effect on the financial condition, assets, liabilities, business, results of operations or prospects of the Alps Holdco Group, on the one hand, or on Parent, on the other hand, taken as a whole; or (ii) prevents or materially impairs or would reasonably be expected to prevent or materially impair the ability of the Alps Holdco Shareholders and the Alps Holdco Group, on the one hand, or on Parent, on the other hand to consummate the Mergers and the other transactions contemplated by this Agreement in accordance with the terms and conditions of this Agreement; provided, however, that a Material Adverse Effect shall not be deemed to include Effects (and solely to the extent of such Effects) resulting from an Excluded Matter.
| 9 |
“Material Contracts” has the meaning set forth in Section 5.15(a). Material Contracts shall not include any Contracts that are also Plans.
“Memorandum and Articles of Association of Alps Holdco” means the Memorandum and Articles of Association of Alps Holdco, as may be amended and/or restated from time to time.
“Mergers” has the meaning set forth in the recitals to this Agreement.
“Merger Consideration Shares” means an aggregate number of Reincorporation Merger Surviving Company Ordinary Shares equal to the product of (i) the Conversion Ratio, multiplied by (ii) the aggregate number of issued and outstanding Alps Holdco Ordinary Shares as of the Closing; provided, however, that the Merger Consideration Shares otherwise payable to Alps Holdco Shareholders are subject to the withholding of the Escrow Shares deposited in the Escrow Account in accordance with Section 3.10, and after the Closing are subject to reduction for the indemnification obligations of the Indemnifying Parties set forth in ARTICLE XI).
“Merger Sub” has the meaning set forth in the Preamble.
“Merger Sub Shareholder Approval” has the meaning set forth in Section 7.7.
“Merger Sub Ordinary Shares” has the meaning set forth in Section 5.5(c).
“NASDAQ” means the Nasdaq Stock Market LLC.
“New Pubco M&A” has the meaning set forth in Section 2.4.
“Offer Documents” has the meaning set forth in Section 7.5(a).
“Order” means any decree, order, judgment, writ, award, injunction, stipulation, determination, award, rule or consent of or by an Authority.
“OSHA” has the meaning set forth in Section 5.20(l).
“Other Filings” means any filings to be made by Parent required under the Exchange Act, Securities Act or any other United States federal, foreign or blue-sky laws, other than the SEC Statement and the other Offer Documents.
“Outside Closing Date” has the meaning set forth in Section 11.1(a).
“Parent” has the meaning set forth in the Preamble.
“Parent Board Recommendation” has the meaning set forth in Section 6.11(a).
“Parent Closing Cash” means (a) the amount of cash available in the Trust Account immediately prior to the Effective Time after deducting the amount required to satisfy the Parent Redemption Amount plus (b) the PIPE Financing Amount actually received by Parent prior to or substantially concurrently with the Closing, net of Parent’s unpaid expenses and liabilities.
| 10 |
“Parent Common Stock” means shares of Common Stock issued by Parent.
“Parent Excluded Shares” has the meaning provided in Section 2.6(d).
“Parent Material Contract” has the meaning set forth in Section 6.22(a).
“Parent Private Unit” means each unit of Parent issued to Public Gold Marketing Sdn. Bhd., a Malaysian private limited company, in a private placement at the time of the consummation of the IPO at a price of $10.00 per Parent Private Unit comprised of (a) one share of Parent Common Stock and (b) one Parent Private Right, and (c) one Parent Private Warrant.
“Parent Private Right” means each right issued as part of the Parent Private Unit, entitling the holder thereof to receive one-tenth (1/10) of one share of Parent Common Stock upon the consummation of the Mergers.
“Parent Private Warrant” means each warrant issued as part of a Parent Private Unit, entitling the holder of one whole warrant to purchase one half (1/2) of a share of Parent Common Stock at an exercise price of $11.50 per whole share.
“Parent Proposals” has the meaning set forth in Section 7.5(e).
“Parent Public Unit” means each unit of Parent issued in the IPO comprised of (a) one share of Parent Common Stock and (b) one Parent Public Right, and (c) one Parent Public Warrant.
“Parent Public Right” means each right issued as part of the Parent Public Unit, entitling the holder thereof to receive one-tenth (1/10) of one share of Parent Common Stock upon the consummation of the Mergers.
“Parent Public Warrant” means each warrant issued as part of a Parent Public Unit or Parent Public Subunit, entitling the holder of one whole warrant to purchase one half (1/2) of a share of Parent Common Stock at an exercise price of $11.50 per whole share.
“Parent Redemption Amount” has the meaning set forth in Section 7.6.
“Parent Representative” has the meaning set forth in the Preamble.
“Parent Representative Documents” has the meaning set forth in Section 13.20(a).
“Parent SEC Documents” has the meaning set forth in Section 6.12(a).
“Parent Stockholder Approval” has the meaning set forth in Section 6.2.
“Parent Stockholder Meeting” has the meaning set forth in Section 7.5(a).
“Parent Support Agreement” has the meaning set forth in the recitals to this Agreement.
| 11 |
“Parent Transaction Expenses” means, without duplication, (a) the fees, costs, expenses, brokerage fees, commissions, finders’ fees and disbursements of financial advisors, investment banks, data room administrators, attorneys, accountants, consultants and other advisors and service providers in connection with Parent’s negotiation, documentation and consummation of this Agreement and the transactions contemplated hereby including any deferred underwriting fees incurred by Parent in connection with its initial public offering, (b) the filing fees payable to the SEC in connection with the registration of the Parent Public Units and the Merger Consideration Shares; provided, however, that “Parent Transaction Expenses” shall not include (i) the filing fee of the HSR Act filing, (ii) any litigation expenses arising from the transactions contemplated hereby including any litigation related fees and expenses of attorneys, accountants and other advisors and (iii) any fees and expenses incurred in connection with the Subscription Agreements.
“Parent Unit” shall mean each Parent Private Unit and Parent Public Unit.
“Parent Right” shall mean each Parent Private Right and Parent Public Right.
“Parent Warrant” shall mean each Parent Private Warrant and Parent Public Warrant.
“Patents” has the meaning set forth in the definition of “Intellectual Property.”
“PCAOB” means the Public Company Accounting Oversight Board.
“Pending Claims” has the meaning set forth in Section 3.10(b).
“Per Share Merger Consideration Amount” means an amount equal to (a) United States Dollar One Billion and Six Hundred Million ($1,600,000,000.00), divided by (b) the number of Fully Diluted Alps Holdco Shares.
“Permit” has the meaning set forth in Section 5.16.
“Permitted Liens” means (a) all defects, exceptions, restrictions, easements, rights of way and encumbrances disclosed in policies of title insurance which have been made available to Parent; (b) mechanics’, carriers’, workers’, repairers’ and similar statutory Liens arising or incurred in the ordinary course of business consistent with past practices for amounts (i) that are not delinquent, (ii) that are not material to the business, operations and financial condition of Alps Holdco so encumbered, either individually or in the aggregate, and (iii) not resulting from a breach, default or violation by Alps Holdco Group of any Contract or Law; (c) liens for Taxes not yet due and payable or which are being contested in good faith by appropriate proceedings (and for which adequate accruals or reserves have been established on the Financial Statements in accordance with IFRS); and (d) the Liens set forth on Schedule 1.1(b).
“Person” means an individual, corporation, partnership (including a general partnership, limited partnership or limited liability partnership), limited liability company, association, trust or other entity or organization, including a government, domestic or foreign, or political subdivision thereof, or an agency or instrumentality thereof.
“Personal Information” means any and all (i) information relating to an individual that either contains data elements that identify the individual or that can be used, directly or indirectly, to identify, contact or locate the individual, (ii) information that enables a person to contact the individual (such as information contained in a cookie or electronic device fingerprint) (iii) “personal data,” “personal information,” “nonpublic personal information” or any similar term as defined under any applicable Laws and (iv) other information, the Processing of which is regulated by an applicable Law in relation to data protection or data privacy.
“PIPE Financing” has the meaning set forth in the recitals to this Agreement.
| 12 |
“PIPE Financing Amount” has the meaning set forth in the recitals to this Agreement.
“PIPE Investors” has the meaning set forth in the recitals to this Agreement.
“PIPE Shares” has the meaning set forth in the recitals to this Agreement.
“Plan” means each “employee benefit plan” within the meaning of Section 3(3) of ERISA and all other material compensation and benefits plans, policies, programs, arrangements or payroll practices, including multiemployer plans within the meaning of Section 3(37) of ERISA, and each other stock purchase, stock option, restricted stock, severance, retention, employment (other than any employment offer letter in such form as previously provided to Parent that is terminable “at will” without any contractual obligation on the part of the Alps Holdco Group to make any severance, termination, change of control, or similar payment), consulting, change-of-control, bonus, incentive, deferred compensation, employee loan and fringe benefit plan, agreement, program, policy, commitment or other arrangement, whether or not subject to ERISA (including any related funding mechanism now in effect or required in the future), whether formal or informal, oral or written, in each case, that is sponsored, maintained, contributed or required to be contributed to by the Alps Holdco Group, or under which the Alps Holdco Group has any current or potential liability, but excluding any statutory plan, program or arrangement that is maintained by an Authority.
“Plan of Merger” has the meaning set forth in Section 3.2.
“Plans of Merger” means the Plan of Merger and the Reincorporation Plan of Merger.
“Pro Rata Share” means, with respect to each Alps Holdco Shareholder, a fraction expressed a percentage equal to (i) the portion of the Merger Consideration Shares payable by Pubco to such Alps Holdco Shareholder in accordance with the terms of this Agreement and the Plans of Merger, divided by (ii) the total Merger Consideration Shares payable by Pubco to all Alps Holdco Shareholders in accordance with the terms of this Agreement and the Plans of Merger.
“Process,” “Processed” or “Processing” means any operation or set of operations performed upon Personal Information or sets of Personal Information, whether or not by automated means, such as collection, recording, organization, structuring, storage, adaptation or alteration, retrieval, consultation, use, disclosure by transmission, dissemination, or otherwise making available, alignment or combination, restriction, erasure, or destruction.
“Prohibited Party” has the meaning set forth in Section 5.29(b).
“Prospectus” has the meaning set forth in Section 13.13.
“Proxy Statement” has the meaning set forth in Section 7.5(a).
“Pubco” has the meaning set forth in the Preamble.
“Pubco Ordinary Shares” has the meaning set forth in Section 5.5(b).
“Pubco Shareholder Approval” has the meaning set forth in Section 7.7.
“Public Distributions” has the meaning set forth in Section 13.13.
| 13 |
“Publicly Available Software” means each of any Software that contains, or is derived in any manner (in whole or in part) from, any Software that is distributed as free software, “copyleft,” open source software (e.g. Linux), or under similar licensing and distribution models, including but not limited to any of the following: (A) the GNU General Public License (GPL) or Lesser/Library GPL (LGPL), (B) the Artistic License (e.g., PERL), (C) the Mozilla Public License, (D) the Netscape Public License, (E) the Sun Community Source License (SCSL), (F) the Sun Industry Source License (SISL) and (G) the Apache Server License, including for the avoidance of doubt all Software licensed under a Copyleft License.
“Real Property” means, collectively, all real properties and interests therein (including the right to use), together with all buildings, fixtures, trade fixtures, plant and other improvements located thereon or attached thereto; all rights arising out of use thereof (including air, water, oil and mineral rights); and all subleases, franchises, licenses, permits, easements and rights-of-way which are appurtenant thereto.
“Registered Exclusively Licensed IP” means all Alps Holdco Exclusively Licensed IP that is the subject of a registration or an application for registration, including issued patents and patent applications.
“Registered IP” means collectively, all Registered Owned IP and Registered Exclusively Licensed IP.
“Registered Owned IP” means all Intellectual Property constituting Alps Holdco Owned IP or filed in the name of any member of the Alps Holdco Group, and in each instance is the subject of a registration or an application for registration, including issued patents and patent applications.
“Reincorporation Intended Tax Treatment” has the meaning provided in Section 2.9.
“Reincorporation Merger” has the meaning provided in the recitals.
“Reincorporation Merger Certificate” has the meaning provided in Section 2.2.
“Reincorporation Merger Effective Time” has the meaning provided in Section 2.2.
“Reincorporation Merger Surviving Company Ordinary Share” means an ordinary share, par value US$0.0001, in the capital of Reincorporation Merger Surviving Company.
“Reincorporation Merger Surviving Company Private Right” means each right issued at the Reincorporation Merger Effective Time, entitling the holder thereof to receive one-tenth (1/10) of one Reincorporation Merger Surviving Company Ordinary Share upon the consummation of the Mergers.
“Reincorporation Merger Surviving Company Private Warrant” means each warrant issued at the Reincorporation Merger Effective Time, entitling the holder of one whole warrant to purchase one half (1/2) of one Reincorporation Merger Surviving Company Ordinary Share at an exercise price of $11.50 per whole share.
| 14 |
“Reincorporation Merger Surviving Company Public Right” means each right issued at the Reincorporation Merger Effective Time, entitling the holder thereof to receive one-tenth (1/10) of one Reincorporation Merger Surviving Company Ordinary Share upon the consummation of the Mergers.
“Reincorporation Merger Surviving Company Public Warrant” means each warrant issued at the Reincorporation Merger Effective Time, entitling the holder thereof to purchase one half (1/2) of one Reincorporation Merger Surviving Company Ordinary Share at a purchase price of $11.50 per whole share.
“Reincorporation Merger Surviving Company Rights” means the Reincorporation Merger Surviving Company Private Rights and Reincorporation Merger Surviving Company Public Rights.
“Reincorporation Merger Surviving Company Warrants” means the Reincorporation Merger Surviving Company Private Warrants and Reincorporation Merger Surviving Company Public Warrants.
“Reincorporation Merger Surviving Company Share Price” means an amount equal to the VWAP of the Reincorporation Merger Surviving Company Ordinary Shares over the twenty (20) Trading Days ending at the close of business on the principal securities exchange or securities market on which the Reincorporation Merger Surviving Company Ordinary Shares are then traded immediately prior to the date of determination, as equitably adjusted for share splits and sub-divisions, share dividends and bonus issues of shares, share combinations and consolidations, recapitalizations and the like after the Closing Date.
“Reincorporation Merger Surviving Company” has the meaning provided in Section 2.1.
“Reincorporation Plan of Merger” has the meaning set forth in Section 2.2.
“Representatives” means a party’s officers, directors, Affiliates, managers, consultant, employees, representatives and agents.
“Resolution Period” has the meaning set forth in Section 13.17.
“Revenue” shall mean the consolidated revenue of Parent and the Alps Holdco Group that is attributable to: (A) sales by the Parent and Alps Holdco Group (present and future); and (B) revenue from any joint venture or entity in which the Parent and/or any Alps Holdco Group has made an equity investment to the extent (and in the amount) that such revenue can be reported as revenue by the aforementioned entities in accordance with US generally accepted accounting principles.
“Sanctions Laws” has the meaning set forth in Section 5.29(a).
“Sarbanes-Oxley Act” means the Sarbanes-Oxley Act of 2002.
“SEC” means the Securities and Exchange Commission.
“SEC Statement” means the Form F-4, including the Proxy Statement, whether in preliminary or definitive form, and any amendments or supplements thereto.
“Securities Act” means the Securities Act of 1933, as amended.
“Securities Filing” has the meaning set forth in Section 3.4(b).
“Seller Representative” has the meaning set forth in the Preamble.
“Seller Representative Documents” has the meaning set forth in Section 13.21(a).
“Software” means computer software, programs, and databases (including development tools, library functions, and compilers) in any form, including in or as Internet Web sites, web content, links, source code, object code, operating systems, database management code, utilities, graphical user interfaces, menus, images, icons, forms, methods of processing, software engines, platforms, and data formats, together with all versions, updates, corrections, enhancements and modifications thereof, and all related specifications, documentation, developer notes, comments, and annotations.
| 15 |
“Sponsor” means GL Sponsor LLC, a Delaware limited liability company.
“Standard Contracts” has the meaning set forth in the definition of IP Contracts.
“Subscription Agreement” has the meaning set forth in the recitals to this Agreement.
“Subsidiary” means, with respect to any Person, each entity of which at least fifty percent (50%) of the capital stock or other equity or voting securities are Controlled or owned, directly or indirectly, by such Person.
“Survival Period” has the meaning set forth in Section 12.1.
“Surviving Company” has the meaning set forth in the recitals to this Agreement.
“Tangible Personal Property” means all tangible personal property and interests therein, including machinery, computers and accessories, furniture, office equipment, communications equipment, automobiles, laboratory equipment and other equipment owned or leased by the Alps Holdco Group and other tangible property.
“Tax Opinion” has the meaning set forth in Section 3.4.
“Tax Return” means any return, information return, declaration, claim for refund or credit, report or any similar statement, and any amendment thereto, including any attached schedule and supporting information, whether on a separate, consolidated, combined, unitary or other basis, that is filed or required to be filed with any Taxing Authority in connection with the determination, assessment, collection or payment of a Tax or the administration of any Law relating to any Tax.
“Tax(es)” means any U.S. federal, state or local or non-U.S. tax, charge, fee, levy, custom, duty, deficiency, or other assessment of any kind or nature imposed by any Taxing Authority (including any income (net or gross), gross receipts, profits, windfall profit, sales, use, goods and services, ad valorem, franchise, license, withholding, employment, social security, workers compensation, unemployment compensation, employment, payroll, transfer, excise, import, real property, personal property, intangible property, occupancy, recording, minimum, alternative minimum), together with any interest, penalty, additions to tax or additional amount imposed with respect thereto.
“Taxing Authority” means the Internal Revenue Service and any other Authority responsible for the collection, assessment or imposition of any Tax or the administration of any Law relating to any Tax.
“Third-Party Claim” has the meaning set forth in Section 12.1.
“Trade Secrets” has the meaning set forth in the definition of “Intellectual Property.”
“Trademarks” has the meaning set forth in the definition of “Intellectual Property.”
“Trading Day” means any day on which shares of Parent Common Stock are actually traded on the principal securities exchange or securities market on which the Parent Common Stock is then traded.
“Transaction Litigation” has the meaning set forth in Section 9.1(c).
“Trust Account” has the meaning set forth in Section 6.9.
| 16 |
“Trust Agreement” has the meaning set forth in Section 6.9.
“Trust Fund” has the meaning set forth in Section 6.9.
“Trustee” has the meaning set forth in Section 6.9.
“U.S. GAAP” means U.S. generally accepted accounting principles, consistently applied.
“USPTO” has the meaning set forth in Section 5.18(c).
“VWAP” means, for any security as of any date(s), the dollar volume-weighted average price for such security on the principal securities exchange or securities market on which such security is then traded during the period beginning at 9:30:01 a.m., New York time, and ending at 4:00:00 p.m., New York time, as reported by Bloomberg through its “HP” function (set to weighted average) or, if the foregoing does not apply, the dollar volume-weighted average price of such security in the over-the-counter market on the electronic bulletin board for such security during the period beginning at 9:30:01 a.m., New York time, and ending at 4:00:00 p.m., New York time, as reported by Bloomberg, or, if no dollar volume-weighted average price is reported for such security by Bloomberg for such hours, the average of the highest closing bid price and the lowest closing ask price of any of the market makers for such security as reported by OTC Markets Group Inc. If the VWAP cannot be calculated for such security on such date(s) on any of the foregoing bases, the VWAP of such security on such date(s) shall be the fair market value as determined reasonably and in good faith by a majority of the disinterested independent directors of the board of directors (or equivalent governing body) of the applicable issuer. All such determinations shall be appropriately adjusted for any stock dividend, stock split, stock combination, recapitalization or other similar transaction during such period.
1.2 Construction.
(a) References to particular sections and subsections, schedules, and exhibits not otherwise specified are cross-references to sections and subsections, schedules, and exhibits of this Agreement. Captions are not a part of this Agreement, but are included for convenience, only.
(b) References to “US$” or “$” are to the lawful currency of the United States of America.
(c) The words “herein,” “hereof,” “hereunder,” and words of similar import refer to this Agreement as a whole and not to any particular provision of this Agreement; and, unless the context requires otherwise, “party” means a party signatory hereto.
(d) Any use of the singular or plural, or the masculine, feminine or neuter gender, includes the others, unless the context otherwise requires; the word “including” means “including without limitation”; the word “or” means “and/or”; the word “any” means “any one, more than one, or all”; and, unless otherwise specified, any financial or accounting term has the meaning of the term under IFRS as consistently applied heretofore by Alps Holdco. Any reference in this Agreement to a Person’s directors shall include any member of such Person’s governing body and any reference in this Agreement to a Person’s officers shall include any Person filling a substantially similar position for such Person. Any reference in this Agreement or any Additional Agreement to a Person’s shareholders or stockholders shall include any applicable owners of the equity interests of such Person, in whatever form.
| 17 |
(e) Unless otherwise specified, any reference to any agreement (including this Agreement), instrument, or other document includes all schedules, exhibits, or other attachments referred to therein, and any reference to a statute or other law means such law as amended, restated, supplemented or otherwise modified from time to time and includes any rule, regulation, ordinance or the like promulgated thereunder, in each case, as amended, restated, supplemented or otherwise modified from time to time.
(f) Any reference to a numbered schedule means the same-numbered section of the Disclosure Schedules. Any reference in a schedule contained in the Disclosure Schedules delivered by a party hereunder shall be deemed to be an exception to (or, as applicable, a disclosure for purposes of) the applicable representations and warranties (or applicable covenants) that are contained in the section or subsection of this Agreement that corresponds to such schedule and any other representations and warranties of such party that are contained in this Agreement to which the relevance of such item thereto is reasonably apparent on its face. The mere inclusion of an item in a schedule as an exception to (or, as applicable, a disclosure for purposes of) a representation or warranty shall not be deemed an admission that such item represents a material exception or material fact, event or circumstance or that such item would have a Material Adverse Effect or establish any standard of materiality to define further the meaning of such terms for purposes of this Agreement. Nothing in the Disclosure Schedules constitutes an admission of any liability or obligation of the disclosing party to any third party or an admission to any third party, including any Authority, against the interest of the disclosing party, including any possible breach of violation of any Contract or Law. Summaries of any written document in the Disclosure Schedules do not purport to be complete and are qualified in their entirety by the written document itself. The disclosures schedules and the information and disclosures contained therein are intended only to qualify and limit the representations and warranties of the parties contained in this Agreement and shall not be deemed to expand in any way the scope or effect of any of such representations and warranties.
(g) If any action is required to be taken or notice is required to be given within a specified number of days following a specific date or event, the day of such date or event is not counted in determining the last day for such action or notice. If any action is required to be taken or notice is required to be given on or before a particular day which is not a Business Day, such action or notice shall be considered timely if it is taken or given on or before the next Business Day.
(h) To the extent that any Contract, document, certificate or instrument is represented and warranted to by Alps Holdco to be given, delivered, provided or made available by Alps Holdco, such Contract, document, certificate or instrument shall be deemed to have been given, delivered, provided and made available to Parent or its Representatives, if such Contract, document, certificate or instrument shall have been posted not later than two (2) Business Days prior to the date of this Agreement to the electronic data site maintained on behalf of Alps Holdco for the benefit of Parent and its Representatives and Parent and its Representatives have been given access to the electronic folders containing such information and received a notice that such Contract, document, certificate or instrument has been so posted.
ARTICLE II
REINCORPORATION MERGER
2.1 Reincorporation Merger. At the Reincorporation Merger Effective Time, and subject to and upon the terms and conditions of this Agreement and the Reincorporation Plan of Merger, and in accordance with the applicable provisions of the Companies Act and the DGCL, respectively, Parent shall be merged with and into Pubco, the separate corporate existence of Parent shall cease and Pubco shall continue as the surviving company in the Reincorporation Merger. Pubco as the surviving company after the Reincorporation Merger is hereinafter sometimes referred to as the “Reincorporation Merger Surviving Company”.
| 18 |
2.2 Reincorporation Merger Effective Time. On a date no later than three (3) Business Days after the satisfaction or waiver of all the conditions set forth in ARTICLE X that are required to be satisfied prior to the Closing Date, the Parties hereto shall cause the Reincorporation Merger to be consummated by filing a certificate of merger (the “Reincorporation Merger Certificate”) with the Secretary of State of the State of Delaware, in accordance with the relevant provisions of the DGCL, and executing and filing a plan of merger (the “Reincorporation Plan of Merger”) and all other documents required by the Companies Act with the Registrar of Companies of the Cayman Islands (the “Cayman Registrar”), in accordance with the relevant provisions of the Companies Act. The effective time of the Reincorporation Merger shall be the later of the acceptance of the Reincorporation Merger Certificate and the date that the Reincorporation Plan of Merger is registered by the Cayman Registrar in accordance with the Companies Act, or such other time as specified in or otherwise in accordance with the Reincorporation Merger Certificate, the DGCL, the Reincorporation Plan of Merger and the Companies Act (such time is herein referred to as the “Reincorporation Merger Effective Time”.)
2.3 Effect of Reincorporation Merger. At the Reincorporation Merger Effective Time, the effect of Reincorporation Merger shall be as provided in this Agreement, the Reincorporation Merger Certificate, the Reincorporation Plan of Merger and the applicable provisions of the DGCL and the Companies Act. Without limiting the generality of the foregoing, and subject thereto, at the Reincorporation Merger Effective Time, all the rights, the property of every description including choses in action, and the business, undertaking, goodwill, benefits, immunities and privileges of Parent and Pubco shall immediately vest in and become the rights, property, business, undertaking, goodwill, benefits, immunities and privileges of the Reincorporation Merger Surviving Company and the Reincorporation Merger Surviving Company shall be liable for and subject, in the same manner as Parent and Pubco, to all mortgages, charges or security interests, and all contracts, obligations, claims, debts, and liabilities of each of Parent and Pubco, which shall include the assumption by the Reincorporation Merger Surviving Company of any and all agreements, covenants, duties and obligations of Parent set forth in this Agreement or any other outstanding agreement to which Parent is a party to be performed after the Closing, and all securities of the Reincorporation Merger Surviving Company issued and outstanding as a result of the conversion under Section 2.6 hereof shall be listed on NASDAQ (on which the Parent Common Stock were trading prior to Reincorporation Merger).
2.4 Charter Documents. At the Reincorporation Merger Effective Time, the memorandum and articles of association of Pubco shall be amended and restated so that they read in their entirety substantially as set forth in Exhibit F annexed hereto (the “New Pubco M&A”), and as so amended and restated, shall become the memorandum and articles of association of the Reincorporation Merger Surviving Company until duly amended in accordance with the terms thereof and the Companies Act. The new name of the Reincorporation Merger Surviving Company will be “Alps Life Sciences” or such other name as provided by Alps Holdco and stated in the Reincorporation Plan of Merger and the New Pubco M&A.
2.5 Directors and Officers of the Reincorporation Merger Surviving Company. As of the Reincorporation Merger Effective Time, the Persons constituting the officers and directors of Parent prior to the Reincorporation Merger Effective Time shall continue to be the officers and directors of the Reincorporation Merger Surviving Company (and holding the same title as held at Parent) until the Effective Time.
2.6 Effect on Issued Securities of Parent.
(a) Conversion of Parent Common Stock.
(i) At the Reincorporation Merger Effective Time, each issued and outstanding share of Parent Common Stock (other than the Parent Excluded Shares and shares of Parent Common Stock redeemed pursuant to Section 8.5(f)) shall be converted automatically into the right to receive one Reincorporation Merger Surviving Company Ordinary Share. At the Reincorporation Merger Effective Time, all Parent Common Stock shall cease to be outstanding and shall automatically be canceled and retired and shall cease to exist. The holders of issued Parent Common Stock immediately prior to the Reincorporation Merger Effective Time shall cease to have any rights with respect to such Parent Common Stock, except as provided herein or by Law. Each certificate (if any) previously evidencing Parent Common Stock (other than the Parent Excluded Shares and Parent Common Stock redeemed pursuant to Section 7.5(f)) shall be exchanged for a certificate representing the same number of Reincorporation Merger Surviving Company Ordinary Shares upon the surrender of such certificate in accordance with Section 2.8.
| 19 |
(ii) Each holder of shares of Parent Common Stock (other than the Parent Excluded Shares and shares of Parent Common Stock redeemed pursuant to Section 8.5(f)) shall thereafter have the right to receive the same number of Reincorporation Merger Surviving Company Ordinary Shares only.
(b) Conversion of Parent Warrants; Treatment of Parent Units. At the Reincorporation Merger Effective Time, (i) all Parent Units will separate into their individual components of Parent Common Stock, Parent Rights and Parent Warrants, and will cease separate existence and trading and (ii) each issued and outstanding Parent Warrant shall cease to be a warrant with respect to Parent Common Stock and be assumed by the Reincorporation Merger Surviving Company and shall be converted into one Reincorporation Merger Surviving Company Warrant, and will cease separate existence and trading. Each of the Reincorporation Merger Surviving Company Warrants shall have, and be subject to, the same terms and conditions set forth in the applicable agreements governing the Parent Warrants that are outstanding immediately prior to the Reincorporation Merger Effective Time. At or prior to the Reincorporation Merger Effective Time, Pubco shall take all corporate action necessary to reserve for future issuance and shall maintain such reservation for so long as any of the Reincorporation Merger Surviving Company Warrants remain outstanding, a sufficient number of Reincorporation Merger Surviving Company Ordinary Shares for delivery upon the exercise of the Reincorporation Merger Surviving Company Warrants after the Reincorporation Merger Effective Time.
(c) Parent Right. At the Reincorporation Merger Effective Time, (i) each outstanding Parent Private Right shall, without any action on the part of the holder thereof, be converted into one Reincorporation Merger Surviving Company Private Right, and (ii) each outstanding Parent Public Right shall be converted into one Reincorporation Merger Surviving Company Public Right, in accordance with the terms of such Parent Right. At the Reincorporation Merger Effective Time, each Parent Right shall cease to be outstanding and shall automatically be cancelled and retired and shall cease to exist. The holders of certificates previously evidencing Parent Rights outstanding immediately prior to the Reincorporation Merger Effective Time shall cease to have any rights with respect to such Parent Right, except as provided herein or by applicable Law. Each certificate formerly representing Parent Right shall thereafter represent only the right to receive Reincorporation Merger Surviving Company Ordinary Shares as set forth herein.
(d) Cancellation of Parent Common Stock Owned by Parent. At the Reincorporation Merger Effective Time, if there are any shares of Parent Common Stock that are owned by Parent as treasury shares or any shares of Parent Common Stock owned by any direct or indirect wholly owned Subsidiary of Parent immediately prior to the Reincorporation Merger Effective Time (the “Parent Excluded Shares”), such shares shall be canceled and extinguished without any conversion thereof or payment therefor. In addition, as of the Reincorporation Merger Effective Time, any shares of Pubco owned by Parent immediately prior to the Reincorporation Merger Effective Time shall be automatically cancelled and extinguished without any conversion or consideration delivered in exchange therefor.
(e) No Liability. Notwithstanding anything to the contrary in this Section 2.6, none of the Reincorporation Merger Surviving Company, Parent or any other Party hereto shall be liable to any person for any amount properly paid to a public official pursuant to any applicable abandoned property, escheat or similar law.
| 20 |
2.7 Surrender of Parent Common Stock. All securities issued upon the surrender of the shares of Parent Common Stock in accordance with the terms hereof, shall be deemed to have been issued in full satisfaction of all rights pertaining to such securities, provided that any restrictions on the sale and transfer of the shares of Parent Common Stock shall also apply to the Reincorporation Merger Surviving Company Ordinary Shares so issued in conversion.
2.8 Lost, Stolen or Destroyed Certificates. In the event any of its certificates shall have been lost, stolen or destroyed, Reincorporation Merger Surviving Company shall issue in exchange for such lost, stolen or destroyed certificates or securities as the case may be, upon the affidavit of that fact by the holder thereof, such securities, as may be required pursuant to Section 2.7; provided, however, that the Reincorporation Merger Surviving Company may, in its discretion and as a condition precedent to the issuance thereof, require the owner of such lost, stolen or destroyed certificates to execute and deliver a deed of indemnity in respect of such lost, stolen or destroyed certificates in the form required by the Reincorporation Merger Surviving Company as indemnity against any claim that may be made against it with respect to the certificates alleged to have been lost, stolen or destroyed.
2.9 Reincorporation Intended Tax Treatment. For U.S. federal income Tax purposes, Parent and Pubco intend that the Reincorporation Merger will constitute a transaction that qualifies as a “reorganization” within the meaning of Section 368(a) of the Code and the Treasury Regulations promulgated thereunder to which each of Parent and Common Stock is a party under Section 368(b) of the Code (the “Reincorporation Intended Tax Treatment”). Parent and Pubco hereby (a) adopt, and the Company acknowledges, this Agreement as a “plan of reorganization” within the meaning of Treasury Regulation Section 1.368-2(g), (b) agree to file and retain such information as shall be required under Treasury Regulation Section 1.368-3, and (c) agree to file all Tax and other informational returns on a basis consistent with the Reincorporation Intended Tax Treatment. Notwithstanding the foregoing or anything else to the contrary contained in this Agreement, the parties acknowledge and agree that no party is making any representation or warranty as to the qualification of Reincorporation Merger for the Reincorporation Intended Tax Treatment or as to the effect, if any, that any transaction consummated on, after or prior to the Reincorporation Merger Effective Time has or may have on any such reorganization status. Each of the parties acknowledges and agrees that each (x) has had the opportunity to obtain independent legal and tax advice with respect to the transactions contemplated by this Agreement, and (y) is responsible for paying its own Taxes, including any adverse Tax consequences that may result if the Reincorporation Merger is determined not to qualify for the Reincorporation Intended Tax Treatment.
2.10 Taking of Necessary Action; Further Action. If, at any time after the Reincorporation Merger Effective Time, any further action is necessary or desirable to carry out the purposes of this Agreement and to vest the Reincorporation Merger Surviving Company with full right, title and possession to all rights, property, business, undertaking, goodwill, benefits, immunities and privileges of Parent and Pubco, the officers and directors of Parent and Pubco are fully authorized in the name of their respective corporations / companies or otherwise to take, and will take, all such lawful and necessary action, so long as such action is not inconsistent with this Agreement.
ARTICLE III
MERGER
3.1 Merger. At the Effective Time, and subject to and upon the terms and conditions set forth in this Agreement and the Plan of Merger, and in accordance with the applicable provisions of the Companies Act, (a) Merger Sub shall be merged with and into Alps Holdco, (b) the separate corporate existence of Merger Sub shall cease and Alps Holdco shall continue as the surviving company in the Merger, and (c) the Surviving Company shall become a wholly-owned Subsidiary of Pubco.
| 21 |
3.2 Merger Effective Time. Subject to the provisions of this Agreement, at the Closing and after the Reincorporation Merger, the Parties hereto shall cause the Merger to be consummated by executing and filing a plan of merger (the “Plan of Merger”) and all other documents required by the Companies Act with the Cayman Registrar, in accordance with the relevant provisions of the Companies Act. The effective time of the Merger shall be the date that the Plan of Merger is registered by the Cayman Registrar in accordance with the Companies Act, or such later time as specified in or otherwise in accordance with the Plan of Merger and the Companies Act (such time is herein referred to as the “Effective Time”).
3.3 Effect of the Merger. At the Effective Time, the effect of the Merger shall be as provided in this Agreement, the Certificate of Merger and the applicable provisions of the Companies Act. Without limiting the generality of the foregoing, and subject thereto, at the Effective Time, all the rights, the property of every description including choses in action, and the business, undertaking, goodwill, benefits, immunities and privileges of Alps Holdco and Merger Sub shall immediately vest in and become the rights, property, business, undertaking, goodwill, benefits, immunities and privileges of the Surviving Company and the Surviving Company shall be liable for and subject, in the same manner as Alps Holdco and Merger Sub, to all mortgages, charges or security interests, and all contracts, obligations, claims, debts, and liabilities of each of Alps Holdco and Merger Sub.
3.4 Tax Opinion. If, in connection with the preparation and filing of the Parent SEC Documents, the Additional Parent SEC Documents, the SEC Statement or any Other Filing (individually, a “Securities Filing”) or the SEC’s review thereof, the SEC requests or requires that a tax opinion (or tax opinions) with respect to the U.S. federal income tax consequences of the Merger be prepared and submitted in such connection (each, a “Tax Opinion”), (i) Alps Holdco and Parent shall each use commercially reasonable efforts to deliver to Hunter Taubman Fischer & Li LLC and a firm to be retained by Alps Holdco, in connection with any Tax Opinion rendered by such counsels, customary Tax representation letters satisfactory to such counsels, dated and executed as of the date such relevant filing shall have been declared effective by the SEC and such other date(s) as determined to be reasonably necessary by such counsels in connection with the preparation and filing of such Securities Filing, and (ii) Alps Holdco shall use commercially reasonable efforts to cause its counsel, and Parent shall use commercially reasonable efforts to cause Hunter Taubman Fischer & Li LLC, to furnish Tax Opinions, subject to customary assumptions and limitations.
3.5 Memorandum and Articles of Association. At the Effective Time, the Memorandum and Articles of Association of Alps Holdco as in effect immediately prior to the Effective Time shall, in accordance with the Plan of Merger and the Companies Act be the memorandum and articles of association of the Surviving Company until duly amended in accordance with the terms thereof and the Companies Act.
3.6 Closing. Unless this Agreement is earlier terminated in accordance with ARTICLE XI, the closing of the Merger (the “Closing”) shall take place virtually at 10:00 a.m. local time, on the second (2nd) Business Day after the consummation of the Reincorporation Merger, given satisfaction or waiver (to the extent permitted by applicable law) of the conditions set forth in ARTICLE X or at such other time, date and location as Parent and Alps Holdco agree in writing. The parties may participate in the Closing via electronic means. The date on which the Closing actually occurs is hereinafter referred to as the “Closing Date”.
3.7 Directors and Officers of Surviving Company.
(a) At the Effective Time, the initial directors of the Surviving Company shall consist of the same persons serving on Alps Holdco’s Board of Directors immediately prior to the Effective Time, and such directors shall hold office until their successors shall have been duly elected or appointed and qualified or until their earlier death, resignation, removal or other vacation of office in accordance with the Surviving Company’s memorandum and articles of association then in effect.
| 22 |
(b) At the Effective Time, the officers of Alps Holdco shall become the initial officers of the Surviving Company and shall hold office until their respective successors are duly elected or appointed and qualified, or until their earlier death, resignation, removal or other vacation of office.
3.8 Directors of Pubco. At the Effective Time, Pubco’s Board of Directors will consist of five (5) directors. Alps Holdco shall have the right to designate all directors (the majority of whom shall qualify as independent directors under the Securities Act and the NASDAQ rules). In accordance with the New Pubco M&A as in effect at the Closing, no director on Pubco’s Board of Directors may be removed without cause. At or prior to the Closing, Pubco will provide each member of Pubco’s post-Closing Board of Directors with a customary director indemnification agreement, in form and substance reasonably acceptable to the directors, to be effective upon the Closing (or if later, such director’s appointment).
3.9 Taking of Necessary Action; Further Action. If, at any time after the Closing, any further action is necessary or desirable to carry out the purposes of this Agreement and to vest the Surviving Company with full right, title and interest in, to and under, or possession of, all assets, property, rights, privileges, powers and franchises of Alps Holdco and Merger Sub, the officers and directors of the Surviving Company are fully authorized in the name and on behalf of Alps Holdco and Merger Sub, to take all lawful action necessary or desirable to accomplish such purpose or acts, so long as such action is not inconsistent with this Agreement.
3.10 No Further Ownership Rights in Alps Holdco Ordinary Shares. If, after the Effective Time, certificates formerly representing Alps Holdco Ordinary Shares (each, an “Alps Holdco Share Certificate”) are presented to the Surviving Company, subject to the terms and conditions set forth herein, they shall be cancelled and exchanged for the Merger Consideration Shares provided for, and in accordance with the procedures set forth, in ARTICLE IV.
ARTICLE IV
EFFECT OF THE MERGER
4.1 Effect of the Merger on Alps Holdco Ordinary Shares and Merger Sub Ordinary Shares. At the Effective Time, as a result of the Merger and without any action on the part of Parent, Merger Sub, Alps Holdco or the holders of any equity securities of any of them:
(a) Conversion of Alps Holdco Ordinary Shares.
(i) Each Alps Holdco Ordinary Share issued and outstanding immediately prior to the Effective Time (other than the Alps Holdco Excluded Shares) shall be converted into the right to receive a number of Reincorporation Merger Surviving Company Ordinary Shares equal to the Conversion Ratio (subject to the withholding of the Escrow Shares). For the avoidance of doubt, from the Effective Time, each such holder of Alps Holdco Ordinary Shares shall cease to have any rights with respect to Alps Holdco Ordinary Shares except the right to receive the Per Share Merger Consideration Amount as provided herein, the rights provided for in Section 4.2 and the Companies Act (if applicable) and such other rights as may be provided by Law.
(ii) If, at the Effective Time, there are any Alps Holdco Ordinary Shares that are owned by Alps Holdco as treasury shares or any Alps Holdco Ordinary Shares owned by any direct or indirect wholly owned Subsidiary of Alps Holdco immediately prior to the Effective Time (the “Alps Holdco Excluded Shares”), such shares shall be canceled and extinguished without any conversion thereof or payment therefor.
| 23 |
(iii) The register of members of Alps Holdco shall be updated promptly at the Effective Time to reflect each such cancellation, and each holder of an Alps Holdco Share Certificate previously representing any such Alps Holdco Ordinary Share shall thereafter cease to have any rights with respect to such securities, except the right to receive the Reincorporation Merger Surviving Company Ordinary Shares into which such Alps Holdco Ordinary Share shall have been converted in the Merger (if applicable), the rights provided for in Section 4.2 and the Companies Act (if applicable) and such other rights as may be provided by Law.
(b) Conversion of Merger Sub Ordinary Shares. Each Merger Sub Ordinary Share issued and outstanding immediately prior to the Effective Time shall be converted into and become one newly issued, fully paid and non-assessable ordinary share of par value US$0.00001 per share in the capital of the Surviving Company.
4.2 Dissenting Shares. Notwithstanding any provision of this Agreement to the contrary, including Section 4.1, and in accordance with the Companies Act, each Alps Holdco Ordinary Share issued and outstanding immediately prior to the Effective Time (other than an Excluded Alps Holdco Ordinary Share) and held by a holder who has validly exercised properly in writing their dissenters’ rights for such Alps Holdco Ordinary Share in accordance with Section 238 of the Companies Act, and has otherwise complied in all respects with all of the provisions of the Companies Act relevant to the exercise and perfection of dissenters’ rights (such Alps Holdco Ordinary Shares being referred to collectively as the “Dissenting Shares” and individually as a “Dissenting Share”) (i) shall not be converted into a right to receive, and the applicable holder shall have no right to receive, the applicable portion of the Merger Consideration Shares (including Escrow Shares) to which such holder would otherwise be entitled pursuant to Section 4.1, and (ii) such Dissenting Shares shall no longer be outstanding and shall automatically be cancelled and extinguished by virtue of the Merger and shall cease to exist; and such holder shall be entitled to only such rights as are granted by Section 238 of the Companies Act; provided, however, that if, after the Effective Time, such holder fails to perfect, withdraws or loses such holder’s dissenters’ rights pursuant to Section 238 of the Companies Act or if a court of competent jurisdiction shall determine that such holder is not entitled to the relief provided by Section 238 of the Companies Act, such Dissenting Shares shall no longer be deemed to be Dissenting Shares and shall be treated as if they had been converted as of the Effective Time into the right to receive the applicable portion of the Merger Consideration Shares to which such holder is entitled pursuant to the applicable subsections of Section 4.1, without interest thereon, upon surrender of the Alps Holdco Share Certificate or Alps Holdco Share Certificates representing such Dissenting Shares in accordance with Section 5.4. Each holder of Dissenting Shares who becomes entitled to payment for his, her or its Dissenting Shares pursuant to the Companies Act shall receive payment therefor from Alps Holdco in accordance with the Companies Act. Alps Holdco shall promptly provide Parent and the Parent Representative prompt written notice of any demands received by Alps Holdco for dissenters’ rights in respect of any Alps Holdco Ordinary Share, any withdrawal of any such demand and any other demand, notice or instrument delivered to Alps Holdco pursuant to the Companies Act that relates to such demand, and Parent and the Parent Representative shall have the opportunity to participate in all negotiations and proceedings with respect to such demands.
| 24 |
4.3 Surrender and Payment.
(a) Exchange Fund. On the Closing Date, Reincorporation Merger Surviving Company shall deposit, or shall cause to be deposited, with Continental Stock Transfer & Trust Company (the “Exchange Agent”) (i) for the benefit of the Alps Holdco Shareholders, for exchange in accordance with this ARTICLE IV, the number of Reincorporation Merger Surviving Company Ordinary Shares sufficient to deliver the aggregate Merger Consideration Shares (less the Escrow Shares, which will be deposited in the Escrow Account in accordance with Section 3.10) and Earn-out Shares payable pursuant to this Agreement (such Reincorporation Merger Surviving Company Ordinary Shares, the “Exchange Fund”), and (ii) for the benefit of the Parent stockholders immediately prior to the Reincorporation Merger Effective Time, for exchange in accordance with Section 2.6, such number of Reincorporation Merger Surviving Company Ordinary Shares equal to the number of issued and outstanding shares of Parent Common Stock (other than the Parent Excluded Shares and shares of Parent Common Stock redeemed pursuant to Section 8.5(f)). Reincorporation Merger Surviving Company shall cause the Exchange Agent, pursuant to irrevocable instructions, to pay the Merger Consideration Shares and Earn-out Shares out of the Exchange Fund in accordance with the Consideration Spreadsheet and the other applicable provisions contained in this Agreement. The Exchange Fund shall not be used for any other purpose other than as contemplated by this Agreement.
(b) Exchange Procedures. As soon as practicable following the Effective Time, and in any event within two (2) Business Days following the Effective Time (but in no event prior to the Effective Time), Reincorporation Merger Surviving Company shall cause the Exchange Agent to deliver to each Alps Holdco Shareholder, as of immediately prior to the Effective Time, represented by certificate or book-entry, a letter of transmittal and instructions for use in exchanging such Alps Holdco Shareholder’s Alps Holdco Ordinary Shares for such Alps Holdco Shareholder’s Pro Rata Share of the Merger Consideration Shares (less the Escrow Shares) and Earn-out Shares from the Exchange Fund, and which shall be in form and contain provisions which Reincorporation Merger Surviving Company may specify and which are reasonably acceptable to Alps Holdco) (a “Letter of Transmittal”), and promptly following receipt of a Alps Holdco Shareholder’s properly executed Letter of Transmittal, update the Reincorporation Merger Surviving Company’s register of members accordingly and deliver such Alps Holdco Shareholder’s Pro Rata Share of the Merger Consideration Shares and Earn-out Shares to such Alps Holdco Shareholder.
(c) Termination of Exchange Fund. Any portion of the Exchange Fund relating to the Merger Consideration Shares and Earn-out Shares and any Escrowed Property disbursed to the Escrow Agent in accordance with the Escrow Agreement that remains undistributed to the Alps Holdco Shareholders for two (2) years after the Effective Time shall be delivered to Reincorporation Merger Surviving Company, upon demand, and any Alps Holdco Shareholders who have not theretofore complied with this Section 4.3 shall thereafter look only to Reincorporation Merger Surviving Company for their portion of the Merger Consideration Shares and Earn-out Shares. Any portion of the Exchange Fund remaining unclaimed by Alps Holdco Shareholders as of a date which is immediately prior to such time as such amounts would otherwise escheat to or become property of any Authority shall, to the extent permitted by applicable Law, become the property of Reincorporation Merger Surviving Company free and clear of any claims or interest of any person previously entitled thereto.
4.4 Consideration Spreadsheet.
(a) At least three (3) Business Days prior to the Closing, Alps Holdco shall deliver to Parent a spreadsheet (the “Consideration Spreadsheet”), prepared by Alps Holdco in good faith and detailing the following, in each case, as of immediately prior to the Effective Time:
(i) the name and address of record of each Alps Holdco Shareholder and the number of Alps Holdco Ordinary Shares held by each;
(ii) the number of Fully Diluted Alps Holdco Shares;
| 25 |
(iii) detailed calculations of each of the following (in each case, determined without regard to withholding):
(A) the Per Share Merger Consideration Amount;
(B) the Conversion Ratio;
(C) the Merger Consideration Shares; and
(iv) any explanatory or supporting information, including calculations, as Parent may reasonably request.
(b) The contents of the Consideration Spreadsheet delivered by Alps Holdco hereunder shall be subject to reasonable review and comment by Parent, but Alps Holdco shall, in all events, remain solely responsible for the contents of the Consideration Spreadsheet. Under no circumstances shall Reincorporation Merger Surviving Company, Parent or Merger Sub be responsible for the calculations or the determinations regarding such calculations in the Consideration Spreadsheet and the parties agree that Reincorporation Merger Surviving Company, Parent and Merger Sub shall be entitled to rely on the Consideration Spreadsheet in making payments under ARTICLE V.
(c) Nothing contained in this Section 4.4 or in the Consideration Spreadsheet shall be construed or deemed to: (i) modify Alps Holdco’s obligations to obtain Reincorporation Merger Surviving Company’s prior consent to the issuance of any securities pursuant to Section 7.1(a)(xviii); or (ii) alter or amend the definition of Per Share Merger Consideration Amount or Merger Consideration Shares.
4.5 Earn-out Shares At the Closing, and as additional consideration for the Mergers, Reincorporation Merger Surviving Company shall issue up to an aggregate of 48,000,000 additional Reincorporation Merger Surviving Company Ordinary Shares (the “Earn-Out Shares”) to the Alps Holdco Shareholders upon the completion of certain milestones, as follows:
(a) If during the Surviving Company’s first full fiscal year following the Closing Date, the Revenue exceeds $7,000,000, the Alps Holdco Shareholders shall be issued an additional 9,600,000 Reincorporation Merger Surviving Company Ordinary Shares.
(b) If during the Surviving Company’s second full fiscal year following the Closing Date, the Revenue exceeds $14,000,000, the Alps Holdco Shareholders shall be issued an additional 9,600,000 Reincorporation Merger Surviving Company Ordinary Shares.
(c) If during the Surviving Company’s third full fiscal year following the Closing Date, the Revenue exceeds $23,000,000, the Alps Holdco Shareholders shall be issued an additional 9,600,000 Reincorporation Merger Surviving Company Ordinary Shares.
(d) If during the Surviving Company’s fourth full fiscal year following the Closing Date, the Revenue exceeds $34,000,000, the Alps Holdco Shareholders shall be issued an additional 9,600,000 Reincorporation Merger Surviving Company Ordinary Shares.
(e) If during the Surviving Company’s fifth full fiscal year following the Closing Date, the Revenue exceeds $56,000,000, the Alps Holdco Shareholders shall be issued an additional 9,600,000 Reincorporation Merger Surviving Company Ordinary Shares.
| 26 |
In the event the Surviving Company fails to achieve the Revenue target as set out above for any particular fiscal year (“Relevant Fiscal Year”), the actual Revenue achieved in the Relevant Fiscal Year and the Earn-Out Shares for the Relevant Fiscal Year (“Relevant Earn-Out Shares”) shall be brought forward to the subsequent fiscal year (“Subsequent Fiscal Year”), whereupon the Relevant Earn-Out Shares can be issued in Subsequent Fiscal Year, subject always that the Surviving Company makes up for the Revenue target for the Relevant Fiscal Year in the next Subsequent Fiscal Year, together with the Revenue target in the next Subsequent Fiscal Year so that at the end of the next Subsequent Fiscal Year the combined Revenue for the Relevant Fiscal Year and the next Subsequent Fiscal Year equals the combined Revenue target for the Relevant Fiscal Year and the next Subsequent Fiscal Year. As an illustration only, if the Surviving Company achieves a Revenue of $6,000,000 in the first full fiscal year, the Earn-Out Shares for the first full fiscal year will not be issued as the Revenue target has not been achieved. However, if the Surviving Company achieves Revenue of $15,000,000 in the second full fiscal year, the Alps Holdco Shareholders shall be entitled to receive the Earn-Out Shares for the first full fiscal year (Relevant Fiscal Year) as well as the second full fiscal year (Subsequent Fiscal Year).
4.6 Adjustment. The Merger Consideration Shares, Earn-out Shares and Conversion Ratio shall be adjusted to reflect appropriately the effect of any stock split or share subdivision, reverse stock split or share consolidation, share dividend or bonus issue of shares or share capitalization, recapitalization, reclassification, combination, exchange of shares or other like change with respect to Reincorporation Merger Surviving Company Ordinary Shares occurring prior to the date the Merger Consideration Shares or the Earn-out Shares are issued.
4.7 No Fractional Shares. No fractional Reincorporation Merger Surviving Company Ordinary Shares, or certificates or scrip representing fractional Reincorporation Merger Surviving Company Ordinary Shares, will be issued upon the conversion of the Alps Holdco Ordinary Shares pursuant to the Merger, and such fractional share interests will not entitle the owner thereof to vote or to any rights of a shareholder of Reincorporation Merger Surviving Company. Any fractional Reincorporation Merger Surviving Company Ordinary Shares will be rounded up or down to the nearest whole number of Reincorporation Merger Surviving Company Ordinary Shares.
4.8 Withholding. Reincorporation Merger Surviving Company and the Surviving Company shall be entitled to deduct and withhold from the consideration otherwise payable to any Person pursuant to this Agreement such amounts as may be required to be deducted or withheld with respect to the making of such payment under the Code, or under any provision of state, local or non-U.S. Tax Law. To the extent that amounts are so deducted and withheld and paid over to the appropriate Taxing Authorities, such amounts shall be treated for all purposes under this Agreement as having been paid to the Person in respect of which such deduction and withholding was made. Notwithstanding the foregoing, Pubco, Parent, Alps Holdco, Reincorporation Merger Surviving Company and the Surviving Company shall use commercially reasonable efforts to reduce or eliminate any such withholding, including providing recipients of consideration a reasonable opportunity to provide documentation establishing exemptions from or reductions of such withholdings.
4.9 Lost, Stolen or Destroyed Certificates. Notwithstanding the foregoing, if any Alps Holdco Share Certificate shall have been lost, stolen or destroyed, then upon the making of a customary affidavit of that fact by the Person claiming such Alps Holdco Share Certificate to be lost, stolen or destroyed in a form reasonably acceptable to Reincorporation Merger Surviving Company, the Exchange Agent shall issue, in exchange for such lost, stolen or destroyed Alps Holdco Share Certificate, the portion of the Merger Consideration Shares or Earn-out Shares to be paid in respect of the Alps Holdco Ordinary Shares formerly represented by such Alps Holdco Share Certificate as contemplated under this ARTICLE IV.
| 27 |
4.10 Escrow.
(a) At or prior to the Closing, the Parent Representative, the Seller Representative and Continental Stock Transfer & Trust Company (or such other escrow agent mutually acceptable to Pubco and Alps Holdco), as escrow agent (the “Escrow Agent”), shall enter into an Escrow Agreement, effective as of the Effective Time, in form and substance reasonably satisfactory to Pubco and Alps Holdco (the “Escrow Agreement”), pursuant to which Reincorporation Merger Surviving Company shall issue to the Escrow Agent a number of Reincorporation Merger Surviving Company Ordinary Shares (with each share valued at $10.00) equal to five percent (5%) of the Merger Consideration (the “Escrow Amount”) (together with any equity securities paid as dividends or distributions with respect to such shares or into which such shares are exchanged or converted, the “Escrow Shares”) to be held, along with any other dividends, distributions or other income on the Escrow Shares (together with the Escrow Shares, the “Escrow Property”), in a segregated escrow account (the “Escrow Account”) and disbursed therefrom in accordance with the terms of ARTICLE XII hereof and the Escrow Agreement. Upon disbursement in accordance with the terms of ARTICLE XII hereof, the Escrow Property shall be allocated among and transferred to Alps Holdco Shareholders pro rata based on their respective Pro Rata Share. The Escrow Property shall serve as the sole source of payment for the obligations of the Alps Holdco Shareholders pursuant to ARTICLE XII. Unless otherwise required by Law, all distributions made from the Escrow Account shall be treated by the parties as an adjustment to the number of Merger Consideration Shares received by the Alps Holdco Shareholders pursuant to ARTICLE XII hereof.
(b) The Escrow Property shall not be subject to any indemnification claim to the extent made after the date which is six (6) months after the Closing Date (the “Expiration Date”); provided, however, with respect to any indemnification claims made in accordance with ARTICLE XII hereof on or prior to the Expiration Date that remain unresolved at the time of the Expiration Date (“Pending Claims”), all or a portion of the Escrow Property reasonably necessary to satisfy such Pending Claims (as determined based on the amount of the indemnification claim included in the Indemnification Notice provided by the Parent Representative under ARTICLE XII and the Reincorporation Merger Surviving Company Share Price as of the Expiration Date) shall remain in the Escrow Account until such time as such Pending Claim shall have been finally resolved and paid pursuant to the provisions of ARTICLE XII. After the Expiration Date, any Escrow Property remaining in the Escrow Account that is not subject to Pending Claims, if any, and not subject to resolved but unpaid claims in favor of an Indemnified Party, shall be transferred by the Escrow Agent to the Alps Holdco Shareholders that have previously delivered the Letter of Transmittal in accordance with Section 4.3(b), with each such Alps Holdco Shareholder receiving its Pro Rata Share of such Escrow Property. Promptly after the final resolution of all Pending Claims and payment of all indemnification obligations in connection therewith, the Escrow Agent shall transfer any remaining Escrow Property remaining in the Escrow Account to the Alps Holdco Shareholders that have previously delivered the Letter of Transmittal in accordance with Section 4.3(b), with each such Alps Holdco Shareholder receiving its Pro Rata Share of such Escrow Property.
ARTICLE V
REPRESENTATIONS AND WARRANTIES OF ALPS HOLDCO, PUBCO AND MERGER SUB
Except as set forth in the Disclosure Schedules delivered by Alps Holdco to Parent prior to the execution of this Agreement (with specific reference to the particular section or subsection of this Agreement to which the information set forth in such Disclosure Schedules relate (which qualify (a) the correspondingly numbered representation, warranty or covenant specified therein and (b) such other representations, warranties or covenants where its relevance as an exception to (or disclosure for purposes of) such other representation, warranty or covenant is reasonably apparent on its face or cross-referenced), each of Alps Holdco, Pubco and Merger Sub hereby severally represents and warrants to Parent that each of the following representations and warranties are true, correct and complete as of the date of this Agreement and as of the Closing Date (except for representations and warranties that are made as of a specific date, which are made only as of such date or representations and warranties that are made with respect to a certain party, which are applicable only to such named party).
| 28 |
5.1 Corporate Existence and Power.
(a) Each of Pubco, Merger Sub, Alps Holdco and each other member of the Alps Holdco Group is an exempted company, corporation or legal entity duly incorporated, registered and/or organized, validly existing and in good standing (with respect to jurisdictions that recognize that concept) under the laws of its jurisdiction of its incorporation, registration and organization, as the case may be. Alps Holdco and each other member of the Alps Holdco Group has all requisite power and authority, corporate and otherwise, to own, lease or otherwise hold and operate its properties and other assets and to carry on the Business as presently conducted and as proposed to be conducted. Alps Holdco and each other member of the Alps Holdco Group is duly licensed or qualified to do business and is in good standing (with respect to jurisdictions that recognize that concept) in each jurisdiction in which the nature of its business or the ownership, leasing or operation of its properties or other assets makes such qualification, licensing or good standing necessary, except where the failure to be so qualified, licensed or in good standing, individually or in the aggregate, has not had and would not reasonably be expected to have a Material Adverse Effect in respect of Alps Holdco Group. Pubco, Merger Sub, Alps Holdco and each other member of the Alps Holdco Group has offices located only at the addresses set forth on Schedule 5.1. Each of Pubco, Merger Sub, and Alps Holdco has made available to Parent, on or prior to the date of this Agreement, complete and accurate copies of its memorandum and articles of association, and the comparable organizational documents of each of its Subsidiaries, in each case as amended to the date hereof (collectively, “Organizational Documents”), and the Organizational Documents are in full force and effect. None of Pubco, Merger Sub, Alps Holdco nor any of Alps Holdco’s Subsidiaries is in violation of its respective Organizational Documents.
(b) Each of Pubco and Merger Sub was formed solely for the purposes of engaging in the transactions contemplated herein and have not engaged in any business activities or conducted any operations other than in connection with or related to the Mergers as contemplated by this Agreement.
5.2 Authorization.
(a) Each of Pubco, Merger Sub and Alps Holdco has all requisite corporate power to execute and deliver this Agreement and the Additional Agreements to which it is a party and to consummate the transactions contemplated hereby and thereby. The execution and delivery by Pubco, Merger Sub and Alps Holdco of this Agreement and the Additional Agreements to which it is a party and the consummation by Alps Holdco of the transactions contemplated hereby and thereby have been duly authorized by all necessary corporate action on the part of Pubco, Merger Sub and Alps Holdco, except that (1) the Board of Directors of Alps Holdco will need to approve the draft Plan of Merger and approve the authorization, filing and registration of the Plan of Merger and the Alps Restructuring, (2) the Board of Directors of Merger Sub will need to approve the draft Plan of Merger and approve the authorization, filing and registration of the Plan of Merger, (3) the Board of Directors of Pubco will need to approve the draft of the Reincorporation Plan of Merger, the authorization, filing and registration of the Reincorporation Plan of Merger, the issue and reservation of Reincorporation Merger Surviving Company Ordinary Shares, the change of directors and officers at the Reincorporation Effective Time and the Effective Time and any other actions to complete Closing as required under this Agreement, (4) the Alps Holdco Shareholder Approval, Pubco Shareholder Approval, and Merger Sub Shareholder Approval will need to be obtained. No other corporate proceedings on the part of Pubco, Merger Sub and Alps Holdco are necessary to authorize this Agreement or the Additional Agreements to which it is a party or to consummate the transactions contemplated by this Agreement (other than the authorization referred to above in this Section 5.2(a)) or the Additional Agreements. This Agreement and the Additional Agreements to which Pubco, Merger Sub and Alps Holdco is a party have been duly executed and delivered by Pubco, Merger Sub and Alps Holdco and, assuming the due authorization, execution and delivery by each of the other parties hereto and thereto, this Agreement and the Additional Agreements to which Pubco, Merger Sub and Alps Holdco is a party constitute a legal, valid and binding obligation of Pubco, Merger Sub and Alps Holdco, enforceable against Pubco, Merger Sub and Alps Holdco in accordance with their respective terms, subject to applicable bankruptcy, insolvency, fraudulent conveyance, reorganization, moratorium and similar Laws affecting or relating to creditors’ rights generally and subject, as to enforceability, to general principles of equity, whether such enforceability is considered in a proceeding in equity or at Law (the “Enforceability Exceptions”).
| 29 |
(b) By resolutions duly passed (and not thereafter modified or rescinded) by the requisite vote of the Board of Directors of Pubco, Merger Sub and Alps Holdco, the Board of Directors of each of Pubco, Merger Sub and Alps Holdco has (i) approved the execution, delivery and performance by Pubco, Merger Sub and Alps Holdco of this Agreement, the Additional Agreements to which it is a party and the consummation of the transactions contemplated hereby and thereby, including the Mergers, on the terms and subject to the conditions set forth herein and therein; (ii) determined that this Agreement, the Additional Agreements to which it is a party, and the transactions contemplated hereby and thereby, upon the terms and subject to the conditions set forth herein, are advisable and fair to and in the best interests of Alps Holdco, Pubco, Merger Sub and their respective shareholders; (iii) directed that the adoption of this Agreement and the approval of the transactions contemplated hereby be submitted to their respective shareholders for consideration and recommended that all of such shareholders adopt this Agreement and approve such transactions. A special resolution (as defined in the Companies Act and the Memorandum and Articles of Association of Alps Holdco) of the Alps Holdco Shareholders, being either (i) a resolution passed by a majority of at least two-thirds of the Alps Holdco Shareholders entitled to vote and voting in person or, where proxies are allowed, by proxy at a general meeting of which notice specifying the intention to propose the resolution as a special resolution has been duly given or (ii) a unanimous written resolution signed by all of the Alps Holdco Shareholders or their duly appointed attorneys, is required to, and shall be sufficient to, approve this Agreement and the transactions contemplated hereby (the “Alps Holdco Shareholder Approval”). The Alps Holdco Shareholder Approval is the only vote or consent of any of the holders of Alps Holdco Ordinary Shares necessary to adopt this Agreement and approve the Mergers and the consummation of the other transactions contemplated hereby by Alps Holdco. The Pubco Shareholder Approval is the only vote or consent of any of the holders of Pubco Ordinary Shares necessary to adopt this Agreement and approve the Mergers and the consummation of the other transactions contemplated hereby by Pubco. The Merger Sub Shareholder Approval is the only vote or consent of any of the holders of shares of Merger Sub necessary to adopt this Agreement and approve the Mergers and the consummation of the other transactions contemplated hereby by Merger Sub.
5.3 Governmental Authorization. Assuming the accuracy of the representations and warranties of Parent set forth in this Agreement, none of the execution, delivery or performance by Alps Holdco, Pubco or Merger Sub of this Agreement or any Additional Agreement to which Alps Holdco, Pubco or Merger Sub is or will be a party, or the consummation of the transactions contemplated hereby or thereby, requires any consent, approval, license, Order or other action by or in respect of, or registration, declaration or filing with, any Authority, except for (a) to the extent applicable, the filing of a premerger notification and report form by Alps Holdco under the HSR Act and the termination of the waiting period required thereunder, and (b) the filing of the Plans of Merger and all other documents required by the Companies Act with the Cayman Registrar pursuant to the Companies Act and the terms hereof.
| 30 |
5.4 Non-Contravention. None of the execution, delivery or performance by Alps Holdco, Pubco or Merger Sub of this Agreement or any Additional Agreement to which Alps Holdco, Pubco or Merger Sub is or will be a party or the consummation by Alps Holdco, Pubco or Merger Sub of the transactions contemplated hereby and thereby does or will (a) contravene or conflict with the Organizational Documents of any member of the Alps Holdco Group, Pubco or Merger Sub (b) contravene or conflict with or constitute a violation of any provision of any Law or Order binding upon or applicable to any member of the Alps Holdco Group, Pubco or Merger Sub or to any of their respective properties, rights or assets, (c) except for the Contracts listed on Schedule 5.8 requiring Alps Holdco Consents (but only as to the need to obtain such Alps Holdco Consents), (i) require consent, approval or waiver under, (ii) constitute a default under or breach of (with or without the giving of notice or the passage of time or both), (iii) violate, (iv) give rise to any right of termination, cancellation, amendment or acceleration of any right or obligation of the Alps Holdco Group or to a loss of any material benefit to which any member of the Alps Holdco Group is entitled, in the case of each of clauses (i) – (iv), under any provision of any Permit, Contract or other instrument or obligations binding upon any member of the Alps Holdco Group or any of their respective properties, rights or assets, (d) result in the creation or imposition of any Lien (except for Permitted Liens) on any of the Alps Holdco Group’s properties, rights or assets, or (e) require any consent, approval or waiver from any Person pursuant to any provision of the Organizational Documents of any member of the Alps Holdco Group, Pubco or Merger Sub, except for such consent, approval or waiver which shall be obtained (and a copy provided to Parent) prior to the Closing.
5.5 Capitalization.
(a) As of the date hereof, (i) the authorized share capital of Alps Holdco is US$5,000 divided into 500,000,000 Alps Holdco Ordinary Shares and (ii) the issued share capital of Alps Holdco consists of one (1) Alps Holdco Ordinary Share. Prior to Closing, the issued share capital of Alps Holdco shall be increased by the issuance by Alps Holdco of duly authorized, validly issued, fully paid and non-assessable Alps Holdco Ordinary Shares on a one-for-one basis in exchange for the contribution of ordinary shares in Alps Malaysia to Alps Holdco pursuant to the Alps Restructuring, leading to the issued share capital of Alps Holdco comprising of no more than 58,041,998 Alps Holdco Ordinary Shares. No other shares or other securities of Alps Holdco are authorized, issued, reserved for issuance or outstanding. All issued and outstanding Alps Holdco Ordinary Shares are duly authorized, validly issued, fully paid and non-assessable. No Alps Holdco Ordinary Shares are subject to or were issued in violation of any purchase option, right of first refusal, preemptive right, subscription right or any similar right (including under any applicable Laws of the Cayman Islands, the Organizational Documents of Alps Holdco or any Contract to which Alps Holdco is a party or by which Alps Holdco or any of its properties, rights or assets are bound). After the date of this Agreement and prior to the Closing, the Alps Holdco Ordinary Shares will be owned of record by the Persons set forth on Schedule 5.5 in the amounts set forth opposite their respective names.
(b) The authorized share capital of Pubco is US$50,000, divided into 500,000,000 ordinary shares of par value US$0.0001 each (the “Pubco Ordinary Shares”), of which one (1) Pubco Ordinary Share is issued and outstanding as of the date hereof. No other shares or other securities of Pubco are authorized, issued, reserved for issuance or outstanding. All issued and outstanding Pubco Ordinary Shares are duly authorized, validly issued, fully paid and non-assessable. No Pubco Ordinary Shares are subject to or were issued in violation of any purchase option, right of first refusal, preemptive right, subscription right or any similar right (including under any applicable Laws of the Cayman Islands, the Organizational Documents of Pubco or any contract to which Pubco is a party or by which Pubco or any of its properties, rights or assets are bound). As of the date of this Agreement, all Pubco Ordinary Shares are owned of record by the Persons set forth on Schedule 5.5 in the amounts set forth opposite their respective names.
(c) The authorized share capital of Merger Sub is US$5,000, divided into 500,000,000 ordinary shares of par value US$0.00001 each (the “Merger Sub Ordinary Shares”), of which one (1) Merger Sub Ordinary Share is issued and outstanding as of the date hereof. No other shares or other securities of Merger Sub are authorized, issued, reserved for issuance or outstanding. All issued and outstanding Merger Sub Ordinary Shares are duly authorized, validly issued, fully paid and non-assessable. No Merger Sub Ordinary Shares are subject to or were issued in violation of any purchase option, right of first refusal, preemptive right, subscription right or any similar right (including under any applicable Laws of the Cayman Islands, the Organizational Documents of Merger Sub or any contract to which Merger Sub is a party or by which Merger Sub or any of its properties, rights or assets are bound). As of the date of this Agreement, all Merger Sub Ordinary Shares are owned of record by Pubco.
| 31 |
(d) Except as set forth on Schedule 5.5, there are no (i) outstanding warrants, options, agreements, convertible securities, performance units or other commitments or instruments pursuant to which Alps Holdco, Pubco or Merger Sub is or may become obligated to issue or sell any of its Alps Holdco Ordinary Shares or other securities, (ii) outstanding obligations of Alps Holdco, Pubco or Merger Sub to repurchase, redeem or otherwise acquire outstanding shares Alps Holdco, Pubco or Merger Sub, (iii) treasury shares of Alps Holdco, Pubco or Merger Sub, (iv) bonds, debentures, notes or other Indebtedness of Alps Holdco, Pubco or Merger Sub having the right to vote (or convertible into, or exchangeable for, securities having the right to vote) on any matters on which shareholders of Alps Holdco, Pubco or Merger Sub may vote, are issued or outstanding, (v) preemptive or similar rights to purchase or otherwise acquire shares or other securities of Alps Holdco, Pubco or Merger Sub (including pursuant to any provision of Law, the Organizational Documents of Alps Holdco, Pubco or Merger Sub or any Contract to which Alps Holdco is a party), or (vi) Liens (including any right of first refusal, right of first offer, proxy, voting trust, voting agreement or similar arrangement) with respect to the sale or voting of shares or securities of Alps Holdco, Pubco or Merger Sub (whether outstanding or issuable).
(e) The Consideration Spreadsheet, when delivered by Alps Holdco pursuant to Section 4.4(a), will be true, complete and correct in all material respects as of immediately prior to the Effective Time.
5.6 Corporate Records. To the Knowledge of Alps Holdco, Pubco and Merger Sub, all proceedings occurring since the incorporation of Alps Holdco, Pubco and Merger Sub, of the Board of Directors of Alps Holdco, Pubco and Merger Sub, including all committees thereof, and of the Alps Holdco Shareholders and the shareholders of Pubco and Merger Sub, and all resolutions (including resolutions in writing) and consents to actions taken thereby, are accurately reflected in the minutes and records contained in the corporate minute books of Alps Holdco, Pubco or Merger Sub and made available to Parent. The statutory registers of Alps Holdco, Pubco and Merger Sub made available to Parent are true, correct and complete in all respects.
5.7 Subsidiaries. Schedule 5.7 lists each Subsidiary of Alps Holdco, Pubco, Merger Sub and each other entity which Alps Holdco, Pubco, Merger Sub or any Subsidiary owns any equity of similar interest, together with the jurisdiction of incorporation or formation of each Subsidiary or such other entity and the percentage of the equity interest of each Subsidiary or such other entity that is owned by Alps Holdco, Pubco, Merger Sub and each other Subsidiary. As at the date of this Agreement, Alps Malaysia is not a subsidiary of Alps Holdco. After the date of this Agreement and prior to the Closing, it is intended that Alps Malaysia will become a Subsidiary of Alps Holdco pursuant to the Alps Restructuring. All the issued and outstanding shares of capital stock of, or other equity interests in, each Subsidiary of Alps Holdco, Pubco and Merger Sub have been validly issued and are fully paid and non-assessable and are owned directly or indirectly Alps Holdco free and clear of all Liens. Except for the Subsidiaries of Alps Holdco, Alps Holdco does not own, directly or indirectly, as of the date hereof, (i) any capital stock of, or other voting securities or other equity or voting interests in, any Person or (ii) any other interest or participation that confers on Alps Holdco or any Subsidiary of Alps Holdco the right to receive (A) a share of the profits and losses of, or distributions of assets of, any other Person or (B) any economic benefit or right similar to, or derived from, the economic benefits and rights occurring to holders of capital stock of any other Person. Except for Merger Sub, Pubco does not own, directly or indirectly, as of the date hereof, (i) any capital stock of, or other voting securities or other equity or voting interests in, any Person or (ii) any other interest or participation that confers on Pubco the right to receive (A) a share of the profits and losses of, or distributions of assets of, any other Person or (B) any economic benefit or right similar to, or derived from, the economic benefits and rights occurring to holders of capital stock of any other Person. Merger Sub does not own, directly or indirectly, as of the date hereof, (i) any capital stock of, or other voting securities or other equity or voting interests in, any Person or (ii) any other interest or participation that confers on Merger Sub the right to receive (A) a share of the profits and losses of, or distributions of assets of, any other Person or (B) any economic benefit or right similar to, or derived from, the economic benefits and rights occurring to holders of capital stock of any other Person.
| 32 |
5.8 Consents. The Contracts listed on Schedule 5.8 are the only Material Contracts requiring a consent, approval, authorization, order or other action of or filing with any Person as a result of the execution, delivery and performance of this Agreement or any Additional Agreement to which the Alps Holdco Group is or will be a party or the consummation of the transactions contemplated hereby or thereby (each of the foregoing, a “Alps Holdco Consent”).
5.9 Financial Statements.
(a) The audited consolidated balance sheets of the Alps Holdco Group, and the related statements of operations, changes in shareholders’ equity and cash flows, for the fiscal year ended March 31, 2023 including the notes thereto, prepared under IFRS and audited by a qualified auditor in accordance with requirements of IFRS (collectively, the “Annual Financial Statements”, and together with, when delivered in accordance with Section 8.3, the Final March 31, 2024 Financial Statements, the “Alps Holdco Financial Statements”) have been prepared in conformity with IFRS applied on a consistent basis. Alps Holdco Financial Statements fairly present, in all material respects, the financial position of the Alps Holdco Group as of the dates thereof and the results of operations of Alps Holdco for the periods reflected therein. The Alps Holdco Financial Statements were prepared from the Books and Records of the Alps Holdco Group in all material respects. Since March 31, 2024 (the “Balance Sheet Date”), except as required by applicable Law or IFRS, there has been no change in any accounting principle, procedure or practice followed by Alps Holdco or in the method of applying any such principle, procedure or practice.
(b) Except as: (i) specifically disclosed, reflected or fully reserved against on the Balance Sheets; (ii) liabilities and obligations incurred in the ordinary course of business consistent with past practices since the date of the Balance Sheets that are not material; (iii) liabilities that are executory obligations arising under Contracts to which a member of the Alps Holdco Group is a party (none of which, with respect to the liabilities described in clause (ii) and this clause (iii) results from, arises out of, or relates to any breach or violation of, or default under, a Contract or applicable Law); (iv) expenses incurred in connection with the negotiation, execution and performance of this Agreement, any Additional Agreement or any of the transactions contemplated hereby or thereby; and (v) liabilities set forth on Schedule 5.9(b), the Alps Holdco Group does not have any material liabilities, debts or obligations of any nature (whether accrued, fixed or contingent, liquidated or unliquidated or asserted or, to the Knowledge of Alps Holdco, unasserted).
(c) Except as set forth on Schedule 5.9(c), the Alps Holdco Group does not have any Indebtedness for borrowed money or any other material Indebtedness.
5.10 Books and Records. The Books and Records accurately and fairly, in reasonable detail, reflect the transactions and dispositions of assets of and the providing of services by the Alps Holdco Group. The Books and Records of Alps Holdco have been maintained, in all material respects in accordance with reasonable business practices.
5.11 Internal Accounting Controls. Except as set forth on Schedule 5.11, the Alps Holdco Group has established a system of internal accounting controls sufficient to provide reasonable assurance that: (a) transactions are executed in accordance with management’s general or specific authorizations; (b) transactions are recorded as necessary to permit preparation of financial statements in conformity with IFRS, and the Alps Holdco Group’s historical practices and to maintain asset accountability; (c) access to assets is permitted only in accordance with management’s general or specific authorization; and (d) the recorded accountability for assets is compared with the existing assets at reasonable intervals and appropriate action is taken with respect to any differences.
| 33 |
5.12 Absence of Certain Changes. To Alps Holdco’s Knowledge, from the Balance Sheet Date until the date of this Agreement, (a) each member of the Alps Holdco Group have conducted their respective businesses in the ordinary course and in a manner consistent with past practice; (b) there has not been any Material Adverse Effect in respect of the Alps Holdco Group; and (c) neither Alps Holdco nor any other member of the Alps Holdco Group has taken any action that, if taken after the date of this Agreement and prior to the consummation of the Mergers, would require the consent of Parent pursuant to Section 7.1 and Parent has not given consent.
5.13 Properties; Title to Alps Holdco’s Assets.
(a) To Alps Holdco’s Knowledge, all items of Tangible Personal Property in the aggregate are in good operating condition and repair and function in all material respects in the aggregate in accordance with their intended uses (ordinary wear and tear excepted).
(b) Alps Holdco or a Subsidiary has good, valid and marketable title in and to, or in the case of Real Property leasehold interests and the material assets which are leased or licensed pursuant to Contracts, a valid leasehold interest or license in or a right to use the aggregate tangible assets reflected on the Balance Sheets. Except as set forth on Schedule 5.13(b) and the Balance Sheets, no such tangible asset is subject to any Lien other than Permitted Liens. The Alps Holdco Group’s assets constitute all of the rights, properties, and assets of any kind or description whatsoever, including goodwill, reasonably necessary for the Alps Holdco Group to operate the Business immediately after the Closing in substantially the same manner as the Business is currently being conducted.
5.14 Litigation. Except as set forth on Schedule 5.14, there is no Action that is otherwise material to the Alps Holdco Group, Pubco, or Merger Sub that is pending or, to the Knowledge of Alps Holdco, threatened against or affecting the Alps Holdco Group, Pubco, or Merger Sub, any of the officers or directors of the Alps Holdco Group, Pubco, or Merger Sub, any of the rights, properties or assets of Alps Holdco Group, Pubco, or Merger Sub or any Contract before any Authority or which in any manner challenges or seeks to prevent, enjoin, alter or delay the transactions contemplated by this Agreement or any Additional Agreement. There are no outstanding judgments against the Alps Holdco Group, Pubco, or Merger Sub. None of the Alps Holdco Group, Pubco, or Merger Sub is subject to any Action by any Authority.
5.15 Contracts.
(a) Schedule 5.15(a) sets forth a true, complete and accurate list, as of the date of this Agreement, of all of the following Contracts as amended to date which are currently in effect (collectively, “Material Contracts”):
(i) all Contracts that require annual payments or expenses incurred by, or annual payments or income to, the Alps Holdco Group, Pubco or Merger Sub, of $50,000 or more (other than standard purchase and sale orders entered into in the ordinary course of business consistent with past practices);
(ii) all sales, advertising, agency, lobbying, broker, sales promotion, market research, marketing or similar Contracts that require annual payments of $100,000 or more;
| 34 |
(iii) each Contract with any current officer, director, employee or consultant of the Alps Holdco Group, Pubco or Merger Sub, under which the Alps Holdco Group, Pubco or Merger Sub (A) has continuing obligations for payment of an annual compensation of at least $50,000, and which is not terminable for any reason or no reason upon reasonable notice without payment of any penalty, severance or other obligation; (B) has severance or post-termination obligations to such Person in excess of $50,000 (other than COBRA obligations); or (C) has an obligation to make a payment upon consummation of the transactions contemplated hereby;
(iv) all Contracts with third parties creating a limited liability company or legal partnership arrangement or a material joint venture or strategic alliance to which the Alps Holdco Group, Pubco or Merger Sub is a party;
(v) all Contracts relating to any acquisitions or dispositions of material assets by the Alps Holdco Group, Pubco or Merger Sub (other than acquisitions or dispositions of inventory in the ordinary course of business consistent with past practices) under which the Alps Holdco Group, Pubco or Merger Sub has any continuing obligations (other than confidentiality requirements);
(vi) all IP Contracts under which the Alps Holdco Group, Pubco or Merger Sub is obligated to pay royalties thereunder in excess of $50,000 per year, all IP Contracts under which the Alps Holdco Group, Pubco or Merger Sub is entitled to receive royalties in excess of $50,000 per year thereunder, all IP Contracts under which Alps Holdco, Pubco or Merger Sub has granted or has received an exclusive license or right to Intellectual Property, all IP Contracts involving an agreement not to assert or sue with respect to Intellectual Property (other than non-exclusive licenses), and all settlement agreements and co-existence agreements involving Intellectual Property;
(vii) all Contracts limiting the freedom of the Alps Holdco Group, Pubco or Merger Sub to compete in any line of business or industry, with any Person or in any geographic area;
(viii) all Contracts providing for guarantees, indemnification arrangements and other hold harmless arrangements made or provided by the Alps Holdco Group, Pubco or Merger Sub other than commercial arrangements where it is not the primary purpose of the agreement;
(ix) all Contracts with or pertaining to the Alps Holdco Group, Pubco or Merger Sub which any Affiliate of the Alps Holdco Group, Pubco or Merger Sub is a party, other than any Contracts (A) relating to such Affiliate’s status as an Alps Holdco Shareholder or shareholder of Pubco or Merger Sub, (B) relating to employment or service as a director or (C) among the Alps Holdco and/or its Subsidiaries, Pubco or Merger Sub;
(x) all Contracts relating to property or assets (whether real or personal, tangible or intangible) in which the Alps Holdco Group, Pubco or Merger Sub holds a leasehold interest and which involve payments to the lessor thereunder in excess of $50,000 per year;
(xi) all Contracts creating or otherwise relating to outstanding Indebtedness for borrowed money or any other material Indebtedness (other than intercompany Indebtedness);
(xii) all Contracts relating to the voting or control of the equity interests of the Alps Holdco Group or the election of directors of the Alps Holdco Group, Pubco or Merger Sub (other than the organizational documents of the Alps Holdco Group, Pubco or Merger Sub);
| 35 |
(xiii) all Contracts not cancellable by the Alps Holdco Group, Pubco or Merger Sub with no more than sixty (60) days’ notice if the effect of such cancellation would result in monetary penalty to the Alps Holdco Group, Pubco or Merger Sub in excess of $50,000 per the terms of such contract;
(xiv) all material Contracts that may be terminated, or the provisions of which may be altered, as a result of the consummation of the transactions contemplated by this Agreement or any Additional Agreement;
(xv) all Contracts under which any of the benefits, compensation or payments (or the vesting thereof) will be increased or accelerated by the consummation of the transactions contemplated by this Agreement or any Additional Agreement; and
(xvi) all collective bargaining agreements or other agreement with a labor union or labor organization.
(b) Except as set forth on Schedule 5.15(b), each Material Contract is (i) a valid and binding agreement, (ii) in full force and effect and (iii) enforceable by and against Alps Holdco, Pubco or Merger Sub or any Subsidiary of Alps Holdco and each counterparty that is party thereto, subject, in the case of this clause (iii), to the Enforceability Exceptions. None of the Alps Holdco Group, Pubco, Merger Sub nor, to the Knowledge of Alps Holdco, Pubco or Merger Sub, any other party to a Material Contract is in material breach or default (whether with or without the passage of time or the giving of notice or both) under the terms of any such Material Contract. None of the Alps Holdco Group, Pubco or Merger Sub has assigned, delegated or otherwise transferred any of its rights or obligations under any Material Contract or granted any power of attorney with respect thereto.
(c) Each of the Alps Holdco Group, Pubco and Merger Sub is in compliance in all material respects with all covenants, including all financial covenants, in all notes, indentures, bonds and other instruments or Contracts establishing or evidencing any Indebtedness. The consummation and closing of the transactions contemplated by this Agreement shall not cause or result in an event of default under any instruments or Contracts establishing or evidencing any Indebtedness.
5.16 Licenses and Permits. Schedule 5.16 sets forth a true, complete and correct list of each material license, franchise, permit, order or approval or other similar authorization required under applicable Law to carry out the Business as currently conducted, together with the name of the Authority issuing the same (the “Permits”). The Alps Holdco Group has all such Permits, and each such Permit is in full force and effect and none of such Permits will be terminated or materially impaired as a result of the transactions contemplated hereby. Alps Holdco is not in breach or violation of, or default under, any such Permit, and, to Alps Holdco’s Knowledge, no basis (including the execution of this Agreement and the other Additional Agreements to which Alps Holdco is a party and the consummation of the transactions contemplated by this Agreement) reasonably exists which, with notice or lapse of time or both, would reasonably constitute any such breach, violation or default or give any Authority grounds to suspend, revoke or terminate any such Permit. Alps Holdco has not received any written (or, to Alps Holdco’s Knowledge, oral) notice from any Authority regarding any material violation of any Permit. There has not been and there is not any pending or, to Alps Holdco’s Knowledge, threatened Action, investigation or disciplinary proceeding by or from any Authority against Alps Holdco involving any Permit.
| 36 |
5.17 Compliance with Laws.
(a) None of the Alps Holdco Group, Pubco and Merger Sub, nor to the Knowledge of Alps Holdco, Pubco or Merger Sub, any Representative or other Person acting on behalf of the Alps Holdco Group, Pubco or Merger Sub, is in violation in any material respect of, and, since its respective formation date, no such Person has failed to be in compliance with, all applicable Laws and Orders in all material respects. Since inception (i) no event has occurred or circumstance exists that (with or without notice or due to lapse of time) would reasonably constitute or result in a material violation by the Alps Holdco Group, Pubco or Merger Sub, as applicable, of, or failure on the part of the Alps Holdco Group, Pubco or Merger Sub to comply in all material respects with, or any liability suffered or incurred by the Alps Holdco Group, Pubco or Merger Sub in respect of any material violation of or material noncompliance with, any Laws, Orders or policies by Authority that are or were applicable to it or the conduct or operation of its business or the ownership or use of any of its assets and (ii) no Action is pending, or to the Knowledge of the Alps Holdco, Pubco and Merger Sub threatened, alleging any such violation or noncompliance by a member of the Alps Holdco Group, Pubco or Merger Sub. Since inception, none of the Alps Holdco Group, Pubco or Merger Sub has been threatened in writing or, to the Knowledge of Alps Holdco, Pubco and Merger Sub, orally to be charged with, or given written or, to the Knowledge of Alps Holdco, Pubco and Merger Sub, oral notice of any violation of any Law or any judgment, order or decree entered by any Authority. Without limiting the generality of the foregoing, each of the Alps Holdco Group, Pubco and Merger Sub is, and since inception, has been, in compliance in all material respects with: (i) every Law applicable to the Alps Holdco Group, Pubco or Merger Sub due to the specific nature of the Business as currently conducted, including Data Protection Laws; (ii) the Foreign Corrupt Practices Act of 1977 (the “Foreign Corrupt Practices Act”) and any comparable or similar Law of any jurisdiction applicable to the Alps Holdco Group, Pubco or Merger Sub; and (iii) every Law regulating or covering conduct in the workplace, including regarding sexual harassment or, on any legally impermissible basis, a hostile work environment. Since inception, none of the Alps Holdco Group, Pubco or Merger Sub has been threatened or charged in writing (or to the Knowledge of Alps Holdco, Pubco or Merger Sub orally) with or given written (or to the Knowledge of Alps Holdco, Pubco or Merger Sub oral) notice of any violation of any Data Protection Law, the Foreign Corrupt Practices Act or any other Law referred to in or generally described in foregoing sentence and, to the Knowledge of Alps Holdco, Pubco or Merger Sub, none of the Alps Holdco Group, Pubco or Merger Sub is not under any investigations with respect to any such Law.
(b) None of the Alps Holdco Group, Pubco, nor Merger Sub nor, to the Knowledge of Alps Holdco, Pubco, and Merger Sub, any Representative or other Person acting on behalf of the Alps Holdco Group, Pubco or Merger Sub is currently subject to any U.S. sanctions administered by the Office of Foreign Assets Control of the U.S. Treasury Department.
5.18 Intellectual Property.
(a) The Alps Holdco Group is the sole and exclusive owner of each item of Alps Holdco Owned IP, free and clear of any Liens. To the Knowledge Alps Holdco, the Alps Holdco Group is the sole and exclusive licensee of in each item of Alps Holdco Exclusively Licensed IP, free and clear of any Liens. To the Knowledge of Alps Holdco, the Alps Holdco Group has a valid right to use the Alps Holdco Licensed IP. Pubco and Merger Sub do not own any Intellectual Property, Registered IP, unregistered or registered Trademarks, or Domain Names.
(b) Schedule 5.18(b) sets forth a true, correct and complete list of all (i) Registered Owned IP; (ii) Registered Exclusively Licensed IP; (iii) unregistered Trademarks constituting Alps Holdco Owned IP; and (iv) Domain Names constituting Alps Holdco Owned IP; accurately specifying as to each of the foregoing, as applicable: (A) the application number, issuance or registration number, or other identifying details; (B) the owner and nature of the ownership; and (C) the jurisdictions by or in which such Registered Owned IP has been issued, registered, or in which an application for such issuance or registration has been filed or for unregistered material Trademarks, used by the Alps Holdco Group.
| 37 |
(c) Except for any Alps Holdco Owned IP appearing on Schedule 5.18(b) that the Alps Holdco Group may have intentionally abandoned or let expire, all Registered Owned IP is subsisting and, to the Knowledge of Alps Holdco, valid and enforceable. To the Knowledge of the Alps Holdco, all Registered Exclusively Licensed IP is subsisting, valid and enforceable. To the Knowledge of Alps Holdco, all Persons (including members of the Alps Holdco Group) have, in connection with the prosecution of all Patents before the United States Patent and Trademark Office (the “USPTO”) and, where applicable, other similar offices in other jurisdictions complied with the applicable obligations of candor owed to the USPTO and, where applicable, such other offices. No Registered Owned IP, and to the Knowledge of the Alps Holdco no Registered Exclusively Licensed IP, is or has been involved in any interference, opposition, reissue, reexamination, revocation or equivalent proceeding, and no such proceeding has been threatened in writing with respect thereto. Since Alps Holdco’s incorporation, not including any office actions or other responses issued by the USPTO or other similar offices in other jurisdictions in the ordinary course of ex parte prosecution, there have been no claims filed, served or threatened in writing, or to the Knowledge of Alps Holdco orally threatened, against Alps Holdco contesting the validity, use, ownership, enforceability, patentability, registrability, or scope of any Registered IP. All registration, maintenance and renewal fees currently due in connection with any Registered Owned IP, and to the Knowledge of Alps Holdco all Registered Exclusively Licensed IP, have been paid and all documents, recordations and certificates in connection therewith have been filed with the authorities in the United States or foreign jurisdictions, as the case may be, for the purposes of prosecuting, maintaining and perfecting such rights and recording the Alps Holdco Group’s ownership or interests therein.
(d) To the Knowledge of Alps Holdco, the operation of the Business as currently conducted and as conducted since the date of formation of the first member of the Alps Holdco Group do not conflict with, infringe, misappropriate or otherwise violate any Intellectual Property of any third Person. Since the date of formation of the first member of the Alps Holdco Group, there have been no claims filed, served or threatened in writing, or to the Knowledge of Alps Holdco orally threatened, against Alps Holdco alleging any conflict with, infringement, misappropriation, or other violation of any Intellectual Property of a third Person (including any unsolicited written offers to license any such Intellectual Property). There are no Actions pending that involve a claim against a member of the Alps Holdco Group by a third Person alleging infringement or misappropriation of such third Person’s Intellectual Property. To the Knowledge of Alps Holdco, since the date of formation of the first member of the Alps Holdco Group, no third Person has conflicted with, infringed, misappropriated, or otherwise violated any Alps Holdco IP.
(e) Since the date of formation of the first member of the Alps Holdco Group, no member of Alps Holdco Group has filed, served, or threatened a third Person with any claims alleging any conflict with, infringement, misappropriation, or other violation of any Alps Holdco IP. There are no Actions pending that involve a claim against a third Person by a member of the Alps Holdco Group alleging infringement or misappropriation of Alps Holdco IP. The Alps Holdco Group is not subject to any Order that adversely restricts the use, transfer, registration or licensing of any such Intellectual Property by the Alps Holdco Group.
(f) Except as disclosed on Schedule 5.18(f), each employee, agent, consultant and contractor who has contributed to or participated in the creation or development of any Intellectual Property on behalf of the Alps Holdco Group or any predecessor in interest thereto has executed a form of proprietary information and/or inventions agreement or similar written Contract with the Alps Holdco Group under which such Person: (i) has assigned all right, title and interest in and to such Intellectual Property to the Alps Holdco Group (or such predecessor in interest, as applicable); and (ii) is obligated to maintain the confidentiality of the Alps Holdco Group’s confidential information both during and after the term of such Person’s employment or engagement. To the extent any such proprietary information and/or inventions agreement or other similar written Contract permitted such employee, agent, consultant or contractor to exclude from the scope of such agreement or Contract any Intellectual Property in existence prior to the date of the employment or relationship, no such employee, agent, consultant or contractor excluded Intellectual Property that was related to the Business as currently conducted. To the Knowledge of the Alps Holdco, no employee, agent, consultant or contractor of the Alps Holdco Group is or has been in violation of any term of any such Contract.
| 38 |
(g) To the Knowledge of Alps Holdco, none of the execution, delivery or performance by Alps Holdco of this Agreement or any of the Additional Agreements to which Alps Holdco is or will be a party or the consummation by Alps Holdco of the transactions contemplated hereby or thereby will (i) cause any item of Alps Holdco Owned IP, or any item of Alps Holdco Licensed IP immediately prior to the Closing, to not be owned, licensed or available for use by the Alps Holdco Group on substantially the same terms and conditions immediately following the Closing or (ii) require any additional material payment obligations by the Alps Holdco Group in order to use or exploit any other such Intellectual Property to the same extent as the Alps Holdco Group was permitted immediately before the Closing.
(h) Except with respect to the Material Contracts listed on Schedule 5.15(a)(vi), the Alps Holdco Group is not obligated under any Contract to make any payments by way of royalties, fees, or otherwise to any owner or licensor of, or other claimant to, any Intellectual Property.
(i) The Alps Holdco Group has exercised reasonable efforts necessary to maintain, protect and enforce the secrecy, confidentiality and value of all Trade Secrets constituting Alps Holdco Owned IP and all other material Confidential Information, in each instance that are at least consistent with efforts undertaken by third Persons in the industry within which the Business is a part. No Alps Holdco IP is subject to any technology or source code escrow arrangement or obligation, and (ii) no person other than the Alps Holdco Group and their employees and contractors has a right to access or possess any source code of the Software constituting the Alps Holdco Owned IP or will be entitled to obtain access to or possession of such source code as a result of the execution, delivery and performance by Alps Holdco of this Agreement. The Alps Holdco Group is in actual possession and control of the source code of any Software constituting Alps Holdco Owned IP and all related documentation and materials.
(j) To Alps Holdco’s Knowledge, the Software that constitutes Alps Holdco Owned IP and all Software that is used by the Alps Holdco Group is free of all viruses, worms, Trojan horses and other material known contaminants and does not contain any bugs, errors, or problems of a material nature that would disrupt its operation or have an adverse impact on the operation of other Software.
(k) The Alps Holdco Group has implemented and maintained (or, where applicable, has required its vendors to maintain), consistent with commercially reasonable and industry practices and in compliance in all material respects with its contractual obligations to other Persons, reasonable security measures designed to protect, preserve and maintain the performance, security and integrity of all computers, servers, equipment, hardware, networks, Software and systems used, owned, leased or licensed by the Alps Holdco Group in connection with the operation of the Business as currently conducted (the “Alps Holdco Information Systems”). To the Alps Holdco’s Knowledge, there has been no unauthorized access to or use of the Alps Holdco Information Systems nor has there been, in the past twenty-four (24) months, any downtime or unavailability of the Alps Holdco Information Systems that resulted in a material disruption of the Business. The Alps Holdco Information Systems are adequate and sufficient (including with respect to working condition and capacity) for the operations of the Business as currently conducted. There has been no failure with respect to any Alps Holdco Information System that has had a material effect on the operations of the Alps Holdco Group.
(l) Intellectual Property exclusively licensed or assigned to customers by Alps Holdco (and derivatives thereof) is not and has not been included in any current or past products or offerings of Alps Holdco, and is not and has not been disclosed to any third party.
| 39 |
5.19 Accounts Payable; Affiliate Loans.
(a) The accounts payable of the Alps Holdco Group reflected on the Alps Holdco Financial Statements, and all accounts payable arising subsequent to the date thereof, arose from bona fide transactions in the ordinary course consistent with past practice.
(b) The information set forth on Schedule 5.19(b) separately identifies any and all accounts, receivables or notes of the Alps Holdco Group, Pubco and Merger Sub which are owed by any Affiliate of the Alps Holdco Group, Pubco or Merger Sub (excluding another member of the Alps Holdco Group). Except as set forth on Schedule 5.19(b), the Alps Holdco Group, Pubco and Merger Sub do not owe any Indebtedness to any of its Affiliates and no Affiliates owe any Indebtedness to the Alps Holdco Group, Pubco or Merger Sub (excluding any Indebtedness among Alps Holdco and/or its Subsidiaries or Pubco or Merger Sub).
5.20 Employees; Employment Matters.
(a) Schedule 5.20(a) sets forth a true, correct and complete list of each of the five highest compensated officers or employees of the Alps Holdco Group, as of the date hereof, setting forth the name, title, current base salary or hourly rate for each such person and total compensation (including bonuses and commissions) paid to each such person for the year ended March 31, 2024. Pubco and Merger Sub do not have any employees. None of the officers or directors of Pubco or Merger Sub have received compensation for their services.
(b) The Alps Holdco Group is not a party to any collective bargaining agreement, and since the formation of the first entity within the Alps Holdco Group, there has been no activity or proceeding by a labor union or representative thereof to organize any employees of the Alps Holdco Group. There is no labor strike, material slowdown or material work stoppage or lockout pending or, to the Knowledge of Alps Holdco, threatened against the Alps Holdco Group, and, since the formation of the first entity within the Alps Holdco Group, the Alps Holdco Group has not experienced any strike, material slowdown, material work stoppage or lockout by or with respect to its employees. To the Knowledge of Alps Holdco, the Alps Holdco Group is not subject to any attempt by any union to represent Alps Holdco Group employees as a collective bargaining agent.
(c) There are no pending or, to the Knowledge of Alps Holdco, threatened material Actions against Alps Holdco Group under any worker’s compensation policy or long-term disability policy. There is no material unfair labor practice charge or complaint pending or, to the Knowledge of Alps Holdco, threatened before any applicable Authority relating to employees of the Alps Holdco Group. Except as set forth on Schedule 5.20(c), since the formation of the first entity within the Alps Holdco Group, the Alps Holdco Group has not engaged in, and is not currently contemplating, any location closing, employee layoff, or relocation activities that would reasonably be expected to trigger the Worker Adjustment Retraining and Notification Act of 1988, as amended, or any similar Law, state or local statute, rule or regulation.
| 40 |
(d) The Alps Holdco Group is, and since the formation of the first entity within the Alps Holdco Group, has been, in material compliance in all material respects with all applicable Laws relating to employment of labor, including all applicable Laws relating to wages, hours, overtime, collective bargaining, equal employment opportunity, anti-discrimination, anti-harassment (including, but not limited to sexual harassment), anti-retaliation, immigration, leaves, disability rights or benefits, employment and reemployment rights of members and veterans of the uniformed services, paid time off/vacation, unemployment insurance, safety and health, workers’ compensation, pay equity, restrictive covenants, child labor, whistleblower rights, classification of employees and independent contractors, meal and rest breaks, business expenses, and the collection and payment of withholding or social security Taxes. Since the formation of the first entity within the Alps Holdco Group, no audits have been conducted, or are currently being conducted, or, to the Knowledge of the Alps Holdco, are threatened to be conducted by any Authority with respect to applicable Laws regarding employment or labor Laws. The Alps Holdco Group has complied, in all material respects, with all Laws relating to the verification of identity and employment authorization of individuals employed in Malaysia and each of the jurisdictions in which the Alps Holdco Group has employees, and none of the Alps Holdco Group currently employs, or since the formation of the first entity within the Alps Holdco Group, has employed, any Person who was not permitted to work in Malaysia or such other jurisdiction in which such Person was employed. No audit by any Authority is currently being conducted, pending or, to the Knowledge of the Alps Holdco, threatened to be conducted in respect to any foreign workers employed by the Alps Holdco Group. Except as set forth on Schedule 5.14, no employee of the Alps Holdco Group has, since the formation of the first entity within the Alps Holdco Group, brought or, to the Knowledge of Alps Holdco, threatened to bring a claim that remains pending for unpaid compensation, including overtime amounts.
(e) To the Knowledge of Alps Holdco, no key employee or officer of the Alps Holdco Group is a party to or is bound by any non-competition agreement (with any Person) that would materially interfere with the performance by such officer or key employee of any of his or her duties or responsibilities as an officer or employee of the Alps Holdco Group. To the Knowledge of Alps Holdco, no key employee or officer of the Alps Holdco Group has given written notice of their definite intent to terminate their employment with Alps Holdco, nor does Alps Holdco have any present intention to terminate the employment of any of the foregoing.
(f) Except as set forth on Schedule 5.20(f), the employment of each of the key employees is terminable at will without any penalty or severance obligation on the part of the Alps Holdco Group. All material sums due for employee compensation and all vacation time owing to any employees of the Alps Holdco Group, and all fees owing to any independent contractors and consultants, have been duly accrued on the accounting records of the Alps Holdco Group.
(g) Each current and former employee and officer, and where appropriate, each independent contractor and consultant, of the Alps Holdco Group has executed a form of proprietary information and/or inventions agreement or similar agreement. To the Knowledge of Alps Holdco, no current or former employees, officers or consultants are or were, as the case may be, in violation thereof in any material respect. Other than with respect to exclusions previously accepted by Alps Holdco involving works or inventions unrelated to the business of the Alps Holdco Group or that are otherwise immaterial, no current or former employee, officer or consultant of the Alps Holdco Group has disclosed excluded works or inventions made prior to his or her employment or consulting relationship with the Alps Holdco Group from his, her or its assignment of inventions pursuant to such employee, officer or consultant’s proprietary information and inventions agreement.
(h) With regard to any individual who performs or performed services for the Alps Holdco Group and who is not treated as an employee for Tax purposes by the Alps Holdco Group, the Alps Holdco Group has complied in all material respects with applicable Laws concerning independent contractors, including for Tax withholding purposes or Plan purposes, and the Alps Holdco Group does not have any material Liability by reason of any individual who performs or performed services for the Alps Holdco Group, in any capacity, being improperly excluded from participating in any Plan. Each individual engaged by the Alps Holdco Group as an independent contractor or consultant is, and since the formation of the first entity within the Alps Holdco Group, has been, properly classified by the Alps Holdco Group as an independent contractor, and the Alps Holdco Group has not received any notice from any Authority or Person disputing such classification.
| 41 |
(i) Except as set forth on Schedule 5.14, there is no, and since the formation of the first entity within the Alps Holdco Group, there has been no, written notice provided to the Alps Holdco Group of any pending or, to the Knowledge of the Alps Holdco, threatened claim or litigation relating to, or any complaint or allegation of, discrimination, retaliation, wrongful termination, constructive termination, harassment (including sexual harassment), sexual misconduct, or wage and hour violation against the Alps Holdco Group that remains pending; nor there is any pending obligation for the Alps Holdco Group under any settlement or out-of-court or pre-litigation arrangement relating to such matters.
(j) To the Knowledge of Alps Holdco, since the formation of the first entity within the Alps Holdco Group, the Alps Holdco Group has investigated all workplace harassment (including sexual harassment), discrimination, retaliation, and workplace violence written claims relating to current and/or former employees of the Alps Holdco Group or third-parties who interacted with current and/or former employees of the Alps Holdco Group. With respect to each such written claim with potential merit, the Alps Holdco Group has taken corrective actions. Further, to the Knowledge of the Alps Holdco, since the formation of the first entity within the Alps Holdco Group, no allegations of sexual harassment have been made to the Alps Holdco Group against any individual in his or her capacity as director or an executive officer of the Alps Holdco Group.
(k) The Alps Holdco Group has complied in all material respects with all applicable Laws regarding the COVID-19 pandemic in Malaysia regarding shelters-in-place, or similar Orders in effect as of the date hereof and have taken appropriate precautions regarding its employees. The Alps Holdco Group has promptly and thoroughly investigated all occupational safety and health complaints, issues, or inquiries related to the COVID-19 pandemic. With respect to each material occupational safety and health complaint, issue, or inquiry related to the COVID-19 pandemic, the Alps Holdco Group has taken prompt corrective action that is reasonably calculated to prevent the spread of COVID-19 within the Alps Holdco Group’s workplace.
(l) To the Knowledge of the Alps Holdco as of the date hereof and since the formation of the first entity within the Alps Holdco Group, there have been no material audits by any Authority, nor have there been any charges, fines, or penalties, including those pending or threatened, under any applicable federal, state or local occupational safety and health Law and Orders (collectively, “OSHA”) against the Alps Holdco Group. The Alps Holdco Group is in compliance in all material respects with OSHA and there are no pending appeals of any Authority’s decision or fines issued in relation to OSHA.
(m) Except as set forth on Schedule 5.20(m), the Alps Holdco Group has not paid or promised to pay any bonus to any employee in connection with the consummation of the transactions contemplated hereby.
5.21 Withholding. Except as disclosed on Schedule 5.21, all obligations of the Alps Holdco Group applicable to its employees, whether arising by operation of Law, by Contract, or attributable to payments by the Alps Holdco Group to trusts or other funds or to any Authority, with respect to unemployment compensation benefits or social security benefits for its employees through the date hereof, have been paid or adequate accruals therefor have been made on the Alps Holdco Financial Statements. Except as disclosed on Schedule 5.21, all reasonably anticipated material obligations of the Alps Holdco Group with respect to such employees (except for those related to wages during the pay period immediately prior to the Closing Date and arising in the ordinary course of business consistent with past practices), whether arising by operation of Law, by contract, by past custom, or otherwise, for salaries and holiday pay, bonuses and other forms of compensation payable to such employees in respect of the services rendered by any of them prior to the date hereof have been or will be paid by the Alps Holdco Group prior to the Closing Date.
| 42 |
5.22 Employee Benefits.
(a) Schedule 5.22(a) sets forth a correct and complete list of all Plans. With respect to each Plan, the Alps Holdco has made available to Parent and its counsel a true and complete copy, to the extent applicable, of: (i) each writing constituting a part of such Plan and all amendments thereto, including all plan documents, benefit schedules, trust agreements, and insurance contracts and other funding vehicles; (ii) the three (3) most recent annual reports on Form 5500 and accompanying schedules; (iii) the current summary plan description and any material modifications thereto; (iv) the most recent annual financial and actuarial reports; (v) the most recent determination or advisory letter received by the Alps Holdco Group from the tax authorities in Malaysia regarding the tax-qualified status of such Plan and (vi) the three (3) most recent written results of all required compliance testing.
(b) There are no pending or, to the Knowledge of the Alps Holdco, threatened Actions against or relating to the Plans, the assets of any of the trusts under such Plans or the Plan sponsor or the Plan administrator, or against any fiduciary of any Plan with respect to the operation of such Plan (other than routine benefits claims). No Plan is presently under audit or examination (nor has written notice been received of a potential audit or examination) by any Authority.
(c) Each Plan has been established, administered and funded in accordance with its terms and in compliance in all material respects with the applicable provisions of applicable Laws. All premiums due or payable with respect to insurance policies funding any Plan have been made or paid in full or, to the extent not required to be made or paid on or before the date hereof, have been fully reflected on the Alps Holdco Financial Statements.
(d) Neither the execution and delivery of this Agreement nor the consummation of the transactions contemplated hereby will (either alone or in combination with another event) (i) result in any payment becoming due, or increase the amount of any compensation or benefits due, to any current or former employee of the Alps Holdco and its Subsidiaries or with respect to any Plan; (ii) increase any benefits otherwise payable under any Plan; (iii) result in the acceleration of the time of payment or vesting of any such compensation or benefits.
(e) All Plans subject to the laws of any jurisdiction outside of the United States (i) if they are intended to qualify for special tax treatment, meet all material requirements for such treatment, and (ii) if they are intended to be funded and/or book-reserved, are fully funded and/or book reserved, as appropriate, based upon reasonable actuarial assumptions.
5.23 Real Property.
(a) Except as set forth on Schedule 5.23 and the Balance Sheets, none of the Alps Holdco Group, Pubco and Merger Sub owns, or otherwise has an interest in, any Real Property, including under any Real Property lease, sublease, space sharing, license or other occupancy agreement. The Alps Holdco Group has good, valid and subsisting title to its respective leasehold estates in the offices described on Schedule 5.23, free and clear of all Liens (other than Permitted Liens). The Alps Holdco Group has not breached or violated, in any material respect, any local zoning ordinance, and no written, or to the Alps Holdco’s Knowledge, oral notice from any Person has been received by the Alps Holdco Group or served upon the Alps Holdco Group claiming any violation of any local zoning ordinance. Pubco and Merger Sub do not hold, lease, sublease, share space, license, or have any other occupancy agreement for any real property.
| 43 |
(b) With respect to each Real Property lease set forth on Schedule 5.23: (i) it is valid, binding and in full force and effect, subject to the Enforceability Exceptions; (ii) all rents and additional rents and other sums, expenses and charges due thereunder have been paid; (iii) Alps Holdco has been in peaceable possession of the premises leased thereunder since the commencement of the original term thereof; (iv) no waiver, indulgence or postponement of Alps Holdco’s obligations thereunder has been granted by the lessor; (v) there exist no material default or event of default thereunder by the Alps Holdco Group or, to Alps Holdco’s Knowledge, by any other party thereto; (vi) there exists, to Alps Holdco’s Knowledge, no occurrence, condition or act which, with the giving of notice, the lapse of time or the happening of any further event or condition, would reasonably be expected to become a material default or event of default by the Alps Holdco Group thereunder; and (vii) there are no outstanding claims of breach or indemnification or notice of default or termination thereunder.
5.24 Tax Matters. Except as set forth on Schedule 5.24:
(a) (i) The Alps Holdco Group has duly and timely filed all Tax Returns which are required to be filed by or with respect to it, and has paid all Taxes (whether or not shown on such Tax Returns) which have become due; (ii) all such Tax Returns are true, correct and complete and accurate in all material respects; (iii) there is no Action, pending or proposed in writing, with respect to Taxes of the Alps Holdco Group; (iv) no statute of limitations in respect of the assessment or collection of any Taxes of the Alps Holdco Group for which a Lien may be imposed on any of the Alps Holdco Group’s assets has been waived or extended, which waiver or extension is in effect; (v) the Alps Holdco Group has complied in all respects with all applicable Laws relating to the reporting, payment, collection and withholding of Taxes and has duly and timely withheld or collected, paid over to the applicable Taxing Authority and reported all Taxes (including income, social, security and other payroll Taxes) required to be withheld or collected by the Alps Holdco Group; (vi) no stock transfer Tax, sales Tax, use Tax, real estate transfer Tax or other similar Tax will be imposed on the transfer of the Alps Holdco Ordinary Shares by the Parent’s stockholders pursuant to this Agreement; (vii) there is no outstanding request for a ruling from any Taxing Authority, request for consent by a Taxing Authority for a change in a method of accounting, subpoena or request for information by any Taxing Authority or agreement with any Taxing Authority with respect to the Alps Holdco Group; (viii) there is no Lien (other than Permitted Liens) for Taxes upon any of the assets of the Alps Holdco Group; (ix) no claim has ever been made by a Taxing Authority in a jurisdiction where the Alps Holdco Group has not paid any Tax or filed Tax Returns, asserting that the Alps Holdco Group is or may be subject to Tax in such jurisdiction, the Alps Holdco Group is not nor has it ever been subject to Tax in any country other than the respective countries of incorporation or formation of the Alps Holdco Group members by virtue of having a permanent establishment or other place of business in that country, and the members of the Alps Holdco Group are and have always been tax residents solely in their respective countries of incorporation or formation; (x) the Alps Holdco Group has provided to Parent true, complete and correct copies of all Tax Returns relating to, and all audit reports and correspondence relating to each proposed adjustment, if any, made by any Taxing Authority with respect to, any taxable period since the Alps Holdco’s inception; (xi) is not, and has ever been, a party to any Tax sharing, Tax indemnity or Tax allocation Contract; (xii) the Alps Holdco has not been a member of an “affiliated group” within the meaning of Section 1504(a) of the Code filing a consolidated federal income Tax Return (other than a group the common parent of which was the Alps Holdco); (xiii) the Alps Holdco Group has no liability for the Taxes of any other Person: (1) under Treasury Regulation Section 1.1502-6 (or any similar provision of applicable Law), (2) as a transferee or successor or by contract or (3) otherwise by operation of applicable Law; (xiv) to the Knowledge of Alps Holdco, no issue has been raised by a Taxing Authority in any prior Action relating to the Alps Holdco Group with respect to any Tax for any period which, by application of the same or similar principles, could reasonably be expected to result in a proposed Tax deficiency of the Alps Holdco Group for any other period; (xv) the Alps Holdco Group has not requested any extension of time within which to file any Tax Return, which Tax Return has since not been filed; (xvi) Alps Holdco is not a “United States real property holding corporation” within the meaning of Section 897(c)(2) of the Code during the applicable period specified in Section 897(c)(1)(A)(ii) of the Code; and (xvii) the Alps Holdco has not disclosed on its Tax Returns any Tax reporting position taken in any Tax Return which could result in the imposition of penalties under Section 6662 of the Code (or any comparable provisions of state, local or foreign Law) and Alps Holdco has not been a party to any “reportable transaction” or “listed transaction” as defined in Section 6707A(c) of the Code and Treasury Regulation Section 1.6011-4(b).
| 44 |
(b) The Alps Holdco Group will not be required to include any item of income or exclude any item of deduction for any taxable period ending after the Closing Date as a result of: (i) the use of, or change in, a method of accounting with respect to any transaction that occurred on or before the Closing Date; (ii) any closing agreement described in Section 7121 of the Code (or similar provision of state, local or foreign Law); (iii) any installment sale or open sale transaction disposition made in a pre-Closing Tax period; (iv) any prepaid amount received in a pre-Closing Tax period; or (v) any intercompany transaction or excess loss account described in Treasury Regulations under Section 1502 of the Code (or any corresponding or similar provision of state, local or non-U.S. income Tax law).
(c) The unpaid Taxes of the Alps Holdco Group (i) did not, as of the most recent fiscal month end, exceed the reserve for Tax liability (rather than any reserve for deferred Taxes established to reflect timing differences between book and Tax income) set forth on the Alps Holdco Financial Statements and (ii) will not exceed that reserve as adjusted for the passage of time through the Closing Date in accordance with the past custom and practice of the Alps Holdco in filing its Tax Return.
(d) The Alps Holdco Group has been in compliance in all respects with all applicable transfer pricing laws and legal requirements.
(e) Alps Holdco is not aware of any fact or circumstance that would reasonably be expected to prevent the Mergers from qualifying as a “reorganization” within the meaning of Section 368(a) of the Code.
(f) The Alps Holdco Group has not deferred the withholding or remittance of any Applicable Taxes related or attributable to any Applicable Wages for any employees of the Alps Holdco and shall not defer the withholding or remittance any Applicable Taxes related or attributable to Applicable Wages for any employees of Alps Holdco up to and through and including Closing Date, notwithstanding IRS Notice 2020-65 (or any comparable regime for state or local Tax purposes).
5.25 Environmental Laws. Since the formation of the first entity within the Alps Holdco Group, the Alps Holdco Group has complied and is in compliance with all Environmental Laws, and there are no Actions pending or, to the Knowledge of the Alps Holdco Group, threatened against the Alps Holdco Group alleging any failure to so comply.
5.26 Finders’ Fees. Except as set forth on Schedule 5.26, there is no investment banker, broker, finder or other intermediary which has been retained by or is authorized to act on behalf of the Alps Holdco Group, Pubco or Merger Sub, or any of its Affiliates who might be entitled to any fee or commission from Alps Holdco, Merger Sub, Pubco, Parent or any of their Affiliates upon consummation of the transactions contemplated by this Agreement.
5.27 Powers of Attorney, Suretyships and Bank Accounts. None of the Alps Holdco Group, Pubco or Merger Sub has any general or special powers of attorney outstanding (whether as grantor or grantee thereof) or any obligation or liability (whether actual, accrued, accruing, contingent or otherwise) as guarantor, surety, co-signer, endorser, co-maker, indemnitor or otherwise in respect of the obligation of any Person.
5.28 Directors and Officers. Schedule 5.28 sets forth a true, correct and complete list of all directors and officers of the Alps Holdco Group, Pubco and Merger Sub.
| 45 |
5.29 Anti-Money Laundering Laws.
(a) Each of the Alps Holdco Group, Pubco and Merger Sub currently is and, as applicable, since inception, has been, in compliance in all material respects with applicable Laws related to (i) anti-corruption or anti-bribery, including the U.S. Foreign Corrupt Practices Act of 1977, 15 U.S.C. §§ 78dd-1, et seq., and any other equivalent or comparable Laws of other countries (collectively, “Anti-Corruption Laws”), (ii) economic sanctions administered, enacted or enforced by any Authority (collectively, “Sanctions Laws”), (iii) export controls, including the U.S. Export Administration Regulations, 15 C.F.R. §§ 730, et seq., and any other equivalent or comparable Laws of other countries (collectively, “Export Control Laws”), (iv) anti-money laundering, including the Money Laundering Control Act of 1986, 18 U.S.C. §§ 1956, 1957, and any other equivalent or comparable Laws of other countries; (v) anti-boycott regulations, as administered by the U.S. Department of Commerce; and (vi) importation of goods, including Laws administered by the U.S. Customs and Border Protection, Title 19 of the U.S.C. and C.F.R., and any other equivalent or comparable Laws of other countries (collectively, “International Trade Control Laws”).
(b) None of the Alps Holdco Group, Pubco or Merger Sub nor, to the Knowledge of the Alps Holdco, Pubco and Merger Sub, any Representative of the Alps Holdco Group, Pubco or Merger Sub (acting on behalf of the Alps Holdco Group, Pubco or Merger Sub, as applicable), is or is acting under the direction of, on behalf of or for the benefit of a Person that is, (i) the subject of Sanctions Laws or identified on any sanctions or similar lists administered by an Authority, including the U.S. Department of the Treasury’s Specially Designated Nationals List, the U.S. Department of Commerce’s Denied Persons List and Entity List, the U.S. Department of State’s Debarred List, HM Treasury’s Consolidated List of Financial Sanctions Targets and the Investment Bank List, or any similar list enforced by any other relevant Authority, as amended from time to time, or any Person owned or controlled by any of the foregoing (collectively, “Prohibited Party”); (ii) the target of any Sanctions Laws; (iii) located, organized or resident in a country or territory that is, or whose government is, the target of comprehensive trade sanctions under Sanctions Laws, including, as of the date of this Agreement, Crimea, Cuba, Iran, North Korea, Sudan and Syria; or (iv) an officer or employee of any Authority or public international organization, or officer of a political party or candidate for political office. None of the Alps Holdco Group, Pubco or Merger Sub nor, to the Knowledge of Alps Holdco, Pubco and Merger Sub, any Representative of the Alps Holdco Group, Pubco or Merger Sub (acting on behalf of the Alps Holdco Group, Pubco or Merger Sub, as applicable), (A) has participated in any transaction involving a Prohibited Party, or a Person who is the target of any Sanctions Laws, or any country or territory that was during such period or is, or whose government was during such period or is, the target of comprehensive trade sanctions under Sanctions Laws, (B) to the Knowledge of Alps Holdco, Pubco and Merger Sub, has exported (including deemed exportation) or re-exported, directly or indirectly, any commodity, software, technology, or services in violation of any applicable Export Control Laws or (C) has participated in any transaction in violation of or connected with any purpose prohibited by Anti-Corruption Laws or any applicable International Trade Control Laws, including support for international terrorism and nuclear, chemical, or biological weapons proliferation.
(c) None of Alps Holdco, Pubco or Merger Sub has received written notice of, nor, to the Knowledge of Alps Holdco, Pubco and Merger Sub, any of its Representatives is or has been the subject of, any investigation, inquiry or enforcement proceedings by any Authority regarding any offense or alleged offense under Anti-Corruption Laws, Sanctions Laws, Export Control Laws or International Trade Control Laws (including by virtue of having made any disclosure relating to any offense or alleged offense) and, to the Knowledge of Alps Holdco, Pubco and Merger Sub there are no circumstances likely to give rise to any such investigation, inquiry or proceeding.
| 46 |
5.30 Insurance. All insurance policies owned or held by and insuring the Alps Holdco Group are set forth on Schedule 5.30, and such policies are in full force and effect. All premiums with respect to such policies covering all periods up to and including the Closing Date have been or will be paid when due, no notice of cancellation or termination has been received with respect to any such policy which was not replaced on substantially similar terms prior to the date of such cancellation or termination and there is no claim by the Alps Holdco Group or, to Alps Holdco’s Knowledge, any other Person pending under any of such insurance policies as to which coverage has been denied by the underwriters or issuers of such policies. There is no existing default or event which, with or without the passage of time or the giving of notice or both, would constitute noncompliance with, or a default under, any such policy that would entitle any insurer to terminate or cancel any such policy. The Alps Holdco Group does not have any self-insurance arrangements. Pubco and Merger Sub do not hold any insurance policies.
5.31 Related Party Transactions. Except as set forth in Schedule 5.31, as contemplated by this Agreement or as provided in the Alps Holdco Financial Statements, no Affiliate of the Alps Holdco Group, Pubco or Merger Sub, current or former director, manager or officer of any Person in the Alps Holdco Group or of Pubco and Merger Sub, or any immediate family member or Affiliate of any of the foregoing (a) is a party to any Contract, or has otherwise entered into any transaction, understanding or arrangement, with the Alps Holdco Group, Pubco or Merger Sub (in each case, excluding any employment or service agreements as an employee or director of the Alps Holdco Group, Pubco or Merger Sub), (b) owns any asset, property or right, tangible or intangible, which is used by the Alps Holdco Group, Pubco or Merger Sub, or (c) is a borrower or lender, as applicable, under any Indebtedness owed by or to the Alps Holdco Group, Pubco or Merger Sub.
5.32 Information Supplied. None of the information supplied or to be supplied by the Alps Holdco Group, Pubco or Merger Sub expressly for inclusion or incorporation by reference in the Form F-4 or the Proxy Statement, as applicable, and mailings to Parent’s stockholders with respect to the solicitation of proxies to approve the transactions contemplated by this Agreement and the Additional Agreements, if applicable, will, when filed, made available, mailed or distributed, as the case may be, contain any untrue statement of a material fact or omit to state any material fact required to be stated therein or necessary in order to make the statements therein, in light of the circumstances under which they are made, not misleading (subject to the qualifications and limitations set forth in the materials provided by the Alps Holdco Group, Pubco or Merger Sub or included in the Parent SEC Documents, the Additional Parent SEC Documents, the SEC Statement or any Other Filing). This Section 5.32 does not address projections and other forward-looking information, as to which no representations are provided herein.
5.33 Derogation. In derogation to all the representations and warranties as set out under this ARTICLE V, and except as expressly set forth in this Agreement, Alps Holdco does not represent and warrant that it or any other member of the Alps Holdco Group comply with the Law of any jurisdiction other than the jurisdiction of Malaysia.
5.34 Disclosure Letter. At any time after the date of this Agreement and before the Closing Date, Alps Holdco, Pubco and Merger Sub shall be entitled to update and modify the Disclosure Schedules by way of a disclosure letter, subject always that any such update and modification shall require the Parent’s prior written consent. The disclosure letter constitutes full, frank and formal disclosure to the Parent of facts and circumstances which may be inconsistent with, or in contradiction to, the representations and warranties of Alps Holdco, Pubco or Merger Sub, as applicable, under this Agreement and/or the Disclosure Schedule. The contents of the disclosure letter may qualify such representations and warranties and accordingly, the Parent shall not be entitled to claim that any fact, matter or circumstance causes or results in any damage or loss to the Parent or any breach of the Agreement, if such fact, matter or circumstance is disclosed in the Disclosure Schedules by way of a disclosure letter to which Parent has consented in writing.
| 47 |
ARTICLE VI
REPRESENTATIONS AND WARRANTIES OF PARENT
Except as disclosed in the Parent SEC Documents filed with or furnished to the SEC prior to the date of this Agreement and available to public access through the EDGAR database (other than any risk factor disclosures or other similar cautionary or predictive statements therein), or except as set forth in the Parent’s disclosure schedules delivered by Parent to Alps Holdco on the date of this Agreement (“Parent Disclosure Schedules”) (with specific reference to the particular section or subsection of this Agreement to which the information set forth in such Parent Disclosure Schedules relate (which qualify (a) the correspondingly numbered representation, warranty or covenant specified therein and (b) such other representations, warranties or covenants where its relevance as an exception to (or disclosure for purposes of) such other representation, warranty or covenant is reasonably apparent on its face or cross-referenced), Parent hereby represents and warrants to Alps Holdco and Pubco that each of the following representations and warranties relating to Parent are true, correct and complete as of the date of this Agreement and as of the Closing Date:
6.1 Corporate Existence and Power. Parent is a corporation duly incorporated, validly existing and in good standing under the laws of the State of Delaware.
6.2 Corporate Authorization. Parent has all requisite corporate power and authority to execute and deliver this Agreement and the Additional Agreements to which it is a party and to consummate the transactions contemplated hereby and thereby, in the case of the Mergers, subject to receipt of the Parent Stockholder Approval. The execution and delivery by Parent of this Agreement and the Additional Agreements to which it is a party and the consummation by Parent of the transactions contemplated hereby and thereby have been duly authorized by all necessary corporate action on the part of Parent. No other corporate proceedings on the part of Parent are necessary to authorize this Agreement or the Additional Agreements to which it is a party or to consummate the transactions contemplated by this Agreement (other than, in the case of the Mergers, the receipt of the Stockholder Approval) or the Additional Agreements. This Agreement and the Additional Agreements to which Parent is a party have been duly executed and delivered by Parent and, assuming the due authorization, execution and delivery by each of the other parties hereto and thereto (other than Parent), this Agreement and the Additional Agreements to which Parent is a party constitute a legal, valid and binding obligation of Parent, enforceable against Parent in accordance with their respective terms, subject to the Enforceability Exceptions. The affirmative vote of holders of a majority of the then outstanding shares of Parent Common Stock present in person or by proxy and entitled to vote at the Parent Stockholder Meeting, assuming a quorum is present (the “Parent Stockholder Approval”), is the only vote of the holders of any of Parent’s capital stock necessary to adopt this Agreement and approve the Mergers and the consummation of the other transactions contemplated hereby, except that the approval of the New PubCo M&A requires the approval of a majority of the issued and outstanding shares of Parent Common Stock and approval of the members of the Board of Directors of Parent immediately after the Closing requires a plurality of the votes cast. As of the Closing Date, the affirmative vote or written consent of Parent is the only vote of the holders of any of Merger Sub’s share capital necessary to adopt this Agreement and approve the Mergers and the consummation of the other transactions contemplated hereby.
6.3 Governmental Authorization. Assuming the accuracy of the representations and warranties of Alps Holdco set forth in Section 6.3, none of the execution, delivery or performance of this Agreement or any Additional Agreement by Parent or the consummation by Parent of the transactions contemplated hereby and thereby requires any consent, approval, license or other action by or in respect of, or registration, declaration or filing with any Authority except for (a) the filing of a premerger notification and report form by Alps Holdco under the HSR Act and the termination of the waiting period required thereunder, and (b) the filing of the Certificate of Merger with the Cayman Registrar pursuant to the Companies Act and the filing required pursuant to the HSR Act.
| 48 |
6.4 Non-Contravention. The execution, delivery and performance by Parent of this Agreement or the consummation by Parent of the transactions contemplated hereby and thereby do not and will not (a) contravene or conflict with the organizational or constitutive documents of Parent, or (b) contravene or conflict with or constitute a violation of any provision of any Law or any Order binding upon Parent; or (c) violate, conflict with, result in the loss of any benefit under, constitute a default (or an event which, with notice or lapse of time, or both, would constitute a default) under, result in the termination of or a right of termination or cancellation under, accelerate the performance required by, or result in the creation of any Lien upon any of the properties or assets of Parent under any of the terms, conditions or provisions of any contract or other agreement to which Parent is a party, or by which it or its properties or assets may be bound or affected.
6.5 Finders’ Fees. Except for the Persons identified on Schedule 6.5, there is no investment banker, broker, finder or other intermediary which has been retained by or is authorized to act on behalf of Parent or their Affiliates who might be entitled to any fee or commission from Alps Holdco or any of its Affiliates upon consummation of the transactions contemplated by this Agreement or any of the Additional Agreements.
6.6 Issuance of Shares. The Merger Consideration Shares, when issued in accordance with this Agreement, will be duly authorized and validly issued, and will be fully paid and nonassessable, free and clear of all Liens, other than restrictions arising from applicable securities Laws, any applicable Lock-Up Agreement, the Escrow Agreement, and any Liens incurred by any Alps Holdco Shareholder, and the issuance and sale of such Reincorporation Merger Surviving Company Ordinary Shares pursuant hereto will not be subject to or give rise to any preemptive rights or rights of first refusal.
6.7 Capitalization. As of the date hereof, the authorized capital stock of Parent consists of 500,000,000 shares of Parent Common Stock, par value US$0.001 per share, of which 6,007,567 are issued and outstanding, and 5,000,000 shares of preferred stock, par value US$0.001 per share, of which none is issued and outstanding. In addition, as of the date hereof, 12,070,000 Parent Warrants exercisable for 12,070,000 shares of Parent Common Stock are issued and outstanding and 12,070,000 Parent Rights are issued and outstanding. As of the date hereof, no other shares of capital stock, options, warrants, convertible securities or other rights, agreements or commitments of any character relating to the capital stock or other securities of Parent are issued, reserved for issuance or outstanding. All issued and outstanding shares of Parent Common Stock are duly authorized, validly issued, fully paid and nonassessable and are not subject to, and were not issued in violation of, any purchase option, right of first refusal, preemptive right, subscription right or any similar right under any applicable Laws of the State of Delaware, Parent’s organizational documents or any contract to which Parent is a party or by which Parent is bound. Except as set forth in Parent’s organizational documents, there are no outstanding contractual obligations of Parent to repurchase, redeem or otherwise acquire any shares of Parent Common Stock or any capital equity of Parent. There are no outstanding contractual obligations of Parent to provide funds to, or make any investment (in the form of a loan, capital contribution or otherwise) in, any other Person.
6.8 Information Supplied. None of the information supplied or to be supplied by Parent expressly for inclusion or incorporation by reference in the filings with the SEC and mailings to Parent’s stockholders with respect to the solicitation of proxies to approve the transactions contemplated by this Agreement and the Additional Agreements, if applicable, will, at the date of filing or mailing, at the time of the Parent Stockholder Meeting or at the Effective Time, as the case may be, contain any untrue statement of a material fact or omit to state any material fact required to be stated therein or necessary in order to make the statements therein, in light of the circumstances under which they are made, not misleading (subject to the qualifications and limitations set forth in the materials provided by Parent or included in the Parent SEC Documents, the Additional Parent SEC Documents, the SEC Statement or any Other Filing).
| 49 |
6.9 Trust Fund. As of the day immediately prior to the date of this Agreement, Parent has at least $$28,990,232.20 in the trust fund established by Parent for the benefit of its public stockholders (the “Trust Fund”) in a trust account (the “Trust Account”) maintained by Continental Stock Transfer & Trust Company (the “Trustee”) at Citibank, N.A., held in trust by the Trustee in cash pursuant to the Investment Management Trust Agreement dated as of December 6, 2021, as amended, between Parent and the Trustee (the “Trust Agreement”). The Trust Agreement is valid and in full force and effect and enforceable in accordance with its terms, except as may be limited by the Enforceability Exceptions, and has not been amended or modified. There are no separate agreements, side letters or other agreements or understandings (whether written or unwritten, express or implied) that would cause the description of the Trust Agreement in the Parent SEC Documents to be inaccurate in any material respect or that would entitle any Person (other than stockholders of Parent holding shares of Parent Common Stock sold in Parent’s IPO who shall have elected to redeem their shares of Parent Common Stock pursuant to Parent’s amended and restated certificate of incorporation, as amended) to any portion of the proceeds in the Trust Account. Prior to the Closing, none of the funds held in the Trust Account may be released except in accordance with the Trust Agreement and Parent’s amended and restated certificate of incorporation, as amended. Parent has performed all material obligations required to be performed by it to date under, and is not in material default or delinquent in performance or any other respect (claimed or actual) in connection with, the Trust Agreement, and, to the knowledge of Parent, no event has occurred which, with due notice or lapse of time or both, would reasonably be expected to constitute such a material default thereunder. There are no claims or proceedings pending with respect to the Trust Account.
6.10 Listing. The Parent Common Stock, Parent Units, Parent Public Warrants and Parent Public Rights are listed on the NASDAQ, with trading tickers “GLLI,” “GLLIU,” “GLLIW” and “GLLIR.”
6.11 Board Approval. Parent’s Board of Directors (including any required committee or subgroup of such board) has, as of the date of this Agreement, unanimously (i) declared the advisability of the transactions contemplated by this Agreement, (ii) determined that the transactions contemplated hereby are in the best interests of the stockholders of Parent and (iii) determined that the transactions contemplated hereby constitutes a “Business Combination” as such term is defined in Parent’s second amended and restated certificate of incorporation and bylaws (“Business Combination”) and (iv) recommended to Parent’s stockholders to adopt and approve each of the Parent Proposals (“Parent Board Recommendation”).
6.12 Parent SEC Documents and Financial Statements.
(a) Parent has filed all forms, reports, schedules, statements and other documents, including any exhibits thereto, required to be filed or furnished by Parent with the SEC since Parent’s formation under the Exchange Act or the Securities Act, together with any amendments, restatements or supplements thereto, and will use commercially reasonable efforts to file all such forms, reports, schedules, statements and other documents required to be filed subsequent to the date of this Agreement (the “Additional Parent SEC Documents”). Parent has made available to Alps Holdco copies in the form filed with the SEC of all of the following, except to the extent available in full without redaction on the SEC’s website through EDGAR for at least two (2) Business Days prior to the date of this Agreement: (i) Parent’s Annual Reports on Form 10-K for each fiscal year of Parent beginning with the first year that Parent was required to file such a form, (ii) all proxy statements relating to Parent’s meetings of stockholders (whether annual or special) held, and all information statements relating to stockholder consents, since the beginning of the first fiscal year referred to in clause (i) above, (iii) its Form 8-Ks filed since the beginning of the first fiscal year referred to in clause (i) above, and (iv) all other forms, reports, registration statements and other documents (other than preliminary materials if the corresponding definitive materials have been provided to Alps Holdco pursuant to this Section 6.12) filed by Parent with the SEC since Parent’s formation (the forms, reports, registration statements and other documents referred to in clauses (i) through (iv) above, whether or not available through EDGAR, collectively, the “Parent SEC Documents”).
| 50 |
(b) Except as to the accounting relating to the Parent Warrants, Parent SEC Documents were, and the Additional Parent SEC Documents will be, prepared in all material respects in accordance with the requirements of the Securities Act, the Exchange Act, and the Sarbanes-Oxley Act, as the case may be, and the rules and regulations thereunder. Except as to the accounting relating to the Parent Warrants, Parent SEC Documents did not, and the Additional Parent SEC Documents will not, at the time they were or are filed, as the case may be, with the SEC (except to the extent that information contained in any Parent SEC Document or Additional Parent SEC Document has been or is revised or superseded by a later filed Parent SEC Document or Additional Parent SEC Document, then on the date of such filing) contain any untrue statement of a material fact or omit to state a material fact required to be stated therein or necessary in order to make the statements made therein, in the light of the circumstances under which they were made, not misleading; provided, however, that the foregoing does not apply to statements in or omissions in any information supplied or to be supplied by the Alps Holdco Group expressly for inclusion or incorporation by reference in the SEC Statement or Other Filing. Except as to the accounting relating to the Parent Warrants, each of the financial statements (including, in each case, any notes thereto) contained or incorporated by reference in the Parent SEC Documents and Additional Parent SEC Documents is in conformity with U.S. GAAP (applied on a consistent basis), Regulation S-X and Regulation S-K, as applicable, throughout the periods indicated and each is complete and fairly presents, in all material respects, the financial position, results of operations and cash flows of Parent as at the respective dates thereof and for the respective periods indicated therein.
(c) Except as to the accounting relating to the Parent Warrants, Parent does not have any material liabilities, debts or obligations of any nature (whether accrued, fixed or contingent, liquidated or unliquidated or asserted or, to the Knowledge of Parent, unasserted), except: (a) liabilities provided for in or otherwise disclosed in the balance sheet included in the most recent financial statements of Parent or in the notes thereto, and (b) such liabilities arising in the ordinary course of Parent’s business since the date of the most recent financial statement, none of which, individually or in the aggregate, would have a Material Adverse Effect on Parent.
6.13 Certain Business Practices. Neither Parent, nor any Representative of Parent has (a) used any funds for unlawful contributions, gifts, entertainment or other unlawful expenses relating to political activity, (b) made any unlawful payment to foreign or domestic government officials, employees or political parties or campaigns, (c) violated any provision of the Foreign Corrupt Practices Act or (d) made any other unlawful payment. Neither Parent, nor any director, officer, agent or employee of Parent (nor any Person acting on behalf of any of the foregoing, but solely in his or her capacity as a director, officer, employee or agent of Parent) has, since the IPO, directly or indirectly, given or agreed to give any gift or similar benefit in any material amount to any customer, supplier, governmental employee or other Person who is or may be in a position to help or hinder Parent or assist Parent in connection with any actual or proposed transaction, which, if not given or continued in the future, would reasonably be expected to (i) adversely affect the business of Parent and (ii) subject Parent to suit or penalty in any private or governmental Action.
6.14 Anti-Money Laundering Laws. The operations of Parent are and have at all times been conducted in compliance with the Money Laundering Laws, and no Action involving Parent with respect to the Money Laundering Laws is pending or, to the knowledge of Parent, threatened.
| 51 |
6.15 Compliance with Laws. Parent is, and has since its formation been, in all material respects in compliance with all Laws applicable to it and the conduct of its business, and Parent has not received written notice alleging any violation of applicable Law in any material respect by Parent.
6.16 Affiliate Transactions. Except as contemplated by this Agreement, the Mergers and the other transactions contemplated by this Agreement, including the Additional Agreements, there are no transactions, agreements, arrangements or understandings between Parent or any of its subsidiaries, on the one hand, and any director, officer, employee, stockholder, warrant holder or Affiliate of Parent or any of its subsidiaries, on the other hand.
6.17 Litigation; Permits. There is no (a) Action pending, or, to the Knowledge of Parent, threatened against Parent or any of its Subsidiaries or that affects its or their assets or properties, or (b) Order outstanding against Parent or any of its Subsidiaries or that affects its or their assets or properties. Neither Parent nor any of its Subsidiaries is party to a settlement or similar agreement regarding any of the matters set forth in the preceding sentence that contains any ongoing obligations, restrictions or liabilities (of any nature) that are material to Parent and its Subsidiaries. Parent holds all material Permits necessary to lawfully conduct its business as presently conducted, and to own, lease and operate its assets and properties, all of which are in full force and effect.
6.18 Indebtedness. Except for as set forth in Schedule 6.18, Parent does not have any Indebtedness.
6.19 Absence of Certain Changes. As of the date of this Agreement, except as set forth in Schedule 6.19, Parent has, (a) since its formation, conducted no business other than its formation, the public offering of its securities (and the related private offerings), public reporting and its search for an initial Business Combination as described in the Prospectus (including the investigation of the Alps Holdco Group and the negotiation and execution of this Agreement) and related activities and (b) since December 31, 2022, not been subject to any Effect that would have a Material Adverse Effect on Parent.
6.20 Employees and Employee Benefit Plans. Parent does not (a) have any paid employees or (b) maintain, sponsor, contribute to or otherwise have any liability under, any employee benefit plans.
6.21 Properties. Parent does not own, license or otherwise have any right, title or interest in any material Intellectual Property. Parent does not own or lease any material real property or material personal or tangible property.
6.22 Parent Material Contracts.
(a) Except as set forth on Schedule 6.22(a), other than this Agreement, the Additional Agreements and confidentiality agreements entered into by Parent in the ordinary course of business, there are no Contracts to which Parent is a party or by which any of its properties or assets may be bound, subject or affected, which (i) creates or imposes a liability greater than $100,000, (ii) may not be cancelled by Parent on less than sixty (60) days’ prior notice without payment of a material penalty or termination fee, (iii) prohibits, prevents, restricts or impairs in any material respect any business practice of Parent as its business is currently conducted, any acquisition of material property by Parent, or restricts in any material respect the ability of Parent to solicit the personnel or customers of another Person or engage in business or compete with any other Person or (iv) otherwise imposes any obligations or restrictions on Parent after the Closing (other than customary confidentiality or indemnification obligations) (each, a “Parent Material Contract”). All Parent Material Contracts have been made available to Alps Holdco.
| 52 |
(b) With respect to each Parent Material Contract: (i) such Parent Material Contract was entered into at arms’ length and in the ordinary course of business; (ii) such Parent Material Contract is legal, valid, binding and enforceable in all material respects against Parent and, to the Knowledge of Parent, the other parties thereto, and is in full force and effect (except, in each case, as such enforcement may be limited by the Enforceability Exceptions); (iii) Parent is not in breach or default in any material respect, and no event has occurred that with the passage of time or giving of notice or both would constitute such a breach or default in any material respect by Parent, or permit termination or acceleration by the other party, under such Parent Material Contract; and (iv) to the Knowledge of Parent, no other party to any Parent Material Contract is in breach or default in any material respect, and no event has occurred that with the passage of time or giving of notice or both would constitute such a breach or default by such other party, or permit termination or acceleration by Parent under any Parent Material Contract.
6.23 Insurance. Schedule 6.23 lists all material insurance policies (by policy number, insurer, coverage period, coverage amount, annual premium and type of policy) held by Parent relating to Parent or its business, properties, assets, directors, officers and employees, copies of which have been provided to Alps Holdco. All premiums due and payable under all such insurance policies have been timely paid and Parent is otherwise in material compliance with the terms of such insurance policies. All such insurance policies are in full force and effect, and to the Knowledge of Parent, there is no threatened termination of, or material premium increase with respect to, any of such insurance policies. There have been no insurance claims made by Parent. Parent has each reported to its insurers all material claims and pending circumstances that would reasonably be expected to result in a material claim.
6.24 Investment Company Act. Parent is not an “investment company” or a Person directly or indirectly “controlled” by or acting on behalf of an “investment company”, or required to register as an “investment company”, in each case within the meaning of the Investment Company Act of 1940, as amended.
6.25 Tax Matters.
(a) (i) Parent has filed all income and all other material Tax Returns which are required to be filed by or with respect to it, and has paid all Taxes which have become due; (ii) all such Tax Returns are true, correct and complete and accurate in all material respects; (iii) there is no Action, pending or proposed in writing, with respect to Taxes of Parent; (iv) no statute of limitations in respect of the assessment or collection of any Taxes of Parent for which a Lien may be imposed on any of Parent’s assets has been waived or extended, which waiver or extension is in effect; (v) there is no outstanding request for a ruling from any Taxing Authority, request for consent by a Taxing Authority for a change in a method of accounting, subpoena or request for information by any Taxing Authority or agreement with any Taxing Authority with respect to Parent; (vi) there is no Lien (other than Permitted Liens) for Taxes upon any of the assets of Parent; (vii) no claim has ever been made by a Taxing Authority in a jurisdiction where Parent has not paid any Tax or filed Tax Returns, asserting that Parent is or may be subject to Tax in such jurisdiction, Parent is not nor has it ever been subject to Tax in any country other than the respective countries of incorporation or formation of Parent members by virtue of having a permanent establishment or other place of business in that country, and the members of Parent are and have always been tax residents solely in their respective countries of incorporation or formation; (viii) Parent has made available to Alps Holdco true, complete and correct copies of all Tax Returns relating to, and all audit reports and correspondence relating to each proposed adjustment, if any, made by any Taxing Authority with respect to, any taxable period since its formation; (ix) there is no outstanding power of attorney from Parent authorizing anyone to act on behalf of Parent in connection with any Tax, Tax Return or Action relating to any Tax or Tax Return of Parent; (x) Parent is not, and has ever been, a party to any Tax sharing, Tax indemnity or Tax allocation Contract (other than a contract entered into in the ordinary course of business consistent with past practices, the primary purpose of which is not related to Taxes); (xi) Parent has not been a member of an “affiliated group” within the meaning of Section 1504(a) of the Code filing a consolidated federal income Tax Return (other than a group the common parent of which was Parent); (xii) Parent has no liability for the Taxes of any other Person: (1) under Treasury Regulation Section 1.1502-6 (or any similar provision of applicable Law), (2) as a transferee or successor or by contract or (3) otherwise by operation of applicable Law; (xiii) Parent has not requested any extension of time within which to file any Tax Return, which Tax Return has since not been filed; (xiv) Parent is not a “United States real property holding corporation” within the meaning of Section 897(c)(2) of the Code during the applicable period specified in Section 897(c)(1)(A)(ii) of the Code; and (xv) Parent has not been a party to any “listed transaction” as defined in Section 6707A(c)(2) of the Code and Treasury Regulation Section 1.6011-4(b)(2).
| 53 |
(b) Parent will not be required to include any item of income or exclude any item of deduction for any taxable period ending after the Closing Date as a result of: (i) the use of, or change in, a method of accounting with respect to any transaction that occurred on or before the Closing Date (ii) any closing agreement described in Section 7121 of the Code (or similar provision of state, local or foreign Law); (iii) any installment sale or open sale transaction disposition made in a pre-Closing Tax period; (iv) any prepaid amount received in a pre-Closing Tax period; or (v) any intercompany transaction or excess loss account described in Treasury Regulations under Section 1502 of the Code (or any corresponding or similar provision of state, local or non-U.S. income Tax law).
(c) The unpaid Taxes of Parent (i) did not, as of the most recent fiscal month end, exceed the reserve for Tax liability (rather than any reserve for deferred Taxes established to reflect timing differences between book and Tax income) set forth on the Alps Holdco Financial Statements and (ii) will not exceed that reserve as adjusted for the passage of time through the Closing Date in accordance with the past custom and practice of Parent in filing its Tax Return.
(d) Parent has been in compliance in all respects with all applicable transfer pricing laws and legal requirements.
(e) Parent has not taken any action (nor permitted any action to be taken), and to Parent’s Knowledge, there is no fact or circumstance that would reasonably be expected to prevent the Mergers from qualifying as a “reorganization” within the meaning of Section 368(a) of the Code.
6.26 PIPE Financing. Parent has executed Subscription Agreements with the PIPE Investors for them to purchase the PIPE Shares for an aggregate investment equal to the PIPE Financing Amount. Each of the PIPE Investors has represented that it is an “accredited investor” (within the meaning of Rule 501(a) under the Securities Act) or a “qualified institutional buyer” (within the meaning of Rule 144A under the Securities Act). True and complete executed copies of each Subscription Agreement have been delivered to Alps Holdco on or prior to the date hereof. Each of the Subscription Agreements (a) have been duly authorized, executed and delivered by Parent, (b) are in full force and effect and have not been withdrawn, terminated or otherwise amended or modified (and no such withdrawal, termination, amendment or modification is contemplated by Parent) and (c) constitute a legal, valid and binding obligation of Parent, enforceable against Parent, and, to the knowledge of Parent, the other parties thereto, in accordance with their terms. There are no other agreements, side letters, or arrangements between Parent and any PIPE Investor relating to any Subscription Agreement or the purchase by such PIPE Investor of securities of Parent, that could affect the obligation of the PIPE Investors to acquire their portion of the PIPE Shares as set forth in the applicable Subscription Agreement, and, as of the date hereof, Parent does not know of any facts or circumstances that may reasonably be expected to result in any of the conditions set forth in any Subscription Agreements not being satisfied, or the PIPE Investment Amount not being available to Parent, on the Closing Date. No event has occurred that, with or without notice, lapse of time or both, would constitute a default or breach on the part of Parent under any material term or condition of any Subscription Agreement and, as of the date hereof, Parent has no reason to believe that it will be unable to satisfy in all material respects on a timely basis any term or condition of closing to be satisfied by it contained in any Subscription Agreement. The Subscription Agreements contain all of the conditions precedent to the obligations of the PIPE Investors to acquire the PIPE Shares on the terms set forth therein. No fees, consideration (other than PIPE Shares) or other discounts are payable or have been agreed by Parent to any PIPE Investor in respect of its PIPE Shares to be acquired under its Subscription Agreement.
| 54 |
6.27 CFIUS Foreign Person Status. Parent represents and warrants that Sponsor is not a “foreign person” within the meaning of the Defense Production Act of 1950, as amended, including all implementing regulations thereof.
ARTICLE VII
COVENANTS OF THE PARTIES PENDING CLOSING
7.1 Conduct of the Business. Each of Alps Holdco and Parent covenants and agrees that:
(a) Except as expressly contemplated by this Agreement or the Additional Agreements or as set forth on Schedule 7.1(a) , from the date hereof until the earlier of the Closing Date and the termination of this Agreement in accordance with its terms (the “Interim Period”), each party shall (I) conduct its business only in the ordinary course (including the payment of accounts payable and the collection of accounts receivable), consistent with past practices, (II) duly and timely file all material Tax Returns required to be filed (or obtain a permitted extension with respect thereto) with the applicable Taxing Authorities and pay any and all Taxes due and payable during such time period; (III) duly observe and conform in with all applicable Law, including the Exchange Act, and Orders, in each case, in all material respects, and (IV) use commercially reasonable efforts to preserve intact its business relationships with employees, clients, suppliers, contract manufacturing organizations, contract research organizations and other third parties. Without limiting the generality of the foregoing, and except as expressly contemplated by this Agreement or the Additional Agreements, as required by applicable Law, or as set forth on Schedule 7.1(a) , from the date hereof until the earlier of the Closing Date and the termination of this Agreement in accordance with its terms, without the other party’s prior written consent (which shall not be unreasonably conditioned, withheld or delayed), neither Alps Holdco nor Parent shall, or permit its Subsidiaries to:
(i) amend, modify or supplement its memorandum or articles of association, certificate of incorporation or bylaws or other organizational or governing documents except as contemplated hereby, or engage in any reorganization, reclassification, liquidation, dissolution or similar transaction;
(ii) amend, waive any provision of, terminate prior to its scheduled expiration date, or otherwise compromise in any material way or relinquish any material right under, any material contract, agreement, lease, license or other right or asset of such party or its Subsidiaries;
(iii) other than in the ordinary course of business, modify, amend or enter into any contract, agreement, lease, license or commitment, including for capital expenditures, that extends for a term of one year or more and obligates the payment by it of more than $100,000 (individually or in the aggregate);
(iv) make any capital expenditures in excess of $100,000 (individually or in the aggregate);
| 55 |
(v) sell, lease, license or otherwise dispose of any of its material assets, except pursuant to existing contracts or commitments disclosed herein or in the ordinary course of business consistent with past practices;
(vi) solely in the case of Alps Holdco, sell, lease, license or otherwise dispose of any of any material Alps Holdco Owned IP outside of the ordinary course of business consistent with past practices;
(vii) solely in the case of Alps Holdco, permit any material Registered Owned IP to go abandoned or expire for failure to make an annuity or maintenance fee payment, or file any necessary paper or action to maintain such rights;
(viii) (A) pay, declare or promise to pay any dividends or other distributions with respect to its capital stock or other equity securities; or (B) except as contemplated hereby or by any Additional Agreement, amend any material term, right or obligation with respect to any outstanding shares of its capital stock or other equity securities;
(ix) (A) make any loan, advance or capital contribution in excess of $100,000 to any Person; (B) incur any Indebtedness in excess of $100,000 including drawings under the lines of credit, if any, other than (1) loans evidenced by promissory notes made to Parent as working capital advances as described in the Prospectus and (2) intercompany Indebtedness; or (C) repay or satisfy any Indebtedness in excess of $100,000, other than the repayment of Indebtedness in accordance with the terms thereof;
(x) suffer or incur any Lien in excess of $100,000, except for Permitted Liens, on its assets;
(xi) delay, accelerate or cancel, or waive any material right with respect to, any receivables or Indebtedness in excess of $100,000 owed to it, or write off or make reserves in excess of $250,000 against the same (other than, in the case of the Alps Holdco Group, in the ordinary course of business consistent with past practices);
(xii) merge or consolidate or enter a similar transaction with, or acquire all or substantially all of the assets or business of, any other Person; make any material investment in any Person; or be acquired by any other Person;
(xiii) terminate or allow to lapse any material insurance policy protecting any of its material assets, unless simultaneously with such termination or lapse, a replacement policy underwritten by an insurance company of nationally recognized standing having comparable deductions and providing coverage equal to or greater than the coverage under the terminated or lapsed policy for substantially similar premiums or less is in full force and effect;
(xiv) adopt any severance, retention or other employee plan in excess of $100,000 in the aggregate, or fail to continue to make timely contributions to each Plan in accordance with the terms thereof;
(xv) institute, settle or agree to settle any Action before any Authority, in each case in excess of $100,000 (exclusive of any amounts covered by insurance) or that imposes material injunctive or other material non-monetary relief on it;
| 56 |
(xvi) except as required by IFRS, make any material change in its accounting principles, methods or practices or write down in any material respect the value of its assets;
(xvii) change its principal place of business or jurisdiction of organization;
(xviii) issue, redeem or repurchase any shares, capital stock, membership interests or other securities, or issue any securities exchangeable for or convertible into any shares of its capital stock or other securities, other than with respect to the Alps Holdco Group, the issuance of Alps Holdco Ordinary Shares upon the exercise or conversion of outstanding Alps Holdco convertible securities;
(xix) (A) make, change or revoke any material Tax election; (B) change any annual Tax accounting periods in any material respect; (C) settle or compromise any material claim, notice, audit report or assessment in respect of Taxes of the Alps Holdco Group; (D) enter into any Tax allocation, Tax sharing, Tax indemnity or other closing agreement relating to any Taxes of the Alps Holdco Group; or (E) surrender or forfeit any right to claim a material Tax refund;
(xx) enter into any material transaction with or distribute or advance any material assets or property to any of its Affiliates (other than its Subsidiaries), other than the payment of salary and benefits in the ordinary course;
(xxi) other than as required by a Plan, (A) materially increase or change the compensation or benefits of any employee or service provider of the Alps Holdco Group other than in the ordinary course of business, (B) accelerate the vesting or payment of any material compensation or benefits of any employee or service provider of the Alps Holdco Group, (C) make any loan to any present or former employee or other individual service provider of the Alps Holdco Group, other than advancement of expenses in the ordinary course of business consistent with past practices, or (D) enter into, amend or terminate any collective bargaining agreement or other material agreement with a labor union or labor organization;
(xxii) fail to duly observe and conform to any applicable Laws and Orders in any material respect; or
(xxiii) agree or commit to do any of the foregoing.
(b) Notwithstanding the foregoing, Alps Holdco and Parent and their respective Subsidiaries shall be permitted to take any and all actions required to comply in all material respects with the quarantine, “shelter in place,” “stay at home,” workforce reduction, social distancing, shut down, closure, sequester or any other Law, directive, guidelines or recommendations by any Authority (including the Centers for Disease Control and the World Health Organization) in each case in connection with, related to or in response to COVID-19, including the Coronavirus Aid, Relief, and Economic Security (CARES) Act or any changes thereto.
(c) Nothing contained herein shall be deemed to give Parent or Parent Representative, directly or indirectly, the right to control or direct Alps Holdco or any operations of Alps Holdco prior to the Closing. Prior to the Closing, Alps Holdco and Parent shall exercise, consistent with the terms and conditions hereof, control over their respective businesses and operations.
| 57 |
7.2 Exclusivity.
(a) During the Interim Period, neither Alps Holdco, on the one hand, nor Parent, on the other hand, shall, and such Persons shall cause each of their respective Representatives not to, without the prior written consent of the other party (which consent may be withheld in the sole and absolute discretion of the party asked to provide consent), directly or indirectly, (i) encourage, solicit, initiate, engage or participate in negotiations with any Person concerning any Alternative Transaction, (ii) take any other action intended or designed to facilitate the efforts of any Person relating to a possible Alternative Transaction or (iii) approve, recommend or enter into any Alternative Transaction or any contract or agreement related to any Alternative Transaction. Immediately following the execution of this Agreement, Alps Holdco, on the one hand, and Parent, on the other hand, shall, and shall cause each of its Representatives, to terminate any existing discussion or negotiations with any Persons other than Alps Holdco or Parent, as applicable, concerning any Alternative Transaction. Each of Alps Holdco and Parent shall be responsible for any acts or omissions of any of its respective Representatives that, if they were the acts or omissions of Alps Holdco or Parent, as applicable, would be deemed a breach of such party’s obligations hereunder (it being understood that such responsibility shall be in addition to and not by way of limitation of any right or remedy Alps Holdco or Parent, as applicable, may have against such Representatives with respect to any such acts or omissions). For purposes of this Agreement, the term “Alternative Transaction” means any of the following transactions involving Alps Holdco or Parent or their respective Subsidiaries (other than the transactions contemplated by this Agreement or the Additional Agreements): (A) any merger, consolidation, share exchange, business combination or other similar transaction (other than between or among such party and/or its wholly-owned Subsidiaries), (B) any sale, lease, exchange, transfer or other disposition of all or a material portion of the assets of such Person or any material portion of the capital stock or other equity interests of such party or its Subsidiaries in a single transaction or series of transactions or (C) with respect to Parent, any other Business Combination.
(b) In the event that there is an unsolicited proposal for, or an indication of interest in entering into, an Alternative Transaction, communicated in writing to Alps Holdco or Parent or any of their respective Representatives (each, an “Alternative Proposal”), such party shall as promptly as practicable (and in any event within three (3) Business Days after receipt thereof) advise the other parties to this Agreement, orally and in writing, of such Alternative Proposal and the material terms and conditions thereof (including any changes thereto) and the identity of the Person making any such Alternative Proposal. Alps Holdco and Parent shall keep each other informed on a reasonably current basis of material developments with respect to any such Alternative Proposal. As used herein with respect to Parent, the term “Alternative Proposal” shall not include the receipt by Parent of any unsolicited communications (including the receipt of draft non-disclosure agreements) in the ordinary course of business consistent with past practices inquiring as to Parent’s interest in a potential target for a Business Combination; provided, however, that Parent shall inform the person initiating such communication of the existence of this Agreement and its obligations hereunder.
7.3 Access to Information. During the Interim Period, Alps Holdco and Parent shall each, use commercially reasonable efforts to, (a) continue to give the other party, its legal counsel and its other Representatives reasonable access to the offices, properties and Books and Records, (b) furnish to the other party, its legal counsel and its other Representatives such information relating to the business of the Alps Holdco Group and Parent as such Persons may reasonably request and (c) cause its employees, legal counsel, accountants and other Representatives to reasonably cooperate with the other party in its investigation of the Business (in the case of Alps Holdco) or the business of Parent (in the case of Parent); provided, that no investigation pursuant to this Section 7.3 (or any investigation made prior to the date hereof) shall affect any representation or warranty given by Alps Holdco or Parent; and provided, further, that any investigation pursuant to this Section 7.3 shall be conducted in such manner as not to interfere unreasonably with the conduct of the Business of the Alps Holdco Group. Notwithstanding anything to the contrary expressed or implied in this Agreement, neither party shall be required to provide the access described above or disclose any information to the other party if doing so is, in such party’s reasonable judgement, reasonably likely to (i) result in a waiver of attorney-client privilege, work product doctrine or similar privilege or (ii) violate any contract to which it is a party or to which it is subject or applicable Law.
| 58 |
7.4 Notices of Certain Events. During the Interim Period, each of Parent and Alps Holdco shall promptly notify the other party of:
(a) any notice from any Person alleging or raising the possibility that the consent of such Person is or may be required in connection with the transactions contemplated by this Agreement or that the transactions contemplated by this Agreement might give rise to any Action or other rights by or on behalf of such Person or result in the loss of any rights or privileges of Alps Holdco (or Pubco, post-Closing) to any such Person or create any Lien on any of the Alps Holdco Group’s or Parent’s assets;
(b) any notice or other material communication from any Authority in connection with the transactions contemplated by this Agreement or the Additional Agreements;
(c) any Actions commenced or threatened in writing against, relating to or involving or otherwise affecting either party or any of their stockholders or their equity, assets or business and that relate to the consummation of the transactions contemplated by this Agreement or the Additional Agreements;
(d) the occurrence of any fact or circumstance which constitutes or results, or would reasonably be expected to constitute or result in a Material Adverse Effect, upon having actual Knowledge of such occurrence; and
(e) any inaccuracy of any representation or warranty of such party contained in this Agreement at any time during the term hereof, or any failure of such party to comply with or satisfy any covenant, condition or agreement to be complied with or satisfied by it hereunder, in either case, upon having actual Knowledge of such occurrence, that would reasonably be expected to cause any of the conditions set forth in Sections 10.2(a), 10.2(c) or 10.2(c), with respect to Alps Holdco’s obligations under this Section 7.4(e), or Sections 10.3(a) or 10.3(b) with respect to Parent’s obligations under this Section 7.4(e), not to be satisfied.
7.5 Cooperation with Form F-4/Proxy Statement; Other Filings.
(a) As soon as reasonably practicable following the date of this Agreement, (i) Parent (with the assistance and cooperation of Alps Holdco) shall prepare and Pubco shall file with the SEC a registration statement on Form F-4 (as amended or supplemented from time to time, and including the Proxy Statement contained therein, the “Registration Statement”) in connection with the registration under the Securities Act of the Reincorporation Merger Surviving Company Ordinary Shares to be issued under this Agreement to the holders of Alps Holdco Ordinary Shares and holders of the Parent Common Stock prior to the Reincorporation Merger Effective Time and Effective Time, which Registration Statement will also contain a proxy statement of Parent (as amended, the “Proxy Statement”) for the purpose of soliciting proxies or votes from holders of Parent Common Stock sufficient to obtain Parent Stockholder Approval at a meeting of holders of Parent Common Stock to be called and held for such purpose (the “Parent Stockholder Meeting”). The Proxy Statement shall be included in the Registration Statement filed by Parent and Pubco with the SEC, pursuant to which the Reincorporation Merger Surviving Company Ordinary Shares issuable in the Mergers shall be registered. Parent and Pubco shall promptly respond to any SEC comments on the Registration Statement, and which the Registration Statement will also include a consent solicitation for the Company Shareholders to obtain the Company Shareholder Approval pursuant to the Company Shareholder Written Consent. The Proxy Statement, the Registration Statement and the documents included or referred to therein, together with any supplements, amendments or exhibits thereto, are referred to herein as the “Offer Documents”.
| 59 |
(b) Parent and Pubco (i) shall permit Alps Holdco and its counsel to review and comment on the Offer Documents and any exhibits, amendments or supplements thereto (or other related documents); (ii) shall consider any such comments reasonably and in good faith; and (iii) shall not file the Offer Documents or any exhibit, amendment or supplement thereto without the prior written consent of Alps Holdco (not to be unreasonably withheld, delayed or conditioned). As promptly as practicable after receipt thereof, Parent shall provide to Alps Holdco and its counsel notice and a copy of all correspondence (or, to the extent such correspondence is oral, a summary thereof), including any comments from the SEC or its staff, between Parent or any of its Representatives, on the one hand, and the SEC or its staff or other government officials, on the other hand, with respect to the Offer Documents, and, in each case, shall consult with Alps Holdco and its counsel concerning any such correspondence. Parent and Pubco shall not file any response letters to any comments from the SEC without the prior written consent of Alps Holdco (not to be unreasonably withheld, delayed or conditioned). Parent and Pubco will use their reasonable efforts to permit Alps Holdco’s counsel to participate in any calls, meetings or other communications with the SEC or its staff. Parent and Pubco will advise Alps Holdco, promptly after it receives notice thereof, of the time when the Offer Documents or any amendment or supplement thereto has been filed with the SEC and the time when the Registration Statement declared effective or any stop order relating to the Registration Statement is issued.
(c) As soon as practicable following the date on which the Registration Statement is declared effective by the SEC (the “F-4 Effective Date”), Parent shall distribute the Proxy Statement to the holders of Parent Common Stock and Alps Holdco Shareholders and, pursuant thereto, shall call the Parent Stockholder Meeting in accordance with its organizational documents and the laws of the State of Delaware and, subject to the other provisions of this Agreement, solicit proxies from such holders to vote in favor of the adoption and approval of this Agreement and the transactions contemplated hereby and the other matters presented to the Parent Stockholders for approval or adoption at the Parent Stockholder Meeting, including the matters described in Section 7.5(e).
(d) Parent and Alps Holdco shall comply with all applicable provisions of and rules under the Securities Act and Exchange Act and all applicable Laws of the State of Delaware or the Cayman Islands, as applicable, and the NASDAQ, in the preparation, filing and distribution of the Registration Statement and the Proxy Statement (or any amendment or supplement thereto), as applicable, the solicitation of proxies under the Proxy Statement and the calling and holding of the Parent Stockholder Meeting. Without limiting the foregoing, Parent shall ensure that each of the Registration Statement, as of the F-4 Effective Date, and the Proxy Statement, as of the date on which it is first distributed to Parent Stockholders, and as of the date of the Parent Stockholder Meeting, does not contain any untrue statement of a material fact or omit to state a material fact necessary in order to make the statements made, in light of the circumstances under which they were made, not misleading (provided, that Parent shall not be responsible for the accuracy or completeness of any information relating to Alps Holdco (or any other information) that is furnished by Alps Holdco expressly for inclusion in the Proxy Statement). If at any time prior to the Effective Time, a change in the information relating to Alps Holdco or any other information furnished by Parent, Pubco, Merger Sub or Alps Holdco for inclusion in the Proxy Statement, which would make the preceding sentence incorrect, should be discovered by Parent, Pubco, Merger Sub or Alps Holdco, as applicable, such party shall promptly notify the other parties of such change or discovery and an appropriate amendment or supplement describing such information shall be promptly filed with the SEC and, to the extent required by Law, disseminated to Parent’s stockholders. In connection therewith, Parent, Pubco, Merger Sub and Alps Holdco shall instruct their respective employees, counsel, financial advisors, auditors and other authorized representatives to reasonably cooperate with Parent as relevant if required to achieve the foregoing.
| 60 |
(e) In accordance with Parent’s second amended and restated certificate of incorporation and applicable securities laws, rules and regulations, including the Delaware General Corporation Law and rules and regulations of the NASDAQ, in the Proxy Statement, Parent shall seek from the holders of shares of Parent Common Stock the approval the following proposals: (i) the Parent Stockholder Approval of the Mergers; (ii) adoption and approval of the New Pubco M&A; (iii) approval on a non-binding advisory basis, several separate proposals relating to the differences between Parent’s charter as currently in effect and the New Pubco M&A; (iv) approval of the members of the Board of Directors of Pubco immediately after the Closing, (v) approval of the issuance of more than 20% of the issued and outstanding Reincorporation Merger Surviving Company Ordinary Shares to (a) Alps Holdco Shareholders in connection with the Merger and (b) the PIPE Investors in connection with the PIPE Financing, under applicable exchange listing rules; (vi) approval to adjourn the Parent Stockholder Meeting, if necessary; (vii) all required approvals under the NASDAQ rules of the issuance of the Reincorporation Merger Surviving Company Ordinary Shares in connection with the Subscription Agreements; and (viii) approval to obtain any and all other approvals necessary or advisable to effect the consummation of the Mergers as reasonably determined by Alps Holdco and Parent (the proposals set forth in the forgoing clauses (i) through (viii) collectively, the “Parent Proposals”).
(f) Parent and Pubco, with the assistance of Alps Holdco, shall use their reasonable efforts to cause the Registration Statement and the Proxy Statement to “clear” comments from the SEC and the Registration Statement to become effective as promptly as reasonably practicable thereafter. As soon as practicable after the Registration Statement and the Proxy Statement is “cleared” by the SEC, Parent shall cause the Proxy Statement, together will all other Offer Documents, to be disseminated to holders of shares of Parent Common Stock. The Offer Documents shall provide the public stockholders of Parent with the opportunity to redeem all or a portion of their public shares of Parent Common Stock, up to that number of shares of Parent Common Stock that would permit Parent to maintain consolidated net tangible assets of at least $5,000,001 either immediately prior to or upon the consummation of the Mergers, at a price per share equal to the pro rata share of the funds in the Trust Account, all in accordance with and as required by Parent’s amended and restated certificate of incorporation, as amended, the Trust Agreement, applicable Law and any applicable rules and regulations of the SEC. In accordance with Parent’s amended and restated certificate of incorporation, as amended, the proceeds held in the Trust Account will first be used for the redemption of the shares of Parent Common Stock held by Parent’s public stockholders who have elected to redeem such shares.
(g) Parent shall call and hold the Parent Stockholder Meeting as promptly as practicable after the F-4 Effective Date for the purpose of seeking the approval of each of the Parent Proposals, and Parent shall consult in good faith with Alps Holdco with respect to the date on which such meeting is to be held. Parent shall not postpone or adjourn the Parent Stockholder Meeting without the prior written consent of Alps Holdco (which shall not be unreasonably withheld, delayed or conditioned) unless the requisite quorum is not obtained. Parent shall use reasonable efforts to solicit from its stockholders proxies in favor of the approval and adoption of the Mergers and this Agreement and the other Parent Proposals. Parent’s Board of Directors shall recommend that the Parent Stockholders vote in favor of the Parent Proposals, and neither Parent’s Board of Directors, nor any committee thereof, shall withhold, withdraw, amend, modify, change or propose or resolve to withhold, withdraw, amend, modify or change, in each case in a manner adverse to Alps Holdco, such recommendation.
(h) In connection with the preparation and filing of the Registration Statement and any amendments thereto, Alps Holdco Group shall cooperate with Parent and Pubco and shall make their directors, officers and appropriate senior employees reasonably available to Parent and Pubco and their respective counsel in connection with the drafting of such filings and mailings and responding in a timely manner to comments from the SEC.
| 61 |
(i) Notwithstanding anything else to the contrary in this Agreement or any Additional Agreements, Parent and Pubco may make any public filing with respect to the Mergers to the extent required by applicable Law, provided that prior to making any filing that includes information regarding the Alps Holdco Group, Parent and Pubco shall provide a copy of the filing to Alps Holdco and permit Alps Holdco to make revisions to protect confidential or proprietary information of the Alps Holdco Group.
7.6 Trust Account. Parent covenants that it shall cause the funds in the Trust Account to be disbursed in accordance with the Trust Agreement, including for the payment of (a) all amounts payable to public holders of shares of Parent Common Stock and/or Parent Public Rights who shall have validly redeemed their shares of Parent Common Stock and/or Parent Public Rights (the “Parent Redemption Amount”), (b) deferred underwriting commissions and the expenses of Parent and the Alps Holdco Group to the third parties to which they are owed, and (c) the remaining monies in the Trust Account to Pubco or the Reincorporation Merger Surviving Company after the Closing.
7.7 Obligations of Merger Sub and Pubco. Alps Holdco and Pubco shall take all action necessary to cause Pubco and Merger Sub to perform their respective obligations under this Agreement and to consummate the transactions contemplated under this Agreement, upon the terms and subject to the conditions set forth in this Agreement, including the delivery to Parent as promptly as reasonably practicable (but in any event within three (3) Business Days following the execution of this Agreement or such further period as may be mutually agreed upon by Alps Holdco, Merger Sub and Pubco) of (i) the unanimous resolutions in writing of the shareholders of Merger Sub approving, in accordance with Merger Sub’s memorandum and articles of association and the Companies Act, this Agreement, the Merger and the other transactions contemplated by this Agreement and the performance of the obligations of Merger Sub hereunder (the “Merger Sub Shareholder Approval”) and (ii) the unanimous resolutions in writing of the shareholders of Pubco approving, in accordance with Pubco’s memorandum and articles of association and the Companies Act, this Agreement, the Mergers, the draft of the Reincorporation Plan of Merger, the authorization, filing and registration of the Reincorporation Plan of Merger, the adoption of the New Pubco M&A and the relevant change of authorized share capital and change of company name, and the other transactions contemplated by this Agreement and the performance of the obligations of Pubco hereunder (the “Pubco Shareholder Approval”).
7.8 Cooperation with Antitrust Law Approvals. Promptly after the date hereof, to the extent required by applicable Law, Parent and Alps Holdco shall each prepare and file the notification required of it under the HSR Act in connection with the transactions contemplated by this Agreement and shall promptly and in good faith respond to all information requested of it by the U.S. Federal Trade Commission, U.S. Department of Justice, or any other Authority in connection with such notification and otherwise cooperate in good faith with each other and such Authorities. Each party will promptly furnish to the other such information and assistance as the other may reasonably request in connection with its preparation of any filing or submission that is necessary under the HSR Act will use commercially reasonable efforts to cause the expiration or termination of the applicable waiting periods as soon as practicable, including, if appropriate, by requesting early termination of the HSR waiting period. Without limiting the foregoing, Parent and Alps Holdco shall: (i) promptly inform the other of any communication to or from the U.S. Federal Trade Commission, the U.S. Department of Justice or any other Authority with respect to Antitrust Laws regarding the transactions contemplated by this Agreement; (ii) permit each other to review reasonably in advance any proposed substantive written communication to any such Authority and incorporate reasonable comments thereto; (iii) give the other prompt written notice of the commencement of any Action with respect to such transactions under Antitrust Laws; (iv) not agree to participate in any substantive meeting or discussion with any such Authority in respect of any filing, investigation or inquiry concerning this Agreement or the transactions contemplated by this Agreement with respect to Antitrust Laws unless, to the extent reasonably practicable, it consults with the other party in advance and, to the extent permitted by such Authority, gives the other party the opportunity to attend; (v) keep the other reasonably informed as to the status of any such Action; and (vi) promptly furnish each other with copies of all correspondence, filings (except for filings made under the HSR Act) and written communications (and memoranda setting forth the substance of all substantive oral communications) between such party and their Subsidiaries and their respective Representatives and advisors, on one hand, and any such Authority, on the other hand, in each case, with respect to this Agreement and the transactions contemplated by this Agreement with respect to Antitrust Laws; provided that materials required to be supplied pursuant to this section may be redacted (1) as necessary to comply with contractual arrangements, (2) as necessary to comply with applicable Law, and (3) as necessary to address reasonable privilege or confidentiality concerns; provided further, that a party may reasonably designate any competitively sensitive material provided to another party under this Section 7.8 as “Outside Counsel Only”.
| 62 |
7.9 Subscription Agreements. Unless otherwise approved in writing by Alps Holdco (not to be unreasonably withheld, conditioned or delayed), Parent shall not (a) reduce the PIPE Financing Amount or the amount of PIPE Shares under Any Subscription Agreement or reduce or impair the rights of Parent under any Subscription Agreement or (b) permit any amendment or modification to be made to, any waiver (in whole or in part) of, or provide consent to modify (including consent to terminate), any provision or remedy under, or any replacements of, any of the Subscription Agreements, in each case, other than any assignment or transfer contemplated therein or expressly permitted thereby (without any further amendment, modification or waiver to such assignment or transfer provision); provided, that, in the case of any such assignment or transfer, the initial party to such Subscription Agreement remains bound by its obligations with respect thereto in the event that the transferee or assignee, as applicable, does not comply with its obligations thereunder. Subject to the conditions in the Subscription Agreements having been satisfied, Parent and Pubco shall use commercially reasonable efforts to take, or to cause to be taken, all actions required, necessary or that it otherwise deems to be proper or advisable to consummate the transactions contemplated by the Subscription Agreements on the terms described therein, including using commercially reasonable efforts to enforce its rights under each Subscription Agreement to cause the applicable PIPE Investor to pay to Parent (either directly or to an escrow account established by Parent) for the PIPE Shares under such PIPE Investor’s Subscription Agreement in accordance with its terms.
ARTICLE VIII
COVENANTS OF ALPS HOLDCO
8.1 No Insider Trading. During the Interim Period, Alps Holdco shall not, and it shall direct its Representatives to not, directly or indirectly, (a) purchase or sell (including entering into any hedge transaction with respect to) any shares of Parent Common Stock, Parent Warrant, Parent Right, Parent Unit, Reincorporation Merger Surviving Company Ordinary Shares, Reincorporation Merger Surviving Company Warrant or Reincorporation Merger Surviving Company Right except in compliance with all applicable securities Laws, including Regulation M under the Exchange Act, in all material respects; or (ii) use or disclose or encourage any other Person to use or disclose any information that Parent or its Affiliates has made or makes available to Alps Holdco and its Representatives in violation of the Exchange Act, the Securities Act or any other applicable securities Law in any material respect.
8.2 Alps Holdco’s Shareholders Approval.
(a) As promptly as reasonably practicable after the F-4 Effective Date and in any event within five (5) Business Days following the F-4 Effective Date (the “Alps Holdco Shareholder Written Consent Deadline”), Alps Holdco shall obtain and deliver to Parent a true and correct copy of (i) unanimous written resolutions duly executed by all of the Alps Holdco Shareholders or their duly appointed attorney or (ii) the minutes of a duly called and held general meeting of the Alps Holdco Shareholders, in each case in form and substance reasonably satisfactory to Parent and evidencing the Alps Holdco Shareholder Approval (the “Alps Holdco Shareholder Written Consent”).
| 63 |
(b) Alps Holdco’s Board of Directors shall recommend that the Alps Holdco Shareholders vote in favor of this Agreement, the Additional Agreements to which Alps Holdco is or will be a party, the transactions contemplated hereby (including, without limitation, the Mergers) and thereby and other related matters, and neither Alps Holdco’s Board of Directors, nor any committee thereof, shall withhold, withdraw, amend, modify, change or propose or resolve to withhold, withdraw, amend, modify or change, in each case in a manner adverse to Parent, such recommendation.
8.3 Additional Financial Information. Alps Holdco shall use its commercially reasonable efforts to provide Parent (i) on or prior to May 15, 2024, with the Annual Financial Statements, and (ii) on or prior to May 31, 2024, with the audited consolidated balance sheet of Alps Holdco Group as of March 31, 2024 and the related unaudited statements of operations, changes in shareholders’ equity and cash flows for the fiscal year ended March 31, 2024, each prepared under IFRS and reviewed by a PCAOB qualified auditor in accordance with the requirements of the PCAOB for public companies (the “Final March 31, 2024 Financial Statements”). Additionally, during the Interim Period, Alps Holdco shall use its commercially reasonable efforts to provide Parent with Alps Holdco Group’s consolidated interim financial information for each quarterly period beginning after March 31, 2024 no later than forty (40) calendar days following the end of each quarterly period, each prepared under IFRS and reviewed by a PCAOB qualified auditor in accordance with the requirements of the PCAOB for public companies. All of the financial statements to be delivered pursuant to this Section 8.3 shall be accompanied by a certificate of the Chief Executive Officer or Chief Financial Officer of Alps Holdco to the effect that all such financial statements fairly present the consolidated financial position and consolidated results of operations of Alps Holdco Group as of the date or for the periods indicated, in accordance with IFRS, except as otherwise indicated in such statements and subject to year-end audit adjustments. Alps Holdco will promptly provide Parent with additional Alps Holdco Group financial information reasonably requested by Parent for inclusion in the Proxy Statement and any other filings to be made by Parent with the SEC.
8.4 Lock-Up Agreements. Prior to the Closing, Parent shall cause the Identified Parent Stockholders to enter into a Lock-Up Agreement, and Alps Holdco shall cause those persons set forth on Schedule 8.4 who have not entered into a Lock-Up Agreement concurrently with this Agreement, to enter into a Lock-Up Agreement with Parent to be effective as of the Closing, pursuant to which the Merger Consideration Shares shall be subject to a lock-up in accordance with the terms and conditions more fully set forth in the Lock-Up Agreement.
ARTICLE IX
COVENANTS OF ALL PARTIES HERETO
9.1 Commercially Reasonable Efforts; Further Assurances; Governmental Consents.
(a) Subject to the terms and conditions of this Agreement, each party shall use commercially reasonable efforts to take, or cause to be taken, all actions and to do, or cause to be done, all things necessary or desirable under applicable Laws, or as reasonably requested by the other parties, to consummate and implement expeditiously each of the transactions contemplated by this Agreement, including using commercially reasonable efforts to (i) obtain all necessary actions, nonactions, waivers, consents, approvals and other authorizations from all applicable Authorities prior to the Effective Time; (ii) avoid an Action by any Authority, and (iii) execute and deliver any additional instruments necessary to consummate the transactions contemplated by this Agreement. The parties shall execute and deliver such other documents, certificates, agreements and other writings and take such other actions as may be necessary or desirable in order to consummate or implement expeditiously each of the transactions contemplated by this Agreement.
| 64 |
(b) Other than with respect to Antitrust Laws, which shall be governed by Section 7.8, and subject to applicable Law, each of Alps Holdco and Parent agrees to (i) reasonably cooperate and consult with the other regarding obtaining and making all notifications and filings with Authorities, (ii) furnish to the other such information and assistance as the other may reasonably request in connection with its preparation of any notifications or filings, (iii) keep the other reasonably apprised of the status of matters relating to the completion of the transactions contemplated by this Agreement, including promptly furnishing the other with copies of notices and other communications received by such party from, or given by such party to, any third party or any Authority with respect to such transactions, (iv) permit the other party to review and incorporate the other party’s reasonable comments in any communication to be given by it to any Authority with respect to any filings required to be made with, or action or non-actions, waivers, expirations or terminations of waiting periods, clearances, consents or orders required to be obtained from, such Authority in connection with execution and delivery of this Agreement and the consummation of the transactions contemplated by this Agreement and (v) to the extent reasonably practicable, consult with the other in advance of and not participate in any meeting or discussion relating to the transactions contemplated by this Agreement, either in person or by telephone, with any Authority in connection with the proposed transactions unless it gives the other party the opportunity to attend and observe; provided, however, that, in each of clauses (iii) and (iv) above, that materials may be redacted (A) as necessary to comply with contractual arrangements or applicable Laws, and (B) as necessary to address reasonable attorney-client or other privilege or confidentiality concerns.
(c) During the Interim Period, Parent, on the one hand, and Alps Holdco, on the other hand, shall each notify the other in writing promptly after learning of any stockholder demands or other stockholder Action (including derivative claims) relating to this Agreement, any of the Additional Agreements or any matters relating thereto commenced against Parent or its Representative in its capacity as a representative of Parent or against any member of the Alps Holdco Group (collectively, the “Transaction Litigation”). Parent shall control the negotiation, defense and settlement of any such Transaction Litigation brought against Parent, the Merger Sub or members of the boards of directors of Parent or Merger Sub and Alps Holdco shall control the negotiation, defense and settlement of any such Transaction Litigation brought against any member of the Alps Holdco Group or the members of their boards of directors; provided, however, that in no event shall Alps Holdco or Parent settle, compromise or come to any arrangement with respect to any Transaction Litigation, or agree to do the same, without the prior written consent of the other party (not to be unreasonably withheld, conditioned or delayed; provided, that it shall be deemed to be reasonable for Parent (if Alps Holdco is controlling the Transaction Litigation) or Alps Holdco (if Parent is controlling the Transaction Litigation) to withhold, condition or delay its consent if any such settlement or compromise (A) does not provide for a legally binding, full, unconditional and irrevocable release of Parent(if Alps Holdco is controlling the Transaction Litigation) or Alps Holdco and its Subsidiaries and related parties (if Parent is controlling the Transaction Litigation) and its respective Representative that is the subject of such Transaction Litigation, (B) provides for any non-monetary, injunctive, equitable or similar relief against Parent (if Alps Holdco is controlling the Transaction Litigation) or Alps Holdco and its Subsidiaries and related parties (if Parent is controlling the Transaction Litigation) or (C) contains an admission of wrongdoing or Liability by Parent (if Alps Holdco is controlling the Transaction Litigation) or Alps Holdco and its Subsidiaries and related parties (if Parent is controlling the Transaction Litigation) and its respective Representative that is the subject of such Transaction Litigation. Parent and Alps Holdco shall each (i) keep the other reasonably informed regarding any Transaction Litigation, (ii) give the other the opportunity to, at its own cost and expense, participate in the defense, settlement and compromise of any such Transaction Litigation and reasonably cooperate with the other in connection with the defense, settlement and compromise of any such Transaction Litigation, (iii) consider in good faith the other’s advice with respect to any such Transaction Litigation and (iv) reasonably cooperate with each other.
| 65 |
(d) During the Interim Period, Parent shall use commercially reasonable efforts to timely obtain the fairness opinion referred to in Section 10.2(k).
9.2 Compliance with SPAC Agreements. Except to the extent Alps Holdco agrees otherwise in writing, during the Interim Period, Parent shall (a) comply with the Trust Agreement, the Underwriting Agreement, dated as of December 6, 2021, by and between Parent and Chardan Capital Markets, LLC, and (b) enforce the terms of (i) the letter agreement, dated as of December 6, 2021, by and among Parent and each of the officers, directors and stockholders of Parent named therein, and (ii) the Stock Escrow Agreement, dated as of December 6, 2021, by and among Parent, Continental Stock Transfer & Trust Company, as escrow agent, and the initial stockholders of Parent named therein.
9.3 Confidentiality. Except as necessary to complete the SEC Statement, the other Offer Documents or any Other Filings, Alps Holdco, on the one hand, and Parent and Merger Sub, on the other hand, shall comply with the Confidentiality Agreement.
9.4 Directors’ and Officers’ Indemnification and Liability Insurance.
(a) All rights to indemnification for acts or omissions occurring through the Closing Date now existing in favor of the current directors and officers of Alps Holdco or its Subsidiaries or Parent and Persons who served as a director, officer, member, trustee or fiduciary of another corporation, partnership, joint venture, trust, pension or other employee benefit plan or enterprise at the request of Alps Holdco or its Subsidiaries or Parent, as provided in their respective organizational documents or in any indemnification agreements shall survive the Mergers and shall continue in full force and effect in accordance with their terms. For a period of six (6) years after the Effective Time, the parties hereto shall cause the organizational documents of Reincorporation Merger Surviving Company and the Surviving Company and their respective Subsidiaries to contain provisions no less favorable with respect to exculpation and indemnification of and advancement of expenses than are set forth as of the date of this Agreement in the organizational documents of, with respect to Parent, Parent, and with respect to the Surviving Company and its Subsidiaries, Alps Holdco and its Subsidiaries, as applicable, to the extent permitted by applicable Law.
(b) Prior to the Closing, Parent and Alps Holdco shall reasonably cooperate in order to obtain (at Parent’s expense) directors’ and officers’ liability insurance for Reincorporation Merger Surviving Company and Alps Holdco that shall be effective as of Closing and will cover those Persons who will be the directors and officers of Reincorporation Merger Surviving Company and its Subsidiaries (including the Surviving Company after the Effective Time) at and after the Closing on terms not less favorable than the better of (x) the terms of the current directors’ and officers’ liability insurance in place for Alps Holdco’s directors and officers and (y) the terms of a typical directors’ and officers’ liability insurance policy for a company whose equity is listed on NASDAQ which policy has a scope and amount of coverage that is reasonably appropriate for a company of similar characteristics (including the line of business, anticipated market capitalization and revenues) as Alps Holdco.
(c) The provisions of this Section 9.4 are intended to be for the benefit of, and shall be enforceable by, each Person who will have been a director or officer of Alps Holdco or Parent for all periods ending on or before the Closing Date and may not be changed with respect to any officer or director without his or her written consent.
| 66 |
(d) At or prior to the Effective Time, the Pubco shall obtain a policy for the extension of the directors’ and officers’ liability coverage of Alps Holdco’s existing directors’ and officers’ liability insurance policies, for claims reporting or discovery period of six years from and after the Effective Time, on terms and conditions providing coverage retentions, limits and other material terms (other than premiums payable) substantially equivalent to the current policies of directors’ and officers’ liability insurance maintained by Alps Holdco with respect to matters arising on or before the Effective Time, covering without limitation the transactions contemplated hereby.
9.5 Parent Public Filings; NASDAQ. During the Interim Period, Parent will keep current and timely file all of its public filings with the SEC and otherwise comply in all material respects with applicable securities Laws, and shall use commercially reasonable efforts prior to the Closing to maintain the listing of the Parent Common Stock, Parent Public Units, the Parent Public Rights and the Parent Public Warrants on NASDAQ. During the Interim Period, Alps Holdco shall use commercially reasonable efforts to cause (a) Pubco’s initial listing application with NASDAQ in connection with the transactions contemplated by this Agreement to have been approved; (b) all applicable initial and continuing listing requirements of NASDAQ to be satisfied; and (c) the Reincorporation Merger Surviving Company Ordinary Shares, including the Merger Consideration Shares and the Earn-out Shares, and the Reincorporation Merger Surviving Company Warrants to be approved for listing on NASDAQ, subject to official notice of issuance, in each case, as promptly as reasonably practicable after the date of this Agreement and in any event prior to the Effective Time.
9.6 Certain Tax Matters. Each of Parent and Alps Holdco shall use commercially reasonable efforts to cause the Mergers to qualify as a “reorganization” within the meaning of Section 368(a) of the Code. Neither Parent nor Alps Holdco shall take any action, or fail to take any action, that could reasonably be expected to cause the Mergers to fail to qualify as a “reorganization” within the meaning of Section 368(a) of the Code. Parent and Alps Holdco intend to report and, except to the extent otherwise required by a change in Law, shall report, for U.S. federal income tax purposes, the Mergers as a “reorganization” within the meaning of Section 368(a) of the Code, unless otherwise required by applicable Law.
9.7 [Intentionally Omitted.]
9.8 Parent Expenses. Parent shall use commercially reasonable efforts to minimize the Parent Transaction Expenses such that the total of all Parent Transaction Expenses incurred or payable as of the Closing by Parent or its Subsidiaries in connection with the consummation of the transactions contemplated hereby shall not exceed $1,000,000 (excluding the cost of any insurance required under Section 9.4(b) above).
ARTICLE X
CONDITIONS TO CLOSING
10.1 Condition to the Obligations of the Parties. The obligations of all of the parties to consummate the Closing are subject to the satisfaction or written waiver (where permissible) by Parent and Alps Holdco of all the following conditions:
(a) No provisions of any applicable Law and no Order shall restrain or prohibit or impose any condition on the consummation of the transactions contemplated hereby, including the Mergers.
(b) (i) all applicable waiting periods under the HSR Act with respect to the Merger shall have expired or been terminated, and (ii) each consent, approval or authorization of any Authority required of Parent, Pubco, Merger Sub, Alps Holdco or any of their respective Subsidiaries to consummate the Mergers that are set forth on Schedule 10.1(b) shall have been obtained and shall be in full force and effect.
| 67 |
(c) There shall not be any pending Action brought by any Authority to enjoin or otherwise restrict the consummation of the Closing.
(d) After giving effect to any redemption of shares of Parent Common Stock in connection with the transactions contemplated by this Agreement, Parent shall have net tangible assets of at least $5,000,001 immediately prior to, and Pubco shall have net tangible assets of at least $5,000,001 upon consummation of the Mergers.
(e) Alps Holdco Shareholder Approval shall have been obtained.
(f) Each of the Parent Proposals shall have been approved at the Parent Stockholder Meeting.
(g) Pubco’s initial listing application with NASDAQ in connection with the transactions contemplated by this Agreement shall have been conditionally approved and, immediately following the Effective Time, Pubco shall satisfy any applicable initial and continuing listing requirements of NASDAQ, and Pubco shall not have received any notice of non-compliance therewith, and the Reincorporation Merger Surviving Company Ordinary Shares and the Reincorporation Merger Surviving Company Warrants shall have been approved for listing on NASDAQ.
(h) The Form F-4 shall have become effective in accordance with the provisions of the Securities Act, no stop order suspending the effectiveness of the Form F-4 shall have been issued by the SEC that remains in effect and no proceeding seeking such a stop order shall have been initiated by the SEC and not withdrawn.
10.2 Conditions to Obligations of Parent. The obligation of Parent to consummate the Closing is subject to the satisfaction, or the waiver in Parent’s sole and absolute discretion, of all the following further conditions:
(a) Alps Holdco, Pubco and Merger Sub shall have duly performed or complied with, in all material respects, all of their respective obligations and covenants hereunder required to be performed or complied with (without giving effect to any materiality or similar qualifiers contained therein) by Alps Holdco, Pubco and Merger Sub at or prior to the Closing Date.
(b) The representations and warranties of Alps Holdco, Pubco and Merger Sub contained in this Agreement (disregarding all qualifications and exceptions contained therein relating to materiality or Material Adverse Effect), shall be true and correct in all respects at and as of the date of this Agreement and as of the Closing Date as if made as of such date (except to the extent that any such representation and warranty is expressly made as of a specific date, in which case such representation and warranty shall be true and correct at and as of such specific date), except, in each case, for any failure of such representations and warranties (disregarding all qualifications and exceptions contained therein relating to materiality or Material Adverse Effect) to be so true and correct that would not in the aggregate have or reasonably be expected to have a Material Adverse Effect in respect of the Alps Holdco Group as a whole.
(c) Since the date of this Agreement, there shall not have occurred any Effect in respect of the Alps Holdco Group, that individually, or together with any other Effect since the date of this Agreement, has had or would reasonably be expected to have a Material Adverse Effect in respect of the Alps Holdco Group as a whole which is continuing and uncured.
| 68 |
(d) Parent shall have received a certificate, dated as of the Closing Date, signed by the Chief Executive Officer or another officer or a director of Alps Holdco certifying the accuracy of the provisions of the foregoing clauses (a), (b) and (c) of this Section 10.2.
(e) Parent shall have received a certificate, dated as of the Closing Date, signed by the Secretary or another officer or a director of Alps Holdco attaching true, correct and complete copies of (i) the Memorandum and Articles of Association of Alps Holdco as then in force, as filed with and stamped by the Cayman Registrar; (ii) copies of resolutions duly passed by the Board of Directors of Alps Holdco authorizing this Agreement, the Additional Agreements to which Alps Holdco is a party and the transactions contemplated hereby and thereby and the Alps Holdco Shareholder Written Consent; and (iii) a certificate of good standing of Alps Holdco, issued as of a recent date (not less than three (3) days before the Closing Date) by the Cayman Registrar.
(f) Parent shall have received a certificate, dated as of the Closing Date, signed by the Secretary or another officer or a director of Pubco attaching true, correct and complete copies of (i) the memorandum and articles of association of Pubco as then in force, as filed with and stamped by the Cayman Registrar; (ii) copies of resolutions duly passed by the Board of Directors of Pubco authorizing this Agreement, the Additional Agreements to which Pubco is a party and the transactions contemplated hereby and thereby; (iii) the Pubco Shareholder Approval, in each case evidencing the approval of the shareholders of Pubco, and (iv) a certificate of good standing of Pubco, issued as of a recent date (not less than three (3) days before the Closing Date) by the Cayman Registrar.
(g) Parent shall have received a certificate, dated as of the Closing Date, signed by the Secretary or another officer or a director of Merger Sub attaching true, correct and complete copies of (i) the memorandum and articles of association of Merger Sub as then in force, as filed with and stamped by the Cayman Registrar; (ii) copies of resolutions duly passed by the Board of Directors of Merger Sub authorizing this Agreement, the Additional Agreements to which Merger Sub is a party and the transactions contemplated hereby and thereby; (iii) the Merger Sub Shareholder Approval duly executed by all of the shareholders of Merger Sub, and (iv) a certificate of good standing of Merger Sub, issued as of a recent date (not less than three (3) days before the Closing Date) by the Cayman Registrar.
(h) Each of Alps Holdco and the Alps Holdco Shareholders, as applicable, shall have executed and delivered to Parent a copy of each Additional Agreement to which Alps Holdco or such Alps Holdco Shareholder, as applicable, is a party and that was not otherwise executed and delivered prior to the Closing.
(i) The size and composition as set forth in Section 3.8 of the post-Closing Reincorporation Merger Surviving Company Board of Directors shall have been approved, with effect from the Effective Time, by resolutions of the Pubco Board of Directors.
(j) Parent shall have received a copy of the Escrow Agreement, duly executed by the Seller Representative and the Escrow Agent.
(k) Parent shall obtain an opinion from an independent investment bank or other financial advisory firm mutually acceptable to Parent and Alps Holdco as to the fairness from a financial point of view, as of the date of such opinion, of the Merger Consideration Shares to be paid to the Alps Holdco Shareholders pursuant to this Agreement.
| 69 |
10.3 Conditions to Obligations of Alps Holdco, Pubco and Merger Sub. The obligations of Alps Holdco to consummate the Closing is subject to the satisfaction, or the waiver in Alps Holdco’s sole and absolute discretion, of all of the following further conditions:
(a) Parent shall have duly performed or complied with, in all material respects, all of its obligations and covenants hereunder required to be performed or complied with (without giving effect to any materiality or similar qualifiers contained therein) by Parent at or prior to the Closing Date.
(b) The representations and warranties of Parent contained in this Agreement (disregarding all qualifications contained therein relating to materiality or Material Adverse Effect), shall be true and correct as of the date of this Agreement and as of the Closing Date in all material respects, as if made at and as of such date (except to the extent that any such representation and warranty is made as of an earlier date, in which case such representation and warranty shall be true and correct in all material respects at and as of such earlier date).
(c) Since the date of this Agreement, there shall not have occurred any Effect in respect of Parent that individually, or together with any other Effect since the date of this Agreement, has had or would reasonably be expected to have a Material Adverse Effect in respect of Parent which is continuing and uncured.
(d) Alps Holdco shall have received a certificate, dated as of the Closing Date, signed by the Chief Executive Officer of Parent certifying the accuracy of the provisions of the foregoing clauses (a), (b) and (c) of this Section 10.3.
(e) Alps Holdco shall have received a certificate, dated as of the Closing Date, signed by the Secretary or another officer or a director of the Reincorporation Merger Surviving Company attaching true, correct and complete copies of (i) the memorandum and articles of association of the Reincorporation Merger Surviving Company then in effect, as filed with and (if received prior to the date of such certificate) stamped by the Cayman Registrar; (ii) copies of resolutions duly adopted by the Board of Directors of Parent authorizing this Agreement, the Additional Agreements to which Parent is a party and the transactions contemplated hereby and thereby and the Parent Proposals; and (iii) a certificate of good standing of Pubco, issued as of a recent date by the Cayman Registrar.
(f) Each of Parent, Sponsor or other stockholder of Parent, as applicable, shall have executed and delivered to Alps Holdco a copy of each Additional Agreement to which Parent, Sponsor or such other stockholder of Parent, as applicable, is a party.
(g) Alps Holdco shall have received a copy of the Escrow Agreement, duly executed by Pubco, the Pubco Representative and the Escrow Agent.
ARTICLE XI
TERMINATION
11.1 Termination Without Default.
(a) In the event that (i) the Closing of the transactions contemplated hereunder has not occurred by the six (6)-month anniversary of the date of this Agreement (the “Outside Closing Date”) (provided that, if the SEC has not declared the Proxy Statement/Form F-4 effective on or prior to the five (5)-month anniversary of the date of this Agreement, the Outside Closing Date shall be automatically extended by one (1) month); and (ii) the material breach or violation of any representation, warranty, covenant or obligation under this Agreement by the party (i.e., Parent, on one hand, or Alps Holdco, Pubco or Merger Sub on the other hand) seeking to terminate this Agreement was not the cause of, or resulted in, the failure of the Closing to occur on or before the Outside Closing Date, then Parent or Alps Holdco, as applicable, shall have the right, at its sole option, to terminate this Agreement without liability to the other party. Such right may be exercised by Parent or Alps Holdco, as the case may be, giving written notice to the other at any time after the Outside Closing Date.
| 70 |
(b) In the event an Authority shall have issued an Order or enacted a Law, having the effect of permanently restraining, enjoining or otherwise prohibiting the Mergers, which Order or Law is final and non-appealable, Parent or Alps Holdco shall have the right, at its sole option, to terminate this Agreement without liability to the other party; provided, however, that the right to terminate this Agreement pursuant to this Section shall not be available to Alps Holdco or Parent if the failure by such party or its Affiliates to comply with any provision of this Agreement has been a substantial cause of, or substantially resulted in, such action by such Authority.
(c) This Agreement may be terminated at any time by mutual written consent of Alps Holdco and Parent duly authorized by each of their respective boards of directors.
(d) Parent may terminate this Agreement by giving written notice to Alps Holdco in the event that Alps Holdco (i) does not deliver to Parent the Final March 31, 2024 Financial Statements on or prior to May 31, 2024 or such further period as mutually agreed between Parent and Alps Holdco in writing; provided, that upon delivery by Alps Holdco to Parent of the Final March 31, 2024 Financial Statements, Parent shall no longer have the right to terminate under this Section 11.1(d)(i), and (ii) the Final March 31, 2024 Financial Statements reveal a change or modification from the Annual Financial Statements that has or would reasonably be expected to have a Material Adverse Effect on the business of the Surviving Company.
11.2 Termination Upon Default.
(a) Parent may terminate this Agreement by giving written (email shall suffice) notice to Alps Holdco, without prejudice to any rights or obligations Parent may have: (i) at any time prior to the Closing Date if (x) Alps Holdco, Pubco or Merger Sub shall have breached any representation, warranty, agreement or covenant contained herein to be performed on or prior to the Closing Date, which has rendered or would reasonably be expected to render the satisfaction of any of the conditions set forth in Section 10.2(a) or 10.2(c) impossible; and (y) such breach cannot be cured or is not cured by the earlier of the Outside Closing Date and thirty (30) days following receipt by Alps Holdco of a written notice from Parent describing in reasonable detail the nature of such breach; (ii) at any time prior to the Closing Date if there shall have been any Effect in respect of the Alps Holdco Group, that individually, or together with any other Effect since the date of this Agreement, has had or would reasonably be expected to have a Material Adverse Effect in respect of the Alps Holdco Group as a whole which is uncurable or is not cured within thirty (30) days following the date such Effect initially happened; or (iii) at any time after the Alps Holdco Shareholder Written Consent Deadline if Alps Holdco has not previously received the Alps Holdco Shareholder Approval (provided, that upon Alps Holdco receiving the Alps Holdco Shareholder Approval, Parent shall no longer have any right to terminate this Agreement under this clause (iii)).
(b) Alps Holdco may terminate this Agreement by giving written (email shall suffice) notice to Parent, without prejudice to any rights or obligations Alps Holdco may have, at any time prior to the Closing Date, if: (i) (x) Parent shall have breached any of its covenants, agreements, representations, and warranties contained herein to be performed on or prior to the Closing Date, which has rendered or reasonably would render the satisfaction of any of the conditions set forth in Section 10.3(a) or 10.3(b) impossible; and (y) such breach cannot be cured or is not cured by the earlier of the Outside Closing Date and thirty (30) days following receipt by Parent of a written notice from Alps Holdco describing in reasonable detail the nature of such breach; or (ii) there shall have been any Effect in respect of Parent, that individually, or together with any other Effect since the date of this Agreement, has had or would reasonably be expected to have a Material Adverse Effect in respect of Parent which is uncurable and continuing.
| 71 |
11.3 Effect of Termination. If this Agreement is terminated pursuant to this ARTICLE XI (other than termination pursuant to Section 11.1(c)), this Agreement shall become void and of no further force or effect without liability of any party (or any shareholder, director, officer, employee, Affiliate, agent, consultant or representative of such party) to the other parties hereto; provided that, if such termination shall result from the willful breach by a party or its Affiliate of its covenants and agreements hereunder or fraud in connection with the transactions contemplated by this Agreement, such party shall not be relieved of liability to the other parties for any such willful breach or fraud. The provisions of Section 9.3, this Section 11.3 and ARTICLE XIII, and the Confidentiality Agreement, shall survive any termination hereof pursuant to this ARTICLE XI.
ARTICLE XII
INDEMNIFICATION
12.1 Indemnification of Parent. Subject to the terms and conditions of this ARTICLE XII and for a period commencing from the Closing Date and continuing until 12 months thereafter, the Alps Holdco Shareholders (the “Indemnifying Parties”) hereby jointly and severally agree to indemnify and hold harmless Parent (the “Indemnified Party”), against and in respect of any and all out-of-pocket loss, cost, payment, demand, penalty, forfeiture, expense, liability, judgment, deficiency or damage, and diminution in value or claim (including actual costs of investigation and attorneys’ fees and other costs and expenses) (all of the foregoing collectively, “Losses”) incurred or sustained by the Indemnified Party as a result of or in connection with any breach or inaccuracy of the representations and warranties of Alps Holdco contained in Sections 5.1 (Corporate Existence and Power), 5.2 (Authorization), 5.3 (Governmental Authorization), 5.4 (Non-Contravention), 5.5 (Capitalization), 5.7 (Subsidiaries), 5.8 (Consents), 5.9 (Financial Statements), 5.10 (Books and Records), 5.12 (Absence of Certain Changes), 5.13 (Properties; Title to Alps Holdco’s Assets), 5.14 (Litigation), 5.16 (Licenses and Permits), 5.17 (Compliance with Laws), 5.23 (Real Property), 5.24 (Tax Matters), 5.26 (Finders’ Fees), 5.29 (Anti-Money Laundering Laws), and 5.31 (Related Party Transactions) (collectively, the “Alps Holdco Surviving Representations”). Except for Fraud Claims against Alps Holdco, the Indemnified Party shall not assert any claim, and shall not be entitled to indemnification, unless and until the aggregate amount of all Losses indemnifiable hereunder exceeds an amount equal to $1,000,000, in which event the Indemnifying Party shall be responsible for the aggregate amount of all Losses from the first dollar, regardless of such threshold. Notwithstanding anything to the contrary herein, except for Fraud Claims against Alps Holdco, the maximum aggregate amount of indemnification payments which the Indemnifying Parties will be obligated to pay under this ARTICLE XI shall not exceed the amount of the Escrow Property in the Escrow Account at such time.
12.2 Procedure. The following shall apply with respect to all claims by the Indemnified Party for indemnification:
(a) The Parent Representative shall have the sole right to act on behalf of the Indemnified Party with respect to any indemnification claims made pursuant to this ARTICLE XII, including bringing and settling any indemnification claims hereunder and receiving any notices on behalf of the Indemnified Party. The Seller Representative shall have the sole right to act on behalf of the Indemnifying Parties with respect to any indemnification claims made pursuant to this ARTICLE XII, including defending and settling any indemnification claims hereunder and receiving any notices on behalf of the Indemnifying Parties.
| 72 |
(b) The Parent Representative on behalf of an Indemnified Party shall give the Seller Representative on behalf of the Indemnifying Parties and the Escrow Agent prompt written notice (an “Indemnification Notice”) of any third-party action with respect to which the Indemnified Party seeks indemnification pursuant to Section 12.1 (a “Third-Party Claim”), which shall describe in reasonable detail the Loss that has been or may be suffered by the Indemnified Party; provided that the copy of such Indemnification Notice provided to the Escrow Agent shall be redacted for any confidential or proprietary information of the Indemnifying Party or the Indemnified Party. The failure to give the Indemnification Notice shall not impair any of the rights or benefits of such Indemnified Party under Section 12.1, except to the extent such failure materially and adversely affects the ability of the Indemnifying Parties to defend such claim or increases the amount of such liability.
(c) In the case of any Third-Party Claims as to which indemnification is sought by the Indemnified Party, the Seller Representative will be entitled, at the sole expense and liability of the Seller Representative, to exercise full control of the defense, compromise or settlement of any Third-Party Claim unless the Indemnifying Parties, within a reasonable time after the giving of an Indemnification Notice by the Parent Representative (but in any event within ten (10) days thereafter), shall (i) deliver a written confirmation to the Parent Representative that the indemnification provisions of Section 12.1 are applicable to such action and the Indemnifying Parties will indemnify such Indemnified Party in respect of such action pursuant to the terms of Section 12.1 and, notwithstanding anything to the contrary, shall do so without asserting any challenge, defense, limitation on the Indemnifying Parties’ liability for Losses, counterclaim or offset, (ii) notify the Parent Representative in writing of the intention of the Seller Representative to assume the defense thereof, and (iii) retain legal counsel selected by the Seller Representative to conduct the defense of such Third-Party Claim.
(d) If the Seller Representative assumes the defense of any such Third-Party Claim pursuant to Section 12.2(b), then the Parent Representative and the Indemnified Party shall cooperate with the Seller Representative in any manner reasonably requested in connection with the defense, and the Parent Representative shall have the right to be kept fully informed by the Seller Representative and its legal counsel with respect to the status of any legal proceedings, to the extent not inconsistent with the preservation of attorney-client or work product privilege. If the Seller Representative so assumes the defense of any such Third-Party Claim, the Parent Representative shall have the right to employ separate counsel and to participate in (but not control) the defense, compromise, or settlement thereof, but the fees and expenses of such counsel employed by the Parent Representative shall be at the expense of the Parent Representative unless (i) the Seller Representative has agreed to pay such fees and expenses, or (ii) the named parties to any such Third-Party Claim (including any impleaded parties) include an Indemnified Party and an Indemnifying Party and such Indemnified Party shall have been advised by its counsel that there may be a conflict of interest between such Indemnified Party and the Indemnifying Parties in the conduct of the defense thereof, and in any such case the reasonable fees and expenses of such separate counsel shall be borne by the Indemnifying Parties.
(e) If the Seller Representative elects to assume the defense of any Third-Party Claim pursuant to Section 12.2(b), the Indemnified Party shall not pay, or permit to be paid, any part of any claim or demand arising from such asserted liability unless the Seller Representative withdraws from or fails to vigorously prosecute the defense of such asserted liability, or unless a judgment is entered against the Seller Representative for such liability. If the Seller Representative does not elect to defend, or if, after commencing or undertaking any such defense, the Seller Representative fails to adequately prosecute or withdraw such defense, the Parent Representative shall have the right to undertake the defense or settlement thereof, at the Indemnifying Parties’ expense. Notwithstanding anything to the contrary, the Seller Representative shall not be entitled to control, but may participate in, and the Parent Representative (at the expense of the Indemnifying Parties) shall be entitled to have sole control over, the defense or settlement of (x) that part of any Third-Party Claim (i) that seeks a temporary restraining order, a preliminary or permanent injunction or specific performance against the Indemnified Party, or (ii) to the extent such Third-Party Claim involves criminal allegations against the Indemnified Party or (y) the entire Third-Party Claim if such Third-Party Claim would impose liability on the part of the Indemnified Party in an amount which is greater than the amount as to which the Indemnified Party is entitled to indemnification under this Agreement. In the event the Parent Representative retains control of the Third-Party Claim, the Parent Representative will not settle the subject claim without the prior written consent of the Seller Representative, which consent will not be unreasonably withheld or delayed.
| 73 |
(f) If the Parent Representative undertakes the defense of any such Third-Party Claim pursuant to Section 12.1 and proposes to settle the same prior to a final judgment thereon or to forgo appeal with respect thereto, then the Parent Representative shall give the Seller Representative prompt written notice thereof and the Seller Representative shall have the right to participate in the settlement, assume or reassume the defense thereof or prosecute such appeal, in each case at its expense. The Seller Representative shall not, without the prior written consent of the Parent Representative settle or compromise or consent to entry of any judgment with respect to any such Third-Party Claim (i) in which any relief other than the payment of money damages is or may be sought against the Indemnified Party, (ii) in which such Third-Party Claim could be reasonably expected to impose or create a monetary liability on the part of the Indemnified Party (such as an increase in the Indemnified Party’s income Tax) other than the monetary claim of the third party in such Third-Party Claim being paid pursuant to such settlement or judgment, or (iii) which does not include as an unconditional term thereof the giving by the claimant, person conducting such investigation or initiating such hearing, plaintiff or petitioner to the Indemnified Party of a release from all liability with respect to such Third-Party Claim and all other actions (known or unknown) arising or which might arise out of the same facts.
(g) Except for Fraud Claims against Alps Holdco, any indemnification claims against the Indemnifying Parties shall be satisfied solely by the Escrow Property (with such indemnification first be applied against the Escrow Shares and then against any other Escrow Property), and no Indemnifying Party shall be required to make any out-of-pocket payment for indemnification. Any indemnification obligation of an Indemnifying Party under this ARTICLE XII will be paid within five (5) Business Days after the determination of such obligation in accordance with this ARTICLE XII (and the Parent Representative and the Seller Representative will provide or cause to be provided to the Escrow Agent any written instructions or other information or documents required by the Escrow Agent to do so). Notwithstanding anything to the contrary contained herein, any indemnification payments will be made to Parent or its successors. With respect to any indemnification payment, the value of each Escrow Share or any other share of Parent Common Stock for purposes of determining the indemnification payment shall be the Reincorporation Merger Surviving Company Share Price on the date that the indemnification claim is finally determined in accordance with this ARTICLE XI. Any Escrow Shares or other shares of Parent Common Stock received by Parent as an indemnification payment shall be promptly canceled by Parent after its receipt thereof.
12.3 Insurance. Any indemnification payments hereunder shall take into account any insurance proceeds or other third party reimbursement actually received.
12.4 Survival of Indemnification Rights. The Alps Holdco Surviving Representations shall survive until the Expiration Date. Except for the Alps Holdco Surviving Representations, no representations or warranties contained in this in this Agreement (including all schedules and exhibits hereto and all certificates, documents, instruments and undertakings furnished pursuant to this Agreement) shall survive the Closing.
12.5 Sole and Exclusive Remedy. The remedies provided in this ARTICLE XII shall be deemed the sole and exclusive remedies of the Indemnified Party, from and after the Closing Date, with respect to any and all claims arising out of or related to this Agreement or in connection with the transactions contemplated hereby.
| 74 |
ARTICLE XIII
MISCELLANEOUS
13.1 Notices. Any notice hereunder shall be sent in writing, addressed as specified below, and shall be deemed given: (a) if by hand or nationally recognized overnight courier service, by 5:00 PM Eastern Time on a Business Day, addressee’s day and time, on the date of delivery, and if delivered after 5:00 PM Eastern Time, on the first Business Day after such delivery; (b) if by email, on the date of transmission with affirmative confirmation of receipt; or (c) three (3) Business Days after mailing by prepaid certified or registered mail, return receipt requested. Notices shall be addressed to the respective parties as follows (excluding telephone numbers, which are for convenience only), or to such other address as a party shall specify to the others in accordance with these notice provisions:
if to Alps Holdco, Pubco or Merger Sub (or, following the Closing, Pubco, the Reincorporation Merger Surviving Company or the Surviving Company), to:
Alps Life Sciences Inc
Unit E-18-01 & E-18-02 Level 18
Icon Tower (East)
No. 1 Jalan 1/68F, Jalan Tun Razak
50400 Kuala Lumpur
Wilayah Persekutuan, Malaysia
Attn: Dr. Tham Seng Kong; Low Wei Sim; Christie Elizabeth
E-mail: drtham@alpsmedical.com; amanda@alpsmedical.com; christie@alpsmedical.com
if to Parent (prior to the Closing):
Globalink Investment Inc.
200 Continental Drive, Suite 401
Newark, Delaware, 19713
Attn: Say Leong Lim
E-mail: slim@globalinkinvestment.com
with a copy (which shall not constitute notice) to:
Hunter Taubman Fischer & Li LLC
950 Third Avenue, 19th Floor
New York, NY 10022
Attention: Ying Li, Esq.; Guillaume de Sampigny, Esq.
E-mail: yli@htflawyers.com; gdesampigny@htflawyers.com
if to the Parent Representative:
GL Sponsor LLC
1180 Avenue of the Americas, 8th Floor,
New York, NY 10036.
Attn: Ng Yan Xun
E-mail: glsponsor.jerryng@gmail.com
| 75 |
with a copy (which shall not constitute notice) to:
Hunter Taubman Fischer & Li LLC
950 Third Avenue, 19th Floor
New York, NY 10022
Attention: Ying Li, Esq.; Guillaume de Sampigny, Esq.
E-mail: yli@htflawyers.com; gdesampigny@htflawyers.com
if to the Seller Representative:
Dr. Tham Seng Kong
Unit E-18-01 & E-18-02 Level 18
Icon Tower (East)
No. 1 Jalan 1/68F, Jalan Tun Razak
50400 Kuala Lumpur
Wilayah Persekutuan, Malaysia
Email: drtham@alpsmedical.com
13.2 Amendments; No Waivers; Remedies.
(a) This Agreement cannot be amended, except by a writing signed by Parent, Alps Holdco, the Parent Representative and the Seller Representative, and cannot be terminated orally or by course of conduct. No provision hereof can be waived, except by a writing signed by the party against whom such waiver is to be enforced (or by the Parent Representative or Seller Representative, as applicable, in lieu of such party to the extent provided in this Agreement), and any such waiver shall apply only in the particular instance in which such waiver shall have been given. Notwithstanding the foregoing, any waiver of any provision of this Agreement after the Closing shall also require the prior written consent of the Parent Representative and the Seller Representative.
(b) Neither any failure or delay in exercising any right or remedy hereunder or in requiring satisfaction of any condition herein nor any course of dealing shall constitute a waiver of or prevent any party from enforcing any right or remedy or from requiring satisfaction of any condition. No notice to or demand on a party waives or otherwise affects any obligation of that party or impairs any right of the party giving such notice or making such demand, including any right to take any action without notice or demand not otherwise required by this Agreement. No exercise of any right or remedy with respect to a breach of this Agreement shall preclude exercise of any other right or remedy, as appropriate to make the aggrieved party whole with respect to such breach, or subsequent exercise of any right or remedy with respect to any other breach.
(c) Except as otherwise expressly provided herein, no statement herein of any right or remedy shall impair any other right or remedy stated herein or that otherwise may be available.
(d) Notwithstanding anything to the contrary contained herein, no shall any party seek, nor shall any party be liable for, punitive or exemplary damages under any tort, contract, equity or other legal theory with respect to any breach (or alleged breach) of this Agreement or any provision hereof or any matter otherwise relating hereto or arising in connection herewith.
| 76 |
13.3 Arm’s Length Bargaining; No Presumption Against Drafter. This Agreement has been negotiated at arm’s-length by parties of equal bargaining strength, each represented by counsel or having had but declined the opportunity to be represented by counsel and having participated in the drafting of this Agreement. This Agreement creates no fiduciary or other special relationship between the parties, and no such relationship otherwise exists. No presumption in favor of or against any party in the construction or interpretation of this Agreement or any provision hereof shall be made based upon which Person might have drafted this Agreement or such provision.
13.4 Publicity. Except as required by law or applicable stock exchange rules and except with respect to the Additional Parent SEC Documents, the parties agree that neither they nor their Representatives shall issue any press release or make any other public disclosure concerning the transactions contemplated hereunder without the prior approval of the other party hereto. If a party is required to make such a disclosure as required by law or applicable stock exchange rules, the party making such determination will, if practicable in the circumstances, use reasonable commercial efforts to allow the other party reasonable time to comment on such disclosure in advance of its issuance.
13.5 Expenses. The costs and expenses in connection with this Agreement and the transactions contemplated hereby incurred by each party shall be borne by such party, regardless of whether the Closing takes place.
13.6 No Assignment or Delegation. No party may assign any right or delegate any obligation hereunder, including by merger, consolidation, operation of law or otherwise, without the written consent of Parent and Alps Holdco (and after the Closing, the Parent Representative and the Seller Representative). Any purported assignment or delegation without such consent shall be void, in addition to constituting a material breach of this Agreement.
13.7 Governing Law. This Agreement and all disputes or controversies arising out of or relating to this Agreement or the transactions contemplated hereby, including the applicable statute of limitations, shall be governed by and construed in accordance with the Laws of the State of Delaware, without giving effect to any choice of law or conflict of law provision or rule (whether of the State of Delaware or any other jurisdiction) that would cause the application of the Law of any jurisdiction other than the State of Delaware; provided that matters that as a matter of the laws of the Cayman Islands are required to be governed by the laws of the Cayman Islands (including, without limitation, the effects of the Mergers and the fiduciary duties that may apply to the directors and officers of the Parties that are incorporated under the laws of the Cayman Islands) shall be governed by, and construed in accordance with, the laws of the Cayman Islands, without regard to laws that may be applicable under conflicts of laws principles that would cause the application of the laws of any jurisdiction other than the Cayman Islands to such matters.
13.8 Counterparts; Facsimile Signatures. This Agreement may be executed in counterparts, each of which shall constitute an original, but all of which shall constitute one agreement. This Agreement shall become effective upon delivery to each party of an executed counterpart or the earlier delivery to each party of original, photocopied, or electronically transmitted signature pages that together (but need not individually) bear the signatures of all other parties.
13.9 Entire Agreement. This Agreement, together with the Additional Agreements, sets forth the entire agreement of the parties with respect to the subject matter hereof and thereof and supersedes all prior and contemporaneous understandings and agreements related thereto (whether written or oral), all of which are merged herein. No provision of this Agreement or any Additional Agreement may be explained or qualified by any agreement, negotiations, understanding, discussion, conduct or course of conduct or by any trade usage. Except as otherwise expressly stated herein or in any Additional Agreement, there is no condition precedent to the effectiveness of any provision hereof or thereof. Notwithstanding the foregoing, the Confidentiality Agreement is not superseded by this Agreement or merged herein and shall continue in accordance with its terms, including in the event of any termination of this Agreement, until the Closing.
| 77 |
13.10 Severability. A determination by a court or other legal authority that any provision that is not of the essence of this Agreement is legally invalid shall not affect the validity or enforceability of any other provision hereof. The parties shall cooperate in good faith to substitute (or cause such court or other legal authority to substitute) for any provision so held to be invalid a valid provision, as alike in substance to such invalid provision as is lawful.
13.11 Further Assurances. Each party shall execute and deliver such documents and take such action, as may reasonably be considered within the scope of such party’s obligations hereunder, necessary to effectuate the transactions contemplated by this Agreement.
13.12 Third Party Beneficiaries. Except as provided in Section 9.4 and Section 13.19, neither this Agreement nor any provision hereof confers any benefit or right upon or may be enforced by any Person not a signatory hereto.
13.13 Waiver. Reference is made to the final prospectus of Parent, dated December 6, 2021 (the “Prospectus”). Alps Holdco understands that Parent has established the Trust Account for the benefit of the public shareholders of Parent and the underwriters of the IPO pursuant to the Trust Agreement and that, except for a portion of the interest earned on the amounts held in the Trust Account, Parent may disburse monies from the Trust Account only for the purposes set forth in the Trust Agreement. For and in consideration of Parent agreeing to enter into this Agreement, Alps Holdco, for itself and on behalf of the Alps Holdco Shareholders, hereby agrees that it does not now and shall not at any time hereafter prior to the Closing have any right, title, interest or claim of any kind in or to any monies in the Trust Account as a result of, or arising out of, any negotiations, contracts or agreements with Parent and hereby agrees that it will not seek recourse against the Trust Account for any reason. Notwithstanding the foregoing, nothing contained in this Section 13.13 shall serve to limit or prohibit (x) Alps Holdco’s right to pursue a claim for a breach for legal relief against assets held outside the Trust Account (other than distributions therefrom to Parent’s public shareholders that elect to redeem their shares in connection with the consummation of Parent’s initial business combination or an extension of Parent’s deadline to consummate its initial business combination or that receive proceeds from the Trust Account upon the liquidation of Parent if it fails to meet its deadline to consummate its initial business combination (“Public Distributions”), for specific performance or other non-monetary relief, or (y) any claims that Alps Holdco may have in the future against assets or funds that are not held in the Trust Account (including any funds that have been released from such trust account and any assets that have been purchased or acquired with any such funds, but excluding Public Distributions).
| 78 |
13.14 No Other Representations; No Reliance.
(a) NONE OF ALPS HOLDCO, ANY ALPS HOLDCO SHAREHOLDER NOR ANY OF THEIR RESPECTIVE REPRESENTATIVES HAS MADE ANY REPRESENTATIONS OR WARRANTIES, EXPRESS OR IMPLIED, OF ANY NATURE WHATSOEVER RELATING TO ALPS HOLDCO OR THE BUSINESS OR OTHERWISE IN CONNECTION WITH THE TRANSACTIONS CONTEMPLATED BY THIS AGREEMENT OR ANY ADDITIONAL AGREEMENT, OTHER THAN THOSE REPRESENTATIONS AND WARRANTIES EXPRESSLY SET FORTH IN ARTICLE V, IN EACH CASE, AS MODIFIED BY THE SCHEDULES TO THIS AGREEMENT. Without limiting the generality of the foregoing, neither Alps Holdco, any Alps Holdco Shareholder nor any of their respective representatives has made, and shall not be deemed to have made, any representations or warranties in the materials relating to Alps Holdco made available to Parent and its representatives, including due diligence materials, or in any presentation of the business of Alps Holdco by management of Alps Holdco or others in connection with the transactions contemplated hereby, and no statement contained in any of such materials or made in any such presentation shall be deemed a representation or warranty hereunder or otherwise or deemed to be relied upon by Parent in executing, delivering and performing this Agreement, the Additional Agreements or the transactions contemplated hereby or thereby, in each case except for the representations and warranties set forth in ARTICLE V as modified by the Schedules to this Agreement. It is understood that any cost estimates, projections or other predictions, any data, any financial information or any memoranda or offering materials or presentations, including any offering memorandum or similar materials made available by Alps Holdco, any Alps Holdco Shareholder or their respective representatives are not and shall not be deemed to be or to include representations or warranties of Alps Holdco or any Alps Holdco Shareholder, and are not and shall not be deemed to be relied upon by Parent in executing, delivering and performing this Agreement, the Additional Agreement and the transactions contemplated hereby or thereby, in each case except for the representations and warranties set forth in ARTICLE V, in each case, as modified by the Schedules to this Agreement. Except for the specific representations and warranties expressly made by Alps Holdco in ARTICLE V, in each case as modified by the Schedules: (a) Parent acknowledges and agrees that: (i) neither Alps Holdco, the Alps Holdco Shareholders nor any of their respective representatives is making or has made any representation or warranty, express or implied, at law or in equity, in respect of Alps Holdco, the business, assets, liabilities, operations, prospects or condition (financial or otherwise) of Alps Holdco, the nature or extent of any liabilities of Alps Holdco, the effectiveness or the success of any operations of Alps Holdco or the accuracy or completeness of any confidential information memoranda, projections, forecasts or estimates of earnings, or other information (financial or otherwise) regarding Alps Holdco furnished to Parent or its representatives or made available to Parent and its representatives in any “data rooms,” “virtual data rooms,” management presentations or any other form in expectation of, or in connection with, the transactions contemplated hereby, or in respect of any other matter or thing whatsoever; and (ii) no representative of any Alps Holdco Shareholder or Alps Holdco has any authority, express or implied, to make any representations, warranties or agreements not specifically set forth in ARTICLE V and subject to the limited remedies herein provided; (b) each of Parent and Merger Sub specifically disclaims that it is relying upon or has relied upon any such other representations or warranties that may have been made by any Person, and acknowledges and agrees that the Alps Holdco Shareholders and Alps Holdco have specifically disclaimed and do hereby specifically disclaim any such other representation or warranty made by any Person; and (c) none of Alps Holdco, the Alps Holdco Shareholders nor any other Person shall have any liability to Parent or any other Person with respect to any such other representations or warranties, including projections, forecasts, estimates, plans or budgets of future revenue, expenses or expenditures, future results of operations, future cash flows or the future financial condition of Alps Holdco or the future business, operations or affairs of Alps Holdco.
| 79 |
(b) NEITHER PARENT NOR ANY OF ITS REPRESENTATIVES HAS MADE ANY REPRESENTATIONS OR WARRANTIES, EXPRESS OR IMPLIED, OF ANY NATURE WHATSOEVER RELATING TO PARENT OR OTHERWISE IN CONNECTION WITH THE TRANSACTIONS CONTEMPLATED BY THIS AGREEMENT OR ANY ADDITIONAL AGREEMENT, OTHER THAN THOSE REPRESENTATIONS AND WARRANTIES EXPRESSLY SET FORTH IN ARTICLE VI, IN EACH CASE, AS MODIFIED BY THE SCHEDULES TO THIS AGREEMENT AND THE PARENT SEC DOCUMENTS. Without limiting the generality of the foregoing, neither Parent nor any of its representatives has made, and shall not be deemed to have made, any representations or warranties in the materials relating to Parent made available to Alps Holdco and the Alps Holdco Shareholders and their representatives, including due diligence materials, or in any presentation of the business of Parent by management of Parent or others in connection with the transactions contemplated hereby, and no statement contained in any of such materials or made in any such presentation shall be deemed a representation or warranty hereunder or otherwise or deemed to be relied upon by Alps Holdco and the Alps Holdco Shareholders in executing, delivering and performing this Agreement, the Additional Agreements or the transactions contemplated hereby or thereby, in each case except for the representations and warranties set forth in ARTICLE VI as modified by the Schedules to this Agreement and the Parent SEC Documents. It is understood that any cost estimates, projections or other predictions, any data, any financial information or any memoranda or offering materials or presentations, including any offering memorandum or similar materials made available by Parent or its representatives are not and shall not be deemed to be or to include representations or warranties of Parent, and are not and shall not be deemed to be relied upon by Alps Holdco or Alps Holdco Shareholders in executing, delivering and performing this Agreement, the Additional Agreement and the transactions contemplated hereby or thereby, in each case except for the representations and warranties set forth in ARTICLE VI, in each case, as modified by the Schedules to this Agreement and the Parent SEC Documents. Except for the specific representations and warranties expressly made by Parent in ARTICLE VI, in each case as modified by the Schedules and Parent SEC Documents: (a) Alps Holdco acknowledges and agrees that: (i) neither Parent nor any of its representatives is making or has made any representation or warranty, express or implied, at law or in equity, in respect of Parent, the business, assets, liabilities, operations, prospects or condition (financial or otherwise) of Parent, the nature or extent of any liabilities of Parent or Merger Sub, the effectiveness or the success of any operations of Parent or the accuracy or completeness of any confidential information memoranda, projections, forecasts or estimates of earnings, or other information (financial or otherwise) regarding Parent furnished to Alps Holdco, the Alps Holdco Shareholders or their respective representatives or made available to Alps Holdco, the Alps Holdco Shareholders and their representatives in any “data rooms,” “virtual data rooms,” management presentations or any other form in expectation of, or in connection with, the transactions contemplated hereby, or in respect of any other matter or thing whatsoever; and (ii) no representative of Parent has any authority, express or implied, to make any representations, warranties or agreements not specifically set forth in ARTICLE VI and subject to the limited remedies herein provided; (b) Alps Holdco specifically disclaims that it is relying upon or has relied upon any such other representations or warranties that may have been made by any Person, and acknowledges and agrees that Parent has specifically disclaimed and do hereby specifically disclaim any such other representation or warranty made by any Person; and (c) neither Parent nor any other Person shall have any liability to Alps Holdco, the Alps Holdco Shareholders or any other Person with respect to any such other representations or warranties, including projections, forecasts, estimates, plans or budgets of future revenue, expenses or expenditures, future results of operations, future cash flows or the future financial condition of Parent or the future business, operations or affairs of Parent.
13.15 Waiver of Jury Trial. THE PARTIES EACH HEREBY WAIVES, TO THE FULLEST EXTENT PERMITTED BY LAW, ANY RIGHT TO TRIAL BY JURY OF ANY PROCEEDING (I) ARISING UNDER THIS AGREEMENT OR UNDER ANY ADDITIONAL AGREEMENT OR (II) IN ANY WAY CONNECTED WITH OR RELATED OR INCIDENTAL TO THE DEALINGS OF THE PARTIES IN RESPECT OF THIS AGREEMENT OR ANY ADDITIONAL AGREEMENT OR ANY OF THE TRANSACTIONS RELATED HERETO OR THERETO OR ANY FINANCING IN CONNECTION WITH THE TRANSACTIONS CONTEMPLATED HEREBY OR ANY OF THE TRANSACTIONS CONTEMPLATED THEREBY, IN EACH CASE, WHETHER NOW EXISTING OR HEREAFTER ARISING, AND WHETHER IN CONTRACT, TORT, EQUITY, OR OTHERWISE. THE PARTIES EACH HEREBY AGREES AND CONSENTS THAT ANY SUCH PROCEEDING SHALL BE DECIDED BY COURT TRIAL WITHOUT A JURY AND THAT THE PARTIES MAY FILE AN ORIGINAL COUNTERPART OF A COPY OF THIS AGREEMENT WITH ANY COURT AS WRITTEN EVIDENCE OF THE CONSENT OF THE PARTIES HERETO TO THE WAIVER OF THEIR RIGHT TO TRIAL BY JURY. EACH PARTY CERTIFIES AND ACKNOWLEDGES THAT (A) NO REPRESENTATIVE, AGENT OR ATTORNEY OF ANY OTHER PARTY HAS REPRESENTED, EXPRESSLY OR OTHERWISE, THAT SUCH OTHER PARTY WOULD NOT, IN THE EVENT OF LITIGATION, SEEK TO ENFORCE THE FOREGOING WAIVER, (B) EACH SUCH PARTY UNDERSTANDS AND HAS CONSIDERED THE IMPLICATIONS OF THIS WAIVER, (C) EACH SUCH PARTY MAKES THIS WAIVER VOLUNTARILY AND (D) EACH SUCH PARTY HAS BEEN INDUCED TO ENTER INTO THIS AGREEMENT BY, AMONG OTHER THINGS, THE MUTUAL WAIVERS AND CERTIFICATIONS IN THIS SECTION 13.15.
| 80 |
13.16 Submission to Jurisdiction. Subject to the provisions of Section 13.17, each of the parties irrevocably and unconditionally submits to the exclusive jurisdiction of the Chancery Court of the State of Delaware (or, if the Chancery Court of the State of Delaware does not have jurisdiction, a federal court sitting in Wilmington, Delaware) (or any appellate courts thereof), for the purposes of any Action (a) arising under this Agreement or under any Additional Agreement or (b) in any way connected with or related or incidental to the dealings of the parties in respect of this Agreement or any Additional Agreement or any of the transactions contemplated hereby or thereby, and irrevocably and unconditionally waives any objection to the laying of venue of any such Action in any such court, and further irrevocably and unconditionally waives and agrees not to plead or claim in any such court that any such Action has been brought in an inconvenient forum. Each Party hereby irrevocably and unconditionally waives, and agrees not to assert, by way of motion or as a defense, counterclaim or otherwise, in any Action (i) arising under this Agreement or under any Additional Agreement or (ii) in any way connected with or related or incidental to the dealings of the parties in respect of this Agreement or any Additional Agreement or any of the transactions contemplated hereby or thereby, (A) any claim that it is not personally subject to the jurisdiction of the courts as described in this Section 13.16 for any reason, (B) that it or its property is exempt or immune from the jurisdiction of any such court or from any Action commenced in such courts (whether through service of notice, attachment prior to judgment, attachment in aid of execution of judgment, execution of judgment or otherwise) and (C) that (x) the Action in any such court is brought in an inconvenient forum, (y) the venue of such Action is improper or (z) this Agreement, or the subject matter hereof, may not be enforced in or by such courts. Each Party agrees that service of any process, summons, notice or document by registered mail to such Party’s respective address set forth in Section 13.1 shall be effective service of process for any such Action.
13.17 Arbitration. Any and all disputes, controversies and claims (other than applications for a temporary restraining order, preliminary injunction, permanent injunction or other equitable relief or application for enforcement of a resolution under this Section 13.17), arising out of, related to, or in connection with this Agreement or the transactions contemplated hereby (a “Dispute”) shall be governed by this Section 13.17. A party must, in the first instance, provide written notice of any Disputes to the other parties subject to such Dispute, which notice must provide a reasonably detailed description of the matters subject to the Dispute. The parties involved in such Dispute shall seek to resolve the Dispute on an amicable basis within ten (10) Business Days of the notice of such Dispute being received by such other parties subject to such Dispute (the “Resolution Period”); provided, that if any Dispute would reasonably be expected to have become moot or otherwise irrelevant if not decided within sixty (60) days after the occurrence of such Dispute, then there shall be no Resolution Period with respect to such Dispute. Any Dispute that is not resolved during the Resolution Period may immediately be referred to the American Arbitration Association (the “AAA”) and finally resolved by arbitration pursuant to the then-existing Expedited Procedures (as defined in the AAA Procedures) of the Commercial Arbitration Rules (the “AAA Procedures”) of the AAA. Any party involved in such Dispute may submit the Dispute to the AAA to commence the proceedings after the Resolution Period. To the extent that the AAA Procedures and this Agreement are in conflict, the terms of this Agreement shall control. The arbitration shall be conducted by one arbitrator nominated by the AAA promptly (but in any event within five (5) Business Days) after the submission of the Dispute to the AAA and reasonably acceptable to each party subject to the Dispute, which arbitrator shall be a commercial lawyer with substantial experience arbitrating disputes under acquisition agreements. The arbitrator shall accept his or her appointment and begin the arbitration process promptly (but in any event within five (5) Business Days) after his or her nomination and acceptance by the parties subject to the Dispute. The proceedings shall be streamlined and efficient. The arbitrator shall decide the Dispute in accordance with the substantive law of the State of Delaware. Time is of the essence. Each party subject to the Dispute shall submit a proposal for resolution of the Dispute to the arbitrator within twenty (20) days after confirmation of the appointment of the arbitrator. The arbitrator shall have the power to order any party to do, or to refrain from doing, anything consistent with this Agreement, the Additional Agreements and applicable Law, including to perform its contractual obligation(s); provided, that the arbitrator shall be limited to ordering pursuant to the foregoing power (and, for the avoidance of doubt, shall order) the relevant party (or parties, as applicable) to comply with only one or the other of the proposals. The arbitrator’s award shall be in writing and shall include a reasonable explanation of the arbitrator’s reason(s) for selecting one or the other proposal. The seat of arbitration shall be in Kuala Lumpur, Malaysia. The language of the arbitration shall be English.
| 81 |
13.18 Remedies. Except as otherwise expressly provided herein, any and all remedies provided herein will be deemed cumulative with and not exclusive of any other remedy conferred hereby, or by law or equity upon such Party, and the exercise by a Party of any one remedy will not preclude the exercise of any other remedy. The parties agree that irreparable damage for which monetary damages, even if available, would not be an adequate remedy, would occur in the event that the parties do not perform their respective obligations under the provisions of this Agreement (including failing to take such actions as are required of them hereunder to consummate the transactions contemplated by this Agreement) in accordance with their specific terms or otherwise breach such provisions. It is accordingly agreed that the parties shall be entitled to an injunction or injunctions, specific performance and other equitable relief to prevent breaches of this Agreement and to enforce specifically the terms and provisions of this Agreement, in each case, without posting a bond or undertaking and without proof of damages and this being in addition to any other remedy to which they are entitled at law or in equity. Each of the parties agrees that it will not oppose the granting of an injunction, specific performance and other equitable relief when expressly available pursuant to the terms of this Agreement on the basis that the other parties have an adequate remedy at law or an award of specific performance is not an appropriate remedy for any reason at law or equity.
13.19 Non-Recourse. This Agreement may be enforced only against, and any dispute, claim or controversy based upon, arising out of or related to this Agreement or the transactions contemplated hereby may be brought only against, the entities that are expressly named as parties hereto and then only with respect to the specific obligations set forth in this Agreement with respect to such party. No past, present or future director, officer, employee, incorporator, member, partner, shareholder, agent, attorney, advisor, lender or representative or Affiliate of any named party to this Agreement (which Persons are intended third party beneficiaries of this Section 13.19) shall have any liability (whether in contract or tort, at law or in equity or otherwise, or based upon any theory that seeks to impose liability of an entity party against its owners or Affiliates) for any one or more of the representations, warranties, covenants, agreements or other obligations or liabilities of such named party or for any dispute, claim or controversy based on, arising out of, or related to this Agreement or the transactions contemplated hereby.
13.20 Parent Representative.
(a) Parent, on behalf of itself and its Subsidiaries, successors and assigns, by execution and delivery of this Agreement, hereby irrevocably appoints GL Sponsor LLC, in the capacity as the Parent Representative, as each such Person’s agent, attorney-in-fact and representative, with full power of substitution to act in the name, place and stead of such Person, to act on behalf of such Person from and after the Closing in connection with: (i) bringing, managing, controlling, defending and settling on behalf of the Indemnified Party any indemnification claims by any of them under ARTICLE XI; (ii) acting on behalf of such Person under the Escrow Agreement; (iii) terminating, amending or waiving on behalf of such Person any provision of this Agreement or any Additional Agreements to which the Parent Representative is a party or otherwise has rights in such capacity (together with this Agreement, the “Parent Representative Documents”); (iv) signing on behalf of such Person any releases or other documents with respect to any dispute or remedy arising under any Parent Representative Documents; (vi) employing and obtaining the advice of legal counsel, accountants and other professional advisors as the Parent Representative, in its reasonable discretion, deems necessary or advisable in the performance of its duties as the Parent Representative and to rely on their advice and counsel; (vi) incurring and paying reasonable out-of-pocket costs and expenses, including fees of brokers, attorneys and accountants incurred pursuant to the transactions contemplated hereby, and any other out-of-pocket fees and expenses allocable or in any way relating to such transaction or any indemnification claim; and (vii) otherwise enforcing the rights and obligations of any such Persons under any Parent Representative Documents, including giving and receiving all notices and communications hereunder or thereunder on behalf of such Person; provided, that the parties acknowledge that the Parent Representative is specifically authorized and directed to act on behalf of, and for the benefit of, the holders of Parent’s securities (other than the Alps Holdco Shareholders immediately prior to the Effective Time and their respective successors and assigns). All decisions and actions by the Parent Representative, including any agreement between the Parent Representative and Alps Holdco, Seller Representative, any Alps Holdco Shareholders or Indemnifying Party relating to the defense or settlement of any claims for which an Indemnifying Party may be required to indemnify an Indemnified Party pursuant to ARTICLE XII, shall be binding upon Parent and its Subsidiaries, successors and assigns, and neither they nor any other Party shall have the right to object, dissent, protest or otherwise contest the same. The provisions of this Section 13.20 are irrevocable and coupled with an interest. The Parent Representative hereby accepts its appointment and authorization as the Parent Representative under this Agreement.
| 82 |
(b) The Parent Representative shall not be liable for any act done or omitted under any Parent Representative Document as the Parent Representative while acting in good faith and without willful misconduct or gross negligence, and any act done or omitted pursuant to the advice of counsel shall be conclusive evidence of such good faith. Parent shall indemnify, defend and hold harmless the Parent Representative from and against any and all Losses incurred without gross negligence, bad faith or willful misconduct on the part of the Parent Representative (in its capacity as such) and arising out of or in connection with the acceptance or administration of the Parent Representative’s duties under any Parent Representative Document, including the reasonable fees and expenses of any legal counsel retained by the Parent Representative. In no event shall the Parent Representative in such capacity be liable under or in connection with any Parent Representative Document for any indirect, punitive, special or consequential damages. The Parent Representative shall be fully protected in relying upon any written notice, demand, certificate or document that it in good faith believes to be genuine, including facsimiles or copies thereof, and no Person shall have any liability for relying on the Parent Representative in the foregoing manner. In connection with the performance of its rights and obligations hereunder, the Parent Representative shall have the right at any time and from time to time to select and engage, at the cost and expense of Parent, attorneys, accountants, investment bankers, advisors, consultants and clerical personnel and obtain such other professional and expert assistance, maintain such records and incur other out-of-pocket expenses, as the Parent Representative may deem necessary or appropriate from time to time. All of the indemnities, immunities, releases and powers granted to the Parent Representative under this Section 13.20 shall survive the Closing and continue indefinitely.
(c) The Person serving as the Parent Representative may resign upon ten (10) days’ prior written notice to Parent and the Seller Representative, provided, that the Parent Representative appoints in writing a replacement Parent Representative. Each successor Parent Representative shall have all of the power, authority, rights and privileges conferred by this Agreement upon the original Parent Representative, and the term “Parent Representative” as used herein shall be deemed to include any such successor Parent Representatives.
| 83 |
13.21 Seller Representative.
(a) Each Alps Holdco Shareholder, by delivery of a Letter of Transmittal, on behalf of itself and its successors and assigns, hereby irrevocably constitutes and appoints Dr. Tham Seng Kong, in his capacity as the Seller Representative, as the true and lawful agent and attorney-in-fact of such Persons with full powers of substitution to act in the name, place and stead of thereof with respect to the performance on behalf of such Person under the terms and provisions of this Agreement and the Additional Agreements to which the Seller Representative is a party or otherwise has rights in such capacity (together with this Agreement, the “Seller Representative Documents”), as the same may be from time to time amended, and to do or refrain from doing all such further acts and things, and to execute all such documents on behalf of such Person, if any, as the Seller Representative will deem necessary or appropriate in connection with any of the transactions contemplated under the Seller Representative Documents, including: (i) making on behalf of such Person and taking all actions on their behalf relating to the achievement of the requirements for the Earn-out Shares and any disputes with respect thereto; (ii) managing, controlling, defending and settling on behalf of an Indemnifying Party any indemnification claims against any of them under ARTICLE XII, including controlling, defending, managing, settling and participating in any Third-Party Claim thereunder; (iii) acting on behalf of such Person under the Escrow Agreement; (iv) terminating, amending or waiving on behalf of such Person any provision of any Seller Representative Document (provided, that any such action, if material to the rights and obligations of the Alps Holdco Shareholders in the reasonable judgment of the Seller Representative, will be taken in the same manner with respect to all Alps Holdco Shareholders unless otherwise agreed by each Alps Holdco Shareholder who is subject to any disparate treatment of a potentially material and adverse nature); (v) signing on behalf of such Person any releases or other documents with respect to any dispute or remedy arising under any Seller Representative Document; (vi) employing and obtaining the advice of legal counsel, accountants and other professional advisors as the Seller Representative, in its reasonable discretion, deems necessary or advisable in the performance of its duties as the Seller Representative and to rely on their advice and counsel; (vii) incurring and paying reasonable costs and expenses, including fees of brokers, attorneys and accountants incurred pursuant to the transactions contemplated hereby, and any other reasonable fees and expenses allocable or in any way relating to such transaction or any indemnification claim, whether incurred prior or subsequent to Closing; (viii) receiving all or any portion of the consideration provided to the Alps Holdco Shareholders under this Agreement and to distribute the same to the Alps Holdco Shareholders in accordance with their Pro Rata Share; and (ix) otherwise enforcing the rights and obligations of any such Persons under any Seller Representative Document, including giving and receiving all notices and communications hereunder or thereunder on behalf of such Person. All decisions and actions by the Seller Representative, including any agreement between the Seller Representative and the Parent Representative, Parent or any Indemnified Party relating to the defense or settlement of any claims for which an Indemnifying Party may be required to indemnify an Indemnified Party pursuant to ARTICLE XII, shall be binding upon each Alps Holdco Shareholder and their respective successors and assigns, and neither they nor any other party shall have the right to object, dissent, protest or otherwise contest the same. The provisions of this Section 13.21 are irrevocable and coupled with an interest. The Seller Representative hereby accepts its appointment and authorization as the Seller Representative under this Agreement.
(b) Any other Person, including the Parent Representative, Parent, Alps Holdco and the Indemnified Party and the Indemnifying Parties may conclusively and absolutely rely, without inquiry, upon any actions of the Seller Representative as the acts of the Alps Holdco Shareholders under any Seller Representative Documents. The Parent Representative, Parent, Alps Holdco and each Indemnified Party and Indemnifying Party shall be entitled to rely conclusively on the instructions and decisions of the Seller Representative as to (i) the settlement of any indemnification claims by an Indemnified Party pursuant to ARTICLE XI, (ii) any payment instructions provided by the Seller Representative or (iii) any other actions required or permitted to be taken by the Seller Representative hereunder, and no Alps Holdco Shareholder nor any Indemnifying Party shall have any cause of action against the Parent Representative, Parent or any other Indemnified Party for any action taken by any of them in reliance upon the instructions or decisions of the Seller Representative. The Parent Representative and Parent shall not have any Liability to any Alps Holdco Shareholder or Indemnifying Party for any allocation or distribution among the Alps Holdco Shareholders by the Seller Representative of payments made to or at the direction of the Seller Representative. All notices or other communications required to be made or delivered to a Alps Holdco Shareholder under any Seller Representative Document shall be made to the Seller Representative for the benefit of such Alps Holdco Shareholder, and any notices so made shall discharge in full all notice requirements of the other parties hereto or thereto to such Alps Holdco Shareholder with respect thereto. All notices or other communications required to be made or delivered by an Alps Holdco Shareholder shall be made by the Seller Representative (except for a notice under Section 13.21(d) of the replacement of the Seller Representative).
| 84 |
(c) The Seller Representative will act for the Alps Holdco Shareholders on all of the matters set forth in this Agreement in the manner the Seller Representative believes to be in the best interest of the Alps Holdco Shareholders, but the Seller Representative will not be responsible to the Alps Holdco Shareholders for any Losses that any Alps Holdco Shareholder or any Indemnifying Party may suffer by reason of the performance by the Seller Representative of the Seller Representative’s duties under this Agreement, other than Losses arising from the bad faith, gross negligence or willful misconduct by the Seller Representative in the performance of its duties under this Agreement. From and after the Closing, the Alps Holdco Shareholders shall jointly and severally indemnify, defend and hold the Seller Representative harmless from and against any and all Losses reasonably incurred without gross negligence, bad faith or willful misconduct on the part of the Seller Representative (in its capacity as such) and arising out of or in connection with the acceptance or administration of the Seller Representative’s duties under any Seller Representative Document, including the reasonable fees and expenses of any legal counsel retained by the Seller Representative. In no event shall the Seller Representative in such capacity be liable hereunder or in connection herewith for any indirect, punitive, special or consequential damages. The Seller Representative shall not be liable for any act done or omitted under any Seller Representative Document as the Seller Representative while acting in good faith and without willful misconduct or gross negligence, and any act done or omitted pursuant to the advice of counsel shall be conclusive evidence of such good faith. The Seller Representative shall be fully protected in relying upon any written notice, demand, certificate or document that it in good faith believes to be genuine, including facsimiles or copies thereof, and no Person shall have any liability for relying on the Seller Representative in the foregoing manner. In connection with the performance of its rights and obligations hereunder, the Seller Representative shall have the right at any time and from time to time to select and engage, at the reasonable cost and expense of the Alps Holdco Shareholders, attorneys, accountants, investment bankers, advisors, consultants and clerical personnel and obtain such other professional and expert assistance, maintain such records and incur other reasonable out-of-pocket expenses, as the Seller Representative may reasonably deem necessary or appropriate from time to time. All of the indemnities, immunities, releases and powers granted to the Seller Representative under this Section 13.21 shall survive the Closing and continue indefinitely.
(d) If the Seller Representative shall die, become disabled, dissolve, resign or otherwise be unable or unwilling to fulfill its responsibilities as representative and agent of Alps Holdco Shareholders, then the Alps Holdco Shareholders shall, within ten (10) days after such death, disability, dissolution, resignation or other event, appoint a successor Seller Representative (by vote or written consent of the Alps Holdco Shareholders holding in the aggregate a Pro Rata Share in excess of fifty percent (50%)), and promptly thereafter (but in any event within two (2) Business Days after such appointment) notify the Parent Representative and Parent in writing of the identity of such successor. Any such successor so appointed shall become the “Seller Representative” for purposes of this Agreement.
[Signature Pages Follow]
| 85 |
IN WITNESS WHEREOF, the parties hereto have caused this Agreement to be duly executed as of the day and year first above written.
| Parent: | ||
| GLOBALINK INVESTMENT INC. | ||
| By: | /s/ Say Leong Lim | |
| Name: | Say Leong Lim | |
| Title: | Chairman & CEO | |
| Alps Holdco: | ||
| ALPS LIFE SCIENCES INC | ||
| By: | /s/ Tham Seng Kong | |
| Name: | Dr. Tham Seng Kong | |
| Title: | ||
| PubCo: | ||
| ALPS GLOBAL HOLDING PUBCO | ||
| By: | /s/ Tham Seng Kong | |
| Name: | Dr. Tham Seng Kong | |
| Title: | Director | |
| Merger Sub: | ||
| ALPS BIOSCIENCES MERGER SUB | ||
| By: | /s/ Tham Seng Kong | |
| Name: | Dr. Tham Seng Kong | |
| Title: | Director | |
{Signature page to Merger Agreement}
| Parent Representative: | ||
| GL SPONSOR LLC, in the capacity as the Parent Representative | ||
| By: | /s/ Yan Xun Ng | |
| Name: | Yan Xun Ng | |
| Title: | Manager | |
| Seller Representative: | ||
| /s/ Tham Seng Kong | ||
| Dr. Tham Seng Kong, in the capacity as the Seller Representative | ||
ANNEX B
Companies Act (Revised)
Company Limited by Shares
amended and restated
memorandum of association
OF
ALPS GROUP
Adopted by special resolution on [ ] and effective on [ ]

| B-1 |
Companies Act (Revised)
Company Limited by Shares
Amended and Restated
Memorandum of Association
of
ALPS Group
Adopted by special resolution on [ ] and effective on [ ]
| 1 | The name of the Company is ALPS Group, including such name as may be changed from time to time . |
| 2 | The Company’s registered office will be situated at the office of Ogier Global (Cayman) Limited, 89 Nexus Way, Camana Bay, Grand Cayman, KY1-9009, Cayman Islands or at such other place in the Cayman Islands as the directors may at any time decide. |
| 3 | The Company’s objects are unrestricted. As provided by section 7(4) of the Companies Act (Revised), the Company has full power and authority to carry out any object not prohibited by any law of the Cayman Islands. |
| 4 | The Company has unrestricted corporate capacity. Without limitation to the foregoing, as provided by section 27 (2) of the Companies Act (Revised), the Company has and is capable of exercising all the functions of a natural person of full capacity irrespective of any question of corporate benefit. |
| 5 | Nothing in any of the preceding paragraphs permits the Company to carry on any of the following businesses without being duly licensed, namely: |
| (a) | the business of a bank or trust company without being licensed in that behalf under the Banks and Trust Companies Act (Revised); or |
| (b) | insurance business from within the Cayman Islands or the business of an insurance manager, agent, sub-agent or broker without being licensed in that behalf under the Insurance Act (Revised); or |
| (c) | the business of company management without being licensed in that behalf under the Companies Management Act (Revised). |
| 6 | The Company will not trade in the Cayman Islands with any person, firm or corporation except in furtherance of its business carried on outside the Cayman Islands. Despite this, the Company may effect and conclude contracts in the Cayman Islands and exercise in the Cayman Islands any of its powers necessary for the carrying on of its business outside the Cayman Islands. |
| 7 | The Company is a company limited by shares and accordingly the liability of each member is limited to the amount (if any) unpaid on that member’s shares. |
| 8 | The share capital of the Company is US$50,000 divided into 500,000,000 Shares comprising (i) 495,000,000 Ordinary Shares of US$0.0001 par value each; and (ii) 5,000,000 Preferred Shares of US$0.0001 par value each of such class or classes (however designated) as the Board may determine in accordance with Article 2.4 of the Articles. However, subject to the Companies Act (Revised) and the Company’s articles of association, the Company has power to do any one or more of the following: |
| (a) | redeem or repurchase any of its shares; |
| (b) | increase or reduce its capital; |
| (c) | issue any part of its capital (whether original, redeemed, increased or reduced): |
| (i) | with or without any preferential, deferred, qualified or special rights, privileges or conditions; or |
| (ii) | subject to any limitations or restrictions |
and unless the condition of issue expressly declares otherwise, every issue of shares (whether declared to be ordinary, preference or otherwise) is subject to this power; and
| (d) | alter any of those rights, privileges, conditions, limitations or restrictions. |
| 9 | The Company has power to register by way of continuation as a body corporate limited by shares under the laws of any jurisdiction outside the Cayman Islands and to be deregistered in the Cayman Islands. |
Companies Act (Revised)
Company Limited By Shares
AMENDED AND RESTATED
articles of association
of
ALPS GROUP
(Adopted by special resolution passed on [ ] and effective on [ ])

Contents
| 1 | Definitions, interpretation and exclusion of Table A | 1 |
| Definitions | 1 | |
| Interpretation | 4 | |
| Exclusion of Table A Articles | 5 | |
| 2 | Shares | 5 |
| Power to issue Shares and options, with or without special rights | 5 | |
| Power to pay commissions and brokerage fees | 7 | |
| Trusts not recognized | 7 | |
| Security interests | 7 | |
| Power to vary class rights | 8 | |
| Effect of new Share issue on existing class rights | 8 | |
| No bearer Shares or warrants | 8 | |
| Treasury Shares | 8 | |
| Rights attaching to Treasury Shares and related matters | 8 | |
| Register of Members | 9 | |
| Annual Return | 9 | |
| 3 | Share certificates | 9 |
| Issue of share certificates | 9 | |
| Renewal of lost or damaged share certificates | 10 | |
| 4 | Lien on Shares | 10 |
| Nature and scope of lien | 10 | |
| Company may sell Shares to satisfy lien | 11 | |
| Authority to execute instrument of transfer | 11 | |
| Consequences of sale of Shares to satisfy lien | 11 | |
| Application of proceeds of sale | 12 | |
| 5 | Calls on Shares and forfeiture | 12 |
| Power to make calls and effect of calls | 12 | |
| Time when call made | 12 | |
| Liability of joint holders | 13 | |
| Interest on unpaid calls | 13 | |
| Deemed calls | 13 | |
| Power to accept early payment | 13 | |
| Power to make different arrangements at time of issue of Shares | 13 | |
| Notice of default | 13 | |
| Forfeiture or surrender of Shares | 14 | |
| Disposal of forfeited or surrendered Share and power to cancel forfeiture or surrender | 14 | |
| Effect of forfeiture or surrender on former Member | 14 | |
| Evidence of forfeiture or surrender | 15 | |
| Sale of forfeited or surrendered Shares | 15 | |
| 6 | Transfer of Shares | 15 |
| Form of Transfer | 15 | |
| Power to refuse registration for Shares not listed on a Designated Stock Exchange | 15 | |
| Suspension of transfers | 16 | |
| Company may retain instrument of transfer | 16 | |
| Notice of refusal to register | 16 | |
| 7 | Transmission of Shares | 16 |
| Persons entitled on death of a Member | 16 | |
| Registration of transfer of a Share following death or bankruptcy | 17 | |
| Indemnity | 17 | |
| Rights of person entitled to a Share following death or bankruptcy | 17 | |
| 8 | Alteration of capital | 17 |
| Increasing, consolidating, converting, dividing and cancelling share capital | 17 | |
| Dealing with fractions resulting from consolidation of Shares | 18 | |
| Reducing share capital | 18 | |
| 9 | Redemption and purchase of own Shares | 19 |
| Power to issue redeemable Shares and to purchase own Shares | 19 | |
| Power to pay for redemption or purchase in cash or in specie | 19 | |
| Effect of redemption or purchase of a Share | 19 | |
| 10 | Meetings of Members | 20 |
| Annual and extraordinary general meetings | 20 | |
| Power to call meetings | 20 | |
| Content of notice | 21 | |
| Period of notice | 21 | |
| Persons entitled to receive notice | 21 | |
| Accidental omission to give notice or non-receipt of notice | 22 | |
| 11 | Proceedings at meetings of Members | 22 |
| Quorum | 22 | |
| Lack of quorum | 22 | |
| Chairman | 23 | |
| Right of a Director to attend and speak | 23 | |
| Accommodation of Members attending meeting virtually | 23 | |
| Security | 23 | |
| Adjournment, postponement and cancellation | 24 | |
| Method of voting | 24 | |
| Taking of a poll | 24 | |
| Chairman’s casting vote | 24 | |
| Written resolutions | 24 | |
| Sole-Member Company | 25 | |
| 12 | Voting rights of Members | 25 |
| Right to vote | 25 | |
| Rights of joint holders | 26 | |
| Representation of corporate Members | 26 | |
| Member with mental disorder | 26 | |
| Objections to admissibility of votes | 27 | |
| Form of proxy | 27 | |
| How and when proxy is to be delivered | 27 | |
| Voting by proxy | 29 | |
| 13 | Number of Directors | 29 |
| 14 | Appointment, disqualification and removal of Directors | 29 |
| No age limit | 29 | |
| Corporate Directors | 29 | |
| No shareholding qualification | 29 | |
| Appointment of Directors | 29 | |
| Board’s power to appoint Directors | 30 | |
| Term of appointment | 30 | |
| Removal of Directors | 30 | |
| Resignation of Directors | 30 | |
| Termination of the office of Director | 31 | |
| 15 | Alternate Directors | 31 |
| Appointment and removal | 31 | |
| Notices | 32 | |
| Rights of alternate Director | 32 | |
| Appointment ceases when the appointor ceases to be a Director | 32 | |
| Status of alternate Director | 33 | |
| Status of the Director making the appointment | 33 | |
| 16 | Powers of Directors | 33 |
| Powers of Directors | 33 | |
| Directors below the minimum number | 33 | |
| Appointments to office | 33 | |
| Provisions for employees | 34 | |
| Exercise of voting rights | 34 | |
| Remuneration | 34 | |
| Disclosure of information | 35 | |
| 17 | Delegation of powers | 35 |
| Power to delegate any of the Directors’ powers to a committee | 35 | |
| Local boards | 36 | |
| Power to appoint an agent of the Company | 36 | |
| Power to appoint an attorney or authorised signatory of the Company | 37 | |
| Borrowing Powers | 37 | |
| Corporate Governance | 37 | |
| 18 | Meetings of Directors | 38 |
| Regulation of Directors’ meetings | 38 | |
| Calling meetings | 38 | |
| Notice of meetings | 38 | |
| Use of technology | 38 | |
| Quorum | 38 | |
| Chairman or deputy to preside | 38 | |
| Voting | 39 | |
| Recording of dissent | 39 | |
| Written resolutions | 39 | |
| Validity of acts of Directors in spite of formal defect | 39 | |
| 19 | Permissible Directors’ interests and disclosure | 40 |
| 20 | Minutes | 40 |
| 21 | Accounts and audit | 40 |
| Auditors | 41 | |
| 22 | Record dates | 41 |
| 23 | Dividends | 42 |
| Source of dividends | 42 | |
| Declaration of dividends by Members | 42 | |
| Payment of interim dividends and declaration of final dividends by Directors | 42 | |
| Apportionment of dividends | 43 | |
| Right of set off | 43 | |
| Power to pay other than in cash | 43 | |
| How payments may be made | 43 | |
| Dividends or other monies not to bear interest in absence of special rights | 44 | |
| Dividends unable to be paid or unclaimed | 44 | |
| 24 | Capitalisation of profits | 44 |
| Capitalisation of profits or of any share premium account or capital redemption reserve; | 44 | |
| Applying an amount for the benefit of Members | 45 | |
| 25 | Share Premium Account | 45 |
| Directors to maintain share premium account | 45 | |
| Debits to share premium account | 45 | |
| 26 | Seal | 46 |
| Company seal | 46 | |
| Duplicate seal | 46 | |
| When and how seal is to be used | 46 | |
| If no seal is adopted or used | 46 | |
| Power to allow non-manual signatures and facsimile printing of seal | 46 | |
| Validity of execution | 46 | |
| 27 | Indemnity | 47 |
| Release | 47 | |
| Insurance | 48 | |
| 28 | Notices | 48 |
| Form of notices | 48 | |
| Electronic communications | 48 | |
| Persons entitled to notices | 49 | |
| Persons authorised to give notices | 50 | |
| Delivery of written notices | 50 | |
| Joint holders | 50 | |
| Signatures | 50 | |
| Giving notice to a deceased or bankrupt Member | 50 | |
| Date of giving notices | 51 | |
| Saving provision | 51 | |
| 29 | Authentication of Electronic Records | 51 |
| Application of Articles | 51 | |
| Authentication of documents sent by Members by Electronic means | 51 | |
| Authentication of document sent by the Secretary or Officers of the Company by Electronic means | 52 | |
| Manner of signing | 52 | |
| Saving provision | 52 | |
| 30 | Transfer by way of continuation | 53 |
| 31 | Winding up | 53 |
| Distribution of assets in specie | 53 | |
| No obligation to accept liability | 53 | |
| 32 | Amendment of Memorandum and Articles | 54 |
| Power to change name or amend Memorandum | 54 | |
| Power to amend these Articles | 54 | |
| 33 | Mergers and Consolidations | 54 |
Companies Act (Revised)
Company Limited by Shares
Amended and Restated
Articles of Association
of
ALPS Group
(Adopted by special resolution passed on [ ] and effective on [ ])
| 1 | Definitions, interpretation and exclusion of Table A |
Definitions
| 1.1 | In these Articles, the following definitions apply: |
Act means the Companies Act (Revised) of the Cayman Islands, including any statutory modification or re-enactment thereof for the time being in force;
Articles means, as appropriate:
| (a) | these articles of association as amended from time to time: or |
| (b) | two or more particular articles of these Articles; |
and Article refers to a particular article of these Articles;
Auditors means the auditor or auditors for the time being of the Company;
Board means the board of Directors from time to time;
Business Day means a day when banks in Grand Cayman, the Cayman Islands are open for the transaction of normal banking business and for the avoidance of doubt, shall not include a Saturday, Sunday or public holiday in the Cayman Islands;
Cayman Islands means the British Overseas Territory of the Cayman Islands;
Class A Director means a Director so designated pursuant to Article 14.4.
Class B Director means a Director so designated pursuant to Article 14.4.
Clear Days, in relation to a period of notice, means that period of calendar days excluding:
| (a) | the calendar day when the notice is given or deemed to be given; and |
| 1 |
| (b) | the calendar day for which it is given or on which it is to take effect; |
Commission means Securities and Exchange Commission of the United States of America or other federal agency for the time being administering the U.S. Securities Act;
Company means the above-named company;
Default Rate means ten per cent per annum;
Designated Stock Exchanges means the Nasdaq Capital Market in the United States of America for so long as the Company’s Shares are there listed and any other stock exchange on which the Company’s Shares are listed for trading;
Designated Stock Exchange Rules means the relevant code, rules and regulations, as amended, from time to time, applicable as a result of the original and continued listing of any Shares or on the Designated Stock Exchanges;
Directors means the directors for the time being of the Company and the expression Director shall be construed accordingly;
Electronic has the meaning given to that term in the Electronic Transactions Act (Revised) of the Cayman Islands;
Electronic Record has the meaning given to that term in the Electronic Transactions Act (Revised) of the Cayman Islands;
Electronic Signature has the meaning given to that term in the Electronic Transactions Act (Revised) of the Cayman Islands;
Fully Paid Up means:
| (a) | in relation to a Share with par value, means that the par value for that Share and any premium payable in respect of the issue of that Share, has been fully paid or credited as paid in money or money’s worth; and |
| (b) | in relation to a Share without par value, means that the agreed issue price for that Share has been fully paid or credited as paid in money or money’s worth; |
general meeting means a general meeting of the Company duly constituted in accordance with the Articles;
Independent Director means a Director who is an independent director as defined in the Designated Stock Exchange Rules as determined by the Board;
Member means any person or persons entered on the register of Members from time to time as the holder of a Share;
| 2 |
Memorandum means the memorandum of association of the Company as amended from time to time;
month means a calendar month;
Officer means a person appointed to hold an office in the Company including a Director, alternate Director or liquidator and excluding the Secretary;
Ordinary Resolution means a resolution of a general meeting passed by a simple majority of Members who (being entitled to do so) vote in person or by proxy or, in the case of corporations, by their duly authorised representatives, at that meeting. The expression includes a unanimous written resolution signed by all of the Members entitled to vote at such meeting;
Ordinary Share means an ordinary share in the capital of the Company;
Partly Paid Up means:
| (a) | in relation to a Share with par value, that the par value for that Share and any premium payable in respect of the issue of that Share, has not been fully paid or credited as paid in money or money’s worth; and |
| (b) | in relation to a Share without par value, means that the agreed issue price for that Share has not been fully paid or credited as paid in money or money’s worth; |
Preferred Share means a preferred share in the capital of the Company;
Secretary means a person appointed to perform the duties of the secretary of the Company, including a joint, assistant or deputy secretary;
Share means a share in the share capital of the Company and the expression:
| (a) | includes stock (except where a distinction between shares and stock is expressed or implied); and |
| (b) | where the context permits, also includes a fraction of a Share; |
Special Resolution means a resolution of a general meeting or a resolution of a meeting of the holders of any class of Shares in a class meeting duly constituted in accordance with the Articles in each case passed by a majority of not less than two-thirds of Members who (being entitled to do so) vote in person or by proxy at that meeting. The expression includes a unanimous written resolution signed by all of the Members entitled to vote at such meeting;
Treasury Shares means Shares held in treasury pursuant to the Act and Article 2.12; and
U.S. Securities Act means the Securities Act of 1933 of the United States of America, as amended, or any similar federal statute and the rules and regulations of the Commission thereunder, all as the same shall be in effect at the time.
| 3 |
Interpretation
| 1.2 | In the interpretation of these Articles, the following provisions apply unless the context otherwise requires: |
| (a) | A reference in these Articles to a statute is a reference to a statute of the Cayman Islands as known by its short title, and includes: |
| (i) | any statutory modification, amendment or re-enactment; and |
| (ii) | any subordinate legislation or regulations issued under that statute. |
Without limitation to the preceding sentence, a reference to a revised Act of the Cayman Islands is taken to be a reference to the revision of that Act in force from time to time as amended from time to time.
| (b) | Headings are inserted for convenience only and do not affect the interpretation of these Articles, unless there is ambiguity. |
| (c) | If a day on which any act, matter or thing is to be done under these Articles is not a Business Day, the act, matter or thing must be done on the next Business Day. |
| (d) | A word which denotes the singular also denotes the plural, a word which denotes the plural also denotes the singular, and a reference to any gender also denotes the other genders. |
| (e) | A reference to a person includes, as appropriate, a company, trust, partnership, joint venture, association, body corporate or government agency. |
| (f) | Where a word or phrase is given a defined meaning another part of speech or grammatical form in respect to that word or phrase has a corresponding meaning. |
| (g) | All references to time are to be calculated by reference to time in the place where the Company’s registered office is located. |
| (h) | The words written and in writing include all modes of representing or reproducing words in a visible form, but do not include an Electronic Record where the distinction between a document in writing and an Electronic Record is expressed or implied. |
| (i) | The words including, include and in particular or any similar expression are to be construed without limitation. |
| 1.3 | The headings in these Articles are intended for convenience only and shall not affect the interpretation of these Articles. |
| 4 |
Exclusion of Table A Articles
| 1.4 | The regulations contained in Table A in the First Schedule of the Act and any other regulations contained in any statute or subordinate legislation are expressly excluded and do not apply to the Company. |
| 2 | Shares |
Power to issue Shares and options, with or without special rights
| 2.1 | Subject to the provisions of the Act and these Articles (including provisions about the redemption and purchase of the Shares, the Directors have general and unconditional authority to: |
| (a) | allot (with or without confirming rights of renunciation), grant options over or otherwise deal with any unissued Shares to such persons, at such times and on such terms and conditions as they may decide; and/or |
| (b) | grant rights over Shares or other securities to be issued in one or more classes as they deem necessary or appropriate and determine the designations, powers, preferences, privileges and other rights attaching to such Shares or securities, including dividend rights, voting rights, conversion rights, terms of redemption and liquidation preferences, any or all of which may be greater than the powers, preferences, privileges and rights associated with the then issued and outstanding Shares, at such times and on such other terms as they think proper. |
No Share may be issued at a discount except in accordance with the provisions of the Act.
| 2.2 | Without limitation to the preceding Article, the Directors may so deal with the unissued Shares: |
| (a) | either at a premium or at par; or |
| (b) | with or without preferred, deferred or other special rights or restrictions, whether in regard to dividend, voting, return of capital or otherwise. |
| 2.3 | Without limitation to the two preceding Articles: |
| (a) | the Company may issue rights, options, warrants or convertible securities or securities of similar nature conferring the right upon the holders thereof to subscribe for, purchase or receive any class of Shares or other securities in the Company at such times and on such terms and conditions as the Directors may decide; |
| (b) | the Directors may refuse to accept any application for Shares, and may accept any application in whole or in part, for any reason or for no reason; and |
| (c) | subject to the Act, the Company may issue fractions of a Share of any class. A fraction of a Share shall be subject to and carry the corresponding fraction of liabilities (whether with respect to calls or otherwise), limitations, preferences, privileges, qualifications, restrictions, rights and other attributes of a Share of that class of Shares. |
| 5 |
| 2.4 | Subject to the provisions of the Act and these Articles (including provisions about the redemption and purchase of the Shares), the Directors may authorise the division of Shares into any number of classes and the different classes shall be authorised, established and designated (or re-designated as the case may be) and the variations in the relative rights (including, without limitation, voting, dividend and redemption rights), restrictions, preferences, privileges and payment obligations as between the different classes (if any) may be fixed and determined by the Directors or by an Ordinary Resolution. The Directors may issue Shares with such preferred or other rights, all or any of which may be greater than the rights of Ordinary Shares, at such time and on such terms as they may think appropriate. Notwithstanding Article 2.11, the Directors may issue from time to time, out of the authorised share capital of the Company (other than the authorised but unissued Ordinary Shares), series of Preferred Shares in their absolute discretion and without approval of the Members; provided, however, before any Preferred Shares of any series are issued, the Directors shall fix, by resolution or resolutions, the following provisions of such series: |
| (a) | the designation of such series and the number of Preferred Shares to constitute such series; |
| (b) | whether the shares of such series shall have voting rights, in addition to any voting rights provided by Act, and, if so, the terms of such voting rights, which may be general or limited; |
| (c) | the dividends, if any, payable on such series, whether any such dividends shall be cumulative, and, if so, from what dates, the conditions and dates upon which such dividends shall be payable, the preference or relation which such dividends shall bear to the dividends payable on any Shares of any other class of Shares or any other series of Preferred Shares; |
| (d) | whether the Preferred Shares or such series shall be subject to redemption by the Company, and, if so, the times, prices and other conditions of such redemption; |
| (e) | the amount or amounts payable upon Preferred Shares of such series upon, and the rights of the holders of such series in, a voluntary or involuntary liquidation, dissolution or winding up, or upon any distribution of the assets, of the Company; |
| (f) | whether the Preferred Shares of such series shall be subject to the operation of a retirement or sinking fund and, if so, the extent to and manner in which any such retirement or sinking fund shall be applied to the purchase or redemption of the Preferred Shares of such series for retirement or other corporate purposes and the terms and provisions relative to the operation of the retirement or sinking fund; |
| 6 |
| (g) | whether the Preferred Shares of such series shall be convertible into, or exchangeable for, Shares of any other class of Shares or any other series of Preferred Shares or any other securities and, if so, the price or prices or the rate or rates of conversion or exchange and the method, if any, of adjusting the same, and any other terms and conditions of conversion or exchange; |
| (h) | the limitations and restrictions, if any, to be effective while any Preferred Shares or such series are outstanding upon the payment of dividends or the making of other distributions on, and upon the purchase, redemption or other acquisition by the Company of, the existing Shares or Shares of any other class of Shares or any other series of Preferred Shares; |
| (i) | the conditions or restrictions, if any, upon the creation of indebtedness of the Company or upon the issue of any additional Shares, including additional shares of such series or of any other class of Shares or any other series of Preferred Shares; and |
| (j) | any other powers, preferences and relative, participating, optional and other special rights, and any qualifications, limitations and restrictions of any other class of Shares or any other series of Preferred Shares. |
Power to pay commissions and brokerage fees
| 2.5 | The Company may pay a commission to any person in consideration of that person: |
| (a) | subscribing or agreeing to subscribe, whether absolutely or conditionally; or |
| (b) | procuring or agreeing to procure subscriptions, whether absolute or conditional, |
for any Shares. That commission may be satisfied by the payment of cash or the allotment of Fully Paid Up or Partly Paid Up Shares or partly in one way and partly in another.
| 2.6 | The Company may employ a broker in the issue of its capital and pay him any proper commission or brokerage. |
Trusts not recognised
| 2.7 | Except as required by law: |
| (a) | no person shall be recognised by the Company as holding any Share on any trust; and |
| (b) | no person other than the Member shall be recognised by the Company as having any right in a Share. |
Security interests
| 2.8 | Notwithstanding the preceding Article, the Company may (but shall not be obliged to) recognise a security interest of which it has actual notice over shares. The Company shall not be treated as having recognised any such security interest unless it has so agreed in writing with the secured party. |
| 7 |
Power to vary class rights
| 2.9 | If the share capital is divided into different classes of Shares then, unless the terms on which a class of Shares was issued state otherwise, the rights attaching to a class of Shares may only be varied if one of the following applies: |
| (a) | the Members holding not less than two-thirds of the issued Shares of that class consent in writing to the variation; or |
| (b) | the variation is made with the sanction of a Special Resolution passed at a separate general meeting of the Members holding the issued Shares of that class. |
| 2.10 | For the purpose of Article 2.9(b), all the provisions of these Articles relating to general meetings apply, mutatis mutandis, to every such separate meeting except that the necessary quorum shall be one or more persons holding, or representing by proxy, not less than one third of the issued Shares of the class. |
Effect of new Share issue on existing class rights
| 2.11 | Unless the terms on which a class of Shares was issued state otherwise, the rights conferred on the Member holding Shares of any class shall not be deemed to be varied by the creation or issue of further Shares ranking pari passu with the existing Shares of that class. |
No bearer Shares or warrants
| 2.12 | The Company shall not issue Shares or warrants to bearers. |
Treasury Shares
| 2.13 | Shares that the Company purchases, redeems or acquires by way of surrender in accordance with the Act shall be held as Treasury Shares and not treated as cancelled if: |
| (a) | the Directors so determine prior to the purchase, redemption or surrender of those shares; and |
| (b) | the relevant provisions of the Memorandum and Articles and the Act are otherwise complied with. |
Rights attaching to Treasury Shares and related matters
| 2.14 | No dividend may be declared or paid, and no other distribution (whether in cash or otherwise) of the Company’s assets (including any distribution of assets to Members on a winding up) may be made to the Company in respect of a Treasury Share. |
| 2.15 | The Company shall be entered in the register of Members as the holder of the Treasury Shares. However: |
| (a) | the Company shall not be treated as a Member for any purpose and shall not exercise any right in respect of the Treasury Shares, and any purported exercise of such a right shall be void; and |
| 8 |
| (b) | a Treasury Share shall not be voted, directly or indirectly, at any meeting of the Company and shall not be counted in determining the total number of issued shares at any given time, whether for the purposes of these Articles or the Act. |
| 2.16 | Nothing in Article 2.15 prevents an allotment of Shares as Fully Paid Up bonus shares in respect of a Treasury Share and Shares allotted as Fully Paid Up bonus shares in respect of a Treasury Share shall be treated as Treasury Shares. |
| 2.17 | Treasury Shares may be disposed of by the Company in accordance with the Act and otherwise on such terms and conditions as the Directors determine. |
Register of Members
| 2.18 | The Directors shall keep or cause to be kept a register of Members as required by the Act and may cause the Company to maintain one or more branch registers as contemplated by the Act, provided that where the Company is maintaining one or more branch registers, the Directors shall ensure that a duplicate of each branch register is kept with the Company’s principal register of Members and updated within such number of days of any amendment having been made to such branch register as may be required by the Act. |
| 2.19 | The title to Shares listed on a Designated Stock Exchange may be evidenced and transferred in accordance with the laws applicable to the rules and regulations of the Designated Stock Exchange and, for these purposes, the register of Members may be maintained in accordance with Section 40B of the Act. |
Annual Return
| 2.20 | The Directors in each calendar year shall prepare or cause to be prepared an annual return and declaration setting forth the particulars required by the Act and shall deliver a copy thereof to the registrar of companies for the Cayman Islands. |
| 3 | Share certificates |
Issue of share certificates
| 3.1 | A Member shall only be entitled to a share certificate if the Directors resolve that share certificates shall be issued. Share certificates representing Shares, if any, shall be in such form as the Directors may determine. If the Directors resolve that share certificates shall be issued, upon being entered in the register of Members as the holder of a Share, the Directors may issue to any Member: |
| (a) | without payment, one certificate for all the Shares of each class held by that Member (and, upon transferring a part of the Member’s holding of Shares of any class, to a certificate for the balance of that holding); and |
| 9 |
| (b) | upon payment of such reasonable sum as the Directors may determine for every certificate after the first, several certificates each for one or more of that Member’s Shares. |
| 3.2 | Every certificate shall specify the number, class and distinguishing numbers (if any) of the Shares to which it relates and whether they are Fully Paid Up or Partly Paid Up. A certificate may be executed under seal or executed in such other manner as the Directors determine. |
| 3.3 | Every certificate shall bear legends required under the applicable laws, including the U.S. Securities Act (to the extent applicable). |
| 3.4 | The Company shall not be bound to issue more than one certificate for Shares held jointly by several persons and delivery of a certificate for a Share to one joint holder shall be a sufficient delivery to all of them. |
Renewal of lost or damaged share certificates
| 3.5 | If a share certificate is defaced, worn-out, lost or destroyed, it may be renewed on such terms (if any) as to: |
| (a) | evidence; |
| (b) | indemnity; |
| (c) | payment of the expenses reasonably incurred by the Company in investigating the evidence; and |
| (d) | payment of a reasonable fee, if any for issuing a replacement share certificate, |
as the Directors may determine, and (in the case of defacement or wearing-out) on delivery to the Company of the old certificate.
| 4 | Lien on Shares |
Nature and scope of lien
| 4.1 | The Company has a first and paramount lien on all Shares (whether Fully Paid Up or not) registered in the name of a Member (whether solely or jointly with others). The lien is for all monies payable to the Company by the Member or the Member’s estate: |
| (a) | either alone or jointly with any other person, whether or not that other person is a Member; and |
| 10 |
| (b) | whether or not those monies are presently payable. |
| 4.2 | At any time the Board may declare any Share to be wholly or partly exempt from the provisions of this Article. |
Company may sell Shares to satisfy lien
| 4.3 | The Company may sell any Shares over which it has a lien if all of the following conditions are met: |
| (a) | the sum in respect of which the lien exists is presently payable; |
| (b) | the Company gives notice to the Member holding the Share (or to the person entitled to it in consequence of the death or bankruptcy of that Member) demanding payment and stating that if the notice is not complied with the Shares may be sold; and |
| (c) | that sum is not paid within fourteen Clear Days after that notice is deemed to be given under these Articles, |
and Shares to which this Article 4.3 applies shall be referred to as Lien Default Shares.
| 4.4 | The Lien Default Shares may be sold in such manner as the Board determines. |
| 4.5 | To the maximum extent permitted by law, the Directors shall incur no personal liability to the Member concerned in respect of the sale. |
Authority to execute instrument of transfer
| 4.6 | To give effect to a sale, the Directors may authorise any person to execute an instrument of transfer of the Lien Default Shares sold to, or in accordance with the directions of, the purchaser. |
| 4.7 | The title of the transferee of the Lien Default Shares shall not be affected by any irregularity or invalidity in the proceedings in respect of the sale. |
Consequences of sale of Shares to satisfy lien
| 4.8 | On a sale pursuant to the preceding Articles: |
| (a) | the name of the Member concerned shall be removed from the register of Members as the holder of those Lien Default Shares; and |
| (b) | that person shall deliver to the Company for cancellation the certificate (if any) for those Lien Default Shares. |
| 11 |
| 4.9 | Notwithstanding the provisions of Article 4.8, such person shall remain liable to the Company for all monies which, at the date of sale, were presently payable by him to the Company in respect of those Lien Default Shares. That person shall also be liable to pay interest on those monies from the date of sale until payment at the rate at which interest was payable before that sale or, failing that, at the Default Rate. The Board may waive payment wholly or in part or enforce payment without any allowance for the value of the Lien Default Shares at the time of sale or for any consideration received on their disposal. |
Application of proceeds of sale
| 4.10 | The net proceeds of the sale, after payment of the costs, shall be applied in payment of so much of the sum for which the lien exists as is presently payable. Any residue shall be paid to the person whose Lien Default Shares have been sold: |
| (a) | if no certificate for the Lien Default Shares was issued, at the date of the sale; or |
| (b) | if a certificate for the Lien Default Shares was issued, upon surrender to the Company of that certificate for cancellation |
but, in either case, subject to the Company retaining a like lien for all sums not presently payable as existed on the Lien Default Shares before the sale.
| 5 | Calls on Shares and forfeiture |
Power to make calls and effect of calls
| 5.1 | Subject to the terms of allotment, the Board may make calls on the Members in respect of any monies unpaid on their Shares including any premium. The call may provide for payment to be by instalments. Subject to receiving at least 14 Clear Days’ notice specifying when and where payment is to be made, each Member shall pay to the Company the amount called on his Shares as required by the notice. |
| 5.2 | Before receipt by the Company of any sum due under a call, that call may be revoked in whole or in part and payment of a call may be postponed in whole or in part. Where a call is to be paid in instalments, the Company may revoke the call in respect of all or any remaining instalments in whole or in part and may postpone payment of all or any of the remaining instalments in whole or in part. |
| 5.3 | A Member on whom a call is made shall remain liable for that call notwithstanding the subsequent transfer of the Shares in respect of which the call was made. He shall not be liable for calls made after he is no longer registered as Member in respect of those Shares. |
Time when call made
| 5.4 | A call shall be deemed to have been made at the time when the resolution of the Directors authorising the call was passed. |
| 12 |
Liability of joint holders
| 5.5 | Members registered as the joint holders of a Share shall be jointly and severally liable to pay all calls in respect of the Share. |
Interest on unpaid calls
| 5.6 | If a call remains unpaid after it has become due and payable the person from whom it is due and payable shall pay interest on the amount unpaid from the day it became due and payable until it is paid: |
| (a) | at the rate fixed by the terms of allotment of the Share or in the notice of the call; or |
| (b) | if no rate is fixed, at the Default Rate. |
The Directors may waive payment of the interest wholly or in part.
Deemed calls
| 5.7 | Any amount payable in respect of a Share, whether on allotment or on a fixed date or otherwise, shall be deemed to be payable as a call. If the amount is not paid when due the provisions of these Articles shall apply as if the amount had become due and payable by virtue of a call. |
Power to accept early payment
| 5.8 | The Company may accept from a Member the whole or a part of the amount remaining unpaid on Shares held by him although no part of that amount has been called up. |
Power to make different arrangements at time of issue of Shares
| 5.9 | Subject to the terms of allotment, the Directors may make arrangements on the issue of Shares to distinguish between Members in the amounts and times of payment of calls on their Shares. |
Notice of default
| 5.10 | If a call remains unpaid after it has become due and payable the Directors may give to the person from whom it is due not less than 14 Clear Days’ notice requiring payment of: |
| (a) | the amount unpaid; |
| (b) | any interest which may have accrued; and |
| (c) | any expenses which have been incurred by the Company due to that person’s default. |
| 5.11 | The notice shall state the following: |
| (a) | the place where payment is to be made; and |
| 13 |
| (b) | a warning that if the notice is not complied with the Shares in respect of which the call is made will be liable to be forfeited. |
Forfeiture or surrender of Shares
| 5.12 | If the notice given pursuant to Article 5.10 is not complied with, the Directors may, before the payment required by the notice has been received, resolve that any Share the subject of that notice be forfeited. The forfeiture shall include all dividends or other monies payable in respect of the forfeited Share and not paid before the forfeiture. Despite the foregoing, the Board may determine that any Share the subject of that notice be accepted by the Company as surrendered by the Member holding that Share in lieu of forfeiture. |
Disposal of forfeited or surrendered Share and power to cancel forfeiture or surrender
| 5.13 | A forfeited or surrendered Share may be sold, re-allotted or otherwise disposed of on such terms and in such manner as the Board determine either to the former Member who held that Share or to any other person. The forfeiture or surrender may be cancelled on such terms as the Directors think fit at any time before a sale, re-allotment or other disposition. Where, for the purposes of its disposal, a forfeited or surrendered Share is to be transferred to any person, the Directors may authorise some person to execute an instrument of transfer of the Share to the transferee. |
Effect of forfeiture or surrender on former Member
| 5.14 | On forfeiture or surrender: |
| (a) | the name of the Member concerned shall be removed from the register of Members as the holder of those Shares and that person shall cease to be a Member in respect of those Shares; and |
| (b) | that person shall surrender to the Company for cancellation the certificate (if any) for the forfeited or surrendered Shares. |
| 5.15 | Despite the forfeiture or surrender of his Shares, that person shall remain liable to the Company for all monies which at the date of forfeiture or surrender were presently payable by him to the Company in respect of those Shares together with: |
| (a) | all expenses; and |
| (b) | interest from the date of forfeiture or surrender until payment: |
| (i) | at the rate of which interest was payable on those monies before forfeiture; or |
| (ii) | if no interest was so payable, at the Default Rate. |
The Directors, however, may waive payment wholly or in part.
| 14 |
Evidence of forfeiture or surrender
| 5.16 | A declaration, whether statutory or under oath, made by a Director or the Secretary shall be conclusive evidence of the following matters stated in it as against all persons claiming to be entitled to forfeited Shares: |
| (a) | that the person making the declaration is a Director or Secretary of the Company, and |
| (b) | that the particular Shares have been forfeited or surrendered on a particular date. |
Subject to the execution of an instrument of transfer, if necessary, the declaration shall constitute good title to the Shares.
Sale of forfeited or surrendered Shares
| 5.17 | Any person to whom the forfeited or surrendered Shares are disposed of shall not be bound to see to the application of the consideration, if any, of those Shares nor shall his title to the Shares be affected by any irregularity in, or invalidity of the proceedings in respect of, the forfeiture, surrender or disposal of those Shares. |
| 6 | Transfer of Shares |
Form of Transfer
| 6.1 | Subject to the following Articles about the transfer of Shares, and provided that such transfer complies with applicable rules of the Designated Stock Exchange, a Member may freely transfer Shares to another person by completing an instrument of transfer in a common form or in a form prescribed by the Designated Stock Exchange or in any other form approved by the Directors, executed: |
| (a) | where the Shares are Fully Paid, by or on behalf of that Member; and |
| (b) | where the Shares are partly paid, by or on behalf of that Member and the transferee. |
| 6.2 | The transferor shall be deemed to remain the holder of a Share until the name of the transferee is entered into the register of Members. |
Power to refuse registration for Shares not listed on a Designated Stock Exchange
| 6.3 | Where the Shares in question are not listed on or subject to the rules of any Designated Stock Exchange, the Directors may in their absolute discretion decline to register any transfer of such Shares which are not Fully Paid Up or on which the Company has a lien. The Directors may also, but are not required to, decline to register any transfer of any such Share unless: |
| (a) | the instrument of transfer is lodged with the Company, accompanied by the certificate (if any) for the Shares to which it relates and such other evidence as the Board may reasonably require to show the right of the transferor to make the transfer; |
| 15 |
| (b) | the instrument of transfer is in respect of only one class of Shares; |
| (c) | the instrument of transfer is properly stamped, if required; |
| (d) | in the case of a transfer to joint holders, the number of joint holders to whom the Share is to be transferred does not exceed four; |
| (e) | the Shares transferred are Fully Paid Up and free of any lien in favour of the Company; and |
| (f) | any applicable fee of such maximum sum as the Designated Stock Exchanges may determine to be payable, or such lesser sum as the Board may from time to time require, related to the transfer is paid to the Company. |
Suspension of transfers
| 6.4 | The registration of transfers may, on 14 Clear Days’ notice being given by advertisement in such one or more newspapers or by electronic means, be suspended and the register of Members closed at such times and for such periods as the Directors may, in their absolute discretion, from time to time determine, provided always that such registration of transfer shall not be suspended nor the register of Members closed for more than 30 Clear Days in any year. |
Company may retain instrument of transfer
| 6.5 | All instruments of transfer that are registered shall be retained by the Company. |
Notice of refusal to register
| 6.6 | If the Directors refuse to register a transfer of any Shares not listed on a Designated Stock Exchange, they shall within one month after the date on which the instrument of transfer was lodged with the Company send to each of the transferor and the transferee notice of the refusal. |
| 7 | Transmission of Shares |
Persons entitled on death of a Member
| 7.1 | If a Member dies, the only persons recognised by the Company as having any title to the deceased Members’ interest are the following: |
| (a) | where the deceased Member was a joint holder, the survivor or survivors; and |
| (b) | where the deceased Member was a sole holder, that Member’s personal representative or representatives. |
| 7.2 | Nothing in these Articles shall release the deceased Member’s estate from any liability in respect of any Share, whether the deceased was a sole holder or a joint holder. |
| 16 |
Registration of transfer of a Share following death or bankruptcy
| 7.3 | A person becoming entitled to a Share in consequence of the death or bankruptcy of a Member may elect to do either of the following: |
| (a) | to become the holder of the Share; or |
| (b) | to transfer the Share to another person. |
| 7.4 | That person must produce such evidence of his entitlement as the Directors may properly require. |
| 7.5 | If the person elects to become the holder of the Share, he must give notice to the Company to that effect. For the purposes of these Articles, that notice shall be treated as though it were an executed instrument of transfer. |
| 7.6 | If the person elects to transfer the Share to another person then: |
| (a) | if the Share is Fully Paid Up, the transferor must execute an instrument of transfer; and |
| (b) | if the Share is nil or Partly Paid Up, the transferor and the transferee must execute an instrument of transfer. |
| 7.7 | All the Articles relating to the transfer of Shares shall apply to the notice or, as appropriate, the instrument of transfer. |
Indemnity
| 7.8 | A person registered as a Member by reason of the death or bankruptcy of another Member shall indemnify the Company and the Directors against any loss or damage suffered by the Company or the Directors as a result of that registration. |
Rights of person entitled to a Share following death or bankruptcy
| 7.9 | A person becoming entitled to a Share by reason of the death or bankruptcy of a Member shall have the rights to which he would be entitled if he were registered as the holder of the Share. But, until he is registered as Member in respect of the Share, he shall not be entitled to attend or vote at any meeting of the Company or at any separate meeting of the holders of that class of Shares. |
| 8 | Alteration of capital |
Increasing, consolidating, converting, dividing and cancelling share capital
| 8.1 | To the fullest extent permitted by the Act, the Company may by Ordinary Resolution do any of the following and amend its Memorandum for that purpose: |
| (a) | increase its share capital by new Shares of the amount fixed by that Ordinary Resolution and with the attached rights, priorities and privileges set out in that Ordinary Resolution; |
| 17 |
| (b) | consolidate and divide all or any of its share capital into Shares of larger amount than its existing Shares; |
| (c) | convert all or any of its Paid Up Shares into stock, and reconvert that stock into Paid Up Shares of any denomination; |
| (d) | sub-divide its Shares or any of them into Shares of an amount smaller than that fixed by the Memorandum, so, however, that in the sub-division, the proportion between the amount paid and the amount, if any, unpaid on each reduced Share shall be the same as it was in case of the Share from which the reduced Share is derived; and |
| (e) | cancel Shares which, at the date of the passing of that Ordinary Resolution, have not been taken or agreed to be taken by any person, and diminish the amount of its share capital by the amount of the Shares so cancelled or, in the case of Shares without nominal par value, diminish the number of Shares into which its capital is divided. |
Dealing with fractions resulting from consolidation of Shares
| 8.2 | Whenever, as a result of a consolidation of Shares, any Members would become entitled to fractions of a Share the Directors may on behalf of those Members deal with the fractions as it thinks fit, including (without limitation): |
| (a) | sell the Shares representing the fractions for the best price reasonably obtainable to any person (including, subject to the provisions of the Act, the Company); and |
| (b) | distribute the net proceeds in due proportion among those Members. |
| 8.3 | For the purposes of Article 8.2, the Directors may authorise some person to execute an instrument of transfer of the Shares to, in accordance with the directions of, the purchaser. The transferee shall not be bound to see to the application of the purchase money nor shall the transferee’s title to the Shares be affected by any irregularity in, or invalidity of, the proceedings in respect of the sale. |
Reducing share capital
| 8.4 | Subject to the Act and to any rights for the time being conferred on the Members holding a particular class of Shares, the Company may, by Special Resolution, reduce its share capital in any way. |
| 18 |
| 9 | Redemption and purchase of own Shares |
Power to issue redeemable Shares and to purchase own Shares
| 9.1 | Subject to the Act and to any rights for the time being conferred on the Members holding a particular class of Shares, the Company may by its Directors: |
| (a) | issue Shares that are to be redeemed or liable to be redeemed, at the option of the Company or the Member holding those redeemable Shares, on the terms and in the manner its Directors determine before the issue of those Shares; |
| (b) | with the consent by Special Resolution of the Members holding Shares of a particular class, vary the rights attaching to that class of Shares so as to provide that those Shares are to be redeemed or are liable to be redeemed at the option of the Company on the terms and in the manner which the Directors determine at the time of such variation; and |
| (c) | purchase all or any of its own Shares of any class including any redeemable Shares on the terms and in the manner which the Directors determine at the time of such purchase. |
The Company may make a payment in respect of the redemption or purchase of its own Shares in any manner authorised by the Act, including out of any combination of the following: capital, its profits and the proceeds of a fresh issue of Shares.
Power to pay for redemption or purchase in cash or in specie
| 9.2 | When making a payment in respect of the redemption or purchase of Shares, the Directors may make the payment in cash or in specie (or partly in one and partly in the other) if so authorised by the terms of the allotment of those Shares or by the terms applying to those Shares in accordance with Article 9.1, or otherwise by agreement with the Member holding those Shares. |
Effect of redemption or purchase of a Share
| 9.3 | Upon the date of redemption or purchase of a Share: |
| (a) | the Member holding that Share shall cease to be entitled to any rights in respect of the Share other than the right to receive: |
| (i) | the price for the Share; and |
| (ii) | any dividend declared in respect of the Share prior to the date of redemption or purchase; |
| (b) | the Member’s name shall be removed from the register of Members with respect to the Share; and |
| 19 |
| (c) | the Share shall be cancelled or held as a Treasury Share, as the Directors may determine. |
| 9.4 | For the purpose of Article 9.3, the date of redemption or purchase is the date when the Member’s name is removed from the register of Members with respect to the Shares the subject of the redemption or purchase. |
| 10 | Meetings of Members |
Annual and extraordinary general meetings
| 10.1 | The Company may, but shall not (unless required by the Designated Stock Exchange Rules) be obligated to, in each year hold a general meeting as an annual general meeting, which, if held, shall be convened by the Board, in accordance with these Articles. |
| 10.2 | All general meetings other than annual general meetings shall be called extraordinary general meetings. |
Power to call meetings
| 10.3 | A majority of all the Directors or all of the Class A Directors may call a general meeting at any time. |
| 10.4 | If there are insufficient Directors to constitute a quorum and the remaining Directors are unable to agree on the appointment of additional Directors, the Directors must call a general meeting for the purpose of appointing additional Directors. |
| 10.5 | A majority of all the Directors or all of the Class A Directors must also call a general meeting if requisitioned in the manner set out in the next two Articles. |
| 10.6 | The requisition must be in writing and given by one or more Members who together hold at least ten per cent of the rights to vote at such general meeting. |
| 10.7 | The requisition must also: |
| (a) | specify the purpose of the meeting. |
| (b) | be signed by or on behalf of each requisitioner (and for this purpose each joint holder shall be obliged to sign). The requisition may consist of several documents in like form signed by one or more of the requisitioners; and |
| (c) | be delivered in accordance with the notice provisions. |
| 10.8 | Should the majority of all the Directors or all of the Class A Directors fail to call a general meeting within 21 Clear Days’ from the date of receipt of a requisition, the requisitioners or any of them may call a general meeting within three months after the end of that period. |
| 20 |
| 10.9 | Without limitation to the foregoing, if there are insufficient Directors to constitute a quorum and the remaining Directors are unable to agree on the appointment of additional Directors, any one or more Members who together hold at least five per cent of the rights to vote at a general meeting may call a general meeting for the purpose of considering the business specified in the notice of meeting which shall include as an item of business the appointment of additional Directors. |
| 10.10 | If the Members call a meeting under the above provisions, the Company shall reimburse their reasonable expenses. |
Content of notice
| 10.11 | Notice of a general meeting shall specify each of the following: |
| (a) | the date and the hour of the meeting; |
| (b) | whether the meeting will be held virtually, at a physical place or both; |
| (c) | if the meeting is to be held in any part at a physical place, the address of such place; |
| (d) | if the meeting is to be held in two or more places or in any part virtually, the technology that will be used to facilitate the meeting; |
| (e) | subject to paragraph (f) and the requirements of the Designated Stock Exchange Rules (to the extent applicable), the general nature of the business to be transacted; and |
| (f) | if a resolution is proposed as a Special Resolution, the text of that resolution. |
| 10.12 | In each notice there shall appear with reasonable prominence the following statements: |
| (a) | that a Member who is entitled to attend and vote is entitled to appoint one or more proxies to attend and vote instead of that Member; and |
| (b) | that a proxyholder need not be a Member. |
Period of notice
| 10.13 | At least five (5) Clear Days’ notice shall be given to Members for any general meeting. |
| 10.14 | Subject to the Act, a meeting may be convened on shorter notice, subject to the Act with the consent of the Member or Members who, individually or collectively, hold not less than 75 per cent of the voting rights of all those who have a right to vote at that meeting. |
Persons entitled to receive notice
| 10.15 | Subject to the provisions of these Articles and to any restrictions imposed on any Shares, the notice shall be given to the following people: |
| (a) | the Members |
| 21 |
| (b) | persons entitled to a Share in consequence of the death or bankruptcy of a Member; |
| (c) | the Directors; and |
| (d) | the Auditors (if appointed). |
| 10.16 | The Board may determine that the Members entitled to receive notice of, attend and vote at a meeting are those persons entered on the register of Members at the close of business on a day determined by the Board. |
Accidental omission to give notice or non-receipt of notice
| 10.17 | Proceedings at a meeting shall not be invalidated by the following: |
| (a) | an accidental failure to give notice of the meeting to any person entitled to notice; or |
| (b) | non-receipt of notice of the meeting by any person entitled to notice. |
| 10.18 | In addition, where a notice of meeting is published on a website proceedings at the meeting shall not be invalidated merely because it is accidentally published: |
| (a) | in a different place on the website; or |
| (b) | for part only of the period from the date of the notification until the conclusion of the meeting to which the notice relates. |
| 11 | Proceedings at meetings of Members |
Quorum
| 11.1 | Save as provided in the following Article, no business shall be transacted at any meeting unless a quorum is present in person or by proxy. A quorum is as follows: |
| (a) | if the Company has only one Member: that Member; |
| (b) | if the Company has more than one Member: one or more Members holding Shares that represent not less than one-third of the outstanding Shares carrying the right to vote at such general meeting. |
Lack of quorum
| 11.2 | If a quorum is not present within fifteen minutes of the time appointed for the meeting, or if at any time during the meeting it becomes inquorate, then the following provisions apply: |
| (a) | If the meeting was requisitioned by Members, it shall be cancelled. |
| 22 |
| (b) | In any other case, the meeting shall stand adjourned to the same time and place seven days hence, or to such other time or place as is determined by the Directors. If a quorum is not present within fifteen minutes of the time appointed for the adjourned meeting, then the Members present in person or by proxy shall constitute a quorum. |
Chairman
| 11.3 | The chairman of a general meeting shall be the chairman of the Board or such other Director as the Directors may determine. Absent any such person being present within fifteen minutes of the time appointed for the meeting, the Directors present shall elect one of their number to chair the meeting. |
| 11.4 | If no Director is present within fifteen minutes of the time appointed for the meeting, or if no Director is willing to act as chairman, the Members present in person or by proxy and entitled to vote shall choose one of their number to chair the meeting. |
Right of a Director to attend and speak
| 11.5 | Even if a Director is not a Member, he shall be entitled to attend and speak at any general meeting and at any separate meeting of Members holding a particular class of Shares. |
Accommodation of Members attending meeting virtually
| 11.6 | A Member entitled to receive notice and attend a meeting will be deemed to be in attendance at such meeting despite their attendance being virtual if adequate facilities are available to ensure that the Member is able to: |
| (a) | participate in the business for which the meeting has been convened; and |
| (b) | hear all that happens at the meeting. |
Security
| 11.7 | In addition to any measures which the Board may be required to take due to the location or venue of the meeting, the Board may make any arrangement and impose any restriction it considers appropriate and reasonable in the circumstances to ensure the security of a meeting including, without limitation, the searching of any person attending the meeting and the imposing of restrictions on the items of personal property that may be taken into the meeting place. The Board may refuse entry to, or eject from, a meeting a person who refuses to comply with any such arrangements or restrictions. |
| 23 |
Adjournment, postponement and cancellation
| 11.8 | A meeting may be: |
| (a) | postponed or cancelled prior to the meeting at the discretion of the Directors by written notice provided to all persons entitled to attend the meeting, unless the meeting was requisitioned by Members or otherwise called by Members pursuant to Article 10; or |
| (b) | adjourned, with or without an appointed date for resumption, at any time during the meeting at the discretion of the chairman with the consent of the Members constituting a quorum. |
| 11.9 | The chairman must adjourn the meeting if so directed by Members constituting a quorum at the meeting. No business, however, can be transacted at an adjourned or postponed meeting other than business which might properly have been transacted at the original meeting. |
| 11.10 | Should a meeting be adjourned for more than 7 Clear Days, whether because of a lack of quorum or otherwise, Members shall be given at least seven Clear Days’ notice of the date, time and place of the adjourned meeting and the general nature of the business to be transacted. Otherwise it shall not be necessary to give any notice of the adjournment. |
Method of voting
| 11.11 | A resolution put to the vote of the meeting shall be decided on a poll. |
Taking of a poll
| 11.12 | A poll on the question of adjournment shall be taken immediately. |
| 11.13 | A poll shall be taken in such manner as the chairman directs. He may appoint scrutineers (who need not be Members) and fix a place and time for declaring the result of the poll. If, through the aid of technology, the meeting is held virtually or in more than one place, the chairman may appoint scrutineers virtually and in more than one place; but if he considers that the poll cannot be effectively monitored at that meeting, the chairman shall adjourn the holding of the poll to a date, place and time when that can occur. |
Chairman’s casting vote
| 11.14 | In the case of an equality of votes, the chairman of the meeting at which the poll is taken shall be entitled to a second or casting vote. |
Written resolutions
| 11.15 | Members may pass a resolution in writing without holding a meeting if the following conditions are met: |
| (a) | all Members entitled to vote are given notice of the resolution as if the same were being proposed at a meeting of Members; |
| 24 |
| (b) | all Members entitled so to vote; |
| (i) | sign a document; or |
| (ii) | sign several documents in the like form each signed by one or more of those Members; and |
| (c) | the signed document or documents is or are delivered to the Company, including, if the Company so nominates, by delivery of an Electronic Record by Electronic means to the address specified for that purpose. |
| (d) | Such written resolution shall be as effective as if it had been passed at a meeting of the Members entitled to vote duly convened and held. |
| 11.16 | If a written resolution is described as a Special Resolution or as an Ordinary Resolution, it has effect accordingly. |
| 11.17 | The Directors may determine the manner in which written resolutions shall be put to Members. In particular, they may provide, in the form of any written resolution, for each Member to indicate, out of the number of votes the Member would have been entitled to cast at a meeting to consider the resolution, how many votes he wishes to cast in favour of the resolution and how many against the resolution or to be treated as abstentions. The result of any such written resolution shall be determined on the same basis as on a poll. |
Sole-Member Company
| 11.18 | If the Company has only one Member, and the Member records in writing his decision on a question, that record shall constitute both the passing of a resolution and the minute of it. |
| 12 | Voting rights of Members |
Right to vote
| 12.1 | Unless their Shares carry no right to vote, or unless a call or other amount presently payable has not been paid, all Members are entitled to vote at a general meeting, and all Members holding Shares of a particular class of Shares are entitled to vote at a meeting of the holders of that class of Shares. |
| 12.2 | Members may vote in person or by proxy. |
| 12.3 | On a poll a Member shall have one vote for each Share he holds, unless any Share carries special voting rights. |
| 12.4 | No Member is bound to vote on his Shares or any of them; nor is he bound to vote each of his Shares in the same way. |
| 25 |
Rights of joint holders
| 12.5 | If Shares are held jointly, only one of the joint holders may vote. If more than one of the joint holders tenders a vote, the vote of the holder whose name in respect of those Shares appears first in the register of Members shall be accepted to the exclusion of the votes of the other joint holder. |
Representation of corporate Members
| 12.6 | Save where otherwise provided, a corporate Member must act by a duly authorised representative. |
| 12.7 | A corporate Member wishing to act by a duly authorised representative must identify that person to the Company by notice in writing. |
| 12.8 | The authorisation may be for any period of time, and must be delivered to the Company before the commencement of the meeting at which it is first used. |
| 12.9 | The Directors of the Company may require the production of any evidence which they consider necessary to determine the validity of the notice. |
| 12.10 | Where a duly authorised representative is present at a meeting that Member is deemed to be present in person; and the acts of the duly authorised representative are personal acts of that Member. |
| 12.11 | A corporate Member may revoke the appointment of a duly authorised representative at any time by notice to the Company; but such revocation will not affect the validity of any acts carried out by the duly authorised representative before the Directors of the Company had actual notice of the revocation. |
Member with mental disorder
| 12.12 | A Member in respect of whom an order has been made by any court having jurisdiction (whether in the Cayman Islands or elsewhere) in matters concerning mental disorder may vote by that Member’s receiver, curator bonis or other person authorised in that behalf appointed by that court. |
| 12.13 | For the purpose of the preceding Article, evidence to the satisfaction of the Directors of the authority of the person claiming to exercise the right to vote must be received not less than 24 hours before holding the relevant meeting or the adjourned meeting in any manner specified for the delivery of forms of appointment of a proxy, whether in writing or by Electronic means. In default, the right to vote shall not be exercisable. |
| 26 |
Objections to admissibility of votes
| 12.14 | An objection to the validity of a person’s vote may only be raised at the meeting or at the adjourned meeting at which the vote is sought to be tendered. Any objection duly made shall be referred to the chairman whose decision shall be final and conclusive. |
Form of proxy
| 12.15 | An instrument appointing a proxy shall be in any common form or in any other form approved by the Directors. |
| 12.16 | The instrument must be in writing and signed in one of the following ways: |
| (a) | by the Member; or |
| (b) | by the Member’s authorised attorney; or |
| (c) | if the Member is a corporation or other body corporate, under seal or signed by an authorised officer, secretary or attorney. |
If the Directors so resolve, the Company may accept an Electronic Record of that instrument delivered in the manner specified below and otherwise satisfying the Articles about authentication of Electronic Records.
| 12.17 | The Directors may require the production of any evidence which they consider necessary to determine the validity of any appointment of a proxy. |
| 12.18 | A Member may revoke the appointment of a proxy at any time by notice to the Company duly signed in accordance with Article 12.16. |
| 12.19 | No revocation by a Member of the appointment of a proxy made in accordance with Article 12.18 will affect the validity of any acts carried out by the relevant proxy before the Directors of the Company had actual notice of the revocation. |
How and when proxy is to be delivered
| 12.20 | Subject to the following Articles, the Directors may, in the notice convening any meeting or adjourned meeting, or in an instrument of proxy sent out by the Company, specify the manner by which the instrument appointing a proxy shall be deposited and the place and the time (being not later than the time appointed for the commencement of the meeting or adjourned meeting to which the proxy relates) at which the instrument appointing a proxy shall be deposited. In the absence of any such direction from the Directors in the notice convening any meeting or adjourned meeting or in an instrument of proxy sent out by the Company, the form of appointment of a proxy and any authority under which it is signed (or a copy of the authority certified notarially or in any other way approved by the Directors) must be delivered so that it is received by the Company before the time for holding the meeting or adjourned meeting at which the person named in the form of appointment of proxy proposes to vote. They must be delivered in either of the following ways: |
| (a) | In the case of an instrument in writing, it must be left at or sent by post: |
| (i) | to the registered office of the Company; or |
| 27 |
| (ii) | to such other place within the Cayman Islands specified in the notice convening the meeting or in any form of appointment of proxy sent out by the Company in relation to the meeting. |
| (b) | If, pursuant to the notice provisions, a notice may be given to the Company in an Electronic Record, an Electronic Record of an appointment of a proxy must be sent to the address specified pursuant to those provisions unless another address for that purpose is specified: |
| (i) | in the notice convening the meeting; or |
| (ii) | in any form of appointment of a proxy sent out by the Company in relation to the meeting; or |
| (iii) | in any invitation to appoint a proxy issued by the Company in relation to the meeting. |
| (c) | Notwithstanding Article 12.20(a) and Article 12.20(b), the chairman of the Company may, in any event at his discretion, direct that an instrument of proxy shall be deemed to have been duly deposited. |
| 12.21 | If the form of appointment of proxy is not delivered on time, it is invalid. |
| 12.22 | When two or more valid but differing appointments of proxy are delivered or received in respect of the same Share for use at the same meeting and in respect of the same matter, the one which is last validly delivered or received (regardless of its date or of the date of its execution) shall be treated as replacing and revoking the other or others as regards that Share. If the Company is unable to determine which appointment was last validly delivered or received, none of them shall be treated as valid in respect of that Share. |
| 12.23 | The Board may at the expense of the Company send forms of appointment of proxy to the Members by post (that is to say, pre-paying and posting a letter), or by Electronic communication or otherwise (with or without provision for their return by pre-paid post) for use at any general meeting or at any separate meeting of the holders of any class of Shares, either blank or nominating as proxy in the alternative any one or more of the Directors or any other person. If for the purpose of any meeting invitations to appoint as proxy a person or one of a number of persons specified in the invitations are issued at the Company’s expense, they shall be issued to all (and not to some only) of the Members entitled to be sent notice of the meeting and to vote at it. The accidental omission to send such a form of appointment or to give such an invitation to, or the non-receipt of such form of appointment by, any Member entitled to attend and vote at a meeting shall not invalidate the proceedings at that meeting |
| 28 |
Voting by proxy
| 12.24 | A proxy shall have the same voting rights at a meeting or adjourned meeting as the Member would have had except to the extent that the instrument appointing him limits those rights. Notwithstanding the appointment of a proxy, a Member may attend and vote at a meeting or adjourned meeting. If a Member votes on any resolution a vote by his proxy on the same resolution, unless in respect of different Shares, shall be invalid. |
| 12.25 | The appointment of a proxy shall not confer on that proxy any right to speak at the meeting, except with the permission of the chairman of the meeting. |
| 13 | Number of Directors |
| 13.1 | There shall be a Board consisting of not less than one person provided however that the Company may by Ordinary Resolution increase or reduce the limits in the number of Directors. Unless fixed by Ordinary Resolution, the maximum number of Directors shall be unlimited. |
| 14 | Appointment, disqualification and removal of Directors |
No age limit
| 14.1 | There is no age limit for Directors save that they must be at least eighteen years of age. |
Corporate Directors
| 14.2 | Unless prohibited by law, a body corporate may be a Director. If a body corporate is a Director, the Articles about representation of corporate Members at general meetings apply, mutatis mutandis, to the Articles about Directors’ meetings. |
No shareholding qualification
| 14.3 | Unless a shareholding qualification for Directors is fixed by Ordinary Resolution, no Director shall be required to own Shares as a condition of his appointment. |
Appointment of Directors
| 14.4 | The Directors shall be divided into two classes: Class A and Class B. An executive Director shall be a Class A Director (a Class A Director). Each Independent Director shall be a Class B Director (a Class B Director) . Any Director that is not an Independent Director but is non-executive shall also be a Class B Director. Upon the adoption of the Articles, the existing Directors shall by resolution classify themselves as Class A or Class B as applicable. A Director may be appointed by Ordinary Resolution or by the Directors. Any appointment may be to fill a vacancy or as an additional Director. |
| 29 |
| 14.5 | The remaining Director(s) may appoint a Director even though there is not a quorum of Directors. |
| 14.6 | No appointment can cause the number of Directors to exceed the maximum (if one is set); and any such appointment shall be invalid. |
| 14.7 | For so long as Shares are listed on a Designated Stock Exchange, the Directors shall include at least such number of Independent Directors as applicable law, rules or regulations or the Designated Stock Exchange Rules require as determined by the Board. |
Board’s power to appoint Directors
| 14.8 | Without prejudice to the Company’s power to appoint a person to be a Director pursuant to these Articles, the Board shall have power at any time to appoint any person who is willing to act as a Director, either to fill a vacancy or as an addition to the existing Board, subject to the total number of Directors not exceeding any maximum number fixed by or in accordance with these Articles. |
Term of appointment
| 14.9 | Subject to all applicable laws and the Designated Stock Exchange Rules: |
| (a) | each Class A Director shall hold office until such Director resigns, is removed from office, or otherwise vacates the office. There shall be no requirement for any Class A Director to retire or be re-elected at any annual general meeting of the Company or upon any specified event. A Class A Director elected to fill a vacancy resulting from the death, resignation or removal of a Director shall serve for the remainder of the full term of the Director whose death, resignation or removal shall have created such vacancy and until his successor shall have been elected and qualified. |
| (b) | an appointment of a Class B Director may be on terms that the Director shall automatically retire from office (unless he has sooner vacated office) at the next or a subsequent annual general meeting or upon any specified event or after any specified period in a written agreement between the Company and the Class B Director, if any; but no such term shall be implied in the absence of express provision. Each Class B Director whose term of office expires shall be eligible for re-election at a meeting of the Shareholders or re-appointment by the Board. |
Removal of Directors
| 14.10 | A Director may be removed by Ordinary Resolution. |
Resignation of Directors
| 14.11 | A Director may at any time resign office by giving to the Company notice in writing or, if permitted pursuant to the notice provisions, in an Electronic Record delivered in either case in accordance with those provisions. |
| 30 |
| 14.12 | Unless the notice specifies a different date, the Director shall be deemed to have resigned on the date that the notice is delivered to the Company. |
Termination of the office of Director
| 14.13 | A Director may retire from office as a Director by giving notice in writing to that effect to the Company at the registered office, which notice shall be effective upon such date as may be specified in the notice, failing which upon delivery to the registered office. |
| 14.14 | Without prejudice to the provisions in these Articles for retirement (by rotation or otherwise), a Director’s office shall be terminated forthwith if: |
| (a) | he is prohibited by the law of the Cayman Islands from acting as a Director; or |
| (b) | he is made bankrupt or makes an arrangement or composition with his creditors generally; or |
| (c) | he resigns his office by notice to the Company; or |
| (d) | he only held office as a Director for a fixed term and such term expires; or |
| (e) | in the opinion of a registered medical practitioner by whom he is being treated he becomes physically or mentally incapable of acting as a Director; or |
| (f) | he is given notice by the majority of the other Directors (not being less than two in number) to vacate office (without prejudice to any claim for damages for breach of any agreement relating to the provision of the services of such Director); or |
| (g) | he is made subject to any law relating to mental health or incompetence, whether by court order or otherwise; or |
| (h) | without the consent of the other Directors, he is absent from meetings of Directors for a continuous period of six months. |
| 15 | Alternate Directors |
Appointment and removal
| 15.1 | Any Director may appoint any other person, including another Director, to act in his place as an alternate Director. No appointment shall take effect until the Director has given notice of the appointment to the Board. |
| 15.2 | A Director may revoke his appointment of an alternate at any time. No revocation shall take effect until the Director has given notice of the revocation to the Board. |
| 31 |
| 15.3 | A notice of appointment or removal of an alternate Director shall be effective only if given to the Company by one or more of the following methods: |
| (a) | by notice in writing in accordance with the notice provisions contained in these Articles; |
| (b) | if the Company has a facsimile address for the time being, by sending by facsimile transmission to that facsimile address a facsimile copy or, otherwise, by sending by facsimile transmission to the facsimile address of the Company’s registered office a facsimile copy (in either case, the facsimile copy being deemed to be the notice unless Article 29.7 applies), in which event notice shall be taken to be given on the date of an error-free transmission report from the sender’s fax machine; |
| (c) | if the Company has an email address for the time being, by emailing to that email address a scanned copy of the notice as a PDF attachment or, otherwise, by emailing to the email address provided by the Company’s registered office a scanned copy of the notice as a PDF attachment (in either case, the PDF version being deemed to be the notice unless Article 29.7 applies), in which event notice shall be taken to be given on the date of receipt by the Company or the Company’s registered office (as appropriate) in readable form; or |
| (d) | if permitted pursuant to the notice provisions, in some other form of approved Electronic Record delivered in accordance with those provisions in writing. |
Notices
| 15.4 | All notices of meetings of Directors shall continue to be given to the appointing Director and not to the alternate. |
Rights of alternate Director
| 15.5 | An alternate Director shall be entitled to attend and vote at any Board meeting or meeting of a committee of the Directors at which the appointing Director is not personally present, and generally to perform all the functions of the appointing Director in his absence. An alternate Director, however, is not entitled to receive any remuneration from the Company for services rendered as an alternate Director. |
Appointment ceases when the appointor ceases to be a Director
| 15.6 | An alternate Director shall cease to be an alternate Director if: |
| (a) | the Director who appointed him ceases to be a Director; or |
| (b) | the Director who appointed him revokes his appointment by notice delivered to the Board or to the registered office of the Company or in any other manner approved by the Board; or |
| (c) | in any event happens in relation to him which, if he were a Director of the Company, would cause his office as Director to be vacated. |
| 32 |
Status of alternate Director
| 15.7 | An alternate Director shall carry out all functions of the Director who made the appointment. |
| 15.8 | Save where otherwise expressed, an alternate Director shall be treated as a Director under these Articles. |
| 15.9 | An alternate Director is not the agent of the Director appointing him. |
| 15.10 | An alternate Director is not entitled to any remuneration for acting as alternate Director. |
Status of the Director making the appointment
| 15.11 | A Director who has appointed an alternate is not thereby relieved from the duties which he owes the Company. |
| 16 | Powers of Directors |
Powers of Directors
| 16.1 | Subject to the provisions of the Act, the Memorandum and these Articles the business of the Company shall be managed by the Directors who may for that purpose exercise all the powers of the Company. |
| 16.2 | No prior act of the Directors shall be invalidated by any subsequent alteration of the Memorandum or these Articles. However, to the extent allowed by the Act, Members may, by Special Resolution, validate any prior or future act of the Directors which would otherwise be in breach of their duties. |
Directors below the minimum number
| 16.3 | If the number of Directors is less than the minimum prescribed in accordance with these Articles, the remaining Director or Directors shall act only for the purposes of appointing an additional Director or Directors to make up such minimum or of convening a general meeting of the Company for the purpose of making such appointment. If there are no Director or Directors able or willing to act, any two Members may summon a general meeting for the purpose of appointing Directors. Any additional Director so appointed shall hold office (subject to these Articles) only until the dissolution of the annual general meeting next following such appointment unless he is re-elected during such meeting. |
Appointments to office
| 16.4 | The Directors may appoint a Director: |
| (a) | as chairman of the Board; |
| (b) | as managing Director; |
| 33 |
| (c) | to any other executive office, |
for such period, and on such terms, including as to remuneration as they think fit.
| 16.5 | The appointee must consent in writing to holding that office. |
| 16.6 | Where a chairman is appointed he shall, unless unable to do so, preside at every meeting of Directors. |
| 16.7 | If there is no chairman, or if the chairman is unable to preside at a meeting, that meeting may select its own chairman; or the Directors may nominate one of their number to act in place of the chairman should he ever not be available. |
| 16.8 | Subject to the provisions of the Act, the Directors may also appoint and remove any person, who need not be a Director: |
| (a) | as Secretary; and |
| (b) | to any office that may be required |
for such period and on such terms, including as to remuneration, as they think fit. In the case of an Officer, that Officer may be given any title the Directors decide.
| 16.9 | The Secretary or Officer must consent in writing to holding that office. |
| 16.10 | A Director, Secretary or other Officer of the Company may not the hold the office, or perform the services, of auditor. |
Provisions for employees
| 16.11 | The Board may make provision for the benefit of any persons employed or formerly employed by the Company or any of its subsidiary undertakings (or any member of his family or any person who is dependent on him) in connection with the cessation or the transfer to any person of the whole or part of the undertaking of the Company or any of its subsidiary undertakings. |
Exercise of voting rights
| 16.12 | The Board may exercise the voting power conferred by the Shares in any body corporate held or owned by the Company in such manner in all respects as it thinks fit (including, without limitation, the exercise of that power in favour of any resolution appointing any Director as a Director of such body corporate, or voting or providing for the payment of remuneration to the Directors of such body corporate). |
Remuneration
| 16.13 | Every Director may be remunerated by the Company for the services he provides for the benefit of the Company, whether as Director, employee or otherwise, and shall be entitled to be paid for the expenses incurred in the Company’s business including attendance at Directors’ meetings. |
| 34 |
| 16.14 | Until otherwise determined by the Company by Ordinary Resolution, the Directors (other than alternate Directors) shall be entitled to such remuneration by way of fees for their services in the office of Director as the Directors may determine. |
| 16.15 | Remuneration may take any form and may include arrangements to pay pensions, health insurance, death or sickness benefits, whether to the Director or to any other person connected to or related to him. |
| 16.16 | Unless his fellow Directors determine otherwise, a Director is not accountable to the Company for remuneration or other benefits received from any other company which is in the same group as the Company or which has common shareholdings. |
Disclosure of information
| 16.17 | Subject to the compliance with applicable laws, including the federal securities laws of the United States, the Directors may release or disclose to a third party any information regarding the affairs of the Company, including any information contained in the register of Members relating to a Member, (and they may authorise any Director, Officer or other authorised agent of the Company to release or disclose to a third party any such information in his possession) if: |
| (a) | the Company or that person, as the case may be, is lawfully required to do so under the laws of any jurisdiction to which the Company is subject; or |
| (b) | such disclosure is in compliance with the Designated Stock Exchange Rules; or |
| (c) | such disclosure is in accordance with any contract entered into by the Company; or |
| (d) | the Directors are of the opinion such disclosure would assist or facilitate the Company’s operations. |
| 17 | Delegation of powers |
Power to delegate any of the Directors’ powers to a committee
| 17.1 | The Directors may delegate any of their powers to any committee consisting of one or more persons who need not be Members. Persons on the committee may include non-Directors so long as the majority of those persons are Directors. For so long as Shares are listed on a Designated Stock Exchange, any such committee shall be made up of such number of Independent Directors as required from time to time by the Designated Stock Exchange Rules or otherwise required by applicable law. |
| 17.2 | The delegation may be collateral with, or to the exclusion of, the Directors’ own powers. |
| 35 |
| 17.3 | The delegation may be on such terms as the Directors think fit, including provision for the committee itself to delegate to a sub-committee; save that any delegation must be capable of being revoked or altered by the Directors at will. |
| 17.4 | Unless otherwise permitted by the Directors, a committee must follow the procedures prescribed for the taking of decisions by Directors. |
| 17.5 | For so long as Shares are listed on a Designated Stock Exchange, the Board shall establish an audit committee, a compensation committee and a nominating and corporate governance committee. Each of these committees shall be empowered to do all things necessary to exercise the rights of such committee set forth in these Articles. Each of the audit committee, compensation committee and nominating and corporate governance committee shall consist of at least three Directors (or such larger minimum number as may be required from time to time by the Designated Stock Exchange Rules). The committees shall be made up of such number of Independent Directors as required from time to time by the Designated Stock Exchange Rules or otherwise required by applicable law, subject to any exemptions permitted under the Designated Stock Exchange Rules and other applicable laws. |
Local boards
| 17.6 | The Board may establish any local or divisional board or agency for managing any of the affairs of the Company whether in the Cayman Islands or elsewhere and may appoint any persons to be members of a local or divisional Board, or to be managers or agents, and may fix their remuneration. |
| 17.7 | The Board may delegate to any local or divisional board, manager or agent any of its powers and authorities (with power to sub-delegate) and may authorise the members of any local or divisional board or any of them to fill any vacancies and to act notwithstanding vacancies. |
| 17.8 | Any appointment or delegation under this Article 17.8 may be made on such terms and subject to such conditions as the Board thinks fit and the Board may remove any person so appointed, and may revoke or vary any delegation. |
Power to appoint an agent of the Company
| 17.9 | The Directors may appoint any person, either generally or in respect of any specific matter, to be the agent of the Company with or without authority for that person to delegate all or any of that person’s powers. The Directors may make that appointment: |
| (a) | by causing the Company to enter into a power of attorney or agreement; or |
| (b) | in any other manner they determine. |
| 36 |
Power to appoint an attorney or authorised signatory of the Company
| 17.10 | The Directors may appoint any person, whether nominated directly or indirectly by the Directors, to be the attorney or the authorised signatory of the Company. The appointment may be: |
| (a) | for any purpose; |
| (b) | with the powers, authorities and discretions; |
| (c) | for the period; and |
| (d) | subject to such conditions |
as they think fit. The powers, authorities and discretions, however, must not exceed those vested in, or exercisable, by the Directors under these Articles. The Directors may do so by power of attorney or any other manner they think fit.
| 17.11 | Any power of attorney or other appointment may contain such provision for the protection and convenience for persons dealing with the attorney or authorised signatory as the Directors think fit. Any power of attorney or other appointment may also authorise the attorney or authorised signatory to delegate all or any of the powers, authorities and discretions vested in that person. |
| 17.12 | The Board may remove any person appointed under Article 17.10 and may revoke or vary the delegation. |
Borrowing Powers
| 17.13 | The Directors may exercise all the powers of the Company to borrow money and to mortgage or charge its undertaking, property and assets both present and future and uncalled capital, or any part thereof, and to issue debentures and other securities, whether outright or as collateral security for any debt, liability or obligation of the Company or its parent undertaking (if any) or any subsidiary undertaking of the Company or of any third party. |
Corporate Governance
| 17.14 | The Board may, from time to time, and except as required by applicable law or the Designated Stock Exchange Rules, adopt, institute, amend, modify or revoke the corporate governance policies or initiatives of the Company, which shall be intended to set forth the guiding principles and policies of the Company and the Board on various corporate governance related matters as the Board shall determine by resolution from time to time. |
| 37 |
| 18 | Meetings of Directors |
Regulation of Directors’ meetings
| 18.1 | Subject to the provisions of these Articles, the Directors may regulate their proceedings as they think fit. |
Calling meetings
| 18.2 | Any Director may call a meeting of Directors at any time. The Secretary must call a meeting of the Directors if requested to do so by a Director. |
Notice of meetings
| 18.3 | Notice of a Board meeting may be given to a Director personally or by word of mouth or given in writing or by Electronic communications at such address as he may from time to time specify for this purpose (or, if he does not specify an address, at his last known address). A Director may waive his right to receive notice of any meeting either prospectively or retrospectively. |
Use of technology
| 18.4 | A Director may participate in a meeting of Directors through the medium of conference telephone, video or any other form of communications equipment providing all persons participating in the meeting are able to hear and speak to each other throughout the meeting. |
| 18.5 | A Director participating in this way is deemed to be present in person at the meeting. |
Quorum
| 18.6 | The quorum for the transaction of business at a meeting of Directors shall be two unless the Directors fix some other number. |
Chairman or deputy to preside
| 18.7 | The chairman of the Board shall be Dr. Tham Seng Kong, as long as Dr. Tham Seng Kong is a Director. In the event that Dr. Tham Seng Kong is no longer a Director, the Board may appoint a chairman and one or more deputy chairman or chairmen and may at any time revoke any such appointment by the affirmative vote of a simple majority of the Directors then in office, and the period for which the chairman will hold office will also be determined by the affirmative vote of a simple majority of the Directors then in office. |
| 18.8 | The chairman, or failing him any deputy chairman (the longest in office taking precedence if more than one is present), shall preside at all Board meetings. If no chairman or deputy chairman has been appointed, or if he is not present within five minutes after the time fixed for holding the meeting, or is unwilling to act as chairman of the meeting, the Directors present shall choose one of their number to act as chairman of the meeting. |
| 38 |
Voting
| 18.9 | A question which arises at a Board meeting shall be decided by a majority of votes. If votes are equal the chairman may, if he wishes, exercise a casting vote. |
Recording of dissent
| 18.10 | A Director present at a meeting of Directors shall be presumed to have assented to any action taken at that meeting unless: |
| (a) | his dissent is entered in the minutes of the meeting; or |
| (b) | he has filed with the meeting before it is concluded signed dissent from that action; or |
| (c) | he has forwarded to the Company as soon as practical following the conclusion of that meeting signed dissent. |
A Director who votes in favour of an action is not entitled to record his dissent to it.
Written resolutions
| 18.11 | The Directors may pass a resolution in writing without holding a meeting if all Directors sign a document or sign several documents in the like form each signed by one or more of those Directors. |
| 18.12 | A written resolution signed by a validly appointed alternate Director need not also be signed by the appointing Director. |
| 18.13 | A written resolution signed personally by the appointing Director need not also be signed by his alternate. |
| 18.14 | A resolution in writing passed pursuant to Article 18.11, Article 18.12 and/or Article 18.13 shall be as effective as if it had been passed at a meeting of the Directors duly convened and held; and it shall be treated as having been passed on the day and at the time that the last Director signs (and for the avoidance of doubt, such day may or may not be a Business Day). |
Validity of acts of Directors in spite of formal defect
| 18.15 | All acts done by a meeting of the Board, or of a committee of the Board, or by any person acting as a Director or an alternate Director, shall, notwithstanding that it is afterwards discovered that there was some defect in the appointment of any Director or alternate Director or member of the committee, or that any of them were disqualified or had vacated office or were not entitled to vote, be as valid as if every such person had been duly appointed and qualified and had continued to be a Director or alternate Director and had been entitled to vote. |
| 39 |
| 19 | Permissible Directors’ interests and disclosure |
| 19.1 | A Director who is in any way, whether directly or indirectly, interested in a contract or transaction or proposed contract or transaction with the Company shall declare the nature of his interest at a meeting of the Directors. A general notice given to the Directors by any Director to the effect that he is a member of any specified company or firm and is to be regarded as interested in any contract or transaction which may thereafter be made with that company or firm shall be deemed a sufficient declaration of interest in regard to any contract so made or transaction so consummated. Subject to the Designated Stock Exchange Rules and disqualification by the chairman of the relevant Board meeting, a Director may vote in respect of any contract or transaction or proposed contract or transaction notwithstanding that he may be interested therein and if he does so his vote shall be counted and he may be counted in the quorum at any meeting of the Directors at which any such contract or transaction or proposed contract or transaction shall come before the meeting for consideration. |
| 19.2 | For the purposes of the preceding Article: |
| (a) | a general notice that a Director gives to the other Directors that he is to be regarded as having an interest of the nature and extent specified in the notice in any transaction or arrangement in which a specified person or class of persons is interested shall be deemed to be a disclosure that he has an interest in or duty in relation to any such transaction of the nature and extent so specified; and |
| (b) | an interest of which a Director has no knowledge and of which it is unreasonable to expect him to have knowledge shall not be treated as an interest of his. |
| 20 | Minutes |
| 20.1 | The Company shall cause minutes to be made in books of: |
| (a) | all appointments of Officers and committees made by the Board and of any such Officer’s remuneration; and |
| (b) | the names of Directors present at every meeting of the Directors, a committee of the Board, the Company or the holders of any class of shares or debentures, and all orders, resolutions and proceedings of such meetings. |
| 20.2 | Any such minutes, if purporting to be signed by the chairman of the meeting at which the proceedings were held or by the chairman of the next succeeding meeting or the Secretary, shall be prima facie evidence of the matters stated in them. |
| 21 | Accounts and audit |
| 21.1 | The Directors must ensure that proper accounting and other records are kept, and that accounts and associated reports are distributed in accordance with the requirements of the Act. |
| 40 |
| 21.2 | The books of account shall be kept at the registered office of the Company and shall always be open to inspection by the Directors. No Member (other than a Director) shall have any right of inspecting any account or book or document of the Company except as conferred by the Act or as authorised by the Directors or by Ordinary Resolution. |
| 21.3 | Unless the Directors otherwise prescribe, the financial year of the Company shall end on 31 March in each year and begin on 1 April in each year. |
Auditors
| 21.4 | The Directors may appoint an Auditor of the Company who shall hold office on such terms as the Directors determine. |
| 21.5 | At any general meeting convened and held at any time in accordance with these Articles, the Members may, by Ordinary Resolution, remove the Auditor before the expiration of his term of office. If they do so, the Members shall, by Ordinary Resolution, at that meeting appoint another Auditor in his stead for the remainder of his term. |
| 21.6 | The Auditors shall examine such books, accounts and vouchers; as may be necessary for the performance of their duties. |
| 21.7 | The Auditors shall, if so requested by the Directors, make a report on the accounts of the Company during their tenure of office at the next annual general meeting following their appointment, and at any time during their term of office, upon request of the Directors or any general meeting of the Company. |
| 22 | Record dates |
| 22.1 | Except to the extent of any conflicting rights attached to Shares, the resolution declaring a dividend on Shares of any class, whether it be an Ordinary Resolution of the Members or a Director’s resolution, may specify that the dividend is payable or distributable to the persons registered as the holders of those Shares at the close of business on a particular date, notwithstanding that the date may be a date prior to that on which the resolution is passed. |
| 22.2 | If the resolution does so specify, the dividend shall be payable or distributable to the persons registered as the holders of those Shares at the close of business on the specified date in accordance with their respective holdings so registered, but without prejudice to the rights inter se in respect of the dividend of transferors and transferees of any of those Shares. |
| 22.3 | The provisions of this Article apply, mutatis mutandis, to bonuses, capitalisation issues, distributions of realised capital profits or offers or grants made by the Company to the Members. |
| 41 |
| 23 | Dividends |
Source of dividends
| 23.1 | Dividends may be declared and paid out of any funds of the Company lawfully available for distribution. |
| 23.2 | Subject to the requirements of the Act regarding the application of a company’s Share premium account and with the sanction of an Ordinary Resolution, dividends may also be declared and paid out of any share premium account. |
Declaration of dividends by Members
| 23.3 | Subject to the provisions of the Act, the Company may by Ordinary Resolution declare dividends in accordance with the respective rights of the Members but no dividend shall exceed the amount recommended by the Directors. |
Payment of interim dividends and declaration of final dividends by Directors
| 23.4 | The Directors may declare and pay interim dividends or recommend final dividends in accordance with the respective rights of the Members if it appears to them that they are justified by the financial position of the Company and that such dividends may lawfully be paid. |
| 23.5 | Subject to the provisions of the Act, in relation to the distinction between interim dividends and final dividends, the following applies: |
| (a) | Upon determination to pay a dividend or dividends described as interim by the Directors in the dividend resolution, no debt shall be created by the declaration until such time as payment is made. |
| (b) | Upon declaration of a dividend or dividends described as final by the Directors in the dividend resolution, a debt shall be created immediately following the declaration, the due date to be the date the dividend is stated to be payable in the resolution. |
If the resolution fails to specify whether a dividend is final or interim, it shall be assumed to be interim.
| 23.6 | In relation to Shares carrying differing rights to dividends or rights to dividends at a fixed rate, the following applies: |
| (a) | If the share capital is divided into different classes, the Directors may pay dividends on Shares which confer deferred or non-preferred rights with regard to dividends as well as on Shares which confer preferential rights with regard to dividends but no dividend shall be paid on Shares carrying deferred or non-preferred rights if, at the time of payment, any preferential dividend is in arrears. |
| 42 |
| (b) | The Directors may also pay, at intervals settled by them, any dividend payable at a fixed rate if it appears to them that there are sufficient funds of the Company lawfully available for distribution to justify the payment. |
| (c) | If the Directors act in good faith, they shall not incur any liability to the Members holding Shares conferring preferred rights for any loss those Members may suffer by the lawful payment of the dividend on any Shares having deferred or non-preferred rights. |
Apportionment of dividends
| 23.7 | Except as otherwise provided by the rights attached to Shares all dividends shall be declared and paid according to the amounts Paid Up on the Shares on which the dividend is paid. All dividends shall be apportioned and paid proportionately to the amount Paid Up on the Shares during the time or part of the time in respect of which the dividend is paid. But if a Share is issued on terms providing that it shall rank for dividend as from a particular date, that Share shall rank for dividend accordingly. |
Right of set off
| 23.8 | The Directors may deduct from a dividend or any other amount payable to a person in respect of a Share any amount due by that person to the Company on a call or otherwise in relation to a Share. |
Power to pay other than in cash
| 23.9 | If the Directors so determine, any resolution declaring a dividend may direct that it shall be satisfied wholly or partly by the distribution of assets. If a difficulty arises in relation to the distribution, the Directors may settle that difficulty in any way they consider appropriate. For example, they may do any one or more of the following: |
| (a) | issue fractional Shares; |
| (b) | fix the value of assets for distribution and make cash payments to some Members on the footing of the value so fixed in order to adjust the rights of Members; and |
| (c) | vest some assets in trustees. |
How payments may be made
| 23.10 | A dividend or other monies payable on or in respect of a Share may be paid in any of the following ways: |
| (a) | if the Member holding that Share or other person entitled to that Share nominates a bank account for that purpose - by wire transfer to that bank account; or |
| (b) | by cheque or warrant sent by post to the registered address of the Member holding that Share or other person entitled to that Share. |
| 43 |
| 23.11 | For the purposes of Article 23.10(a), the nomination may be in writing or in an Electronic Record and the bank account nominated may be the bank account of another person. For the purposes of Article 23.10(b), subject to any applicable law or regulation, the cheque or warrant shall be made to the order of the Member holding that Share or other person entitled to the Share or to his nominee, whether nominated in writing or in an Electronic Record, and payment of the cheque or warrant shall be a good discharge to the Company. |
| 23.12 | If two or more persons are registered as the holders of the Share or are jointly entitled to it by reason of the death or bankruptcy of the registered holder (Joint Holders), a dividend (or other amount) payable on or in respect of that Share may be paid as follows: |
| (a) | to the registered address of the Joint Holder of the Share who is named first on the register of Members or to the registered address of the deceased or bankrupt holder, as the case may be; or |
| (b) | to the address or bank account of another person nominated by the Joint Holders, whether that nomination is in writing or in an Electronic Record. |
| 23.13 | Any Joint Holder of a Share may give a valid receipt for a dividend (or other amount) payable in respect of that Share. |
Dividends or other monies not to bear interest in absence of special rights
| 23.14 | Unless provided for by the rights attached to a Share, no dividend or other monies payable by the Company in respect of a Share shall bear interest. |
Dividends unable to be paid or unclaimed
| 23.15 | If a dividend cannot be paid to a Member or remains unclaimed within six weeks after it was declared or both, the Directors may pay it into a separate account in the Company’s name. If a dividend is paid into a separate account, the Company shall not be constituted trustee in respect of that account and the dividend shall remain a debt due to the Member. |
| 23.16 | A dividend that remains unclaimed for a period of six years after it became due for payment shall be forfeited to, and shall cease to remain owing by, the Company. |
| 24 | Capitalisation of profits |
Capitalisation of profits or of any share premium account or capital redemption reserve;
| 24.1 | The Directors may resolve to capitalise: |
| (a) | any part of the Company’s profits not required for paying any preferential dividend (whether or not those profits are available for distribution); or |
| (b) | any sum standing to the credit of the Company’s share premium account or capital redemption reserve, if any. |
| 44 |
| 24.2 | The amount resolved to be capitalised must be appropriated to the Members who would have been entitled to it had it been distributed by way of dividend and in the same proportions. The benefit to each Member so entitled must be given in either or both of the following ways: |
| (a) | by paying up the amounts unpaid on that Member’s Shares; |
| (b) | by issuing Fully Paid Up Shares, debentures or other securities of the Company to that Member or as that Member directs. The Directors may resolve that any Shares issued to the Member in respect of Partly Paid Up Shares (Original Shares) rank for dividend only to the extent that the Original Shares rank for dividend while those Original Shares remain Partly Paid Up. |
Applying an amount for the benefit of Members
| 24.3 | The amount capitalised must be applied to the benefit of Members in the proportions to which the Members would have been entitled to dividends if the amount capitalised had been distributed as a dividend. |
| 24.4 | Subject to the Act, if a fraction of a Share, a debenture or other security is allocated to a Member, the Directors may issue a fractional certificate to that Member or pay him the cash equivalent of the fraction. |
| 25 | Share Premium Account |
Directors to maintain share premium account
| 25.1 | The Directors shall establish a share premium account in accordance with the Act. They shall carry to the credit of that account from time to time an amount equal to the amount or value of the premium paid on the issue of any Share or capital contributed or such other amounts required by the Act. |
Debits to share premium account
| 25.2 | The following amounts shall be debited to any share premium account: |
| (a) | on the redemption or purchase of a Share, the difference between the nominal value of that Share and the redemption or purchase price; and |
| (b) | any other amount paid out of a share premium account as permitted by the Act. |
| 25.3 | Notwithstanding the preceding Article, on the redemption or purchase of a Share, the Directors may pay the difference between the nominal value of that Share and the redemption purchase price out of the profits of the Company or, as permitted by the Act, out of capital. |
| 45 |
| 26 | Seal |
Company seal
| 26.1 | The Company may have a seal if the Directors so determine. |
Duplicate seal
| 26.2 | Subject to the provisions of the Act, the Company may also have a duplicate seal or seals for use in any place or places outside the Cayman Islands. Each duplicate seal shall be a facsimile of the original seal of the Company. However, if the Directors so determine, a duplicate seal shall have added on its face the name of the place where it is to be used. |
When and how seal is to be used
| 26.3 | A seal may only be used by the authority of the Directors. Unless the Directors otherwise determine, a document to which a seal is affixed must be signed in one of the following ways: |
| (a) | by a Director (or his alternate) and the Secretary; or |
| (b) | by a single Director (or his alternate). |
If no seal is adopted or used
| 26.4 | If the Directors do not adopt a seal, or a seal is not used, a document may be executed in the following manner: |
| (a) | by a Director (or his alternate) and the Secretary; or |
| (b) | by a single Director (or his alternate); or |
| (c) | in any other manner permitted by the Act. |
Power to allow non-manual signatures and facsimile printing of seal
| 26.5 | The Directors may determine that either or both of the following applies: |
| (a) | that the seal or a duplicate seal need not be affixed manually but may be affixed by some other method or system of reproduction; |
| (b) | that a signature required by these Articles need not be manual but may be a mechanical or Electronic Signature. |
Validity of execution
| 26.6 | If a document is duly executed and delivered by or on behalf of the Company, it shall not be regarded as invalid merely because, at the date of the delivery, the Secretary, or the Director, or other Officer or person who signed the document or affixed the seal for and on behalf of the Company ceased to be the Secretary or hold that office and authority on behalf of the Company. |
| 46 |
| 27 | Indemnity |
| 27.1 | To the extent permitted by law, the Company shall indemnify each existing or former Director (including alternate Director), Secretary and other Officer of the Company (including an investment adviser or an administrator or liquidator) and their personal representatives against: |
| (a) | all actions, proceedings, costs, charges, expenses, losses, damages or liabilities incurred or sustained by the existing or former Director (including alternate Director), Secretary or Officer in or about the conduct of the Company’s business or affairs or in the execution or discharge of the existing or former Director’s (including alternate Director’s), Secretary’s or Officer’s duties, powers, authorities or discretions; and |
| (b) | without limitation to paragraph (a), all costs, expenses, losses or liabilities incurred by the existing or former Director (including alternate Director), Secretary or Officer in defending (whether successfully or otherwise) any civil, criminal, administrative or investigative proceedings (whether threatened, pending or completed) concerning the Company or its affairs in any court or tribunal, whether in the Cayman Islands or elsewhere. |
No such existing or former Director (including alternate Director), Secretary or Officer, however, shall be indemnified in respect of any matter arising out of his own dishonesty, wilful default, wilful neglect or fraud.
| 27.2 | To the extent permitted by law, the Company may make a payment, or agree to make a payment, whether by way of advance, loan or otherwise, for any legal costs incurred by an existing or former Director (including alternate Director), Secretary or Officer of the Company in respect of any matter identified in Article 27.1 on condition that the Director (including alternate Director), Secretary or Officer must repay the amount paid by the Company to the extent that it is ultimately found not liable to indemnify the Director (including alternate Director), Secretary or that Officer for those legal costs. |
Release
| 27.3 | To the extent permitted by law, the Company may by Special Resolution release any existing or former Director (including alternate Director), Secretary or other Officer of the Company from liability for any loss or damage or right to compensation which may arise out of or in connection with the execution or discharge of the duties, powers, authorities or discretions of his office; but there may be no release from liability arising out of or in connection with that person’s own dishonesty, wilful default, wilful neglect or fraud. |
| 47 |
Insurance
| 27.4 | To the extent permitted by law, the Company may pay, or agree to pay, a premium in respect of a contract insuring each of the following persons against risks determined by the Directors, other than liability arising out of that person’s own dishonesty, wilful default, wilful neglect or fraud: |
| (a) | an existing or former Director (including alternate Director), Secretary or Officer or auditor of: |
| (i) | the Company; |
| (ii) | a company which is or was a subsidiary of the Company; |
| (iii) | a company in which the Company has or had an interest (whether direct or indirect); and |
| (b) | a trustee of an employee or retirement benefits scheme or other trust in which any of the persons referred to in paragraph (a) is or was interested. |
| 28 | Notices |
Form of notices
| 28.1 | Save where these Articles provide otherwise, and subject to the Designated Stock Exchange Rules, any notice to be given to or by any person pursuant to these Articles shall be: |
| (a) | in writing signed by or on behalf of the giver in the manner set out below for written notices; or |
| (b) | subject to the next Article, in an Electronic Record signed by or on behalf of the giver by Electronic Signature and authenticated in accordance with Articles about authentication of Electronic Records; or |
| (c) | where these Articles expressly permit, by the Company by means of a website. |
Electronic communications
| 28.2 | A notice may only be given to the Company in an Electronic Record if: |
| (a) | the Directors so resolve or otherwise accept the notice; or |
| (b) | any Director or Officer provides the giver of the notice an electronic address to which the notice may be sent and a notice is sent to that address within a reasonable period of time. |
| 48 |
| 28.3 | A notice may not be given by Electronic Record to a person other than the Company unless the recipient has provided the giver of the notice with an Electronic address to which notice may be sent. |
| 28.4 | Subject to the Act, the Designated Stock Exchange Rules and to any other rules which the Company is bound to follow, the Company may also send any notice or other document pursuant to these Articles to a Member by publishing that notice or other document on a website where: |
| (a) | the Company and the Member have agreed to his having access to the notice or document on a website (instead of it being sent to him); |
| (b) | the notice or document is one to which that agreement applies; |
| (c) | the Member is notified (in accordance with any requirements laid down by the Act and, in a manner for the time being agreed between him and the Company for the purpose) of: |
| (i) | the publication of the notice or document on a website; |
| (ii) | the address of that website; and |
| (iii) | the place on that website where the notice or document may be accessed, and how it may be accessed; and |
| (d) | the notice or document is published on that website throughout the publication period, provided that, if the notice or document is published on that website for a part, but not all of, the publication period, the notice or document shall be treated as being published throughout that period if the failure to publish that notice of document throughout that period is wholly attributable to circumstances which it would not be reasonable to have expected the Company to prevent or avoid. For the purposes of this Article 28.4 “publication period” means a period of not less than twenty-one days, beginning on the day on which the notification referred to in Article 28.4(c) is deemed sent. |
Persons entitled to notices
| 28.5 | Any notice or other document to be given to a Member may be given by reference to the register of Members as it stands at any time within the period of twenty-one days before the day that the notice is given or (where and as applicable) within any other period permitted by, or in accordance with the requirements of, (to the extent applicable) the Designated Stock Exchange Rules and/or the Designated Stock Exchanges. No change in the register of Members after that time shall invalidate the giving of such notice or document or require the Company to give such item to any other person. |
| 49 |
Persons authorised to give notices
| 28.6 | A notice by either the Company or a Member pursuant to these Articles may be given on behalf of the Company or a Member by a Director or company secretary of the Company or a Member. |
Delivery of written notices
| 28.7 | Save where these Articles provide otherwise, a notice in writing may be given personally to the recipient, or left at (as appropriate) the Member’s or Director’s registered address or the Company’s registered office, or posted to that registered address or registered office. |
Joint holders
| 28.8 | Where Members are joint holders of a Share, all notices shall be given to the Member whose name first appears in the register of Members. |
Signatures
| 28.9 | A written notice shall be signed when it is autographed by or on behalf of the giver, or is marked in such a way as to indicate its execution or adoption by the giver. |
| 28.10 | An Electronic Record may be signed by an Electronic Signature. |
Evidence of transmission
| 28.11 | A notice given by Electronic Record shall be deemed sent if an Electronic Record is kept demonstrating the time, date and content of the transmission, and if no notification of failure to transmit is received by the giver. |
| 28.12 | A notice given in writing shall be deemed sent if the giver can provide proof that the envelope containing the notice was properly addressed, pre-paid and posted, or that the written notice was otherwise properly transmitted to the recipient. |
| 28.13 | A Member present, either in person or by proxy, at any meeting of the Company or of the holders of any class of Shares shall be deemed to have received due notice of the meeting and, where requisite, of the purposes for which it was called. |
Giving notice to a deceased or bankrupt Member
| 28.14 | A notice may be given by the Company to the persons entitled to a Share in consequence of the death or bankruptcy of a Member by sending or delivering it, in any manner authorised by these Articles for the giving of notice to a Member, addressed to them by name, or by the title of representatives of the deceased, or trustee of the bankrupt or by any like description, at the address, if any, supplied for that purpose by the persons claiming to be so entitled. |
| 28.15 | Until such an address has been supplied, a notice may be given in any manner in which it might have been given if the death or bankruptcy had not occurred. |
| 50 |
Date of giving notices
| 28.16 | A notice is given on the date identified in the following table |
| Method for giving notices | When taken to be given | |
| (A) Personally | At the time and date of delivery | |
| (B) By leaving it at the Member’s registered address | At the time and date it was left | |
| (C) By posting it by prepaid post to the street or postal address of that recipient | 48 hours after the date it was posted | |
| (D) By Electronic Record (other than publication on a website), to recipient’s Electronic address | 48 hours after the date it was sent | |
| (E) By publication on a website | 24 hours after the date on which the Member is deemed to have been notified of the publication of the notice or document on the website |
Saving provision
| 28.17 | None of the preceding notice provisions shall derogate from the Articles about the delivery of written resolutions of Directors and written resolutions of Members. |
| 29 | Authentication of Electronic Records |
Application of Articles
| 29.1 | Without limitation to any other provision of these Articles, any notice, written resolution or other document under these Articles that is sent by Electronic means by a Member, or by the Secretary, or by a Director or other Officer of the Company, shall be deemed to be authentic if either Article 29.2 or Article 29.4 applies. |
Authentication of documents sent by Members by Electronic means
| 29.2 | An Electronic Record of a notice, written resolution or other document sent by Electronic means by or on behalf of one or more Members shall be deemed to be authentic if the following conditions are satisfied: |
| (a) | the Member or each Member, as the case may be, signed the original document, and for this purpose Original Document includes several documents in like form signed by one or more of those Members; and |
| (b) | the Electronic Record of the Original Document was sent by Electronic means by, or at the direction of, that Member to an address specified in accordance with these Articles for the purpose for which it was sent; and |
| 51 |
| (c) | Article 29.7 does not apply. |
| 29.3 | For example, where a sole Member signs a resolution and sends the Electronic Record of the original resolution, or causes it to be sent, by facsimile transmission to the address in these Articles specified for that purpose, the facsimile copy shall be deemed to be the written resolution of that Member unless Article 29.7 applies. |
Authentication of document sent by the Secretary or Officers of the Company by Electronic means
| 29.4 | An Electronic Record of a notice, written resolution or other document sent by or on behalf of the Secretary or an Officer or Officers of the Company shall be deemed to be authentic if the following conditions are satisfied: |
| (a) | the Secretary or the Officer or each Officer, as the case may be, signed the original document, and for this purpose Original Document includes several documents in like form signed by the Secretary or one or more of those Officers; and |
| (b) | the Electronic Record of the Original Document was sent by Electronic means by, or at the direction of, the Secretary or that Officer to an address specified in accordance with these Articles for the purpose for which it was sent; and |
| (c) | Article 29.7 does not apply. |
This Article 29.4 applies whether the document is sent by or on behalf of the Secretary or Officer in his own right or as a representative of the Company.
| 29.5 | For example, where a sole Director signs a resolution and scans the resolution, or causes it to be scanned, as a PDF version which is attached to an email sent to the address in these Articles specified for that purpose, the PDF version shall be deemed to be the written resolution of that Director unless Article 29.7 applies. |
Manner of signing
| 29.6 | For the purposes of these Articles about the authentication of Electronic Records, a document will be taken to be signed if it is signed manually or in any other manner permitted by these Articles. |
Saving provision
| 29.7 | A notice, written resolution or other document under these Articles will not be deemed to be authentic if the recipient, acting reasonably: |
| (a) | believes that the signature of the signatory has been altered after the signatory had signed the original document; or |
| 52 |
| (b) | believes that the original document, or the Electronic Record of it, was altered, without the approval of the signatory, after the signatory signed the original document; or |
| (c) | otherwise doubts the authenticity of the Electronic Record of the document |
and the recipient promptly gives notice to the sender setting the grounds of its objection. If the recipient invokes this Article, the sender may seek to establish the authenticity of the Electronic Record in any way the sender thinks fit.
| 30 | Transfer by way of continuation |
| 30.1 | The Company may, by Special Resolution, resolve to be registered by way of continuation in a jurisdiction outside: |
| (a) | the Cayman Islands; or |
| (b) | such other jurisdiction in which it is, for the time being, incorporated, registered or existing. |
| 30.2 | To give effect to any resolution made pursuant to the preceding Article, the Directors may cause the following: |
| (a) | an application be made to the Registrar of Companies of the Cayman Islands to deregister the Company in the Cayman Islands or in the other jurisdiction in which it is for the time being incorporated, registered or existing; and |
| (b) | all such further steps as they consider appropriate to be taken to effect the transfer by way of continuation of the Company. |
| 31 | Winding up |
Distribution of assets in specie
| 31.1 | If the Company is wound up the Members may, subject to these Articles and any other sanction required by the Act, pass a Special Resolution allowing the liquidator to do either or both of the following: |
| (a) | to divide in specie among the Members the whole or any part of the assets of the Company and, for that purpose, to value any assets and to determine how the division shall be carried out as between the Members or different classes of Members; and/or |
| (b) | to vest the whole or any part of the assets in trustees for the benefit of Members and those liable to contribute to the winding up. |
No obligation to accept liability
| 31.2 | No Member shall be compelled to accept any assets if an obligation attaches to them. |
| 53 |
| 31.3 | The Directors are authorised to present a winding up petition |
| 31.4 | The Directors have the authority to present a petition for the winding up of the Company to the Grand Court of the Cayman Islands on behalf of the Company without the sanction of a resolution passed at a general meeting. |
| 32 | Amendment of Memorandum and Articles |
Power to change name or amend Memorandum
| 32.1 | Subject to the Act, the Company may, by Special Resolution: |
| (a) | change its name; or |
| (b) | change the provisions of its Memorandum with respect to its objects, powers or any other matter specified in the Memorandum. |
Power to amend these Articles
| 32.2 | Subject to the Act and as provided in these Articles, the Company may, by Special Resolution, amend these Articles in whole or in part. |
| 33 | Mergers and Consolidations |
| 33.1 | The Company shall have the power to merge or consolidate with one or more other constituent companies (as defined in the Companies Act) upon such terms as the Directors may determine and (to the extent required by the Companies Act) with the approval of a Special Resolution. |
| 54 |
ANNEX C
FORM OF GLOBALINK SUPPORT AGREEMENT
This PARENT STOCKHOLDER SUPPORT AGREEMENT, dated as of May ___, 2024 (this “Support Agreement”), is entered into by and among the stockholder named on the signature page hereto (the “Stockholder”), Alps Life Sciences Inc, a Cayman Islands exempted company (“Alps Holdco”) and Globalink Investment Inc., a Delaware corporation (“Parent”). Capitalized terms used but not defined in this Support Agreement shall have the meanings ascribed to them in the Merger Agreement (as defined below).
WHEREAS, Parent and Alps Holdco are parties to that certain Amended and Restated Merger Agreement, dated as of the date hereof (as amended, modified or supplemented from time to time, the “Merger Agreement”), which provides, among other things, that concurrently therewith or immediately after the Reincorporation Merger, Merger Sub will merge with and into Alps Holdco with Alps Holdco being the Surviving Company and a wholly-owned subsidiary of Pubco, in accordance with the Merger Agreement, the Plan of Merger and the Companies Act; , and as a result of which, among other matters, all of the issued ordinary shares of Alps Holdco as of the Effective Time shall be converted into the right to receive the Merger Consideration Shares as set forth in the Merger Agreement, all upon the terms and subject to the conditions set forth in the Merger Agreement and in accordance with the applicable provisions of Cayman laws and regulations;
WHEREAS, as of the date hereof, the Stockholder owns the number of shares of Parent’s common stock, par value $0.001 (“Parent Common Stock”), as set forth underneath the Stockholder’s name on the signature page hereto (all such shares, or any successor or additional shares of Parent of which ownership of record or the power to vote is hereafter acquired by the Stockholder prior to the termination of this Support Agreement being referred to herein as the “Stockholder Shares”);
WHEREAS, the Board of Directors of the Parent has (a) approved and declared advisable the Merger Agreement, the Additional Agreements, the Reincorporation Merger, the Merger and the other transactions contemplated by any such documents (collectively, the “Transactions”), (b) determined that the Transactions are fair to and in the best interests of the Parent and its stockholders (the “Parent Stockholders”) and (c) recommended the approval and the adoption by each of the Parent Stockholders of the Merger Agreement, the Additional Agreements, the Reincorporation Merger, the Merger and the other Transactions; and
WHEREAS, in order to induce Alps Holdco to enter into the Merger Agreement, Stockholder is executing and delivering this Support Agreement to Alps Holdco.
NOW, THEREFORE, in consideration of the foregoing and of the mutual covenants and agreements contained herein, and intending to be legally bound hereby, the parties hereby agree as follows:
1. Voting Agreements. Stockholder, solely in its capacity as a stockholder of Parent, agrees that, during the term of this Support Agreement, at the Parent Stockholder Meeting, at any other meeting of the Parent Stockholders related to the Transactions (whether annual or special and whether or not an adjourned or postponed meeting, however called and including any adjournment or postponement thereof) and/or in connection with any written consent of the Parent Stockholders related to the Transactions (the Parent Stockholder Meeting and all other meetings or consents related to the Merger Agreement, collectively referred to herein as the “Meeting”), Stockholder shall:
(a) when the Meeting is held, appear at the Meeting or otherwise cause the Stockholder Shares to be counted as present thereat for the purpose of establishing a quorum;
| C-1 |
(b) vote (or execute and return an action by written consent), or cause to be voted at the Meeting (or validly execute and return and cause such consent to be granted with respect to), all of the Stockholder Shares in favor of the Merger Agreement and the Transactions and each of the Parent Proposals; and
(c) vote (or execute and return an action by written consent), or cause to be voted at the Meeting (or validly execute and return and cause such consent to be granted with respect to), all of the Stockholder Shares against any other action that would reasonably be expected to (x) materially impede, interfere with, delay, postpone or adversely affect the Mergers or any of the Transactions, (y) result in a breach of any covenant, representation or warranty or other obligation or agreement of Parent under the Merger Agreement or (z) result in a breach of any covenant, representation or warranty or other obligation or agreement of the Stockholder contained in this Support Agreement.
2. Restrictions on Transfer. The Stockholder agrees that, during the term of this Support Agreement, it shall not sell, assign or otherwise transfer any of the Stockholder Shares unless the buyer, assignee or transferee thereof executes a joinder agreement to this Support Agreement in a form reasonably acceptable to Alps Holdco and Parent. Parent shall not, and shall not permit Parent’s transfer agent to, register any sale, assignment or transfer of the Stockholder Shares on Parent’s stock ledger (book entry or otherwise) that is not in compliance with this Section 2.
3. No Redemption. Stockholder hereby agrees that, during the term of this Agreement, it shall not redeem, or submit a request to Parent’s transfer agent or otherwise exercise any right to redeem, any Stockholder Shares.
4. New Securities. During the term of this Support Agreement, in the event that, (a) any shares of Parent Common Stock or other equity securities of Parent are issued to the Stockholder after the date of this Support Agreement as a result of the Reincorporation Merger or pursuant to any stock dividend, stock split, recapitalization, reclassification, combination or exchange of Parent securities owned by the Stockholder, (b) the Stockholder purchases or otherwise acquires beneficial ownership of any shares of Parent Common Stock or other equity securities of Parent after the date of this Support Agreement, or (c) the Stockholder acquires the right to vote or share in the voting of any Parent Common Stock or other equity securities of Parent after the date of this Support Agreement (such Parent Common Stock or other equity securities of Parent, collectively the “New Securities”), then such New Securities acquired or purchased by the Stockholder shall be subject to the terms of this Support Agreement to the same extent as if they constituted the Stockholder Shares as of the date hereof.
5. No Challenge. Stockholder agrees not to commence, join in, facilitate, assist or encourage, and agrees to take all actions necessary to opt out of any class in any class action with respect to, any claim, derivative or otherwise, against Parent, Pubco, Merger Sub, Alps Holdco or any of their respective successors or directors (a) challenging the validity of, or seeking to enjoin the operation of, any provision of this Support Agreement or the Merger Agreement or (b) alleging a breach of any fiduciary duty of any Person in connection with the evaluation, negotiation or entry into the Merger Agreement.
6. Consent to Disclosure. Stockholder hereby consents to the publication and disclosure in the Form F-4 and the Proxy Statement (and, as and to the extent otherwise required by applicable securities Laws or the SEC or any other securities authorities, any other documents or communications provided by Parent or Alps Holdco to any Authority or to securityholders of Parent or the Company) of Stockholder’s identity and beneficial ownership of Stockholder Shares and the nature of Stockholder’s commitments, arrangements and understandings under and relating to this Support Agreement and, if deemed appropriate by Parent or Alps Holdco, a copy of this Support Agreement. Stockholder will promptly provide any information reasonably requested by Parent or Alps Holdco for any regulatory application or filing made or approval sought in connection with the Transactions (including filings with the SEC). Stockholder shall not issue any press release or otherwise make any public statements with respect to the Transactions or the transactions contemplated herein without the prior written approval of Alps Holdco and Parent.
| C-2 |
7. Stockholder Representations: Stockholder represents and warrants to Parent and Alps Holdco, as of the date hereof, that:
(a) Stockholder has never been suspended or expelled from membership in any securities or commodities exchange or association or had a securities or commodities license or registration denied, suspended or revoked;
(b) Stockholder has full right and power, without violating any agreement to which it is bound (including any non-competition or non-solicitation agreement with any employer or former employer), to enter into this Support Agreement;
(c) (i) if Stockholder is not an individual, Stockholder is duly organized, validly existing and in good standing under the Laws of the jurisdiction in which it is organized, and the execution, delivery and performance of this Support Agreement and the consummation of the transactions contemplated hereby are within the Stockholder’s organizational powers and have been duly authorized by all necessary organizational actions on the part of the Stockholder and (ii) if Stockholder is an individual, the signature on this Support Agreement is genuine, and Stockholder has legal competence and capacity to execute the same;
(d) this Support Agreement has been duly executed and delivered by Stockholder and, assuming due authorization, execution and delivery by the other parties to this Support Agreement, this Support Agreement constitutes a legally valid and binding obligation of Stockholder, enforceable against Stockholder in accordance with the terms hereof (except as enforceability may be limited by bankruptcy Laws, other similar Laws affecting creditors’ rights and general principles of equity affecting the availability of specific performance and other equitable remedies);
(e) the execution and delivery of this Support Agreement by Stockholder does not, and the performance by Stockholder of its obligations hereunder will not, (i) conflict with or result in a violation of the organizational documents of Stockholder, or (ii) require any consent or approval from any third party that has not been given or other action that has not been taken by any third party, in each case, to the extent such consent, approval or other action would prevent, enjoin or materially delay the performance by Stockholder of its obligations under this Support Agreement;
(f) there are no Actions pending against Stockholder or, to the knowledge of Stockholder, threatened against Stockholder, before (or, in the case of threatened Actions, that would be before) any Authority, which in any manner challenges or seeks to prevent, enjoin or materially delay the performance by Stockholder of Stockholder’s obligations under this Support Agreement;
(g) no broker, finder, investment banker or other Person is entitled to any brokerage fee, finders’ fee or other commission in connection with this Support Agreement or any of the respective transactions contemplated hereby, based upon arrangements made by or on behalf of the Stockholder;
| C-3 |
(h) Stockholder has had the opportunity to read the Merger Agreement and this Support Agreement and has had the opportunity to consult with Stockholder’s tax and legal advisors;
(i) Stockholder has not entered into, and shall not enter into, any agreement that would prevent Stockholder from performing any of Stockholder’s obligations hereunder;
(j) Stockholder has good title to the Stockholder Shares underneath Stockholder’s name on the signature page hereto, free and clear of any Liens other than Permitted Liens and Liens under Parent’s Organizational Documents, and Stockholder has the sole power to vote or cause to be voted the Stockholder Shares; and
(k) the Stockholder Shares set forth underneath Stockholder’s name on the signature page to this Support Agreement are the only shares of Parent’s outstanding capital stock owned of record or beneficially owned by the Stockholder as of the date hereof, and none of the Stockholder Shares are subject to any proxy, voting trust or other agreement or arrangement with respect to the voting of the Stockholder Shares that is inconsistent with Stockholder’s obligations pursuant to this Support Agreement.
8. Specific Performance. The Stockholder hereby agrees and acknowledges that (a) Parent and Alps Holdco would be irreparably injured in the event of a breach by the Stockholder of its obligations under this Support Agreement, (b) monetary damages may not be an adequate remedy for such breach and (c) Parent and Alps Holdco shall be entitled to obtain injunctive relief, in addition to any other remedy that such party may have in law or in equity, in the event of such breach or anticipated breach, without the requirement to post any bond or other security or to prove that money damages would be inadequate.
9. Entire Agreement; Amendment; Waiver. This Support Agreement and the other agreements referenced herein constitute the entire agreement and understanding of the parties hereto in respect of the subject matter hereof and supersede all prior understandings, agreements or representations by or among the parties hereto, written or oral, to the extent they relate in any way to the subject matter hereof or the transactions contemplated hereby provided, that, for the avoidance of doubt, the foregoing shall not affect the rights and obligations of the parties under the Merger Agreement or any Additional Agreement. This Support Agreement may not be changed, amended, modified or waived (other than to correct a typographical error) as to any particular provision, except by a written instrument executed by all parties hereto. No failure or delay by a party in exercising any right hereunder shall operate as a waiver thereof. No waivers of or exceptions to any term, condition, or provision of this Support Agreement, in any one or more instances, shall be deemed to be or construed as a further or continuing waiver of any such term, condition, or provision.
10. Binding Effect; Assignment; Third Parties. This Support Agreement and all of the provisions hereof shall be binding upon and inure to the benefit of the parties hereto and their respective permitted successors and assigns. This Support Agreement and all obligations of Stockholder are personal to Stockholder and may not be assigned, transferred or delegated by Stockholder at any time without the prior written consent of Parent and Alps Holdco, and any purported assignment, transfer or delegation without such consent shall be null and void ab initio. Nothing contained in this Support Agreement or in any instrument or document executed by any party in connection with the transactions contemplated hereby shall create any rights in, or be deemed to have been executed for the benefit of, any Person that is not a party hereto or thereto or a successor or permitted assign of such a party.
11. Counterparts. This Support Agreement may be executed in any number of original, electronic or facsimile counterparts and each of such counterparts shall for all purposes be deemed to be an original, and all such counterparts shall together constitute but one and the same instrument.
| C-4 |
12. Severability. This Support Agreement shall be deemed severable, and the invalidity or unenforceability of any term or provision hereof shall not affect the validity or enforceability of this Support Agreement or of any other term or provision hereof. Furthermore, in lieu of any such invalid or unenforceable term or provision, the parties hereto intend that there shall be added as a part of this Support Agreement a provision as similar in terms to such invalid or unenforceable provision as may be possible and be valid and enforceable.
13. Governing Law; Jury Trial Waiver. Sections 13.7, 13.15, 13.16 and 13.17 of the Merger Agreement are incorporated by reference herein to apply with full force to any disputes arising under this Support Agreement.
14. Notice. Any notice, consent or request to be given in connection with any of the terms or provisions of this Support Agreement shall be in writing and shall be sent or given in accordance with the terms of Section 13.1 of the Merger Agreement to the applicable party, with respect to Alps Holdco and Parent, at the respective addresses set forth in Section 13.1 of the Merger Agreement, and, with respect to the Stockholder, at the address set forth underneath Stockholder’s name on the signature page hereto.
15. Termination. This Support Agreement become effective upon the date hereof and shall automatically terminate, and none of Parent, Alps Holdco or Stockholder shall have any rights or obligations hereunder, on the earliest of (i) the mutual written consent of Parent, Alps Holdco and the Stockholder, (ii) the Closing (following the performance of the obligations of the parties hereunder required to be performed at or prior to the Closing), or (iii) the termination of the Merger Agreement in accordance with its terms. No such termination shall relieve the Stockholder, Parent or Alps Holdco from any liability resulting from a breach of this Support Agreement occurring prior to such termination. Notwithstanding anything to the contrary herein, the provisions of this Section 15 shall survive the termination of this Support Agreement.
16. Adjustment for Stock Split. If, and as often as, there are any changes in the Stockholder Shares by way of stock split, stock dividend, combination or reclassification, or through merger, consolidation, reorganization, recapitalization or business combination, or by any other means, equitable adjustment shall be made to the provisions of this Support Agreement as may be required so that the rights, privileges, duties and obligations hereunder shall continue with respect to the Stockholder, Parent, Alps Holdco, the Stockholder Shares as so changed.
17. Further Actions. Each of the parties hereto agrees to execute and deliver hereafter any further document, agreement or instrument of assignment, transfer or conveyance as may be necessary or desirable to effectuate the purposes hereof and as may be reasonably requested in writing by another party hereto.
18. Expenses. Each party shall be responsible for its own fees and expenses (including the fees and expenses of investment bankers, accountants and counsel) in connection with the entering into of this Support Agreement, the performance of its obligations hereunder and the consummation of the transactions contemplated hereby; provided, that in the event of any Action arising out of or relating to this Support Agreement, the non-prevailing party in any such Action will pay its own expenses and the reasonable documented out-of-pocket expenses, including reasonable attorneys’ fees and costs, reasonably incurred by the prevailing party.
| C-5 |
19. Interpretation. The titles and subtitles used in this Support Agreement are for convenience only and are not to be considered in construing or interpreting this Support Agreement. In this Support Agreement, unless the context otherwise requires: (i) any pronoun used shall include the corresponding masculine, feminine or neuter forms, and the singular form of nouns, pronouns and verbs shall include the plural and vice versa; (ii) the term “including” (and with correlative meaning “include”) shall be deemed in each case to be followed by the words “without limitation”; and (iii) the words “herein,” “hereto,” and “hereby” and other words of similar import shall be deemed in each case to refer to this Support Agreement as a whole and not to any particular section or other subdivision of this Support Agreement. The parties have participated jointly in the negotiation and drafting of this Support Agreement. Consequently, in the event an ambiguity or question of intent or interpretation arises, this Support Agreement shall be construed as if drafted jointly by the parties hereto, and no presumption or burden of proof shall arise favoring or disfavoring any party by virtue of the authorship of any provision of this Support Agreement.
20. No Partnership, Agency or Joint Venture. This Support Agreement is intended to create a contractual relationship among Stockholder, Alps Holdco and Parent, and is not intended to create, and does not create, any agency, partnership, joint venture or any like relationship among the parties hereto or among any other Parent Stockholders entering into support agreements with Alps Holdco or Parent. Stockholder has acted independently regarding its decision to enter into this Support Agreement. Nothing contained in this Support Agreement shall be deemed to vest in Alps Holdco or Parent any direct or indirect ownership or incidence of ownership of or with respect to any Stockholder Shares.
21. Capacity as Stockholder. Stockholder signs this Support Agreement solely in Stockholder’s capacity as a stockholder of Parent, and not in any other capacity, including, if applicable, as a director, officer or employee of Parent or any of its Subsidiaries. Nothing herein shall be construed to limit or affect any actions or inactions by Stockholder or any representative of Stockholder, as applicable, serving as a director of Parent or any Subsidiary of Parent, acting in such Person’s capacity as a director of Parent or any Subsidiary of Parent.
[Signature Pages Follow]
| C-6 |
IN WITNESS WHEREOF, the parties have executed this Parent Stockholder Support Agreement as of the date first written above.
| Alps Holdco: | ||
| Alps Life Sciences Inc | ||
| By: | ||
| Name: | ||
| Title: | ||
| Parent: | ||
| Globalink Investment Inc. | ||
| By: | ||
| Name: | ||
| Title: | ||
[Signature Page to Parent Stockholder Support Agreement]
| C-7 |
| Stockholder: | ||
| By: | ||
| Name: | ||
| Title: | ||
Number of Shares:
Shares of Parent Common Stock: __________________________________________
Address for Notice:
Address:
Facsimile No.:
Telephone No.:
Email:
[Signature Page to Parent Stockholder Support Agreement]
| C-8 |
ANNEX D
FORM OF ALPS HOLDCO SUPPORT AGREEMENT
This COMPANY SHAREHOLDERS SUPPORT AGREEMENT, dated as of May 20, 2024 (this “Support Agreement”), is entered into by and among the Shareholders named on the signature page hereto (each, a “Shareholder”), Alps Life Sciences Inc, a Cayman Islands exempted company (the “Company”), and Globalink Investment Inc., a Delaware corporation (“Parent”). Capitalized terms used but not defined in this Support Agreement shall have the meanings ascribed to them in the Merger Agreement (as defined below).
WHEREAS, Parent and the Company are parties to that certain Amended and Restated Merger Agreement, dated as of the date hereof (as amended, modified or supplemented from time to time, the “Merger Agreement”), which provides, among other things, that concurrently therewith or immediately after the Reincorporation Merger, Merger Sub will merge with and into the Company with the Company being the Surviving Company and a wholly-owned subsidiary of Pubco, in accordance with the Merger Agreement, the Plan of Merger and the Companies Act, and as a result of which, among other matters, all of the issued ordinary shares of the Company as of the Effective Time shall be converted into the right to receive the Merger Consideration Shares as set forth in the Merger Agreement, all upon the terms and subject to the conditions set forth in the Merger Agreement and in accordance with the applicable provisions of Cayman laws and regulations;
WHEREAS, as of the date hereof, each Shareholder owns such number of the Company’s ordinary shares (“Company Ordinary Shares”), as set forth underneath such Shareholder’s name on the signature page hereto (all such shares, or any successor or additional shares of the Company of which ownership of record or the power to vote is hereafter acquired by the such Shareholder prior to the termination of this Support Agreement being referred to herein as the “Shareholder Shares”);
WHEREAS, the Board of Directors of the Company has (a) approved and declared advisable the Merger Agreement, the Additional Agreements, the Mergers and the other transactions contemplated by any such documents (collectively, the “Transactions”), (b) determined that the Transactions are fair to and in the best interests of the Company and its Shareholders (the “Company Shareholders”) and (c) recommended the approval and the adoption by each of the Company Shareholders of the Merger Agreement, the Additional Agreements, the Mergers and the other Transactions; and
WHEREAS, in order to induce Parent to enter into the Merger Agreement, Shareholder is executing and delivering this Support Agreement to Parent.
NOW, THEREFORE, in consideration of the foregoing and of the mutual covenants and agreements contained herein, and intending to be legally bound hereby, the parties hereby agree as follows:
1. Voting Agreements. Each Shareholder, solely in its capacity as a shareholder of the Company, agrees that, during the term of this Support Agreement, at any meeting of the Company Shareholders related to the Transactions (whether annual or special and whether or not an adjourned or postponed meeting, however called and including any adjournment or postponement thereof), and/or in connection with any written consent of the Company Shareholders related to the Transactions (all meetings or consents related to the Merger Agreement, collectively referred to herein as the “Meeting”), such Shareholder shall:
(a) when the Meeting is held, appear at the Meeting or otherwise cause the Shareholder Shares to be counted as present thereat for the purpose of establishing a quorum;
(b) vote (or execute and return an action by written consent), or cause to be voted at the Meeting (or validly execute and return and cause such consent to be granted with respect to), all of the Shareholder Shares in favor of the Merger Agreement and the Transactions; and
(c) vote (or execute and return an action by written consent), or cause to be voted at the Meeting (or validly execute and return and cause such consent to be granted with respect to), all of the Shareholder Shares against any other action that would reasonably be expected to (x) materially impede, interfere with, delay, postpone or adversely affect the Mergers or any of the Transactions, or (y) result in a breach of any covenant, representation or warranty or other obligation or agreement of the Shareholder contained in this Support Agreement.
| D-1 |
2. Restrictions on Transfer. Each Shareholder agrees that, during the term of this Support Agreement, it shall not sell, assign or otherwise transfer any of the Shareholder Shares unless the buyer, assignee or transferee thereof executes a joinder agreement to this Support Agreement in a form reasonably acceptable to Parent and the Company. the Company shall not register any sale, assignment or transfer of the Shareholder Shares on the Company’s stock ledger (book entry or otherwise) that is not in compliance with this Section 2.
3. New Securities. During the term of this Support Agreement, in the event that, (a) any shares of the Company’s share capital or other equity securities of the Company are issued to a Shareholder after the date of this Support Agreement pursuant to any stock dividend, stock split, recapitalization, reclassification, combination or exchange of the Company securities owned by such Shareholder, (b) a Shareholder purchases or otherwise acquires beneficial ownership of any shares of the Company’s share capital or other equity securities of the Company after the date of this Support Agreement, or (c) a Shareholder acquires the right to vote or share in the voting of any the Company’s share capital or other equity securities of the Company after the date of this Support Agreement (such the Company’s share capital or other equity securities of the Company, collectively the “New Securities”), then such New Securities acquired or purchased by such Shareholder shall be subject to the terms of this Support Agreement to the same extent as if they constituted the Shareholder Shares as of the date hereof.
4. No Challenge. Each Shareholder agrees not to commence, join in, facilitate, assist or encourage, and agrees to take all actions necessary to opt out of any class in any class action with respect to, any claim, derivative or otherwise, against Parent, Pubco, Merger Sub, the Company or any of their respective successors or directors (a) challenging the validity of, or seeking to enjoin the operation of, any provision of this Support Agreement or the Merger Agreement or (b) alleging a breach of any fiduciary duty of any Person in connection with the evaluation, negotiation or entry into the Merger Agreement.
5. Waiver. Each Shareholder hereby irrevocably and unconditionally waives any rights of appraisal, dissenter’s rights and any similar rights relating to the Merger Agreement and the consummation by the parties of the Transactions, including the Mergers, that Shareholder may have under applicable law.
6. Consent to Disclosure. Each Shareholder hereby consents to the publication and disclosure in the Form F-4 and the Proxy Statement (and, as and to the extent otherwise required by applicable securities Laws or the SEC or any other securities authorities, any other documents or communications provided by Parent, Pubco or The Company to any Authority or to securityholders of Parent, Pubco or the Company) of Shareholder’s identity and beneficial ownership of such Shareholder Shares and the nature of Shareholder’s commitments, arrangements and understandings under and relating to this Support Agreement and, if deemed appropriate by Parent, Pubco or the Company, a copy of this Support Agreement. Each Shareholder will promptly provide any information reasonably requested by Parent, Pubco or the Company for any regulatory application or filing made or approval sought in connection with the Transactions (including filings with the SEC). Each Shareholder shall not issue any press release or otherwise make any public statements with respect to the Transactions or the transactions contemplated herein without the prior written approval of the Company, Pubco and Parent.
7. Shareholder Representations: Each Shareholder represents and warrants to Parent and the Company, as of the date hereof, that:
(a) such Shareholder has never been suspended or expelled from membership in any securities or commodities exchange or association or had a securities or commodities license or registration denied, suspended or revoked;
(b) such Shareholder has full right and power, without violating any agreement to which it is bound (including any non-competition or non-solicitation agreement with any employer or former employer), to enter into this Support Agreement;
(c) (i) if such Shareholder is not an individual, such Shareholder is duly organized, validly existing and in good standing under the Laws of the jurisdiction in which it is organized, and the execution, delivery and performance of this Support Agreement and the consummation of the transactions contemplated hereby are within the Shareholder’s organizational powers and have been duly authorized by all necessary organizational actions on the part of the Shareholder and (ii) if such Shareholder is an individual, the signature on this Support Agreement is genuine, and such Shareholder has legal competence and capacity to execute the same;
| D-2 |
(d) this Support Agreement has been duly executed and delivered by such Shareholder and, assuming due authorization, execution and delivery by the other parties to this Support Agreement, this Support Agreement constitutes a legally valid and binding obligation of Shareholder, enforceable against Shareholder in accordance with the terms hereof (except as enforceability may be limited by bankruptcy Laws, other similar Laws affecting creditors’ rights and general principles of equity affecting the availability of specific performance and other equitable remedies);
(e) the execution and delivery of this Support Agreement by such Shareholder does not, and the performance by the Shareholder of its obligations hereunder will not, (i) conflict with or result in a violation of the organizational documents of such Shareholder, or (ii) require any consent or approval from any third party that has not been given or other action that has not been taken by any third party, in each case, to the extent such consent, approval or other action would prevent, enjoin or materially delay the performance by the Shareholder of its obligations under this Support Agreement;
(f) there are no Actions pending against such Shareholder or, to the knowledge of the Shareholder, threatened against the Shareholder, before (or, in the case of threatened Actions, that would be before) any Authority, which in any manner challenges or seeks to prevent, enjoin or materially delay the performance by the Shareholder of Shareholder’s obligations under this Support Agreement;
(g) no broker, finder, investment banker or other Person is entitled to any brokerage fee, finders’ fee or other commission in connection with this Support Agreement or any of the respective transactions contemplated hereby, based upon arrangements made by or on behalf of such Shareholder;
(h) such Shareholder has had the opportunity to read the Merger Agreement and this Support Agreement and has had the opportunity to consult with such Shareholder’s tax and legal advisors;
(i) such Shareholder has not entered into, and shall not enter into, any agreement that would prevent the Shareholder from performing any of Shareholder’s obligations hereunder;
(j) such Shareholder has good title to the Shareholder Shares underneath Shareholder’s name on the signature page hereto, free and clear of any Liens other than Permitted Liens and Liens under the Company’s Organizational Documents, and Shareholder has the sole power to vote or cause to be voted the Shareholder Shares; and
(k) the Shareholder Shares set forth underneath such Shareholder’s name on the signature page to this Support Agreement are the only shares of the Company’s outstanding share capital owned of record or beneficially owned by the Shareholder as of the date hereof, and none of the Shareholder Shares are subject to any proxy, voting trust or other agreement or arrangement with respect to the voting of the Shareholder Shares that is inconsistent with Shareholder’s obligations pursuant to this Support Agreement.
8. Specific Performance. Each Shareholder hereby agrees and acknowledges that (a) Parent and the Company would be irreparably injured in the event of a breach by the Shareholder of its obligations under this Support Agreement, (b) monetary damages may not be an adequate remedy for such breach and (c) Parent and the Company shall be entitled to obtain injunctive relief, in addition to any other remedy that such party may have in law or in equity, in the event of such breach or anticipated breach, without the requirement to post any bond or other security or to prove that money damages would be inadequate.
| D-3 |
9. Entire Agreement; Amendment; Waiver. This Support Agreement and the other agreements referenced herein constitute the entire agreement and understanding of the parties hereto in respect of the subject matter hereof and supersede all prior understandings, agreements or representations by or among the parties hereto, written or oral, to the extent they relate in any way to the subject matter hereof or the transactions contemplated hereby provided, that, for the avoidance of doubt, the foregoing shall not affect the rights and obligations of the parties under the Merger Agreement or any Additional Agreement. This Support Agreement may not be changed, amended, modified or waived (other than to correct a typographical error) as to any particular provision, except by a written instrument executed by all parties hereto. No failure or delay by a party in exercising any right hereunder shall operate as a waiver thereof. No waivers of or exceptions to any term, condition, or provision of this Support Agreement, in any one or more instances, shall be deemed to be or construed as a further or continuing waiver of any such term, condition, or provision.
10. Binding Effect; Assignment; Third Parties. This Support Agreement and all of the provisions hereof shall be binding upon and inure to the benefit of the parties hereto and their respective permitted successors and assigns. This Support Agreement and all obligations of a Shareholder are personal to such Shareholder and may not be assigned, transferred or delegated by a Shareholder at any time without the prior written consent of Parent and the Company, and any purported assignment, transfer or delegation without such consent shall be null and void ab initio. Nothing contained in this Support Agreement or in any instrument or document executed by any party in connection with the transactions contemplated hereby shall create any rights in, or be deemed to have been executed for the benefit of, any Person that is not a party hereto or thereto or a successor or permitted assign of such a party.
11. Counterparts. This Support Agreement may be executed in any number of original, electronic or facsimile counterparts and each of such counterparts shall for all purposes be deemed to be an original, and all such counterparts shall together constitute but one and the same instrument.
12. Severability. This Support Agreement shall be deemed severable, and the invalidity or unenforceability of any term or provision hereof shall not affect the validity or enforceability of this Support Agreement or of any other term or provision hereof. Furthermore, in lieu of any such invalid or unenforceable term or provision, the parties hereto intend that there shall be added as a part of this Support Agreement a provision as similar in terms to such invalid or unenforceable provision as may be possible and be valid and enforceable.
13. Governing Law; Jury Trial Waiver. Sections 13.7, 13.15, 13.16 and 13.17 of the Merger Agreement are incorporated by reference herein to apply with full force to any disputes arising under this Support Agreement.
14. Notice. Any notice, consent or request to be given in connection with any of the terms or provisions of this Support Agreement shall be in writing and shall be sent or given in accordance with the terms of Section 13.1 of the Merger Agreement to the applicable party, with respect to the Company and Parent, at the respective addresses set forth in Section 13.1 of the Merger Agreement, and, with respect to a Shareholder, at the address set forth underneath such Shareholder’s name on the signature page hereto.
15. Termination. This Support Agreement become effective upon the date hereof and shall automatically terminate, and none of Parent, the Company or a Shareholder shall have any rights or obligations hereunder, on the earliest of (i) the mutual written consent of Parent, the Company and the such Shareholder, (ii) the Closing (following the performance of the obligations of the parties hereunder required to be performed at or prior to the Closing), or (iii) the termination of the Merger Agreement in accordance with its terms. No such termination shall relieve a Shareholder, Parent or the Company from any liability resulting from a breach of this Support Agreement occurring prior to such termination. Notwithstanding anything to the contrary herein, the provisions of this Section 15 shall survive the termination of this Support Agreement.
16. Adjustment for Stock Split. If, and as often as, there are any changes in the Shareholder Shares by way of stock split, stock dividend, combination or reclassification, or through merger, consolidation, reorganization, recapitalization or business combination, or by any other means, equitable adjustment shall be made to the provisions of this Support Agreement as may be required so that the rights, privileges, duties and obligations hereunder shall continue with respect to such Shareholder, Parent, the Company, the Shareholder Shares as so changed.
17. Further Actions. Each of the parties hereto agrees to execute and deliver hereafter any further document, agreement or instrument of assignment, transfer or conveyance as may be necessary or desirable to effectuate the purposes hereof and as may be reasonably requested in writing by another party hereto.
| D-4 |
18. Expenses. Each party shall be responsible for its own fees and expenses (including the fees and expenses of investment bankers, accountants and counsel) in connection with the entering into of this Support Agreement, the performance of its obligations hereunder and the consummation of the transactions contemplated hereby; provided, that in the event of any Action arising out of or relating to this Support Agreement, the non-prevailing party in any such Action will pay its own expenses and the reasonable documented out-of-pocket expenses, including reasonable attorneys’ fees and costs, reasonably incurred by the prevailing party.
19. Interpretation. The titles and subtitles used in this Support Agreement are for convenience only and are not to be considered in construing or interpreting this Support Agreement. In this Support Agreement, unless the context otherwise requires: (i) any pronoun used shall include the corresponding masculine, feminine or neuter forms, and the singular form of nouns, pronouns and verbs shall include the plural and vice versa; (ii) the term “including” (and with correlative meaning “include”) shall be deemed in each case to be followed by the words “without limitation”; and (iii) the words “herein,” “hereto,” and “hereby” and other words of similar import shall be deemed in each case to refer to this Support Agreement as a whole and not to any particular section or other subdivision of this Support Agreement. The parties have participated jointly in the negotiation and drafting of this Support Agreement. Consequently, in the event an ambiguity or question of intent or interpretation arises, this Support Agreement shall be construed as if drafted jointly by the parties hereto, and no presumption or burden of proof shall arise favoring or disfavoring any party by virtue of the authorship of any provision of this Support Agreement.
20. No Partnership, Agency or Joint Venture. This Support Agreement is intended to create a contractual relationship among the Shareholders, the Company and Parent, and is not intended to create, and does not create, any agency, partnership, joint venture or any like relationship among the parties hereto or among any other the Company shareholders entering into support agreements with the Company or Parent. Each Shareholder has acted independently regarding its decision to enter into this Support Agreement. Nothing contained in this Support Agreement shall be deemed to vest in the Company or Parent any direct or indirect ownership or incidence of ownership of or with respect to any Shareholder Shares.
21. Capacity as Shareholder. Each Shareholder signs this Support Agreement solely in Shareholder’s capacity as a shareholder of the Company, and not in any other capacity, including, if applicable, as a director, officer or employee of the Company or any of its Subsidiaries. Nothing herein shall be construed to limit or affect any actions or inactions by a Shareholder or any representative of such Shareholder, as applicable, serving as a director of the Company or any Subsidiary of the Company, acting in such Person’s capacity as a director of the Company or any Subsidiary of the Company.
[Signature Page Follows]
| D-5 |
IN WITNESS WHEREOF, the parties have executed this Company Shareholders Support Agreement as of the date first written above.
| The Company: | ||
| Alps Life Sciences Inc | ||
| By: | ||
| Name: | ||
| Title: | ||
| Parent: | ||
| Globalink Investment Inc. | ||
| By: | ||
| Name: | ||
| Title: | ||
[Signature Page to Company Shareholders Support Agreement]
| D-6 |
| Shareholder: | ||
| By: | ||
| Name: | ||
| Number of Shares: | |
| Shares of Company Ordinary Shares: |
| Address for Notice: | ||
| Address: | ||
| Facsimile No.: | ||
| Telephone No.: | ||
| Email: |
[Signature Page to Company Shareholders Support Agreement]
| D-7 |
ANNEX E
FORM OF PIPE SUBSCRIPTION AGREEMENT
[_____], 2024
Globalink Investment Inc.
200 Continental Drive, Suite 401
Newark, Delaware, 19713
Attn: Say Leong Lim, CEO
Alps Global Holding Pubco
Unit E-18-01 & E-18-02 Level 18
Icon Tower (East)
No. 1 Jalan 1/68F, Jalan Tun Razak
50400 Kuala Lumpur
Wilayah Persekutuan, Malaysia
Attn: Dr. Tham Seng Kong
Alps Life Sciences Inc
Unit E-18-01 & E-18-02 Level 18
Icon Tower (East)
No. 1 Jalan 1/68F, Jalan Tun Razak
50400 Kuala Lumpur
Wilayah Persekutuan, Malaysia
Attn: Dr. Tham Seng Kong; Low Wei Sim; Christie Elizabeth
Ladies and Gentlemen:
In connection with the proposed business combination (the “Transaction”) by and among Globalink Investment Inc. a Delaware corporation (“Parent”), Alps Global Holding Pubco, a Cayman Islands exempted company (“Pubco”), Alps Biosciences Merger Sub, a Cayman Islands exempted company and a wholly owned subsidiary of Pubco (“Merger Sub”), Alps Life Sciences Inc, a Cayman Islands exempted company (“Target”), , pursuant to that certain Amended and Restated Merger Agreement, dated as of May ___, 2024 (as it may be amended, modified or supplemented from time to time, the “Transaction Agreement”), by and among Parent, Pubco, Merger Sub, Target and certain other parties named therein, Pubco is seeking commitments from interested investors to purchase its ordinary shares, par value $0.0001 per share (the “Pubco Ordinary Shares”), for a purchase price of $10.00 per share (the “Per Share Purchase Price” and the aggregate purchase price for all Securities being referred to herein as the “Purchase Price”), in a private placement to be conducted by Pubco (the “Offering”).
Pursuant to the Transaction Agreement, upon the consummation of the Transaction Closing (as defined below), among other things, (a) Parent will redomicile into the Cayman Islands via a merger between Parent and Pubco, with Pubco continuing as the surviving company (the “Reincorporation Merger”); and (b) concurrently therewith or immediately after the Reincorporation Merger, Merger Sub will merge with and into Target, with Alps Holdco being the surviving company and a wholly-owned subsidiary of Pubco. Upon provision of written notification by the Seller Representative (as defined in the Transaction Agreement): (i) the definition of “Parent” under this Subscription Agreement shall be automatically replaced and substituted to mean Reincorporation Merger Surviving Company (as defined in the Transaction Agreement); and (ii) references to the Securities shall mean the Reincorporation Merger Surviving Company Ordinary Shares (as defined in the Transaction Agreement) into which each issued and outstanding Parent Common Stock (as defined in the Transaction Agreement) and Alps Holdco Ordinary Share will convert into the Reincorporation Merger Surviving Company Ordinary Shares upon the Effective Date (as defined in the Transaction Agreement) in accordance with the terms of the Transaction Agreement.
| E-1 |
In connection therewith, the undersigned subscriber (“Subscriber”), Parent and Target agree in this subscription agreement (this “Subscription Agreement”) as follows:
1. Subscription. As of the date first written above (the “Subscription Date”), the Subscriber hereby irrevocably subscribes for and agrees to purchase from Pubco, and Pubco hereby agrees to issue and sell to the Subscriber upon payment of the Purchase Price, such number of Pubco Ordinary Shares as is set forth on the signature page of this Subscription Agreement (the “Securities”) at the Per Share Purchase Price per Pubco Ordinary Share on the terms and subject to the conditions provided for herein.
2. Closing; Issuance of Shares.
(a) The closing of the sale of the Securities contemplated hereby (the “Closing”, and the date that the Closing actually occurs, the “Closing Date”) is contingent upon and substantially concurrent with consummation of the Transaction (the “Transaction Closing”). The Closing shall occur on the date of, and immediately prior to, the Transaction Closing.
(b) Parent and Pubco shall provide written notice (which may be via email) to the Subscriber (the “Payment Notice”), which Payment Notice shall contain wire instructions for an escrow account (the “Escrow Account”) established by Parent and Pubco with a third party escrow agent (the “Escrow Agent”) to be identified in the Payment Notice. The Subscriber shall deliver to the Escrow Account the aggregate Purchase Price for the Securities subscribed by wire transfer of United States dollars in immediately available funds in accordance with the instructions as set out in the Payment Notice. Upon the Closing, Parent and Pubco shall provide instructions to the Escrow Agent to release the funds in the Escrow Account to Pubco against the issuance to the Subscriber of the Securities, free and clear of any liens or other restrictions whatsoever (other than those arising under state or federal securities laws), in book-entry form as set forth in Section 2(c) below. If this Subscription Agreement is terminated prior to the Closing and any funds have already been sent by the Subscriber to the Escrow Account, then promptly after such termination, Parent and Pubco shall instruct the Escrow Agent to promptly return such funds to the Subscriber.
(c) On the Closing Date, promptly after the Closing, Pubco shall deliver (or cause the delivery of) the Securities in book-entry form with restrictive legends in the amount as set forth on the signature page to the Subscriber as indicated on the signature page or to a custodian designated by the Subscriber, as applicable, as indicated below.
(d) On the Closing Date, Pubco shall deliver (or cause the delivery of) the Securities in the amount as set forth on the signature page to the Subscriber as indicated on the signature page or to a custodian designated by the Subscriber, as applicable, as indicated below.
(e) Simultaneously with the execution and delivery of this Subscription Agreement, Subscriber is delivering to Parent and Pubco a duly completed and executed U.S. Internal Revenue Service Form W-9 or appropriate Form W-8.
3. Closing Conditions. In addition to the condition set forth in the first sentence of Section 2(a) above:
(a) The Closing is also subject to the satisfaction or valid waiver by each of Parent, Pubco and Subscriber of the conditions that, on the Closing Date:
| E-2 |
(i) no suspension of the qualification of the Securities for offering or sale or trading in any jurisdiction, or initiation or threatening of any proceedings for any of such purposes, shall have occurred;
(ii) no applicable governmental authority shall have enacted, issued, promulgated, enforced or entered any judgment, order, law, rule or regulation (whether temporary, preliminary or permanent) which is then in effect and has the effect of making consummation of the transactions contemplated hereby illegal or otherwise restraining or prohibiting consummation of the transactions contemplated hereby, and no governmental authority shall have instituted or threatened in writing a proceeding seeking to impose any such restraint or prohibition; and
(iii) all material conditions precedent to the Transaction Closing set forth in the Transaction Agreement shall have been satisfied or waived (other than those conditions which, by their nature, are to be satisfied at the Transaction Closing).
(b) The obligations of Parent and Pubco to consummate the Closing are also subject to the satisfaction or valid waiver by Parent and Pubco of the additional conditions that, on the Closing Date:
(i) all representations and warranties of the Subscriber contained in this Subscription Agreement shall be true and correct in all material respects (other than representations and warranties that are qualified as to materiality or Material Adverse Effect (as defined herein), which representations and warranties shall be true in all respects) at and as of the Closing Date (except for representations and warranties made as of a specific date, which shall be true and correct in all material respects (other than representations and warranties that are qualified as to materiality or Material Adverse Effect, which representations and warranties shall be true in all respects) as of such date), and consummation of the Closing, shall constitute a reaffirmation by the Subscriber of each of the representations, warranties and agreements of the Subscriber contained in this Subscription Agreement as of the Closing Date; and
(ii) the Subscriber shall have performed, satisfied and complied in all material respects with all covenants, agreements and conditions required by this Subscription Agreement to be performed, satisfied or complied with by it at or prior to Closing.
(c) The obligations of the Subscriber to consummate the Closing are also subject to the satisfaction or valid waiver by the Subscriber of the additional conditions that, on the Closing Date:
(i) all representations and warranties of Parent, Pubco and Target contained in this Subscription Agreement shall be true and correct in all material respects (other than representations and warranties that are qualified as to materiality or Material Adverse Effect (as defined herein), which representations and warranties shall be true in all respects) at and as of the Closing Date (except for representations and warranties made as of a specific date, which shall be true and correct in all material respects (other than representations and warranties that are qualified as to materiality or Material Adverse Effect, which representations and warranties shall be true in all respects) as of such date), and consummation of the Closing, shall constitute a reaffirmation by Parent, Pubco and Target of each of the representations, warranties and agreements of Parent, Pubco and Target contained in this Subscription Agreement as of the Closing Date; and
(ii) Parent, Pubco and Target each shall have performed, satisfied and complied in all material respects with all covenants, agreements and conditions required by this Subscription Agreement to be performed, satisfied or complied with by it at or prior to Closing.
| E-3 |
4. Pubco Representations and Warranties. Pubco represents and warrants to the Subscriber that:
(a) As of the date hereof, Pubco is, and as of the Closing, Pubco will be, a corporation duly incorporated, registered and/or organized, validly existing and in good standing under the laws of the jurisdiction of its incorporation, registration or organization. Pubco has the corporate power and authority to own, lease and operate its properties and conduct its business as presently conducted and to enter into, deliver and perform its obligations under this Subscription Agreement. This Subscription Agreement has been duly authorized, executed and delivered by Pubco and is enforceable against Pubco in accordance with its terms, except as may be limited or otherwise affected by (i) bankruptcy, insolvency, fraudulent conveyance, reorganization, moratorium or other laws relating to or affecting the rights of creditors generally, and (ii) principles of equity, whether considered at law or equity.
(b) The Securities have been duly authorized and, when issued and delivered to the Subscriber against full payment therefor in accordance with the terms of this Subscription Agreement, the Securities will be validly issued, fully paid and non-assessable and will not have been issued in violation of or subject to any preemptive or similar rights created under the memorandum and articles of association of Pubco (together, and in each case as amended, modified or supplemented from time to time, the “PubCo Charter”) or under the laws of the Cayman Islands.
(c) The issuance and sale of the Securities and the compliance by Pubco with all of the provisions of this Subscription Agreement and the consummation of the transactions herein will be done in accordance with the Nasdaq marketplace rules and will not conflict with or result in a material breach or material violation of any of the terms or provisions of, or constitute a material default under, or result in the creation or imposition of any lien, charge or encumbrance upon any of the property or assets of Pubco or any of its subsidiaries pursuant to the terms of (i) any indenture, mortgage, deed of trust, loan agreement, license, lease or any other agreement or instrument to which Pubco or any of its subsidiaries is a party or by which Pubco or any of its subsidiaries is bound or to which any of the property or assets of Pubco is subject, which would have a material adverse effect on the business, properties, financial condition, stockholders’ equity or results of operations of Pubco (a “Material Adverse Effect”) or materially affect the validity of the Securities or the legal authority of Pubco to comply in all material respects with the terms of this Subscription Agreement; (ii) result in any material violation of the provisions of the organizational documents of Pubco; or (iii) result in any violation of any statute or any judgment, order, rule or regulation of any court or governmental agency or body, domestic or foreign, having jurisdiction over Pubco or any of its properties that would have a Material Adverse Effect or materially affect the validity of the Securities or the legal authority of Pubco to comply with this Subscription Agreement; subject, in the case of the foregoing clauses (i) and (iii) with respect to the consummation of the transactions therein contemplated.
(d) Pubco has not entered into any agreement or arrangement entitling any agent, broker, investment banker, financial advisor or other person to any broker’s or finder’s fee or any other commission or similar fee in connection with the transactions contemplated by this Subscription Agreement for which the Subscriber could become liable. [ Other than [TBD] (the “Placement Agent”), which is acting as placement agent to Pubco,]1 Pubco is not aware of any person that has been or will be paid (directly or indirectly) remuneration for solicitation of purchasers in connection with the sale of any Securities in the Offering.
(e) Pubco is not, and immediately after receipt of payment for the Securities, will not be, an “investment company” within the meaning of the Investment Company Act of 1940, as amended.
1 NTD: To be determined whether the offering will involve a placement agent.
| E-4 |
(f) Assuming the accuracy of the Subscriber’s representations and warranties set forth in Section 6, in connection with the offer, sale and issuance of the Securities in the manner contemplated by this Subscription Agreement, it is not necessary to register the Securities under the Securities Act of 1933, as amended (the “Securities Act”).
(g) Pubco understands that the foregoing representations and warranties shall be deemed material to and have been relied upon by the Subscriber.
5. Parent and Target Representations and Warranties. Each of Parent and Target represents and warrants to the Subscriber that:
(a) As of the date hereof, each of Parent and Target is a company duly organized, validly existing and in good standing under the laws of the jurisdiction of its incorporation, registration or organization. Each of Parent and Target has the corporate power and authority to own, lease and operate its properties and conduct its business as presently conducted and to enter into, deliver and perform its obligations under this Subscription Agreement. This Subscription Agreement has been duly authorized, executed and delivered by each of Parent and Target and is enforceable against each of Parent and Target in accordance with its terms, except as may be limited or otherwise affected by (i) bankruptcy, insolvency, fraudulent conveyance, reorganization, moratorium or other laws relating to or affecting the rights of creditors generally, and (ii) principles of equity, whether considered at law or equity.
(b) Neither Parent nor Target has entered into any agreement or arrangement entitling any agent, broker, investment banker, financial advisor or other person to any broker’s or finder’s fee or any other commission or similar fee in connection with the transactions contemplated by this Subscription Agreement for which the Subscriber could become liable. [Other than the Placement Agent,] Parent and Target are not aware of any person that has been or will be paid (directly or indirectly) remuneration for solicitation of purchasers in connection with the sale of any Securities in the Offering.
(c) The execution and delivery of this Subscription Agreement by each of Parent and Target, and the compliance by Parent and Target with all of the provisions of this Subscription Agreement and the consummation of the transactions herein will not conflict with or result in a material breach or material violation of any of the terms or provisions of, or constitute a material default under, or result in the creation or imposition of any lien, charge or encumbrance upon any of the property or assets of pursuant to the terms of (i) any indenture, mortgage, deed of trust, loan agreement, license, lease or any other agreement or instrument to which either Parent or Target is a party or by which Parent or Target is bound or to which any of the property or assets of Parent or Target is subject, which would have a Material Adverse Effect or materially affect the legal authority of Parent or Target to comply in all material respects with the terms of this Subscription Agreement; (ii) result in any material violation of the provisions of the organizational documents of Parent or Target; or (iii) result in any violation of any statute or any judgment, order, rule or regulation of any court or governmental agency or body, domestic or foreign, having jurisdiction over Parent or Target or any of its properties that would have a Material Adverse Effect or materially affect the legal authority of Parent or Target to comply with this Subscription Agreement; subject, in the case of the foregoing clauses (i) and (iii) with respect to the consummation of the transactions therein contemplated.
(d) Each of Parent and Target understands that the foregoing representations and warranties shall be deemed material to and have been relied upon by the Subscriber.
| E-5 |
6. Subscriber Representations, Warranties and Covenants. The Subscriber represents and warrants to, and covenants with Parent, Pubco and Target that:
(a) The Subscriber is either a U.S. investor or non-U.S. investor as set forth under its name on the signature page hereto, and accordingly represents the applicable additional matters under clause (i) or (ii) below:
(i) Applicable to U.S. investors: At the time the Subscriber was offered the Securities, it was, and as of the date hereof, the Subscriber is (A) a “qualified institutional buyer” (within the meaning of Rule 144A under the Securities Act) or an “accredited investor” (within the meaning of Rule 501(a) of Regulation D under the Securities Act) as indicated in the questionnaire attached as Exhibit B hereto, and (B) is acquiring the Securities only for its own account and (C) not for the account of others, and not on behalf of any other account or person or with a view to, or for offer or sale in connection with, any distribution thereof in violation of the Securities Act. The Subscriber is not an entity formed for the specific purpose of acquiring the Securities.
(ii) Applicable to non-U.S. investors: The Subscriber understands that the sale of the Securities is made pursuant to and in reliance upon Regulation S promulgated under the Securities Act (“Regulation S”). The Subscriber is not a U.S. Person (as defined in Regulation S), it is acquiring the Securities in an offshore transaction in reliance on Regulation S, and it has received all the information that it considers necessary and appropriate to decide whether to acquire the Securities hereunder. The Subscriber is not relying on any statements or representations made in connection with the transactions contemplated hereby other than representations contained in this Subscription Agreement. The Subscriber understands and agrees that Securities sold pursuant to Regulation S may be subject to restrictions thereunder, including compliance with the distribution compliance period provisions therein.
(b) The Subscriber understands that the Securities are being offered in a transaction not involving any public offering within the meaning of the Securities Act and that the Securities issued at the Closing have not been registered under the Securities Act. The Subscriber understands that the Securities may not be resold, transferred, pledged or otherwise disposed of by the Subscriber absent an effective registration statement under the Securities Act except (i) to Pubco or a subsidiary thereof, (ii) to non-U.S. persons pursuant to offers and sales that occur outside the United States within the meaning of Regulation S under the Securities Act or (iii) pursuant to another applicable exemption from the registration requirements of the Securities Act, and in each of cases (i) and (iii) in accordance with any applicable securities laws of the states and other jurisdictions of the United States, and that any certificates (if any) or any book-entry shares representing the Securities issued at the Closing shall contain a legend or restrictive notation to such effect. The Subscriber acknowledges that the Securities will not be eligible for resale pursuant to Rule 144A promulgated under the Securities Act. The Subscriber understands and agrees that the Securities, until registered under an effective registration statement, will be subject to transfer restrictions and, as a result of these transfer restrictions, the Subscriber may not be able to readily resell the Securities and may be required to bear the financial risk of an investment in the Securities for an indefinite period of time. The Subscriber understands that it has been advised to consult legal counsel prior to making any offer, resale, pledge or transfer of any of the Securities.
(c) The Subscriber understands and agrees that the Subscriber is purchasing Securities directly from Pubco. The Subscriber further acknowledges that there have been no representations, warranties, covenants and agreements made to the Subscriber by Pubco, Parent, Target, or any of their respective officers or directors, or any other person, expressly (other than those representations, warranties, covenants and agreements included in this Subscription Agreement) or by implication.
| E-6 |
(d) The Subscriber acknowledges and agrees that the Subscriber has received such information as the Subscriber deems necessary in order to make an investment decision with respect to the Securities. Without limiting the generality of the foregoing, the Subscriber acknowledges that it has received and carefully reviewed the following items (collectively, the “Disclosure Documents”): (i) the final prospectus of Parent, dated as of December 6, 2021 and filed with the SEC (File No. 333-261222) on December 8, 2021 (the “Prospectus”), (ii) each filing made by Parent and Pubco with the SEC following the filing of the Prospectus through the date of this Subscription Agreement, (iii) the Transaction Agreement, a copy of which will be filed by Parent with the SEC [and (iv) the investor presentation by Parent and Target, a copy of which will be furnished by Parent to the SEC]2. The Subscriber understands the significant extent to which certain of the disclosures contained in items (i) and (ii) above shall not apply following the Transaction Closing. The Subscriber represents and agrees that the Subscriber and the Subscriber’s professional advisor(s), if any, have had the full opportunity to ask Parent’s and Target’s management questions, receive such answers and obtain such information as the Subscriber and such Subscriber’s professional advisor(s), if any, have deemed necessary to make an investment decision with respect to the Securities. The Subscriber has conducted its own investigation of Parent, Target and the Securities and the Subscriber has made its own assessment and have satisfied itself concerning the relevant tax and other economic considerations relevant to its investment in the Securities. The Subscriber further acknowledges that the information contained in the Disclosure Documents is subject to change, and that any changes to the information contained in the Disclosure Documents, including any changes based on updated information or changes in terms of the Transaction, shall in no way affect the Subscriber’s obligation to purchase the Securities hereunder, except as otherwise provided herein.
(e) The Subscriber became aware of this Offering of the Securities solely by means of direct contact between the Subscriber and Pubco, Parent, Target, [the Placement Agent] or a representative of Pubco, Parent, Target or [the Placement Agent,] and the Securities were offered to the Subscriber solely by direct contact between the Subscriber and Pubco, Parent, Target, [the Placement Agent] or a representative of Pubco, Parent, Target or [the applicable Placement Agent.] The Subscriber acknowledges that Pubco represents and warrants that the Securities (i) were not offered by any form of general solicitation or general advertising and (ii) are not being offered in a manner involving a public offering under, or in a distribution in violation of, the Securities Act, or any state securities laws. The Subscriber has a substantive pre-existing relationship with Pubco, Parent, Target or their respective affiliates or [the Placement Agent] for this Offering of the Securities. Neither the Subscriber, nor any of its directors, officers, employees, agents, stockholders or partners has either directly or indirectly, including through a broker or finder, (i) to its knowledge, engaged in any general solicitation, or (ii) published any advertisement in connection with the Offering.
(f) The Subscriber acknowledges that it is aware that there are substantial risks incident to the purchase and ownership of the Securities, including those set forth in the Disclosure Documents and in Parent’s and Pubco’s filings with the SEC. The Subscriber is able to fend for itself in the transactions contemplated herein and has such knowledge and experience in financial and business matters as to be capable of evaluating the merits and risks of an investment in the Securities, and the Subscriber has sought such accounting, legal and tax advice as the Subscriber has considered necessary to make an informed investment decision.
(g) Alone, or together with any professional advisor(s), the Subscriber has adequately analyzed and fully considered the risks of an investment in the Securities and determined that the Securities are a suitable investment for the Subscriber and that the Subscriber is able at this time and in the foreseeable future to bear the economic risk of a total loss of the Subscriber’s investment in Pubco (and after the Transaction Closing, Reincorporation Merger Surviving Company). The Subscriber acknowledges specifically that a possibility of total loss exists.
2 Subject to negotiation of the parties.
| E-7 |
(h) In making its decision to purchase the Securities, the Subscriber has relied solely upon independent investigation made by the Subscriber and the representations and warranties of Pubco, Parent and Target set forth herein. [Without limiting the generality of the foregoing, the Subscriber has not relied on any statements or other information provided by the Placement Agent concerning Pubco, Parent, Target or the Securities or the offer and sale of the Securities.]
(i) The Subscriber understands and agrees that no federal or state agency has passed upon or endorsed the merits of this Offering of the Securities or made any findings or determination as to the fairness of this investment or the accuracy or adequacy of the Disclosure Documents.
(j) If an entity, the Subscriber has been duly formed or incorporated and is validly existing in good standing under the laws of its jurisdiction of incorporation or formation. The execution, delivery and performance by the Subscriber of this Subscription Agreement are within the powers of the Subscriber, have been duly authorized and will not constitute or result in a breach or default under or conflict with any law, statute, rule or regulation applicable to the Subscriber, any order, ruling or regulation of any court or other tribunal or of any governmental commission or agency, or any agreement or other undertaking, to which the Subscriber is a party or by which the Subscriber is bound, and, if the Subscriber is not an individual, will not violate any provisions of the Subscriber’s organizational documents. The signature on this Subscription Agreement is genuine, and the signatory, if the Subscriber is an individual, has legal competence and capacity to execute the same or, if the Subscriber is not an individual the signatory has been duly authorized to execute the same, and this Subscription Agreement constitutes a legal, valid and binding obligation of the Subscriber, enforceable against the Subscriber in accordance with its terms.
(k) Neither the due diligence investigation conducted by the Subscriber in connection with making its decision to acquire the Securities nor any representations and warranties made by the Subscriber herein shall modify, amend or affect the Subscriber’s right to rely on the truth, accuracy and completeness of Parent’s or Target’s representations and warranties contained herein.
(l) The Subscriber is not (i) a person named on the List of Specially Designated Nationals and Blocked Persons administered by the U.S. Treasury Department’s Office of Foreign Assets Control (“OFAC”) or in any Executive Order issued by the President of the United States and administered by OFAC (“OFAC List”), or a person prohibited by any OFAC sanctions program, (ii) a Designated National as defined in the Cuban Assets Control Regulations, 31 C.F.R. Part 515, or (iii) a non-U.S. shell bank or providing banking services indirectly to a non-U.S. shell bank (collectively, a “Prohibited Investor”). The Subscriber agrees to provide law enforcement agencies, if requested thereby, such records as required by applicable law, provided that the Subscriber is permitted to do so under applicable law. If the Subscriber is a financial institution subject to the Bank Secrecy Act (31 U.S.C. Section 5311 et seq.), as amended by the USA PATRIOT Act of 2001, and its implementing regulations (collectively, the “BSA/PATRIOT Act”), the Subscriber maintains policies and procedures reasonably designed to comply with applicable obligations under the BSA/PATRIOT Act. To the extent required, it maintains policies and procedures reasonably designed for the screening of its investors against the OFAC sanctions programs, including the OFAC List. To the extent required, it maintains policies and procedures reasonably designed to ensure that the funds held by the Subscriber and used to purchase the Securities were legally derived.
(m) As of the date hereof and as of the Closing Date, neither the Subscriber, nor, to the extent it has them, any of its equity holders, managers, general or limited partners, directors, affiliates or executive officers (collectively with the Subscriber, the “Covered Persons”), are subject to any of the “Bad Actor” disqualifications described in Rule 506(d) under the Securities Act (a “Disqualification Event”), except for a Disqualification Event covered by Rule 506(d)(2) or (d)(3). The Subscriber has exercised reasonable care to determine whether any Covered Person is subject to a Disqualification Event. The acquisition of Securities by the Subscriber will not subject Parent or Target to any Disqualification Event.
| E-8 |
(n) [No disclosure or offering document has been prepared by the Placement Agent in connection with the offer and sale of the Securities. The Placement Agent and each of its respective members, directors, officers, employees, representatives and controlling persons have made no independent investigation with respect to Pubco, Parent, Target or the Securities or the accuracy, completeness or adequacy of any information supplied to the Subscriber by Pubco, Parent or Target. In connection with the issue and purchase of the Securities, the Placement Agent has not acted as the Subscriber’s financial advisor or fiduciary.]
(o) The Subscriber acknowledges its obligations under applicable securities laws with respect to the treatment of non-public information relating to Pubco and Parent.
(p) At the Closing, the Subscriber will have sufficient funds to pay the Purchase Price.
7. Registration Rights.
(a) Pubco agrees that, within sixty (60) calendar days after the Transaction Closing, Pubco will file with the SEC (at Target’s sole cost and expense) a registration statement registering the resale of the Securities (the “Registration Statement”), and Pubco shall use its commercially reasonable efforts to have the Registration Statement declared effective as soon as practicable after the filing thereof. Pubco agrees that Pubco will cause such Registration Statement or another registration statement (which may be a “shelf” registration statement) to remain effective until the earlier of (i) two years from the issuance of the Securities, (ii) the date on which the Subscriber ceases to hold the Securities covered by such Registration Statement, or (iii) on the first date on which the Subscriber can sell all of its Securities (or shares received in exchange therefor) under Rule 144 of the Securities Act without limitation as to the manner of sale or the amount of such securities that may be sold. The Subscriber agrees to disclose its beneficial ownership, as determined in accordance with Rule 13d-3 of the Exchange Act, of the Securities to Pubco (or its successor) upon request to assist Pubco in making the determination described above. Pubco’s obligations to include the Securities in the Registration Statement are contingent upon the Subscriber furnishing in writing to Pubco such information regarding the Subscriber, the securities of Pubco held by the Subscriber and the intended method of disposition of the Securities as shall be reasonably requested by Pubco to effect the registration of the Securities, and shall execute such documents in connection with such registration as Pubco may reasonably request that are customary of a selling stockholder in similar situations.
(b) Pubco may delay filing or suspend the use of any such registration statement if it determines that in order for the registration statement to not contain a material misstatement or omission, an amendment thereto would be needed, or if such filing or use could materially affect a bona fide business or financing transaction of Pubco or would require premature disclosure of information that could materially adversely affect Pubco (each such circumstance, a “Suspension Event”); provided, that Pubco shall use commercially reasonable efforts to make such registration statement available for the sale by the Subscriber of such securities as soon as practicable thereafter. Upon receipt of any written notice from Pubco of the happening of any Suspension Event during the period that the Registration Statement is effective or if as a result of a Suspension Event the Registration Statement or related prospectus contains any untrue statement of a material fact or omits to state any material fact required to be stated therein or necessary to make the statements therein, in light of the circumstances under which they were made (in the case of the prospectus) not misleading, the Subscriber agrees that it will (i) immediately discontinue offers and sales of the Securities under the Registration Statement until the Subscriber receives (A) (x) copies of a supplemental or amended prospectus that corrects the misstatement(s) or omission(s) referred to above and (y) notice that any post-effective amendment has become effective or (B) notice from Pubco that it may resume such offers and sales, and (ii) maintain the confidentiality of any information included in such written notice delivered by Parent unless otherwise required by applicable law. If so directed by Pubco, the Subscriber will deliver to Pubco or destroy all copies of the prospectus covering the Securities in the Subscriber’s possession; provided, however, that this obligation to deliver or destroy all copies of the prospectus covering the Securities shall not apply to (i) the extent the Subscriber is required to retain a copy of such prospectus (A) in order to comply with applicable legal, regulatory, self-regulatory or professional requirements or (B) in accordance with a bona fide pre-existing document retention policy or (ii) copies stored electronically on archival servers as a result of automatic data back-up.
| E-9 |
8. Termination. This Subscription Agreement shall terminate and be void and of no further force and effect, and all rights and obligations of the parties hereunder shall terminate without any further liability on the part of any party in respect thereof, upon the earlier to occur of: (a) the mutual written agreement of Parent, Pubco, the Subscriber and Target to terminate this Subscription Agreement; (b) such date and time as the Transaction Agreement is terminated in accordance with its terms; or (c) if any of the conditions to Closing set forth in Section 3 are not satisfied on or prior to the Closing Date and, as a result thereof, the transactions contemplated by this Subscription Agreement are not consummated at the Closing, or (d) unanimous written notice by Parent, Pubco and Target to the Subscriber to terminate this Subscription Agreement if the transactions contemplated by this Subscription Agreement are not consummated on or prior to the Outside Closing Date (as defined in the Transaction Agreement); provided that (i) nothing herein will relieve any party from liability for any willful breach hereof prior to the time of termination, and each party will be entitled to any remedies at law or in equity to recover losses, liabilities or damages arising from such breach. Pubco shall notify the Subscriber of the termination of the Transaction Agreement promptly after the termination thereof and (ii) the provisions of Sections 8 through 11 of this Subscription Agreement will survive any termination of this Subscription Agreement and continue indefinitely.
9. Trust Account Waiver. The Subscriber hereby represents and warrants that it has read the Prospectus and understands that Parent has established a trust account (the “Trust Account”) containing the proceeds of its IPO and the overallotment securities acquired by its underwriters and from certain private placements occurring simultaneously with the IPO (including interest accrued from time to time thereon) for the benefit of Parent’s public stockholders (including overallotment shares acquired by Parent’s underwriters, the “Public Stockholders”), and that, except as otherwise described in the Prospectus, Parent may disburse monies from the Trust Account only: (a) to the Public Stockholders in the event they elect to redeem their Parent shares in connection with the consummation of Parent’s initial business combination (as such term is used in the Prospectus) (the “Business Combination”) or in connection with an extension of its deadline to consummate a Business Combination, (b) to the Public Stockholders if Parent fails to consummate a Business Combination within the time limit prescribed in Parent’s organizational documents, (c) with respect to any interest earned on the amounts held in the Trust Account, amounts necessary to pay for any taxes and up to $100,000 in dissolution expenses, or (d) to Parent after or concurrently with the consummation of a Business Combination. For and in consideration of Parent entering into this Subscription Agreement, and for other good and valuable consideration, the receipt and sufficiency of which is hereby acknowledged, the Subscriber hereby agrees on behalf of itself and its affiliates that, notwithstanding anything to the contrary in this Subscription Agreement, neither the Subscriber nor any of its affiliates do now or shall at any time hereafter have any right, title, interest or claim of any kind in or to any monies in the Trust Account or distributions therefrom, or make any claim against the Trust Account (including any distributions therefrom), regardless of whether such claim arises as a result of, in connection with or relating in any way to, this Subscription Agreement or any proposed or actual business relationship between Parent or its Representatives, on the one hand, and the Subscriber or its Representatives, on the other hand, or any other matter, and regardless of whether such claim arises based on contract, tort, equity or any other theory of legal liability (collectively, the “Released Claims”). The Subscriber on behalf of itself and its affiliates hereby irrevocably waives any Released Claims that the Subscriber or any of its affiliates may have against the Trust Account (including any distributions therefrom) now or in the future as a result of, or arising out of, any negotiations, contracts or agreements with Parent or its Representatives and will not seek recourse against the Trust Account (including any distributions therefrom) for any reason whatsoever (including for an alleged breach of this Subscription Agreement or any other agreement with Parent or its affiliates). The Subscriber agrees and acknowledges that such irrevocable waiver is material to this Subscription Agreement and specifically relied upon by Parent and its affiliates to induce Parent to enter into this Subscription Agreement, and the Subscriber further intends and understands such waiver to be valid, binding and enforceable against the Subscriber and each of its affiliates under applicable law. To the extent the Subscriber or any of its affiliates commences any action or proceeding based upon, in connection with, relating to or arising out of any matter relating to Parent or its Representatives, which proceeding seeks, in whole or in part, monetary relief against Parent or its Representatives, the Subscriber hereby acknowledges and agrees that the Subscriber’s and its affiliates’ sole remedy shall be against funds held outside of the Trust Account and that such claim shall not permit the Subscriber or its affiliates (or any person claiming on any of their behalves or in lieu of any of them) to have any claim against the Trust Account (including any distributions therefrom) or any amounts contained therein. In the event the Subscriber or any of its affiliates commences any action or proceeding based upon, in connection with, relating to or arising out of any matter relating to Parent or its Representatives, which proceeding seeks, in whole or in part, relief against the Trust Account (including any distributions therefrom) or the Public Stockholders, whether in the form of money damages or injunctive relief, Parent and its Representatives, as applicable, shall be entitled to recover from the Subscriber and its affiliates the associated legal fees and costs in connection with any such action in the event Parent or its Representatives, as applicable, prevails in such action or proceeding. Notwithstanding the foregoing, this Section 9 shall not affect any rights of Subscriber or its affiliates as a Public Stockholder to receive distributions from the Trust Account in its capacity as a Public Stockholder. For purposes of this Subscription Agreement, “Representatives” with respect to any person shall mean such person’s affiliates and its and its affiliate’s respective directors, officers, employees, consultants, advisors, agents and other representatives. Notwithstanding anything to the contrary contained in this Subscription Agreement, the provisions of this Section 9 shall survive the Closing or any termination of this Subscription Agreement and last indefinitely.
| E-10 |
10. Miscellaneous.
(a) Neither this Subscription Agreement nor any rights that may accrue to the Subscriber hereunder (other than the Securities acquired hereunder, if any, subject to applicable securities laws) may be transferred or assigned by the Subscriber without the prior written consent of Pubco, Parent and Target, and any purported transfer or assignment without such consent shall be null and void ab initio.
(b) Parent and Pubco may request from the Subscriber such additional information as Parent and Pubco may reasonably deem necessary to evaluate the eligibility of the Subscriber to acquire the Securities, and the Subscriber shall provide such information to Parent and Pubco as may be reasonably requested, it being understood by the Subscriber that Parent and Pubco may without any liability hereunder reject the Subscriber’s subscription prior to the Closing Date in the event the Subscriber fails to provide such additional information requested by Parent and Pubco to evaluate the Subscriber’s eligibility or Parent determines that the Subscriber is not eligible.
(c) The Subscriber acknowledges that Pubco, Parent, Target, [the Placement Agent] and others will rely on the acknowledgments, understandings, agreements, representations and warranties of the Subscriber contained in this Subscription Agreement as if they were made directly to them. Prior to the Closing, the Subscriber agrees to promptly notify Pubco, Parent and Target if any of the acknowledgments, understandings, agreements, representations and warranties set forth herein are no longer accurate. The Subscriber agrees that the purchase by the Subscriber of Securities from Pubco will constitute a reaffirmation of the acknowledgments, understandings, agreements, representations and warranties herein (as modified by any such notice) by the Subscriber as of the time of such purchase. [The Subscriber acknowledges and agrees that the Placement Agent is a third-party beneficiary of the representations, warranties and covenants of the Subscriber contained in Section 6 of this Subscription Agreement.] [Each of Pubco, Parent and Target acknowledges and agrees that the Placement Agent is a third-party beneficiary of the representations, warranties and covenants of Parent and Target contained in Sections 4 and 5, respectively, of this Subscription Agreement in performing their respective services to Pubco in connection with the sale of the Securities.] Except as expressly set forth herein, this Subscription Agreement shall not confer any rights or remedies upon any person other than the parties hereto, and their respective successor and assigns.
(d) Each of Pubco, Parent, Target and Subscriber is entitled to rely upon this Subscription Agreement and is irrevocably authorized to produce this Subscription Agreement or a copy hereof to any interested party in any administrative or legal proceeding or official inquiry with respect to the matters covered hereby to the extent required by law or regulatory bodies or applicable stock exchange requirements. The Subscriber shall not issue any press release or make any other similar public statement with respect to the transactions contemplated hereby without the prior written consent of Pubco, Parent and Target (such consent not to be unreasonably withheld or delayed).
(e) All the agreements, representations and warranties made by each party hereto in this Subscription Agreement shall survive the Closing.
(f) This Subscription Agreement may not be amended, modified or terminated except by an instrument in writing, signed by Pubco, Parent, Target and Subscriber. This Subscription Agreement may not be waived except by an instrument in writing signed by the party against whom enforcement of such modification, waiver, or termination is sought. No failure or delay in exercising any right, power or privilege hereunder will operate as a waiver thereof, nor will any single or partial exercise thereof preclude any other or further exercise thereof or the exercise of any right, power or privilege hereunder.
(g) This Subscription Agreement constitutes the entire agreement, and supersedes all other prior agreements, understandings, representations and warranties, both written and oral, among the parties, with respect to the subject matter hereof (other than any confidentiality agreement entered into by Parent or Target and the Subscriber in connection with the Offering).
| E-11 |
(h) This Subscription Agreement shall be binding upon, and inure to the benefit of the parties hereto and their heirs, executors, administrators, successors, legal representatives, and permitted assigns, and the agreements, representations, warranties, covenants and acknowledgments contained herein shall be deemed to be made by, and be binding upon, such heirs, executors, administrators, successors, legal representatives and permitted assigns.
(i) If any provision of this Subscription Agreement shall be invalid, illegal or unenforceable, the validity, legality or enforceability of the remaining provisions of this Subscription Agreement shall not in any way be affected or impaired thereby and shall continue in full force and effect. Upon such determination that any provision is invalid, illegal or unenforceable, the parties will substitute for any invalid, illegal or unenforceable provision a suitable and equitable provision that carries out, so far as may be valid, legal and enforceable, the intent and purpose of such invalid, illegal or unenforceable provision.
(j) This Subscription Agreement may be executed in one or more counterparts (including by facsimile or electronic mail or in .pdf) and by different parties in separate counterparts, with the same effect as if all parties hereto had signed the same document. All counterparts so executed and delivered shall be construed together and shall constitute one and the same agreement.
(k) The parties hereto agree that irreparable damage would occur in the event that any of the provisions of this Subscription Agreement were not performed in accordance with their specific terms or were otherwise breached. It is accordingly agreed that the parties shall be entitled to an injunction or injunctions to prevent breaches of this Subscription Agreement and to enforce specifically the terms and provisions of this Subscription Agreement, this being in addition to any other remedy to which such party is entitled at law, in equity, in contract, in tort or otherwise.
(l) This Subscription Agreement shall be governed by, and construed in accordance with, the laws of the State of Delaware, without regard to principles relating to conflict of laws. Each party hereby irrevocably and unconditionally submits, for itself and its property, to the exclusive jurisdiction of the Chancery Court of the State of Delaware (or, if the Chancery Court of the State of Delaware does not have jurisdiction, a federal court sitting in Wilmington, Delaware) (and any appellate courts thereof) in any action or proceeding arising out of or relating to this Subscription Agreement, and each of the parties hereby irrevocably and unconditionally (a) agrees not to commence any such action or proceeding except in such courts, (b) agrees that any claim in respect of any such action or proceeding may be heard and determined in such court, (c) waives, to the fullest extent it may legally and effectively do so, any objection which it may now or hereafter have to the laying of venue of any such action or proceeding in any such court, and (d) waives, to the fullest extent permitted by law, the defense of an inconvenient forum to the maintenance of such action or proceeding in any such court. Each party agrees that a final judgment in any such action or proceeding shall be conclusive and may be enforced in other jurisdictions by suit on the judgment or in any other manner provided by law. Each party irrevocably consents to the service of the summons and complaint and any other process in any other Proceeding relating to the transactions contemplated by this Subscription Agreement, on behalf of itself, or its property, by personal delivery of copies of such process to such party at the applicable address set forth in Section 10(m). Nothing in this Section 10(l) shall affect the right of any party to serve legal process in any other manner permitted by law. Each party hereby knowingly, voluntarily and intentionally irrevocably waives the right to a trial by jury in respect to any litigation, dispute, claim, legal action or other legal proceeding based hereon, or arising out of, under, or in connection with, this Subscription Agreement.
| E-12 |
(m) All notices, consents, waivers and other communications hereunder shall be in writing and shall be deemed to have been duly given (i) when delivered in person, (ii) when delivered by facsimile or email, with affirmative confirmation of receipt, (iii) one business day after being sent, if sent by reputable, internationally recognized overnight courier service or (iv) three (3) business days after being mailed, if sent by registered or certified mail, prepaid and return receipt requested, in each case to the applicable party at the following addresses (or at such other address for a party as shall be specified by like notice):
If to Pubco at or prior to the Transaction Closing, to:
Alps Global Holding Pubco Unit E-18-01 & E-18-02 Level 18 Icon Tower (East) No. 1 Jalan 1/68F, Jalan Tun Razak 50400 Kuala Lumpur Wilayah Persekutuan, Malaysia Attn: Dr. Tham Seng Kong E-mail: drtham@alpsmedical.com; |
||
If to Parent at or prior to the Transaction Closing, to:
Globalink Investment Inc. 200 Continental Drive, Suite 401 Newark, Delaware, 19713 Attn: Say Leong Lim E-mail: slim@globalinkinvestment.com |
with a copy (which shall not constitute notice) to:
Hunter Taubman Fischer & Li LLC 950 Third Avenue, 19th Floor New York, NY 10022 Attn: Ying Li, Esq.; Guillaume de Sampigny, Esq. Email: yli@htflawyers.com; gdesampigny@htflawyers.com | |
If to Pubco after the Transaction Closing or to Target, to:
Alps Life Sciences Inc. Unit E-18-01 & E-18-02 Level 18 Icon Tower (East) No. 1 Jalan 1/68F, Jalan Tun Razak 50400 Kuala Lumpur Wilayah Persekutuan, Malaysia Attn: Dr. Tham Seng Kong; Low Wei Sim; Christie Elizabeth E-mail: drtham@alpsmedical.com; amanda@alpsmedical.com; christie@alpsmedical.com |
||
| Notice to the Subscriber shall be given to the address underneath the Subscriber’s name on the signature page hereto. | ||
| E-13 |
(n) The headings set forth in this Subscription Agreement are for convenience of reference only and shall not be used in interpreting this Subscription Agreement. In this Subscription Agreement, unless the context otherwise requires: (i) whenever required by the context, any pronoun used in this Subscription Agreement shall include the corresponding masculine, feminine or neuter forms, and the singular form of nouns, pronouns and verbs shall include the plural and vice versa; (ii) “including” (and with correlative meaning “include”) means including without limiting the generality of any description preceding or succeeding such term and shall be deemed in each case to be followed by the words “without limitation”; and (iii) the words “herein”, “hereto” and “hereby” and other words of similar import in this Subscription Agreement shall be deemed in each case to refer to this Subscription Agreement as a whole and not to any particular portion of this Subscription Agreement. As used in this Subscription Agreement, the term: (x) “business day” shall mean any day other than a Saturday, Sunday or a legal holiday on which commercial banking institutions in New York, New York are authorized to close for business (excluding as a result of “stay at home”, “shelter-in-place”, “non-essential employee” or any other similar orders or restrictions or the closure of any physical branch locations at the direction of any governmental authority so long as the electronic funds transfer systems, including for wire transfers, of commercially banking institutions in New York, New York are generally open for use by customers on such day); (y) “person” shall refer to any individual, corporation, partnership, trust, limited liability company or other entity or association, including any governmental or regulatory body, whether acting in an individual, fiduciary or any other capacity; and (z) “affiliate” shall mean, with respect to any specified person, any other person or group of persons acting together that, directly or indirectly, through one or more intermediaries controls, is controlled by or is under common control with such specified person (where the term “control” (and any correlative terms) means the possession, direct or indirect, of the power to direct or cause the direction of the management and policies of such person, whether through the ownership of voting securities, by contract or otherwise). For the avoidance of doubt, any reference in this Subscription Agreement to an affiliate of Parent will include Parent’s sponsor, GL Sponsor LLC.
(o) At Closing, the parties hereto shall execute and deliver such additional documents and take such additional actions as the parties may reasonably deem practical and necessary in order to consummate the Offering as contemplated by this Subscription Agreement.
11. Non-Reliance and Exculpation. The Subscriber acknowledges that it is not relying upon, and has not relied upon, any statement, representation or warranty made by any person other than the statements, representations and warranties contained in this Subscription Agreement in making its investment or decision to invest in Parent. The Subscriber agrees that neither (i) any other purchaser pursuant to other subscription agreements entered into in connection with the Offering (including the controlling persons, members, officers, directors, partners, agents, or employees of any such other purchaser) [nor (ii) the Placement Agent, its affiliates or any of its or its affiliates’ respective control persons, officers, directors or employees, shall be liable to the Subscriber pursuant to this Subscription Agreement for any action heretofore or hereafter taken or omitted to be taken by any of them in connection with the purchase of the Securities.] [The Subscriber acknowledges that neither the Placement Agent, nor any of its Representatives (a) shall be liable to the Subscriber for any improper payment made in accordance with the information provided by Parent; (b) makes any representation or warranty, or has any responsibilities as to the validity, accuracy, value or genuineness of any information, certificates or documentation delivered by or on behalf of Parent or Target pursuant to this Subscription Agreement or the Transaction Agreement (together with any related documents, the “Transaction Documents”); or (c) shall be liable to the Subscriber (x) for any action taken, suffered or omitted by any of them in good faith and reasonably believed to be authorized or within the discretion or rights or powers conferred upon it by this Subscription Agreement or any Transaction Document or (y) for anything which any of them may do or refrain from doing in connection with this Subscription Agreement or any Transaction Document, except for their gross negligence, willful misconduct or bad faith.]
12. Cutback. Notwithstanding anything to the contrary herein, Pubco, with the prior written consent of Parent, shall have the right to reduce the number of Securities to be issued to the Subscriber pursuant to this Subscription Agreement, as long as Pubco is reducing the number of Securities to be issued and sold to all investors pursuant to the other subscription agreements, on a pro rata basis. Pubco [or the Placement Agent] shall notify the Subscriber in writing at least two (2) business days in advance of Closing if Pubco elects to reduce the number of Securities to be issued and sold to the Subscriber pursuant to this Section 12.
[SIGNATURE PAGES FOLLOW]
| E-14 |
IN WITNESS WHEREOF, the parties hereto have caused this Subscription Agreement to be duly executed by their respective authorized signatories as of the date first indicated above.
| GLOBALINK INVESTMENT INC. | ||
| By: | ||
| Name: | ||
| Title: | ||
| ALPS GLOBAL HOLDING PUBCO | ||
| By: | ||
| Name: | ||
| Title: | ||
| ALPS LIFE SCIENCES INC | ||
| By: | ||
| Name: | ||
| Title: | ||
[Signature Page to Subscription Agreement]
{SUBSCRIBER SIGNATURE PAGE TO THE SUBSCRIPTION AGREEMENT}
IN WITNESS WHEREOF, the undersigned has caused this Subscription Agreement to be duly executed by its authorized signatory as of the date first indicated above.
| Name(s) of Subscriber: | |
| Signature of Authorized Signatory of Subscriber: | |
| Name of Authorized Signatory: | |
| Title of Authorized Signatory: |
| Address for Notice to Subscriber: | |
| Attention: | ||
| Email: | ||
| Facsimile No.: | ||
| Telephone No.: |
| Address for Delivery of Securities to Subscriber (if not same as address for notice): | |
| Purchase Price: | $ | |
| Number of Securities: |
| Subscriber status (mark one): ☐ U.S. investor ☐ Non-U.S. investor |
| EIN Number: |
Exhibit A
Accredited Investor Questionnaire
Capitalized terms used and not defined in this Exhibit A shall have the meanings given in the Subscription Agreement to which this Exhibit A is attached. The undersigned represents and warrants that the undersigned is an “accredited investor” (an “Accredited Investor”) as such term is defined in Rule 501(a) of Regulation D under the U.S. Securities Act of 1933, as amended (the “Securities Act”), for one or more of the reasons specified below (please check all boxes that apply):
| _______ (i) | A natural person whose net worth, either individually or jointly with such person’s spouse or spousal equivalent, at the time of the Subscriber’s purchase, exceeds $1,000,000; |
| The term “net worth” means the excess of total assets over total liabilities (including personal and real property, but excluding the estimated fair market value of the Subscriber’s primary home). For the purposes of calculating joint net worth with the person’s spouse or spousal equivalent, joint net worth can be the aggregate net worth of the Subscriber and spouse or spousal equivalent; assets need not be held jointly to be included in the calculation. There is no requirement that securities be purchased jointly. A spousal equivalent means a cohabitant occupying a relationship generally equivalent to a spouse. | |
| _______ (ii) | A natural person who had an individual income in excess of $200,000, or joint income with the Subscriber’s spouse or spousal equivalent in excess of $300,000, in each of the two most recent years and reasonably expects to reach the same income level in the current year; |
| In determining individual “income,” the Subscriber should add to the Subscriber’s individual taxable adjusted gross income (exclusive of any spousal or spousal equivalent income) any amounts attributable to tax exempt income received, losses claimed as a limited partner in any limited partnership, deductions claimed for depletion, contributions to an IRA or Keogh retirement plan, alimony payments, and any amount by which income from long-term capital gains has been reduced in arriving at adjusted gross income. | |
| _______ (iii) | A director or executive officer of Parent; |
| _______ (iv) | A natural person holding in good standing with one or more professional certifications or designations or other credentials from an accredited educational institution that the U.S. Securities Exchange Commission (“SEC”) has designated as qualifying an individual for accredited investor status; |
| The SEC has designated the General Securities Representative license (Series 7), the Private Securities Offering Representative license (Series 82) and the Licensed Investment Adviser Representative (Series 65) as the initial certifications that qualify for accredited investor status. | |
| _______ (v) | A natural person who is a “knowledgeable employee” as defined in Rule 3c-5(a)(4) under the Investment Company Act of 1940 (the “Investment Company Act”), of the issuer of the securities being offered or sold where the issuer would be an investment company, as defined in section 3 of the Investment Company Act, but for the exclusion provided by either section 3(c)(1) or section 3(c)(7) of the Investment Company Act; |
| _______ (vi) | A bank as defined in Section 3(a)(2) of the Securities Act, or any savings and loan association or other institution as defined in Section 3(a)(5)(A) of the Securities Act, whether acting in its individual or fiduciary capacity; |
| A-1 |
| _______ (vii) | A broker or dealer registered pursuant to Section 15 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”); |
| _______ (viii) | An investment adviser registered pursuant to section 203 of the Investment Advisers Act of 1940 (the “Investment Advisers Act”) or registered pursuant to the laws of a state, or an investment adviser relying on the exemption from registering with the SEC under the section 203(l) or (m) of the Investment Advisers Act; |
| _______ (ix) | An insurance company as defined in section 2(13) of the Exchange Act; |
| _______ (x) | An investment company registered under the Investment Company Act or a business development company as defined in Section 2(a)(48) of that Act; |
| _______ (xi) | A Small Business Investment Company licensed by the U.S. Small Business Administration under Section 301(c) or (d) of the Small Business Investment Act of 1958; |
| _______ (xii) | A Rural Business Investment Company as defined in section 384A of the Consolidated Farm and Rural Development Act; |
| _______ (xiii) | A plan established and maintained by a state, its political subdivisions, or any agency or instrumentality of a state, or its political subdivisions for the benefit of its employees, if such plan has total assets in excess of $5,000,000; |
| _______ (xiv) | An employee benefit plan within the meaning of the Employee Retirement Income Security Act of 1974, if the investment decision is made by a plan fiduciary, as defined in Section 3(21) of such act, which is either a bank, savings and loan association, insurance company, or registered investment adviser, or if the employee benefit plan has total assets in excess of $5,000,000 or, if a self-directed plan, with investment decisions made solely by persons that are accredited investors; |
| _______ (xv) | A private business development company as defined in Section 202(a)(22) of the Investment Advisers Act of 1940; |
| _______ (xvi) | An organization described in Section 501(c)(3) of the Internal Revenue Code, or a corporation, business trust, partnership, or limited liability company, or any other entity not formed for the specific purpose of acquiring the Securities, with total assets in excess of $5,000,000; |
| _______ (xvii) | A trust, with total assets in excess of $5,000,000, not formed for the specific purpose of acquiring the Securities, whose purchase is directed by a sophisticated person who has such knowledge and experience in financial and business matters that such person is capable of evaluating the merits and risks of investing in Parent; |
| _______ (xviii) | A “family office” as defined in Rule 202(a)(11)(G)-1 under the Investment Advisers Act with assets under management in excess of $5,000,000 that is not formed for the specific purpose of acquiring the securities offered and whose prospective investment is directed by a person who has such knowledge and experience in financial and business matters that such family office is capable of evaluating the merits and risks of the prospective investment; |
| _______ (xix) | A “family client” as defined in Rule 202(a)(11)(G)-1 under the Investment Advisers Act, of a family office meeting the requirements set forth in (xviii) and whose prospective investment in the issuer is directed by a person from a family office that is capable of evaluating the merits and risks of the prospective investment; |
| A-2 |
| _______ (xx) | A “qualified institutional buyer” as defined in Rule 144A under the Securities Act; |
| _______ (xxi) | An entity, of a type not listed above, not formed for the specific purpose of acquiring the securities offered, owning investments in excess of $5,000,000; and/or |
| _______ (xxii) | An entity in which all of the equity owners qualify as an accredited investor under any of the above subparagraphs. |
| _______ (xxiii) | The Subscriber does not qualify under any of the investor categories set forth in (i) through (xxii) above. |
| 2.1 | Type of the Subscriber. Indicate the form of entity of the Subscriber: |
| ☐ | Individual | ☐ | Limited Partnership | |
| ☐ | Corporation | ☐ | General Partnership | |
| ☐ | Revocable Trust | ☐ | Limited Liability Company | |
| ☐ | Other Type of Trust (indicate type): ________________________________ | |||
| ☐ | Other (indicate form of organization):________________________________ | |||
| 2.2.1 | If the Subscriber is not an individual, indicate the approximate date the Subscriber entity was formed: _____________________. |
| 2.2.2 | If the Subscriber is not an individual, initial the line below which correctly describes the application of the following statement to the Subscriber’s situation: the Subscriber (x) was not organized or reorganized for the specific purpose of acquiring the Securities and (y) has made investments prior to the date hereof, and each beneficial owner thereof has and will share in the investment in proportion to his or her ownership interest in the Subscriber. |
__________ True __________ False
If the “False” line is initialed, each person participating in the entity will be required to fill out a Subscription Agreement.
| Subscriber: | ||
| Subscriber Name: | ||
| By: | ||
| Signatory Name: | ||
| Signatory Title: | ||
| Date: | ||
| A-3 |
ANNEX F
FORM OF LOCK-UP AGREEMENT
This LOCK-UP AGREEMENT (this “Agreement”) is dated as of [*], 2024 by and between the undersigned stockholder (the “Holder”), Alps Global Holding Pubco, a Cayman Islands exempted company (“Pubco”) and Globalink Investment Inc., a Delaware corporation (the “Parent”) and GL Sponsor LLC, in the capacity as the Parent representative (the “Parent Representative”).
A. Parent, Pubco, Alps Life Sciences Inc, a Cayman Islands exempted company (the “Alps Holdco”), Alps Biosciences Merger Sub, a Cayman Islands exempted company and a wholly owned subsidiary of Pubco (“Merger Sub”), and certain other parties thereto, entered into an Amended and Restated Merger Agreement dated as of May 20, 2024 (the “Merger Agreement”). Capitalized terms used, but not otherwise defined herein, shall have the meanings ascribed to such terms in the Merger Agreement.
B. Pubco is an entity newly formed for the purposes of the merger of Parent with and into Pubco (the “Reincorporation Merger”), in which Pubco will be the surviving company, in accordance with the Merger Agreement.
C. Concurrently therewith or immediately after the Reincorporation Merger, Merger Sub will merge with and into Alps Holdco (the “Merger”, the Reincorporation Merger and the Merger are collectively referred to as the “Mergers”), with Alps Holdco being the surviving company (the “Surviving Company”) and a wholly-owned subsidiary of Pubco, in accordance with the Merger Agreement.
D. Upon provision of written notification by the Seller Representative (as defined in the Merger Agreement): (i) the definition of “Parent” under this Agreement shall be automatically replaced and substituted to mean Pubco; and (ii) references to Parent Common Stock shall mean Pubco’s ordinary shares into which the Parent Common Stock will convert upon the effective date of the Reincorporation Merger in accordance with the terms of the Merger Agreement.
E. [The Holder is the record and/or beneficial owner of certain shares of Alps Holdco Ordinary Shares, which will be exchanged for Reincorporation Merger Surviving Company Ordinary Shares pursuant to the Merger Agreement.]1 [The Holder is an Identified Parent Stockholders and the holder of certain shares of Parent Common Stock.]2
F. As a condition of, and as a material inducement for the Parent to enter into and consummate the transactions contemplated by the Merger Agreement, the Holder has agreed to execute and deliver this Agreement.
NOW, THEREFORE, for and in consideration of the mutual covenants and agreements set forth herein, and other good and valuable consideration, the receipt and sufficiency of which is hereby acknowledged, the parties, intending to be legally bound, agree as follows:
1 For Company shareholders.
2 For Parent stockholders.
| F-1 |
AGREEMENT
1. Lock-Up.
(a) Subject to this Section 1 and Section 3 below, during the Lock-Up Period, the Holder agrees that it, he or she will not offer, sell, contract to sell, pledge or otherwise dispose of, directly or indirectly, any of the Lock-Up Shares (as defined below), enter into a transaction that would have the same effect, or enter into any swap, hedge or other arrangement that transfers, in whole or in part, any of the economic consequences of ownership of the Lock-Up Shares or otherwise, publicly disclose the intention to make any offer, sale, pledge or disposition, or to enter into any transaction, swap, hedge or other arrangement, or engage in any Short Sales (as defined below) with respect to the Lock-Up Shares (any of the foregoing, a “Prohibited Transfer”).
(b) In furtherance of the foregoing, during the Lock-Up Period, Pubco shall (i) place a stop order on all the Lock-Up Shares, including those which may be covered by a registration statement, and (ii) notify Pubco’s transfer agent in writing of the stop order and the restrictions on the Lock-Up Shares under this Agreement and direct Pubco’s transfer agent not to process any attempts by the Holder to resell or transfer any Lock-Up Shares, except in compliance with this Agreement.
(c) For purposes hereof, “Short Sales” include all “short sales” as defined in Rule 200 promulgated under Regulation SHO under the Securities Exchange Act of 1934, as amended (the “Exchange Act”), and all types of direct and indirect stock pledges, forward sale contracts, options, puts, calls, swaps and similar arrangements (including on a total return basis), and sales and other transactions through non-US broker dealers or foreign regulated brokers.
(d) The term “Lock-Up Period” (i) (w) with respect to one hundred percent (100%) of the Lock-Up Shares, the date from the Closing until the last date that is six (6) months after the date of the Closing, (x) with respect to ninety percent (90%) of the Lock-Up Shares, the 1st day of the 7th month from the Closing until the date that is nine (9) months after the date of the Closing, (y) with respect to seventy percent (70%) of the Lock-Up Shares, the 1st day of the 10th month from the Closing until the date that is twelve (12) months after the date of the Closing, (z) with respect to forty percent (40%) of the Lock-Up Shares, the 1st day of the 13th month from the Closing until the date that is fifteen (15) months after the date of the Closing, or (ii) if sooner, the date after the Closing on which Pubco consummates a liquidation, merger, share exchange or other similar transaction with an unaffiliated third party that results in all of Pubco’s shareholders having the right to exchange their equity holdings in Pubco for cash, securities or other property. For the avoidance of doubt, no Lock-up Shares shall be subject to the terms of this Agreement from and after the date that is fifteen (15) months after the date of the Closing.
2. Beneficial Ownership. [The Holder hereby represents and warrants that as of the date of this Agreement, it, he or she does not beneficially own, directly or through its, his or her nominees (as determined in accordance with Section 13(d) of the Exchange Act, and the rules and regulations promulgated thereunder), any ordinary shares of Pubco, or any economic interest in or derivative of such shares, other than those ordinary shares of Pubco issued pursuant to the Merger Agreement (“Merger Shares”) or shares that it, he or she may acquire through the PIPE Financing. For purposes of this Agreement, the Merger Shares beneficially owned by the Holder, together with any securities paid as dividends or distributions with respect to such securities or into which such securities are exchanged or converted, are collectively referred to as the “Lock-Up Shares” (for the avoidance of doubt, Lock-Up Shares shall not include ordinary shares of Pubco acquired by such Holder in open market transactions or PIPE Shares).]3 [The Holder hereby represents and warrants that as of the date of this Agreement, it does not beneficially own, directly or through its nominees (as determined in accordance with Section 13(d) of the Exchange Act, and the rules and regulations promulgated thereunder), any shares of capital equity of Pubco, or any economic interest in or derivative of such equity, other than those shares of capital equity of Pubco specified on the signature page hereto (“the Lock-Up Shares”); for the avoidance of doubt, Lock-Up Shares shall not include ordinary shares of Pubco acquired by such Holder in open market transactions or PIPE Shares.]4
3 For Company shareholders.
4 For Parent stockholders.
| F-2 |
3. Permitted Transfers. Notwithstanding the foregoing, and subject to the conditions below, a Prohibited Transfer will not include, and the undersigned may transfer Lock-Up Shares in connection with (a) transfers or distributions to the Holder’s direct or indirect affiliates (within the meaning of Rule 405 under the Securities Act of 1933, as amended (the “Securities Act”)) or to the estates of any of the foregoing; (b) transfers by bona fide gift to a member of the Holder’s immediate family (for purposes of this Agreement, “immediate family” shall mean with respect to any natural person, any of the following: such person’s spouse, the siblings of such person and his or her spouse, and the direct descendants and ascendants (including adopted and step children and parents) of such person and his or her spouses and siblings) or to a trust, the beneficiary of which is the Holder or a member of the Holder’s immediate family for estate planning purposes; (c) by virtue of the laws of descent and distribution upon death of the Holder; (d) pursuant to a qualified domestic relations order, (e) transfers to Pubco’s officers, directors or their affiliates, (f) transfers as a dividend or distribution to limited partners, shareholders, members of, or owners of similar equity interests in the Holder, (g) pledges of Lock-Up Shares as security or collateral in connection with a borrowing or the incurrence of any indebtedness by the Holder, provided, however, that such borrowing or incurrence of indebtedness is secured by either a portfolio of assets or equity interests issued by multiple issuers, (h) transfers pursuant to a bona fide third-party tender offer, merger, stock sale, recapitalization, consolidation or other transaction involving a change of control of Pubco; provided, however, that in the event that such tender offer, merger, recapitalization, consolidation or other such transaction is not completed, the Lock-Up Shares subject to this Agreement shall remain subject to this Agreement, (i) the establishment of a trading plan pursuant to Rule 10b5-1 promulgated under the Exchange Act; provided, however, that such plan does not provide for the transfer of Lock-Up Shares during the Lock-Up Period, (j) transfers to satisfy tax withholding obligations in connection with the exercise of options to purchase ordinary shares of Pubco or the vesting of share-based awards; and (k) transfers in payment on a “net exercise” or “cashless” basis of the exercise or purchase price with respect to the exercise of options to purchase ordinary shares of Pubco; provided, however, that, in the case of any transfer pursuant to the foregoing (a) through (f) clauses, it shall be a condition to any such transfer that (i) the transferee/donee agrees to be bound by the terms of this Agreement (including the restrictions set forth in Section 1) to the same extent as if the transferee/donee were a party hereto; and (ii) each party (donor, donee, transferor or transferee) shall not be required by law (including the disclosure requirements of the Securities Act and the Exchange Act) to make, and shall agree to not voluntarily make, any filing or public announcement of the transfer or disposition prior to the expiration of the Lock-Up Period.
4. Representations and Warranties. Each of the parties hereto, by their respective execution and delivery of this Agreement, hereby represents and warrants to the other that (a) such party has the full right, capacity and authority to enter into, deliver and perform its respective obligations under this Agreement, (b) this Agreement has been duly executed and delivered by such party and is a binding and enforceable obligation of such party and, enforceable against such party in accordance with the terms of this Agreement, and (c) the execution, delivery and performance of such party’s obligations under this Agreement will not conflict with or breach the terms of any other agreement, contract, commitment or understanding to which such party is a party or to which the assets or securities of such party are bound. The Holder has independently evaluated the merits of his/her/its decision to enter into and deliver this Agreement, and such Holder confirms that he/she/it has not relied on the advice of Alps Holdco, its legal counsel, or any other person.
5. No Additional Fees/Payment. Other than the consideration specifically referenced herein to be issued in connection with the Merger Agreement, the parties hereto agree that no fee, payment or additional consideration in any form has been or will be paid to the Holder in connection with this Agreement.
| F-3 |
6. Notices. Any notice hereunder shall be sent in writing, addressed as specified below, and shall be deemed given: (a) if by hand or nationally recognized overnight courier service, by 5:00 PM Eastern Time on a Business Day, addressee’s day and time, on the date of delivery, and if delivered after 5:00 PM Eastern Time, on the first Business Day after such delivery; (b) if by email, on the date of transmission with affirmative confirmation of receipt; or (c) three (3) Business Days after mailing by prepaid certified or registered mail, return receipt requested. Notices shall be addressed to the respective parties as follows, or to such other address as a party shall specify to the others in accordance with these notice provisions:
if to Pubco, to:
Alps Global Holding Pubco
Unit E-18-01 & E-18-02 Level 18
Icon Tower (East)
No. 1 Jalan 1/68F, Jalan Tun Razak
50400 Kuala Lumpur
Wilayah Persekutuan, Malaysia
Attn: Dr. Tham Seng Kong; Low Wei Sim; Christie Elizabeth
E-mail: drtham@alpsmedical.com; amanda@alpsmedical.com; christie@alpsmedical.com
if to Parent prior to the Closing:
Globalink Investment Inc.
200 Continental Drive, Suite 401
Newark, Delaware, 19713
Attn: Say Leong Lim
E-mail: slim@globalinkinvestment.com
with a copy (which shall not constitute notice) to:
Hunter Taubman Fischer & Li LLC
950 Third Avenue, 19th Floor
New York, NY 10022
Attention: Ying Li, Esq; Guillaume de Sampigny, Esq.
E-mail: yli@htflawyers.com; gdesampigny@htflawyers.com
If to the Parent Representative:
GL Sponsor LLC
1180 Avenue of the Americas, 8th Floor,
New York, NY 10036.
Attn: Ng Yan Xun
E-mail: glsponsor.jerryng@gmail.com
with a copy (which shall not constitute notice) to:
Hunter Taubman Fischer & Li LLC
950 Third Avenue, 19th Floor
New York, NY 10022
Attention: Ying Li, Esq; Guillaume de Sampigny, Esq.
E-mail: yli@htflawyers.com; gdesampigny@htflawyers.com
if to the Holder, to the address set forth on the Holder’s signature page hereto.
| F-4 |
7. Termination of Merger Agreement. This Agreement shall be binding upon the Holder upon the Holder’s execution and delivery of this Agreement, but this Agreement shall only become effective upon the Closing. Notwithstanding anything to the contrary contained herein, in the event that the Merger Agreement is terminated in accordance with its terms prior to the Closing, this Agreement and all rights and obligations of the parties hereunder shall automatically terminate and be of no further force or effect.
8. Enumeration and Headings; Interpretation. The enumeration and headings contained in this Agreement are for convenience of reference only and shall not control or affect the meaning or construction of any of the provisions of this Agreement. The titles and subtitles used in this Agreement are for convenience only and are not to be considered in construing or interpreting this Agreement. In this Agreement, unless the context otherwise requires: (i) any pronoun used in this Agreement shall include the corresponding masculine, feminine or neuter forms, and the singular form of nouns, pronouns and verbs shall include the plural and vice versa; (ii) “including” (and with correlative meaning “include”) means including without limiting the generality of any description preceding or succeeding such term and shall be deemed in each case to be followed by the words “without limitation”; and (iii) the words “herein,” “hereto,” and “hereby” and other words of similar import shall be deemed in each case to refer to this Agreement as a whole and not to any particular section or other subdivision of this Agreement.
9. Counterparts. This Agreement may be executed in facsimile (including by email in pdf) and in any number of counterparts, each of which when so executed and delivered shall be deemed an original, but all of which shall together constitute one and the same agreement.
10. Successors and Assigns. This Agreement and the terms, covenants, provisions and conditions hereof shall be binding upon, and shall inure to the benefit of, the respective heirs, successors and assigns of the parties hereto. The Holder hereby acknowledges and agrees that this Agreement is entered into for the benefit of and is enforceable by Pubco and its successors and assigns.
11. No Third Parties. Nothing contained in this Agreement or in any instrument or document executed by any party in connection with the transactions contemplated hereby shall create any rights in, or be deemed to have been executed for the benefit of, any person or entity that is not a party hereto or thereto or a successor or permitted assign of such a party; provided, that Parent and Alps Holdco are express third party beneficiaries of this Agreement and shall have the rights to enforce this Agreement against the parties hereto prior to the Closing.
12. Severability. If any provision of this Agreement is held to be invalid or unenforceable for any reason, such provision will be conformed to prevailing law rather than voided, if possible, in order to achieve the intent of the parties and, in any event, the remaining provisions of this Agreement shall remain in full force and effect and shall be binding upon the parties hereto.
13. Amendment and Waivers. This Agreement may be amended or modified, or any provision hereof waived, by written agreement executed by each of the parties hereto (including prior to the Closing, Alps Holdco, Parent and Pubco). No failure or delay by a party in exercising any right hereunder shall operate as a waiver thereof. No waivers of or exceptions to any term, condition, or provision of this Agreement, in any one or more instances, shall be deemed to be or construed as a further or continuing waiver of any such term, condition, or provision.
14. Further Assurances. Each party shall do and perform, or cause to be done and performed, all such further acts and things, and shall execute and deliver all such other agreements, certificates, instruments and documents, as any other party may reasonably request in order to carry out the intent and accomplish the purposes of this Agreement and the consummation of the transactions contemplated hereby.
15. No Strict Construction. The language used in this Agreement will be deemed to be the language chosen by the parties to express their mutual intent, and no rules of strict construction will be applied against any party.
| F-5 |
16. Dispute Resolution. Section 13.15, 13.16 and 13.17 of the Merger Agreement is incorporated by reference herein to apply with full force to any disputes arising under this Agreement.
17. Governing Law. Section 13.7 of the Merger Agreement is incorporated by reference herein to apply with full force to any disputes arising under this Agreement.
18. Entire Agreement; Controlling Agreement. This Agreement constitutes the full and entire understanding and agreement among the parties with respect to the subject matter hereof, and any other written or oral agreement relating to the subject matter hereof existing between the parties is expressly canceled; provided, that, for the avoidance of doubt, the foregoing shall not affect the rights and obligations of the parties under the Merger Agreement or any Additional Agreement. To the extent the terms of this Agreement (as amended, supplemented, restated or otherwise modified from time to time) directly conflicts with a provision in the Merger Agreement, the terms of this Agreement shall control.
[Signature Page Follows]
| F-6 |
IN WITNESS WHEREOF, the parties hereto have caused this Lock-Up Agreement to be duly executed by their respective authorized signatories as of the date first indicated above.
| Globalink Investment Inc. | ||
| By: | ||
| Name: | ||
| Title: | ||
| Alps Global Holding Pubco | ||
| By: | ||
| Name: | ||
| Title: | ||
| GL Sponsor LLC | ||
| By: | ||
| Name: | ||
| Title: | ||
[Signature Page to Lock-Up Agreement]
| F-7 |
IN WITNESS WHEREOF, the parties hereto have caused this Lock-Up Agreement to be duly executed by their respective authorized signatories as of the date first indicated above.
| HOLDER: | ||
| By: | ||
| Name: | ||
| Title: |
| Address for Notice: | ||
| Address: | ||
| Facsimile No.: | ||
| Telephone No.: | ||
| Email: | ||
| Number of Lock-Up Shares: | ||
[Signature Page to Lock-Up Agreement]
| F-8 |
ANNEX G
FORM OF AMENDED AND RESTATED REGISTRATION RIGHTS AGREEMENT
THIS AMENDED AND RESTATED REGISTRATION RIGHTS AGREEMENT (this “Agreement”), is made and entered into as of [●], 2024, and shall be effective as of the Closing (as defined below), is made and entered into by and among (i) Alps Global Holding Pubco (successor to Globalink Investment Inc.), a Cayman Islands exempted (the “Pubco”), (ii) GL Sponsor LLC, a Delaware limited liability company (the “Sponsor”), (iii) each of the undersigned parties listed as Pre-BC Investors on the signature page hereto (each such party, together with the Sponsor and any person or entity deemed a “Pre-BC Investor” and collectively the “Pre-BC Investors”), and (iv) each of the former shareholders of Alps Life Sciences Inc, a Cayman Islands exempted company (“Alps Holdco”) whose names are listed on Exhibit B hereto (each a “Alps Holdco Shareholder” and collectively the “Alps Holdco Shareholders”) (each of the foregoing parties (other than Pubco) and any Person (as defined below) who hereafter becomes a party to this Agreement pursuant to Section 6.2 of this Agreement, an “Investor” and collectively, the “Investors”).
WHEREAS, each of Globalink Investment Inc., a Delaware corporation (“Globalink”) and the Pre-BC Investors are a party to a certain Registration Rights Agreement, dated December 6, 2021 (the “Original Registration Rights Agreement”), pursuant to which Globalink granted the Pre-BC Investors certain registration rights with respect to certain securities of Pubco, as set forth therein;
WHEREAS, Globalink, Pubco, Alps Holdco, Alps Biosciences Merger Sub, a Cayman Islands exempted company and a wholly owned subsidiary of Pubco (“Merger Sub”), and certain other parties thereto have entered into that certain Amended and Restated Merger Agreement (as it may be amended, modified or supplemented from time to time, the “Merger Agreement”), dated as of May 20, 2024, which provides, among other things, that Globalink will redomicile into the Cayman Islands via a merger between Globalink and Pubco, with Pubco continuing as the surviving company (the “Reincorporation Merger”), in accordance with the Merger Agreement, the Plan of Merger and the Companies Act (as such terms are defined in the Merger Agreement);
WHEREAS, concurrently therewith or immediately after the Reincorporation Merger, Merger Sub will merge with and into Alps Holdco (the “Merger”, the Reincorporation Merger and the Merger are collectively referred to as the “Mergers”), with Alps Holdco being the surviving company (the “Surviving Company”) and a wholly-owned subsidiary of Pubco, and the outstanding shares of Alps Holdco’s share capital will convert into the right to receive Merger Consideration Shares (as defined in the Merger Agreement) described in the Merger Agreement; and
WHEREAS, the Investors and Pubco desire to enter into this Agreement in connection with the Closing to amend and restate the Original Registration Rights Agreement to provide the Investors with certain rights relating to the registration of the securities held by them as of the Closing on the terms and conditions set forth in this Agreement.
| G-1 |
NOW, THEREFORE, in consideration of the mutual covenants and agreements set forth herein, and for other good and valuable consideration, the receipt and sufficiency of which are hereby acknowledged, the parties hereto agree as follows:
1. DEFINITIONS. The following capitalized terms used herein have the following meanings:
“Agreement” means this Agreement, as amended, restated, supplemented, or otherwise modified from time to time.
“Alps Holdco Investors” is defined in the preamble to this Agreement.
“Alps Holdco” is defined in the preamble to this Agreement.
“Blackout Period” is defined in Section 3.1.1.
“Business Day” means a day other than Saturday, Sunday or other day on which commercial banks in New York, New York are authorized or required by law to close, excluding as a result of “stay at home”, “shelter-in-place”, “non-essential employee” or any other similar orders or restrictions or the closure of any physical branch locations at the direction of any governmental authority so long as the electronic funds transfer systems, including for wire transfers, of commercially banking institutions in New York, New York are generally open for use by customers on such day.
“Closing” shall have the meaning given in the Merger Agreement.
“Closing Date” shall have the meaning given in the Merger Agreement.
“Commission” means the U.S. Securities and Exchange Commission, or any other Federal agency then administering the Securities Act or the Exchange Act.
“Common Stock” means the Common Stock of Globalink, par value $0.001 per share.
“Demand Registration” is defined in Section 2.1.1.
“Demanding Holder” is defined in Section 2.1.1.
“Exchange Act” means the Securities Exchange Act of 1934, as amended, and the rules and regulations of the Commission promulgated thereunder, all as the same shall be in effect at the time.
“Form F-3” is defined in Section 2.3.
“Indemnified Party” is defined in Section 4.3.
“Indemnifying Party” is defined in Section 4.3.
“Initial Shares” means all of the outstanding shares of Common Stock issued prior to the consummation of the IPO.
“Investor” is defined in the preamble to this Agreement.
“Investor Indemnified Party” is defined in Section 4.1.
“IPO” means Globalink’s initial public offering.
“IPO Escrow Agreement” means the Stock Escrow Agreement dated as of December 6, 2021 by and among Globalink, certain of the Investors and Continental Stock Transfer & Trust Company.
“Lock-up Agreement” is defined in Section 2.1.1.
“Maximum Number of Shares” is defined in Section 2.1.4.
“Merger” is defined in the preamble to this Agreement.
| G-2 |
“Merger Agreement” is defined in the preamble to this Agreement.
“Merger Sub” is defined in the preamble to this Agreement.
“Original Registration Rights Agreement” is defined in the preamble to this Agreement.
“Person” means a company, corporation, association, partnership, limited liability company, organization, joint venture, trust or other legal entity, an individual, a government or political subdivision thereof or a governmental agency.
“Piggy-Back Registration” is defined in Section 2.2.1(a).
“PIPE Subscription Agreements” means the Subscription Agreements, dated as of [●], [●], by and among Pubco and the subscribers thereto (as may be amended from time to time).
“Pre-BC Investor” is defined in the preamble to this Agreement.
“Private Unit” means each unit of the aggregate of 570,000 units Globalink sold to Public Gold Marketing Sdn. Bhd., a Malaysian private limited company, simultaneously with the consummation of Globalink’s initial public offering.
“Pro Rata” is defined in Section 2.1.4.
“Prospectus” shall mean the prospectus included in any Registration Statement, as supplemented by any and all prospectus supplements and as amended by any and all post-effective amendments and including all material incorporated by reference in such prospectus.
“Pubco” is defined in the preamble to this Agreement.
“Pubco Ordinary Shares” means ordinary shares of Pubco, par value US$0.0001.
“Pubco Underwritten Offering” is defined in Section 2.2.1(b).
“Pubco Underwritten Shelf Offering Requesting Holder” is defined in Section 2.2.1(b).
“Register,” “Registered” and “Registration” mean a registration effected by preparing and filing a registration statement or similar document in compliance with the requirements of the Securities Act, and the applicable rules and regulations promulgated thereunder, and such registration statement becoming effective.
“Registrable Securities” means (i) the Initial Shares, (ii) the Private Units (and underlying securities), (iii) any securities issued upon the conversion at or prior to the Closing of working capital loans from Pre-BC Investors to Globalink, if any, (iv) the Pubco Ordinary Shares issued or hereafter acquired pursuant to the Merger Agreement (including Earnout Shares (as defined in the Merger Agreement)), and (v) any other equity security of Pubco issued or issuable with respect to any such securities by way of a stock dividend, stock split, or other distribution or in connection with a combination of shares, recapitalization, merger, consolidation or reorganization. As to any particular Registrable Securities, such securities shall cease to be Registrable Securities when: (a) a Registration Statement with respect to the sale of such securities shall have become effective under the Securities Act and such securities shall have been sold, transferred, disposed of or exchanged in accordance with such Registration Statement; (b) such securities shall have been otherwise transferred, new certificates for them not bearing a legend restricting further transfer shall have been delivered by Pubco and subsequent public distribution of them shall not require registration under the Securities Act; (c) such securities shall have ceased to be outstanding, or (d) the Registrable Securities are freely saleable by the applicable Investor under Rule 144 without volume limitations, requirements of current public information, manner of sale or any other restrictions under Rule 144.
| G-3 |
“Registration Statement” means a registration statement filed by Pubco with the Commission in compliance with the Securities Act and the rules and regulations promulgated thereunder for a public offering and sale of equity securities, or securities or other obligations exercisable or exchangeable for, or convertible into, equity securities (other than a registration statement on Form S-4 or Form S-8, or their successors, or any registration statement covering only securities proposed to be issued in exchange for securities or assets of another entity).
“Release Date” means the date on which the Initial Shares are disbursed from escrow pursuant to Section 3 of the IPO Escrow Agreement.
“Rule 144” means Rule 144 as promulgated under the Securities Act.
“Securities Act” means the Securities Act of 1933, as amended, and the rules and regulations of the Commission promulgated thereunder, all as the same shall be in effect at the time.
“Sponsor” means GL Sponsor LLC.
“Underwriter” means, solely for the purposes of this Agreement, a securities dealer who purchases any Registrable Securities as principal in an underwritten offering and not as part of such dealer’s market-making activities.
“Underwritten Offering” means a Registration in which securities of Pubco are sold to the Underwriter in a firm commitment underwriting for distribution to the public.
2. REGISTRATION RIGHTS.
2.1 Demand Registration.
2.1.1 Request for Demand Registration. At any time and from time to time on or after (i) three months prior to the first possible Release Date with respect to the Initial Shares that are Registrable Securities and subject to the IPO Escrow Agreement, or (ii) three months prior to the first possible date on which the restrictions on transfer may lapse under the Lock-up Agreement entered into in connection with the Merger Agreement (the “Lock-up Agreement”) with respect to the Private Units (or underlying securities), all Registrable Securities held by the Alps Holdco Shareholders, the holders of a majority-in-interest of such Registrable Securities held by (x) the Pre-BC Investors, on the one hand, or (y) the Alps Holdco Shareholders, on the other hand, as the case may be, held by such Investors, or the transferees of such Investors, may make a written demand, on no more than one occasion in any twelve month period for each of the Pre-BC Investors and the Alps Holdco Shareholders, for registration under the Securities Act of all or part of their Registrable Securities, as the case may be (a “Demand Registration”). Any demand for a Demand Registration shall specify the number of shares of Registrable Securities proposed to be sold and the intended method(s) of distribution thereof. Pubco will notify all holders of Registrable Securities of the demand, and each holder of Registrable Securities who wishes to include all or a portion of such holder’s Registrable Securities in the Demand Registration (each such holder including shares of Registrable Securities in such registration, a “Demanding Holder”) shall so notify Pubco within ten (10) days after the receipt by the holder of the notice from Pubco. Upon any such request, the Demanding Holders shall be entitled to have their Registrable Securities included in the Demand Registration, subject to Section 2.1.4 and the provisos set forth in Section 3.1.1. Pubco shall not be obligated to effect more than an aggregate of two (2) Demand Registrations (up to one (1) Demand Registration initiated by a majority-in-interest of the Pre-BC Investors, and up to one (1) Demand Registration initiated by a majority-in-interest of the Alps Holdco Shareholders) under this Section 2.1.1 in respect of all Registrable Securities.
| G-4 |
2.1.2 Effective Registration. A registration will not count as a Demand Registration until (i) the Registration Statement filed with the Commission with respect to such Demand Registration has been declared effective, and (ii) Pubco has complied with all of its obligations under this Agreement with respect thereto; provided, however, that if, after such Registration Statement has been declared effective, the offering of Registrable Securities pursuant to a Demand Registration is interfered with by any stop order or injunction of the Commission or any other governmental agency or court, the Registration Statement with respect to such Demand Registration will be deemed not to have been declared effective, unless and until, (i) such stop order or injunction is removed, rescinded or otherwise terminated, and (ii) a majority-in-interest of the Demanding Holders thereafter elect to continue the offering; provided, further, that Pubco shall not be obligated to file a second Registration Statement until a Registration Statement that has been filed is counted as a Demand Registration or is terminated.
2.1.3 Underwritten Offering pursuant to Demand Registration. If a majority-in-interest of the Demanding Holders so elect and such holders so advise Pubco as part of their written demand for a Demand Registration, the offering of such Registrable Securities pursuant to such Demand Registration, or a portion thereof, shall be in the form of an Underwritten Offering; provided, however, that the aggregate offering price for any such Underwritten Offering may not be less than $25,000,000, unless Pubco is eligible to register such Pubco Ordinary Shares on Form F-3, or subsequent similar form, in a manner which does not require inclusion of any information concerning Pubco other than to incorporate by reference (including forward incorporation by reference) its filings under the Exchange Act, in which case the aggregate offering price for any such Underwritten Offering may not be less than $10,000,000. All such Demanding Holders proposing to distribute their Registrable Securities through such Underwritten Offering under this Section 2.1.3 shall, at the time of any such Underwritten Offering, enter into an underwriting agreement in customary form with the Underwriter(s) selected by a majority-in-interest of the Demanding Holders; provided, however, that such Underwriter(s) is reasonably satisfactory to Pubco; provided, further, that any obligation of any such Investor to indemnify any Person pursuant to any such underwriting agreement shall be several, not joint, among such Investors selling Registrable Securities, and such liability shall be limited to the net amount received by any such Investor from the sale of his, her or its Registrable Securities pursuant to such Underwritten Offering, and the relative liability of each such Investor shall be in proportion to such net amounts.
2.1.4 Reduction of Offering in Connection with Demand Registration. If the managing Underwriter(s) in an Underwritten Offering effected pursuant to a Demand Registration in good faith advises Pubco and the Demanding Holders in writing that the dollar amount or number of shares of Registrable Securities which the Demanding Holders desire to sell, taken together with all other Pubco Ordinary Shares or other securities which Pubco desires to sell and the Pubco Ordinary Shares or other securities, if any, as to which a registration has been requested pursuant to separate written contractual piggy-back registration rights held by other stockholders of Pubco who desire to sell, exceeds the maximum dollar amount or maximum number of shares that can be sold in such offering without adversely affecting the proposed offering price, the timing, the distribution method, or the probability of success of such offering (such maximum dollar amount or maximum number of shares, as applicable, the “Maximum Number of Shares”), then Pubco shall include in such registration: (i) first, the Registrable Securities as to which Demand Registration has been requested by the Demanding Holders (pro rata in accordance with the number of shares that each such Person has requested be included in such registration, regardless of the number of shares held by each such Person (such proportion is referred to herein as “Pro Rata”)) up to the maximum amount that can be sold without exceeding the Maximum Number of Shares; (ii) second, to the extent that the Maximum Number of Shares has not been reached under the foregoing clause (i), the Pubco Ordinary Shares or other securities that Pubco desires to sell that can be sold without exceeding the Maximum Number of Shares; (iii) third, to the extent that the Maximum Number of Shares has not been reached under the foregoing clauses (i) and (ii), the Pubco Ordinary Shares or other securities for the account of other Persons that Pubco is obligated to register pursuant to then other written contractual arrangements with such Persons and that can be sold without exceeding the Maximum Number of Shares.
| G-5 |
2.1.5 Demand Registration Withdrawal.
(a) If a majority-in-interest of the Demanding Holders disapprove of the terms of any underwriting or are not entitled to include all of their Registrable Securities in any offering, such majority-in-interest of the Demanding Holders may elect to withdraw from such offering by giving written notice to Pubco and the Underwriter or Underwriters of their request to withdraw prior to the effectiveness of the Registration Statement filed with the Commission with respect to such Demand Registration. If the majority-in-interest of the Demanding Holders withdraws from a proposed offering relating to a Demand Registration, then such registration shall not count as a Demand Registration provided for in this Section 2.1. Notwithstanding the forgoing, an Investor may withdraw all or any portion of its Registrable Securities included in a Demand Registration from such Demand Registration at any time prior to the effectiveness of the applicable Registration Statement; provided that such withdrawal shall be irrevocable and, after making such withdrawal, an Investor shall no longer have any right to include Registrable Securities in the Demand Registration as to which such withdrawal was made.
(b) Notwithstanding anything to the contrary in this Agreement, Pubco shall be responsible for the registration expenses described in Section 3.3 incurred in connection with a Registration pursuant to a Demand Registration or an Underwritten Offering prior to its withdrawal under this Section 2.1.5.
2.2 Piggy-Back Registration.
2.2.1 Piggy-Back Rights.
(a) If at any time on or after the Closing, Pubco proposes to file a Registration Statement under the Securities Act with respect to an offering of equity securities, or securities or other obligations exercisable or exchangeable for, or convertible into, equity securities, by Pubco for its own account or for shareholders of Pubco for their account (or by Pubco and by shareholders of Pubco including, without limitation, pursuant to Section 2.1), other than a Registration Statement (i) filed in connection with any employee stock option or other benefit plan, (ii) for an exchange offer or offering of securities solely to Pubco’s existing shareholders, (iii) for an offering of debt that is convertible into equity securities of Pubco, (iv) for a dividend reinvestment plan, (v) that is on Form F-4 (as promulgated under the Securities Act) relating to equity securities to be issued solely in connection with any acquisition of any entity or business or their then equivalents, or (vi) filed relating to the resale of equity securities to be issued under the PIPE Subscription Agreements; provided, however, that the limitation under clause (vi) shall only apply to the first Registration Statement filed by Pubco as required under the PIPE Subscription Agreements, then Pubco shall (x) give written notice of such proposed filing to the holders of Registrable Securities as soon as practicable but in no event less than ten (10) days before the anticipated filing date, which notice shall describe the amount and type of securities to be included in such offering, the intended method(s) of distribution, and the name of the proposed managing Underwriter or Underwriters, if any, of the offering, and (y) offer to the holders of Registrable Securities in such notice the opportunity to register the sale of such number of shares of Registrable Securities as such holders may request in writing within five (5) days following receipt of such notice (a “Piggy-Back Registration”). Pubco shall cause such Registrable Securities to be included in such Piggy-back Registration.
| G-6 |
(b) If at any time on or after the Closing, Pubco proposes to effect an Underwritten Offering for its own account or for the account of stockholders of Pubco (a “Pubco Underwritten Offering”), Pubco shall notify, in writing, all Investors holding Registrable Securities of such demand, and such Investor who thereafter wishes to include all or a portion of such Investor’s Registrable Securities in such Underwritten Offering (each such Investor, a “Pubco Underwritten Shelf Offering Requesting Holder”) shall so notify Pubco, in writing, within five (5) days after the receipt by such Investor of the notice from Pubco. Upon receipt by Pubco of any such written notification, such Pubco Underwritten Shelf Offering Requesting Holder shall be entitled, subject to Sections 2.2.2 and 3.1.1 hereof, to have its Registrable Securities included in such Pubco Underwritten Offering. Pubco shall use its commercially reasonable efforts to cause the managing Underwriter or Underwriters of a proposed Underwritten Offering to permit the Registrable Securities requested to be included in a Piggy-Back Registration on the same terms and conditions as any similar securities of Pubco and to permit the sale or other disposition of such Registrable Securities in accordance with the intended method(s) of distribution thereof. All holders of Registrable Securities proposing to distribute their securities through a Piggy-Back Registration that involves an Underwriter or Underwriters shall enter into an underwriting agreement in customary form with the Underwriter or Underwriters selected for such Piggy-Back Registration; provided, however, that any obligation of any such Investor to indemnify any Person pursuant to any such underwriting agreement shall be several, not joint, among such Investors selling Registrable Securities, and such liability shall be limited to the net amount received by any such Investor from the sale of its Registrable Securities pursuant to such Underwritten Offering, and the relative liability of each such Investor shall be in proportion to such net amounts. Notwithstanding the provisions set forth in the immediately preceding sentences, the right to a Piggy-Back Registration set forth under this Section 2.2.1 with respect to the Registrable Securities shall terminate on the tenth (10th) anniversary of the Closing.
2.2.2 Reduction of Underwritten Offering in Connection with Piggy-Back Registration. If the managing Underwriter or Underwriters for a Piggy-Back Registration that is to be an Underwritten Offering advises Pubco and the holders of Registrable Securities participating in the Underwritten Offering in writing that the dollar amount or number of Ordinary Shares which Pubco desires to sell in such Underwritten Offering, taken together with the Ordinary Shares, if any, as to which inclusion in such Underwritten Offering has been demanded pursuant to separate written contractual arrangements with Persons other than the holders of Registrable Securities hereunder, the Registrable Securities as to which inclusion in such Underwritten Offering has been requested under Section 2.2.1 above, and the Ordinary Shares or other securities, if any, as to which inclusion in such Underwritten Offering has been requested pursuant to separate written contractual Piggy-Back Registration rights of other stockholders of Pubco, exceeds the Maximum Number of Shares, then Pubco shall include in any such registration:
(a) If the Underwritten Offering is undertaken for Pubco’s account: (A) first, the Pubco Ordinary Shares or other equity securities that Pubco desires to sell in such Underwritten Offering that can be sold without exceeding the Maximum Number of Shares; (B) second, to the extent that the Maximum Number of Shares has not been reached under the foregoing clause (A), the Pubco Ordinary Shares or other securities, if any, comprised of Registrable Securities, as to which registration has been requested pursuant to the applicable written contractual piggy-back registration rights of such security holders, Pro Rata, that can be sold without exceeding the Maximum Number of Shares; and (C) third, to the extent that the Maximum Number of Shares has not been reached under the foregoing clauses (A) and (B), the Pubco Ordinary Shares or other securities for the account of other Persons that Pubco is obligated to register pursuant to written contractual piggy-back registration rights with such Persons and that can be sold without exceeding the Maximum Number of Shares;
| G-7 |
(b) If the registration is a “demand” registration undertaken at the demand of Persons other than either the holders of Registrable Securities, (A) first, the Pubco Ordinary Shares or other securities for the account of the demanding Persons and the Pubco Ordinary Shares or other securities comprised of Registrable Securities, Pro Rata, as to which registration has been requested pursuant to the terms hereof, that can be sold without exceeding the Maximum Number of Shares; (B) second, to the extent that the Maximum Number of Shares has not been reached under the foregoing clause (A), the Pubco Ordinary Shares or other securities that Pubco desires to sell that can be sold without exceeding the Maximum Number of Shares; (C) third, to the extent that the Maximum Number of Shares has not been reached under the foregoing clauses (A) and (B), the Pubco Ordinary Shares or other securities for the account of other Persons that Pubco is obligated to register pursuant to written contractual arrangements with such Persons, that can be sold without exceeding the Maximum Number of Shares.
2.2.3 Piggy-Back Registration Withdrawal. Any holder of Registrable Securities may elect to withdraw such holder’s request for inclusion of Registrable Securities in any Piggy-Back Registration by giving written notice to Pubco and the Underwriter(s) (if any) of such request to withdraw prior to the effectiveness of the Registration Statement. Pubco (whether on its own determination or as the result of a withdrawal by Persons making a demand pursuant to separate written contractual obligations) may withdraw a Registration Statement filed with the Commission in connection with a Piggy-back Registration at any time prior to the effectiveness of such Registration Statement. In the case of any Underwritten Offering in connection with any Piggy-back Registration, any participating Investor shall have the right to withdraw their respective Registrable Securities from such Underwritten Offering prior to the pricing of such Underwritten Offering. Notwithstanding anything to the contrary in this Agreement, Pubco shall pay all expenses incurred by the holders of Registrable Securities in connection with such Piggy-Back Registration or Underwritten Offering prior to its withdrawal as provided in Section 3.3.
2.2.4 Unlimited Piggy-back Registration Rights. For purposes of clarity, any Registration or Underwritten Offering effected pursuant to Section 2.2. hereof shall not be counted as a Registration pursuant to a Demand Registration effected under Section 2.1 hereof.
2.3 Resale Shelf Registration Rights.
2.3.1 Registration Statement Covering Resale of Registrable Securities. Pubco shall prepare and file or cause to be prepared and filed with the Commission, no later than ninety (90) days following the Closing Date (the “Filing Deadline”), a Registration Statement for an offering to be made on a continuous basis pursuant to Rule 415 of the Securities Act or any successor thereto registering the resale from time to time by holders of all of the Registrable Securities held by the Holders (the “Resale Shelf Registration Statement”). The Resale Shelf Registration Statement shall be on Form F-3 (or, if Form F-3 is not available to be used by Pubco at such time, on Form F-1 or another appropriate form permitting Registration of such Registrable Securities for resale). If the Resale Shelf Registration Statement is initially filed on Form F-1 and thereafter Pubco becomes eligible to use Form F-3 for secondary sales, Pubco shall, as promptly as practicable, cause such Resale Shelf Registration Statement to be amended, or shall file a new replacement Resale Shelf Registration Statement, such that the Resale Shelf Registration Statement is on Form F-3. Pubco shall use commercially reasonable efforts to cause the Resale Shelf Registration Statement to be declared effective as soon as possible after filing, but in no event later than ninety (90) days following the Filing Deadline (the “Effectiveness Deadline”); provided, however, that the Effectiveness Deadline shall be extended to one hundred and twenty (120) days after the Filing Deadline if the Registration Statement is reviewed by, and receives comments from, the Commission. Notwithstanding the foregoing, Pubco’s obligations to include the Registrable Securities held by a holder in the Resale Shelf Registration Statement are contingent upon such holder furnishing in writing to Pubco such information regarding the holder, the securities of Pubco held by the holder and the intended method of disposition of the Registrable Securities as shall be reasonably requested by Pubco to effect the registration of the Registrable Securities, and the holder’s execution and delivery of such documents in connection with such registration as Pubco may reasonably request that are customary of a selling stockholder in similar situations. Once effective, Pubco shall use commercially reasonable efforts to keep the Resale Shelf Registration Statement and Prospectus included therein continuously effective and to be supplemented and amended to the extent necessary to ensure that such Registration Statement is available or, if not available, to ensure that another Registration Statement is available, under the Securities Act at all times until the earliest of (i) the date on which all Registrable Securities and other securities covered by such Registration Statement have been disposed of in accordance with the intended method(s) of distribution set forth in such Registration Statement and (ii) the date on which all Registrable Securities and other securities covered by such Registration Statement have ceased to be Registrable Securities. The Registration Statement filed with the Commission pursuant to this subsection 2.3.1 shall contain a Prospectus in such form as to permit any holder to sell such Registrable Securities pursuant to Rule 415 under the Securities Act (or any successor or similar provision adopted by the Commission then in effect) at any time beginning on the effective date for such Registration Statement (subject to lock-up restrictions under the Lock-up Agreement and the Release Date under the IPO Escrow Agreement), and shall provide that such Registrable Securities may be sold pursuant to any method or combination of methods legally available to, and requested by, holders of the Registrable Securities.
| G-8 |
2.3.2 Amendments and Supplements. Subject to the provisions of Section 2.3.1 above, Pubco shall promptly prepare and file with the Commission from time to time such amendments and supplements to the Resale Shelf Registration Statement and Prospectus used in connection therewith as may be necessary to keep the Resale Shelf Registration Statement effective and to comply with the provisions of the Securities Act with respect to the disposition of all the Registrable Securities. If any Resale Shelf Registration Statement filed pursuant to Section 2.3.1 is filed on Form F-3 and thereafter Pubco becomes ineligible to use Form F-3 for secondary sales, Pubco shall promptly notify the holders of such ineligibility and use its commercially reasonable efforts to file a shelf registration on an appropriate form as promptly as practicable to replace the shelf registration statement on Form F-3 and have such replacement Resale Shelf Registration Statement declared effective as promptly as practicable and to cause such replacement Resale Shelf Registration Statement to remain effective, and to be supplemented and amended to the extent necessary to ensure that such Resale Shelf Registration Statement is available or, if not available, that another Resale Shelf Registration Statement is available, for the resale of all the Registrable Securities held by the holders until all such Registrable Securities have ceased to be Registrable Securities; provided, however, that at any time Pubco once again becomes eligible to use Form F-3, Pubco shall cause such replacement Resale Shelf Registration Statement to be amended, or shall file a new replacement Resale Shelf Registration Statement, such that the Resale Shelf Registration Statement is once again on Form F-3.
2.3.3 SEC Cutback. Notwithstanding the registration obligations set forth in this Section 2.3, in the event the Commission informs Pubco that all of the Registrable Securities cannot, as a result of the application of Rule 415, be registered for resale as a secondary offering on a single registration statement, Pubco agrees to promptly (i) inform each of the holders thereof and use its commercially reasonable efforts to file amendments to the Resale Shelf Registration Statement as required by the Commission and/or (ii) withdraw the Resale Shelf Registration Statement and file a new registration statement (a “New Registration Statement”) on Form F-3, or if Form F-3 is not then available to Pubco for such registration statement, on such other form available to register for resale the Registrable Securities as a secondary offering; provided, however, that prior to filing such amendment or New Registration Statement, Pubco shall use its commercially reasonable efforts to advocate with the Commission for the registration of all of the Registrable Securities in accordance with any publicly-available written or oral guidance, comments, requirements or requests of the Commission staff (the “SEC Guidance”). Notwithstanding any other provision of this Agreement, if any SEC Guidance sets forth a limitation on the number of Registrable Securities permitted to be registered on a particular Registration Statement as a secondary offering (and notwithstanding that Pubco used diligent efforts to advocate with the Commission for the registration of all or a greater number of Registrable Securities), unless otherwise directed in writing by a holder as to further limit its Registrable Securities to be included on the Registration Statement, the number of Registrable Securities to be registered on such Registration Statement will be reduced Pro Rata among all such selling shareholders whose securities are included in such Registration Statement, subject to a determination by the Commission that certain holders must be reduced first based on the number of Registrable Securities held by such holders. In the event Pubco amends the Resale Shelf Registration Statement or files a New Registration Statement, as the case may be, Pubco will use its commercially reasonable efforts to file with the Commission, as promptly as allowed by Commission or SEC Guidance provided to Pubco or to registrants of securities in general, one or more registration statements on Form F-3 or such other form available to register for resale those Registrable Securities that were not registered for resale on the Resale Shelf Registration Statement, as amended, or the New Registration Statement.
| G-9 |
2.3.4 Underwritten Shelf Takedown. At any time and from time to time after a Resale Shelf Registration Statement has been declared effective by the Commission, the holders of Registrable Securities may request to sell all or any portion of the Registrable Securities in an underwritten offering that is registered pursuant to the Resale Shelf Registration Statement (each, an “Underwritten Shelf Takedown”); provided, however, that Pubco shall only be obligated to effect an Underwritten Shelf Takedown if such offering shall include securities with a total offering price (including securities added to such registration through piggyback registration rights and before deduction of underwriting discounts) reasonably expected to exceed, in the aggregate, $10,000,000. At least fifteen (15) days prior to the public announcement of such Underwritten Shelf Takedown, Pubco shall notify all holders of Registrable Securities that it intends to conduct an Underwritten Shelf Takedown, and all requests for Underwritten Shelf Takedowns shall be made by giving written notice to Pubco at least ten (10) days prior to the public announcement of such Underwritten Shelf Takedown, which shall specify the approximate number of Registrable Securities proposed to be sold in the Underwritten Shelf Takedown and the expected price range (net of underwriting discounts and commissions) of such Underwritten Shelf Takedown. Pubco shall give written notice of such request to all holders of Registrable Securities promptly (but in any event within five (5) days after receipt of such request for an Underwritten Shelf Takedown) and shall include in any Underwritten Shelf Takedown the securities requested to be included by any holder (each a “Takedown Requesting Holder”) at least forty-eight (48) hours prior to the public announcement of such Underwritten Shelf Takedown pursuant to written contractual piggyback registration rights of such holder (including those set forth herein). All such holders proposing to distribute their Registrable Securities through an Underwritten Shelf Takedown under this Section 2.3.4 shall enter into an underwriting agreement in customary form with the Underwriter(s) selected for such Underwritten Offering by the majority-in-interest of the Takedown Requesting Holders initiating the Underwritten Shelf Takedown.
2.3.5 Reduction of Underwritten Shelf Takedown. If the managing Underwriter(s) in an Underwritten Shelf Takedown, in good faith, advise Pubco and the Takedown Requesting Holders in writing that the dollar amount or number of Registrable Securities that the Takedown Requesting Holders desire to sell, taken together with all other Pubco Ordinary Shares or other equity securities that Pubco desires to sell, exceeds the Maximum Number of Shares, then Pubco shall include in such Underwritten Shelf Takedown, as follows: (i) first, the Registrable Securities of the Takedown Requesting Holders, on a Pro Rata basis, that can be sold without exceeding the Maximum Number of Shares; and (ii) second, to the extent that the Maximum Number of Shares has not been reached under the foregoing clause (i), the Pubco Ordinary Shares or other equity securities that Pubco desires to sell, which can be sold without exceeding the Maximum Number of Shares.
2.3.6 Limits on Underwritten Shelf Takedowns. Registrations effected pursuant to this Section 2.3 shall not be counted as Demand Registrations effected pursuant to Section 2.1. Under no circumstances shall Pubco be obligated to effect more than an aggregate of two (2) Underwritten Shelf Takedowns in any 12-month period.
| G-10 |
3. REGISTRATION PROCEDURES.
3.1 Filings; Information. Whenever Pubco is required to effect the registration of any Registrable Securities pursuant to Section 2, Pubco shall use its commercially reasonable efforts to effect the registration and sale of such Registrable Securities in accordance with the intended method(s) of distribution thereof as expeditiously as practicable, and in connection with any such request:
3.1.1 Filing Registration Statement; Restriction on Registration Rights. Pubco shall use its commercially reasonable efforts to, as expeditiously as possible after receipt of a request for a Demand Registration pursuant to Section 2.1, prepare and file with the Commission a Registration Statement on any form for which Pubco then qualifies or which counsel for Pubco shall deem appropriate and which form shall be available for the sale of all Registrable Securities to be registered thereunder in accordance with the intended method(s) of distribution thereof, and shall use its commercially reasonable efforts to cause such Registration Statement to become effective and use its commercially reasonable efforts to keep it effective for the period required by Section 3.1.3; provided, however, that Pubco shall not be obligated to (but may, at its sole option) (a) effect any Demand Registration or an Underwritten Offering or (b) file a Registration Statement (or any amendment thereto) or effect an Underwritten Offering if Pubco has determined in good faith that the sale of Registrable Securities pursuant a Registration Statement would require disclosure of material non-public information not otherwise required to be disclosed under applicable securities laws (i) which disclosure would have a material adverse effect on Pubco or (ii) relating to a material transaction involving Pubco (any such period, a “Blackout Period”); provided, however, that in no event shall any Blackout Period together with other Blackout Periods exceed an aggregate of 90 days in any consecutive 12-month period. Notwithstanding the foregoing, Pubco shall not exercise its rights under this Section 3.1.1 to invoke a Blackout Period unless it applies the same Blackout Period restrictions contained herein to all other securityholders of Pubco with contractual registration rights.
3.1.2 Copies. Pubco shall, prior to filing a Registration Statement or Prospectus, or any amendment or supplement thereto, furnish without charge to the holders of Registrable Securities included in such registration, and such holders’ legal counsel, copies of such Registration Statement as proposed to be filed, each amendment and supplement to such Registration Statement (in each case including all exhibits thereto and documents incorporated by reference therein), the Prospectus included in such Registration Statement, and such other documents as the holders of Registrable Securities included in such registration or legal counsel for any such holders may request in order to facilitate the disposition of the Registrable Securities owned by such holders.
3.1.3 Amendments and Supplements. Pubco shall prepare and file with the Commission such amendments, including post-effective amendments, and supplements to such Registration Statement and the Prospectus used in connection therewith as may be necessary to keep such Registration Statement effective and in compliance with the provisions of the Securities Act until all Registrable Securities and other securities covered by such Registration Statement have been disposed of in accordance with the intended method(s) of distribution set forth in such Registration Statement or such securities have been withdrawn.
| G-11 |
3.1.4 Notification. After the filing of a Registration Statement, Pubco shall promptly, and in no event more than five (5) Business Days after such filing, notify the holders of Registrable Securities included in such Registration Statement of such filing, and shall further notify such holders promptly and confirm such advice in writing in all events within five (5) Business Days of the occurrence of any of the following: (i) when such Registration Statement becomes effective; (ii) when any post-effective amendment to such Registration Statement becomes effective; (iii) the issuance or threatened issuance by the Commission of any stop order (and Pubco shall take all actions required to prevent the entry of such stop order or to remove it if entered); and (iv) any written comments by the Commission or any request by the Commission for any amendment or supplement to such Registration Statement or any Prospectus relating thereto or for additional information or of the occurrence of an event requiring the preparation of a supplement or amendment to such Prospectus so that, as thereafter delivered to the purchasers of the securities covered by such Registration Statement, such Prospectus will not contain an untrue statement of a material fact or omit to state any material fact required to be stated therein or necessary to make the statements therein not misleading, and promptly make available to the holders of Registrable Securities included in such Registration Statement any such supplement or amendment; except that not less than two (2) Business Days before filing with the Commission a Registration Statement or not less than one (1) Business Day before the filing of any related Prospectus or any amendment or supplement thereto, including documents incorporated by reference, Pubco shall (x) furnish to the holders of Registrable Securities included in such Registration Statement and to the legal counsel for any such holders, copies of all such documents proposed to be filed and (y) reasonably cooperate with such holders and their counsel and consider in good faith any comments received by such holders or their counsel with respect to the Registration Statement or Prospectus. Pubco shall not file any Registration Statement or Prospectus or amendment or supplement thereto, including documents incorporated by reference, to which such holders or their legal counsel shall object in good faith, provided that, Pubco is notified of such objection in writing no later than two (2) Business Days after the holders have been so furnished copies of a Registration Statement or one (1) Business Day after the holders have been so furnished copies of any related Prospectus or amendments or supplements thereto.
3.1.5 State Securities Laws Compliance. Pubco shall use its commercially reasonable efforts to (i) register or qualify the Registrable Securities covered by the Registration Statement under such securities or “blue sky” laws of such jurisdictions in the United States as the holders of Registrable Securities included in such Registration Statement (in light of their intended plan of distribution) may request and (ii) take such action necessary to cause such Registrable Securities covered by the Registration Statement to be registered with or approved by such other governmental authorities as may be necessary by virtue of the business and operations of Pubco and do any and all other acts and things that may be necessary or advisable to enable the holders of Registrable Securities included in such Registration Statement to consummate the disposition of such Registrable Securities in such jurisdictions; provided, however, that Pubco shall not be required to qualify generally to do business in any jurisdiction where it would not otherwise be required to qualify but for this paragraph or subject itself to taxation in any such jurisdiction.
3.1.6 Agreements for Disposition. Pubco shall enter into customary agreements (including, if applicable, an underwriting agreement in customary form) and take such other actions as are reasonably required in order to expedite or facilitate the disposition of such Registrable Securities. The representations, warranties and covenants of Pubco in any underwriting agreement which are made to or for the benefit of any Underwriters, to the extent applicable, shall also be made to and for the benefit of the holders of Registrable Securities included in such registration statement. No holder of Registrable Securities included in such registration statement shall be required to make any representations or warranties in the underwriting agreement except, if applicable, with respect to such holder’s organization, good standing, authority, title to Registrable Securities, lack of conflict of such sale with such holder’s material agreements and organizational documents, and with respect to written information relating to such holder that such holder has furnished in writing expressly for inclusion in such Registration Statement.
3.1.7 Cooperation. The principal executive officer of Pubco, the principal financial officer of Pubco, the principal accounting officer of Pubco and all other officers and members of the management of Pubco shall cooperate fully in any offering of Registrable Securities hereunder, which cooperation shall include, without limitation, the preparation of the Registration Statement with respect to such offering and all other offering materials and related documents, and participation in meetings with Underwriters, attorneys, accountants and potential investors.
| G-12 |
3.1.8 Records. Pubco shall make available for inspection by the holders of Registrable Securities included in such Registration Statement, any Underwriter participating in any disposition pursuant to such registration statement and any attorney, accountant or other professional retained by any holder of Registrable Securities included in such Registration Statement or any Underwriter, all financial and other records, pertinent corporate documents and properties of Pubco, as shall be necessary to enable them to exercise their due diligence responsibility, and cause Pubco’s officers, directors and employees to supply all information requested by any of them in connection with such Registration Statement.
3.1.9 Opinions and Comfort Letters. Upon written request, Pubco shall furnish to each holder of Registrable Securities included in any Registration Statement a signed counterpart, addressed to such holder, of (i) any opinion of counsel to Pubco delivered to any Underwriter and (ii) any comfort letter from Pubco’s independent public accountants delivered to any Underwriter. In the event no legal opinion is delivered to any Underwriter, Pubco shall furnish to each holder of Registrable Securities included in such Registration Statement, at any time that such holder elects to use a prospectus, an opinion of counsel to Pubco to the effect that the Registration Statement containing such prospectus has been declared effective and that no stop order is in effect.
3.1.10 Earnings Statement. Pubco shall comply with all applicable rules and regulations of the Commission and the Securities Act, and make available to its shareholders, as soon as practicable, an earnings statement covering a period of twelve (12) months, which earnings statement shall satisfy the provisions of Section 11(a) of the Securities Act and Rule 158 thereunder.
3.1.11 Listing. Pubco shall use its commercially reasonable efforts to cause all Registrable Securities included in any registration to be listed on such exchanges or otherwise designated for trading in the same manner as similar securities issued by Pubco are then listed or designated or, if no such similar securities are then listed or designated, in a manner satisfactory to the holders of a majority of the Registrable Securities included in such registration.
3.1.12 Road Show. If the registration involves the registration of Registrable Securities involving gross proceeds in excess of $25,000,000, Pubco shall use its reasonable efforts to make available senior executives of Pubco to participate in customary “road show” presentations that may be reasonably requested by the Underwriter in any Underwritten Offering.
3.1.13 Regulation M. Pubco shall take no direct or indirect action prohibited by Regulation M under the Exchange Act; provided, that, to the extent that any prohibition is applicable to Pubco, Pubco will take all reasonable action to make any such prohibition inapplicable.
3.2 Obligation to Suspend Distribution. Upon receipt of any notice from Pubco of the happening of any event of the kind described in Section 3.1.4(iv), or, in the case of a resale registration on Form F-3 pursuant to Section 2.3 hereof, upon any suspension by Pubco, pursuant to a written insider trading compliance program adopted by Pubco’s Board of Directors, of the ability of all “insiders” covered by such program to transact in Pubco’s securities because of the existence of material non-public information, each holder of Registrable Securities included in any registration shall immediately discontinue disposition of such Registrable Securities pursuant to the Registration Statement covering such Registrable Securities until such holder receives the supplemented or amended Prospectus contemplated by Section 3.1.4(iv) or the restriction on the ability of “insiders” to transact in Pubco’s securities is removed, as applicable, and, if so directed by Pubco, each such holder will deliver to Pubco all copies, other than permanent file copies then in such holder’s possession, of the most recent Prospectus covering such Registrable Securities at the time of receipt of such notice.
| G-13 |
3.3 Registration Expenses. Pubco shall bear all costs and expenses incurred in connection with any Demand Registration pursuant to Section 2.1, any Piggy-Back Registration pursuant to Section 2.2, and any registration on Form F-3 effected pursuant to Section 2.3, and all expenses incurred in performing or complying with its other obligations under this Agreement, whether or not the Registration Statement becomes effective, including, without limitation: (i) all registration and filing fees; (ii) fees and expenses of compliance with securities or “blue sky” laws (including fees and disbursements of counsel in connection with blue sky qualifications of the Registrable Securities); (iii) printing expenses; (iv) Pubco’s internal expenses (including, without limitation, all salaries and expenses of its officers and employees); (v) the fees and expenses incurred in connection with the listing of the Registrable Securities as required by Section 3.1.11; (vi) Financial Industry Regulatory Authority fees; (vii) fees and disbursements of counsel for Pubco and fees and expenses for independent certified public accountants retained by Pubco (including the expenses or costs associated with the delivery of any opinions or comfort letters requested pursuant to Section 3.1.9); (viii) the reasonable fees and expenses of any special experts retained by Pubco in connection with such registration; and (ix) the reasonable fees and expenses of one legal counsel selected by the holders of a majority-in-interest of the Registrable Securities included in such registration in an amount not to exceed $25,000. Pubco shall have no obligation to pay any underwriting discounts or selling commissions attributable to the Registrable Securities being sold by the holders thereof or any fees and disbursements of its counsel in excess of $25,000 in the aggregate in connection therewith, which underwriting discounts or selling commissions and fees and disbursements of its counsel in excess of $25,000 in the aggregate shall be borne by such holders. Additionally, in an Underwritten Offering, all selling shareholders and Pubco shall bear the expenses of the Underwriter pro rata in proportion to the respective amount of shares each is selling in such offering.
3.4 Holders’ Information. The holders of Registrable Securities shall provide such information as may reasonably be requested by Pubco, or the managing Underwriter, if any, in connection with the preparation of any Registration Statement, including amendments and supplements thereto, in order to effect the registration of any Registrable Securities under the Securities Act pursuant to Section 2 and in connection with Pubco’s obligation to comply with Federal and applicable state securities laws. Pubco’s obligations to include the Registrable Securities in any Registration Statement under this Agreement are contingent upon each holder of Registrable Securities furnishing in writing to Pubco such information regarding such holder, the securities of Pubco held by holder and the intended method of disposition of the Registrable Securities as shall be reasonably requested by Pubco to effect the registration of the Registrable Securities, and such holder shall execute such documents in connection with such registration as Pubco may reasonably request that are customary of a selling stockholder in similar situations.
4. INDEMNIFICATION AND CONTRIBUTION.
4.1 Indemnification by Pubco. Pubco agrees to indemnify and hold harmless each Investor and each other holder of Registrable Securities, and each of their respective officers, employees, affiliates, directors, partners, members, attorneys and agents, and each Person, if any, who controls an Investor and each other holder of Registrable Securities (within the meaning of Section 15 of the Securities Act or Section 20 of the Exchange Act) (each, an “Investor Indemnified Party”), from and against any expenses, losses, judgments, claims, damages or liabilities, whether joint or several, arising out of or based upon any untrue statement (or allegedly untrue statement) of a material fact contained in (or incorporated by reference in) any Registration Statement under which the sale of such Registrable Securities was registered under the Securities Act, any Prospectus contained in the Registration Statement, or free writing prospectus (as defined in Rule 405 under the Securities Act or any successor rule thereto), or any amendment or supplement to such Registration Statement, or any filing under any state securities law required to be filed or furnished, or arising out of or based upon any omission (or alleged omission) to state a material fact required to be stated therein or necessary to make the statements therein not misleading, or any violation by Pubco of the Securities Act or any rule or regulation promulgated thereunder applicable to Pubco and relating to action or inaction required of Pubco in connection with any such registration; and Pubco shall promptly reimburse the Investor Indemnified Party for any legal and any other expenses reasonably incurred by such Investor Indemnified Party in connection with investigating and defending any such expense, loss, judgment, claim, damage, liability or action; provided, however, that Pubco will not be liable in any such case to the extent that any such expense, loss, claim, damage or liability arises out of or is based upon any untrue statement or allegedly untrue statement or omission or alleged omission made in such Registration Statement, Prospectus, or free writing prospectus, or any such amendment or supplement, in reliance upon and in conformity with information furnished to Pubco, in writing, by such selling holder expressly for use therein, and shall reimburse Pubco, its directors and officers, and each other selling holder or controlling Person for any legal or other expenses reasonably incurred by any of them in connection with investigation or defending any such loss, claim, damage, liability or action. Pubco also shall indemnify any Underwriter of the Registrable Securities, their officers, affiliates, directors, partners, members and agents and each Person who controls such Underwriter (within the meaning of the Securities Act or the Exchange Act, as applicable) on substantially the same basis as that of the indemnification provided above in this Section 4.1.
| G-14 |
4.2 Indemnification by Holders of Registrable Securities. Each selling holder of Registrable Securities will, in the event that any registration is being effected under the Securities Act pursuant to this Agreement of any Registrable Securities held by such selling holder, indemnify and hold harmless Pubco, each of its directors, officers, agents and employees, each Person, if any, who controls Pubco (within the meaning of Section 15 of the Securities Act and Section 20 of the Exchange Act), each Underwriter (if any), and each other selling holder and each other Person, if any, who controls another selling holder or such Underwriter within the meaning of the Securities Act, and the directors, officers, agents or employees of such controlling Persons, to the fullest extent permitted by applicable law, against any losses, claims, judgments, damages or liabilities, whether joint or several, insofar as such losses, claims, judgments, damages or liabilities (or actions in respect thereof) (including, without limitation, reasonable attorneys’ fees and other expenses) arise out of or are based upon any untrue statement or allegedly untrue statement of a material fact contained in any Registration Statement under which the sale of such Registrable Securities was registered under the Securities Act, any Prospectus contained in the Registration Statement, or any amendment or supplement to the Registration Statement, or arise out of or are based upon any omission or the alleged omission to state a material fact required to be stated therein or necessary to make the statement therein not misleading, if the statement or omission was made in reliance upon and in conformity with information furnished in writing to Pubco by such selling holder expressly for use therein, and shall reimburse Pubco, its directors and officers, and each other selling holder or controlling Person for any legal or other expenses reasonably incurred by any of them in connection with investigation or defending any such loss, claim, damage, liability or action. Each selling holder’s indemnification obligations hereunder shall be several and not joint and shall be limited to the amount of any net proceeds (after payment of any underwriting fees, discounts, commissions or taxes) actually received by such selling holder.
4.3 Conduct of Indemnification Proceedings. Promptly after receipt by any Person of any notice of any loss, claim, damage or liability or any action in respect of which indemnity may be sought pursuant to Section 4.1 or 4.2, such Person (the “Indemnified Party”) shall, if a claim in respect thereof is to be made against any other Person for indemnification hereunder, notify such other Person (the “Indemnifying Party”) in writing of the loss, claim, judgment, damage, liability or action; provided, however, that the failure by the Indemnified Party to notify the Indemnifying Party shall not relieve the Indemnifying Party from any liability which the Indemnifying Party may have to such Indemnified Party hereunder, except and solely to the extent the Indemnifying Party is actually prejudiced by such failure. If the Indemnified Party is seeking indemnification with respect to any claim or action brought against the Indemnified Party, then the Indemnifying Party shall be entitled to participate in such claim or action, and, to the extent that it wishes, jointly with all other Indemnifying Parties, to assume control of the defense thereof with counsel satisfactory to the Indemnified Party. After notice from the Indemnifying Party to the Indemnified Party of its election to assume control of the defense of such claim or action, the Indemnifying Party shall not be liable to the Indemnified Party for any legal or other expenses subsequently incurred by the Indemnified Party in connection with the defense thereof other than reasonable costs of investigation; provided, however, that in any action in which both the Indemnified Party and the Indemnifying Party are named as defendants, the Indemnified Party shall have the right to employ separate counsel (but no more than one such separate counsel) to represent the Indemnified Party and its controlling Persons who may be subject to liability arising out of any claim in respect of which indemnity may be sought by the Indemnified Party against the Indemnifying Party, with the fees and expenses of such counsel to be paid by such Indemnifying Party if, based upon the written opinion of counsel of such Indemnified Party, representation of both parties by the same counsel would be inappropriate due to actual or potential differing interests between them. No Indemnifying Party shall, without the prior written consent of the Indemnified Party, consent to entry of judgment or effect any settlement of any claim or pending or threatened proceeding in respect of which the Indemnified Party is or could have been a party and indemnity could have been sought hereunder by such Indemnified Party, unless such judgment or settlement includes an unconditional release of such Indemnified Party from all liability arising out of such claim or proceeding.
| G-15 |
4.4 Contribution.
4.4.1 If the indemnification provided for in the foregoing Sections 4.1, 4.2 and 4.3 is unavailable to any Indemnified Party in respect of any loss, claim, damage, liability or action referred to herein, then each such Indemnifying Party, in lieu of indemnifying such Indemnified Party, shall contribute to the amount paid or payable by such Indemnified Party as a result of such loss, claim, damage, liability or action in such proportion as is appropriate to reflect the relative fault of the Indemnified Parties and the Indemnifying Parties in connection with the actions or omissions which resulted in such loss, claim, damage, liability or action, as well as any other relevant equitable considerations. The relative fault of any Indemnified Party and any Indemnifying Party shall be determined by reference to, among other things, whether the untrue or alleged untrue statement of a material fact or the omission or alleged omission to state a material fact relates to information supplied by such Indemnified Party or such Indemnifying Party and the parties’ relative intent, knowledge, access to information and opportunity to correct or prevent such statement or omission.
4.4.2 The parties hereto agree that it would not be just and equitable if contribution pursuant to this Section 4.4 were determined by pro rata allocation or by any other method of allocation which does not take account of the equitable considerations referred to in the immediately preceding Section 4.4.1.
4.4.3 The amount paid or payable by an Indemnified Party as a result of any loss, claim, damage, liability or action referred to in the immediately preceding paragraph shall be deemed to include, subject to the limitations set forth above, any legal or other expenses incurred by such Indemnified Party in connection with investigating or defending any such action or claim. Notwithstanding the provisions of this Section 4.4, no holder of Registrable Securities shall be required to contribute any amount in excess of the dollar amount of the net proceeds (after payment of any underwriting fees, discounts, commissions or taxes) actually received by such selling holder from the sale of Registrable Securities which gave rise to such contribution obligation. No Person guilty of fraudulent misrepresentation (within the meaning of Section 11(f) of the Securities Act) shall be entitled to contribution from any Person who was not guilty of such fraudulent misrepresentation.
5. RULE 144.
5.1 Rule 144. Pubco covenants that it shall file any reports required to be filed by it under the Securities Act and the Exchange Act and shall take such further action as the holders of Registrable Securities may reasonably request, all to the extent required from time to time to enable such holders to sell Registrable Securities without registration under the Securities Act within the limitation of the exemptions provided by Rule 144 under the Securities Act, as such Rules may be amended from time to time, or any similar rule or regulation hereafter adopted by the Commission.
| G-16 |
6. MISCELLANEOUS.
6.1 Other Registration Rights. Pubco represents and warrants that, except for registration rights granted to the investors pursuant to the PIPE Subscription Agreements, no Person, other than the holders of the Registrable Securities, has any right to require Pubco to register any of Pubco’s share capital for sale or to include Pubco’s share capital in any registration filed by Pubco for the sale of share capital for its own account or for the account of any other Person. The Investors hereby acknowledge that Pubco has granted resale registration rights to the purchasers of Pubco’s securities in the PIPE Subscription Agreements, and that nothing herein shall restrict the ability of Pubco to fulfill its resale registration obligations under the PIPE Subscription Agreements.
6.2 Assignment; No Third Party Beneficiaries. This Agreement and the rights, duties and obligations of Pubco hereunder may not be assigned or delegated by Pubco in whole or in part. This Agreement and the rights, duties and obligations of the holders of Registrable Securities hereunder may be freely assigned or delegated by such holder of Registrable Securities in conjunction with and to the extent of any legally permitted transfer of Registrable Securities by any such holder (subject to lock-up restrictions under the Lock-up Agreement and the Release Date under the IPO Escrow Agreement). This Agreement and the provisions hereof shall be binding upon and shall inure to the benefit of each of the parties, to the permitted assigns of the Investors or holder of Registrable Securities or of any assignee of the Investors or holder of Registrable Securities. This Agreement is not intended to confer any rights or benefits on any Persons that are not party hereto other than as expressly set forth in this. Section 6.2.
6.3 Notices. Any notice hereunder shall be sent in writing, addressed as specified below, and shall be deemed given: (a) if by hand or nationally recognized overnight courier service, by 5:00 PM Eastern Time on a Business Day, addressee’s day and time, on the date of delivery, and if delivered after 5:00 PM Eastern Time, on the first Business Day after such delivery; (b) if by email, on the date of transmission with affirmative confirmation of receipt; or (c) three (3) Business Days after mailing by prepaid certified or registered mail, return receipt requested. Notices shall be addressed to the respective parties as follows (excluding telephone numbers, which are for convenience only), or to such other address as a party shall specify to the others in accordance with these notice provisions:
If to Pubco, to:
Alps Global Holding Pubco
Unit E-18-01 & E-18-02 Level 18
Icon Tower (East)
No. 1 Jalan 1/68F, Jalan Tun Razak
50400 Kuala Lumpur
Wilayah Persekutuan, Malaysia
Attn:
Dr. Tham Seng Kong
E-mail: drtham@alpsmedical.com
If to an Investor, to the address set forth below such Investor’s name on Exhibit A hereto.
| G-17 |
6.4 Severability. This Agreement shall be deemed severable, and the invalidity or unenforceability of any term or provision hereof shall not affect the validity or enforceability of this Agreement or of any other term or provision hereof. Furthermore, in lieu of any such invalid or unenforceable term or provision, the parties hereto intend that there shall be added as a part of this Agreement a provision as similar in terms to such invalid or unenforceable provision as may be possible that is valid and enforceable.
6.5 Counterparts. This Agreement may be executed in multiple counterparts, each of which shall be deemed an original, and all of which taken together shall constitute one and the same instrument. Delivery of a signed counterpart of this Agreement by facsimile or email/pdf transmission shall constitute valid and sufficient delivery thereof.
6.6 Entire Agreement. This Agreement (including all agreements entered into pursuant hereto and all certificates and instruments delivered pursuant hereto and thereto) constitute the entire agreement of the parties with respect to the subject matter hereof and supersede all prior and contemporaneous agreements, representations, understandings, negotiations and discussions between the parties, whether oral or written; provided, that, for the avoidance of doubt, the foregoing shall not affect the rights and obligations of the parties under the Merger Agreement or any Additional Agreement. Without limiting the foregoing, the Pre-BC Investors hereby acknowledge and agree that this Agreement amends and restates and supersedes the Original Registration Rights Agreement in its entirety.
6.7 Modifications and Amendments. Any term of this Agreement may be amended or modified with the written consent of Pubco and the holders of a majority of the Registrable Securities then outstanding; provided that no such amendment or modification may affect any Investor in a manner material and disproportionately adverse to other Investors without the prior written consent of such Investor.
6.8 Titles and Headings; Interpretation. Titles and headings of sections of this Agreement are for convenience only and shall not affect the construction of any provision of this Agreement. In this Agreement, unless the context otherwise requires: (i) any pronoun used in this Agreement shall include the corresponding masculine, feminine or neuter forms, and the singular form of nouns, pronouns and verbs shall include the plural and vice versa; (ii) “including” (and with correlative meaning “include”) means including without limiting the generality of any description preceding or succeeding such term and shall be deemed in each case to be followed by the words “without limitation”; (iii) the words “herein,” “hereto,” and “hereby” and other words of similar import in this Agreement shall be deemed in each case to refer to this Agreement as a whole and not to any particular section or other subdivision of this Agreement; and (iv) the term “or” means “and/or”. The parties have participated jointly in the negotiation and drafting of this Agreement. Consequently, in the event an ambiguity or question of intent or interpretation arises, this Agreement shall be construed as if drafted jointly by the parties hereto, and no presumption or burden of proof shall arise favoring or disfavoring any party by virtue of the authorship of any provision of this Agreement.
6.9 Waivers and Extensions. Any party to this Agreement may waive any right, breach or default which such party has the right to waive, provided that such waiver will not be effective against the waiving party unless it is in writing, is signed by such party, and specifically refers to this Agreement. Waivers may be made in advance or after the right waived has arisen or the breach or default waived has occurred. Any waiver may be conditional. No waiver of any breach of any agreement or provision herein contained shall be deemed a waiver of any preceding or succeeding breach thereof nor of any other agreement or provision herein contained. No waiver or extension of time for performance of any obligations or acts shall be deemed a waiver or extension of the time for performance of any other obligations or acts.
| G-18 |
6.10 Remedies Cumulative. In the event that Pubco fails to observe or perform any covenant or agreement to be observed or performed under this Agreement, the Investor or any other holder of Registrable Securities may proceed to protect and enforce its rights by suit in equity or action at law, whether for specific performance of any term contained in this Agreement or for an injunction against the breach of any such term or in aid of the exercise of any power granted in this Agreement or to enforce any other legal or equitable right, or to take any one or more of such actions, without being required to post a bond. None of the rights, powers or remedies conferred under this Agreement shall be mutually exclusive, and each such right, power or remedy shall be cumulative and in addition to any other right, power or remedy, whether conferred by this Agreement or now or hereafter available at law, in equity, by statute or otherwise.
6.11 Governing Law. This Agreement shall be governed by, interpreted under, and construed in accordance with the internal laws of the State of Delaware applicable to agreements made, without giving effect to any choice-of-law provisions thereof that would compel the application of the substantive laws of any other jurisdiction.
6.12 Consent to Jurisdiction; Waiver of Trial by Jury. The parties hereto agree to submit any matter or dispute resulting from or arising out of the execution, performance, interpretation, breach or termination of this Agreement to the exclusive jurisdiction of the Chancery Court of the State of Delaware (or, if the Chancery Court of the State of Delaware does not have jurisdiction, a federal court sitting in Wilmington, Delaware) (or any appellate courts thereof). Each of the parties agrees that service of any process, summons, notice or document in the manner set forth in Section 6.3 hereof or in such other manner as may be permitted by applicable law, shall be effective service of process for any proceeding in the State of New York with respect to any matters to which it has submitted to jurisdiction in this Section 6.12. Each party hereby irrevocably and unconditionally submits, for itself and its property, to the exclusive jurisdiction of the Chancery Court of the State of Delaware (or, if the Chancery Court of the State of Delaware does not have jurisdiction, a federal court sitting in Wilmington, Delaware) (and any appellate courts thereof) in any action or proceeding arising out of or relating to this Agreement, and each of the parties hereby irrevocably and unconditionally (a) agrees not to commence any such action or proceeding except in such courts, (b) agrees that any claim in respect of any such action or proceeding may be heard and determined in such court, (c) waives, to the fullest extent it may legally and effectively do so, any objection which it may now or hereafter have to the laying of venue of any such action or proceeding in any such court, and (d) waives, to the fullest extent permitted by law, the defense of an inconvenient forum to the maintenance of such action or proceeding in any such court. Each party agrees that a final judgment in any such action or proceeding shall be conclusive and may be enforced in other jurisdictions by suit on the judgment or in any other manner provided by law.
6.13 Termination of Merger Agreement. This Agreement shall be binding upon each party upon such party’s execution and delivery of this Agreement, but this Agreement shall only become effective upon the Closing. In the event that the Merger Agreement is validly terminated in accordance with its terms prior to the Closing, this Agreement shall automatically terminate and become null and void and be of no further force or effect, and the parties shall have no obligations hereunder.
6.14 Term. This Agreement shall terminate upon the earlier of (i) the fifth anniversary of the date of this Agreement or, (ii) on a holder of Registrable Securities-by-holder of Registrable Securities basis, on the date as of which (A) all of the Registrable Securities held by such holder have been sold pursuant to a Registration Statement (but in no event prior to the applicable period referred to in Section 4(a)(3) of the Securities Act and Rule 174 thereunder (or any successor rule promulgated thereafter by the Commission)) or (B) such holder of Registrable Securities is permitted to sell all of its Registrable Securities under Rule 144 (or any similar provision) under the Securities Act without limitation on the amount of securities sold or the manner of sale.
[SIGNATURE
PAGE FOLLOWS]
| G-19 |
IN WITNESS WHEREOF, the parties have caused this Registration Rights Agreement to be executed and delivered by their duly authorized representatives as of the date first written above.
| PUBCO: | ||
| Alps Global Holding Pubco | ||
| By: | ||
| Name: | ||
| Title: | ||
| PRE-BC INVESTORS: | ||
| GL Sponsor LLC | ||
| By: | ||
| Name: | ||
| Title: | ||
| Public Gold Marketing Sdn. Bhd | ||
| By: | ||
| Name: | ||
| Title: | ||
| By: | ||
| Name: | Kelvin (Zeng Yenn) Chin | |
| By: | ||
| Name: | Hui Liang Wong | |
| By: | ||
| Name: | Say Leong Lim | |
| By: | ||
| Name: | Hong Shien Beh | |
| By: | ||
| Name: | Kian Huat Lai | |
| ALPS HOLDCO SHAREHOLDER: | ||
| Name: | ||
| Title: | ||
[Signature Page to Amended and Restated Registration Rights Agreement]
| G-20 |
EXHIBIT A
Name and Address of Pre-BC Investors
| Name of Investor | Address | |
| Public Gold Marketing Sdn. Bhd | Plot 21, Technoplex, Medan Bayan Lepas, Taman Perindustrian Bayan Lepas, Phase IV, 11900 Penang | |
| GL Sponsor LLC | 1180 Avenue of the Americas, 8th Floor New York, NY 10036 | |
| Say Leong Lim | 200 Continental Drive, Suite 401, Newark, Delaware, NY 19713. | |
| Kelvin (Zeng Yenn) Chin | 200 Continental Drive, Suite 401, Newark, Delaware, NY 19713. | |
| Hong Shien Beh | 200 Continental Drive, Suite 401, Newark, Delaware, NY 19713. | |
| Kian Huat Lai | 200 Continental Drive, Suite 401, Newark, Delaware, NY 19713. | |
| Hui Liang Wong | 200 Continental Drive, Suite 401, Newark, Delaware, NY 19713. |
| G-21 |
EXHIBIT B
Alps Holdco Shareholders
| 1. | Affluxury Berhad |
| 2. | Agnes Wong Ling Lee |
| 3. | Alphacap Berhad |
| 4. | Ang Chai Kian |
| 5. | Ang Hee Yeou |
| 6. | Antara Emas Sdn. Bhd. |
| 7. | Ashley Wong Po Yin |
| 8. | Aslan Bacho |
| 9. | Azizul Bin Abdul Rahman |
| 10. | Ben Iskandar Bin Mohd. Razef |
| 11. | Benjamin George |
| 12. | Brian Henry Scott |
| 13. | Chair Ngar Kieng |
| 14. | Chan Kuo Wei |
| 15. | Chan Ten Weng |
| 16. | Chan Yoke Ying |
| 17. | Chang Chun Hee |
| 18. | Chang Hao Ker |
| 19. | Chang Woei Jou |
| 20. | Che Rahim Bin Che Daud |
| 21. | Chee Tuck Weng |
| 22. | Chen Lai Koon |
| 23. | Chew Chin Hoong |
| 24. | Chew Seow Boi |
| 25. | Chew Soon Keong |
| 26. | Chew Thiar Loon @Chew Thiang Soon |
| 27. | Chew Yoke Ling |
| 28. | Chia Kok Khaw |
| 29. | Chiang, Ruei-Hong |
| 30. | Chiew June Heng |
| 31. | Chok Kwee Wah |
| 32. | Chong Kok Liang |
| 33. | Chou Hai Cheng |
| 34. | Christie Tan Poh Chu |
| 35. | Chua Boon Ping |
| 36. | Chua Giat Lee |
| 37. | Chua Su Chong |
| 38. | Chua Swee Peng |
| 39. | Chua Boon Yi |
| 40. | Chua Boon Chun |
| 41. | Crystal Propel Sdn. Bhd. |
| 42. | CIMB Islamic Trustee Berhad as the executor and trustee for the estate of Dato’ Yusli Bin Mohamed Yusoff |
| 43. | Daulah Holding Sdn. Bhd. |
| 44. | Deltacap Sdn. Bhd. |
| 45. | Ding Ke Xiang |
| 46. | Dr Barath A/L Malla Naidu |
| G-22 |
| 47. | Dr. Ng Chin Leong |
| 48. | Dr. Wan Sazrina Binti Wan Zaid |
| 49. | Dr. Wong Jeh Shyan |
| 50. | Dr. Tham Seng Kong |
| 51. | Engku Nur Zakiah Binti Syed Mohamed |
| 52. | Eugene Lee |
| 53. | Faizal Bin Jamat |
| 54. | Faridah Hanim Binti Haron |
| 55. | Fatanah Ventures Berhad |
| 56. | Fong Wern Chern |
| 57. | Fovty Tech Sdn. Bhd. |
| 58. | Gan Kian Chong |
| 59. | Gao, Yuan |
| 60. | Germaine Phoon Synn Munn |
| 61. | Goh Jasmine |
| 62. | Goh Jeffrey |
| 63. | Hayati Binti Ismail |
| 64. | Hazimah Binti Malek |
| 65. | Heng Lek Nee |
| 66. | Heng Yue Yue |
| 67. | Hew Pik Wai |
| 68. | Ho Kian Hui |
| 69. | Hon Joe Hang |
| 70. | Hui Wai Kwok |
| 71. | IPO House Berhad |
| 72. | Irewards Global Sdn. Bhd. |
| 73. | Jamal Abdul Nasser Bin Zakaria |
| 74. | Jashida Binti Mohamed Kamal |
| 75. | Jescelynn Wong Fung Yee |
| 76. | Joey Lee |
| 77. | Khong Lian Kiew |
| 78. | Khoo Kiat Beng |
| 79. | Khoo Shuh Mey |
| 80. | Khor Chin Poey |
| 81. | Koay Ee Boon |
| 82. | Koh Kuan Yew |
| 83. | Kok Weng Siong |
| 84. | Kong Siew Hoong |
| 85. | Kua Siau Ying |
| 86. | Lai Jiunn Wen |
| 87. | Lam Hui Sim |
| 88. | Lau Moo Leong |
| 89. | Lau Yung Liem |
| 90. | Lee Chai Ling |
| G-23 |
| 91. | Lee Jou Yee |
| 92. | Lee Yuk Choi |
| 93. | Leong Kon Ming |
| 94. | Leong Nelson |
| 95. | Li, Cui |
| 96. | Lian Ooi Loon |
| 97. | Liang Yoke Ling |
| 98. | Lim Hong Khin |
| 99. | Lim Huai Guo |
| 100. | Lim Kuang Sia |
| 101. | Lim Lai Ping |
| 102. | Lim Ooi Chow |
| 103. | Lim Seok Ping |
| 104. | Lim Siow Wee |
| 105. | Lim Tian Noi |
| 106. | Lim Wai Hong |
| 107. | Lim Wai Kuok |
| 108. | Lin Fook Sing |
| 109. | Lo Yiing Shin |
| 110. | Loi Kheng Seong |
| 111. | Low Lai Chu |
| 112. | Low Lai Keng |
| 113. | Low Lai Youn |
| 114. | Low Wei Sim |
| 115. | Low Yock Chen |
| 116. | Ma Jing |
| 117. | MOC Capital Berhad |
| 118. | Mohamad Sabri Bin Mohd Shaffi |
| 119. | Mohd Razef Bin Abdullah |
| 120. | Mok See Zhuang |
| 121. | Mok Yim Yin |
| 122. | Mong Li Shun |
| 123. | Mong Yong Shin |
| 124. | Muhammad Hanif Bin Azmi |
| 125. | Nah Yoke Eng |
| 126. | Nazar Bin Mohd Idrus |
| 127. | Nazran Bin Nazar |
| 128. | Neo Khean Lee |
| 129. | Neo Kian Huat |
| 130. | Ng Leong Bee |
| 131. | Ng See Kee |
| 132. | Ngo Kee Fook |
| 133. | Noor Zamani Binti Baharuddin |
| 134. | Noorhaniza Binti Abdullah |
| 135. | Norshida Binti Ali |
| G-24 |
| 136. | Nur Dayana Binti Azman Wong |
| 137. | Onetwo Choose Sdn. Bhd. |
| 138. | Ong Kean Leong |
| 139. | Ong Moh Ching |
| 140. | Ong Tee Keat |
| 141. | Ong Yeow Hwee David |
| 142. | Oo Lin Thai |
| 143. | Pang Mei Fong |
| 144. | Phan Thi Ngoc Bich |
| 145. | Phoo Mun Chan @ Phoon Mun Chan |
| 146. | Phoon Khai Foh |
| 147. | Phoon Ngah Wan |
| 148. | Poh Kok Keong |
| 149. | Poon Kian Huat |
| 150. | Precession Capital Berhad |
| 151. | Putri Zharifa Binti Amdun |
| 152. | Qin Lihui |
| 153. | Razim Faris Bin Malek @ Othman |
| 154. | Redzuan Bin Abd Aziz |
| 155. | Roselfida Salmah Binti Daud |
| 156. | Saw Sang Loong |
| 157. | Say Choong Seng @ Chia Choong Seng |
| 158. | Seu Ting Wai |
| 159. | Shahirul Basir Bin Mohamed Sultan |
| 160. | Sinoman International Capital Berhad |
| 161. | Siti Maria Binti Juwaini |
| 162. | Siti Nooruhani Binti Ibrahim |
| 163. | So Bee Hwe |
| 164. | So Jing Theng |
| 165. | Solo Labeller Technology Sdn. Bhd. |
| 166. | Soon Siew Yin |
| 167. | Tai Chee Chong |
| 168. | Tan Ai Hong |
| 169. | Tan Chong Hwa |
| 170. | Tan Chor Lee |
| 171. | Tan Hui Lian |
| 172. | Tan Keat Ying |
| 173. | Tan Mee Su |
| 174. | Tan Weoi Tar |
| 175. | Tan Yen Lean |
| 176. | Tan Yu Yun (Chen Yuying) |
| 177. | Tay Hong Son |
| 178. | Tay Hwee Leck |
| 179. | Tay May Chan |
| 180. | Tay Tong Kui |
| 181. | Tee Siew Poh |
| 182. | Teh Kok Hui |
| 183. | Teh Kok Kuan |
| 184. | Teh Swee Bee |
| 185. | Teh Yen Peng |
| G-25 |
| 186. | Teh Yen Shing |
| 187. | Teng Seng Wen |
| 188. | Tham Chong Wai |
| 189. | Tham Yang Xi |
| 190. | Tiew Tze Tong |
| 191. | Ting Ju Teen |
| 192. | Uf Engineering Supply Sdn. Bhd. |
| 193. | Venture Equity Berhad |
| 194. | Wan Sazril Bin Wan Zaid |
| 195. | Wan Sazriz Bin Wan Zaid |
| 196. | Wan Sazrudeen Bin Wan Zaid |
| 197. | Wealthpier Berhad |
| 198. | Wong Chau |
| 199. | Wong E Zung |
| 200. | Wong Fuat Jun |
| 201. | Wong Man Ling Brenda |
| 202. | Wong Sai Mei |
| 203. | Wong Sim Kuan |
| 204. | Wong Sui Ting |
| 205. | Wong Yeow Men |
| 206. | Wong Yin Hui |
| 207. | Wong Yu Han |
| 208. | Yang Loong Marketing Sdn. Bhd. |
| 209. | Yap Mou Sing |
| 210. | Yap Poh Lan |
| 211. | Yap Tek Sang |
| 212. | Yap Yee Men |
| 213. | Ye Yongle |
| 214. | Yee Yun Jek |
| 215. | Zhang, Yongming |
| 216. | Zuo Xia Lin |
| G-26 |
ANNEX H
PROXY CARD
H-1 |
ANNEX J

| J-1 |

| J-2 |

| J-3 |

| J-4 |

| J-5 |

| J-6 |

| J-7 |

| J-8 |

| J-9 |

| J-10 |
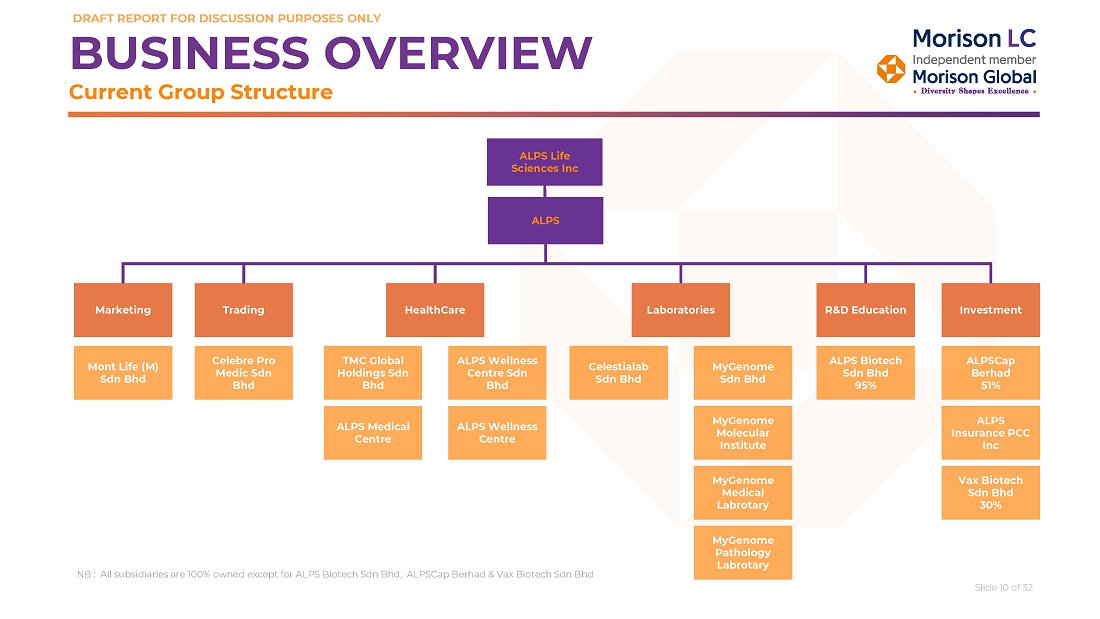
| J-11 |
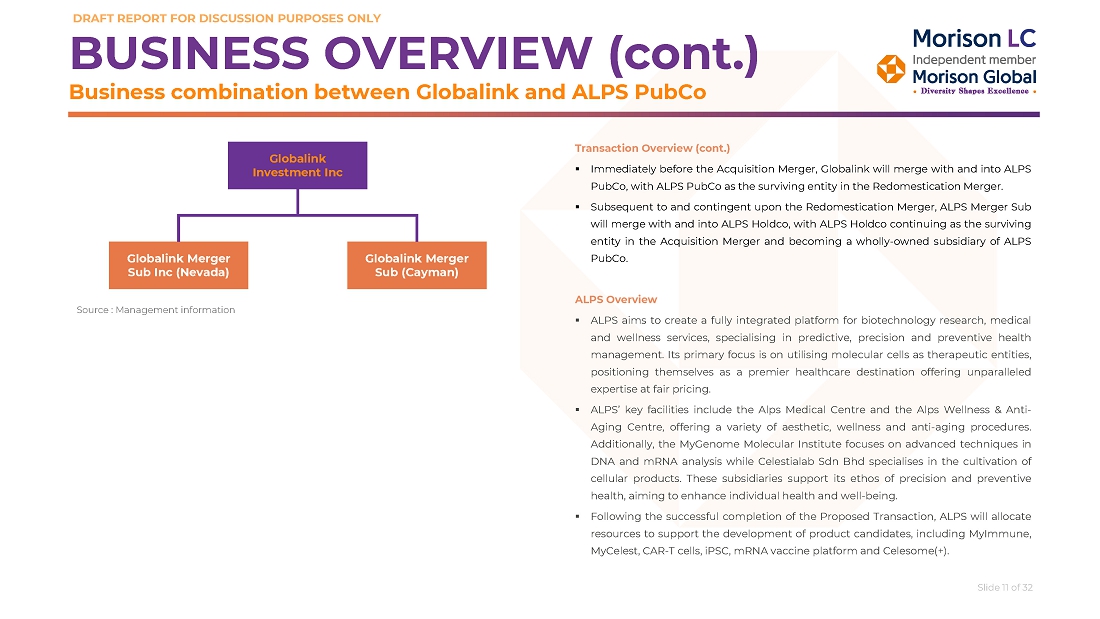
| J-12 |

| J-13 |

| J-14 |

| J-15 |

| J-16 |

| J-17 |

| J-18 |

| J-19 |

| J-20 |

| J-21 |

| J-22 |
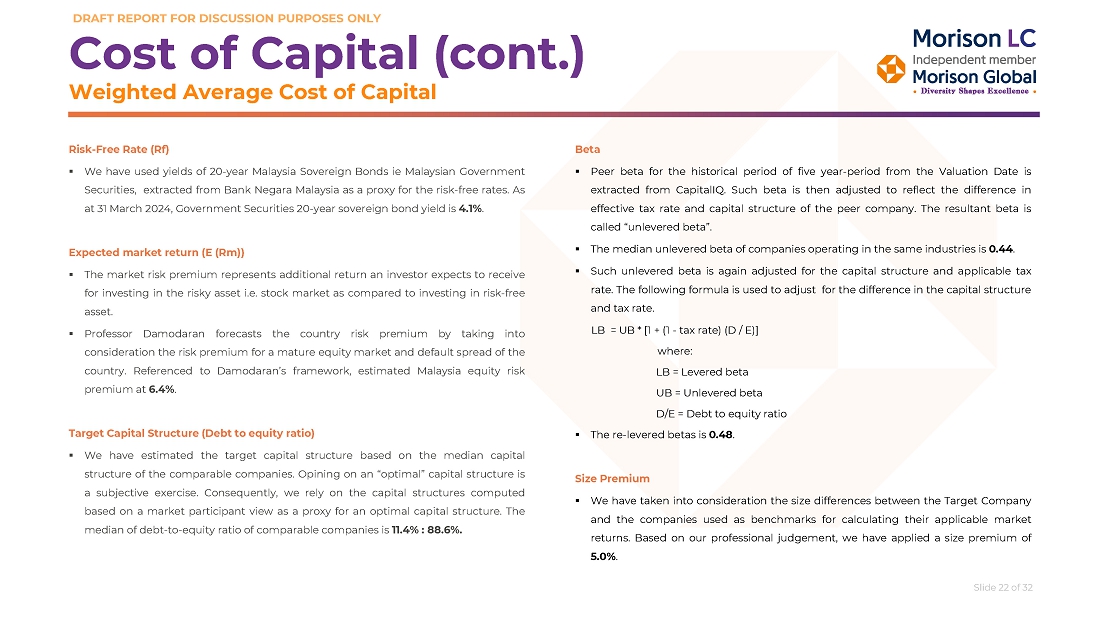
| J-23 |

| J-24 |

| J-25 |
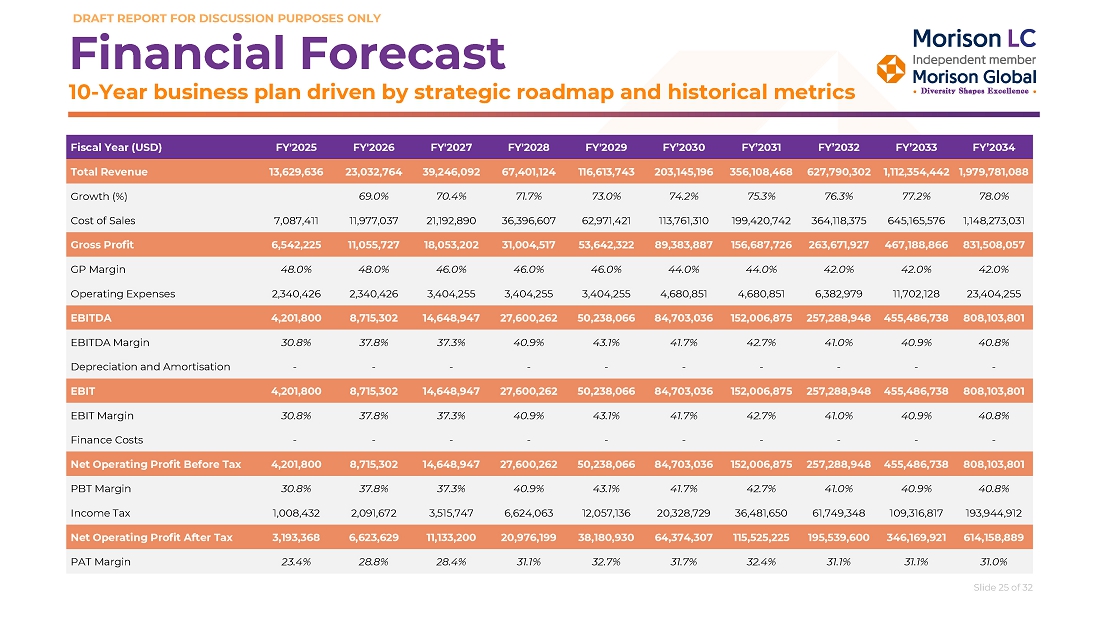
| J-26 |
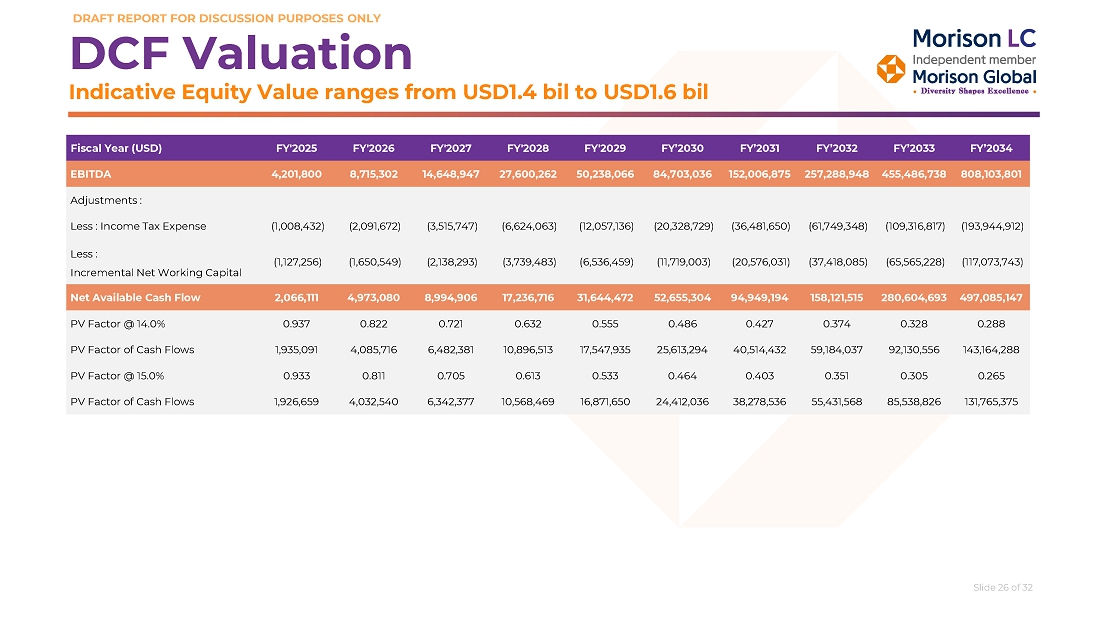
| J-27 |

| J-28 |
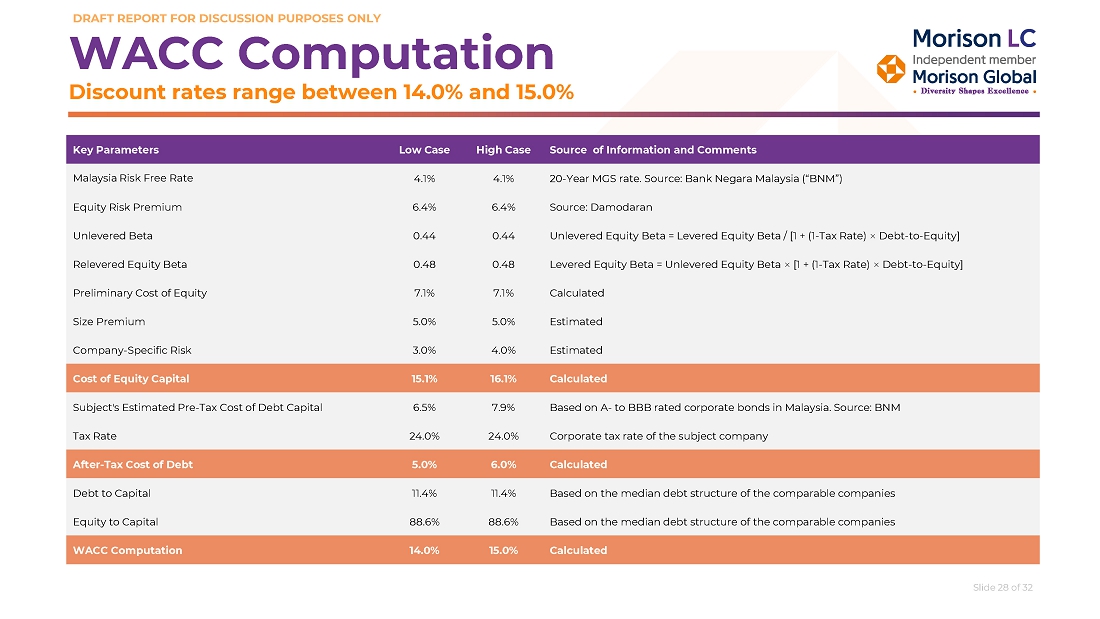
| J-29 |

| J-30 |

| J-31 |

| J-32 |

| J-33 |

| J-34 |
PART II
INFORMATION NOT REQUIRED IN THE PROSPECTUS
Item 20. Indemnification of Directors and Officers.
PubCo A&R Memorandum and Articles of Association, provide that, to the extent permitted by law, PubCo shall indemnify each existing or former secretary, director (including alternate director), and any other officers (including an investment adviser or an administrator or liquidator) and their personal representatives against:
(a) all actions, proceedings, costs, charges, expenses, losses, damages, or liabilities incurred or sustained by the existing or former director (including alternate director), secretary, or officer in or about the conduct of the PubCo business or affairs or in the execution or discharge of the existing or former director (including alternate director)’s, secretary’s, or officer’s duties, powers, authorities or discretions; and
(b) without limitation to paragraph (a) above, all costs, expenses, losses, or liabilities incurred by the existing or former director (including alternate director), secretary, or officer in defending (whether successfully or otherwise) any civil, criminal, administrative or investigative proceedings (whether threatened, pending or completed) concerning the PubCo or its affairs in any court or tribunal, whether in the Cayman Islands or elsewhere.
No such existing or former director (including alternate director), secretary, or officer, however, shall be indemnified in respect of any matter arising out of his own dishonesty, willful default, willful neglect or fraud.
To the extent permitted by law, PubCo may make a payment, or agree to make a payment, whether by way of advance, loan or otherwise, for any legal costs incurred by an existing or former director (including alternate director), secretary or officer of the company in respect of any matter identified above on condition that the director (including alternate director), secretary or officer must repay the amount paid by the company to the extent that it is ultimately found not liable to indemnify the director (including alternate director), secretary or that officer for those legal costs.
Item 21. Exhibits and Financial Statements Schedules
| II-1 |
| * | Indicates management contract or compensatory plan or arrangement. |
| + | To be filed by amendment. |
| # | Certain of the exhibits and schedules to this Exhibit have been omitted in accordance with Regulation S-K Item 601. The Registrant agrees to furnish a copy of all omitted exhibits and schedules to the SEC upon its request. |
| ** | Certain confidential portions (indicated by brackets and asterisks) have been omitted from this exhibit. |
| II-2 |
Item 22. Undertakings
| A. | PubCo hereby undertakes: |
| (1) | To file, during any period in which offers or sales are being made, a post-effective amendment to this registration statement: |
| (i) | To include any prospectus required by Section 10(a)(3) of the Securities Act of 1933; | |
| (ii) | To reflect in the prospectus any facts or events arising after the effective date of the registration statement (or the most recent post-effective amendment thereof) which, individually or in the aggregate, represent a fundamental change in the information set forth in the registration statement. Notwithstanding the foregoing, any increase or decrease in volume of securities offered (if the total dollar value of securities offered would not exceed that which was registered) and any deviation from the low or high end of the estimated maximum offering range may be reflected in the form of prospectus filed with the Commission pursuant to Rule 424(b) if, in the aggregate, the changes in volume and price represent no more than 20 percent change in the maximum aggregate offering price set forth in the “Calculation of Registration Fee” table in the effective registration statement. | |
| (iii) | To include any material information with respect to the plan of distribution not previously disclosed in the registration statement or any material change to such information in the registration statement. |
| (2) | That, for the purpose of determining any liability under the Securities Act of 1933, as amended, each such post-effective amendment shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof. | |
| (3) | To remove from registration by means of a post-effective amendment any of the securities being registered which remain unsold at the termination of the offering. | |
|
(4) |
To file a post-effective amendment to the registration statement to include any financial statements required by Item 8.A of Form 20-F at the start of any delayed offering or throughout a continuous offering. | |
| (5) | For purposes of determining any liability under the Securities Act of 1933, each filing of the registrant’s annual report pursuant to section 13(a) or section 15(d) of the Securities Exchange Act of 1934 (and, where applicable, each filing of an employee benefit plan’s annual report pursuant to section 15(d) of the Securities Exchange Act of 1934) that is incorporated by reference in the registration statement shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof. |
| B. | PubCo hereby undertakes: |
| (1) | that prior to any public reoffering of the securities registered hereunder through use of a prospectus which is a part of this registration statement, by any person or party who is deemed to be an underwriter within the meaning of Rule 145(c), that such reoffering prospectus will contain the information called for by the applicable registration form with respect to reofferings by persons who may be deemed underwriters, in addition to the information called for by the other items of the applicable form. | |
| (2) | that every prospectus: (i) that is filed pursuant to paragraph (1) immediately preceding, or (ii) that purports to meet the requirements of Section 10(a)(3) of the Act and is used in connection with an offering of securities subject to Rule 415, will be filed as a part of an amendment to the registration statement and will not be used until such amendment is effective, and that, for purposes of determining any liability under the Securities Act of 1933, each such post-effective amendment shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof. |
| C. | Insofar as indemnification for liabilities arising under the Securities Act of 1933 may be permitted to directors, officers and controlling persons of the registrant pursuant to the foregoing provisions, or otherwise, the registrant has been advised that in the opinion of the Securities and Exchange Commission such indemnification is against public policy as expressed in the Act and is, therefore, unenforceable. In the event that a claim for indemnification against such liabilities (other than the payment by the registrant of expenses incurred or paid by a director, officer or controlling person of the registrant in the successful defense of any action, suit or proceeding) is asserted by such director, officer or controlling person in connection with the securities being registered, the registrant will, unless in the opinion of its counsel the matter has been settled by controlling precedent, submit to a court of appropriate jurisdiction the question whether such indemnification by it is against public policy as expressed in the Act and will be governed by the final adjudication of such issue. |
| D. | The undersigned registrant hereby undertakes (i) to respond to requests for information that is incorporated by reference into the prospectus pursuant to Items 4, 10(b), 11, or 13 of this Form, within one business day of receipt of such request, and to send the incorporated documents by first class mail or other equally prompt means. This includes information contained in documents filed subsequent to the effective date of the registration statement through the date of responding to the request. |
| E. | The undersigned registrant hereby undertakes to supply by means of a post-effective amendment all information concerning a transaction and the company being acquired involved therein, that was not the subject of and included in the registration statement when it became effective. |
| II-3 |
SIGNATURES
Pursuant to the requirements of the Securities Act, the undersigned co-registrant has duly caused this registration statement to be signed on its behalf by the undersigned, thereunto duly authorized, in Kuala Lumpur, Malaysia, on December 23, 2024.
| Alps Global Holding Pubco | ||
| By: | /s/ Dr. Tham Seng Kong | |
| Name: | Dr. Tham Seng Kong | |
| Title: | Director | |
Power of Attorney
Each person whose signature appears below constitutes and appoints Dr. Tham Seng Kong as attorney-in-fact with full power of substitution, for him or her in any and all capacities, to do any and all acts and all things and to execute any and all instruments which said attorney and agent may deem necessary or desirable to enable the registrant to comply with the Securities Act, and any rules, regulations, and requirements of the Securities and Exchange Commission thereunder, in connection with the registration under the Securities Act of ordinary shares of the registrant, including, without limitation, the power and authority to sign the name of each of the undersigned in the capacities indicated below to the Registration Statement on Form F-4 (the “Registration Statement”) to be filed with the Securities and Exchange Commission with respect to such common stock, to any and all amendments or supplements to such Registration Statement, whether such amendments or supplements are filed before or after the effective date of such Registration Statement, to any related Registration Statement filed pursuant to Rule 462(b) under the Securities Act, and to any and all instruments or documents filed as part of or in connection with such Registration Statement or any and all amendments thereto, whether such amendments are filed before or after the effective date of such Registration Statement; and each of the undersigned hereby ratifies and confirms all that such attorney and agent shall do or cause to be done by virtue hereof.
Pursuant to the requirements of the Securities Act of 1933, this registration statement has been signed by the following persons in the capacities indicated on December 23, 2024.
| Signatures | Title | |
/s/ Dr. Tham Seng Kong |
Managing Director; Group Chief Executive Officer; Group Chief Research Scientist, Interim Chief Financial Officer | |
| Dr Tham Seng Kong | (Principal Executive Officer, Financial Officer and Accounting Officer) |
| II-4 |
AUTHORIZED REPRESENTATIVE
Pursuant to the requirement of the Securities Act of 1933, the undersigned, solely in his capacity as the duly authorized representative of Alps Global Holding Pubco, has signed this registration statement in the City of New York, New York, on December 23, 2024.
| COGENCY GLOBAL INC. | ||
| By: | /s/ Colleen A. DeVries | |
| Name: | Colleen A. DeVries | |
| Title: | Senior Vice President on behalf of Cogency Global Inc | |
| II-5 |
SIGNATURES
Pursuant to the requirements of the Securities Act, the undersigned co-registrant has duly caused this registration statement to be signed on its behalf by the undersigned, thereunto duly authorized, in Kuala Lumpur, Malaysia, on the December 23, 2024.
| Alps Life Sciences Inc | ||
| By: | /s/ Dr. Tham Seng Kong | |
| Name: | Dr. Tham Seng Kong | |
| Title: | Director | |
Power of Attorney
Each person whose signature appears below constitutes and appoints Dr. Tham Seng Kong as attorney-in-fact with full power of substitution, for him or her in any and all capacities, to do any and all acts and all things and to execute any and all instruments which said attorney and agent may deem necessary or desirable to enable the registrant to comply with the Securities Act, and any rules, regulations, and requirements of the Securities and Exchange Commission thereunder, in connection with the registration under the Securities Act of ordinary shares of the registrant, including, without limitation, the power and authority to sign the name of each of the undersigned in the capacities indicated below to the Registration Statement on Form F-4 (the “Registration Statement”) to be filed with the Securities and Exchange Commission with respect to such common stock, to any and all amendments or supplements to such Registration Statement, whether such amendments or supplements are filed before or after the effective date of such Registration Statement, to any related Registration Statement filed pursuant to Rule 462(b) under the Securities Act, and to any and all instruments or documents filed as part of or in connection with such Registration Statement or any and all amendments thereto, whether such amendments are filed before or after the effective date of such Registration Statement; and each of the undersigned hereby ratifies and confirms all that such attorney and agent shall do or cause to be done by virtue hereof.
Pursuant to the requirements of the Securities Act of 1933, this registration statement has been signed by the following persons in the capacities indicated on December 23, 2024.
| Signatures | Title | |
| /s/ Dr. Tham Seng Kong | Managing Director; Group Chief Executive Officer; Group Chief Research Scientist | |
| Dr. Tham Seng Kong | (Principal Executive Officer) | |
| /s/ Lisa Teoh @ Teoh Lee Eng | Chief Operating Officer | |
| Lisa Teoh @ Teoh Lee Eng | (Principal Financial Officer and Accounting Officer) | |
| /s/ Chew Yoke Ling | Director | |
| Chew Yoke Ling | ||
| /s/ Mohd Razef bin Abdullah | Director | |
| Mohd Razef bin Abdullah |
AUTHORIZED REPRESENTATIVE
Pursuant to the requirement of the Securities Act of 1933, the undersigned, solely in his capacity as the duly authorized representative of Alps Life Sciences Inc., has signed this registration statement in the City of New York, New York, on December 23, 2024.
| COGENCY GLOBAL INC. | ||
| By: | /s/ Colleen A. DeVries | |
| Name: | Colleen A. DeVries | |
| Title: | Senior Vice President on behalf of Cogency Global Inc | |
| II-6 |